Loncastuximab Tesirine in the Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma
Abstract
1. Background
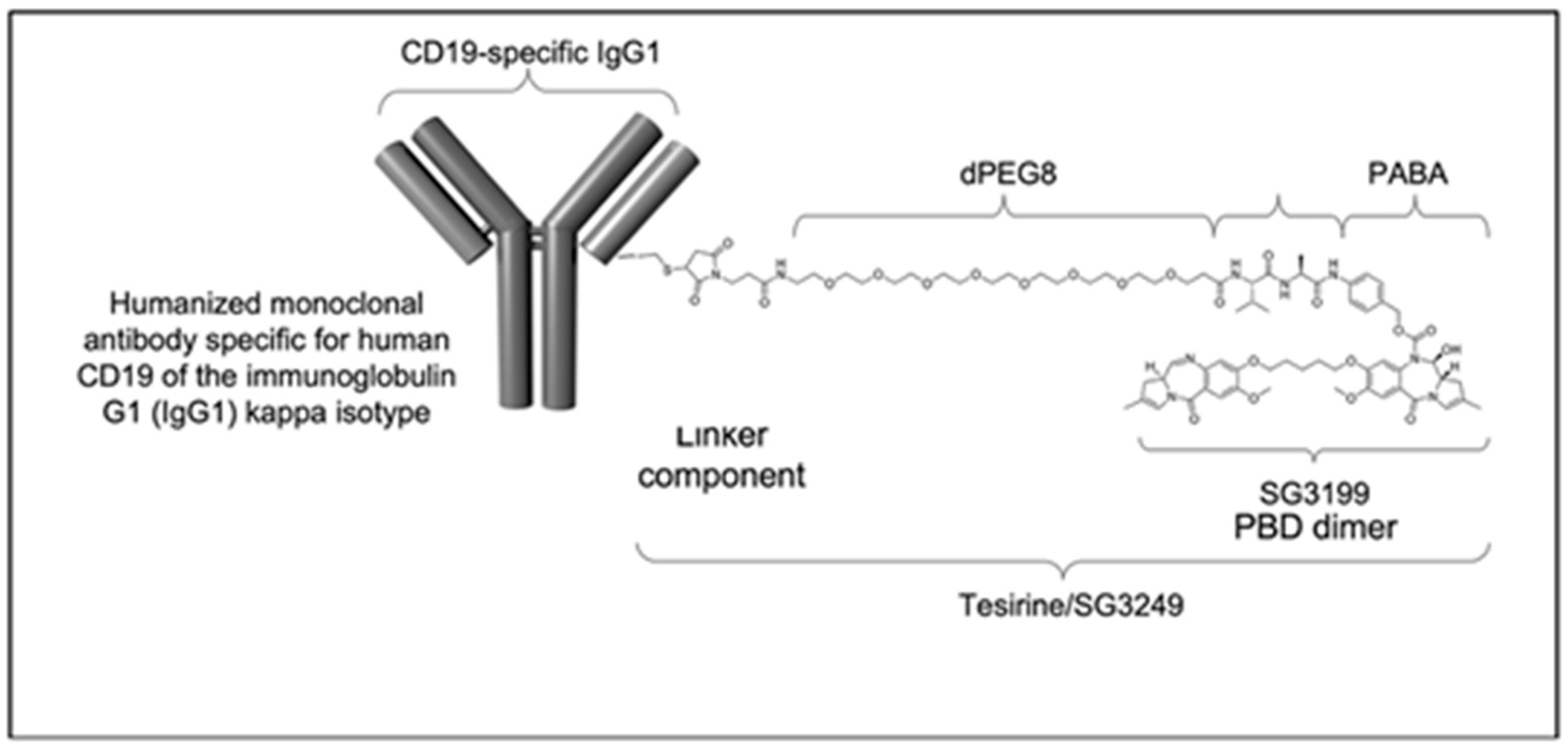
2. Mechanism of Action
3. Clinical Trial Data
4. Toxicity Profile
5. Future Clinical Trials
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sehn, L.H.; Salles, G. Diffuse Large B-Cell Lymphoma. N. Engl. J. Med. 2021, 384, 842–858. [Google Scholar] [CrossRef] [PubMed]
- Coiffier, B.; Lepage, E.; Brière, J.; Herbrecht, R.; Tilly, H.; Bouabdallah, R.; Morel, P.; Gisselbrecht, C.; Van Den Neste, E.; Salles, G.; et al. CHOP Chemotherapy plus Rituximab Compared with CHOP Alone in Elderly Patients with Diffuse Large-B-Cell Lymphoma. N. Engl. J. Med. 2002, 346, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M.J.; Ghesquières, H.; Jais, J.-P.; Witzig, T.E.; Haioun, C.; Thompson, C.A.; Delarue, R.; Micallef, I.N.; Peyrade, F.; Macon, W.R.; et al. Event-free survival at 24 months is a robust end point for disease-related outcome in diffuse large B-cell lymphoma treated with immunochemotherapy. J. Clin. Oncol. 2014, 32, 1066–1073. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Horning, S.J.; Coiffier, B.; Shipp, M.A.; Fisher, R.I.; Connors, J.M.; Lister, T.A.; Vose, J.; Grillo-López, A.; Hagenbeek, A.; et al. Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphomas. NCI Sponsored International Working Group. J. Clin. Oncol. 1999, 17, 1244. [Google Scholar] [CrossRef] [PubMed]
- Crump, M.; Neelapu, S.S.; Farooq, U.; Van Den Neste, E.; Kuruvilla, J.; Westin, J.; Link, B.K.; Hay, A.; Cerhan, J.R.; Zhu, L.; et al. Outcomes in refractory diffuse large B-cell lymphoma: Results from the international SCHOLAR-1 study. Blood 2017, 130, 1800–1808. [Google Scholar] [CrossRef] [PubMed]
- Crump, M.; Kuruvilla, J.; Couban, S.; MacDonald, D.A.; Kukreti, V.; Kouroukis, C.T.; Rubinger, M.; Buckstein, R.; Imrie, K.R.; Federico, M.; et al. Randomized comparison of gemcitabine, dexamethasone, and cisplatin versus dexamethasone, cytarabine, and cisplatin chemotherapy before autologous stem-cell transplantation for relapsed and refractory aggressive lymphomas: NCIC-CTG LY.12. J. Clin. Oncol. 2014, 32, 3490–3496. [Google Scholar] [CrossRef] [PubMed]
- Gisselbrecht, C.; Glass, B.; Mounier, N.; Gill, D.S.; Linch, D.C.; Trneny, M.; Bosly, A.; Ketterer, N.; Shpilberg, O.; Hagberg, H.; et al. Salvage regimens with autologous transplantation for relapsed large B-cell lymphoma in the rituximab era. J. Clin. Oncol. 2010, 28, 4184–4190. [Google Scholar] [CrossRef] [PubMed]
- Chapuy, B.; Stewart, C.; Dunford, A.J.; Kim, J.; Kamburov, A.; Redd, R.A.; Lawrence, M.S.; Roemer, M.G.M.; Li, A.J.; Ziepert, M.; et al. Molecular subtypes of diffuse large B cell lymphoma are associated with distinct pathogenic mechanisms and outcomes. Nat. Med. 2018, 24, 679–690. [Google Scholar] [CrossRef] [PubMed]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N. Engl. J. Med. 2017, 377, 2531–2544. [Google Scholar] [CrossRef] [PubMed]
