Ca2+-Activated K+ Channels in Progenitor Cells of Musculoskeletal Tissues: A Narrative Review
Abstract
1. Introduction
1.1. Progenitor Cells in Musculoskeletal Tissues
1.2. Musculoskeletal Diseases
2. Ion Channels Involved in Progenitor Cell Differentiation
2.1. Ion Channels That Mediate Chondrogenesis
2.1.1. Calcium Signalling Pathways
2.1.2. Potassium Channels
2.1.3. Sodium Channels
2.1.4. Chloride Channels
2.1.5. Aquaporin Water Channels
2.2. Altered Ionic Homeostasis in Chondrocytes of OA-Afflicted Cartilage
2.3. Ion Channels That Mediate Osteogenesis
2.4. Ion Channels That Mediate Muscle Differentiation
3. Calcium-Dependent Potassium Channels
3.1. Calcium-Dependent Potassium Channels in MSCs, CPCs, and Chondrocytes: Expression and Function
3.1.1. KCa Channels in MSCs
3.1.2. KCa Channels in CPCs
3.1.3. KCa Channels in Chondrocytes
3.2. Calcium-Dependent Potassium Channels in CPCs and Osteocytes: Expression and Function
3.3. Calcium-Dependent Potassium Channels in Muscle Progenitor Cells and Myogenesis: Expression and Function
3.4. Calcium-Dependent Potassium Channels in Cells of the Synovium: Expression and Function
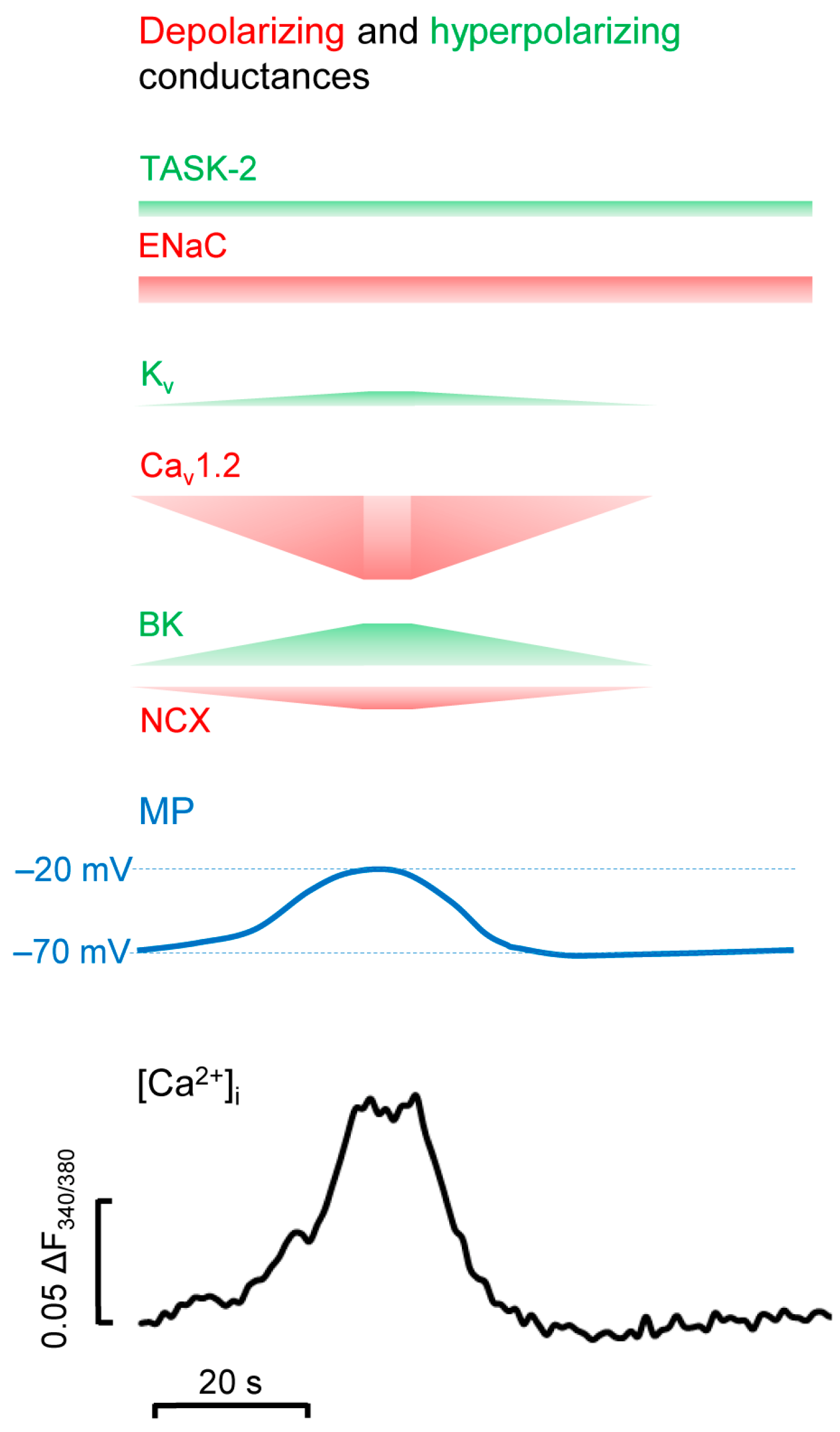
4. BK Channels in Calcium Oscillations in CPCs
5. KCa Channels in Musculoskeletal Pathologies
6. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nguyen, H.Q.; Callegari, E.; Obach, R.S. The Use of In Vitro Data and Physiologically-Based Pharmacokinetic Modeling to Predict Drug Metabolite Exposure: Desipramine Exposure in Cytochrome P4502D6 Extensive and Poor Metabolizers Following Administration of Imipramine. Drug Metab. Dispos. 2016, 44, 1569–1578. [Google Scholar] [CrossRef]
- Bruno Garza, J.L.; Eijckelhof, B.H.; Johnson, P.W.; Raina, S.M.; Rynell, P.W.; Huysmans, M.A.; van Dieen, J.H.; van der Beek, A.J.; Blatter, B.M.; Dennerlein, J.T. Observed differences in upper extremity forces, muscle efforts, postures, velocities and accelerations across computer activities in a field study of office workers. Ergonomics 2012, 55, 670–681. [Google Scholar] [CrossRef] [PubMed]
- Roshdy, H.S.; Soliman, M.H.; El-Dosouky, I.I.; Ghonemy, S. Skin aging parameters: A window to heart block. Clin. Cardiol. 2018, 41, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Balat, A. From past to present: Traditional herbs used in the treatment of nephrologic diseases in southeast Turkey. J. Nephrol. 2013, 26, 187–191. [Google Scholar] [PubMed]
- Sun, Y.; Wan, B.; Wang, R.; Zhang, B.; Luo, P.; Wang, D.; Nie, J.J.; Chen, D.; Wu, X. Mechanical Stimulation on Mesenchymal Stem Cells and Surrounding Microenvironments in Bone Regeneration: Regulations and Applications. Front. Cell Dev. Biol. 2022, 10, 808303. [Google Scholar] [CrossRef] [PubMed]
- Camernik, K.; Barlic, A.; Drobnic, M.; Marc, J.; Jeras, M.; Zupan, J. Mesenchymal Stem Cells in the Musculoskeletal System: From Animal Models to Human Tissue Regeneration? Stem Cell Rev. Rep. 2018, 14, 346–369. [Google Scholar] [CrossRef]
- Li, W.J.; Jiao, H.; Walczak, B.E. Emerging opportunities for induced pluripotent stem cells in orthopaedics. J. Orthop. Translat. 2019, 17, 73–81. [Google Scholar] [CrossRef]
- Liu, S.; Wang, B.; Fan, S.; Wang, Y.; Zhan, Y.; Ye, D. Global burden of musculoskeletal disorders and attributable factors in 204 countries and territories: A secondary analysis of the Global Burden of Disease 2019 study. BMJ Open 2022, 12, e062183. [Google Scholar] [CrossRef]
- Kyu, H.H.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national disability-adjusted life-years (DALYs) for 359 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1859–1922. [Google Scholar] [CrossRef]
- Angelini, F.; Widera, P.; Mobasheri, A.; Blair, J.; Struglics, A.; Uebelhoer, M.; Henrotin, Y.; Marijnissen, A.C.; Kloppenburg, M.; Blanco, F.J.; et al. Osteoarthritis endotype discovery via clustering of biochemical marker data. Ann. Rheum. Dis. 2022, 81, 666–675. [Google Scholar] [CrossRef]
- Lachiewicz, P.F.; Soileau, E.S. Ten-year survival and clinical results of constrained components in primary total knee arthroplasty. J. Arthroplast. 2006, 21, 803–808. [Google Scholar] [CrossRef] [PubMed]
- Chimutengwende-Gordon, M.; Donaldson, J.; Bentley, G. Current solutions for the treatment of chronic articular cartilage defects in the knee. EFORT Open Rev. 2020, 5, 156–163. [Google Scholar] [CrossRef]
- Andrade, R.; Vasta, S.; Pereira, R.; Pereira, H.; Papalia, R.; Karahan, M.; Oliveira, J.M.; Reis, R.L.; Espregueira-Mendes, J. Knee donor-site morbidity after mosaicplasty—A systematic review. J. Exp. Orthop. 2016, 3, 31. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, Y.; Mukai, S.; Setoguchi, Y.; Goto, T.; Furukawa, T.; Nakamura, T. Clinical Outcomes of Donor Sites After Osteochondral Graft Harvest from Healthy Knees. Orthop. J. Sport. Med. 2017, 5, 2325967117732525. [Google Scholar] [CrossRef] [PubMed]
- Maumus, M.; Pers, Y.M.; Ruiz, M.; Jorgensen, C.; Noel, D. Mesenchymal stem cells and regenerative medicine: Future perspectives in osteoarthritis. Med. Sci. 2018, 34, 1092–1099. [Google Scholar] [CrossRef]
- Barry, F. MSC Therapy for Osteoarthritis: An Unfinished Story. J. Orthop. Res. 2019, 37, 1229–1235. [Google Scholar] [CrossRef]
- Adams, M.E.; Huang, D.Q.; Yao, L.Y.; Sandell, L.J. Extraction and isolation of mRNA from adult articular cartilage. Anal. Biochem. 1992, 202, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Asmar, A.; Barrett-Jolley, R.; Werner, A.; Kelly, R., Jr.; Stacey, M. Membrane channel gene expression in human costal and articular chondrocytes. Organogenesis 2016, 12, 94–107. [Google Scholar] [CrossRef]
- Barrett-Jolley, R.; Lewis, R.; Fallman, R.; Mobasheri, A. The emerging chondrocyte channelome. Front. Physiol. 2010, 1, 135. [Google Scholar] [CrossRef]
- Mobasheri, A.; Matta, C.; Uzieliene, I.; Budd, E.; Martin-Vasallo, P.; Bernotiene, E. The chondrocyte channelome: A narrative review. Jt. Bone Spine 2019, 86, 29–35. [Google Scholar] [CrossRef]
- Fellows, C.R.; Matta, C.; Zakany, R.; Khan, I.M.; Mobasheri, A. Adipose, Bone Marrow and Synovial Joint-Derived Mesenchymal Stem Cells for Cartilage Repair. Front. Genet. 2016, 7, 213. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.M.; Kalamegam, G.; Pushparaj, P.N.; Matta, C.; Memic, A.; Khademhosseini, A.; Mobasheri, R.; Poletti, F.L.; Hoyland, J.A.; Mobasheri, A. Mesenchymal stem cells in regenerative medicine: Focus on articular cartilage and intervertebral disc regeneration. Methods 2016, 99, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Matta, C.; Lewis, R.; Fellows, C.; Diszhazi, G.; Almassy, J.; Miosge, N.; Dixon, J.; Uribe, M.C.; May, S.; Poliska, S.; et al. Transcriptome-based screening of ion channels and transporters in a migratory chondroprogenitor cell line isolated from late-stage osteoarthritic cartilage. J. Cell. Physiol. 2021, 236, 7421–7439. [Google Scholar] [CrossRef] [PubMed]
- Matta, C.; Boocock, D.J.; Fellows, C.R.; Miosge, N.; Dixon, J.E.; Liddell, S.; Smith, J.; Mobasheri, A. Molecular phenotyping of the surfaceome of migratory chondroprogenitors and mesenchymal stem cells using biotinylation, glycocapture and quantitative LC-MS/MS proteomic analysis. Sci. Rep. 2019, 9, 9018. [Google Scholar] [CrossRef]
