Dopamine Pharmacodynamics: New Insights
Abstract
1. Introduction
2. The Dopamine System
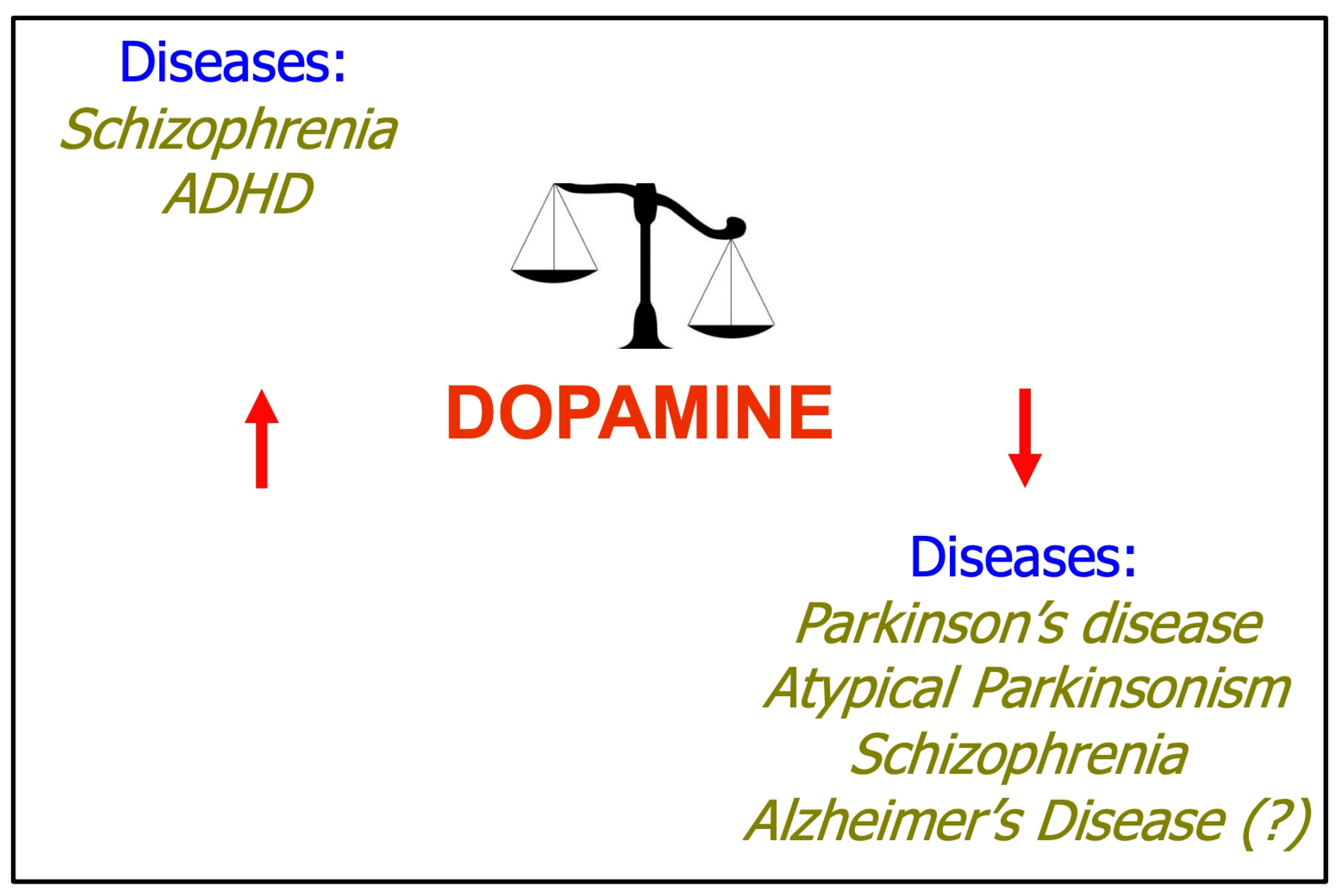
3. Dopamine System and Human Disease
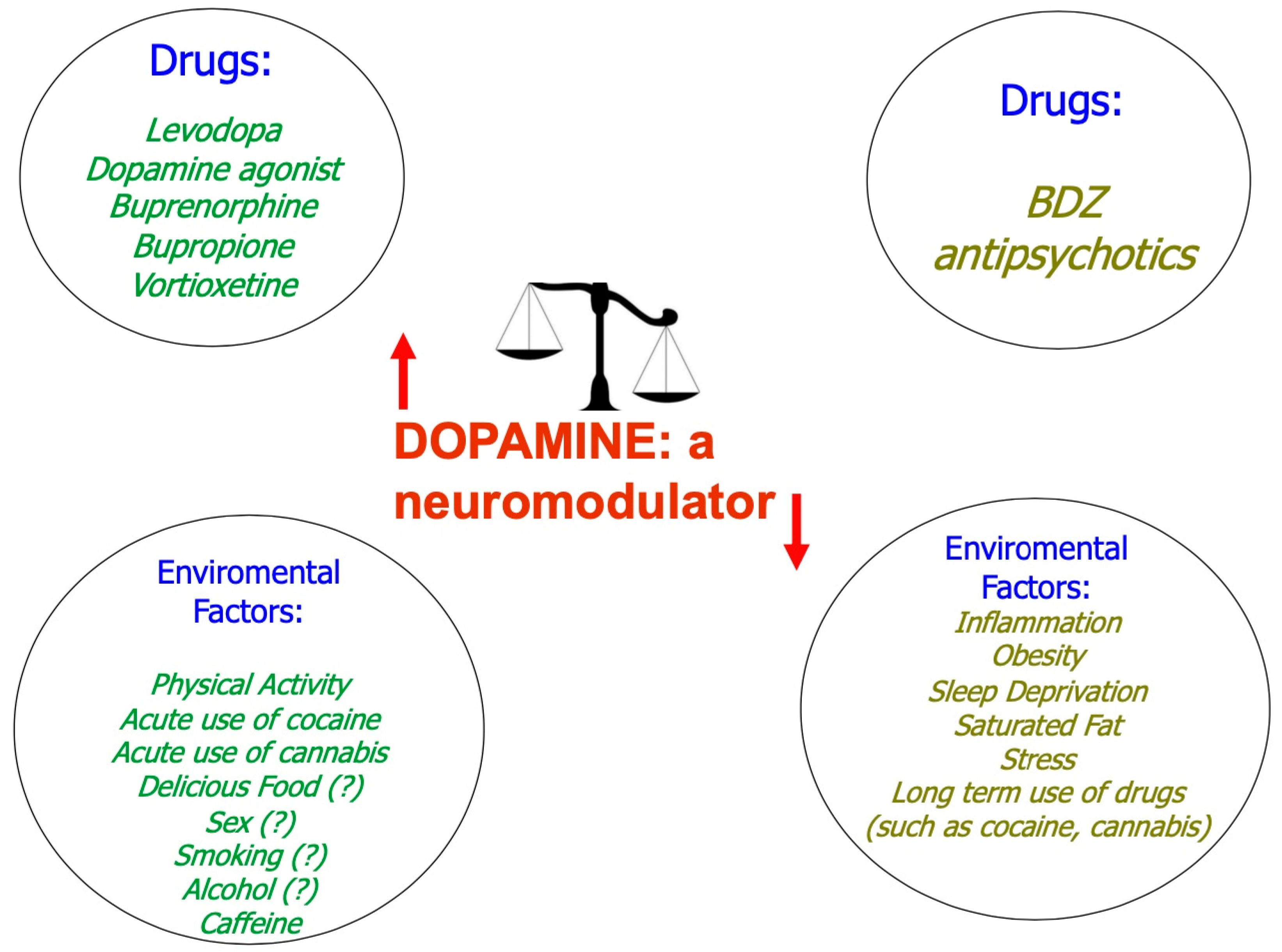
4. Dopamine as a Neuromodulator
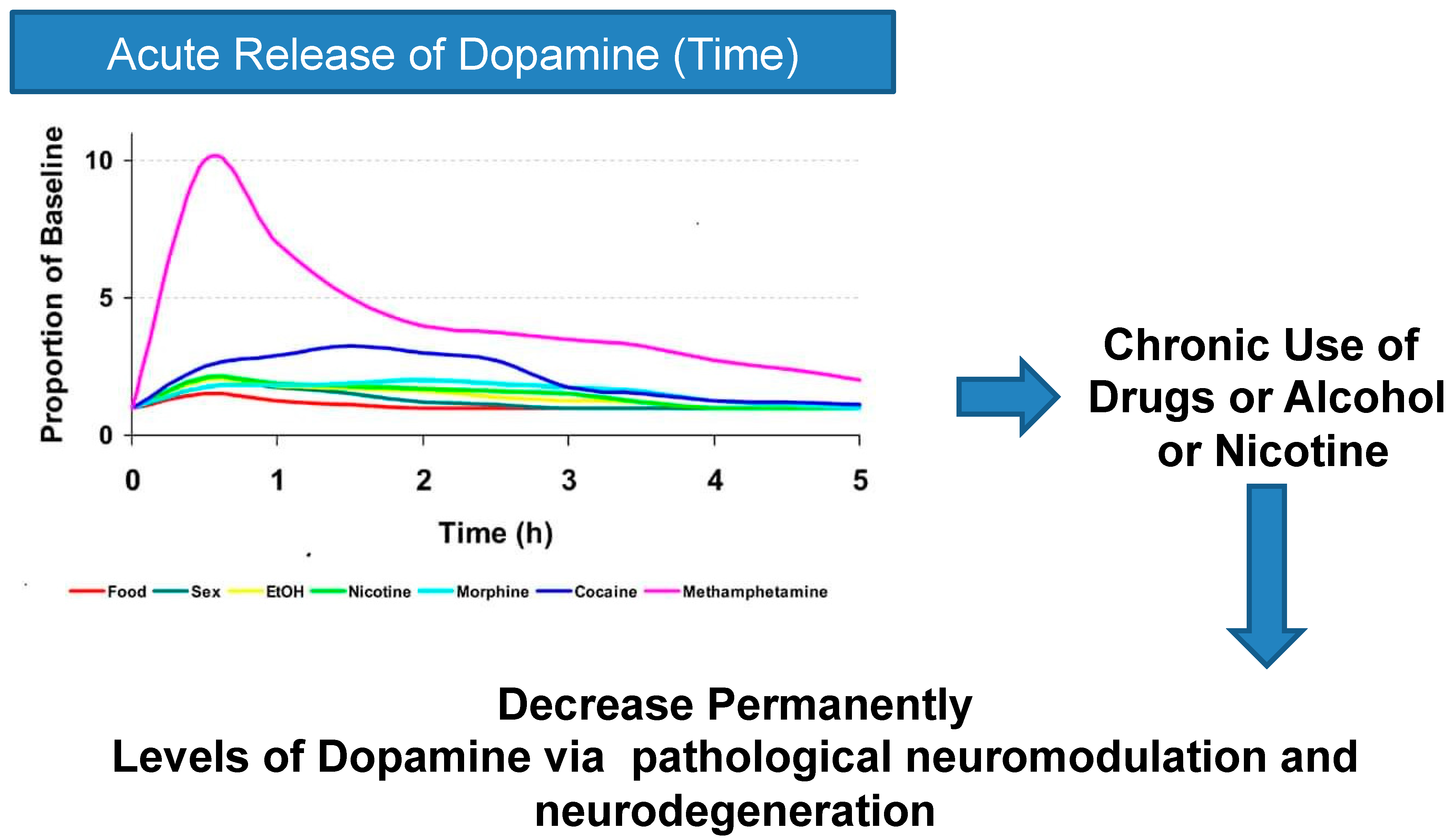
5. From Bad Neuromodulation to Neurotoxicity
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Carlsson, A.; Lindqvist, M.; Magnusson, T. 3,4-Dihydroxyphenylalanine and 5-hydroxytryptophan as reserpine antagonists. Nature 1957, 180, 1200. [Google Scholar] [CrossRef]
- Carlsson, A.; Lindqvist, M.; Magnusson, T.; Waldeck, B. On the presence of 3-hydroxytyramine in brain. Science 1958, 127, 471. [Google Scholar] [CrossRef] [PubMed]
- Lauretani, F.; Testa, C.; Salvi, M.; Zucchini, I.; Lorenzi, B.; Tagliaferri, S.; Cattabiani, C.; Maggio, M. Reward System Dysfunction and the Motoric-Cognitive Risk Syndrome in Older Persons. Biomedicines 2022, 10, 808. [Google Scholar] [CrossRef]
- Liu, C.; Cai, X.; Ritzau-Jost, A.; Kramer, P.F.; Li, Y.; Khaliq, Z.M.; Hallermann, S.; Kaeser, P.S. An action potential initiation mechanism in distal axons for the control of dopamine release. Science 2022, 375, 1378–1385. [Google Scholar] [CrossRef]
- Lauretani, F.; Ruffini, L.; Testa, C.; Salvi, M.; Scarlattei, M.; Baldari, G.; Zucchini, I.; Lorenzi, B.; Cattabiani, C.; Maggio, M. Cognitive and Behavior Deficits in Parkinson’s Disease with Alteration of FDG-PET Irrespective of Age. Geriatrics 2021, 6, 110. [Google Scholar] [CrossRef] [PubMed]
- Chinta, S.J.; Andersen, J.K. Dopaminergic neurons. Int. J. Biochem Cell. Biol. 2005, 37, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Costa, K.M.; Schoenbaum, G. Dopamine. Curr. Biol. 2022, 32, R817–R824. [Google Scholar] [CrossRef] [PubMed]
- Schultz, W. Neuronal Reward and Decision Signals: From Theories to Data. Physiol. Rev. 2015, 95, 853–951. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.O.; Battagello, D.S.; Cardoso, A.R.; Hauser, D.N.; Bittencourt, J.C. Dopamine: Functions, Signaling, and Association with Neurological Diseases. Cell. Mol. Neurobiol. 2019, 39, 31–59. [Google Scholar] [CrossRef]
- Seeman, P.; Van Tol, H.H. Dopamine receptor pharmacology. Curr. Opin. Neurol. Neurosurg. 1993, 6, 602–608. [Google Scholar] [CrossRef]
- Zhang, M.; Liang, L.H.; Lu, Y.J.; Cao, X. G protein-coupled receptor-associated sorting proteins: Function and relevant disorders. Yi Chuan 2020, 42, 713–724. [Google Scholar] [PubMed]
- Neve, K.A.; Seamans, J.K.; Trantham-Davidson, H. Dopamine receptor signaling. J. Recept. Signal. Transduct. Res. 2004, 24, 165–205. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Chen, F.; Li, B.; Hu, Z. Neurophysiology of HCN channels: From cellular functions to multiple regulations. Prog. Neurobiol. 2014, 112, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Salatino-Oliveira, A.; Rohde, L.A.; Hutz, M.H. The dopamine transporter role in psychiatric phenotypes. Am. J. Med. Genet. Part B Neuropsychiatr. Genet. 2018, 177, 211–231. [Google Scholar] [CrossRef] [PubMed]
- Andersen, P.H.; Gingrich, J.A.; Bates, M.D.; Dearry, A.; Falardeau, P.; Senogles, S.E.; Caron, M.G. Dopamine receptor subtypes: Beyond the D1/D2 classification. Trends Pharmacol. Sci. 1990, 11, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Tong, Q.; Cui, X.; Xu, H.; Zhang, X.; Hu, S.; Huang, F.; Xiao, L. D1 receptor-expressing neurons in ventral tegmental area alleviate mouse anxiety-like behaviors via glutamatergic projection to lateral septum. Mol. Psychiatry 2022, 28, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Tavares, G.; Marques, D.; Barra, C.; Rosendo-Silva, D.; Costa, A.; Rodrigues, T.; Gasparini, P.; Melo, B.; Sacramento, J.; Seiça, R.; et al. Dopamine D2 receptor agonist, bromocriptine, remodels adipose tissue dopaminergic signalling and upregulates catabolic pathways, improving metabolic profile in type 2 diabetes. Mol. Metab. 2021, 51, 101241. [Google Scholar] [CrossRef] [PubMed]
- Heidbreder, C.A.; Gardner, E.L.; Xi, Z.-X.; Thanos, P.K.; Mugnaini, M.; Hagan, J.J.; Ashby, C.R. The role of central dopamine D3 receptors in drug addiction: A review of pharmacological evidence. Brain Res. Rev. 2005, 49, 77–105. [Google Scholar] [CrossRef]
- Altamirano-Espinoza, A.H.; González-Hernández, A.; Manrique-Maldonado, G.; A Marichal-Cancino, B.; Ruiz-Salinas, I.; Villalón, C.M. The role of dopamine D2, but not D3 or D4, receptor subtypes, in quinpirole-induced inhibition of the cardioaccelerator sympathetic outflow in pithed rats. Br. J. Pharmacol. 2013, 170, 1102–1111. [Google Scholar] [CrossRef]
