The Anti-Tumorigenic Role of Cannabinoid Receptor 2 in Non-Melanoma Skin Cancer
Abstract
1. Introduction
2. Results
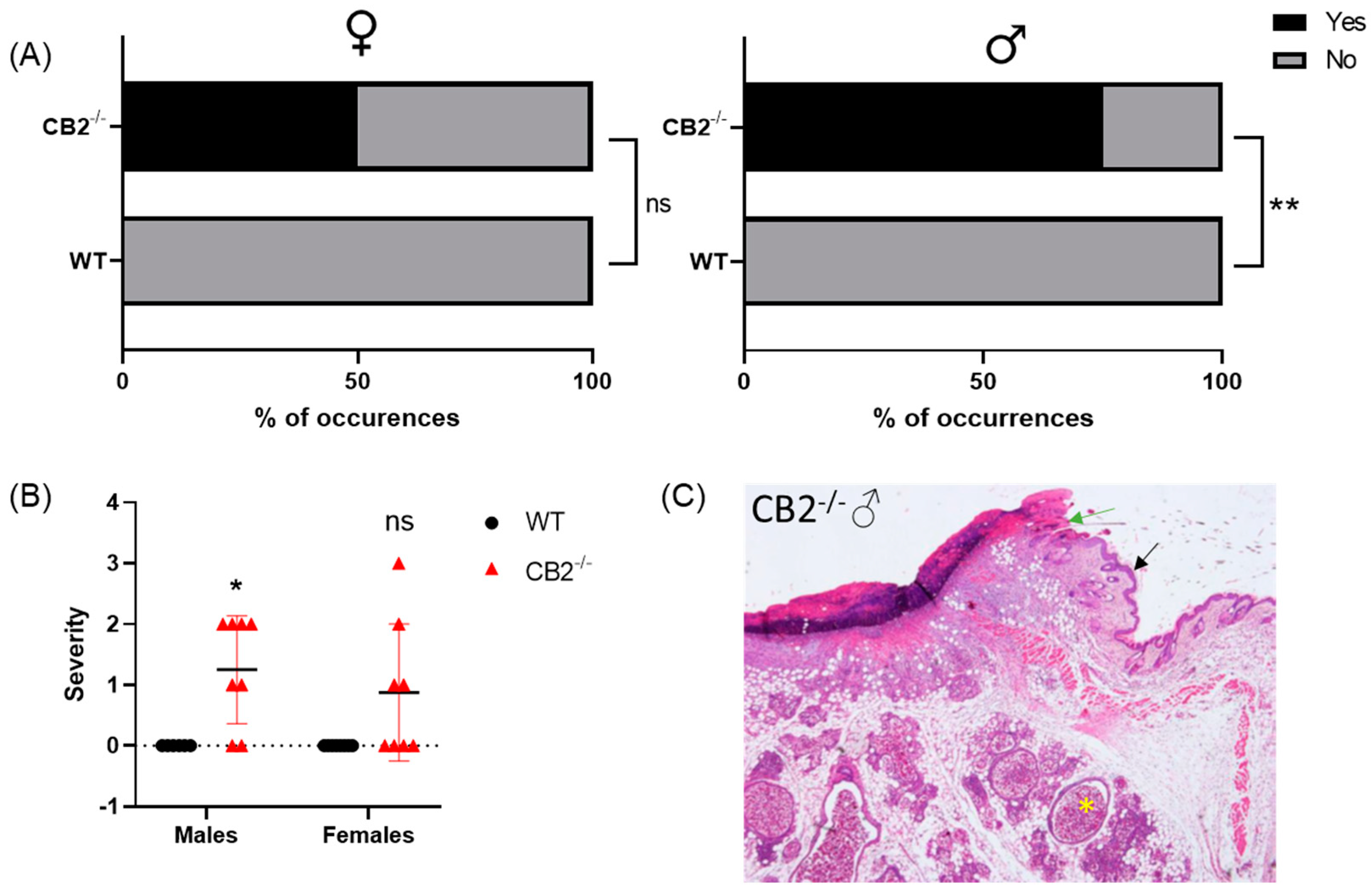
2.1. CB2-/- Is Associated with an Increased Risk for Spontaneous Skin Cancer in Males
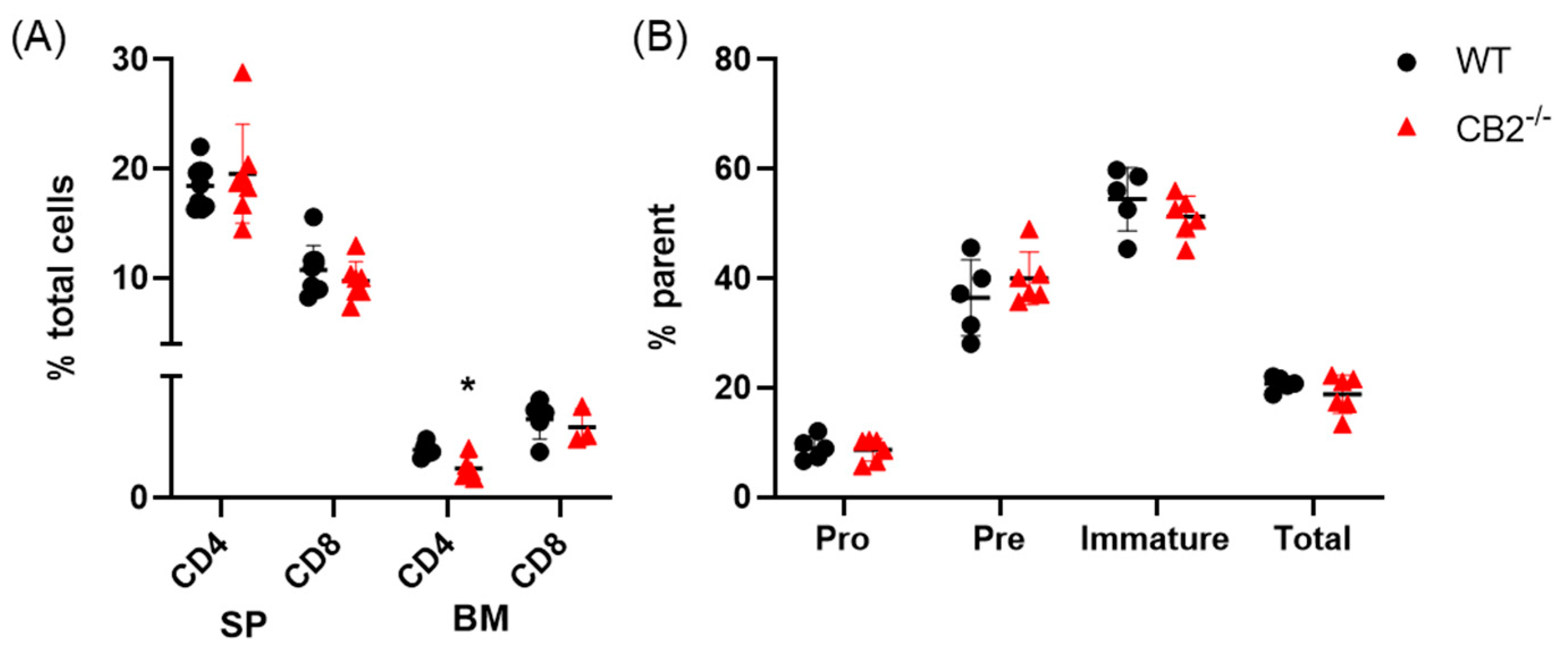
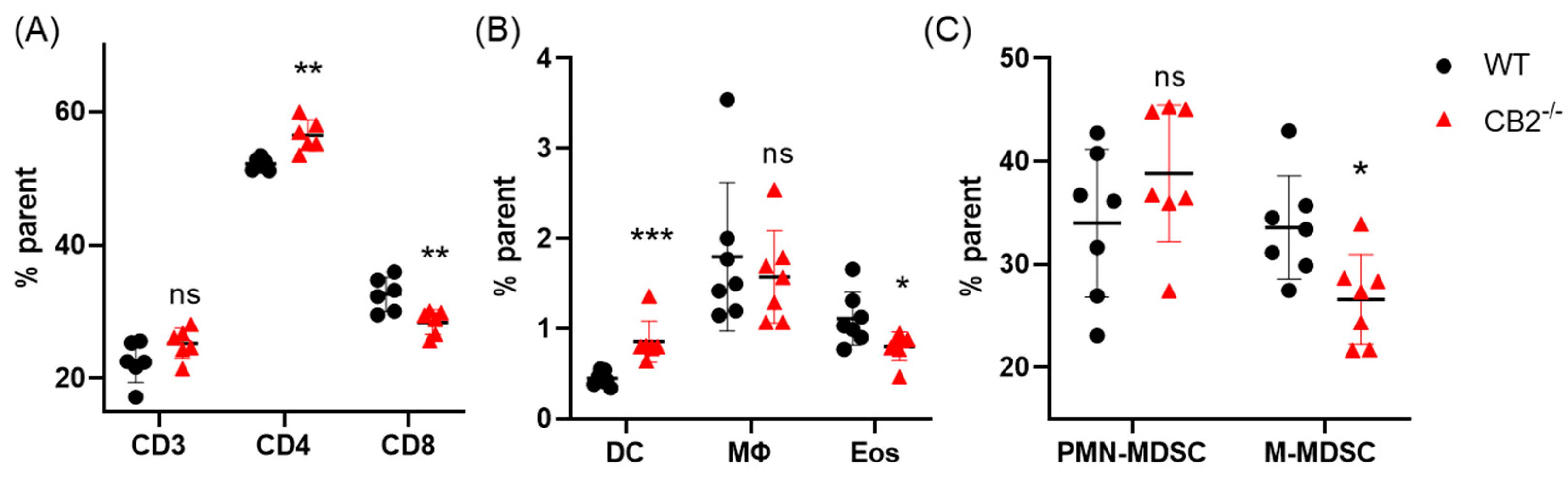
2.2. CB2-/- and WT Mice Have Similar Lymphoid Compartments in the Spleen and Bone Marrow at Baseline
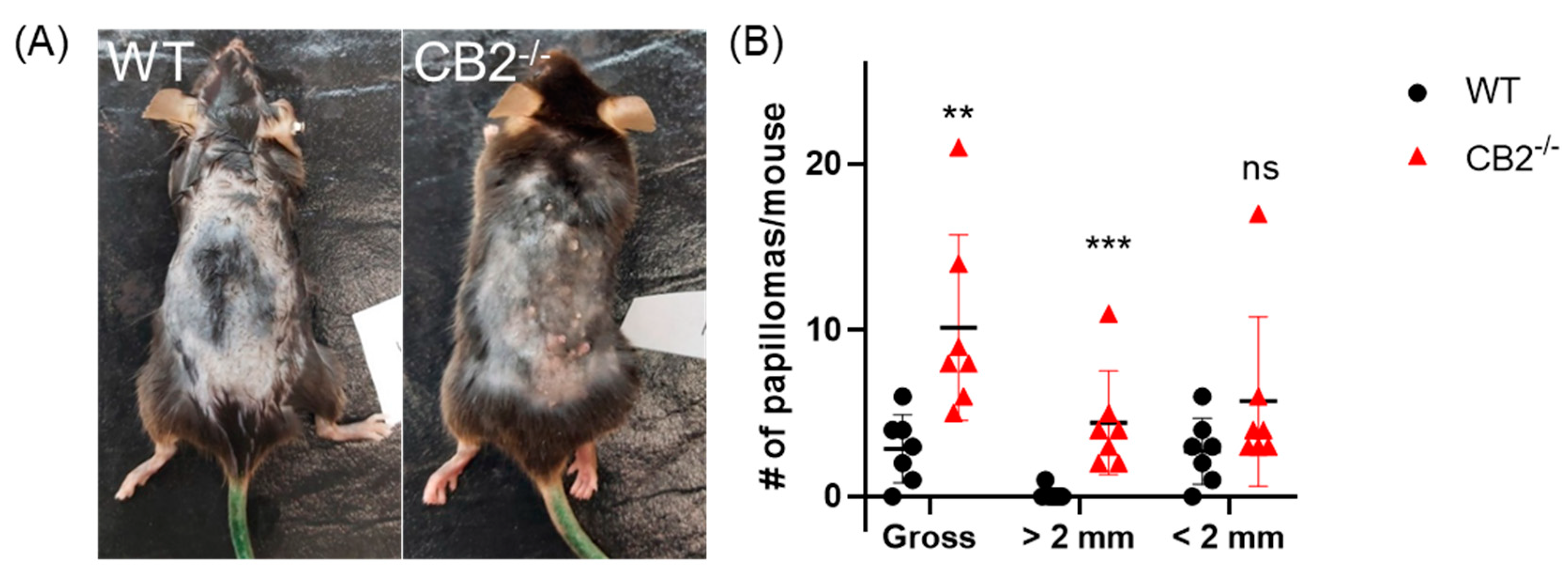
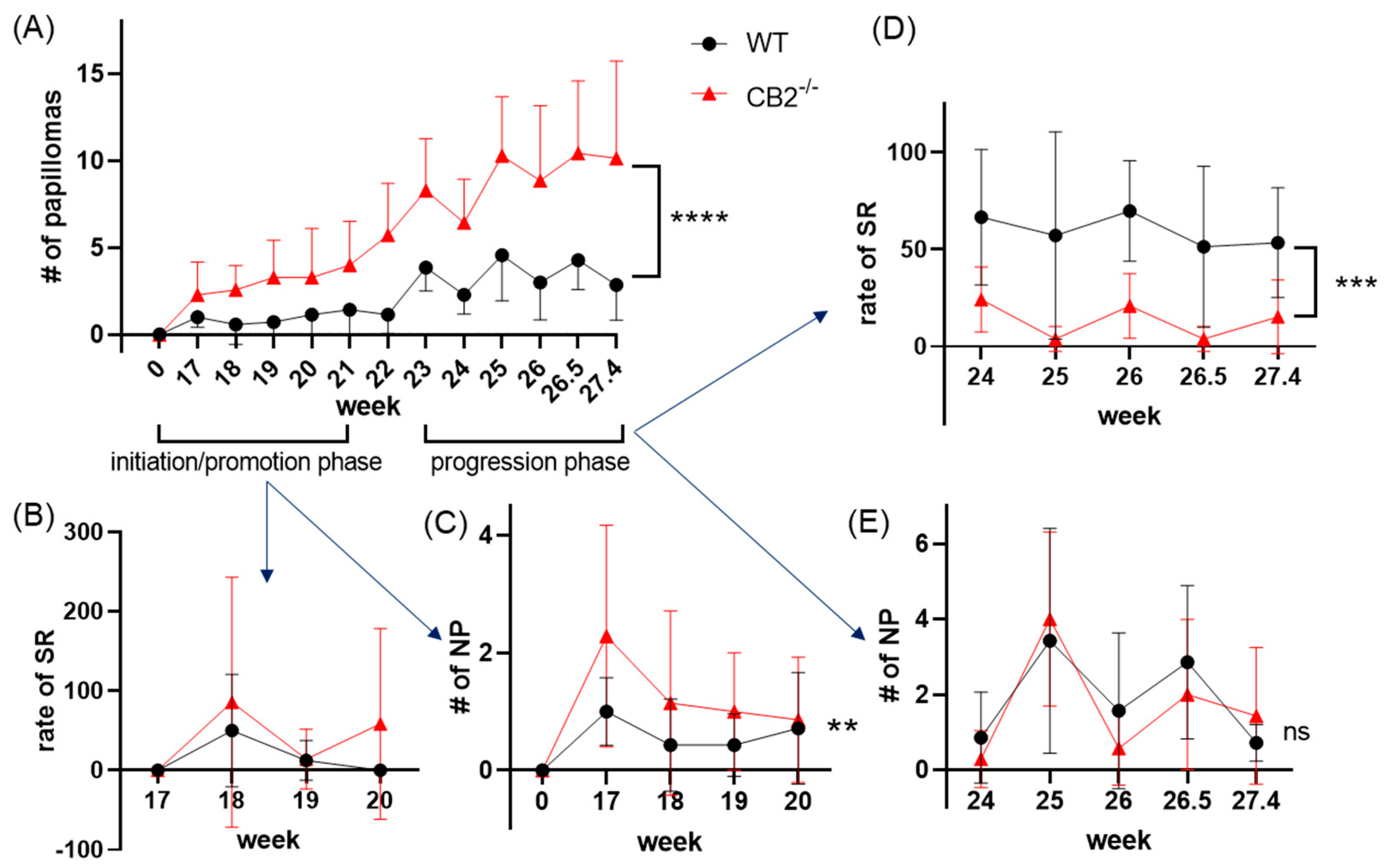
2.3. CB2-/- Mice Have Heightened Tumor Susceptibility in the DMBA/TPA Model
2.4. CB2-/- Mice Have Depleted Spontaneous Regression of Papillomas in the DMBA/TPA Model
2.5. CB2-/- and WT Mice Treated with DMBA/TPA Have Similar Tumor Microenvironments
2.6. CB2 Expression Is Upregulated in Carcinogen-Exposed Skin of WT Mice
2.7. CB2-/- Mice Treated with DMBA/TPA Have an Altered Systemic Immune Profile
3. Discussion
4. Materials and Methods
4.1. Skin Cancer Induction
4.2. Flow Cytometry
4.3. Histology
4.4. RNA Extraction, cDNA Synthesis, and Quantitative Real-Time Polymerase Chain Reaction (qPCR)
4.5. Genotyping
4.6. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Griffin, L.L.; Ali, F.R.; Lear, J.T. Non-melanoma skin cancer. Clin. Med. 2016, 16, 62. [Google Scholar] [CrossRef]
- Linares, M.A.; Zakaria, A.; Nizran, P. Skin cancer. Prim. Care Clin. Off. Pract. 2015, 42, 645–659. [Google Scholar] [CrossRef] [PubMed]
- Diepgen, T.L.; Mahler, V. The epidemiology of skin cancer. Br. J. Dermatol. 2002, 146, 1–6. [Google Scholar] [CrossRef]
- Lomas, A.; Leonardi-Bee, J.; Bath-Hextall, F. A systematic review of worldwide incidence of nonmelanoma skin cancer. Br. J. Dermatol. 2012, 166, 1069–1080. [Google Scholar] [CrossRef]
- Weinberg, A.S.; Ogle, C.A.; Shim, E.K. Metastatic cutaneous squamous cell carcinoma: An update. Dermatol. Surg. 2007, 33, 885–899. [Google Scholar] [CrossRef] [PubMed]
- Abel, E.L.; Angel, J.M.; Kiguchi, K.; DiGiovanni, J. Multi-stage chemical carcinogenesis in mouse skin: Fundamentals and applications. Nat. Protoc. 2009, 4, 1350–1362. [Google Scholar] [CrossRef]
- Kong, Y.-H.; Xu, S.-P. Salidroside prevents skin carcinogenesis induced by DMBA/TPA in a mouse model through suppression of inflammation and promotion of apoptosis. Oncol. Rep. 2018, 39, 2513–2526. [Google Scholar] [CrossRef]
