Novel Strategy in Searching for Natural Compounds with Anti-Aging and Rejuvenating Potential
Abstract
1. Introduction
2. Results
2.1. Anti-Aging Strategies, Rejuvenation, Immortality
2.1.1. Age Assessment at the Body, Organ and Tissue Levels
2.1.2. Age Assessment for Cells and Cell Populations
2.1.3. Aging and Rejuvenation of a Cell Population as Reflected in the Age Profile
2.1.4. Aging and Rejuvenation of the Proliferation Niche
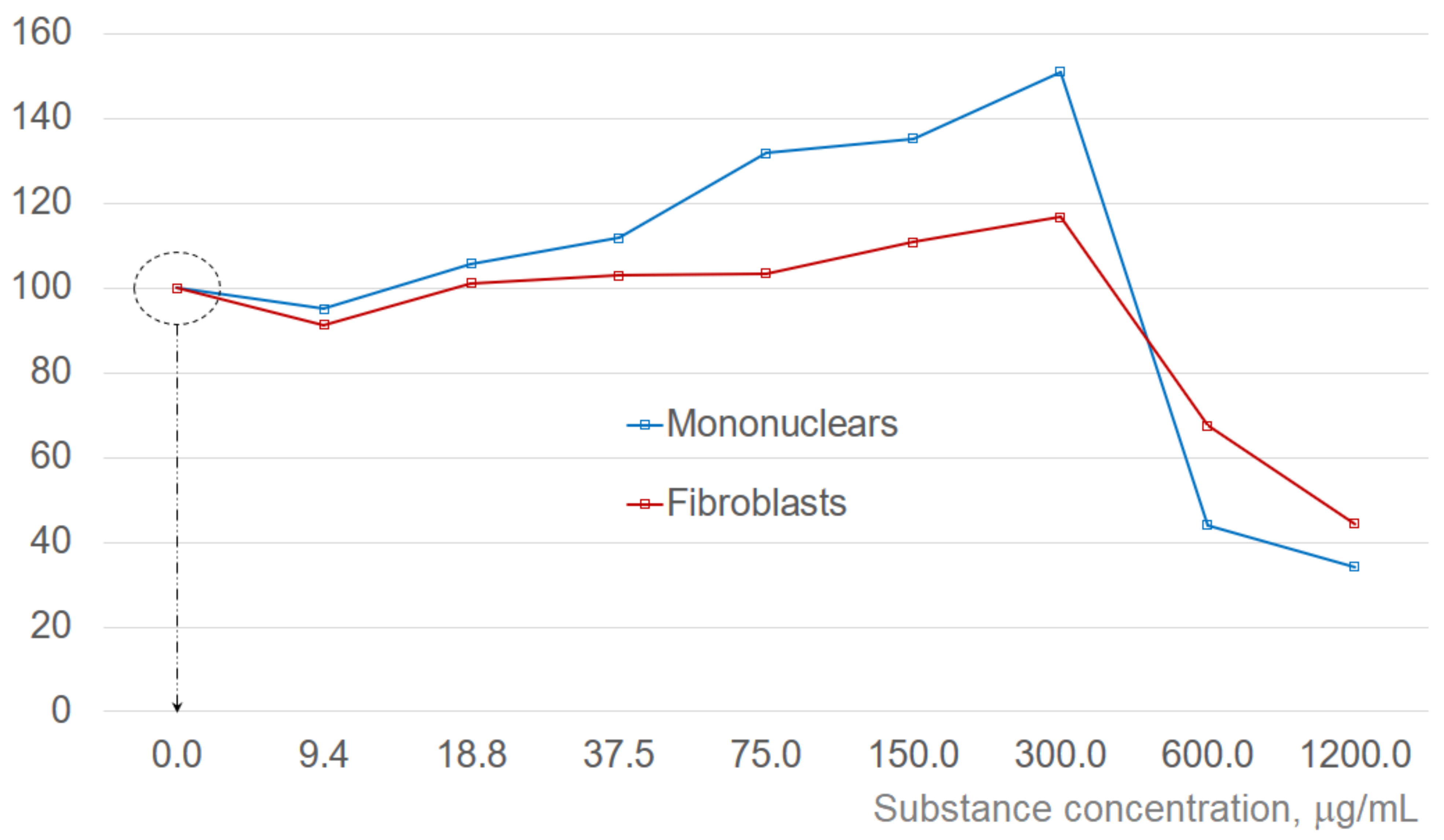
2.2. Screening for the Substances with Anti-Aging and Rejuvenation Potential
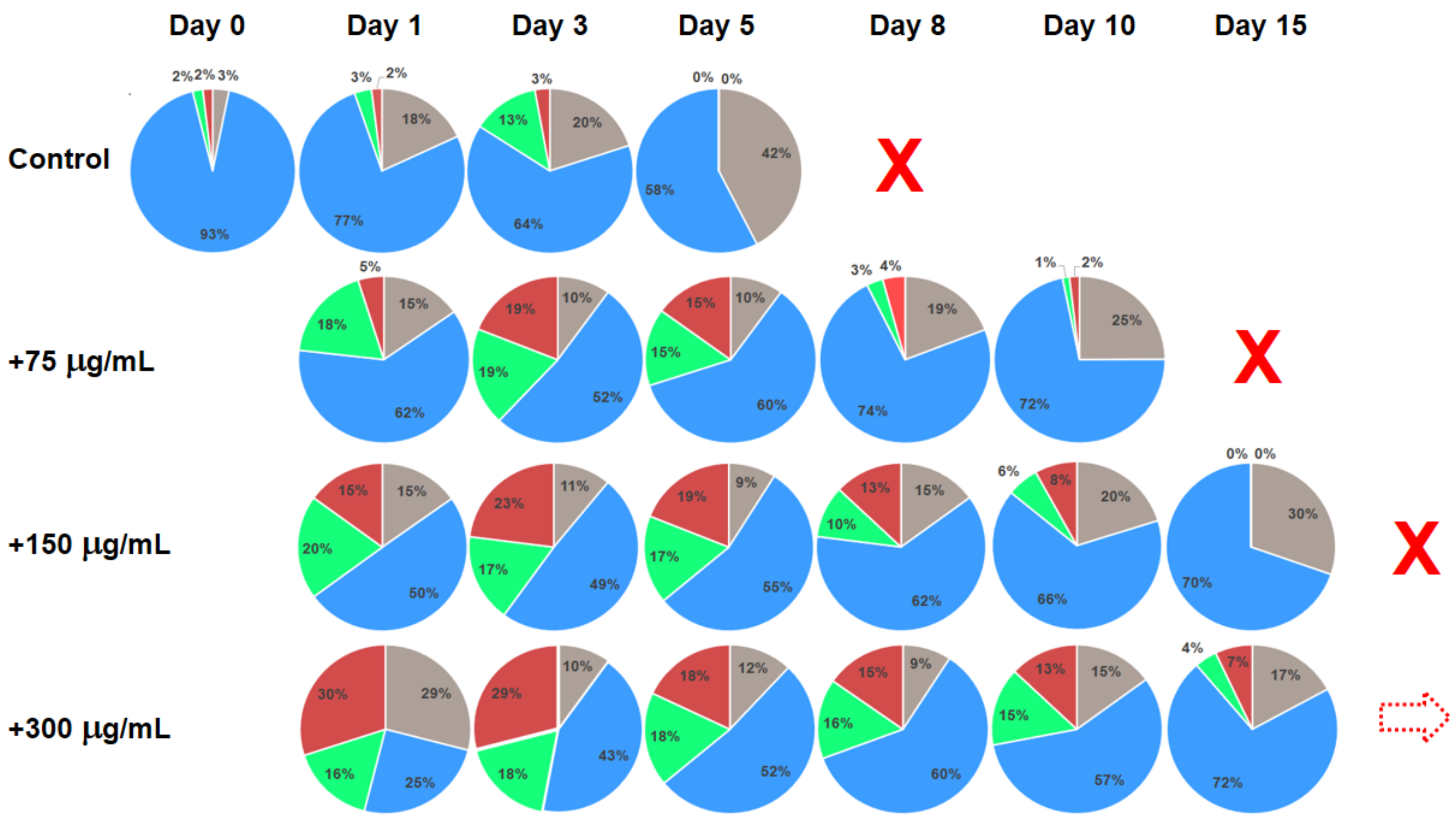
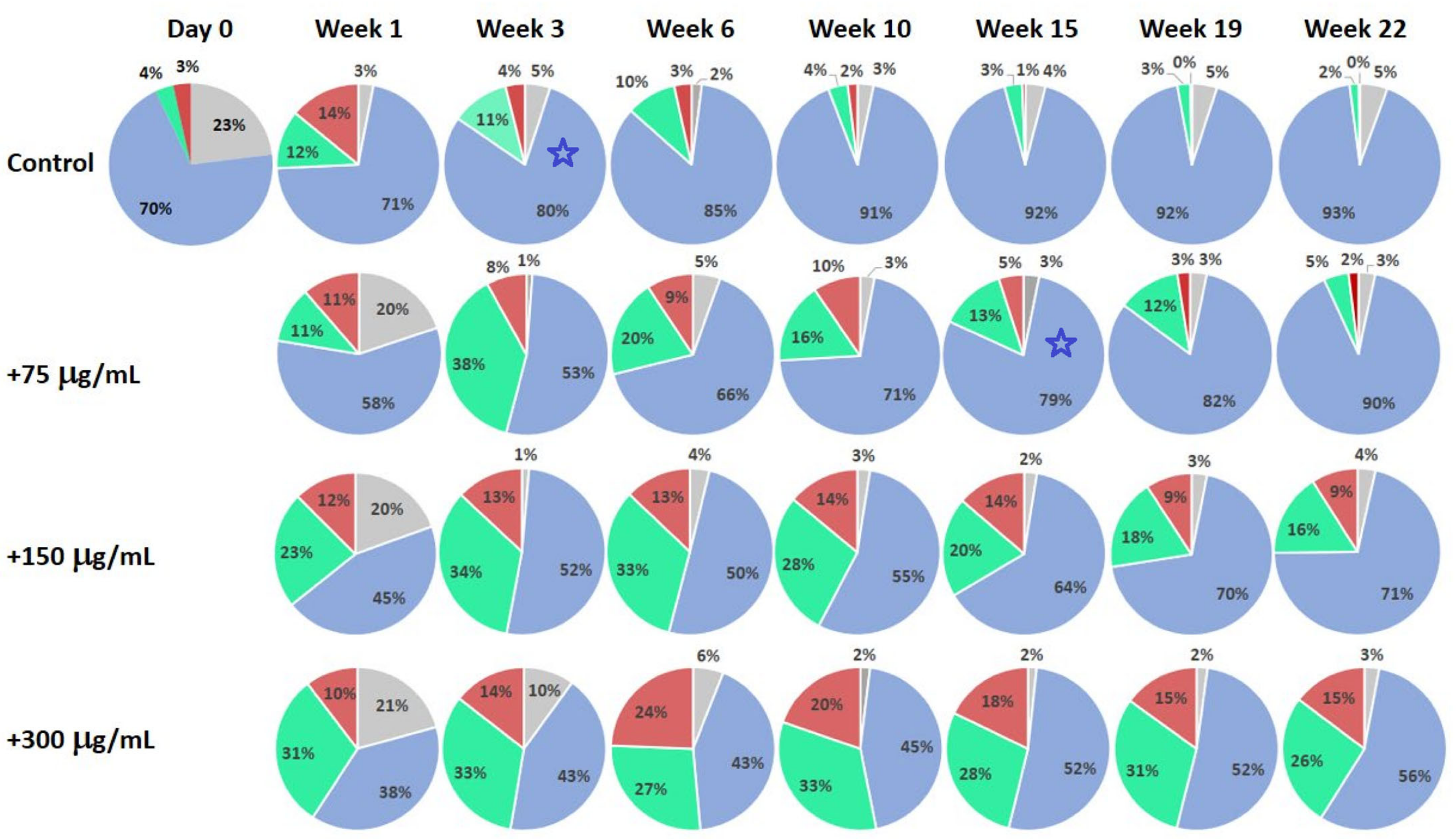
2.3. Example of Testing Natural Substances with Claimed Anti-Aging and Rejuvenation Potential
3. Discussion
4. Materials and Methods
4.1. Extraction and Cultivation of Human Blood Mononuclear Cells
