Immunological Landscapes in Lung Transplantation: Insights from T Cell Profiling in BAL and PBMC
Abstract
1. Introduction
2. Results
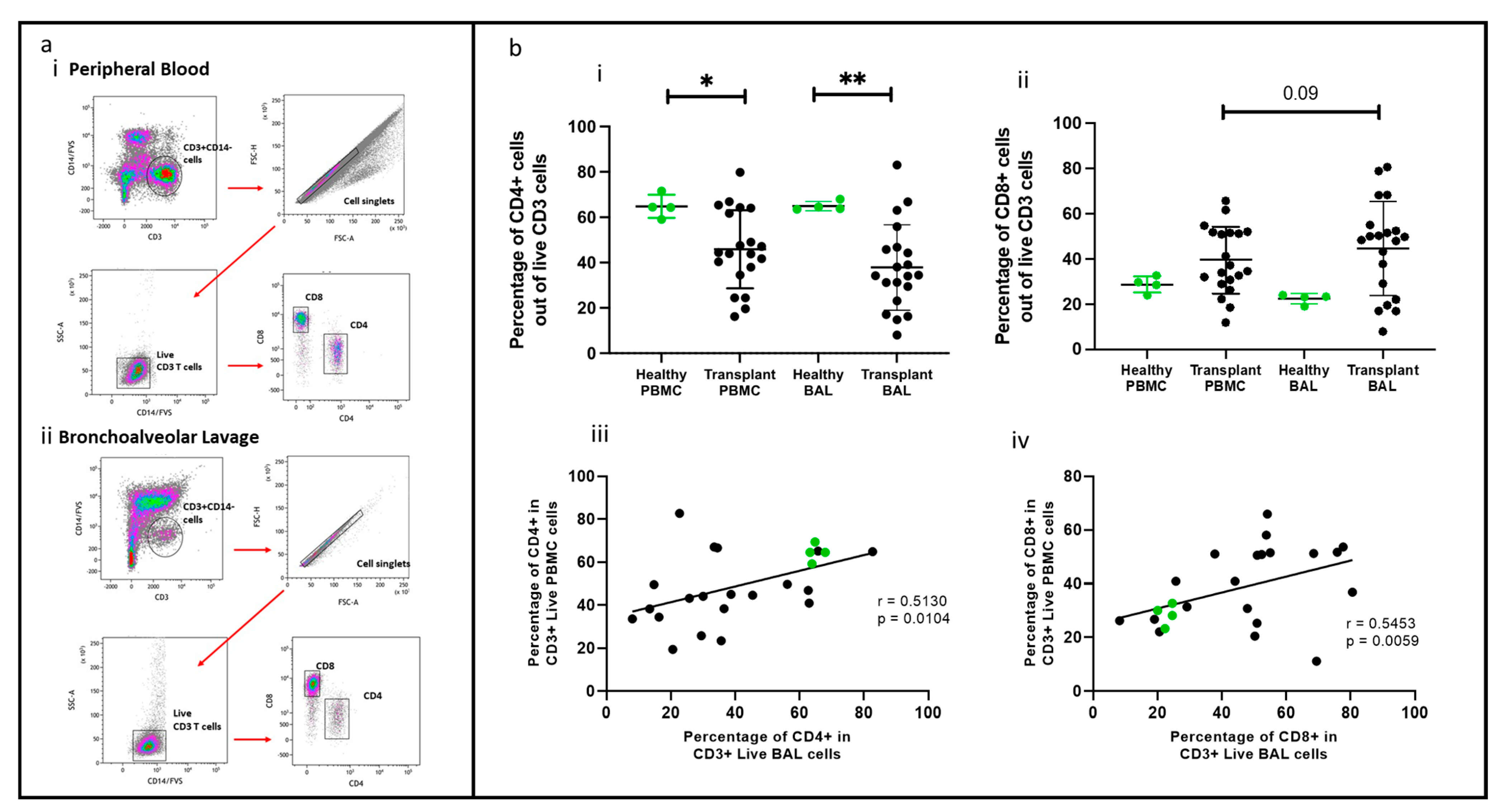
2.1. Multi-Colour Flow Cytometric Panel to Identify T Cell Subsets Comparing PBMC and BAL
2.2. CD4 and CD8 T Cells Comparing PBMC and BAL
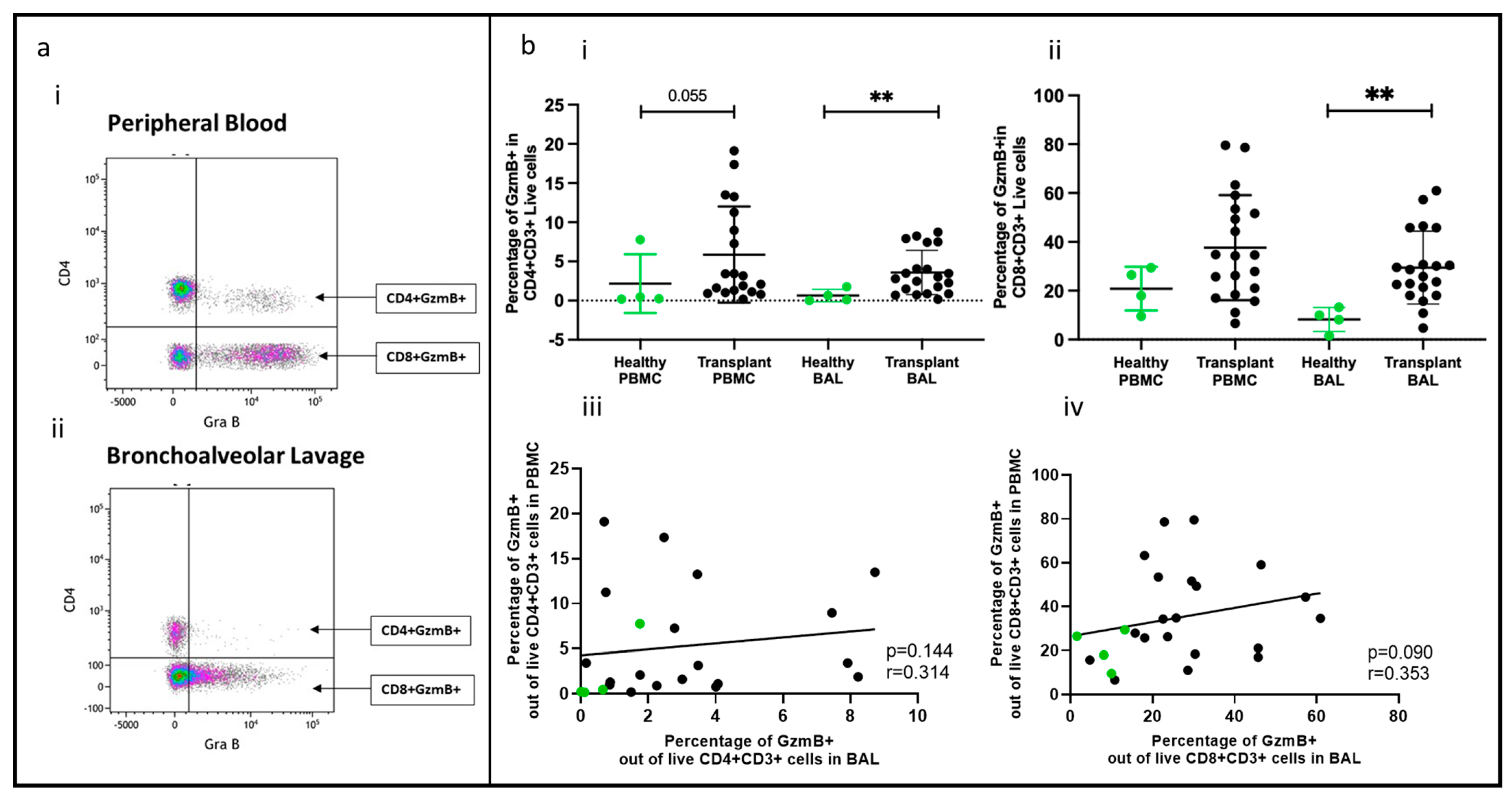
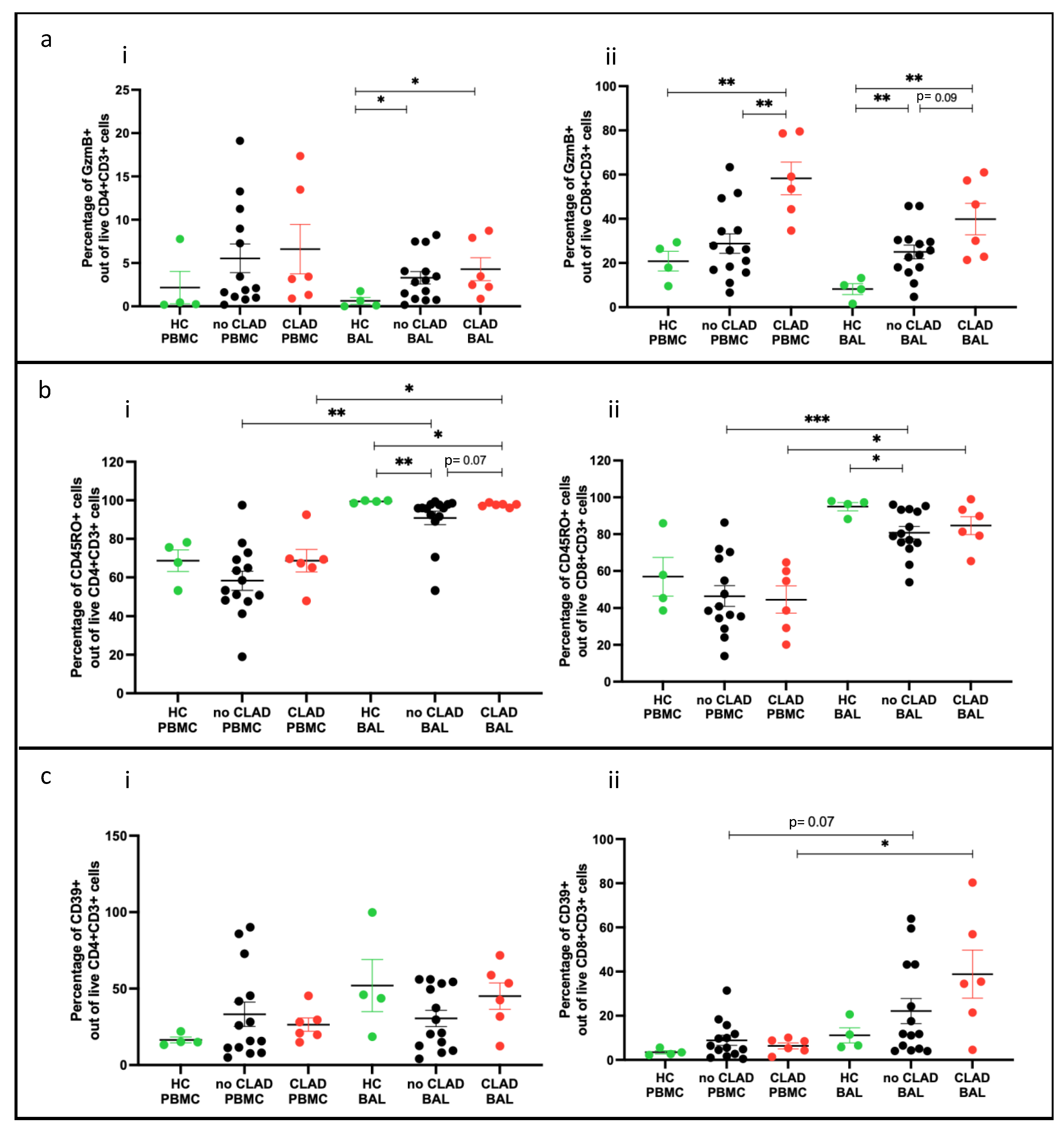
2.3. Effector T Cells
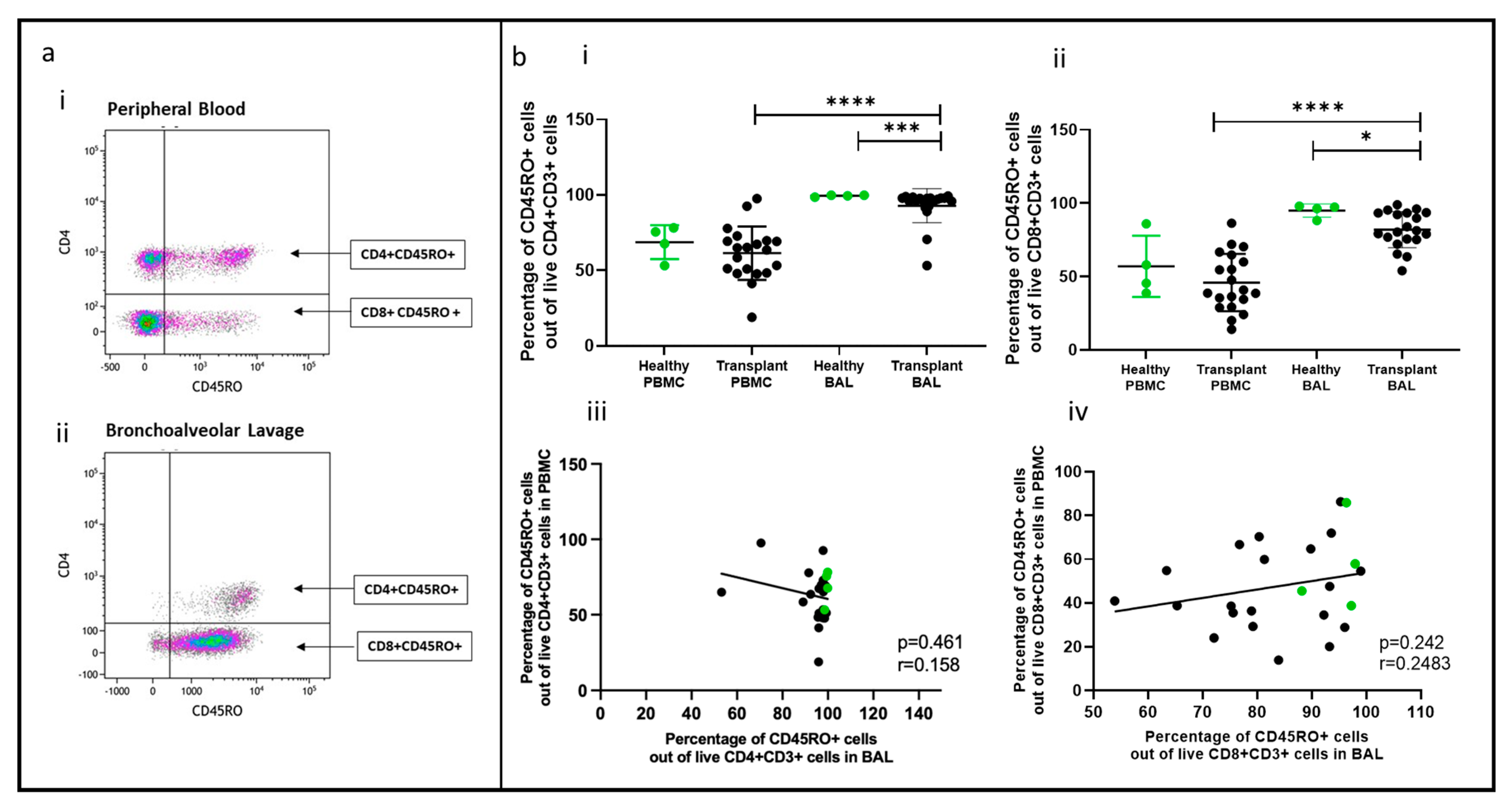
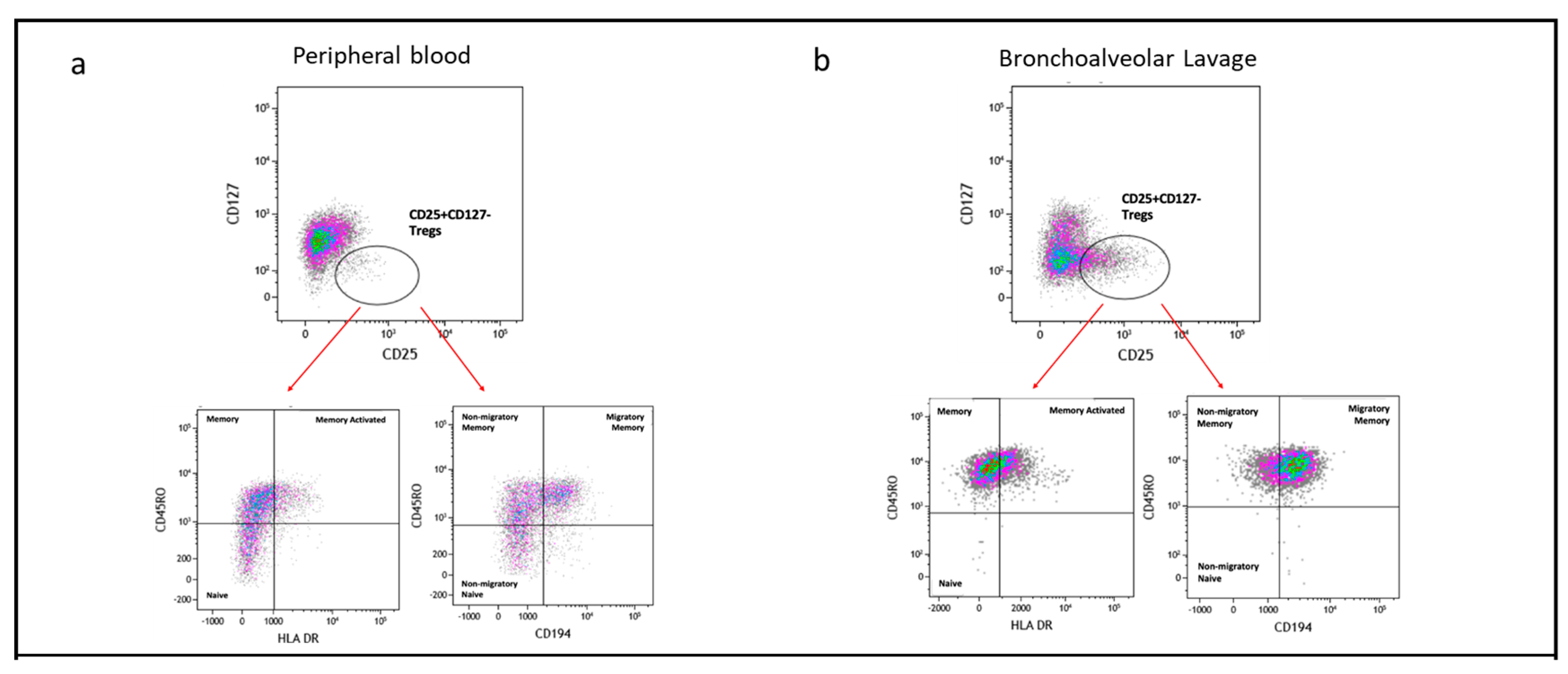
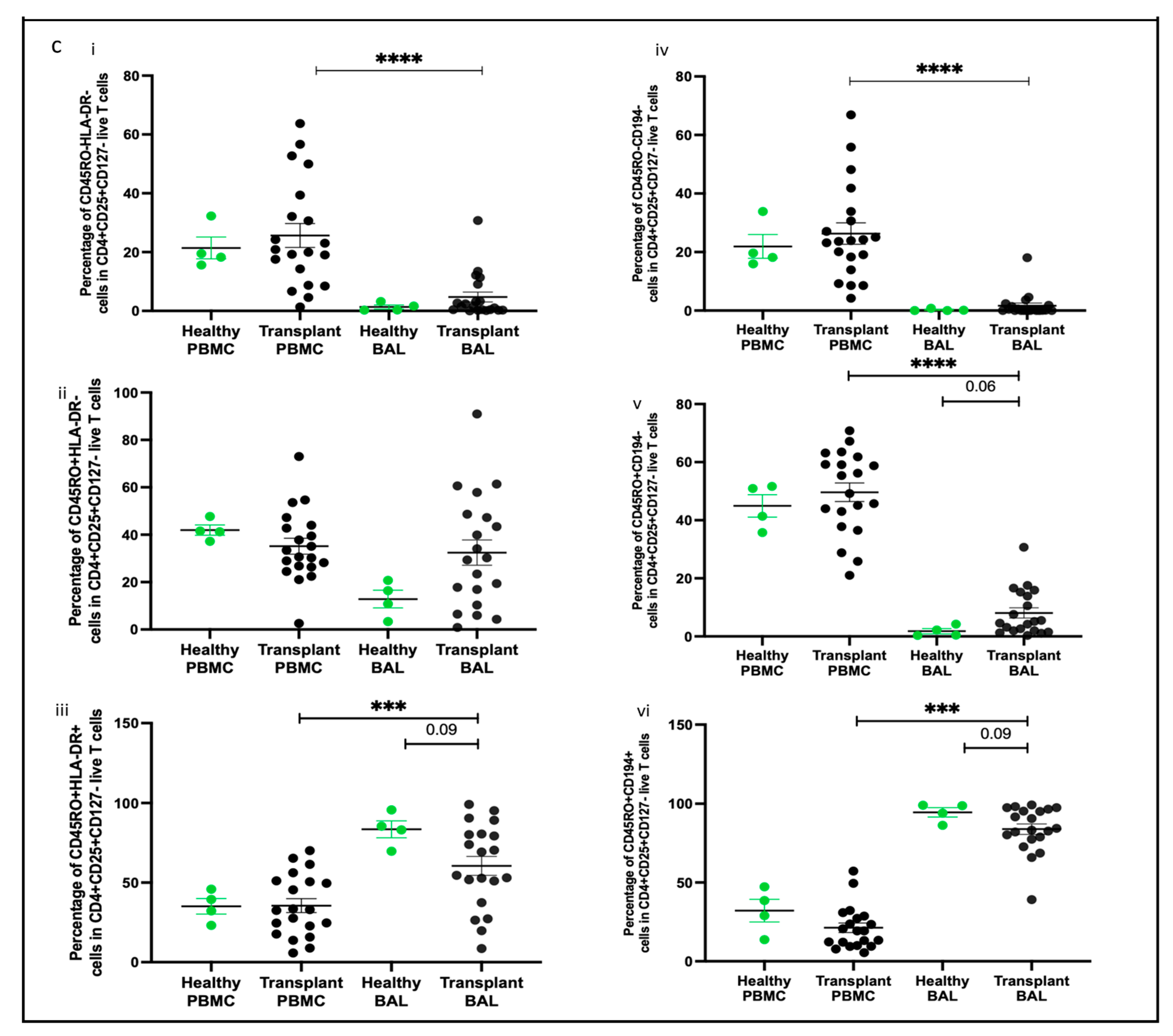
2.4. Memory T Cell Populations Comparing PBMC and BAL
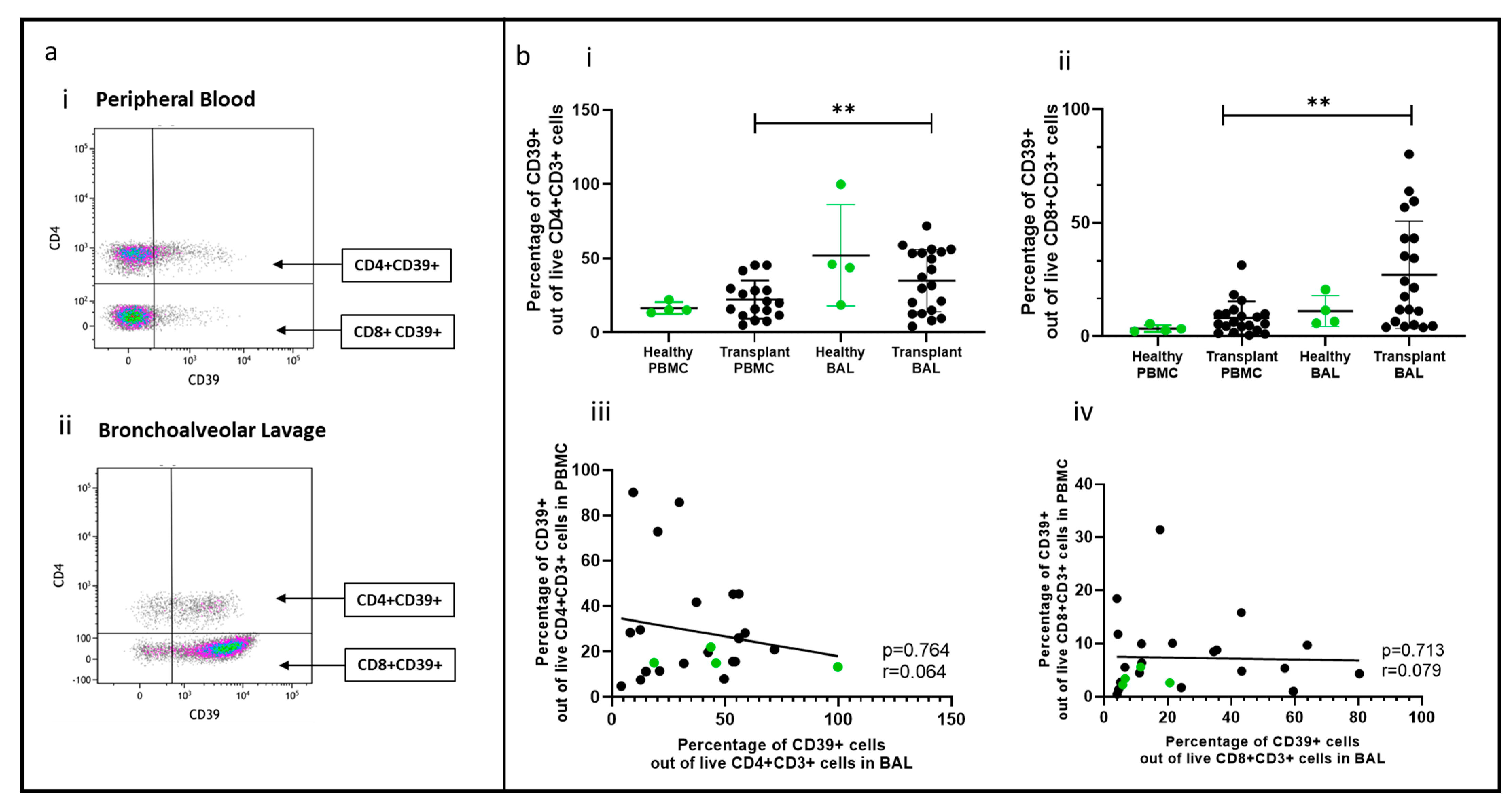
2.5. Non-Tregs Expressing CD39 Comparing PBMC and BAL
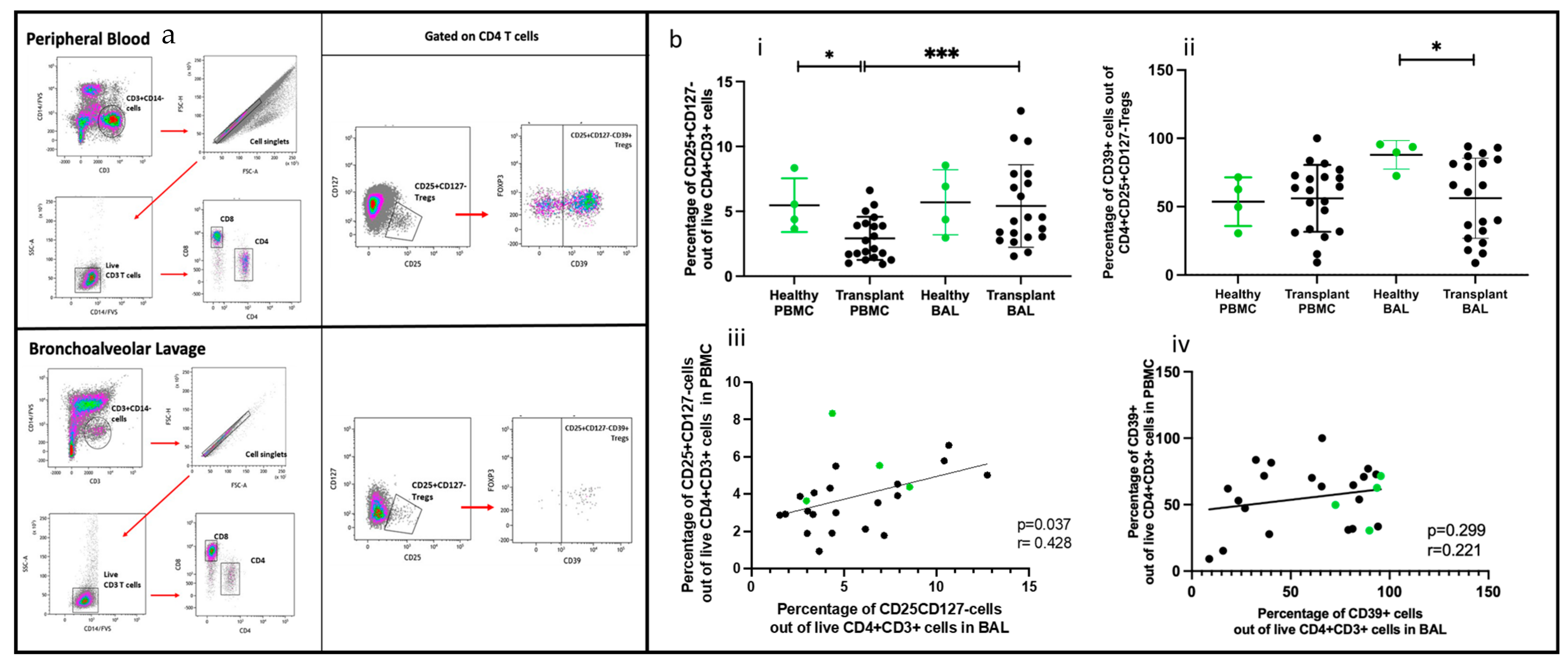
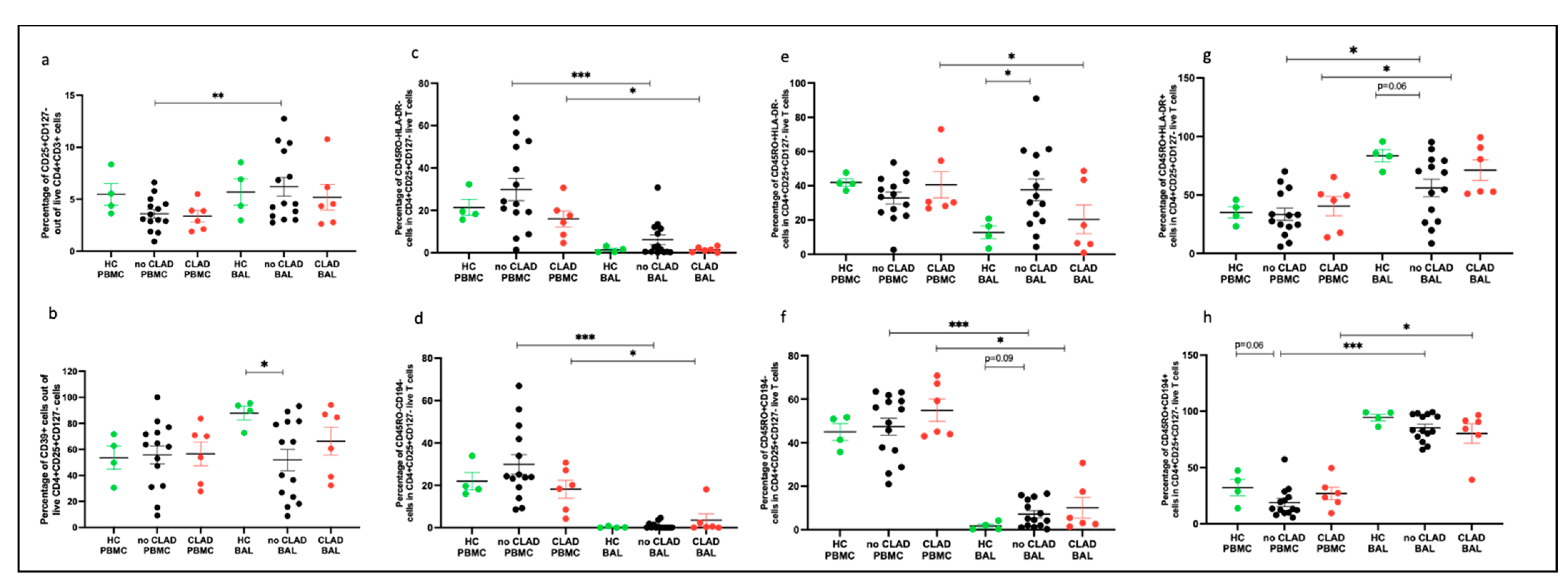
2.6. Regulatory T Cells Comparing PBMC and BAL
2.7. Subpopulation of Tregs Comparing PBMC and BAL
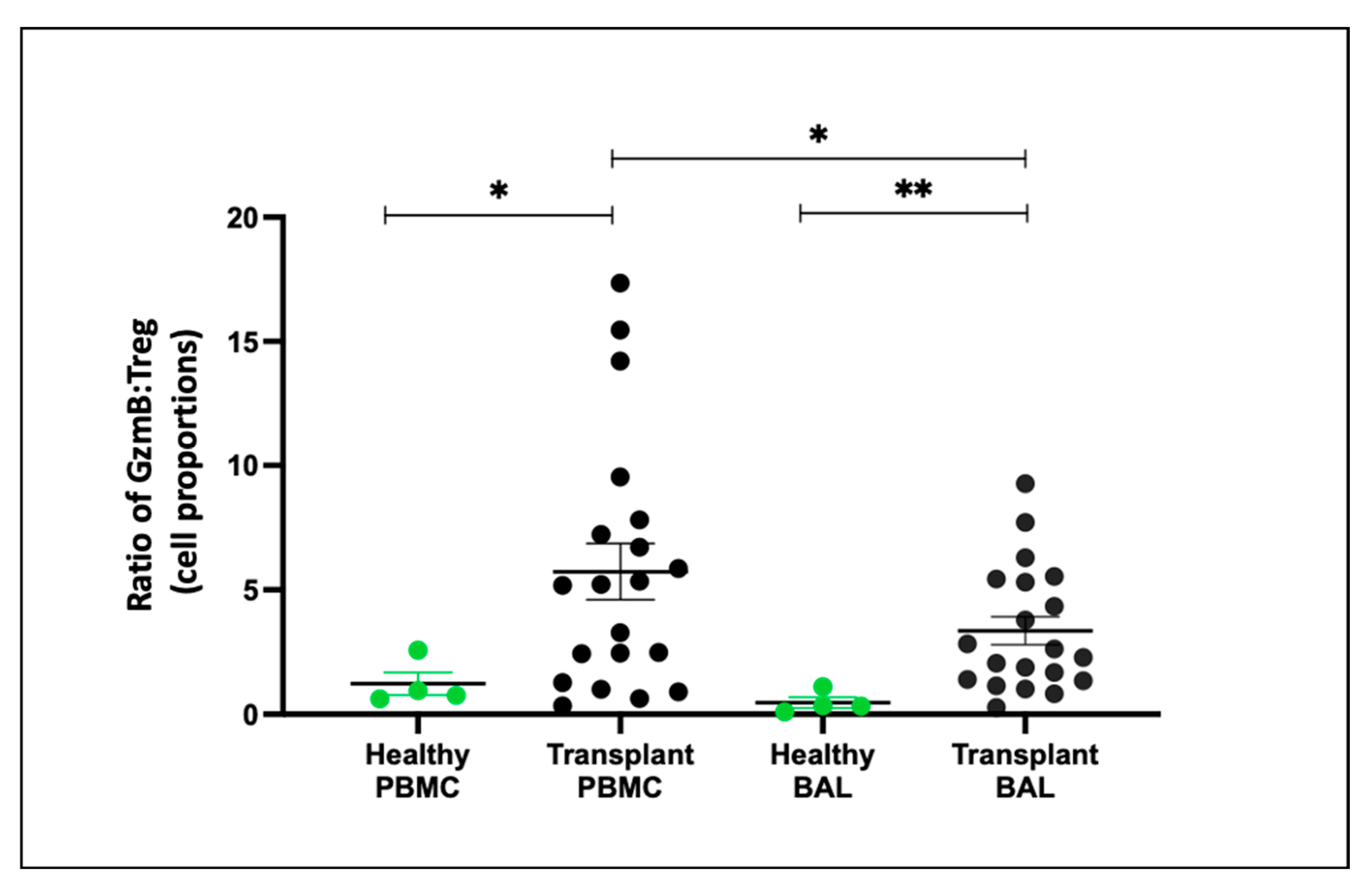
2.8. Teff and Treg Balance (Granzyme B:Treg Ratio) Comparing PBMC and BAL
2.9. T Cell Subsets and Association with CLAD
2.9.1. Effector T Cells (Teffs) and Memory T Cells (Tmems) Comparing PBMC and BAL
2.9.2. Treg Populations Comparing PBMC and BAL
2.9.3. Teffs and Tregs Balance (Granzyme B–Treg Ratio) Comparing PBMC and BAL
3. Discussion
4. Methods
4.1. Patient Demographics
4.2. Ethics Approval
4.3. Isolation of PBMCs from Peripheral PBMC
4.4. Isolation of Immune Cells from BAL
4.5. Flow Cytometric Analysis
4.6. Lung Function Parameters
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McDermott, J.K.; Girgis, R.E. Individualizing immunosuppression in lung transplantation. Glob. Cardiol. Sci. Pract. 2018, 2018, 5. [Google Scholar] [CrossRef][Green Version]
- Neujahr, D.C.; Cardona, A.C.; Ulukpo, O.; Rigby, M.; Pelaez, A.; Ramirez, A.; Gal, A.A.; Force, S.D.; Lawrence, E.C.; Kirk, A.D.; et al. Dynamics of human regulatory T cells in lung lavages of lung transplant recipients. Transplantation 2009, 88, 521–527. [Google Scholar] [CrossRef]
