The Role of Fibrogenesis and Extracellular Matrix Proteins in the Pathogenesis of Graves’ Ophthalmopathy
Abstract
1. Introduction
2. Cell Mediators in GO
3. Adipogenesis and ECM of GO
4. Transforming Growth Factor-β1 (TGF-β1)-Induced Myofibroblast Transdifferentiation and ECM in GO
5. Hyaluronan Formation by Orbital Fibroblasts
6. Oxidative Stress and Smoking in the ECM of GO
7. Endoplasmic Reticulum (ER) Stress in the ECM of GO
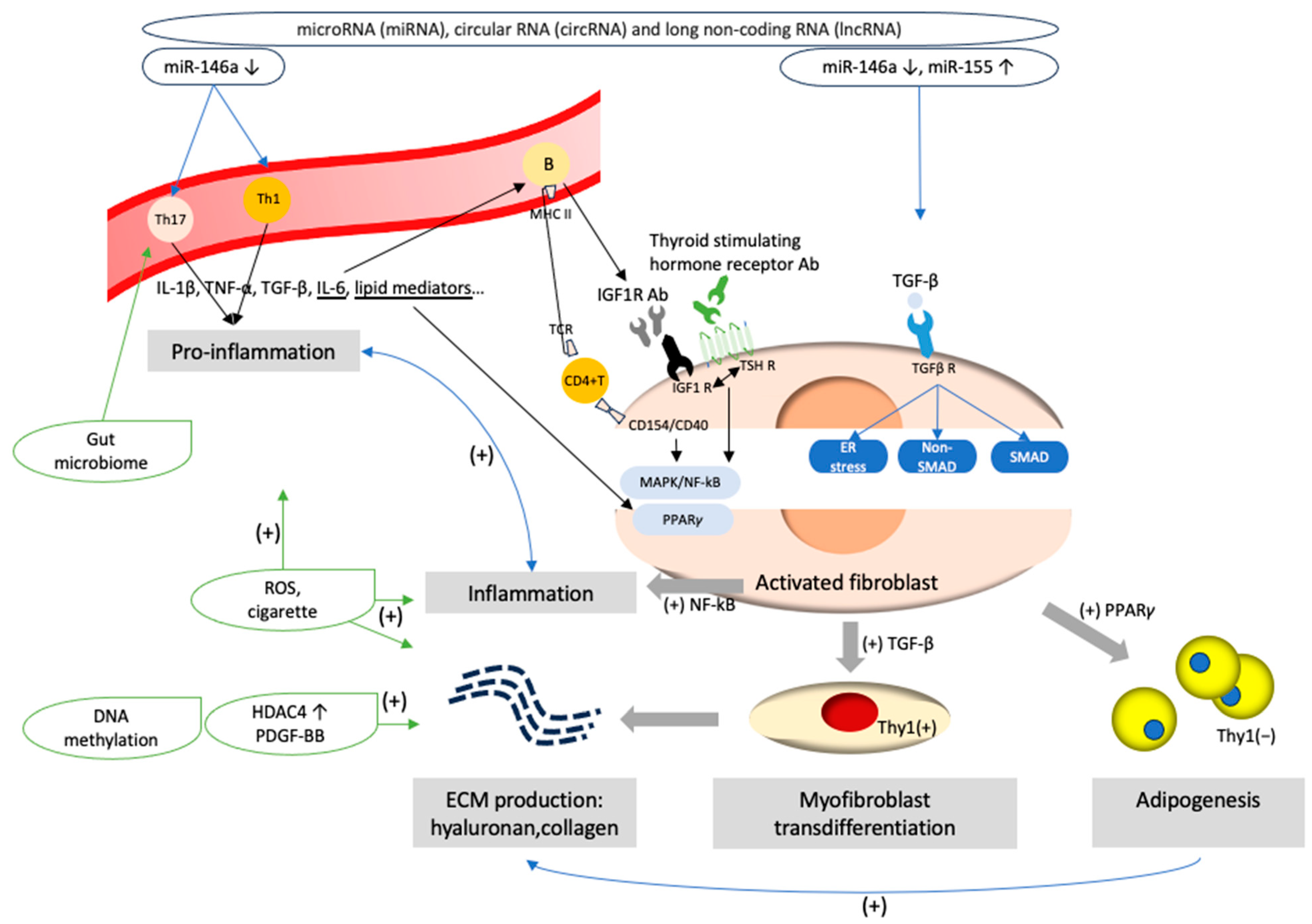
8. Epigenetics and the Gut Microbiome in the ECM of GO
8.1. Histone Deacetylases (HDACs) and DNA Methylation
8.2. MicroRNA in GO
8.3. Other Non-Coding RNAs in GO
8.4. Gut Microbiome in GO
9. Treatment for Tissue Remodeling in GO
9.1. Biologic Agents
9.2. Potential Therapeutic Target—Antioxidants
9.2.1. Selenium (Se)
9.2.2. Pentoxifylline
9.2.3. Nicotinamide and Allopurinol
9.3. Potential Therapeutic Target—Pirfenidone
9.4. Other Potential Targets
10. Concluding Remarks
Funding
Data Availability Statement
Conflicts of Interest
References
- Taylor, P.N.; Zhang, L.; Lee, R.W.J.; Muller, I.; Ezra, D.G.; Dayan, C.M.; Kahaly, G.J.; Ludgate, M. New insights into the pathogenesis and nonsurgical management of Graves orbitopathy. Nat. Rev. Endocrinol. 2020, 16, 104–116. [Google Scholar] [CrossRef]
- Douglas, R.S.; Kahaly, G.J.; Patel, A.; Sile, S.; Thompson, E.H.Z.; Perdok, R.; Fleming, J.C.; Fowler, B.T.; Marcocci, C.; Marino, M.; et al. Teprotumumab for the Treatment of Active Thyroid Eye Disease. N. Engl. J. Med. 2020, 382, 341–352. [Google Scholar] [CrossRef]
- Sears, C.M.; Azad, A.D.; Amarikwa, L.; Pham, B.H.; Men, C.J.; Kaplan, D.N.; Liu, J.; Hoffman, A.R.; Swanson, A.; Alyono, J.; et al. Hearing Dysfunction After Treatment with Teprotumumab for Thyroid Eye Disease. Am. J. Ophthalmol. 2022, 240, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bhargav, P.R.K.; Sabaretnam, M.; Kumar, S.C.; Zwalitha, S.; Devi, N.V. Regression of Ophthalmopathic Exophthalmos in Graves’ Disease After Total Thyroidectomy: A Prospective Study of a Surgical Series. Indian J. Surg. 2017, 79, 521–526. [Google Scholar] [CrossRef]
- Liu, Z.W.; Masterson, L.; Fish, B.; Jani, P.; Chatterjee, K. Thyroid surgery for Graves’ disease and Graves’ ophthalmopathy. Cochrane Database Syst. Rev. 2015, CD010576. [Google Scholar] [CrossRef]
- Ryan, H. Thyroidectomy and thyrotropic exophthalmos (exophthalmic ophthalmoplegia) a review of 1001 thyroidectomies. Br. J. Ophthalmol. 1949, 33, 769–773. [Google Scholar] [CrossRef][Green Version]
- Feldon, S.E.; Park, D.J.; O’Loughlin, C.W.; Nguyen, V.T.; Landskroner-Eiger, S.; Chang, D.; Thatcher, T.H.; Phipps, R.P. Autologous T-lymphocytes stimulate proliferation of orbital fibroblasts derived from patients with Graves’ ophthalmopathy. Investig. Ophthalmol. Vis. Sci. 2005, 46, 3913–3921. [Google Scholar] [CrossRef]
- Bahn, R.S. Graves’ ophthalmopathy. N. Engl. J. Med. 2010, 362, 726–738. [Google Scholar] [CrossRef]
- Smith, T.J.; Hegedus, L.; Douglas, R.S. Role of insulin-like growth factor-1 (IGF-1) pathway in the pathogenesis of Graves’ orbitopathy. Best. Pract. Res. Clin. Endocrinol. Metab. 2012, 26, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, G.M.; Woeller, C.F.; Pollock, S.J.; O’Loughlin, C.W.; Gupta, S.; Feldon, S.E.; Phipps, R.P. Novel anti-adipogenic activity produced by human fibroblasts. Am. J. Physiol. Cell Physiol. 2010, 299, C672–C681. [Google Scholar] [CrossRef]
- Kumar, S.; Coenen, M.J.; Scherer, P.E.; Bahn, R.S. Evidence for enhanced adipogenesis in the orbits of patients with Graves’ ophthalmopathy. J. Clin. Endocrinol. Metab. 2004, 89, 930–935. [Google Scholar] [CrossRef]
- Zhao, P.; Deng, Y.; Gu, P.; Wang, Y.; Zhou, H.; Hu, Y.; Chen, P.; Fan, X. Insulin-like growth factor 1 promotes the proliferation and adipogenesis of orbital adipose-derived stromal cells in thyroid-associated ophthalmopathy. Exp. Eye Res. 2013, 107, 65–73. [Google Scholar] [CrossRef]
