Efficiency of Promoters of Human Genes FAP and CTGF at Organism Level in a Danio rerio Model
Abstract
1. Introduction
2. Results
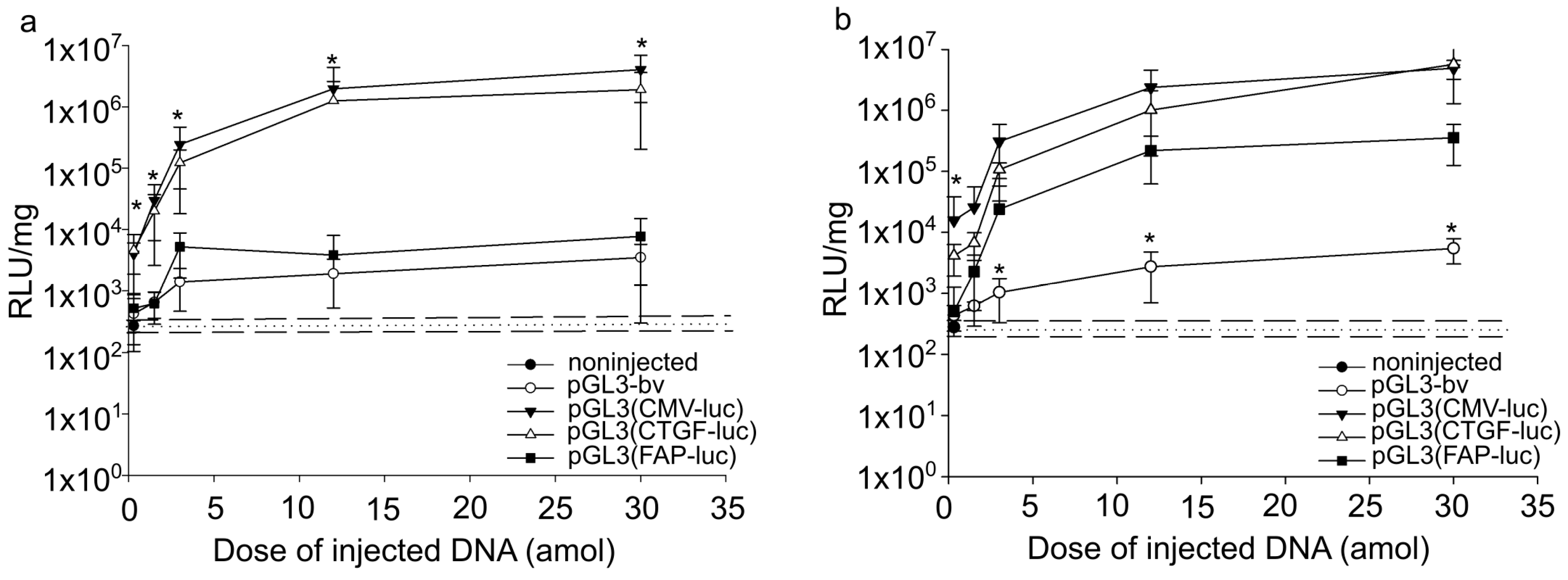
2.1. Quantitative Analysis of Expression Vector Efficiencies at Organism Level
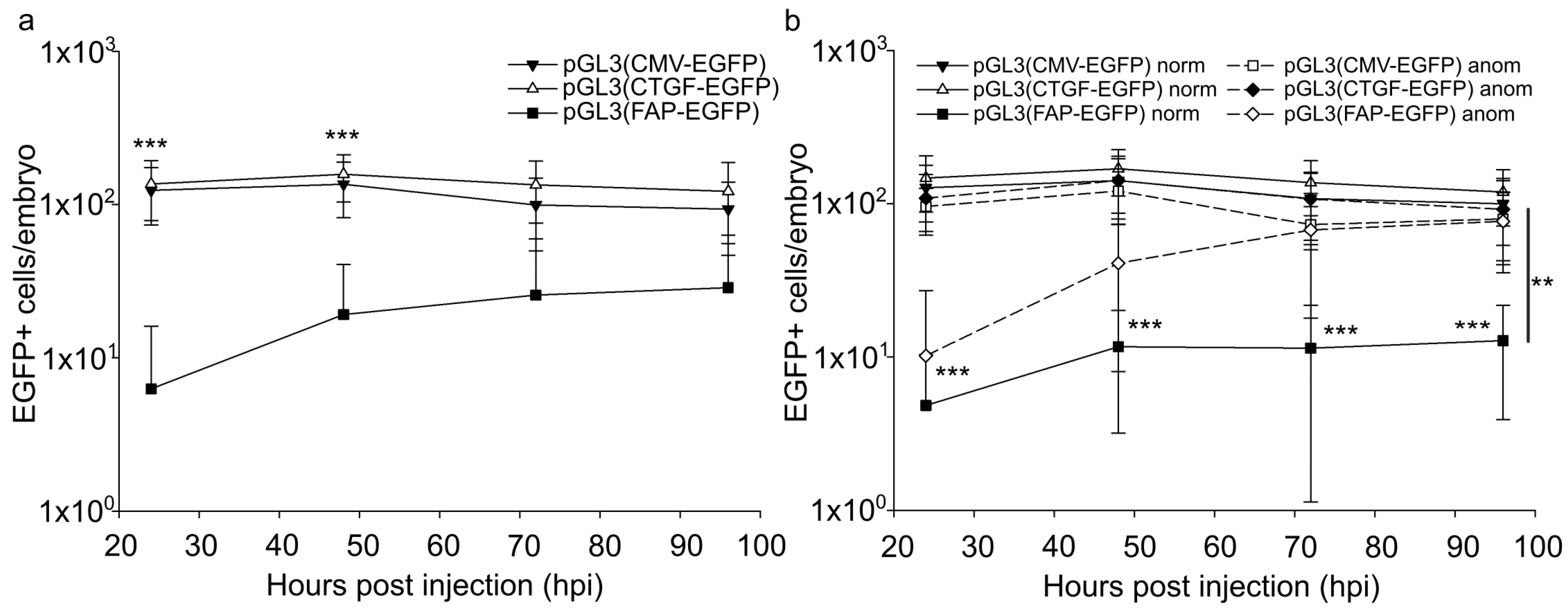
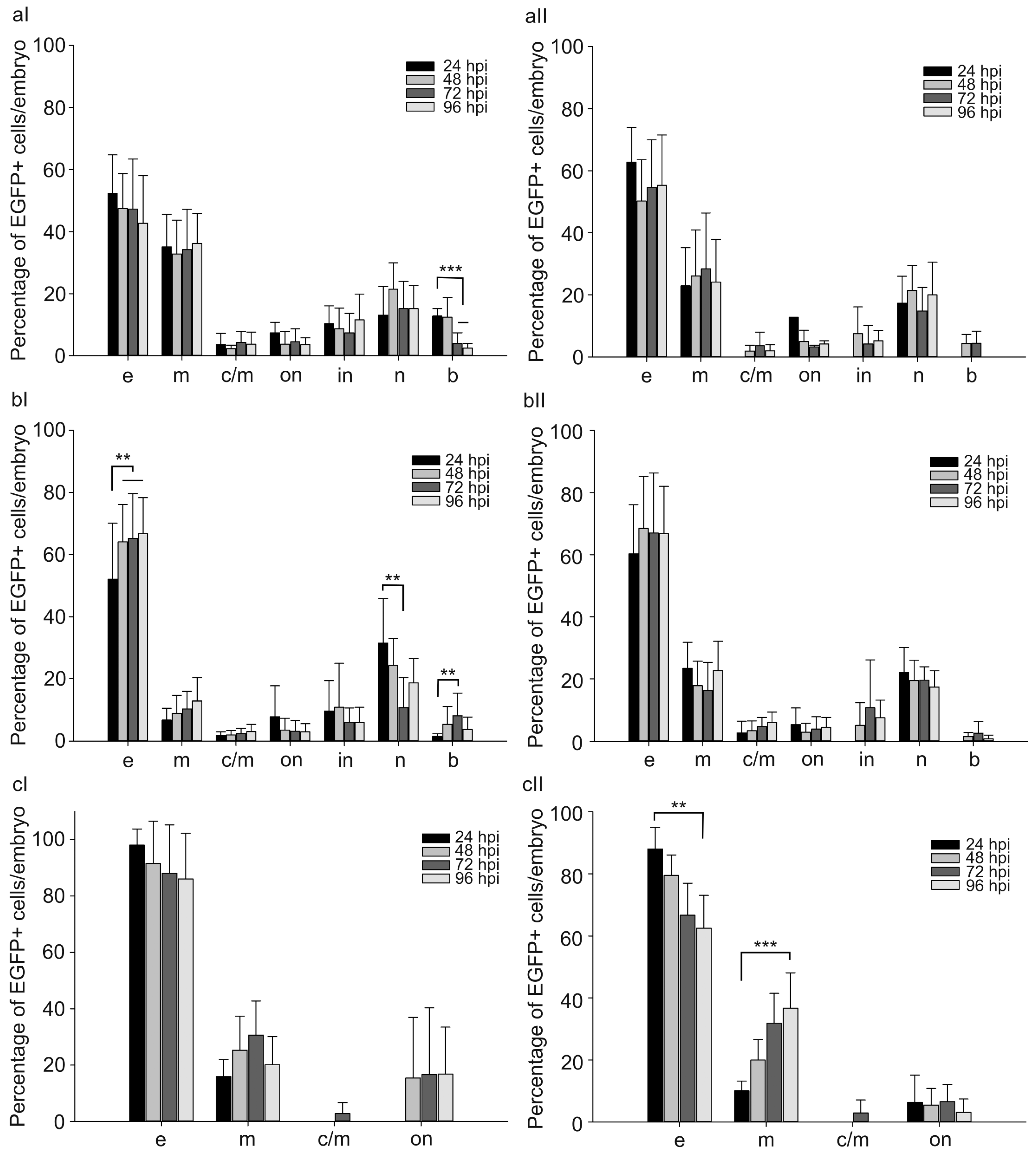
2.2. Quantitation of Embryonic Cells Expressing Marker Protein
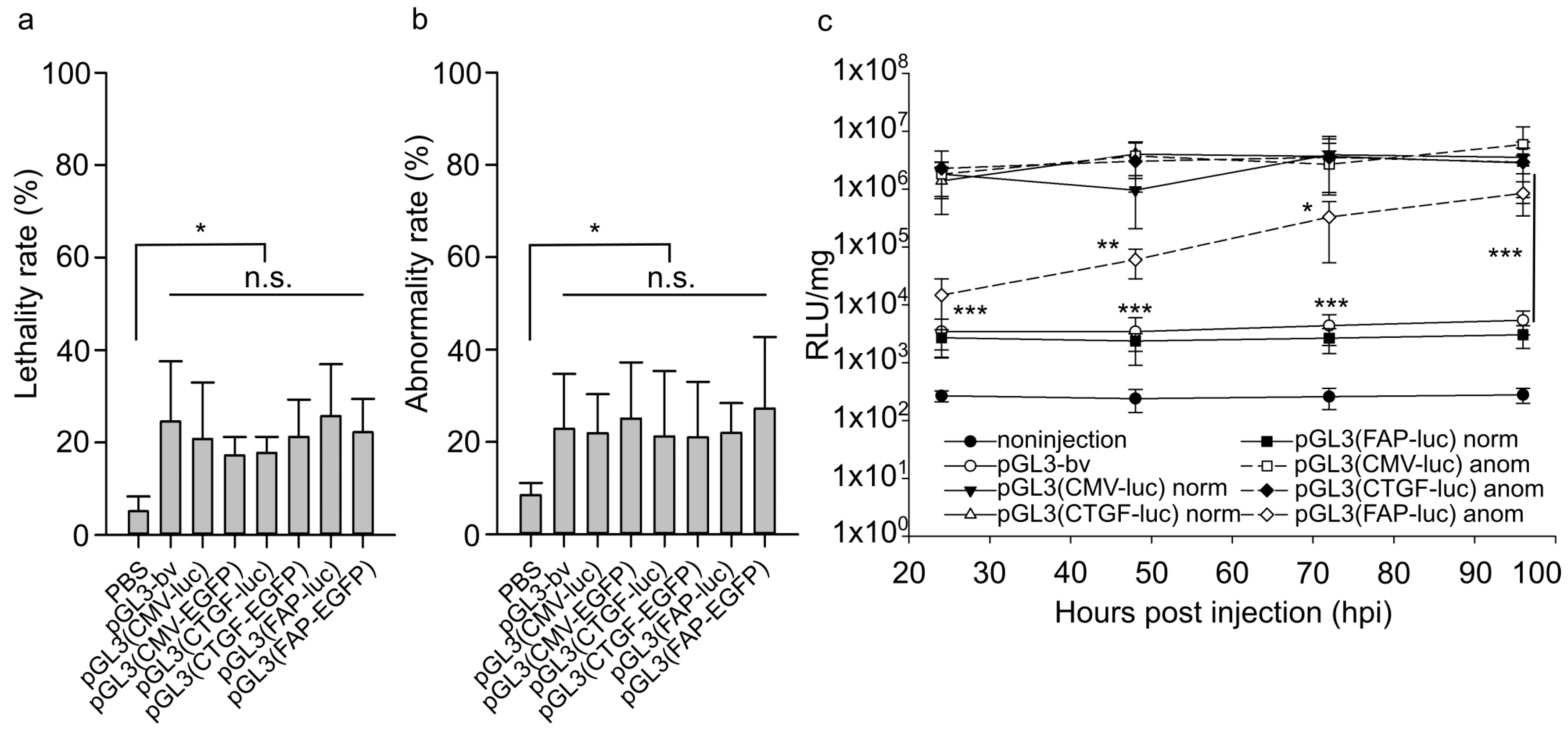
2.3. Lethality and Abnormality Rates in Developing Embryos Injected with Studied Vectors
2.4. Reporter Protein Accumulation in Abnormally Developing Injected Animals
3. Discussion
4. Materials and Methods
4.1. Genetic Constructs
4.2. Organismic Model
4.3. Vector DNA Microinjection into Danio rerio Eggs
4.4. Quantitation of Embryotoxic Effect of Vector DNA Injection into Zebrafish Embryos
4.5. Quantitation of Specific Luciferase Activity in Zebrafish Embryos
4.6. Detection of EGFP-Positive Cells in Developing Zebrafish Embryo and Bioimaging
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Denton, A.E.; Roberts, E.W.; Fearon, D.T. Stromal cells in the tumor microenvironment. Adv. Exp. Med. Biol. 2018, 1060, 99–114. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, J.; Zeng, G.; Lu, L.; Jarett, M.J.; Zhao, Y.; Yao, Q.; Chen, X.; Yu, K. Overexpression of connective tissue growth factor is associated with tumor progression and unfavorable prognosis in endometrial cancer. Cancer Biomark. 2019, 25, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Lacle, M.M.; van Diest, P.J.; Goldschmeding, R.; van der Wall, E.; Nguyen, T.Q. Expression of connective tissue growth factor in male breast cancer: Clinicopathologic correlations and prognostic value. PLoS ONE 2015, 10, e0118957. [Google Scholar] [CrossRef] [PubMed]
- Garcia, P.; Leal, P.; Alvarez, H.; Brebi, P.; Ili, C.; Tapia, O.; Roa, J.C. Connective tissue growth factor immunohistochemical expression is associated with gallbladder cancer progression. Arch Pathol. Lab. Med. 2013, 137, 245–250. [Google Scholar] [CrossRef]
