The RING-Finger Protein NbRFP1 Contributes to Regulating the Host Hypersensitive Response Induced by Oat Dwarf Virus RepA
Abstract
1. Introduction
2. Results
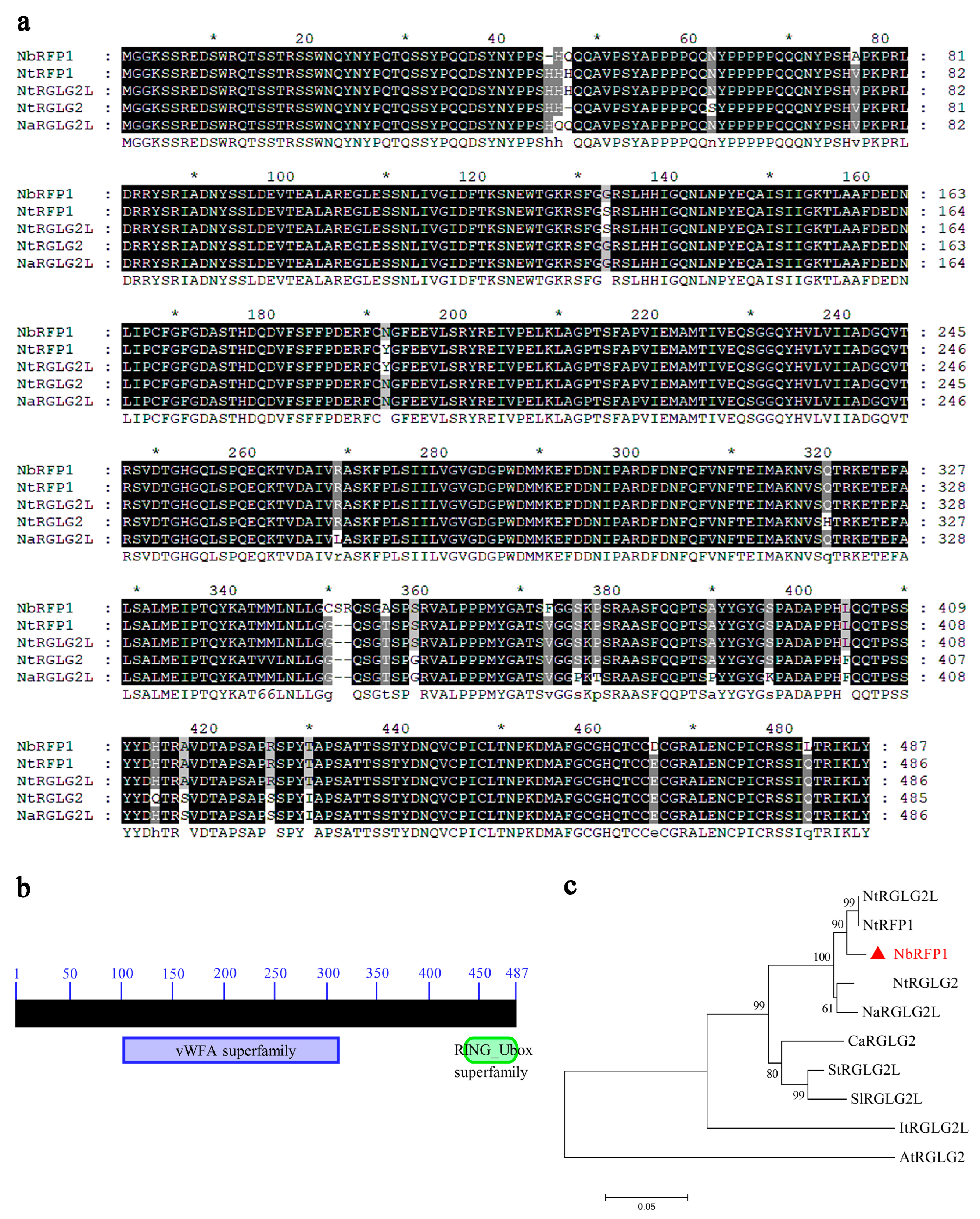
2.1. The Isolation and Sequence Analysis of NbRFP1
2.2. Expression Pattern and Subcellular Localization of NbRFP1
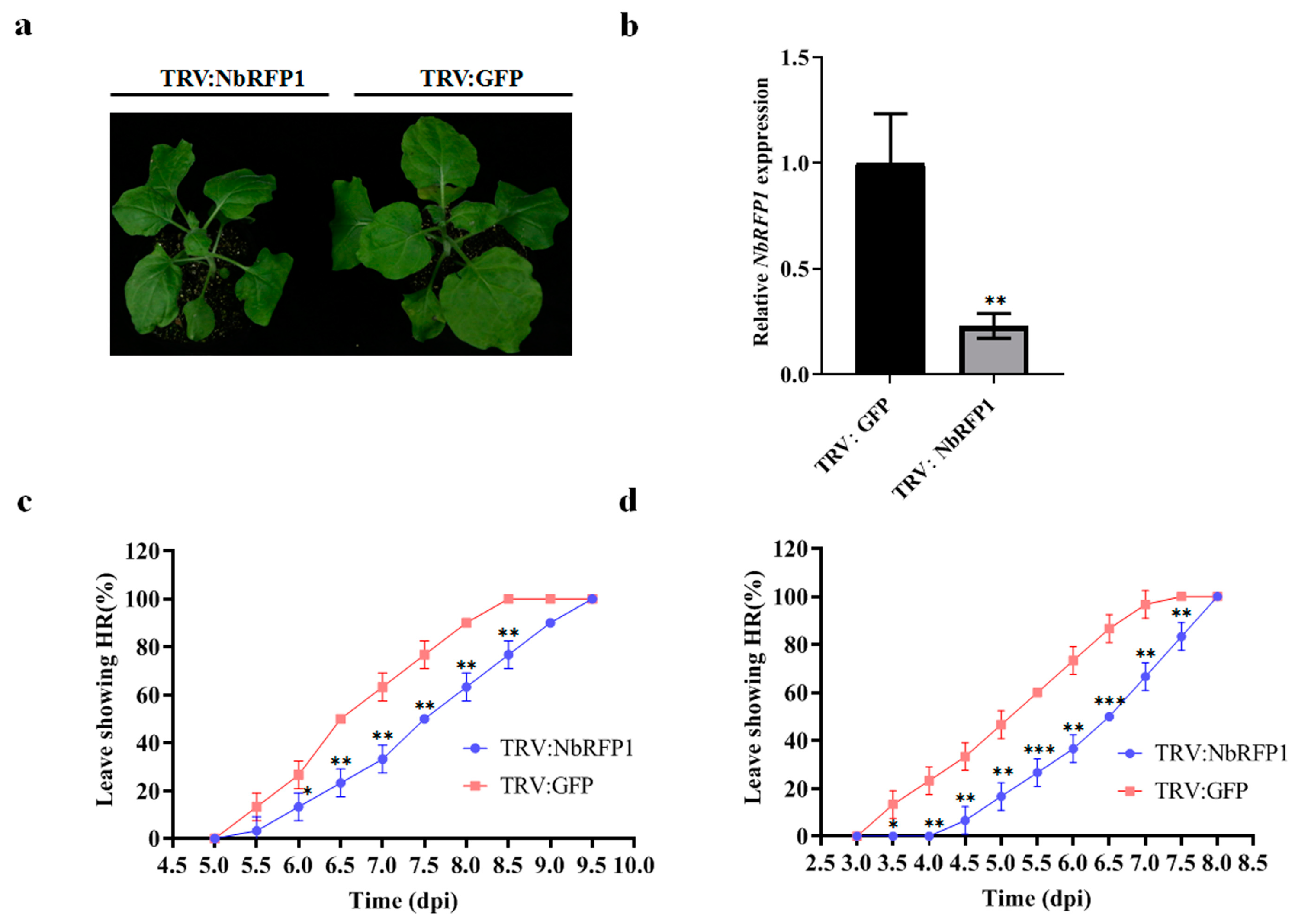
2.3. Silencing of NbRFP1 Delays ODV RepA-Induced HR
2.4. The Overexpression of NbRFP1 Enhances ODV RepA-Induced HR
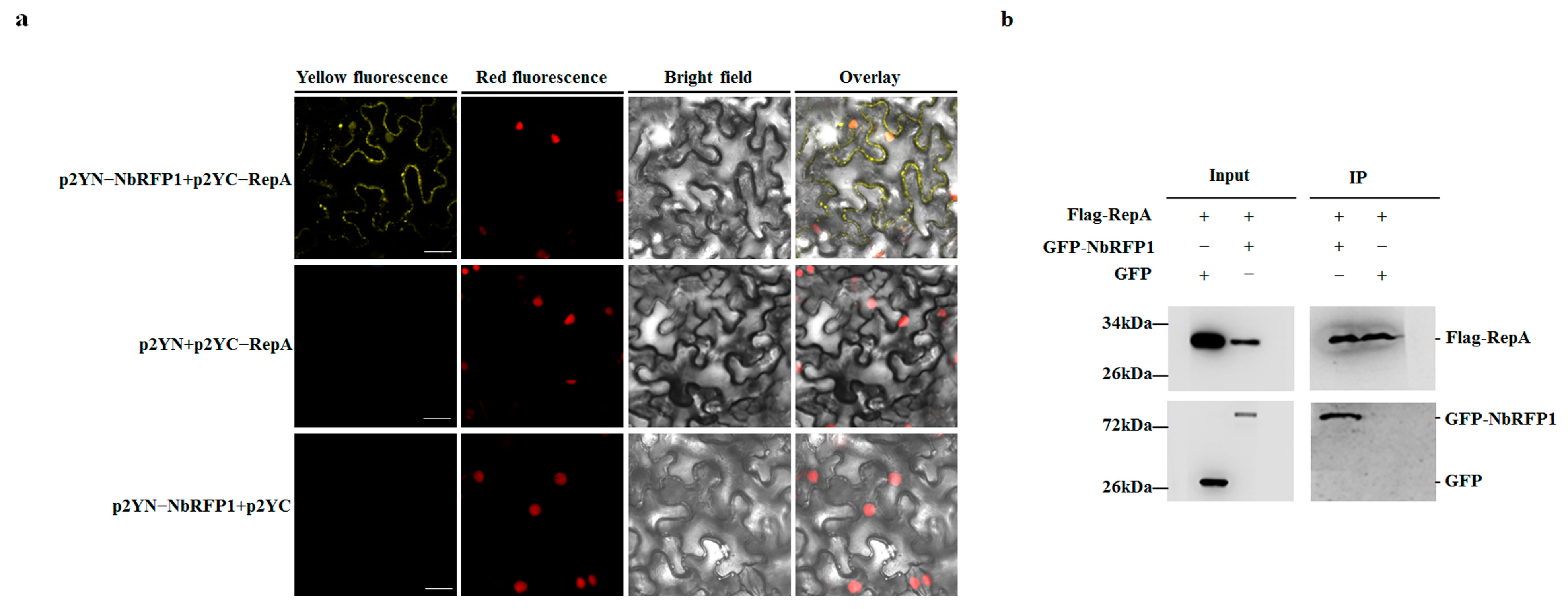
2.5. ODV RepA Interacts with NbRFP1
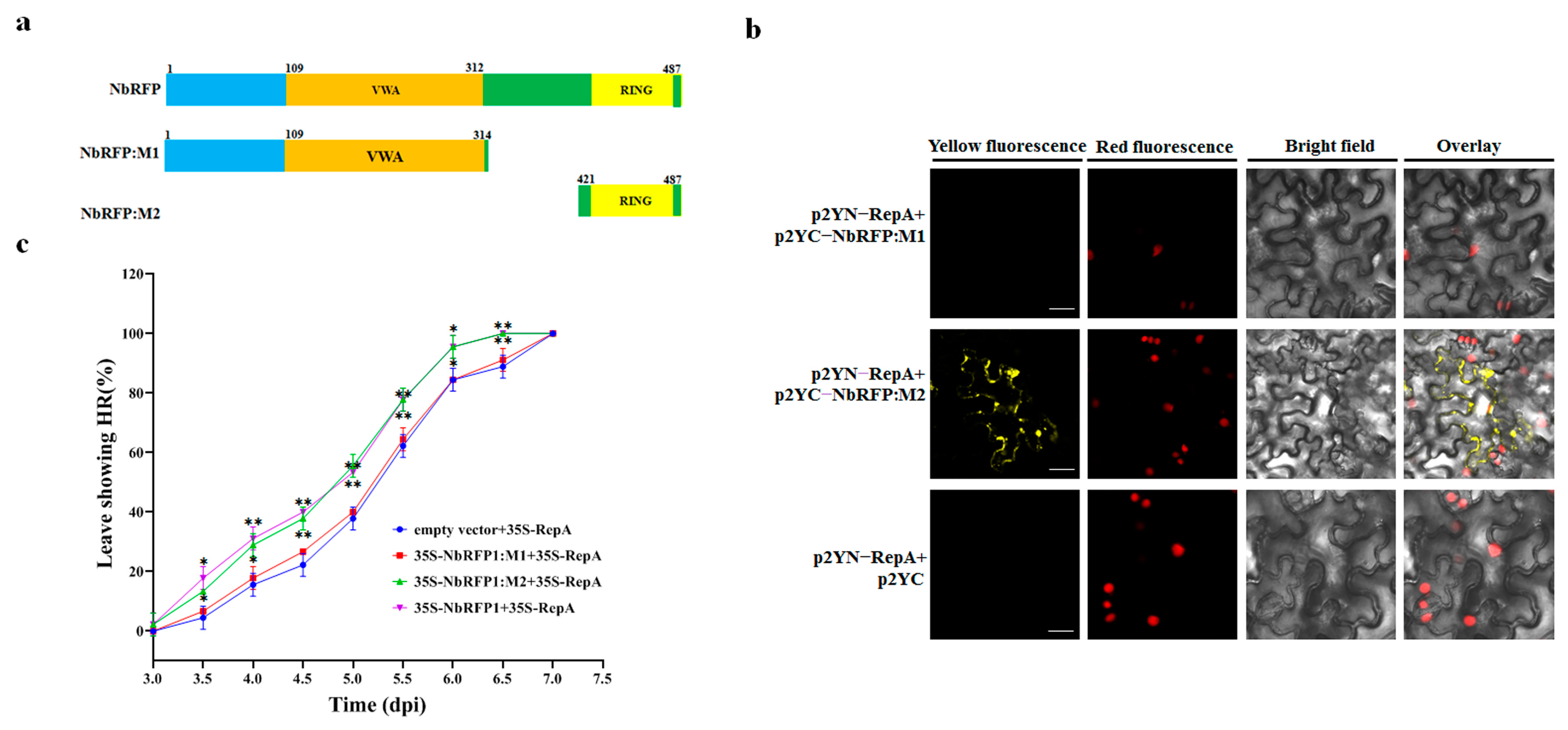
2.6. The RING-Finger Domain in NbRFP1 Was Involved in the Interaction with RepA and Positively Regulated RepA-Induced HR
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. RNA Extraction, RT-PCR, and qPCR Analyses
4.3. Subcellular Localization
4.4. BiFC Assay
4.5. Protein Extraction and Western Blotting Analysis
4.6. CoIP Assay
4.7. Silencing of Endogenous NbRFP1
4.8. Transient Co-Expression of RepA and NbRFP1
4.9. Phylogenetic Analysis
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, P.; Day, B. Battlefield cytoskeleton: Turning the tide on plant immunity. Mol. Plant Microbe Interact. 2019, 32, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Davies, L.J.; Brown, C.R.; Elling, A.A. Calcium is involved in the RMc1(blb)-mediated hypersensitive response against Meloidogyne chitwoodi in potato. Plant Cell Rep. 2015, 34, 167–177. [Google Scholar] [CrossRef]
- Mur, L.A.J.; Kenton, P.; Lloyd, A.J.; Ougham, H.; Prats, E. The hypersensitive response; the centenary is upon us but how much do we know? J. Exp. Bot. 2008, 59, 501–520. [Google Scholar] [CrossRef]
- Kunstler, A.; Bacso, R.; Gullner, G.; Hafez, Y.M.; Kiraly, L. Staying alive-is cell death dispensable for plant disease resistance during the hypersensitive response? Physiol. Mol. Plant Pathol. 2016, 93, 75–84. [Google Scholar] [CrossRef]