- Lehmann, G.M.; Feldon, S.E.; Smith, T.J.; Phipps, R.P. Immune mechanisms in thyroid eye disease. Thyroid 2008, 18, 959–965. [Google Scholar] [CrossRef]
- Heufelder, A.E.; Bahn, R.S. Modulation of Graves’ orbital fibroblast proliferation by cytokines and glucocorticoid receptor agonists. Investig. Ophthalmol. Vis. Sci. 1994, 35, 120–127. [Google Scholar]
- Koumas, L.; Smith, T.J.; Feldon, S.; Blumberg, N.; Phipps, R.P. Thy-1 expression in human fibroblast subsets defines myofibroblastic or lipofibroblastic phenotypes. Am. J. Pathol. 2003, 163, 1291–1300. [Google Scholar] [CrossRef]
- Tan, J.; Tong, B.D.; Wu, Y.J.; Xiong, W. MicroRNA-29 mediates TGFbeta1-induced extracellular matrix synthesis by targeting wnt/beta-catenin pathway in human orbital fibroblasts. Int. J. Clin. Exp. Pathol. 2014, 7, 7571–7577. [Google Scholar]
- Frangogiannis, N. Transforming growth factor-beta in tissue fibrosis. J. Exp. Med. 2020, 217, e20190103. [Google Scholar] [CrossRef]
- Robertson, I.B.; Horiguchi, M.; Zilberberg, L.; Dabovic, B.; Hadjiolova, K.; Rifkin, D.B. Latent TGF-beta-binding proteins. Matrix Biol. 2015, 47, 44–53. [Google Scholar] [CrossRef]
- Pardo, A.; Cabrera, S.; Maldonado, M.; Selman, M. Role of matrix metalloproteinases in the pathogenesis of idiopathic pulmonary fibrosis. Respir. Res. 2016, 17, 23. [Google Scholar] [CrossRef]
- Budi, E.H.; Duan, D.; Derynck, R. Transforming Growth Factor-beta Receptors and Smads: Regulatory Complexity and Functional Versatility. Trends Cell Biol. 2017, 27, 658–672. [Google Scholar] [CrossRef]
- Finnson, K.W.; Almadani, Y.; Philip, A. Non-canonical (non-SMAD2/3) TGF-beta signaling in fibrosis: Mechanisms and targets. Semin. Cell Dev. Biol. 2020, 101, 115–122. [Google Scholar] [CrossRef]
- Hou, T.Y.; Wu, S.B.; Kau, H.C.; Tsai, C.C. JNK and p38 Inhibitors Prevent Transforming Growth Factor-beta1-Induced Myofibroblast Transdifferentiation in Human Graves’ Orbital Fibroblasts. Int. J. Mol. Sci. 2021, 22, 2952. [Google Scholar] [CrossRef]
- Tsai, C.C.; Wu, S.B.; Kau, H.C.; Wei, Y.H. Essential role of connective tissue growth factor (CTGF) in transforming growth factor-beta1 (TGF-beta1)-induced myofibroblast transdifferentiation from Graves’ orbital fibroblasts. Sci. Rep. 2018, 8, 7276. [Google Scholar] [CrossRef]
- Huang, Y.M.; Chang, P.C.; Wu, S.B.; Kau, H.C.; Tsai, C.C.; Liu, C.J.; Wei, Y.H. Expression and clinical significance of connective tissue growth factor (CTGF) in Graves’ ophthalmopathy. Br. J. Ophthalmol. 2017, 101, 676–680. [Google Scholar] [CrossRef]
- Tsai, C.C.; Wu, S.B.; Chang, P.C.; Wei, Y.H. Alteration of Connective Tissue Growth Factor (CTGF) Expression in Orbital Fibroblasts from Patients with Graves’ Ophthalmopathy. PLoS ONE 2015, 10, e0143514. [Google Scholar] [CrossRef]
- Horan, G.S.; Wood, S.; Ona, V.; Li, D.J.; Lukashev, M.E.; Weinreb, P.H.; Simon, K.J.; Hahm, K.; Allaire, N.E.; Rinaldi, N.J.; et al. Partial inhibition of integrin alpha(v)beta6 prevents pulmonary fibrosis without exacerbating inflammation. Am. J. Respir. Crit. Care Med. 2008, 177, 56–65. [Google Scholar] [CrossRef]
- Saito, A.; Horie, M.; Nagase, T. TGF-beta Signaling in Lung Health and Disease. Int. J. Mol. Sci. 2018, 19, 2460. [Google Scholar] [CrossRef]
- Smith, T.J.; Bahn, R.S.; Gorman, C.A. Connective tissue, glycosaminoglycans, and diseases of the thyroid. Endocr. Rev. 1989, 10, 366–391. [Google Scholar] [CrossRef]
- Zhang, L.; Bowen, T.; Grennan-Jones, F.; Paddon, C.; Giles, P.; Webber, J.; Steadman, R.; Ludgate, M. Thyrotropin receptor activation increases hyaluronan production in preadipocyte fibroblasts: Contributory role in hyaluronan accumulation in thyroid dysfunction. J. Biol. Chem. 2009, 284, 26447–26455. [Google Scholar] [CrossRef]
