Development and Characterization of Curcumin-Loaded TPGS/F127/P123 Polymeric Micelles as a Potential Therapy for Colorectal Cancer
Abstract
1. Introduction
2. Results
2.1. Development and Characterization of P123:F127:TPGS Polymeric Micelles
2.1.1. Particle Size, Polydispersity Index, and Zeta Potential
2.1.2. Quantification of Curcumin Using UV—vis Spectroscopy
2.1.3. Drug Loading (DL%) and Encapsulation Efficiency (EE%) Percentage Analysis by UV—vis Spectroscopy
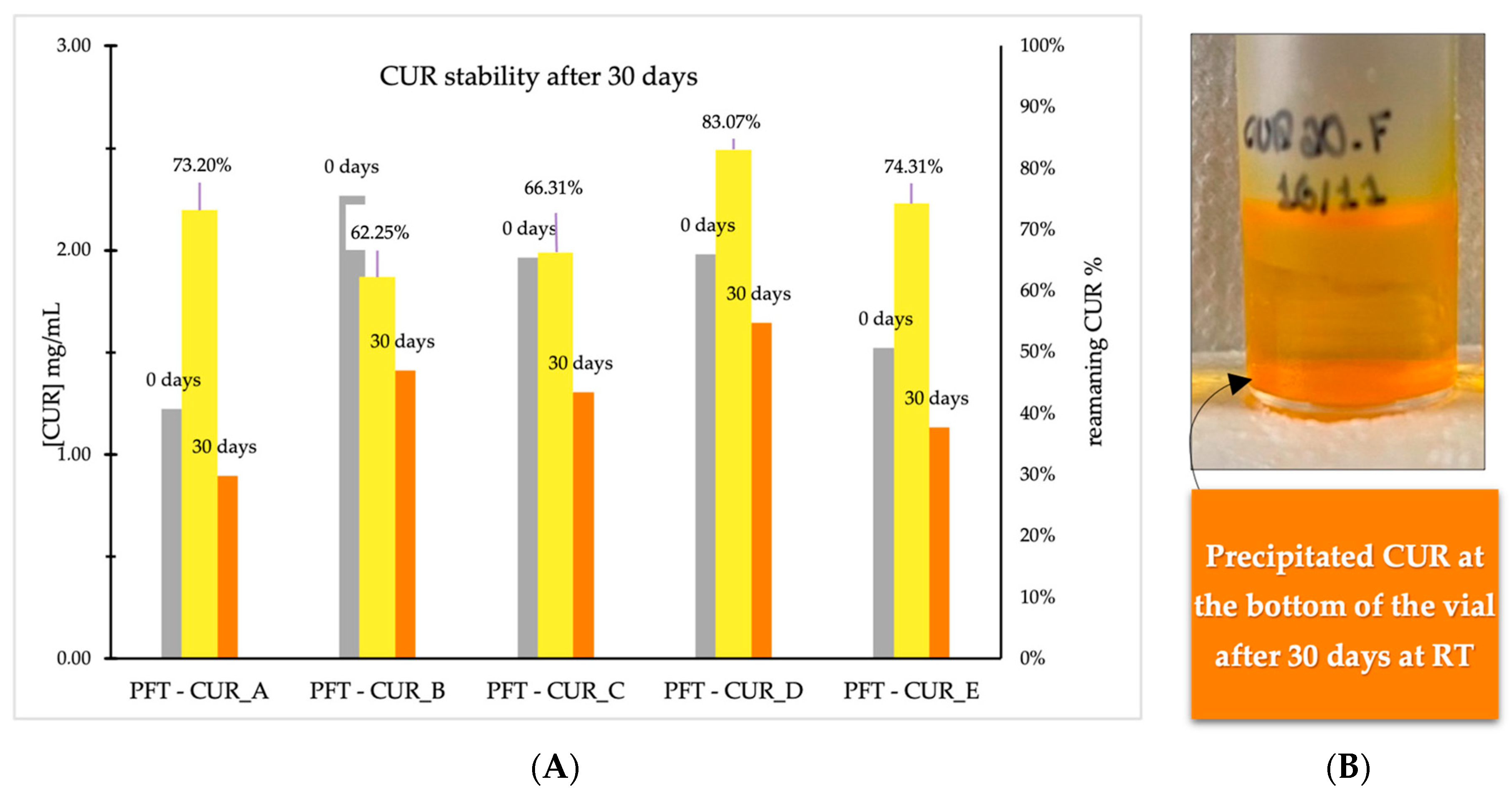
2.1.4. Storage Stability
2.2. Development and Characterization of Lyophilized P123:F127:TPGS Polymeric Micelles
2.2.1. Cryoprotectors (Pre-Lyophilization)
2.2.2. Re-Constitution (Post-Lyophilization)
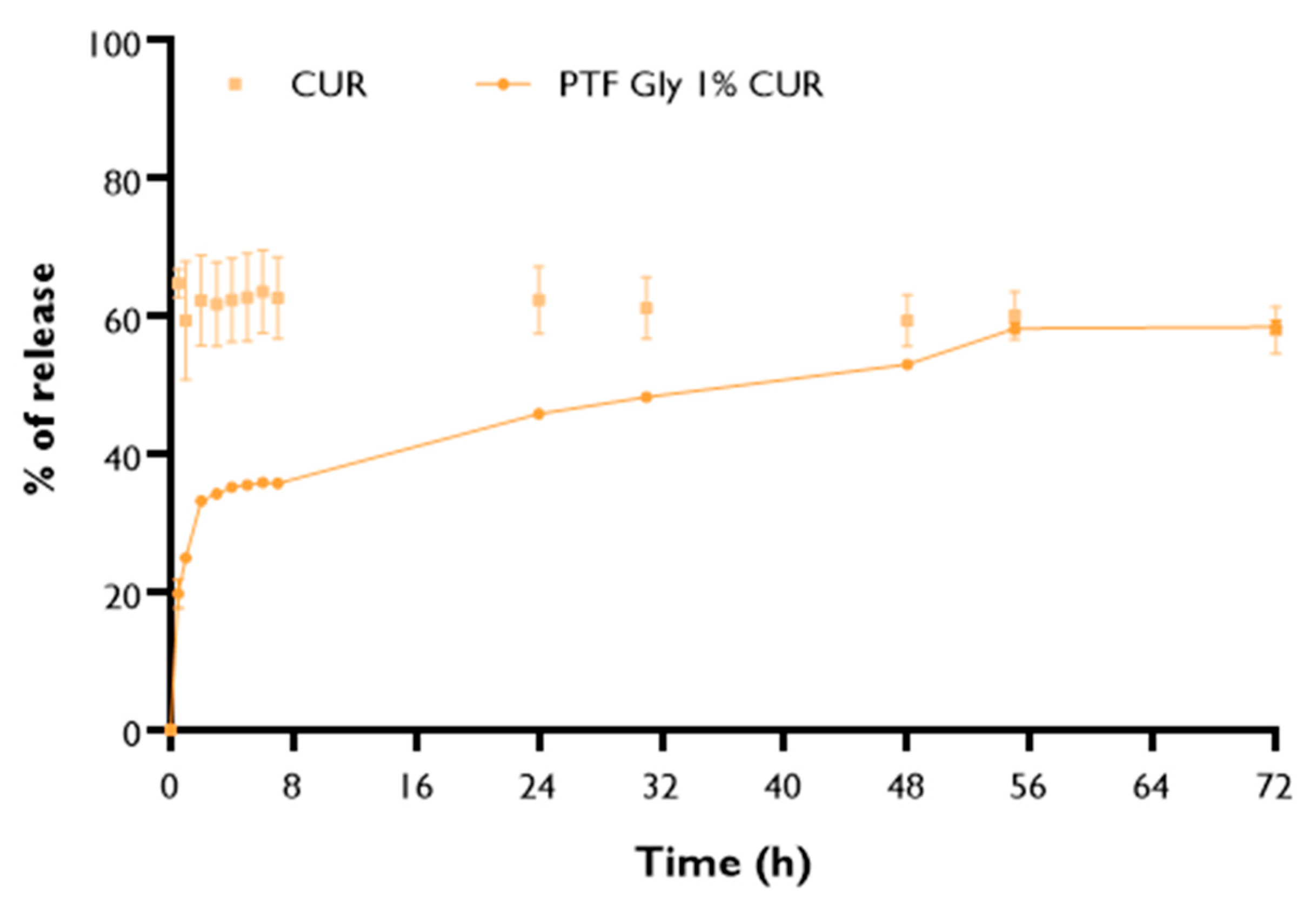
2.2.3. In Vitro Drug Release
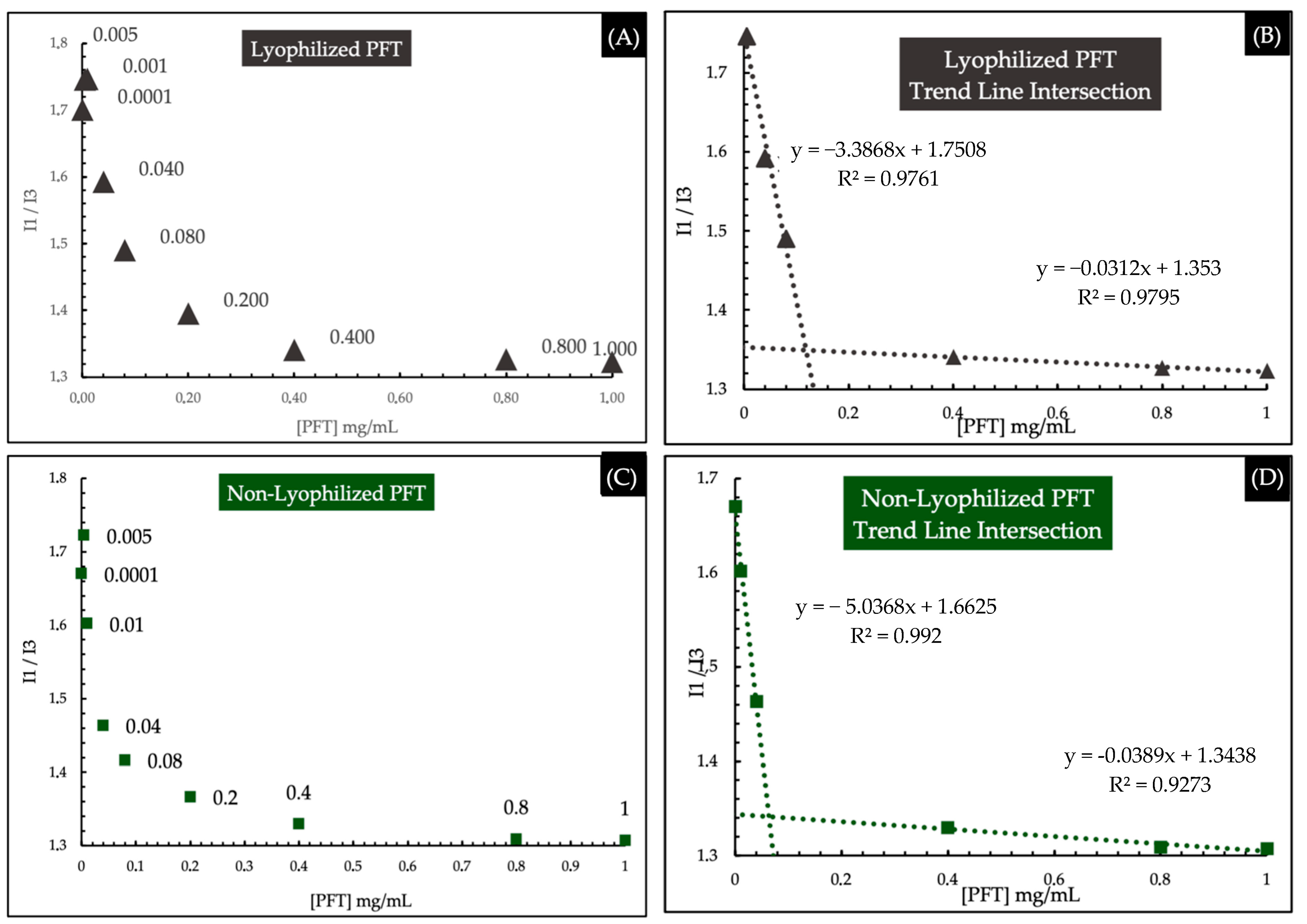
2.2.4. Critical Micelle Concentration (CMC)
2.2.5. Storage Stability
2.3. In Vitro Studies of the Optimized Nanosystems—Lyophilized Polymeric Micelles
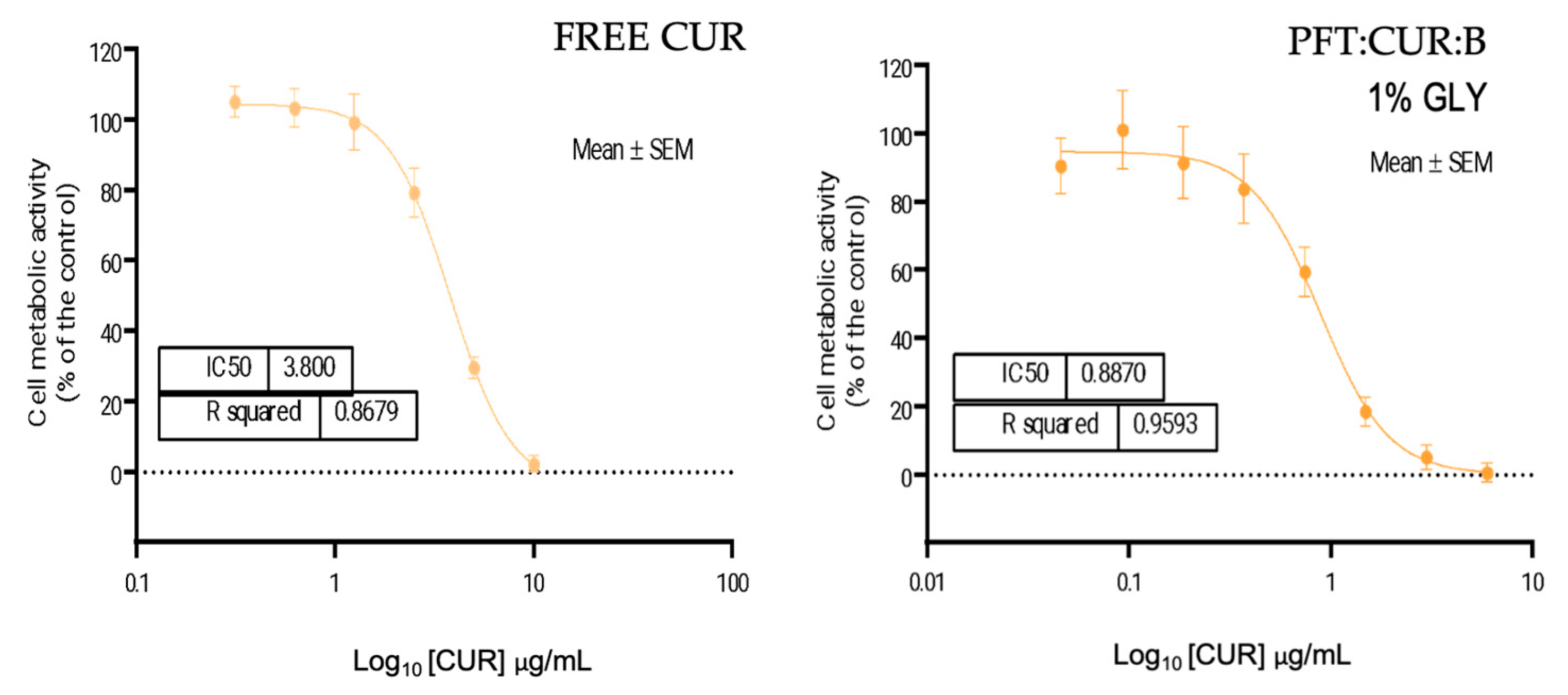
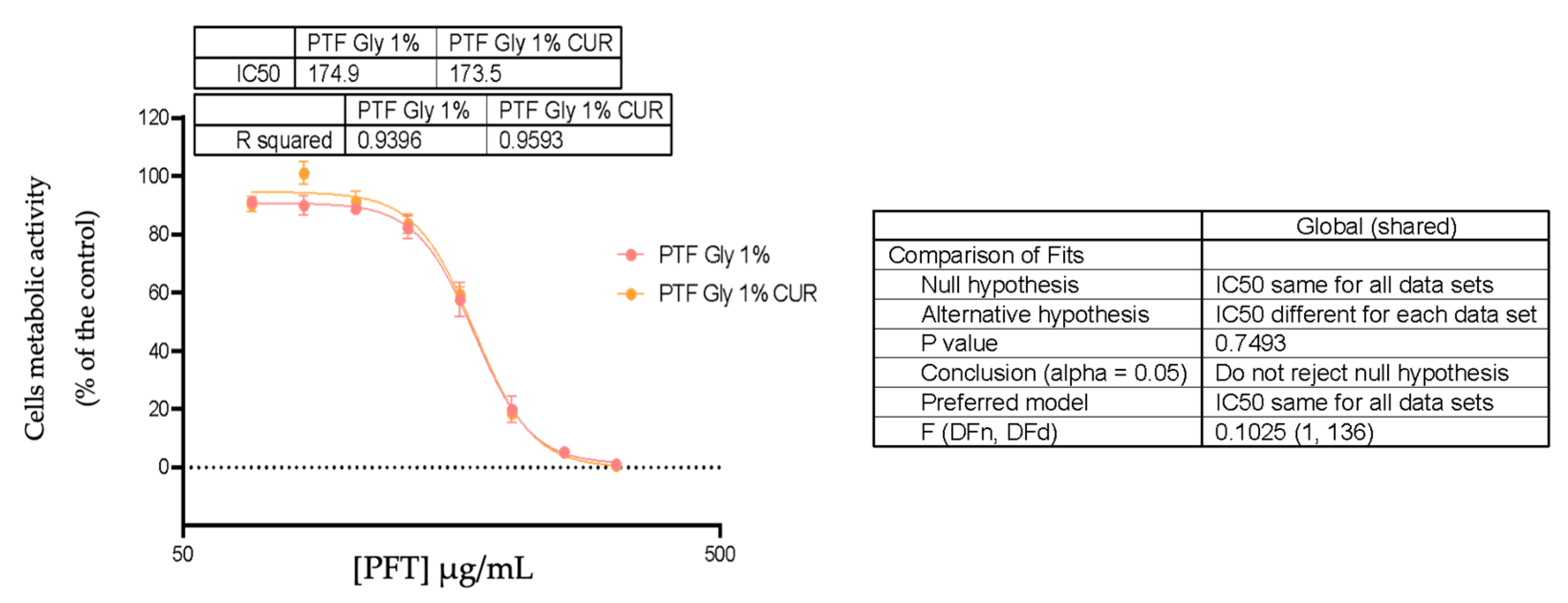
Cell Viability
3. Discussion
- Improving CUR entrapment by removing thermal processing in micelle production, more specifically, in the hydration step (Section 2.1.);
- Optimization of the lyophilization protocol by adding 1% GLY to the formulation, and post-lyophilization, reconstitute the lyophilized powder using a water/EtOH mixture (Section 2.2).
