The Effects of Intermittent Cold Exposure on Adipose Tissue
Abstract
:1. Introduction
2. Overview of Included Literature
3. ICE Effects on Body Weight, Energy Expenditure, and Adipose Tissue
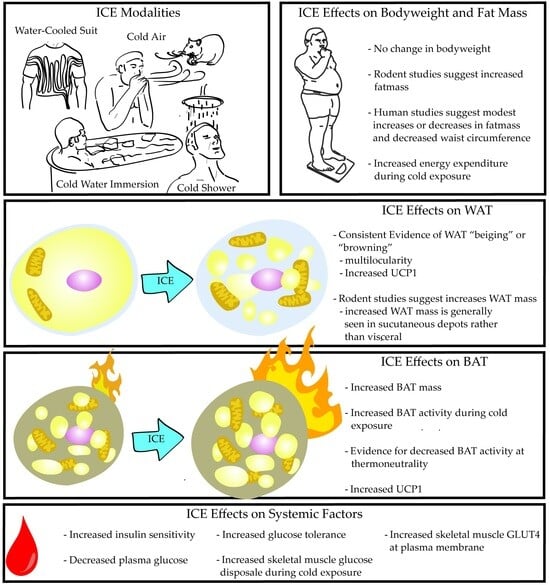
3.1. Body Weight and Energy Expenditure
3.2. White Adipose Tissue Weight and Morphology
3.3. Brown Adipose Tissue Weight, Morphology, and Energy Expenditure
3.4. Overall Conclusions from Table 2
4. ICE Effects on Adipose Tissue Gene and Protein Expression
4.1. Brown Adipose Tissue Gene and Protein Expression
4.2. White Adipose Tissue Gene and Protein Expression
4.3. Overall Conclusions from Table 3
5. ICE Effects on Systemic Factors Related to Adipose Tissue and Metabolism
Insulin, Glucose Homeostasis, and Adipokines
6. Effects of Exercise during ICE
7. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Grundy, S.M. Adipose Tissue and Metabolic Syndrome: Too Much, Too Little or Neither. Eur. J. Clin. Investig. 2015, 45, 1209. [Google Scholar] [CrossRef] [PubMed]
- Fahed, G.; Aoun, L.; Zerdan, M.B.; Allam, S.; Zerdan, M.B.; Bouferraa, Y.; Assi, H.I. Metabolic Syndrome: Updates on Pathophysiology and Management in 2021. Int. J. Mol. Sci. 2022, 23, 786. [Google Scholar] [CrossRef] [PubMed]
- Qian, T.; Sheng, X.; Shen, P.; Fang, Y.; Deng, Y.; Zou, G. Mets-IR as a Predictor of Cardiovascular Events in the Middle-Aged and Elderly Population and Mediator Role of Blood Lipids. Front. Endocrinol. 2023, 14, 1224967. [Google Scholar] [CrossRef] [PubMed]
- Kouvari, M.; D’cunha, N.M.; Travica, N.; Sergi, D.; Zec, M.; Marx, W.; Naumovski, N. Metabolic Syndrome, Cognitive Impairment and the Role of Diet: A Narrative Review. Nutrients 2022, 14, 333. [Google Scholar] [CrossRef]
- Sakers, A.; De Siqueira, M.K.; Seale, P.; Villanueva, C.J. Adipose-Tissue Plasticity in Health and Disease. Cell 2022, 185, 419–446. [Google Scholar] [CrossRef] [PubMed]
- Hauner, H. The Mode of Action of Thiazolidinediones. Diabetes Metab. Res. Rev. 2002, 18 (Suppl. S2), S10–S15. [Google Scholar] [CrossRef]
- Ahmadian, M.; Suh, J.M.; Hah, N.; Liddle, C.; Atkins, A.R.; Downes, M.; Evans, R.M. PPARγ Signaling and Metabolism: The Good, the Bad and the Future. Nat. Med. 2013, 19, 557–566. [Google Scholar] [CrossRef]
- Jaacks, L.M.; Vandevijvere, S.; Pan, A.; McGowan, C.J.; Wallace, C.; Imamura, F.; Mozaffarian, D.; Swinburn, B.; Ezzati, M. The Obesity Transition: Stages of the Global Epidemic. Lancet Diabetes Endocrinol. 2019, 7, 231. [Google Scholar] [CrossRef]
- Néma, J.; Zdara, J.; Lašák, P.; Bavlovič, J.; Bureš, M.; Pejchal, J.; Schvach, H. Impact of Cold Exposure on Life Satisfaction and Physical Composition of Soldiers. BMJ Mil Health 2023, e002237. [Google Scholar] [CrossRef]
- Van Der Lans, A.A.J.J.; Boon, M.R.; Haks, M.C.; Quinten, E.; Schaart, G.; Ottenhoff, T.H.; van Marken Lichtenbelt, W.D. Cold Acclimation Affects Immune Composition in Skeletal Muscle of Healthy Lean Subjects. Physiol. Rep. 2015, 3, e12394. [Google Scholar] [CrossRef]