- Monga, J.; Aggarwal, V.; Suthar, S.K.; Nongalleima, K.; Sharma, M. Topical (+)-catechin emulsified gel prevents DMBA/TPA-induced squamous cell carcinoma of the skin by modulating antioxidants and inflammatory biomarkers in BALB/c mice. Food Funct. 2014, 5, 3197–3207. [Google Scholar] [CrossRef]
- Yang, A.Y.; Lee, J.H.; Shu, L.; Zhang, C.; Su, Z.-Y.; Lu, Y.; Huang, M.-T.; Ramirez, C.; Pung, D.; Huang, Y. Genome-wide analysis of DNA methylation in UVB-and DMBA/TPA-induced mouse skin cancer models. Life Sci. 2014, 113, 45–54. [Google Scholar] [CrossRef]
- Zambrano-Román, M.; Padilla-Gutiérrez, J.R.; Valle, Y.; Muñoz-Valle, J.F.; Valdés-Alvarado, E. Non-Melanoma Skin Cancer: A Genetic Update and Future Perspectives. Cancers 2022, 14, 2371. [Google Scholar] [CrossRef]
- Ito, T.; Wang, Y.-H.; Duramad, O.; Hori, T.; Delespesse, G.J.; Watanabe, N.; Qin, F.X.-F.; Yao, Z.; Cao, W.; Liu, Y.-J. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J. Exp. Med. 2005, 202, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Soumelis, V.; Reche, P.A.; Kanzler, H.; Yuan, W.; Edward, G.; Homey, B.; Gilliet, M.; Ho, S.; Antonenko, S.; Lauerma, A. Human epithelial cells trigger dendritic cell–mediated allergic inflammation by producing TSLP. Nat. Immunol. 2002, 3, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Demehri, S.; Turkoz, A.; Manivasagam, S.; Yockey, L.J.; Turkoz, M.; Kopan, R. Elevated epidermal thymic stromal lymphopoietin levels establish an antitumor environment in the skin. Cancer Cell 2012, 22, 494–505. [Google Scholar] [CrossRef]
- Cipolat, S.; Hoste, E.; Natsuga, K.; Quist, S.R.; Watt, F.M. Epidermal barrier defects link atopic dermatitis with altered skin cancer susceptibility. Elife 2014, 3, e01888. [Google Scholar] [CrossRef]
- Dontje, W.; Schotte, R.; Cupedo, T.; Nagasawa, M.; Scheeren, F.; Gimeno, R.; Spits, H.; Blom, B. Delta-like1-induced Notch1 signaling regulates the human plasmacytoid dendritic cell versus T-cell lineage decision through control of GATA-3 and Spi-B. Blood 2006, 107, 2446–2452. [Google Scholar] [CrossRef]
- Ouyang, W.; Ranganath, S.H.; Weindel, K.; Bhattacharya, D.; Murphy, T.L.; William, C.S.; Murphy, K.M. Inhibition of Th1 development mediated by GATA-3 through an IL-4-independent mechanism. Immunity 1998, 9, 745–755. [Google Scholar] [CrossRef]
