Clinical and Real-World Effectiveness of Mogamulizumab: A Narrative Review
Abstract
1. Introduction
2. Mechanism of Action
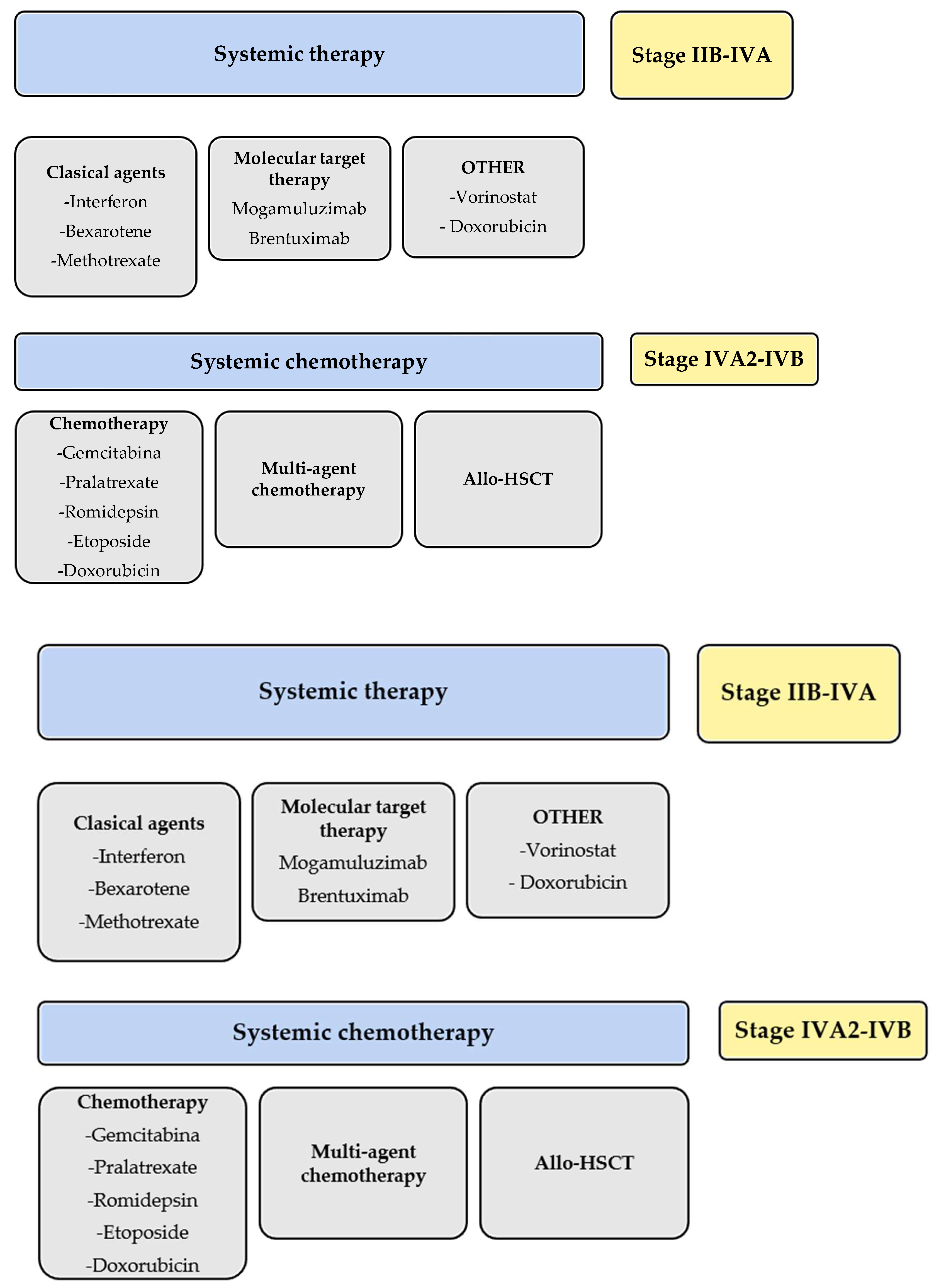
3. Mogamulizumab in Clinical Guidelines
4. Clinical Trials Involving Mogamulizumab
5. Real-World Results of Mogamulizumab
6. Safety of Mogamulizumab in Clinical Assays
7. Mogamulizumab Results with Combined Treatments in ATLL
8. Mogamulizumab Results with Combined Treatments in Cutaneous Lymphomas/Dermatology
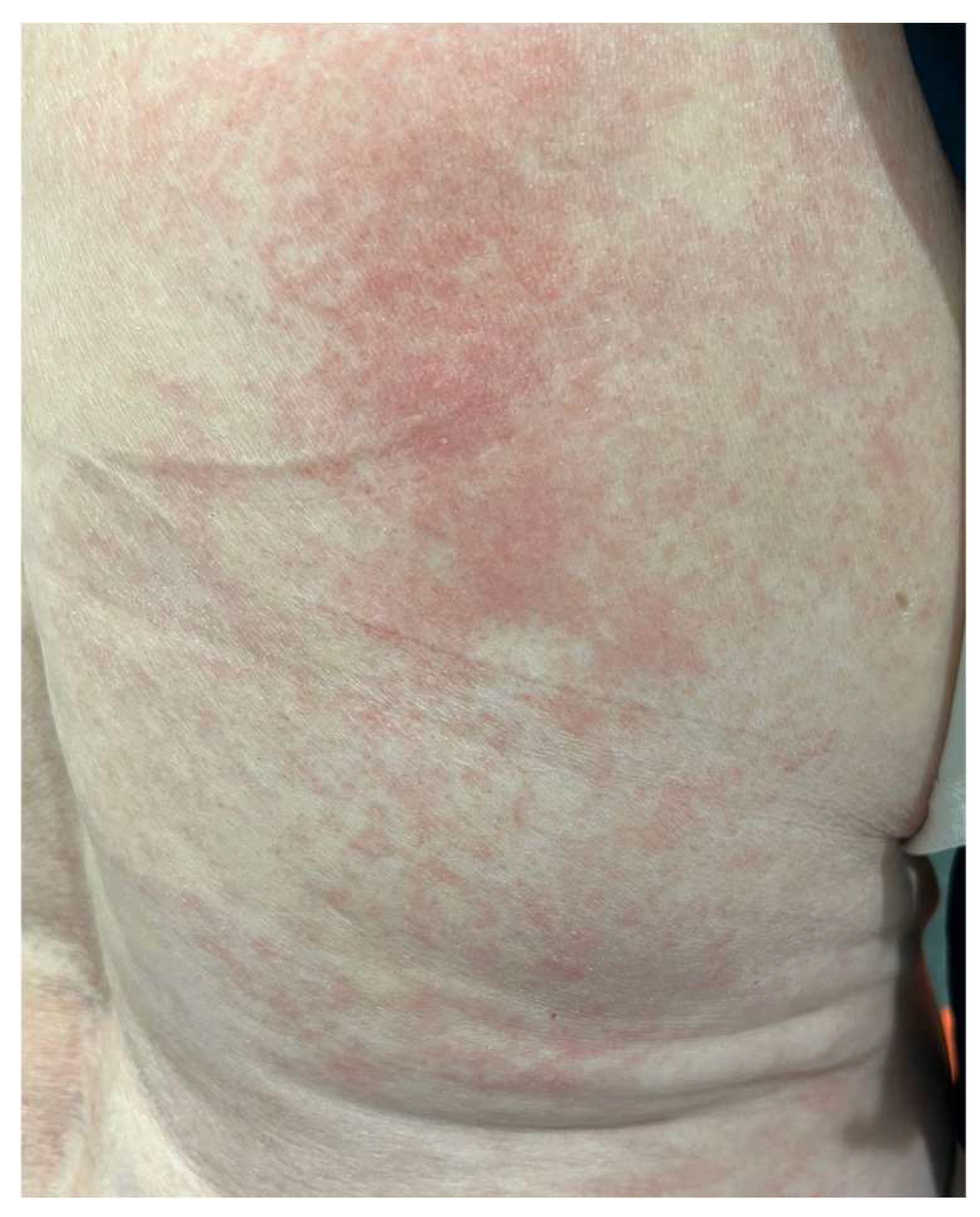
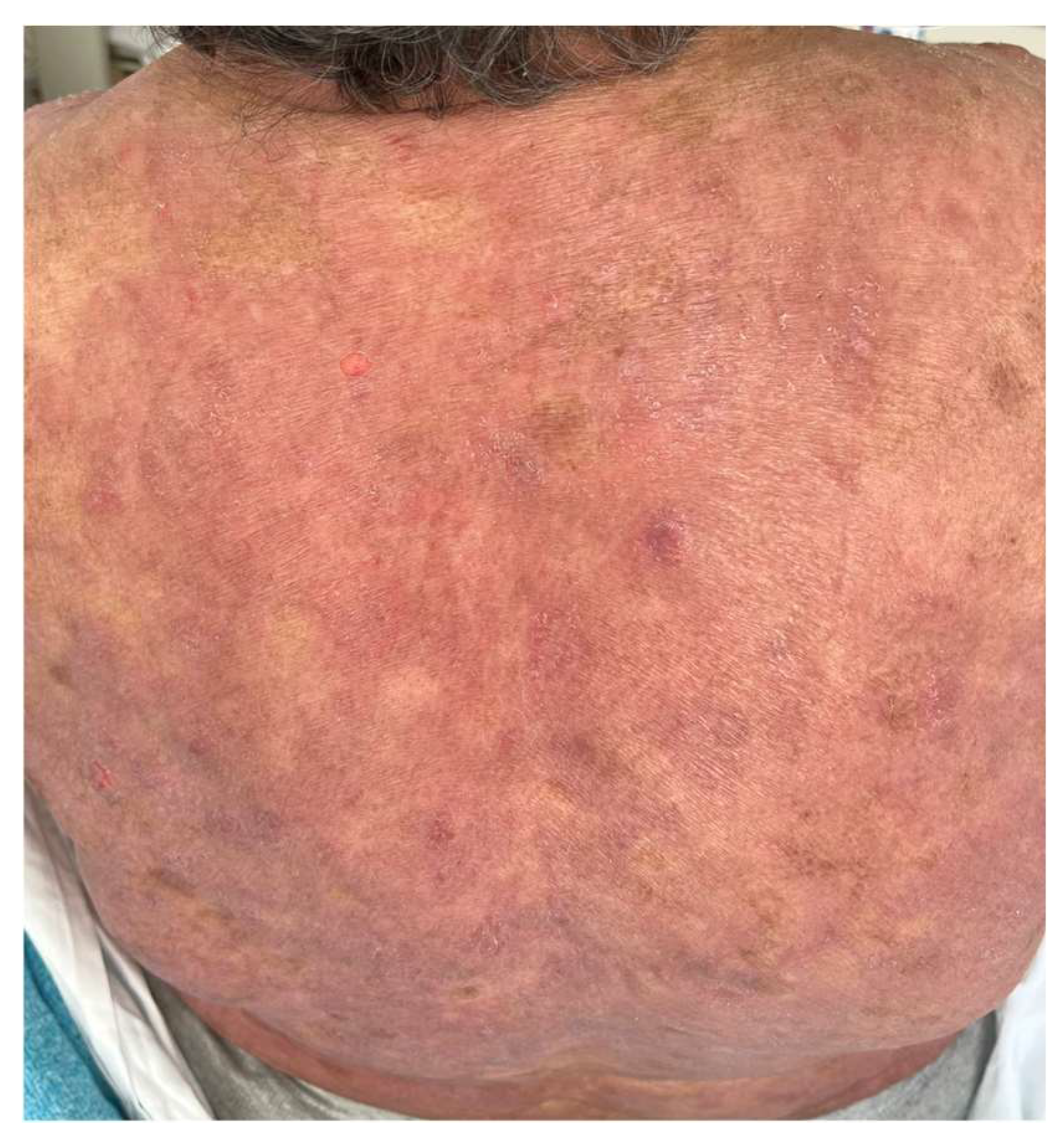
9. Cutaneous Adverse Events
10. Conclusions
11. Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Latzka, J.; Assaf, C.; Bagot, M.; Cozzio, A.; Dummer, R.; Guenova, E.; Gniadecki, R.; Hodak, E.; Jonak, C.; Klemke, C.D.; et al. EORTC consensus recommendations for the treatment of mycosis fungoides/Sezary syndrome—Update 2023. Eur. J. Cancer 2023, 195, 113343. [Google Scholar] [CrossRef]
- Quadri, I.; Reneau, J.C.; Hanel, W.; Chung, C.G. Advancements in the treatment of mycosis fungoides and Sezary syndrome: Monoclonal antibodies, immunotherapies, and Janus kinase inhibitors. Front. Immunol. 2023, 14, 1291259. [Google Scholar] [CrossRef]
- Stuver, R.; Geller, S. Advances in the treatment of mycoses fungoides and Sezary syndrome: A narrative update in skin-directed therapies and immune-based treatments. Front. Immunol. 2023, 14, 1284045. [Google Scholar] [CrossRef]
- Beygi, S.; Duran, G.E.; Fernandez-Pol, S.; Rook, A.H.; Kim, Y.H.; Khodadoust, M.S. Resistance to mogamulizumab is associated with loss of CCR4 in cutaneous T-cell lymphoma. Blood 2022, 139, 3732–3736. [Google Scholar] [CrossRef] [PubMed]
- Mukai, M.; Maeda, H.; Narushima, K.; Mould, D.R.; Greene, D. Population Pharmacokinetic Modeling of Mogamulizumab in Adults With Cutaneous T-Cell Lymphoma or Adult T-Cell Lymphoma. J. Clin. Pharmacol. 2020, 60, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Tobinai, K.; Takahashi, T.; Akinaga, S. Targeting chemokine receptor CCR4 in adult T-cell leukemia-lymphoma and other T-cell lymphomas. Curr. Hematol. Malig. Rep. 2012, 7, 235–240. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yoshie, O.; Fujisawa, R.; Nakayama, T.; Harasawa, H.; Tago, H.; Izawa, D.; Hieshima, K.; Tatsumi, Y.; Matsushima, K.; Hasegawa, H.; et al. Frequent expression of CCR4 in adult T-cell leukemia and human T-cell leukemia virus type 1-transformed T cells. Blood 2002, 99, 1505–1511. [Google Scholar] [CrossRef] [PubMed]
- Cartron, G.; Dacheux, L.; Salles, G.; Solal-Celigny, P.; Bardos, P.; Colombat, P.; Watier, H. Therapeutic activity of humanized anti-CD20 monoclonal antibody and polymorphism in IgG Fc receptor FcgammaRIIIa gene. Blood 2002, 99, 754–758. [Google Scholar] [CrossRef] [PubMed]
- Clynes, R.A.; Towers, T.L.; Presta, L.G.; Ravetch, J.V. Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nat. Med. 2000, 6, 443–446. [Google Scholar] [CrossRef]
- Voso, M.T.; Pantel, G.; Rutella, S.; Weis, M.; D’Alo, F.; Urbano, R.; Leone, G.; Haas, R.; Hohaus, S. Rituximab reduces the number of peripheral blood B-cells in vitro mainly by effector cell-mediated mechanisms. Haematologica 2002, 87, 918–925. [Google Scholar] [PubMed]
- Jefferis, R.; Lund, J.; Pound, J.D. IgG-Fc-mediated effector functions: Molecular definition of interaction sites for effector ligands and the role of glycosylation. Immunol. Rev. 1998, 163, 59–76. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, T. Engineered therapeutic antibodies with enhanced effector functions: Clinical application of the Potelligent(R) Technology. Korean J. Hematol. 2011, 46, 148–150. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shinkawa, T.N.K.; Yamane, N.; Shoji-Hosaka, E.; Kanda, Y.; Sakurada, M.; Uchida, K.; Anazawa, H.; Satoh, M.; Yamasaki, M.; Hanai, N.; et al. The absence of fucose but not the presence of galactose or bisecting N-acetylglucosamine of human IgG1 complex-type oligosaccharides shows the critical role of enhancing antibody-dependent cellular cytotoxicity. J. Biol. Chem. 2003, 278, 3466–3473. [Google Scholar] [CrossRef] [PubMed]
- Iida, S.M.H.; Inoue, M.; Shibata, M.; Nakano, R.; Yamane-Ohnuki, N.; Wakitani, M.; Yano, K.; Shitara, K.; Satoh, M. Nonfucosylated therapeutic IgG1 antibody can evade the inhibitory effect of serum immunoglobulin G on antibody-dependent cellular cytotoxicity through its high binding to FcgammaRIIIa. Clin. Cancer Res. 2006, 12, 2879–2887. [Google Scholar] [CrossRef] [PubMed]
