Revolution in Cancer Treatment: How Are Intelligently Designed Nanostructures Changing the Game?
Abstract
1. Introduction
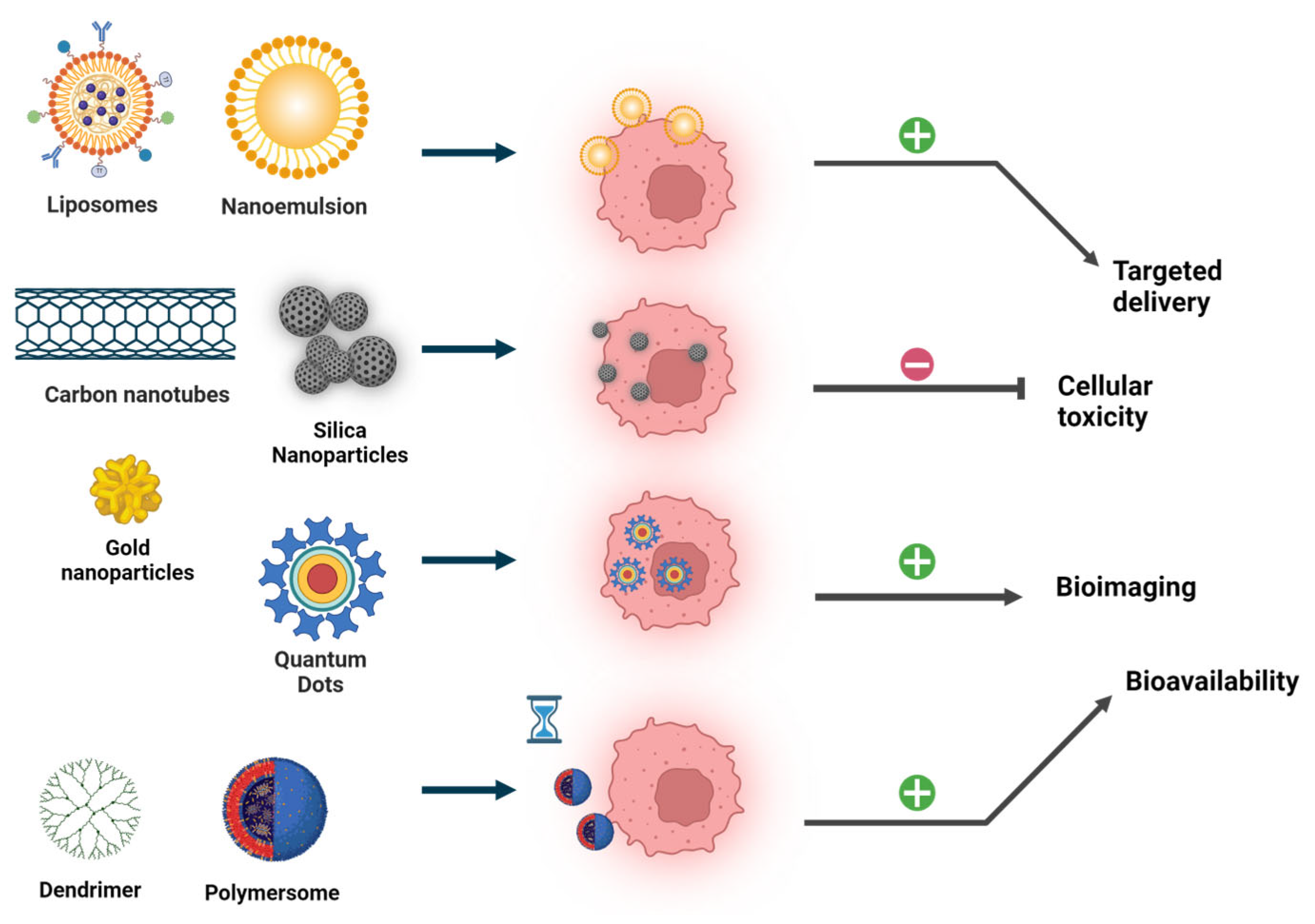
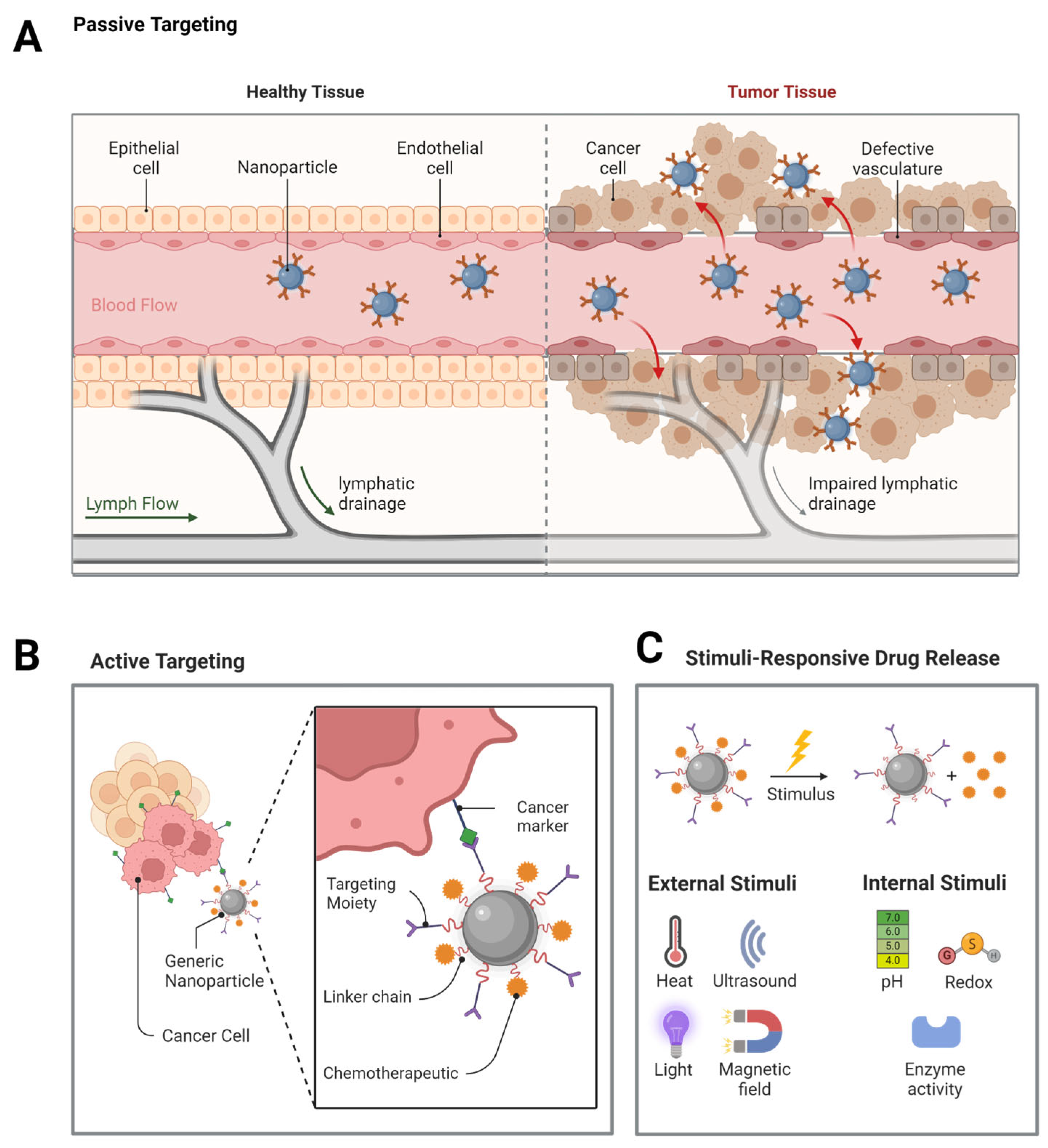
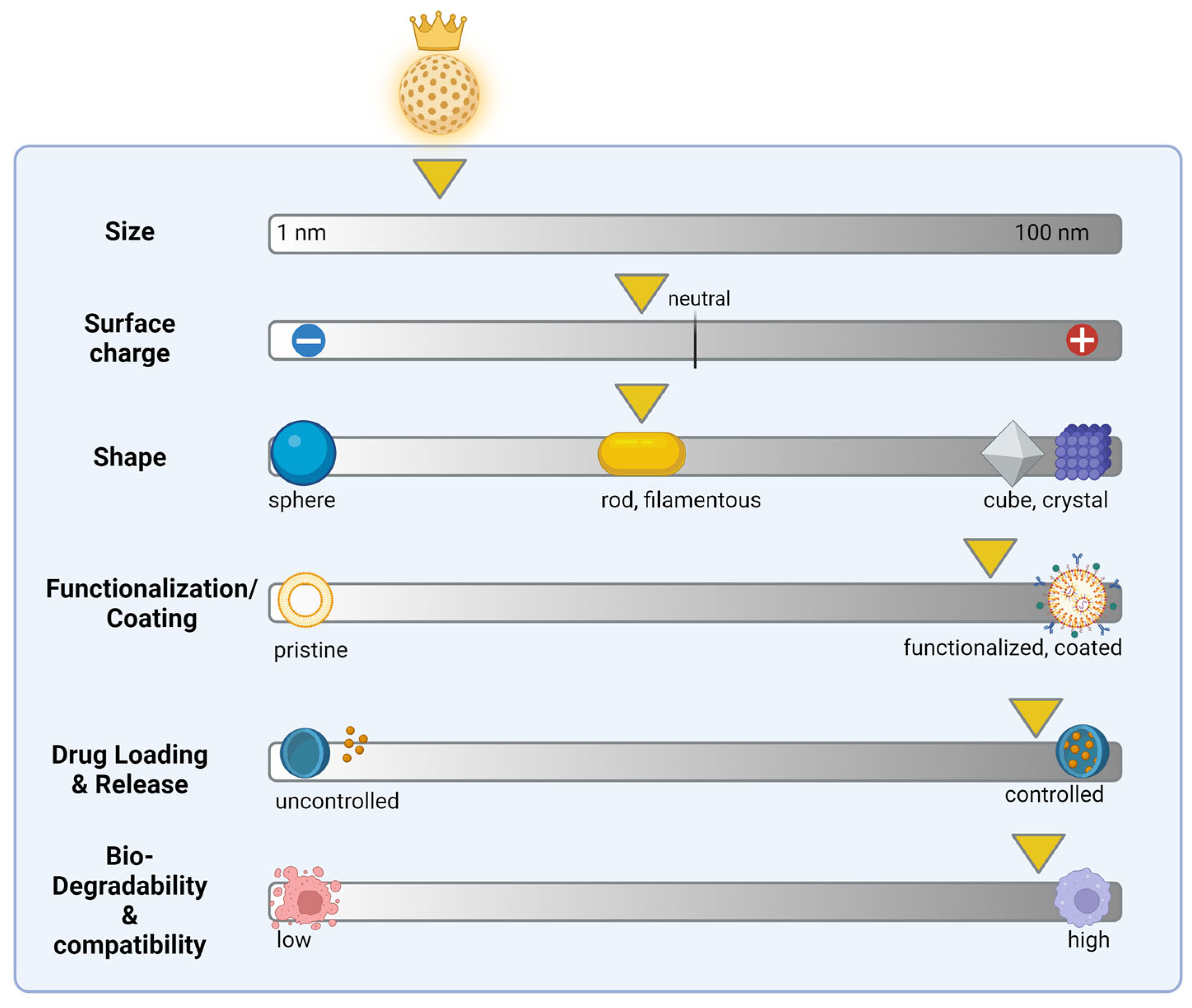
2. Selection and Design Nanostructures
3. The Protein Corona—A Crucial Factor in Nanomedicine
3.1. Size
3.2. Shape
3.3. Charge and Chemistry
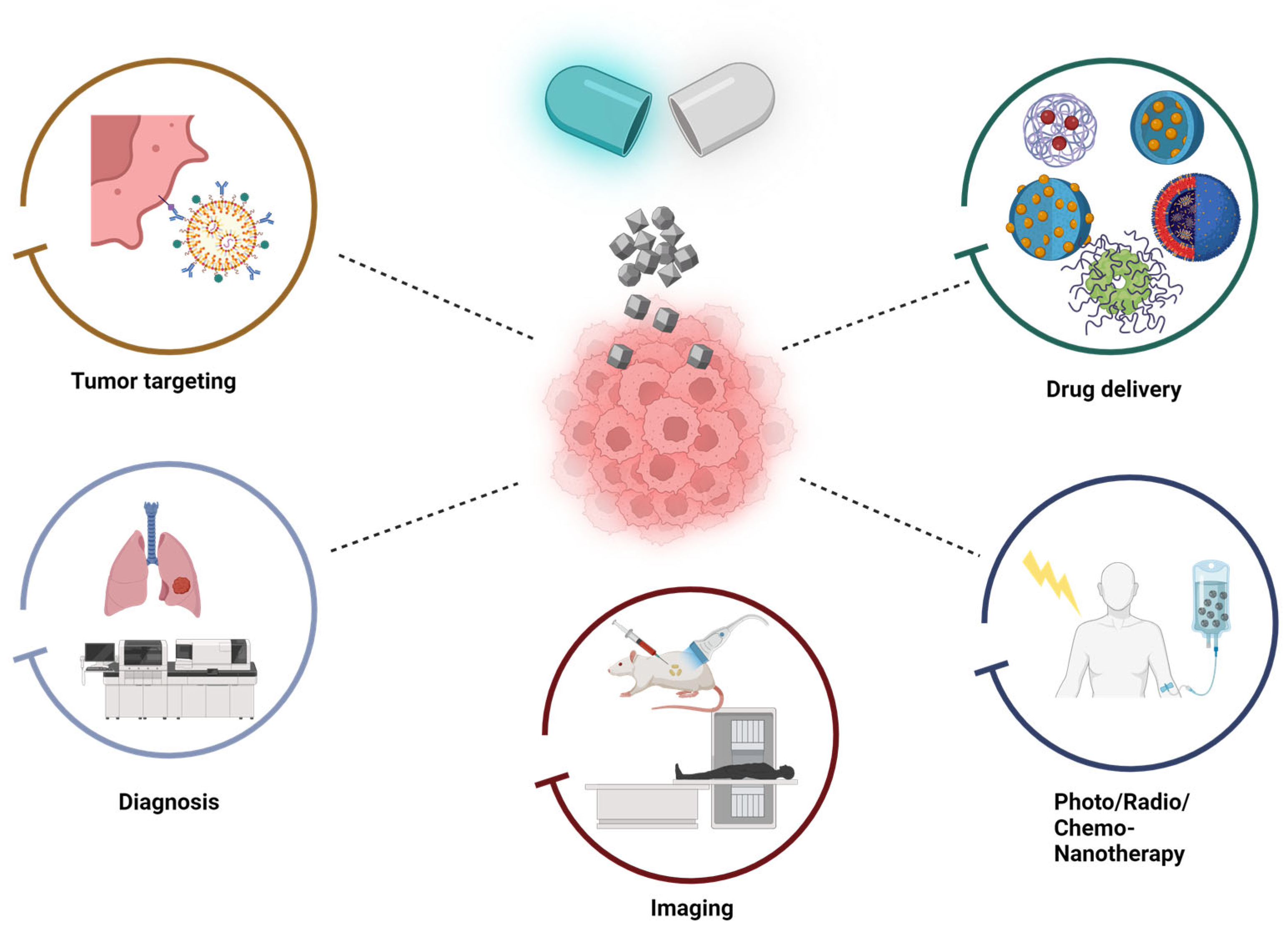
4. Specialized Nanostructures for Cancer Treatment
Applications
5. Shortcomings, Current Trends, and Future Perspectives
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering Precision Nanoparticles for Drug Delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124. [Google Scholar] [CrossRef] [PubMed]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of Nanoparticle Design for Overcoming Biological Barriers to Drug Delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef] [PubMed]
- Kou, L.; Bhutia, Y.D.; Yao, Q.; He, Z.; Sun, J.; Ganapathy, V. Transporter-Guided Delivery of Nanoparticles to Improve Drug Permeation across Cellular Barriers and Drug Exposure to Selective Cell Types. Front. Pharmacol. 2018, 9, 27. [Google Scholar] [CrossRef] [PubMed]
- Mitragotri, S.; Lammers, T.; Bae, Y.H.; Schwendeman, S.; De Smedt, S.; Leroux, J.-C.; Peer, D.; Kwon, I.C.; Harashima, H.; Kikuchi, A.; et al. Drug Delivery Research for the Future: Expanding the Nano Horizons and Beyond. J. Control. Release 2017, 246, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Wechsler, M.E.; Vela Ramirez, J.E.; Peppas, N.A. 110th Anniversary: Nanoparticle Mediated Drug Delivery for the Treatment of Alzheimer’s Disease: Crossing the Blood–Brain Barrier. Ind. Eng. Chem. Res. 2019, 58, 15079–15087. [Google Scholar] [CrossRef] [PubMed]
- Hua, S.; de Matos, M.B.C.; Metselaar, J.M.; Storm, G. Current Trends and Challenges in the Clinical Translation of Nanoparticulate Nanomedicines: Pathways for Translational Development and Commercialization. Front. Pharmacol. 2018, 9, 790. [Google Scholar] [CrossRef] [PubMed]
- Anselmo, A.C.; Mitragotri, S. Nanoparticles in the Clinic: An Update. Bioeng. Transl. Med. 2019, 4, e10143. [Google Scholar] [CrossRef]
- Liu, Y.; Bhattarai, P.; Dai, Z.; Chen, X. Photothermal Therapy and Photoacoustic Imaging via Nanotheranostics in Fighting Cancer. Chem. Soc. Rev. 2019, 48, 2053–2108. [Google Scholar] [CrossRef]
- Raheem, M.A.; Rahim, M.A.; Gul, I.; Zhong, X.; Xiao, C.; Zhang, H.; Wei, J.; He, Q.; Hassan, M.; Zhang, C.Y.; et al. Advances in Nanoparticles-Based Approaches in Cancer Theranostics. OpenNano 2023, 12, 100152. [Google Scholar] [CrossRef]
- Wang, S.; Liu, Y.; Feng, Y.; Zhang, J.; Swinnen, J.; Li, Y.; Ni, Y. A Review on Curability of Cancers: More Efforts for Novel Therapeutic Options Are Needed. Cancers 2019, 11, 1782. [Google Scholar] [CrossRef]
- Chan, H.-K.; Ismail, S. Side Effects of Chemotherapy among Cancer Patients in a Malaysian General Hospital: Experiences, Perceptions and Informational Needs from Clinical Pharmacists. Asian Pac. J. Cancer Prev. 2014, 15, 5305–5309. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Gao, L.; Chen, K.; Zhang, W.; Zhang, Q.; Li, Q.; Hu, K. Nanoparticles: A New Approach to Upgrade Cancer Diagnosis and Treatment. Nanoscale Res. Lett. 2021, 16, 88. [Google Scholar] [CrossRef] [PubMed]
- Roma-Rodrigues, C.; Pombo, I.; Raposo, L.; Pedrosa, P.; Fernandes, A.R.; Baptista, P.V. Nanotheranostics Targeting the Tumor Microenvironment. Front. Bioeng. Biotechnol. 2019, 7, 197. [Google Scholar] [CrossRef] [PubMed]
- Benoit, D.S.; Koo, H. Targeted, Triggered Drug Delivery to Tumor and Biofilm Microenvironments. Nanomedicine 2016, 11, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Koppers-Lalic, D.; Hogenboom, M.M.; Middeldorp, J.M.; Pegtel, D.M. Virus-Modified Exosomes for Targeted RNA Delivery; A New Approach in Nanomedicine. Adv. Drug Deliv. Rev. 2013, 65, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Elumalai, K.; Srinivasan, S.; Shanmugam, A. Review of the Efficacy of Nanoparticle-Based Drug Delivery Systems for Cancer Treatment. Biomed. Technol. 2024, 5, 109–122. [Google Scholar] [CrossRef]
- Trigueros, S. Nanoscale Metal Particles as Nanocarriers in Targeted Drug Delivery System. J. Nanomed. Res. 2016, 4, 00086. [Google Scholar] [CrossRef]
- Jaiswal, P.; Gidwani, B.; Vyas, A. Nanostructured Lipid Carriers and Their Current Application in Targeted Drug Delivery. Artif. Cells Nanomed. Biotechnol. 2016, 44, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Carrese, B.; Sanità, G.; Lamberti, A. Nanoparticles Design for Theranostic Approach in Cancer Disease. Cancers 2022, 14, 4654. [Google Scholar] [CrossRef]
