Metformin and Glucose Concentration as Limiting Factors in Retinal Pigment Epithelial Cell Viability and Proliferation
Abstract
1. Introduction
2. Results
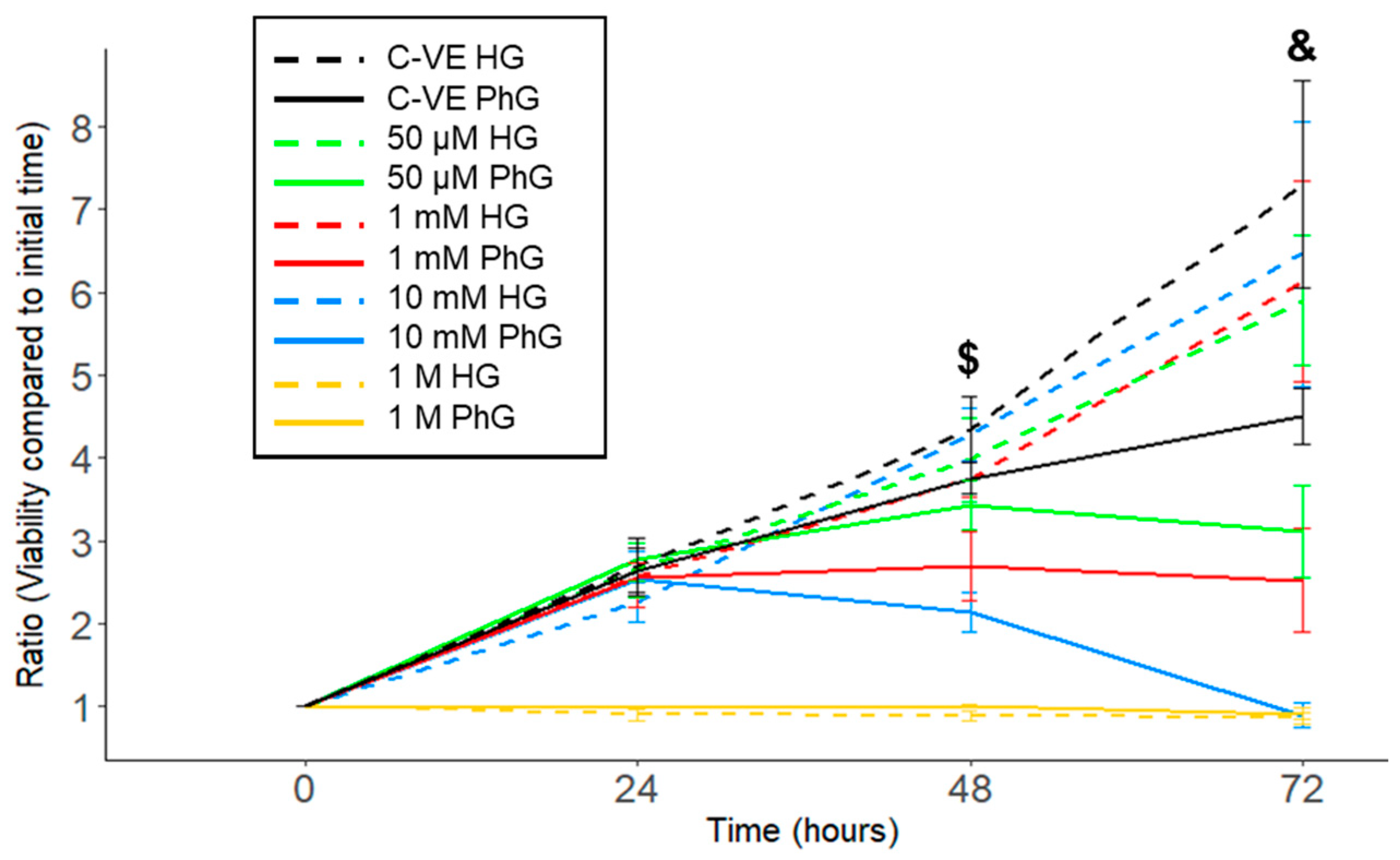
2.1. Influence of Glucose Media Concentration in hTERT RPE-1 Cell Viability
2.2. Metformin Concentration Affects Growth in a Glucose-Dependent Manner
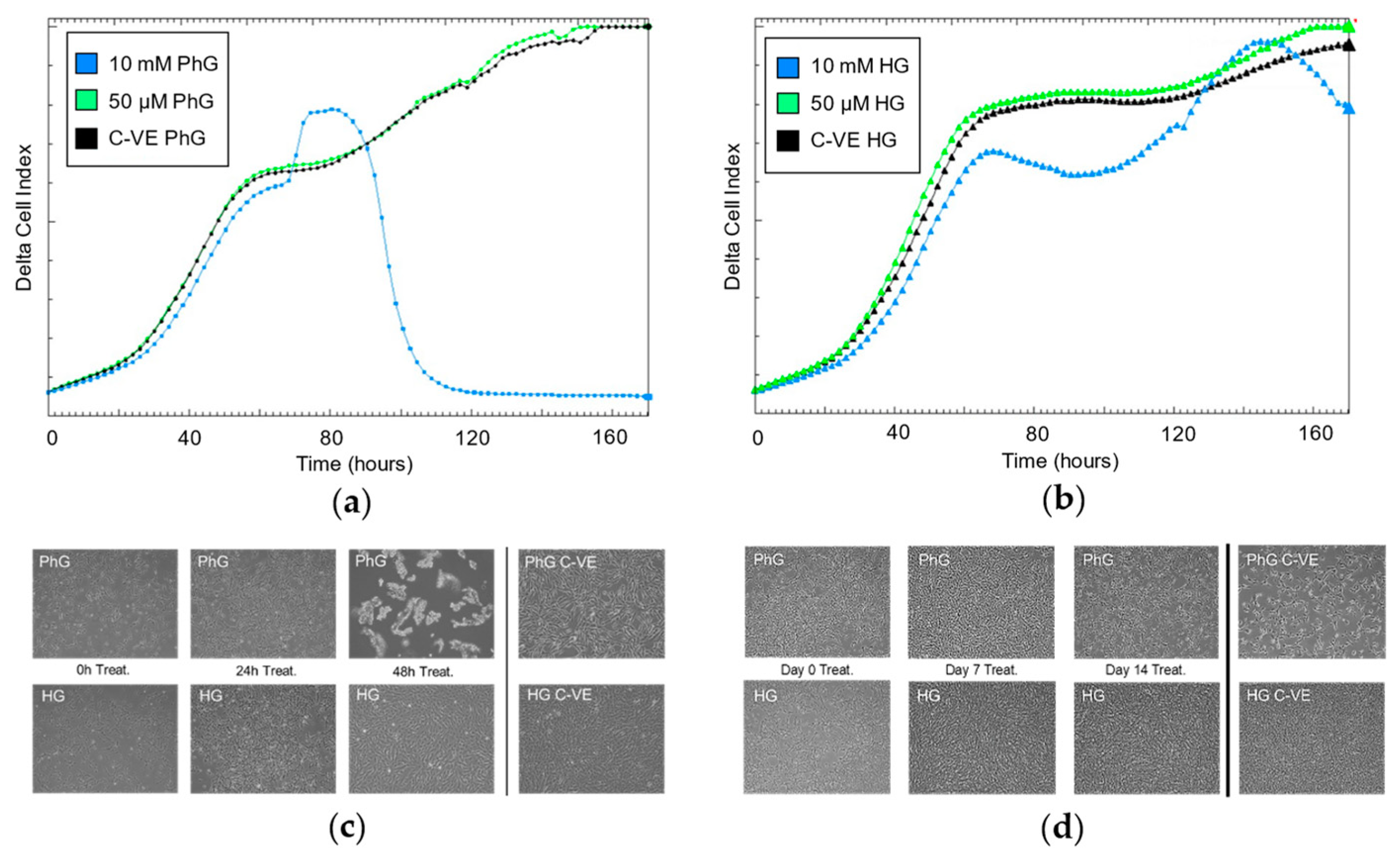
2.3. Influence of Glucose and Metformin Concentration on Cell Proliferation Rate
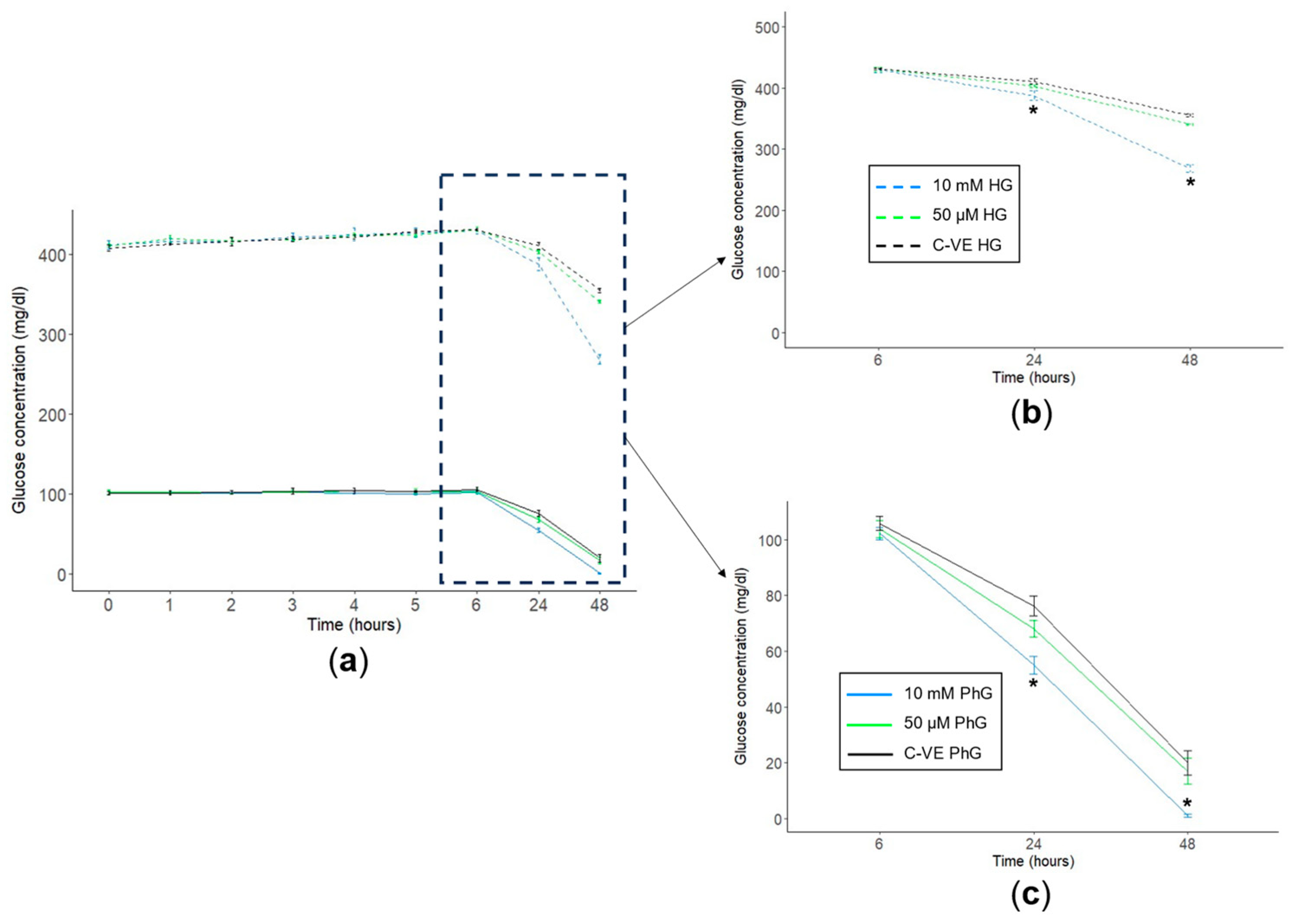
2.4. Metformin Concentration Influences Glucose Consumption in Cell Cultures
3. Discussion
Limitation of the Study
4. Materials and Methods
4.1. Cell Line
4.2. Cell Viability Assays
4.3. Cell Cultures
4.4. Proliferation Analysis
4.5. Glucose Concentration
4.6. Statistics
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bailey, C.J. Metformin: Historical Overview. Diabetologia 2017, 60, 1566–1576. [Google Scholar] [CrossRef]
- Sterne, J. Du nouveau dans les antidiabétiques. La NN dimethylamine guanyl guanidine (N.N.D.G.). Maroc. Med. 1957, 36, 1295–1296. (In French) [Google Scholar]
- Inzucchi, S.E.; Bergenstal, R.M.; Buse, J.B.; Diamant, M.; Ferrannini, E.; Nauck, M.; Peters, A.L.; Tsapas, A.; Wender, R.; Matthews, D.R. Management of Hyperglycemia in Type 2 Diabetes: A Patient-Centered Approach. Diabetes Care 2012, 35, 1364–1379. [Google Scholar] [CrossRef]
- IDF Diabetes Atlas. Diabetes around the World in 2021. Available online: https://diabetesatlas.org/ (accessed on 20 December 2023).