- Shields, R.L.; Lai, J.; Keck, R.; O’Connell, L.Y.; Hong, K.; Meng, Y.G.; Weikert, S.H.; Presta, L.G. Lack of fucose on human IgG1 N-linked oligosaccharide improves binding to human Fcgamma RIII and antibody-dependent cellular toxicity. J. Biol. Chem. 2002, 277, 26733–26740. [Google Scholar] [CrossRef]
- Ishii, T.; Ishida, T.; Utsunomiya, A.; Inagaki, A.; Yano, H.; Komatsu, H.; Iida, S.; Imada, K.; Uchiyama, T.; Akinaga, S.; et al. Defucosylated humanized anti-CCR4 monoclonal antibody KW-0761 as a novel immunotherapeutic agent for adult T-cell leukemia/lymphoma. Clin. Cancer Res. 2010, 16, 1520–1531. [Google Scholar] [CrossRef]
- Wilcox, R.A. Mogamulizumab: 2 birds, 1 stone. Blood 2015, 125, 1847–1848. [Google Scholar] [CrossRef]
- Ishida, T.; Joh, T.; Uike, N.; Yamamoto, K.; Utsunomiya, A.; Yoshida, S.; Saburi, Y.; Miyamoto, T.; Takemoto, S.; Suzushima, H.; et al. Defucosylated anti-CCR4 monoclonal antibody (KW-0761) for relapsed adult T-cell leukemia-lymphoma: A multicenter phase II study. J. Clin. Oncol. 2012, 30, 837–842. [Google Scholar] [CrossRef]
- Yamamoto, K.; Utsunomiya, A.; Tobinai, K.; Tsukasaki, K.; Uike, N.; Uozumi, K.; Yamaguchi, K.; Yamada, Y.; Hanada, S.; Tamura, K.; et al. Phase I study of KW-0761, a defucosylated humanized anti-CCR4 antibody, in relapsed patients with adult T-cell leukemia-lymphoma and peripheral T-cell lymphoma. J. Clin. Oncol. 2010, 28, 1591–1598. [Google Scholar] [CrossRef] [PubMed]
- Gilson, D.; Whittaker, S.J.; Child, F.J.; Scarisbrick, J.J.; Illidge, T.M.; Parry, E.J.; Mohd Mustapa, M.F.; Exton, L.S.; Kanfer, E.; Rezvani, K.; et al. British Association of Dermatologists and U.K. Cutaneous Lymphoma Group guidelines for the management of primary cutaneous lymphomas 2018. Br. J. Dermatol. 2019, 180, 496–526. [Google Scholar] [CrossRef] [PubMed]
- Mehta-Shah, N.; Horwitz, S.M.; Ansell, S.; Ai, W.Z.; Barnes, J.; Barta, S.K.; Clemens, M.W.; Dogan, A.; Fisher, K.; Goodman, A.M.; et al. NCCN Guidelines Insights: Primary Cutaneous Lymphomas, Version 2.2020. J. Natl. Compr. Cancer Netw. 2020, 18, 522–536. [Google Scholar] [CrossRef]
- Ohtsuka, M.; Hamada, T.; Miyagaki, T.; Shimauchi, T.; Yonekura, K.; Kiyohara, E.; Fujita, H.; Izutsu, K.; Okuma, K.; Kawai, K.; et al. Outlines of the Japanese guidelines for the management of primary cutaneous lymphomas 2020. J. Dermatol. 2021, 48, e49–e71. [Google Scholar] [CrossRef] [PubMed]
- Calzado-Villareal, L.; Fernandez de Misa, R.; Lopez-Lerma, I.; Marin-Niebla, A.; Marín-Palomo, A.; Muniesa-Montserrat, C.; Novella-Canales, S.; Rodriguez Pinilla, S.M.; Zapatero, A. Guía Multidisciplinar Para El Abordaje Diagnóstico Y Terapéutico Del Paciente Con Linfoma Cutáneo Primario; Ósea GEdLyTAdM, (GELTAMO): Madrid, Spain, 2021. [Google Scholar]
- Sugaya, M. Clinical Guidelines and New Molecular Targets for Cutaneous Lymphomas. Int. J. Mol. Sci. 2021, 22, 11079. [Google Scholar] [CrossRef] [PubMed]
- Kamijo, H.; Miyagaki, T. Mycosis Fungoides and Sézary Syndrome: Updates and Review of Current Therapy. Curr. Treat. Opt. Oncol. 2021, 22, 10. [Google Scholar] [CrossRef] [PubMed]
- Duvic, M.; Pinter-Brown, L.C.; Foss, F.M.; Sokol, L.; Jorgensen, J.L.; Challagundla, P.; Dwyer, K.M.; Zhang, X.; Kurman, M.R.; Ballerini, R.; et al. Phase 1/2 study of mogamulizumab, a defucosylated anti-CCR4 antibody, in previously treated patients with cutaneous T-cell lymphoma. Blood 2015, 125, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Ogura, M.; Ishida, T.; Hatake, K.; Taniwaki, M.; Ando, K.; Tobinai, K.; Fujimoto, K.; Yamamoto, K.; Miyamoto, T.; Uike, N.; et al. Multicenter phase II study of mogamulizumab (KW-0761), a defucosylated anti-cc chemokine receptor 4 antibody, in patients with relapsed peripheral T-cell lymphoma and cutaneous T-cell lymphoma. J. Clin. Oncol. 2014, 32, 1157–1163. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.T.; Kartan, S.; Sokol, K.; Nikbakht, N.; Porcu, P. Clinical characteristics and outcomes of black patients with mycosis fungoides and Sézary syndrome: A subgroup analysis of the phase III MAVORIC trial. Leuk. Lymphoma 2021, 62, 1877–1883. [Google Scholar] [CrossRef]
- Kasamon, Y.L.; Chen, H.; de Claro, R.A.; Nie, L.; Ye, J.; Blumenthal, G.M.; Farrell, A.T.; Pazdur, R. FDA Approval Summary: Mogamulizumab-kpkc for Mycosis Fungoides and Sézary Syndrome. Clin. Cancer Res. 2019, 25, 7275–7280. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Sun, J.; Li, J.; Zhao, Y.; Zhang, T.; Yang, R.; Ma, X. Safety and efficacy profile of mogamulizumab (Poteligeo) in the treatment of cancers: An update evidence from 14 studies. BMC Cancer 2021, 21, 618. [Google Scholar] [CrossRef]