- Chambers, D.C.; Cherikh, W.S.; Goldfarb, S.B.; Hayes, D.; Kucheryavaya, A.Y.; Toll, A.E.; Khush, K.K.; Levvey, B.J.; Meiser, B.; Rossano, J.W.; et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-fifth adult lung and heart-lung transplant report-2018; Focus theme: Multiorgan Transplantation. J. Heart Lung Transplant. 2018, 37, 1169–1183. [Google Scholar] [CrossRef]
- Studer, S.M.; Levy, R.D.; McNeil, K.; Orens, J.B. Lung transplant outcomes: A review of survival, graft function, physiology, health-related quality of life and cost-effectiveness. Eur. Respir. J. 2004, 24, 674–685. [Google Scholar] [CrossRef]
- Glanville, A.R.; Verleden, G.M.; Todd, J.L.; Benden, C.; Calabrese, F.; Gottlieb, J.; Hachem, R.R.; Levine, D.; Meloni, F.; Palmer, S.M.; et al. Chronic lung allograft dysfunction: Definition update of restrictive allograft syndrome-A consensus report from the Pulmonary Council of the ISHLT. J. Heart Lung Transplant. 2019, 38, 483–492. [Google Scholar] [CrossRef]
- Vaikunthanathan, T.; Safinia, N.; Boardman, D.; Lechler, R.I.; Lombardi, G. Regulatory T cells: Tolerance induction in solid organ transplantation. Clin. Exp. Immunol. 2017, 189, 197–210. [Google Scholar] [CrossRef] [PubMed]
- Ius, F.; Salman, J.; Knöfel, A.; Nakagiri, T.; Sommer, W.; Siemeni, T.; Kühn, C.; Welte, T.; Falk, C.; Haverich, A.; et al. Increased Frequency of regulatory CD127low T Cells and of IL2+ T Cells Early after Lung Transplant is Associated with Improved Graft Survival. J. Heart Lung Transplant. 2019, 38, S28–S29. [Google Scholar] [CrossRef]
- Mamessier, E.; Lorec, A.M.; Thomas, P.; Badier, M.; Magnan, A.; Reynaud-Gaubert, M. T regulatory cells in stable posttransplant bronchiolitis obliterans syndrome. Transplantation 2007, 84, 908–916. [Google Scholar] [CrossRef] [PubMed]
