A2AR as a Prognostic Marker and a Potential Immunotherapy Target in Human Glioma
Abstract
1. Introduction
2. Results
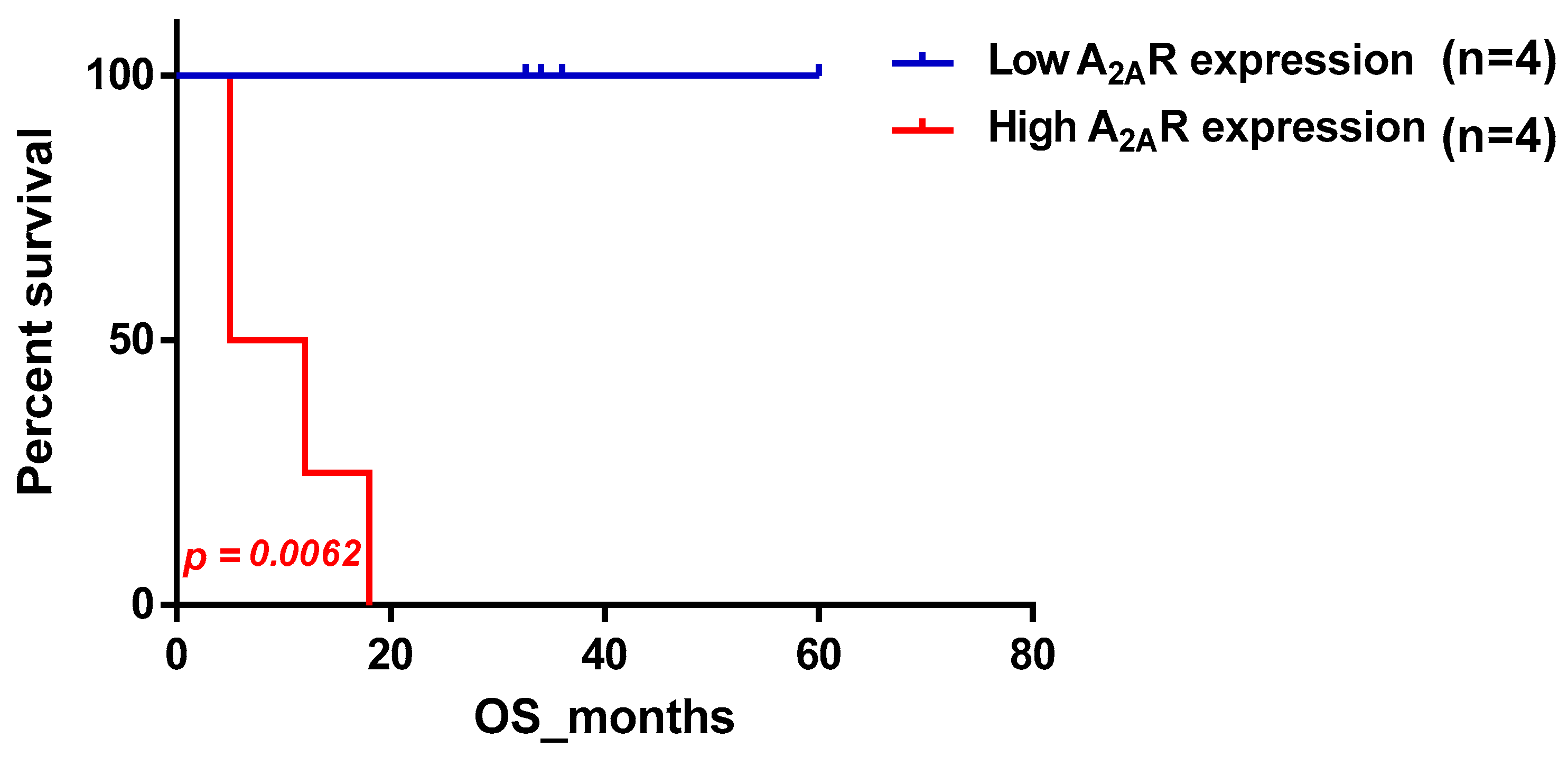
2.1. Evaluation of A2AR Expression According to Clinico-Pathological Parameters in a Moroccan Glioma Patient Cohort
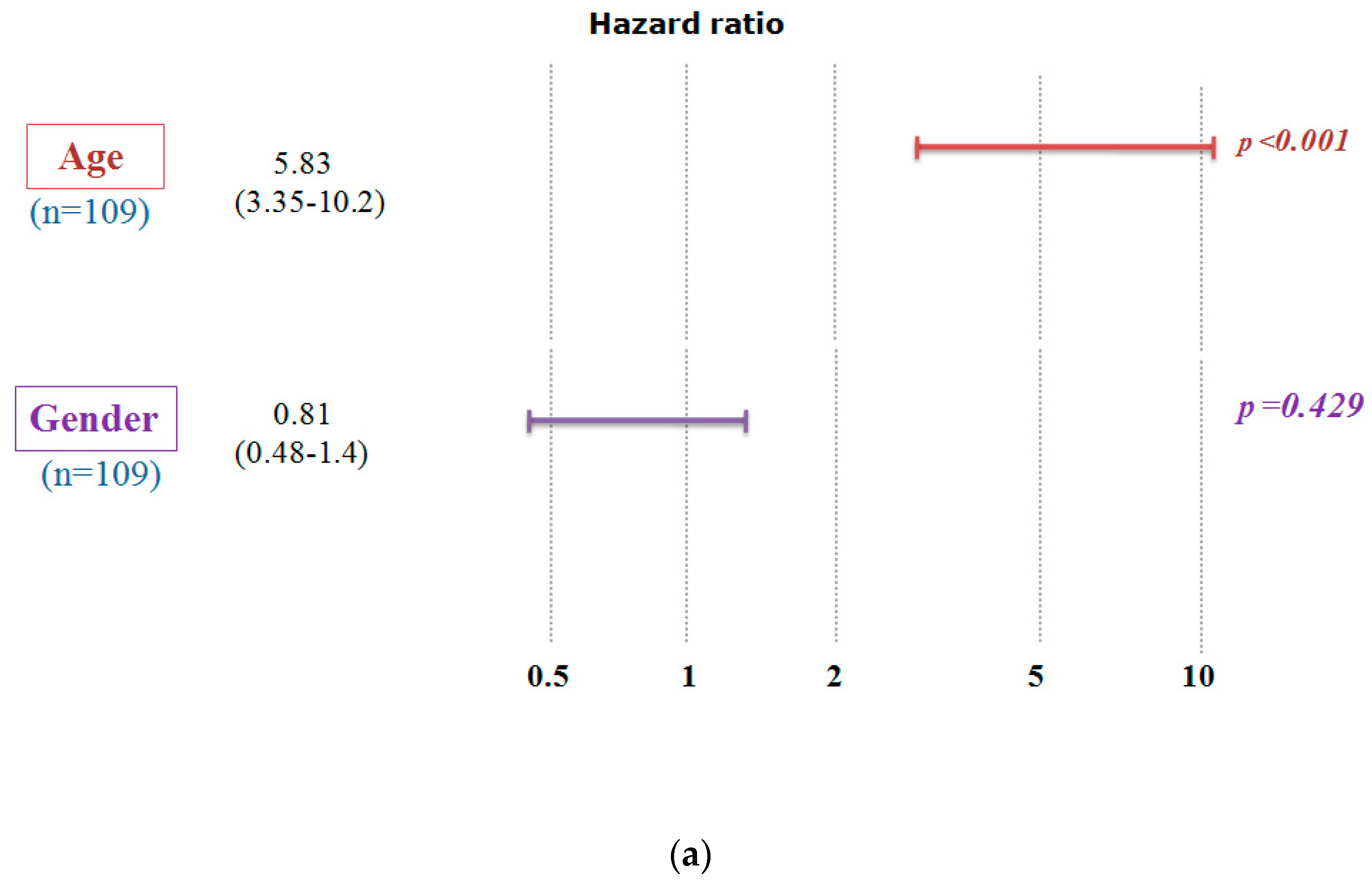
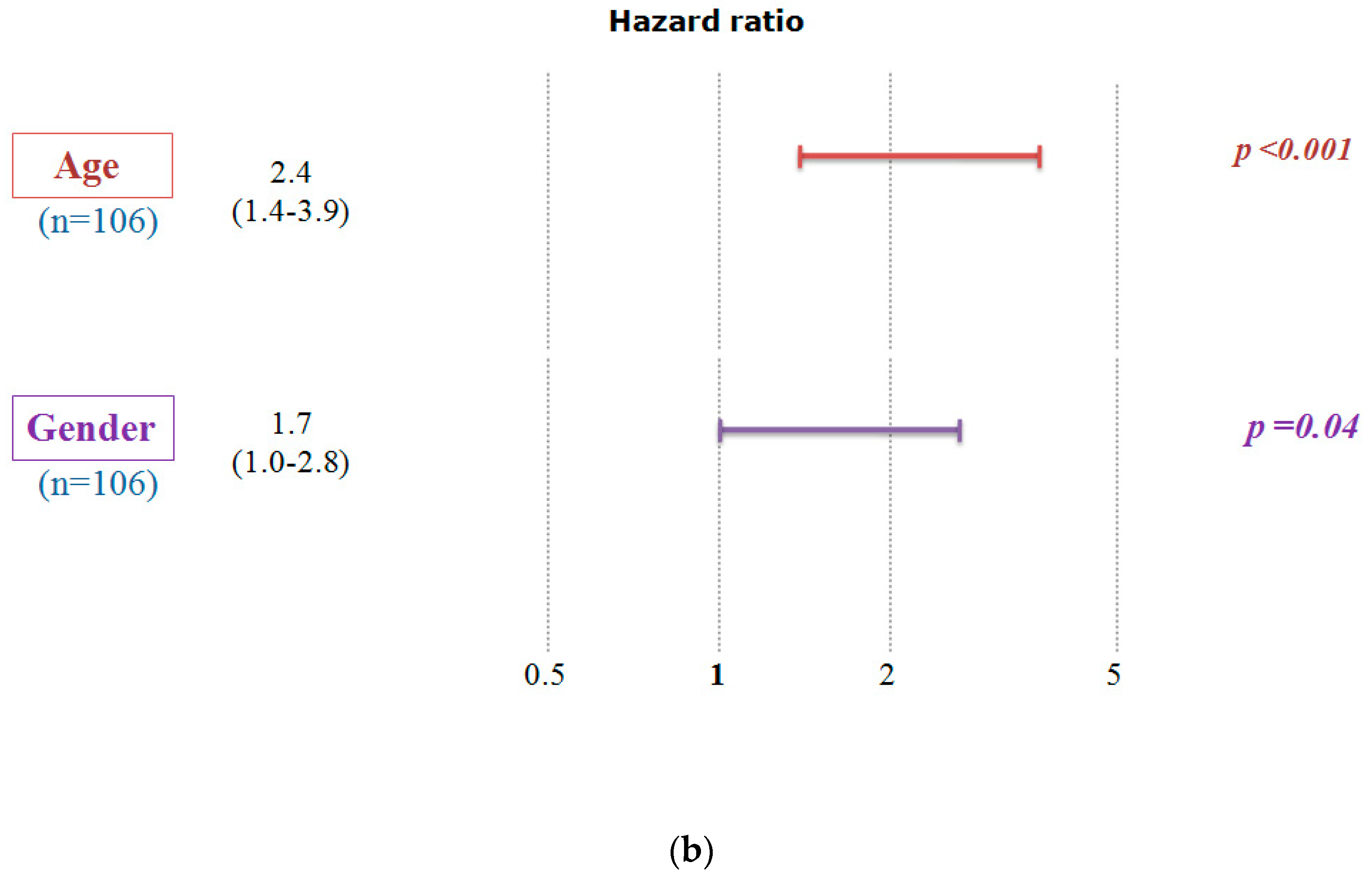
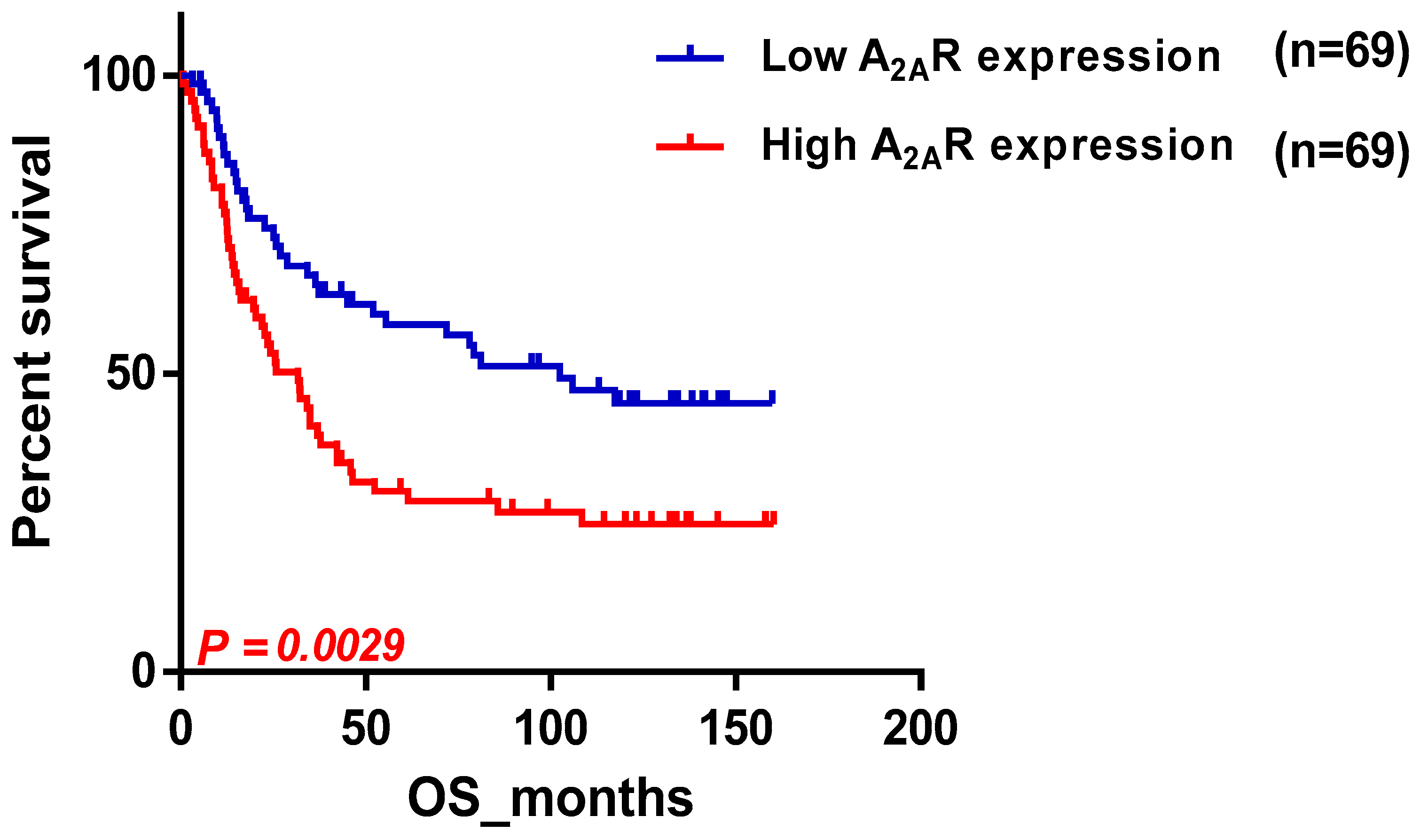
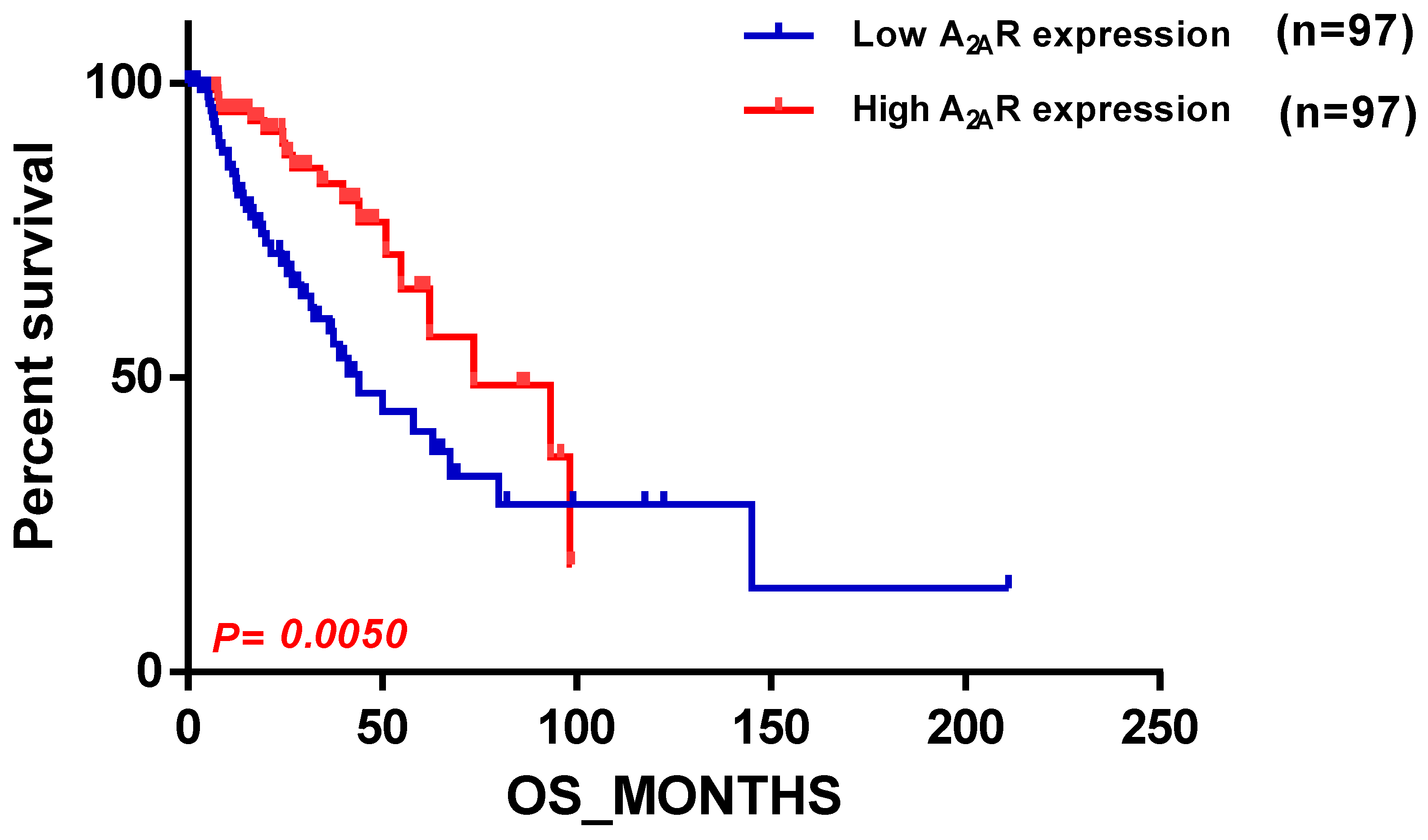
2.2. Confirmation of the Correlation between A2AR Gene Expression and Prognosis Using the Chinese Glioma Database (CGGA)
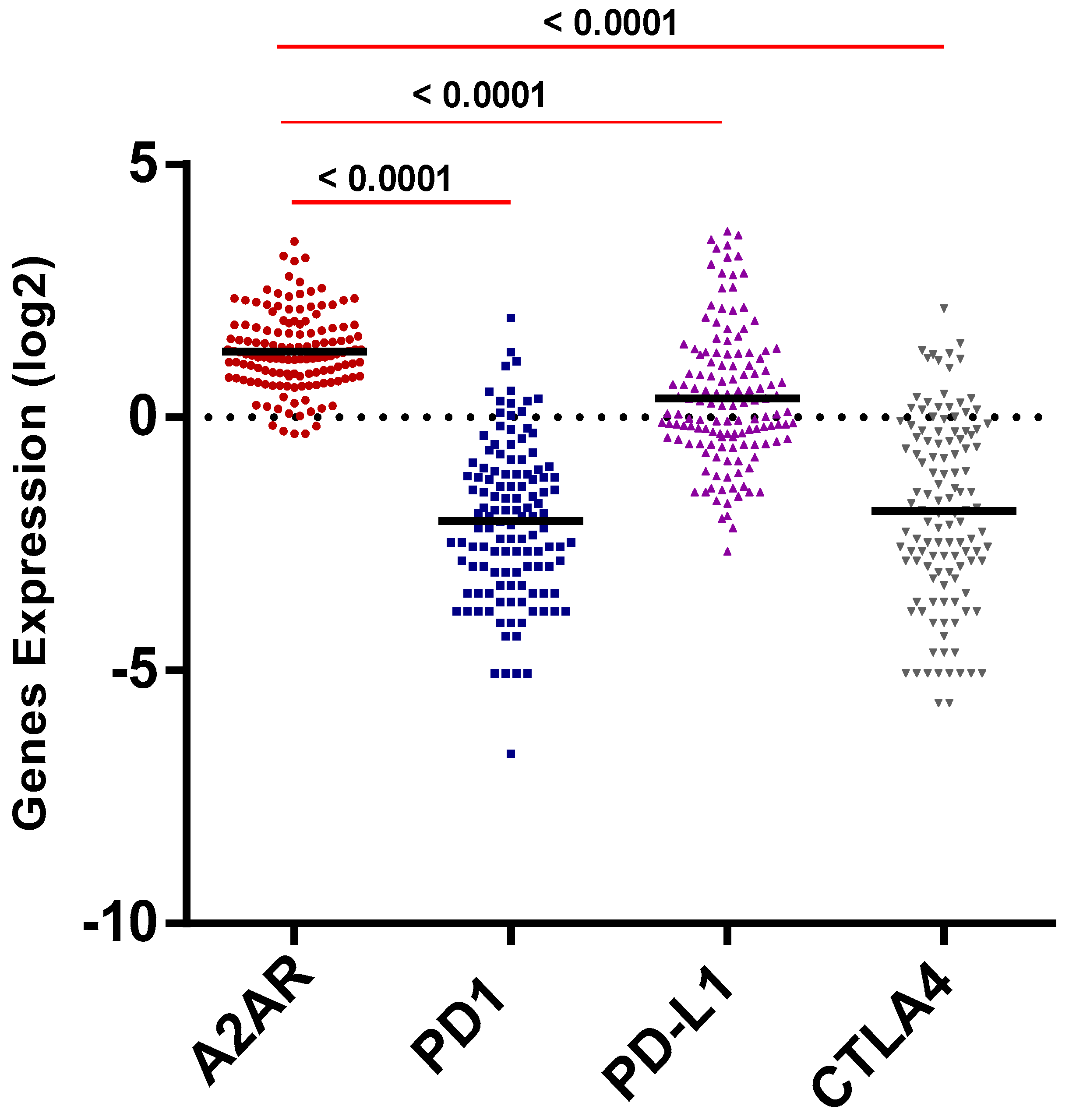
2.3. A2AR Expression Is Most Elevated Compared to Other Immune Checkpointsin Male CGGA
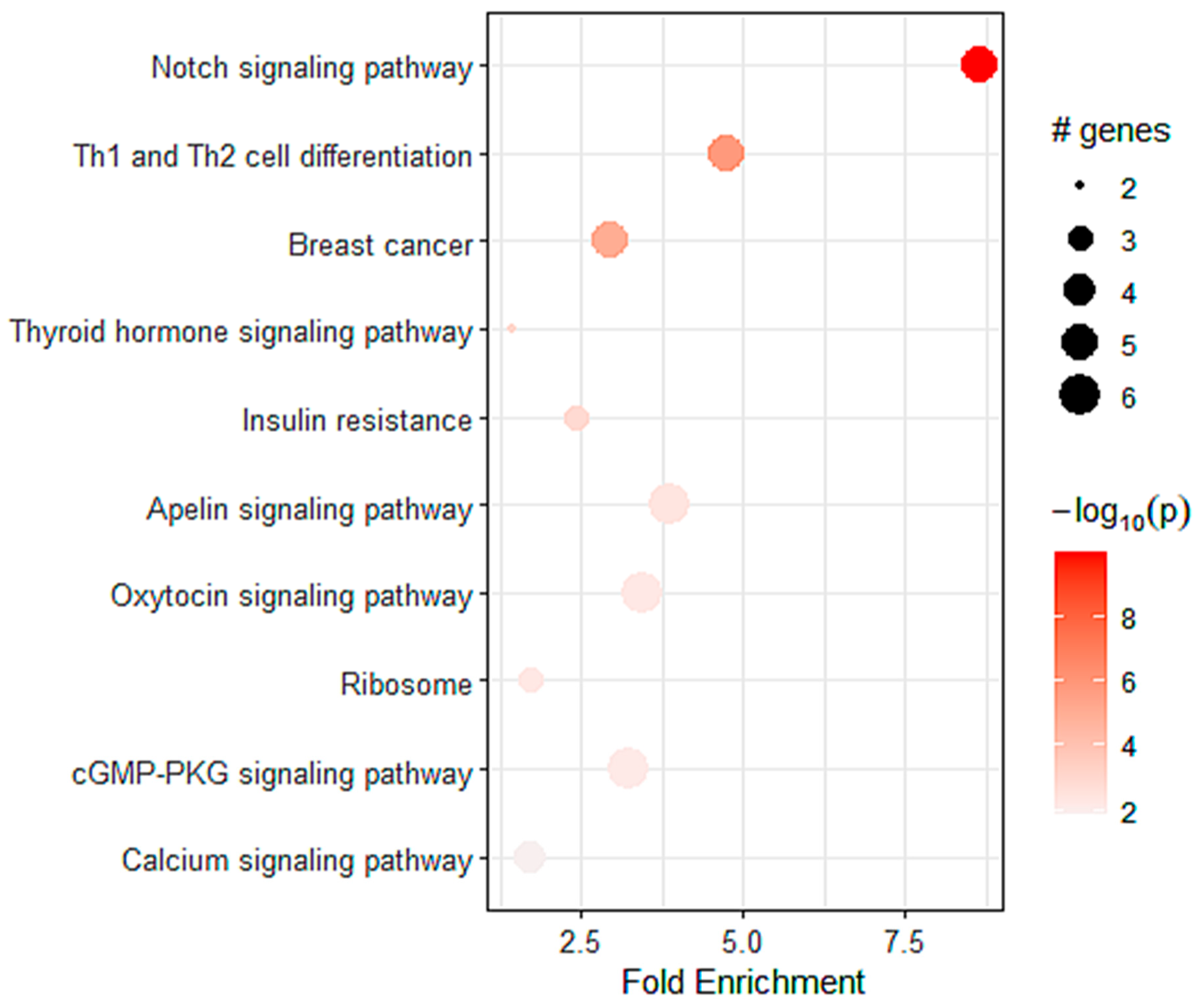
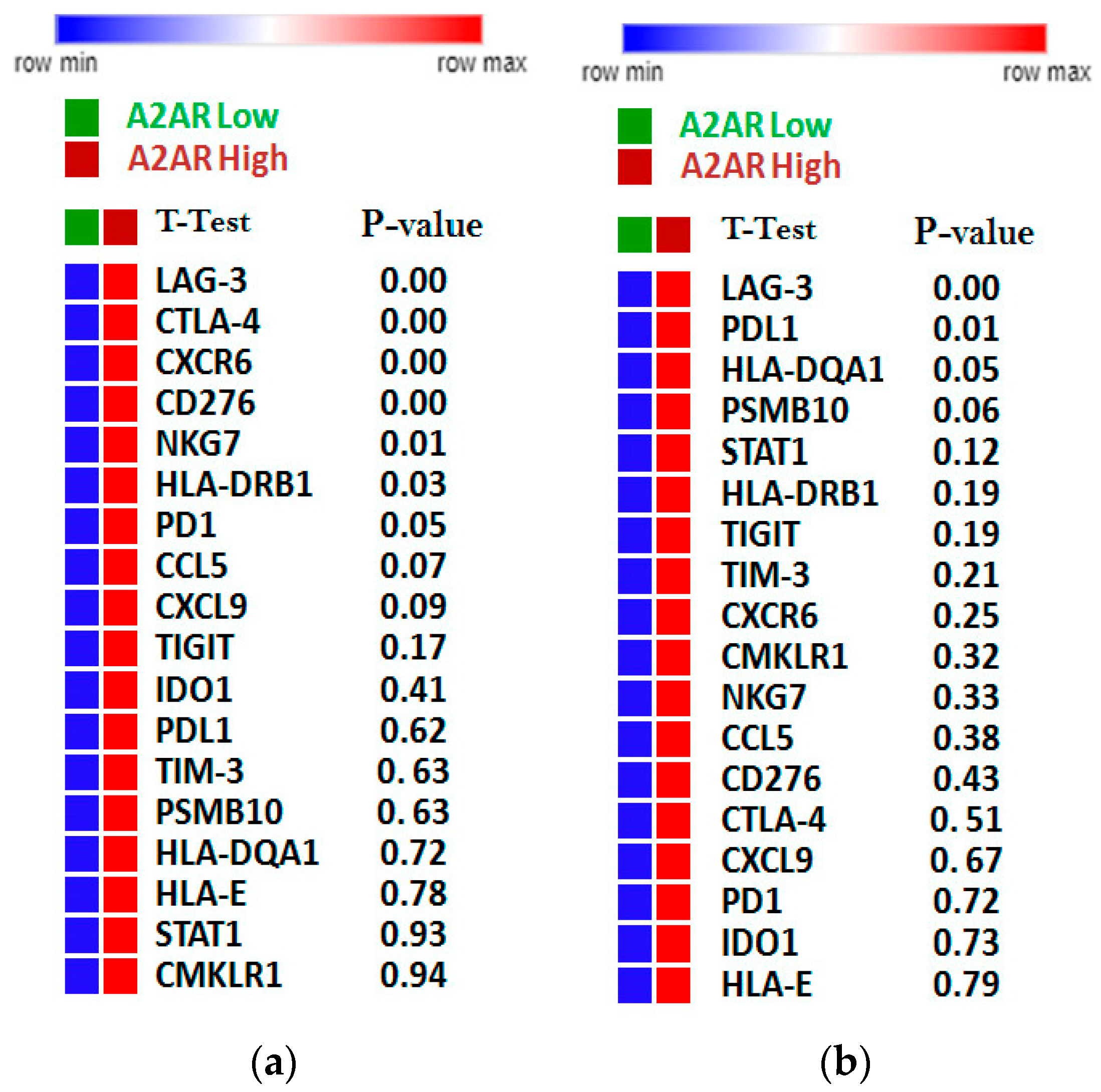
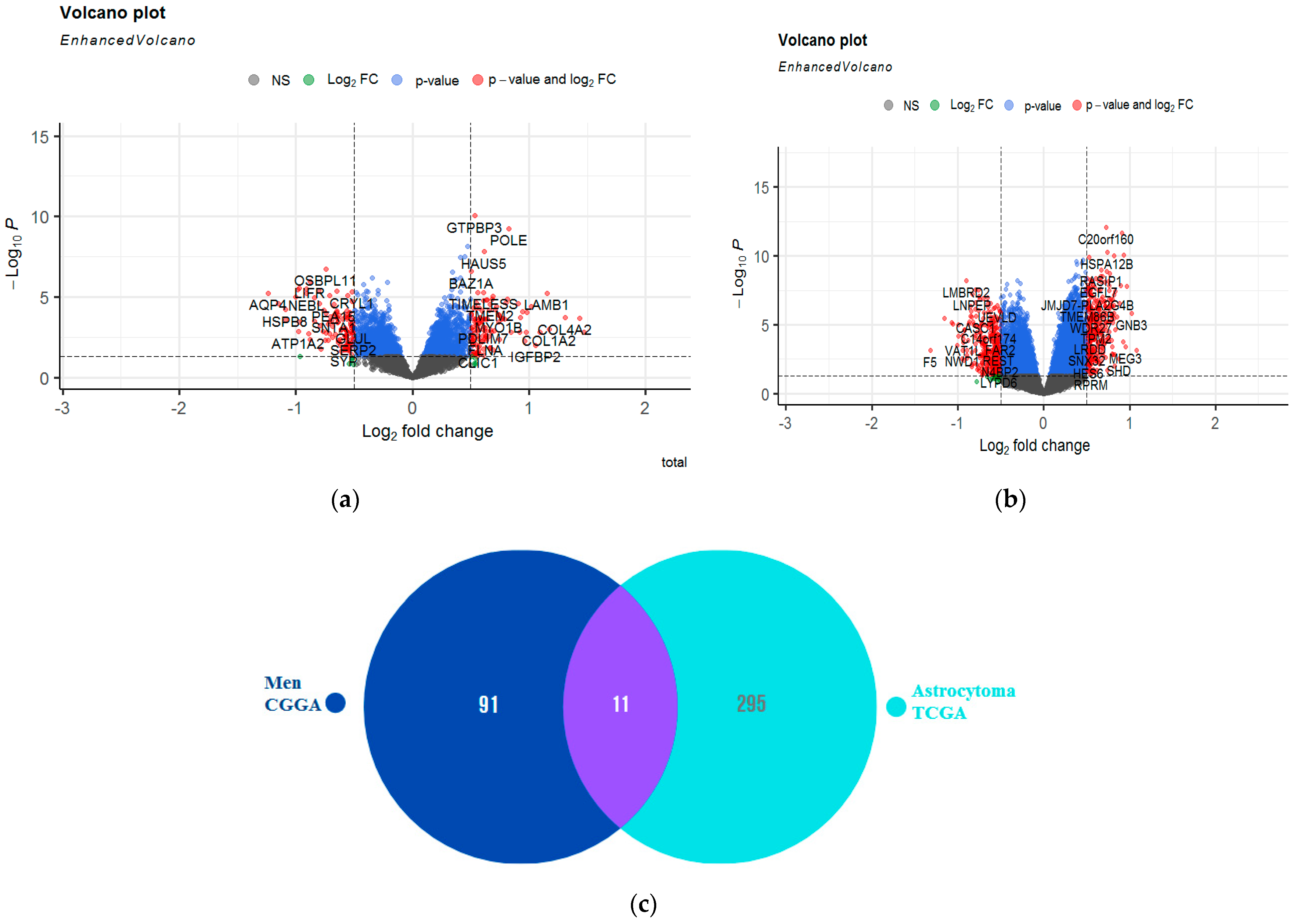