- Horwitz, S.; Zinzani, P.L.; Bagot, M.; Kim, Y.H.; Moskowitz, A.J.; Porcu, P.; Dwyer, K.; Sun, W.; Herr, F.M.; Scarisbrick, J. Lack of impact of type and extent of prior therapy on outcomes of mogamulizumab therapy in patients with cutaneous T cell lymphoma in the MAVORIC trial. Leuk. Lymphoma 2021, 62, 3109–3118. [Google Scholar] [CrossRef]
- Kitadate, A.; Ikeda, S.; Abe, F.; Takahashi, N.; Shimizu, N.; Matsue, K.; Tagawa, H. Histone deacetylase inhibitors downregulate CCR4 expression and decrease mogamulizumab efficacy in CCR4-positive mature T-cell lymphomas. Haematologica 2018, 103, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Shichijo, T.; Nosaka, K.; Tatetsu, H.; Higuchi, Y.; Endo, S.; Inoue, Y.; Toyoda, K.; Kikukawa, Y.; Kawakita, T.; Yasunaga, J.I.; et al. Beneficial impact of first-line mogamulizumab-containing chemotherapy in adult T-cell leukaemia-lymphoma. Br. J. Haematol. 2022, 198, 983–987. [Google Scholar] [CrossRef] [PubMed]
- Satake, A.; Konishi, A.; Azuma, Y.; Tsubokura, Y.; Yoshimura, H.; Hotta, M.; Nakanishi, T.; Fujita, S.; Nakaya, A.; Ito, T.; et al. Clinical efficacy of mogamulizumab for relapsed/refractory aggressive adult T-cell leukemia/lymphoma: A retrospective analysis. Eur. J. Haematol. 2020, 105, 704–711. [Google Scholar] [CrossRef]
- Fujimura, T.; Kambayashi, Y.; Ohuchi, K.; Amagai, R.; Muto, Y.; Aiba, S. Successful treatment of CCR4+ mycosis fungoides palmaris et plantaris with mogamulizumab monotherapy. Dermatol. Ther. 2020, 33, e13731. [Google Scholar] [CrossRef]
- Kim, Y.H.; Khodadoust, M.; de Masson, A.; Moins-Teisserenc, H.; Ito, T.; Dwyer, K.; Herr, F.; Bagot, M. Patient characteristics of long-term responders to mogamulizumab: Results from the MAVORIC study. Eur. J. Cancer 2021, 156 (Suppl. 1), S48–S49. [Google Scholar] [CrossRef] [PubMed]
- Fujii, K.; Sakamoto, Y.; Masaki, A.; Murase, T.; Tashiro, Y.; Yonekura, K.; Utsunomiya, A.; Ito, A.; Kusumoto, S.; Iida, S.; et al. Immunohistochemistry for CCR4 C-terminus predicts CCR4 mutations and mogamulizumab efficacy in adult T-cell leukemia/lymphoma. J. Pathol. Clin. Res. 2021, 7, 52–60. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Ishida, T.; Masaki, A.; Murase, T.; Yonekura, K.; Tashiro, Y.; Tokunaga, M.; Utsunomiya, A.; Ito, A.; Kusumoto, S.; et al. CCR4 mutations associated with superior outcome of adult T-cell leukemia/lymphoma under mogamulizumab treatment. Blood 2018, 132, 758–761. [Google Scholar] [CrossRef]
- Fujimura, T.; Furudate, S.; Tanita, K.; Sato, Y.; Kambayashi, Y.; Hidaka, T.; Aiba, S. Successful treatment of relapsed folliculotropic mycosis fungoides with mogamulizumab followed by intensity-modulated radiotherapy. J. Dermatol. 2018, 45, e84–e85. [Google Scholar] [CrossRef]
- Hisamoto, T.; Suga, H.; Kawana, Y.; Oka, T.; Miyagaki, T.; Sugaya, M.; Sato, S. A case of mycosis fungoides successfully treated with combination of bexarotene and mogamulizumab. Dermatol. Ther. 2021, 34, e14805. [Google Scholar] [CrossRef]
- Amagai, R.; Kambayashi, Y.; Ohuchi, K.; Furudate, S.; Hashimoto, A.; Asano, Y.; Fujimura, T. Cutaneous T cell lymphoma treated with mogamulizumab monotherapy and mogamulizumab plus etoposide combined therapy: A real-world case series. Dermatol. Ther. 2022, 35, e15858. [Google Scholar] [CrossRef]
- Caruso, L.; Castellino, A.; Dessi, D.; Flenghi, L.; Giordano, A.; Ibatici, A.; Massone, C.; Pileri, A.; Proietti, I.; Pupo, L.; et al. Italian Real-Life Experience on the Use of Mogamulizumab in Patients with Cutaneous T-Cell Lymphomas. Cancer Manag. Res. 2022, 14, 3205–3221. [Google Scholar] [CrossRef]
- Molloy, K.; Vico, C.; Ortiz-Romero, P.L.; Scarisbrick, J.J. Real-world experience of using mogamulizumab in relapsed/refractory mycosis fungoides/Sézary syndrome. Br. J. Dermatol. 2021, 184, 978–981. [Google Scholar] [CrossRef]
- Jouandet, M.; Nakouri, I.; Nadin, L.; Kieny, A.; Samimi, M.; Adamski, H.; Quereux, G.; Chaby, G.; Dompmartin, A.; L’Orphelin, J.M. Impact of Mogamulizumab in Real-Life Advanced Cutaneous T-Cell Lymphomas: A Multicentric Retrospective Cohort Study. Cancers 2022, 14, 1659. [Google Scholar] [CrossRef]