- Chowdhury, R.N.; Lasky, D.; Karki, H.; Zhang, Z.Y.; Goyer, A.; Halterman, D.; Rakotondrafara, A.M. HCPro suppression of callose deposition contributes to strain-specific resistance against potato virus Y. Phytopathology 2020, 110, 164–173. [Google Scholar] [CrossRef]
- Gamage, S.; McGrath, D.J.; Persley, D.M.; Dietzgen, R.G. Transcriptome analysis of capsicum chlorosis virus-induced hypersensitive resistance response in bell capsicum. PLoS ONE 2016, 11, e0159085. [Google Scholar]
- Lukan, T.; Zupanic, A.; Povalej, T.M.; Brunkard, J.O.; Kmetic, M.; Jutersek, M.; Baebler, S.; Gruden, K. Chloroplast redox state changes mark cell-to-cell signaling in the hypersensitive response. New Phytol. 2023, 237, 548–562. [Google Scholar] [CrossRef]
- Qian, Y.; Hou, H.; Shen, Q.; Cai, X.; Sunter, G.; Zhou, X. RepA protein encoded by oat dwarf virus elicits a temperature-sensitive hypersensitive response-type cell death that involves jasmonic acid-dependent signaling. Mol. Plant Microbe Interact. 2016, 29, 5–21. [Google Scholar] [CrossRef]
- Xu, H.Y.; Zhang, C.; Li, Z.C.; Wang, Z.R.; Jiang, X.X.; Shi, Y.F.; Tian, S.N.; Braun, E.; Mei, Y.; Qiu, W.L.; et al. The MAPK kinase kinase GmMEKK1 regulates cell death and defense responses. Plant Physiol. 2018, 178, 907–922. [Google Scholar] [CrossRef]
- Chen, Y.M.; Song, Y.Y.; Liu, J.; Xu, G.Y.; Dou, D.L. Ubiquitination of receptorsomes, frontline of plant immunity. Int. J. Mol. Sci. 2022, 23, 2937. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.Y.; Tang, D.Z.; Wang, W. The role of ubiquitination in plant immunity: Fine-tuning immune signaling and beyond. Plant Cell Physiol. 2022, 63, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Lu, D.P.; Shan, L.B. Ubiquitination of pattern recognition receptors in plant innate immunity. Mol. Plant Pathol. 2014, 15, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Pickart, C.M.; Eddins, M.J. Ubiquitin: Structures, functions, mechanisms. Biochim. Biophys. Acta 2004, 1695, 55–72. [Google Scholar] [CrossRef]