- Matta, C.; Zakany, R. Calcium signalling in chondrogenesis: Implications for cartilage repair. Front. Biosci. 2013, 5, 305–324. [Google Scholar] [CrossRef]
- Zhang, M.; Che, C.; Cheng, J.; Li, P.; Yang, Y. Ion channels in stem cells and their roles in stem cell biology and vascular diseases. J. Mol. Cell. Cardiol. 2022, 166, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Muramatsu, S.; Wakabayashi, M.; Ohno, T.; Amano, K.; Ooishi, R.; Sugahara, T.; Shiojiri, S.; Tashiro, K.; Suzuki, Y.; Nishimura, R.; et al. Functional gene screening system identified TRPV4 as a regulator of chondrogenic differentiation. J. Biol. Chem. 2007, 282, 32158–32167. [Google Scholar] [CrossRef]
- Somogyi, C.S.; Matta, C.; Foldvari, Z.; Juhasz, T.; Katona, E.; Takacs, A.R.; Hajdu, T.; Dobrosi, N.; Gergely, P.; Zakany, R. Polymodal Transient Receptor Potential Vanilloid (TRPV) Ion Channels in Chondrogenic Cells. Int. J. Mol. Sci. 2015, 16, 18412–18438. [Google Scholar] [CrossRef]
- Guilak, F.; Leddy, H.A.; Liedtke, W. Transient receptor potential vanilloid 4: The sixth sense of the musculoskeletal system? Ann. N. Y. Acad. Sci. 2010, 1192, 404–409. [Google Scholar] [CrossRef]
- Willard, V.P.; Leddy, H.A.; Palmer, D.; Wu, C.L.; Liedtke, W.; Guilak, F. Transient receptor potential vanilloid 4 as a regulator of induced pluripotent stem cell chondrogenesis. Stem Cells 2021, 39, 1447–1456. [Google Scholar] [CrossRef]
- Nasrollahzadeh, N.; Karami, P.; Wang, J.; Bagheri, L.; Guo, Y.; Abdel-Sayed, P.; Laurent-Applegate, L.; Pioletti, D.P. Temperature evolution following joint loading promotes chondrogenesis by synergistic cues via calcium signaling. eLife 2022, 11, e72068. [Google Scholar] [CrossRef]
- Zhang, Z.; Sa, G.; Wang, Z.; Wei, Z.; Zheng, L.; Zhang, R.; Zhu, X.; Yang, X. Piezo1 and IFT88 synergistically regulate mandibular condylar chondrocyte differentiation under cyclic tensile strain. Tissue Cell 2022, 76, 101781. [Google Scholar] [CrossRef] [PubMed]
- Ramage, L.; Martel, M.A.; Hardingham, G.E.; Salter, D.M. NMDA receptor expression and activity in osteoarthritic human articular chondrocytes. Osteoarthr. Cartil. 2008, 16, 1576–1584. [Google Scholar] [CrossRef] [PubMed]
- Salter, D.M.; Wright, M.O.; Millward-Sadler, S.J. NMDA receptor expression and roles in human articular chondrocyte mechanotransduction. Biorheology 2004, 41, 273–281. [Google Scholar] [PubMed]
- Alhilali, M.; Hearn, J.I.; Rong, J.; Jain, L.; Bolam, S.M.; Monk, A.P.; Munro, J.T.; Dalbeth, N.; Poulsen, R.C. IL-1beta induces changes in expression of core circadian clock components PER2 and BMAL1 in primary human chondrocytes through the NMDA receptor/CREB and NF-kappaB signalling pathways. Cell. Signal. 2021, 87, 110143. [Google Scholar] [CrossRef]
- Kalev-Zylinska, M.L.; Hearn, J.I.; Rong, J.; Zhu, M.; Munro, J.; Cornish, J.; Dalbeth, N.; Poulsen, R.C. Altered N-methyl D-aspartate receptor subunit expression causes changes to the circadian clock and cell phenotype in osteoarthritic chondrocytes. Osteoarthr. Cartil. 2018, 26, 1518–1530. [Google Scholar] [CrossRef]
- Matta, C.; Juhasz, T.; Fodor, J.; Hajdu, T.; Katona, E.; Szucs-Somogyi, C.; Takacs, R.; Vago, J.; Olah, T.; Bartok, A.; et al. N-methyl-D-aspartate (NMDA) receptor expression and function is required for early chondrogenesis. Cell Commun. Signal. 2019, 17, 166. [Google Scholar] [CrossRef]
- Fodor, J.; Matta, C.; Olah, T.; Juhasz, T.; Takacs, R.; Toth, A.; Dienes, B.; Csernoch, L.; Zakany, R. Store-operated calcium entry and calcium influx via voltage-operated calcium channels regulate intracellular calcium oscillations in chondrogenic cells. Cell Calcium 2013, 54, 1–16. [Google Scholar] [CrossRef]
- Matta, C.; Zakany, R.; Mobasheri, A. Voltage-dependent calcium channels in chondrocytes: Roles in health and disease. Curr. Rheumatol. Rep. 2015, 17, 43. [Google Scholar] [CrossRef]
- Atsuta, Y.; Tomizawa, R.R.; Levin, M.; Tabin, C.J. L-type voltage-gated Ca2+ channel CaV1.2 regulates chondrogenesis during limb development. Proc. Natl. Acad. Sci. USA 2019, 116, 21592–21601. [Google Scholar] [CrossRef]
- Steward, A.J.; Kelly, D.J.; Wagner, D.R. The role of calcium signalling in the chondrogenic response of mesenchymal stem cells to hydrostatic pressure. Eur. Cells Mater. 2014, 28, 358–371. [Google Scholar] [CrossRef]
- Matta, C.; Fodor, J.; Miosge, N.; Takacs, R.; Juhasz, T.; Rybaltovszki, H.; Toth, A.; Csernoch, L.; Zakany, R. Purinergic signalling is required for calcium oscillations in migratory chondrogenic progenitor cells. Pflug. Arch. 2015, 467, 429–442. [Google Scholar] [CrossRef]
- Fodor, J.; Matta, C.; Juhasz, T.; Olah, T.; Gonczi, M.; Szijgyarto, Z.; Gergely, P.; Csernoch, L.; Zakany, R. Ionotropic purinergic receptor P2X4 is involved in the regulation of chondrogenesis in chicken micromass cell cultures. Cell Calcium 2009, 45, 421–430. [Google Scholar] [CrossRef]
- Meyer, M.P.; Swann, K.; Burnstock, G.; Clarke, J.D. The extracellular ATP receptor, cP2Y1, inhibits cartilage formation in micromass cultures of chick limb mesenchyme. Dev. Dyn. 2001, 222, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.B.; Hatano, N.; Kondo, C.; Belke, D.D.; Brown, B.S.; Kumar, S.; Votta, B.J.; Giles, W.R. Voltage-gated K+ currents in mouse articular chondrocytes regulate membrane potential. Channels 2010, 4, 179–191. [Google Scholar] [CrossRef]
- Mobasheri, A.; Gent, T.C.; Womack, M.D.; Carter, S.D.; Clegg, P.D.; Barrett-Jolley, R. Quantitative analysis of voltage-gated potassium currents from primary equine (Equus caballus) and elephant (Loxodonta africana) articular chondrocytes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005, 289, R172–R180. [Google Scholar] [CrossRef] [PubMed]
- Mobasheri, A.; Gent, T.C.; Nash, A.I.; Womack, M.D.; Moskaluk, C.A.; Barrett-Jolley, R. Evidence for functional ATP-sensitive (KATP) potassium channels in human and equine articular chondrocytes. Osteoarthr. Cartil. 2007, 15, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Mobasheri, A.; Lewis, R.; Maxwell, J.E.; Hill, C.; Womack, M.; Barrett-Jolley, R. Characterization of a stretch-activated potassium channel in chondrocytes. J. Cell. Physiol. 2010, 223, 511–518. [Google Scholar] [CrossRef]
- Wright, M.; Jobanputra, P.; Bavington, C.; Salter, D.M.; Nuki, G. Effects of intermittent pressure-induced strain on the electrophysiology of cultured human chondrocytes: Evidence for the presence of stretch-activated membrane ion channels. Clin. Sci. 1996, 90, 61–71. [Google Scholar] [CrossRef]
- Varga, Z.; Juhasz, T.; Matta, C.; Fodor, J.; Katona, E.; Bartok, A.; Olah, T.; Sebe, A.; Csernoch, L.; Panyi, G.; et al. Switch of voltage-gated K+ channel expression in the plasma membrane of chondrogenic cells affects cytosolic Ca2+-oscillations and cartilage formation. PLoS ONE 2011, 6, e27957. [Google Scholar] [CrossRef]