- Trum, N.A.; Zain, J.; Martinez, X.U.; Parekh, V.; Afkhami, M.; Abdulla, F.; Carson, K.R.; Rosen, S.T.; Bennett, C.L.; Querfeld, C. Mogamulizumab efficacy is underscored by its associated rash that mimics cutaneous T-cell lymphoma: A retrospective single-centre case series. Br. J. Dermatol. 2022, 186, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Beylot-Barry, M.; Quereux, G.; Nardin, C.; Duval-Modeste, A.B.; Dereure, O.; Dalac-Rat, S.; Dobos, G.; Pham-Ledard, A.; Ram-Wolff, C.; D’Incan, M.; et al. Effectiveness of mogamulizumab in patients with Mycosis Fungoides or Sezary syndrome: A multicentre, retrospective, real-world French study. J. Eur. Acad. Dermatol. Venereol. 2023, 37, 1777–1784. [Google Scholar] [CrossRef] [PubMed]
- Remak, E.; Hawkins, N.; Jones, T.; Otley, M.; Twigger, R.; Prince, M. Understanding relative survival outcomes for patients with cutaneous T-cell lymphoma (CTCL) subtypes mycosis fungoides and Sezary syndrome treated with mogamulizumab or vorinostat: Combining Australian real-world evidence and MAVORIC phase 3 trial data. Eur. J. Cancer 2021, 156 (Suppl. 1), S18. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Bagot, M.; Pinter-Brown, L.; Rook, A.H.; Porcu, P.; Horwitz, S.M.; Whittaker, S.; Tokura, Y.; Vermeer, M.; Zinzani, P.L.; et al. Mogamulizumab versus vorinostat in previously treated cutaneous T-cell lymphoma (MAVORIC): An international, open-label, randomised, controlled phase 3 trial. Lancet Oncol. 2018, 19, 1192–1204. [Google Scholar] [CrossRef] [PubMed]
- Lewis, D.J.; Rook, A.H. Mogamulizumab in the treatment of advanced mycosis fungoides and Sézary syndrome: Safety and efficacy. Expert Rev. Anticancer Ther. 2020, 20, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Khodadoust, M.S.; Rook, A.H.; Porcu, P.; Foss, F.; Moskowitz, A.J.; Shustov, A.; Shanbhag, S.; Sokol, L.; Fling, S.P.; Ramchurren, N.; et al. Pembrolizumab in Relapsed and Refractory Mycosis Fungoides and Sezary Syndrome: A Multicenter Phase II Study. J. Clin. Oncol. 2020, 38, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, P.; Battistella, M.; Roelens, M.; Ram-Wolff, C.; Herms, F.; Frumholtz, L.; Bouaziz, J.D.; Brice, P.; Moins-Teisserenc, H.; Bagot, M.; et al. Association of autoimmunity and long-term complete remission in patients with Sezary syndrome treated with mogamulizumab. Br. J. Dermatol. 2019, 180, 419–420. [Google Scholar] [CrossRef] [PubMed]
- Porcu, P.; Hudgens, S.; Horwitz, S.; Quaglino, P.; Cowan, R.; Geskin, L.; Beylot-Barry, M.; Floden, L.; Bagot, M.; Tsianakas, A.; et al. Quality of Life Effect of the Anti-CCR4 Monoclonal Antibody Mogamulizumab Versus Vorinostat in Patients With Cutaneous T-cell Lymphoma. Clin. Lymphoma Myeloma Leuk. 2021, 21, 97–105. [Google Scholar] [CrossRef]
- Ishitsuka, K.; Yurimoto, S.; Tsuji, Y.; Iwabuchi, M.; Takahashi, T.; Tobinai, K. Safety and effectiveness of mogamulizumab in relapsed or refractory adult T-cell leukemia-lymphoma. Eur. J. Haematol. 2019, 102, 407–415. [Google Scholar] [CrossRef]
- Fukushima, T.; Nomura, S.; Shimoyama, M.; Shibata, T.; Imaizumi, Y.; Moriuchi, Y.; Tomoyose, T.; Uozumi, K.; Kobayashi, Y.; Fukushima, N.; et al. Japan Clinical Oncology Group (JCOG) prognostic index and characterization of long-term survivors of aggressive adult T-cell leukaemia-lymphoma (JCOG0902A). Br. J. Haematol. 2014, 166, 739–748. [Google Scholar] [CrossRef]
- Katsuya, H.; Yamanaka, T.; Ishitsuka, K.; Utsunomiya, A.; Sasaki, H.; Hanada, S.; Eto, T.; Moriuchi, Y.; Saburi, Y.; Miyahara, M.; et al. Prognostic index for acute- and lymphoma-type adult T-cell leukemia/lymphoma. J. Clin. Oncol. 2012, 30, 1635–1640. [Google Scholar] [CrossRef]
- Dai, J.; Almazan, T.H.; Hong, E.K.; Khodadoust, M.S.; Arai, S.; Weng, W.K.; Kim, Y.H. Potential Association of Anti-CCR4 Antibody Mogamulizumab and Graft-vs-Host Disease in Patients With Mycosis Fungoides and Sézary Syndrome. JAMA Dermatol. 2018, 154, 728–730. [Google Scholar] [CrossRef]
- Hosoi, H.; Mushino, T.; Nishikawa, A.; Hashimoto, H.; Murata, S.; Hatanaka, K.; Tamura, S.; Hanaoka, N.; Shimizu, N.; Sonoki, T. Severe graft-versus-host disease after allogeneic hematopoietic stem cell transplantation with residual mogamulizumab concentration. Int. J. Hematol. 2018, 107, 717–719. [Google Scholar] [CrossRef]