- Jia, X.; Cheng, H.; Li, H.; Zhu, Y.; Li, Y.; Feng, Z.; Zhang, J. Inhibition of connective tissue growth factor overexpression decreases growth of hepatocellular carcinoma cells in vitro and in vivo. Chin. Med. J. 2011, 124, 3794–3799. [Google Scholar]
- Vlachostergios, P.J.; Karathanasis, A.; Tzortzis, V. Expression of fibroblast activation protein is enriched in neuroendocrine prostate cancer and predicts worse survival. Genes 2022, 13, 135. [Google Scholar] [CrossRef]
- Mei, S.; Zhang, Y.; Yu, L.; Chen, G.; Zi, F. Expression and role of fibroblast activation protein α in acute myeloid leukemia. Oncol. Rep. 2021, 45, 641–651. [Google Scholar] [CrossRef]
- Solano-Iturri, J.D.; Errarte, P.; Etxezarraga, M.C.; Echevarria, E.; Angulo, J.; Lopez, J.I.; Larrinaga, G. Altered Tissue and Plasma Levels of Fibroblast Activation Protein-α (FAP) in Renal Tumours. Cancers 2020, 12, 3393. [Google Scholar] [CrossRef]
- Li, M.; Cheng, X.; Rong, R.; Gao, Y.; Tang, X.; Chen, Y. High expression of fibroblast activation protein (FAP) predicts poor outcome in high-grade serous ovarian cancer. BMC Cancer 2020, 20, 1032. [Google Scholar] [CrossRef]
- Wang, X.M.; Yu, D.M.; McCaughan, G.W.; Gorrell, M.D. Fibroblast activation protein increases apoptosis, cell adhesion, and migration by the LX-2 human stellate cell line. Hepatology 2005, 42, 935–945. [Google Scholar] [CrossRef]
- Yang, W.; Han, W.; Ye, S.; Liu, D.; Wu, J.; Liu, H.; Li, C.; Chen, H. Fibroblast activation protein-α promotes ovarian cancer cell proliferation and invasion via extracellular and intracellular signaling mechanisms. Exp. Mol. Pathol. 2013, 95, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Heng, E.C.; Huang, Y.; Black, S.A.; Trackman, P.C. CCN2, connective tissue growth factor, stimulates collagen deposition by gingival fibroblasts via module 3 and alpha6- and beta1 integrins. J. Cell Biochem. 2006, 98, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.H.; Tsai, H.C.; Cheng, Y.C.; Lin, C.Y.; Huang, Y.L.; Tsai, C.H.; Xu, G.H.; Wang, S.W.; Fong, Y.C.; Tang, C.H. CTGF promotes osteosarcoma angiogenesis by regulating miR-543/angiopoietin 2 signaling. Cancer Lett. 2017, 391, 28–37. [Google Scholar] [CrossRef]
