Transcriptomic and Epigenomic Responses to Cortisol-Mediated Stress in Rainbow Trout (Oncorhynchus mykiss) Skeletal Muscle
Abstract
1. Introduction
2. Results
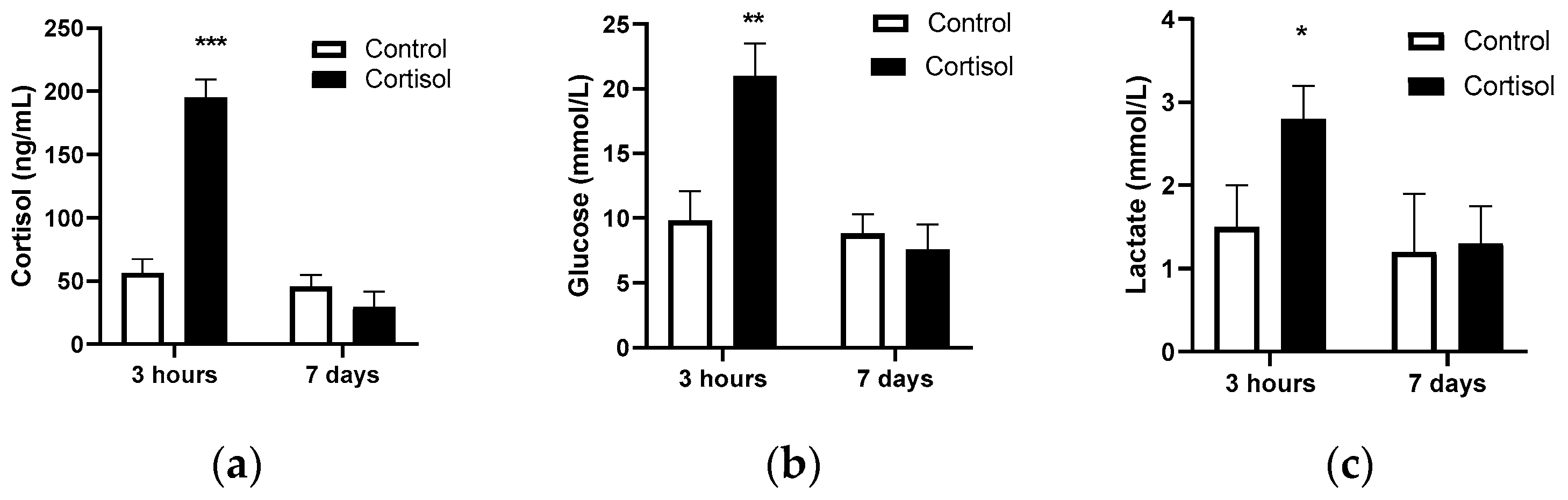
2.1. Assessment of Physiological and Transcriptomic Response to Stress
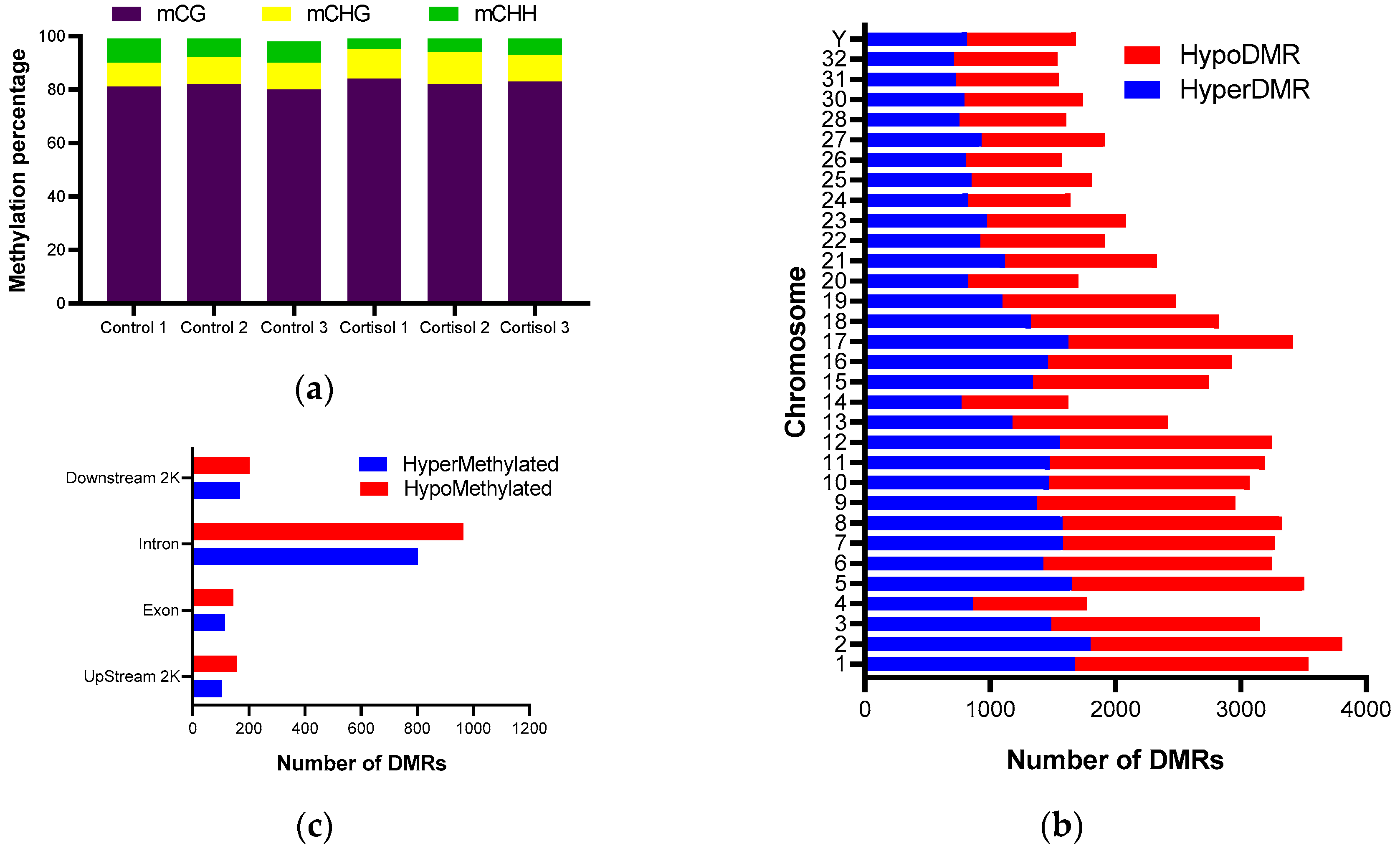
2.2. Analysis of DNA Methylation Induced by Cortisol
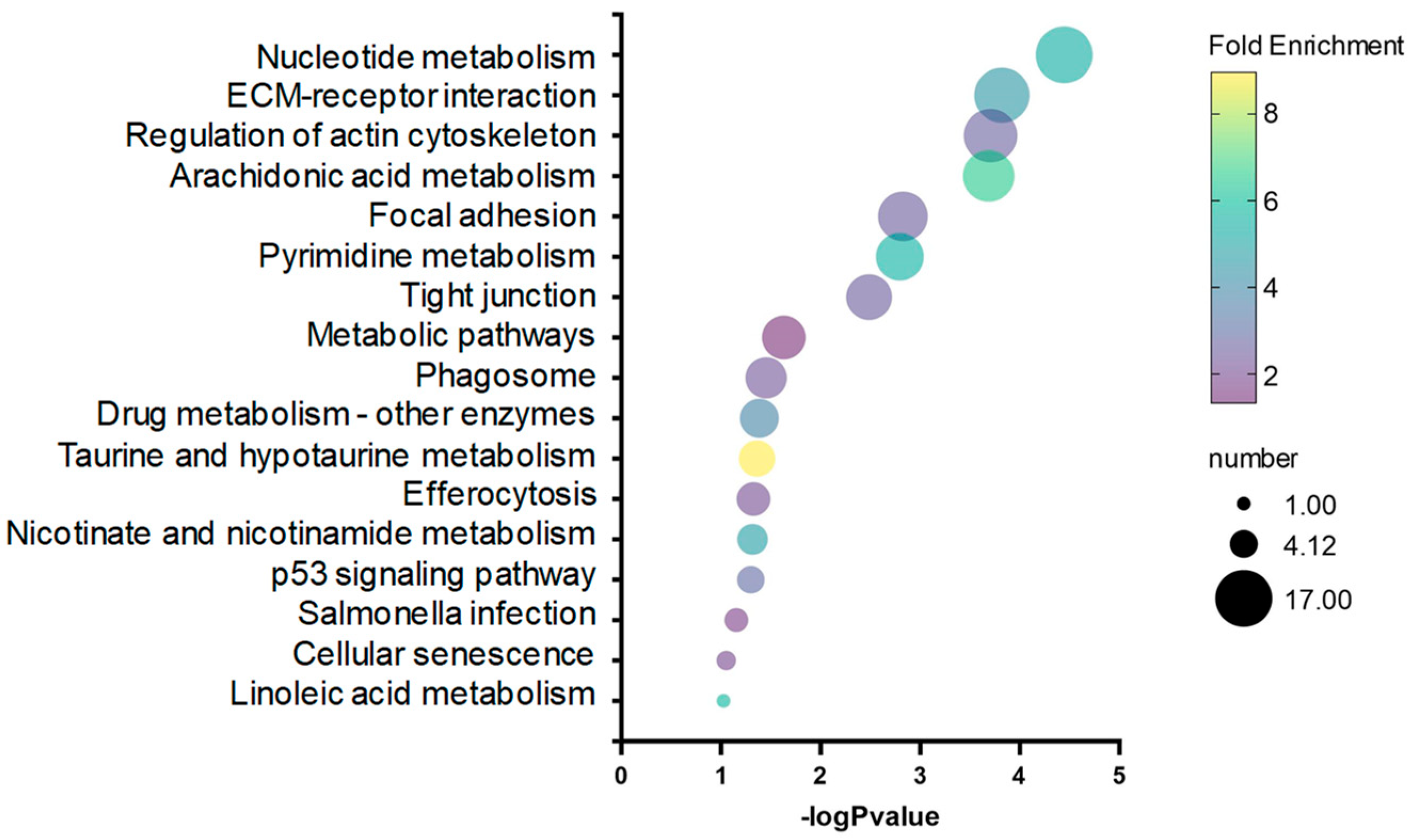
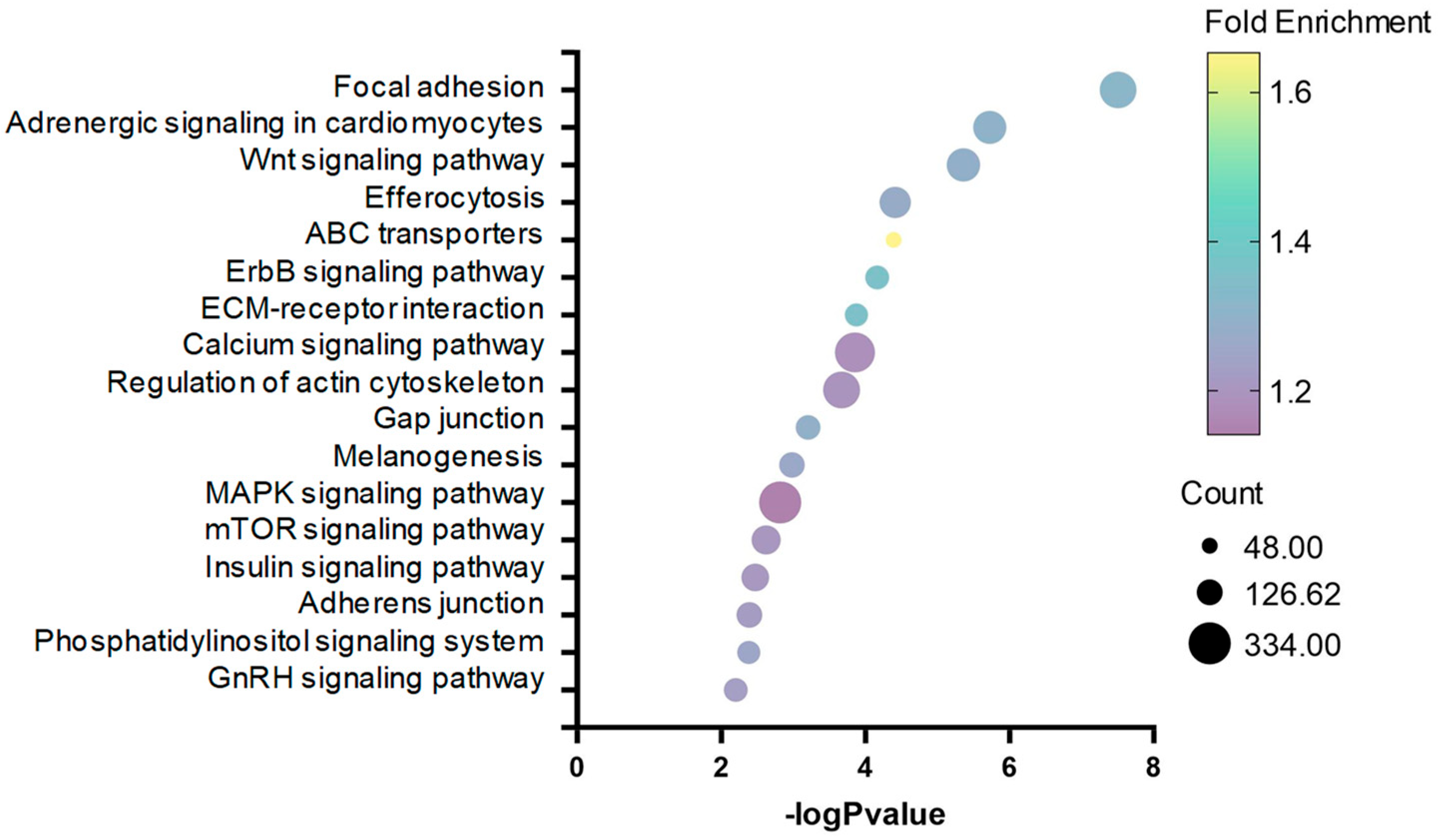
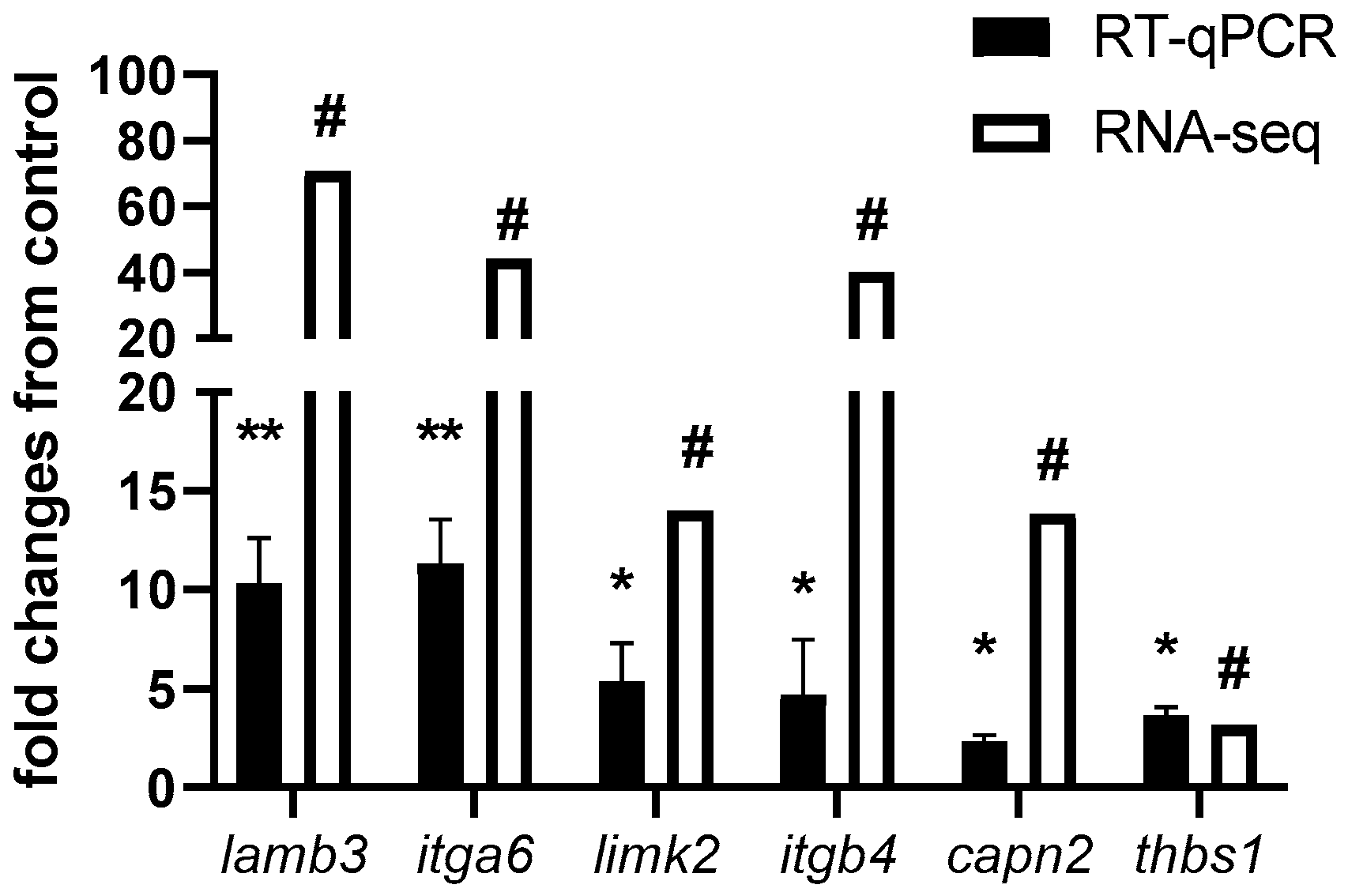
2.3. Correlation and Validation between DNA Methylation and Gene Expression
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Protocol Experiment
4.3. RNA Extraction, Library Construction, Sequencing and RNA-Seq Analysis
4.4. Real-Time qPCR Validation
4.5. DNA Extraction, Library Construction, Ssequencing, and WGBS Analysis
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Flores-Kossack, C.; Montero, R.; Köllner, B.; Maisey, K. Chilean Aquaculture and the New Challenges: Pathogens, Immune Response, Vaccination and Fish Diversification. Fish Shellfish Immunol. 2020, 98, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhou, T.; Gao, D. Genetic and Epigenetic Regulation of Growth, Reproduction, Disease Resistance and Stress Responses in Aquaculture. Front. Genet. 2022, 13, 994471. [Google Scholar] [CrossRef] [PubMed]
- Ellis, T.; Yildiz, H.Y.; López-Olmeda, J.; Spedicato, M.T.; Tort, L.; Øverli, Ø.; Martins, C.I.M. Cortisol and Finfish Welfare. Fish Physiol. Biochem. 2012, 38, 163–188. [Google Scholar] [CrossRef]
- Aluru, N.; Vijayan, M.M. Stress Transcriptomics in Fish: A Role for Genomic Cortisol Signaling. Gen. Comp. Endocrinol. 2009, 164, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Lemos, L.; Angarica, L.; Hauser-Davis, R.; Quinete, N. Cortisol as a Stress Indicator in Fish: Sampling Methods, Analytical Techniques, and Organic Pollutant Exposure Assessments. Int. J. Environ. Res. Public Health 2023, 20, 6237. [Google Scholar] [CrossRef] [PubMed]
- Balasch, J.C.; Tort, L. Netting the Stress Responses in Fish. Front. Endocrinol. 2019, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Molina, A.; Dettleff, P.; Valenzuela-Muñoz, V.; Gallardo-Escarate, C.; Valdés, J.A. High-Temperature Stress Induces Autophagy in Rainbow Trout Skeletal Muscle. Fishes 2023, 8, 303. [Google Scholar] [CrossRef]
- Aedo, J.E.; Maldonado, J.; Aballai, V.; Estrada, J.M.; Bastias-Molina, M.; Meneses, C.; Gallardo-Escarate, C.; Silva, H.; Molina, A.; Valdés, J.A. mRNA-Seq Reveals Skeletal Muscle Atrophy in Response to Handling Stress in a Marine Teleost, the Red Cusk-Eel (Genypterus chilensis). BMC Genom. 2015, 16, 1024. [Google Scholar] [CrossRef]
