P2Y12 Receptor Inhibitor for Antiaggregant Therapies: From Molecular Pathway to Clinical Application
Abstract
1. Introduction
2. Search Strategy and Selection Criteria
3. Purinergic Receptors’ Mechanism of Action: Thienopyridine vs. Nonthienopyridine P2Y12 Inhibitors
Effective Signaling Pathways and Platelet Inhibitory Purinergic Receptors
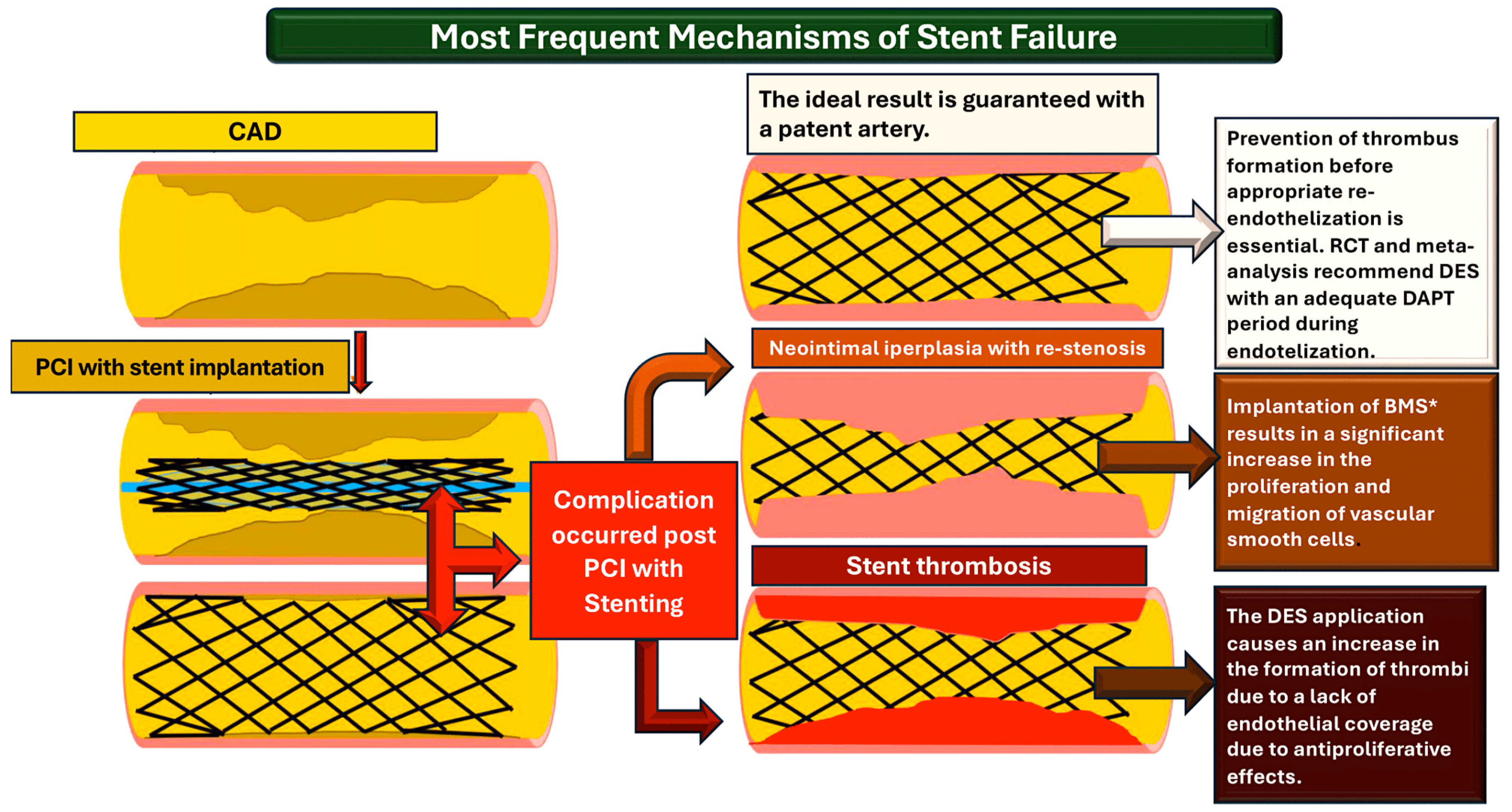
4. Current Therapies on P2Y12 Receptor Inhibitor
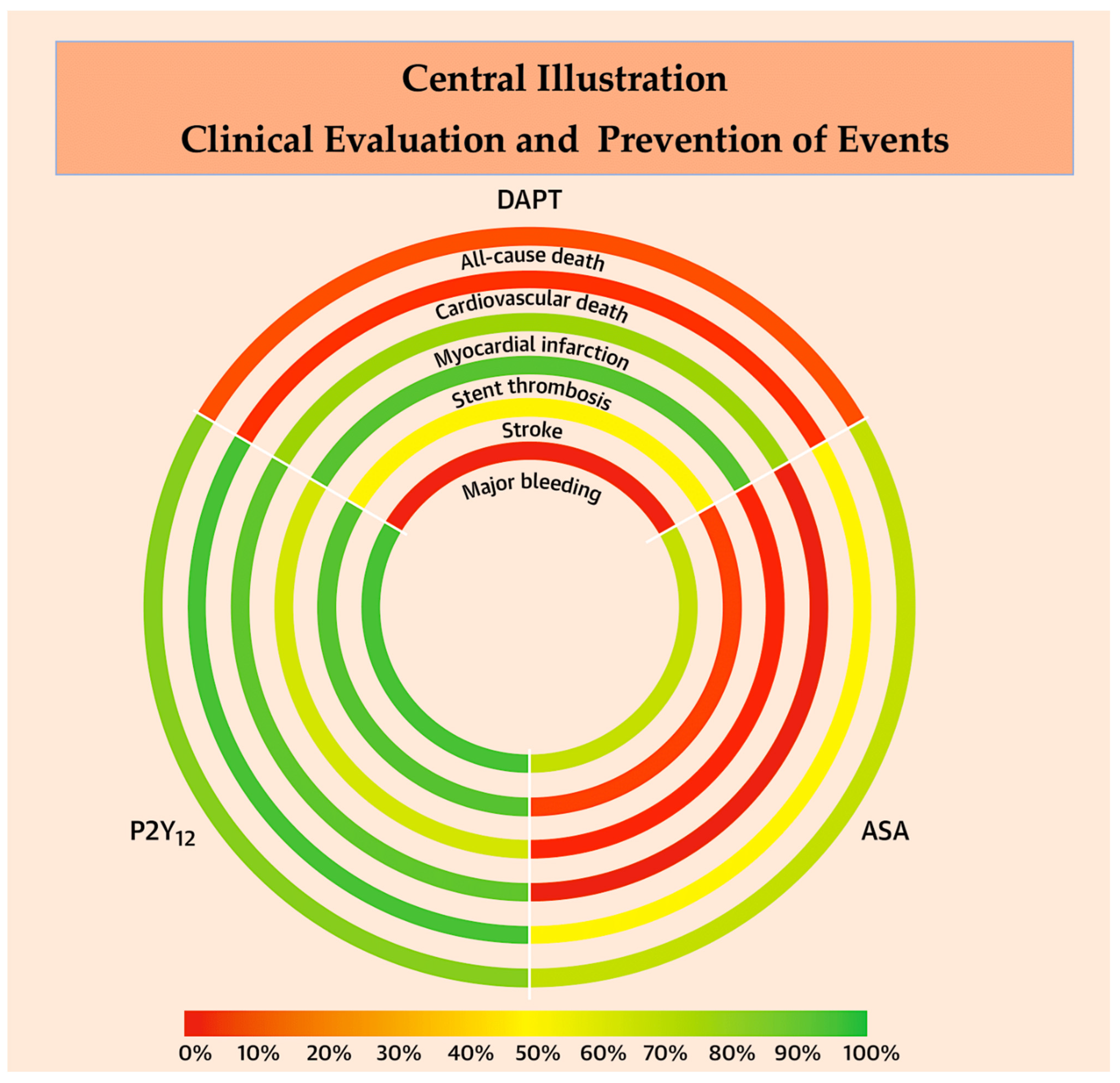
4.1. Assessing Meta-Analyses Investigating DAPT vs. SAPT for Balancing High Risk of Bleeding and Thrombosis
4.2. Thrombotic Complications Receptor Signaling: Emerging Evidence from RCTs and Meta-Analysis
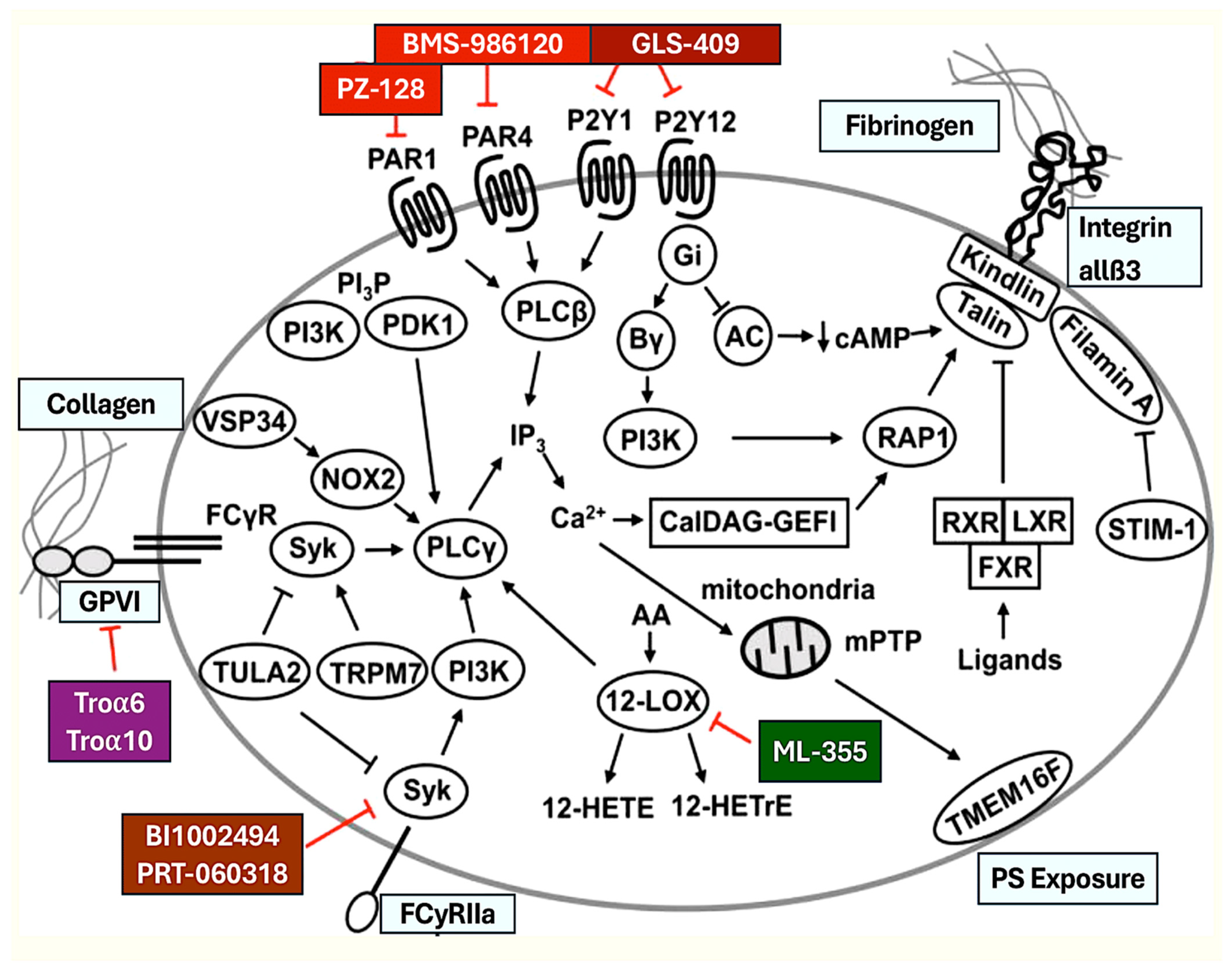
5. Future Direction
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mehran, R.; Rao, S.V.; Bhatt, D.L.; Gibson, C.M.; Caixeta, A.; Eikelboom, J.; Kaul, S.; Wiviott, S.D.; Menon, V.; Nikolsky, E.; et al. Standardized bleeding definitions for cardiovascular clinical trials: A consensus report from the Bleeding Academic Research Consortium. Circulation 2011, 123, 2736–2747. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Delaney, M.K.; O’Brien, K.A.; Du, X. Signaling during platelet adhesion and activation. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2341–2349. [Google Scholar] [CrossRef] [PubMed]
- Nieswandt, B.; Pleines, I.; Bender, M. Platelet adhesion and activation mechanisms in arterial thrombosis and ischaemic stroke. J. Thromb. Haemost. 2011, 9 (Suppl. S1), 92–104. [Google Scholar] [CrossRef] [PubMed]
- Bye, A.P.; Unsworth, A.J.; Gibbins, J.M. Platelet signaling: A complex interplay between inhibitory and activatory networks. J. Thromb. Haemost. 2016, 14, 918–930. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, M.; Gragnano, F.; Branca, M.; Franzone, A.; da Costa, B.R.; Baber, U.; Kimura, T.; Jang, Y.; Hahn, J.Y.; Zhao, Q.; et al. Single Versus Dual Antiplatelet Therapy (Sidney-3) Collaboration. Ticagrelor or Clopidogrel Monotherapy vs Dual Antiplatelet Therapy After Percutaneous Coronary Intervention: A Systematic Review and Patient-Level Meta-Analysis. JAMA Cardiol. 2024, 20, e240133. [Google Scholar] [CrossRef]
- Gragnano, F.; Cao, D.; Pirondini, L.; Franzone, A.; Kim, H.S.; von Scheidt, M.; Pettersen, A.R.; Zhao, Q.; Woodward, M.; Chiarito, M.; et al. P2Y12 inhibitor or aspirin monotherapy for secondary prevention of coronary events. J. Am. Coll. Cardiol. 2023, 82, 89–105. [Google Scholar] [CrossRef]
- Steinhubl, S.R.; Berger, P.B.; Mann, J.T., 3rd; Fry, E.T.; DeLago, A.; Wilmer, C.; Topol, E.J.; CREDO Investigators. Clopidogrel for the Reduction of Events During Observation. Early and sustained dual oral antiplatelet therapy following percutaneous coronary intervention: A randomized controlled trial. JAMA 2002, 288, 2411–2420. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.R.; Yusuf, S. Short- and long-term oral antiplatelet therapy in acute coronary syndromes and percutaneous coronary intervention. J. Am. Coll. Cardiol. 2003, 41 (Suppl. S4), 79S–88S. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.R.; Yusuf, S.; Peters, R.J.; Bertrand, M.E.; Lewis, B.S.; Natarajan, M.K.; Malmberg, K.; Rupprecht, H.-J.; Zhao, F.; Chrolavicius, S.; et al. Effects of pretreatment with clopidogrel and aspirin followed by long-term therapy in patients undergoing percutaneous coronary intervention: The PCI-CURE study. Lancet 2001, 358, 527–533. [Google Scholar] [CrossRef]
- Angiolillo, D.J.; Guzman, L.A.; Bass, T.A. Current antiplatelet therapies: Benefits and limitations. Am. Heart J. 2008, 156 (Suppl. S2), S3–S9. [Google Scholar] [CrossRef]
- Grover, S.P.; Bergmeier, W.; Mackman, N. Platelet Signaling Pathways and New Inhibitors. Arterioscler. Thromb. Vasc. Biol. 2018, 38, e28–e35. [Google Scholar] [CrossRef] [PubMed]
- Hahn, J.Y.; Song, Y.B.; Oh, J.H.; Cho, D.K.; Lee, J.B.; Doh, J.H.; Kim, S.H.; Jeong, J.O.; Bae, J.H.; Kim, B.O.; et al. 6-Month versus 12-month or longer dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndrome (SMART-DATE): A randomised, open-label, non-inferiority trial. Lancet 2018, 391, 1274–1284. [Google Scholar] [CrossRef] [PubMed]
- Laudani, C.; Greco, A.; Occhipinti, G.; Ingala, S.; Calderone, D.; Scalia, L.; Agnello, F.; Legnazzi, M.; Mauro, M.S.; Rochira, C.; et al. Short duration of DAPT versus de-escalation after percutaneous coronary intervention for acute coronary syndromes. JACC Cardiovasc. Interv. 2022, 15, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Palmerini, T.; Della Riva, D.; Benedetto, U.; Bacchi Reggiani, L.; Feres, F.; Abizaid, A.; Gilard, M.; Morice, M.C.; Valgimigli, M.; Hong, M.K.; et al. Three, six, or twelve months of dual antiplatelet therapy after DES implantation in patients with or without acute coronary syndromes: An individual patient data pairwise and network meta-analysis of six randomized trials and 11 473 patients. Eur. Heart J. 2017, 38, 1034–1043. [Google Scholar] [CrossRef] [PubMed]
- Franzone, A.; McFadden, E.; Leonardi, S.; Piccolo, R.; Vranckx, P.; Serruys, P.W.; Benit, E.; Liebetrau, C.; Janssens, L.; Ferrario, M.; et al. Ticagrelor alone versus dual antiplatelet therapy from 1 month after drug-eluting coronary stenting. J. Am. Coll. Cardiol. 2019, 74, 2223–2234. [Google Scholar] [CrossRef] [PubMed]