- van Steensel, L.; Paridaens, D.; van Meurs, M.; van Hagen, P.M.; van den Bosch, W.A.; Kuijpers, R.W.; Drexhage, H.A.; Hooijkaas, H.; Dik, W.A. Orbit-infiltrating mast cells, monocytes, and macrophages produce PDGF isoforms that orchestrate orbital fibroblast activation in Graves’ ophthalmopathy. J. Clin. Endocrinol. Metab. 2012, 97, E400–E408. [Google Scholar] [CrossRef]
- van Steensel, L.; Hooijkaas, H.; Paridaens, D.; van den Bosch, W.A.; Kuijpers, R.W.; Drexhage, H.A.; van Hagen, P.M.; Dik, W.A. PDGF enhances orbital fibroblast responses to TSHR stimulating autoantibodies in Graves’ ophthalmopathy patients. J. Clin. Endocrinol. Metab. 2012, 97, E944–E953. [Google Scholar] [CrossRef]
- Galgoczi, E.; Katko, M.; Papp, F.R.; Csiki, R.; Csiha, S.; Erdei, A.; Bodor, M.; Ujhelyi, B.; Steiber, Z.; Gyory, F.; et al. Glucocorticoids Directly Affect Hyaluronan Production of Orbital Fibroblasts; A Potential Pleiotropic Effect in Graves’ Orbitopathy. Molecules 2022, 28, 15. [Google Scholar] [CrossRef]
- Lim, H.S.; Back, K.O.; Kim, H.J.; Choi, Y.H.; Park, Y.M.; Kook, K.H. Hyaluronic acid induces COX-2 expression via CD44 in orbital fibroblasts from patients with thyroid-associated ophthalmopathy. Investig. Ophthalmol. Vis. Sci. 2014, 55, 7441–7450. [Google Scholar] [CrossRef][Green Version]
- Zhang, L.; Grennan-Jones, F.; Lane, C.; Rees, D.A.; Dayan, C.M.; Ludgate, M. Adipose tissue depot-specific differences in the regulation of hyaluronan production of relevance to Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 2012, 97, 653–662. [Google Scholar] [CrossRef] [PubMed]
- Guo, N.; Baglole, C.J.; O’Loughlin, C.W.; Feldon, S.E.; Phipps, R.P. Mast cell-derived prostaglandin D2 controls hyaluronan synthesis in human orbital fibroblasts via DP1 activation: Implications for thyroid eye disease. J. Biol. Chem. 2010, 285, 15794–15804. [Google Scholar] [CrossRef] [PubMed]
- Marcocci, C.; Leo, M.; Altea, M.A. Oxidative stress in graves’ disease. Eur. Thyroid J. 2012, 1, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Lanzolla, G.; Marcocci, C.; Marino, M. Oxidative Stress in Graves Disease and Graves Orbitopathy. Eur. Thyroid J. 2020, 9 (Suppl. S1), 40–50. [Google Scholar] [CrossRef]
- Tsai, C.C.; Kao, S.C.; Cheng, C.Y.; Kau, H.C.; Hsu, W.M.; Lee, C.F.; Wei, Y.H. Oxidative stress change by systemic corticosteroid treatment among patients having active graves ophthalmopathy. Arch. Ophthalmol. 2007, 125, 1652–1656. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.C.; Wu, S.B.; Cheng, C.Y.; Kao, S.C.; Kau, H.C.; Chiou, S.H.; Hsu, W.M.; Wei, Y.H. Increased oxidative DNA damage, lipid peroxidation, and reactive oxygen species in cultured orbital fibroblasts from patients with Graves’ ophthalmopathy: Evidence that oxidative stress has a role in this disorder. Eye 2010, 24, 1520–1525. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.C.; Wu, S.B.; Cheng, C.Y.; Kao, S.C.; Kau, H.C.; Lee, S.M.; Wei, Y.H. Increased response to oxidative stress challenge in Graves’ ophthalmopathy orbital fibroblasts. Mol. Vis. 2011, 17, 2782–2788. [Google Scholar] [PubMed]
- Akarsu, E.; Buyukhatipoglu, H.; Aktaran, S.; Kurtul, N. Effects of pulse methylprednisolone and oral methylprednisolone treatments on serum levels of oxidative stress markers in Graves’ ophthalmopathy. Clin. Endocrinol. 2011, 74, 118–124. [Google Scholar] [CrossRef]
- Abalovich, M.; Llesuy, S.; Gutierrez, S.; Repetto, M. Peripheral parameters of oxidative stress in Graves’ disease: The effects of methimazole and 131 iodine treatments. Clin. Endocrinol. 2003, 59, 321–327. [Google Scholar] [CrossRef]
- Bednarek, J.; Wysocki, H.; Sowinski, J. Oxidative stress peripheral parameters in Graves’ disease: The effect of methimazole treatment in patients with and without infiltrative ophthalmopathy. Clin. Biochem. 2005, 38, 13–18. [Google Scholar] [CrossRef]
- Yuksel, N.; Yaman, D.; Tugce Pasaoglu, O.; Pasaoglu, H. The Effect of Smoking on Mitochondrial Biogenesis in Patients with Graves Ophthalmopathy. Ophthalmic Plast. Reconstr. Surg. 2020, 36, 172–177. [Google Scholar] [CrossRef]