- Zeng, C.; Yang, Z.; Asico, L.D.; Jose, P.A. Regulation of blood pressure by D5 dopamine receptors. Cardiovasc. Hematol. Agents Med. Chem. 2007, 5, 241–248. [Google Scholar] [CrossRef]
- Simon, H.H.; Bhatt, L.; Gherbassi, D.; Sgadó, P.; Alberí, L. Midbrain dopaminergic neurons: Determination of their developmental fate by transcription factors. Ann. N. Y. Acad. Sci. 2003, 991, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Floresca, C.Z.; Schetz, J.A. Dopamine receptor microdomains involved in molecular recognition and the regulation of drug affinity and function. J. Recept. Signal Transduct. Res. 2004, 24, 207–239, Erratum in J. Recept. Signal Transduct. Res. 2006, 26, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Everett, A.C.; Graul, B.E.; Ronström, J.W.; Robinson, J.K.; Watts, D.B.; España, R.A.; Siciliano, C.A.; Yorgason, J.T. Effectiveness and Relationship between Biased and Unbiased Measures of Dopamine Release and Clearance. ACS Chem. Neurosci. 2022, 13, 1534–1548. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, S.; Mortensen, O.V. Discovery and Development of Monoamine Transporter Ligands. Adv. Neurobiol. 2023, 30, 101–129. [Google Scholar]
- Schultz, W. Dopamine neurons and their role in reward mechanisms. Curr. Opin. Neurobiol. 1997, 7, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Courtney, C.D.; Pamukcu, A.; Chan, C.S. Cell and circuit complexity of the external globus pallidus. Nat. Neurosci. 2023, 26, 1147–1159. [Google Scholar] [CrossRef] [PubMed]
- Floresco, S.B.; West, A.R.; Ash, B.; Moore, H.; Grace, A.A. Afferent modulation of dopamine neuron firing differentially regulates tonic and phasic dopamine transmission. Nat. Neurosci. 2003, 6, 968–973. [Google Scholar] [CrossRef] [PubMed]
- Feltenstein, M.W.; See, R.E.; Fuchs, R.A. Neural Substrates and Circuits of Drug Addiction. Cold Spring Harb. Perspect. Med. 2021, 11, a039628. [Google Scholar] [CrossRef] [PubMed]
- Pierce, R.C.; Kumaresan, V. The mesolimbic dopamine system: The final common pathway for the reinforcing effect of drugs of abuse? Neurosci. Biobehav. Rev. 2006, 30, 215–238. [Google Scholar] [CrossRef]
- Engel, J.A.; Jerlhag, E. Alcohol: Mechanisms along the mesolimbic dopamine system. Prog. Brain Res. 2014, 211, 201–233. [Google Scholar]
- McCutcheon, R.A.; Abi-Dargham, A.; Howes, O.D. Schizophrenia, Dopamine and the Striatum: From Biology to Symptoms. Trends Neurosci. 2019, 42, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Korchounov, A.; Meyer, M.F.; Krasnianski, M. Postsynaptic nigrostriatal dopamine receptors and their role in movement regulation. J. Neural Transm. 2010, 117, 1359–1369. [Google Scholar] [CrossRef] [PubMed]
- Prensa, L.; Giménez-Amaya, J.M.; Parent, A.; Bernácer, J.; Cebrián, C. The nigrostriatal pathway: Axonal collateralization and compartmental specificity. J. Neural. Transm. Suppl. 2009, 73, 49–58. [Google Scholar]
- Seip-Cammack, K.M.; Young, J.J.; Young, M.E.; Shapiro, M.L. Partial lesion of the nigrostriatal dopamine pathway in rats impairs egocentric learning but not spatial learning or behavioral flexibility. Behav. Neurosci. 2017, 131, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Hagelberg, N.; Jääskeläinen, S.K.; Martikainen, I.K.; Mansikka, H.; Forssell, H.; Scheinin, H.; Hietala, J.; Pertovaara, A. Striatal dopamine D2 receptors in modulation of pain in humans: A review. Eur. J. Pharmacol. 2004, 500, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Gorelova, N.; Mulholland, P.J.; Chandler, L.J.; Seamans, J.K. The glutamatergic component of the mesocortical pathway emanating from different subregions of the ventral midbrain. Cereb. Cortex 2012, 22, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Knable, M.B.; Weinberger, D.R. Dopamine, the prefrontal cortex and schizophrenia. J. Psychopharmacol. 1997, 11, 123–131. [Google Scholar] [CrossRef]
- Mohamadian, M.; Fallah, H.; Ghofrani-Jahromi, Z.; Rahimi-Danesh, M.; Shokouhi Qare Saadlou, M.S.; Vaseghi, S. Mood and behavior regulation: Interaction of lithium and dopaminergic system. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2023, 396, 1339–1359. [Google Scholar] [CrossRef] [PubMed]
- Grattan, D.R. 60 years of neuroendocrinology: The hypothalamo-prolactin axis. J. Endocrinol. 2015, 226, T101–T122. [Google Scholar] [CrossRef]
- Gudelsky, G.A.; Berry, S.A.; Meltzer, H.Y. Actions of typical and atypical antipsychotics on tuberoinfundibular dopamine neurons. Psychopharmacol. Bull. 1989, 25, 377–382. [Google Scholar]
- Volkow, N.D.; Wise, R.A.; Baler, R. The dopamine motive system: Implications for drug and food addiction. Nat. Rev. Neurosci. 2017, 18, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.H. Stress and the dopaminergic reward system. Exp. Mol. Med. 2020, 52, 1879–1890. [Google Scholar] [CrossRef] [PubMed]