- Suzuki, Y.; Yamamura, H.; Imaizumi, Y.; Clark, R.B.; Giles, W.R. K+ and Ca2+ Channels Regulate Ca2+ Signaling in Chondrocytes: An Illustrated Review. Cells 2020, 9, 1577. [Google Scholar] [CrossRef]
- Pini, J.; Giuliano, S.; Matonti, J.; Gannoun, L.; Simkin, D.; Rouleau, M.; Bendahhou, S. Osteogenic and Chondrogenic Master Genes Expression Is Dependent on the Kir2.1 Potassium Channel Through the Bone Morphogenetic Protein Pathway. J. Bone Miner. Res. 2018, 33, 1826–1841. [Google Scholar] [CrossRef] [PubMed]
- Kachroo, U.; Livingston, A.; Vinod, E.; Sathishkumar, S.; Boopalan, P. Comparison of Electrophysiological Properties and Gene Expression between Human Chondrocytes and Chondroprogenitors Derived from Normal and Osteoarthritic Cartilage. Cartilage 2020, 11, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, T.; Yoshino, M.; Nagao, M.; Ishii, S.; Yabu, H. Voltage-gated ionic channels in cultured rabbit articular chondrocytes. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1996, 115, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Duan, Y.; Duan, X. Chloride channels regulate chondrogenesis in chicken mandibular mesenchymal cells. Arch. Oral Biol. 2010, 55, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Graziano, A.C.E.; Avola, R.; Pannuzzo, G.; Cardile, V. Aquaporin1 and 3 modification as a result of chondrogenic differentiation of human mesenchymal stem cell. J. Cell. Physiol. 2018, 233, 2279–2291. [Google Scholar] [CrossRef]
- Kotelsky, A.; Carrier, J.S.; Aggouras, A.; Richards, M.S.; Buckley, M.R. Evidence that reduction in volume protects in situ articular chondrocytes from mechanical impact. Connect. Tissue Res. 2020, 61, 360–374. [Google Scholar] [CrossRef]
- Hall, A.C. The Role of Chondrocyte Morphology and Volume in Controlling Phenotype-Implications for Osteoarthritis, Cartilage Repair, and Cartilage Engineering. Curr. Rheumatol. Rep. 2019, 21, 38. [Google Scholar] [CrossRef]
- Lewis, R.; Feetham, C.H.; Barrett-Jolley, R. Cell volume regulation in chondrocytes. Cell. Physiol. Biochem. 2011, 28, 1111–1122. [Google Scholar] [CrossRef]
- Manzano, S.; Manzano, R.; Doblare, M.; Doweidar, M.H. Altered swelling and ion fluxes in articular cartilage as a biomarker in osteoarthritis and joint immobilization: A computational analysis. J. R. Soc. Interface 2015, 12, 20141090. [Google Scholar] [CrossRef]
- Gao, W.; Hasan, H.; Anderson, D.E.; Lee, W. The Role of Mechanically-Activated Ion Channels Piezo1, Piezo2, and TRPV4 in Chondrocyte Mechanotransduction and Mechano-Therapeutics for Osteoarthritis. Front. Cell Dev. Biol. 2022, 10, 885224. [Google Scholar] [CrossRef]
- Lewis, R.; Barrett-Jolley, R. Changes in Membrane Receptors and Ion Channels as Potential Biomarkers for Osteoarthritis. Front. Physiol. 2015, 6, 357. [Google Scholar] [CrossRef]
- Jeremiasse, B.; Matta, C.; Fellows, C.R.; Boocock, D.J.; Smith, J.R.; Liddell, S.; Lafeber, F.; van Spil, W.E.; Mobasheri, A. Alterations in the chondrocyte surfaceome in response to pro-inflammatory cytokines. BMC Mol. Cell Biol. 2020, 21, 47. [Google Scholar] [CrossRef] [PubMed]
- Kito, H.; Ohya, S. Role of K+ and Ca2+-Permeable Channels in Osteoblast Functions. Int. J. Mol. Sci. 2021, 22, 10459. [Google Scholar] [CrossRef] [PubMed]
- Savadipour, A.; Palmer, D.; Ely, E.V.; Collins, K.H.; Garcia-Castorena, J.M.; Harissa, Z.; Kim, Y.S.; Oestreich, A.; Qu, F.; Rashidi, N.; et al. The Role of PIEZO Ion Channels in the Musculoskeletal System. Am. J. Physiol. Cell Physiol. 2023, 324, C728–C740. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, M.; Suzuki, H.; Hatano, N.; Nakano, S.; Muraki, Y.; Miyazawa, K.; Goto, S.; Muraki, K. PIEZO1 and TRPV4, which Are Distinct Mechano-Sensors in the Osteoblastic MC3T3-E1 Cells, Modify Cell-Proliferation. Int. J. Mol. Sci. 2019, 20, 4960. [Google Scholar] [CrossRef]
- Liu, Y.S.; Liu, Y.A.; Huang, C.J.; Yen, M.H.; Tseng, C.T.; Chien, S.; Lee, O.K. Mechanosensitive TRPM7 mediates shear stress and modulates osteogenic differentiation of mesenchymal stromal cells through Osterix pathway. Sci. Rep. 2015, 5, 16522. [Google Scholar] [CrossRef]
- Xiao, E.; Yang, H.Q.; Gan, Y.H.; Duan, D.H.; He, L.H.; Guo, Y.; Wang, S.Q.; Zhang, Y. Brief reports: TRPM7 Senses mechanical stimulation inducing osteogenesis in human bone marrow mesenchymal stem cells. Stem Cells 2015, 33, 615–621. [Google Scholar] [CrossRef]
- Yao, H.; Zhang, L.; Yan, S.; He, Y.; Zhu, H.; Li, Y.; Wang, D.; Yang, K. Low-intensity pulsed ultrasound/nanomechanical force generators enhance osteogenesis of BMSCs through microfilaments and TRPM7. J. Nanobiotechnol. 2022, 20, 378. [Google Scholar] [CrossRef]
- Shin, M.; Mori, S.; Mizoguchi, T.; Arai, A.; Kajiya, H.; Okamoto, F.; Bartlett, J.D.; Matsushita, M.; Udagawa, N.; Okabe, K. Mesenchymal cell TRPM7 expression is required for bone formation via the regulation of chondrogenesis. Bone 2023, 166, 116579. [Google Scholar] [CrossRef]
- Shao, Y.; Alicknavitch, M.; Farach-Carson, M.C. Expression of voltage sensitive calcium channel (VSCC) L-type Cav1.2 (α1C) and T-type Cav3.2 (α1H) subunits during mouse bone development. Dev. Dyn. 2005, 234, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Nagai, S.; Kitamura, K.; Kimura, M.; Yamamoto, H.; Katakura, A.; Shibukawa, Y. Functional Expression of Mechanosensitive Piezo1/TRPV4 Channels in Mouse Osteoblasts. Bull. Tokyo Dent. Coll. 2023, 64, 1–11. [Google Scholar] [CrossRef]
- Orriss, I.R.; Davies, B.K.; Bourne, L.E.; Arnett, T.R. Modulation of osteoblast differentiation and function by the P2X4 receptor. Purinergic Signal. 2022. [Google Scholar] [CrossRef] [PubMed]
- Bergamin, L.S.; Penolazzi, L.; Lambertini, E.; Falzoni, S.; Sarti, A.C.; Molle, C.M.; Gendron, F.P.; De Bonis, P.; Di Virgilio, F.; Piva, R. Expression and function of the P2X7 receptor in human osteoblasts: The role of NFATc1 transcription factor. J. Cell. Physiol. 2021, 236, 641–652. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Arredondo, H.M.; Wang, N. P2Y Receptors in Bone—Anabolic, Catabolic, or Both? Front. Endocrinol. 2021, 12, 818499. [Google Scholar] [CrossRef] [PubMed]
- Alvarenga, E.C.; Rodrigues, R.; Caricati-Neto, A.; Silva-Filho, F.C.; Paredes-Gamero, E.J.; Ferreira, A.T. Low-intensity pulsed ultrasound-dependent osteoblast proliferation occurs by via activation of the P2Y receptor: Role of the P2Y1 receptor. Bone 2010, 46, 355–362. [Google Scholar] [CrossRef]
- Hoebertz, A.; Meghji, S.; Burnstock, G.; Arnett, T.R. Extracellular ADP is a powerful osteolytic agent: Evidence for signaling through the P2Y1 receptor on bone cells. FASEB J. 2001, 15, 1139–1148. [Google Scholar] [CrossRef]
- Li, W.; Wei, S.; Liu, C.; Song, M.; Wu, H.; Yang, Y. Regulation of the osteogenic and adipogenic differentiation of bone marrow-derived stromal cells by extracellular uridine triphosphate: The role of P2Y2 receptor and ERK1/2 signaling. Int. J. Mol. Med. 2016, 37, 63–73. [Google Scholar] [CrossRef]
- Orriss, I.R.; Knight, G.E.; Ranasinghe, S.; Burnstock, G.; Arnett, T.R. Osteoblast responses to nucleotides increase during differentiation. Bone 2006, 39, 300–309. [Google Scholar] [CrossRef]