- Bueso, E.; Rodriguez, L.; Lorenzo-Orts, L.; Gonzalez-Guzman, M.; Sayas, E.; Munoz-Bertomeu, J.; Ibanez, C.; Serrano, R.; Rodriguez, P.L. The single-subunit RING-type E3 ubiquitin ligase RSL1 targets PYL4 and PYR1 ABA receptors in plasma membrane to modulate abscisic acid signaling. Plant J. 2014, 80, 1057–1071. [Google Scholar] [CrossRef] [PubMed]
- Smalle, J.; Vierstra, R.D. The ubiquitin 26S proteasome proteolytic pathway. Annu. Rev. Plant Biol. 2004, 55, 555–590. [Google Scholar] [CrossRef]
- Shen, Q.T.; Hu, T.; Bao, M.; Cao, L.G.; Zhang, H.W.; Song, F.M.; Xie, Q.; Zhou, X.P. Tobacco RING E3 ligase NtRFP1 mediates ubiquitination and proteasomal degradation of a geminivirus-encoded beta C1. Mol. Plant 2016, 9, 911–925. [Google Scholar] [CrossRef]
- Fu, S.; Wang, K.; Ma, T.; Liang, Y.; Ma, Z.; Wu, J.; Xu, Y.; Zhou, X. An evolutionarily conserved C4HC3-type E3 ligase regulates plant broad-spectrum resistance against pathogens. Plant Cell 2022, 34, 1822–1843. [Google Scholar] [CrossRef]
- Deshaies, R.J.; Joazeiro, C.A.P. RING domain E3 ubiquitin ligases. Annu. Rev. Biochem. 2009, 78, 399–434. [Google Scholar] [CrossRef]
- Mazzucotelli, E.; Belloni, S.; Marone, D.; De Leonardis, A.M.; Guerra, D.; Fonzo, N.; Cattivelli, L.; Mastrangelo, A.M. The E3 ubiquitin ligase gene family in plants: Regulation by degradation. Curr. Genom. 2006, 7, 509–522. [Google Scholar] [CrossRef]
- Zhang, H.J.; Zheng, D.W.; Song, F.M.; Jiang, M. Expression patterns and functional analysis of 11 E3 ubiquitin ligase genes in rice. Front. Plant Sci. 2022, 13, 840360. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Choi, H.W.; Hwang, B.K. The pepper E3 ubiquitin ligase RING1 gene, CaRING1, is required for cell death and the salicylic acid-dependent defense response. Plant Physiol. 2011, 156, 2011–2025. [Google Scholar] [CrossRef]
- Li, W.; Zhong, S.; Li, G.; Li, Q.; Mao, B.; Deng, Y.; Zhang, H.; Zeng, L.; Song, F.; He, Z. Rice RING protein OsBBI1 with E3 ligase activity confers broad-spectrum resistance against Magnaporthe oryzae by modifying the cell wall defence. Cell Res. 2011, 21, 835–848. [Google Scholar] [CrossRef]