- Fan, W.H.; Karnovsky, M.J. Increased MMP-2 expression in connective tissue growth factor over-expression vascular smooth muscle cells. J. Biol. Chem. 2002, 277, 9800–9805. [Google Scholar] [CrossRef]
- Yamanaka, O.; Saika, S.; Ikeda, K.; Miyazaki, K.; Kitano, A.; Ohnishi, Y. Connective tissue growth factor modulates extracellular matrix production in human subconjunctival fibroblasts and their proliferation and migration in vitro. Jpn. J. Ophthalmol. 2008, 52, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Sun, Z.; Liu, H.; Ren, Y.; Shao, D.; Zhang, W.; Lin, J.; Wolfram, J.; Wang, F.; Nie, S. Connective tissue growth factor stimulates the proliferation, migration and differentiation of lung fibroblasts during paraquat-induced pulmonary fibrosis. Mol. Med. Rep. 2015, 12, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Ball, D.K.; Perbal, B.; Brigstock, D.R. Connective tissue growth factor induces c-fos gene activation and cell proliferation through p44/42 MAP kinase in primary rat hepatic stellate cells. J. Hepatol. 2004, 40, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Lu, X.; Wang, H. Downregulation of miR-18a induces CTGF and promotes proliferation and migration of sodium hyaluronate treated human corneal epithelial cells. Gene 2016, 591, 129–136. [Google Scholar] [CrossRef]
- Lee, C.H.; Moioli, E.K.; Mao, J.J. Fibroblastic differentiation of human mesenchymal stem cells using connective tissue growth factor. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2006, 2006, 775–778. [Google Scholar] [CrossRef]