- Meloni, F.; Vitulo, P.; Bianco, A.M.; Paschetto, E.; Morosini, M.; Cascina, A.; Mazzucchelli, I.; Ciardelli, L.; Oggionni, T.; Fietta, A.M.; et al. Regulatory CD4+CD25+ T Cells in The Peripheral Blood of Lung Transplant Recipients: Correlation with Transplant Outcome. Transplantation 2004, 77, 762–766. [Google Scholar] [CrossRef] [PubMed]
- Piloni, D.; Morosini, M.; Magni, S.; Balderacchi, A.; Scudeller, L.; Cova, E.; Oggionni, T.; Stella, G.; Tinelli, C.; Antonacci, F.; et al. Analysis of long term CD4+CD25highCD127− T-reg cells kinetics in peripheral blood of lung transplant recipients. BMC Pulm. Med. 2017, 17, 102. [Google Scholar] [CrossRef] [PubMed]
- Durand, M.; Lacoste, P.; Danger, R.; Jacquemont, L.; Brosseau, C.; Durand, E.; Tilly, G.; Loy, J.; Foureau, A.; Royer, P.J.; et al. High circulating CD4+CD25hiFOXP3+ T-cell sub-population early after lung transplantation is associated with development of bronchiolitis obliterans syndrome. J. Heart Lung Transplant. 2018, 37, 770–781. [Google Scholar] [CrossRef] [PubMed]
- Salman, J.; Ius, F.; Knoefel, A.-K.; Sommer, W.; Siemeni, T.; Kuehn, C.; Tudorache, I.; Avsar, M.; Nakagiri, T.; Preissler, G.; et al. Association of Higher CD4+CD25highCD127low, FoxP3+, and IL-2+ T Cell Frequencies Early After Lung Transplantation with Less Chronic Lung Allograft Dysfunction at Two Years. Am. J. Transplant. 2017, 17, 1637–1648. [Google Scholar] [CrossRef] [PubMed]
- Bhorade, S.M.; Chen, H.; Molinero, L.; Liao, C.; Garrity, E.R.; Vigneswaran, W.T.; Shilling, R.; Sperling, A.; Chong, A.; Alegre, M.-L. Decreased percentage of CD4+FoxP3+ cells in bronchoalveolar lavage from lung transplant recipients correlates with development of bronchiolitis obliterans syndrome. Transplantation 2010, 90, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Gregson, A.L.; Hoji, A.; Palchevskiy, V.; Hu, S.; Weigt, S.S.; Liao, E.; Derhovanessian, A.; Saggar, R.; Song, S.; Elashoff, R.; et al. Protection against bronchiolitis obliterans syndrome is associated with allograft CCR7+CD45RA− T regulatory cells. PLoS ONE 2010, 5, e11354. [Google Scholar] [CrossRef] [PubMed]