- Yu, G.; Derkacheva, M.; Rufian, J.S.; Brillada, C.; Kowarschik, K.; Jiang, S.S.; Derbyshire, P.; Ma, M.M.; DeFalco, T.A.; Morcillo, R.J.L.; et al. The Arabidopsis E3 ubiquitin ligase PUB4 regulates BIK1 and is targeted by a bacterial type-III effector. EMBO J. 2022, 41, e107257. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Chen, S.; Shirsekar, G.; Zhou, B.; Khang, C.H.; Songkumarn, P.; Afzal, A.J.; Ning, Y.; Wang, R.; Bellizzi, M.; et al. The Magnaporthe oryzae effector AvrPiz-t targets the RING E3 ubiquitin ligase APIP6 to suppress pathogen-associated molecular pattern-triggered immunity in rice. Plant Cell 2012, 24, 4748–4762. [Google Scholar] [CrossRef] [PubMed]
- Fiallo-Olive, E.; Lett, J.M.; Martin, D.P.; Roumagnac, P.; Varsani, A.; Zerbini, F.M.; Navas-Castillo, J.; Consortium, I.R. ICTV virus taxonomy profile: Geminiviridae 2021. J. Gen. Virol. 2021, 102, 001696. [Google Scholar] [CrossRef] [PubMed]
- Schubert, J.; Habekuss, A.; Kazmaier, K.; Jeske, H. Surveying cereal-infecting geminiviruses in Germany-diagnostics and direct sequencing using rolling circle amplification. Virus Res. 2007, 127, 61–70. [Google Scholar] [CrossRef]
- Hou, H.; Hu, Y.; Wang, Q.; Xu, X.; Qian, Y.; Zhou, X. Gene expression profiling shows that NbFDN1 is involved in modulating the hypersensitive response-like cell death induced by the oat dwarf virus RepA protein. Mol. Plant-Microbe Interact. 2018, 31, 1006–1020. [Google Scholar] [CrossRef] [PubMed]
- Kashihara, S.; Nishimura, T.; Noutoshi, Y.; Yamamoto, M.; Toyoda, K.; Ichinose, Y.; Matsui, H. HopAZ1, a type III effector of Pseudomonas amygdali pv. tabaci, induces a hypersensitive response in tobacco wildfire-resistant Nicotiana tabacum ‘N509’. Mol. Plant Pathol. 2022, 23, 885–894. [Google Scholar] [CrossRef]