- Kievit, F.M.; Zhang, M. Cancer Nanotheranostics: Improving Imaging and Therapy by Targeted Delivery Across Biological Barriers. Adv. Mater. 2011, 23, H217–H247. [Google Scholar] [CrossRef]
- Pandit, C.; Roy, A.; Ghotekar, S.; Khusro, A.; Islam, M.N.; Emran, T.B.; Lam, S.E.; Khandaker, M.U.; Bradley, D.A. Biological Agents for Synthesis of Nanoparticles and Their Applications. J. King Saud. Univ. Sci. 2022, 34, 101869. [Google Scholar] [CrossRef]
- Janjua, T.I.; Cao, Y.; Yu, C.; Popat, A. Clinical Translation of Silica Nanoparticles. Nat. Rev. Mater. 2021, 6, 1072–1074. [Google Scholar] [CrossRef]
- Anselmo, A.C.; Mitragotri, S. Nanoparticles in the Clinic. Bioeng. Transl. Med. 2016, 1, 10–29. [Google Scholar] [CrossRef] [PubMed]
- Elsabahy, M.; Wooley, K.L. Design of Polymeric Nanoparticles for Biomedical Delivery Applications. Chem. Soc. Rev. 2012, 41, 2545. [Google Scholar] [CrossRef] [PubMed]
- Schlenoff, J.B. Zwitteration: Coating Surfaces with Zwitterionic Functionality to Reduce Nonspecific Adsorption. Langmuir 2014, 30, 9625–9636. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Lai, S.K. Anti-PEG Immunity: Emergence, Characteristics, and Unaddressed Questions. WIREs Nanomed. Nanobiotechnol. 2015, 7, 655–677. [Google Scholar] [CrossRef] [PubMed]
- Graván, P.; Peña-Martín, J.; de Andrés, J.L.; Pedrosa, M.; Villegas-Montoya, M.; Galisteo-González, F.; Marchal, J.A.; Sánchez-Moreno, P. Exploring the Impact of Nanoparticle Stealth Coatings in Cancer Models: From PEGylation to Cell Membrane-Coating Nanotechnology. ACS Appl. Mater. Interfaces 2024, 16, 2058–2074. [Google Scholar] [CrossRef] [PubMed]
- Acet, Ö. Design of Enhanced Smart Delivery Systems for Therapeutic Enzymes: Kinetic and Release Performance of Dual Effected Enzyme-Loaded Nanopolymers. Catal. Lett. 2023, 153, 3174–3184. [Google Scholar] [CrossRef]
- Lin, G.; Zhang, H.; Huang, L. Smart Polymeric Nanoparticles for Cancer Gene Delivery. Mol. Pharm. 2015, 12, 314–321. [Google Scholar] [CrossRef]
- Sun, T.; Zhang, Y.S.; Pang, B.; Hyun, D.C.; Yang, M.; Xia, Y. Engineered Nanoparticles for Drug Delivery in Cancer Therapy. Angew. Chem. Int. Ed. 2014, 53, 12320–12364. [Google Scholar] [CrossRef]
- Oerlemans, C.; Bult, W.; Bos, M.; Storm, G.; Nijsen, J.F.W.; Hennink, W.E. Polymeric Micelles in Anticancer Therapy: Targeting, Imaging and Triggered Release. Pharm. Res. 2010, 27, 2569–2589. [Google Scholar] [CrossRef]
- Torchilin, V.P. Micellar Nanocarriers: Pharmaceutical Perspectives. Pharm. Res. 2006, 24, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Yokoyama, M. Polymeric Micelles as a New Drug Carrier System and Their Required Considerations for Clinical Trials. Expert. Opin. Drug Deliv. 2010, 7, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Alsehli, M. Polymeric Nanocarriers as Stimuli-Responsive Systems for Targeted Tumor (Cancer) Therapy: Recent Advances in Drug Delivery. Saudi Pharm. J. 2020, 28, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Prabhakar, U.; Maeda, H.; Jain, R.K.; Sevick-Muraca, E.M.; Zamboni, W.; Farokhzad, O.C.; Barry, S.T.; Gabizon, A.; Grodzinski, P.; Blakey, D.C. Challenges and Key Considerations of the Enhanced Permeability and Retention Effect for Nanomedicine Drug Delivery in Oncology. Cancer Res. 2013, 73, 2412–2417. [Google Scholar] [CrossRef] [PubMed]
- Yildirim, M.; Acet, Ö. Immunomodulatory Activities of PH/Temperature Sensitive Smart Naringenin-Loaded Nanopolymers on the Mammalian Macrophages. Appl. Surf. Sci. Adv. 2023, 18, 100527. [Google Scholar] [CrossRef]
- Kaushik, A.; Khan, S.; Pharasi, N.; Mani, S. Dual PH and Ultrasound Responsive Nanocarriers: A Smart Approach in Cancer Theranostics. J. Drug Deliv. Sci. Technol. 2024, 95, 105560. [Google Scholar] [CrossRef]
- Yıldırım, M.; Acet, Ö.; Yetkin, D.; Acet, B.Ö.; Karakoc, V.; Odabası, M. Anti-Cancer Activity of Naringenin Loaded Smart Polymeric Nanoparticles in Breast Cancer. J. Drug Deliv. Sci. Technol. 2022, 74, 103552. [Google Scholar] [CrossRef]
- Truong, N.P.; Whittaker, M.R.; Mak, C.W.; Davis, T.P. The Importance of Nanoparticle Shape in Cancer Drug Delivery. Expert. Opin. Drug Deliv. 2015, 12, 129–142. [Google Scholar] [CrossRef]
