The Role of the Piezo1 Mechanosensitive Channel in the Musculoskeletal System
Abstract
1. Introduction
2. Mechanosensation—Mechanosensitive and Mechanically Activated Channels
Mechanosensitive Ion Channels
3. Piezo Channels in the Animal Kingdom
4. Structure and Function of Piezo1
4.1. Channel Structure
4.2. Regulation
4.3. Pharmacology
4.3.1. Agonists
4.3.2. Antagonists
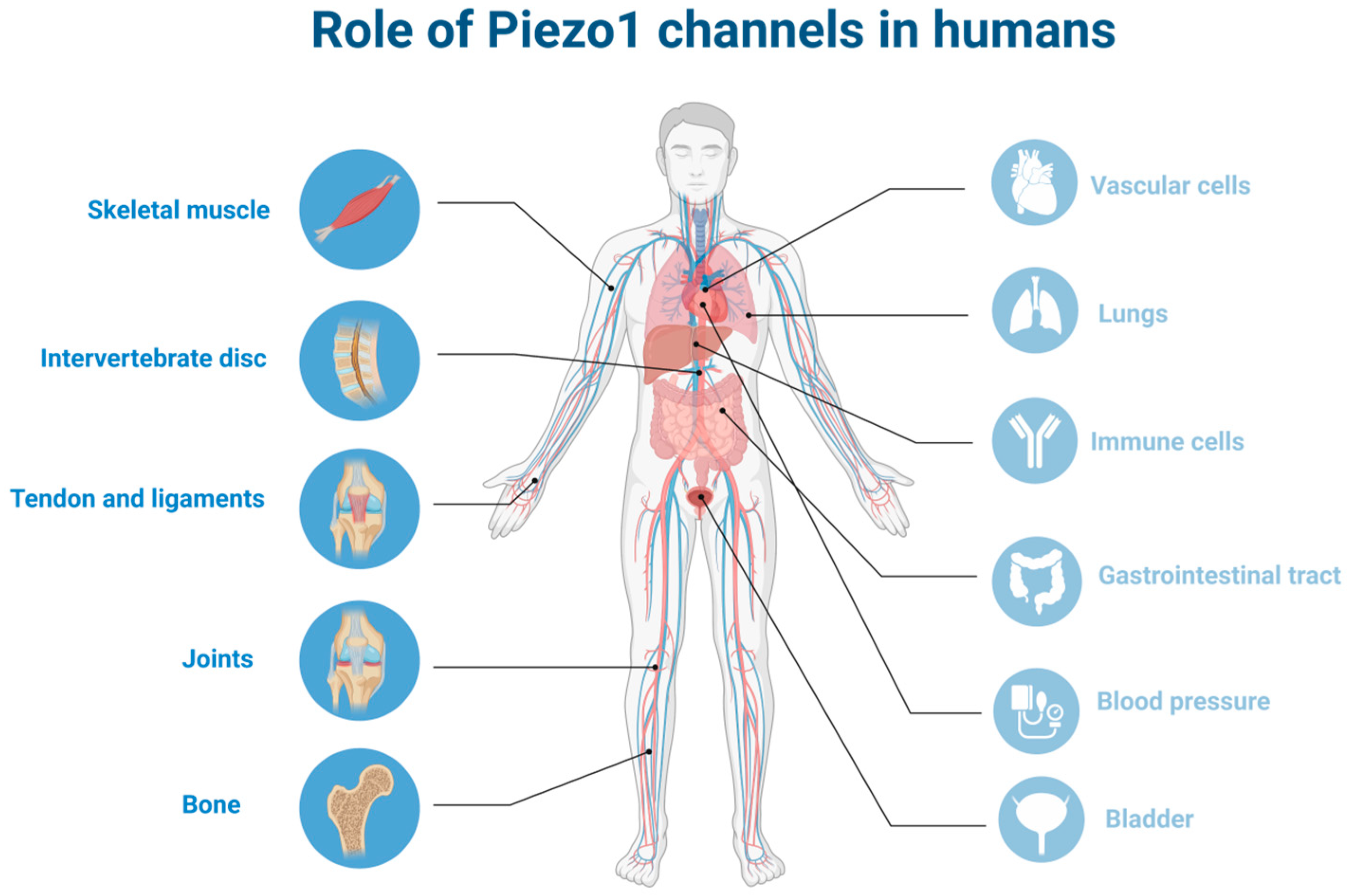
5. Piezo1 Channels in the Musculoskeletal System
5.1. Skeletal Muscle
5.2. Bone
5.3. Tendons and Ligaments
5.4. Joints
Piezo1 in Chondrocytes
5.5. Intervertebral Discs
6. Disorders in the Musculoskeletal System Associated with Piezo1 Channels
7. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Coste, B.; Mathur, J.; Schmidt, M.; Earley, T.J.; Ranade, S.; Petrus, M.J.; Dubin, A.E.; Patapoutian, A. Activated Cation Channels. Science 2010, 330, 7–12. [Google Scholar]
- Li, J.; Hou, B.; Tumova, S.; Muraki, K.; Bruns, A.; Ludlow, M.J.; Sedo, A.; Hyman, A.J.; McKeown, L.; Young, R.S.; et al. Piezo1 integration of vascular architecture with physiological force. Nature 2014, 515, 279–282. [Google Scholar] [CrossRef] [PubMed]
- Ranade, S.S.; Qiu, Z.; Woo, S.H.; Hur, S.S.; Murthy, S.E.; Cahalan, S.M.; Xu, J.; Mathur, J.; Bandell, M.; Coste, B.; et al. Piezo1, a mechanically activated ion channel, is required for vascular development in mice. Proc. Natl. Acad. Sci. USA 2014, 111, 10347–10352. [Google Scholar] [CrossRef] [PubMed]
- Douguet, D.; Patel, A.; Xu, A.; Vanhoutte, P.M.; Honoré, E. Piezo Ion Channels in Cardiovascular Mechanobiology. Trends Pharmacol. Sci. 2019, 40, 956–970. [Google Scholar] [CrossRef] [PubMed]
- Retailleau, K.; Duprat, F.; Arhatte, M.; Ranade, S.S.; Peyronnet, R.; Martins, J.R.; Jodar, M.; Moro, C.; Offermanns, S.; Feng, Y.; et al. Piezo1 in Smooth Muscle Cells Is Involved in Hypertension-Dependent Arterial Remodeling. Cell Rep. 2015, 13, 1161–1171. [Google Scholar] [CrossRef]
- Szabó, L.; Balogh, N.; Tóth, A.; Angyal, Á.; Gönczi, M.; Csiki, D.M.; Tóth, C.; Balatoni, I.; Jeney, V.; Csernoch, L.; et al. The mechanosensitive Piezo1 channels contribute to the arterial medial calcification. Front. Physiol. 2022, 13, 2338. [Google Scholar] [CrossRef]
- Swain, S.M.; Romac, J.M.J.; Vigna, S.R.; Liddle, R.A. Piezo1-mediated stellate cell activation causes pressure-induced pancreatic fibrosis in mice. JCI Insight 2022, 7, e158288. [Google Scholar] [CrossRef]