- Goyal, R.; Jialal, I. Diabetes Mellitus Type 2; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Shukla, U.V.; Tripathy, K. Diabetic Retinopathy; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Diabetic Retinopathy-Europe. Available online: https://www.aao.org/education/topic-detail/diabetic-retinopathy-europe (accessed on 31 January 2024).
- Heng, L.Z.; Comyn, O.; Peto, T.; Tadros, C.; Ng, E.; Sivaprasad, S.; Hykin, P.G. Diabetic Retinopathy: Pathogenesis, Clinical Grading, Management and Future Developments. Diabet. Med. 2013, 30, 640–650. [Google Scholar] [CrossRef]
- Kern, T.S.; Berkowitz, B.A. Photoreceptors in Diabetic Retinopathy. J. Diabetes Investig. 2015, 6, 371–380. [Google Scholar] [CrossRef]
- Du, Y.; Veenstra, A.; Palczewski, K.; Kern, T.S. Photoreceptor Cells Are Major Contributors to Diabetes-Induced Oxidative Stress and Local Inflammation in the Retina. Proc. Natl. Acad. Sci. USA 2013, 110, 16586–16591. [Google Scholar] [CrossRef]
- Tonade, D.; Liu, H.; Kern, T.S. Photoreceptor Cells Produce Inflammatory Mediators That Contribute to Endothelial Cell Death in Diabetes. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4264–4271. [Google Scholar] [CrossRef]
- Tonade, D.; Kern, T.S. Photoreceptor Cells and RPE Contribute to the Development of Diabetic Retinopathy. Prog. Retin. Eye Res. 2021, 83, 100919. [Google Scholar] [CrossRef]
- Yerevanian, A.; Soukas, A.A. Metformin: Mechanisms in Human Obesity and Weight Loss. Curr. Obes. Rep. 2019, 8, 156–164. [Google Scholar] [CrossRef]
- Picón-César, M.J.; Molina-Vega, M.; Suárez-Arana, M.; González-Mesa, E.; Sola-Moyano, A.P.; Roldan-López, R.; Romero-Narbona, F.; Olveira, G.; Tinahones, F.J.; González-Romero, S. Metformin for Gestational Diabetes Study: Metformin vs Insulin in Gestational Diabetes: Glycemic Control and Obstetrical and Perinatal Outcomes: Randomized Prospective Trial. Am. J. Obstet. Gynecol. 2021, 225, 517.e1–517.e17. [Google Scholar] [CrossRef]
- Barthelmess, E.K.; Naz, R.K. Polycystic Ovary Syndrome: Current Status and Future Perspective. Front. Biosci. (Elite Ed.) 2014, 6, 104–119. [Google Scholar] [CrossRef]
- Guarnaccia, L.; Marfia, G.; Masseroli, M.M.; Navone, S.E.; Balsamo, M.; Caroli, M.; Valtorta, S.; Moresco, R.M.; Campanella, R.; Garzia, E.; et al. Frontiers in Anti-Cancer Drug Discovery: Challenges and Perspectives of Metformin as Anti-Angiogenic Add-On Therapy in Glioblastoma. Cancers 2021, 14, 112. [Google Scholar] [CrossRef]
- Urpilainen, E.; Puistola, U.; Boussios, S.; Karihtala, P. Metformin and Ovarian Cancer: The Evidence. Ann. Transl. Med. 2020, 8, 1711. [Google Scholar] [CrossRef]
- Ng, C.-A.W.; Jiang, A.A.; Toh, E.M.S.; Ng, C.H.; Ong, Z.H.; Peng, S.; Tham, H.Y.; Sundar, R.; Chong, C.S.; Khoo, C.M. Metformin and Colorectal Cancer: A Systematic Review, Meta-Analysis and Meta-Regression. Int. J. Colorectal Dis. 2020, 35, 1501–1512. [Google Scholar] [CrossRef]
- Chae, Y.K.; Arya, A.; Malecek, M.-K.; Shin, D.S.; Carneiro, B.; Chandra, S.; Kaplan, J.; Kalyan, A.; Altman, J.K.; Platanias, L.; et al. Repurposing Metformin for Cancer Treatment: Current Clinical Studies. Oncotarget 2016, 7, 40767–40780. [Google Scholar] [CrossRef]