- Solus, J.F.; Hassan, K.; Lee, S.J.; Hsi, A.C.; Rosman, I.S.; Dehmeri, S.; Schaffer, A. Cutaneous squamous cell carcinoma progression is associated with decreased GATA-3 immunohistochemical staining. J. Cutan. Pathol. 2016, 43, 347–353. [Google Scholar] [CrossRef]
- He, D.; Li, H.; Yusuf, N.; Elmets, C.A.; Athar, M.; Katiyar, S.K.; Xu, H. IL-17 mediated inflammation promotes tumor growth and progression in the skin. PLoS ONE 2012, 7, e32126. [Google Scholar] [CrossRef]
- Fujimura, T.; Aiba, S. Significance of immunosuppressive cells as a target for immunotherapies in melanoma and non-melanoma skin cancers. Biomolecules 2020, 10, 1087. [Google Scholar] [CrossRef]
- Endrizzi, B.; Ahmed, R.L.; Ray, T.; Dudek, A.; Lee, P. Capecitabine to reduce nonmelanoma skin carcinoma burden in solid organ transplant recipients. Dermatol. Surg. 2013, 39, 634–645. [Google Scholar] [CrossRef]
- Chockalingam, R.; Downing, C.; Tyring, S.K. Cutaneous squamous cell carcinomas in organ transplant recipients. J. Clin. Med. 2015, 4, 1229–1239. [Google Scholar] [CrossRef] [PubMed]
- Alpini, G.; DeMorrow, S. Changes in the endocannabinoid system may give insight into new and effective treatments for cancer. Vitam. Horm. 2009, 81, 469–485. [Google Scholar] [PubMed]
- Corroon, J.; Felice, J.F. The Endocannabinoid System and its Modulation by Cannabidiol (CBD). Altern. Ther. Health Med. 2019, 25, 6–14. [Google Scholar] [PubMed]
- Huang, S.J.; Hijnen, D.; Murphy, G.F.; Kupper, T.S.; Calarese, A.W.; Mollet, I.G.; Schanbacher, C.F.; Miller, D.M.; Schmults, C.D.; Clark, R.A. Imiquimod enhances IFN-γ production and effector function of T cells infiltrating human squamous cell carcinomas of the skin. J. Investig. Dermatol. 2009, 129, 2676–2685. [Google Scholar] [CrossRef] [PubMed]
- Becker, W.; Alrafas, H.R.; Wilson, K.; Miranda, K.; Culpepper, C.; Chatzistamou, I.; Cai, G.; Nagarkatti, M.; Nagarkatti, P.S. Activation of cannabinoid receptor 2 prevents colitis-associated colon cancer through myeloid cell de-activation upstream of IL-22 production. Iscience 2020, 23, 101504. [Google Scholar] [CrossRef]
- Louvet, A.; Teixeira-Clerc, F.; Chobert, M.N.; Deveaux, V.; Pavoine, C.; Zimmer, A.; Pecker, F.; Mallat, A.; Lotersztajn, S. Cannabinoid CB2 receptors protect against alcoholic liver disease by regulating Kupffer cell polarization in mice. Hepatology 2011, 54, 1217–1226. [Google Scholar] [CrossRef]
