Biotechnologies in Perfume Manufacturing: Metabolic Engineering of Terpenoid Biosynthesis
Abstract
1. Introduction
2. Engineering of the Isoprenoid Pathways
2.1. Genetic Engineering of Plants for the Production of Terpenoids
2.2. Microbial Terpenoid Production
2.2.1. Escherichia coli
| Compound | Host | Approach a | Titer b | Ref. |
|---|---|---|---|---|
| Limonene | E. coli | Introduction of the Mentha spicata LS; heterologous expression of MVA pathway | 3.63 g/L 7.3 g/L c | [40] |
| Sclareol | E. coli | Introduction of the S. sclarea DTSs; heterologous expression of MVA pathway | 1.46 g/L | [42] |
| Santalene | E. coli | Overexpression of E. coli FPPS (IspA); introduction of plant (Clausena lansium) STS; optimization of RBSs; heterologous expression of MVA pathway; removal of competitive indole synthesis by tnaA deletion | 0.60 g/L | [44] |
| E. coli | FPPSs screening to introduce the selected S. cerevisiae mutated FPPS (Erg20F96W); tailored mutagenesis of C. lansium STS to introduce the selected STS variant (STSS533A) and fusion to a solubilization enhancing tag; heterologous expression of MVA pathway | 2.92 g/L | [45] | |
| α-santalol β-santalol | S. cerevisiae | Introduction of S. album CYP (CYP736A167), CPR (CPR2) and STS; manipulation of MVA pathway for the use of galactose-based regulation system | 1.18 g/L | [47] |
| Patchoulol | E. coli | Introduction of the P. cablin PTS; heterologous expression of MVA pathway | 0.040 g/L | [43] |
| Patchoulol | S. cerevisiae | Fusion of FPPS (Erg20) and P. cablin PTS to increase the utilization of the FPP precursor; manipulation of MVA pathway to enhance its flux to FPP by overexpressing HMGR (tHMG1), IDI (IDI1), and UPC2-1, and by repressing competitive steps | 0.47 g/L | [9] |
| Geraniol | S. cerevisiae | Introduction of Ocimum basilicum codon-optimized GS; manipulation of MVA pathway to funnel it to GPP production by overexpressing HMGR (tHMG1), IDI (IDI1), MAF1 and mutated FPPS (Erg20K197G) catalytic domains | 0.036 g/L | [48] |