- Mehran, R.; Baber, U.; Sharma, S.K.; Cohen, D.J.; Angiolillo, D.J.; Briguori, C.; Cha, J.Y.; Collier, T.; Dangas, G.; Dudek, D.; et al. Ticagrelor with or without aspirin in high-risk patients after PCI. N. Engl. J. Med. 2019, 381, 2032–2042. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Domei, T.; Morimoto, T.; Natsuaki, M.; Shiomi, H.; Toyota, T.; Ohya, M.; Suwa, S.; Takagi, K.; Nanasato, M.; et al. Effect of 1-month dual antiplatelet therapy followed by clopidogrel vs 12-month dual antiplatelet therapy on cardiovascular and bleeding events in patients receiving PCI: The STOPDAPT-2 randomized clinical trial. JAMA 2019, 321, 2414–2427. [Google Scholar] [CrossRef] [PubMed]
- Hahn, J.Y.; Song, Y.B.; Oh, J.H.; Chun, W.J.; Park, Y.H.; Jang, W.J.; Im, E.S.; Jeong, J.O.; Cho, B.R.; Oh, S.K.; et al. Effect of P2Y12 inhibitor monotherapy vs dual antiplatelet therapy on cardiovascular events in patients undergoing percutaneous coronary intervention: The SMART-CHOICE randomized clinical trial. JAMA 2019, 321, 2428–2437. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Hong, S.J.; Cho, Y.H.; Yun, K.H.; Kim, Y.H.; Suh, Y.; Cho, J.Y.; Her, A.Y.; Cho, S.; Jeon, D.W.; et al. Effect of ticagrelor monotherapy vs ticagrelor with aspirin on major bleeding and cardiovascular events in patients with acute coronary syndrome: The TICO randomized clinical trial. JAMA 2020, 323, 2407–2416. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhu, Y.; Xu, Z.; Cheng, Z.; Mei, J.; Chen, X.; Wang, X. Effect of ticagrelor plus aspirin, ticagrelor alone, or aspirin alone on saphenous vein graft patency 1 year after coronary artery bypass grafting: A randomized clinical trial. JAMA 2018, 319, 1677–1686. [Google Scholar] [CrossRef]
- Valgimigli, M.; Gragnano, F.; Branca, M.; Franzone, A.; Baber, U.; Jang, Y.; Kimura, T.; Hahn, J.-Y.; Zhao, Q.; Windecker, S.; et al. P2Y12 inhibitor monotherapy or dual antiplatelet therapy after coronary revascularisation: Individual patient level meta-analysis of randomised controlled trials. BMJ 2021, 373, n1332. [Google Scholar] [CrossRef] [PubMed]
- Sibbing, D.; Aradi, D.; Alexopoulos, D.; Ten Berg, J.; Bhatt, D.L.; Bonello, L.; Collet, J.P.; Cuisset, T.; Franchi, F.; Gross, L.; et al. Updated expert consensus statement on platelet function and genetic testing for guiding P2Y12 receptor inhibitor treatment in percutaneous coronary intervention. JACC Cardiovasc. Interv. 2019, 12, 1521–1537. [Google Scholar] [CrossRef] [PubMed]
- Brandt, J.T.; Close, S.L.; Iturria, S.J.; Payne, C.D.; Farid, N.A.; Ernest, C.S., 2nd; Lachno, D.R.; Salazar, D.; Winters, K.J. Common polymorphisms of CYP2C19 and CYP2C9 affect the pharmacokinetic and pharmacodynamic response to clopidogrel but not prasugrel. J. Thromb. Haemost. 2007, 5, 2429–2436. [Google Scholar] [CrossRef] [PubMed]
- Mega, J.L.; Close, S.L.; Wiviott, S.D.; Shen, L.; Hockett, R.D.; Brandt, J.T.; Walker, J.R.; Antman, E.M.; Macias, W.L.; Braunwald, E.; et al. Cytochrome P450 genetic polymorphisms and the response to prasugrel: Relationship to pharmacokinetic, pharmacodynamic, and clinical outcomes. Circulation 2009, 119, 2553–2560. [Google Scholar] [CrossRef]
- Kelly, R.P.; Close, S.L.; Farid, N.A.; Winters, K.J.; Shen, L.; Natanegara, F.; Jakubowski, J.A.; Ho, M.; Walker, J.R.; Small, D.S. Pharmacokinetics and pharmacodynamics following maintenance doses of prasugrel and clopidogrel in Chinese carriers of CYP2C19 variants. Br. J. Clin. Pharmacol. 2012, 73, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, H.; Morimoto, T.; Natsuaki, M.; Yamamoto, K.; Obayashi, Y.; Ogita, M.; Suwa, S.; Isawa, T.; Domei, T.; Yamaji, K.; et al. Comparison of clopidogrel monotherapy after 1 to 2 months of dual antiplatelet therapy with 12 months of dual antiplatelet therapy in patients with acute coronary syndrome: The STOPDAPT-2 ACS randomized clinical trial. JAMA Cardiol. 2022, 7, 407–417. [Google Scholar] [CrossRef]
- Franchi, F.; Rollini, F.; Rivas, A.; Wali, M.; Briceno, M.; Agarwal, M.; Shaikh, Z.; Nawaz, A.; Silva, G.; Been, L.; et al. Platelet Inhibition With Cangrelor and Crushed Ticagrelor in Patients With ST-Segment-Elevation Myocardial Infarction Undergoing Primary Percutaneous Coronary Intervention. Circulation 2019, 139, 1661–1670. [Google Scholar] [CrossRef] [PubMed]
- Akkaif, M.A.; Daud, N.A.A.; Noor, D.A.M.; Sha’aban, A.; Kader, M.A.S.A.; Ibrahim, B. The Impact of CYP2C19 Genotype on the Platelet Reactivity Index (PRI) among Chronic Coronary Syndromes (CCS) Patients Undergoing Percutaneous Coronary Intervention (PCI): Affectability of Rapid Genetic Testing. Cardiovasc. Drugs Ther. 2024. online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Holmes, M.V.; Perel, P.; Shah, T.; Hingorani, A.D.; Casas, J.P. CYP2C19 genotype, clopidogrel metabolism, platelet function, and cardiovascular events: A systematic review and meta-analysis. JAMA 2011, 306, 2704–2714. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, D.; Song, S.; Huang, X.; Ding, Y.; Meng, R. Efficacy and Safety of Long-Term Dual Antiplatelet Therapy: A Systematic Review and Meta-Analysis. Clin. Appl. Thromb. Hemost. 2024, 30, 10760296241244772. [Google Scholar] [CrossRef]
