Na+ Lattice Doping Induces Oxygen Vacancies to Achieve High Capacity and Mitigate Voltage Decay of Li-Rich Cathodes
Abstract
1. Introduction
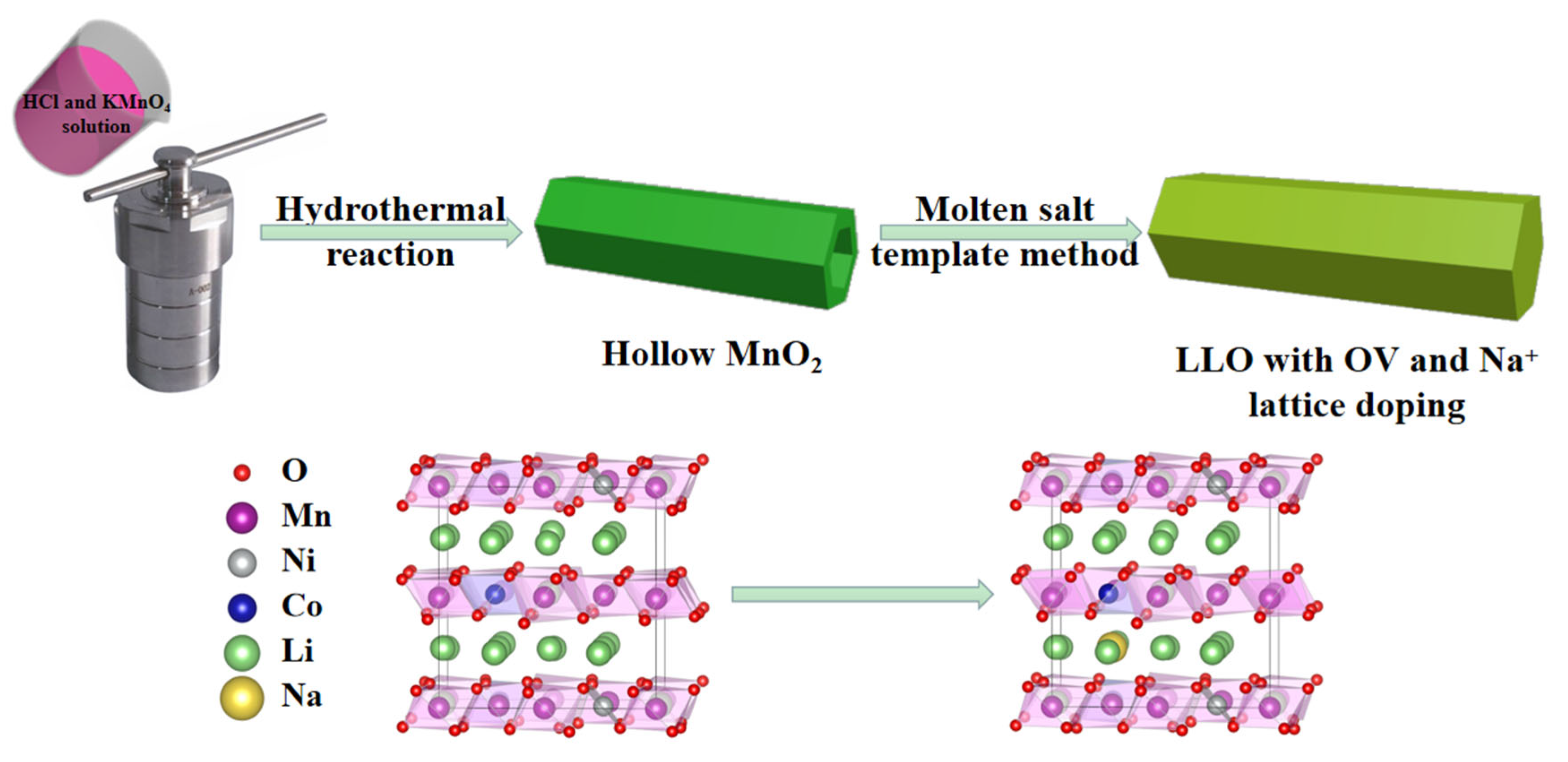
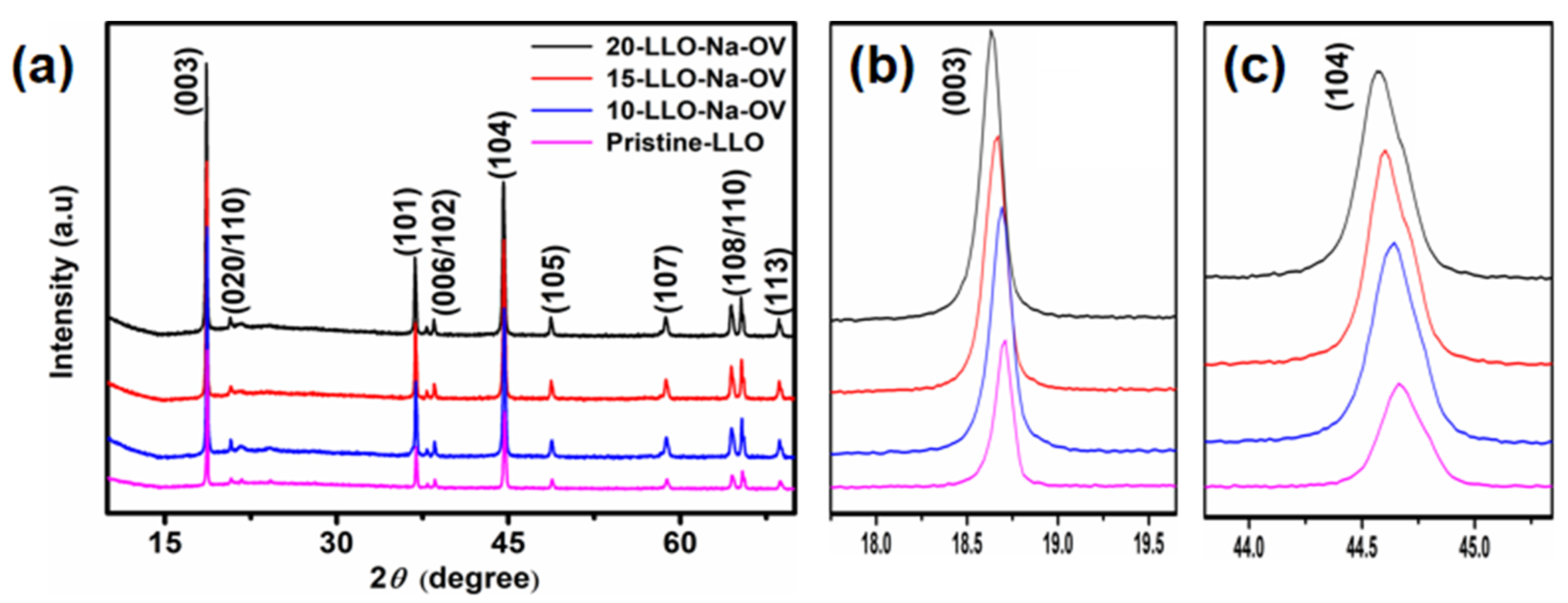
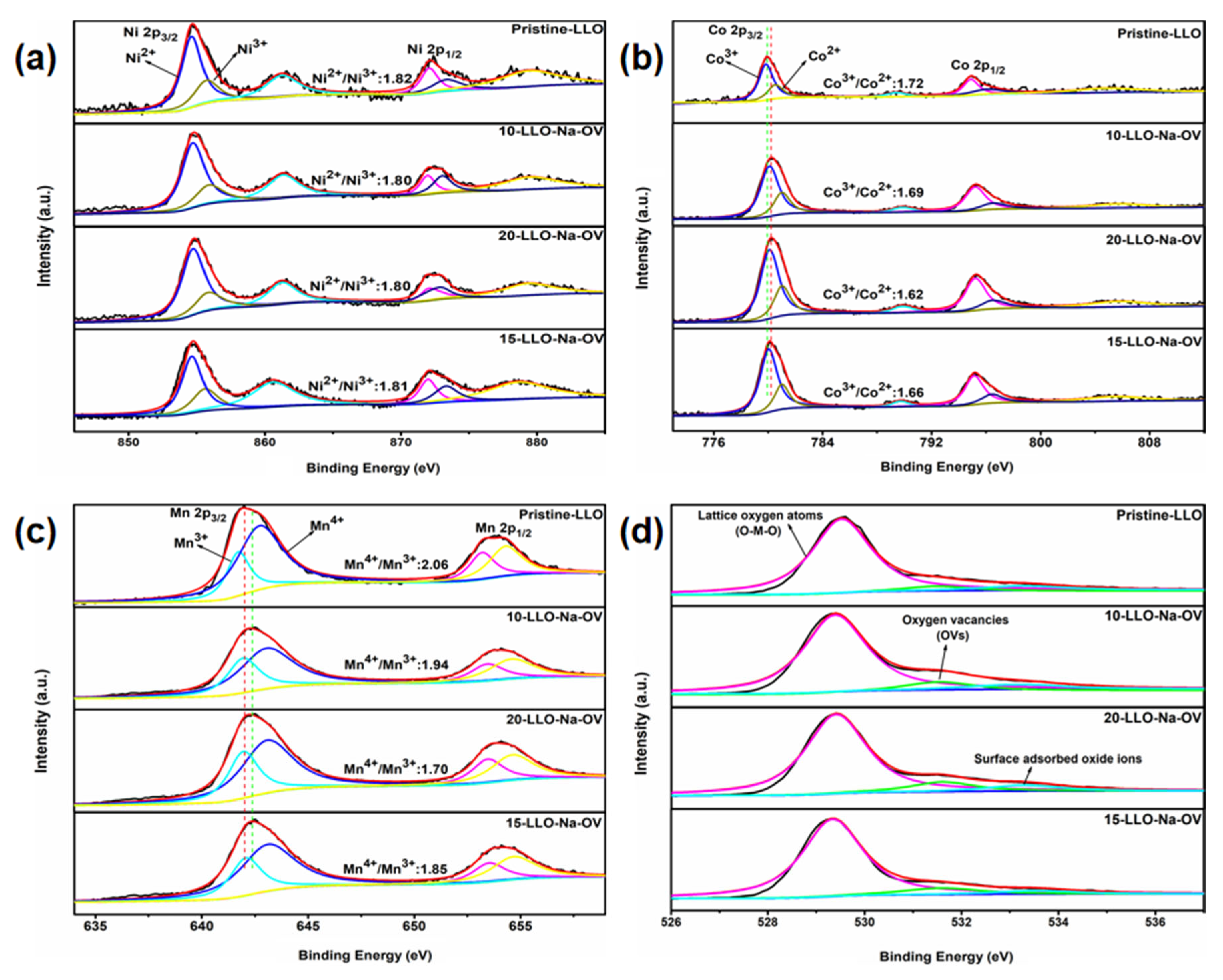
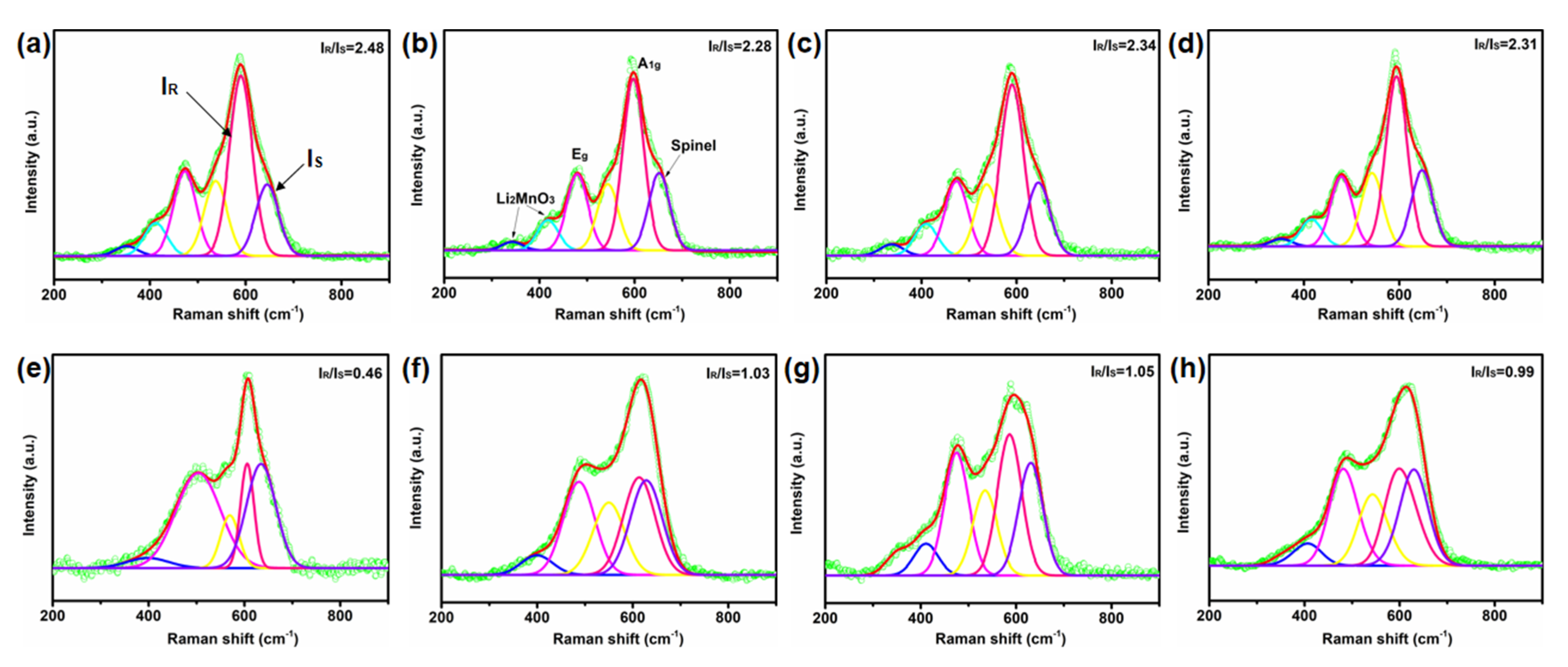
2. Results and Discussions
3. Materials and Methods
3.1. Materials and Chemical Reagents
3.2. Synthetic Process
3.3. Material Characterization
3.4. Electrochemical Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Armand, M.; Tarascon, J. Building better batteries. Nature 2008, 451, 652. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.; Myeong, S.; Hwang, J.; Jang, H.; Sung, J.; Yoo, Y.; Kim, M.; Cho, J. Unraveling the rapid redox behavior of Li-excess 3d-transition metal oxides for high rate capability. Adv. Energy Mater. 2020, 10, 1904092. [Google Scholar] [CrossRef]
- Richard, S.; Ralf, W.; Gerhard, H.; Tobias, P.; Martin, W. Performance and cost of materials for lithium-based rechargeable automotive batteries. Nat. Energy 2018, 3, 267–278. [Google Scholar]
- Choi, J.W.; Aurbach, D. Promise and reality of post-lithium-ion batteries with high energy densities. Nat. Rev. Mater. 2016, 1, 16013. [Google Scholar] [CrossRef]
- Kim, T.; Song, W.T.; Son, D.Y.; Ono, L.K.; Qi, Y.B. Lithium-ion batteries: Outlook on present, future, and hybridized technologies. J. Mater. Chem. A 2019, 7, 2942–2964. [Google Scholar] [CrossRef]
- McCalla, E.; Abakumov, A.M.; Saubanere, M.; Foix, D.; Berg, E.J.; Rousse, G.; Doublet, M.L.; Gonbeau, D.; Novak, P.; Tendeloo, G.V.; et al. Visualization of O-O peroxo-like dimers in high-capacity layered oxides for Li-ion batteries. Science 2015, 350, 1516–1521. [Google Scholar] [CrossRef]
- Croy, J.R.; Iddir, H.; Gallagher, K.; Johnson, C.S.; Benedek, R.; Balasubramanian, M. First-charge instabilities of layered-layered lithium-ion-battery materials. Phys. Chem. Chem. Phys. 2015, 17, 24382–24391. [Google Scholar] [CrossRef]