| Linalool | S. cerevisiae | Introduction of Mentha citrata LIS variant (t67OMcLISE343D/E352H) generated by directed evolution d; overexpression of MVA pathway and mutated FPPS (Erg20F96W/N127W) | 0.053 g/L | [49] |
2.2.2. Saccharomyces cerevisiae
2.3. Comparison of Plant- and Microorganism-Based Systems
3. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sadgrove, N.; Jones, G. A Contemporary Introduction to Essential Oils: Chemistry, Bioactivity and Prospects for Australian Agriculture. Agriculture 2015, 5, 48–102. [Google Scholar] [CrossRef]
- Ríos, J.-L. Essential Oils: What They Are and How the Terms Are Used and Defined. In Essential Oils in Food Preservation, Flavor and Safety; Academic Press: Cambridge, MA, USA, 2016; pp. 3–10. [Google Scholar] [CrossRef]
- Irshad, M.; Ali Subhani, M.; Ali, S.; Hussain, A. Biological Importance of Essential Oils. In Essential Oils—Oils of Nature; El-Shemy, H., Ed.; IntechOpen: London, UK, 2020; pp. 1–7. [Google Scholar]
- Wang, S.; Alseekh, S.; Fernie, A.R.; Luo, J. The Structure and Function of Major Plant Metabolite Modifications. Mol. Plant 2019, 12, 899–919. [Google Scholar] [CrossRef] [PubMed]
- Croteau, R.; Karp, F. Origin of Natural Odorants. In Perfumes; Springer: Dordrecht, The Netherlands, 1994; pp. 101–126. [Google Scholar] [CrossRef]
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of Plant Volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Milc, J.A.; Pecchioni, N.; Chen, L.-Q. Genetic aspects of floral fragrance in plants. Biochem. 2007, 72, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Kim, S.-W. Shaking up ancient scents: Insights into santalol synthesis in engineered Escherichia coli. Process Biochem. 2015, 50, 1177–1183. [Google Scholar] [CrossRef]
- Ma, B.; Liu, M.; Li, Z.-H.; Tao, X.; Wei, D.-Z.; Wang, F.-Q. Significantly Enhanced Production of Patchoulol in Metabolically Engineered Saccharomyces cerevisiae. J. Agric. Food Chem. 2019, 67, 8590–8598. [Google Scholar] [CrossRef]
