Current View on PPAR-α and Its Relation to Neurosteroids in Alzheimer’s Disease and Other Neuropsychiatric Disorders: Promising Targets in a Therapeutic Strategy
Abstract
1. Introduction
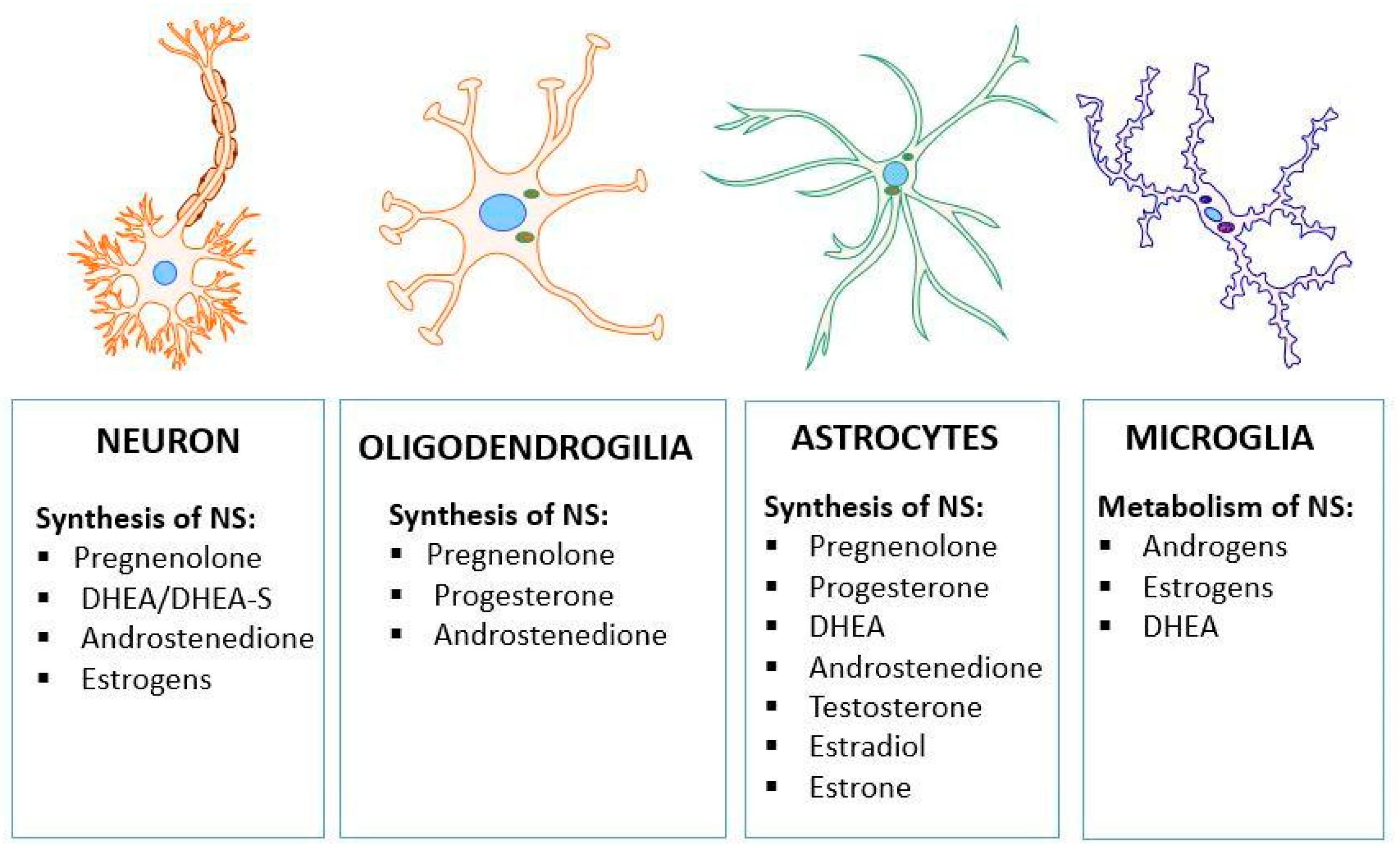
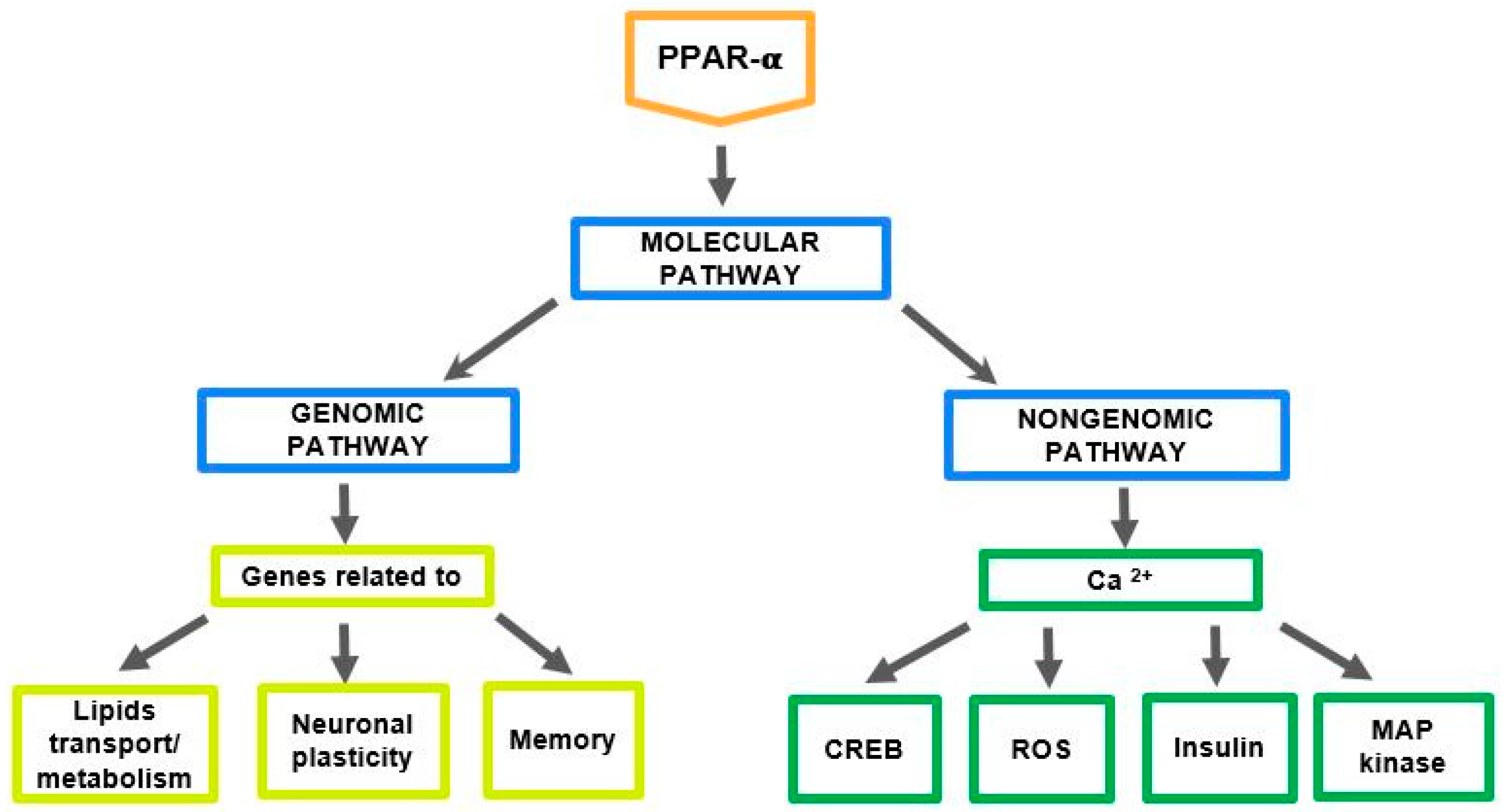
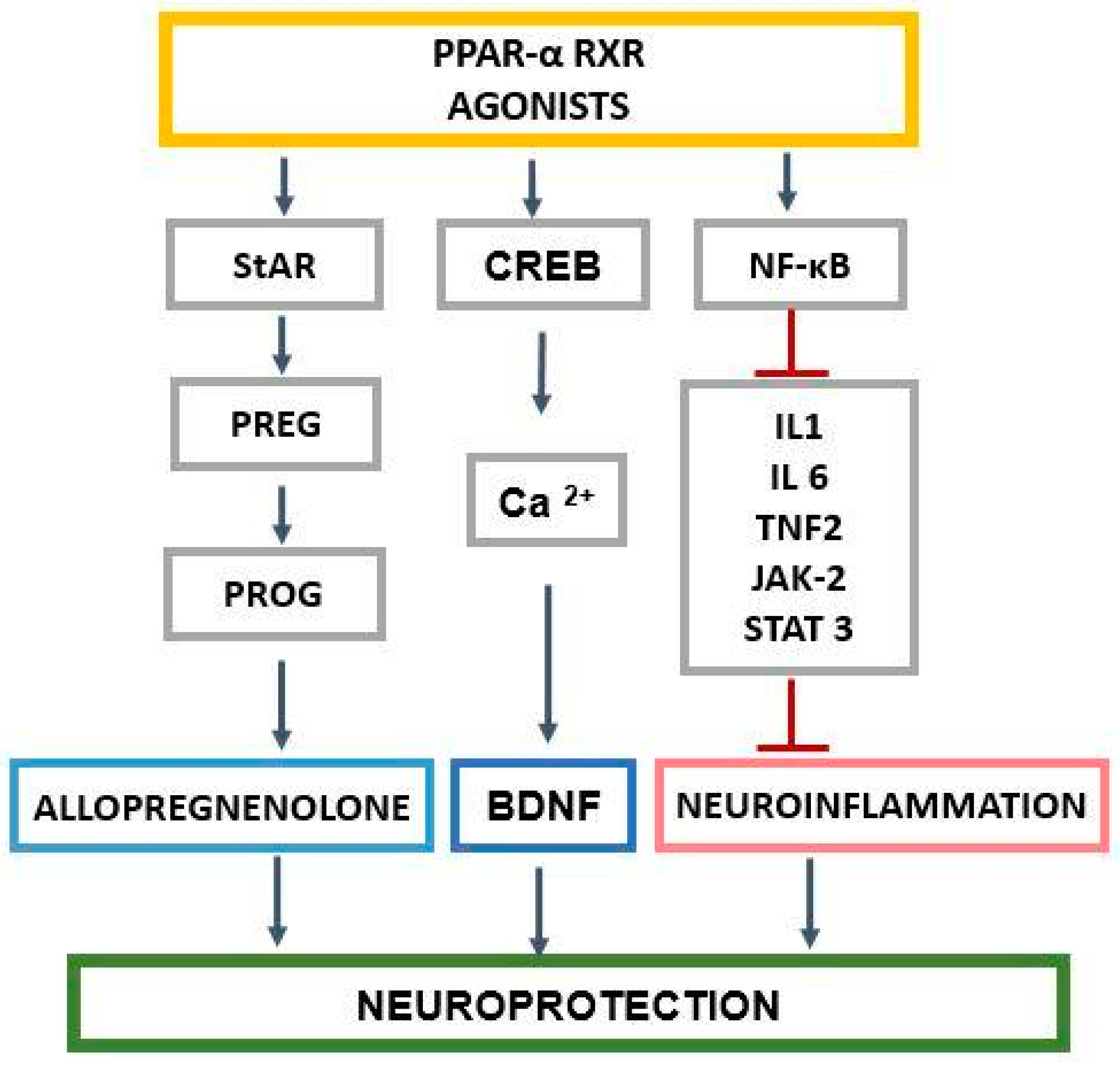
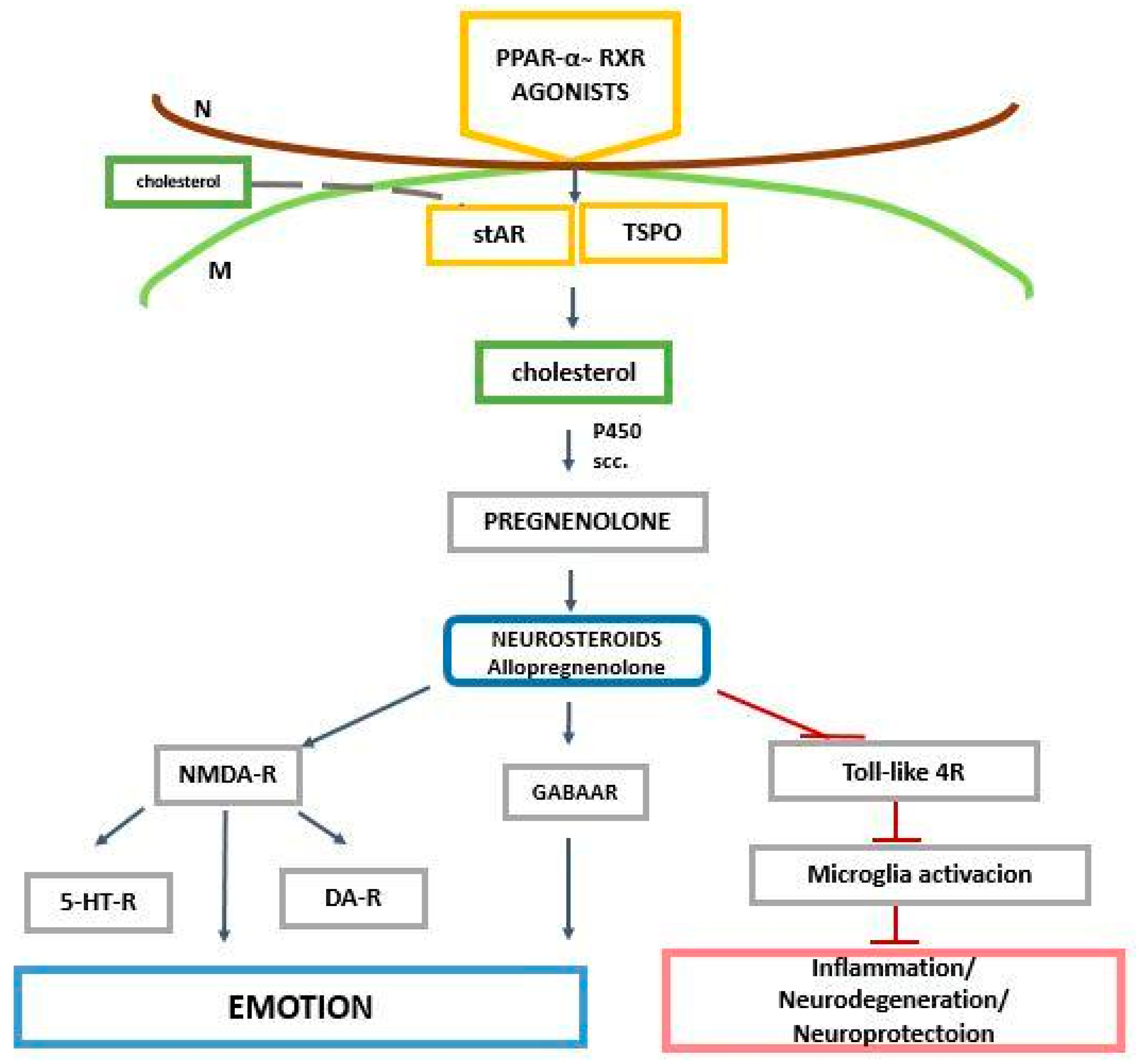
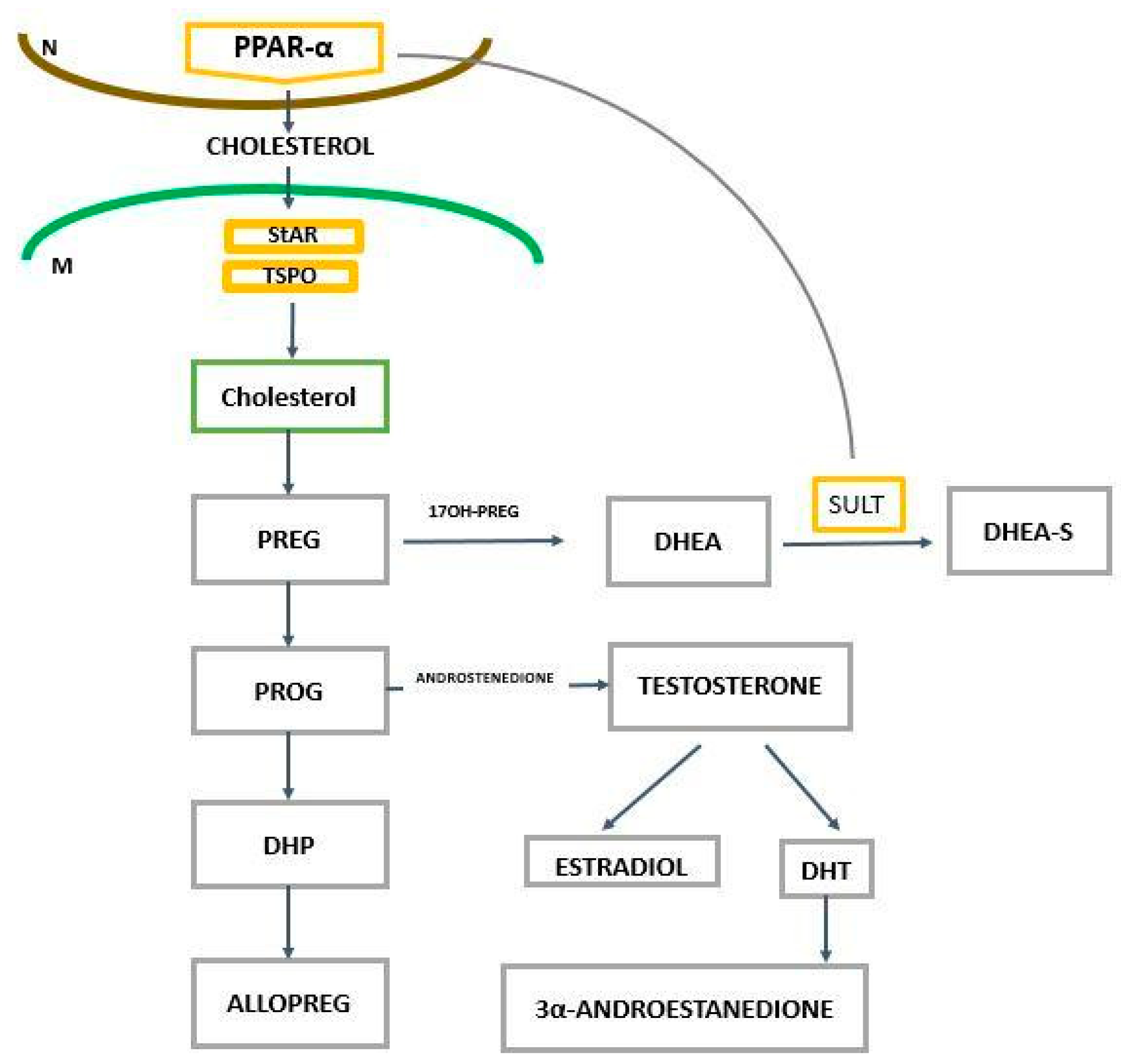
2. The Role of PPAR-α in the Regulation of Genes Related to Cholesterol and Neurosteroids
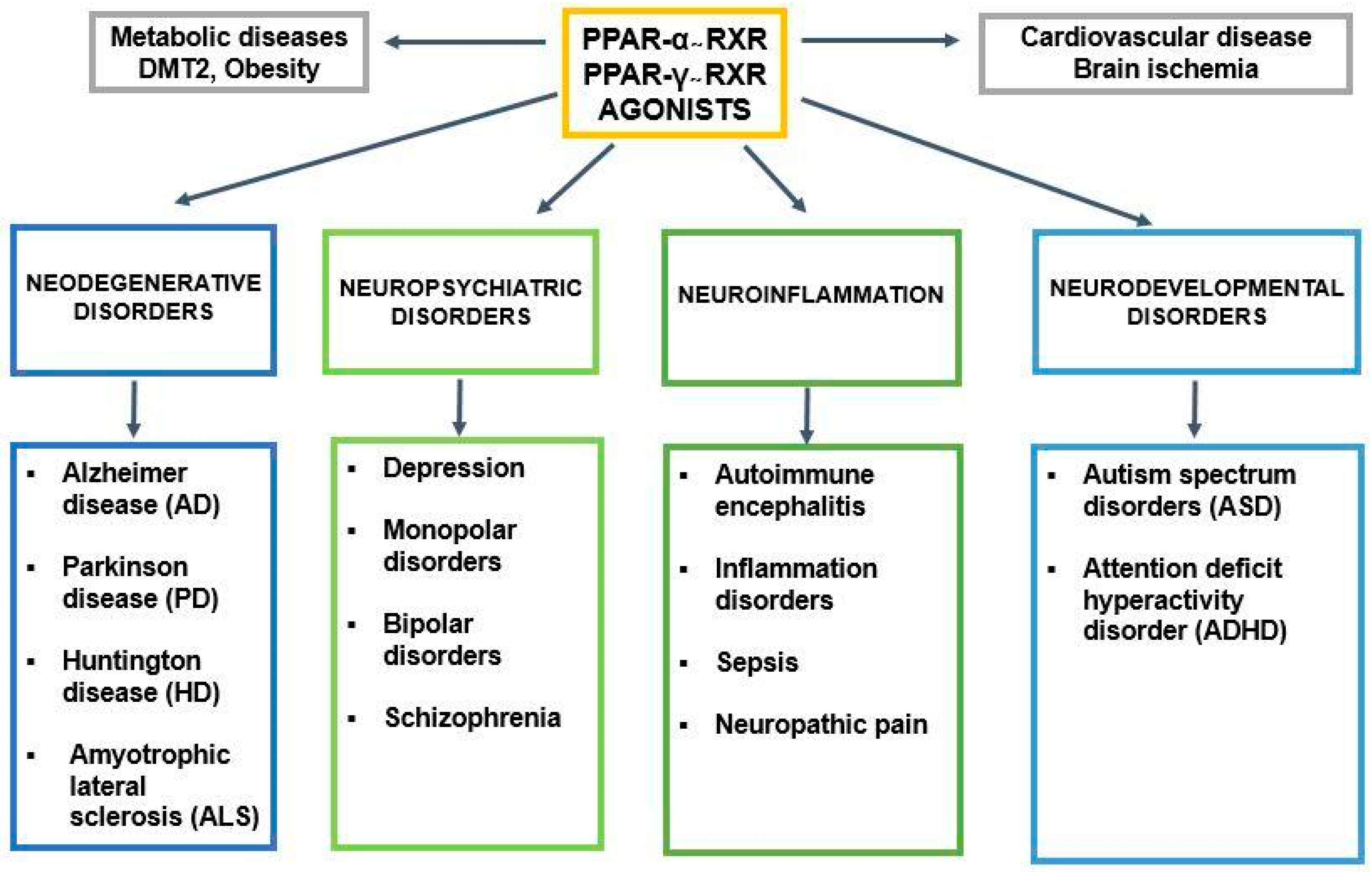
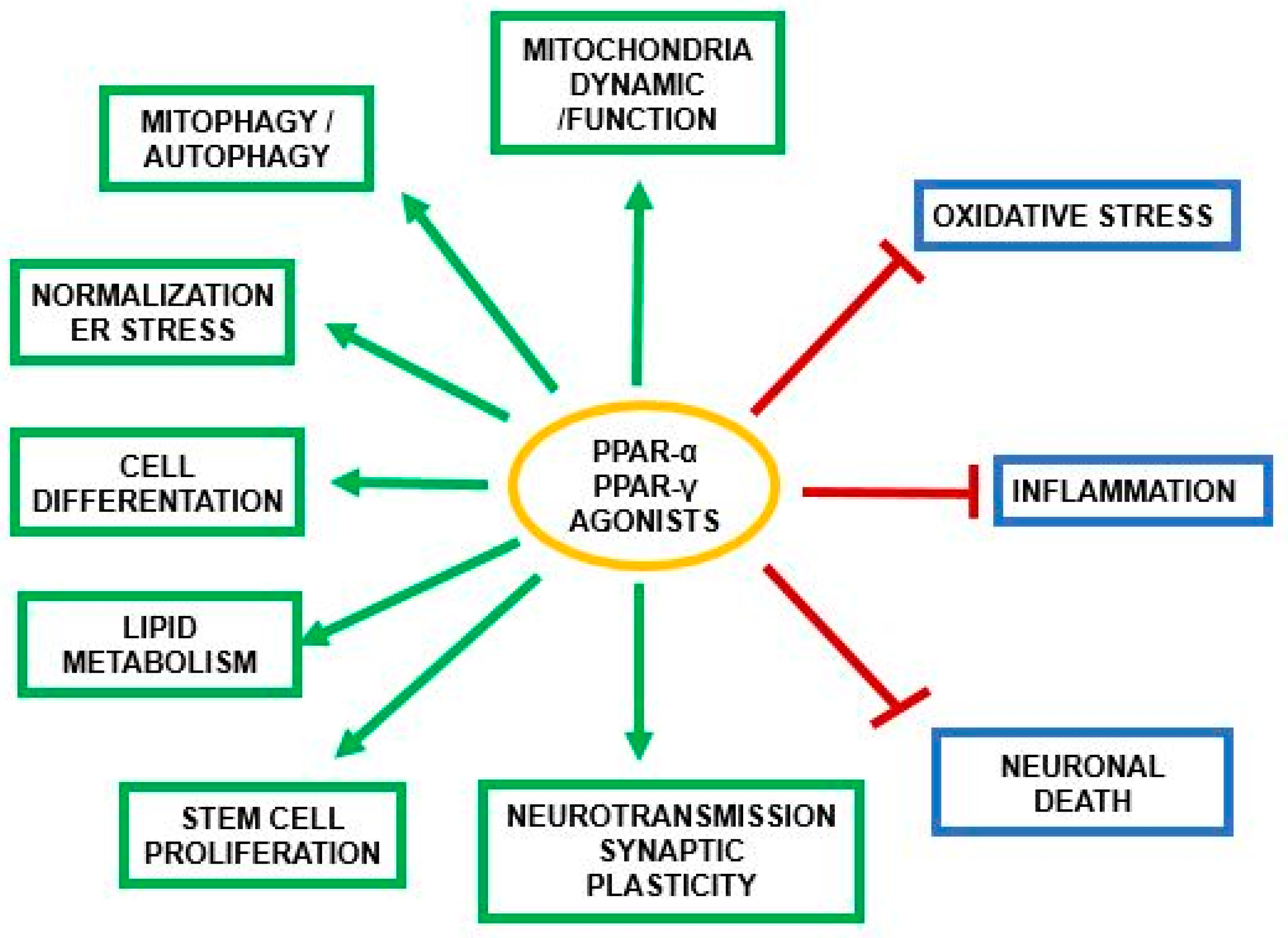
3. PPAR-α and Neurosteroids in Preclinical and Clinical Therapeutic Approaches of Neurodegenerative/Neuropsychiatric Disorders
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Corbett, A.; Pickett, J.; Burns, A.; Corcoran, J.; Dunnett, S.B.; Edison, P.; Hagan, J.J.; Holmes, C.; Jones, E.; Katona, C.; et al. Drug repositioning for Alzheimer’s disease. Nat. Rev. Drug Discov. 2012, 11, 833–846. [Google Scholar] [CrossRef] [PubMed]
- Ballard, C.; Aarsland, D.; Cummings, J.; O’Brien, J.; Mills, R.; Molinuevo, J.L.; Fladby, T.; Williams, G.; Doherty, P.; Corbett, A.; et al. Drug repositioning and repurposing for Alzheimer disease. Nat. Rev. Neurol. 2020, 16, 661–673. [Google Scholar] [CrossRef] [PubMed]
- Cummings, J.; Zhou, Y.; Lee, G.; Zhong, K.; Fonseca, J.; Cheng, F. Alzheimer’s disease drug development pipeline: 2023. Alzheimer Dement. 2023, 9, e12385. [Google Scholar] [CrossRef] [PubMed]
- Tondo, G.; Aprile, D.; Tesser, F.; Comi, C. Increased Prevalence of Neuropsychiatric Disorders during COVID-19 Pandemic in People Needing a Non-Deferrable Neurological Evaluation. J. Clin. Med. 2021, 10, 5169. [Google Scholar] [CrossRef]
- McGrath, J.J.; Al-Hamzawi, A.; Alonso, J.; Altwaijri, Y.; Andrade, L.H.; Bromet, E.J.; Bruffaerts, R.; de Almeida, J.M.C.; Chardoul, S.; Chiu, W.T.; et al. Age of onset and cumulative risk of mental disorders: A cross-national analysis of population surveys from 29 countries. Lancet. Psychiatry 2023, 10, 668–681. [Google Scholar] [CrossRef]
- Solmi, M.; Radua, J.; Olivola, M.; Croce, E.; Soardo, L.; Salazar de Pablo, G.; Il Shin, J.; Kirkbride, J.B.; Jones, P.; Kim, J.H.; et al. Age at onset of mental disorders worldwide: Large-scale meta-analysis of 192 epidemiological studies. Mol. Psychiatry 2022, 27, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Vasishta, S.; Umakanth, S.; Adiga, P.; Joshi, M.B. Extrinsic and intrinsic factors influencing metabolic memory in type 2 diabetes. Vasc. Pharmacol. 2022, 142, 106933. [Google Scholar] [CrossRef]
- Wencel, P.L.; Blecharz-Klin, K.; Piechal, A.; Pyrzanowska, J.; Mirowska-Guzel, D.; Strosznajder, R.P. Fingolimod Modulates the Gene Expression of Proteins Engaged in Inflammation and Amyloid-Beta Metabolism and Improves Exploratory and Anxiety-Like Behavior in Obese Mice. Neurotherapeutics 2023, 20, 1388–1404. [Google Scholar] [CrossRef]
- Teo, C.H.Y.; Lin, M.T.; Lee, I.X.Y.; Koh, S.K.; Zhou, L.; Goh, D.S.; Choi, H.; Koh, H.W.L.; Lam, A.Y.R.; Lim, P.S.; et al. Oral Peroxisome Proliferator-Activated Receptor-α Agonist Enhances Corneal Nerve Regeneration in Patients With Type 2 Diabetes. Diabetes 2023, 72, 932–946. [Google Scholar] [CrossRef]
- Blanquart, C.; Barbier, O.; Fruchart, J.C.; Staels, B.; Glineur, C. Peroxisome proliferator-activated receptors: Regulation of transcriptional activities and roles in inflammation. J. Steroid Biochem. Mol. Biol. 2003, 85, 267–273. [Google Scholar] [CrossRef]
- Bordet, R.; Gelé, P.; Duriez, P.; Fruchart, J.C. PPARs: A new target for neuroprotection. J. Neurol. Neurosurg. Psychiatry 2006, 77, 285–287. [Google Scholar] [CrossRef] [PubMed]
- Wójtowicz, S.; Strosznajder, A.K.; Jeżyna, M.; Strosznajder, J.B. The Novel Role of PPAR Alpha in the Brain: Promising Target in Therapy of Alzheimer’s Disease and Other Neurodegenerative Disorders. Neurochem. Res. 2020, 45, 972–988. [Google Scholar] [CrossRef] [PubMed]
- Strosznajder, A.K.; Wójtowicz, S.; Jeżyna, M.J.; Sun, G.Y.; Strosznajder, J.B. Recent Insights on the Role of PPAR-β/δ in Neuroinflammation and Neurodegeneration, and Its Potential Target for Therapy. Neuromolecular Med. 2021, 23, 86–98. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Jana, M.; Corbett, G.T.; Ramaswamy, S.; Kordower, J.H.; Gonzalez, F.J.; Pahan, K. Regulation of cyclic AMP response element binding and hippocampal plasticity-related genes by peroxisome proliferator-activated receptor α. Cell Rep. 2013, 4, 724–737. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Pahan, K. PPARα signaling in the hippocampus: Crosstalk between fat and memory. J. Neuroimmune Pharmacol. 2015, 10, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Bougarne, N.; Weyers, B.; Desmet, S.J.; Deckers, J.; Ray, D.W.; Staels, B.; De Bosscher, K. Molecular Actions of PPARα in Lipid Metabolism and Inflammation. Endocr. Rev. 2018, 39, 760–802. [Google Scholar] [CrossRef]