- Yusuf, A.; Almotairy, A.R.Z.; Henidi, H.; Alshehri, O.Y.; Aldughaim, M.S. Nanoparticles as Drug Delivery Systems: A Review of the Implication of Nanoparticles’ Physicochemical Properties on Responses in Biological Systems. Polymers 2023, 15, 1596. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Ge, X.; Wang, L.-J.; Lee, S.S.; Paulsen, M.H.; Khan, Q.M.; Khalid, Z.M.; Bhalli, J.A.; Waheed, U.; Simpson, C.D.; et al. An Ultra Low-Cost Smartphone Device for in-Situ Monitoring of Acute Organophosphorus Poisoning for Agricultural Workers. Sens. Actuators B Chem. 2018, 275, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Kang, C.; Yang, E.; Ge, X.; Du, D.; Lin, Y. A Sensitive Magnetic Nanoparticle-Based Immunoassay of Phosphorylated Acetylcholinesterase Using Protein Cage Templated Lead Phosphate for Signal Amplification with Graphite Furnace Atomic Absorption Spectrometry Detection. Analyst 2016, 141, 2278–2283. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Mei, K.; Guan, P.; Hu, X.; Zhao, Y. Protein-Based Artificial Nanosystems in Cancer Therapy. Small 2020, 16, 1907256. [Google Scholar] [CrossRef] [PubMed]
- Wan, Z.-L.; Guo, J.; Yang, X.-Q. Plant Protein-Based Delivery Systems for Bioactive Ingredients in Foods. Food Funct. 2015, 6, 2876–2889. [Google Scholar] [CrossRef] [PubMed]
- Lohcharoenkal, W.; Wang, L.; Chen, Y.C.; Rojanasakul, Y. Protein Nanoparticles as Drug Delivery Carriers for Cancer Therapy. Biomed. Res. Int. 2014, 2014, 180549. [Google Scholar] [CrossRef] [PubMed]
- Salatin, S.; Jelvehgari, M.; Maleki-Dizaj, S.; Adibkia, K. A Sight on Protein-Based Nanoparticles as Drug/Gene Delivery Systems. Ther. Deliv. 2015, 6, 1017–1029. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, J.; Hekmatara, T.; Dreis, S.; Vogel, T.; Gelperina, S.; Langer, K. Covalent Attachment of Apolipoprotein A-I and Apolipoprotein B-100 to Albumin Nanoparticles Enables Drug Transport into the Brain. J. Control. Release 2007, 118, 54–58. [Google Scholar] [CrossRef]
- Yoo, J.-W.; Irvine, D.J.; Discher, D.E.; Mitragotri, S. Bio-Inspired, Bioengineered and Biomimetic Drug Delivery Carriers. Nat. Rev. Drug Discov. 2011, 10, 521–535. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, T.; Jiang, C. Biomacromolecules as Carriers in Drug Delivery and Tissue Engineering. Acta Pharm. Sin. B 2018, 8, 34–50. [Google Scholar] [CrossRef]
- Saif, A.; Anjum, L.; Faisal, Z.; Akram, N.; Shah, Y.A.; Irfan, R.; Saeed, F.; Afzaal, M.; Asif Shah, M. Recent Advances in Protein-Based Nanoparticles and Their Applications in the Delivery of Bioactive Compounds. Int. J. Food Prop. 2023, 26, 2866–2880. [Google Scholar] [CrossRef]
- Bor, G.; Mat Azmi, I.D.; Yaghmur, A. Nanomedicines for Cancer Therapy: Current Status, Challenges and Future Prospects. Ther. Deliv. 2019, 10, 113–132. [Google Scholar] [CrossRef] [PubMed]
- Nienhaus, K.; Nienhaus, G.U. Mechanistic Understanding of Protein Corona Formation around Nanoparticles: Old Puzzles and New Insights. Small 2023, 19, 2301663. [Google Scholar] [CrossRef] [PubMed]
- Guo, F.; Luo, S.; Wang, L.; Wang, M.; Wu, F.; Wang, Y.; Jiao, Y.; Du, Y.; Yang, Q.; Yang, X.; et al. Protein Corona, Influence on Drug Delivery System and Its Improvement Strategy: A Review. Int. J. Biol. Macromol. 2024, 256, 128513. [Google Scholar] [CrossRef] [PubMed]
- Docter, D.; Westmeier, D.; Markiewicz, M.; Stolte, S.; Knauer, S.K.; Stauber, R.H. The Nanoparticle Biomolecule Corona: Lessons Learned—Challenge Accepted? Chem. Soc. Rev. 2015, 44, 6094–6121. [Google Scholar] [CrossRef] [PubMed]
- Coglitore, D.; Janot, J.-M.; Balme, S. Protein at Liquid Solid Interfaces: Toward a New Paradigm to Change the Approach to Design Hybrid Protein/Solid-State Materials. Adv. Colloid. Interface Sci. 2019, 270, 278–292. [Google Scholar] [CrossRef] [PubMed]
- Vu, V.P.; Gifford, G.B.; Chen, F.; Benasutti, H.; Wang, G.; Groman, E.V.; Scheinman, R.; Saba, L.; Moghimi, S.M.; Simberg, D. Immunoglobulin Deposition on Biomolecule Corona Determines Complement Opsonization Efficiency of Preclinical and Clinical Nanoparticles. Nat. Nanotechnol. 2019, 14, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Hadjidemetriou, M.; Al-Ahmady, Z.; Kostarelos, K. Time-Evolution of in Vivo Protein Corona onto Blood-Circulating PEGylated Liposomal Doxorubicin (DOXIL) Nanoparticles. Nanoscale 2016, 8, 6948–6957. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wang, G.; Griffin, J.I.; Brenneman, B.; Banda, N.K.; Holers, V.M.; Backos, D.S.; Wu, L.; Moghimi, S.M.; Simberg, D. Complement Proteins Bind to Nanoparticle Protein Corona and Undergo Dynamic Exchange in Vivo. Nat. Nanotechnol. 2017, 12, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Elechalawar, C.K.; Hossen, M.N.; McNally, L.; Bhattacharya, R.; Mukherjee, P. Analysing the Nanoparticle-Protein Corona for Potential Molecular Target Identification. J. Control. Release 2020, 322, 122–136. [Google Scholar] [CrossRef]
- Maiorano, G.; Sabella, S.; Sorce, B.; Brunetti, V.; Malvindi, M.A.; Cingolani, R.; Pompa, P.P. Effects of Cell Culture Media on the Dynamic Formation of Protein−Nanoparticle Complexes and Influence on the Cellular Response. ACS Nano 2010, 4, 7481–7491. [Google Scholar] [CrossRef]