- Daviet, L.; Schalk, M.; Bicchi, C.; Rubiolo, P. Plenary Lecture Biotechnology in plant essential oil production: Progress and perspective in metabolic engineering of the terpene pathway. Flavour Fragr. J. 2010, 25, 123–127. [Google Scholar] [CrossRef]
- Braga, A.; Guerreiro, C.; Belo, I. Generation of Flavors and Fragrances Through Biotransformation and De Novo Synthesis. Food Bioprocess Technol. 2018, 11, 2217–2228. [Google Scholar] [CrossRef]
- Welsh, F.W.; Murray, W.D.; Williams, R.E.; Katz, I. Microbiological and Enzymatic Production of Flavor and Fragrance Chemicals. Crit. Rev. Biotechnol. 1989, 9, 105–169. [Google Scholar] [CrossRef]
- Lecourt, M.; Antoniotti, S. Chemistry, Sustainability and Naturality of Perfumery Biotech Ingredients. Chemsuschem 2020, 13, 5599. [Google Scholar] [CrossRef] [PubMed]
- Vilela, A.; Bacelar, E.; Pinto, T.; Anjos, R.; Correia, E.; Gonçalves, B.; Cosme, F. Beverage and Food Fragrance Biotechnology, Novel Applications, Sensory and Sensor Techniques: An Overview. Foods 2019, 8, 643. [Google Scholar] [CrossRef] [PubMed]
- Chatzivasileiou, A.O.; Ward, V.; Edgar, S.M.B.; Stephanopoulos, G. Two-step pathway for isoprenoid synthesis. Proc. Natl. Acad. Sci. USA 2019, 116, 506–511. [Google Scholar] [CrossRef]
- Mahmoud, S.S.; Croteau, R.B. Strategies for transgenic manipulation of monoterpene biosynthesis in plants. Trends Plant Sci. 2002, 7, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Vainstein, A.; Lewinsohn, E.; Pichersky, E.; Weiss, D. Floral Fragrance. New Inroads into an Old Commodity. Plant Physiol. 2001, 127, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Leresche, J.E.; Meyer, H.-P. Chemocatalysis and Biocatalysis (Biotransformation): Some Thoughts of a Chemist and of a Biotechnologist. Org. Process Res. Dev. 2006, 10, 572–580. [Google Scholar] [CrossRef]
- de Carvalho, C.C.C.R.; da Fonseca, M.M.R. Biotransformation of terpenes. Biotechnol. Adv. 2006, 24, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Gupta, C. A Biotechnological Approach to Microbial Based Perfumes and Flavours. J. Microbiol. Exp. 2015, 2, 11–18. [Google Scholar] [CrossRef]