- Valenzuela, C.A.; Zuloaga, R.; Mercado, L.; Einarsdottir, I.E.; Björnsson, B.T.; Valdés, J.A.; Molina, A. Chronic Stress Inhibits Growth and Induces Proteolytic Mechanisms through Two Different Nonoverlapping Pathways in the Skeletal Muscle of a Teleost Fish. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2018, 314, R102–R113. [Google Scholar] [CrossRef]
- De Boeck, G.; Alsop, D.; Wood, C. Cortisol Effects on Aerobic and Anaerobic Metabolism, Nitrogen Excretion, and Whole-Body Composition in Juvenile Rainbow Trout. Physiol. Biochem. Zool. 2001, 74, 858–868. [Google Scholar] [CrossRef]
- Milligan, C.L. A Regulatory Role for Cortisol in Muscle Glycogen Metabolism in Rainbow Trout Oncorhynchus mykiss Walbaum. J. Exp. Biol. 2003, 206, 3167–3173. [Google Scholar] [CrossRef] [PubMed]
- Frolow, J.; Milligan, C.L. Hormonal Regulation of Glycogen Metabolism in White Muscle Slices from Rainbow Trout (Oncorhynchus mykiss Walbaum). Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2004, 287, R1344–R1353. [Google Scholar] [CrossRef] [PubMed]
- Bernier, N.J.; Bedard, N.; Peter, R.E. Effects of Cortisol on Food Intake, Growth, and Forebrain Neuropeptide Y and Corticotropin-Releasing Factor Gene Expression in Goldfish. Gen. Comp. Endocrinol. 2004, 135, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Sadoul, B.; Vijayan, M.M. Stress and Growth. In Fish Physiology; Elsevier: Amsterdam, The Netherlands, 2016; Volume 35, pp. 167–205. ISBN 978-0-12-802728-8. [Google Scholar]
- Glass, D.J. Signaling Pathways Perturbing Muscle Mass. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela, C.A.; Ponce, C.; Zuloaga, R.; González, P.; Avendaño-Herrera, R.; Valdés, J.A.; Molina, A. Effects of Crowding on the Three Main Proteolytic Mechanisms of Skeletal Muscle in Rainbow Trout (Oncorhynchus mykiss). BMC Vet. Res. 2020, 16, 294. [Google Scholar] [CrossRef] [PubMed]
- Aravena-Canales, D.; Aedo, J.E.; Molina, A.; Valdés, J.A. Regulation of the Early Expression of MAFbx/Atrogin-1 and MuRF1 through Membrane-Initiated Cortisol Action in the Skeletal Muscle of Rainbow Trout. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2021, 253, 110565. [Google Scholar] [CrossRef] [PubMed]
- Faught, E.; Vijayan, M.M. Mechanisms of Cortisol Action in Fish Hepatocytes. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2016, 199, 136–145. [Google Scholar] [CrossRef] [PubMed]
- Das, C.; Thraya, M.; Vijayan, M.M. Nongenomic Cortisol Signaling in Fish. Gen. Comp. Endocrinol. 2018, 265, 121–127. [Google Scholar] [CrossRef]
- Guinand, B.; Samaras, A. Epigenetics of Stress in Farmed Fish: An Appraisal. In Epigenetics in Aquaculture; Piferrer, F., Wang, H., Eds.; Wiley: Hoboken, NJ, USA, 2023; pp. 263–285. ISBN 978-1-119-82191-5. [Google Scholar]
- Metzger, D.C.H.; Schulte, P.M. Epigenomics in Marine Fishes. Mar. Genom. 2016, 30, 43–54. [Google Scholar] [CrossRef]