- Mergiya, T.F.; Gundersen, J.E.T.; Kanhema, T.; Brighter, G.; Ishizuka, Y.; Bramham, C.R. Detection of Arc/Arg3.1 oligomers in rat brain: Constitutive and synaptic activity-evoked dimer expression in vivo. Front. Mol. Neurosci. 2023, 16, 1142361. [Google Scholar] [CrossRef] [PubMed]
- Tzingounis, A.V.; Nicoll, R.A. Arc/Arg3.1: Linking gene expression to synaptic plasticity and memory. Neuron 2006, 52, 403–407. [Google Scholar] [CrossRef]
- Sibarov, D.A.; Tsytsarev, V.; Volnova, A.; Vaganova, A.N.; Alves, J.; Rojas, L.; Sanabria, P.; Ignashchenkova, A.; Savage, E.D.; Inyushin, M. Arc protein, a remnant of ancient retrovirus, forms virus-like particles, which are abundantly generated by neurons during epileptic seizures, and affects epileptic susceptibility in rodent models. Front. Neurol. 2023, 14, 1201104. [Google Scholar] [CrossRef]
- Żulińska, S.; Strosznajder, A.K.; Strosznajder, J.B. The role of synthetic ligand of PPARα in regulation of transcription of genes related to mitochondria biogenesis and dynamic in an animal model of Alzheimer’s disease. Folia Neuropathol. 2023, 61, 138–143. [Google Scholar] [CrossRef]
- Scarpulla, R.C.; Vega, R.B.; Kelly, D.P. Transcriptional integration of mitochondrial biogenesis. Trends Endocrinol. Metab. 2012, 23, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Nierenberg, A.A.; Ghaznavi, S.A.; Sande Mathias, I.; Ellard, K.K.; Janos, J.A.; Sylvia, L.G. Peroxisome Proliferator-Activated Receptor Gamma Coactivator-1 Alpha as a Novel Target for Bipolar Disorder and Other Neuropsychiatric Disorders. Biol. Psychiatry 2018, 83, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Nisbett, K.E.; Pinna, G. Emerging Therapeutic Role of PPAR–α in Cognition and Emotions. Front. Pharmacol. 2018, 9, 400177. [Google Scholar] [CrossRef] [PubMed]
- Pinna, G. Role of PPAR-Allopregnanolone Signaling in Behavioral and Inflammatory Gut-Brain Axis Communications. Biol. Psychiatry 2023, 94, 609–618. [Google Scholar] [CrossRef] [PubMed]
- Ratner, M.H.; Kumaresan, V.; Farb, D.H. Neurosteroid Actions in Memory and Neurologic/Neuropsychiatric Disorders. Front. Endocrinol. 2019, 10, 169. [Google Scholar] [CrossRef] [PubMed]
- Monnet, F.P.; Maurice, T. The sigma1 protein as a target for the non-genomic effects of neuro(active)steroids: Molecular, physiological, and behavioral aspects. J. Pharmacol. Sci. 2006, 100, 93–118. [Google Scholar] [CrossRef] [PubMed]
- Lambert, J.J.; Cooper, M.A.; Simmons, R.D.; Weir, C.J.; Belelli, D. Neurosteroids: Endogenous allosteric modulators of GABA(A) receptors. Psychoneuroendocrinology 2009, 34 (Suppl. 1), S48–S58. [Google Scholar] [CrossRef] [PubMed]
- Vallée, M.; Mayo, W.; Darnaudéry, M.; Corpéchot, C.; Young, J.; Koehl, M.; Le Moal, M.; Baulieu, E.E.; Robel, P.; Simon, H. Neurosteroids: Deficient cognitive performance in aged rats depends on low pregnenolone sulfate levels in the hippocampus. Proc. Natl. Acad. Sci. USA 1997, 94, 14865–14870. [Google Scholar] [CrossRef] [PubMed]
- Weill-Engerer, S.; David, J.P.; Sazdovitch, V.; Liere, P.; Eychenne, B.; Pianos, A.; Schumacher, M.; Delacourte, A.; Baulieu, E.E.; Akwa, Y. Neurosteroid quantification in human brain regions: Comparison between Alzheimer’s and nondemented patients. J. Clin. Endocrinol. Metab. 2002, 87, 5138–5143. [Google Scholar] [CrossRef]
- van Wingen, G.; van Broekhoven, F.; Verkes, R.J.; Petersson, K.M.; Bäckström, T.; Buitelaar, J.; Fernández, G. How progesterone impairs memory for biologically salient stimuli in healthy young women. J. Neurosci. 2007, 27, 11416–11423. [Google Scholar] [CrossRef]
- Scullin, C.S.; Partridge, L.D. Modulation by pregnenolone sulfate of filtering properties in the hippocampal trisynaptic circuit. Hippocampus 2012, 22, 2184–2198. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.C.; Gibbs, T.T.; Farb, D.H. Pregnenolone sulfate as a modulator of synaptic plasticity. Psychopharmacology 2014, 231, 3537–3556. [Google Scholar] [CrossRef] [PubMed]
- Kreinin, A.; Bawakny, N.; Ritsner, M.S. Adjunctive Pregnenolone Ameliorates the Cognitive Deficits in Recent-Onset Schizophrenia: An 8-Week, Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Schizophr. Relat. Psychoses 2017, 10, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Reddy, D.S.; Gangisetty, O.; Wu, X. PR-independent neurosteroid regulation of α2-GABA-A receptors in the hippocampus subfields. Brain Res. 2017, 1659, 142–147. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.; García-Ruiz, C.M.; Fernandez-Checa, J.C. Mitochondrial Cholesterol in Alzheimer’s Disease and Niemann-Pick Type C Disease. Front. Neurol. 2019, 10, 1168. [Google Scholar] [CrossRef] [PubMed]