4.2. Cultivation of the Human Fibroblasts
4.3. Flow Cytometry Analysis
4.4. Cell Viability Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Biesalski, H.-K.; Dragsted, L.O.; Elmadfa, I.; Grossklaus, R.; Müller, M.; Schrenk, D.; Walter, P.; Weber, P. Bioactive compounds: Definition and assessment of activity. Nutrition 2009, 25, 1202–1205. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.; Häder, D.-P. (Eds.) Natural Bioactive Compounds: Technological Advancements; Academic Press: London, UK, 2020. [Google Scholar] [CrossRef]
- Pai, S.; Hebbar, A.; Selvaraj, S. A critical look at challenges and future scopes of bioactive compounds and their incorporations in the food, energy, and pharmaceutical sector. Environ. Sci. Pollut. Res. 2022, 29, 35518–35541. [Google Scholar] [CrossRef] [PubMed]
- Apetrei, C. Frontiers in Bioactive Compounds. Natural Sources, Physicochemical Characterization and Applications; Bentham Science: Sharjah, United Arab Emirates, 2016; Volume 1. [Google Scholar] [CrossRef]
- Market Data Forecast: Bioactive Ingredients Market. Available online: https://www.marketdataforecast.com/market-reports/global-bioactive-ingredients-market (accessed on 30 January 2023).
- Market Insights on Biomaterials Covering Sales Outlook, Demand Forecast & Up-to-Date Key Trends. Biomaterials Market by Product, Application & Region—Forecast 2022–2032. Available online: https://www.futuremarketinsights.com/reports/biomaterials-market?utm_source=adwords&utm_medium=ppc&utm_source=adwords&utm_medium=ppc&id=5&gclid=Cj0KCQiA4aacBhCUARIsAI55maG_2teivaB2_99xJTsBzQPfRtMMydO0_qwZS99CDamcAaBp8N8OvGcaAmf0EALw_wcB (accessed on 30 January 2023).