- Liu, H.; Hu, J.; Zheng, Q.; Feng, X.; Zhan, F.; Wang, X.; Xu, G.; Hua, F. Piezo1 Channels as Force Sensors in Mechanical Force-Related Chronic Inflammation. Front. Immunol. 2022, 13, 816149. [Google Scholar] [CrossRef]
- Xu, Y.; Bai, T.; Xiong, Y.; Liu, C.; Liu, Y.; Hou, X.; Song, J. Mechanical stimulation activates Piezo1 to promote mucin2 expression in goblet cells. J. Gastroenterol. Hepatol. 2021, 36, 3127–3139. [Google Scholar] [CrossRef]
- Rode, B.; Shi, J.; Endesh, N.; Drinkhill, M.J.; Webster, P.J.; Lotteau, S.J.; Bailey, M.A.; Yuldasheva, N.Y.; Ludlow, M.J.; Cubbon, R.M.; et al. Piezo1 channels sense whole body physical activity to reset cardiovascular homeostasis and enhance performance. Nat. Commun. 2017, 8, 350. [Google Scholar] [CrossRef]
- Eisenhoffer, G.T.; Loftus, P.D.; Yoshigi, M.; Otsuna, H.; Chien, C.B.; Morcos, P.A.; Rosenblatt, J. Crowding induces live cell extrusion to maintain homeostatic cell numbers in epithelia. Nature 2012, 484, 546–549. [Google Scholar] [CrossRef] [PubMed]
- Solis, A.G.; Bielecki, P.; Steach, H.R.; Sharma, L.; Harman, C.C.D.; Yun, S.; de Zoete, M.R.; Warnock, J.N.; To, S.D.F.; York, A.G.; et al. Mechanosensation of cyclical force by PIEZO1 is essential for innate immunity. Nature 2019, 573, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Lukacs, V.; Mathur, J.; Mao, R.; Bayrak-Toydemir, P.; Procter, M.; Cahalan, S.M.; Kim, H.J.; Bandell, M.; Longo, N.; Day, R.W.; et al. Impaired PIEZO1 function in patients with a novel autosomal recessive congenital lymphatic dysplasia. Nat. Commun. 2015, 6, 8329. [Google Scholar] [CrossRef] [PubMed]
- Cox, C.D.; Bavi, N.; Martinac, B. Cytoskeleton-Associated Proteins Modulate the Tension Sensitivity of Piezo1. Biophys. J. 2018, 114, 111a. [Google Scholar] [CrossRef]
- Lehtonen, J.Y.A.; Kinnunen, P.K.J. Phospholipase A2 as a Mechanosensor. Biophys. Soc. 1995, 68, 1888–1894. [Google Scholar] [CrossRef] [PubMed]
- Storch, U.; Mederos, M.; Gudermann, T. G protein-mediated stretch reception. Am. J. Physiol. Heart Circ. Physiol. 2012, 302, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Arnadóttir, J.; Chalfie, M. Eukaryotic mechanosensitive channels. Annu. Rev. Biophys. 2010, 39, 111–137. [Google Scholar] [CrossRef]
- Ranade, S.S.; Syeda, R.; Patapoutian, A. Mechanically Activated Ion Channels. Neuron 2015, 87, 1162–1179. [Google Scholar] [CrossRef]