- Baulieu, E.E.; Thomas, G.; Legrain, S.; Lahlou, N.; Roger, M.; Debuire, B.; Faucounau, V.; Girard, L.; Hervy, M.P.; Latour, F.; et al. Dehydroepiandrosterone (DHEA), DHEA sulfate, and aging: Contribution of the DHEAge Study to a sociobiomedical issue. Proc. Natl. Acad. Sci. USA 2000, 97, 4279–4284. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea, L.; Conde de la Rosa, L.; Torres, S.; García-Ruiz, C.; Fernández-Checa, J.C. Mitochondrial cholesterol: Metabolism and impact on redox biology and disease. Redox Biol. 2023, 61, 102643. [Google Scholar] [CrossRef] [PubMed]
- di Gregorio, M.C.; Cautela, J.; Galantini, L. Physiology and Physical Chemistry of Bile Acids. Int. J. Mol. Sci. 2021, 22, 1870. [Google Scholar] [CrossRef] [PubMed]
- Wood, W.G.; Li, L.; Müller, W.E.; Eckert, G.P. Cholesterol as a causative factor in Alzheimer’s disease: A debatable hypothesis. J. Neurochem. 2014, 129, 559–572. [Google Scholar] [CrossRef]
- Refolo, L.M.; Malester, B.; LaFrancois, J.; Bryant-Thomas, T.; Wang, R.; Tint, G.S.; Sambamurti, K.; Duff, K.; Pappolla, M.A. Hypercholesterolemia accelerates the Alzheimer’s amyloid pathology in a transgenic mouse model. Neurobiol. Dis. 2000, 7, 321–331. [Google Scholar] [CrossRef]
- Woodruff-Pak, D.S.; Agelan, A.; Del Valle, L. A rabbit model of Alzheimer’s disease: Valid at neuropathological, cognitive, and therapeutic levels. J. Alzheimers Dis. 2007, 11, 371–383. [Google Scholar] [CrossRef]
- Cutler, R.G.; Kelly, J.; Storie, K.; Pedersen, W.A.; Tammara, A.; Hatanpaa, K.; Troncoso, J.C.; Mattson, M.P. Involvement of oxidative stress-induced abnormalities in ceramide and cholesterol metabolism in brain aging and Alzheimer’s disease. Proc. Natl. Acad. Sci. USA 2004, 101, 2070–2075. [Google Scholar] [CrossRef]
- Ehehalt, R.; Keller, P.; Haass, C.; Thiele, C.; Simons, K. Amyloidogenic processing of the Alzheimer beta-amyloid precursor protein depends on lipid rafts. J. Cell Biol. 2003, 160, 113–123. [Google Scholar] [CrossRef]
- Testa, G.; Giannelli, S.; Sottero, B.; Staurenghi, E.; Giaccone, G.; Caroppo, P.; Gamba, P.; Leonarduzzi, G. 24-Hydroxycholesterol Induces Tau Proteasome-Dependent Degradation via the SIRT1/PGC1α/Nrf2 Pathway: A Potential Mechanism to Counteract Alzheimer’s Disease. Antioxid. 2023, 12, 631. [Google Scholar] [CrossRef]
- Zwain, I.H.; Yen, S.S. Neurosteroidogenesis in astrocytes, oligodendrocytes, and neurons of cerebral cortex of rat brain. Endocrinology 1999, 140, 3843–3852. [Google Scholar] [CrossRef]
- Gago, N.; Akwa, Y.; Sananès, N.; Guennoun, R.; Baulieu, E.E.; El-Etr, M.; Schumacher, M. Progesterone and the oligodendroglial lineage: Stage-dependent biosynthesis and metabolism. Glia 2001, 36, 295–308. [Google Scholar] [CrossRef]
- Alexaki, V.I.; Fodelianaki, G.; Neuwirth, A.; Mund, C.; Kourgiantaki, A.; Ieronimaki, E.; Lyroni, K.; Troullinaki, M.; Fujii, C.; Kanczkowski, W.; et al. DHEA inhibits acute microglia-mediated inflammation through activation of the TrkA-Akt1/2-CREB-Jmjd3 pathway. Mol. Psychiatry 2018, 23, 1410–1420. [Google Scholar] [CrossRef]
- Runge-Morris, M.; Kocarek, T.A. Regulation of sulfotransferase and UDP-glucuronosyltransferase gene expression by the PPARs. PPAR Res. 2009, 2009, 728941. [Google Scholar] [CrossRef]
- Yilmaz, C.; Karali, K.; Fodelianaki, G.; Gravanis, A.; Chavakis, T.; Charalampopoulos, I.; Alexaki, V.I. Neurosteroids as regulators of neuroinflammation. Front. Neuroendocrinol. 2019, 55, 100788. [Google Scholar] [CrossRef]
- Luo, R.; Su, L.-Y.; Li, G.; Yang, J.; Liu, Q.; Yang, L.-X.; Zhang, D.-F.; Zhou, H.; Xu, M.; Fan, Y.; et al. Activation of PPARA-mediated autophagy reduces Alzheimer disease-like pathology and cognitive decline in a murine model. Autophagy 2020, 16, 52–69. [Google Scholar] [CrossRef]
- Suzuki, R.; Ferris, H.A.; Chee, M.J.; Maratos-Flier, E.; Kahn, C.R. Reduction of the cholesterol sensor SCAP in the brains of mice causes impaired synaptic transmission and altered cognitive function. PLoS Biol. 2013, 11, e1001532. [Google Scholar] [CrossRef] [PubMed]
- Ujiie, M.; Dickstein, D.L.; Carlow, D.A.; Jefferies, W.A. Blood-brain barrier permeability precedes senile plaque formation in an Alzheimer disease model. Microcirculation 2003, 10, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Martín, M.G.; Pfrieger, F.; Dotti, C.G. Cholesterol in brain disease: Sometimes determinant and frequently implicated. EMBO Rep. 2014, 15, 1036–1052. [Google Scholar] [CrossRef]
- Martín-Segura, A.; Ahmed, T.; Casadomé-Perales, Á.; Palomares-Perez, I.; Palomer, E.; Kerstens, A.; Munck, S.; Balschun, D.; Dotti, C.G. Age-associated cholesterol reduction triggers brain insulin resistance by facilitating ligand-independent receptor activation and pathway desensitization. Aging Cell 2019, 18, e12932. [Google Scholar] [CrossRef] [PubMed]