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Development of P123:F127:TPGS Polymeric Micelles
4.2.2. Physicochemical Characterization of P123: F127: TPGS Polymeric Micelles
Particle Size, Polydispersity Index, and Zeta Potential
Quantification of Curcumin by UV—vis Spectroscopy
Drug Loading (DL%) and Encapsulation Efficiency (EE%) Analysis by UV—vis Spectroscopy
Storage Stability
Lyophilization of P123:F127:TPGS Polymeric Micelles
In Vitro Drug Release
Critical Micelle Concentration (CMC)
4.2.3. In Vitro Studies of the Optimized Nanosystem
Cell Culture
Cell Viability
4.3. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Abbreviations
| ACN | Acetonitrile |
| CDs | Cyclodextrins |
| CMC | Critical micellar concentration |
| CRC | Colorectal cancer |
| CUR | Curcumin |
| DL % | Drug loading percentage |
| EE % | Encapsulation efficiency percentage |
| EtOH | Ethanol |
| GLY | Glycine |
| I1/I3 | Intensity ratio between first and third peak of fluorescence of pyrene |
| IC50 | Half maximal inhibitory concentration |
| Mw | Molecular weight |
| nano-DDS | Nanotechnology-based drug delivery system |
| NPs | Nanoparticles |
| PBS | Phosphate-buffered saline |
| PDI | Polydispersity index |
| PEO | Poly(ethylene oxide) |
| PFTs | P123:F127:TPGS polymeric micelles |
| PFT:CUR | CUR-loaded P123:F127:TPGS polymeric micelles |
| PFT:CUR:B_L 1%GLY | CUR-loaded P123:F127:TPGS lyophilized with 1% of GLY |