- Orriss, I.R.; Guneri, D.; Hajjawi, M.O.R.; Shaw, K.; Patel, J.J.; Arnett, T.R. Activation of the P2Y2 receptor regulates bone cell function by enhancing ATP release. J. Endocrinol. 2017, 233, 341–356. [Google Scholar] [CrossRef]
- Orriss, I.R.; Wang, N.; Burnstock, G.; Arnett, T.R.; Gartland, A.; Robaye, B.; Boeynaems, J.M. The P2Y6 receptor stimulates bone resorption by osteoclasts. Endocrinology 2011, 152, 3706–3716. [Google Scholar] [CrossRef] [PubMed]
- Korcok, J.; Raimundo, L.N.; Du, X.; Sims, S.M.; Dixon, S.J. P2Y6 nucleotide receptors activate NF-κB and increase survival of osteoclasts. J. Biol. Chem. 2005, 280, 16909–16915. [Google Scholar] [CrossRef] [PubMed]
- Noronha-Matos, J.B.; Costa, M.A.; Magalhaes-Cardoso, M.T.; Ferreirinha, F.; Pelletier, J.; Freitas, R.; Neves, J.M.; Sevigny, J.; Correia-de-Sa, P. Role of ecto-NTPDases on UDP-sensitive P2Y6 receptor activation during osteogenic differentiation of primary bone marrow stromal cells from postmenopausal women. J. Cell. Physiol. 2012, 227, 2694–2709. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Riquelme, M.A.; Tian, Y.; Zhao, D.; Acosta, F.M.; Gu, S.; Jiang, J.X. ATP Inhibits Breast Cancer Migration and Bone Metastasis through Down-Regulation of CXCR4 and Purinergic Receptor P2Y11. Cancers 2021, 13, 4293. [Google Scholar] [CrossRef]
- Biver, G.; Wang, N.; Gartland, A.; Orriss, I.; Arnett, T.R.; Boeynaems, J.M.; Robaye, B. Role of the P2Y13 receptor in the differentiation of bone marrow stromal cells into osteoblasts and adipocytes. Stem Cells 2013, 31, 2747–2758. [Google Scholar] [CrossRef]
- Lee, S.A.; Park, J.H.; Lee, S.Y. Selective induction of P2Y14 receptor by RANKL promotes osteoclast formation. Mol. Cells 2013, 36, 273–277. [Google Scholar] [CrossRef]
- Mikolajewicz, N.; Komarova, S.V. Role of UDP-Sugar Receptor P2Y14 in Murine Osteoblasts. Int. J. Mol. Sci. 2020, 21, 2747. [Google Scholar] [CrossRef]
- Syberg, S.; Brandao-Burch, A.; Patel, J.J.; Hajjawi, M.; Arnett, T.R.; Schwarz, P.; Jorgensen, N.R.; Orriss, I.R. Clopidogrel (Plavix), a P2Y12 receptor antagonist, inhibits bone cell function in vitro and decreases trabecular bone in vivo. J. Bone Miner. Res. 2012, 27, 2373–2386. [Google Scholar] [CrossRef]
- Duan, X.Q.; Li, Y.H.; Zhang, X.Y.; Zhao, Z.T.; Wang, Y.; Wang, H.; Li, G.S.; Jing, L. Mechanisms of Intracellular Calcium Homeostasis in MC3T3-E1 Cells and Bone Tissues of Sprague-Dawley Rats Exposed to Fluoride. Biol. Trace Elem. Res. 2016, 170, 331–339. [Google Scholar] [CrossRef]
- Huang, X.; Li, Z.; Liu, P.; Wu, M.; Liu, A.Q.; Hu, C.; Liu, X.; Guo, H.; Yang, X.; Guo, X.; et al. Gli1+ Cells Residing in Bone Sutures Respond to Mechanical Force via IP3R to Mediate Osteogenesis. Stem Cells Int. 2021, 2021, 8138374. [Google Scholar] [CrossRef]
- Volodarsky, M.; Markus, B.; Cohen, I.; Staretz-Chacham, O.; Flusser, H.; Landau, D.; Shelef, I.; Langer, Y.; Birk, O.S. A deletion mutation in TMEM38B associated with autosomal recessive osteogenesis imperfecta. Hum. Mutat. 2013, 34, 582–586. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Ichimura, A.; Qian, N.; Iida, T.; Yamazaki, D.; Noma, N.; Asagiri, M.; Yamamoto, K.; Komazaki, S.; Sato, C.; et al. Mice lacking the intracellular cation channel TRIC-B have compromised collagen production and impaired bone mineralization. Sci. Signal. 2016, 9, ra49. [Google Scholar] [CrossRef] [PubMed]
- Mikolajewicz, N.; Smith, D.; Komarova, S.V.; Khadra, A. High-affinity P2Y2 and low-affinity P2X7 receptor interaction modulates ATP-mediated calcium signaling in murine osteoblasts. PLoS Comput. Biol. 2021, 17, e1008872. [Google Scholar] [CrossRef] [PubMed]
- Mikolajewicz, N.; Sehayek, S.; Wiseman, P.W.; Komarova, S.V. Transmission of Mechanical Information by Purinergic Signaling. Biophys. J. 2019, 116, 2009–2022. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, B.L.; Penuela, S. Pannexin 3 channels in health and disease. Purinergic Signal. 2021, 17, 577–589. [Google Scholar] [CrossRef]
- Ishikawa, M.; Iwamoto, T.; Nakamura, T.; Doyle, A.; Fukumoto, S.; Yamada, Y. Pannexin 3 functions as an ER Ca2+ channel, hemichannel, and gap junction to promote osteoblast differentiation. J. Cell Biol. 2011, 193, 1257–1274. [Google Scholar] [CrossRef]
- Ishikawa, M.; Williams, G.; Forcinito, P.; Ishikawa, M.; Petrie, R.J.; Saito, K.; Fukumoto, S.; Yamada, Y. Pannexin 3 ER Ca2+ channel gating is regulated by phosphorylation at the Serine 68 residue in osteoblast differentiation. Sci. Rep. 2019, 9, 18759. [Google Scholar] [CrossRef]
- Carluccio, M.; Ziberi, S.; Zuccarini, M.; Giuliani, P.; Caciagli, F.; Di Iorio, P.; Ciccarelli, R. Adult mesenchymal stem cells: Is there a role for purine receptors in their osteogenic differentiation? Purinergic Signal. 2020, 16, 263–287. [Google Scholar] [CrossRef]
- Ma, Y.; Di, R.; Zhao, H.; Song, R.; Zou, H.; Liu, Z. P2X7 receptor knockdown suppresses osteoclast differentiation by inhibiting autophagy and Ca2+/calcineurin signaling. Mol. Med. Rep. 2022, 25, 160. [Google Scholar] [CrossRef]
- Sun, W.; Guo, S.; Li, Y.; Li, J.; Liu, C.; Chen, Y.; Wang, X.; Tan, Y.; Tian, H.; Wang, C.; et al. Anoctamin 1 controls bone resorption by coupling Cl− channel activation with RANKL-RANK signaling transduction. Nat. Commun. 2022, 13, 2899. [Google Scholar] [CrossRef]
- Lu, X.; Li, W.; Wang, H.; Cao, M.; Jin, Z. The role of the Smad2/3/4 signaling pathway in osteogenic differentiation regulation by ClC-3 chloride channels in MC3T3-E1 cells. J. Orthop. Surg. Res. 2022, 17, 338. [Google Scholar] [CrossRef]
- Baghdadi, M.B.; Tajbakhsh, S. Regulation and phylogeny of skeletal muscle regeneration. Dev. Biol. 2018, 433, 200–209. [Google Scholar] [CrossRef]
- Chen, L.; Hassani Nia, F.; Stauber, T. Ion Channels and Transporters in Muscle Cell Differentiation. Int. J. Mol. Sci. 2021, 22, 13615. [Google Scholar] [CrossRef]
- Liu, J.H.; Konig, S.; Michel, M.; Arnaudeau, S.; Fischer-Lougheed, J.; Bader, C.R.; Bernheim, L. Acceleration of human myoblast fusion by depolarization: Graded Ca2+ signals involved. Development 2003, 130, 3437–3446. [Google Scholar] [CrossRef] [PubMed]
- Olah, T.; Fodor, J.; Ruzsnavszky, O.; Vincze, J.; Berbey, C.; Allard, B.; Csernoch, L. Overexpression of transient receptor potential canonical type 1 (TRPC1) alters both store operated calcium entry and depolarization-evoked calcium signals in C2C12 cells. Cell Calcium 2011, 49, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Singareddy, S.S.; Roessler, H.I.; McClenaghan, C.; Ikle, J.M.; Tryon, R.C.; van Haaften, G.; Nichols, C.G. ATP-sensitive potassium channels in zebrafish cardiac and vascular smooth muscle. J. Physiol. 2022, 600, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Ando, K.; Tong, L.; Peng, D.; Vazquez-Liebanas, E.; Chiyoda, H.; He, L.; Liu, J.; Kawakami, K.; Mochizuki, N.; Fukuhara, S.; et al. KCNJ8/ABCC9-containing K-ATP channel modulates brain vascular smooth muscle development and neurovascular coupling. Dev. Cell 2022, 57, 1383–1399. [Google Scholar] [CrossRef] [PubMed]