- Moghadam, H.K.; Johnsen, H.; Robinson, N.; Andersen, Ø.H.; Jørgensen, E.; Johnsen, H.K.; Bæhr, V.J.; Tveiten, H. Impacts of Early Life Stress on the Methylome and Transcriptome of Atlantic Salmon. Sci. Rep. 2017, 7, 5023. [Google Scholar] [CrossRef]
- Uren Webster, T.M.; Rodriguez-Barreto, D.; Martin, S.A.M.; Van Oosterhout, C.; OrozcoterWengel, P.; Cable, J.; Hamilton, A.; Garcia De Leaniz, C.; Consuegra, S. Contrasting Effects of Acute and Chronic Stress on the Transcriptome, Epigenome, and Immune Response of Atlantic Salmon. Epigenetics 2018, 13, 1191–1207. [Google Scholar] [CrossRef] [PubMed]
- López-Patiño, M.A.; Hernández-Pérez, J.; Gesto, M.; Librán-Pérez, M.; Míguez, J.M.; Soengas, J.L. Short-Term Time Course of Liver Metabolic Response to Acute Handling Stress in Rainbow Trout, Oncorhynchus mykiss. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2014, 168, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.-T.; Lee, T.-H. Rapid Response of Osmotic Stress Transcription Factor 1 (OSTF1) Expression to Salinity Challenge in Gills of Marine Euryhaline Milkfish (Chanos chanos). PLoS ONE 2022, 17, e0271029. [Google Scholar] [CrossRef] [PubMed]
- Aedo, J.; Aravena-Canales, D.; Ruiz-Jarabo, I.; Oyarzún, R.; Molina, A.; Martínez-Rodríguez, G.; Valdés, J.A.; Mancera, J.M. Differential Metabolic and Transcriptional Responses of Gilthead Seabream (Sparus aurata) Administered with Cortisol or Cortisol-BSA. Animals 2021, 11, 3310. [Google Scholar] [CrossRef]
- Mommsen, T.P.; Vijayan, M.M.; Moon, T.W. Cortisol in teleosts: Dynamics, mechanisms of action, and metabolic regulation. Rev. Fish Biol. Fish. 1999, 9, 211–268. [Google Scholar] [CrossRef]
- Aedo, J.E.; Zuloaga, R.; Bastías-Molina, M.; Meneses, C.; Boltaña, S.; Molina, A.; Valdés, J.A. Early Transcriptomic Responses Associated with the Membrane-Initiated Action of Cortisol in the Skeletal Muscle of Rainbow Trout (Oncorhynchus mykiss). Physiol. Genom. 2019, 51, 596–606. [Google Scholar] [CrossRef]
- Aedo, J.E.; Zuloaga, R.; Aravena-Canales, D.; Molina, A.; Valdés, J.A. Role of Glucocorticoid and Mineralocorticoid Receptors in Rainbow Trout (Oncorhynchus mykiss) Skeletal Muscle: A Transcriptomic Perspective of Cortisol Action. Front. Physiol. 2023, 13, 1048008. [Google Scholar] [CrossRef]
- Aedo, J.E.; Fuentes-Valenzuela, M.; Molina, A.; Valdés, J.A. Quantitative Proteomics Analysis of Membrane Glucocorticoid Receptor Activation in Rainbow Trout Skeletal Muscle. Comp. Biochem. Physiol. Part D Genom. Proteom. 2019, 32, 100627. [Google Scholar] [CrossRef]
- Espinoza, M.B.; Aedo, J.E.; Zuloaga, R.; Valenzuela, C.; Molina, A.; Valdés, J.A. Cortisol Induces Reactive Oxygen Species Through a Membrane Glucocorticoid Receptor in Rainbow Trout Myotubes. J. Cell. Biochem. 2017, 118, 718–725. [Google Scholar] [CrossRef]