- Sabatine, M.S.; Cannon, C.P.; Gibson, C.M.; López-Sendón, J.L.; Montalescot, G.; Theroux, P.; Lewis, B.S.; Murphy, S.A.; McCabe, C.H.; Braunwald, E.; et al. Effect of clopidogrel pretreatment before percutaneous coronary intervention in patients with ST-elevation myocardial infarction treated with fibrinolytics: The PCI-CLARITY study. JAMA 2005, 294, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Rodés-Cabau, J.; Masson, J.-B.; Welsh, R.C.; del Blanco, B.G.; Pelletier, M.; Webb, J.G.; Al-Qoofi, F.; Généreux, P.; Maluenda, G.; Thoenes, M.; et al. Aspirin Versus Aspirin Plus Clopidogrel as Antithrombotic Treatment Following Transcatheter Aortic Valve Replacement With a Balloon-Expandable Valve: The ARTE (Aspirin Versus Aspirin + Clopidogrel Following Transcatheter Aortic Valve Implantation) Randomized Clinical Trial. JACC Cardiovasc. Interv. 2017, 10, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Keller, T.T.; Squizzato, A.; Middeldorp, S. Clopidogrel plus aspirin versus aspirin alone for preventing cardiovascular disease. Cochrane Database Syst. Rev. 2007, 3, CD005158. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Fox, K.A.; Hacke, W.; Berger, P.B.; Black, H.R.; Boden, W.E.; Cacoub, P.; Cohen, E.A.; Creager, M.A.; Easton, J.D.; et al. Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. N. Engl. J. Med. 2006, 354, 1706–1717. [Google Scholar] [CrossRef] [PubMed]
- Diener, H.-C.; Bogousslavsky, J.; Brass, L.M.; Cimminiello, C.; Csiba, L.; Kaste, M.; Leys, D.; Matias-Guiu, J.; Rupprecht, H.-J. Aspirin and clopidogrel compared with clopidogrel alone after recent ischaemic stroke or transient ischaemic attack in high-risk patients (MATCH): Randomised, double-blind, placebo-controlled trial. Lancet 2004, 364, 331–337. [Google Scholar] [CrossRef]
- Estevez, B.; Du, X. New concepts and mechanisms of platelet activation signaling. Physiology 2017, 32, 162–177. [Google Scholar] [CrossRef] [PubMed]
- Tantry, U.S.; Bonello, L.; Aradi, D.; Price, M.J.; Jeong, Y.H.; Angiolillo, D.J.; Stone, G.W.; Curzen, N.; Geisler, T.; Berg, J.T.; et al. Consensus and update on the definition of on-treatment platelet reactivity to adenosine diphosphate associated with ischemia and bleeding. J. Am. Coll. Cardiol. 2013, 62, 2261–2273. [Google Scholar] [CrossRef]
- Armstrong, P.C.; Hoefer, T.; Knowles, R.B.; Tucker, A.T.; Hayman, M.A.; Ferreira, P.M.; Chan, M.V.; Warner, T.D. Newly formed reticulated platelets undermine pharmacokinetically short-lived antiplatelet therapies. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Gremmel, T.; Yanachkov, I.B.; Yanachkova, M.I.; Wright, G.E.; Wider, J.; Undyala, V.V.; Michelson, A.D.; Frelinger, A.L.I.I.I.; Przyklenk, K. Synergistic inhibition of both P2Y1 and P2Y12 adenosine diphosphate receptors as novel approach to rapidly attenuate platelet-mediated thrombosis. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Yanachkov, I.B.; Chang, H.; Yanachkova, M.I.; Dix, E.J.; Berny-Lang, M.A.; Gremmel, T.; Michelson, A.D.; Wright, G.E.; Frelinger, A.L.I.I.I. New highly active antiplatelet agents with dual specificity for platelet P2Y1 and P2Y12 adenosine diphosphate receptors. Eur. J. Med. Chem. 2016, 107, 204–218. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.L.; Hulot, J.S.; Moliterno, D.J.; Harrington, R.A. Antiplatelet and anticoagulation therapy for acute coronary syndromes. Circ. Res. 2014, 114, 1929–1943. [Google Scholar] [CrossRef]
- Guthikonda, S.; Alviar, C.L.; Vaduganathan, M.; Arikan, M.; Tellez, A.; DeLao, T.; Granada, J.F.; Dong, J.F.; Kleiman, N.S.; Lev, E.I. Role of reticulated platelets and platelet size heterogeneity on platelet activity after dual antiplatelet therapy with aspirin and clopidogrel in patients with stable coronary artery disease. J. Am. Coll. Cardiol. 2008, 52, 743–749. [Google Scholar] [CrossRef] [PubMed]
- Vaduganathan, M.; Bhatt, D.L. Simultaneous platelet P2Y12 and P2Y1 ADP receptor blockade: Are two better than one? Arterioscler. Thromb. Vasc. Biol. 2016, 36, 427–428. [Google Scholar] [CrossRef]
- Bhatt, D.L. Intensifying platelet inhibition–navigating between Scylla and Charybdis. N. Engl. J. Med. 2007, 357, 2078–2081. [Google Scholar] [CrossRef]
- Bhatt, D.L.; Stone, G.W.; Mahaffey, K.W.; Gibson, C.M.; Steg, P.G.; Hamm, C.W.; Price, M.J.; Leonardi, S.; Gallup, D.; Bramucci, E.; et al. Effect of platelet inhibition with cangrelor during PCI on ischemic events. N. Engl. J. Med. 2013, 368, 1303–1313. [Google Scholar] [CrossRef]
- Liverani, E.; Rico, M.C.; Tsygankov, A.Y.; Kilpatrick, L.E.; Kunapuli, S.P. P2Y12 receptor modulates sepsis-induced inflammation. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 961–971. [Google Scholar] [CrossRef]
- Rahman, M.; Gustafsson, D.; Wang, Y.; Thorlacius, H.; Braun, O.Ö. Ticagrelor reduces neutrophil recruitment and lung damage in abdominal sepsis. Platelets 2014, 25, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Chiarito, M.; Sanz-Sánchez, J.; Cannata, F.; Cao, D.; Sturla, M.; Panico, C.; Godino, C.; Regazzoli, D.; Reimers, B.; De Caterina, R.; et al. Monotherapy with a P2Y12 inhibitor or aspirin for secondary prevention in patients with established atherosclerosis: A systematic review and meta-analysis. Lancet 2020, 395, 1487–1495. [Google Scholar] [CrossRef] [PubMed]