- Hong, J.H.; Gent, W.E.; Xiao, P.H.; Lim, K.; Seo, D.H.; Wu, J.P.; Csernica, P.M.; Takacs, C.J.; Nordlund, D.; Sun, C.J.; et al. Metal-oxygen decoordination stabilizes anion redox in Li-rich oxides. Nat. Mater. 2019, 18, 256–265. [Google Scholar] [CrossRef]
- Shi, J.L.; Xiao, D.D.; Ge, M.Y.; Yu, X.Q.; Chu, Y.; Huang, X.J.; Zhang, X.D.; Yin, Y.X.; Yang, X.Q.; Guo, Y.G. High-capacity cathode material with high voltage for Li-ion batteries. Adv. Mater. 2018, 30, 1705575. [Google Scholar] [CrossRef]
- Erickson, E.M.; Sclar, H.; Schipper, F.; Liu, J.; Tian, R.; Ghanty, C.; Burstein, L.; Leifer, N.; Grinblat, J.; Talianker, M.; et al. High-temperature treatment of Li-rich cathode materials with ammonia: Improved capacity and mean voltage stability during cycling. Adv. Energy Mater. 2017, 7, 1700708. [Google Scholar] [CrossRef]
- Sathiya, M.; Abakumov, A.M.; Foix, D.; Rousse, G.; Ramesha, K.; Sauban, M.; Doublet, M.; Vezin, H.; Laisa, C.; Prakash, A. Origin of voltage decay in high-capacity layered oxide electrodes. Nat. Mater. 2015, 14, 230–238. [Google Scholar] [CrossRef]
- Lee, S.; Jin, W.; Kim, S.H.; Joo, S.H.; Nam, G.; Oh, P.; Kim, Y.; Kwak, S.K.; Cho, J. Oxygen Vacancy Diffusion and Condensation in Lithium-Ion Battery Cathode Materials. Angew. Chem. Int. Edit. 2019, 58, 10478–10485. [Google Scholar] [CrossRef]
- Qiu, B.; Zhang, M.; Wu, L.; Wang, J.; Xia, Y.; Qian, D.; Liu, H.D.; Hy, S.; Chen, Y.; An, K.; et al. Gas-solid interfacial modification of oxygen activity in layered oxide cathodes for lithium-ion batteries. Nat. Commun. 2016, 7, 12108. [Google Scholar] [CrossRef]
- An, J.; Shi, L.; Chen, G.; Li, M.; Liu, H.; Yuan, S.; Chen, S.; Zhang, D. Insights into the stable layered structure of a Li-rich cathode material for lithium-ion batteries. J. Mater. Chem. A 2017, 5, 19738–19744. [Google Scholar] [CrossRef]