- Agnihotri, T.G.; Alexander, A.; Agrawal, M.; Dubey, S.K.; Jain, A. In Vitro-in Vivo Correlation in Nanocarriers: From Protein Corona to Therapeutic Implications. J. Control. Release 2023, 354, 794–809. [Google Scholar] [CrossRef] [PubMed]
- García Vence, M.; Chantada-Vázquez, M.d.P.; Vázquez-Estévez, S.; Manuel Cameselle-Teijeiro, J.; Bravo, S.B.; Núñez, C. Potential Clinical Applications of the Personalized, Disease-Specific Protein Corona on Nanoparticles. Clin. Chim. Acta 2020, 501, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J. Protein–Nanoparticle Interaction: Corona Formation and Conformational Changes in Proteins on Nanoparticles. Int. J. Nanomed. 2020, 15, 5783–5802. [Google Scholar] [CrossRef] [PubMed]
- Klein, J. Probing the Interactions of Proteins and Nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 2029–2030. [Google Scholar] [CrossRef] [PubMed]
- Tenzer, S.; Docter, D.; Rosfa, S.; Wlodarski, A.; Kuharev, J.; Rekik, A.; Knauer, S.K.; Bantz, C.; Nawroth, T.; Bier, C.; et al. Nanoparticle Size Is a Critical Physicochemical Determinant of the Human Blood Plasma Corona: A Comprehensive Quantitative Proteomic Analysis. ACS Nano 2011, 5, 7155–7167. [Google Scholar] [CrossRef] [PubMed]
- Lacerda, S.H.D.P.; Park, J.J.; Meuse, C.; Pristinski, D.; Becker, M.L.; Karim, A.; Douglas, J.F. Interaction of Gold Nanoparticles with Common Human Blood Proteins. ACS Nano 2010, 4, 365–379. [Google Scholar] [CrossRef]
- Partikel, K.; Korte, R.; Mulac, D.; Humpf, H.-U.; Langer, K. Serum Type and Concentration Both Affect the Protein-Corona Composition of PLGA Nanoparticles. Beilstein J. Nanotechnol. 2019, 10, 1002–1015. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Sharifi, M.; Gleghorn, J.P.; Babadaei, M.M.N.; Bloukh, S.H.; Edis, Z.; Amin, M.; Bai, Q.; ten Hagen, T.L.M.; Falahati, M.; et al. Artificial Engineering of the Protein Corona at Bio-Nano Interfaces for Improved Cancer-Targeted Nanotherapy. J. Control. Release 2022, 348, 127–147. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.J.; Mortimer, G.; Schiller, T.; Musumeci, A.; Martin, D.; Minchin, R.F. Differential Plasma Protein Binding to Metal Oxide Nanoparticles. Nanotechnology 2009, 20, 455101. [Google Scholar] [CrossRef]
- Bewersdorff, T.; Glitscher, E.A.; Bergueiro, J.; Eravci, M.; Miceli, E.; Haase, A.; Calderón, M. The Influence of Shape and Charge on Protein Corona Composition in Common Gold Nanostructures. Mater. Sci. Eng. C 2020, 117, 111270. [Google Scholar] [CrossRef]
- García-Álvarez, R.; Hadjidemetriou, M.; Sánchez-Iglesias, A.; Liz-Marzán, L.M.; Kostarelos, K. In Vivo Formation of Protein Corona on Gold Nanoparticles. The Effect of Their Size and Shape. Nanoscale 2018, 10, 1256–1264. [Google Scholar] [CrossRef]
- Madathiparambil Visalakshan, R.; González García, L.E.; Benzigar, M.R.; Ghazaryan, A.; Simon, J.; Mierczynska-Vasilev, A.; Michl, T.D.; Vinu, A.; Mailänder, V.; Morsbach, S.; et al. The Influence of Nanoparticle Shape on Protein Corona Formation. Small 2020, 16, 2000285. [Google Scholar] [CrossRef] [PubMed]
- Cho, E.C.; Au, L.; Zhang, Q.; Xia, Y. The Effects of Size, Shape, and Surface Functional Group of Gold Nanostructures on Their Adsorption and Internalization by Cells. Small 2010, 6, 517–522. [Google Scholar] [CrossRef] [PubMed]
- Karaman, D.S.; Desai, D.; Senthilkumar, R.; Johansson, E.M.; Råtts, N.; Odén, M.; Eriksson, J.E.; Sahlgren, C.; Toivola, D.M.; Rosenholm, J.M. Shape Engineering vs Organic Modification of Inorganic Nanoparticles as a Tool for Enhancing Cellular Internalization. Nanoscale Res. Lett. 2012, 7, 358. [Google Scholar] [CrossRef]
- Gratton, S.E.A.; Ropp, P.A.; Pohlhaus, P.D.; Luft, J.C.; Madden, V.J.; Napier, M.E.; DeSimone, J.M. The Effect of Particle Design on Cellular Internalization Pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 11613–11618. [Google Scholar] [CrossRef] [PubMed]
- Olfati, A.-H.; Safary, A.; Akbarzadeh-Khiavi, M.; Adibkia, K. Recent Advances in Protein Corona and Practical Applications of Nanoparticle-Based Therapeutic Agents. J. Drug Deliv. Sci. Technol. 2023, 87, 104844. [Google Scholar] [CrossRef]
- Giulimondi, F.; Digiacomo, L.; Pozzi, D.; Palchetti, S.; Vulpis, E.; Capriotti, A.L.; Chiozzi, R.Z.; Laganà, A.; Amenitsch, H.; Masuelli, L.; et al. Interplay of Protein Corona and Immune Cells Controls Blood Residency of Liposomes. Nat. Commun. 2019, 10, 3686. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, P.; Hall, J.B.; McLeland, C.B.; Dobrovolskaia, M.A.; McNeil, S.E. Nanoparticle Interaction with Plasma Proteins as It Relates to Particle Biodistribution, Biocompatibility and Therapeutic Efficacy. Adv. Drug Deliv. Rev. 2009, 61, 428–437. [Google Scholar] [CrossRef]