- Kumar Sharma, M.; Kaushik, P. (Eds.) Frontiers in Bioactive Compounds. Therapeutic Implications of Natural Bioactive Compounds; Bentham Science Publishers: Sharjah, United Arab Emirates, 2022. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Tringali, C. (Ed.) Chapter 1—Strategies and Methods for a Sustainable Search for Bioactive Compounds. In Bioactive Compounds from Natural Sources. Natural Products as Lead Compounds in Drug Discovery, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2012; pp. 1–36. [Google Scholar] [CrossRef]
- Tringali, C. (Ed.) Bioactive Compounds from Natural Sources. Isolation, Characterisation and Biological Properties, 1st ed.; Taylor & Francis: London, UK; New York, NY, USA, 2001; ISBN 978-0748408900. [Google Scholar]
- Gallo, M. Bioactive Compounds from Various Sources: Beneficial Effects and Technological Applications. Appl. Sci. 2022, 12, 8955. [Google Scholar] [CrossRef]
- Chang, C.-W.T.; Takemoto, J.Y.; Zhan, J. Natural Bioactive Compounds. Isr. J. Chem. 2019, 59, 325–326. [Google Scholar] [CrossRef]
- Hernandez, D.F.; Cervantes, E.L.; Luna-Vital, D.A. Food-derived bioactive compounds with anti-aging potential for nutricosmetic and cosmeceutical products. Crit. Rev. Food Sci. Nutr. 2021, 61, 3740–3755. [Google Scholar] [CrossRef]
- Bjørklund, G.; Shanaida, M.; Lysiuk, R.; Butnariu, M.; Peana, M.; Sarac, I.; Strus, O.; Smetanina, K.; Chirumbolo, S. Natural Compounds and Products from an Anti-aging Perspective. Molecules 2022, 27, 7084. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef]
- Chakraborty, A.; Sahoo, M.; Roy, S.D.; Dasgupta, R.K. Anti-aging natural herbs: A Systematic Review. Indian Res. J. Pharm. Sci. 2018, 5, 1589–1598. [Google Scholar] [CrossRef]
- Shen, C.-Y.; Jiang, J.-G.; Yang, L.; Wang, D.-W.; Zhu, W. Anti-ageing active ingredients from herbs and nutraceuticals used in traditional Chinese medicine: Pharmacological mechanisms and implications for drug discovery. Br. J. Pharmacol. 2017, 174, 1395–1425. [Google Scholar] [CrossRef]
- Sharafzadeh, S. Medicinal Plants as Anti-Ageing Materials: A Review. Glob. J. Med. Plant Res. 2013, 1, 234–236. Available online: http://www.aensiweb.com/old/GJMPR/2013/234-236.pdf (accessed on 26 February 2023).
- González-Minero, F.J.; Bravo-Díaz, L. The Use of Plants in Skin-Care Products, Cosmetics and Fragrances: Past and Present. Cosmetics 2018, 5, 50. [Google Scholar] [CrossRef]
- Mechchate, H.; El Allam, A.; El Omari, N.; El Hachlafi, N.; Shariati, M.A.; Wilairatana, P.; Mubarak, M.S.; Bouyahya, A. Vegetables and Their Bioactive Compounds as Anti-aging Drugs. Molecules 2022, 27, 2316. [Google Scholar] [CrossRef]
- Sahatpure, N.S.; Daud, F.S. Anti-ageing herbs-A Review. Int. J. Sci. Eng. Dev. Res. 2018, 3, 51–61. Available online: http://www.ijsdr.org/papers/IJSDR1810010.pdf (accessed on 26 February 2023).
- Chondrogianni, N.; Kapeta, S.; Chinou, I.; Vassilatou, K.; Papassideri, I.; Gonos, E.S. Anti-ageing and rejuvenating effects of quercetin. Exp. Gerontol. 2010, 45, 763–771. [Google Scholar] [CrossRef]