- Li, N.; He, Y.S.; Wang, X.; Zhang, W.; Ma, Z.F.; Zhang, D. Incorporation of rubidium cations into Li1.2Mn0.54Co0.13Ni0.13O2 layered oxide cathodes for improved cycling stability. Electrochim. Acta 2017, 231, 363. [Google Scholar] [CrossRef]
- Hashem, A.M.; Abdel-Ghany, A.E.; El-Tawil, R.S.; Mauger, A.; Julien, C.M. Effect of Na Doping on the Electrochemical Performance of Li1.2Ni0.13Co0.13Mn0.54O2 Cathode for Lithium-Ion Batteries. Sustain. Chem. 2022, 3, 131–148. [Google Scholar] [CrossRef]
- Kim, S.; Kim, M.; Kwak, D.; Kim, D.; Lee, G.; Choe, H.; Park, K. Highly stable TiO2 coated Li2MnO3 cathode materials for lithium-ion batteries. J. Power Sources 2016, 304, 119–127. [Google Scholar] [CrossRef]
- Song, J.H.; Kapylou, A.; Choi, H.S.; Yu, B.Y.; Matulevich, E.; Kang, S.H. Suppression of irreversible capacity loss in Li-rich layered oxide by fluorine doping. J. Power Sources 2016, 313, 65–72. [Google Scholar] [CrossRef]
- Kim, H.; Choi, A.; Doo, S.W.; Lim, J.; Kim, Y.; Lee, K.T. Role of Na+ in the Cation Disorder of [Li1-xNax]NiO2 as a Cathode for Lithium-Ion Batteries. J. Electrochem. Soc. 2018, 165, A201. [Google Scholar] [CrossRef]
- Park, H.G.; Min, K.; Park, K. A Synergistic Effect of Na+ and Al3+ Dual Doping on Electrochemical Performance and Structural Stability of LiNi0.88Co0.08Mn0.04O2 Cathodes for Li-Ion Batteries. ACS Appl. Mater. Interfaces 2022, 14, 5168–5176. [Google Scholar] [CrossRef]
- Hu, G.; Zhang, M.; Liang, L.; Peng, Z.; Du, K.; Cao, Y. Mg-Al-B Co-substitution LiNi0.5Co0.2Mn0.3O2 Cathode Materials with Improved Cycling Performance for Lithium-ion Battery under High Cutoff Voltage. Electrochim. Acta 2016, 190, 264–275. [Google Scholar] [CrossRef]
- Vanaphuti, P.; Chen, J.; Cao, J.; Bigham, K.; Chen, B.; Yang, L.; Chen, H.; Wang, Y. Enhanced Electrochemical Performance of the Lithium-Manganese-Rich Cathode for Li-Ion Batteries with Na and F CoDoping. Acs Appl. Mater. Interfaces 2019, 11, 37842–37849. [Google Scholar] [CrossRef] [PubMed]
