Experimental and Thermodynamic Study on the Temperature-Dependent Surface Activity of Some Polyether Siloxane Surfactants at the Water–Air Interface
Abstract
1. Introduction
2. Results and Discussion
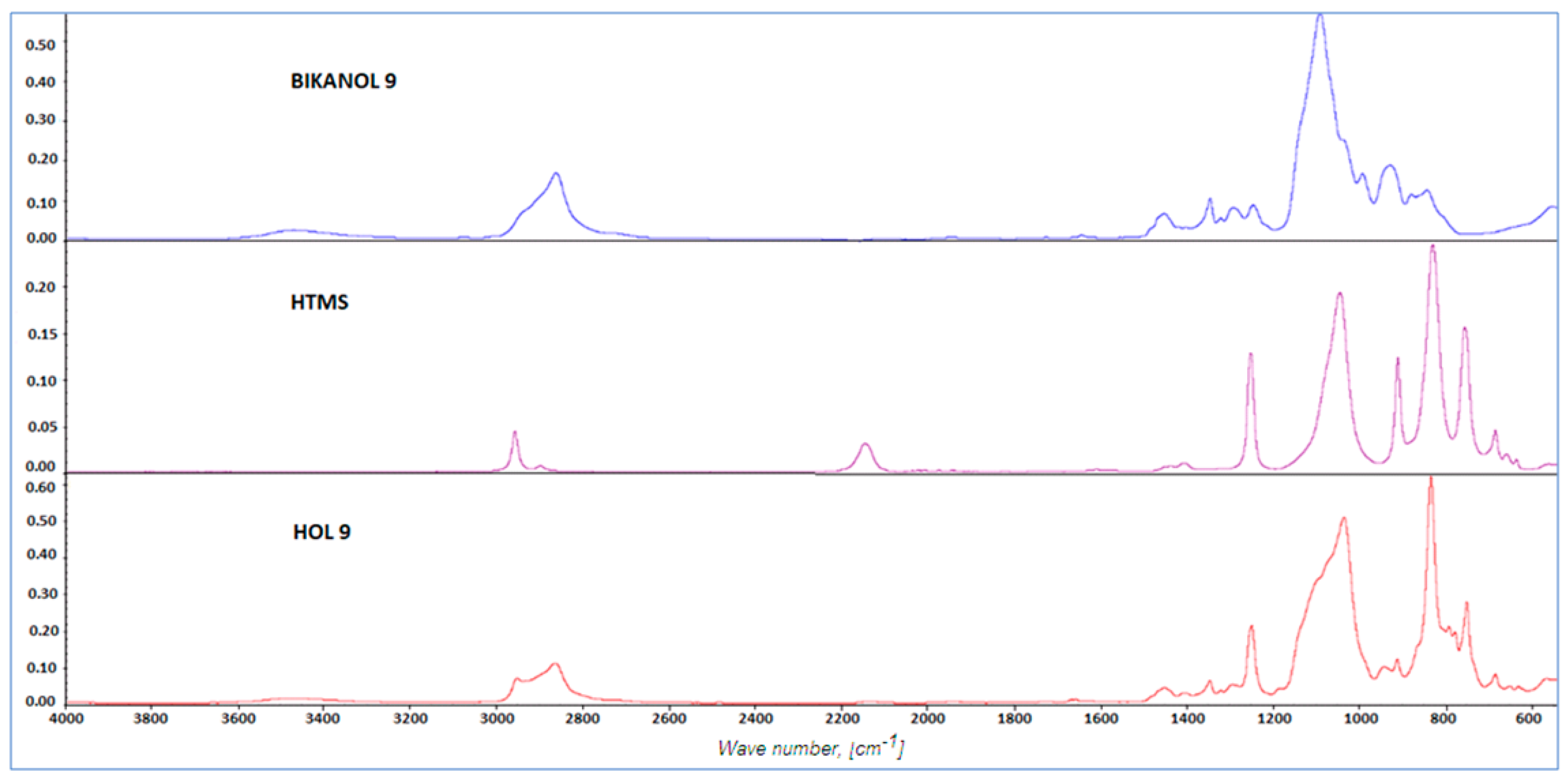
2.1. Results of Synthesis
- 1H NMR (CDCl3, TMS) δ (ppm): −0.05 (CDCl3, TMS) δ (ppm): −0.74 (–SiCH3); 1.49 (–Si(CH3)3); 13.11 (–SiCH2–); 22.79 (–CH2CH2CH2–); 61.19 (–CH2O–); 70.18 (–OCH2CH2–).
- 1Si NMR (CDCl3, TMS) δ (ppm): −21.76 (–Si(CH3)CH2); 6.98 (–Si(CH3)3).