- Lega, I.C.; Austin, P.C.; Gruneir, A.; Goodwin, P.J.; Rochon, P.A.; Lipscombe, L.L. Association between Metformin Therapy and Mortality after Breast Cancer: A Population-Based Study. Diabetes Care 2013, 36, 3018–3026. [Google Scholar] [CrossRef]
- He, L.; Wondisford, F.E. Metformin Action: Concentrations Matter. Cell Metab. 2015, 21, 159–162. [Google Scholar] [CrossRef]
- LaMoia, T.E.; Shulman, G.I. Cellular and Molecular Mechanisms of Metformin Action. Endocr. Rev. 2020, 42, 77–96. [Google Scholar] [CrossRef]
- He, L. Metformin and Systemic Metabolism. Trends Pharmacol. Sci. 2020, 41, 868–881. [Google Scholar] [CrossRef]
- Foretz, M.; Guigas, B.; Viollet, B. Metformin: Update on Mechanisms of Action and Repurposing Potential. Nat. Rev. Endocrinol. 2023, 19, 460–476. [Google Scholar] [CrossRef]
- Metformin. In LiverTox: Clinical and Research Information on Drug-Induced Liver Injury; National Institute of Diabetes and Digestive and Kidney Diseases: Bethesda, MD, USA, 2012.
- Zheng, Y.; Zhu, J.; Zhang, H.; Liu, Y.; Sun, H. Metformin plus First-Line Chemotherapy versus Chemotherapy Alone in the Treatment of Epithelial Ovarian Cancer: A Prospective Open-Label Pilot Trial. Cancer Chemother. Pharmacol. 2019, 84, 1349–1357. [Google Scholar] [CrossRef]
- Marini, C.; Cossu, V.; Bauckneht, M.; Lanfranchi, F.; Raffa, S.; Orengo, A.M.; Ravera, S.; Bruno, S.; Sambuceti, G. Metformin and Cancer Glucose Metabolism: At the Bench or at the Bedside? Biomolecules 2021, 11, 1231. [Google Scholar] [CrossRef]
- Mohamed Suhaimi, N.-A.; Phyo, W.M.; Yap, H.Y.; Choy, S.H.Y.; Wei, X.; Choudhury, Y.; Tan, W.J.; Tan, L.A.P.Y.; Foo, R.S.Y.; Tan, S.H.S.; et al. Metformin Inhibits Cellular Proliferation and Bioenergetics in Colorectal Cancer Patient–Derived Xenografts. Mol. Cancer Ther. 2017, 16, 2035–2044. [Google Scholar] [CrossRef]
- Hua, Y.; Zheng, Y.; Yao, Y.; Jia, R.; Ge, S.; Zhuang, A. Metformin and Cancer Hallmarks: Shedding New Lights on Therapeutic Repurposing. J. Transl. Med. 2023, 21, 403. [Google Scholar] [CrossRef]
- Bayrak, B.; Koroglu, P.; Karabulut Bulan, O.; Yanardag, R. Metformin Protects against Diabetes-Induced Heart Injury and Dunning Prostate Cancer Model. Hum. Exp. Toxicol. 2021, 40, 297–309. [Google Scholar] [CrossRef]
- Alhourani, A.H.; Tidwell, T.R.; Bokil, A.A.; Røsland, G.V.; Tronstad, K.J.; Søreide, K.; Hagland, H.R. Metformin Treatment Response Is Dependent on Glucose Growth Conditions and Metabolic Phenotype in Colorectal Cancer Cells. Sci. Rep. 2021, 11, 10487. [Google Scholar] [CrossRef]
- Ma, L.; Wei, J.; Wan, J.; Wang, W.; Wang, L.; Yuan, Y.; Yang, Z.; Liu, X.; Ming, L. Low Glucose and Metformin-Induced Apoptosis of Human Ovarian Cancer Cells Is Connected to ASK1 via Mitochondrial and Endoplasmic Reticulum Stress-Associated Pathways. J. Exp. Clin. Cancer Res. 2019, 38, 77. [Google Scholar] [CrossRef]
- Ma, M.; Ma, C.; Li, P.; Ma, C.; Ping, F.; Li, W.; Xu, L.; Zhang, H.; Sun, Q.; Li, Y. Low Glucose Enhanced Metformin’s Inhibitory Effect on Pancreatic Cancer Cells by Suppressing Glycolysis and Inducing Energy Stress via up-Regulation of miR-210-5p. Cell Cycle 2020, 19, 2168–2181. [Google Scholar] [CrossRef]
- Diabetic Retinopathy. Available online: https://www.nhs.uk/conditions/diabetic-retinopathy/ (accessed on 27 November 2023).