- Krustrup, D.; Madsen, C.B.; Iversen, M.; Engelholm, L.; Ryder, L.P.; Andersen, C.B. The number of regulatory T cells in transbronchial lung allograft biopsies is related to FoxP3 mRNA levels in bronchoalveolar lavage fluid and to the degree of acute cellular rejection. Transpl. Immunol. 2013, 29, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Krustrup, D.; Iversen, M.; Martinussen, T.; Andersen, C.B. Time elapsed after transplantation influences the relationship between the number of regulatory T cells in lung allograft biopsies and subsequent acute rejection episodes. Transpl. Immunol. 2014, 31, 42–47. [Google Scholar] [CrossRef]
- Krustrup, D.; Iversen, M.; Martinussen, T.; Schultz, H.H.L.; Andersen, C.B. The number of FoxP3+ cells in transbronchial lung allograft biopsies does not predict bronchiolitis obliterans syndrome within the first five years after transplantation. Clin. Transplant. 2015, 29, 179–184. [Google Scholar] [CrossRef]
- White, M.; Levy, L.; Moshkelgosha, S.; Zehong, G.; Keshavjee, S.; Martinu, T.; Juvet, S. Regulatory T Cells in Asymptomatic ISHLT Grade A1 Rejection and the Risk of Chronic Lung Allograft Dysfunction. J. Heart Lung Transplant. 2019, 38, S17. [Google Scholar] [CrossRef]
- Martinu, T.; Koutsokera, A.; Benden, C.; Cantu, E.; Chambers, D.; Cypel, M.; Edelman, J.; Emtiazjoo, A.; Fisher, A.J.; Greenland, J.R.; et al. International Society for Heart and Lung Transplantation consensus statement for the standardization of bronchoalveolar lavage in lung transplantation. J. Heart Lung Transplant. 2020, 39, 1171–1190. [Google Scholar] [CrossRef]
- Kennedy, V.E.; Todd, J.L.; Palmer, S.M. Bronchoalveolar Lavage as a Tool to Predict, Diagnose and Understand Bronchiolitis Obliterans Syndrome. Am. Soc. Transplant. Am. Soc. Oftransplant Surg. 2013, 13, 552–561. [Google Scholar] [CrossRef]