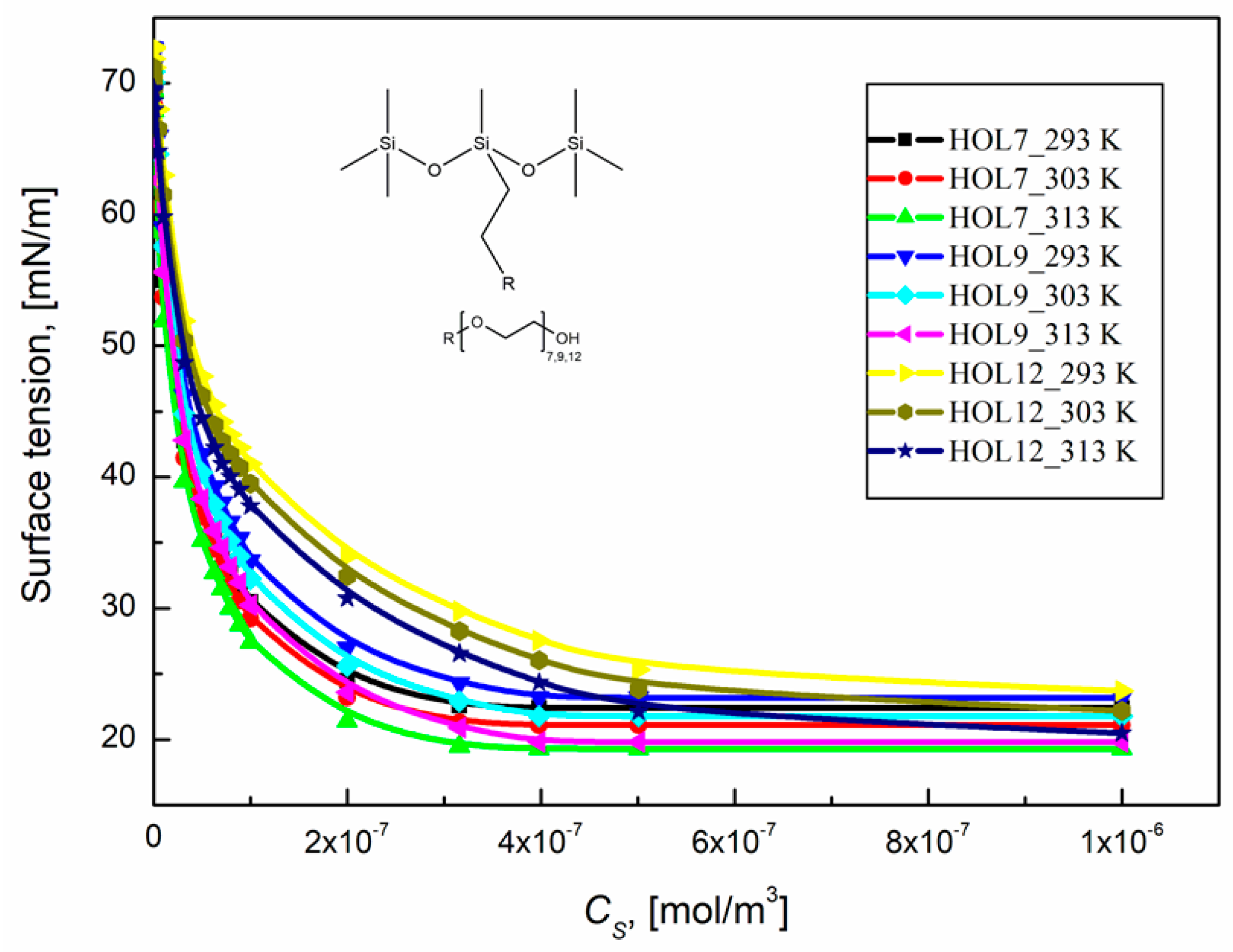
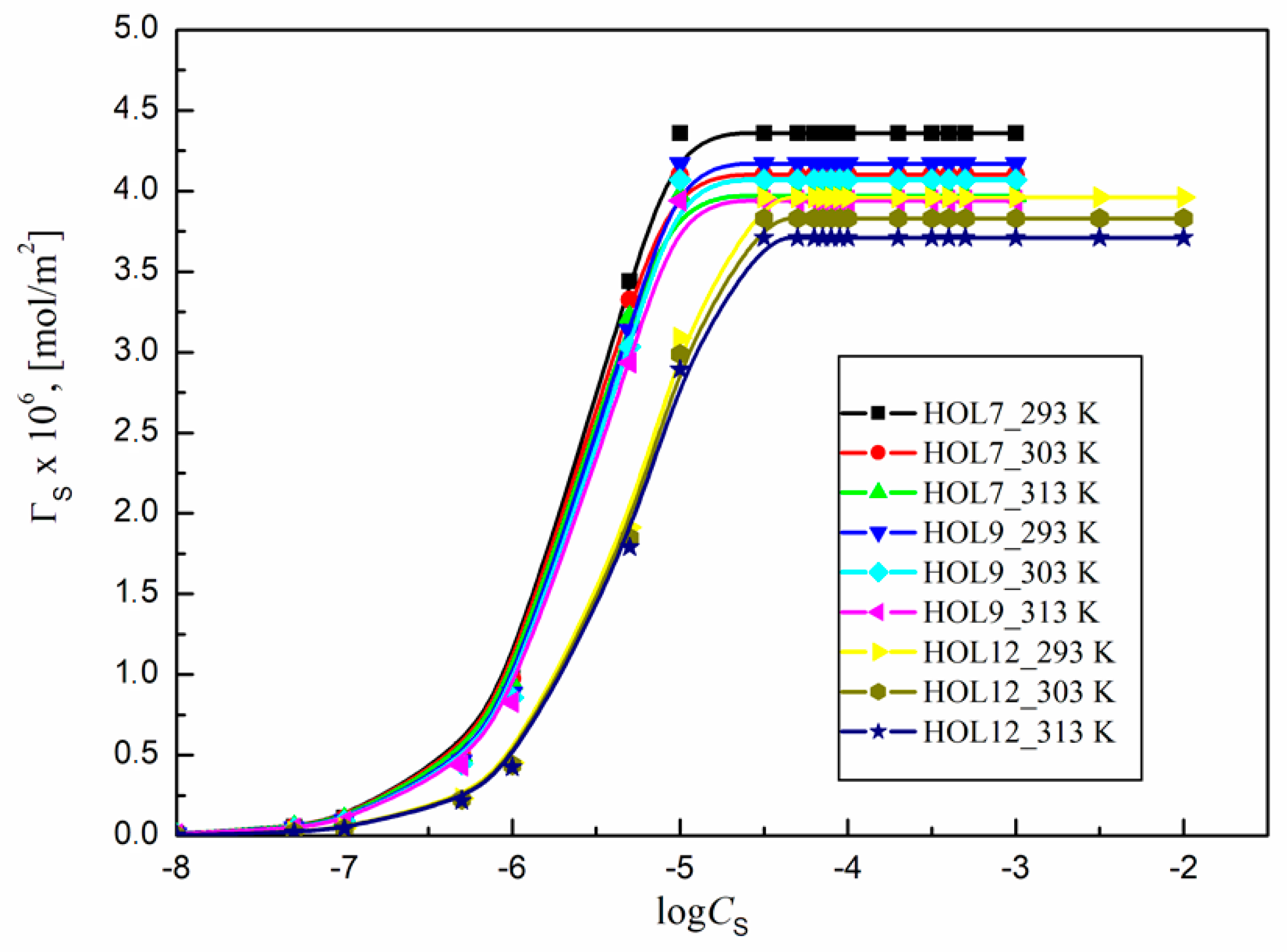
2.2. Tendency of Some Trisiloxane Polyether Surfactants to Adsorb at the Water–Air Interface at Various Temperatures
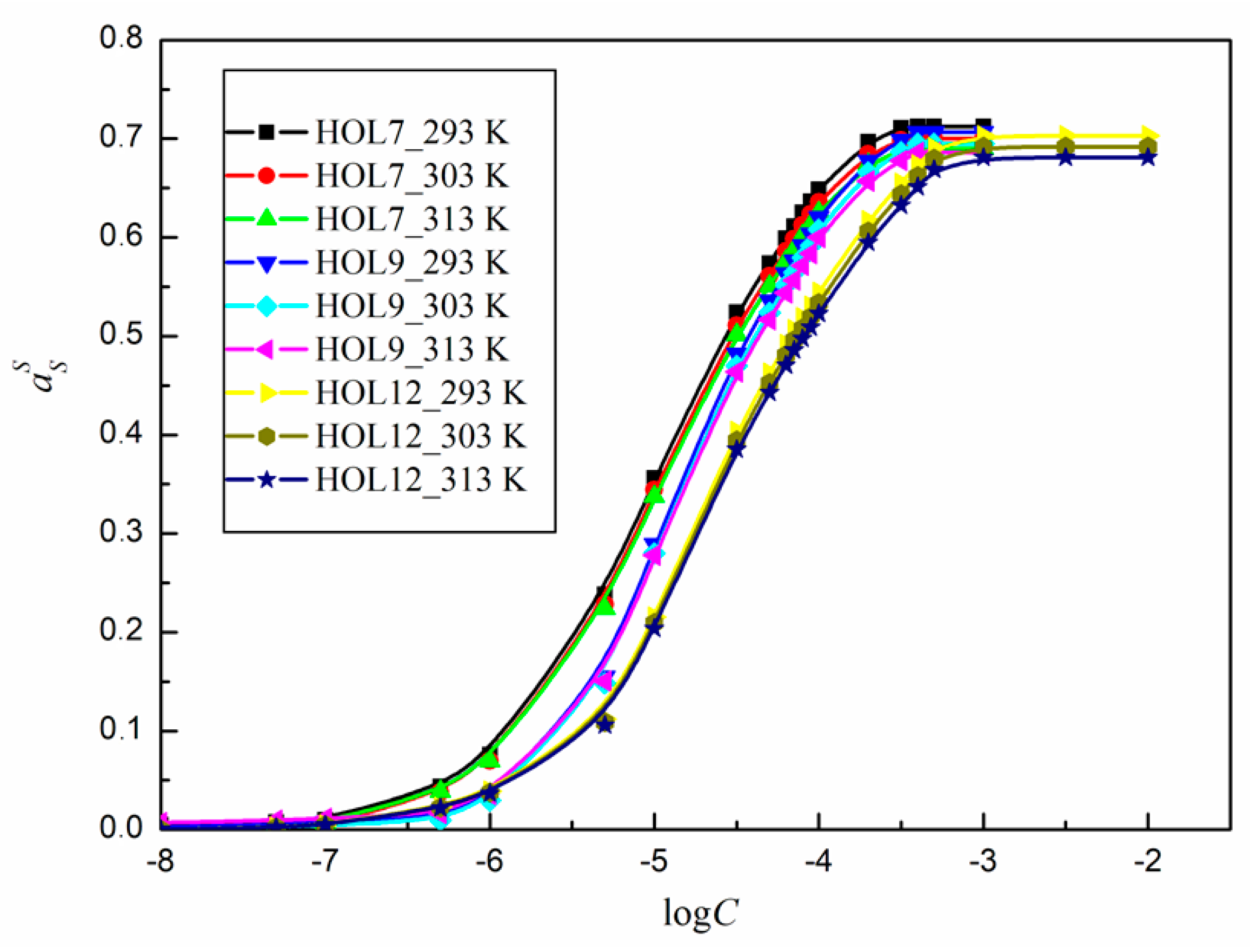
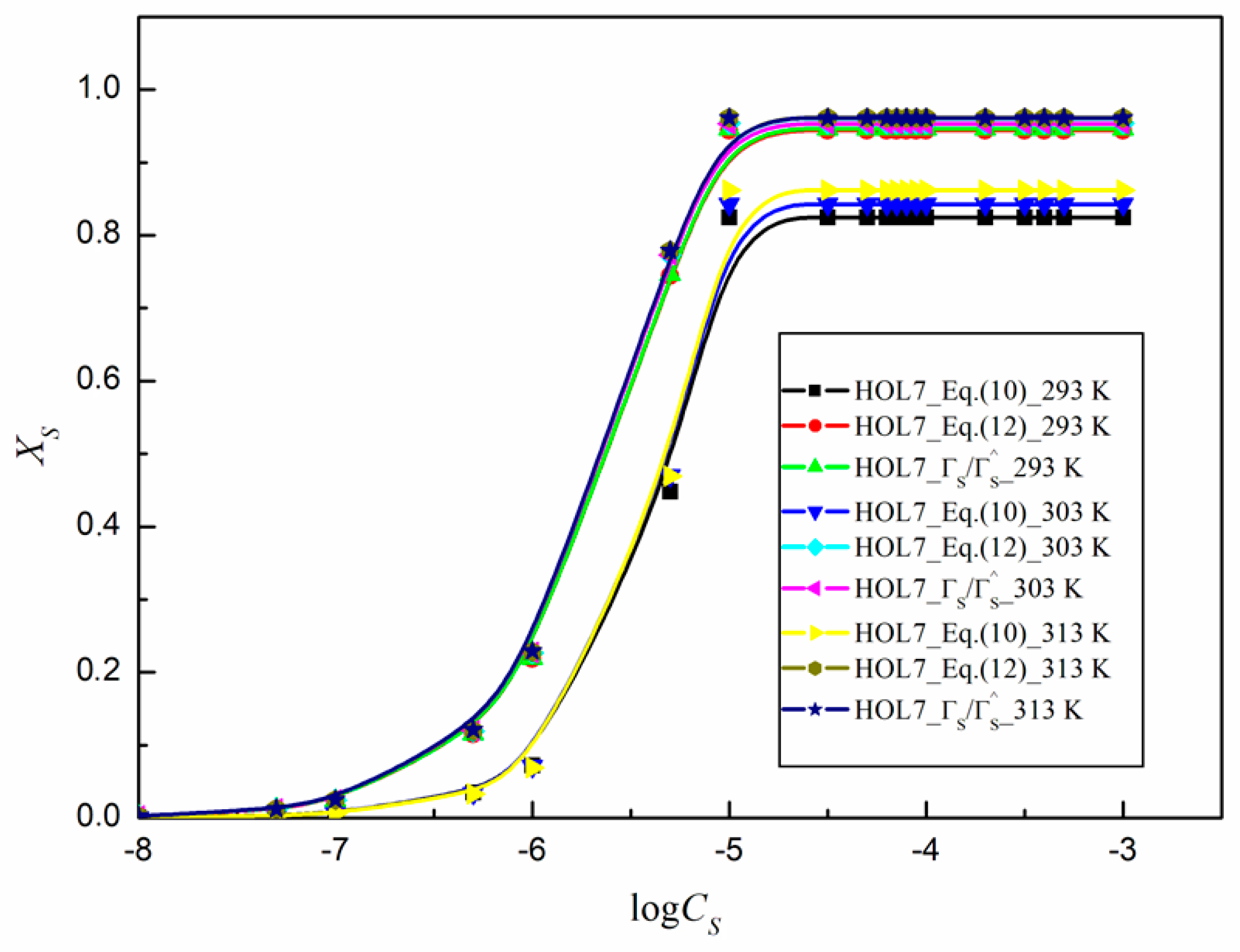
2.3. Activity of Surfactants at the Water–Air Interface at Different Temperatures
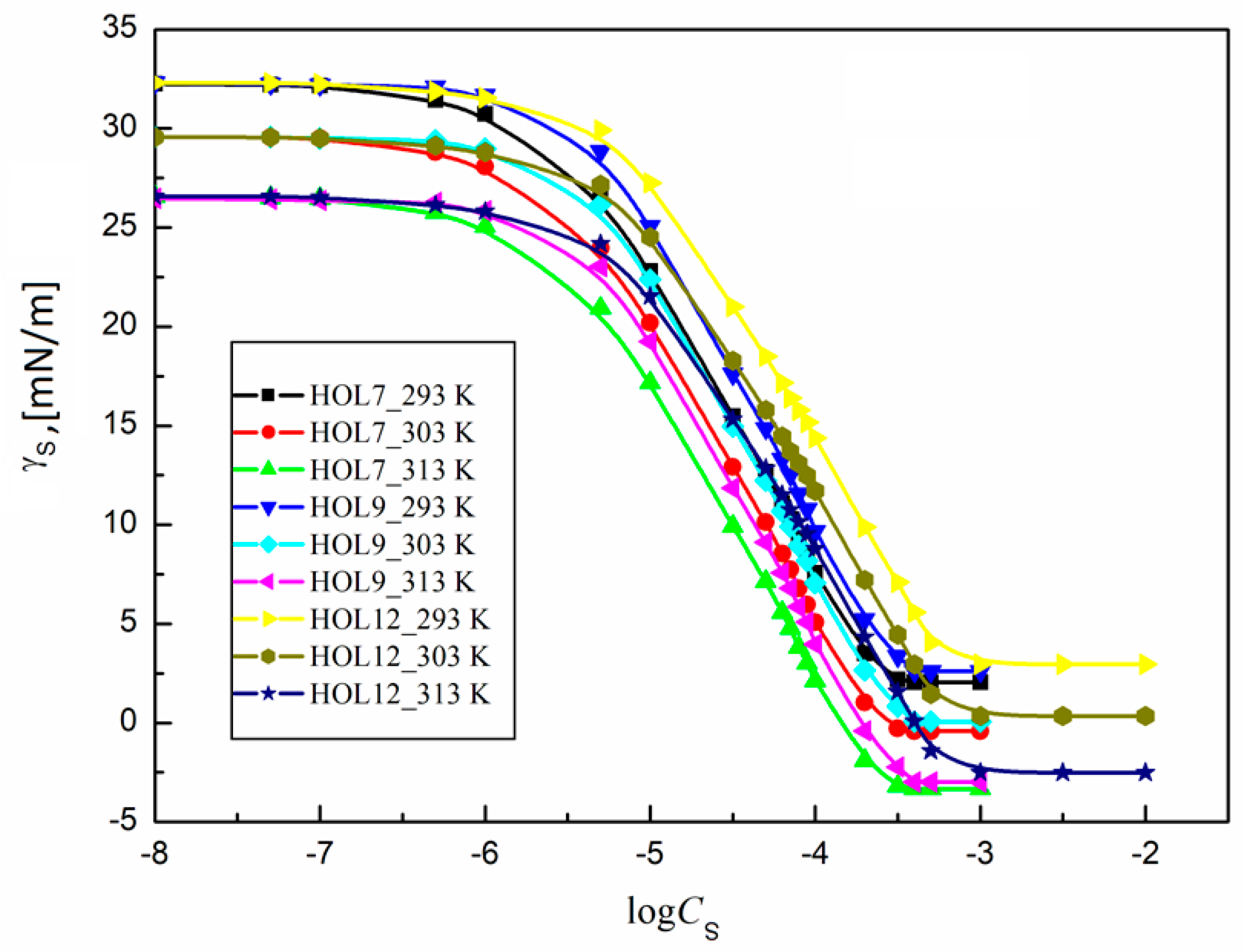
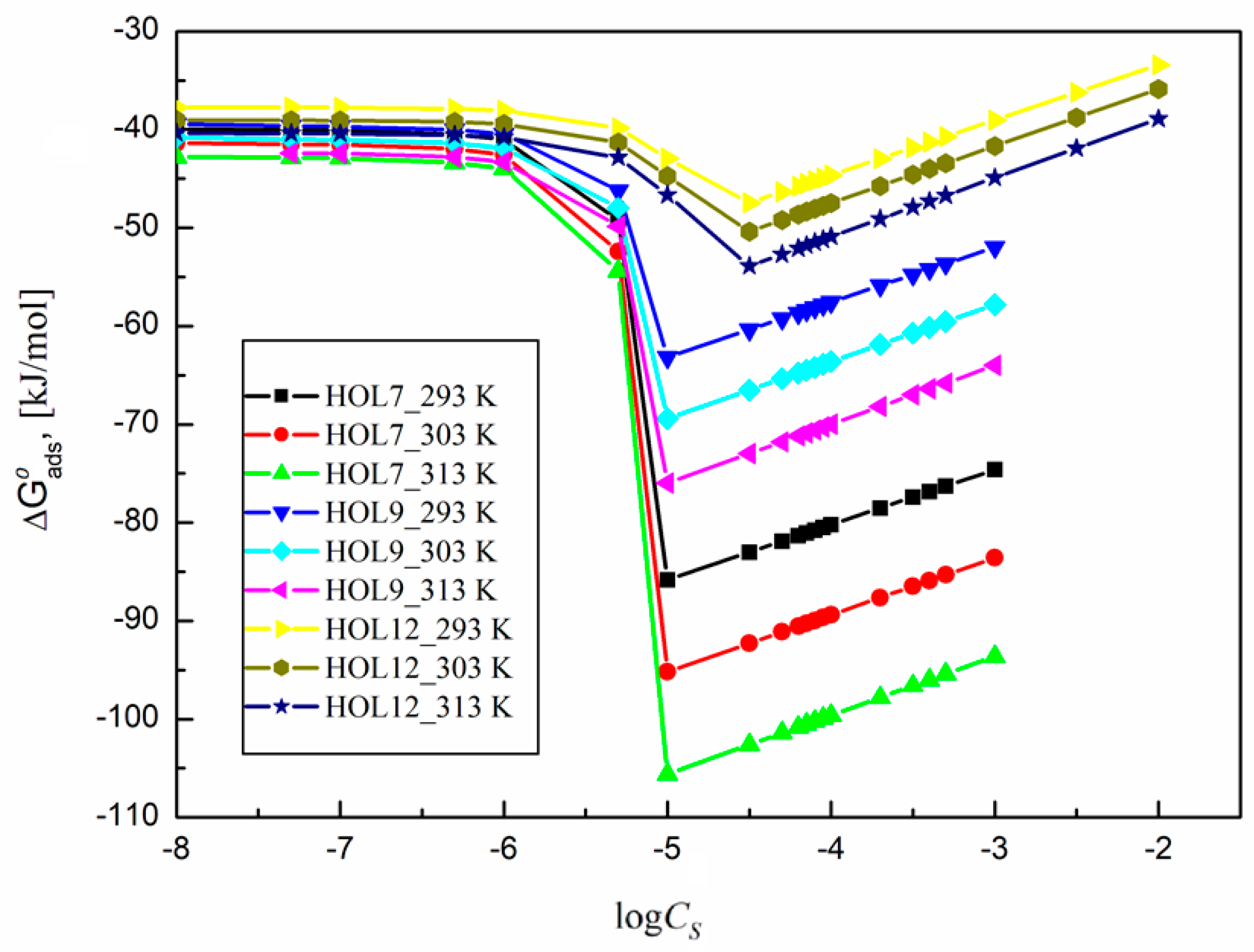
2.4. Thermodynamic Parameters of the Trisiloxane Surfactants at the Interface Between Water and Air
2.5. Thermodynamics of the Adsorption of Some Trisiloxane Surfactants at the Water–Air Interface
3. Materials and Methods
3.1. Chemicals
3.2. Synthesis of 3-[3-(Hydroxy)(polyethoxy)propyl]-1,1,1,3,5,5,5-heptamethyltrisiloxane (HOL7, HOL9, and HOL12) Surfactants
3.3. Physicochemical Studies
3.4. Measurements of the Surface Tension
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ramis-Ramos, G.; Simó-Alfonso, E.F.; Escrig-Domenech, A.; Beneito-Cambra, M. Liquid Chromatography of Surfactants. In Reference Module in Chemistry, Molecular Sciences and Chemical Engineering; Elsevier: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Hill, R.M. Siloxane surfactants. In Specialist Surfactants; Robb, I.O., Ed.; Blackie Academic and Professional, an imprint of Chapman & Hall: London, UK, 1997; p. 143. [Google Scholar]
- Hill, R.M. Silicone (Siloxane) surfactants. Encyclopedia of Physical Science and Technology, 3rd ed.; Meyers, R.A., Ed.; Academic Press: Academic, MA, USA, 2003; pp. 793–804. [Google Scholar]