- Sáez-Orellana, F.; Leroy, T.; Ribeiro, F.; Kreis, A.; Leroy, K.; Lalloyer, F.; Baugé, E.; Staels, B.; Duyckaerts, C.; Brion, J.P.; et al. Regulation of PPARα by APP in Alzheimer disease affects the pharmacological modulation of synaptic activity. JCI Insight 2021, 6, e150099. [Google Scholar] [CrossRef]
- Compagnone, N.A.; Mellon, S.H. Neurosteroids: Biosynthesis and function of these novel neuromodulators. Front. Neuroendocrinol. 2000, 21, 1–56. [Google Scholar] [CrossRef]
- Mellon, S.H.; Griffin, L.D. Neurosteroids: Biochemistry and clinical significance. Trends Endocrinol. Metab. 2002, 13, 35–43. [Google Scholar] [CrossRef]
- Prough, R.A.; Clark, B.J.; Klinge, C.M. Novel mechanisms for DHEA action. J. Mol. Endocrinol. 2016, 56, R139–R155. [Google Scholar] [CrossRef]
- Maguire, J.L.; Mennerick, S. Neurosteroids: Mechanistic considerations and clinical prospects. Neuropsychopharmacology 2024, 49, 73–82. [Google Scholar] [CrossRef]
- Gunay, A.; Pinna, G. The novel rapid-acting neurosteroid-based antidepressant generation. Curr. Opin. Endocr. Metab. Res. 2022, 24, 100340. [Google Scholar] [CrossRef]
- Puia, G.; Ravazzini, F.; Castelnovo, L.F.; Magnaghi, V. PKCε and allopregnanolone: Functional cross-talk at the GABAA receptor level. Front. Cell. Neurosci. 2015, 9, 83. [Google Scholar] [CrossRef]
- Brown, R.C.; Cascio, C.; Papadopoulos, V. Pathways of neurosteroid biosynthesis in cell lines from human brain: Regulation of dehydroepiandrosterone formation by oxidative stress and beta-amyloid peptide. J. Neurochem. 2000, 74, 847–859. [Google Scholar] [CrossRef]
- Covey, D.F.; Evers, A.S.; Izumi, Y.; Maguire, J.L.; Mennerick, S.J.; Zorumski, C.F. Neurosteroid enantiomers as potentially novel neurotherapeutics. Neurosci. Biobehav. Rev. 2023, 149, 105191. [Google Scholar] [CrossRef]
- Vitku, J.; Hill, M.; Kolatorova, L.; Kubala Havrdova, E.; Kancheva, R. Steroid Sulfation in Neurodegenerative Diseases. Front. Mol. Biosci. 2022, 9, 839887. [Google Scholar] [CrossRef]
- Grimm, A.; Schmitt, K.; Lang, U.E.; Mensah-Nyagan, A.G.; Eckert, A. Improvement of neuronal bioenergetics by neurosteroids: Implications for age-related neurodegenerative disorders. Biochim. Biophys. Acta 2014, 1842, 2427–2438. [Google Scholar] [CrossRef]
- Magnaghi, V.; Ballabio, M.; Cavarretta, I.T.; Froestl, W.; Lambert, J.J.; Zucchi, I.; Melcangi, R.C. GABAB receptors in Schwann cells influence proliferation and myelin protein expression. Eur. J. Neurosci. 2004, 19, 2641–2649. [Google Scholar] [CrossRef]
- Mendell, A.L.; MacLusky, N.J. Neurosteroid Metabolites of Gonadal Steroid Hormones in Neuroprotection: Implications for Sex Differences in Neurodegenerative Disease. Front. Mol. Neurosci. 2018, 11, 359. [Google Scholar] [CrossRef]
- Gaignard, P.; Liere, P.; Thérond, P.; Schumacher, M.; Slama, A.; Guennoun, R. Role of Sex Hormones on Brain Mitochondrial Function, with Special Reference to Aging and Neurodegenerative Diseases. Front. Aging Neurosci. 2017, 9, 406. [Google Scholar] [CrossRef]
- Grimm, A.; Lim, Y.A.; Mensah-Nyagan, A.G.; Götz, J.; Eckert, A. Alzheimer’s disease, oestrogen and mitochondria: An ambiguous relationship. Mol. Neurobiol. 2012, 46, 151–160. [Google Scholar] [CrossRef]
- Rivera-Meza, M.; Muñoz, D.; Jerez, E.; Quintanilla, M.E.; Salinas-Luypaert, C.; Fernandez, K.; Karahanian, E. Fenofibrate Administration Reduces Alcohol and Saccharin Intake in Rats: Possible Effects at Peripheral and Central Levels. Front. Behav. Neurosci. 2017, 11, 133. [Google Scholar] [CrossRef]
- Le Foll, B.; Di Ciano, P.; Panlilio, L.V.; Goldberg, S.R.; Ciccocioppo, R. Peroxisome proliferator-activated receptor (PPAR) agonists as promising new medications for drug addiction: Preclinical evidence. Curr. Drug Targets 2013, 14, 768–776. [Google Scholar] [CrossRef]
- Blednov, Y.A.; Benavidez, J.M.; Black, M.; Ferguson, L.B.; Schoenhard, G.L.; Goate, A.M.; Edenberg, H.J.; Wetherill, L.; Hesselbrock, V.; Foroud, T.; et al. Peroxisome proliferator-activated receptors α and γ are linked with alcohol consumption in mice and withdrawal and dependence in humans. Alcohol. Clin. Exp. Res. 2015, 39, 136–145. [Google Scholar] [CrossRef]
- Lee, Y.; Cho, J.H.; Lee, S.; Lee, W.; Chang, S.C.; Chung, H.Y.; Moon, H.R.; Lee, J. Neuroprotective effects of MHY908, a PPAR α/γ dual agonist, in a MPTP-induced Parkinson’s disease model. Brain Res. 2019, 1704, 47–58. [Google Scholar] [CrossRef]
- Kwon, H.S.; Koh, S.H. Neuroinflammation in neurodegenerative disorders: The roles of microglia and astrocytes. Transl. Neurodegener. 2020, 9, 42. [Google Scholar] [CrossRef]
- Kreisl, W.C.; Kim, M.J.; Coughlin, J.M.; Henter, I.D.; Owen, D.R.; Innis, R.B. PET imaging of neuroinflammation in neurological disorders. Lancet Neurol. 2020, 19, 940–950. [Google Scholar] [CrossRef] [PubMed]