- Sattar, T.; Lee, S.; Sim, S.; Jin, B.; Kim, H. Effect of Mg-doping on the electrochemical performance of LiNi0.84Co0.11Mn0.05O2 cathode for lithium ion batteries. Int. J. Hydrog. Energy 2020, 45, 19567–19576. [Google Scholar] [CrossRef]
- Jeong, S.; Choi, K.; Ho, V.; Cho, J.; Bae, J.; Nam, S.C.; Yim, T.; Mun, J. Crucial role of Ni-doping to interfacial Li2MnO3 layer of High-performance Ni-rich layered cathode in Lithium-Ion batteries. Chem. Eng. J. 2022, 434, 134577. [Google Scholar] [CrossRef]
- Sanad, M.M.S.; Toghan, A. Unveiling the role of trivalent cation incorporation in Li-rich Mn-based layered cathode materials for low-cost lithium-ion batteries. Applied Physics A 2021, 127, 733. [Google Scholar] [CrossRef]
- Qing, R.P.; Shi, J.L.; Xiao, D.D.; Zhang, X.D.; Yin, Y.X.; Zhai, Y.B.; Gu, L.; Guo, Y.G. Enhancing the kinetics of Li-rich cathode materials through the pinning effects of gradient surface Na+ doping. Adv. Energy Mater. 2016, 6, 1501914. [Google Scholar] [CrossRef]
- Shaju, K.M.; Subba-Rao, G.V.; Chowdari, B.V.R. Performance of layered Li(Ni1/3Co1/3Mn1/3)O2 as cathode for Li-ion batteries. Electrochim. Acta 2002, 48, 145–151. [Google Scholar] [CrossRef]
- Ramakrishnan, S.; Park, B.; Wu, J.; Yang, W.; McCloskey, B.D. Extended Interfacial Stability through Simple Acid Rinsing in a Li-Rich Oxide Cathode Material. J. Am. Chem. Soc. 2020, 142, 8522–8531. [Google Scholar] [CrossRef]
- Sathiya, M.; Rousse, G.; Ramesha, K.; Laisa, C.P.; Vezin, H.; Sougrati, M.T.; Doublet, M.L.; Foix, D.; Gonbeau, D.; Walker, W.; et al. Reversible anionic redox chemistry in high-capacity layered-oxide electrodes. Nat. Mater. 2013, 12, 827–835. [Google Scholar] [CrossRef]
- Vahdatkhah, P.; Voznyy, O.; Sadrnezhaad, S.K. Engineering of surface oxygen vacancies in Co-free concentration-gradient Li-rich cathodes for high-capacity batteries. Mater. Today Sustain. 2023, 21, 100309. [Google Scholar] [CrossRef]