- Hogea, S.P.; Tudorache, E.; Pescaru, C.; Marc, M.; Oancea, C. Bronchoalveolar lavage: Role in the evaluation of pulmonary interstitial disease. Expert. Rev. Respir. Med. 2020, 14, 1117–1130. [Google Scholar] [CrossRef]
- Marino, J.; Paster, J.; Benichou, G. Allorecognition by T Lymphocytes and Allograft Rejection. Front. Immunol. 2016, 7, 582. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Fan, H.; Jiang, S. CD4+ T-cell subsets in transplantation. Immunol. Rev. 2013, 252, 183–191. [Google Scholar] [CrossRef]
- Bueno, V.; Pestana, J.O.M. The role of CD8+ T cells during allograft rejection. Braz. J. Med. Biol. Res. 2002, 35, 1247–1258. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Putnam, A.L.; Xu-Yu, Z.; Szot, G.L.; Lee, M.R.; Zhu, S.; Gottlieb, P.A.; Kapranov, P.; Gingeras, T.R.; de St Groth, B.F.; et al. CD127 expression inversely correlates with FoxP3 and suppressive function of human CD4+ T reg cells. J. Exp. Med. 2006, 203, 1701–1711. [Google Scholar] [CrossRef] [PubMed]
- Hartigan-O’Connor, D.J.; Poon, C.; Sinclair, E.; McCune, J.M. Human CD4+ regulatory T cells express lower levels of the IL-7 receptor alpha chain (CD127), allowing consistent identification and sorting of live cells. J. Immunol. Methods 2007, 319, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Seddiki, N.; Santner-Nanan, B.; Martinson, J.; Zaunders, J.; Sasson, S.; Landay, A.; Solomon, M.; Selby, W.; Alexander, S.I.; Nanan, R.; et al. Expression of interleukin (IL)-2 and IL-7 receptors discriminates between human regulatory and activated T cells. J. Exp. Med. 2006, 203, 1693–1700. [Google Scholar] [CrossRef]
- Yu, N.; Li, X.; Song, W.; Li, D.; Yu, D.; Zeng, X.; Li, M.; Leng, X.; Li, X. CD4+CD25+CD127low/− T cells: A more specific Treg population in human peripheral blood. Inflammation 2012, 35, 1773–1780. [Google Scholar] [CrossRef]