- Kravanja, K.A.; Finšgar, M.; Knez, Ž.; Marevci., M.K. Evaluation of Natural Extracts as Promising Components of Bioactive Coatings for Orthopedic Implants. Front. Mater. 2022, 9, 878176. [Google Scholar] [CrossRef]
- Barba-Ostria, C.; Carrera-Pacheco, S.E.; Gonzalez-Pastor, R.; Heredia-Moya, J.; Mayorga-Ramos, A.; Rodríguez-Pólit, C.; Zúñiga-Miranda, J.; Arias-Almeida, B.; Guamán, L.P. Evaluation of Biological Activity of Natural Compounds: Current Trends and Methods. Molecules 2022, 27, 4490. [Google Scholar] [CrossRef]
- Hayflick, L. ”Anti-agingAnti-aging” is an oxymoron. J. Gerontol. A Biol. Sci. Med. Sci. 2004, 59, B573–B578. [Google Scholar] [CrossRef]
- Chmielewski, P.P. Human ageing as a dynamic, emergent and malleable process: From disease-oriented to health-oriented approaches. Biogerontology 2020, 21, 125–130. [Google Scholar] [CrossRef]
- Rae, M.J. All hype, no hope? Excessive pessimism in the “anti-aging medicine” special sections. J. Gerontol. A Biol. Sci. Med. Sci. 2005, 60, 139–140. [Google Scholar] [CrossRef]
- Stambler, I. The Unexpected Outcomes of Anti-agingAnti-aging, Rejuvenation, and Life Extension Studies: An Origin of Modern Therapies. Rejuvenat. Res. 2014, 17, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Stambler, I. Has aging ever been considered healthy? Front. Genet. 2015, 6, 202. [Google Scholar] [CrossRef] [PubMed]
- Stambler, I. Chapter 26—Health and Immortality. In Explaining Health across the Sciences, Healthy Ageing and Longevity, 1st ed.; Sholl, J., Rattan, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2020. [Google Scholar] [CrossRef]
- Zhang, B.; Trapp, A.; Kerepesi, C.; Gladyshev, V.N. Emerging rejuvenation strategies—Reducing the biological age. Aging Cell 2022, 21, e13538. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, S.; Xu, L.; Brunet, A. Turning back time with emerging rejuvenation strategies. Nat. Cell Biol. 2019, 21, 32–43. [Google Scholar] [CrossRef]
- Zhang, W.; Qu, J.; Liu, G.-H.; Belmonte, J.C.I. The ageing epigenome and its rejuvenation. Nat. Rev. Mol. Cell Biol. 2020, 21, 137–150. [Google Scholar] [CrossRef]
- O’Loghlen, A. The potential of aging rejuvenation. Cell Cycle 2022, 21, 111–116. [Google Scholar] [CrossRef]
- Sheldrake, A.R. Cellular senescence, rejuvenation and potential immortality. Rev. Proc. R. Soc. B 2022, 289, 20212434. [Google Scholar] [CrossRef]
- Lizard, G.; Hammami, M.; Poli, G. Pharmacological and Nutraceutical Activation of Rejuvenation, Geroprotection and Cytoprotection: Proofs of Concept. Cells 2022, 11, 3786. [Google Scholar] [CrossRef]
- Aldag, C.; Teixeira, D.N.; Leventhal, P.S. Skin rejuvenation using cosmetic products containing growth factors, cytokines, and matrikines: A review of the literature. Clin. Cosmet. Investig. Dermatol. 2016, 9, 411–419. [Google Scholar] [CrossRef]
- Antonio, C.R.; Antonio, J.R.; Graciano, C.S.; Trídico, L.A. Hormones in Rejuvenation: A Review of Their True Effectiveness. Surg. Cosmet. Dermatol. 2012, 4, 322–330. Available online: http://www.surgicalcosmetic.org.br/details/229 (accessed on 26 February 2023).