- Maqoud, F.; Scala, R.; Hoxha, M.; Zappacosta, B.; Tricarico, D. ATP-sensitive Potassium Channel Subunits in Neuroinflammation: Novel Drug Targets in Neurodegenerative Disorders. CNS Neurol. Disord. Drug Targets 2022, 21, 130–149. [Google Scholar] [CrossRef]
- Scala, R.; Maqoud, F.; McClenaghan, C.; Harter, T.M.; Perrone, M.G.; Scilimati, A.; Nichols, C.G.; Tricarico, D. Zoledronic Acid Blocks Overactive Kir6.1/SUR2-Dependent KATP Channels in Skeletal Muscle and Osteoblasts in a Murine Model of Cantu Syndrome. Cells 2023, 12, 928. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, N.S.; Robertson, G.A.; Ganetzky, B. A component of calcium-activated potassium channels encoded by the Drosophila slo locus. Science 1991, 253, 551–555. [Google Scholar] [CrossRef]
- Marty, A.; Tan, Y.P.; Trautmann, A. Three types of calcium-dependent channel in rat lacrimal glands. J. Physiol. 1984, 357, 293–325. [Google Scholar] [CrossRef] [PubMed]
- Latorre, R.; Miller, C. Conduction and selectivity in potassium channels. J. Membr. Biol. 1983, 71, 11–30. [Google Scholar] [CrossRef]
- Park, Y.B. Ion selectivity and gating of small conductance Ca2+-activated K+ channels in cultured rat adrenal chromaffin cells. J. Physiol. 1994, 481 Pt 3, 555–570. [Google Scholar] [CrossRef]
- Barrett, J.N.; Magleby, K.L.; Pallotta, B.S. Properties of single calcium-activated potassium channels in cultured rat muscle. J. Physiol. 1982, 331, 211–230. [Google Scholar] [CrossRef] [PubMed]
- Brown, B.M.; Shim, H.; Christophersen, P.; Wulff, H. Pharmacology of Small- and Intermediate-Conductance Calcium-Activated Potassium Channels. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 219–240. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Perez, V.; Lingle, C.J. Regulation of BK Channels by Beta and Gamma Subunits. Annu. Rev. Physiol. 2019, 81, 113–137. [Google Scholar] [CrossRef]
- Schreiber, M.; Salkoff, L. A novel calcium-sensing domain in the BK channel. Biophys. J. 1997, 73, 1355–1363. [Google Scholar] [CrossRef]
- Schumacher, M.A.; Rivard, A.F.; Bachinger, H.P.; Adelman, J.P. Structure of the gating domain of a Ca2+-activated K+ channel complexed with Ca2+/calmodulin. Nature 2001, 410, 1120–1124. [Google Scholar] [CrossRef]
- Xia, X.M.; Fakler, B.; Rivard, A.; Wayman, G.; Johnson-Pais, T.; Keen, J.E.; Ishii, T.; Hirschberg, B.; Bond, C.T.; Lutsenko, S.; et al. Mechanism of calcium gating in small-conductance calcium-activated potassium channels. Nature 1998, 395, 503–507. [Google Scholar] [CrossRef]
- Haidar, O.; O’Neill, N.; Staunton, C.A.; Bavan, S.; O’Brien, F.; Zouggari, S.; Sharif, U.; Mobasheri, A.; Kumagai, K.; Barrett-Jolley, R. Pro-inflammatory Cytokines Drive Deregulation of Potassium Channel Expression in Primary Synovial Fibroblasts. Front. Physiol. 2020, 11, 226. [Google Scholar] [CrossRef]
- Nelson, M.T.; Quayle, J.M. Physiological roles and properties of potassium channels in arterial smooth muscle. Am. J. Physiol. 1995, 268, C799–C822. [Google Scholar] [CrossRef] [PubMed]
- Feetham, C.H.; O’Brien, F.; Barrett-Jolley, R. Ion Channels in the Paraventricular Hypothalamic Nucleus (PVN); Emerging Diversity and Functional Roles. Front. Physiol. 2018, 9, 760. [Google Scholar] [CrossRef] [PubMed]
- Lewis, R.; May, H.; Mobasheri, A.; Barrett-Jolley, R. Chondrocyte channel transcriptomics: Do microarray data fit with expression and functional data? Channels 2013, 7, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Staunton, C.A.; Owen, E.D.; Hemmings, K.; Vasilaki, A.; McArdle, A.; Barrett-Jolley, R.; Jackson, M.J. Skeletal muscle transcriptomics identifies common pathways in nerve crush injury and ageing. Skelet. Muscle 2022, 12, 3. [Google Scholar] [CrossRef]
- Takacs, R.; Vago, J.; Poliska, S.; Pushparaj, P.N.; Ducza, L.; Kovacs, P.; Jin, E.J.; Barrett-Jolley, R.; Zakany, R.; Matta, C. The temporal transcriptomic signature of cartilage formation. Nucleic Acids Res. 2023, gkad210. [Google Scholar] [CrossRef]
- Abdul Kadir, L.; Stacey, M.; Barrett-Jolley, R. Emerging Roles of the Membrane Potential: Action Beyond the Action Potential. Front. Physiol. 2018, 9, 1661. [Google Scholar] [CrossRef]
- McFerrin, M.B.; Turner, K.L.; Cuddapah, V.A.; Sontheimer, H. Differential role of IK and BK potassium channels as mediators of intrinsic and extrinsic apoptotic cell death. Am. J. Physiol. Cell Physiol. 2012, 303, C1070–C1078. [Google Scholar] [CrossRef]
- Lewis, R.; Asplin, K.E.; Bruce, G.; Dart, C.; Mobasheri, A.; Barrett-Jolley, R. The role of the membrane potential in chondrocyte volume regulation. J. Cell. Physiol. 2011, 226, 2979–2986. [Google Scholar] [CrossRef]
- Feetham, C.H.; Nunn, N.; Lewis, R.; Dart, C.; Barrett-Jolley, R. TRPV4 and KCa ion channels functionally couple as osmosensors in the paraventricular nucleus. Br. J. Pharmacol. 2015, 172, 1753–1768. [Google Scholar] [CrossRef]
- Nilius, B.; Droogmans, G. Ion channels and their functional role in vascular endothelium. Physiol. Rev. 2001, 81, 1415–1459. [Google Scholar] [CrossRef]
- Tao, R.; Lau, C.P.; Tse, H.F.; Li, G.R. Functional ion channels in mouse bone marrow mesenchymal stem cells. Am. J. Physiol. Cell Physiol. 2007, 293, C1561–C1567. [Google Scholar] [CrossRef]
- Deng, X.L.; Lau, C.P.; Lai, K.; Cheung, K.F.; Lau, G.K.; Li, G.R. Cell cycle-dependent expression of potassium channels and cell proliferation in rat mesenchymal stem cells from bone marrow. Cell Prolif. 2007, 40, 656–670. [Google Scholar] [CrossRef] [PubMed]
- Echeverry, S.; Grismaldo, A.; Sanchez, C.; Sierra, C.; Henao, J.C.; Granados, S.T.; Sutachan, J.J.; Torres, Y.P. Activation of BK Channel Contributes to PL-Induced Mesenchymal Stem Cell Migration. Front. Physiol. 2020, 11, 210. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Ma, J.; Pan, Z.; Song, Y.H.; Freyberg, S.; Yan, Y.; Vykoukal, D.; Alt, E. Electrophysiological properties of human adipose tissue-derived stem cells. Am. J. Physiol. Cell Physiol. 2007, 293, C1539–C1550. [Google Scholar] [CrossRef]
- Zhang, Y.Y.; Yue, J.; Che, H.; Sun, H.Y.; Tse, H.F.; Li, G.R. BKCa and hEag1 channels regulate cell proliferation and differentiation in human bone marrow-derived mesenchymal stem cells. J. Cell. Physiol. 2014, 229, 202–212. [Google Scholar] [CrossRef]
- Zhao, Y.; Wei, H.; Kong, G.; Shim, W.; Zhang, G. Hydrogen sulfide augments the proliferation and survival of human induced pluripotent stem cell-derived mesenchymal stromal cells through inhibition of BKCa. Cytotherapy 2013, 15, 1395–1405. [Google Scholar] [CrossRef]
- Vigneault, P.; Naud, P.; Qi, X.; Xiao, J.; Villeneuve, L.; Davis, D.R.; Nattel, S. Calcium-dependent potassium channels control proliferation of cardiac progenitor cells and bone marrow-derived mesenchymal stem cells. J. Physiol. 2018, 596, 2359–2379. [Google Scholar] [CrossRef]
- Li, G.R.; Sun, H.; Deng, X.; Lau, C.P. Characterization of ionic currents in human mesenchymal stem cells from bone marrow. Stem Cells 2005, 23, 371–382. [Google Scholar] [CrossRef]