- van der Veeken, J.; Gonzalez, A.J.; Cho, H.; Arvey, A.; Hemmers, S.; Leslie, C.S.; Rudensky, A.Y. Memory of Inflammation in Regulatory T Cells. Cell 2016, 166, 977–990. [Google Scholar] [CrossRef]
- Liston, A.; Gray, D.H. Homeostatic control of regulatory T cell diversity. Nat. Rev. Immunol. 2014, 14, 154–165. [Google Scholar] [CrossRef]
- Shevyrev, D.; Tereshchenko, V. Treg Heterogeneity, Function, and Homeostasis. Front. Immunol. 2020, 10, 3100. [Google Scholar] [CrossRef]
- Gu, J.; Ni, X.; Pan, X.; Lu, H.; Lu, Y.; Zhao, J.; Zheng, S.G.; Hippen, K.L.; Wang, X.; Lu, L. Human CD39hi regulatory T cells present stronger stability and function under inflammatory conditions. Cell. Mol. Immunol. 2017, 14, 521–528. [Google Scholar] [CrossRef] [PubMed]
- Schuler, P.J.; Saze, Z.; Hong, C.-S.; Muller, L.; Gillespie, D.G.; Cheng, D.; Harasymczuk, M.; Mandapathil, M.; Lang, S.; Jackson, E.K.; et al. Human CD4+CD39+ regulatory T cells produce adenosine upon co-expression of surface CD73 or contact with CD73+ or CD73+ cells. Clin. Exp. Immunol. 2014, 177, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Wang, X.; Gu, J.; Lu, H.; Zhang, F.; Li, X.; Qian, X.; Wang, X.; Lu, L. iTreg induced from CD39+ naive T cells demonstrate enhanced proliferate and suppressive ability. Int. Immunopharmacol. 2015, 28, 925–930. [Google Scholar] [CrossRef]
- Moesta, A.K.; Li, X.Y.; Smyth, M.J. Targeting CD39 in cancer. Nat. Rev. Immunol. 2020, 20, 739–755. [Google Scholar] [CrossRef]
- Moncrieffe, H.; Nistala, K.; Kamhieh, Y.; Evans, J.; Eddaoudi, A.; Eaton, S.; Wedderburn, L.R. High expression of the ectonucleotidase CD39 on T cells from the inflamed site identifies two distinct populations, one regulatory and one memory T cell population. J. Immunol. 2010, 185, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Yu, M.; Cavanagh, M.M.; Saunders, J.H.; Qi, Q.; Ye, Z.; Le Saux, S.; Sultan, W.; Turgano, E.; Dekker, C.L.; et al. Expression of CD39 on Activated T Cells Impairs their Survival in Older Individuals. Cell Rep. 2016, 14, 1218–1231. [Google Scholar] [CrossRef] [PubMed]