- Aedo, J.E.; Zuloaga, R.; Boltaña, S.; Molina, A.; Valdés, J.A. Membrane-Initiated Cortisol Action Modulates Early Pyruvate Dehydrogenase Kinase 2 (Pdk2) Expression in Fish Skeletal Muscle. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2019, 233, 24–29. [Google Scholar] [CrossRef]
- Blondeau-Bidet, E.; Banousse, G.; L’Honoré, T.; Farcy, E.; Cosseau, C.; Lorin-Nebel, C. The Role of Salinity on Genome-wide DNA Methylation Dynamics in European Sea Bass Gills. Mol. Ecol. 2023, 32, 5089–5109. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, M.; Zhou, T.; Li, Q.; Lin, Z. Transcriptome and Methylome Dynamics in the Gills of Large Yellow Croaker (Larimichthys crocea) during Low-Salinity Adaption. Front. Mar. Sci. 2023, 10, 1082655. [Google Scholar] [CrossRef]
- Lin, Y.L.; Zhu, Z.X.; Ai, C.H.; Xiong, Y.Y.; De Liu, T.; Lin, H.R.; Xia, J.H. Transcriptome and DNA Methylation Responses in the Liver of Yellowfin Seabream Under Starvation Stress. Mar. Biotechnol. 2023, 25, 150–160. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Wang, Y.; Zhang, B.; Zeng, J.; Wang, Q.; Wang, C.; Peng, S. Whole-Genome Methylation Sequencing of Large Yellow Croaker (Larimichthys crocea) Liver Under Hypoxia and Acidification Stress. Mar. Biotechnol. 2023, 25, 567–579. [Google Scholar] [CrossRef] [PubMed]
- Robinson, N.A.; Johnsen, H.; Moghadam, H.; Andersen, Ø.; Tveiten, H. Early Developmental Stress Affects Subsequent Gene Expression Response to an Acute Stress in Atlantic Salmon: An Approach for Creating Robust Fish for Aquaculture? G3 Genes Genomes Genet. 2019, 9, 1597–1611. [Google Scholar] [CrossRef] [PubMed]
- Valenzuela-Muñoz, V.; Wanamaker, S.; Núñez-Acuña, G.; Roberts, S.; Garcia, A.; Valdés, J.A.; Valenzuela-Miranda, D.; Gallardo-Escárate, C. Environmental Influence on the Atlantic Salmon Transcriptome and Methylome during Sea Lice Infestations. Fish Shellfish Immunol. 2024, 151, 109692. [Google Scholar] [CrossRef]
- Paes, T.; Feelders, R.A.; Hofland, L.J. Epigenetic Mechanisms Modulated by Glucocorticoids with a Focus on Cushing Syndrome. J. Clin. Endocrinol. Metab. 2024, 109, e1424–e1433. [Google Scholar] [CrossRef]
- Liu, W.; Mohan, S.P.; Nagaraj, N.R.; Sundar Jaganathan, S.; Wen, Y.; Ramasubramanyan, S.; Irudayaraj, J. Epigenetic Alterations Associated with Dexamethasone Sodium Phosphate through DNMT and TET in RPE Cells. Mol. Vis. 2021, 27, 643–655. [Google Scholar]
- Zhao, J.; Ma, X.; Ma, J.; Sun, L.; Lu, B.; Wang, Y.; Xing, G.; Wang, Y.; Dong, B.; Xu, L.; et al. TET3 Mediates Alterations in the Epigenetic Marker 5hmC and Akt Pathway in Steroid-Associated Osteonecrosis. J. Bone Miner. Res. 2017, 32, 319–332. [Google Scholar] [CrossRef]