- Wu, S.; Schalk, M.; Clark, A.; Miles, R.B.; Coates, R.; Chappell, J. Redirection of cytosolic or plastidic isoprenoid precursors elevates terpene production in plants. Nat. Biotechnol. 2006, 24, 1441–1447. [Google Scholar] [CrossRef] [PubMed]
- Ohara, K.; Matsunaga, E.; Nanto, K.; Yamamoto, K.; Sasaki, K.; Ebinuma, H.; Yazaki, K. Monoterpene engineering in a woody plant Eucalyptus camaldulensis using a limonene synthase cDNA. Plant Biotechnol. J. 2010, 8, 28–37. [Google Scholar] [CrossRef]
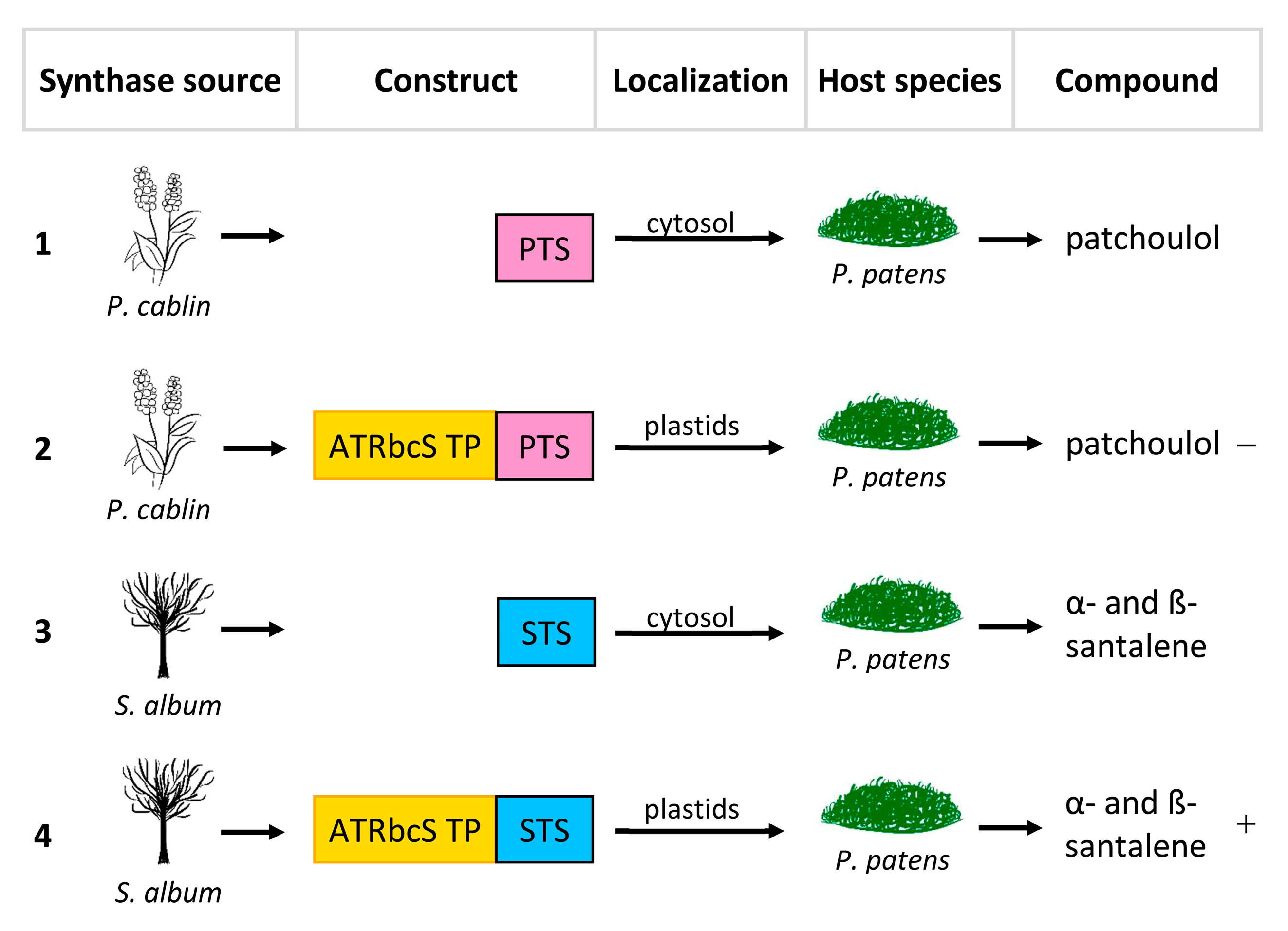
- Zhan, X.; Zhang, Y.-H.; Chen, D.-F.; Simonsen, H.T. Metabolic engineering of the moss Physcomitrella patens to produce the sesquiterpenoids patchoulol and α/β-santalene. Front. Plant Sci. 2014, 5, 636. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, L.; Tang, K.; Xu, M.; Miao, Z. Matching is the Key Factor to Improve the Production of Patchoulol in the Plant Chassis of Marchantia paleacea. ACS Omega 2020, 5, 33028–33038. [Google Scholar] [CrossRef]
- van Beek, T.A.; Joulain, D. The essential oil of patchouli, Pogostemon cablin: A review. Flavour Fragr. J. 2018, 33, 6–51. [Google Scholar] [CrossRef]
- Shukla, V.; Phulara, S.C. Impact of Culture Condition Modulation on the High-Yield, High-Specificity, and Cost-Effective Production of Terpenoids from Microbial Sources: A Review. Appl. Environ. Microbiol. 2021, 87, e02369-20. [Google Scholar] [CrossRef]
- Lee, J.-W.; Trinh, C.T. Towards renewable flavors, fragrances, and beyond. Curr. Opin. Biotechnol. 2020, 61, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Schempp, F.M.; Drummond, L.; Buchhaupt, M.; Schrader, J. Microbial Cell Factories for the Production of Terpenoid Flavor and Fragrance Compounds. J. Agric. Food Chem. 2017, 66, 2247–2258. [Google Scholar] [CrossRef]
- Ncube, E.N.; Steenkamp, P.A.; van der Westhuyzen, C.W.; Steenkamp, L.H.; Dubery, I.A. Metabolomics-Guided Analysis of the Biocatalytic Conversion of Sclareol to Ambradiol by Hyphozyma roseoniger. Catalysts 2022, 12, 55. [Google Scholar] [CrossRef]