- Schuster, S.J.; Bishop, M.R.; Tam, C.S.; Waller, E.K.; Borchmann, P.; McGuirk, J.P.; Jäger, U.; Jaglowski, S.; Andreadis, C.; Westin, J.R.; et al. Tisagenlecleucel in Adult Relapsed or Refractory Diffuse Large B-Cell Lymphoma. N. Engl. J. Med. 2019, 380, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Chau, C.H.; Steeg, P.S.; Figg, W.D. Antibody-drug conjugates for cancer. Lancet 2019, 394, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Sehn, L.H.; Herrera, A.F.; Flowers, C.R.; Kamdar, M.K.; McMillan, A.; Hertzberg, M.; Assouline, S.; Kim, T.M.; Kim, W.S.; Ozcan, M.; et al. Polatuzumab Vedotin in Relapsed or Refractory Diffuse Large B-Cell Lymphoma. J. Clin. Oncol. 2020, 38, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Kalakonda, N.; Maerevoet, M.; Cavallo, F.; Follows, G.; Goy, A.; Vermaat, J.S.P.; Casasnovas, O.; Hamad, N.; Zijlstra, J.M.; Bakhshi, S.; et al. Selinexor in patients with relapsed or refractory diffuse large B-cell lymphoma (SADAL): A single-arm, multinational, multicentre, open-label, phase 2 trial. Lancet Haematol. 2020, 7, e511–e522. [Google Scholar] [CrossRef] [PubMed]
- Salles, G.; Duell, J.; Barca, E.G.; Tournilhac, O.; Jurczak, W.; Liberati, A.M.; Nagy, Z.; Obr, A.; Gaidano, G.; André, M.; et al. Tafasitamab plus lenalidomide in relapsed or refractory diffuse large B-cell lymphoma (L-MIND): A multicentre, prospective, single-arm, phase 2 study. Lancet Oncol. 2020, 21, 978–988. [Google Scholar] [CrossRef]
- Caimi, P.F.; Ai, W.; Alderuccio, J.P.; Ardeshna, K.M.; Hamadani, M.; Hess, B.; Kahl, B.S.; Radford, J.; Solh, M.; Stathis, A.; et al. Loncastuximab tesirine in relapsed or refractory diffuse large B-cell lymphoma (LOTIS-2): A multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol. 2021, 22, 790–800. [Google Scholar] [CrossRef] [PubMed]
- McMillan, A. A new warhead in lymphoma therapy? Blood 2021, 137, 2568–2570. [Google Scholar] [CrossRef] [PubMed]
- Wolska-Washer, A.; Robak, P.; Smolewski, P.; Robak, T. Emerging antibody-drug conjugates for treating lymphoid malignancies. Expert Opin. Emerg. Drugs 2017, 22, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Calabretta, E.; Hamadani, M.; Zinzani, P.L.; Caimi, P.; Carlo-Stella, C. The antibody-drug conjugate loncastuximab tesirine for the treatment of diffuse large B-cell lymphoma. Blood 2022, 140, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Liu, D. Cancer biomarkers for targeted therapy. Biomark. Res. 2019, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Hartley, J.A. The development of pyrrolobenzodiazepines as antitumour agents. Expert Opin. Investig. Drugs 2011, 20, 733–744. [Google Scholar] [CrossRef] [PubMed]