- Janský, L.; Pospíšilová, D.; Honzová, S.; Uličný, B.; Šrámek, P.; Zeman, V.; Kamínková, J. Immune System of Cold-Exposed and Cold-Adapted Humans. Eur. J. Appl. Physiol. Occup. Physiol. 1996, 72, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Van Tulleken, C.; Tipton, M.; Massey, H.; Harper, C.M. Open Water Swimming as a Treatment for Major Depressive Disorder. BMJ Case Rep. 2018, 2018, bcr-2018-225007. [Google Scholar] [CrossRef]
- Castellani, J.W.; Young, A.J. Human Physiological Responses to Cold Exposure: Acute Responses and Acclimatization to Prolonged Exposure. Auton. Neurosci. 2016, 196, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Fenzl, A.; Kiefer, F.W. Brown Adipose Tissue and Thermogenesis. Horm. Mol. Biol. Clin. Investig. 2014, 19, 25–37. [Google Scholar] [CrossRef]
- Blondin, D.P.; Labbé, S.M.; Phoenix, S.; Guérin, B.; Turcotte, É.E.; Richard, D.; Carpentier, A.C.; Haman, F. Contributions of White and Brown Adipose Tissues and Skeletal Muscles to Acute Cold-Induced Metabolic Responses in Healthy Men. J. Physiol. 2015, 593, 701–714. [Google Scholar] [CrossRef] [PubMed]
- Søberg, S.; Löfgren, J.; Philipsen, F.E.; Jensen, M.; Hansen, A.E.; Ahrens, E.; Nystrup, K.B.; Nielsen, R.D.; Sølling, C.; Wedell-Neergaard, A.S.; et al. Altered Brown Fat Thermoregulation and Enhanced Cold-Induced Thermogenesis in Young, Healthy, Winter-Swimming Men. Cell Rep. Med. 2021, 2, 100408. [Google Scholar] [CrossRef]
- Cheng, L.; Wang, J.; Dai, H.; Duan, Y.; An, Y.; Shi, L.; Lv, Y.; Li, H.; Wang, C.; Ma, Q.; et al. Brown and Beige Adipose Tissue: A Novel Therapeutic Strategy for Obesity and Type 2 Diabetes Mellitus. Adipocyte 2021, 10, 48–65. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Z.; Song, Y.; Xie, H.; Dong, M. An Update on Brown Adipose Tissue and Obesity Intervention: Function, Regulation and Therapeutic Implications. Front. Endocrinol. 2023, 13, 1065263. [Google Scholar] [CrossRef]
- Arnold, J.; Richard, D. Exercise during Intermittent Cold Exposure Prevents Acclimation to Cold Rats. J. Physiol. 1987, 390, 45–54. [Google Scholar] [CrossRef]
- Deshaies, Y.; Arnold, J.; Richard, D. Lipoprotein Lipase in Adipose Tissues of Rats Running during Cold Exposure. J. Appl. Physiol. 1988, 65, 549–554. [Google Scholar] [CrossRef]
- Doi, K.; Kuroshima, A. Lasting Effect of Infantile Cold Experience on Cold Tolerance in Adult Rats. Jpn. J. Physiol. 1979, 29, 139–150. [Google Scholar] [CrossRef] [PubMed]
- Harri, M.; Dannenberg, T.; Oksanen-Rossi, R.; Hohtola, E.; Sundin, U. Related and Unrelated Changes in Response to Exercise and Cold in Rats: A Reevaluation. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1984, 57, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Yahata, T.; Kuroshima, A. Metabolic Cold Acclimation after Repetitive Intermittent Cold Exposure in Rat. Jpn. J. Physiol. 1989, 39, 215–228. [Google Scholar] [CrossRef] [PubMed]