2.4. Identification of Molecular Profile Related to A2AR Expression in Male Glioma Patients (CGGA)
2.5. Correlation between A2AR Gene Expression and Prognosis of TCGA Glioma Patients
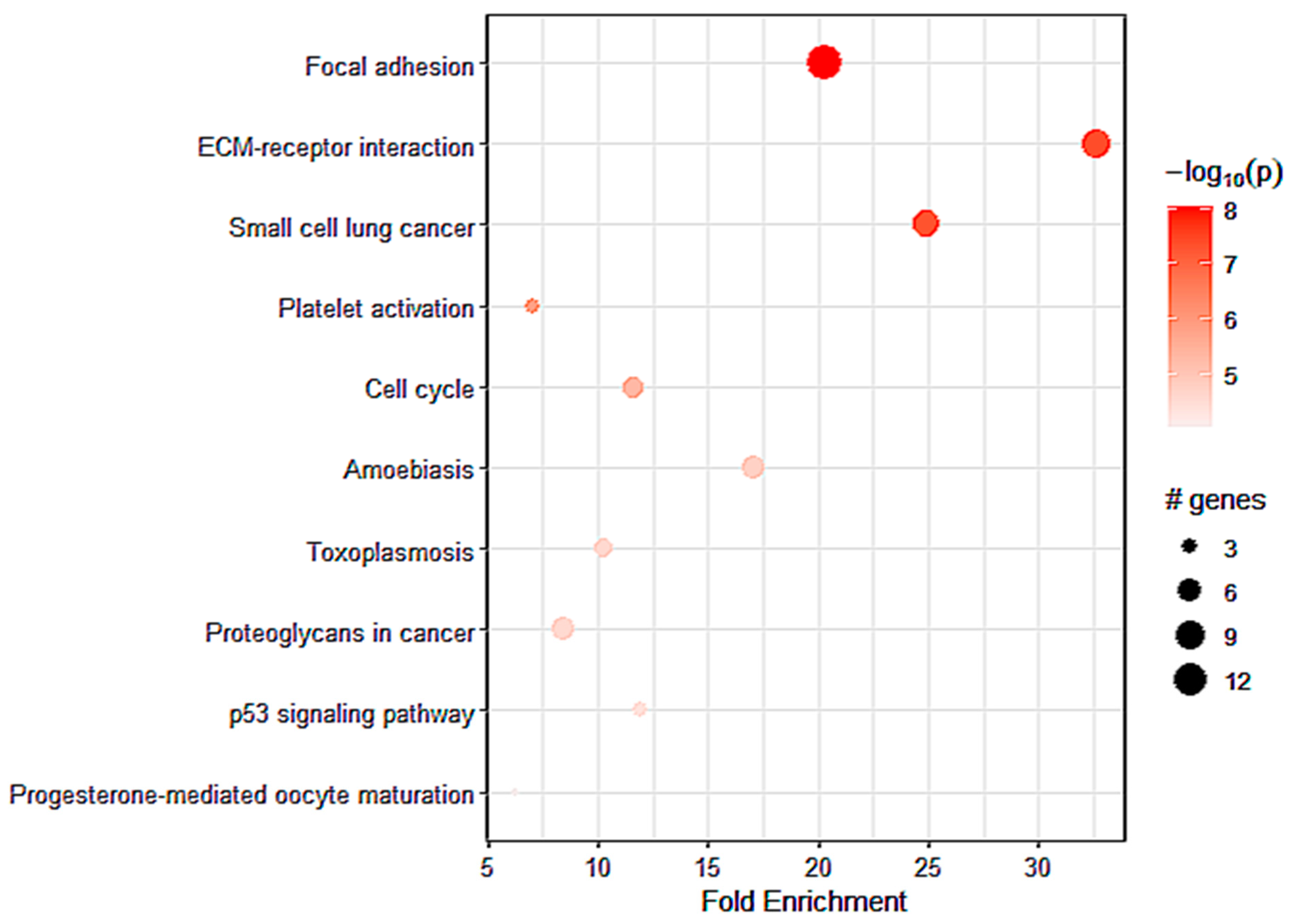
2.6. Identification of Pathways Related to A2AR Expression in Astrocytoma Glioma Patients (TCGA)
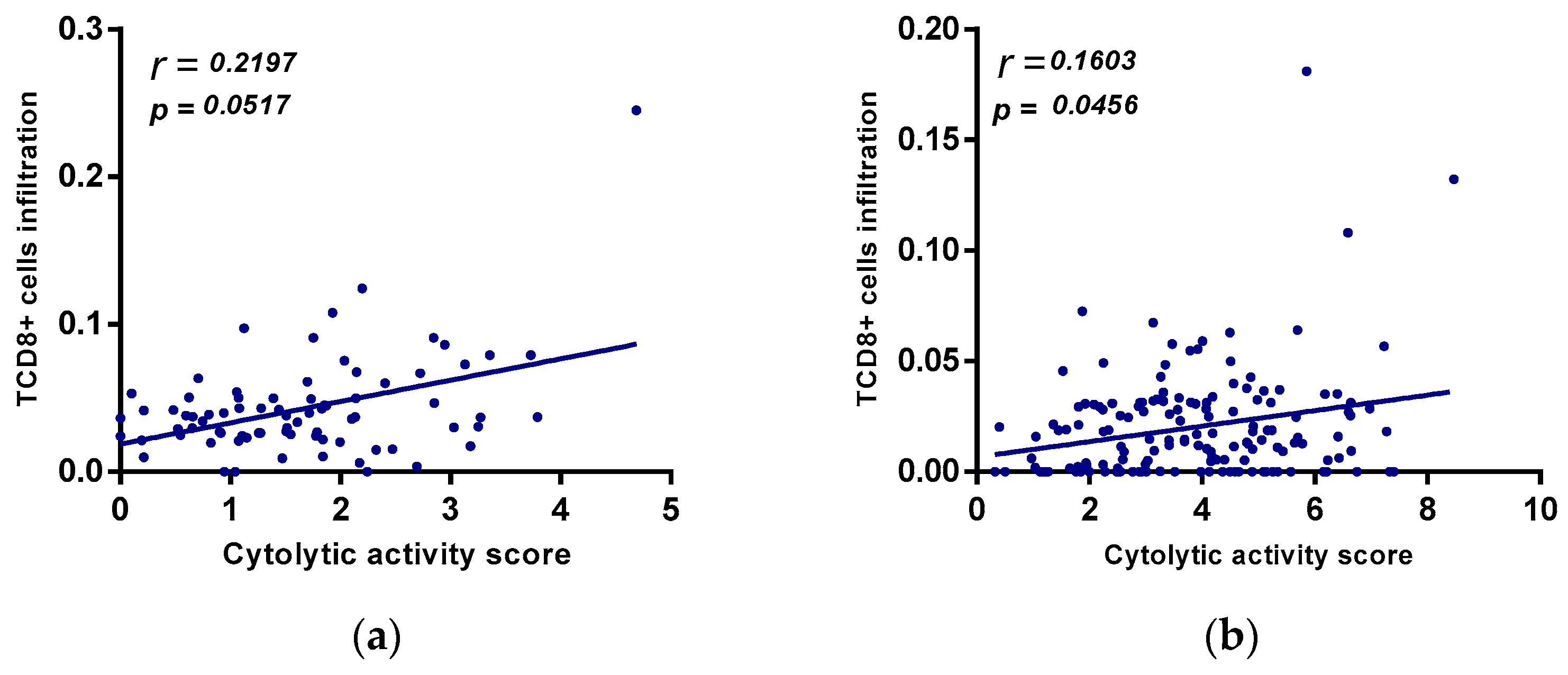
2.7. Relation between A2AR Expression and T Cell Exhaustion in Male CGGA and TCGA Astrocytoma Patients
2.8. Evaluation of A2AR Expression with Genes Responsible for Extracellular Adenosine Levels (CGGA Males and TCGA Astrocytoma)
3. Discussion
4. Materials and Methods
4.1. Patients and Samples
4.2. Total RNA Isolation and Reverse Transcription (RT)
4.3. RT-PCR (Real-Time—Polymerase Chain Reaction)
- β-actine: Forward: 5′ TGGAATCCTGTGGCATCCATGAAAC-3′
- β-actine: Reverse: 5′-TAAAACGCAGCTCAGTAACAGTCCG-3′