- Giacoppo, D.; Matsuda, Y.; Fovino, L.N.; D’amico, G.; Gargiulo, G.; A Byrne, R.; Capodanno, D.; Valgimigli, M.; Mehran, R.; Tarantini, G. Short dual antiplatelet therapy followed by P2Y12 inhibitor monotherapy vs. prolonged dual antiplatelet therapy after percutaneous coronary intervention with second-generation drug-eluting stents: A systematic review and meta-analysis of randomized clinical trials. Eur. Heart J. 2021, 42, 308–319. [Google Scholar]
- Lahu, S.; Bristot, P.; Gewalt, S.; Goedel, A.; Giacoppo, D.; Schüpke, S.; Schunkert, H.; Kastrati, A.; Sarafoff, N. Meta-analysis of short vs. prolonged dual antiplatelet therapy after drug-eluting stent implantation and role of continuation with either aspirin or a P2Y12 inhibitor thereafter. J. Atheroscler. Thromb. 2022, 29, 1001–1019. [Google Scholar] [CrossRef]
- Pettersen, A.Å.; Seljeflot, I.; Abdelnoor, M.; Arnesen, H. High On-Aspirin Platelet Reactivity and Clinical Outcome in Patients With Stable Coronary Artery Disease: Results From ASCET (Aspirin Nonresponsiveness and Clopidogrel Endpoint Trial). J. Am. Heart Assoc. 2012, 1, e000703. [Google Scholar] [CrossRef]
- Woodward, M.; Lowe, G.D.O.; Francis, L.M.A.; Rumley, A.; Cobbe, S.M.; CADET Study Investigators. A randomized comparison of the effects of aspirin and clopidogrel on thrombotic risk factors and Creactive protein following myocardial infarction: The CADET trial. J. Thromb. Haemost. 2004, 2, 1934–1940. [Google Scholar] [CrossRef] [PubMed]
- CAPRIE Steering Committee. A randomised, blinded, trial of clopidogrel versus aspirin in patients at risk of ischaemic events (CAPRIE). CAPRIE Steering Committee. Lancet 1996, 348, 1329–1339. [Google Scholar] [CrossRef] [PubMed]
- Vranckx, P.; Valgimigli, M.; Odutayo, A.; Serruys, P.W.; Hamm, C.; Steg, P.G.; Heg, D.; Mc Fadden, E.P.; Onuma, Y.; Benit, E.; et al. Efficacy and Safety of Ticagrelor Monotherapy by Clinical Presentation: Pre-Specified Analysis of the GLOBAL LEADERS Trial. J. Am. Heart Assoc. 2021, 10, e015560. [Google Scholar] [CrossRef] [PubMed]
- Koo, B.-K.; Kang, J.; Park, K.W.; Rhee, T.-M.; Yang, H.-M.; Won, K.-B.; Rha, S.-W.; Bae, J.-W.; Lee, N.H.; Hur, S.-H.; et al. Aspirin versus clopidogrel for chronic maintenance monotherapy after percutaneous coronary intervention (HOST-EXAM): An investigator-initiated, prospective, randomised, open-label, multicentre trial. Lancet 2021, 397, 2487–2496. [Google Scholar] [CrossRef] [PubMed]
- Gargiulo, G.; Windecker, S.; Vranckx, P.; Gibson, C.M.; Mehran, R.; Valgimigli, M. A critical appraisal of aspirin in secondary prevention. Circulation 2016, 134, 1881–1906. [Google Scholar] [CrossRef] [PubMed]
- Capodanno, D.; Mehran, R.; Valgimigli, M.; Baber, U.; Windecker, S.; Vranckx, P.; Dangas, G.; Rollini, F.; Kimura, T.; Collet, J.P.; et al. Aspirin-free strategies in cardiovascular disease and cardioembolic stroke prevention. Nat. Rev. Cardiol. 2018, 15, 480–496. [Google Scholar] [CrossRef] [PubMed]
- Capodanno, D.; Baber, U.; Bhatt, D.L.; Collet, J.-P.; Dangas, G.; Franchi, F.; Gibson, C.M.; Gwon, H.-C.; Kastrati, A.; Kimura, T.; et al. P2Y12 inhibitor monotherapy in patients undergoing percutaneous coronary intervention. Nat. Rev. Cardiol. 2022, 19, 829–844. [Google Scholar] [CrossRef] [PubMed]
- Schunkert, H.; Boening, A.; von Scheidt, M.; Lanig, C.; Gusmini, F.; de Waha, A.; Kuna, C.; Fach, A.; Grothusen, C.; Oberhoffer, M.; et al. Randomized trial of ticagrelor vs. aspirin in patients after coronary artery bypass grafting: The TiCAB trial. Eur. Heart J. 2019, 40, 2432–2440. [Google Scholar] [CrossRef]
- Gorelick, P.B.; Richardson, D.; Kelly, M.; Ruland, S.; Hung, E.; Harris, Y.; Kittner, S.; Leurgans, S.; African American Antiplatelet Stroke Prevention Study Investigators. Aspirin and ticlopidine for prevention of recurrent stroke in black patients: A randomized trial. JAMA 2003, 289, 2947–2957. [Google Scholar] [CrossRef]
- Scrutinio, D.; Cimminiello, C.; Marubini, E.; Pitzalis, M.V.; Di Biase, M.; Rizzon, P. Ticlopidine versus aspirin after myocardial infarction (STAMI) trial. J. Am. Coll. Cardiol. 2001, 37, 1259–1265. [Google Scholar] [CrossRef] [PubMed]
- Hass, W.K.; Easton, J.D.; Adams, H.P., Jr.; Pryse-Phillips, W.; Molony, B.A.; Anderson, S.; Kamm, B.; for the Ticlopidine Aspirin Stroke Study Group. A randomized trial comparing ticlopidine hydrochloride with aspirin for the prevention of stroke in high-risk patients. N. Engl. J. Med. 1989, 321, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Alberts, M.J.; Bhatt, D.L.; Mas, J.-L.; Ohman, E.M.; Hirsch, A.T.; Röther, J.; Salette, G.; Goto, S.; Smith, S.C.; Liau, C.-S.; et al. Three-year follow-up and event rates in the international REduction of Atherothrombosis for Continued Health registry. Eur. Heart J. 2009, 30, 2318–2326. [Google Scholar] [CrossRef] [PubMed]
- Johnston, S.C.; Amarenco, P.; Albers, G.W.; Denison, H.; Easton, J.D.; Evans, S.R.; Held, P.; Jonasson, J.; Minematsu, K.; Molina, C.A.; et al. Ticagrelor versus aspirin in acute stroke or transient ischemic attack. N. Engl. J. Med. 2016, 375, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Rothwell, P.M.; Villagra, R.; Gibson, R.; Donders, R.C.J.M.; Warlow, C.P. Evidence of a chronic systemic cause of instability of atherosclerotic plaques. Lancet 2000, 355, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Sabeti, S.; Schlager, O.; Exner, M.; Mlekusch, W.; Amighi, J.; Dick, P.; Maurer, G.; Huber, K.; Koppensteiner, R.; Wagner, O.; et al. Progression of carotid stenosis detected by duplex ultrasonography predicts adverse outcomes in cardiovascular high-risk patients. Stroke 2007, 38, 2887–2894. [Google Scholar] [CrossRef] [PubMed]