- Yang, X.; Ewald, E.R.; Huo, Y.; Tamashiro, K.L.; Salvatori, R.; Sawa, A.; Wand, G.S.; Lee, R.S. Glucocorticoid-Induced Loss of DNA Methylation in Non-Neuronal Cells and Potential Involvement of DNMT1 in Epigenetic Regulation of Fkbp5. Biochem. Biophys. Res. Commun. 2012, 420, 570–575. [Google Scholar] [CrossRef]
- Chen, C.S.; Alonso, J.L.; Ostuni, E.; Whitesides, G.M.; Ingber, D.E. Cell Shape Provides Global Control of Focal Adhesion Assembly. Biochem. Biophys. Res. Commun. 2003, 307, 355–361. [Google Scholar] [CrossRef]
- Boncompagni, S.; Arthurton, L.; Akujuru, E.; Pearson, T.; Steverding, D.; Protasi, F.; Mutungi, G. Membrane Glucocorticoid Receptors Are Localised in the Extracellular Matrix and Signal through the MAPK Pathway in Mammalian Skeletal Muscle Fibres. J. Physiol. 2015, 593, 2679–2692. [Google Scholar] [CrossRef]
- Wang, J.; Koganti, P.P.; Yao, J.; Wei, S.; Cleveland, B. Comprehensive Analysis of lncRNAs and mRNAs in Skeletal Muscle of Rainbow Trout (Oncorhynchus mykiss) Exposed to Estradiol. Sci. Rep. 2017, 7, 11780. [Google Scholar] [CrossRef]
- Leo, J.C.L.; Guo, C.; Woon, C.T.; Aw, S.E.; Lin, V.C.L. Glucocorticoid and Mineralocorticoid Cross-Talk with Progesterone Receptor to Induce Focal Adhesion and Growth Inhibition in Breast Cancer Cells. Endocrinology 2004, 145, 1314–1321. [Google Scholar] [CrossRef] [PubMed]
- Lowin, T.; Straub, R.H.; Neumann, E.; Bosserhoff, A.; Vogel, C.; Moissl, C.; Anders, S.; Müller-Ladner, U.; Schedel, J. Glucocorticoids Increase A5 Integrin Expression and Adhesion of Synovial Fibroblasts but Inhibit ERK Signaling, Migration, and Cartilage Invasion. Arthritis Rheum. 2009, 60, 3623–3632. [Google Scholar] [CrossRef] [PubMed]
- Thorsteinsdóttir, S.; Deries, M.; Cachaço, A.S.; Bajanca, F. The Extracellular Matrix Dimension of Skeletal Muscle Development. Dev. Biol. 2011, 354, 191–207. [Google Scholar] [CrossRef] [PubMed]
- Rallière, C.; Branthonne, A.; Rescan, P. Formation of Intramuscular Connective Tissue Network in Fish: First Insight from the Rainbow Trout (Oncorhynchus mykiss). J. Fish Biol. 2018, 93, 1171–1177. [Google Scholar] [CrossRef]
- Zhao, N.; Wang, X.; Wang, T.; Xu, X.; Liu, Q.; Li, J. Genome-Wide Identification of Laminin Family Related to Follicular Pseudoplacenta Development in Black Rockfish (Sebastes schlegelii). Int. J. Mol. Sci. 2022, 23, 10523. [Google Scholar] [CrossRef]
- Al-Obaide, M.; Ishmakej, A.; Brown, C.; Mazzella, M.; Agosta, P.; Perez-Cruet, M.; Chaudhry, G.R. The Potential Role of Integrin Alpha 6 in Human Mesenchymal Stem Cells. Front. Genet. 2022, 13, 968228. [Google Scholar] [CrossRef]
- Wilschut, K.J.; Van Tol, H.T.A.; Arkesteijn, G.J.A.; Haagsman, H.P.; Roelen, B.A.J. Alpha 6 Integrin Is Important for Myogenic Stem Cell Differentiation. Stem Cell Res. 2011, 7, 112–123. [Google Scholar] [CrossRef]