- Zolezzi, J.M.; Santos, M.J.; Bastías-Candia, S.; Pinto, C.; Godoy, J.A.; Inestrosa, N.C. PPARs in the central nervous system: Roles in neurodegeneration and neuroinflammation. Biol. Rev. 2017, 92, 2046–2069. [Google Scholar] [CrossRef]
- Spychala, M.S.; Honarpisheh, P.; McCullough, L.D. Sex differences in neuroinflammation and neuroprotection in ischemic stroke. J. Neurosci. Res. 2017, 95, 462–471. [Google Scholar] [CrossRef]
- Kanmogne, M.; Klein, R.S. Neuroprotective versus Neuroinflammatory Roles of Complement: From Development to Disease. Trends Neurosci. 2021, 44, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Katharesan, V.; Deery, S.; Johnson, I.P. Neuroprotective effect of acute prior inflammation with lipopolysaccharide for adult male rat facial motoneurones. Brain Res. 2018, 1696, 56–62. [Google Scholar] [CrossRef]
- del Rey, A.; Balschun, D.; Wetzel, W.; Randolf, A.; Besedovsky, H.O. A cytokine network involving brain-borne IL-1β, IL-1ra, IL-18, IL-6, and TNFα operates during long-term potentiation and learning. Brain Behav. Immun. 2013, 33, 15–23. [Google Scholar] [CrossRef]
- Kipnis, J.; Gadani, S.; Derecki, N.C. Pro-cognitive properties of T cells. Nat. Rev. Immunol. 2012, 12, 663–669. [Google Scholar] [CrossRef] [PubMed]
- Marion-Letellier, R.; Savoye, G.; Ghosh, S. Fatty acids, eicosanoids and PPAR gamma. Eur. J. Pharmacol. 2016, 785, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Shen, W.J.; Bittner, S.; Kraemer, F.B.; Azhar, S. PPARs: Regulators of metabolism and as therapeutic targets in cardiovascular disease. Part II: PPAR-β/δ and PPAR-γ. Future Cardiol. 2017, 13, 279–296. [Google Scholar] [CrossRef] [PubMed]
- Chigurupati, S.; Dhanaraj, S.A.; Balakumar, P. A step ahead of PPARγ full agonists to PPARγ partial agonists: Therapeutic perspectives in the management of diabetic insulin resistance. Eur. J. Pharmacol. 2015, 755, 50–57. [Google Scholar] [CrossRef]
- Avarachan, J.; Augustine, A.; Shinde, P.; Gunasekaran, V. A Mechanistic approach of Peroxisome Proliferator-Activated Receptors and its subtypes on Clinical and preclinical model of Neurodegenerative disorders. Res. J. Pharm. Technol. 2021, 14, 3967–3975. [Google Scholar] [CrossRef]
- Mirza, A.Z.; Althagafi, I.I.; Shamshad, H. Role of PPAR receptor in different diseases and their ligands: Physiological importance and clinical implications. Eur. J. Med. Chem. 2019, 166, 502–513. [Google Scholar] [CrossRef]
- Contreras, A.V.; Torres, N.; Tovar, A.R. PPAR-α as a key nutritional and environmental sensor for metabolic adaptation. Adv. Nutr. 2013, 4, 439–452. [Google Scholar] [CrossRef]
- Kersten, S.; Desvergne, B.; Wahli, W. Roles of PPARs in health and disease. Nature 2000, 405, 421–424. [Google Scholar] [CrossRef]
- Vamecq, J.; Latruffe, N. Medical significance of peroxisome proliferator-activated receptors. Lancet 1999, 354, 141–148. [Google Scholar] [CrossRef]
- Fruchart, J.C. Peroxisome proliferator-activated receptor-alpha (PPARalpha): At the crossroads of obesity, diabetes and cardiovascular disease. Atherosclerosis 2009, 205, 1–8. [Google Scholar] [CrossRef]
- Semple, R.K.; Chatterjee, V.K.; O’Rahilly, S. PPAR gamma and human metabolic disease. J. Clin. Investig. 2006, 116, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Abulaban, A.A.; Al-Kuraishy, H.M.; Al-Gareeb, A.I.; Elekhnawy, E.; Alanazi, A.; Alexiou, A.; Papadakis, M.; Batiha, G.E. Role of fenofibrate in multiple sclerosis. Eur. J. Med. Res. 2024, 29, 113. [Google Scholar] [CrossRef]
- Ibáñez, C.; Acuña, T.; Quintanilla, M.E.; Pérez-Reytor, D.; Morales, P.; Karahanian, E. Fenofibrate Decreases Ethanol-Induced Neuroinflammation and Oxidative Stress and Reduces Alcohol Relapse in Rats by a PPAR-α-Dependent Mechanism. Antioxidants 2023, 12, 1758. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Lalonde, K.; Truesdell, A.; Gomes Welter, P.; Brocardo, P.S.; Rosenstock, T.R.; Gil-Mohapel, J. New Avenues for the Treatment of Huntington’s Disease. Int. J. Mol. Sci. 2021, 22, 8363. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Shen, W.-J.; Bittner, S.; Kraemer, F.B.; Azhar, S. PPARs: Regulators of metabolism and as therapeutic targets in cardiovascular disease. Part I: PPAR-α. Future Cardiol. 2017, 13, 259–278. [Google Scholar] [CrossRef]
- Dotson, A.L.; Wang, J.; Chen, Y.; Manning, D.; Nguyen, H.; Saugstad, J.A.; Offner, H. Sex differences and the role of PPAR alpha in experimental stroke. Metab. Brain Dis. 2016, 31, 539–547. [Google Scholar] [CrossRef]
- Dotson, A.L.; Wang, J.; Saugstad, J.; Murphy, S.J.; Offner, H. Splenectomy reduces infarct volume and neuroinflammation in male but not female mice in experimental stroke. J. Neuroimmunol. 2015, 278, 289–298. [Google Scholar] [CrossRef]