- Zammarchi, F.; Corbett, S.; Adams, L.; Tyrer, P.C.; Kiakos, K.; Janghra, N.; Marafioti, T.; Britten, C.E.; Havenith, C.E.G.; Chivers, S.; et al. ADCT-402, a PBD dimer-containing antibody drug conjugate targeting CD19-expressing malignancies. Blood 2018, 131, 1094–1105. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Stock, W.; Zeidan, A.; Atallah, E.; McCloskey, J.; Heffner, L.; Tomlinson, B.; Bhatnagar, B.; Feingold, J.; Ungar, D.; et al. Loncastuximab tesirine, an anti-CD19 antibody-drug conjugate, in relapsed/refractory B-cell acute lymphoblastic leukemia. Blood Adv. 2020, 4, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Hamadani, M.; Radford, J.; Carlo-Stella, C.; Caimi, P.F.; Reid, E.; O’connor, O.A.; Feingold, J.M.; Ardeshna, K.M.; Townsend, W.; Solh, M.; et al. Final results of a phase 1 study of loncastuximab tesirine in relapsed/refractory B-cell non-Hodgkin lymphoma. Blood 2021, 137, 2634–2645. [Google Scholar] [CrossRef] [PubMed]
- Kahl, B.S.; Hamadani, M.; Radford, J.; Carlo-Stella, C.; Caimi, P.; Reid, E.; Feingold, J.M.; Ardeshna, K.M.; Solh, M.; Heffner, L.T.; et al. A Phase I Study of ADCT-402 (Loncastuximab Tesirine), a Novel Pyrrolobenzodiazepine-Based Antibody-Drug Conjugate, in Relapsed/Refractory B-Cell Non-Hodgkin Lymphoma. Clin. Cancer Res. 2019, 25, 6986–6994. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Hamadani, M. Evaluating efficacy and safety of loncastuximab tesirine injection for the treatment of adult patients with relapsed or refractory large B-cell lymphoma. Expert Rev. Anticancer Ther. 2021, 21, 1313–1320. [Google Scholar] [CrossRef] [PubMed]
- Furqan, F.; Hamadani, M. Loncastuximab tesirine in relapsed or refractory diffuse large B-cell lymphoma: A review of clinical data. Ther. Adv. Hematol. 2022, 13, 20406207221087511. [Google Scholar] [CrossRef] [PubMed]
- Hamadani, M.; Chen, L.; Song, Y.; Xu, M.K.; Liao, L.; Caimi, P.F.; Carlo-Stella, C. Matching-adjusted Indirect Comparison of the Efficacy of Loncastuximab Tesirine Versus Treatment in the Chemoimmunotherapy Era for Relapsed/Refractory Diffuse Large B-cell Lymphoma. Clin. Lymphoma Myeloma Leuk. 2022, 22, e738–e744. [Google Scholar] [CrossRef] [PubMed]
- Hamadani, M.; Spira, A.; Zhou, X.; Liao, L.; Chen, L.; Radford, J.; Ai, W.; Solh, M.; Ardeshna, K.M.; Hess, B.; et al. Clinical outcomes of older and younger patients treated with loncastuximab tesirine in the LOTIS-2 clinical trial. Blood Adv. 2024, 8, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Caimi, P.F.; Ai, W.Z.; Alderuccio, J.P.; Ardeshna, K.M.; Hamadani, M.; Hess, B.; Kahl, B.S.; Radford, J.; Solh, M.; Stathis, A.; et al. Loncastuximab tesirine in relapsed/refractory diffuse large B-cell lymphoma: Long-term efficacy and safety from the phase II LOTIS-2 study. Haematologica 2024, 109, 1184–1193. [Google Scholar] [CrossRef] [PubMed]