- Matic, S.; Pegoraro, M.; Noris, E. The C2 protein of tomato yellow leaf curl Sardinia virus acts as a pathogenicity determinant and a 16-amino acid domain is responsible for inducing a hypersensitive response in plants. Virus Res. 2016, 215, 12–19. [Google Scholar] [CrossRef]
- Mesarich, C.H.; Stergiopoulos, I.; Beenen, H.G.; Cordovez, V.; Guo, Y.N.; Jashni, M.K.; Bradshaw, R.E.; de Wit, P. A conserved proline residue in Dothideomycete Avr4 effector proteins is required to trigger a Cf-4-dependent hypersensitive response. Mol. Plant Pathol. 2016, 17, 84–95. [Google Scholar] [CrossRef]
- Brugiere, N.; Zhang, W.; Xu, Q.; Scolaro, E.J.; Lu, C.; Kahsay, R.Y.; Kise, R.; Trecker, L.; Williams, R.W.; Hakimi, S.; et al. Overexpression of RING domain E3 ligase ZmXerico1 confers drought tolerance through regulation of ABA homeostasis. Plant Physiol. 2017, 175, 1350–1369. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.Y.; Park, H.L.; Park, C.; Chen, Y.C.; Yoon, G.M. Reciprocal antagonistic regulation of E3 ligases controls ACC synthase stability and responses to stress. Proc. Natl. Acad. Sci. USA. 2021, 118, e2011900118. [Google Scholar] [CrossRef]
- Liu, H.X.; Ravichandran, S.; Teh, O.K.; McVey, S.; Lilley, C.; Teresinski, H.J.; Gonzalez-Ferrer, C.; Mullen, R.T.; Hofius, D.; Prithiviraj, B.; et al. The RING-type E3 ligase XBAT35.2 is involved in cell death induction and pathogen response. Plant Physiol. 2017, 175, 1469–1483. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Xie, X.; Yao, W.; Wang, J.; Ma, F.; Wang, C.; Yang, Y.; Tong, W.; Zhang, J.; Xu, Y.; et al. RING-H2-type E3 gene VpRH2 from Vitis pseudoreticulata improves resistance to Powdery mildew by interacting with VpGRP2A. J. Exp. Bot. 2017, 68, 1669–1687. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lim, S.D.; Jung, K.H.; Jang, C.S. Overexpression of a C3HC4-type RING E3 ligase gene, OsRFPHC-13, improves salinity resistance in rice, Oryza sativa, by altering the expression of Na+/K+ transporter genes. Environ. Exp. Bot. 2023, 207, 925–939. [Google Scholar] [CrossRef]