- Levine, G.N.; Bates, E.R.; Bittl, J.A.; Brindis, R.G.; Fihn, S.D.; Fleisher, L.A.; Granger, C.B.; Lange, R.A.; Mack, M.J.; Mauri, L.; et al. 2016 ACC/AHA guideline focused update on duration of dual antiplatelet therapy in patients with coronary artery disease: A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J. Am. Coll. Cardiol. 2016, 68, 1082–1115. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, G. Aspirin-induced gastric mucosal injury: Lessons learned from animal models. Gastroenterology 1989, 96 (Suppl. S2), 606–614. [Google Scholar] [CrossRef] [PubMed]
- Andò, G.; De Santis, G.A.; Greco, A.; Pistelli, L.; Francaviglia, B.; Capodanno, D.; De Caterina, R.; Capranzano, P. P2Y12 inhibitor or aspirin following dual antiplatelet therapy after percutaneous coronary intervention. J. Am. Coll. Cardiol. Intv. 2022, 15, 2239–2249. [Google Scholar] [CrossRef]
- Kim, B.K.; Hong, M.K.; Shin, D.H.; Nam, C.M.; Kim, J.S.; Ko, Y.G.; Choi, D.; Kang, T.S.; Park, B.E.; Kang, W.C.; et al. A new strategy for discontinuation of dual antiplatelet therapy: The RESET Trial (REal Safety and Efficacy of 3-month dual antiplatelet Therapy following Endeavor zotarolimus-eluting stent implantation). J. Am. Coll. Cardiol. 2012, 60, 1340–1348. [Google Scholar] [CrossRef]
- Feres, F.; Costa, R.A.; Abizaid, A.; Leon, M.B.; Marin-Neto, J.A.; Botelho, R.V.; King, S.B., 3rd; Negoita, M.; Liu, M.; de Paula, J.E.; et al. Three vs twelve months of dual antiplatelet therapy after zotarolimus-eluting stents: The OPTIMIZE randomized trial. JAMA 2013, 310, 2510–2522. [Google Scholar] [CrossRef] [PubMed]
- De Luca, G.; Damen, S.A.; Camaro, C.; Benit, E.; Verdoia, M.; Rasoul, S.; Liew, H.B.; Polad, J.; Ahmad, W.A.; Zambahari, R.; et al. Final results of the randomised evaluation of short-term dual antiplatelet therapy in patients with acute coronary syndrome treated with a new-generation stent (REDUCE trial). EuroIntervention 2019, 15, e990–e998. [Google Scholar] [CrossRef] [PubMed]
- Vranckx, P.; Valgimigli, M.; Jüni, P.; Hamm, C.; Steg, P.G.; Heg, D.; van Es, G.A.; McFadden, E.P.; Onuma, Y.; van Meijeren, C.; et al. Ticagrelor plus aspirin for 1 month, followed by ticagrelor monotherapy for 23 months vs aspirin plus clopidogrel or ticagrelor for 12 months, followed by aspirin monotherapy for 12 months after implantation of a drug-eluting stent: A multicentre, open-label, randomised superiority trial. Lancet 2018, 392, 940–949. [Google Scholar] [CrossRef] [PubMed]
- Colombo, A.; Chieffo, A.; Frasheri, A.; Garbo, R.; Masotti-Centol, M.; Salvatella, N.; Dominguez, J.F.O.; Steffanon, L.; Tarantini, G.; Presbitero, P.; et al. Sec- ond-generation drug-eluting stent implantation followed by 6- versus 12-month dual antiplatelet therapy: The SECURITY randomized clinical trial. J. Am. Coll. Cardiol. 2014, 64, 2086–2097. [Google Scholar] [CrossRef] [PubMed]
- Gilard, M.; Barragan, P.; Noryani, A.A.; Noor, H.A.; Majwal, T.; Hovasse, T.; Castellant, P.; Schneeberger, M.; Maillard, L.; Bressolette, E.; et al. 6- versus 24-month dual antiplatelet therapy after implantation of drug-eluting stents in patients nonresistant to aspirin: The randomized, multicenter ITALIC trial. J. Am. Coll. Cardiol. 2015, 65, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.; Xu, K.; Jing, Q.; Li, J.; Li, Y. Six versus 12 months of dual antiplatelet therapy after implantation of biodegradable polymer sirolimus-eluting stent: Randomized substudy of the I-LOVE-IT 2 trial. Circ. Cardiovasc. Interv. 2016, 9, e003145. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.J.; Shin, D.H.; Kim, J.S.; Kim, B.K.; Ko, Y.G.; Choi, D.; Her, A.Y.; Kim, Y.H.; Jang, Y.; Hong, M.K.; et al. 6-Month Versus 12-Month Dual-Antiplatelet Therapy Following Long Everolimus-Eluting Stent Implantation: The IVUS-XPL Randomized Clinical Trial. JACC Cardiovasc. Interv. 2016, 9, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Kedhi, E.; Fabris, E.; Van Der Ent, M.; Buszman, P.; Von Birgelen, C.; Roolvink, V.; Zurakowski, A.; Schotborgh, C.E.; Hoorntje, J.C.A.; Eek, C.H.; et al. Six months versus 12 months dual antiplatelet therapy after drug-eluting stent implantation in ST- elevation myocardial infarction (DAPT-STEMI): Randomised, multicentre, non-inferiority trial. BMJ 2018, 363, k3793. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.J.; Kim, J.S.; Hong, S.J.; Lim, D.S.; Lee, S.Y.; Yun, K.H.; Park, J.K.; Kang, W.C.; Kim, Y.H.; Yoon, H.J.; et al. 1-Month Dual-Antiplatelet Therapy Followed by Aspirin Monotherapy After Polymer-Free Drug-Coated Stent Implantation: One-Month DAPT Trial. One-Mon. DAPT Investig. JACC Cardiovasc. Interv. 2021, 14, 1801–1811. [Google Scholar] [CrossRef]
- Li, L.; Geraghty, O.C.; Mehta, Z.; Rothwell, P.M. Age-specific risks, severity, time course, and outcome of bleeding on long-term antiplatelet treatment after vascular events: A population-based cohort study. Lancet 2017, 390, 490–499. [Google Scholar] [CrossRef]