- Ochsner, A.M.; Sonntag, F.; Buchhaupt, M.; Schrader, J.; Vorholt, J.A. Methylobacterium extorquens: Methylotrophy and biotechnological applications. Appl. Microbiol. Biotechnol. 2015, 99, 517–534. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Wei, L.-J.; Lin, J.-Y.; Hua, Q. Enhancing linalool production by engineering oleaginous yeast Yarrowia lipolytica. Bioresour. Technol. 2017, 245, 1641–1644. [Google Scholar] [CrossRef]
- Jia, D.; Xu, S.; Sun, J.; Zhang, C.; Li, D.; Lu, W. Yarrowia lipolytica construction for heterologous synthesis of α-santalene and fermentation optimization. Appl. Microbiol. Biotechnol. 2019, 103, 3511–3520. [Google Scholar] [CrossRef] [PubMed]
- Davies, F.K.; Jinkerson, R.E.; Posewitz, M.C. Toward a photosynthetic microbial platform for terpenoid engineering. Photosynth. Res. 2015, 123, 265–284. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Gonzalez, L.; Mozumder, S.I.; Dubreuil, M.; Volcke, E.I.; De Wever, H. Sustainable autotrophic production of polyhydroxybutyrate (PHB) from CO2 using a two-stage cultivation system. Catal. Today 2015, 257, 237–245. [Google Scholar] [CrossRef]
- Troost, K.; Loeschcke, A.; Hilgers, F.; Özgür, A.Y.; Weber, T.M.; Santiago-Schübel, B.; Svensson, V.; Hage-Hülsmann, J.; Habash, S.S.; Grundler, F.M.W.; et al. Engineered Rhodobacter capsulatus as a Phototrophic Platform Organism for the Synthesis of Plant Sesquiterpenoids. Front. Microbiol. 2019, 10, 1998. [Google Scholar] [CrossRef]
- Guan, Z.; Xue, D.; Abdallah, I.I.; Dijkshoorn, L.; Setroikromo, R.; Lv, G.; Quax, W.J. Metabolic engineering of Bacillus subtilis for terpenoid production. Appl. Microbiol. Biotechnol. 2015, 99, 9395–9406. [Google Scholar] [CrossRef] [PubMed]
- Fraatz, M.A.; Berger, R.G.; Zorn, H. Nootkatone—A biotechnological challenge. Appl. Microbiol. Biotechnol. 2009, 83, 35–41. [Google Scholar] [CrossRef]