- Dunn, S.E.; Ousman, S.S.; Sobel, R.A.; Zuniga, L.; Baranzini, S.E.; Youssef, S.; Crowell, A.; Loh, J.; Oksenberg, J.; Steinman, L. Peroxisome proliferator-activated receptor (PPAR)alpha expression in T cells mediates gender differences in development of T cell-mediated autoimmunity. J. Exp. Med. 2007, 204, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Ouk, T.; Gautier, S.; Pétrault, M.; Montaigne, D.; Maréchal, X.; Masse, I.; Devedjian, J.-C.; Deplanque, D.; Bastide, M.; Nevière, R.; et al. Effects of the PPAR-α agonist fenofibrate on acute and short-term consequences of brain ischemia. J. Cereb. Blood Flow. Metab. 2014, 34, 542–551. [Google Scholar] [CrossRef]
- Czupryniak, L.; Joshi, S.R.; Gogtay, J.A.; Lopez, M. Effect of micronized fenofibrate on microvascular complications of type 2 diabetes: A systematic review. Expert. Opin. Pharmacother. 2016, 17, 1463–1473. [Google Scholar] [CrossRef]
- Stewart, S.; Lois, N. Fenofibrate for Diabetic Retinopathy. Asia-Pac. J. Ophthalmol. 2018, 7, 422–426. [Google Scholar] [CrossRef]
- Kataoka, S.Y.; Lois, N.; Kawano, S.; Kataoka, Y.; Inoue, K.; Watanabe, N. Fenofibrate for diabetic retinopathy. Cochrane Database Syst. Rev. 2023, 6, Cd013318. [Google Scholar] [CrossRef]
- Raciti, L.; Formica, C.; Raciti, G.; Quartarone, A.; Calabrò, R.S. Gender and Neurosteroids: Implications for Brain Function, Neuroplasticity and Rehabilitation. Int. J. Mol. Sci. 2023, 24, 4758. [Google Scholar] [CrossRef] [PubMed]
- Hogervorst, E.; Williams, J.; Budge, M.; Barnetson, L.; Combrinck, M.; Smith, A.D. Serum total testosterone is lower in men with Alzheimer’s disease. Neuro Endocrinol. Lett. 2001, 22, 163–168. [Google Scholar]
- Hogervorst, E.; De Jager, C.; Budge, M.; Smith, A.D. Serum levels of estradiol and testosterone and performance in different cognitive domains in healthy elderly men and women. Psychoneuroendocrinology 2004, 29, 405–421. [Google Scholar] [CrossRef]
- Yeap, B.B.; Hyde, Z.; Almeida, O.P.; Norman, P.E.; Chubb, S.A.; Jamrozik, K.; Flicker, L.; Hankey, G.J. Lower testosterone levels predict incident stroke and transient ischemic attack in older men. J. Clin. Endocrinol. Metab. 2009, 94, 2353–2359. [Google Scholar] [CrossRef]
- Dong, X.; Jiang, H.; Li, S.; Zhang, D. Low Serum Testosterone Concentrations Are Associated With Poor Cognitive Performance in Older Men but Not Women. Front. Aging Neurosci. 2021, 13, 712237. [Google Scholar] [CrossRef] [PubMed]
- Pike, C.J. Sex and the development of Alzheimer’s disease. J. Neurosci. Res. 2017, 95, 671–680. [Google Scholar] [CrossRef]
- Choleris, E.; Galea, L.A.M.; Sohrabji, F.; Frick, K.M. Sex differences in the brain: Implications for behavioral and biomedical research. Neurosci. Biobehav. Rev. 2018, 85, 126–145. [Google Scholar] [CrossRef]
- Giatti, S.; Diviccaro, S.; Serafini, M.M.; Caruso, D.; Garcia-Segura, L.M.; Viviani, B.; Melcangi, R.C. Sex differences in steroid levels and steroidogenesis in the nervous system: Physiopathological role. Front. Neuroendocrinol. 2020, 56, 100804. [Google Scholar] [CrossRef]
- Combs, C.K.; Johnson, D.E.; Karlo, J.C.; Cannady, S.B.; Landreth, G.E. Inflammatory mechanisms in Alzheimer’s disease: Inhibition of beta-amyloid-stimulated proinflammatory responses and neurotoxicity by PPARgamma agonists. J. Neurosci. 2000, 20, 558–567. [Google Scholar] [CrossRef] [PubMed]
- Combs, C.K.; Bates, P.; Karlo, J.C.; Landreth, G.E. Regulation of beta-amyloid stimulated proinflammatory responses by peroxisome proliferator-activated receptor alpha. Neurochem. Int. 2001, 39, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Fuenzalida, K.; Quintanilla, R.; Ramos, P.; Piderit, D.; Fuentealba, R.A.; Martinez, G.; Inestrosa, N.C.; Bronfman, M. Peroxisome proliferator-activated receptor gamma up-regulates the Bcl-2 anti-apoptotic protein in neurons and induces mitochondrial stabilization and protection against oxidative stress and apoptosis. J. Biol. Chem. 2007, 282, 37006–37015. [Google Scholar] [CrossRef]
- Sagheddu, C.; Melis, M.; Muntoni, A.L.; Pistis, M. Repurposing Peroxisome Proliferator-Activated Receptor Agonists in Neurological and Psychiatric Disorders. Pharmaceuticals 2021, 14, 1025. [Google Scholar] [CrossRef] [PubMed]
- Chakravarthy, M.V.; Lodhi, I.J.; Yin, L.; Malapaka, R.R.; Xu, H.E.; Turk, J.; Semenkovich, C.F. Identification of a physiologically relevant endogenous ligand for PPARalpha in liver. Cell 2009, 138, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Esposito, E.; Mazzon, E.; Paterniti, I.; Dal Toso, R.; Pressi, G.; Caminiti, R.; Cuzzocrea, S. PPAR-alpha Contributes to the Anti-Inflammatory Activity of Verbascoside in a Model of Inflammatory Bowel Disease in Mice. PPAR Res. 2010, 2010, 917312. [Google Scholar] [CrossRef]