- Putzien, S.; Nuyken, O.; Kühn, F.E. Functionalized polysilalkylene siloxanes (polycarbosiloxanes) by hydrosilylation—Catalysis and synthesis. Prog. Polym. Sci. 2010, 35, 687–713. [Google Scholar] [CrossRef]
- Shi, Q.; Qiao, W. Synthesis of siloxane polyether surfactants and their solubility in supercritical CO2. J. Surfactants Deterg. 2017, 20, 453–458. [Google Scholar] [CrossRef]
- Fu, K.; Xu, M.; Zhong, R.; Yang, Z.; Zhou, A. Preparation, characterization and application of polyether and long-chain alkyl co-modified polydimethylsiloxane. J. Polymer Res. 2019, 26, 261–269. [Google Scholar] [CrossRef]
- Kim, D.-W.; Noh, S.-T. Synthesis and surface-active properties of a trisiloxane modified oligo(propylene oxide-block-ethylene oxide) wetting agent. J. Appl. Polymer Sci. 2004, 92, 3292–3302. [Google Scholar] [CrossRef]
- Tan, J.; Xiong, X.; He, Z.; Cao, F.; Sun, D. Aggregation behavior of polyether based Siloxane Surfactants in Aqueous Solutions: Effect of Alkyl Groups and Steric Hindrance. J. Phys. Chem. B 2019, 123, 1390–1399. [Google Scholar] [CrossRef]
- Tan, J.; He, Z.; Miao, Y.; Lin, M. Synthesis and Surface Properties of Polyether-Based Silicone Surfactants with Different Siloxane Groups. J. Surfactants Deterg. 2019, 22, 875–883. [Google Scholar] [CrossRef]
- Tan, J.; Liu, Y.; Ye, Z. Synthesis, aggregation behavior of polyether based carbosilane surfactants in aqueous solution. J. Mol. Liq. 2019, 279, 657–661. [Google Scholar] [CrossRef]
- Cao, J.; Zuo, Y.; Wang, D.; Zhang, J.; Feng, S. Functional polysiloxanes: A novel synthesismethod and hydrophilic applications. New J. Chem. 2017, 41, 8546–8553. [Google Scholar] [CrossRef]