- Canale, F.P.; Ramello, M.C.; Núñez, N.; Furlan, C.L.; Bossio, S.N.; Serrán, M.G.; Boari, J.T.; Del Castillo, A.; Ledesma, M.; Sedlik, C.; et al. CD39 Expression Defines Cell Exhaustion in Tumor-Infiltrating CD8+ T Cells. Cancer Res. 2018, 78, 115–128. [Google Scholar] [CrossRef]
- de Silva, T.A.; Apte, S.; Voisey, J.; Spann, K.; Tan, M.; Divithotawela, C.; Chambers, D.; O’Sullivan, B. Single-Cell Profiling of Cells in the Lung of a Patient with Chronic Hypersensitivity Pneumonitis Reveals Inflammatory Niche with Abundant CD39+ T Cells with Functional ATPase Phenotype: A Case Study. Int. J. Mol. Sci. 2023, 24, 14442. [Google Scholar] [CrossRef]
- Choy, J.C. Granzymes and perforin in solid organ transplant rejection. Cell Death Differ. 2010, 17, 567–576. [Google Scholar] [CrossRef]

| Transplant (n = 20) | Non-Transplant (n = 4) | |
|---|---|---|
| Gender | ||
| Male | 10 | 3 |
| Female | 10 | 1 |
| Initial disease condition for transplant | ||
| Cystic Fibrosis | 11 | N/A |
| COPD | 4 | N/A |
| IPF | 4 | N/A |
| Histiocytosis | 1 | N/A |
| Type of transplant | ||
| Bilateral | 19 | N/A |
| Heart and Lung | 1 | N/A |
| Time post-transplant (months) | 52.83 (SEM ± 10.84) | N/A |
| Age at bronchoscopy (years) | 43.66 (SEM ± 3.78) | 46.96 (SEM ± 9.38) |
| Marker | Purpose | Antibody Used |
|---|---|---|
| CD3 | Marker for T lymphocytes | anti-CD3-AF700 (BioLegend) |
| CD4 | Marker for CD4 T cells | anti-CD4-BV510 (BioLegend) |
| CD8 | Marker for CD8 T cells | anti-CD8-APC-H7 (BD Biosciences) |
| CD14 | Marker of macrophages and granulocytes. | anti-CD14-PE-Cy 7 (BD Biosciences) |
| CD25 | Regulatory T cells express high CD25 | anti-CD25-BV421 (BD Biosciences) |
| CD127 | Regulatory T cells express low CD127 | anti-CD127-FITC (BD Biosciences) |
| CD39 | Immunosuppressive ecto-enzyme converts ATP to ADP | anti-CD39-BV711 (BioLegend) |