- Inoue, Y.; Nishimura, N.; Murai, M.; Matsumoto, M.; Watanabe, M.; Yamada, A.; Izaki, M.; Nosaka, K.; Matsuoka, M. Prevention of acute graft-versus-host disease in adult T-cell leukemia-lymphoma patients who received mogamulizumab before allogeneic hematopoietic cell transplantation. Int. J. Hematol. 2022, 115, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Nosaka, K.; Crawford, B.; Yi, J.; Kuan, W.; Matsumoto, T.; Takahashi, T. Systematic review of survival outcomes for relapsed or refractory adult T-cell leukemia-lymphoma. Eur. J. Haematol. 2022, 108, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Mori, S.; Kiyotani, K.; Ota, Y.; Gotoh, O.; Kusumoto, S.; Nakano, N.; Suehiro, Y.; Ito, A.; Choi, I.; et al. Genomic determinants impacting the clinical outcome of mogamulizumab treatment for adult T-cell leukemia/lymphoma. Haematologica 2022, 107, 2418–2431. [Google Scholar] [CrossRef] [PubMed]
- Alsina, M.; Becker, P.S.; Zhong, X.; Adams, A.; Hari, P.; Rowley, S.; Stadtmauer, E.A.; Vesole, D.H.; Logan, B.; Weisdorf, D.; et al. Lenalidomide maintenance for high-risk multiple myeloma after allogeneic hematopoietic cell transplantation. Biol. Blood Marrow Transplant. 2014, 20, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Hirosawa, M.; Goto, M.; Oku, M.; Akao, K.; Kitamura, N.; Nakanishi, T.; Tanaka, A.; Niino, D.; Higashi, T.; Morimoto, H.; et al. Mogamulizumab for post-transplant relapse of adult T-cell leukemia/lymphoma: A case study. Int. J. Hematol. 2023, 117, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Inoue, Y.; Endo, S.; Matsuno, N.; Kikukawa, Y.; Shichijo, T.; Koga, K.; Takaki, A.; Iwanaga, K.; Nishimura, N.; Fuji, S.; et al. Safety of mogamulizumab for relapsed ATL after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2019, 54, 338–342. [Google Scholar] [CrossRef] [PubMed]
- Kneppers, E.; van der Holt, B.; Kersten, M.J.; Zweegman, S.; Meijer, E.; Huls, G.; Cornelissen, J.J.; Janssen, J.J.; Huisman, C.; Cornelisse, P.B.; et al. Lenalidomide maintenance after nonmyeloablative allogeneic stem cell transplantation in multiple myeloma is not feasible: Results of the HOVON 76 Trial. Blood 2011, 118, 2413–2419. [Google Scholar] [CrossRef] [PubMed]
- Kroger, N.; Zabelina, T.; Klyuchnikov, E.; Kropff, M.; Pfluger, K.H.; Burchert, A.; Stubig, T.; Wolschke, C.; Ayuk, F.; Hildebrandt, Y.; et al. Toxicity-reduced, myeloablative allograft followed by lenalidomide maintenance as salvage therapy for refractory/relapsed myeloma patients. Bone Marrow Transplant. 2013, 48, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, H.; Itonaga, H.; Sawayama, Y.; Furumoto, T.; Fujioka, M.; Chiwata, M.; Toriyama, E.; Kasai, S.; Nakashima, J.; Horai, M.; et al. Treatment with mogamulizumab or lenalidomide for relapsed adult T-cell leukemia/lymphoma after allogeneic hematopoietic stem cell transplantation: The Nagasaki transplant group experience. Hematol. Oncol. 2020, 38, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Jo, T.; Takemoto, S.; Suzushima, H.; Suehiro, Y.; Choi, I.; Yoshimitsu, M.; Saburi, Y.; Nosaka, K.; Utsunomiya, A.; et al. Follow-up of a randomised phase II study of chemotherapy alone or in combination with mogamulizumab in newly diagnosed aggressive adult T-cell leukaemia-lymphoma: Impact on allogeneic haematopoietic stem cell transplantation. Br. J. Haematol. 2019, 184, 479–483. [Google Scholar] [CrossRef]
- Jo, T.; Matsuzaka, K.; Shioya, H.; Tominaga, H.; Sakai, T.; Kaneko, Y.; Hayashi, S.; Matsuo, M.; Taguchi, J.; Abe, K.; et al. Mogamulizumab Plus EPOCH Therapy for Patients With Newly Diagnosed Aggressive Adult T-cell Leukemia/lymphoma. Anticancer Res. 2020, 40, 5237–5243. [Google Scholar] [CrossRef]
- Masuda, Y.; Tatsuno, K.; Kitano, S.; Miyazawa, H.; Ishibe, J.; Aoshima, M.; Shimauchi, T.; Fujiyama, T.; Ito, T.; Tokura, Y. Mogamulizumab-induced photosensitivity in patients with mycosis fungoides and other T-cell neoplasms. J. Eur. Acad. Dermatol. Venereol. 2018, 32, 1456–1460. [Google Scholar] [CrossRef]
- Schmiedel, B.J.; Singh, D.; Madrigal, A.; Valdovino-Gonzalez, A.G.; White, B.M.; Zapardiel-Gonzalo, J.; Ha, B.; Altay, G.; Greenbaum, J.A.; McVicker, G.; et al. Impact of Genetic Polymorphisms on Human Immune Cell Gene Expression. Cell 2018, 175, 1701–1715.e16. [Google Scholar] [CrossRef] [PubMed]
- Fong, S.; Hong, E.K.; Khodadoust, M.S.; Li, S.; Hoppe, R.T.; Kim, Y.H.; Hiniker, S.M. Low-Dose Total Skin Electron Beam Therapy Combined With Mogamulizumab for Refractory Mycosis Fungoides and Sezary Syndrome. Adv. Radiat. Oncol. 2021, 6, 100629. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Hoppe, R.T.; Rook, A.H.; Maity, A.; Geskin, L.J.; Horowitz, D.P.; Finnegan, G.; Khodadoust, M.; Weng, W.K.; Lares, A.; et al. A Single-Arm PHASE 2A Study of NM-IL-12 (rHu-IL12) in Patients with Mycosis Fungoides-Type CTCL (MF) Undergoing Low-Dose TOTAL Skin Electron BEAM Therapy (LD-TSEBT). Blood 2016, 128, 4165. [Google Scholar] [CrossRef]