- Patel, K.D.; Davison, J.S.; Pittman, Q.J.; Sharkey, K.A. Cannabinoid CB2 receptors in health and disease. Curr. Med. Chem. 2010, 17, 1394–1410. [Google Scholar] [CrossRef]
- Schwarz, R.; Ramer, R.; Hinz, B. Targeting the endocannabinoid system as a potential anticancer approach. Drug Metab. Rev. 2018, 50, 26–53. [Google Scholar] [CrossRef]
- Wang, J.; Xu, Y.; Zhu, L.; Zou, Y.; Kong, W.; Dong, B.; Huang, J.; Chen, Y.; Xue, W.; Huang, Y. Cannabinoid receptor 2 as a novel target for promotion of renal cell carcinoma prognosis and progression. J. Cancer Res. Clin. Oncol. 2018, 144, 39–52. [Google Scholar] [CrossRef]
- Martínez-Martínez, E.; Gómez, I.; Martín, P.; Sánchez, A.; Román, L.; Tejerina, E.; Bonilla, F.; Merino, A.G.; de Herreros, A.G.; Provencio, M. Cannabinoids receptor type 2, CB2, expression correlates with human colon cancer progression and predicts patient survival. Oncoscience 2015, 2, 131. [Google Scholar] [CrossRef]
- Eagleston, L.R.; Kalani, N.K.; Patel, R.R.; Flaten, H.K.; Dunnick, C.A.; Dellavalle, R.P. Cannabinoids in dermatology: A scoping review. Dermatol. Online J. 2018, 24, 13030. [Google Scholar] [CrossRef]
- Rao, M.; Chen, D.; Zhan, P.; Jiang, J. MDA19, a novel CB2 agonist, inhibits hepatocellular carcinoma partly through inactivation of AKT signaling pathway. Biol. Direct 2019, 14, 9. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Martínez, E.; Martín-Ruiz, A.; Martín, P.; Calvo, V.; Provencio, M.; García, J.M. CB2 cannabinoid receptor activation promotes colon cancer progression via AKT/GSK3β signaling pathway. Oncotarget 2016, 7, 68781. [Google Scholar] [CrossRef] [PubMed]
- Iden, J.A.; Raphael-Mizrahi, B.; Awida, Z.; Naim, A.; Zyc, D.; Liron, T.; Kasher, M.; Livshits, G.; Vered, M.; Gabet, Y. The Anti-Tumorigenic Role of Cannabinoid Receptor 2 in Colon Cancer: A Study in Mice and Humans. Int. J. Mol. Sci. 2023, 24, 4060. [Google Scholar] [CrossRef]
- Kapellos, T.S.; Recio, C.; Greaves, D.R.; Iqbal, A.J. Cannabinoid receptor 2 modulates neutrophil recruitment in a murine model of endotoxemia. Mediat. Inflamm. 2017, 2017, 4315412. [Google Scholar] [CrossRef] [PubMed]
- Hensel, J.A.; Khattar, V.; Ponnazhagan, S. Characterization of immune cell subtypes in three commonly used mouse strains reveals gender and strain-specific variations. Lab. Investig. 2019, 99, 93–106. [Google Scholar] [CrossRef]