| PMs | Polymeric micelle |
| PPO | Poly(propylene oxide) |
| RC | Retention rate of curcumin |
| RT | Room temperature |
| SDS | Sodium Dodecyl Sulfate |
| TPGS | Vitamin E TPGS or D-ɑ-tocopherol polyethylene glycol succinate |
| UV—vis | Ultraviolet–visible spectroscopy |
| ZP | Zeta potential |
References
- Baidoun, F.; Elshiwy, K.; Elkeraie, Y.; Merjaneh, Z.; Khoudari, G.; Sarmini, M.T.; Gad, M.; Al-Husseini, M.; Saad, A. Colorectal Cancer Epidemiology: Recent Trends and Impact on Outcomes. Curr. Drug Targets 2020, 22, 998–1009. [Google Scholar] [CrossRef]
- Morgan, E.; Arnold, M.; Gini, A.; Lorenzoni, V.; Cabasag, C.J.; Laversanne, M.; Vignat, J.; Ferlay, J.; Murphy, N.; Bray, F. Global Burden of Colorectal Cancer in 2020 and 2040: Incidence and Mortality Estimates from GLOBOCAN. Gut 2023, 72, 338–344. [Google Scholar] [CrossRef]
- Shin, A.E.; Giancotti, F.G.; Rustgi, A.K. Metastatic Colorectal Cancer: Mechanisms and Emerging Therapeutics. Trends Pharmacol. Sci. 2023, 44, 222–236. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.B.; Majumdar, A.P. Synergistic Role of Curcumin With Current Therapeutics in Colorectal Cancer: Minireview. Physiol. Behav. 2009, 61, 842–846. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). IARC Handbooks Colorectal Cancer; International Agency for Research on Cancer (IARC): Lyon, France, 2018; Volume 17, ISBN 9789283230229.