- Kumbhani, D.J.; de Lemos, J.A. The Optimal Antiplatelet Agent for Secondary Prevention of Coronary Artery Disease: Ring Out the Old? J. Am. Coll. Cardiol. 2023, 82, 106–108. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, M.L.; Murphy, S.A.; Sabatine, M.S. The safety and efficacy of aspirin discontinuation on a background of a P2Y12 inhibitor in patients after percutaneous coronary intervention: A systematic review and meta-analysis. Circulation 2020, 142, 538–545. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, M.; Frigoli, E.; Heg, D.; Tijssen, J.; Jüni, P.; Vranckx, P.; Ozaki, Y.; Morice, M.-C.; Chevalier, B.; Onuma, Y.; et al. Dual antiplatelet therapy after PCI in patients at high bleeding risk. N. Engl. J. Med. 2021, 385, 1643–1655. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, M.; Costa, F.; Lokhnygina, Y.; Clare, R.M.; Wallentin, L.; Moliterno, D.J.; Armstrong, P.W.; White, H.D.; Held, C.; Aylward, P.E.; et al. Trade-off of myocardial infarction vs. bleeding types on mortality after acute coronary syndrome: Lessons from the Thrombin Receptor Antagonist for Clinical Event Reduction in Acute Coronary Syndrome (TRACER) randomized trial. Eur. Heart J. 2017, 38, 804–810. [Google Scholar] [CrossRef]
- Leonardi, S.; Gragnano, F.; Carrara, G.; Gargiulo, G.; Frigoli, E.; Vranckx, P.; Di Maio, D.; Spedicato, V.; Monda, E.; Fimiani, L.; et al. Prognostic implications of declining hemoglobin content in patients hospitalized with acute coronary syndromes. J. Am. Coll. Cardiol. 2021, 77, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Aradi, D.; Kirtane, A.; Bonello, L.; Gurbel, P.A.; Tantry, U.S.; Huber, K.; Freynhofer, M.K.; ten Berg, J.; Janssen, P.; Angiolillo, D.J.; et al. Bleeding and stent thrombosis on P2Y12-inhibitors: Collaborative analysis on the role of platelet reactivity for risk stratification after percutaneous coronary intervention. Eur. Heart J. 2015, 36, 1762–1771. [Google Scholar] [CrossRef] [PubMed]
- Tamargo, J.; Kaski, J.C.; Kimura, T.; Barton, J.C.; Yamamoto, K.; Komiyama, M.; Drexel, H.; Lewis, B.S.; Agewall, S.; Hasegawa, K. Racial and ethnic differences in pharmacotherapy to prevent coronary artery disease and thrombotic events. Eur. Heart J. Cardiovasc. Pharmacother. 2022, 8, 738–751. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, M.; Smits, P.C.; Frigoli, E.; Bongiovanni, D.; Tijssen, J.; Hovasse, T.; Mafragi, A.; Ruifrok, W.T.; Karageorgiev, D.; Aminian, A.; et al. Duration of antiplatelet therapy after complex percutaneous coronary intervention in patients at high bleeding risk: A MASTER DAPT trial sub-analysis. Eur. Heart J. 2022, 43, 3100–3114. [Google Scholar] [CrossRef] [PubMed]
- Gurbel, P.A.; Bliden, K.P.; Turner, S.E.; Tantry, U.S.; Gesheff, M.G.; Barr, T.P.; Covic, L.; Kuliopulos, A. Cell-penetrating pepducin therapy targeting PAR1 in subjects with coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 189–197. [Google Scholar] [CrossRef]
- Wilson, S.J.; Ismat, F.A.; Wang, Z.; Cerra, M.; Narayan, H.; Raftis, J.; Gray, T.J.; Connell, S.; Garonzik, S.; Ma, X.; et al. PAR4 (Protease- Activated Receptor 4) antagonism with BMS-986120 inhibits human ex vivo thrombus formation. Arterioscler. Thromb. Vasc. Biol. 2018, 38, 448–456. [Google Scholar] [CrossRef]
- Chang, C.H.; Chung, C.H.; Tu, Y.S.; Tsai, C.C.; Hsu, C.C.; Peng, H.C.; Tseng, Y.J.; Huang, T.F. Trowaglerix venom polypeptides as a novel antithrombotic agent by targeting immunoglobulin-like domains of glycoprotein VI in platelet. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1307–1314. [Google Scholar] [CrossRef] [PubMed]
- van Eeuwijk, J.M.; Stegner, D.; Lamb, D.J.; Kraft, P.; Beck, S.; Thielmann, I.; Kiefer, F.; Walzog, B.; Stoll, G.; Nieswandt, B. The Novel Oral Syk Inhibitor, Bl1002494, protects mice from arterial thrombosis and thromboinflamma- tory brain infarction. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Reilly, M.P.; Sinha, U.; André, P.; Taylor, S.M.; Pak, Y.; Deguzman, F.R.; Nanda, N.; Pandey, A.; Stolla, M.; Bergmeier, W.; et al. PRT-060318, a novel Syk inhibitor, prevents heparin-induced thrombocytopenia and thrombosis in a transgenic mouse model. Blood 2011, 117, 2241–2246. [Google Scholar] [CrossRef] [PubMed]
- Adili, R.; Tourdot, B.E.; Mast, K.; Yeung, J.; Freedman, J.C.; Green, A.; Luci, D.K.; Jadhav, A.; Simeonov, A.; Maloney, D.J.; et al. First selective 12-LOX inhibitor, ML355, impairs thrombus formation and vessel occlusion in vivo with minimal effects on hemostasis. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1828–1839. [Google Scholar] [CrossRef] [PubMed]
- Morrow, D.A.; Braunwald, E.; Bonaca, M.P.; Ameriso, S.F.; Dalby, A.J.; Fish, M.P.; Fox, K.A.A.; Lipka, L.J.; Liu, X.; Nicolau, J.C.; et al. Vorapaxar in the secondary prevention of atherothrombotic events. N. Engl. J. Med. 2012, 366, 1404–1413. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, M.L.; Bhatt, D.L.; Wiviott, S.D.; Goodman, S.G.; Fitzgerald, D.J.; Angiolillo, D.J.; Goto, S.; Montalescot, G.; Zeymer, U.; Aylward, P.E.; et al. Safety and tolerability of atopaxar in the treatment of patients with acute coronary syndromes: The lessons from antagonizing the cellular effects of Thrombin–Acute Coronary Syndromes Trial. Circulation. 2011, 123, 1843–1853. [Google Scholar] [CrossRef] [PubMed]