- Chubinskiy-Nadezhdin, V.I.; Vasileva, V.Y.; Pugovkina, N.A.; Vassilieva, I.O.; Morachevskaya, E.A.; Nikolsky, N.N.; Negulyaev, Y.A. Local calcium signalling is mediated by mechanosensitive ion channels in mesenchymal stem cells. Biochem. Biophys. Res. Commun. 2017, 482, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Hei, H.; Gao, J.; Dong, J.; Tao, J.; Tian, L.; Pan, W.; Wang, H.; Zhang, X. BK Knockout by TALEN-Mediated Gene Targeting in Osteoblasts: KCNMA1 Determines the Proliferation and Differentiation of Osteoblasts. Mol. Cells 2016, 39, 530–535. [Google Scholar] [CrossRef]
- Chubinskiy-Nadezhdin, V.I.; Sudarikova, A.V.; Shilina, M.A.; Vasileva, V.Y.; Grinchuk, T.M.; Lyublinskaya, O.G.; Nikolsky, N.N.; Negulyaev, Y.A. Cell Cycle-Dependent Expression of Bk Channels in Human Mesenchymal Endometrial Stem Cells. Sci. Rep. 2019, 9, 4595. [Google Scholar] [CrossRef]
- Jia, X.; Su, H.; Chen, X.; Huang, Y.; Zheng, Y.; Ji, P.; Gao, C.; Gong, X.; Huang, Y.; Jiang, L.H.; et al. A critical role of the KCa3.1 channel in mechanical stretch-induced proliferation of rat bone marrow-derived mesenchymal stem cells. J. Cell. Mol. Med. 2020, 24, 3739–3744. [Google Scholar] [CrossRef] [PubMed]
- Tao, R.; Lau, C.P.; Tse, H.F.; Li, G.R. Regulation of cell proliferation by intermediate-conductance Ca2+-activated potassium and volume-sensitive chloride channels in mouse mesenchymal stem cells. Am. J. Physiol. Cell Physiol. 2008, 295, C1409–C1416. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.R.; Duncan, N.A.; Giles, W.R.; Clark, R.B. A voltage-dependent K+ current contributes to membrane potential of acutely isolated canine articular chondrocytes. J. Physiol. 2004, 557, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.B.; Kondo, C.; Belke, D.D.; Giles, W.R. Two-pore domain K+ channels regulate membrane potential of isolated human articular chondrocytes. J. Physiol. 2011, 589, 5071–5089. [Google Scholar] [CrossRef]
- Mobasheri, A.; Lewis, R.; Ferreira-Mendes, A.; Rufino, A.; Dart, C.; Barrett-Jolley, R. Potassium channels in articular chondrocytes. Channels 2012, 6, 416–425. [Google Scholar] [CrossRef]
- Lee, H.S.; Millward-Sadler, S.J.; Wright, M.O.; Nuki, G.; Salter, D.M. Integrin and mechanosensitive ion channel-dependent tyrosine phosphorylation of focal adhesion proteins and beta-catenin in human articular chondrocytes after mechanical stimulation. J. Bone Miner. Res. 2000, 15, 1501–1509. [Google Scholar] [CrossRef]
- Hdud, I.M.; Mobasheri, A.; Loughna, P.T. Effects of cyclic equibiaxial mechanical stretch on α-BK and TRPV4 expression in equine chondrocytes. Springerplus 2014, 3, 59. [Google Scholar] [CrossRef]
- Sanchez, J.C.; Lopez-Zapata, D.F. Effects of osmotic challenges on membrane potential in human articular chondrocytes from healthy and osteoarthritic cartilage. Biorheology 2010, 47, 321–331. [Google Scholar] [CrossRef]
- Miyazaki, Y.; Ichimura, A.; Kitayama, R.; Okamoto, N.; Yasue, T.; Liu, F.; Kawabe, T.; Nagatomo, H.; Ueda, Y.; Yamauchi, I.; et al. C-type natriuretic peptide facilitates autonomic Ca2+ entry in growth plate chondrocytes for stimulating bone growth. eLife 2022, 11, 71931. [Google Scholar] [CrossRef]
- Valhmu, W.B.; Raia, F.J. myo-Inositol 1,4,5-trisphosphate and Ca2+/calmodulin-dependent factors mediate transduction of compression-induced signals in bovine articular chondrocytes. Biochem. J. 2002, 361, 689–696. [Google Scholar] [CrossRef]
- Hou, S.; Heinemann, S.H.; Hoshi, T. Modulation of BKCa channel gating by endogenous signaling molecules. Physiology 2009, 24, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Bolduc, J.A.; Collins, J.A.; Loeser, R.F. Reactive oxygen species, aging and articular cartilage homeostasis. Free Radic. Biol. Med. 2019, 132, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, J.C.; Lopez-Zapata, D.F. The role of BKCa channels on hyperpolarization mediated by hyperosmolarity in human articular chondrocytes. Gen. Physiol. Biophys. 2011, 30, 20–27. [Google Scholar] [CrossRef]
- Hirukawa, K.; Muraki, K.; Ohya, S.; Imaizumi, Y.; Togari, A. Electrophysiological properties of a novel Ca2+-activated K+ channel expressed in human osteoblasts. Calcif. Tissue Int. 2008, 83, 222–229. [Google Scholar] [CrossRef]
- Wang, Y.; Guo, Q.; Hei, H.; Tao, J.; Zhou, Y.; Dong, J.; Xin, H.; Cai, H.; Gao, J.; Yu, K.; et al. BK ablation attenuates osteoblast bone formation via integrin pathway. Cell Death Dis. 2019, 10, 738. [Google Scholar] [CrossRef]
- Rezzonico, R.; Cayatte, C.; Bourget-Ponzio, I.; Romey, G.; Belhacene, N.; Loubat, A.; Rocchi, S.; Van Obberghen, E.; Girault, J.A.; Rossi, B.; et al. Focal adhesion kinase pp125FAK interacts with the large conductance calcium-activated hSlo potassium channel in human osteoblasts: Potential role in mechanotransduction. J. Bone Miner. Res. 2003, 18, 1863–1871. [Google Scholar] [CrossRef]
- Henney, N.C.; Li, B.; Elford, C.; Reviriego, P.; Campbell, A.K.; Wann, K.T.; Evans, B.A. A large-conductance (BK) potassium channel subtype affects both growth and mineralization of human osteoblasts. Am. J. Physiol. Cell Physiol. 2009, 297, C1397–C1408. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Yang, Q.; Gao, J.; Yang, J.; He, J.; Xin, H.; Zhang, X. BK Channel Deficiency in Osteoblasts Reduces Bone Formation via the Wnt/β-Catenin Pathway. Mol. Cells 2021, 44, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Ren, M.S.; Xie, H.H.; Ding, Y.; Li, Z.H.; Liu, B. Er-xian decoction drug-containing serum promotes Mc3t3-e1 cell proliferation and osteogenic differentiation via regulating BK channel. J. Ethnopharmacol. 2023, 302, 115887. [Google Scholar] [CrossRef] [PubMed]
- Ning, F.L.; Tao, J.; Li, D.D.; Tian, L.L.; Wang, M.L.; Reilly, S.; Liu, C.; Cai, H.; Xin, H.; Zhang, X.M. Activating BK channels ameliorates vascular smooth muscle calcification through Akt signaling. Acta Pharmacol. Sin. 2022, 43, 624–633. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Dai, H.; Yu, G.; Song, C.; Liu, J.; Xu, J. Inhibiting KCNMA1-AS1 promotes osteogenic differentiation of HBMSCs via miR-1303/cochlin axis. J. Orthop. Surg. Res. 2023, 18, 73. [Google Scholar] [CrossRef]
- Kito, H.; Morihiro, H.; Sakakibara, Y.; Endo, K.; Kajikuri, J.; Suzuki, T.; Ohya, S. Downregulation of the Ca2+-activated K+ channel KCa3.1 in mouse preosteoblast cells treated with vitamin D receptor agonist. Am. J. Physiol. Cell Physiol. 2020, 319, C345–C358. [Google Scholar] [CrossRef] [PubMed]
- Freise, C.; Querfeld, U. Inhibition of vascular calcification by block of intermediate conductance calcium-activated potassium channels with TRAM-34. Pharmacol. Res. 2014, 85, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Grossinger, E.M.; Kang, M.; Bouchareychas, L.; Sarin, R.; Haudenschild, D.R.; Borodinsky, L.N.; Adamopoulos, I.E. Ca2+-Dependent Regulation of NFATc1 via KCa3.1 in Inflammatory Osteoclastogenesis. J. Immunol. 2018, 200, 749–757. [Google Scholar] [CrossRef]
- Gu, Y.; Preston, M.R.; El Haj, A.J.; Howl, J.D.; Publicover, S.J. Three types of K+ currents in murine osteocyte-like cells (MLO-Y4). Bone 2001, 28, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, P.; Body, J.J.; Boonen, S.; Boutsen, Y.; Devogelaer, J.P.; Goemaere, S.; Kaufman, J.; Reginster, J.Y.; Rozenberg, S. Loading and skeletal development and maintenance. J. Osteoporos. 2011, 2011, 786752. [Google Scholar] [CrossRef]