- Sun, Z.; Xu, L.; Dong, C.; Zhang, H.; Zhang, L.; Ma, Y.; Liu, Y.; Li, Z.; Zhou, Y.; Chen, Y. A facile gaseous sulfur treatment strategy for Li-rich and Ni-rich cathode materials with high cycling and rate performance. Nano Energy 2019, 63, 103887. [Google Scholar] [CrossRef]
- Lee, E.; Persson, K.A. Structural and chemical evolution of the layered Li-excess LixMnO3 as a function of Li content from first-principles calculations. Adv. Energy Mater. 2014, 4, 1400498. [Google Scholar] [CrossRef]
- Momeni, M.; Mashhour, H.Y.; Kalantarian, M.M. New approaches to consider electrical properties. band gaps and rate capability of same-structured cathode materials using density of states diagrams: Layered oxides as a case study. J. Alloys Compd. 2019, 787, 738–743. [Google Scholar] [CrossRef]
- Gao, Y.; Ma, J.; Wang, X.; Lu, X.; Bai, Y.; Wang, Z.; Chen, L. Improved electron/Li-ion transport and oxygen stability of Mo-doped Li2MnO3. J. Mater. Chem. A 2014, 2, 4811–4818. [Google Scholar] [CrossRef]
- Kim, J.S.; Johnson, C.S.; Thackeray, M.M. Layered xLiMO2·(1-x)Li2M′O3 electrodes for lithium batteries: A study of 0.95LiMn0.5Ni0.5O2·0.05Li2TiO3. Electrochem. Commun. 2002, 4, 205–209. [Google Scholar] [CrossRef]
- Muhammad, S.; Kim, H.; Kim, Y.; Kim, D.; Song, J.H.; Yoon, J.; Park, J.; Ahn, S.; Kang, S.; Thackeray, M.M.; et al. Evidence of reversible oxygen participation in anomalously high capacity Li- and Mn-rich cathodes for Li-ion batteries. Nano Energy 2016, 21, 172–184. [Google Scholar] [CrossRef]
- Wang, J.; He, X.; Paillard, E.; Laszczynski, N.; Li, J.; Passerini, S. Lithium- and Manganese-Rich Oxide Cathode Materials for High-Energy Lithium Ion Batteries. Adv. Energy Mater. 2016, 6, 1600906. [Google Scholar] [CrossRef]
- Basu, S.; Dahiya, P.P.; Akhtar, M.; Ray, S.K.; Chang, J.K.; Majumder, S.B. High energy density layered-spinel hybrid cathodes for lithium ion rechargeable batteries. Mat. Sci. Eng. B 2016, 213, 148–156. [Google Scholar] [CrossRef]