- Patra, J.K.; Shukla, A.C.; Das, G. Advances in Pharmaceutical Biotechnology: Recent Progress and Future Applications; Springer: Berlin/Heidelberg, Germany, 2020; ISBN 9789811521959. [Google Scholar]
- Li, L.; Ahmed, B.; Mehta, K.; Kurzrock, R. Liposomal Curcumin with and without Oxaliplatin: Effects on Cell Growth, Apoptosis, and Angiogenesis in Colorectal Cancer. Mol. Cancer Ther. 2007, 6, 1276–1282. [Google Scholar] [CrossRef] [PubMed]
- Weng, W.; Goel, A. Curcumin and Colorectal Cancer: An Update and Current Perspective on This Natural Medicine. Semin. Cancer Biol. 2022, 80, 73–86. [Google Scholar] [CrossRef] [PubMed]
- Gunther, J.R.; Chadha, A.S.; Guha, S.; Raju, G.S.; Maru, D.M.; Munsell, M.F.; Jiang, Y.; Yang, P.; Felix, E.; Clemons, M.; et al. A Phase II Randomized Double Blinded Trial Evaluating the Efficacy of Curcumin with Pre-Operative Chemoradiation for Rectal Cancer. J. Gastrointest. Oncol. 2022, 13, 2938–2950. [Google Scholar] [CrossRef] [PubMed]
- Riesco-Martinez, M.C.; Modrego, A.; Espinosa-Olarte, P.; La Salvia, A.; Garcia-Carbonero, R. Perioperative Chemotherapy for Liver Metastasis of Colorectal Cancer: Lessons Learned and Future Perspectives. Curr. Treat. Opt. Oncol. 2022, 23, 1320–1337. [Google Scholar] [CrossRef] [PubMed]
- Ruiz de Porras, V.; Layos, L.; Martínez-Balibrea, E. Curcumin: A Therapeutic Strategy for Colorectal Cancer? Semin. Cancer Biol. 2021, 73, 321–330. [Google Scholar] [CrossRef]
- Urošević, M.; Nikolić, L.; Gajić, I.; Nikolić, V.; Dinić, A.; Miljković, V. Curcumin: Biological Activities and Modern Pharmaceutical Forms. Antibiotics 2022, 11, 135. [Google Scholar] [CrossRef]
- Galen, E.V.; Kroes, B.; Llorente, G.G.; Ensink, E.; Van Galen, E.; Kroes, B. Assessment Report on Curcuma longa L. (C. Domestica valeton), Rhizoma. Eur. Med. Agency 2017, 8, 1–34. [Google Scholar]
- Mahran, R.I.; Hagras, M.M.; Sun, D.; Brenner, D.E. Bringing Curcumin to the Clinic in Cancer Prevention: A Review of Strategies to Enhance Bioavailability and Efficacy. AAPS J. 2017, 19, 54–81. [Google Scholar] [CrossRef] [PubMed]
- Hafez Ghoran, S.; Calcaterra, A.; Abbasi, M.; Taktaz, F.; Nieselt, K.; Babaei, E. Curcumin-Based Nanoformulations: A Promising Adjuvant towards Cancer Treatment. Molecules 2022, 27, 5236. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.-H.; Lu, P.W.-A.; Lu, E.W.-H.; Lin, C.-W.; Yang, S.-F. Curcumin and Its Analogs and Carriers: Potential Therapeutic Strategies for Human Osteosarcoma. Int. J. Biol. Sci. 2023, 19, 1241–1265. [Google Scholar] [CrossRef] [PubMed]
- Shuvalov, O.; Kirdeeva, Y.; Daks, A.; Fedorova, O.; Parfenyev, S.; Simon, H.-U.; Barlev, N.A. Phytochemicals Target Multiple Metabolic Pathways in Cancer. Antioxidants 2023, 12, 2012. [Google Scholar] [CrossRef] [PubMed]
- Park, J. Anti-Carcinogenic Properties of Curcumin on Colorectal Cancer. World J. Gastrointest. Oncol. 2010, 2, 169. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Hu, X.; Zuo, X.; Wang, M. Chemopreventive Effects of Some Popular Phytochemicals on Human Colon Cancer: A Review. Food Funct. 2018, 9, 4548–4568. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Aggarwal, B.B. Therapeutic Roles of Curcumin: Lessons Learned from Clinical Trials. AAPS J. 2013, 15, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.J.; Mukhtar, H. Curcumin for Chemoprevention of Colon Cancer. Cancer Lett. 2007, 255, 170–181. [Google Scholar] [CrossRef]
- Pricci, M.; Girardi, B.; Giorgio, F.; Losurdo, G.; Ierardi, E.; Di Leo, A. Curcumin and Colorectal Cancer: From Basic to Clinical Evidences. Int. J. Mol. Sci. 2020, 21, 2364. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Joshi, S.; Semwal, D.; Bisht, A.; Paliwal, S.; Dwivedi, J.; Sharma, S. Curcumin: An Insight into Molecular Pathways Involved in Anticancer Activity. Mini Rev. Med. Chem. 2021, 21, 2420–2457. [Google Scholar] [CrossRef] [PubMed]
- Zagami, R.; Barattucci, A.; Monsù Scolaro, L.; Viale, M.; Raffaini, G.; Maria Bonaccorsi, P.; Mazzaglia, A. Curcumin/Amphiphilic Cyclodextrin Nanoassemblies: Theoretical and Spectroscopic Studies to Address Their Debut in Anticancer Therapy. J. Mol. Liq. 2023, 389, 122841. [Google Scholar] [CrossRef]
- Kasi, P.D.; Tamilselvam, R.; Skalicka-Woźniak, K.; Nabavi, S.F.; Daglia, M.; Bishayee, A.; Pazoki-Toroudi, H.; Nabavi, S.M. Molecular Targets of Curcumin for Cancer Therapy: An Updated Review. Tumor Biol. 2016, 37, 13017–13028. [Google Scholar] [CrossRef] [PubMed]
- Sardou, H.S.; Nazari, S.E.; Abbaspour, M.; Akhgari, A.; Sheikh, A.; Kesharwani, P.; Sahebkar, A. Nano-Curcumin Formulations for Targeted Therapy of Colorectal Cancer. J. Drug Deliv. Sci. Technol. 2023, 88, 104943. [Google Scholar] [CrossRef]
- Schneider, C.; Gordon, O.N.; Edwards, R.L.; Luis, P.B. Degradation of Curcumin: From Mechanism to Biological Implications. J. Agric. Food Chem. 2015, 63, 7606–7614. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.E.; Ngai, S.C.; Chan, K.G.; Lee, L.H.; Goh, B.H.; Chuah, L.H. Curcumin Nanoformulations for Colorectal Cancer: A Review. Front. Pharmacol. 2019, 10, 152. [Google Scholar] [CrossRef] [PubMed]
- Mundekkad, D.; Cho, W.C. Applications of Curcumin and Its Nanoforms in the Treatment of Cancer. Pharmaceutics 2023, 15, 2223. [Google Scholar] [CrossRef] [PubMed]
- Beyene, A.M.; Moniruzzaman, M.; Karthikeyan, A.; Min, T. Curcumin Nanoformulations with Metal Oxide Nanomaterials for Biomedical Applications. Nanomaterials 2021, 11, 460. [Google Scholar] [CrossRef]
- Tefas, L.R.; Sylvester, B.; Tomuta, I.; Sesarman, A.; Licarete, E.; Banciu, M.; Porfire, A. Development of Antiproliferative Long-Circulating Liposomes Co-Encapsulating Doxorubicin and Curcumin, through the Use of a Quality-by-Design Approach. Drug Des. Devel Ther. 2017, 11, 1605–1621. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.T.; Rahman, M.H.; Akter, R.; Behl, T.; Kaushik, D.; Mittal, V.; Pandey, P.; Akhtar, M.F.; Saleem, A.; Albadrani, G.M.; et al. Potential Role of Curcumin and Its Nanoformulations to Treat Various Types of Cancers. Biomolecules 2021, 11, 392. [Google Scholar] [CrossRef] [PubMed]
- de Freitas, C.F.; Kimura, E.; Rubira, A.F.; Muniz, E.C. Curcumin and Silver Nanoparticles Carried out from Polysaccharide-Based Hydrogels Improved the Photodynamic Properties of Curcumin through Metal-Enhanced Singlet Oxygen Effect. Mater. Sci. Eng. C 2020, 112, 110853. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.; Li, Y.; Deng, X.; Wang, X.; Cui, X.; Lu, J.; Pan, Z.; Wu, Y. Self-Assembled Fluorescent Hybrid Nanoparticles-Mediated Collaborative LncRNA CCAT1 Silencing and Curcumin Delivery for Synchronous Colorectal Cancer Theranostics. J. Nanobiotechnol. 2021, 19, 238. [Google Scholar] [CrossRef] [PubMed]
- Alexandridis, P. Poly(Ethylene Oxide)/Poly(Propylene Oxide) Block Copolymer Surfactants. Curr. Opin. Colloid Interface Sci. 1997, 2, 478–489. [Google Scholar] [CrossRef]
- Bodratti, A.M.; Alexandridis, P. Formulation of Poloxamers for Drug Delivery. J. Funct. Biomater. 2018, 9, 11. [Google Scholar] [CrossRef] [PubMed]