- Fuest, S.; Post, C.; Balbach, S.T.; Jabar, S.; Neumann, I.; Schimmelpfennig, S.; Sargin, S.; Nass, E.; Budde, T.; Kailayangiri, S.; et al. Relevance of Abnormal KCNN1 Expression and Osmotic Hypersensitivity in Ewing Sarcoma. Cancers 2022, 14, 4819. [Google Scholar] [CrossRef] [PubMed]
- He, C.; Li, X.; Wang, M.; Zhang, S.; Liu, H. Deletion of BK channels decreased skeletal and cardiac muscle function but increased smooth muscle contraction in rats. Biochem. Biophys. Res. Commun. 2021, 570, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Park, W.S.; Heo, S.C.; Jeon, E.S.; Hong, D.H.; Son, Y.K.; Ko, J.H.; Kim, H.K.; Lee, S.Y.; Kim, J.H.; Han, J. Functional expression of smooth muscle-specific ion channels in TGF-β1-treated human adipose-derived mesenchymal stem cells. Am. J. Physiol. Cell Physiol. 2013, 305, C377–C391. [Google Scholar] [CrossRef]
- Fioretti, B.; Pietrangelo, T.; Catacuzzeno, L.; Franciolini, F. Intermediate-conductance Ca2+-activated K+ channel is expressed in C2C12 myoblasts and is downregulated during myogenesis. Am. J. Physiol. Cell Physiol. 2005, 289, C89–C96. [Google Scholar] [CrossRef]
- Pietrangelo, T.; Fioretti, B.; Mancinelli, R.; Catacuzzeno, L.; Franciolini, F.; Fano, G.; Fulle, S. Extracellular guanosine-5′-triphosphate modulates myogenesis via intermediate Ca2+-activated K+ currents in C2C12 mouse cells. J. Physiol. 2006, 572, 721–733. [Google Scholar] [CrossRef]
- Iseki, Y.; Ono, Y.; Hibi, C.; Tanaka, S.; Takeshita, S.; Maejima, Y.; Kurokawa, J.; Murakawa, M.; Shimomura, K.; Sakamoto, K. Opening of Intermediate Conductance Ca2+-Activated K+ Channels in C2C12 Skeletal Muscle Cells Increases the Myotube Diameter via the Akt/Mammalian Target of Rapamycin Pathway. J. Pharmacol. Exp. Ther. 2021, 376, 454–462. [Google Scholar] [CrossRef]
- Beeton, C. KCa1.1 channels as therapeutic targets for rheumatoid arthritis. Expert Opin. Ther. Targets 2017, 21, 1077–1081. [Google Scholar] [CrossRef]
- Bottini, N.; Firestein, G.S. Duality of fibroblast-like synoviocytes in RA: Passive responders and imprinted aggressors. Nat. Rev. Rheumatol. 2013, 9, 24–33. [Google Scholar] [CrossRef]
- Bartok, B.; Firestein, G.S. Fibroblast-like synoviocytes: Key effector cells in rheumatoid arthritis. Immunol. Rev. 2010, 233, 233–255. [Google Scholar] [CrossRef] [PubMed]
- Tanner, M.R.; Pennington, M.W.; Chauhan, S.S.; Laragione, T.; Gulko, P.S.; Beeton, C. KCa1.1 and Kv1.3 channels regulate the interactions between fibroblast-like synoviocytes and T lymphocytes during rheumatoid arthritis. Arthritis Res. Ther. 2019, 21, 6. [Google Scholar] [CrossRef] [PubMed]
- Petho, Z.; Tanner, M.R.; Tajhya, R.B.; Huq, R.; Laragione, T.; Panyi, G.; Gulko, P.S.; Beeton, C. Different expression of beta subunits of the KCa1.1 channel by invasive and non-invasive human fibroblast-like synoviocytes. Arthritis Res. Ther. 2016, 18, 103. [Google Scholar] [CrossRef] [PubMed]
- Tanner, M.R.; Hu, X.; Huq, R.; Tajhya, R.B.; Sun, L.; Khan, F.S.; Laragione, T.; Horrigan, F.T.; Gulko, P.S.; Beeton, C. KCa1.1 inhibition attenuates fibroblast-like synoviocyte invasiveness and ameliorates disease in rat models of rheumatoid arthritis. Arthritis Rheumatol. 2015, 67, 96–106. [Google Scholar] [CrossRef]
- Tanner, M.R.; Pennington, M.W.; Laragione, T.; Gulko, P.S.; Beeton, C. KCa1.1 channels regulate β1-integrin function and cell adhesion in rheumatoid arthritis fibroblast-like synoviocytes. FASEB J. 2017, 31, 3309–3320. [Google Scholar] [CrossRef]
- Hu, X.; Laragione, T.; Sun, L.; Koshy, S.; Jones, K.R.; Ismailov, I.I.; Yotnda, P.; Horrigan, F.T.; Gulko, P.S.; Beeton, C. KCa1.1 potassium channels regulate key proinflammatory and invasive properties of fibroblast-like synoviocytes in rheumatoid arthritis. J. Biol. Chem. 2012, 287, 4014–4022. [Google Scholar] [CrossRef] [PubMed]
- Bertram, K.L.; Banderali, U.; Tailor, P.; Krawetz, R.J. Ion channel expression and function in normal and osteoarthritic human synovial fluid progenitor cells. Channels 2016, 10, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Kawano, S.; Otsu, K.; Kuruma, A.; Shoji, S.; Yanagida, E.; Muto, Y.; Yoshikawa, F.; Hirayama, Y.; Mikoshiba, K.; Furuichi, T. ATP autocrine/paracrine signaling induces calcium oscillations and NFAT activation in human mesenchymal stem cells. Cell Calcium 2006, 39, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Kawano, S.; Otsu, K.; Shoji, S.; Yamagata, K.; Hiraoka, M. Ca2+ oscillations regulated by Na+-Ca2+ exchanger and plasma membrane Ca2+ pump induce fluctuations of membrane currents and potentials in human mesenchymal stem cells. Cell Calcium 2003, 34, 145–156. [Google Scholar] [CrossRef]
- Kawano, S.; Shoji, S.; Ichinose, S.; Yamagata, K.; Tagami, M.; Hiraoka, M. Characterization of Ca2+ signaling pathways in human mesenchymal stem cells. Cell Calcium 2002, 32, 165–174. [Google Scholar] [CrossRef]
- Matta, C.; Fodor, J.; Szijgyarto, Z.; Juhasz, T.; Gergely, P.; Csernoch, L.; Zakany, R. Cytosolic free Ca2+ concentration exhibits a characteristic temporal pattern during in vitro cartilage differentiation: A possible regulatory role of calcineurin in Ca-signalling of chondrogenic cells. Cell Calcium 2008, 44, 310–323. [Google Scholar] [CrossRef]
- Shakibaei, M.; Mobasheri, A. Beta1-integrins co-localize with Na, K-ATPase, epithelial sodium channels (ENaC) and voltage activated calcium channels (VACC) in mechanoreceptor complexes of mouse limb-bud chondrocytes. Histol. Histopathol. 2003, 18, 343–351. [Google Scholar] [CrossRef]
- Cui, J.; Yang, H.; Lee, U.S. Molecular mechanisms of BK channel activation. Cell. Mol. Life Sci. 2009, 66, 852–875. [Google Scholar] [CrossRef]
- Dopico, A.M.; Bukiya, A.N.; Jaggar, J.H. Calcium- and voltage-gated BK channels in vascular smooth muscle. Pflug. Arch. 2018, 470, 1271–1289. [Google Scholar] [CrossRef]
- Papp, F.; Hajdu, P.; Tajti, G.; Toth, A.; Nagy, E.; Fazekas, Z.; Kovacs, S.; Vamosi, G.; Varga, Z.; Panyi, G. Periodic Membrane Potential and Ca2+ Oscillations in T Cells Forming an Immune Synapse. Int. J. Mol. Sci. 2020, 21, 1568. [Google Scholar] [CrossRef]
- Shah, K.R.; Guan, X.; Yan, J. Structural and Functional Coupling of Calcium-Activated BK Channels and Calcium-Permeable Channels within Nanodomain Signaling Complexes. Front. Physiol. 2021, 12, 796540. [Google Scholar] [CrossRef]
- Almassy, J.; Nanasi, P.P. Brief structural insight into the allosteric gating mechanism of BK (Slo1) channel. Can. J. Physiol. Pharmacol. 2019, 97, 498–502. [Google Scholar] [CrossRef]
- Phan, M.N.; Leddy, H.A.; Votta, B.J.; Kumar, S.; Levy, D.S.; Lipshutz, D.B.; Lee, S.H.; Liedtke, W.; Guilak, F. Functional characterization of TRPV4 as an osmotically sensitive ion channel in porcine articular chondrocytes. Arthritis Rheum. 2009, 60, 3028–3037. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Lukowski, R.; Sausbier, M.; Zhang, D.D.; Sisignano, M.; Schuh, C.D.; Kuner, R.; Ruth, P.; Geisslinger, G.; Schmidtko, A. BKCa channels expressed in sensory neurons modulate inflammatory pain in mice. Pain 2014, 155, 556–565. [Google Scholar] [CrossRef] [PubMed]