- Belujon, P.; Grace, A.A. Dopamine System Dysregulation in Major Depressive Disorders. Int. J. Neuropsychopharmacol. 2017, 20, 1036–1046. [Google Scholar] [CrossRef]
- Kahn, R.S.; Sommer, I.E.; Murray, R.M.; Meyer-Lindenberg, A.; Weinberger, D.R.; Cannon, T.D.; O’Donnovan, M.; Correll, C.U.; Kane, J.M.; van Os, J. Schizophrenia. Nat. Rev. Dis. Primers 2015, 1, 15067. [Google Scholar] [CrossRef]
- Howes, O.D.; Williams, M.; Ibrahim, K.; Leung, G.; Egerton, A.; McGuire, P.K.; Turkheimer, F. Midbrain dopamine function in schizophrenia and depression: A post-mortem and positron emission tomographic imaging study. Brain 2013, 136 Pt 11, 3242–3251. [Google Scholar] [CrossRef] [PubMed]
- Posner, J.; Polanczyk, G.V.; Sonuga-Barke, E. Attention-deficit hyperactivity disorder. Lancet 2020, 395, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Grayson, M. Parkinson’s disease. Nature 2016, 538, S1. [Google Scholar] [CrossRef]
- Lauretani, F.; Testa, C.; Salvi, M.; Zucchini, I.; Giallauria, F.; Maggio, M. Clinical Evaluation of Sleep Disorders in Parkinson’s Disease. Brain Sci. 2023, 13, 609. [Google Scholar] [CrossRef]
- Armstrong, M.J.; Okun, M.S. Diagnosis and Treatment of Parkinson Disease: A Review. JAMA 2020, 323, 548–560. [Google Scholar] [CrossRef]
- Dunlop, B.W.; Nemeroff, C.B. The role of dopamine in the pathophysiology of depression. Arch. Gen. Psychiatry 2007, 64, 327–337. [Google Scholar] [CrossRef]
- Morimoto, S.; Takao, M.; Hatsuta, H.; Nishina, Y.; Komiya, T.; Sengoku, R.; Nakano, Y.; Uchino, A.; Sumikura, H.; Saito, Y.; et al. Homovanillic acid and 5-hydroxyindole acetic acid as biomarkers for dementia with Lewy bodies and coincident Alzheimer’s disease: An autopsy-confirmed study. PLoS ONE 2017, 12, e0171524. [Google Scholar] [CrossRef] [PubMed]
- Reitz, C.; Mayeux, R. Alzheimer disease: Epidemiology, diagnostic criteria, risk factors and biomarkers. Biochem. Pharmacol. 2014, 88, 640–651. [Google Scholar] [CrossRef] [PubMed]
- Cepeda, C.; Murphy, K.P.; Parent, M.; Levine, M.S. The role of dopamine in Huntington’s disease. Prog. Brain Res. 2014, 211, 235–254. [Google Scholar] [PubMed]
- The Huntington’s Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 1993, 72, 971–983. [Google Scholar] [CrossRef]
- Nikolaus, S.; Antke, C.; Müller, H.W. In vivo imaging of synaptic function in the central nervous system: I. Movement disorders and dementia. Behav. Brain Res. 2009, 204, 1–31. [Google Scholar] [CrossRef] [PubMed]
- Jakel, R.J.; Maragos, W.F. Neuronal cell death in Huntington’s disease: A potential role for dopamine. Trends Neurosci. 2000, 23, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Cyr, M.; Sotnikova, T.D.; Gainetdinov, R.R.; Caron, M.G. Dopamine enhances motor and neuropathological consequences of polyglutamine expanded huntingtin. FASEB J. 2006, 20, 2541–2543. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Le Moal, M. Drug abuse: Hedonic homeostatic dysregulation. Science 1997, 278, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Sanna, P.P.; Bloom, F.E. Neuroscience of addiction. Neuron 1998, 21, 467–476. [Google Scholar] [CrossRef]
- Nutt, D.J.; Lingford-Hughes, A.; Erritzoe, D.; Stokes, P.R. The dopamine theory of addiction: 40 years of highs and lows. Nat. Rev. Neurosci. 2015, 16, 305–312. [Google Scholar] [CrossRef]
- Wise, R.A.; Robble, M.A. Dopamine and Addiction. Annu. Rev. Psychol. 2020, 71, 79–106. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A.; Jordan, C.J. Dopamine, behavior, and addiction. J. Biomed Sci. 2021, 28, 83. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Nong, Z.; Li, Y.; Huang, J.; Chen, C.; Huang, L. Role of Dopamine Signaling in Drug Addiction. Curr. Top. Med. Chem. 2017, 17, 2440–2455. [Google Scholar] [CrossRef] [PubMed]
- Kenny, P.J. Common cellular and molecular mechanisms in obesity and drug addiction. Nat. Rev. Neurosci. 2011, 12, 638–651. [Google Scholar] [CrossRef] [PubMed]
- Johnson, P.M.; Kenny, P.J. Dopamine D2 receptors in addiction-like reward dysfunction and compulsive eating in obese rats. Nat. Neurosci. 2010, 13, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Baik, J.H. Dopamine signaling in food addiction: Role of dopamine D2 receptors. BMB Rep. 2013, 46, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Puig, S.; Noble, F.; Benturquia, N. Short- and long-lasting behavioral and neurochemical adaptations: Relationship with patterns of cocaine administration and expectation of drug effects in rats. Transl. Psychiatry 2012, 2, e175. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Michaelides, M.; Baler, R. The Neuroscience of Drug Reward and Addiction. Physiol. Rev. 2019, 99, 2115–2140. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; Fowler, J.S.; Wang, G.J.; Swanson, J.M. Dopamine in drug abuse and addiction: Results from imaging studies and treatment implications. Mol. Psychiatry 2004, 9, 557–569. [Google Scholar] [CrossRef]