- Cawood, T.J.; Moriarty, P.; O’Farrelly, C.; O’Shea, D. Smoking and thyroid-associated ophthalmopathy: A novel explanation of the biological link. J. Clin. Endocrinol. Metab. 2007, 92, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Tehrani, M.J.; Bagher Abtahi, S.M. Mini Review on the Effect of Smoking on Retrobulbar Blood Flow in Thyroid Eye Disease. J. Clin. Exp. Ophthalmol. 2018, 9, 5. [Google Scholar] [CrossRef]
- Tamaki, Y.; Araie, M.; Nagahara, M.; Tomita, K. Acute effects of cigarette smoking on tissue circulation in human optic nerve head and choroid-retina. Ophthalmology 1999, 106, 564–569. [Google Scholar] [CrossRef]
- Gortz, G.E.; Horstmann, M.; Aniol, B.; Reyes, B.D.; Fandrey, J.; Eckstein, A.; Berchner-Pfannschmidt, U. Hypoxia-Dependent HIF-1 Activation Impacts on Tissue Remodeling in Graves’ Ophthalmopathy-Implications for Smoking. J. Clin. Endocrinol. Metab. 2016, 101, 4834–4842. [Google Scholar] [CrossRef] [PubMed]
- Kau, H.C.; Wu, S.B.; Tsai, C.C.; Liu, C.J.; Wei, Y.H. Cigarette Smoke Extract-Induced Oxidative Stress and Fibrosis-Related Genes Expression in Orbital Fibroblasts from Patients with Graves’ Ophthalmopathy. Oxid. Med. Cell Longev. 2016, 2016, 4676289. [Google Scholar] [CrossRef]
- Hung, C.T.; Tsai, Y.W.; Wu, Y.S.; Yeh, C.F.; Yang, K.C. The novel role of ER protein TXNDC5 in the pathogenesis of organ fibrosis: Mechanistic insights and therapeutic implications. J. Biomed. Sci. 2022, 29, 63. [Google Scholar] [CrossRef]
- Huang, J.; Chen, M.; Liang, Y.; Hu, Y.; Xia, W.; Zhang, Y.; Zhao, C.; Wu, L. Integrative metabolic analysis of orbital adipose/connective tissue in patients with thyroid-associated ophthalmopathy. Front. Endocrinol. 2022, 13, 1001349. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.; Kim, J.Y.; Kyoung Chae, M.; Jig Lee, E.; Sook Yoon, J. PERK mediates oxidative stress and adipogenesis in Graves’ orbitopathy pathogenesis. J. Mol. Endocrinol. 2021, 66, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ma, X.M.; Wang, X.; Sun, X.; Wang, L.J.; Li, X.Q.; Liu, X.Y.; Yu, H.S. Emerging Insights Into the Role of Epigenetics and Gut Microbiome in the Pathogenesis of Graves’ Ophthalmopathy. Front. Endocrinol. 2021, 12, 788535. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Duan, H.; You, S.; Liang, B.; Chen, Y.; Huang, H. Research progress on the pathogenesis of Graves’ ophthalmopathy: Based on immunity, noncoding RNA and exosomes. Front. Immunol. 2022, 13, 952954. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.L.; Lin, Z.J.; Li, C.C.; Lin, X.; Shan, S.K.; Guo, B.; Zheng, M.H.; Li, F.; Yuan, L.Q.; Li, Z.H. Epigenetic regulation in metabolic diseases: Mechanisms and advances in clinical study. Signal Transduct. Target. Ther. 2023, 8, 98. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, J.S. A short guide to histone deacetylases including recent progress on class II enzymes. Exp. Mol. Med. 2020, 52, 204–212. [Google Scholar] [CrossRef]
- Haberland, M.; Montgomery, R.L.; Olson, E.N. The many roles of histone deacetylases in development and physiology: Implications for disease and therapy. Nat. Rev. Genet. 2009, 10, 32–42. [Google Scholar] [CrossRef]
- Limbach, M.; Saare, M.; Tserel, L.; Kisand, K.; Eglit, T.; Sauer, S.; Axelsson, T.; Syvanen, A.C.; Metspalu, A.; Milani, L.; et al. Epigenetic profiling in CD4+ and CD8+ T cells from Graves’ disease patients reveals changes in genes associated with T cell receptor signaling. J. Autoimmun. 2016, 67, 46–56. [Google Scholar] [CrossRef]
- Sacristan-Gomez, P.; Serrano-Somavilla, A.; Gonzalez-Amaro, R.; Martinez-Hernandez, R.; Marazuela, M. Analysis of Expression of Different Histone Deacetylases in Autoimmune Thyroid Disease. J. Clin. Endocrinol. Metab. 2021, 106, 3213–3227. [Google Scholar] [CrossRef]
- Ekronarongchai, S.; Palaga, T.; Saonanon, P.; Pruksakorn, V.; Hirankarn, N.; van Hagen, P.M.; Dik, W.A.; Virakul, S. Histone Deacetylase 4 Controls Extracellular Matrix Production in Orbital Fibroblasts from Graves’ Ophthalmopathy Patients. Thyroid 2021, 31, 1566–1576. [Google Scholar] [CrossRef] [PubMed]