- Tricarico, D.; Mele, A.; Conte Camerino, D. Phenotype-dependent functional and pharmacological properties of BK channels in skeletal muscle: Effects of microgravity. Neurobiol. Dis. 2005, 20, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Tricarico, D.; Barbieri, M.; Mele, A.; Carbonara, G.; Camerino, D.C. Carbonic anhydrase inhibitors are specific openers of skeletal muscle BK channel of K+-deficient rats. FASEB J. 2004, 18, 760–761. [Google Scholar] [CrossRef]
- Tricarico, D.; Petruzzi, R.; Camerino, D.C. Changes of the biophysical properties of calcium-activated potassium channels of rat skeletal muscle fibers during aging. Pflug. Arch. 1997, 434, 822–829. [Google Scholar] [CrossRef] [PubMed]
- Tricarico, D.; Capriulo, R.; Camerino, D.C. Involvement of KCa2+ channels in the local abnormalities and hyperkalemia following the ischemia-reperfusion injury of rat skeletal muscle. Neuromuscul. Disord. 2002, 12, 258–265. [Google Scholar] [CrossRef]
- Tricarico, D.; Mele, A.; Calzolaro, S.; Cannone, G.; Camerino, G.M.; Dinardo, M.M.; Latorre, R.; Conte Camerino, D. Emerging role of calcium-activated potassium channel in the regulation of cell viability following potassium ions challenge in HEK293 cells and pharmacological modulation. PLoS ONE 2013, 8, e69551. [Google Scholar] [CrossRef]
- Kim, J.B.; Kim, S.J.; Kang, S.Y.; Yi, J.W.; Kim, S.M. The large-conductance calcium-activated potassium channel holds the key to the conundrum of familial hypokalemic periodic paralysis. Korean J. Pediatr. 2014, 57, 445–450. [Google Scholar] [CrossRef]
- Maqoud, F.; Cetrone, M.; Mele, A.; Tricarico, D. Molecular structure and function of big calcium-activated potassium channels in skeletal muscle: Pharmacological perspectives. Physiol. Genom. 2017, 49, 306–317. [Google Scholar] [CrossRef] [PubMed]
- Jurkat-Rott, K.; Weber, M.A.; Fauler, M.; Guo, X.H.; Holzherr, B.D.; Paczulla, A.; Nordsborg, N.; Joechle, W.; Lehmann-Horn, F. K+-dependent paradoxical membrane depolarization and Na+ overload, major and reversible contributors to weakness by ion channel leaks. Proc. Natl. Acad. Sci. USA 2009, 106, 4036–4041. [Google Scholar] [CrossRef] [PubMed]
- Tricarico, D.; Camerino, D.C. Recent advances in the pathogenesis and drug action in periodic paralyses and related channelopathies. Front. Pharmacol. 2011, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Senti, M.; Fernandez-Fernandez, J.M.; Tomas, M.; Vazquez, E.; Elosua, R.; Marrugat, J.; Valverde, M.A. Protective effect of the KCNMB1 E65K genetic polymorphism against diastolic hypertension in aging women and its relevance to cardiovascular risk. Circ. Res. 2005, 97, 1360–1365. [Google Scholar] [CrossRef]
- Gollasch, M.; Tank, J.; Luft, F.C.; Jordan, J.; Maass, P.; Krasko, C.; Sharma, A.M.; Busjahn, A.; Bahring, S. The BK channel beta1 subunit gene is associated with human baroreflex and blood pressure regulation. J. Hypertens. 2002, 20, 927–933. [Google Scholar] [CrossRef]
- Cetrone, M.; Mele, A.; Tricarico, D. Effects of the antidiabetic drugs on the age-related atrophy and sarcopenia associated with diabetes type II. Curr. Diabetes Rev. 2014, 10, 231–237. [Google Scholar] [CrossRef]
- Tomas, M.; Vazquez, E.; Fernandez-Fernandez, J.M.; Subirana, I.; Plata, C.; Heras, M.; Vila, J.; Marrugat, J.; Valverde, M.A.; Senti, M. Genetic variation in the KCNMA1 potassium channel alpha subunit as risk factor for severe essential hypertension and myocardial infarction. J. Hypertens. 2008, 26, 2147–2153. [Google Scholar] [CrossRef]
- Noh, A.S.M.; Ismail, C.A.N. A Review on Chronic Pain in Rheumatoid Arthritis: A Focus on Activation of NR2B Subunit of N-Methyl-D-Aspartate Receptors. Malays. J. Med. Sci. 2020, 27, 6–21. [Google Scholar] [CrossRef]
- Hayashi, Y.; Morinaga, S.; Zhang, J.; Satoh, Y.; Meredith, A.L.; Nakata, T.; Wu, Z.; Kohsaka, S.; Inoue, K.; Nakanishi, H. BK channels in microglia are required for morphine-induced hyperalgesia. Nat. Commun. 2016, 7, 11697. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, Y.; Hou, P.; Yan, Z.; Kong, W.; Liu, B.; Li, X.; Yao, J.; Zhang, Y.; Qin, F.; et al. TRPV1 channels are functionally coupled with BK(mSlo1) channels in rat dorsal root ganglion (DRG) neurons. PLoS ONE 2013, 8, e78203. [Google Scholar] [CrossRef]
- Shoudai, K.; Nonaka, K.; Maeda, M.; Wang, Z.M.; Jeong, H.J.; Higashi, H.; Murayama, N.; Akaike, N. Effects of various K+ channel blockers on spontaneous glycine release at rat spinal neurons. Brain Res. 2007, 1157, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.R.; Cai, Y.Q.; Pan, H.L. Plasticity and emerging role of BKCa channels in nociceptive control in neuropathic pain. J. Neurochem. 2009, 110, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Xue, Q.; Li, C.; Wang, Q.; Han, S.; Zhou, Y.; Yang, T.; Xie, Y.; Fu, H.; Lu, C.; et al. Upregulation of Beta4 subunit of BKCa channels in the anterior cingulate cortex contributes to mechanical allodynia associated anxiety-like behaviors. Mol. Brain 2020, 13, 22. [Google Scholar] [CrossRef] [PubMed]
- Sancho, M.; Kyle, B.D. The Large-Conductance, Calcium-Activated Potassium Channel: A Big Key Regulator of Cell Physiology. Front. Physiol. 2021, 12, 750615. [Google Scholar] [CrossRef] [PubMed]
- Shipston, M.J.; Tian, L. Posttranscriptional and Posttranslational Regulation of BK Channels. Int. Rev. Neurobiol. 2016, 128, 91–126. [Google Scholar] [CrossRef]
- Contreras, G.F.; Castillo, K.; Enrique, N.; Carrasquel-Ursulaez, W.; Castillo, J.P.; Milesi, V.; Neely, A.; Alvarez, O.; Ferreira, G.; Gonzalez, C.; et al. A BK (Slo1) channel journey from molecule to physiology. Channels 2013, 7, 442–458. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takács, R.; Kovács, P.; Ebeid, R.A.; Almássy, J.; Fodor, J.; Ducza, L.; Barrett-Jolley, R.; Lewis, R.; Matta, C. Ca2+-Activated K+ Channels in Progenitor Cells of Musculoskeletal Tissues: A Narrative Review. Int. J. Mol. Sci. 2023, 24, 6796. https://doi.org/10.3390/ijms24076796
Takács R, Kovács P, Ebeid RA, Almássy J, Fodor J, Ducza L, Barrett-Jolley R, Lewis R, Matta C. Ca2+-Activated K+ Channels in Progenitor Cells of Musculoskeletal Tissues: A Narrative Review. International Journal of Molecular Sciences. 2023; 24(7):6796. https://doi.org/10.3390/ijms24076796
Chicago/Turabian StyleTakács, Roland, Patrik Kovács, Rana Abdelsattar Ebeid, János Almássy, János Fodor, László Ducza, Richard Barrett-Jolley, Rebecca Lewis, and Csaba Matta. 2023. "Ca2+-Activated K+ Channels in Progenitor Cells of Musculoskeletal Tissues: A Narrative Review" International Journal of Molecular Sciences 24, no. 7: 6796. https://doi.org/10.3390/ijms24076796
APA StyleTakács, R., Kovács, P., Ebeid, R. A., Almássy, J., Fodor, J., Ducza, L., Barrett-Jolley, R., Lewis, R., & Matta, C. (2023). Ca2+-Activated K+ Channels in Progenitor Cells of Musculoskeletal Tissues: A Narrative Review. International Journal of Molecular Sciences, 24(7), 6796. https://doi.org/10.3390/ijms24076796