- Yoneshiro, T.; Aita, S.; Matsushita, M.; Kayahara, T.; Kameya, T.; Kawai, Y.; Iwanaga, T.; Saito, M. Recruited Brown Adipose Tissue as an Antiobesity Agent in Humans. J. Clin. Investig. 2013, 123, 3404–3408. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.S.; Qiao, L.; Bosco, C.; Leong, L.H.; Lytle, N.; Feng, G.S.; Chi, N.W.; Shao, J. Intermittent Cold Exposure Enhances Fat Accumulation in Mice. PLoS ONE 2014, 9, e96432. [Google Scholar] [CrossRef]
- Qiao, L.; Yoo, H.S.; Bosco, C.; Lee, B.; Feng, G.S.; Schaack, J.; Chi, N.W.; Shao, J. Adiponectin Reduces Thermogenesis by Inhibiting Brown Adipose Tissue Activation in Mice. Diabetologia 2014, 57, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Ravussin, Y.; Xiao, C.; Gavrilova, O.; Reitman, M.L. Effect of Intermittent Cold Exposure on Brown Fat Activation, Obesity, and Energy Homeostasis in Mice. PLoS ONE 2014, 9, e85876. [Google Scholar] [CrossRef]
- Blondin, D.P.; Labbé, S.M.; Tingelstad, H.C.; Noll, C.; Kunach, M.; Phoenix, S.; Guérin, B.; Turcotte, É.E.; Carpentier, A.C.; Richard, D.; et al. Increased Brown Adipose Tissue Oxidative Capacity in Cold-Acclimated Humans. J. Clin. Endocrinol. Metab. 2014, 99, E438. [Google Scholar] [CrossRef]
- Wang, T.Y.; Liu, C.; Wang, A.; Sun, Q. Intermittent Cold Exposure Improves Glucose Homeostasis Associated with Brown and White Adipose Tissues in Mice. Life Sci. 2015, 139, 153. [Google Scholar] [CrossRef]
- Bai, Z.; Wuren, T.; Liu, S.; Han, S.; Chen, L.; McClain, D.; Ge, R.L. Intermittent Cold Exposure Results in Visceral Adipose Tissue “Browning” in the Plateau Pika (Ochotona curzoniae). Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2015, 184, 171–178. [Google Scholar] [CrossRef]
- Hanssen, M.J.W.; Hoeks, J.; Brans, B.; Van Der Lans, A.A.J.J.; Schaart, G.; Van Den Driessche, J.J.; Jörgensen, J.A.; Boekschoten, M.V.; Hesselink, M.K.C.; Havekes, B.; et al. Short-Term Cold Acclimation Improves Insulin Sensitivity in Patients with Type 2 Diabetes Mellitus. Nat. Med. 2015, 21, 863–865. [Google Scholar] [CrossRef]
- Gibas-Dorna, M.; Checinska, Z.; Korek, E.; Kupsz, J.; Sowinska, A.; Krauss, H. Cold Water Swimming Beneficially Modulates Insulin Sensitivity in Middle-Aged Individuals. J. Aging Phys. Act. 2016, 24, 547–554. [Google Scholar] [CrossRef]
- Tsibul’nikov, S.Y.; Maslov, L.N.; Naryzhnaya, N.V.; Ivanov, V.V.; Lishmanov, Y.B. Specific Features of Adaptation of Rats to Chronic Cold Treatment. Dokl. Biol. Sci. 2016, 470, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Hanssen, M.J.W.; Van Der Lans, A.A.J.J.; Brans, B.; Hoeks, J.; Jardon, K.M.C.; Schaart, G.; Mottaghy, F.M.; Schrauwen, P.; Van Marken Lichtenbelt, W.D. Short-Term Cold Acclimation Recruits Brown Adipose Tissue in Obese Humans. Diabetes 2016, 65, 1179–1189. [Google Scholar] [CrossRef]