- Woeller, C.F.; Roztocil, E.; Hammond, C.; Feldon, S.E. TSHR Signaling Stimulates Proliferation Through PI3K/Akt and Induction of miR-146a and miR-155 in Thyroid Eye Disease Orbital Fibroblasts. Investig. Ophthalmol. Vis. Sci. 2019, 60, 4336–4345. [Google Scholar] [CrossRef]
- Jang, S.Y.; Park, S.J.; Chae, M.K.; Lee, J.H.; Lee, E.J.; Yoon, J.S. Role of microRNA-146a in regulation of fibrosis in orbital fibroblasts from patients with Graves’ orbitopathy. Br. J. Ophthalmol. 2018, 102, 407–414. [Google Scholar] [CrossRef]
- Wei, H.; Guan, M.; Qin, Y.; Xie, C.; Fu, X.; Gao, F.; Xue, Y. Circulating levels of miR-146a and IL-17 are significantly correlated with the clinical activity of Graves’ ophthalmopathy. Endocr. J. 2014, 61, 1087–1092. [Google Scholar] [CrossRef]
- Li, K.; Du, Y.; Jiang, B.L.; He, J.F. Increased microRNA-155 and decreased microRNA-146a may promote ocular inflammation and proliferation in Graves’ ophthalmopathy. Med. Sci. Monit. 2014, 20, 639–643. [Google Scholar]
- Eissa, M.G.; Artlett, C.M. The MicroRNA miR-155 Is Essential in Fibrosis. Noncoding RNA 2019, 5, 23. [Google Scholar] [CrossRef] [PubMed]
- Hammond, C.L.; Roztocil, E.; Gonzalez, M.O.; Feldon, S.E.; Woeller, C.F. MicroRNA-130a Is Elevated in Thyroid Eye Disease and Increases Lipid Accumulation in Fibroblasts Through the Suppression of AMPK. Investig. Ophthalmol. Vis. Sci. 2021, 62, 29. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.Y.; Chae, M.K.; Lee, J.H.; Lee, E.J.; Yoon, J.S. MicroRNA-27 inhibits adipogenic differentiation in orbital fibroblasts from patients with Graves’ orbitopathy. PLoS ONE 2019, 14, e0221077. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Yun, M.; Paik, J.S.; Lee, S.B.; Yang, S.W. PDGF-BB Enhances the Proliferation of Cells in Human Orbital Fibroblasts by Suppressing PDCD4 Expression Via Up-Regulation of microRNA-21. Investig. Ophthalmol. Vis. Sci. 2016, 57, 908–913. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B.; Jensen, T.I.; Clausen, B.H.; Bramsen, J.B.; Finsen, B.; Damgaard, C.K.; Kjems, J. Natural RNA circles function as efficient microRNA sponges. Nature 2013, 495, 384–388. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Zhou, R.; Diao, J.; Chen, X.; Huang, J.; Xu, K.; Ling, L.; Xia, W.; Liang, Y.; Liu, G.; et al. Differentially expressed circular RNAs in orbital adipose/connective tissue from patients with thyroid-associated ophthalmopathy. Exp. Eye Res. 2020, 196, 108036. [Google Scholar] [CrossRef] [PubMed]
- Christensen, N.J.; Habekost, G.; Bratholm, P. A RNA transcript (Heg) in mononuclear cells is negatively correlated with CD14 mRNA and TSH receptor autoantibodies. Clin. Exp. Immunol. 2008, 154, 209–215. [Google Scholar] [CrossRef]
- Wu, L.; Li, L.; Liang, Y.; Chen, X.; Mou, P.; Liu, G.; Sun, X.; Qin, B.; Zhang, S.; Zhao, C. Identification of differentially expressed long non-coding RNAs and mRNAs in orbital adipose/connective tissue of thyroid-associated ophthalmopathy. Genomics 2021, 113 Pt 2, 440–449. [Google Scholar] [CrossRef]
- Su, X.; Yin, X.; Liu, Y.; Yan, X.; Zhang, S.; Wang, X.; Lin, Z.; Zhou, X.; Gao, J.; Wang, Z.; et al. Gut Dysbiosis Contributes to the Imbalance of Treg and Th17 Cells in Graves’ Disease Patients by Propionic Acid. J. Clin. Endocrinol. Metab. 2020, 105, 3526–3547. [Google Scholar] [CrossRef] [PubMed]
- Masetti, G.; Moshkelgosha, S.; Kohling, H.L.; Covelli, D.; Banga, J.P.; Berchner-Pfannschmidt, U.; Horstmann, M.; Diaz-Cano, S.; Goertz, G.E.; Plummer, S.; et al. Gut microbiota in experimental murine model of Graves’ orbitopathy established in different environments may modulate clinical presentation of disease. Microbiome 2018, 6, 97. [Google Scholar] [CrossRef] [PubMed]