- Kimura, T.; Monslow, J.; Klampatsa, A.; Leibowits, M.; Sun, J.; Liousia, M.; Woodruff, P.; Moon, E.; Todd, L.; Pure, E.; et al. Loss of cells expressing fibroblast activation protein has variable effects in models of TGF-β and chronic bleomycin-induced fibrosis. Am. J. Physiol. Lung. Cell Mol. Physiol. 2019, 317, L271–L282. [Google Scholar] [CrossRef]
- Mousavi, M.J.; Farhadi, E.; Vodjgani, M.; Karami, J.; Tahmasebi, M.N.; Vaziri, A.S.; Asgari, M.; Rezaei, N.; Mostafaei, S.; Jamshidi, A.; et al. Role of Fibroblast Activation Protein Alpha in Fibroblast-like Synoviocytes of Rheumatoid Arthritis. Iran. J. Allergy Asthma Immunol. 2021, 20, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Milner, J.M.; Kevorkian, L.; Young, D.A.; Jones, D.; Wait, R.; Donell, S.T.; Barksby, E.; Patterson, A.M.; Middleton, J.; Cravat, B.F.; et al. Fibroblast activation protein alpha is expressed by chondrocytes following a pro-inflammatory stimulus and is elevated in osteoarthritis. Arthritis Res. Ther. 2006, 8, R23. [Google Scholar] [CrossRef] [PubMed]
- Kawase, T.; Yasui, Y.; Nishina, S.; Hara, Y.; Yanatori, I.; Tomiyama, Y.; Nakashima, Y.; Yoshida, K.; Kishi, F.; Nakamura, M.; et al. Fibroblast activation protein-α-expressing fibroblasts promote the progression of pancreatic ductal adenocarcinoma. BMC Gastroenterol. 2015, 15, 109. [Google Scholar] [CrossRef]
- Jia, J.; Martin, T.A.; Ye, L.; Meng, L.; Xia, N.; Jiang, W.G.; Zhang, X. Fibroblast activation protein-α promotes the growth and migration of lung cancer cells via the PI3K and sonic hedgehog pathways. Int. J. Mol. Med. 2018, 41, 275–283. [Google Scholar] [CrossRef]
- Dorn, L.E.; Petrosino, J.M.; Wright, P.; Accornero, F. CTGF/CCN2 is an autocrine regulator of cardiac fibrosis. J. Mol. Cell Cardiol. 2018, 121, 205–211. [Google Scholar] [CrossRef]
- Pickles, M.; Leask, A. Analysis of CCN2 promoter activity in PANC-1 cells: Regulation by ras/MEK/ERK. J. Cell Commun. Signal. 2007, 1, 85–90. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.J.; Rhee, H.; Yoo, J.E.; Ko, J.E.; Lee, J.S.; Kim, H.; Choi, J.S.; Park, Y.N. Increased expression of CCN2, epithelial membrane antigen, and fibroblast activation protein in hepatocellular carcinoma with fibrous stroma showing aggressive behavior. PLoS ONE 2014, 9, e105094. [Google Scholar] [CrossRef] [PubMed]