- Blondin, D.P.; Daoud, A.; Taylor, T.; Tingelstad, H.C.; Bézaire, V.; Richard, D.; Carpentier, A.C.; Taylor, A.W.; Harper, M.-E.; Aguer, C.; et al. Four-Week Cold Acclimation in Adult Humans Shifts Uncoupling Thermogenesis from Skeletal Muscles to Brown Adipose Tissue. J. Physiol. 2017, 595, 2099–2113. [Google Scholar] [CrossRef] [PubMed]
- Presby, D.M.; Jackman, M.R.; Rudolph, M.C.; Sherk, V.D.; Foright, R.M.; Houck, J.A.; Johnson, G.C.; Orlicky, D.J.; Melanson, E.L.; Higgins, J.A.; et al. Compensation for Cold-Induced Thermogenesis during Weight Loss Maintenance and Regain. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E977–E986. [Google Scholar] [CrossRef]
- Zhang, L.; An, G.; Wu, S.; Wang, J.; Yang, D.; Zhang, Y.; Li, X. Long-Term Intermittent Cold Exposure Affects Peri-Ovarian Adipose Tissue and Ovarian Microenvironment in Rats. J. Ovarian Res. 2021, 14, 107. [Google Scholar] [CrossRef] [PubMed]
- McKie, G.L.; Shamshoum, H.; Hunt, K.L.; Thorpe, H.H.A.; Dibe, H.A.; Khokhar, J.Y.; Doucette, C.A.; Wright, D.C. Intermittent Cold Exposure Improves Glucose Homeostasis despite Exacerbating Diet-Induced Obesity in Mice Housed at Thermoneutrality. J. Physiol. 2022, 600, 829–845. [Google Scholar] [CrossRef]
- Weng, X.; Wang, C.; Yuan, Y.; Wang, Z.; Kuang, J.; Yan, X.; Chen, H. Effect of Cold Exposure and Exercise on Insulin Sensitivity and Serum Free Fatty Acids in Obese Rats. Med. Sci. Sports Exerc. 2023, 55, 1409–1415. [Google Scholar] [CrossRef]
- Brychta, R.J.; Chen, K.Y. Cold-Induced Thermogenesis in Humans. Eur. J. Clin. Nutr. 2017, 71, 345. [Google Scholar] [CrossRef]
- van Marken Lichtenbelt, W.D.; Schrauwen, P. Implications of Nonshivering Thermogenesis for Energy Balance Regulation in Humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011, 301, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Zeyl, A.; Stocks, J.M.; Taylor, N.A.S.; Jenkins, A.B. Interactions between Temperature and Human Leptin Physiology in Vivo and in Vitro. Eur. J. Appl. Physiol. 2004, 92, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Gibas-Dorna, M.; Checinska, Z.; Korek, E.; Kupsz, J.; Sowinska, A.; Wojciechowska, M.; Krauss, H.; Piątek, J. Variations in Leptin and Insulin Levels within One Swimming Season in Non-Obese Female Cold Water Swimmers. Scand. J. Clin. Lab. Investig. 2016, 76, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.-H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige Adipocytes Are a Distinct Type of Thermogenic Fat Cell in Mouse and Human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef] [PubMed]
- González-García, I.; Milbank, E.; Diéguez, C.; López, M.; Contreras, C. Glucagon, GLP-1 and Thermogenesis. Int. J. Mol. Sci. 2019, 20, 3445. [Google Scholar] [CrossRef] [PubMed]