- Tan, J.; He, Z.; Bai, Y.; Yan, P. Synthesis, characterization and surface properties of novel polyether based siloxane surfactants. J. Disp. Sci. Technol. 2020, 41, 188–194. [Google Scholar] [CrossRef]
- Schmidt, G.L.F. Industrial Applications of Surfactants; Karsa, D.R., Ed.; Royal Society of Chemistry: London, UK, 1987; p. 24. [Google Scholar]
- Schmidt, G. Silicone Surfactants; Part III/Silikon-Tensider; Teil III. Tenside Surfactants Deterg. 1990, 27, 324–328. [Google Scholar] [CrossRef]
- Knoche, M. Organosilicone surfactant performance in agricultural spray application: A review. Weed Res. 1994, 34, 221–239. [Google Scholar] [CrossRef]
- Knoche, M.; Tamura, H.; Bukovac, J. Performance and stability of the organosilicon surfactant L-77: Effect of pH, concentration, and temperature. J. Agric. Food Chem. 1994, 39, 202–206. [Google Scholar] [CrossRef]
- Stevens, P.I.G. Organosilicone surfactants as adjuvants for agrochemicals. Pestic. Sci. 1993, 38, 103–122. [Google Scholar] [CrossRef]
- Fink, H.F. Silicone Surfactants, Part IV*: Silicone Surfactants as Paint Additives. Tenside Surfactants Deterg. 1991, 28, 306–312. [Google Scholar] [CrossRef]
- Karasiewicz, J.; Olszyński, R.M.; Nowicka-Krawczyk, P.; Krawczyk, J. Siloxane containing polyether groups-synthesis and use as an anti-biocorrosion coating. Int. J. Mol. Sci. 2024, 25, 6801. [Google Scholar] [CrossRef]
- Karasiewicz, J.; Krawczyk, J. Thermodynamic Analysis of Trisiloxane Surfactant Adsorption and Aggregation Process. Molecules 2020, 25, 5669. [Google Scholar] [CrossRef] [PubMed]