- Price, P.W.; Cerny, J. Characterization of CD4+ T cells in mouse bone marrow. I. Increased activated/memory phenotype and altered TCR Vβ repertoire. Eur. J. Immunol. 1999, 29, 1051–1056. [Google Scholar] [CrossRef]
- Zhao, E.; Xu, H.; Wang, L.; Kryczek, I.; Wu, K.; Hu, Y.; Wang, G.; Zou, W. Bone marrow and the control of immunity. Cell. Mol. Immunol. 2012, 9, 11–19. [Google Scholar] [CrossRef]
- Miettinen, M.; Cue, P.A.M.; Sarlomo-Rikala, M.; Rys, J.; Czapiewski, P.; Wazny, K.; Langfort, R.; Waloszczyk, P.; Biernat, W.; Lasota, J. GATA 3–a multispecific but potentially useful marker in surgical pathology–a systematic analysis of 2500 epithelial and non-epithelial tumors. Am. J. Surg. Pathol. 2014, 38, 13. [Google Scholar] [CrossRef]
- Gasparoto, T.H.; de Oliveira, C.E.; de Freitas, L.T.; Pinheiro, C.R.; Ramos, R.N.; da Silva, A.L.; Garlet, G.P.; da Silva, J.S.; Campanelli, A.P. Inflammatory events during murine squamous cell carcinoma development. J. Inflamm. 2012, 9, 46. [Google Scholar] [CrossRef]
- Palazuelos, J.; Davoust, N.; Julien, B.; Hatterer, E.; Aguado, T.; Mechoulam, R.; Benito, C.; Romero, J.; Silva, A.; Guzman, M. The CB2 cannabinoid receptor controls myeloid progenitor trafficking: Involvement in the pathogenesis of an animal model of multiple sclerosis. J. Biol. Chem. 2008, 283, 13320–13329. [Google Scholar] [CrossRef] [PubMed]
- Csóka, B.; Németh, Z.H.; Mukhopadhyay, P.; Spolarics, Z.; Rajesh, M.; Federici, S.; Deitch, E.A.; Bátkai, S.; Pacher, P.; Haskó, G. CB2 cannabinoid receptors contribute to bacterial invasion and mortality in polymicrobial sepsis. PLoS ONE 2009, 4, e6409. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, K.; Goel, S.; Funakoshi, A.; Honda, T.; Nagao, K. Flow cytometry analysis of the subpopulations of mouse keratinocytes and skin immune cells. STAR Protoc. 2022, 3, 101052. [Google Scholar] [CrossRef] [PubMed]
- Lança, T.; Silva-Santos, B. The split nature of tumor-infiltrating leukocytes: Implications for cancer surveillance and immunotherapy. Oncoimmunology 2012, 1, 717–725. [Google Scholar] [CrossRef]
- Yusuf, N.; Nasti, T.H.; Katiyar, S.K.; Jacobs, M.K.; Seibert, M.D.; Ginsburg, A.C.; Timares, L.; Xu, H.; Elmets, C.A. Antagonistic roles of CD4+ and CD8+ T-cells in 7, 12-dimethylbenz (a) anthracene cutaneous carcinogenesis. Cancer Res. 2008, 68, 3924–3930. [Google Scholar] [CrossRef]