- Timmons, J.A.; Wennmalm, K.; Larsson, O.; Walden, T.B.; Lassmann, T.; Petrovic, N.; Hamilton, D.L.; Gimeno, R.E.; Wahlestedt, C.; Baar, K.; et al. Myogenic Gene Expression Signature Establishes That Brown and White Adipocytes Originate from Distinct Cell Lineages. Proc. Natl. Acad. Sci. USA 2007, 104, 4401–4406. [Google Scholar] [CrossRef]
- Rosen, E.D.; Spiegelman, B.M. What We Talk About When We Talk About Fat. Cell 2014, 156, 20–44. [Google Scholar] [CrossRef]
- Straub, L.G.; Scherer, P.E. Metabolic Messengers: Adiponectin. Nat. Metab. 2019, 1, 334–339. [Google Scholar] [CrossRef]
- Ye, L.; Wu, J.; Cohen, P.; Kazak, L.; Khandekar, M.J.; Jedrychowski, M.P.; Zeng, X.; Gygi, S.P.; Spiegelman, B.M. Fat Cells Directly Sense Temperature to Activate Thermogenesis. Proc. Natl. Acad. Sci. USA 2013, 110, 12480–12485. [Google Scholar] [CrossRef]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-Dependent Myokine That Drives Brown-Fat-like Development of White Fat and Thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef]
- Rosenwald, M.; Perdikari, A.; Rülicke, T.; Wolfrum, C. Bi-Directional Interconversion of Brite and White Adipocytes. Nat. Cell Biol. 2013, 15, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.K.; Hevener, A.L.; Barnard, R.J. Metabolic Syndrome and Insulin Resistance: Underlying Causes and Modification by Exercise Training. In Comprehensive Physiology; Wiley: Hoboken, NJ, USA, 2013; pp. 1–58. [Google Scholar]
- Bornfeldt, K.E.; Tabas, I. Insulin Resistance, Hyperglycemia, and Atherosclerosis. Cell Metab. 2011, 14, 575–585. [Google Scholar] [CrossRef] [PubMed]
| Year | First Author | Citation | Methods | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Model | Groups | ICE Modality | ICE Freq. | ICE Intensity | ICE Duration | ICE Intervention | |||
| 1979 | Doi | [21] | Rats | 1. Control Newborn 2. Control Adult 3. ICE Newborn 4. ICE Adult | Air | daily | 5 °C | 4 h | 14 days |
| 1984 | Harri | [22] | Rats | 1. Control 2. CCE 3. ICE 4. Control Exercise 5. CCE Exercise | Air | 5 d/wk | −20 °C | 1 h | ≥6 wks |
| 1986 | Arnold | [19] | Rats | 1. Control 2. ICE 3. Control Exercise 4. ICE Exercise | Air | daily | −5 °C | 2 h | 28 days |
| 1988 | Deshaies | [20] | Rats | 1. Control 2. ICE 3. Control Exercise 4. ICE Exercise | Air | daily | −5 °C | 2 h | 28 days |
| 1989 | Yahata | [23] | Rats | 1. Control 2. CCE 3. ICE | Air | daily | 5 °C | 6 h or 2 h | 28 days |
| 2013 | Yoneshiro | [24] | Humans (Low or No BAT Activity During Initial ACE) | 1. Control Pre vs. Post 2. ICE Pre vs. Post | Air | daily | 17 °C | 2 h | 6 wks |
| 2014 | Yoo | [25] | Mice | 1. Control 2. ACE 3. ICE | Air | daily | 4 °C | 6 h | 12–14 days |
| 2014 | Qiao | [26] | Mice | 1. Control ICE 2. Adipoq−/− ICE | Air | daily | 4 °C | 6 h | 10 days |
| 2014 | Ravussin | [27] | Mice | 1. Control 2. ICE 1 h 3. ICE 4 h 4. ICE 8 h | Air | 3 d/wk | 4 °C | 1, 4, or 8 h | 10–11 wks |