- Rosen, J.M. Surfactants and Interfacial Phenomena, 3rd ed.; Wiley: New York, NY, USA, 2004. [Google Scholar]
- He, M.; Hill, R.M. Structure and Flow in Surfactant Solutions; Herb, C.A., Prudhomme, R.K., Eds.; American Chemical Society: New York, NY, USA, 1994. [Google Scholar]
- Pukale, D.D.; Bansode, A.S.; Jadhav, N.L.; Pinjari, D.V.; Kulkarni, R.R. Review on Silicone Surfactants: Silicone-based Gemini Surfactants. Physicochemical Properties and Applications. Tenside Surfactants Deterg. 2019, 56, 268–278. [Google Scholar] [CrossRef]
- Gradzielski, M.; Hoffman, H.; Robisch, P.; Ulbricht, W.; Gruning, B. Aggregation behaviour of silicone surfactants in aqueous solutions. Tenside Surfactants Deterg. 1990, 27, 366–374. [Google Scholar] [CrossRef]
- Gentle, T.E.; Snow, S.A. Adsorption of Small Silicone Polyether Surfactants at the air-water Interface. Langmuir 1995, 11, 2905–2910. [Google Scholar] [CrossRef]
- Zdziennicka, A.; Szymczyk, K.; Krawczyk, J.; Jańczuk, B. Activity and thermodynamic parameters of some surfactants adsorption at the water-air interface. Fluid. Phase Equil. 2012, 318, 25–33. [Google Scholar] [CrossRef]
- Szymczyk, K.; Zdziennicka, A.; Krawczyk, J.; Jańczuk, B. Behaviour of cetyltrimethylammonium bromide, Triton X-100 and Triton X-114 in mixed monolayer at the water-air interface. J. Chem. Therm. 2014, 69, 85–92. [Google Scholar] [CrossRef]
- Krawczyk, J. Temperature impact on the water-air interfacial activity of n-octyl and n-dodecyl-β-D-glucopyranosides. Colloids Surf. A 2017, 533, 61–67. [Google Scholar] [CrossRef]