- Robinson, R.H.; Meissler, J.J.; Breslow-Deckman, J.M.; Gaughan, J.; Adler, M.W.; Eisenstein, T.K. Cannabinoids inhibit T-cells via cannabinoid receptor 2 in an in vitro assay for graft rejection, the mixed lymphocyte reaction. J. Neuroimmune Pharmacol. 2013, 8, 1239–1250. [Google Scholar] [CrossRef] [PubMed]
- Woodworth, C.D.; Michael, E.; Smith, L.; Vijayachandra, K.; Glick, A.; Hennings, H.; Yuspa, S.H. Strain-dependent differences in malignant conversion of mouse skin tumors is an inherent property of the epidermal keratinocyte. Carcinogenesis 2004, 25, 1771–1778. [Google Scholar] [CrossRef][Green Version]
- Fujiwara, K.; Inagaki, Y.; Soma, M.; Ozaki, T.; Nagase, H. Mapping of new skin tumor susceptibility loci by a phenotype-driven congenic approach. Oncol. Lett. 2018, 16, 6670–6676. [Google Scholar] [CrossRef]
- Nagase, H.; Bryson, S.; Cordell, H.; Kemp, C.J.; Fee, F.; Balmain, A. Distinct genetic loci control development of benign and malignant skin tumours in mice. Nat. Genet. 1995, 10, 424–429. [Google Scholar] [CrossRef]
- Nagase, H.; Mao, J.-H.; Balmain, A. A subset of skin tumor modifier loci determines survival time of tumor-bearing mice. Proc. Natl. Acad. Sci. USA 1999, 96, 15032–15037. [Google Scholar] [CrossRef]
- Blázquez, C.; Casanova, M.L.; Planas, A.; Gómez del Pulgar, T.; Villanueva, C.; Fernández-Aceñero, M.J.; Aragonés, J.; Huffman, J.W.; Jorcano, J.L.; Guzmán, M. Inhibition of tumor angiogenesis by cannabinoids. FASEB J. 2003, 17, 529–531. [Google Scholar] [CrossRef]
- Casanova, M.L.; Blázquez, C.; Martínez-Palacio, J.; Villanueva, C.; Fernández-Aceñero, M.J.; Huffman, J.W.; Jorcano, J.L.; Guzmán, M. Inhibition of skin tumor growth and angiogenesis in vivo by activation of cannabinoid receptors. J. Clin. Investig. 2003, 111, 43–50. [Google Scholar] [CrossRef]
- Van Dross, R.T. Metabolism of anandamide by COX-2 is necessary for endocannabinoid-induced cell death in tumorigenic keratinocytes. Mol. Carcinog. Publ. Coop. Univ. Tex. MD Cancer Cent. 2009, 48, 724–732. [Google Scholar] [CrossRef]
- Halliday, G.M.; Patel, A.; Hunt, M.J.; Tefany, F.J.; Barnetson, R.S.C. Spontaneous regression of human melanoma/nonmelanoma skin cancer: Association with infiltrating CD4+ T cells. World J. Surg. 1995, 19, 352–358. [Google Scholar] [CrossRef]