- A2AR: Forward:5′ ATC GCC ATT GAC CGC TAC AT3′
- A2AR: Reverse: 5′ GCT GAC CGC AGT TGT TCC A3′
4.4. TCGA and CGGA Clinical and Transcriptomic Data Collection
4.5. Differential Analyses
4.6. Enrichment Analyses
4.7. Analysis of Intra-Tumor Immune Cell Composition
4.8. Statistical Analysis, Survival, and Graph Generation
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Martínez-Vélez, N.; Gomez-Manzano, C.; Fueyo, J.; Patiño-García, A.; Alonso, M.M. Oncolytic Virotherapy for Gliomas: A Preclinical and Clinical Summary. In Gene Therapy in Neurological Disorders; Elsevier: Amsterdam, The Netherlands, 2018; pp. 357–384. [Google Scholar] [CrossRef]
- Li, C.; Chen, H.; Tan, Q.; Xie, C.; Zhan, W.; Sharma, A.; Sharma, H.S.; Zhang, Z. The Therapeutic and Neuroprotective Effects of an Antiepileptic Drug Valproic Acid in Glioma Patients. Prog. Brain Res. 2020, 258, 369–379. [Google Scholar] [CrossRef]
- AitSsi, S.; Chraa, D.; El Azhary, K.; Sahraoui, S.; Olive, D.; Badou, A. Prognostic Gene Expression Signature in Patients with Distinct Glioma Grades. Front. Immunol. 2021, 12, 685213. [Google Scholar] [CrossRef] [PubMed]
- Immune Checkpoint Blockade: A Common Denominator Approach to Cancer Therapy: Cancer Cell. Available online: https://www.cell.com/cancer-cell/fulltext/S1535-6108(15)00089-6 (accessed on 13 February 2023).
- John, L.B.; Devaud, C.; Duong, C.P.M.; Yong, C.S.; Beavis, P.A.; Haynes, N.M.; Chow, M.T.; Smyth, M.J.; Kershaw, M.H.; Darcy, P.K. Anti-PD-1 Antibody Therapy Potently Enhances the Eradication of Established Tumors by Gene-Modified T Cells. Clin. Cancer Res. 2013, 19, 5636–5646. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Han, H.; He, X.; Li, S.; Wu, C.; Yu, C.; Wang, S. Expression of the Galectin-9-Tim-3 Pathway in Glioma Tissues Is Associated with the Clinical Manifestations of Glioma. Oncol. Lett. 2016, 11, 1829–1834. [Google Scholar] [CrossRef] [PubMed]
- Harris-Bookman, S.; Mathios, D.; Martin, A.M.; Xia, Y.; Kim, E.; Xu, H.; Belcaid, Z.; Polanczyk, M.; Barberi, T.; Theodros, D.; et al. Expression of LAG-3 and Efficacy of Combination Treatment with Anti-LAG-3 and Anti-PD-1 Monoclonal Antibodies in Glioblastoma. Int. J. Cancer 2018, 143, 3201–3208. [Google Scholar] [CrossRef]
- TLR4/IFNγ Pathways Induce Tumor Regression via NOS II-Dependent NO and ROS Production in Murine Breast Cancer Models. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4910700/ (accessed on 13 February 2023).