- Moshkelgosha, S.; Verhasselt, H.L.; Masetti, G.; Covelli, D.; Biscarini, F.; Horstmann, M.; Daser, A.; Westendorf, A.M.; Jesenek, C.; Philipp, S.; et al. Modulating gut microbiota in a mouse model of Graves’ orbitopathy and its impact on induced disease. Microbiome 2021, 9, 45. [Google Scholar] [CrossRef] [PubMed]
- Kahaly, G.J.; Douglas, R.S.; Holt, R.J.; Sile, S.; Smith, T.J. Teprotumumab for patients with active thyroid eye disease: A pooled data analysis, subgroup analyses, and off-treatment follow-up results from two randomised, double-masked, placebo-controlled, multicentre trials. Lancet Diabetes Endocrinol. 2021, 9, 360–372. [Google Scholar] [CrossRef] [PubMed]
- Salvi, M.; Vannucchi, G.; Curro, N.; Campi, I.; Covelli, D.; Dazzi, D.; Simonetta, S.; Guastella, C.; Pignataro, L.; Avignone, S.; et al. Efficacy of B-cell targeted therapy with rituximab in patients with active moderate to severe Graves’ orbitopathy: A randomized controlled study. J. Clin. Endocrinol. Metab. 2015, 100, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Stan, M.N.; Garrity, J.A.; Carranza Leon, B.G.; Prabin, T.; Bradley, E.A.; Bahn, R.S. Randomized controlled trial of rituximab in patients with Graves’ orbitopathy. J. Clin. Endocrinol. Metab. 2015, 100, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Jyonouchi, S.C.; Valyasevi, R.W.; Harteneck, D.A.; Dutton, C.M.; Bahn, R.S. Interleukin-6 stimulates thyrotropin receptor expression in human orbital preadipocyte fibroblasts from patients with Graves’ ophthalmopathy. Thyroid 2001, 11, 929–934. [Google Scholar] [CrossRef]
- Akbarian, S.; Chaibakhsh, S.; Kashkouli, M.B.; Karimi, N.; Abdolalizadeh, P.; Ghahvehchian, H. A Systematic Review on the Role of Antioxidants in Thyroid Eye Disease. J. Curr. Ophthalmol. 2022, 34, 16–24. [Google Scholar]
- Hou, T.Y.; Wu, S.B.; Kau, H.C.; Tsai, C.C. The Role of Oxidative Stress and Therapeutic Potential of Antioxidants in Graves’ Ophthalmopathy. Biomedicines 2021, 9, 1871. [Google Scholar] [CrossRef]
- Lanzolla, G.; Marcocci, C.; Marino, M. Antioxidant Therapy in Graves’ Orbitopathy. Front. Endocrinol. 2020, 11, 608733. [Google Scholar] [CrossRef]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Marcocci, C.; Kahaly, G.J.; Krassas, G.E.; Bartalena, L.; Prummel, M.; Stahl, M.; Altea, M.A.; Nardi, M.; Pitz, S.; Boboridis, K.; et al. Selenium and the course of mild Graves’ orbitopathy. N. Engl. J. Med. 2011, 364, 1920–1931. [Google Scholar] [CrossRef]
- Kahaly, G.J.; Bartalena, L.; Hegedus, L.; Leenhardt, L.; Poppe, K.; Pearce, S.H. 2018 European Thyroid Association Guideline for the Management of Graves’ Hyperthyroidism. Eur. Thyroid J. 2018, 7, 167–186. [Google Scholar] [CrossRef] [PubMed]
- Ross, D.S.; Burch, H.B.; Cooper, D.S.; Greenlee, M.C.; Laurberg, P.; Maia, A.L.; Rivkees, S.A.; Samuels, M.; Sosa, J.A.; Stan, M.N.; et al. 2016 American Thyroid Association Guidelines for Diagnosis and Management of Hyperthyroidism and Other Causes of Thyrotoxicosis. Thyroid 2016, 26, 1343–1421. [Google Scholar] [CrossRef] [PubMed]
- Karalis, D.T. The Beneficiary Role of Selenium in Type II Diabetes: A Longitudinal Study. Cureus 2019, 11, e6443. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Zeng, C.; Gong, Q.Y.; Yang, H.B.; Li, X.X.; Lei, G.H.; Yang, T.B. The association between dietary selenium intake and diabetes: A cross-sectional study among middle-aged and older adults. Nutr. J. 2015, 14, 18. [Google Scholar] [CrossRef] [PubMed]
- Bednarczuk, T.; Schomburg, L. Challenges and perspectives of selenium supplementation in Graves’ disease and orbitopathy. Hormones 2020, 19, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Lanzolla, G.; Marino, M.; Marcocci, C. Selenium in the Treatment of Graves’ Hyperthyroidism and Eye Disease. Front. Endocrinol. 2020, 11, 608428. [Google Scholar] [CrossRef]
- Chang, C.C.; Chang, T.C.; Kao, S.C.; Kuo, Y.F.; Chien, L.F. Pentoxifylline inhibits the proliferation and glycosaminoglycan synthesis of cultured fibroblasts derived from patients with Graves’ ophthalmopathy and pretibial myxoedema. Acta Endocrinol. 1993, 129, 322–327. [Google Scholar] [CrossRef] [PubMed]