- Simões, S.M.N.; Figueiras, A.R.; Veiga, F.; Concheiro, A.; Alvarez-Lorenzo, C. Polymeric Micelles for Oral Drug Administration Enabling Locoregional and Systemic Treatments. Expert Opin. Drug Deliv. 2015, 12, 297–318. [Google Scholar] [CrossRef]
- Zarrintaj, P.; Ramsey, J.D.; Samadi, A.; Atoufi, Z.; Yazdi, M.K.; Ganjali, M.R.; Amirabad, L.M.; Zangene, E.; Farokhi, M.; Formela, K.; et al. Poloxamer: A Versatile Tri-Block Copolymer for Biomedical Applications. Acta Biomater. 2020, 110, 37–67. [Google Scholar] [CrossRef] [PubMed]
- Herzberger, J.; Niederer, K.; Pohlit, H.; Seiwert, J.; Worm, M.; Wurm, F.R.; Frey, H. Polymerization of Ethylene Oxide, Propylene Oxide, and Other Alkylene Oxides: Synthesis, Novel Polymer Architectures, and Bioconjugation. Chem. Rev. 2016, 116, 2170–2243. [Google Scholar] [CrossRef] [PubMed]
- Wei, Z.; Hao, J.; Yuan, S.; Li, Y.; Juan, W.; Sha, X.; Fang, X. Paclitaxel-Loaded Pluronic P123/F127 Mixed Polymeric Micelles: Formulation, Optimization and in Vitro Characterization. Int. J. Pharm. 2009, 376, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, C.; Sahoo, S.K. The in Vitro Stability and in Vivo Pharmacokinetics of Curcumin Prepared as an Aqueous Nanoparticulate Formulation. Biomaterials 2010, 31, 6597–6611. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Qiu, H.; Yin, S.; Wang, H.; Li, Y. Polymeric Drug Delivery System Based on Pluronics for Cancer Treatment. Molecules 2021, 26, 3610. [Google Scholar] [CrossRef]
- Xie, Y.J.; Wang, Q.L.; Adu-Frimpong, M.; Liu, J.; Zhang, K.Y.; Xu, X.M.; Yu, J.N. Preparation and Evaluation of Isoliquiritigenin-Loaded F127/P123 Polymeric Micelles. Drug Dev. Ind. Pharm. 2019, 45, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Russo, A.; Pellosi, D.S.; Pagliara, V.; Milone, M.R.; Pucci, B.; Caetano, W.; Hioka, N.; Budillon, A.; Ungaro, F.; Russo, G.; et al. Biotin-Targeted Pluronic® P123/F127 Mixed Micelles Delivering Niclosamide: A Repositioning Strategy to Treat Drug-Resistant Lung Cancer Cells. Int. J. Pharm. 2016, 511, 127–139. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Wu, T.; Qi, Y.; Zhang, Z. Recent Advances in the Application of Vitamin E TPGS for Drug Delivery. Theranostics 2018, 8, 464–485. [Google Scholar] [CrossRef] [PubMed]
- Constantinou, C.; Hyatt, J.A.; Vraka, P.S.; Papas, A.; Papas, K.A.; Neophytou, C.; Hadjivassiliou, V.; Constantinou, A.I. Induction of Caspase-Independent Programmed Cell Death by Vitamin E Natural Homologs and Synthetic Derivatives. Nutr. Cancer 2009, 61, 864–874. [Google Scholar] [CrossRef] [PubMed]
- Saxena, V.; Hussain, M.D. Polymeric Mixed Micelles for Delivery of Curcumin to Multidrug Resistant Ovarian Cancer. J. Biomed. Nanotechnol. 2013, 9, 1146–1154. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, K.U.N.; Zhu, Y.; Wang, D.; Shao, Y.; Zhang, J.U.N. Curcumin Inhibits Hypoxia-Induced Proliferation and Invasion of MG-63 Osteosarcoma Cells via Downregulating Notch1. Mol. Med. Rep. 2017, 15, 1747–1752. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, Q.; Yang, L.; Xia, X.; Zhu, R.; Chen, S.; Wang, M.; Cheng, L.; Wu, X.; Wang, S. Curcumin-Loaded TPGS/F127/P123 Mixed Polymeric Micelles for Cervical Cancer Therapy: Formulation, Characterization, and in Vitro and in Vivo Evaluation. J. Biomed. Nanotechnol. 2017, 13, 1631–1646. [Google Scholar] [CrossRef]
- Fang, C.; Shi, B.; Pei, Y.Y.; Hong, M.H.; Wu, J.; Chen, H.Z. In Vivo Tumor Targeting of Tumor Necrosis Factor-α-Loaded Stealth Nanoparticles: Effect of MePEG Molecular Weight and Particle Size. Eur. J. Pharm. Sci. 2006, 27, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Fonge, H.; Hoang, B.; Reilly, R.M.; Allen, C. The Effects of Particle Size and Molecular Targeting on the Intratumoral and Subcellular Distribution of Polymeric Nanoparticles. Mol. Pharm. 2010, 7, 1195–1208. [Google Scholar] [CrossRef]
- Guo, D.; Ji, X.; Luo, J. Rational Nanocarrier Design towards Clinical Translation of Cancer Nanotherapy. Biomed. Mater. 2021, 16, 032005. [Google Scholar] [CrossRef]
- Ernsting, M.J.; Murakami, M.; Roy, A.; Li, S.-D. Factors Controlling the Pharmacokinetics, Biodistribution and Intratumoral Penetration of Nanoparticles. J. Control Releaseournal Control Release 2013, 23, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Chehelgerdi, M.; Chehelgerdi, M.; Allela, O.Q.B.; Pecho, R.D.C.; Jayasankar, N.; Rao, D.P.; Thamaraikani, T.; Vasanthan, M.; Viktor, P.; Lakshmaiya, N.; et al. Progressing Nanotechnology to Improve Targeted Cancer Treatment: Overcoming Hurdles in Its Clinical Implementation. Mol. Cancer 2023, 22, 169. [Google Scholar] [CrossRef]
- Nakashima, H.; Takatsu, T.; Imai, R. Radiation-Induced Osteosarcoma in the Pubic Bone after Proton Radiotherapy for Prostate Cancer: A Case Report. J. Rural Med. Case 2022, 17, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Wulandari, A.; Sunarti, T.C.; Fahma, F.; Enomae, T. The Potential of Bioactives as Biosensors for Detection of PH. IOP Conf. Ser. Earth Environ. Sci. 2020, 460, 012034. [Google Scholar] [CrossRef]
- Pourreza, N.; Golmohammadi, H. Application of Curcumin Nanoparticles in a Lab-on-Paper Device as a Simple and Green PH Probe. Talanta 2015, 131, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Chuacharoen, T.; Sabliov, C.M. Comparative Effects of Curcumin When Delivered in a Nanoemulsion or Nanoparticle Form for Food Applications: Study on Stability and Lipid Oxidation Inhibition. LWt 2019, 113, 108319. [Google Scholar] [CrossRef]
- Singla, P.; Singh, O.; Sharma, S.; Betlem, K.; Aswal, V.K.; Peeters, M.; Mahajan, R.K. Temperature-Dependent Solubilization of the Hydrophobic Antiepileptic Drug Lamotrigine in Different Pluronic Micelles—A Spectroscopic, Heat Transfer Method, Small-Angle Neutron Scattering, Dynamic Light Scattering, and in Vitro Release Study. ACS Omega 2019, 4, 11251–11262. [Google Scholar] [CrossRef] [PubMed]
- Guttoff, M.; Saberi, A.H.; Mcclements, D.J. Formation of Vitamin D Nanoemulsion-Based Delivery Systems by Spontaneous Emulsification: Factors Affecting Particle Size and Stability. Food Chem. 2015, 171, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Li, C.; Wang, Z.; Zhang, W.; Liu, J. Factors Affecting the Stability of Drug-Loaded Polymeric Micelles and Strategies for Improvement. J. Nanoparticle Res. 2016, 18, 275. [Google Scholar] [CrossRef]
- Naksuriya, O.; Vansteenbergen, M.J.; Torano, J.S.; Okonogi, S.; Hennink, W.E. A Kinetic Degradation Study of Curcumin in Its Free Form and Loaded in Polymeric Micelles. AAPS J. 2016, 18, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Ndong Ntoutoume, G.M.A.; Granet, R.; Mbakidi, J.P.; Brégier, F.; Léger, D.Y.; Fidanzi-Dugas, C.; Lequart, V.; Joly, N.; Liagre, B.; Chaleix, V.; et al. Development of Curcumin-Cyclodextrin/Cellulose Nanocrystals Complexes: New Anticancer Drug Delivery Systems. Bioorganic Med. Chem. Lett. 2016, 26, 941–945. [Google Scholar] [CrossRef] [PubMed]