- Merighi, S.; Battistello, E.; Giacomelli, L.; Varani, K.; Vincenzi, F.; Borea, P.A.; Gessi, S. Targeting A3 and A2A Adenosine Receptors in the Fight against Cancer. Expert Opin. Ther. Targets 2019, 23, 669–678. [Google Scholar] [CrossRef]
- Allard, D.; Turcotte, M.; Stagg, J. Targeting A2 Adenosine Receptors in Cancer. Immunol. Cell Biol. 2017, 95, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Mazziotta, C.; Rotondo, J.C.; Lanzillotti, C.; Campione, G.; Martini, F.; Tognon, M. Cancer Biology and Molecular Genetics of A3 Adenosine Receptor. Oncogene 2022, 41, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Chen, M.-N.; Du, J.; Liu, H.; He, Y.-J.; Li, G.-L.; Li, S.-Y.; Liu, W.-P.; Long, X.-Y. Differential Expression of Adenosine P1 Receptor ADORA1 and ADORA2A Associated with Glioma Development and Tumor-Associated Epilepsy. Neurochem. Res. 2016, 41, 1774–1783. [Google Scholar] [CrossRef] [PubMed]
- Fredholm, B.B.; IJzerman, A.P.; Jacobson, K.A.; Linden, J.; Müller, C.E. International Union of Basic and Clinical Pharmacology. LXXXI. Nomenclature and Classification of Adenosine Receptors—An Update. Pharmacol. Rev. 2011, 63, 1–34. [Google Scholar] [CrossRef]
- Willingham, S.B.; Ho, P.Y.; Hotson, A.; Hill, C.; Piccione, E.C.; Hsieh, J.; Liu, L.; Buggy, J.J.; McCaffery, I.; Miller, R.A. A2AR Antagonism with CPI-444 Induces Antitumor Responses and Augments Efficacy to Anti-PD-(L)1 and Anti-CTLA-4 in Preclinical Models. Cancer Immunol. Res. 2018, 6, 1136–1149. [Google Scholar] [CrossRef] [PubMed]
- Ohta, A.; Gorelik, E.; Prasad, S.J.; Ronchese, F.; Lukashev, D.; Wong, M.K.K.; Huang, X.; Caldwell, S.; Liu, K.; Smith, P.; et al. A2A Adenosine Receptor Protects Tumors from Antitumor T Cells. Proc. Natl. Acad. Sci. USA 2006, 103, 13132–13137. [Google Scholar] [CrossRef] [PubMed]
- Hypoxia-Adenosinergic Immunosuppression: Tumor Protection by T Regulatory Cells and Cancerous Tissue Hypoxia|Clinical Cancer Research. Available online: https://clincancerres.aacrjournals.org/content/14/19/5947 (accessed on 3 January 2022).
- A2A Receptors in Inflammation and Injury: Lessons Learned from Transgenic Animals-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/18160539/ (accessed on 13 February 2023).
- Rafii, S.; Ghallab, Y.E.; Ghouzlani, A.; Kandoussi, S.; Badou, A. Identification of Promising Antagonists of the Tumor Microenvironment Immunosuppressive Adenosine 2A Receptor through a Pharmacoinformatics- Based Approach. J. Bioinform. Syst. Biol. 2021, 4, 122–139. [Google Scholar] [CrossRef]
- Fredholm, B.B.; Arslan, G.; Halldner, L.; Kull, B.; Schulte, G.; Wasserman, W. Structure and Function of Adenosine Receptors and Their Genes. NaunynSchmiedebergs Arch. Pharmacol. 2000, 362, 364–374. [Google Scholar] [CrossRef]
- Xu, S.; Shao, Q.-Q.; Sun, J.-T.; Yang, N.; Xie, Q.; Wang, D.-H.; Huang, Q.-B.; Huang, B.; Wang, X.-Y.; Li, X.-G.; et al. Synergy between the Ectoenzymes CD39 and CD73 Contributes to Adenosinergic Immunosuppression in Human Malignant Gliomas. Neuro. Oncol. 2013, 15, 1160–1172. [Google Scholar] [CrossRef]
- Ott, M.; Tomaszowski, K.-H.; Marisetty, A.; Kong, L.-Y.; Wei, J.; Duna, M.; Blumberg, K.; Ji, X.; Jacobs, C.; Fuller, G.N.; et al. Profiling of Patients with Glioma Reveals the Dominant Immunosuppressive Axis Is Refractory to Immune Function Restoration. JCI Insight 2020, 5, e134386. [Google Scholar] [CrossRef]
- Ma, S.-R.; Deng, W.-W.; Liu, J.-F.; Mao, L.; Yu, G.-T.; Bu, L.-L.; Kulkarni, A.B.; Zhang, W.-F.; Sun, Z.-J. Blockade of Adenosine A2A Receptor Enhances CD8+ T Cells Response and Decreases Regulatory T Cells in Head and Neck Squamous Cell Carcinoma. Mol. Cancer 2017, 16, 99. [Google Scholar] [CrossRef] [PubMed]
- Young, A.; Ngiow, S.F.; Gao, Y.; Patch, A.-M.; Barkauskas, D.S.; Messaoudene, M.; Lin, G.; Coudert, J.D.; Stannard, K.A.; Zitvogel, L.; et al. A2AR Adenosine Signaling Suppresses Natural Killer Cell Maturation in the Tumor Microenvironment. Cancer Res. 2018, 78, 1003–1016. [Google Scholar] [CrossRef]
- Prognostic Impact of Adenosine Receptor 2 (A2aR) and Programmed Cell Death Ligand 1 (PD-L1) Expression in Colorectal Cancer-Recherche Google. Available online: https://www.google.com/search?q=Prognostic+Impact+of+Adenosine+Receptor+2+(A2aR)+and+Programmed+Cell+Death+Ligand+1+(PD-L1)+Expression+in+Colorectal+Cancer&rlz=1C1CHBD_frMA771MA771&oq=Prognostic+Impact+of+Adenosine+Receptor+2+(A2aR)+and+Programmed+Cell+Death+Ligand+1+(PD-L1)+Expression+in+Colorectal+Cancer&aqs=chrome..69i57.578j0j4&sourceid=chrome&ie=UTF-8 (accessed on 13 February 2023).
- Vijayan, D.; Young, A.; Teng, M.W.L.; Smyth, M.J. Targeting Immunosuppressive Adenosine in Cancer. Nat. Rev. Cancer 2017, 17, 709–724. [Google Scholar] [CrossRef]
- Dong, M.; Cioffi, G.; Wang, J.; Waite, K.A.; Ostrom, Q.T.; Kruchko, C.; Lathia, J.D.; Rubin, J.B.; Berens, M.E.; Connor, J.; et al. Sex Differences in Cancer Incidence and Survival: A Pan-Cancer Analysis. Cancer Epidemiol. Biomark. Prev. 2020, 29, 1389–1397. [Google Scholar] [CrossRef]
- Patterns and Disparities of Care in Glioblastoma-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/30740232/ (accessed on 13 February 2023).
- Ostrom, Q.T.; Rubin, J.B.; Lathia, J.D.; Berens, M.E.; Barnholtz-Sloan, J.S. Females Have the Survival Advantage in Glioblastoma. Neuro. Oncol. 2018, 20, 576–577. [Google Scholar] [CrossRef] [PubMed]
- Sex Differences in Time to Treat and Outcomes for Gliomas. Available online: https://www.frontiersin.org/articles/10.3389/fonc.2021.630597/full (accessed on 13 February 2023).
- Impact of Gender on the Survival of Patients with Glioblastoma. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6239255/ (accessed on 13 February 2023).
- Ostrom, Q.T.; Gittleman, H.; Xu, J.; Kromer, C.; Wolinsky, Y.; Kruchko, C.; Barnholtz-Sloan, J.S. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2009–2013. Neuro. Oncol. 2016, 18, v1–v75. [Google Scholar] [CrossRef] [PubMed]
- Jiang, H.; Cui, Y.; Wang, J.; Lin, S. Impact of Epidemiological Characteristics of Supratentorial Gliomas in Adults Brought about by the 2016 World Health Organization Classification of Tumors of the Central Nervous System. Oncotarget 2017, 8, 20354–20361. [Google Scholar] [CrossRef] [PubMed]
- Belousov, A.; Titov, S.; Shved, N.; Garbuz, M.; Malykin, G.; Gulaia, V.; Kagansky, A.; Kumeiko, V. The Extracellular Matrix and Biocompatible Materials in Glioblastoma Treatment. Front. Bioeng. Biotechnol. 2019, 7, 341. [Google Scholar] [CrossRef]
- Yin, W.; Zhu, H.; Tan, J.; Xin, Z.; Zhou, Q.; Cao, Y.; Wu, Z.; Wang, L.; Zhao, M.; Jiang, X.; et al. Identification of Collagen Genes Related to Immune Infiltration and Epithelial-Mesenchymal Transition in Glioma. Cancer Cell Int. 2021, 21, 276. [Google Scholar] [CrossRef]
- Analysis of Gene Expression Profiles Associated with Glioma Progression-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/25845910/ (accessed on 13 February 2023).