- Shiwen, X.; Rajkumar, V.; Denton, C.P.; Leask, A.; Abraham, D.J. Pericytes display increased CCN2 expression upon culturing. J. Cell Commun. Signal. 2009, 3, 61–64. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, K.; Senn, B.C.; Todesco, L.; Flammer, J.; Meyer, P. Regulation of connective tissue growth factor gene expression in retinal vascular endothelial cells by angiogenic growth factors. Graefe’s Arch. Clin. Exp. Ophthalmol. 2000, 238, 910–915. [Google Scholar] [CrossRef]
- Mohmand-Borkowski, A.; Rozmysłowicz, T. Expression of fibroblast activation protein in human coronary vessels. Pol. Merkur. Lekarski. 2021, 49, 5–8. [Google Scholar]
- Eguchi, T.; Kubota, S.; Kondo, S.; Shimo, T.; Nakanishi, T.; Kuboki, T.; Yatani, H.; Takigawa, M. Regulatory mechanism of human connective tissue growth factor (CTGF/Hcs24) gene expression in a human chondrocytic cell line, HCS-2/8. J. Biochem. 2001, 130, 79–87. [Google Scholar] [CrossRef]
- Antonova, D.V.; Zinovyeva, M.V.; Kondratyeva, L.G.; Sass, A.V.; Alekseenko, I.V.; Pleshkan, V.V. Possibility for Transcriptional Targeting of Cancer-Associated Fibroblasts-Limitations and Opportunities. Int. J. Mol. Sci. 2021, 22, 3298. [Google Scholar] [CrossRef] [PubMed]
- Safina, D.R.; Selina, P.I.; Roschina, M.P.; Karaseva, M.A.; Komissarov, A.A.; Demidyuk, I.V.; Sverdlov, E.D.; Kostrov, S.V. Functional efficiency of PCR vectors in vitro and at the organism level. PLoS ONE 2020, 15, e0232045. [Google Scholar] [CrossRef] [PubMed]
- Hwang, G.; Rahman, M.A.; Razak, S.A.; Sohm, F.; Farahmand, H.; Smith, A.; Brooks, C.; Maclean, N. Isolation and characterization of tilapia beta-actin promoter and comparison of its activity with carp beta-actin promoter. Biochim. Biophys. Acta 2003, 1625, 11–18. [Google Scholar] [CrossRef]