- Ivanova, N.; Starov, V.; Rubio, R.; Ritacco, H.; Hilal, N.; Johanson, D. Critical wetting concentrations of trisiloxane surfactants. Colloids Surf. A 2010, 354, 143–148. [Google Scholar] [CrossRef]
- Sankaran, A.; Karakashev, S.I.; Sett, S.; Grozev, N.; Yarin, A.L. On the nature of superspreaders. Adv. Colloid. Int. Sci. 2019, 263, 1–18. [Google Scholar] [CrossRef]
- Silva, S.C.M.; Gambaryan-Roisman, T.; Venzmer, J. Surface tension behavior of superspreading and non-superspreading trisiloxane surfactants. Colloid. Polym. Sci. 2023, 301, 739–744. [Google Scholar] [CrossRef]
- Atkins, P.W. Chemia Fizyczna; Wyd, I., Ed.; PWN: Warszawa, Poland, 2001. [Google Scholar]
- Butler, J.A.V. The thermodynamics of the surfaces of the solutions. Proc. Roy. Soc. Ser. A 1932, 138, 348–375. [Google Scholar]
- Sprow, F.B.; Prausnitz, J.M. Surface thermodynamics of liquid mixtures. Can. J. Chem. Eng. 1967, 45, 25. [Google Scholar] [CrossRef]
- van Oss, C.J.; Constanzo, F.M. Adhesion of anionic surfactants to polymer surfaces and low-energy materials. J. Adhesion Sci. Technol. 1992, 6, 477–487. [Google Scholar] [CrossRef]
- Jańczuk, B.; Zdziennicka, A.; Wójcik, W. The properties of mixtures of two anionic surfactants in water at the water-air interface. Colloids Surf. A 2003, 220, 61–68. [Google Scholar] [CrossRef]
- Chataraj, D.K.; Birdi, K.S.; Kalder, K.; Das, K.P.; Mitra, A. Surface activity coefficients of spread monolayers of behenic acid salts at air–water interface. Adv. Colloid. Interface Sci. 2006, 151, 123–126. [Google Scholar] [CrossRef]