- Radler, U.; Stangl, H.; Lechner, S.; Lienbacher, G.; Krepp, R.; Zeller, E.; Brachinger, M.; Eller-Berndl, D.; Fischer, A.; Anzur, C.; et al. A combination of (ω-3) polyunsaturated fatty acids, polyphenols and L-carnitine reduces the plasma lipid levels and increases the expression of genes involved in fatty acid oxidation in human peripheral blood mononuclear cells and HepG2 cells. Ann. Nutr. Metab. 2011, 58, 133–140. [Google Scholar] [CrossRef]
- Inoue, H.; Jiang, X.-F.; Katayama, T.; Osada, S.; Umesono, K.; Namura, S. Brain protection by resveratrol and fenofibrate against stroke requires peroxisome proliferator-activated receptor α in mice. Neurosci. Lett. 2003, 352, 203–206. [Google Scholar] [CrossRef]
- Tsukamoto, T.; Nakata, R.; Tamura, E.; Kosuge, Y.; Kariya, A.; Katsukawa, M.; Mishima, S.; Ito, T.; Iinuma, M.; Akao, Y.; et al. Vaticanol C, a resveratrol tetramer, activates PPARalpha and PPARbeta/delta in vitro and in vivo. Nutr. Metab. 2010, 7, 46. [Google Scholar] [CrossRef]
- Hardie, D.G. AMPK: A target for drugs and natural products with effects on both diabetes and cancer. Diabetes 2013, 62, 2164–2172. [Google Scholar] [CrossRef]
- Wang, R.; Zhao, J.; Jin, J.; Tian, Y.; Lan, L.; Wang, X.; Zhu, L.; Wang, J. WY-14643 attenuates lipid deposition via activation of the PPARα/CPT1A axis by targeting Gly335 to inhibit cell proliferation and migration in ccRCC. Lipids Health Dis. 2022, 21, 121. [Google Scholar] [CrossRef] [PubMed]
- Yan, T.; Luo, Y.; Yan, N.; Hamada, K.; Zhao, N.; Xia, Y.; Wang, P.; Zhao, C.; Qi, D.; Yang, S.; et al. Intestinal peroxisome proliferator-activated receptor α-fatty acid-binding protein 1 axis modulates nonalcoholic steatohepatitis. Hepatology 2023, 77, 239–255. [Google Scholar] [CrossRef]
- Wen, W.; Wang, J.; Zhang, B.; Wang, J. PPARα Agonist WY-14643 Relieves Neuropathic Pain through SIRT1-Mediated Deacetylation of NF-κB. PPAR Res. 2020, 2020, 6661642. [Google Scholar] [CrossRef] [PubMed]
- Titus, C.; Hoque, M.T.; Bendayan, R. PPAR agonists for the treatment of neuroinflammatory diseases. Trends Pharmacol. Sci. 2024, 45, 9–23. [Google Scholar] [CrossRef]
- Chandra, S.; Pahan, K. Gemfibrozil, a lipid-lowering drug, attenuates amyloid plaque pathology and enhances memory via PPARα. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2018, 14, P1655–P1656. [Google Scholar] [CrossRef]
- Matlock, H.G.; Qiu, F.; Malechka, V.; Zhou, K.; Cheng, R.; Benyajati, S.; Whelchel, A.; Karamichos, D.; Ma, J.X. Pathogenic Role of PPARα Downregulation in Corneal Nerve Degeneration and Impaired Corneal Sensitivity in Diabetes. Diabetes 2020, 69, 1279–1291. [Google Scholar] [CrossRef]
- He, H.; Liang, M.; Li, L.; Luo, S.; Fang, X.; He, H.; Xiao, X.; Wu, H.; Lin, Z. PPAR-α Agonist Fenofibrate Suppressed the Formation of Ocular Surface Squamous Metaplasia Induced by Topical Benzalkonium Chloride. Investig. Ophthalmol. Vis. Sci. 2020, 61, 54. [Google Scholar] [CrossRef]
- Sarahian, N.; Mohammadi, M.T.; Darabi, S.; Faghihi, N. Fenofibrate protects the neurovascular unit and ameliorates plasma corticosterone levels in pentylenetetrazole-induced kindling seizure in mice. Brain Res. 2021, 1758, 147343. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.X.; He, J.H.; Cui, Z.Q.; Yang, T.; Sun, X.H. PPAR-α Agonist GW7647 Protects Against Oxidative Stress and Iron Deposit via GPx4 in a Transgenic Mouse Model of Alzheimer’s Diseases. ACS Chem. Neurosci. 2022, 13, 207–216. [Google Scholar] [CrossRef]
- Jamwal, S.; Blackburn, J.K.; Elsworth, J.D. PPARγ/PGC1α signaling as a potential therapeutic target for mitochondrial biogenesis in neurodegenerative disorders. Pharmacol. Ther. 2021, 219, 107705. [Google Scholar] [CrossRef]
- García-González, L.; Pilat, D.; Baranger, K.; Rivera, S. Emerging Alternative Proteinases in APP Metabolism and Alzheimer’s Disease Pathogenesis: A Focus on MT1-MMP and MT5-MMP. Front. Aging Neurosci. 2019, 11, 244. [Google Scholar] [CrossRef] [PubMed]
- Corbett, G.T.; Gonzalez, F.J.; Pahan, K. Activation of peroxisome proliferator-activated receptor α stimulates ADAM10-mediated proteolysis of APP. Proc. Natl. Acad. Sci. USA 2015, 112, 8445–8450. [Google Scholar] [CrossRef] [PubMed]
- Barbiero, J.K.; Ramos, D.C.; Boschen, S.; Bassani, T.; Da Cunha, C.; Vital, M. Fenofibrate promotes neuroprotection in a model of rotenone-induced Parkinson’s disease. Behav. Pharmacol. 2022, 33, 513–526. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Segura, I.; Santiago-Balmaseda, A.; Rodríguez-Hernández, L.D.; Morales-Martínez, A.; Martínez-Becerril, H.A.; Martínez-Gómez, P.A.; Delgado-Minjares, K.M.; Salinas-Lara, C.; Martínez-Dávila, I.A.; Guerra-Crespo, M.; et al. PPARs and Their Neuroprotective Effects in Parkinson’s Disease: A Novel Therapeutic Approach in α-Synucleinopathy? Int. J. Mol. Sci. 2023, 24, 3264. [Google Scholar] [CrossRef] [PubMed]