- Mohanan, P.A.; Salini, P.S.; John, B.; SarojiniAmma, S.; Thelakkattu, D.M. Synthesis and Electrochemical Characterization of a Li-Rich Li1.17Ni0.34Mn0.5O2 Cathode Material for Lithium-Ion Cells. Energy Fuels 2022, 36, 11186–11193. [Google Scholar] [CrossRef]
- Nguyen, T.L.; Vo, T.N.; Phung, V.D.; Ayalew, K.; Chun, D.; Luu, A.T.; Nguyen, Q.H.; Kim, K.J.; Kim, I.T.; Moon, J. Li-ion storage in orthorhombic hydrated sodium molybdate with oxygen-vacancy defects. Chem. Eng. J. 2022, 446, 137174. [Google Scholar] [CrossRef]
- Guo, H.; Wei, Z.; Jia, K.; Qiu, B.; Yin, C.; Meng, F.; Zhang, Q.; Gu, L.; Han, S.; Liu, Y.; et al. Abundant Nanoscale Defects to Eliminate Voltage Decay in Li-rich Cathode Materials. Energy Storage Mater. 2019, 16, 220–227. [Google Scholar] [CrossRef]
- Freire, M.; Lebedev, O.I.; Maignan, A.; Jordy, C.; Pralong, V. Nanostructured Li2MnO3: A disordered rock salt type structure for high energy density Li ion batteries. J. Mater. Chem. A 2017, 5, 21898–21902. [Google Scholar] [CrossRef]
- Bao, L.; Wei, L.; Fu, N.; Dong, J.; Chen, L.; Su, Y.; Li, N.; Lu, Y.; Li, Y.; Chen, S.; et al. Urea assisted mixed gas treatment on Li-Rich layered oxide with enhanced electrochemical performance. J. Energy Chem. 2022, 66, 123–132. [Google Scholar] [CrossRef]
- Sun, G.; Zhao, C.; Yu, F.-D.; Yu, R.; Wang, J.; Zhou, J.; Shao, G.; Sun, X.; Wang, Z.-B. Insitu surface chemical and structural self-reconstruction strategy enables high performance of Li-rich cathode. Nano Energy 2021, 79, 105459. [Google Scholar] [CrossRef]
- Vivekanantha, M.; Saravanan, R.S.A.; Nayak, P.K.; Prakash, R.; Bharathi, K.K. Synergistic-effect of high Ni content and Na dopant towards developing a highly stable Li-Rich cathode in Li-ion batteries. Chem. Eng. J. 2022, 444, 136503. [Google Scholar] [CrossRef]