- Zhou, J.; Chen, B. Retinal Cell Damage in Diabetic Retinopathy. Cells 2023, 12, 1342. [Google Scholar] [CrossRef]
- Yang, M.; Chen, Y.; Vagionitis, S.; Körtvely, E.; Ueffing, M.; Schmachtenberg, O.; Hu, Z.; Jiao, K.; Paquet-Durand, F. Expression of Glucose Transporter-2 in Murine Retina: Evidence for Glucose Transport from Horizontal Cells to Photoreceptor Synapses. J. Neurochem. 2022, 160, 283–296. [Google Scholar] [CrossRef]
- Hurley, J.B. Retina Metabolism and Metabolism in the Pigmented Epithelium: A Busy Intersection. Annu. Rev. Vis. Sci. 2021, 7, 665–692. [Google Scholar] [CrossRef]
- Menendez, J.A.; Oliveras-Ferraros, C.; Cufí, S.; Corominas-Faja, B.; Joven, J.; Martin-Castillo, B.; Vazquez-Martin, A. Metformin Is Synthetically Lethal with Glucose Withdrawal in Cancer Cells. Cell Cycle 2012, 11, 2782–2792. [Google Scholar] [CrossRef]
- Li, B.; Zhou, P.; Xu, K.; Chen, T.; Jiao, J.; Wei, H.; Yang, X.; Xu, W.; Wan, W.; Xiao, J. Metformin Induces Cell Cycle Arrest, Apoptosis and Autophagy through ROS/JNK Signaling Pathway in Human Osteosarcoma. Int. J. Biol. Sci. 2020, 16, 74–84. [Google Scholar] [CrossRef]
- Zhuang, Y.; Chan, D.K.; Haugrud, A.B.; Miskimins, W.K. Mechanisms by Which Low Glucose Enhances the Cytotoxicity of Metformin to Cancer Cells Both In Vitro and In Vivo. PLoS ONE 2014, 9, e108444. [Google Scholar] [CrossRef]
- Corcoran, C.; Jacobs, T.F. Metformin; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Wang, Y.-W.; He, S.-J.; Feng, X.; Cheng, J.; Luo, Y.-T.; Tian, L.; Huang, Q. Metformin: A Review of Its Potential Indications. Drug Des. Dev. Ther. 2017, 11, 2421–2429. [Google Scholar] [CrossRef]
- Riaz, M.A.; Sak, A.; Erol, Y.B.; Groneberg, M.; Thomale, J.; Stuschke, M. Metformin Enhances the Radiosensitizing Effect of Cisplatin in Non-Small Cell Lung Cancer Cell Lines with Different Cisplatin Sensitivities. Sci. Rep. 2019, 9, 1282. [Google Scholar] [CrossRef] [PubMed]
- Lord, S.R.; Harris, A.L. Is It Still Worth Pursuing the Repurposing of Metformin as a Cancer Therapeutic? Br. J. Cancer 2023, 128, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Fukushi, A.; Kim, H.-D.; Chang, Y.-C.; Kim, C.-H. Revisited Metabolic Control and Reprogramming Cancers by Means of the Warburg Effect in Tumor Cells. Int. J. Mol. Sci. 2022, 23, 10037. [Google Scholar] [CrossRef] [PubMed]
- Zake, D.M.; Kurlovics, J.; Zaharenko, L.; Komasilovs, V.; Klovins, J.; Stalidzans, E. Physiologically Based Metformin Pharmacokinetics Model of Mice and Scale-up to Humans for the Estimation of Concentrations in Various Tissues. PLoS ONE 2021, 16, e0249594. [Google Scholar] [CrossRef] [PubMed]
- Herrgårdh, T.; Li, H.; Nyman, E.; Cedersund, G. An Updated Organ-Based Multi-Level Model for Glucose Homeostasis: Organ Distributions, Timing, and Impact of Blood Flow. Front. Physiol. 2021, 12, 619254. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J. Energy Metabolism of Cancer: Glycolysis versus Oxidative Phosphorylation (Review). Oncol. Lett. 2012, 4, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Sekar, P.; Hsiao, G.; Hsu, S.-H.; Huang, D.-Y.; Lin, W.-W.; Chan, C.