- Gessner, A.; Lieske, A.; Paulke, B.R.; Müller, R.H. Influence of Surface Charge Density on Protein Adsorption on Polymeric Nanoparticles: Analysis by Two-Dimensional Electrophoresis. Eur. J. Pharm. Biopharm. 2002, 54, 165–170. [Google Scholar] [CrossRef]
- Lai, W.; Wang, Q.; Li, L.; Hu, Z.; Chen, J.; Fang, Q. Interaction of Gold and Silver Nanoparticles with Human Plasma: Analysis of Protein Corona Reveals Specific Binding Patterns. Colloids Surf. B Biointerfaces 2017, 152, 317–325. [Google Scholar] [CrossRef]
- Zhang, L.W.; Monteiro-Riviere, N.A. Mechanisms of Quantum Dot Nanoparticle Cellular Uptake. Toxicol. Sci. 2009, 110, 138–155. [Google Scholar] [CrossRef] [PubMed]
- Harush-Frenkel, O.; Debotton, N.; Benita, S.; Altschuler, Y. Targeting of Nanoparticles to the Clathrin-Mediated Endocytic Pathway. Biochem. Biophys. Res. Commun. 2007, 353, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Gessner, A.; Lieske, A.; Paulke, B.; Müller, R.H. Functional Groups on Polystyrene Model Nanoparticles: Influence on Protein Adsorption. J. Biomed. Mater. Res. A 2003, 65A, 319–326. [Google Scholar] [CrossRef]
- Tenzer, S.; Docter, D.; Kuharev, J.; Musyanovych, A.; Fetz, V.; Hecht, R.; Schlenk, F.; Fischer, D.; Kiouptsi, K.; Reinhardt, C.; et al. Rapid Formation of Plasma Protein Corona Critically Affects Nanoparticle Pathophysiology. Nat. Nanotechnol. 2013, 8, 772–781. [Google Scholar] [CrossRef]
- Hata, K.; Higashisaka, K.; Nagano, K.; Mukai, Y.; Kamada, H.; Tsunoda, S.; Yoshioka, Y.; Tsutsumi, Y. Evaluation of Silica Nanoparticle Binding to Major Human Blood Proteins. Nanoscale Res. Lett. 2014, 9, 668. [Google Scholar] [CrossRef] [PubMed]
- Sakulkhu, U.; Mahmoudi, M.; Maurizi, L.; Coullerez, G.; Hofmann-Amtenbrink, M.; Vries, M.; Motazacker, M.; Rezaee, F.; Hofmann, H. Significance of Surface Charge and Shell Material of Superparamagnetic Iron Oxide Nanoparticle (SPION) Based Core/Shell Nanoparticles on the Composition of the Protein Corona. Biomater. Sci. 2015, 3, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Morales, C.S.; Grodzinski, P. Current Landscape of Treating Different Cancers Using Nanomedicines: Trends and Perspectives. WIREs Nanomed. Nanobiotechnol. 2024, 16, e1927. [Google Scholar] [CrossRef] [PubMed]
- Muzaffar, J.; Bari, S.; Kirtane, K.; Chung, C.H. Recent Advances and Future Directions in Clinical Management of Head and Neck Squamous Cell Carcinoma. Cancers 2021, 13, 338. [Google Scholar] [CrossRef]
- de Lima, J.M.; Bonan, P.R.; da Cruz Perez, D.E.; Hier, M.; Alaoui-Jamali, M.A.; da Silva, S.D. Nanoparticle-Based Chemotherapy Formulations for Head and Neck Cancer: A Systematic Review and Perspectives. Nanomaterials 2020, 10, 1938. [Google Scholar] [CrossRef]
- Gavas, S.; Quazi, S.; Karpiński, T.M. Nanoparticles for Cancer Therapy: Current Progress and Challenges. Nanoscale Res. Lett. 2021, 16, 173. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.; Khan, A.I.; Cai, X.; Song, Y.; Lyu, Z.; Du, D.; Dutta, P.; Lin, Y. Overcoming Blood–Brain Barrier Transport: Advances in Nanoparticle-Based Drug Delivery Strategies. Mater. Today 2020, 37, 112–125. [Google Scholar] [CrossRef]
- Sun, L.; Liu, H.; Ye, Y.; Lei, Y.; Islam, R.; Tan, S.; Tong, R.; Miao, Y.-B.; Cai, L. Smart Nanoparticles for Cancer Therapy. Signal Transduct. Target. Ther. 2023, 8, 418. [Google Scholar] [CrossRef] [PubMed]
- Vanbilloen, W.J.F.; Rechberger, J.S.; Anderson, J.B.; Nonnenbroich, L.F.; Zhang, L.; Daniels, D.J. Nanoparticle Strategies to Improve the Delivery of Anticancer Drugs across the Blood–Brain Barrier to Treat Brain Tumors. Pharmaceutics 2023, 15, 1804. [Google Scholar] [CrossRef] [PubMed]
- Sell, M.; Lopes, A.R.; Escudeiro, M.; Esteves, B.; Monteiro, A.R.; Trindade, T.; Cruz-Lopes, L. Application of Nanoparticles in Cancer Treatment: A Concise Review. Nanomaterials 2023, 13, 2887. [Google Scholar] [CrossRef] [PubMed]
- Saraf, S.; Jain, A.; Tiwari, A.; Verma, A.; Panda, P.K.; Jain, S.K. Advances in Liposomal Drug Delivery to Cancer: An Overview. J. Drug Deliv. Sci. Technol. 2020, 56, 101549. [Google Scholar] [CrossRef]
- Gref, R.; Minamitake, Y.; Peracchia, M.T.; Trubetskoy, V.; Torchilin, V.; Langer, R. Biodegradable Long-Circulating Polymeric Nanospheres. Science 1994, 263, 1600–1603. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, P.; Kumar, S. Current Developments in Nanotechnology for Cancer Treatment. Mater. Today Proc. 2022, 48, 1754–1758. [Google Scholar] [CrossRef]