- Robison, A.J.; Nestler, E.J. Transcriptional and epigenetic mechanisms of addiction. Nat. Rev. Neurosci. 2011, 12, 623–637. [Google Scholar] [CrossRef]
- Jones, S.; Bonci, A. Synaptic plasticity and drug addiction. Curr. Opin. Pharmacol. 2005, 5, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Franklin, T.R.; Acton, P.D.; A Maldjian, J.; Gray, J.D.; Croft, J.R.; A Dackis, C.; O’brien, C.P.; Childress, A.R. Decreased gray matter concentration in the insular, orbitofrontal, cingulate, and temporal cortices of cocaine patients. Biol. Psychiatry 2002, 51, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Siciliano, C.A.; Calipari, E.S.; Ferris, M.J.; Jones, S.R. Adaptations of presynaptic dopamine terminals induced by psychostimulant self-administration. ACS Chem. Neurosci. 2015, 6, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Espana, R.A.; Jones, S.R. Presynaptic dopamine modulation by stimulant self-administration. Front. Biosci. (Sch. Ed.) 2013, 5, 261–276. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Haile, C.N.; Kosten, T.R.; Kosten, T.A. Pharmacogenetic treatments for drug addiction: Cocaine, amphetamine and methamphetamine. Am. J. Drug Alcohol Abuse 2009, 35, 161–177. [Google Scholar] [CrossRef] [PubMed]
- Vena, A.A.; Zandy, S.L.; Cofresí, R.U.; Gonzales, R.A. Behavioral, neurobiological, and neurochemical mechanisms of ethanol self-administration: A translational review. Pharmacol. Ther. 2020, 212, 107573. [Google Scholar] [CrossRef] [PubMed]
- Salinas, A.G.; Mateo, Y.; Carlson, V.C.C.; Stinnett, G.S.; Luo, G.; Seasholtz, A.F.; Grant, K.A.; Lovinger, D.M. Long-term alcohol consumption alters dorsal striatal dopamine release and regulation by D2 dopamine receptors in rhesus macaques. Neuropsychopharmacology 2021, 46, 1432–1441. [Google Scholar] [CrossRef] [PubMed]
- Clauss, N.J.; Mayer, F.P.; Owens, W.A.; Vitela, M.; Clarke, K.M.; Bowman, M.A.; Horton, R.E.; Gründemann, D.; Schmid, D.; Holy, M.; et al. Ethanol inhibits dopamine uptake via organic cation transporter 3: Implications for ethanol and cocaine co-abuse. Mol. Psychiatry 2023, 28, 2934–2945. [Google Scholar] [CrossRef] [PubMed]
- Gibb, S.L.; Jeanblanc, J.; Barak, S.; Yowell, Q.V.; Yaka, R.; Ron, D. Lyn Kinase Regulates Mesolimbic Dopamine Release: Implication for Alcohol Reward. J. Neurosci. 2011, 31, 2180–2187. [Google Scholar] [CrossRef]
- Beckley, J.T.; Laguesse, S.; Phamluong, K.; Morisot, N.; Wegner, S.A.; Ron, D. The First Alcohol Drink Triggers mTORC1-Dependent Synaptic Plasticity in Nucleus Accumbens Dopamine D1 Receptor Neurons. J. Neurosci. 2016, 36, 701–713. [Google Scholar] [CrossRef]
- van Erp, A.M.; Miczek, K.A. Increased accumbal dopamine during daily alcohol consumption and subsequent aggressive behavior in rats. Psychopharmacology 2007, 191, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Schindler, A.G.; Soden, M.E.; Zweifel, L.S.; Clark, J.J. Reversal of Alcohol-Induced Dysregulation in Dopamine Network Dynamics May Rescue Maladaptive Decision-making. J. Neurosci. 2016, 36, 3698–3708. [Google Scholar] [CrossRef]
- Schneider, M.L.; Moore, C.F.; Barnhart, T.E.; Larson, J.A.; DeJesus, O.T.; Mukherjee, J.; Nickles, R.J.; Converse, A.K.; Roberts, A.D.; Kraemer, G.W. Moderate-Level Prenatal Alcohol Exposure Alters Striatal Dopamine System Function in Rhesus Monkeys. Alcohol. Clin. Exp. Res. 2005, 29, 1685–1697. [Google Scholar] [CrossRef]
- Anderson, R.I.; Becker, H.C. Role of the Dynorphin/Kappa Opioid Receptor System in the Motivational Effects of Ethanol. Alcohol. Clin. Exp. Res. 2017, 41, 1402–1418. [Google Scholar] [CrossRef]
- Siciliano, C.A.; Calipari, E.S.; Carlson, V.C.C.; Helms, C.M.; Lovinger, D.M.; Grant, K.A.; Jones, S.R. Voluntary Ethanol Intake Predicts κ-Opioid Receptor Supersensitivity and Regionally Distinct Dopaminergic Adaptations in Macaques. J. Neurosci. 2015, 35, 5959–5968. [Google Scholar] [CrossRef]
- Rose, J.H.; Karkhanis, A.N.; Chen, R.; Gioia, D.; Lopez, M.F.; Becker, H.C.; McCool, B.A.; Jones, S.R. Supersensitive Kappa Opioid Receptors Promotes Ethanol Withdrawal-Related Behaviors and Reduce Dopamine Signaling in the Nucleus Accumbens. Int. J. Neuropsychopharmacol. 2015, 19. [Google Scholar] [CrossRef] [PubMed]