- Liu, H.; Hu, X.; Lian, Z.; Luo, Z.; Lv, A.; Tan, J. Focal Adhesion Signaling Pathway Involved in Skin Immune Response of Tongue Sole Cynoglossus semilaevis to Vibrio vulnificus Infection. Fish Shellfish Immunol. 2023, 135, 108651. [Google Scholar] [CrossRef]
- He, Y.; Zhang, L.; He, Y.; Yu, H.; Li, S.; Li, Q. Involvement of LIMK2 in Actin Cytoskeleton Remodeling during the Definitive Endoderm Differentiation. Vitr. Cell. Dev. Biol. Anim. 2021, 57, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Ott, E.B.; Te Velthuis, A.J.W.; Bagowski, C.P. Comparative Analysis of Splice Form-Specific Expression of LIM Kinases during Zebrafish Development. Gene Expr. Patterns 2007, 7, 620–629. [Google Scholar] [CrossRef] [PubMed]
- Baudry, M.; Bi, X. Calpain-1 and Calpain-2: The Yin and Yang of Synaptic Plasticity and Neurodegeneration. Trends Neurosci. 2016, 39, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Vélez, E.J.; Azizi, S.; Verheyden, D.; Salmerón, C.; Lutfi, E.; Sánchez-Moya, A.; Navarro, I.; Gutiérrez, J.; Capilla, E. Proteolytic Systems’ Expression during Myogenesis and Transcriptional Regulation by Amino Acids in Gilthead Sea Bream Cultured Muscle Cells. PLoS ONE 2017, 12, e0187339. [Google Scholar] [CrossRef] [PubMed]
- Atanasova, V.S.; Russell, R.J.; Webster, T.G.; Cao, Q.; Agarwal, P.; Lim, Y.Z.; Krishnan, S.; Fuentes, I.; Guttmann-Gruber, C.; McGrath, J.A.; et al. Thrombospondin-1 Is a Major Activator of TGF-β Signaling in Recessive Dystrophic Epidermolysis Bullosa Fibroblasts. J. Investig. Dermatol. 2019, 139, 1497–1505.e5. [Google Scholar] [CrossRef]
- Ali, A.; Al-Tobasei, R.; Lourenco, D.; Leeds, T.; Kenney, B.; Salem, M. Genome-Wide Identification of Loci Associated with Growth in Rainbow Trout. BMC Genom. 2020, 21, 209. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE Guidelines: Minimum Information for Publication of Quantitative Real-Time PCR Experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Zuloaga, R.; Ahumada-Langer, L.; Aedo, J.E.; Molina, A.; Valdés, J.A. 11-Deoxycorticosterone (DOC)’s Action on the Gill Osmoregulation of Juvenile Rainbow Trout (Oncorhynchus mykiss). Biology 2024, 13, 107. [Google Scholar] [CrossRef]
- Simon, P. Q-Gene: Processing Quantitative Real-Time RT–PCR Data. Bioinformatics 2003, 19, 1439–1440. [Google Scholar] [CrossRef]
| Name | Number of Reads | Avg. Length | Number of Reads after Trimming | Avg. Length after Trimming | GC Content % | Mapping Rate |
|---|---|---|---|---|---|---|
| Control 1 | 57,044,114 | 101 | 57,027,988 | 95.98 | 48.59 | 82.35 |
| Control 2 | 50,668,370 | 101 | 50,653,122 | 95.42 | 47.92 | 86.46 |
| Control 3 | 54,686,284 | 101 | 54,664,282 | 94.58 | 48.15 | 80.34 |
| Cortisol 1 | 54,262,334 | 101 | 54,249,443 | 96.33 | 48.76 | 82.26 |
| Cortisol 2 | 56,252,594 | 101 | 56,234,957 | 95.63 | 47.96 | 80.49 |
| Cortisol 3 | 56,918,044 | 101 | 56,898,861 | 95.21 | 48.02 | 80.25 |