- Denoth-Lippuner, A.; Jessberger, S. Mechanisms of cellular rejuvenation. FEBS Lett. 2019, 593, 3381–3392. [Google Scholar] [CrossRef]
- Shin, J.W.; Kwon, S.H.; Choi, J.Y.; Na, J.-I.; Huh, C.-H.; Choi, H.-R.; Park, K.-C. Molecular Mechanisms of Dermal Aging and Anti-aging Approaches. Int. J. Mol. Sci. 2019, 20, 2126. [Google Scholar] [CrossRef]
- Kim, M.; Park, H.J. Chapter 4—Molecular Mechanisms of Skin Aging and Rejuvenation. In Molecular Mechanisms of the Aging Process and Rejuvenation; Miri, N.S., Jeong, H., Eds.; IntechOpen: London, UK, 2016; Available online: https://www.intechopen.com/chapters/50471 (accessed on 26 February 2023).
- Miastkowska, M.; Sikora, E. Anti-aging Properties of Plant Stem Cell Extracts. Cosmetics 2018, 5, 55. [Google Scholar] [CrossRef]
- Pourang, A.; Rockwell, H.; Karimi, K. New Frontiers in Skin Rejuvenation, Including Stem Cells and Autologous Therapies. Facial Plast. Surg. Clin. N. Am. 2020, 28, 101–117. [Google Scholar] [CrossRef]
- Neves, J.; Sousa-Victor, P.; Jasper, H. Rejuvenating Strategies for Stem Cell-Based Therapies in Aging. Cell Stem Cell 2017, 20, 161–175. [Google Scholar] [CrossRef]
- Atwood, C.S.; Bowen, R.L. The reproductive-cell cycle theory of aging: An update. Exp. Gerontol. 2011, 46, 100–107. [Google Scholar] [CrossRef]
- Mahmoudi, S.; Brunet, A. Aging and reprogramming: A two-way street. Curr. Opin. Cell Biol. 2012, 24, 744–756. [Google Scholar] [CrossRef]
- Brunet, A.; Goodell, M.A.; Rando, T.A. Ageing and rejuvenation of tissue stem cells and their niches. Nat. Rev. Mol. Cell Biol. 2023, 24, 45–62. [Google Scholar] [CrossRef]
- Hansen, M.; von Lindern, M.; van den Akker, E.; Varga, E. Human-induced pluripotent stem cell-derived blood products: State of the art and future directions. FEBS Lett. 2019, 593, 3288–3303. [Google Scholar] [CrossRef]
- West, M.D.; Sternberg, H.; Labat, I.; Janus, J.; Chapman, K.B.; Malik, N.N.; De Grey, A.D.; Larocca, D. Toward a unified theory of aging and regeneration. Rev. Regen. Med. 2019, 14, 867–886. [Google Scholar] [CrossRef]
- Alonso-Fernández, P.; De la Fuente, M. Role of the Immune System in Aging and Longevity. Curr. Aging Sci. 2011, 4, 78–100. [Google Scholar] [CrossRef] [PubMed]
- Fahy, G.M.; Brooke, R.T.; Watson, J.P.; Good, Z.; Vasanawala, S.S.; Maecker, H.; Leipold, M.D.; Lin, D.T.S.; Kobor, M.S.; Horvath, S. Reversal of epigenetic aging and immunosenescent trends in humans. Aging Cell 2019, 18, e13028. [Google Scholar] [CrossRef] [PubMed]
- Castelo-Branco, C.; Soveral, I. The immune system and aging: A review. Gynecol. Endocrinol. 2014, 30, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Naval, J.; Coma, M.; Valls, R.; Mas, J.M.; Pujol, A.; Herranz, M.A.; Alonso, V. Methods for diagnosing perceived age on the basis of an ensemble of phenotypic features. Clin. Cosmet. Investig. Dermatol. 2014, 7, 133–137. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jylhävä, J.; Pedersen, N.L.; Hägg, S. Biological Age Predictors. eBioMEdicine 2017, 21, 29–36. [Google Scholar] [CrossRef]
- Gunn, D.A.; Murray, P.G.; Tomlin, C.C.; Rexbye, H.; Christensen, K.; Mayes, A.E. Perceived age as a biomarker of ageing: A clinical methodology. Biogerontology 2008, 9, 357–364. [Google Scholar] [CrossRef]
- Borkan, G.A.; Norris, A.H. Assessment of Biological Age Using a Profile of Physical Parameters. J. Gerontol. 1980, 35, 177–184. [Google Scholar] [CrossRef]
- Jia, L.; Zhang, W.; Chen, X. Common methods of biological age estimation. Clin. Interv. Aging 2017, 12, 759–772. [Google Scholar] [CrossRef]
- Kim, S.; Jazwinski, S.M. Quantitative measures of healthy aging and biological age. Healthy Aging Res. 2015, 4, 26. [Google Scholar] [CrossRef]
- Klemera, P.; Doubal, S. A new approach to the concept and computation of biological age. Mech. Ageing Dev. 2006, 127, 240–248. [Google Scholar] [CrossRef]
- Cho, I.H.; Park, K.S.; Lim, C.J. An empirical comparative study on biological age estimation algorithms with an application of Work Ability Index (WAI). Mech. Ageing 2010, 131, 69–78. [Google Scholar] [CrossRef]
- Bae, C.-Y.; Kang, Y.G.; Kim, S.; Cho, C.; Kang, H.C.; Yu, B.Y.; Lee, S.-W.; Cho, K.H.; Lee, D.C.; Lee, K.; et al. Development of models for predicting biological age (BA) with physical, biochemical, and hormonal parameters. Arch. Gerontol. Geriatr. 2008, 47, 253–265. [Google Scholar] [CrossRef]
- Justice, J.N.; Ferrucci, L.; Newman, A.B.; Aroda, V.R.; Bahnson, J.L.; Divers, J.; Espeland, M.A.; Marcovina, S.; Pollak, M.N.; Kritchevsky, S.B.; et al. A framework for selection of blood-based biomarkers for geroscience-guided clinical trials: Report from the TAME Biomarkers Workgroup. GeroScience 2018, 40, 419–436. [Google Scholar] [CrossRef]
- Sprott, R.L. Biomarkers of aging and disease: Introduction and definitions. Exp. Gerontol. 2010, 45, 2–4. [Google Scholar] [CrossRef]
- Holly, A.C.; Melzer, D.; Pilling, L.; Henley, W.E.; Hernandez, D.G.; Singleton, A.B.; Bandinelli, S.; Guralnik, J.M.; Ferrucci, L.; Harries, L. Towards a gene expression biomarker set for human biological age. Aging Cell 2013, 12, 324–326. [Google Scholar] [CrossRef]
- Wang, A.S.; Dreesen, O. Biomarkers of Cellular Senescence and Skin Aging. Front. Genet. 2018, 9, 247. [Google Scholar] [CrossRef]
- Wagner, K.-H.; Cameron-Smith, D.; Wessner, B.; Franzke, B. Biomarkers of Aging: From Function to Molecular Biology. Nutrients 2016, 8, 338. [Google Scholar] [CrossRef]
- Kaczmarek, M.; Lasik, E. Correlates of Biological Age in Postmenopausal Life. Przegląd Antropol.-Anthropol. Rev. 2006, 69, 15–26. Available online: https://www.academia.edu/1369676/Correlates_of_biological_age_in_postmenopausal_life (accessed on 26 February 2023).