- Rose, J.H.; Karkhanis, A.N.; Steiniger-Brach, B.; Jones, S.R. Distinct Effects of Nalmefene on Dopamine Uptake Rates and Kappa Opioid Receptor Activity in the Nucleus Accumbens Following Chronic Intermittent Ethanol Exposure. Int. J. Mol. Sci. 2016, 17, 1216. [Google Scholar] [CrossRef] [PubMed]
- Koob, G.F.; Volkow, N.D. Neurobiology of addiction: A neurocircuitry analysis. Lancet Psychiatry 2016, 3, 760–773. [Google Scholar] [CrossRef] [PubMed]
- Colasanti, A.; Searle, G.E.; Long, C.J.; Hill, S.P.; Reiley, R.R.; Quelch, D.; Erritzoe, D.; Tziortzi, A.C.; Reed, L.J.; Lingford-Hughes, A.R.; et al. Endogenous Opioid Release in the Human Brain Reward System Induced by Acute Amphetamine Administration. Biol. Psychiatry 2012, 72, 371–377. [Google Scholar] [CrossRef]
- Matsumura, K.; Nicot, A.; Choi, I.B.; Asokan, M.; Le, N.N.; Natividad, L.A.; Dobbs, L.K. Endogenous opioid system modulates conditioned cocaine reward in a sex-dependent manner. Addict. Biol. 2023, 28, e13328. [Google Scholar] [CrossRef]
- Vink, J.M. Genetics of Addiction: Future Focus on Gene × Environment Interaction? J. Stud. Alcohol Drugs 2016, 77, 684–687. [Google Scholar] [CrossRef] [PubMed]
- Ducci, F.; Goldman, D. The genetic basis of addictive disorders. Psychiatr. Clin. North Am. 2012, 35, 495–519. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Jiang, Y.; Wedow, R.; Li, Y.; Brazel, D.M.; Chen, F.; Datta, G.; Davila-Velderrain, J.; McGuire, D.; Tian, C.; et al. Association studies of up to 1.2 million individuals yield new insights into the genetic etiology of tobacco and alcohol use. Nat. Genet. 2019, 51, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Testa, C.; DI Lorenzo, A.; Parlato, A.; D’Ambrosio, G.; Merolla, A.; Pacileo, M.; Iannuzzo, G.; Gentile, M.; Nugara, C.; Sarullo, F.M. Exercise for slowing the progression of atherosclerotic process: Effects on inflammatory markers. Panminerva Medica 2021, 63, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yuan, T.F. Exercise and substance abuse. Int. Rev. Neurobiol. 2019, 147, 269–280. [Google Scholar] [PubMed]
- Lynch, W.J.; Peterson, A.B.; Sanchez, V.; Abel, J.; Smith, M.A. Exercise as a novel treatment for drug addiction: A neurobiological and stage-dependent hypothesis. Neurosci. Biobehav. Rev. 2013, 37, 1622–1644. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, J.; Farney, M.R.; Elrod, N.M.; Henderson, C.E.; Weiss, E.P. Exercise as an Adjunctive Treatment for Substance Use Disorders: Rationale and Intervention Description. J. Subst. Abus. Treat. 2017, 72, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Nock, N.L.; Minnes, S.; Alberts, J.L. Neurobiology of substance use in adolescents and potential therapeutic effects of exercise for prevention and treatment of substance use disorders. Birth Defects Res. 2017, 109, 1711–1729. [Google Scholar] [CrossRef]
- Foley, P.B. Dopamine in psychiatry: A historical perspective. J. Neural Transm. 2019, 126, 473–479. [Google Scholar] [CrossRef]
- Iversen, S.D.; Iversen, L.L. Dopamine: 50 years in perspective. Trends Neurosci. 2007, 30, 188–193. [Google Scholar] [CrossRef]
- Schultz, W. Dopamine reward prediction error coding. Dialog- Clin. Neurosci. 2016, 18, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Peters, K.Z.; Cheer, J.F.; Tonini, R. Modulating the Neuromodulators: Dopamine, Serotonin, and the Endocannabinoid System. Trends Neurosci. 2021, 44, 464–477. [Google Scholar] [CrossRef] [PubMed]
- Taylor, N.E.; Pei, J.; Zhang, J.; Vlasov, K.Y.; Davis, T.; Taylor, E.; Weng, F.J.; Van Dort, C.J.; Solt, K.; Brown, E.N. The Role of Glutamatergic and Dopaminergic Neurons in the Periaqueductal Gray/Dorsal Raphe: Separating Analgesia and Anxiety. Eneuro 2019, 6, ENEURO.0018-18.2019. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, R.; Johansson, J.; Nordin, K.; Rieckmann, A.; Wåhlin, A.; Nyberg, L.; Bäckman, L.; Salami, A. Dopamine D1-Receptor Organization Contributes to Functional Brain Architecture. J. Neurosci. 2024, 44. [Google Scholar] [CrossRef] [PubMed]
- Brzosko, Z.; Schultz, W.; Paulsen, O. Retroactive modulation of spike timing-dependent plasticity by dopamine. Elife 2015, 4, e09685. [Google Scholar] [CrossRef]
- Ferguson, K.A.; Cardin, J.A. Mechanisms underlying gain modulation in the cortex. Nat. Rev. Neurosci. 2020, 21, 80–92. [Google Scholar] [CrossRef] [PubMed]
- Salinas, E.; Sejnowski, T.J. Gain modulation in the central nervous system: Where behavior, neurophysiology, and computation meet. Neuroscientist 2001, 7, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Seamans, J.K.; Yang, C.R. The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog. Neurobiol. 2004, 74, 1–58, Erratum in Prog. Neurobiol. 2004, 74, 321. [Google Scholar] [CrossRef]