- Liu, Y.; Zeng, W.T.; Zhu, X.F.; Chen, Z.Y.; He, H.; Chen, X.Y.; Yan, W.L. Improving the electrochemical performance of Li1.2Ni0.13Co0.13Mn0.54O2 cathode through sodium doping. Electrochim. Acta 2022, 404, 139775. [Google Scholar]
- Liu, Y.; Ning, D.; Zheng, L.; Zhang, Q.; Gu, L.; Gao, R.; Zhang, J.; Franz, A.; Schumacher, G.; Liu, X. Improving the electrochemical performances of Li-rich Li1.20Ni0.13Co0.13Mn0.54O2 through a cooperative doping of Na+ and PO43- with Na3PO4. J. Power Sources 2018, 375, 1–10. [Google Scholar] [CrossRef]
- Choi, W.; Shin, H.C.; Kim, J.M.; Choi, J.Y.; Yoon, W.S. Modeling and applications of electrochemical impedance spectroscopy (EIS) for lithium-ion batteries. J. Electrochem. Sci. Te. 2020, 11, 1–13. [Google Scholar] [CrossRef]
- Aida, T.; Tsutsui, Y.; Kanada, S.; Okada, J.; Hayashi, K.; Komukai, T. Ammonium tungstate modified Li-rich Li1+xNi0.35Co0.35Mn0.30O2 to improve rate capability and productivity of lithium-ion batteries. J. Solid State Electrochem. 2017, 21, 2047–2054. [Google Scholar] [CrossRef]
- Vanaphuti, P.; Bai, J.; Ma, L.; Ehrlich, S.; Kisslinger, K.; Wang, F.; Wang, Y. Unraveling Na and F coupling effects in stabilizing Li, Mn-rich layered oxide cathodes via local ordering modification. Energy Storage Mater. 2020, 31, 459–469. [Google Scholar] [CrossRef]
- Vanaphuti, P.; Liu, Y.; Ma, X.; Fu, J.; Lin, Y.; Wen, J.; Yang, Z.; Wang, Y. Stabilized Lithium, Manganese-Rich Layered Cathode Materials Enabled by Integrating Co-Doping and Nanocoating. Acs Appl. Mater. Interfaces 2021, 13, 22597–22607. [Google Scholar] [CrossRef]
- Ehi-Eromosele, C.O.; Indris, S.; Melinte, G.; Bergfeldt, T.; Ehrenberg, H. Solution Combustion-Mechanochemical Syntheses of Composites and Core-Shell xLi2MnO3·(1-x)LiNi0.5Mn0.3Co0.2O2 (0≤x≤0.7) Cathode Materials for Lithium-Ion Batteries. Acs Sustain. Chem. Eng. 2020, 8, 18590–18605. [Google Scholar] [CrossRef]