- Maemoto, H.; Ariga, T.; Kusada, T.; Heianna, J.; Manabe, Y.; Miyakawa, A.; Nakachi, S.; Morishima, S.; Iraha, S.; Ganaha, F.; et al. Radiation-induced dermatitis after administration of mogamulizumab for adult T-cell leukaemia/lymphoma: A multi-institutional retrospective study. Jpn. J. Clin. Oncol. 2019, 49, 153–159. [Google Scholar] [CrossRef]
- Ohuchi, K.; Fujimura, T.; Kambayashi, Y.; Amagai, R.; Lyu, C.; Tanita, K.; Sato, Y.; Aiba, S. Successful treatment of mogamulizumab-resistant mycosis fungoides with mogamulizumab plus etoposide combined therapy: Investigation of the immunomodulatory effects of etoposide on the tumor microenvironment. Dermatol. Ther. 2020, 33, e13487. [Google Scholar] [CrossRef]
- Kelly-Sell, M.J.; Kim, Y.H.; Straus, S.; Benoit, B.; Harrison, C.; Sutherland, K.; Armstrong, R.; Weng, W.K.; Showe, L.C.; Wysocka, M.; et al. The histone deacetylase inhibitor, romidepsin, suppresses cellular immune functions of cutaneous T-cell lymphoma patients. Am. J. Hematol. 2012, 87, 354–360. [Google Scholar] [CrossRef]
- Zinzani, P.L.; Horwitz, S.M.; Kim, Y.H.; Moskowitz, A.J.; Porcu, P.; Scarisbrick, J.; Leoni, M.; Dwyer, K.; Sun, W.; Nikonova, E.; et al. Efficacy of mogamulizumab by prior systemic therapy in patients with previously treated cutaneous T-cell lymphoma: Post hoc analysis from the phase 3 MAVORIC study. Blood 2018, 132, 1619. [Google Scholar] [CrossRef]
- Sultan, A.S.; Mostoufi, B.; Papadimitriou, J.C.; Koka, R.; Basile, J.; Younis, R.H. Large Cell Transformation of Oral Mycosis Fungoides. Head. Neck Pathol. 2018, 12, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Teoli, M.; Mandel, V.D.; Franceschini, C.; Saraceni, P.L.; Cicini, M.P.; Ardigo, M. Mogamulizumab and bexarotene are a promising association for the treatment of advanced cutaneous T-cell lymphomas: A case series. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 8118–8128. [Google Scholar] [CrossRef] [PubMed]
- Rook, A.H.; Gelfand, J.M.; Wysocka, M.; Troxel, A.B.; Benoit, B.; Surber, C.; Elenitsas, R.; Buchanan, M.A.; Leahy, D.S.; Watanabe, R.; et al. Topical resiquimod can induce disease regression and enhance T-cell effector functions in cutaneous T-cell lymphoma. Blood 2015, 126, 1452–1461. [Google Scholar] [CrossRef]
- Gottlieb, S.L.; Wolfe, J.T.; Fox, F.E.; DeNardo, B.J.; Macey, W.H.; Bromley, P.G.; Lessin, S.R.; Rook, A.H. Treatment of cutaneous T-cell lymphoma with extracorporeal photopheresis monotherapy and in combination with recombinant interferon alfa: A 10-year experience at a single institution. J. Am. Acad. Dermatol. 1996, 35, 946–957. [Google Scholar] [CrossRef]
- Breen, I.D.; Brumfiel, C.M.; Patel, M.H.; Rosenthal, A.C.; Rule, W.G.; DiCaudo, D.J.; Craig, F.E.; Pittelkow, M.R.; Mangold, A.R. Mogamulizumab-induced interface dermatitis drug rash treated successfully with methotrexate and extracorporeal photopheresis in a patient with Sezary syndrome. JAAD Case Rep. 2021, 9, 24–27. [Google Scholar] [CrossRef] [PubMed]
- Asokan, I.; Singh, S.; Moesch, J.; Ho, J.; Akilov, O.E. Effective treatment of mogamulizumab-induced head and neck dermatitis with fluconazole in a patient with peripheral T-cell lymphoma. JAAD Case Rep. 2022, 20, 44–46. [Google Scholar] [CrossRef] [PubMed]
- Nosbaum, A.; Prevel, N.; Truong, H.A.; Mehta, P.; Ettinger, M.; Scharschmidt, T.C.; Ali, N.H.; Pauli, M.L.; Abbas, A.K.; Rosenblum, M.D. Cutting Edge: Regulatory T Cells Facilitate Cutaneous Wound Healing. J. Immunol. 2016, 196, 2010–2014. [Google Scholar] [CrossRef] [PubMed]
- Saito, M.; Ishii, T.; Urakawa, I.; Matsumoto, A.; Masaki, A.; Ito, A.; Kusumoto, S.; Suzuki, S.; Takahashi, T.; Morita, A.; et al. Robust CD8+ T-cell proliferation and diversification after mogamulizumab in patients with adult T-cell leukemia-lymphoma. Blood Adv. 2020, 4, 2180–2191. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Carson, K.R.; Staser, K.W.; Mehta-Shah, N.; Schaffer, A.; Rosman, I.S.; Musiek, A. Mogamulizumab-Associated Cutaneous Granulomatous Drug Eruption Mimicking Mycosis Fungoides but Possibly Indicating Durable Clinical Response. JAMA Dermatol. 2019, 155, 968–971. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.Y.; Hirotsu, K.E.; Neal, T.M.; Raghavan, S.S.; Kwong, B.Y.; Khodadoust, M.S.; Brown, R.A.; Novoa, R.A.; Kim, Y.H.; Rieger, K.E. Histopathologic Characterization of Mogamulizumab-associated Rash. Am. J. Surg. Pathol. 2020, 44, 1666–1676. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, K.E.; Neal, T.M.; Khodadoust, M.S.; Wang, J.Y.; Rieger, K.E.; Strelo, J.; Hong, E.; Kim, Y.H.; Kwong, B.Y. Clinical Characterization of Mogamulizumab-Associated Rash During Treatment of Mycosis Fungoides or Sézary Syndrome. JAMA Dermatol. 2021, 157, 700–707. [Google Scholar] [CrossRef]