- Yi, S.Y.; Lee, M.Y.J.; Kwon, S.Y.; Kim, W.T.; Lim, Y.P.; Kang, S.Y. RING-type E3 ubiquitin ligases AtRDUF1 and AtRDUF2 positively regulate the expression of PR1 gene and pattern-triggered immunity. Int. J. Mol. Sci. 2022, 23, 14525. [Google Scholar] [CrossRef]
- Kawasaki, T.; Nam, J.; Boyes, D.C.; Holt, B.F.; Hubert, D.A.; Wiig, A.; Dangl, J.L. A duplicated pair of Arabidopsis RING-finger E3 ligases contribute to the RPM1-and RPS2-mediated hypersensitive response. Plant J. 2005, 44, 258–270. [Google Scholar] [CrossRef]
- Gao, M.J.; He, Y.; Yin, X.; Zhong, X.B.; Yan, B.X.; Wu, Y.; Chen, J.; Li, X.Y.; Zhai, K.R.; Huang, Y.F.; et al. Ca2+ sensor-mediated ROS scavenging suppresses rice immunity and is exploited by a fungal effector. Cell 2021, 184, 5391. [Google Scholar] [CrossRef]
- Ghannam, A.; Jacques, A.; de Ruffray, P.; Kauffmann, S. NtRING1, putative RING-finger E3 ligase protein, is a positive regulator of the early stages of elicitin-induced HR in tobacco. Plant Cell Rep. 2016, 35, 415–428. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, C.; Li, Y.; Zheng, N.; Chen, H.; Zhao, Q.; Gao, T.; Guo, H.; Xie, Q. SDIR1 is a RING finger E3 ligase that positively regulates stress-responsive abscisic acid signaling in Arabidopsis. Plant Cell 2007, 19, 1912–1929. [Google Scholar] [CrossRef] [PubMed]
- Stone, S.L.; Williams, L.A.; Farmer, L.M.; Vierstra, R.D.; Callis, J. Keep on going, a RING E3 ligase essential for Arabidopsis growth and development, is involved in abscisic acid signaling. Plant Cell 2006, 18, 3415–3428. [Google Scholar] [CrossRef]
- Park, C.H.; Shirsekar, G.; Bellizzi, M.; Chen, S.B.; Songkumarn, P.; Xie, X.; Shi, X.T.; Ning, Y.S.; Zhou, B.; Suttiviriya, P.; et al. The E3 ligase APIP10 connects the effector AvrPiz-t to the NLR receptor Piz-t in rice. PLoS Pathog. 2016, 12, e1005529. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.F.; Jin, L.; Huang, X.H.; Geng, Y.T.; Li, F.; Qin, Q.Q.; Wang, R.; Ji, S.Y.; Zhao, S.S.; Xie, Q.; et al. OsRFPH2-10, a RING-H2 Finger E3 ubiquitin ligase, is involved in rice antiviral defense in the early stages of rice dwarf virus infection. Mol. Plant 2014, 7, 1057–1060. [Google Scholar] [CrossRef]