- Sukhovei, Y.; Kostolomova, E.; Unger, I.; Koptyug, A.; Kaigorodov, D. Difference between the biologic and chronologic age as an individualized indicator for the skincare intensity selection: Skin cell profile and age difference studies. Biomed. Dermatol. 2019, 3, 10. [Google Scholar] [CrossRef]
- Sukhovei, Y.; Kostolomova, E.; Unger, I.; Koptyug, A.; Kaigorodov, D. Difference between the biologic and chronologic age as an individualized indicator for the skin care intensity selection: Skin topography and immune system state studies, parameter correlations with age difference. Biomed. Dermatol. 2019, 3, 1e. [Google Scholar] [CrossRef]
- Chen, Y.; Geng, A.; Zhang, W.; Qian, Z.; Wan, X.; Jiang, Y.; Mao, Z. Fight to the bitter end: DNA repair and aging. Ageing Res. Rev. 2020, 64, 101154. [Google Scholar] [CrossRef] [PubMed]
- Švenkrtová, A.; Belicova, L.; Volejníková, A. Stratification of Yeast Cells during Chronological Aging by Size Points to the Role of Trehalose in Cell Vitality. Biogerontology 2016, 17, 395–408. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nie, C.; Li, Y.; Li, R.; Yan, Y.; Zhang, D.; Li, T.; Li, Z.; Sun, Y.; Zhen, H.; Ding, J.; et al. Distinct biological ages of organs and systems identified from a multi-omics study. Cell Rep. 2022, 38, 110459. [Google Scholar] [CrossRef] [PubMed]
- Schaum, N.; Lehallier, B.; Hahn, O.; Pálovics, R.; Hosseinzadeh, S.; Lee, S.E.; Sit, R.; Lee, D.P.; Losada, P.M.; Zardeneta, M.E.; et al. Ageing hallmarks exhibit organ-specific temporal signatures. Nature 2020, 583, 596–602. [Google Scholar] [CrossRef]
- Martinez-Agosto, J.A.; Mikkola, H.K.A.; Hartenstein, V.; Banerjee, U. The hematopoietic stem cell and its niche: A comparative view. Genes Dev. 2007, 21, 3044–3060. [Google Scholar] [CrossRef]
- Watt, F.M.; Hogan, B.L.M. Out of Eden: Stem Cells and Their Niches. Science 2000, 287, 1427–1430. Available online: http://www.jstor.org/stable/3074534 (accessed on 26 February 2023). [CrossRef]
- Scadden, D.T. The stem-cell niche as an entity of action. Nature 2006, 2441, 1075–1079. [Google Scholar] [CrossRef]
- Voog, J.; Jones, D.L. Stem cells and the niche: A dynamic duo. Cell Stem Cell 2010, 6, 103–115. [Google Scholar] [CrossRef]
- Ohlstein, B.; Kai, T.; Decotto, E.; Spradling, A. The stem cell niche: Theme and variations. Curr. Opin. Cell Biol. 2004, 16, 693–699. [Google Scholar] [CrossRef]
- Li, L.; Xie, T. Stem cell niche: Structure and function. Annu. Rev. Cell Dev. Biol. 2005, 21, 605–631. [Google Scholar] [CrossRef]
- Yamashita, Y.M.; Fuller, M.T.; Jones, D.L. Signaling in stem cell niches: Lessons from the Drosophila germline. Rev. J. Cell Sci. 2005, 118 Pt 4, 665–672. [Google Scholar] [CrossRef]
- Wang, C.; Chen, J.; Wen, P.; Sun, P.; Xi, R. Stem Cell Niche. In Regenerative Medicine—From Protocol to Patient; Steinhoff, G., Ed.; Springer: Cham, Switzerland, 2016; pp. 57–85. [Google Scholar] [CrossRef]
- Biteau, B.; Hochmuth, C.E.; Jasper, H. Maintaining tissue homeostasis: Dynamic control of somatic stem cell activity. Cell Stem Cell 2011, 9, 402–411. [Google Scholar] [CrossRef]
- Beerman, I.; Seita, J.; Inlay, M.A.; Weissman, I.L.; Rossi, D.J. Quiescent hematopoietic stem cells accumulate DNA damage during aging that is repaired upon entry into cell cycle. Cell Stem Cell 2014, 15, 37–50. [Google Scholar] [CrossRef]
- Lee, J.Y.; Chen, J.Y.; Shaw, J.L.; Chang, K.T. Maintenance of Stem Cell Niche Integrity by a Novel Activator of Integrin Signaling. PLoS Genet. 2016, 12, e1006043. [Google Scholar] [CrossRef]
- Tominaga, K. The emerging role of senescent cells in tissue homeostasis and pathophysiology. Pathobiol. Aging Age Relat. Dis. 2015, 5, 27743. [Google Scholar] [CrossRef]
- Baar, M.P.; Brandt, R.M.C.; Putavet, D.A.; Klein, J.D.D.; Derks, K.W.J.; Bourgeois, B.R.M.; Stryeck, S.; Rijksen, Y.; Van Willigenburg, H.; Feijtel, D.A.; et al. Targeted Apoptosis of Senescent Cells Restores Tissue Homeostasis in Response to Chemotoxicity and Aging. Cell 2017, 169, 132–147.e16. [Google Scholar] [CrossRef]
- Raffetto, J.D.; Leverkus, M.; Park, H.Y.; Menzoian, J.O. Synopsis on cellular senescence and apoptosis. J. Vasc. Surg. 2001, 34, 173–177. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tieu, K.S.; Tieu, R.S.; Martinez-Agosto, J.A.; Sehl, M.E. Stem Cell Niche Dynamics: From Homeostasis to Carcinogenesis. Stem Cells Int. 2012, 2012, 367567. [Google Scholar] [CrossRef] [PubMed]
- Childs, B.G.; Baker, D.J.; Kirkland, J.L.; Campisi, J.; Van Deursen, J.M. Senescence and apoptosis: Dueling or complementary cell fates? EMBO Rep. 2014, 15, 1139–1153. [Google Scholar] [CrossRef]