- Balazs, C.; Kiss, H.; Farid, N.R. Inhibitory effect of pentoxifylline on HLA-DR expression and glycosaminoglycan synthesis by retrobulbar fibroblasts. Horm. Metab. Res. 1998, 30, 496–499. [Google Scholar] [CrossRef]
- Balazs, C.; Kiss, E.; Vamos, A.; Molnar, I.; Farid, N.R. Beneficial effect of pentoxifylline on thyroid associated ophthalmopathy (TAO)*: A pilot study. J. Clin. Endocrinol. Metab. 1997, 82, 1999–2002. [Google Scholar] [CrossRef]
- Finamor, F.E.; Martins, J.R.M.; Nakanami, D.; Paiva, E.R.; Manso, P.G.; Furlanetto, R.P. Pentoxifylline (PTX)—An Alternative Treatment in Graves’ Ophthalmopathy (Inactive Phase): Assessment by a Disease Specific Quality of Life Questionnaire and by Exophthalmometry in a Prospective Randomized Trial. Eur. J. Ophthalmol. 2018, 14, 277–283. [Google Scholar] [CrossRef]
- Hiromatsu, Y.; Yang, D.; Miyake, I.; Koga, M.; Kameo, J.; Sato, M.; Inoue, Y.; Nonaka, K. Nicotinamide decreases cytokine-induced activation of orbital fibroblasts from patients with thyroid-associated ophthalmopathy. J. Clin. Endocrinol. Metab. 1998, 83, 121–124. [Google Scholar] [CrossRef]
- Burch, H.B.; Lahiri, S.; Bahn, R.S.; Barnes, S. Superoxide radical production stimulates retroocular fibroblast proliferation in Graves’ ophthalmopathy. Exp. Eye Res. 1997, 65, 311–316. [Google Scholar] [CrossRef]
- Sautin, Y.Y.; Johnson, R.J. Uric acid: The oxidant-antioxidant paradox. Nucleosides Nucleotides Nucleic Acids 2008, 27, 608–619. [Google Scholar] [CrossRef]
- Makay, O.; Yenisey, C.; Icoz, G.; Genc Simsek, N.; Ozgen, G.; Akyildiz, M.; Yetkin, E. The role of allopurinol on oxidative stress in experimental hyperthyroidism. J. Endocrinol. Investig. 2009, 32, 641–646. [Google Scholar] [CrossRef]
- Bouzas, E.A.; Karadimas, P.; Mastorakos, G.; Koutras, D.A. Antioxidant agents in the treatment of Graves’ ophthalmopathy. Am. J. Ophthalmol. 2000, 129, 618–622. [Google Scholar] [CrossRef]
- Choi, W.; Yang, Y.S.; Chang, D.J.; Chung, Y.W.; Kim, H.; Ko, S.J.; Yoo, S.; Oh, J.S.; Kang, D.Y.; Yang, H.J.; et al. Association between the use of allopurinol and risk of increased thyroid-stimulating hormone level. Sci. Rep. 2021, 11, 20305. [Google Scholar] [CrossRef] [PubMed]
- Chung, S.A.; Jeon, B.K.; Choi, Y.H.; Back, K.O.; Lee, J.B.; Kook, K.H. Pirfenidone attenuates the IL-1beta-induced hyaluronic acid increase in orbital fibroblasts from patients with thyroid-associated ophthalmopathy. Investig. Ophthalmol. Vis. Sci. 2014, 55, 2276–2283. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Choi, Y.H.; Back, K.O.; Kim, H.J.; Lee, S.Y.; Kook, K.H. Pirfenidone attenuates IL-1beta-induced COX-2 and PGE2 production in orbital fibroblasts through suppression of NF-kappaB activity. Exp. Eye Res. 2013, 113, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Choi, Y.H.; Park, S.J.; Lee, S.Y.; Kim, S.J.; Jou, I.; Kook, K.H. Antifibrotic effect of Pirfenidone on orbital fibroblasts of patients with thyroid-associated ophthalmopathy by decreasing TIMP-1 and collagen levels. Investig. Ophthalmol. Vis. Sci. 2010, 51, 3061–3066. [Google Scholar] [CrossRef]
- Wu, S.B.; Hou, T.Y.; Kau, H.C.; Tsai, C.C. Effect of Pirfenidone on TGF-beta1-Induced Myofibroblast Differentiation and Extracellular Matrix Homeostasis of Human Orbital Fibroblasts in Graves’ Ophthalmopathy. Biomolecules 2021, 11, 1424. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, R.; Xiong, W.; Zhang, F.; Wang, N.; Xie, B.; Cao, J.; Chen, Z.; Ma, C. RNA aptamers with specific binding affinity to CD40 (CD40Apt) represents a promising antagonist of the CD40-CD40L signaling for thyroid-associated ophthalmopathy (TAO) treatment in mouse. J. Transl. Med. 2023, 21, 396. [Google Scholar] [CrossRef]
| Therapy, FDA Approved [Reference] | Mechanism | Study Design | Disease Status | Outcome | Adverse Effect |