- Wriessnegger, T.; Augustin, P.; Engleder, M.; Leitner, E.; Müller, M.; Kaluzna, I.; Schürmann, M.; Mink, D.; Zellnig, G.; Schwab, H.; et al. Production of the sesquiterpenoid (+)-nootkatone by metabolic engineering of Pichia pastoris. Metab. Eng. 2014, 24, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liwei, M.; Park, J.-B.; Jeong, S.-H.; Wei, G.; Wang, Y.; Kim, S.-W. Microbial platform for terpenoid production: Escherichia coli and Yeast. Front. Microbiol. 2018, 9, 2460. [Google Scholar] [CrossRef] [PubMed]
- Rolf, J.; Julsing, M.K.; Rosenthal, K.; Lütz, S. A Gram-Scale Limonene Production Process with Engineered Escherichia coli. Molecules 2020, 25, 1881. [Google Scholar] [CrossRef]
- Ncube, E.N.; Steenkamp, L.; Dubery, I.A. Ambrafuran (AmbroxTM) Synthesis from Natural Plant Product Precursors. Molecules 2020, 25, 3851. [Google Scholar] [CrossRef]
- Schalk, M.; Pastore, L.; Mirata, M.A.; Khim, S.; Schouwey, M.; Deguerry, F.; Pineda, V.; Rocci, L.; Daviet, L. Toward a Biosynthetic Route to Sclareol and Amber Odorants. J. Am. Chem. Soc. 2012, 134, 18900–18903. [Google Scholar] [CrossRef]
- Aguilar, F.; Ekramzadeh, K.; Scheper, T.; Beutel, S. Whole-Cell Production of Patchouli Oil Sesquiterpenes in Escherichia coli: Metabolic Engineering and Fermentation Optimization in Solid–Liquid Phase Partitioning Cultivation. ACS Omega 2020, 5, 32436–32446. [Google Scholar] [CrossRef]
- Wang, Y.; Zhou, S.; Liu, Q.; Jeong, S.-H.; Zhu, L.; Yu, X.; Zheng, X.; Wei, G.; Kim, S.-W.; Wang, C. Metabolic Engineering of Escherichia coli for Production of α-Santalene, a Precursor of Sandalwood Oil. J. Agric. Food Chem. 2021, 69, 13135–13142. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, X.; Zhang, X.; Zhang, Y.; Wang, F.; Li, X. Sesquiterpene Synthase Engineering and Targeted Engineering of α-Santalene Overproduction in Escherichia coli. J. Agric. Food Chem. 2022, 70, 5377–5385. [Google Scholar] [CrossRef]
- Hausjell, J.; Halbwirth, H.; Spadiut, O. Recombinant production of eukaryotic cytochrome P450s in microbial cell factories. Biosci. Rep. 2018, 38, 20171290. [Google Scholar] [CrossRef]