| Total | 329,831,740 | 101 | 329,728,653 | 95.53 | 48.23 | 82.03 |
| Category | Go Term | Gene Number | p-Value |
|---|---|---|---|
| Biological Process | Wound-healing | 8 | 1.95 × 10−9 |
| Intermediate filament cytoskeleton organization | 10 | 1.12 × 10−8 | |
| Keratinization | 6 | 3.28 × 10−8 | |
| Intermediate filament organization | 7 | 4.56 × 10−8 | |
| Cell–cell junction assembly | 10 | 3.21 × 10−7 | |
| Cellular Component | Intermediate filament | 22 | 6.42 × 10−17 |
| Extracellular space | 46 | 1.08 × 10−10 | |
| Keratin filament | 8 | 5.57 × 10−8 | |
| Actin cytoskeleton | 14 | 1.03 × 10−7 | |
| Cytoplasm | 101 | 1.04 × 10−6 | |
| Molecular Function | Structural molecule activity | 21 | 7.27 × 10−12 |
| Cadherin-binding | 15 | 9.10 × 10−11 | |
| Structural constituent of epidermis | 6 | 2.30 × 10−8 | |
| Calcium-dependent cysteine-type endopeptidase activity | 8 | 3.23 × 10−6 | |
| Calcium ion binding | 40 | 4.77 × 10−6 |
| Name | Number of Reads | Avg. Length | Number of Reads after Trimming | Avg. Length after Trimming | Mapping Rate | BS Conversion Rate (%) |
|---|---|---|---|---|---|---|
| Control 1 | 76,907,896 | 151 | 76,445,383 | 114.73 | 76.17 | 99.91 |
| Control 2 | 76,721,140 | 151 | 76,304,825 | 114.83 | 75.24 | 99.92 |
| Control 3 | 77,201,332 | 151 | 76,843,936 | 115.51 | 74.31 | 99.91 |
| Cortisol 1 | 77,549,376 | 151 | 77,172,273 | 119.23 | 77.16 | 99.93 |
| Cortisol 2 | 77,471,992 | 151 | 77,090,668 | 118.88 | 77.89 | 99.89 |
| Cortisol 3 | 77,408,864 | 151 | 76,980,571 | 114.97 | 73.88 | 99.95 |
| Total | 463,260,600 | 151 | 460,837,656 | 116.36 | 75.78 | 99.92 |
| Category | Go Term | Gene Number | p-Value |
|---|---|---|---|
| Biological Process | Protein phosphorylation | 464 | 5.07 × 10−41 |
| Axon guidance | 164 | 8.38 × 10−19 | |
| Transmembrane transport | 247 | 2.33 × 10−12 | |
| Multicellular organism development | 157 | 3.24 × 10−12 | |
| Cell migration | 100 | 7.74 × 10−12 | |
| Cellular Component | Plasma membrane | 1748 | 9.82 × 10−56 |
| Cytoplasm | 2167 | 1.11 × 10−44 | |
| Neuron projection | 209 | 1.01 × 10−22 | |
| Cytosol | 600 | 1.82 × 10−22 | |
| Adherens junction | 123 | 2.04 × 10−14 | |
| Molecular Function | Protein binding | 1991 | 2.26 × 10−13 |
| RNA polymerase II transcription factor activity, DNA binding | 732 | 8.20 × 10−13 | |
| Transmitter-gated ion channel activity involved in regulation of P.M.P | 51 | 1.95 × 10−11 | |
| mRNA binding | 159 | 3.37 × 10−11 | |
| RNA polymerase II core promoter proximal region, DNA binding | 738 | 1.10 × 10−10 |
| KEGG Pathway | Number | p-Value | Genes |
|---|---|---|---|
| ECM-receptor interaction | 5 | 2.57 × 10−3 | lamb3, itga6, thbs1, itgb4, itgb3 |
| Arachidonic acid metabolism | 4 | 3.02 × 10−3 | pla2g4f, ptgs2, cyp2j2, ggt5 |