- Carrasquilla-Capilla, A.; Pham, F.; Fontaine, J.; Harou, O.; Dalle, S. Bone granulomatous infiltrate associated with mogamulizumab in a patient with mycosis fungoides. Lancet Haematol. 2023, 10, e155. [Google Scholar] [CrossRef]
- Raval, N.S.; Snowden, C.K.; De Monnin, K.S.; Yokoyama, C.C.; Choi, J.; Mehta-Shah, N.; Rosman, I.S.; Pavlisin, J.; Musiek, A.C. Scarring alopecia developing after mogamulizumab-associated rash. Eur. J. Dermatol. 2021, 31, 841–843. [Google Scholar] [CrossRef]
- Raval, N.S.; Alexander, N.A.; De Monnin, K.; Yokoyama, C.C.; Mehta-Shah, N.; Rosman, I.S.; Musiek, A.C. Alopecia areata after mogamulizumab treatment. JAAD Case Rep. 2022, 19, 68–70. [Google Scholar] [CrossRef]
- de Masson, A.; Darbord, D.; Dobos, G.; Boisson, M.; Roelens, M.; Ram-Wolff, C.; Cassius, C.; Le Buanec, H.; de la Grange, P.; Jouenne, F.; et al. Macrophage-derived CXCL9 and CXCL11, T-cell skin homing, and disease control in mogamulizumab-treated CTCL patients. Blood 2022, 139, 1820–1832. [Google Scholar] [CrossRef]
- Tokunaga, M.; Yonekura, K.; Nakamura, D.; Haraguchi, K.; Tabuchi, T.; Fujino, S.; Hayashida, M.; Maekawa, K.; Arai, A.; Nakano, N.; et al. Clinical significance of cutaneous adverse reaction to mogamulizumab in relapsed or refractory adult T-cell leukaemia-lymphoma. Br. J. Haematol. 2018, 181, 539–542. [Google Scholar] [CrossRef]
- Yonekura, K.; Kanzaki, T.; Gunshin, K.; Kawakami, N.; Takatsuka, Y.; Nakano, N.; Tokunaga, M.; Kubota, A.; Takeuchi, S.; Kanekura, T.; et al. Effect of anti-CCR4 monoclonal antibody (mogamulizumab) on adult T-cell leukemia-lymphoma: Cutaneous adverse reactions may predict the prognosis. J. Dermatol. 2014, 41, 239–244. [Google Scholar] [CrossRef]
- Trager, M.H.; de Clippelé, D.; Ram-Wolff, C.; de Masson, A.; Vignon-Pennamen, M.D.; Battistella, M.; Michel, L.; Bagot, M.; Dobos, G. Mogamulizumab-induced Mucocutaneous Lichenoid Reaction: A Case Report and Short Review. Acta Derm. Venereol. 2020, 100, adv00158. [Google Scholar] [CrossRef]
- Lehner, G.M.; Schacht, V.; Angela, Y.; Grimmelmann, I.; Gutzmer, R. Psoriasis vulgaris triggered by treatment with mogamulizumab in a patient with cutaneous T-cell lymphoma. J. Dtsch. Dermatol. Ges. 2021, 19, 1355–1358. [Google Scholar] [CrossRef]
- Algarni, A.S.; Ram-Wolff, C.; Bagot, M.; De Masson, A. Mogamulizumab-induced vitiligo in patients with Sézary syndrome: Three cases. Eur. J. Dermatol. 2021, 31, 213–216. [Google Scholar] [CrossRef]
- Herrmann, J.L.; Syklawer, E.; Tarrillion, M.; Duvic, M.; Hughey, L.C. Concomitant mycosis fungoides and vitiligo: How mycosis fungoides may contribute to melanocyte destruction. Dermatology 2015, 230, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Hishizawa, M.; Kataoka, T.R.; Ohmori, K.; Takaori-Kondo, A.; Miyachi, Y.; Kabashima, K. Stevens-Johnson Syndrome Associated with Mogamulizumab-induced Deficiency of Regulatory T cells in an Adult T-cell Leukaemia Patient. Acta Derm. Venereol. 2015, 95, 606–607. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Ito, A.; Sato, F.; Kusumoto, S.; Iida, S.; Inagaki, H.; Morita, A.; Akinaga, S.; Ueda, R. Stevens-Johnson Syndrome associated with mogamulizumab treatment of adult T-cell leukemia / lymphoma. Cancer Sci. 2013, 104, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Lazaridou, I.; Calvani, J.; Annabi, E.; Moins-Teisserenc, H.; Ta, V.A.; Rivet, J.; Ram-Wolff, C.; Dumont, M.; Mahevas, T.; Vignon-Pennamen, M.D.; et al. Toxic epidermal necrolysis possibly associated with mogamulizumab in a patient with Sezary syndrome. J. Eur. Acad. Dermatol. Venereol. 2023, 37, e715–e717. [Google Scholar] [CrossRef] [PubMed]
- Tanba, K.; Uoshima, N.; Uchiyama, H.; Kawata, E.; Isa, R.; Yamaguchi, J.; Tsutsumi, Y.; Akaogi, T.; Kobayashi, Y.; Katsura, K.; et al. Toxic epidermal necrolysis in adult T cell leukemia/lymphoma treated with mogamulizumab. Ann. Hematol. 2016, 95, 661–662. [Google Scholar] [CrossRef] [PubMed]