- El-Ghundi, M.; O’Dowd, B.F.; George, S.R. Insights into the role of dopamine receptor systems in learning and memory. Rev. Neurosci. 2007, 18, 37–66. [Google Scholar] [CrossRef]
- Rieckmann, A.; Karlsson, S.; Fischer, H.; Bäckman, L. Caudate dopamine D1 receptor density is associated with individual differences in frontoparietal connectivity during working memory. J. Neurosci. 2011, 31, 14284–14290. [Google Scholar] [CrossRef]
- Papenberg, G.; Jonasson, L.; Karalija, N.; Johansson, J.; Köhncke, Y.; Salami, A.; Andersson, M.; Axelsson, J.; Wahlin, A.; Riklund, K. Mapping the landscape of human dopamine D2/3 receptors with [11C]raclopride. Brain Struct Funct. 2019, 224, 2871–2882. [Google Scholar] [CrossRef] [PubMed]
- Zald, D.H.; Woodward, N.D.; Cowan, R.L.; Riccardi, P.; Ansari, M.S.; Baldwin, R.M.; Smith, C.E.; Hakyemez, H.; Li, R.; Kessler, R.M. The interrelationship of dopamine D2-like receptor availability in striatal and extrastriatal brain regions in healthy humans: A principal component analysis of [18F]fallypride binding. NeuroImage 2010, 51, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Froudist-Walsh, S.; Bliss, D.P.; Ding, X.; Rapan, L.; Niu, M.; Knoblauch, K.; Zilles, K.; Kennedy, H.; Palomero-Gallagher, N.; Wang, X.-J. A dopamine gradient controls access to distributed working memory in the large-scale monkey cortex. Neuron 2021, 109, 3500. [Google Scholar] [CrossRef] [PubMed]
- Nyberg, L.; Karalija, N.; Salami, A.; Andersson, M.; Wåhlin, A.; Kaboovand, N.; Köhncke, Y.; Axelsson, J.; Rieckmann, A.; Papenberg, G. Dopamine D2 receptor availability is linked to hippocampal-caudate functional connectivity and episodic memory. Proc. Natl. Acad. Sci. USA 2016, 113, 7918–7923. [Google Scholar] [CrossRef]
- Roffman, J.L.; Tanner, A.S.; Eryilmaz, H.; Rodriguez-Thompson, A.; Silverstein, N.J.; Ho, N.F.; Nitenson, A.Z.; Chonde, D.B.; Greve, D.N.; Abi-Dargham, A.; et al. Dopamine D1 signaling organizes network dynamics underlying working memory. Sci. Adv. 2016, 2, e1501672. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.; Nordin, K.; Pedersen, R.; Karalija, N.; Papenberg, G.; Andersson, M.; Korkki, S.M.; Riklund, K.; Guitart-Masip, M.; Rieckmann, A. Biphasic patterns of age-related differences in dopamine D1 receptors across the adult lifespan. Cell Rep. 2023, 42, 113107. [Google Scholar] [CrossRef] [PubMed]
- Rieckmann, A.; Karlsson, S.; Karlsson, P.; Brehmer, Y.; Fischer, H.; Farde, L.; Nyberg, L.; Bäckman, L. Dopamine D1 Receptor Associations within and between Dopaminergic Pathways in Younger and Elderly Adults: Links to Cognitive Performance. Cereb. Cortex 2011, 21, 2023–2032. [Google Scholar] [CrossRef] [PubMed]
- Speranza, L.; di Porzio, U.; Viggiano, D.; de Donato, A.; Volpicelli, F. Dopamine: The Neuromodulator of Long-Term Synaptic Plasticity, Reward and Movement Control. Cells 2021, 10, 735. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Kaeser, P.S. Mechanisms and regulation of dopamine release. Curr. Opin. Neurobiol. 2019, 57, 46–53. [Google Scholar] [CrossRef]
- Meissner, W.G.; Remy, P.; Giordana, C.; Maltête, D.; Derkinderen, P.; Houéto, J.-L.; Anheim, M.; Benatru, I.; Boraud, T.; Brefel-Courbon, C.; et al. Trial of Lixisenatide in Early Parkinson’s Disease. N. Engl. J. Med. 2024, 390, 1176–1185. [Google Scholar] [CrossRef]
- McGarry, A.; Rosanbalm, S.; Leinonen, M.; Olanow, C.W.; To, D.; Bell, A.; Lee, D.; Chang, J.; Dubow, J.; Dhall, R. Safety, tolerability, and efficacy of NLY01 in early untreated Parkinson’s disease: A randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2024, 23, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Athauda, D.; Maclagan, K.; Skene, S.S.; Bajwa-Joseph, M.; Letchford, D.; Chowdhury, K.; Hibbert, S.; Budnik, N.; Zampedri, L.; Dickson, J. Exenatide once weekly versus placebo in Parkinson’s disease: A randomised, double-blind, placebo-controlled trial. Lancet 2017, 390, 1664–1675. [Google Scholar] [CrossRef] [PubMed]
- McFarthing, K.; Rafaloff, G.; Baptista, M.; Mursaleen, L.; Fuest, R.; Wyse, R.K.; Stott, S.R. Parkinson’s Disease Drug Therapies in the Clinical Trial Pipeline: 2022 Update. J. Park. Dis. 2022, 12, 1073–1082. [Google Scholar] [CrossRef] [PubMed]