- Geppert, M.; Himly, M. Iron Oxide Nanoparticles in Bioimaging—An Immune Perspective. Front. Immunol. 2021, 12, 688927. [Google Scholar] [CrossRef] [PubMed]
- Acet, Ö.; Shcharbin, D.; Zhogla, V.; Kirsanov, P.; Halets-Bui, I.; Önal Acet, B.; Gök, T.; Bryszewska, M.; Odabaşı, M. Dipeptide Nanostructures: Synthesis, Interactions, Advantages and Biomedical Applications. Colloids Surf. B Biointerfaces 2023, 222, 113031. [Google Scholar] [CrossRef]
- Kalyane, D.; Raval, N.; Maheshwari, R.; Tambe, V.; Kalia, K.; Tekade, R.K. Employment of Enhanced Permeability and Retention Effect (EPR): Nanoparticle-Based Precision Tools for Targeting of Therapeutic and Diagnostic Agent in Cancer. Mater. Sci. Eng. C 2019, 98, 1252–1276. [Google Scholar] [CrossRef] [PubMed]
- Danhier, F. To Exploit the Tumor Microenvironment: Since the EPR Effect Fails in the Clinic, What Is the Future of Nanomedicine? J. Control. Release 2016, 244, 108–121. [Google Scholar] [CrossRef]
- Wilhelm, S.; Tavares, A.J.; Dai, Q.; Ohta, S.; Audet, J.; Dvorak, H.F.; Chan, W.C.W. Analysis of Nanoparticle Delivery to Tumours. Nat. Rev. Mater. 2016, 1, 16014. [Google Scholar] [CrossRef]
- Kundranda, M.; Niu, J. Albumin-Bound Paclitaxel in Solid Tumors: Clinical Development and Future Directions. Drug Des. Devel Ther. 2015, 2015, 3767–3777. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Hu, J.; Liu, S. Efficacy and Safety of Nanoparticle Albumin-Bound Paclitaxel in Non-Small Cell Lung Cancer: A Systematic Review and Meta-Analysis. Artif. Cells Nanomed. Biotechnol. 2019, 47, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, S.; Wang, X.; Chen, Q.; He, Z.; Luo, C.; Sun, J. Smart Transformable Nanomedicines for Cancer Therapy. Biomaterials 2021, 271, 120737. [Google Scholar] [CrossRef] [PubMed]
- Alsahafi, E.; Begg, K.; Amelio, I.; Raulf, N.; Lucarelli, P.; Sauter, T.; Tavassoli, M. Clinical Update on Head and Neck Cancer: Molecular Biology and Ongoing Challenges. Cell Death Dis. 2019, 10, 540. [Google Scholar] [CrossRef] [PubMed]
- Kola, P.; Nagesh, P.K.B.; Roy, P.K.; Deepak, K.; Reis, R.L.; Kundu, S.C.; Mandal, M. Innovative Nanotheranostics: Smart Nanoparticles Based Approach to Overcome Breast Cancer Stem Cells Mediated Chemo- and Radioresistances. WIREs Nanomed. Nanobiotechnology 2023, 15, e1876. [Google Scholar] [CrossRef] [PubMed]
- Duo, Y.; Li, Y.; Chen, C.; Liu, B.; Wang, X.; Zeng, X.; Chen, H. DOX-Loaded PH-Sensitive Mesoporous Silica Nanoparticles Coated with PDA and PEG Induce pro-Death Autophagy in Breast Cancer. RSC Adv. 2017, 7, 39641–39650. [Google Scholar] [CrossRef]
- Zhang, P.; Cui, Y.; Wang, J.; Cheng, J.; Zhu, L.; Liu, C.; Yue, S.; Pang, R.; Guan, J.; Xie, B.; et al. Dual-Stimuli Responsive Smart Nanoprobe for Precise Diagnosis and Synergistic Multi-Modalities Therapy of Superficial Squamous Cell Carcinoma. J. Nanobiotechnol. 2023, 21, 4. [Google Scholar] [CrossRef]
- Lo, Y.-L.; Lin, H.-C.; Tseng, W.-H. Tumor PH-Functionalized and Charge-Tunable Nanoparticles for the Nucleus/Cytoplasm-Directed Delivery of Oxaliplatin and MiRNA in the Treatment of Head and Neck Cancer. Acta Biomater. 2022, 153, 465–480. [Google Scholar] [CrossRef]
- Bhardwaj, P.; Gota, V.; Vishwakarma, K.; Pai, V.; Chaudhari, P.; Mohanty, B.; Thorat, R.; Yadav, S.; Gurjar, M.; Goda, J.S.; et al. Loco-Regional Radiosensitizing Nanoparticles-in-Gel Augments Head and Neck Cancer Chemoradiotherapy. J. Control. Release 2022, 343, 288–302. [Google Scholar] [CrossRef]
- Zhou, Z.; Liang, S.; Zhao, T.; Chen, X.; Cao, X.; Qi, M.; Huang, Y.; Ju, W.; Yang, M.; Zhu, D.; et al. Overcoming Chemotherapy Resistance Using PH-Sensitive Hollow MnO2 Nanoshells That Target the Hypoxic Tumor Microenvironment of Metastasized Oral Squamous Cell Carcinoma. J. Nanobiotechnol. 2021, 19, 157. [Google Scholar] [CrossRef]
- Dehghankelishadi, P.; Maritz, M.F.; Badiee, P.; Thierry, B. High Density Lipoprotein Nanoparticle as Delivery System for Radio-Sensitising MiRNA: An Investigation in 2D/3D Head and Neck Cancer Models. Int. J. Pharm. 2022, 617, 121585. [Google Scholar] [CrossRef]
- Delavarian, Z.; Pakfetrat, A.; Ghazi, A.; Jaafari, M.R.; Homaei Shandiz, F.; Dalirsani, Z.; Mohammadpour, A.H.; Rahimi, H.R. Oral Administration of Nanomicelle Curcumin in the Prevention of Radiotherapy-induced Mucositis in Head and Neck Cancers. Spec. Care Dent. 2019, 39, 166–172. [Google Scholar] [CrossRef]
- Orienti, I.; Nguyen, F.; Guan, P.; Kolla, V.; Calonghi, N.; Farruggia, G.; Chorny, M.; Brodeur, G.M. A Novel Nanomicellar Combination of Fenretinide and Lenalidomide Shows Marked Antitumor Activity in a Neuroblastoma Xenograft Model. Drug Des. Devel Ther. 2019, 13, 4305–4319. [Google Scholar] [CrossRef]