- Carlo-Stella, C.; Kwiatek, M.; Chung, M.; Móciková, H.; Bergua-Burgues, J.M.; Gao, S.; Grosicki, S.; Stathis, A.; Toupin, D.; Wang, Y.; et al. Lotis-5, an Ongoing Phase 3 Randomized Study of Loncastuximab Tesirine with Rituximab (lonca-R) Versus Immunochemotherapy in Patients with R/R Dlbcl. Hematol. Oncol. 2023, 41, 850–851. [Google Scholar] [CrossRef]
- Lin, N.; Sun, X.; Zhou, H.; Zou, L.; Zhou, K.; Liu, L.; Yang, H.; Hu, K.; Cai, Q.; Liu, Y.; et al. Loncastuximab Tesirine Demonstrated Substantial Single-Agent Efficacy and Manageable Safety Profile in Heavily Pretreated Chinese Patients with Relapsed or Refractory (R/R) Diffuse Large B-Cell Lymphoma (DLBCL). Blood 2023, 142 (Suppl. S1), 4469. [Google Scholar] [CrossRef]
- Kwiatek, M.; Grosicki, S.; Jiménez, J.L.; Mariño, S.F.P.; Snauwaert, S.; Kingsley, E.; Zacchetti, G.; Wang, Y.; Wang, L.; Depaus, J. ABCL-515 Updated Results of the Safety Run-In of the Phase 3 LOTIS-5 Trial: Novel Combination of Loncastuximab Tesirine With Rituximab (Lonca-R) Versus Immunochemotherapy in Patients With R/R DLBCL. Clin. Lymphoma Myeloma Leuk. 2023, 23, S439–S440. [Google Scholar] [CrossRef]
- Caimi, P.F.; Ardeshna, K.M.; Reid, E.; Ai, W.; Lunning, M.; Zain, J.; Solh, M.; Kahl, B.S.; Hamadani, M. The AntiCD19 Antibody Drug Immunoconjugate Loncastuximab Achieves Responses in DLBCL Relapsing after AntiCD19 CAR-T Cell Therapy. Clin. Lymphoma Myeloma Leuk. 2022, 22, e335–e339. [Google Scholar] [CrossRef] [PubMed]
- Alarcon Tomas, A.; Fein, J.A.; Fried, S.; Flynn, J.R.; Devlin, S.M.; Fingrut, W.B.; Anagnostou, T.; Alperovich, A.; Shah, N.; Fraint, E.; et al. Outcomes of first therapy after CD19-CAR-T treatment failure in large B-cell lymphoma. Leukemia 2023, 37, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Thapa, B.; Caimi, P.F.; Ardeshna, K.M.; Solh, M.; Carlo-Stella, C.; Kahl, B.S.; Hamadani, M. CD19 antibody-drug conjugate therapy in DLBCL does not preclude subsequent responses to CD19-directed CAR T-cell therapy. Blood Adv. 2020, 4, 3850–3852. [Google Scholar] [CrossRef] [PubMed]

| Trial | N= | Dosing | OR (ORR) | CR; PR CRR; PRR | PFS; OS; Median DOR (Months) |
|---|---|---|---|---|---|
| LOTIS-1 (open-label, single-arm, phase 1 study) 1 [COMPLETED] | 63 | 3 × 3 Dose-escalation design to determine adequate dose | ≤90 µg/kg: 2 (20%) 120 µg/kg: 6 (54.5%) 150 µg/kg: 9 (60.0%) 200 µg/kg: 13 (52.0%) ≥120 µg/kg: 28 (54.9%) | ≤90 µg/kg: 1; 1 120 µg/kg: 4; 2 150 µg/kg: 5; 4 200 µg/kg: 10; 3 ≥120 µg/kg: 19; 9 20 (32.8%); 10 (16.4%) | 2.9 *; 10.1 DOR: CR- not reached PR-3.1 150 µg/kg: 5.5 120 µg/kg: 4.9 200 µg/kg: 4.1 |
| LOTIS-2 (multicenter, open-label, single-arm, phase 2 study) 2 [COMPLETED] | 145 | Q3w 150 µg/kg first 2 cycles 75 µg/kg for subsequent cycles, up to 1 year | 70 (48.3%) [95% CI: 39.9% to 56.7%] | 35; 35 24.1% [95% CI: 17.5% to 31.9%] **; 24% | 4.9 [95% CI: 2.9–8.3]; 9.9 *** [95% CI: 6.7–11.5]; 10.3 **** [95% CI: 6.9-not reached] |