- Hampel, B.; Malisan, F.; Niederegger, H.; Testi, R.; Jansen-Dürr, P. Differential regulation of apoptotic cell death in senescent human cells. Exp. Gerontol. 2004, 39, 1713–1721. [Google Scholar] [CrossRef]
- Gil, J.; Withers, D.J. Ageing: Out with the old. Nature 2016, 530, 164–165. [Google Scholar] [CrossRef] [PubMed]
- Tümpel, S.; Rudolph, K.L. Quiescence: Good and Bad of Stem Cell Aging. Trends Cell Biol. 2019, 29, 672–685. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Chang, J.; Lv, D.; Kim, H.-N.; Almeida, M.; Zhang, X.; Mao, X.; Zhao, X.; Li, L.; Campisi, J.; et al. Long-Term Clearance of Senescent Cells Prevents the Hematopoietic Stem Cell Aging in Naturally Aged Mice. Blood 2019, 134 (Suppl. S1), 1204. [Google Scholar] [CrossRef]
- Zhang, L.; Pitcher, L.E.; Yousefzadeh, M.J.; Niedernhofer, L.J.; Robbins, P.D.; Zhu, Y. Cellular senescence: A key therapeutic target in aging and diseases. J. Clin. Investig. 2022, 132, e158450. [Google Scholar] [CrossRef] [PubMed]
- Campisi, J. Aging, cellular senescence, and cancer. Annu. Rev. Physiol. 2013, 75, 685–705. [Google Scholar] [CrossRef]
- Loro, E.; Rinaldi, F.; Malena, A.; Masiero, E.; Novelli, G.; Angelini, C.; Romeo, V.; Sandri, M.; Botta, A.; Vergani, L. Normal myogenesis and increased apoptosis in myotonic dystrophy type-1 muscle cells. Cell Death Differ. 2010, 17, 1315–1324. [Google Scholar] [CrossRef]
- Waddington, C.H. A discussion of some aspects of theoretical biology. In The Strategy of the Genes; George Allen and Unwin Ltd.: London, UK, 1957; pp. 1905–1975. [Google Scholar]
- Waddington, C.H. Towards a theoretical biology. Nature 1968, 218, 525–527. [Google Scholar] [CrossRef]
- De la Fuente, I.M.; Cortés, J.M.; Valero, E.; Desroches, M.; Rodrigues, S.; Malaina, I.; Martínez, L. On the dynamics of the adenylate energy system: Homeorhesis vs. homeostasis. PLoS ONE 2014, 9, e108676. [Google Scholar] [CrossRef]
- Sukhovei, I.G.; Kaigorodov, D.G.; Kostolomova, E.G.; Unger, I.G. Method for Testing Substances Which Affect Aging Processes by Blood Analysis. Patent RU-2728601-C1, 30 July 2020. Available online: https://testpubchem.ncbi.nlm.nih.gov/patent/RU-2728601-C1 (accessed on 26 February 2023).
- Natural Extracts Ärlisätt Essential. Available online: http://arlisatt.life/tproduct/509014482-978708525511-prirodnii-ekstrakt-rlistt-essential-anti (accessed on 26 February 2023).
- Zaveri, L.; Dhawan, J. Cycling to Meet Fate: Connecting Pluripotency to the Cell Cycle. Front. Cell Dev. Biol. 2018, 6, 57. [Google Scholar] [CrossRef]
- She, S.; Wei, Q.; Kang, B.; Wang, Y.J. Cell cycle and pluripotency: Convergence on octamer-binding transcription factor 4. Mol. Med. Rep. 2017, 16, 6459–6466. [Google Scholar] [CrossRef]
- Barta, T.; Dolezalova, D.; Holubcova, Z.; Hampl, A. Cell cycle regulation in human embryonic stem cells: Links to adaptation to cell culture. Exp. Biol. Med. 2013, 238, 271–275. [Google Scholar] [CrossRef]
- Gupta, G.P.; Purvis, J.E. Evidence that the human cell cycle is a series of uncoupled, memoryless phases. Mol. Syst. Biol. 2019, 15, e8604. [Google Scholar] [CrossRef]
- White, J.; Dalton, S. Cell cycle control of embryonic stem cells. Stem Cell Rev. 2005, 1, 131–138. [Google Scholar] [CrossRef]
- Böyum, A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand. J Clin. Lab. Investig. Suppl. 1968, 97, 77–89. [Google Scholar]
- Strober, W. Trypan Blue Exclusion Test of Cell Viability. Curr. Protoc. Immunol. 2015, 111, A3.B.1–A3.B.3. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koptyug, A.; Sukhovei, Y.; Kostolomova, E.; Unger, I.; Kozlov, V. Novel Strategy in Searching for Natural Compounds with Anti-Aging and Rejuvenating Potential. Int. J. Mol. Sci. 2023, 24, 8020. https://doi.org/10.3390/ijms24098020
Koptyug A, Sukhovei Y, Kostolomova E, Unger I, Kozlov V. Novel Strategy in Searching for Natural Compounds with Anti-Aging and Rejuvenating Potential. International Journal of Molecular Sciences. 2023; 24(9):8020. https://doi.org/10.3390/ijms24098020
Chicago/Turabian StyleKoptyug, Andrey, Yurij Sukhovei, Elena Kostolomova, Irina Unger, and Vladimir Kozlov. 2023. "Novel Strategy in Searching for Natural Compounds with Anti-Aging and Rejuvenating Potential" International Journal of Molecular Sciences 24, no. 9: 8020. https://doi.org/10.3390/ijms24098020
APA StyleKoptyug, A., Sukhovei, Y., Kostolomova, E., Unger, I., & Kozlov, V. (2023). Novel Strategy in Searching for Natural Compounds with Anti-Aging and Rejuvenating Potential. International Journal of Molecular Sciences, 24(9), 8020. https://doi.org/10.3390/ijms24098020