- Vanbuskirk, A.; Oberyszyn, T.M.; Kusewitt, D.F. Depletion of CD8+ or CD4+ lymphocytes enhances susceptibility to transplantable ultraviolet radiation-induced skin tumours. Anticancer Res. 2005, 25, 1963–1967. [Google Scholar]
- Ho, I.-C.; Tai, T.-S.; Pai, S.-Y. GATA3 and the T-cell lineage: Essential functions before and after T-helper-2-cell differentiation. Nat. Rev. Immunol. 2009, 9, 125–135. [Google Scholar] [CrossRef]
- Yanofsky, V.R.; Mitsui, H.; Felsen, D.; Carucci, J.A. Understanding dendritic cells and their role in cutaneous carcinoma and cancer immunotherapy. Clin. Dev. Immunol. 2013, 2013, 624123. [Google Scholar] [CrossRef]
- Gaffal, E.; Kemter, A.M.; Scheu, S.; Leite Dantas, R.; Vogt, J.; Baune, B.; Tüting, T.; Zimmer, A.; Alferink, J. Cannabinoid receptor 2 modulates maturation of dendritic cells and their capacity to induce hapten-induced contact hypersensitivity. Int. J. Mol. Sci. 2020, 21, 475. [Google Scholar] [CrossRef]
- Do, Y.; McKallip, R.J.; Nagarkatti, M.; Nagarkatti, P.S. Activation through cannabinoid receptors 1 and 2 on dendritic cells triggers NF-κB-dependent apoptosis: Novel role for endogenous and exogenous cannabinoids in immunoregulation. J. Immunol. 2004, 173, 2373–2382. [Google Scholar] [CrossRef]
- Neagu, M.; Constantin, C.; Caruntu, C.; Dumitru, C.; Surcel, M.; Zurac, S. Inflammation: A key process in skin tumorigenesis. Oncol. Lett. 2019, 17, 4068–4084. [Google Scholar] [CrossRef]
- Wilkinson, J.D.; Williamson, E.M. Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J. Dermatol. Sci. 2007, 45, 87–92. [Google Scholar] [CrossRef]
- Tóth, B.I.; Dobrosi, N.; Dajnoki, A.; Czifra, G.; Oláh, A.; Szöllősi, A.G.; Juhász, I.; Sugawara, K.; Paus, R.; Bíró, T. Endocannabinoids modulate human epidermal keratinocyte proliferation and survival via the sequential engagement of cannabinoid receptor-1 and transient receptor potential vanilloid-1. J. Investig. Dermatol. 2011, 131, 1095–1104. [Google Scholar] [CrossRef]
- Norooznezhad, A.H.; Norooznezhad, F. Cannabinoids: Possible agents for treatment of psoriasis via suppression of angiogenesis and inflammation. Med. Hypotheses 2017, 99, 15–18. [Google Scholar] [CrossRef]
- Nakajima, J.i.; Nakae, D.; Yasukawa, K. Structure-dependent inhibitory effects of synthetic cannabinoids against 12-O-tetradecanoylphorbol-13-acetate-induced inflammation and skin tumour promotion in mice. J. Pharm. Pharmacol. 2013, 65, 1223–1230. [Google Scholar] [CrossRef]