- Tidyman, W.E.; Sehnert, A.J.; Huq, A.; Agard, J.; Deegan, F.; Stainier, D.Y.R.; Ordahl, C.P. In vivo regulation of the chicken cardiac troponin T gene promoter in zebrafish embryos. Develop. Dyn. 2003, 227, 484–496. [Google Scholar] [CrossRef]
- Li, D.; Li, G.; Wang, K.; Liu, X.; Li, W.; Chen, X.; Wang, Y. Isolation and functional analysis of the promoter of the amphioxus Hsp70a gene. Gene 2012, 510, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Westerfield, M.; Wegner, J.; Jegalian, B.G.; DeRobertis, E.M.; Püschel, A.W. Specific activation of mammalian Hox promoters in mosaic transgenic zebrafish. Genes Develop. 1992, 6, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Valianou, M.; Cheng, J.D. Identification and characterization of the promoter of fibroblast activation protein. Front. Biosci. (Elite Ed.) 2010, 2, 1154–1163. [Google Scholar] [CrossRef]
- Wenlong, L.; Leilei, Y.; Wei, F.; Yi, C.; Jing, T.; Lanzhi, M.; Bing, L.; Yang, L.; Shaoxia, W.; Qiuxia, F.; et al. Luciferase expression is driven by the promoter of fibroblast activation protein-α in murine pulmonary fibrosis. Biotechnol. Lett. 2015, 37, 1757–1763. [Google Scholar] [CrossRef]
- Tillmanns, J.; Hoffmann, D.; Habbaba, Y.; Schmitto, J.D.; Sedding, D.; Fraccarollo, D.; Galuppo, P.; Bauersachs, J. Fibroblast activation protein alpha expression identifies activated fibroblasts after myocardial infarction. J. Mol. Cell Cardiol. 2015, 87, 194–203. [Google Scholar] [CrossRef]
- Segerer, S.E.; Bartmann, C.; Schwab, M.; Kämmerer, U. Expression of the Peptidase “Fibroblast Activation Protein” on Decidual Stromal Cells Facilitating Tissue Remodeling. Gynecol. Obstet. Investig. 2020, 85, 428–436. [Google Scholar] [CrossRef]
- Niedermeyer, J.; Garin-Chesa, P.; Kriz, M.; Hilberg, F.; Mueller, E.; Bamberger, U.; Retting, W.J.; Schnapp, A. Expression of the fibroblast activation protein during mouse embryo development. Int. J. Develop. Biol. 2001, 45, 445–447. [Google Scholar] [PubMed]