- Ali, M.R.M.; Uemura, T.; Ramadan, A.; Adachi, K.; Nemoto, K.; Nozawa, A.; Hoshino, R.; Abe, H.; Sawasaki, T.; Arimura, G.I. The Ring-type E3 ubiquitin ligase JUL1 targets the VQ-Motif protein JAV1 to coordinate jasmonate signaling. Plant Physiol. 2019, 179, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.S.; Shi, X.T.; Wang, R.Y.; Fan, J.B.; Park, C.H.; Zhang, C.Y.; Zhang, T.; Ouyang, X.H.; Li, S.G.; Wang, G.L. OsELF3-2, an ortholog of Arabidopsis ELF3, interacts with the E3 Ligase APIP6 and negatively regulates immunity against Magnaporthe oryzae in Rice. Mol. Plant. 2015, 8, 1679–1682. [Google Scholar] [CrossRef]
- Suh, J.Y.; Kim, S.J.; Oh, T.R.; Cho, S.K.; Yang, S.W.; Kim, W.T. Arabidopsis Toxicos en Levadura 78 (AtATL78) mediates ABA-dependent ROS signaling in response to drought stress. Biochem. Biophys. Res. Commun. 2016, 469, 8–14. [Google Scholar] [CrossRef]
- Trujillo, M.; Shirasu, K. Ubiquitination in plant immunity. Curr. Opin. Plant Biol. 2010, 13, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.B.; Chen, H.; Teng, K.L.; Zhao, Q.Z.; Zhang, Z.H.; Li, Y.; Liang, L.M.; Xia, R.; Wu, Y.R.; Guo, H.S.; et al. RKP, a RING finger E3 ligase induced by BSCTV C4 protein, affects geminivirus infection by regulation of the plant cell cycle. Plant J. 2009, 57, 905–917. [Google Scholar] [CrossRef]
- Lin, Y.; Hu, Q.; Zhou, J.; Yin, W.; Yao, D.; Shao, Y.; Zhao, Y.; Guo, B.; Xia, Y.; Chen, Q.; et al. Phytophthora sojae effector Avr1d functions as an E2 competitor and inhibits ubiquitination activity of GmPUB13 to facilitate infection. Proc. Natl. Acad. Sci. USA 2021, 118, e2018312118. [Google Scholar] [CrossRef]