| LOTIS-3 x (loncastuximab + Ibrutinib: Phase 2 open-label, single-arm study) 3 [TERMINATED] | 35 | 60 µg/kg loncastuximab q3w for 2 cycles + 560mg/day Ibrutinib oral up to 1 year (CR/PR/stable disease patients receive additional loncastuximab cycle 5,6,9,10) | 20 (57.1%) [95% CI: 39.4% to 73.7%] | 12; 8 34.3% [95% CI: 19.1% to 52.2%]; 22.9% # | PFS not reported; OS not reported; 5.49 [95% CI: 6.9- not reached |
| Trial | Trial Identification Number and Trial Dates | N= | Any Available Results |
|---|---|---|---|
| LOTIS-5 (loncastuximab + R-GemOx [rituximab–gemcitabine–oxaliplatin]) 1 | NCT04384484 September 2020– 2028 | ~330 by trial end. Currently n ≤ 20 | Preliminary Data: OR(ORR): 16/20 (80%) [95% CI: 56.3% to 94.3%] CRR; PRR: 50% [95% CI: 27.2% to 72.8%]; 30% [95% CI: 11.9% to 54.3%] PFS; OS; Median DOR (months): 8.31 [95% CI: 4.53—not reached]; OS not reported; 8.02 [95% CI: 3.19—not reached] |
| LOTIS-7 (loncastuximab + various other agents including but not limited to polatuzumab vedotin, glofitamab, and mosunetuzumab for r/r DLBCL) | NCT04970901 December 2021 - May 2027 | ~n = 200 (which includes patients with r/r B-NHL, DLBLCL, HGBCL, FL, MZL) | N/A |
| Chinese Cancer Centers Trial (Replication of LOTIS-2) 2 | ChiCTR2300072058 September 2021 - Ongoing (unknown completion) | ~n = 64 (as of 11 January 2023 data cutoff) | ORR: 51.6% [95% CI: 38.7% to 64.2%] CRR: 23.4% [NO CI provided] PFS; OS; Median DOR: 4.96; 9.33; 6.37 [NO CI provided] |
| MD Anderson (loncastuximab as consolidation therapy in r/r DLBCL) | NCT05464719 September 2022 - January 2026 | ~n = 30 (As of February 2024) | N/A |
| City of Hope (loncastuximab + mosunetuzumab for r/r DLBCL) | NCT05672251 January 2024 - December 2024 | ~n = 36 (As of February 2024) | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juárez-Salcedo, L.M.; Nimkar, S.; Corazón, A.M.; Dalia, S. Loncastuximab Tesirine in the Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma. Int. J. Mol. Sci. 2024, 25, 7580. https://doi.org/10.3390/ijms25147580
Juárez-Salcedo LM, Nimkar S, Corazón AM, Dalia S. Loncastuximab Tesirine in the Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma. International Journal of Molecular Sciences. 2024; 25(14):7580. https://doi.org/10.3390/ijms25147580
Chicago/Turabian StyleJuárez-Salcedo, Luis Miguel, Santosh Nimkar, Ana María Corazón, and Samir Dalia. 2024. "Loncastuximab Tesirine in the Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma" International Journal of Molecular Sciences 25, no. 14: 7580. https://doi.org/10.3390/ijms25147580
APA StyleJuárez-Salcedo, L. M., Nimkar, S., Corazón, A. M., & Dalia, S. (2024). Loncastuximab Tesirine in the Treatment of Relapsed or Refractory Diffuse Large B-Cell Lymphoma. International Journal of Molecular Sciences, 25(14), 7580. https://doi.org/10.3390/ijms25147580