- Zha, W.; An, T.; Li, T.; Zhu, J.; Gao, K.; Sun, Z.; Xu, W.; Lin, P.; Zi, J. Reconstruction of the Biosynthetic Pathway of Santalols under Control of the GAL Regulatory System in Yeast. ACS Synth. Biol. 2020, 9, 449–456. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, W.; Du, G.; Chen, J.; Zhou, J. Overproduction of geraniol by enhanced precursor supply in Saccharomyces cerevisiae. J. Biotechnol. 2013, 168, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Du, Y.; Xu, N.; Yue, C.; Ye, L. Improved linalool production in Saccharomyces cerevisiae by combining directed evolution of linalool synthase and overexpression of the complete mevalonate pathway. Biochem. Eng. J. 2020, 161, 107655. [Google Scholar] [CrossRef]
- Sun, W.; Zhao, Y.-J.; Li, C. De Novo Synthesis of Plant Natural Products in Yeast. In Yeasts in Biotechnology; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Zhou, P.; Du, Y.; Fang, X.; Xu, N.; Yue, C.; Ye, L. Combinatorial Modulation of Linalool Synthase and Farnesyl Diphosphate Synthase for Linalool Overproduction in Saccharomyces cerevisiae. J. Agric. Food Chem. 2021, 69, 1003–1010. [Google Scholar] [CrossRef]
- Basson, M.E.; Thorsness, M.; Rine, J. Saccharomyces cerevisiae contains two functional genes encoding 3-hydroxy-3-methylglutaryl-coenzyme A reductase. Proc. Natl. Acad. Sci. USA 1986, 83, 5563–5567. [Google Scholar] [CrossRef] [PubMed]
- Eichhorn, E.; Locher, E.; Guillemer, S.; Wahler, D.; Fourage, L.; Schilling, B. Biocatalytic Process for (−)-Ambrox Production Using Squalene Hopene Cyclase. Adv. Synth. Catal. 2018, 360, 2339–2351. [Google Scholar] [CrossRef]
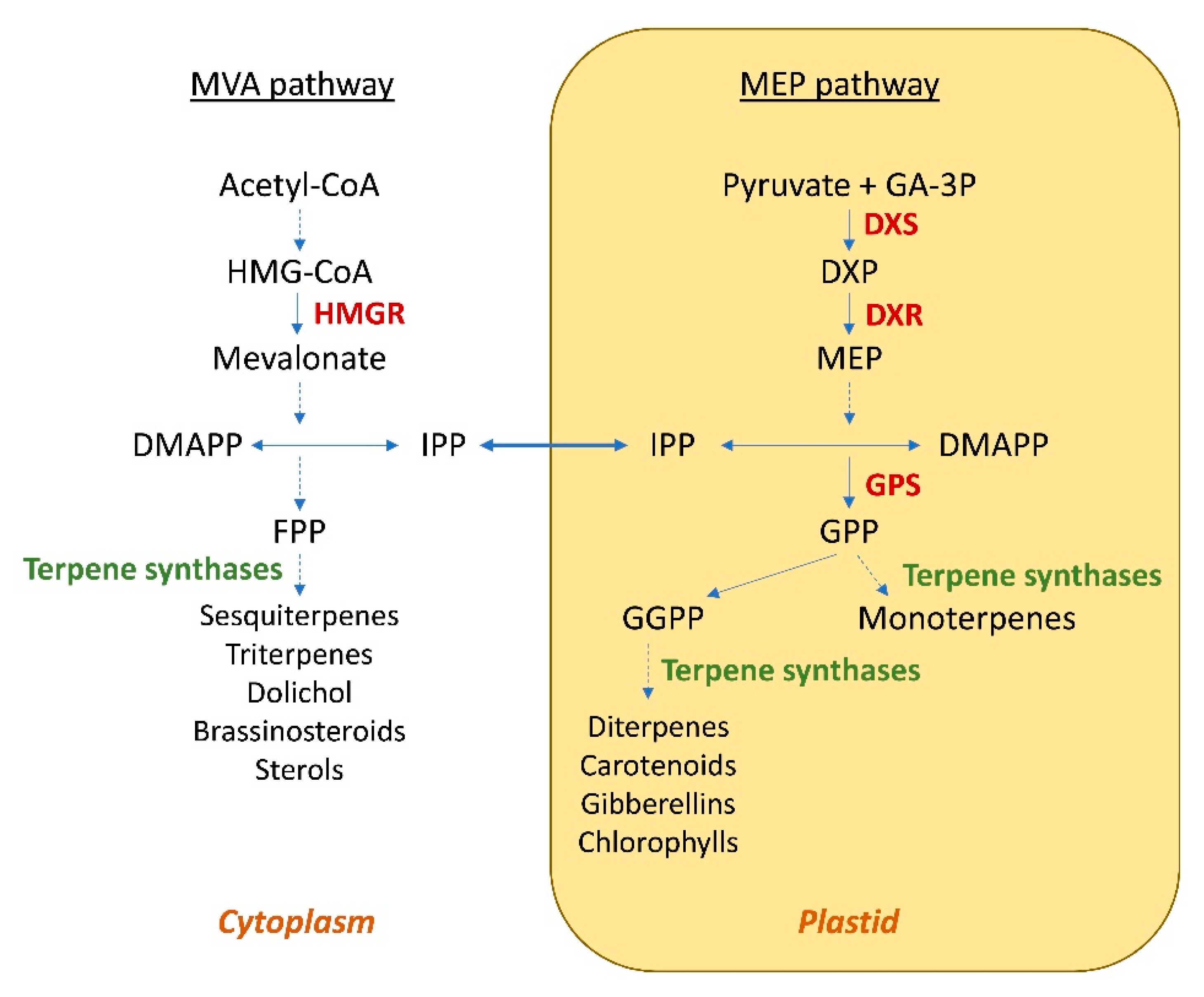

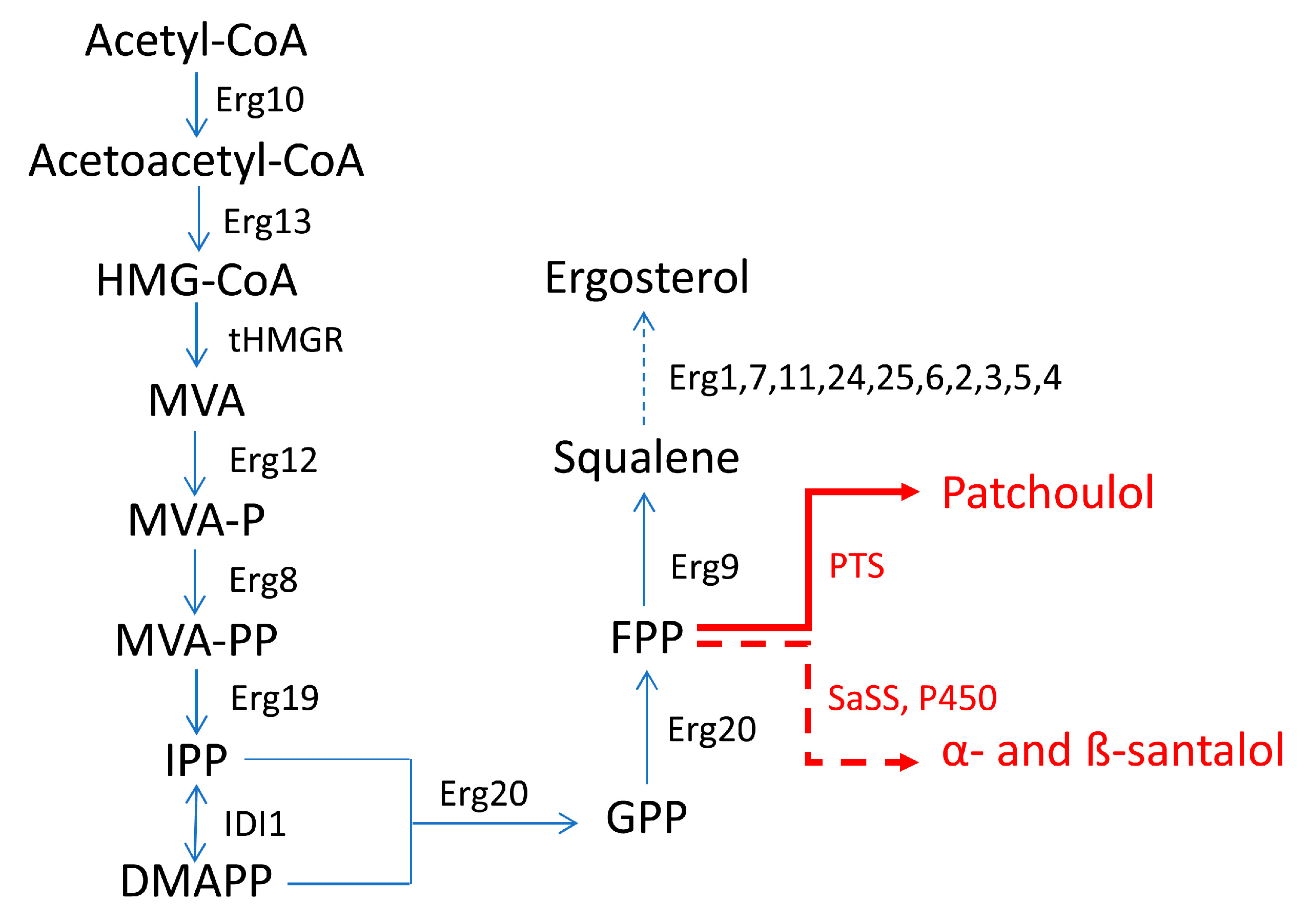
| Compound | Host | Approach | Titer a | Ref. |
|---|---|---|---|---|
| Limonene | Eucalyptus camaldulensis | Native wild-type (not engineered) | 73 µg ⁄g FW b | [22] |
| Introduction of Perilla frutescens LS | 190 µg ⁄g FW b | [22] | ||
| Introduction of P. frutescens tp-deprived LS | 327 µg/g FW b | [22] | ||