| 2014 | Blondin | [28] | Humans | Pre vs. Post ICE | Liquid-cooled suit | 5 d/wk | 18 °C | 2 h | 4 wks |
| 2015 | Wang | [29] | Mice | 1. Control 2. ICE | Air | 5 d/wk | 4 °C | 2 h | 14 wks |
| 2015 | Bai | [30] | Platue Pika (Rodent) Simulated Low Air Pressure (4100 M) | 1. Control 2. ICE | Air | daily | 5–6 °C | 4 h | 15 days |
| 2015 | Hanssen | [31] | Humans (Obese) | Pre vs. Post ICE | Air | daily | 15 °C | 6 h | 10 days |
| 2016 | Gibas-Dorna | [32] | Humans | 1. Controls 2. Winter Swimmers (Pre vs. Post) | Winter swimming | ≥2 d/wk | 1–10 °C | 5–15 min | ~6 months |
| 2016 | Tsibul’nikov | [33] | Rats | 1. Control 2. CCE 3. ICE (1.5 h) 4. ICE (8 h) | Air | daily | 4 °C | 1.5 or 8 h | 4 wks |
| 2016 | Hanssen | [34] | Humans (Overweight, T2DM) | Pre vs. Post ICE | Air | daily | 15 °C | 6 h | 10 days |
| 2017 | Blondin | [35] | Humans | Pre vs. Post ICE | Liquid-cooled suit | daily | 10 °C | 2 h | 4 wks |
| 2019 | Presby | [36] | Obese Mice Caloric Restriction During ICE 24 h ad libitum after ICE | 1. Caloric Restriction 2. ICE + Caloric Restriction | Air | 5 d/wk | 4 °C | 1.5 h | 3 wks |
| 2021 | Soberg | [16] | Humans | 1. Controls 2. Winter Swimmers | Winter swimming | 2–3 d/wk | NA | 11 min | At least 1 season |
| 2021 | Zhang | [37] | Rats | 1. Control 2. ICE | Air | daily | 10 °C | 4 h | 2 wks |
| 2022 | McKie | [38] | Obese Mice | 1. HFD 2. ICE + HFD | Air | 5 d/wk | 4 °C | 1 h | 4 wks |
| 2023 | Weng | [39] | Obese Rats | 1. Control 2. Exercise 3. ACE 4. ACE + Exercise 5. CCE 6. CCE + Exercise 7. ICE 8. ICE + Exercise | Air | daily | 3–4 °C | 4 h | 5 wks |
| 2023 | Nema | [9] | Humans (Soldiers) | 1. Control Pre vs. Post 2. ICE Pre vs. Post | CWI and CS | CWI ≥ 1/wk CS ≥ 4/wk | CWI ≤ 6 °C CS ≤ 10 °C | CWI ≥3 min CS ≥ 30 s | 8 wks |
| Year | First Author | Citation | ICE Outcomes | Model |
|---|---|---|---|---|
| 1979 | Doi | [21] | ↑BW (adults), ↔BW (newborns), ↑BAT, ↑EE in response to NorEp, ↑NST, ↓ST | Rats |
| 1984 | Harri | [22] | ↓Weight gain, ↔BW (trended ↓), ↑BAT, ↑NST, ↓ST | Rats |
| 1986 | Arnold | [19] | ↓BW, ↓FFM, ↓FM, ↑BAT, ↑EE, ↑EI | Rats |
| 1988 | Deshaies | [20] | ↓BW, ↓eWAT and %eWAT, ↑BAT and %BAT | Rats |
| 1989 | Yahata | [23] | ↓BW, ↓eWAT, ↓BAT, ↑EI | Rats |
| 2013 | Yoneshiro | [24] | ↔BW, ↓FM, ↔FFM. ↑BAT activity during CE, ↑EE during CE | Humans (No BAT Activity During Initial ACE) |
| 2014 | Yoo | [25] | ↑BW, ↔FFM, ↑FM, ↑iWAT, ↑iWAT adipocyte size, ↑iWAT beiging, ↑eWAT, ↓eWAT adipocyte size, ↑BAT, ↔EE | Mice |
| 2014 | Qiao | [26] | Adipoq−/−: ↑iWAT beiging, ↓thermogenesis | Mice |
| 2014 | Ravussin | [27] | Cohort 1 (1 or 4 h ICE): ↔BW, ↔FM, ↔FFM, ↔iWAT, ↔eWAT, ↔BAT, ↑EE (4 h), ↑EI (4 h) Cohort 2 (4 or 8h ICE): ↔BW, ↔FM, ↔FFM, ↔iWAT, ↑eWAT (4 & 8 h), ↔BAT, ↑EE (4 h), ↑↑EE (8 h), ↑EI (4 h), ↑↑EI (8 h) | Mice |
| 2014 | Blondin | [28] | ↑BAT activity during CE, ↓skin temp, ↓ST | Humans |