- Adamson, A.W. Physical Chemistry of Surfaces, 5th ed.; Wiley: New York, NY, USA, 1990. [Google Scholar]
- Krawczyk, J. Thermodynamic properties of disaccharide based surfactants adsorption at the water-air interface. Colloids Surf. A 2018, 551, 50–57. [Google Scholar] [CrossRef]
- Zdziennicka, A.; Jańczuk, B. Thermodynamic parameters of some biosurfactants and surfactants adsorption at water-air interface. J. Mol. Liquids 2017, 243, 236–244. [Google Scholar] [CrossRef]
- de Boer, J.H. The Dynamic Character of Adsorption; Oxford University Press: London, UK, 1953. [Google Scholar]
- Pandya, P.; Lad, K.N.; Bahadur, P. Silicone surfactants in aqueous solutions. Tenside Surfactants Deterg. 1996, 33, 374–378. [Google Scholar] [CrossRef]



| Surfactant | CMC [mM/dm3] | ||
|---|---|---|---|
| 293 K | 303 K | 313 K | |
| HOL7 | 0.170 | 0.168 | 0.157 |
| HOL9 | 0.200 | 0.198 | 0.195 |
| HOL12 | 0.312 | 0.253 | 0.208 |
| Surfactant | ×106 [mol/m2] | [Ǻ2] | ×106 [mol/m2] | [Ǻ2] | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 293 K | 303 K | 313 K | 293 K | 303 K | 313 K | 293 K | 303 K | 313 K | 293 K | 303 K | 313 K | |
| HOL7 | 4.36 | 4.10 | 3.97 | 38.08 | 40.50 | 41.82 | 4.61 | 4.30 | 4.13 | 35.95 | 38.50 | 40.00 |
| HOL9 | 4.17 | 4.07 | 3.94 | 39.82 | 40.72 | 42.06 | 4.67 | 4.47 | 4.27 | 35.53 | 37.00 | 38.70 |
| HOL12 | 3.96 | 3.83 | 3.71 | 41.92 | 43.35 | 44.78 | 5.01 | 4.74 | 4.46 | 33.14 | 35.00 | 37.00 |
| Surfactant | [kJ/mol] | [kJ/mol] | [kJ/molK] | |
|---|---|---|---|---|
| HOL7 | 293 K | 39.990 | 0.141 | 1.323 |