- Tricoci, P.; Huang, Z.; Held, C.; Moliterno, D.J.; Armstrong, P.W.; Van de Werf, F.; White, H.D.; Aylward, P.E.; Wallentin, L.; Chen, E.; et al. Thrombin-receptor antagonist vorapaxar in acute coronary syndromes. N. Engl. J. Med. 2012, 366, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Dütting, S.; Bender, M.; Nieswandt, B. Platelet GPVI: A target for antithrombotic therapy?! Trends Pharmacol. Sci. 2012, 33, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Andrews, R.K.; Arthur, J.F.; Gardiner, E.E. Targeting GPVI as a novel antithrombotic strategy. J. Blood Med. 2014, 5, 59–68. [Google Scholar] [CrossRef]
- Yeung, J.; Apopa, P.L.; Vesci, J.; Stolla, M.; Rai, G.; Simeonov, A.; Jadhav, A.; Fernandez-Perez, P.; Maloney, D.J.; Boutaud, O.; et al. 12-lipoxygenase activity plays an important role in PAR4 and GPVI- mediated platelet reactivity. Thromb. Haemost. 2013, 110, 569–581. [Google Scholar] [CrossRef]
- Yeung, J.; Tourdot, B.E.; Fernandez-Perez, P.; Vesci, J.; Ren, J.; Smyrniotis, C.J.; Luci, D.K.; Jadhav, A.; Simeonov, A.; Maloney, D.J.; et al. Platelet 12-LOX is essential for FcγRIIa- mediated platelet activation. Blood. 2014, 124, 2271–2279. [Google Scholar] [CrossRef] [PubMed]
- Tourdot, B.E.; Holinstat, M. Targeting 12-Lipoxygenase as a potential novel antiplatelet therapy. Trends Pharmacol. Sci. 2017, 38, 1006–1015. [Google Scholar] [CrossRef] [PubMed]


| Drug | Method of Delivery | Evolutionary Step | Benefits | Drawbacks | Comment |
|---|---|---|---|---|---|
| P2Y12 inhibitors. Thienopyridines. Clopidogrel bisulfate | Oral | FDA (17 November 1997) and EMEA (1998) approved | Clopidogrel is a widely used drug by cardiologists, emergency department physicians, family practice clinicians, and internists. |
| Patients with any loss of function allele will not effectively metabolize clopidogrel, leading to the inability to inhibit platelet activity. |
| P2Y12 inhibitors. Thienopyridines. Prasugrel | Oral | EMEA and FDA approved |
| This medication may carry a higher risk of bleeding when compared to clopidogrel. | Use cautiously in patients with stroke, TIA, aged 75 years, weighing <60 kg. |
| P2Y12 inhibitors. Thienopyridines. Elinogrel | Oral; iv | Discontinued in January 2012 by Novartis |
| Elinogrel therapy was associated with an increased incidence of dyspnea and incidence of elevated liver transaminases. | The potential use of this as an adjunct or alternative to standard antiplatelet therapy, such as aspirin and/or clopidogrel, was not pursued. |
| P2Y12 inhibitors. Cyclopentyl triazolo- Pyrimidine. Ticagrelor | Oral | EMEA and FDA approved | Exhibits a faster onset and offset of antiplatelet action compared to clopidogrel. | Possible side effects include dyspnea, bradycardia, and elevated uric acid levels. |
|
| P2Y12 inhibitors. ATP analogs. Cangrelor | Iv | EMEA and FDA approved | Exhibits a faster onset and offset of antiplatelet action compared to clopidogrel, prasugrel, and ticagrelor. |
| This is a promising adjunct treatment to fibrinolysis. |
| RCT Author/Reference | Ticagrelor Monotherapy (n = 8790) | Aspirin + P2Y12 Inhibitor (n = 8791) | Clopidogrel Monotherapy (n = 4110) | Aspirin + P2Y12 Inhibitor (n = 4144) | Comment Ticagrelor (HR, 0.89; 95% CI, 0.74–1.06; p for Non-Inferiority = 0.004) Clopidogrel (HR, 0.89; 95% CI, 0.74–1.06; p for Non-Inferiority = 0.004) |
|---|---|---|---|---|---|
| Franzone et al. [33] | 3753 (42.7) | 3756 (42.7) | 0 | 0 | Ticagrelor monotherapy after 1 month of DAPT non-inferior to conventional treatment in the prevention of ischemic events. Superior efficacy not demonstrated. Reduction in the risk of major bleeding in comparison to conventional treatment not observed. p = 0.98 |
| Hahn et al. [34] | 273 (3.1) | 263 (3.0) | 1122 (27.3) | 1143 (27.6) | P2Y12 inhibitor monotherapy after 3 months of DAPT resulted in non-inferior rates of MACCEs compared with prolonged DAPT among patients undergoing PCI. (p = 0.007) |
| Watanabe et al. [35] | 0 | 0 | 1496 (36.4) | 1507 (36.4) | 1 month clopidogrel monotherapy non-inferiority (p < 0.001) and superiority (p = 0.04) to DAPT. Clopidogrel monotherapy more effective than 12 months of DAPT in reducing the rate of a composite of cardiovascular and bleeding events. (HR, 0.26 [95% CI, 0.11–0.64]; p = 0.004 for superiority) |
| Watanabe et al. [36] | 0 | 0 | 1492 (36.3) | 1494 (36.0) | No net clinical benefit with clopidogrel monotherapy after 1 to 2 months of DAPT. Increase in cardiovascular events and decrease in bleeding events (HR, 1.50 [95% CI, 0.99–2.26]) vs. (HR, 0.46 [95% CI, 0.23–0.94]). |
| Kim et al. [37] | 1499 (17.1) | 1505 (17.1) | 0 | 0 | Ticagrelor monotherapy after 3 months of DAPT, modest but statistically significant reduction in a composite outcome of major bleeding and cardiovascular events compared with ticagrelor-based 12-month dual antiplatelet therapy (HR, 0.69 [95% CI, 0.45 to 1.06]; p = 0.09). |
| Mehran et al. [38] | 3265 (37.1) | 3267 (37.2) | 0 | 0 | After 3 months of DAPT, ticagrelor monotherapy was associated with a lower incidence of clinically relevant bleeding than ticagrelor plus aspirin (p < 0.001), with no higher risk of death, myocardial infarction, or stroke (p < 0.001 for non-inferiority) |
| RCT Acronym/Reference | Item | Highlights Findings | Comment |
|---|---|---|---|
| ASCET [39] | Platelet function study clopidogrel vs. ASA (n = 1001) | 73 death/MI/stroke events. No difference. | Not suitable for clinical events. |