| 2015 | Wang | [29] | ↔BW, ↔sWAT, ↓sWAT adipocyte size, ↓vWAT, ↑BAT activity, ↑BAT adipocyte number | Mice |
| 2015 | Bai | [30] | ↔BW, ↔sWAT, ↓sWAT adipocyte size, ↑pericardial WAT, ↓pericardial adipocyte size, ↑pericardial WAT beiging | Platue Pika (Rodent) |
| 2015 | Hanssen | [31] | ↑BAT activity during CE, ↑EE during CE, ↓basal metabolic rate at thermoneutrality | Humans (Overweight,T2D, Males) |
| 2016 | Gibas-Dorna | [32] | Winter swimmers vs. controls: ↑BW, ↑FM, ↓vWAT Winter swimmers (pre vs. post winter swimming season): ↔BW, ↔FM, ↔vWAT | Humans |
| 2016 | Tsibul’nikov | [33] | ↑BW (↑8 h, ↑↑1.5 h), ↑BAT weight (8 h) | Rats |
| 2016 | Hanssen | [34] | ↑BAT activity during CE | Humans (Obese) |
| 2017 | Blondin | [35] | ↔BW, ↑BAT volume, ↑BAT activity with CE, ↓skin temp, ↓ST, ↑EE with CE, ↔fuel utilization during CE | Humans |
| 2019 | Presby | [36] | ↔BW, ↔FM, ↔FFM, ↔iWAT, ↔eWAT, ↑sWAT beiging, ↑BAT weight, ↑BAT adipocyte size, ↑EE during CE, ↓EE during dark cycle, ↑EE during light cycle | Obese Mice Caloric Restriction During ICE 24 h ad libitum after ICE |
| 2021 | Soberg | [16] | Winter swimmers: higher supraclavicular skin temp in response to cold exposure, no BAT glucose uptake at thermoneutrality (controls had glucose uptake at thermoneutrality), ↑glucose uptake in perirenal BAT during cold exposure (not significant for control), ↑REE during cold exposure | Humans |
| 2021 | Zhang | [37] | ↓BW, ↑EI (trend) | Rats |
| 2022 | McKie | [38] | ↑BW, ↑iWAT, ↑eWAT, ↑BAT, ↑EI, ↑↑EI (within 4 h post ICE) | Obese Mice |
| 2023 | Weng | [39] | ↑sWAT, ↔vWAT | Obese Rats |
| 2023 | Nema | [9] | ↔BW, ↔BMI, ↔FM, ↔FFM, ↓Waist Circumference (men only) | Humans (Soldiers) |
| Year | First Author | Citation | ICE Outcomes | Model |
|---|---|---|---|---|
| 1984 | Harri | [22] | ↑Citrate synthase in BAT, ↑cytochrome C oxidase in BAT | Rats |
| 1988 | Deshaies | [20] | ↔eWAT LPL | Rats |
| 1989 | Yahata | [23] | ↑BAT glucagon | Rats |
| 2014 | Yoo | [25] | ↑iWAT Ucp1 and UCP1, ↑BAT UCP1, ↑eWAT lipogenic gene expression (Scd1, Lpl, Pparγ) | Mice |
| 2014 | Qiao | [26] | Adipoq−/−: ↑iWAT UCP1 | Mice |
| 2014 | Ravussin | [27] | Cohort 1 (1 or 4 h ICE): ↑BAT Ucp1 (4 h), ↑BAT Pgc1α (4 h), ↓serum leptin (4 h) Cohort 2 (4 or 8 h ICE): ↑BAT UCP1 (4 & 8 h) | Mice |
| 2015 | Wang | [29] | ↑sWAT UCP1 and PGC1α, ↔BAT UCP1 (trended↑) | Mice |
| 2015 | Bai | [30] | ↑BAT thermogenic gene expression (Pgc1α, Dio2, Cidea) and adipogenic gene expression (Adipoq, Cebpα, Pparγ, Fabp4), ↑pericardial WAT thermogenic gene expression (Ucp1 and Pgc1α), ↑pericardial WAT UCP1 staining and mito activity genes (Cox8 and ATP5α), ↔serum leptin | Platue Pika (Rodent) |
| 2019 | Presby | [36] | ↑sWAT Dio2, ↑sWAT UCP1, ↑BAT Dio2, ↑BAT UCP1 | Obese Mice Caloric Restriction During ICE 24 h ad libitum after ICE |
| 2021 | Zhang | [37] | ↑peri-ovarian adipose thermogenic gene expression (Ucp1, PGC1α, Prdm16, Fndc5) | Rats |