| Regulation of actin cytoskeleton | 7 | 5.85 × 10−3 | limk2, gsn, itga6, arpc1b, itgb4, c7, itgb3 |
| Focal adhesion | 6 | 1.63 × 10−2 | lamb3, itga2, capn2, thbs1, itgb4, itgb3 |
| Efferocytosis | 5 | 2.60 × 10−2 | mapk12, ptgs2, thbs, itgb3, abca1 |
| GnRH signaling pathway | 4 | 2.99 × 10−2 | mapk12, ptk2b, pla2g4e, mmp14 |
| Ether lipid metabolism | 3 | 3.40 × 10−2 | pla2g4e, plpp2, gdpd3 |
| VEGF signaling pathway | 3 | 7.61 × 10−2 | mapk12, pla2g4e, ptgs2 |
| Nucleotide metabolism | 3 | 8.51 × 10−2 | gda, nt5c1a, nt5c2 |
| Category | Go Term | Gene Number | p-Value |
|---|---|---|---|
| Biological Process | Keratinization | 6 | 3.05 × 10−11 |
| Intermediate filament organization | 6 | 1.70 × 10−9 | |
| IRE1-mediated unfolded protein response | 3 | 5.71 × 10−4 | |
| Regulation of RNA metabolic process | 3 | 9.25 × 10−4 | |
| Intrinsic apoptotic signaling pathway in response to ERS | 3 | 9.25 × 10−4 | |
| Cellular Component | Keratin filament | 6 | 2.44 × 10−8 |
| Extracellular space | 13 | 3.03 × 10−4 | |
| IRE1-TRAF2-ASK1 complex | 3 | 5.15 × 10−4 | |
| Plasma membrane | 24 | 7.65 × 10−4 | |
| Bicellular tight junction | 5 | 9.33 × 10−4 | |
| Molecular Function | Structural constituent of epidermis | 6 | 1.90 × 10−11 |
| Calcium ion binding | 15 | 1.07 × 10−4 | |
| Cadherin binding | 4 | 4.31 × 10−3 | |
| Endoribonuclease activity | 3 | 6.31 × 10−3 | |
| Calcium-dependent cysteine-type endopeptidase activity | 3 | 1.03 × 10−2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aravena-Canales, D.; Valenzuela-Muñoz, V.; Gallardo-Escarate, C.; Molina, A.; Valdés, J.A. Transcriptomic and Epigenomic Responses to Cortisol-Mediated Stress in Rainbow Trout (Oncorhynchus mykiss) Skeletal Muscle. Int. J. Mol. Sci. 2024, 25, 7586. https://doi.org/10.3390/ijms25147586
Aravena-Canales D, Valenzuela-Muñoz V, Gallardo-Escarate C, Molina A, Valdés JA. Transcriptomic and Epigenomic Responses to Cortisol-Mediated Stress in Rainbow Trout (Oncorhynchus mykiss) Skeletal Muscle. International Journal of Molecular Sciences. 2024; 25(14):7586. https://doi.org/10.3390/ijms25147586
Chicago/Turabian StyleAravena-Canales, Daniela, Valentina Valenzuela-Muñoz, Cristian Gallardo-Escarate, Alfredo Molina, and Juan Antonio Valdés. 2024. "Transcriptomic and Epigenomic Responses to Cortisol-Mediated Stress in Rainbow Trout (Oncorhynchus mykiss) Skeletal Muscle" International Journal of Molecular Sciences 25, no. 14: 7586. https://doi.org/10.3390/ijms25147586
APA StyleAravena-Canales, D., Valenzuela-Muñoz, V., Gallardo-Escarate, C., Molina, A., & Valdés, J. A. (2024). Transcriptomic and Epigenomic Responses to Cortisol-Mediated Stress in Rainbow Trout (Oncorhynchus mykiss) Skeletal Muscle. International Journal of Molecular Sciences, 25(14), 7586. https://doi.org/10.3390/ijms25147586