- Cox, C.D.; Bavi, N.; Martinac, B. Bacterial Mechanosensors. Annu. Rev. Physiol. 2018, 80, 71–93. [Google Scholar] [CrossRef]
- Chalfie, M. Neurosensory mechanotransduction. Nat. Rev. Mol. Cell Biol. 2009, 10, 44–52. [Google Scholar] [CrossRef]
- Teng, J.; Loukin, S.; Anishkin, A.; Kung, C. The force-from-lipid (FFL) principle of mechanosensitivity, at large and in elements. Pflug. Arch. Eur. J. Physiol. 2015, 467, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.H.; Grandl, J. Mechanical sensitivity of Piezo1 ion channels can be tuned by cellular membrane tension. eLife 2015, 4, e12088. [Google Scholar] [CrossRef]
- Gaub, B.M.; Müller, D.J. Mechanical Stimulation of Piezo1 Receptors Depends on Extracellular Matrix Proteins and Directionality of Force. Nano Lett. 2017, 17, 2064–2072. [Google Scholar] [CrossRef]
- Sukharev, S.; Martinac, B.; Arshavsky, V.Y.; Kung, C. Two Types of Mechanosensitive Channels in the Escherichia coli Cell Envelope: Solubilization and Functional Reconstitution. Biophys. J. 1993, 65, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Guharay, B.Y.F.; Sachs, F. Stretch-Activated Single Ion Channel Currents in Tissue-Cultured Embryonic Chick Skeletal Muscle by. J. Physiol. 1984, 352, 685–701. [Google Scholar] [CrossRef]
- Brohawn, S.G.; Su, Z.; MacKinnon, R. Mechanosensitivity is mediated directly by the lipid membrane in TRAAK and TREK1 K+ channels. Proc. Natl. Acad. Sci. USA 2014, 111, 3614–3619. [Google Scholar] [CrossRef] [PubMed]
- Servin-Vences, M.R.; Moroni, M.; Lewin, G.R.; Poole, K. Direct measurement of TRPV4 and PIEZO1 activity reveals multiple mechanotransduction pathways in chondrocytes. eLife 2017, 6, e21074. [Google Scholar] [CrossRef]
- Ben-Shahar, Y. Sensory Functions for Degenerin/Epithelial Sodium Channels (DEG/ENaC). Adv. Genet. 2011, 76, 1–26. [Google Scholar] [CrossRef]
- Murthy, S.E.; Dubin, A.E.; Whitwam, T.; Jojoa-Cruz, S.; Cahalan, S.M.; Mousavi, S.A.R.; Ward, A.B.; Patapoutian, A. OSCA/TMEM63 are an evolutionarily conserved family of mechanically activated ion channels. eLife 2018, 2018, e41844. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, D.; Kang, Y.; Wu, J.X.; Yao, F.; Pan, C.; Yan, Z.; Song, C.; Chen, L. Structure of the mechanosensitive OSCA channels. Nat. Struct. Mol. Biol. 2018, 25, 850–858. [Google Scholar] [CrossRef]
- Syeda, R.; Florendo, M.N.; Cox, C.D.; Kefauver, J.M.; Santos, J.S.; Martinac, B.; Patapoutian, A. Piezo1 Channels Are Inherently Mechanosensitive. Cell Rep. 2016, 17, 1739–1746. [Google Scholar] [CrossRef] [PubMed]