-M. Metformin Inhibits Methylglyoxal-Induced Retinal Pigment Epithelial Cell Death and Retinopathy via AMPK-Dependent Mechanisms: Reversing Mitochondrial Dysfunction and Upregulating Glyoxalase 1. Redox Biol. 2023, 64, 102786. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Kim, M.; Choi, M.Y.; Lee, D.H.; Roh, G.S.; Kim, H.J.; Kang, S.S.; Cho, G.J.; Kim, S.-J.; Yoo, J.-M.; et al. Metformin Protects against Retinal Cell Death in Diabetic Mice. Biochem. Biophys. Res. Commun. 2017, 492, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Kong, L.; Wang, J.; Ash, J.D. Stimulation of AMPK Prevents Degeneration of Photoreceptors and the Retinal Pigment Epithelium. Proc. Natl. Acad. Sci. USA 2018, 115, 10475–10480. [Google Scholar] [CrossRef] [PubMed]
- Kuan, I.H.S.; Wright, D.F.B.; Duffull, S.B.; Zhu, X. Understanding the Association between Metformin Plasma Concentrations and Lactate. Br. J. Clin. Pharmacol. 2021, 87, 700–701. [Google Scholar] [CrossRef]
- Koons, A.; Amaducci, A.M.; Katz, K.D. Reversible Total Vision Loss Caused by Severe Metformin-Associated Lactic Acidosis: A Case Report. Clin. Pract. Cases Emerg. Med. 2021, 5, 206–209. [Google Scholar] [CrossRef]
- Jeon, J.W.; Choi, W.; Kim, H.R.; Ham, Y.R.; Choi, D.E.; Na, K.R.; Lee, K.W.; Bae, S.Y.; Kim, S.H. Transient Blindness in a Patient with Severe Metformin-Associated Lactic Acidosis (MALA). Electrolyte Blood Press. 2019, 17, 16–20. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villa-Fernández, E.; García, A.V.; Fernández-Fernández, A.; García-Villarino, M.; Ares-Blanco, J.; Pujante, P.; González-Vidal, T.; Fraga, M.F.; Torre, E.M.; Delgado, E.; et al. Metformin and Glucose Concentration as Limiting Factors in Retinal Pigment Epithelial Cell Viability and Proliferation. Int. J. Mol. Sci. 2024, 25, 2637. https://doi.org/10.3390/ijms25052637
Villa-Fernández E, García AV, Fernández-Fernández A, García-Villarino M, Ares-Blanco J, Pujante P, González-Vidal T, Fraga MF, Torre EM, Delgado E, et al. Metformin and Glucose Concentration as Limiting Factors in Retinal Pigment Epithelial Cell Viability and Proliferation. International Journal of Molecular Sciences. 2024; 25(5):2637. https://doi.org/10.3390/ijms25052637
Chicago/Turabian StyleVilla-Fernández, Elsa, Ana Victoria García, Alejandra Fernández-Fernández, Miguel García-Villarino, Jessica Ares-Blanco, Pedro Pujante, Tomás González-Vidal, Mario F. Fraga, Edelmiro Menéndez Torre, Elias Delgado, and et al. 2024. "Metformin and Glucose Concentration as Limiting Factors in Retinal Pigment Epithelial Cell Viability and Proliferation" International Journal of Molecular Sciences 25, no. 5: 2637. https://doi.org/10.3390/ijms25052637
APA StyleVilla-Fernández, E., García, A. V., Fernández-Fernández, A., García-Villarino, M., Ares-Blanco, J., Pujante, P., González-Vidal, T., Fraga, M. F., Torre, E. M., Delgado, E., & Lambert, C. (2024). Metformin and Glucose Concentration as Limiting Factors in Retinal Pigment Epithelial Cell Viability and Proliferation. International Journal of Molecular Sciences, 25(5), 2637. https://doi.org/10.3390/ijms25052637