- Li, S.; Hanlon, R.; Wise, H.; Pal, N.; Brar, H.; Liao, C.; Gao, H.; Perez, E.; Zhou, L.; Tyler, B.M.; et al. Interaction of Phytophthora sojae effector Avr1b with E3 ubiquitin ligase GmPUB1 is required for recognition by soybeans carrying Phytophthora resistance Rps1-b and Rps1-k genes. Front. Plant Sci. 2021, 12, 725571. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2–ΔΔCt method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Xu, Y.; Fu, S.; Liu, Y.; Li, Z.D.; Zhang, T.Z.; Wu, J.X.; Zhou, X.P. The unfolded protein response plays dual roles in rice stripe virus infection through fine-tuning the movement protein accumulation. PLoS Pathog. 2021, 17, e1009370. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Tang, Q.Y.; Zhang, C.X. Data processing system (DPS) software with experimental design, statistical analysis and data mining developed for use in entomological research. Insect Sci. 2013, 20, 254–260. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, Y.; Wang, Z.; Wang, Q.; Zhou, X.; Qian, Y. The RING-Finger Protein NbRFP1 Contributes to Regulating the Host Hypersensitive Response Induced by Oat Dwarf Virus RepA. Int. J. Mol. Sci. 2023, 24, 7697. https://doi.org/10.3390/ijms24097697
Liang Y, Wang Z, Wang Q, Zhou X, Qian Y. The RING-Finger Protein NbRFP1 Contributes to Regulating the Host Hypersensitive Response Induced by Oat Dwarf Virus RepA. International Journal of Molecular Sciences. 2023; 24(9):7697. https://doi.org/10.3390/ijms24097697
Chicago/Turabian StyleLiang, Yanqing, Zhanqi Wang, Qian Wang, Xueping Zhou, and Yajuan Qian. 2023. "The RING-Finger Protein NbRFP1 Contributes to Regulating the Host Hypersensitive Response Induced by Oat Dwarf Virus RepA" International Journal of Molecular Sciences 24, no. 9: 7697. https://doi.org/10.3390/ijms24097697
APA StyleLiang, Y., Wang, Z., Wang, Q., Zhou, X., & Qian, Y. (2023). The RING-Finger Protein NbRFP1 Contributes to Regulating the Host Hypersensitive Response Induced by Oat Dwarf Virus RepA. International Journal of Molecular Sciences, 24(9), 7697. https://doi.org/10.3390/ijms24097697