- Dhule, S.S.; Penfornis, P.; Frazier, T.; Walker, R.; Feldman, J.; Tan, G.; He, J.; Alb, A.; John, V.; Pochampally, R. Curcumin-Loaded γ-Cyclodextrin Liposomal Nanoparticles as Delivery Vehicles for Osteosarcoma. Nanomed. Nanotechnol. Biol. Med. 2012, 8, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H.; Greish, K.; Fang, J. The EPR Effect and Polymeric Drugs: A Paradigm Shift for Cancer Chemotherapy in the 21st Century. Adv. Polym. Sci. 2006, 193, 103–121. [Google Scholar] [CrossRef]
- Owen, S.C.; Chan, D.P.Y.; Shoichet, M.S. Polymeric Micelle Stability. Nano Today 2012, 7, 53–65. [Google Scholar] [CrossRef]
- Degobert, G.; Aydin, D. Lyophilization of Nanocapsules: Instability Sources, Formulation and Process Parameters. Pharmaceutics 2021, 13, 1112. [Google Scholar] [CrossRef] [PubMed]
- Feng, S.; Zhang, Z.; Almotairy, A.; Repka, M.A. Development and Evaluation of Polymeric Mixed Micelles Prepared Using Hot-Melt Extrusion for Extended Delivery of Poorly Water-Soluble Drugs. J. Pharm. Sci. 2023, 112, 2869–2878. [Google Scholar] [CrossRef] [PubMed]
- Suksiriworapong, J.; Rungvimolsin, T.; A-Gomol, A.; Junyaprasert, V.B.; Chantasart, D. Development and Characterization of Lyophilized Diazepam-Loaded Polymeric Micelles. AAPS PharmSciTech 2014, 15, 52–64. [Google Scholar] [CrossRef]
- Sajomsang, W.; Gonil, P.; Saesoo, S.; Ruktanonchai, U.R.; Srinuanchai, W.; Puttipipatkhachorn, S. Synthesis and Anticervical Cancer Activity of Novel PH Responsive Micelles for Oral Curcumin Delivery. Int. J. Pharm. 2014, 477, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Wang, P.; Yang, Z.; Li, R.; Xu, X.; Shen, J. Dual Improvement in Curcumin Encapsulation Efficiency and Lyophilized Complex Dispersibility through Ultrasound Regulation of Curcumin–Protein Assembly. Ultrason. Sonochem. 2022, 90, 106188. [Google Scholar] [CrossRef] [PubMed]
- Fonte, P.; Reis, S.; Sarmento, B. Facts and Evidences on the Lyophilization of Polymeric Nanoparticles for Drug Delivery. J. Control Release 2016, 225, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Bruschi, M.L. Mathematical Models of Drug Release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Elsevier: Amsterdam, The Netherlands, 2015; pp. 63–86. ISBN 9780081000922. [Google Scholar]
- Yang, X.; Li, Z.; Wang, N.; Li, L.; Song, L.; He, T.; Sun, L.; Wang, Z.; Wu, Q.; Luo, N.; et al. Curcumin-Encapsulated Polymeric Micelles Suppress the Development of Colon Cancer in Vitro and in Vivo. Sci. Rep. 2015, 5, 10322. [Google Scholar] [CrossRef] [PubMed]
- Basso, J.; Mendes, M.; Silva, J.; Sereno, J.; Cova, T.; Oliveira, R.; Fortuna, A.; Castelo-Branco, M.; Falcão, A.; Sousa, J.; et al. Peptide-Lipid Nanoconstructs Act Site-Specifically towards Glioblastoma Growth Impairment. Eur. J. Pharm. Biopharm. 2020, 155, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Figueiras, A.; Domingues, C.; Jarak, I.; Santos, A.I.; Parra, A.; Pais, A.; Alvarez-Lorenzo, C.; Concheiro, A.; Kabanov, A.; Cabral, H.; et al. New Advances in Biomedical Application of Polymeric Micelles. Pharmaceutics 2022, 14, 1700. [Google Scholar] [CrossRef] [PubMed]
- Mabrouk, M.M.; Hamed, N.A.; Mansour, F.R. Spectroscopic Methods for Determination of Critical Micelle Concentrations of Surfactants; a Comprehensive Review. Appl. Spectrosc. Rev. 2023, 58, 206–234. [Google Scholar] [CrossRef]
- Kim, S.; Shi, Y.; Kim, J.Y.; Park, K.; Cheng, J.X. Overcoming the Barriers in Micellar Drug Delivery: Loading Efficiency, in Vivo Stability, and Micelle-Cell Interaction. Expert Opin. Drug Deliv. 2010, 7, 49–62. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.H.; Loo, C.Y.; Young, P.M.; Traini, D.; Mason, R.S.; Rohanizadeh, R. Recent Advances in Curcumin Nanoformulation for Cancer Therapy. Expert Opin. Drug Deliv. 2014, 11, 1183–1201. [Google Scholar] [CrossRef] [PubMed]
- Bapat, R.A.; Bedia, S.V.; Bedia, A.S.; Yang, H.J.; Dharmadhikari, S.; Abdulla, A.M.; Chaubal, T.V.; Bapat, P.R.; Abullais, S.S.; Wahab, S.; et al. Current Appraises of Therapeutic Applications of Nanocurcumin: A Novel Drug Delivery Approach for Biomaterials in Dentistry. Environ. Res. 2023, 238, 116971. [Google Scholar] [CrossRef] [PubMed]
- Trenkenschuh, E.; Friess, W. Freeze-Drying of Nanoparticles: How to Overcome Colloidal Instability by Formulation and Process Optimization. Eur. J. Pharm. Biopharm. 2021, 165, 345–360. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Costa, A.P.; Xu, X.; Lee, S.L.; Cruz, C.N.; Bao, Q.; Burgess, D.J. Formulation and Characterization of Curcumin Loaded Polymeric Micelles Produced via Continuous Processing. Int. J. Pharm. 2020, 583, 119340. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.F.; Wan, K.Y.; Cheng, K.K.; Wong, K.W.; Sun, C.C.; Baum, L.; Chow, A.H.L. Development of Highly Stabilized Curcumin Nanoparticles by Flash Nanoprecipitation and Lyophilization. Eur. J. Pharm. Biopharm. 2015, 94, 436–449. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Chiang, Y.T.; Hsu, N.Y.; Yang, C.Y.; Lo, C.L.; Ku, C.A. Vitamin E Containing Polymer Micelles for Reducing Normal Cell Cytotoxicity and Enhancing Chemotherapy Efficacy. Acta Biomater. 2015, 24, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, J.; Chen, J.; Zeng, Y.; Zhu, Z.; Wan, Y. Fabrication of Curcumin-Modified TiO2 Nanoarrays via Cyclodextrin Based Polymer Functional Coatings for Osteosarcoma Therapy. Adv. Healthc. Mater. 2019, 8, e1901031. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.C.; O’Reilly Beringhs, A.; Kim, R.; Zhang, W.; Patel, S.M.; Bogner, R.H.; Lu, X. Impact of Formulation on the Quality and Stability of Freeze-Dried Nanoparticles. Eur. J. Pharm. Biopharm. 2021, 169, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, R.; Domingues, C.; Jarak, I.; Silva, I.; Correias, D.; Valoppi, M.; Veiga, F.; Figueiras, A.R. Development and Characterization of Curcumin-Loaded TPGS/F127/P123 Polymeric Micelles as a Potential Therapy for Osteosarcoma. In Acta Farmacêutica Portuguesa X; Ordem dos Farmacêuticos: Lisboa, Portugal, 2023; pp. 67–68. [Google Scholar]
- Ghezzi, M.; Pescina, S.; Padula, C.; Santi, P.; Del Favero, E.; Cantù, L.; Nicoli, S. Polymeric Micelles in Drug Delivery: An Insight of the Techniques for Their Characterization and Assessment in Biorelevant Conditions. J. Control Release 2021, 332, 312–336. [Google Scholar] [CrossRef]
- Vauthier, C.; Ponchel, G. Polymer Nanoparticles for Nanomedicines; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 9783319414195. [Google Scholar]
- European Directorate for the Quality of Medicines. European Directorate for the Quality of Medicines European Pharmacopeia—2.9.3 Dissolution Test for Solid Dosage Forms. In European Pharmacopoeia; European Directorate for the Quality of Medicines: Strasbourg, France, 2014; pp. 264–288. [Google Scholar]
- Aguiar, J.; Carpena, P.; Molina-Bolívar, J.A.; Carnero Ruiz, C. On the Determination of the Critical Micelle Concentration by the Pyrene 1:3 Ratio Method. J. Colloid Interface Sci. 2003, 258, 116–122. [Google Scholar] [CrossRef]
- ATCC Product Sheet LoVo (ATCC CCL-229). Available online: https://www.atcc.org/products/ccl-229 (accessed on 1 July 2024).