- Bialkowska, A.B.; Ghaleb, A.M.; Nandan, M.O.; Yang, V.W. Improved Swiss-rolling technique for intestinal tissue preparation for immunohistochemical and immunofluorescent analyses. JoVE (J. Vis. Exp.) 2016, 113, e54161. [Google Scholar]
- Regezi, J.A.; Sciubba, J.J.; Jordan, R.C. Oral Pathology: Clinical Pathologic Correlations; Elsevier Health Sciences: Amsterdam, The Netherlands, 2016. [Google Scholar]


| Primer sequence 5′-3′ | |
| GATA3_F | GTGGTCACACTCGGATTCCT |
| GATA3_R | GCAAAAAGGAGGGTTTAGGG |
| TSLP_F | AGGCTACCCTGAAACTGAG |
| TSLP_R | GGAGATTGCATGAAGGAATACC |
| IL-17_F | ACCGCAATGAAGACCCTGAT |
| IL-17_R | TCCCTCCGCATTGACACA |
| IL-23_F | GACAACAGCCAGTTCTGCTT |
| IL-23_R | AGGGAGGTGTGAAGTTGCTC |
| IL-10_F | TGAGGCGCTGTCGTCATCGATTTCTCCC |
| IL-10_R | ACCTGCTCCACTGCCTTGCT |
| IL-6_F | CCGGAGAGGAGACTTCACAG |
| IL-6_R | GGAAATTGGGGTAGGAAGGA |
| β-actin_F | GTCACCCACACTGTGCCCATC |
| β-actin_R | CCGTCAGGCAGCTCATAGCTC |
| β2M_F | TGCTACTCGGCGCTTCAGTC |
| β2M_R | AGGCGGGTGGAACTGTGTTAC |
| CB2_F | TGACCATGACCTTCACAGCC |
| CB2_R | GGTAGGCGGGTAACACAGAC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iden, J.A.; Raphael-Mizrahi, B.; Naim, A.; Kolomansky, A.; Liron, T.; Neumann, D.; Vered, M.; Gabet, Y. The Anti-Tumorigenic Role of Cannabinoid Receptor 2 in Non-Melanoma Skin Cancer. Int. J. Mol. Sci. 2023, 24, 7773. https://doi.org/10.3390/ijms24097773
Iden JA, Raphael-Mizrahi B, Naim A, Kolomansky A, Liron T, Neumann D, Vered M, Gabet Y. The Anti-Tumorigenic Role of Cannabinoid Receptor 2 in Non-Melanoma Skin Cancer. International Journal of Molecular Sciences. 2023; 24(9):7773. https://doi.org/10.3390/ijms24097773
Chicago/Turabian StyleIden, Jennifer Ana, Bitya Raphael-Mizrahi, Aaron Naim, Albert Kolomansky, Tamar Liron, Drorit Neumann, Marilena Vered, and Yankel Gabet. 2023. "The Anti-Tumorigenic Role of Cannabinoid Receptor 2 in Non-Melanoma Skin Cancer" International Journal of Molecular Sciences 24, no. 9: 7773. https://doi.org/10.3390/ijms24097773
APA StyleIden, J. A., Raphael-Mizrahi, B., Naim, A., Kolomansky, A., Liron, T., Neumann, D., Vered, M., & Gabet, Y. (2023). The Anti-Tumorigenic Role of Cannabinoid Receptor 2 in Non-Melanoma Skin Cancer. International Journal of Molecular Sciences, 24(9), 7773. https://doi.org/10.3390/ijms24097773