- Watson, G.S.; Cholerton, B.A.; Reger, M.A.; Baker, L.D.; Plymate, S.R.; Asthana, S.; Fishel, M.A.; Kulstad, J.J.; Green, P.S.; Cook, D.G.; et al. Preserved cognition in patients with early Alzheimer disease and amnestic mild cognitive impairment during treatment with rosiglitazone: A preliminary study. Am. J. Geriatr. Psychiatry Off. J. Am. Assoc. Geriatr. Psychiatry 2005, 13, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Tufano, M.; Pinna, G. Is There a Future for PPARs in the Treatment of Neuropsychiatric Disorders? Molecules 2020, 25, 1062. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, P.; Rosa-Neto, P.; Rochford, J.; Hamel, E. Pioglitazone improves reversal learning and exerts mixed cerebrovascular effects in a mouse model of Alzheimer’s disease with combined amyloid-β and cerebrovascular pathology. PLoS ONE 2013, 8, e68612. [Google Scholar] [CrossRef] [PubMed]
- Risner, M.E.; Saunders, A.M.; Altman, J.F.; Ormandy, G.C.; Craft, S.; Foley, I.M.; Zvartau-Hind, M.E.; Hosford, D.A.; Roses, A.D. Efficacy of rosiglitazone in a genetically defined population with mild-to-moderate Alzheimer’s disease. Pharmacogenomics J. 2006, 6, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Geldmacher, D.S.; Fritsch, T.; McClendon, M.J.; Landreth, G. A randomized pilot clinical trial of the safety of pioglitazone in treatment of patients with Alzheimer disease. Arch. Neurol. 2011, 68, 45–50. [Google Scholar] [CrossRef]
- Galimberti, D.; Scarpini, E. Pioglitazone for the treatment of Alzheimer’s disease. Expert. Opin. Investig. Drugs 2017, 26, 97–101. [Google Scholar] [CrossRef]
- Rasgon, N.L.; Kenna, H.A.; Williams, K.E.; Powers, B.; Wroolie, T.; Schatzberg, A.F. Rosiglitazone add-on in treatment of depressed patients with insulin resistance: A pilot study. Sci. World J. 2010, 10, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Fan, X.; Wang, J.; Liu, D.; Freudenreich, O.; Goff, D.; Henderson, D.C. Rosiglitazone and cognitive function in clozapine-treated patients with schizophrenia: A pilot study. Psychiatry Res. 2012, 200, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Rolland, B.; Marche, K.; Cottencin, O.; Bordet, R. The PPARα Agonist Fenofibrate Reduces Prepulse Inhibition Disruption in a Neurodevelopmental Model of Schizophrenia. Schizophr. Res. Treat. 2012, 2012, 839853. [Google Scholar] [CrossRef]
- De Felice, M.; Melis, M.; Aroni, S.; Muntoni, A.L.; Fanni, S.; Frau, R.; Devoto, P.; Pistis, M. The PPARα agonist fenofibrate attenuates disruption of dopamine function in a maternal immune activation rat model of schizophrenia. CNS Neurosci. Ther. 2019, 25, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Maekawa, M.; Ohnishi, T.; Balan, S.; Matsuoka, S.; Iwamoto, K.; Iwayama, Y.; Ohba, H.; Watanabe, A.; Hisano, Y.; et al. Peroxisome proliferator-activated receptor α as a novel therapeutic target for schizophrenia. EbioMedicine 2020, 62, 103130. [Google Scholar] [CrossRef] [PubMed]
- Duszka, K.; Wahli, W. Enteric Microbiota⁻Gut⁻Brain Axis from the Perspective of Nuclear Receptors. Int. J. Mol. Sci. 2018, 19, 2210. [Google Scholar] [CrossRef] [PubMed]
- Agirman, G.; Yu, K.B.; Hsiao, E.Y. Signaling inflammation across the gut-brain axis. Science 2021, 374, 1087–1092. [Google Scholar] [CrossRef]
- Mulak, A. Bile Acids as Key Modulators of the Brain-Gut-Microbiota Axis in Alzheimer’s Disease. J. Alzheimers Dis. 2021, 84, 461–477. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Żulińska, S.; Strosznajder, A.K.; Strosznajder, J.B. Current View on PPAR-α and Its Relation to Neurosteroids in Alzheimer’s Disease and Other Neuropsychiatric Disorders: Promising Targets in a Therapeutic Strategy. Int. J. Mol. Sci. 2024, 25, 7106. https://doi.org/10.3390/ijms25137106
Żulińska S, Strosznajder AK, Strosznajder JB. Current View on PPAR-α and Its Relation to Neurosteroids in Alzheimer’s Disease and Other Neuropsychiatric Disorders: Promising Targets in a Therapeutic Strategy. International Journal of Molecular Sciences. 2024; 25(13):7106. https://doi.org/10.3390/ijms25137106
Chicago/Turabian StyleŻulińska, Sylwia, Anna K. Strosznajder, and Joanna B. Strosznajder. 2024. "Current View on PPAR-α and Its Relation to Neurosteroids in Alzheimer’s Disease and Other Neuropsychiatric Disorders: Promising Targets in a Therapeutic Strategy" International Journal of Molecular Sciences 25, no. 13: 7106. https://doi.org/10.3390/ijms25137106
APA StyleŻulińska, S., Strosznajder, A. K., & Strosznajder, J. B. (2024). Current View on PPAR-α and Its Relation to Neurosteroids in Alzheimer’s Disease and Other Neuropsychiatric Disorders: Promising Targets in a Therapeutic Strategy. International Journal of Molecular Sciences, 25(13), 7106. https://doi.org/10.3390/ijms25137106