| 2023 | Weng | [39] | ↔iWAT ATGL activity, ↑iWAT LPL activity | Obese Rats |
| Year | First Author | Citation | ICE Outcomes | Model |
|---|---|---|---|---|
| 1988 | Deshaies | [20] | ↓serum cholesterol, ↔HDL | Rats |
| 1989 | Yahata | [23] | ↑plasma glucagon, ↑corticosterone | Rats |
| 2014 | Yoo | [25] | ↔IS, ↑liver TGs and hepatic TG release, ↑DNL | Mice |
| 2014 | Ravussin | [27] | cohort 1 (1 or 4 h ICE): ↑BAT glucose uptake cohort 2 (4 or 8 h ICE): ↑FFA (4 & 8 h), ↓insulin | Mice |
| 2014 | Blondin | [28] | ↓BG, ↓cortisol | Humans |
| 2015 | Wang | [29] | ↑glucose tolerance, ↑insulin sensitivity | Mice |
| 2015 | Bai | [30] | ↔serum glucose, ↔serum TG | Platue Pika (Rodent) |
| 2015 | Hanssen | [31] | ↑IS, ↑GLUT4 translocation in skeletal muscle | Humans (Overweight,T2D, Males) |
| 2016 | Gibas-Dorna | [32] | ↑IS during the portion of the winter swimming season where water temperature was <8 °C | Humans |
| 2016 | Hanssen | [34] | ↑skeletal muscle glucose uptake during ICE, ↑GLUT4 translocation in skeletal muscle | Humans (Obese) |
| 2017 | Blondin | [35] | ↔FG | Humans |
| 2021 | Soberg | [16] | winter swimmers had ↓plasma glucose during IGTT | Humans |
| 2022 | McKie | [38] | ↑glucose tolerance, ↑insulin and c-peptide in response to glucose, ↔IS | Obese Mice |
| 2023 | Weng | [39] | ↔BG, ↔insulin, ↓HOMA-IR, ↔FFA | Obese Rats |
| Year | First Author | Citation | Intervention | Outcomes | Model |
|---|---|---|---|---|---|
| 1986 | Arnold | [19] | ICE | ↓BW, ↓FFM, ↓FM, ↑BAT, ↑EE, ↑EI | Rats |
| Ex | ↓↓BW, ↓FFM, ↓BAT, ↓↓FM, ↔EE, ↓EI | ||||
| ICE + Ex | ↓↓BW, ↓FFM, ↓↓FM, ↔BAT, ↓EI, ↔EE | ||||
| 1988 | Deshaies | [20] | ICE | ↓BW, ↓eWAT, ↑BAT, ↑EI, ↔TG, ↓Cholesterol | Rats |
| Ex | ↓↓BW, ↓↓eWAT, ↔BAT, ↓EI, ↓TG, ↔Cholesterol | ||||
| ICE + Ex | ↓↓BW, ↓↓eWAT, ↔BAT, ↓EI, ↓TG, ↔Cholesterol | ||||
| 2023 | Weng | [39] | ICE | ↔sWAT (trend↑), ↑sWAT LPL activity, ↑muscle PGC1α, ↑muscle p38MAPK | Obese Rats |
| Ex | ↔sWAT, ↑sWAT LPL activity, ↑muscle PGC1α, ↑muscle p38MAPK | ||||
| ICE + Ex | ↔sWAT, ↑↑sWAT LPL activity |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scott, M.C.; Fuller, S. The Effects of Intermittent Cold Exposure on Adipose Tissue. Int. J. Mol. Sci. 2024, 25, 46. https://doi.org/10.3390/ijms25010046
Scott MC, Fuller S. The Effects of Intermittent Cold Exposure on Adipose Tissue. International Journal of Molecular Sciences. 2024; 25(1):46. https://doi.org/10.3390/ijms25010046
Chicago/Turabian StyleScott, Matthew C., and Scott Fuller. 2024. "The Effects of Intermittent Cold Exposure on Adipose Tissue" International Journal of Molecular Sciences 25, no. 1: 46. https://doi.org/10.3390/ijms25010046
APA StyleScott, M. C., & Fuller, S. (2024). The Effects of Intermittent Cold Exposure on Adipose Tissue. International Journal of Molecular Sciences, 25(1), 46. https://doi.org/10.3390/ijms25010046