| Patchoulol | Marchantia paleacea | Introduction of codon-optimized Gallus gallus FPPS and P. cablin PTS, both equipped with tp and driven by the 35S promoter | 3250 µg/g DW c | [24] |
| Physcomitrella patens | Introduction of P. cablin PTS and S. cerevisiae truncated HMGR | 1340 µg/g DW | [23] | |
| Introduction of P. cablin PTS | 830 µg/g DW | [23] | ||
| Introduction of P. cablin PTS equipped with tp | 20 µg/g DW | [23] | ||
| α-santalene β-santalene | Physcomitrella patens | Introduction of S. album STS | n.d. | [23] |
| Introduction of S. album STS and S. cerevisiae truncated HMGR | α: 22 µg/g DW β: 20 µg/g DW | [23] | ||
| Introduction of S. album STS equipped with tp | α: 39 µg/g DW β: 35 µg/g DW | [23] |
| Compound | Host | Titer a |
|---|---|---|
| Limonene | E. camaldulensis | (327 mg/kg FW) 0.33 mg/g FW [22] |
| E. coli | 3630 mg/L (726 mg/g FW) [40] | |
| Patchoulol | M. paleacea | (325 mg/kg FW) 3.25 mg/g DW [24] |
| S. cerevisiae | 470 mg/L (313 mg/g DW) [9] | |
| α-santalene, santalols | P. patens | (3.9 mg/kg FW) b 0.039 mg/g DW b [23] |
| S. cerevisiae | 1180 mg/L (787 mg/g DW) [47] | |
| E. coli | 2920 mg/L b (5840 mg/g DW) b [45] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manina, A.S.; Forlani, F. Biotechnologies in Perfume Manufacturing: Metabolic Engineering of Terpenoid Biosynthesis. Int. J. Mol. Sci. 2023, 24, 7874. https://doi.org/10.3390/ijms24097874
Manina AS, Forlani F. Biotechnologies in Perfume Manufacturing: Metabolic Engineering of Terpenoid Biosynthesis. International Journal of Molecular Sciences. 2023; 24(9):7874. https://doi.org/10.3390/ijms24097874
Chicago/Turabian StyleManina, Alessia Shelby, and Fabio Forlani. 2023. "Biotechnologies in Perfume Manufacturing: Metabolic Engineering of Terpenoid Biosynthesis" International Journal of Molecular Sciences 24, no. 9: 7874. https://doi.org/10.3390/ijms24097874
APA StyleManina, A. S., & Forlani, F. (2023). Biotechnologies in Perfume Manufacturing: Metabolic Engineering of Terpenoid Biosynthesis. International Journal of Molecular Sciences, 24(9), 7874. https://doi.org/10.3390/ijms24097874