- Kefauver, J.M.; Ward, A.B.; Patapoutian, A. Discoveries in structure and physiology of mechanically activated ion channels. Nature 2020, 587, 567–576. [Google Scholar] [CrossRef]
- Morris, C.E.; Prikryl, E.A.; Joós, B. Mechanosensitive gating of Kv channels. PLoS ONE 2015, 10, e0118335. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, B.; Tabarean, I.V.; Juranka, P.; Morris, C.E. Mechanosensitivity of N-type calcium channel currents. Biophys. J. 2002, 83, 2560–2574. [Google Scholar] [CrossRef] [PubMed]
- Beyder, A.; Rae, J.L.; Bernard, C.; Strege, P.R.; Sachs, F.; Farrugia, G. Mechanosensitivity of Nav1.5, a voltage-sensitive sodium channel. J. Physiol. 2010, 588, 4969–4985. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.E.; Juranka, P.F. Nav channel mechanosensitivity: Activation and inactivation accelerate reversibly with stretch. Biophys. J. 2007, 93, 822–833. [Google Scholar] [CrossRef]
- Lin, W.; Laitko, U.; Juranka, P.F.; Morris, C.E. Dual stretch responses of mHCN2 pacemaker channels: Accelerated activation, accelerated deactivation. Biophys. J. 2007, 92, 1559–1572. [Google Scholar] [CrossRef]
- Jia, C. Revealing the hiden mystery of Piezo: A phylogenetic study. IOP Conf. Ser. Earth Environ. Sci. 2021, 657, 012118. [Google Scholar] [CrossRef]
- Fang, X.Z.; Zhou, T.; Xu, J.Q.; Wang, Y.X.; Sun, M.M.; He, Y.J.; Pan, S.W.; Xiong, W.; Peng, Z.K.; Gao, X.H.; et al. Structure, kinetic properties and biological function of mechanosensitive Piezo channels. Cell Biosci. 2021, 11, 13. [Google Scholar] [CrossRef]
- Bagriantsev, S.N.; Gracheva, E.O.; Gallagher, P.G. Piezo proteins: Regulators of mechanosensation and other cellular processes. J. Biol. Chem. 2014, 289, 31673–31681. [Google Scholar] [CrossRef]
- Parpaite, T.; Coste, B. Piezo channels. Curr. Biol. 2017, 27, R250–R252. [Google Scholar] [CrossRef]
- Saotome, K.; Murthy, S.E.; Kefauver, J.M.; Whitwam, T.; Patapoutian, A.; Ward, A.B. Structure of the mechanically activated ion channel Piezo1. Nature 2018, 554, 481–486. [Google Scholar] [CrossRef]
- Murthy, S.E.; Dubin, A.E.; Patapoutian, A. Piezos thrive under pressure: Mechanically activated ion channels in health and disease. Nat. Rev. Mol. Cell Biol. 2017, 18, 771–783. [Google Scholar] [CrossRef]