- Pierce, B. The Thermo Scientific AlamarBlue Cell Viability Assay Reagent. U.S. Patent 6,403,378, 9 March 1999. [Google Scholar]
- Mendes, M.; Branco, F.; Vitorino, R.; Sousa, J.; Pais, A.; Vitorino, C. A Two-Pronged Approach against Glioblastoma: Drug Repurposing and Nanoformulation Design for in Situ-Controlled Release. Drug Deliv. Transl. Res. 2023, 13, 3169–3191. [Google Scholar] [CrossRef] [PubMed]
- Chaibundit, C.; Ricardo, N.M.P.S.; Ricardo, N.M.P.S.; Costa, F.D.M.L.L.; Wong, M.G.P.; Hermida-Merino, D.; Rodriguez-Perez, J.; Hamley, I.W.; Yeates, S.G.; Booth, C. Effect of Ethanol on the Micellization and Gelation of Pluronic P123. Langmuir 2008, 24, 12260–12266. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional da Farmácia e do Medicamento. Farmacopeia Portuguesa 9: Edição Oficial; Infarmed: Lisboa, Portugal, 2009; ISBN 978-972-8425-96-8. [Google Scholar]



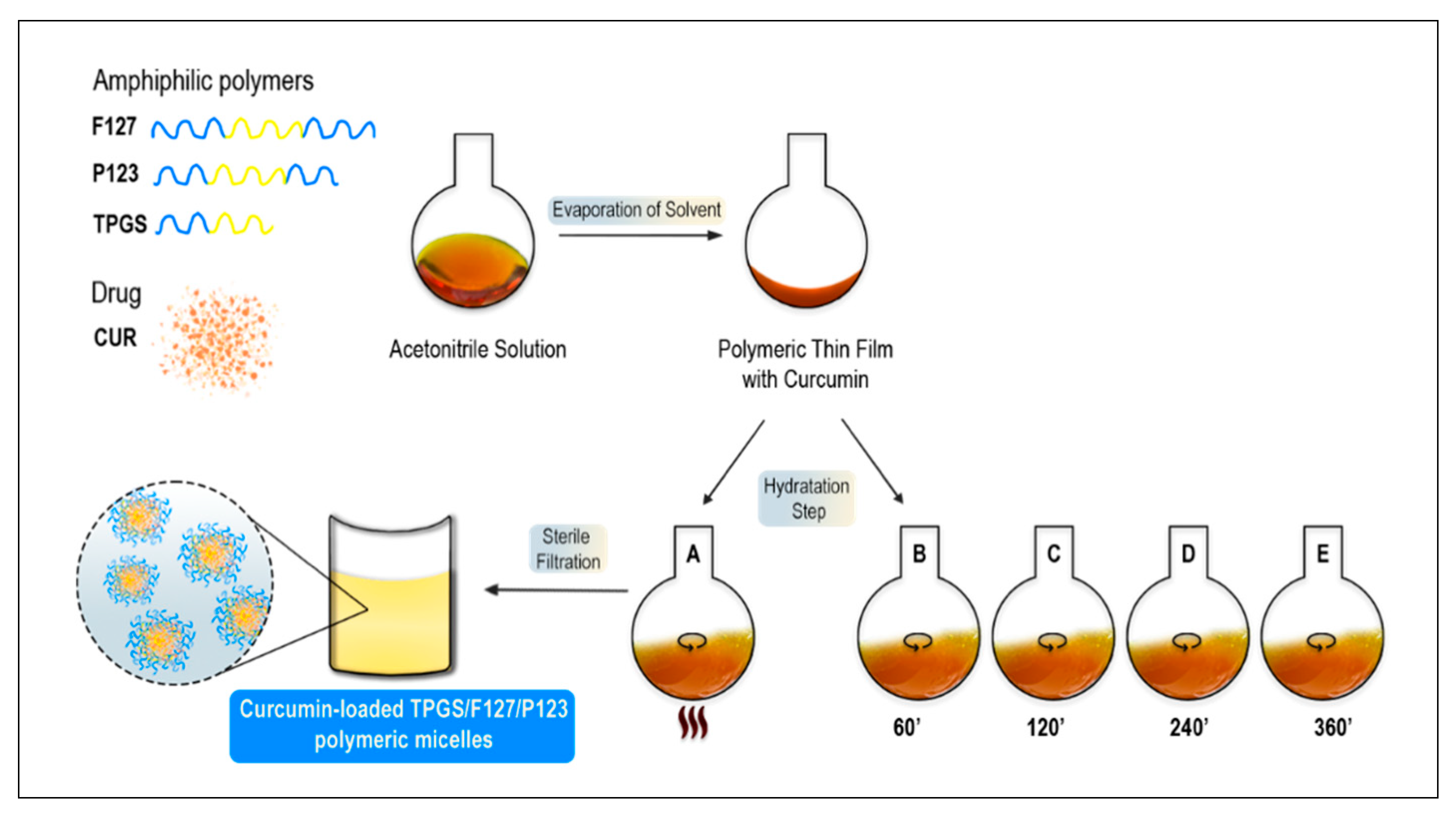

| PEOa—PPOb—PEOa | Mw 1 | Average n° PEO Units 2 | Average n° PPO Units 3 | CMC 4 | HLB 5 | Applications | |
|---|---|---|---|---|---|---|---|
| P123 | PEO20—PPO70—PEO20 | 5750 | 39.2 | 69.4 | 0.0253 | 7–9 | Inhibition of multidrug-resistance Drug delivery |
| F127 | PEO101—PPO56—PEO101 | 12,600 | 200.4 | 65.2 | 0.0353 | 18–23 | Provide stability to NPs Long circulating particles Low release gels Tissue engineering |
| TPGS | - | 1513 | - | - | 0.02 | 13 | Solubilizer of poorly water soluble drugs Enhancer of drug permeability by P-glycoprotein efflux inhibition Stabilizer of amorphous drug dispersion |
| Hydration Conditions | A | B | ||
|---|---|---|---|---|
| Filtered Sample | PFT | PFT:CUR | PFT | PFT:CUR |
| Size (nm) | 22.5 ± 1.2 | 22.9 ± 3.0 | 16.2 ± 0.9 | 15.9 ± 0.3 |
| PDI | 0.401 ± 0.042 | 0.475 ± 0.067 | 0.201 ± 0.065 | 0.251 ± 0.103 |
| Zeta (mV) | −4.2 ± 0.8 | −2.4 ± 1.9 | −1.0 ± 1.8 | −1.5 ± 1.9 |
| EE % | - | 18.197 ± 3.452 | - | 35.083 ± 1.144 |
| DL % | - | 1.880 ± 0.317 | - | 3.217 ± 0.091 |
| [CUR] mg/mL | 1.224 ± 0.224 | 2.265 ± 0.194 | ||
| Filtered Sample | PFT:CUR:A | PFT:CUR:B | PFT:CUR:C | PFT:CUR:D | PFT:CUR:E |
|---|---|---|---|---|---|
| Size (nm) | 22.9 ± 3.0 | 15.9 ± 0.3 | 17.3 ± 2.9 | 15.3 ± 0.6 | 15.9 ± 0.4 |
| 30 days | 51.0 ± 13.4 | 25.7 ± 7.3 | 252.0 ± 25.0 | 113.8 ± 13.8 | 612.9 ± 15.8 |
| PDI | 0.475 ± 0.067 | 0.251 ± 0.103 | 0.204 ± 0.064 | 0.144 ± 0.062 | 0.191 ± 0.025 |
| 30 days | 0.519 ± 0.025 | 0.319 ± 0.092 | 0.475 ± 0.475 | 0.576 ± 0.369 | 0.884 ± 0.148 |
| Zeta (mV) | −2.4 ± 1.9 | −1.5 ± 1.9 | −2.6 ± 1.3 | −0.2 ± 0.7 | −0.8 ± 1.2 |
| 30 days | −4.6 ± 7.0 | −2.1 ± 3.2 | −1.7 ± 0.8 | −1.4 ± 1.2 | −0.6 ± 1.1 |
| EE % | 18.197 ± 3.452 | 35.083 ± 1.144 | 29.153 ± 2.276 | 27.360 ± 5.056 | 24.837 ± 2.106 |
| 30 days | 10.690 ± 3.714 | 20.957 ± 5.309 | 19.350 ± 4.966 | 24.363 ± 4.952 | 16.497 ± 4.233 |
| DL % | 1.880 ± 0.317 | 3.217 ± 0.091 | 2.590 ± 0.320 | 2.553 ± 0.305 | 2.273 ± 0.197 |
| 30 days | 0.975 ± 0.995 | 1.906 ± 0.481 | 1.770 ± 0.450 | 2.230 ± 0.463 | 1.507 ± 0.387 |
| [CUR] mg/mL | 1.224 ± 0.224 | 2.265 ± 0.194 | 1.965 ± 0.164 | 1.979 ± 0.337 | 1.522 ± 0.202 |
| 30 days | 0.896 ± 0.896 | 1.410 ± 0.357 | 1.303 ± 0.331 | 1.644 ± 0.339 | 1.131 ± 0.301 |
| Remaining CUR% | 73.20% | 62.25% | 66.31% | 83.07% | 74.31% |
| (A) | Size (nm) | PDI | Zeta (mV) | EE (%) | DL (%) | [CUR] mg/mL |