- Alcalay, J.; David, M.; Shohat, B.; Sandbank, M. Generalized vitiligo following Sezary syndrome. Br. J. Dermatol. 1987, 116, 851–855. [Google Scholar] [CrossRef]
- Bouloc, A.; Grange, F.; Delfau-Larue, M.H.; Dieng, M.T.; Tortel, M.C.; Avril, M.F.; Revuz, J.; Bagot, M.; Wechsler, J. Leucoderma associated with flares of erythrodermic cutaneous T-cell lymphomas: Four cases. The French Study Group of Cutaneous Lymphomas. Br. J. Dermatol. 2000, 143, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Knol, A.C.; Quereux, G.; Marques-Briand, S.; Pandolfino, M.C.; Khammari, A.; Guilloux, Y.; Dreno, B. Pathogenetic mechanisms of vitiligo in a patient with Sezary syndrome. Br. J. Dermatol. 2005, 153, 1207–1212. [Google Scholar] [CrossRef]
- Sasaki, K.; Iinuma, S.; Fujii, M.; Shibuya, T.; Kanno, K.; Honma, M.; Hatayama, M.; Ishida-Yamamoto, A. Radiation recall dermatitis induced by mogamulizumab. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e107–e108. [Google Scholar] [CrossRef]
- Vico-Alonso, C.; Sánchez-Velázquez, A.; Puerta-Peña, M.; Garrido-Ruiz, M.C.; Velasco-Tamariz, V.; Ortiz-Romero, P.L. Two cases of generalized eruptive lentiginosis in cutaneous T-cell lymphoma following mogamulizumab treatment. Int. J. Dermatol. 2022, 61, e488–e489. [Google Scholar] [CrossRef] [PubMed]
- Barré, M.; Valois, A.; Okhremchuk, I.; Sair, M.; Masbou, J.; Abed, S.; Boye, T.; Morand, J.J. Ecthyma gangrenosum complicating mogamulizumab treatment of Sézary syndrome. Ann. Dermatol. Venereol. 2021, 148, 63–65. [Google Scholar] [CrossRef]
- Kamachi, K.; Shindo, T.; Miyahara, M.; Kitaura, K.; Akashi, M.; Shin, I.T.; Suzuki, R.; Oshima, K.; Kimura, S. Epstein-Barr virus-related diffuse large B-cell lymphoma in mogamulizumab-treated adult T-cell leukemia with incomplete T-cell reconstitution. Int. J. Hematol. 2019, 109, 221–227. [Google Scholar] [CrossRef]
- Tanaka, H.; Aoki, H.; Sugita, Y.; Shimizu, R.; Kiko, K.; Mochida, H.; Suzuki, Y. Development of Epstein-Barr Virus-related Primary Diffuse Large B-cell Lymphoma of the Central Nervous System in a Patient with Peripheral T-cell Lymphoma, Not Otherwise Specified after Mogamulizumab Treatment. Intern. Med. 2017, 56, 2759–2763. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Bargout, M.; Hon, J.D.; Yaghmour, B.; Mohrbacher, A.; Yaghmour, G. The Emergence of Epstein-Barr Virus-Related Diffuse Large B-Cell Lymphoma With Mogamulizumab. World J. Oncol. 2020, 11, 41–43. [Google Scholar] [CrossRef]
- Roelens, M.; de Masson, A.; Andrillon, A.; Ram-Wolff, C.; Biard, L.; Boisson, M.; Mourah, S.; Battistella, M.; Toubert, A.; Bagot, M.; et al. Mogamulizumab induces long-term immune restoration and reshapes tumour heterogeneity in Sézary syndrome. Br. J. Dermatol. 2022, 186, 1010–1025. [Google Scholar] [CrossRef]
- Trum, N.A.; Zain, J.; Abad, C.; Rosen, S.T.; Querfeld, C. Dupilumab as a therapy option for treatment refractory mogamulizumab-associated rash. JAAD Case Rep. 2021, 14, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Musiek, A.C.M.; Rieger, K.E.; Bagot, M.; Choi, J.N.; Fisher, D.C.; Guitart, J.; Haun, P.L.; Horwitz, S.M.; Huen, A.O.; Kwong, B.Y.; et al. Dermatologic Events Associated with the Anti-CCR4 Antibody Mogamulizumab: Characterization and Management. Dermatol. Ther. 2022, 12, 29–40. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Guarino, M.; Ortiz, P.; Gallardo, F.; Llamas-Velasco, M. Clinical and Real-World Effectiveness of Mogamulizumab: A Narrative Review. Int. J. Mol. Sci. 2024, 25, 2203. https://doi.org/10.3390/ijms25042203
Fernández-Guarino M, Ortiz P, Gallardo F, Llamas-Velasco M. Clinical and Real-World Effectiveness of Mogamulizumab: A Narrative Review. International Journal of Molecular Sciences. 2024; 25(4):2203. https://doi.org/10.3390/ijms25042203
Chicago/Turabian StyleFernández-Guarino, Montserrat, Pablo Ortiz, Fernando Gallardo, and Mar Llamas-Velasco. 2024. "Clinical and Real-World Effectiveness of Mogamulizumab: A Narrative Review" International Journal of Molecular Sciences 25, no. 4: 2203. https://doi.org/10.3390/ijms25042203
APA StyleFernández-Guarino, M., Ortiz, P., Gallardo, F., & Llamas-Velasco, M. (2024). Clinical and Real-World Effectiveness of Mogamulizumab: A Narrative Review. International Journal of Molecular Sciences, 25(4), 2203. https://doi.org/10.3390/ijms25042203