| Teprotumumab [2] | Monoclonal antibody against insulin-like growth factor-1 receptor (IGF-1R) | RCT (n = 41) iv 10 mg/kg initially, followed by 20 mg/kg in 3 weeks for 7 additional infusions | Moderate to severe | Improved QOL and proptosis Improvement (83% vs. 10% p < 0.001) after 24 weeks | (1) Hyperlipidemia (2) Hearing impairment |
| Therapeutic agents with RCT [reference] | Mechanism | Study design | Disease Status | Outcome | Adverse effect |
| Rituximab [77] | Monoclonal antibody against CD20 | RCT (n = 31) iv 2000 mg or 500 mg control: iv methylprednisolone | Moderate to severe | Better ocular motility and QOL after 24 weeks | (1) Infusion reaction (2) Transient hypotension |
| [78] | RCT (n = 13) iv 1000 mg twice in 2 weeks control: placebo | Moderate to severe | No benefit | Infectious bronchitis, conjunctivitis, vasculitis, optic neuropathy, gastrointestinal disorder | |
| Selenium [85] | Antioxidant | RCT (n = 54) 200 μg/day for 6 months | Mild | Improved QOL and ocular sign, decreased progression | (−) |
| Pentoxifylline [85] | Antioxidant | RCT (n = 48) 1200 mg/day for 6 months | Mild | No benefit | Mild gastrointestinal and skin disorders |
| [95] | RCT (n = 9) 1200 mg/day for 6 months | Inactive | QOL and proptosis improvement | Mild gastrointestinal disorder | |
| Nicotinamide and Allopurinol [100] | Antioxidant | Nonrandomized comparative study (n = 11) Oral allopurinol (300 mg/day) and nicotinamide (300 mg/day) for 3 months | Mild to moderate | Improved soft tissue inflammation | (−) |
| Potential therapeutic agents without RCT [reference] | Mechanism | ||||
| Tasquinimod [60] | Inhibitor of histone deacetylases 4 (HDAC4), which decreased the mRNA expression of hyaluronan synthase | ||||
| Tocilizumab [79] | Monoclonal interleukin-6 receptor antagonist | ||||
| Pirfenidone [102,103,104,105] | (1) Decreased inflammation by attenuation of COX-2, prostaglandin E2 (2) Decreased ECM production | ||||
| CD40Apt [106] | Specific binding affinity to CD40 represents a promising inhibitor of the CD40-CD40L signaling | ||||
| Potential therapeutic targets [reference] | Mechanism | ||||
| Antagonist of peroxisome proliferator-activated receptor γ (PPARγ) [10,11,12] | Inhibition of adipogenesis | ||||
| Antagonist of transforming growth factor-β1 (TGF-β1) downstream [22,23,24,25] | Inhibition of TGF-β-Induced myofibroblast transdifferentiation in orbital fibroblasts and ECM production | ||||
| Antagonist of thioredoxin domain-containing 5 (TXNDC5) [our study, not published] | Reduction in ER stress and tissue remodeling | ||||
| Non-coding RNA [61,62,63,64,65,66,67,68,69,70,71,72] | Decreased levels of mi146a: decreased inflammation, decreased fibrosis in GO Increased levels of mi155: decreased fibrosis in GO others (miR-29, miR-21, MiR-27a, miR-27b, miR-130a, circRNA, LncRNA) | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, H.-I.; Wu, S.-B.; Tsai, C.-C. The Role of Fibrogenesis and Extracellular Matrix Proteins in the Pathogenesis of Graves’ Ophthalmopathy. Int. J. Mol. Sci. 2024, 25, 3288. https://doi.org/10.3390/ijms25063288
Chiu H-I, Wu S-B, Tsai C-C. The Role of Fibrogenesis and Extracellular Matrix Proteins in the Pathogenesis of Graves’ Ophthalmopathy. International Journal of Molecular Sciences. 2024; 25(6):3288. https://doi.org/10.3390/ijms25063288
Chicago/Turabian StyleChiu, Hsun-I, Shi-Bei Wu, and Chieh-Chih Tsai. 2024. "The Role of Fibrogenesis and Extracellular Matrix Proteins in the Pathogenesis of Graves’ Ophthalmopathy" International Journal of Molecular Sciences 25, no. 6: 3288. https://doi.org/10.3390/ijms25063288
APA StyleChiu, H.-I., Wu, S.-B., & Tsai, C.-C. (2024). The Role of Fibrogenesis and Extracellular Matrix Proteins in the Pathogenesis of Graves’ Ophthalmopathy. International Journal of Molecular Sciences, 25(6), 3288. https://doi.org/10.3390/ijms25063288