- Singhvi, G.; Rapalli, V.K.; Nagpal, S.; Dubey, S.K.; Saha, R.N. Nanocarriers as Potential Targeted Drug Delivery for Cancer Therapy. Nanosci. Med. 2020, 1, 51–88. [Google Scholar]
- Zhu, M.; Chen, S.; Hua, L.; Zhang, C.; Chen, M.; Chen, D.; Dong, Y.; Zhang, Y.; Li, M.; Song, X.; et al. Self-Targeted Salinomycin-Loaded DSPE-PEG-Methotrexate Nanomicelles for Targeting Both Head and Neck Squamous Cell Carcinoma Cancer Cells and Cancer Stem Cells. Nanomedicine 2017, 12, 295–315. [Google Scholar] [CrossRef]
- Ruiz-Pulido, G.; Medina, D.I.; Barani, M.; Rahdar, A.; Sargazi, G.; Baino, F.; Pandey, S. Nanomaterials for the Diagnosis and Treatment of Head and Neck Cancers: A Review. Materials 2021, 14, 3706. [Google Scholar] [CrossRef]
- Mapanao, A.K.; Santi, M.; Voliani, V. Combined Chemo-Photothermal Treatment of Three-Dimensional Head and Neck Squamous Cell Carcinomas by Gold Nano-Architectures. J. Colloid. Interface Sci. 2021, 582, 1003–1011. [Google Scholar] [CrossRef]
- Chen, H.; Deng, J.; Yao, X.; He, Y.; Li, H.; Jian, Z.; Tang, Y.; Zhang, X.; Zhang, J.; Dai, H. Bone-Targeted Erythrocyte-Cancer Hybrid Membrane-Camouflaged Nanoparticles for Enhancing Photothermal and Hypoxia-Activated Chemotherapy of Bone Invasion by OSCC. J. Nanobiotechnol. 2021, 19, 342. [Google Scholar] [CrossRef]
- Pereira, D.; Martins, D.; Mendes, F. Immunotherapy in Head and Neck Cancer When, How, and Why? Biomedicines 2022, 10, 2151. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Zhou, X.; Zhang, Y.; Zhao, T.; Li, J.; Zhong, L.; Pang, Y. Macrophage-Capturing Self-Assembly Photosensitizer Nanoparticles Induces Immune Microenvironment Re-Programming and Golgi-Responsive Immunogenic Cell Death in Head and Neck Carcinoma. Adv. Healthc. Mater. 2024, 2400012. [Google Scholar] [CrossRef]
- Oksel Karakus, C.; Bilgi, E.; Winkler, D.A. Biomedical Nanomaterials: Applications, Toxicological Concerns, and Regulatory Needs. Nanotoxicology 2021, 15, 331–351. [Google Scholar] [CrossRef]
- Mech, A.; Wohlleben, W.; Ghanem, A.; Hodoroaba, V.; Weigel, S.; Babick, F.; Brüngel, R.; Friedrich, C.M.; Rasmussen, K.; Rauscher, H. Nano or Not Nano? A Structured Approach for Identifying Nanomaterials According to the European Commission’s Definition. Small 2020, 16, 2002228. [Google Scholar] [CrossRef]
- Banik, B.L.; Fattahi, P.; Brown, J.L. Polymeric Nanoparticles: The Future of Nanomedicine. WIREs Nanomed. Nanobiotechnol. 2016, 8, 271–299. [Google Scholar] [CrossRef]
- Yu, C.; Li, L.; Wang, S.; Xu, Y.; Wang, L.; Huang, Y.; Hieawy, A.; Liu, H.; Ma, J. Advances in Nanomaterials for the Diagnosis and Treatment of Head and Neck Cancers: A Review. Bioact. Mater. 2023, 25, 430–444. [Google Scholar] [CrossRef]
- Zhang, Z.; Cao, X.; Wang, C.; Huang, Q.; Liu, Y. Nanomaterials in Cancer Treatment. In Handbook of Nanomaterials, Volume 2; Elsevier: Amsterdam, The Netherlands, 2024; pp. 431–472. [Google Scholar]
- Hu, Q.; Li, H.; Wang, L.; Gu, H.; Fan, C. DNA Nanotechnology-Enabled Drug Delivery Systems. Chem. Rev. 2019, 119, 6459–6506. [Google Scholar] [CrossRef]
- Loretan, M.; Domljanovic, I.; Lakatos, M.; Rüegg, C.; Acuna, G.P. DNA Origami as Emerging Technology for the Engineering of Fluorescent and Plasmonic-Based Biosensors. Materials 2020, 13, 2185. [Google Scholar] [CrossRef]
- Udomprasert, A.; Kangsamaksin, T. DNA Origami Applications in Cancer Therapy. Cancer Sci. 2017, 108, 1535–1543. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, F.; Zhao, L.; Wu, Y.; Li, C.; Li, H.; Jiang, Y. Enhancing Cancer Treatment via “Zn2+ Interference” with Zn-Based Nanomaterials. Coord. Chem. Rev. 2024, 500, 215535. [Google Scholar] [CrossRef]
- Zhang, P.; Zhang, L.; Qin, Z.; Hua, S.; Guo, Z.; Chu, C.; Lin, H.; Zhang, Y.; Li, W.; Zhang, X.; et al. Genetically Engineered Liposome-like Nanovesicles as Active Targeted Transport Platform. Adv. Mater. 2018, 30, 1705350. [Google Scholar] [CrossRef] [PubMed]
- Bu, L.-L.; Yan, J.; Wang, Z.; Ruan, H.; Chen, Q.; Gunadhi, V.; Bell, R.B.; Gu, Z. Advances in Drug Delivery for Post-Surgical Cancer Treatment. Biomaterials 2019, 219, 119182. [Google Scholar] [CrossRef]
- Gao, Z.; Li, C.; Shen, J.; Ding, D. Organic Optical Agents for Image-Guided Combined Cancer Therapy. Biomed. Mater. 2021, 16, 042009. [Google Scholar] [CrossRef]
- Fang, J.; Islam, W.; Maeda, H. Exploiting the Dynamics of the EPR Effect and Strategies to Improve the Therapeutic Effects of Nanomedicines by Using EPR Effect Enhancers. Adv. Drug Deliv. Rev. 2020, 157, 142–160. [Google Scholar] [CrossRef]