| Before Lyophilization | 16.9 ± 1.4 | 0.270 ± 0.072 | 0.6 ± 0.9 | 25.825 ± 2.213 | 2.377 ± 0.197 | 1.759 ± 0.142 |
| After Lyophilization No GLY | 21.4 ± 3.4 | 0.256 ± 0.011 | −3.6 ± 1.4 | 20.277 ± 3.842 | 1.702 ± 0.280 | 1.380 ± 0.253 |
| 78.45% | ||||||
| (B) | Size (nm) | PDI | Zeta (mV) | EE (%) | DL (%) | [CUR] mg/mL |
| Before Lyophilization | 17.0 ± 0.8 | 0.264 ± 0.028 | −0.1 ± 0.6 | 27.082 ± 1.857 | 2.504 ± 0.143 | 1.851 ± 0.108 |
| After Lyophilization 1% GLY | 19.3 ± 3.7 | 0.224 ± 0.078 | −1.8 ± 0.6 | 25.926 ± 2.512 | 2.398 ± 0.228 | 1.772 ± 0.168 |
| 95.73% |
| Free CUR | Micelles | ||
|---|---|---|---|
| Zero Order | K | 0.9657 | 0.00872757 |
| b | −0.7111 | 26.7103853 | |
| R2 | 0.9937 | 0.65993668 | |
| Frist Order | K/2.303 | 0.0001 | 8.04 × 10−5 |
| log Q0 | 1.4780 | 1.47796768 | |
| R2 | 0.7272 | 0.7272309 | |
| Higuchi | K | 1.0086 | 0.65853923 |
| b | 29.0437 | 19.0791274 | |
| R2 | 0.8234 | 0.82289694 | |
| Korsmeyer–PEPPAS | K | 0.1971 | 16.6820184 |
| b | 1.2313 | −5.309242 | |
| R2 | 0.9593 | 0.96565865 | |
| (A) | Size (nm) | PDI | Zeta (mV) | ||||||
| 0 Days | 15 Days | 30 Days | 15 Days | 30 Days | 30 Days | 0 Days | 15 Days | 30 Days | |
| No GLY | 21.4 ± 3.4 | 77.3 ± 0.1 | 1093.1 ± 850.0 | 0.256 ± 0.011 | 0.239 ± 0.006 | 0.473 ± 0.429 | −3.6 ± 1.4 | −4.7 ± 4.6 | −4.5 ± 4.5 |
| 1% GLY | 19.3 ± 3.7 | 33.4 ± 3.4 | 37.9 ± 1.5 | 0.224 ± 0.078 | 0.378 ± 0.182 | 0.386 ± 0.259 | −1.8 ± 0.6 | −4.2 ± 2.8 | −4.5 ± 1.7 |
| (B) | EE (%) | DL (%) | [CUR] mg/mL | ||||||
| 0 Days | 15 Days | 30 Days | 15 Days | 0 Days | 15 Days | 30 Days | 15 Days | 0 Days | |
| No GLY | 20.277 ± 3.842 | 18.299 ± 3.208 | 20.616 ± 12.791 | 1.702 ± 0.280 | 1.684 ± 0.290 | 1.899 ± 1.181 | 1.380 ± 0.253 | 1.246 ± 0.212 | 1.406 ± 0.877 |
| 1% GLY | 25.926 ± 2.512 | 20.204 ± 5.636 | 21.913 ± 6.427 | 2.398 ± 0.228 | 1.863 ± 0.497 | 1.719 ± 0.093 | 1.772 ± 0.168 | 1.378 ± 0.369 | 1.271 ± 0.068 |
| IC50 | [CUR] µg/mL | [PFT] µg/mL |
|---|---|---|
| Free CUR | 3.800 | - |
| PFT:CUR:B GLY 1% | 0.887 | 173.5 |
| PFT:B GLY 1% | - | 174.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cerqueira, R.; Domingues, C.; Veiga, F.; Jarak, I.; Figueiras, A. Development and Characterization of Curcumin-Loaded TPGS/F127/P123 Polymeric Micelles as a Potential Therapy for Colorectal Cancer. Int. J. Mol. Sci. 2024, 25, 7577. https://doi.org/10.3390/ijms25147577
Cerqueira R, Domingues C, Veiga F, Jarak I, Figueiras A. Development and Characterization of Curcumin-Loaded TPGS/F127/P123 Polymeric Micelles as a Potential Therapy for Colorectal Cancer. International Journal of Molecular Sciences. 2024; 25(14):7577. https://doi.org/10.3390/ijms25147577
Chicago/Turabian StyleCerqueira, Rita, Cátia Domingues, Francisco Veiga, Ivana Jarak, and Ana Figueiras. 2024. "Development and Characterization of Curcumin-Loaded TPGS/F127/P123 Polymeric Micelles as a Potential Therapy for Colorectal Cancer" International Journal of Molecular Sciences 25, no. 14: 7577. https://doi.org/10.3390/ijms25147577
APA StyleCerqueira, R., Domingues, C., Veiga, F., Jarak, I., & Figueiras, A. (2024). Development and Characterization of Curcumin-Loaded TPGS/F127/P123 Polymeric Micelles as a Potential Therapy for Colorectal Cancer. International Journal of Molecular Sciences, 25(14), 7577. https://doi.org/10.3390/ijms25147577