- Klausen, M.K.; Jensen, M.E.; Møller, M.; Le Dous, N.; Jensen, A.Ø.; Zeeman, V.A.; Johannsen, C.F.; Lee, A.; Thomsen, G.K.; Macoveanu, J. Exenatide once weekly for alcohol use disorder investigated in a randomized, placebo-controlled clinical trial. J. Clin. Investig. 2022, 7, e159863. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.; Li, H.; Kong, X.; Wang, Y.; Sun, T.; Wang, F. Possible Mechanisms Underlying the Effects of Glucagon-Like Peptide-1 Receptor Agonist on Cocaine Use Disorder. Front. Pharmacol. 2022, 13, 819470. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Nestler, E.J. The neural rejuvenation hypothesis of cocaine addiction. Trends Pharmacol. Sci. 2014, 35, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Hattori, A.; Luo, Y.; Umegaki, H.; Munoz, J.; Roth, G.S. Intrastriatal injection of dopamine results in DNA damage and apoptosis in rats. NeuroReport 1998, 9, 2569–2572. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Hattori, A.; Munoz, J.; Qin, Z.H.; Roth, G.S. Intrastriatal dopamine injection induces apoptosis through oxidation-involved activation of transcription factors AP-1 and NF-kappaB in rats. Mol. Pharmacol. 1999, 56, 254–264. [Google Scholar] [CrossRef] [PubMed]
- Cheng, N.-N.; Maeda, T.; Kume, T.; Kaneko, S.; Kochiyama, H.; Akaike, A.; Goshima, Y.; Misu, Y. Differential neurotoxicity induced by l-DOPA and dopamine in cultured striatal neurons. Brain Res. 1996, 743, 278–283. [Google Scholar] [CrossRef]
- Shinkai, T.; Zhang, L.; Mathias, S.A.; Roth, G.S. Dopamine induces apoptosis in cultured rat striatal neurons; possible mechanism of D2-dopamine receptor neuron loss during aging. J. Neurosci. Res. 1997, 47, 393–399. [Google Scholar] [CrossRef]
- McLaughlin, B.A.; Nelson, D.; Erecińska, M.; Chesselet, M.F. Toxicity of dopamine to striatal neurons in vitro and potentiation of cell death by a mitochondrial inhibitor. J. Neurochem. 1998, 70, 2406–2415. [Google Scholar] [CrossRef]
- Iwatsubo, K.; Suzuki, S.; Li, C.; Tsunematsu, T.; Nakamura, F.; Okumura, S.; Sato, M.; Minamisawa, S.; Toya, Y.; Umemura, S.; et al. Dopamine induces apoptosis in young, but not in neonatal, neurons via Ca2+-dependent signal. Am. J. Physiol. Physiol. 2007, 293, C1498–C1508. [Google Scholar] [CrossRef]
- Rosenberg, P.A. Catecholamine toxicity in cerebral cortex in dissociated cell culture. J. Neurosci. 1988, 8, 2887–2894. [Google Scholar] [CrossRef]
- Tanaka, M.; Sotomatsu, A.; Kanai, H.; Hirai, S. Dopa and dopamine cause cultured neuronal death in the presence of iron. J. Neurol. Sci. 1991, 101, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Mytilineou, C.; Han, S.K.; Cohen, G. Toxic and protective effects of L-dopa on mesencephalic cell cultures. J. Neurochem. 1993, 61, 1470–1478. [Google Scholar] [CrossRef] [PubMed]
- Hoyt, K.R.; Reynolds, I.J.; Hastings, T.G. Mechanisms of dopamine-induced cell death in cultured rat forebrain neurons: Interactions with and differences from glutamate-induced cell death. Exp. Neurol. 1997, 143, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Umegaki, H.; Wang, X.; Abe, R.; Roth, G.S. Dopamine induces apoptosis through an oxidation-involved SAPK/JNK activation pathway. J. Biol. Chem. 1998, 273, 3756–3764. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.C.; Gunasekar, P.G.; Borowitz, J.L.; Isom, G.E. Dopamine-induced apoptosis is mediated by oxidative stress and Is enhanced by cyanide in differentiated PC12 cells. J. Neurochem. 2000, 74, 2296–2304. [Google Scholar] [CrossRef]
- Graham, D.G. Oxidative pathways for catecholamines in the genesis of neuromelanin and cytotoxic quinones. Mol. Pharmacol. 1978, 14, 633–643. [Google Scholar]
- Graham, D.G.; Tiffany, S.M.; Bell, W.R., Jr.; Gutknecht, W.F. Autoxidation versus covalent binding of quinones as the mechanism of toxicity of dopamine, 6-hydroxydopamine, and related compounds toward C1300 neuroblastoma cells in vitro. Mol. Pharmacol. 1978, 14, 644–653. [Google Scholar]
- Cadet, J.L.; Brannock, C. Free radicals and the pathobiology of brain dopamine systems. Neurochem. Int. 1998, 32, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Ben-Shachar, D.; Zuk, R.; Gazawi, H.; Ljubuncic, P. Dopamine toxicity involves mitochondrial complex I inhibition: Implications to dopamine-related neuropsychiatric disorders. Biochem. Pharmacol. 2004, 67, 1965–1974. [Google Scholar] [CrossRef] [PubMed]
- Wersinger, C.; Chen, J.; Sidhu, A. Bimodal induction of dopamine-mediated striatal neurotoxicity is mediated through both activation of D1 dopamine receptors and autoxidation. Mol. Cell. Neurosci. 2004, 25, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wersinger, C.; Sidhu, A. Chronic stimulation of D1 dopamine receptors in human SK-N-MC neuroblastoma cells induces nitric-oxide synthase activation and cytotoxicity. J. Biol. Chem. 2003, 278, 28089–28100. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.-X.; Kleitz, H.; Deng, X.; Ladenheim, B.; Peng, X.-Q.; Li, X.; Gardner, E.; Stein, E.; Cadet, J. A single high dose of methamphetamine increases cocaine self-administration by depletion of striatal dopamine in rats. Neuroscience 2009, 161, 392–402. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Cadet, J.L. Methamphetamine administration causes overexpression of nNOS in the mouse striatum. Brain Res. 1999, 851, 254–257. [Google Scholar] [CrossRef]