- Mohammadpour, R.; Dobrovolskaia, M.A.; Cheney, D.L.; Greish, K.F.; Ghandehari, H. Subchronic and Chronic Toxicity Evaluation of Inorganic Nanoparticles for Delivery Applications. Adv. Drug Deliv. Rev. 2019, 144, 112–132. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Li, Z.; Xie, Z.; Sokolova, I.M.; Song, L.; Peijnenburg, W.J.G.M.; Hu, M.; Wang, Y. Rethinking Nano-TiO2 Safety: Overview of Toxic Effects in Humans and Aquatic Animals. Small 2020, 16, 2002019. [Google Scholar] [CrossRef] [PubMed]
- Okesola, B.O.; Mata, A. Multicomponent Self-Assembly as a Tool to Harness New Properties from Peptides and Proteins in Material Design. Chem. Soc. Rev. 2018, 47, 3721–3736. [Google Scholar] [CrossRef]
- Smith, K.H.; Tejeda-Montes, E.; Poch, M.; Mata, A. Integrating Top-down and Self-Assembly in the Fabrication of Peptide and Protein-Based Biomedical Materials. Chem. Soc. Rev. 2011, 40, 4563. [Google Scholar] [CrossRef]
- Manzari, M.T.; Shamay, Y.; Kiguchi, H.; Rosen, N.; Scaltriti, M.; Heller, D.A. Targeted Drug Delivery Strategies for Precision Medicines. Nat. Rev. Mater. 2021, 6, 351–370. [Google Scholar] [CrossRef]
- Yu, C.; Li, L.; Hu, P.; Yang, Y.; Wei, W.; Deng, X.; Wang, L.; Tay, F.R.; Ma, J. Recent Advances in Stimulus-Responsive Nanocarriers for Gene Therapy. Adv. Sci. 2021, 8, 2100540. [Google Scholar] [CrossRef] [PubMed]
- Large, D.E.; Abdelmessih, R.G.; Fink, E.A.; Auguste, D.T. Liposome Composition in Drug Delivery Design, Synthesis, Characterization, and Clinical Application. Adv. Drug Deliv. Rev. 2021, 176, 113851. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Jang, H.; Cho, H.; Choi, J.; Hwang, K.Y.; Choi, Y.; Kim, S.H.; Yang, Y. Recent Advances in Exosome-Based Drug Delivery for Cancer Therapy. Cancers 2021, 13, 4435. [Google Scholar] [CrossRef] [PubMed]
- Giallombardo, M.; Taverna, S.; Alessandro, R.; Hong, D.; Rolfo, C. Exosome-Mediated Drug Resistance in Cancer: The near Future Is Here. Ther. Adv. Med. Oncol. 2016, 8, 320–322. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zheng, Y.; Zhao, M. Exosome-Based Cancer Therapy: Implication for Targeting Cancer Stem Cells. Front. Pharmacol. 2017, 7, 533. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Jiang, L.-H.; Hou, J.-C.; Zhong, S.-L.; Zhu, L.-P.; Wang, D.-D.; Zhou, S.-Y.; Yang, S.-J.; Wang, J.-Y.; Zhang, Q.; et al. Exosome: A Novel Mediator in Drug Resistance of Cancer Cells. Epigenomics 2018, 10, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Wu, D.; Ma, X.; Wang, J.; Hou, W.; Zhang, W. Exosomes as Drug Carriers for Cancer Therapy and Challenges Regarding Exosome Uptake. Biomed. Pharmacother. 2020, 128, 110237. [Google Scholar] [CrossRef]
- Song, H.; Liu, B.; Dong, B.; Xu, J.; Zhou, H.; Na, S.; Liu, Y.; Pan, Y.; Chen, F.; Li, L.; et al. Exosome-Based Delivery of Natural Products in Cancer Therapy. Front. Cell Dev. Biol. 2021, 9, 650426. [Google Scholar] [CrossRef]
- Chehelgerdi, M.; Chehelgerdi, M.; Allela, O.Q.B.; Pecho, R.D.C.; Jayasankar, N.; Rao, D.P.; Thamaraikani, T.; Vasanthan, M.; Viktor, P.; Lakshmaiya, N.; et al. Progressing Nanotechnology to Improve Targeted Cancer Treatment: Overcoming Hurdles in Its Clinical Implementation. Mol. Cancer 2023, 22, 169. [Google Scholar] [CrossRef]
- Torchilin, V.P. Multifunctional, Stimuli-Sensitive Nanoparticulate Systems for Drug Delivery. Nat. Rev. Drug Discov. 2014, 13, 813–827. [Google Scholar] [CrossRef]
- Kinnear, C.; Moore, T.L.; Rodriguez-Lorenzo, L.; Rothen-Rutishauser, B.; Petri-Fink, A. Form Follows Function: Nanoparticle Shape and Its Implications for Nanomedicine. Chem. Rev. 2017, 117, 11476–11521. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gül, D.; Önal Acet, B.; Lu, Q.; Stauber, R.H.; Odabaşı, M.; Acet, Ö. Revolution in Cancer Treatment: How Are Intelligently Designed Nanostructures Changing the Game? Int. J. Mol. Sci. 2024, 25, 5171. https://doi.org/10.3390/ijms25105171
Gül D, Önal Acet B, Lu Q, Stauber RH, Odabaşı M, Acet Ö. Revolution in Cancer Treatment: How Are Intelligently Designed Nanostructures Changing the Game? International Journal of Molecular Sciences. 2024; 25(10):5171. https://doi.org/10.3390/ijms25105171
Chicago/Turabian StyleGül, Désirée, Burcu Önal Acet, Qiang Lu, Roland H. Stauber, Mehmet Odabaşı, and Ömür Acet. 2024. "Revolution in Cancer Treatment: How Are Intelligently Designed Nanostructures Changing the Game?" International Journal of Molecular Sciences 25, no. 10: 5171. https://doi.org/10.3390/ijms25105171
APA StyleGül, D., Önal Acet, B., Lu, Q., Stauber, R. H., Odabaşı, M., & Acet, Ö. (2024). Revolution in Cancer Treatment: How Are Intelligently Designed Nanostructures Changing the Game? International Journal of Molecular Sciences, 25(10), 5171. https://doi.org/10.3390/ijms25105171