| 303 K | 41.400 | 0.141 | 1.323 | |
| 313 K | 42.810 | 0.141 | 1.323 | |
| HOL9 | 293 K | 39.430 | 0.139 | 1.300 |
| 303 K | 40.810 | 0.139 | 1.300 | |
| 313 K | 42.210 | 0.139 | 1.300 | |
| HOL12 | 293 K | 37.960 | 0.130 | 1.262 |
| 303 K | 39.300 | 0.130 | 1.262 | |
| 313 K | 40.640 | 0.130 | 1.262 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krawczyk, J.; Karasiewicz, J.; Wojdat, K. Experimental and Thermodynamic Study on the Temperature-Dependent Surface Activity of Some Polyether Siloxane Surfactants at the Water–Air Interface. Int. J. Mol. Sci. 2025, 26, 5472. https://doi.org/10.3390/ijms26125472
Krawczyk J, Karasiewicz J, Wojdat K. Experimental and Thermodynamic Study on the Temperature-Dependent Surface Activity of Some Polyether Siloxane Surfactants at the Water–Air Interface. International Journal of Molecular Sciences. 2025; 26(12):5472. https://doi.org/10.3390/ijms26125472
Chicago/Turabian StyleKrawczyk, Joanna, Joanna Karasiewicz, and Katarzyna Wojdat. 2025. "Experimental and Thermodynamic Study on the Temperature-Dependent Surface Activity of Some Polyether Siloxane Surfactants at the Water–Air Interface" International Journal of Molecular Sciences 26, no. 12: 5472. https://doi.org/10.3390/ijms26125472
APA StyleKrawczyk, J., Karasiewicz, J., & Wojdat, K. (2025). Experimental and Thermodynamic Study on the Temperature-Dependent Surface Activity of Some Polyether Siloxane Surfactants at the Water–Air Interface. International Journal of Molecular Sciences, 26(12), 5472. https://doi.org/10.3390/ijms26125472