- Zhu, J.P.; Xu, W.; Angulo, J.A. Methamphetamine-induced cell death: Selective vulnerability in neuronal subpopulations of the striatum in mice. Neuroscience 2006, 140, 607–622. [Google Scholar] [CrossRef] [PubMed]
- Krasnova, I.N.; Cadet, J.L. Methamphetamine toxicity and messengers of death. Brain Res. Rev. 2009, 60, 379–407. [Google Scholar] [CrossRef] [PubMed]
- Jayanthi, S.; Deng, X.; Ladenheim, B.; McCoy, M.T.; Cluster, A.; Cai, N.-S.; Cadet, J.L. Calcineurin/NFAT-induced up-regulation of the Fas ligand/Fas death pathway is involved in methamphetamine-induced neuronal apoptosis. Proc. Natl. Acad. Sci. USA 2005, 102, 868–873. [Google Scholar] [CrossRef]
- Guo, C.; Chen, L.; Wang, Y. Substance abuse and neurodegenerative diseases: Focus on ferroptosis. Arch. Toxicol. 2023, 97, 1519–1528. [Google Scholar] [CrossRef]
- Mukherjee, S. Alcoholism and its effects on the central nervous system. Curr. Neurovascular Res. 2013, 10, 256–262. [Google Scholar] [CrossRef]
- Pascual, M.; Calvo-Rodriguez, M.; Núñez, L.; Villalobos, C.; Ureña, J.; Guerri, C. Toll-like receptors in neuroinflammation, neurodegeneration, and alcohol-induced brain damage. IUBMB Life 2021, 73, 900–915. [Google Scholar] [CrossRef]
- Liu, M.; Guo, S.; Huang, D.; Hu, D.; Wu, Y.; Zhou, W.; Song, W. Chronic Alcohol Exposure Alters Gene Expression and Neurodegeneration Pathways in the Brain of Adult Mice. J. Alzheimer’s Dis. 2022, 86, 315–331. [Google Scholar] [CrossRef]
- Anand, S.K.; Ahmad, M.H.; Sahu, M.R.; Subba, R.; Mondal, A.C. Detrimental Effects of Alcohol-Induced Inflammation on Brain Health: From Neurogenesis to Neurodegeneration. Cell. Mol. Neurobiol. 2023, 43, 1885–1904. [Google Scholar] [CrossRef]
- Venkataraman, A.; Kalk, N.; Sewell, G.; Ritchie, C.W.; Lingford-Hughes, A. Alcohol and Alzheimer’s Disease-Does Alcohol Dependence Contribute to Beta-Amyloid Deposition, Neuroinflammation and Neurodegeneration in Alzheimer’s Disease? Alcohol Alcohol. 2017, 52, 151–158, Erratum in Alcohol Alcohol. 2017, 52, 158. [Google Scholar] [PubMed]
- Sinforiani, E.; Zucchella, C.; Pasotti, C.; Casoni, F.; Bini, P.; Costa, A. The effects of alcohol on cognition in the elderly: From protection to neurodegeneration. Funct. Neurol. 2011, 26, 103–106. [Google Scholar]
- Söderpalm, B.; Ericson, M. Alcohol and the dopamine system. Int. Rev. Neurobiol. 2024, 175, 21–73. [Google Scholar]
- Söderpalm, B.; Ericson, M. Neurocircuitry involved in the development of alcohol addiction: The dopamine system and its access points. Curr. Top. Behav. Neurosci. 2013, 13, 127–161. [Google Scholar]
- Daviet, R.; Aydogan, G.; Jagannathan, K.; Spilka, N.; Koellinger, P.D.; Kranzler, H.R.; Nave, G.; Wetherill, R.R. Associations between alcohol consumption and gray and white matter volumes in the UK Biobank. Nat. Commun. 2022, 13, 1175. [Google Scholar] [CrossRef] [PubMed]
- Jeon, K.H.; Han, K.; Jeong, S.M.; Park, J.; Yoo, J.E.; Yoo, J.; Lee, J.; Kim, S.; Shin, D.W. Changes in Alcohol Consumption and Risk of Dementia in a Nationwide Cohort in South Korea. JAMA Netw. Open 2023, 6, e2254771. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lauretani, F.; Giallauria, F.; Testa, C.; Zinni, C.; Lorenzi, B.; Zucchini, I.; Salvi, M.; Napoli, R.; Maggio, M.G. Dopamine Pharmacodynamics: New Insights. Int. J. Mol. Sci. 2024, 25, 5293. https://doi.org/10.3390/ijms25105293
Lauretani F, Giallauria F, Testa C, Zinni C, Lorenzi B, Zucchini I, Salvi M, Napoli R, Maggio MG. Dopamine Pharmacodynamics: New Insights. International Journal of Molecular Sciences. 2024; 25(10):5293. https://doi.org/10.3390/ijms25105293
Chicago/Turabian StyleLauretani, Fulvio, Francesco Giallauria, Crescenzo Testa, Claudia Zinni, Beatrice Lorenzi, Irene Zucchini, Marco Salvi, Raffaele Napoli, and Marcello Giuseppe Maggio. 2024. "Dopamine Pharmacodynamics: New Insights" International Journal of Molecular Sciences 25, no. 10: 5293. https://doi.org/10.3390/ijms25105293
APA StyleLauretani, F., Giallauria, F., Testa, C., Zinni, C., Lorenzi, B., Zucchini, I., Salvi, M., Napoli, R., & Maggio, M. G. (2024). Dopamine Pharmacodynamics: New Insights. International Journal of Molecular Sciences, 25(10), 5293. https://doi.org/10.3390/ijms25105293