- Tsukumo, S.; Yasutomo, K. Regulation of CD8+ T Cells and Antitumor Immunity by Notch Signaling. Front. Immunol. 2018, 9, 101. [Google Scholar] [CrossRef]
- Yu, W.; Wang, Y.; Guo, P. Notch Signaling Pathway Dampens Tumor-Infiltrating CD8+ T Cells Activity in Patients with Colorectal Carcinoma. Biomed. Pharmacother. 2018, 97, 535–542. [Google Scholar] [CrossRef]
- Duval, F.; Mathieu, M.; Labrecque, N. Notch Controls Effector CD8+ T Cell Differentiation. Oncotarget 2015, 6, 21787–21788. [Google Scholar] [CrossRef]
- T-Cell Exhaustion Interrelates with Immune Cytolytic Activity to Shape the Inflamed Tumor Microenvironment. Available online: https://pubmed.ncbi.nlm.nih.gov/32222046/ (accessed on 13 February 2023).
- Johnson, B.J.; Costelloe, E.O.; Fitzpatrick, D.R.; Haanen, J.B.A.G.; Schumacher, T.N.M.; Brown, L.E.; Kelso, A. Single-Cell Perforin and Granzyme Expression Reveals the Anatomical Localization of Effector CD8+ T Cells in Influenza Virus-Infected Mice. Proc. Natl. Acad. Sci. USA 2003, 100, 2657–2662. [Google Scholar] [CrossRef]
- Herbst, R.S.; Soria, J.-C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive Correlates of Response to the Anti-PD-L1 Antibody MPDL3280A in Cancer Patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef]
- Puckett, D.L.; Alquraishi, M.; Chowanadisai, W.; Bettaieb, A. The Role of PKM2 in Metabolic Reprogramming: Insights into the Regulatory Roles of Non-Coding RNAs. Int. J. Mol. Sci. 2021, 22, 1171. [Google Scholar] [CrossRef]
- Zhang, W.; Li, H.; Ogando, D.G.; Li, S.; Feng, M.; Price, F.W.; Tennessen, J.M.; Bonanno, J.A. Glutaminolysis Is Essential for Energy Production and Ion Transport in Human Corneal Endothelium. EBioMedicine 2017, 16, 292–301. [Google Scholar] [CrossRef]
- Yoo, H.C.; Yu, Y.C.; Sung, Y.; Han, J.M. Glutamine Reliance in Cell Metabolism. Exp. Mol. Med. 2020, 52, 1496–1516. [Google Scholar] [CrossRef] [PubMed]
- Di Virgilio, F.; Sarti, A.C.; Falzoni, S.; De Marchi, E.; Adinolfi, E. Extracellular ATP and P2 Purinergic Signalling in the Tumour Microenvironment. Nat. Rev. Cancer 2018, 18, 601–618. [Google Scholar] [CrossRef]
- Li, X.-Y.; Moesta, A.K.; Xiao, C.; Nakamura, K.; Casey, M.; Zhang, H.; Madore, J.; Lepletier, A.; Aguilera, A.R.; Sundarrajan, A.; et al. Targeting CD39 in Cancer Reveals an Extracellular ATP- and Inflammasome-Driven Tumor Immunity. Cancer Discov. 2019, 9, 1754–1773. [Google Scholar] [CrossRef] [PubMed]
- Cui, M.; Bai, X.; Li, T.; Chen, F.; Dong, Q.; Zhao, Y.; Liu, X. Decreased Extracellular Adenosine Levels Lead to Loss of Hypoxia-Induced Neuroprotection after Repeated Episodes of Exposure to Hypoxia. PLoS ONE 2013, 8, e57065. [Google Scholar] [CrossRef] [PubMed]
- Feoktistov, I.; Ryzhov, S.; Zhong, H.; Goldstein, A.E.; Matafonov, A.; Zeng, D.; Biaggioni, I. Hypoxia Modulates Adenosine Receptors in Human Endothelial and Smooth Muscle Cells toward an A2B Angiogenic Phenotype. Hypertension 2004, 44, 649–654. [Google Scholar] [CrossRef]
- Steingold, J.M.; Hatfield, S.M. Targeting Hypoxia-A2A Adenosinergic Immunosuppression of Antitumor T Cells During Cancer Immunotherapy. Front. Immunol. 2020, 11, 2357. [Google Scholar] [CrossRef]
- Bowser, J.L.; Phan, L.H.; Eltzschig, H.K. The Hypoxia–Adenosine Link during Intestinal Inflammation. J. Immunol. 2018, 200, 897–907. [Google Scholar] [CrossRef]
- Boison, D.; Yegutkin, G.G. Adenosine Metabolism: Emerging Concepts for Cancer Therapy. Cancer Cell 2019, 36, 582–596. [Google Scholar] [CrossRef]
- Dzeja, P.; Terzic, A. Adenylate Kinase and AMP Signaling Networks: Metabolic Monitoring, Signal Communication and Body Energy Sensing. Int. J. Mol. Sci. 2009, 10, 1729–1772. [Google Scholar] [CrossRef] [PubMed]
- Kristiansen, S.B.; Skovsted, G.F.; Berchtold, L.A.; Radziwon-Balicka, A.; Dreisig, K.; Edvinsson, L.; Sheykhzade, M.; Haanes, K.A. Role of Pannexin and Adenosine Triphosphate (ATP) Following Myocardial Ischemia/Reperfusion. Scand. Cardiovasc. J. 2018, 52, 340–343. [Google Scholar] [CrossRef] [PubMed]
- Cymer, M.; Adamiak, M.; Skoda, M.; Libura, M.A.; Urbanowska, E.; Kucia, M.; Ratajczak, M.Z. Novel Evidence That the Pannexin 1 Channel Is Involved in Adenosine Triphosphate (ATP) Release from Cells for Optimal Mobilization of Hematopoietic Stem Progenitor Cells, and the Pannexin 1 SNP 5 (Rs3020015) T/C Polymorphism Characterizes Poor Mobilizer Status in Patients. Blood 2019, 134, 3248. [Google Scholar] [CrossRef]
- Shan, Y.; Ni, Y.; Gao, Z. Pannexin-1 Channel Regulates ATP Release in Epilepsy. Neurochem. Res. 2020, 45, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Dvoriantchikova, G.; Pronin, A.; Kurtenbach, S.; Toychiev, A.; Chou, T.-H.; Yee, C.W.; Prindeville, B.; Tayou, J.; Porciatti, V.; Sagdullaev, B.T.; et al. Pannexin 1 Sustains the Electrophysiological Responsiveness of Retinal Ganglion Cells. Sci. Rep. 2018, 8, 5797. [Google Scholar] [CrossRef]
- Feig, J.L.; Mediero, A.; Corciulo, C.; Liu, H.; Zhang, J.; Perez-Aso, M.; Picard, L.; Wilder, T.; Cronstein, B. The Antiviral Drug Tenofovir, an Inhibitor of Pannexin-1-Mediated ATP Release, Prevents Liver and Skin Fibrosis by Downregulating Adenosine Levels in the Liver and Skin. PLoS ONE 2017, 12, e0188135. [Google Scholar] [CrossRef]
- Boison, D. Adenosine Kinase, Epilepsy and Stroke: Mechanisms and Therapies. Trends Pharmacol. Sci. 2006, 27, 652–658. [Google Scholar] [CrossRef]
- Park, J.; Gupta, R.S. Adenosine Metabolism, Adenosine Kinase, and Evolution. In Adenosine: A Key Link between Metabolism and Brain Activity; Masino, S., Boison, D., Eds.; Springer: New York, NY, USA, 2013; pp. 23–54. [Google Scholar] [CrossRef]
- Wang, H.; Gonzalez-Garcia, I.; Traba, J.; Jain, S.; Conteh, S.; Shin, D.-M.; Qi, C.; Gao, Y.; Sun, J.; Kang, S.; et al. ATP-Degrading ENPP1 Is Required for Survival (or Persistence) of Long-Lived Plasma Cells. Sci. Rep. 2017, 7, 17867. [Google Scholar] [CrossRef]
- Quaglino, D.; Boraldi, F.; Lofaro, F.D. Chapter Eight-The Biology of Vascular Calcification. In International Review of Cell and Molecular Biology; Galluzzi, L., Ed.; Academic Press: Cambridge, MA, USA, 2020; Volume 354, pp. 261–353. [Google Scholar] [CrossRef]
- Vigneswaran, K.; Neill, S.; Hadjipanayis, C.G. Beyond the World Health Organization Grading of Infiltrating Gliomas: Advances in the Molecular Genetics of Glioma Classification. Ann. Transl. Med. 2015, 3, 95. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A Summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Ghouzlani, A.; Lakhdar, A.; Rafii, S.; Karkouri, M.; Badou, A. The Immune Checkpoint VISTA Exhibits High Expression Levels in Human Gliomas and Associates with a Poor Prognosis. Sci. Rep. 2021, 11, 21504. [Google Scholar] [CrossRef]
- Ghouzlani, A.; Rafii, S.; Karkouri, M.; Lakhdar, A.; Badou, A. The Promising IgSF11 Immune Checkpoint Is Highly Expressed in Advanced Human Gliomas and Associates to Poor Prognosis. Front. Oncol. 2020, 10, 608609. [Google Scholar] [CrossRef] [PubMed]
- Abstract|High Expression Levels of Foxp3 and VISTA in Advanced Human Gliomas and Impact on Patient’s Prognosis. Available online: https://www.fortunejournals.com/abstract/high-expression-levels-of-foxp3-and-vista-in-advanced-human-gliomas-and-impact-on-patientrsquos-prognosis-1825.html (accessed on 13 February 2023).