| CADET [40] | Thrombogenicity study clopidogrel vs. ASA (n = 184) | No variables showed any differences. | Not suitable for clinical events |
| CAPRIE [41] | Clinical trial clopidogrel vs. ASA (n = 19,185) Recent MI subgroup (n = 6302) Stroke subgroup (n = 6431) PAD subgroup (n = 6452) | No differences were found in the recent (</= 35 day) MI subgroup or stroke subgroup. An additional post hoc subgroup was created, consisting of 2144 patients from the stroke and PAD subgroups who had a distant past history of MI, resulting in a larger 8446 patient subgroup with recent or distant MI. | The PANTHER meta-analysis utilized a post hoc subgroup of patients enrolled 28 years ago from subgroups of a trial that demonstrated no significant difference for patients with recent myocardial infarction or stroke. |
| DACAB [42] | SVG patency trial DAPT vs. ticagrelor vs. ASA (n = 500) | There were no significant differences in the occurrence of death, myocardial infarction, or stroke events between the ticagrelor and ASA subgroups, with a total of 13 events. | Not suitable for clinical events. |
| GLOBAL LEADERS [43] | Clinical trial 1 mo † DAPT/23 mo ticagrelor vs. 12 mo † DAPT/12 mo †† ASA (n = 15,968) | There was no difference in the 2-year endpoint or in the landmark analysis when comparing ticagrelor to ASA after 1 year. However, the GLASSY subgroup analysis of the 20 top enrolling sites (n = 7585) demonstrated a lower MI rate after 1 year with no significant difference in efficacy between the two treatments. | Post hoc subgroup (n = 7065) of a post hoc subgroup (GLASSY) of a negative trial was used for the PANTHER meta-analysis. |
| HOST-EXAM [44] | Clinical trial clopidogrel vs. ASA after 6–18 mo † DAPT for PCI N = 5438 | The study unequivocally demonstrates that the use of clopidogrel leads to a significantly lower incidence of adverse events, including all-cause death, myocardial infarction, stroke, readmission for acute coronary syndrome, and bleeding of type 3 or greater according to the Bleeding Academic Research Consortium (5.7% vs. 7.7%; HR: 0.73; 95% CI: 0.59–0.90; p = 0.0035). | No differences in death, MI, or stent thrombosis rates were observed. The reduced rates of stroke, readmission for ACS, and bleeding were attributed to the benefit. |
| TICAB [45] | Post-CABG clinical trial ticagrelor vs. ASA (n = 1859) | No differences were observed in the 117 death/MI/stroke events. | The study was stopped prematurely due to futility. Additionally, the ticagrelor group had a higher event rate. |
| First Author, Year (Ref. #) | N | Endpoints/Findings | p Value/I2 | HR/OR |
|---|---|---|---|---|
| Valgimigli et al. 2024 [5] | DAPT (12,403) P2Y12 inhibitor plus aspirin vs. SAPT (11,991) SAPT Ticagrelor 8292 Clopidogrel 3654 Prasugrel 45 | Non-inferiority of ticagrelor or clopidogrel monotherapy vs. DAPT on the composite of death, MI, or stroke Ticagrelor monotherapy non-inferior to DAPT for all-cause death, MI, or stroke and superior for major bleeding and NACE | p = 0.004 p < 0.001 | HR: 0.89; HR: 1.00 |
| Gragnano et al. 2023 [6] | SAPT Ticagrelor 4633 Clopidogrel 7545 Aspirin 12,147 | Composite of cardiovascular death, MI, and stroke Secondary endpoint major bleeding and NACE P2Y12 inhibitor monotherapy might be preferred over aspirin monotherapy for long-term secondary prevention | p = 0.012 p < 0.001 for IM p = 0.23 for major bleeding | HR: 0.88 HR: 0.77 for IM |
| Ando et al. 2022 [69] | 73,126 DAPT discontinued with aspirin or P2Y12-I vs. DAPT prolonged with aspirin vs. P2Y12-I | All-cause death; MI; major bleeding | - | HR: I.19 vs. 1.33 vs. 1.10 aspirin vs. P2Y12-I monotherapies |
| Lau et al. 2022 [49] | 20,915 short DAPT † 20,949 prolonged DAPT | Stent thrombosis (ST) and bleeding. DAPT ≤ 3 months followed by SAPT reduces bleeding and is not associated with an increase in ST. | p = 0.26 p < 0.0001 | HR: 1.17 HR: 0.65 |
| Giacoppo et al. 2021 [50] | DAPT (16,088); Short DAPT (16,057) vs. P2Y12I or Aspirin | ST and bleeding. 1–3 months of DAPT followed by P2Y12I SAPT lower major bleeding and similar stent thrombosis, all-cause death, myocardial infarction, and stroke compared with prolonged DAPT. | p = 0.289 p = 0.850 | HR: 1.19 HR: 0.63 |
| Charito et al. 2020 [46] | P2Y12I inhibitor (21,043) Aspirin (21,065) | MI and stroke P2Y12I monotherapy is associated with a risk reduction for MI and a comparable risk of stroke | I2 = 10.9% I2 = 34.5% | OR: 0.81 OR: 0.93 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nappi, F. P2Y12 Receptor Inhibitor for Antiaggregant Therapies: From Molecular Pathway to Clinical Application. Int. J. Mol. Sci. 2024, 25, 7575. https://doi.org/10.3390/ijms25147575
Nappi F. P2Y12 Receptor Inhibitor for Antiaggregant Therapies: From Molecular Pathway to Clinical Application. International Journal of Molecular Sciences. 2024; 25(14):7575. https://doi.org/10.3390/ijms25147575
Chicago/Turabian StyleNappi, Francesco. 2024. "P2Y12 Receptor Inhibitor for Antiaggregant Therapies: From Molecular Pathway to Clinical Application" International Journal of Molecular Sciences 25, no. 14: 7575. https://doi.org/10.3390/ijms25147575
APA StyleNappi, F. (2024). P2Y12 Receptor Inhibitor for Antiaggregant Therapies: From Molecular Pathway to Clinical Application. International Journal of Molecular Sciences, 25(14), 7575. https://doi.org/10.3390/ijms25147575