- Sambrook, J.; Russell, D.W. Molecular Cloning in A laboratory Manual, 3rd ed.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- Selina, P.I.; Karaseva, M.A.; Komissarov, A.A.; Safina, D.R.; Lunina, N.A.; Roschina, M.P.; Sverdlov, E.D.; Demidyuk, I.V.; Kostrov, S.V. Embryotoxic activity of 3C protease of human hepatitis A virus in developing Danio rerio embryos. Sci. Rep. 2021, 11, 18196. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1979, 72, 248–254. [Google Scholar] [CrossRef]
- Gasparov, V.S.; Degtiar’, V.G. Protein determination by binding with the dye Coomassie brilliant blue G250. Biokhimiia 1994, 59, 763–777. [Google Scholar]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selina, P.I.; Alekseenko, I.V.; Kurtova, A.I.; Pleshkan, V.V.; Voronezhskaya, E.E.; Demidyuk, I.V.; Kostrov, S.V. Efficiency of Promoters of Human Genes FAP and CTGF at Organism Level in a Danio rerio Model. Int. J. Mol. Sci. 2023, 24, 7192. https://doi.org/10.3390/ijms24087192
Selina PI, Alekseenko IV, Kurtova AI, Pleshkan VV, Voronezhskaya EE, Demidyuk IV, Kostrov SV. Efficiency of Promoters of Human Genes FAP and CTGF at Organism Level in a Danio rerio Model. International Journal of Molecular Sciences. 2023; 24(8):7192. https://doi.org/10.3390/ijms24087192
Chicago/Turabian StyleSelina, Polina I., Irina V. Alekseenko, Anastasia I. Kurtova, Victor V. Pleshkan, Elena E. Voronezhskaya, Ilya V. Demidyuk, and Sergey V. Kostrov. 2023. "Efficiency of Promoters of Human Genes FAP and CTGF at Organism Level in a Danio rerio Model" International Journal of Molecular Sciences 24, no. 8: 7192. https://doi.org/10.3390/ijms24087192
APA StyleSelina, P. I., Alekseenko, I. V., Kurtova, A. I., Pleshkan, V. V., Voronezhskaya, E. E., Demidyuk, I. V., & Kostrov, S. V. (2023). Efficiency of Promoters of Human Genes FAP and CTGF at Organism Level in a Danio rerio Model. International Journal of Molecular Sciences, 24(8), 7192. https://doi.org/10.3390/ijms24087192