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Tomczak, K.; Czerwińska, P.; Wiznerowicz, M. The Cancer Genome Atlas (TCGA): An Immeasurable Source of Knowledge. Contemp. Oncol. 2015, 19, A68–A77. [Google Scholar] [CrossRef]
- Zhao, Z.; Zhang, K.N.; Wang, Q.; Li, G.; Zeng, F.; Zhang, Y.; Wu, F.; Chai, R.; Wang, Z.; Zhang, C.; et al. Chinese Glioma Genome Atlas (CGGA): A Comprehensive Resource with Functional Genomic Data from Chinese Glioma Patients. Genom. Proteom. Bioinform. 2021, 19, 1–12, ISSN 1672-0229. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.E.; Phipson, B.; Wu, D.I.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and PowerfulApproach to Multiple Testing. J. R. Stat. SocietySer. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Ulgen, E.; Ozisik, O.; Sezerman, O.U. pathfindR: An R Package for Comprehensive Identification of Enriched Pathways in Omics Data Through Active Subnetworks. Front. Genet. 2019, 10, 858. [Google Scholar] [CrossRef]
- Newman, A.M.; Steen, C.B.; Liu, C.L.; Gentles, A.J.; Chaudhuri, A.A.; Scherer, F.; Khodadoust, M.S.; Esfahani, M.S.; Luca, B.A.; Steiner, D.; et al. Determining cell type abundance and expression from bulk tissues with digital cytometry. Nat. Biotechnol. 2019, 37, 773–782. [Google Scholar] [CrossRef]
- Newman, A.M.; Liu, C.L.; Green, M.R.; Gentles, A.J.; Feng, W.; Xu, Y.; Hoang, C.D.; Diehn, M.; Alizadeh, A.A. Robust enumeration of cell subsets from tissue expression profiles. Nat. Methods 2015, 12, 453–457. [Google Scholar] [CrossRef]
- Gao, Z.; Tao, Y.; Lai, Y.; Wang, Q.; Li, Z.; Peng, S.; Chen, J.; Cai, W.; Li, K.; Huang, H. Immune Cytolytic Activity as an Indicator of Immune Checkpoint Inhibitors Treatment for Prostate Cancer. Front. Bioeng. Biotechnol. 2020, 8, 930. [Google Scholar] [CrossRef] [PubMed]
- Rooney, M.S.; Shukla, S.A.; Wu, C.J.; Getz, G.; Hacohen, N. Molecular and Genetic Properties of Tumors Associated with Local Immune Cytolytic Activity. Cell 2015, 160, 48–61. [Google Scholar] [CrossRef] [PubMed]
| Variable | Case(%) | p-Value |
|---|---|---|
| (n = 52) | ||
| Gender | ||
| •Male | 31 (59.62) | 0.0963 |
| •Female | 21 (40.38) | |
| Age | ||
| •≤34 years | 25 (52.083) | 0.3754 |
| •˃34 years | 23 (47.916) | |
| WHO Grade | ||
| •I | 17 (33) | 0.5047 |
| •II | 9 (17) | |
| •III | 6 (12) | |
| •IV | 20 (38) | |
| Histological Type | ||
| •Astrocytomas | 19 (38.775) | 0.3109 |
| •Oligodendrogliomas | 3 (6.122) | |
| •Ependymomas | 7 (14.285) | |
| •Glioblastoma | 20 (40.816) | |
| Smoking status | ||
| •Yes | 11 (26,19) | 0.1985 |
| •No | 31 (73,80) | |
| IDH mutation | ||
| •WT | 11 (73,33) | 0.7824 |
| •IDH mutant | 4 (26,66) |
| Variable | Cas (Low/High) | p-Value |
|---|---|---|
| Overall survival | 9/8 | 0.8232 |
| Gender | ||
| •Male | 4/4 | 0.0062 |
| •Female | 5/4 | 0.0532 |
| Age | ||
| •≤34 years | 4/4 | 0.8629 |
| •˃34 years | 4/4 | 0.7297 |
| WHO Grade | ||
| •LGG | 4/4 | 0.3173 |
| •GBM | 5/4 | 0.8781 |
| Nb Low/High | p-Value | |
|---|---|---|
| Age | ||
| ≤44 years | 62/58 | 0.0239 |
| ˃44 years | 51/51 | 0.7516 |
| Gender | ||
| Male | 69/69 | 0.0029 |
| Female | 43/41 | 0.5453 |
| WHO Grade | ||
| GII | 45/45 | 0.4268 |
| GIII | 24/23 | 0.5753 |
| GIV or GBM | 43/42 | 0.8806 |
| Histological type | ||
| Oligodendroglioma | 22/22 | 0.704 |
| Astrocytoma | 23/23 | 0.887 |
| IDH mutation | ||
| IDH mutant | 57/37 | 0.1249 |
| WT | 55/54 | 0.6984 |
| Enrichment FDR | Hallmark Enrichment |
|---|---|
| 1.3 × 10−20 | Hallmark epithelial mesenchymal transition |
| 2 × 10−4 | Hallmark TGF beta signaling |
| 1.6 × 10−2 | Hallmark angiogenesis |
| 1.4 × 10−6 | Hallmark apoptosis |
| 7.1 × 10−7 | Hallmark G2M checkpoint |
| Nb Low/High | p Value | |
|---|---|---|
| Age | ||
| ≤44 years | 157/157 | 0.6498 |
| ˃44 years | 152/152 | 0.1307 |
| Gender | ||
| •Male | 175/173 | 0.1577 |
| •Female | 135/135 | 0.6536 |
| Grade | ||
| GII | 124/124 | 0.2578 |
| GIII | 132/132 | 0.5293 |
| GIV or GBM | 75/73 | 0.4233 |
| Histological type | ||
| Oligodendroglioma | 95/94 | 0.1064 |
| Oligoastrocytoma | 65/65 | 0.684 |
| Astrocytoma | 97/97 | 0.005 |
| IDH status | ||
| IDH mutant | 212/210 | 0.958 |
| WT | 99/98 | 0.248 |
| Enrichment FDR | Hallmark Enrichment |
|---|---|
| 4.2 × 10−3 | Hallmark myogenesis |
| Enrichment FDR | Pathways Involved |
|---|---|
| 2.385 × 10−7 | Pyruvate kinase deficiency |
| 0.01043 | Glutaminolysis and cancer |
| 0.01043 | Hyperphenylalanemia |
| 0.01043 | Ibutilide action pathway |
| 0.01043 | Metabolism of nucleotide sugars |
| Males CGGA | Astrocytoma TGGA | |||
|---|---|---|---|---|
| p | r | p | r | |
| HIF1A | 0.0452 | 0.1684 | 0.0005 | −0.2464 |
| CD39 | 0.3698 | 0.07582 | 0.9703 | 0.002686 |
| NT5E | 0.2907 | 0.08927 | 0.3233 | 0.07128 |
| ENPP1 | 0.5992 | 0.04513 | <0.0001 | −0.2757 |
| CD38 | 0.0095 | −0.217 | 0.0234 | −0.1627 |
| BST1 | 0.9523 | 0.005137 | 0.7136 | 0.02652 |
| AK1 | 0.0465 | −0.1698 | 0.8975 | −0.009307 |
| ADA | 0.0001 | 0.3198 | 0.0468 | 0.1429 |
| PNP | 0.7196 | −0.03083 | 0.5284 | −0.04554 |
| ADK | 0.9816 | −0.001977 | 0.0483 | 0.142 |
| Connexin | 0.0059 | −0.2299 | 0.018 | −0.1698 |
| Pannexin | 0.0782 | 0.1483 | <0.0001 | −0.2851 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rafii, S.; Ghouzlani, A.; Naji, O.; Ait Ssi, S.; Kandoussi, S.; Lakhdar, A.; Badou, A. A2AR as a Prognostic Marker and a Potential Immunotherapy Target in Human Glioma. Int. J. Mol. Sci. 2023, 24, 6688. https://doi.org/10.3390/ijms24076688
Rafii S, Ghouzlani A, Naji O, Ait Ssi S, Kandoussi S, Lakhdar A, Badou A. A2AR as a Prognostic Marker and a Potential Immunotherapy Target in Human Glioma. International Journal of Molecular Sciences. 2023; 24(7):6688. https://doi.org/10.3390/ijms24076688
Chicago/Turabian StyleRafii, Soumaya, Amina Ghouzlani, Oumayma Naji, Saadia Ait Ssi, Sarah Kandoussi, Abdelhakim Lakhdar, and Abdallah Badou. 2023. "A2AR as a Prognostic Marker and a Potential Immunotherapy Target in Human Glioma" International Journal of Molecular Sciences 24, no. 7: 6688. https://doi.org/10.3390/ijms24076688
APA StyleRafii, S., Ghouzlani, A., Naji, O., Ait Ssi, S., Kandoussi, S., Lakhdar, A., & Badou, A. (2023). A2AR as a Prognostic Marker and a Potential Immunotherapy Target in Human Glioma. International Journal of Molecular Sciences, 24(7), 6688. https://doi.org/10.3390/ijms24076688