- He, W.; Liu, P.F.; Qu, B.H.; Zheng, Z.M.; Zheng, H.F.; Deng, P.; Li, P.; Li, S.Y.; Huang, H.; Wang, L.S.; et al. Uniform Na+ Doping-Induced Defects in Li and Mn-Rich Cathodes for High-Performance Lithium-Ion Batteries. Adv. Sci. 2019, 6, 1802114. [Google Scholar] [CrossRef]
- Urbonaitea, S.; Halldahlb, L.; Svensson, G. Raman spectroscopy studies of carbide derived carbons. Carbon 2008, 46, 1942–1947. [Google Scholar] [CrossRef]
- Pidaparthy, S.; Luo, M.; Rodrigues, M.F.; Zuo, J.M.; Abraham, D.P. Physicochemical Heterogeneity in Silicon Anodes from Cycled Lithium-Ion Cells. ACS Appl. Mater. Interfaces 2022, 14, 38660–38668. [Google Scholar] [CrossRef]
- Hu, Y.Q.; Sun, Z.; Zhang, Z.L.; Liu, S.L.; He, F.B.; Liu, Y.; Zhuang, Z.; Liu, F.Y. Lithium-Rich Li2TiS3 Cathode Enables High-Energy Sulfide All-Solid-State Lithium Batteries. Adv. Energy Mater. 2023, 13, 2202756. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, H.; Zhang, R.; Zhang, Y. Na+ Lattice Doping Induces Oxygen Vacancies to Achieve High Capacity and Mitigate Voltage Decay of Li-Rich Cathodes. Int. J. Mol. Sci. 2023, 24, 8035. https://doi.org/10.3390/ijms24098035
Qiu H, Zhang R, Zhang Y. Na+ Lattice Doping Induces Oxygen Vacancies to Achieve High Capacity and Mitigate Voltage Decay of Li-Rich Cathodes. International Journal of Molecular Sciences. 2023; 24(9):8035. https://doi.org/10.3390/ijms24098035
Chicago/Turabian StyleQiu, Hengrui, Rui Zhang, and Youxiang Zhang. 2023. "Na+ Lattice Doping Induces Oxygen Vacancies to Achieve High Capacity and Mitigate Voltage Decay of Li-Rich Cathodes" International Journal of Molecular Sciences 24, no. 9: 8035. https://doi.org/10.3390/ijms24098035
APA StyleQiu, H., Zhang, R., & Zhang, Y. (2023). Na+ Lattice Doping Induces Oxygen Vacancies to Achieve High Capacity and Mitigate Voltage Decay of Li-Rich Cathodes. International Journal of Molecular Sciences, 24(9), 8035. https://doi.org/10.3390/ijms24098035