| CD73 | Immunosuppressive ecto-enzyme coverts ADP to AMP | anti-CD73-BV605 (BD Biosciences) |
| CD194 | Marker of cell migration, also known as CCR4 | anti-CD194-PE-Cy 7 (BD Biosciences) |
| CD45RO | Marker of memory cells | anti-CD45RO-BV650 (BD Biosciences) |
| HLA-DR | Marker of MHC II and upregulated in activated Treg | anti-HLA-DR-BV785 (BioLegend) |
| Ki67 | Marker of cellular proliferation | anti-Ki67-PerCp- Cy 5.5 (BioLegend) |
| FOXP3 | Transcription factor specific to regulatory T cells | anti-FoxP3-PE (BD Biosciences) |
| Granzyme B | Serine protease commonly found in the granules of cytotoxic T cells | anti-granzyme B-APC (Invitrogen) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Silva, T.A.; Apte, S.; Voisey, J.; Spann, K.; Tan, M.; Chambers, D.; O’Sullivan, B. Immunological Landscapes in Lung Transplantation: Insights from T Cell Profiling in BAL and PBMC. Int. J. Mol. Sci. 2024, 25, 2476. https://doi.org/10.3390/ijms25052476
de Silva TA, Apte S, Voisey J, Spann K, Tan M, Chambers D, O’Sullivan B. Immunological Landscapes in Lung Transplantation: Insights from T Cell Profiling in BAL and PBMC. International Journal of Molecular Sciences. 2024; 25(5):2476. https://doi.org/10.3390/ijms25052476
Chicago/Turabian Stylede Silva, Tharushi Ayanthika, Simon Apte, Joanne Voisey, Kirsten Spann, Maxine Tan, Daniel Chambers, and Brendan O’Sullivan. 2024. "Immunological Landscapes in Lung Transplantation: Insights from T Cell Profiling in BAL and PBMC" International Journal of Molecular Sciences 25, no. 5: 2476. https://doi.org/10.3390/ijms25052476
APA Stylede Silva, T. A., Apte, S., Voisey, J., Spann, K., Tan, M., Chambers, D., & O’Sullivan, B. (2024). Immunological Landscapes in Lung Transplantation: Insights from T Cell Profiling in BAL and PBMC. International Journal of Molecular Sciences, 25(5), 2476. https://doi.org/10.3390/ijms25052476