- Datkhaeva, I.; Arboleda, V.A.; Senaratne, T.N.; Nikpour, G.; Meyerson, C.; Geng, Y.; Afshar, Y.; Scibetta, E.; Goldstein, J.; Quintero-Rivera, F.; et al. Identification of novel PIEZO1 variants using prenatal exome sequencing and correlation to ultrasound and autopsy findings of recurrent hydrops fetalis. Am. J. Med. Genet. Part A 2018, 176, 2829–2834. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Zeng, R.; He, E.; Zhang, I.; Ding, C.; Zhang, A. Piezo-Type Mechanosensitive Ion Channel Component 1 (Piezo1): A Promising Therapeutic Target and Its Modulators. J. Med. Chem. 2022, 65, 6441–6453. [Google Scholar] [CrossRef] [PubMed]
- Ge, J.; Li, W.; Zhao, Q.; Li, N.; Chen, M.; Zhi, P.; Li, R.; Gao, N.; Xiao, B.; Yang, M. Architecture of the mammalian mechanosensitive Piezo1 channel. Nature 2015, 527, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Zhou, H.; Chi, S.; Wang, Y.; Wang, J.; Geng, J.; Wu, K.; Liu, W.; Zhang, T.; Dong, M.Q.; et al. Structure and mechanogating mechanism of the Piezo1 channel. Nature 2018, 554, 487–492. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhou, H.; Li, X.; Xiao, B. The mechanosensitive Piezo1 channel: A three-bladed propeller-like structure and a lever-like mechanogating mechanism. FEBS J. 2019, 286, 2461–2470. [Google Scholar] [CrossRef]
- Gnanasambandam, R.; Bae, C.; Gottlieb, P.A.; Sachs, F. Ionic selectivity and permeation properties of human PIEZO1 channels. PLoS ONE 2015, 10, e0125503. [Google Scholar] [CrossRef]
- Gnanasambandam, R.; Ghatak, C.; Yasmann, A.; Nishizawa, K.; Sachs, F.; Ladokhin, A.S.; Sukharev, S.I.; Suchyna, T.M. GsMTx4: Mechanism of Inhibiting Mechanosensitive Ion Channels. Biophys. J. 2017, 112, 31–45. [Google Scholar] [CrossRef]
- Cox, C.D.; Bae, C.; Ziegler, L.; Hartley, S.; Nikolova-Krstevski, V.; Rohde, P.R.; Ng, C.A.; Sachs, F.; Gottlieb, P.A.; Martinac, B. Removal of the mechanoprotective influence of the cytoskeleton reveals PIEZO1 is gated by bilayer tension. Nat. Commun. 2016, 7, 10366. [Google Scholar] [CrossRef]
- Bae, C.; Sachs, F.; Gottlieb, P.A. Protonation of the human PIEZO1 ion channel stabilizes inactivation. J. Biol. Chem. 2015, 290, 5167–5173. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.R.; MacKinnon, R. Structure-based membrane dome mechanism for piezo mechanosensitivity. eLife 2017, 6, e33660. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Guo, Y.R.; Miyagi, A.; Levring, J.; MacKinnon, R.; Scheuring, S. Force-induced conformational changes in PIEZO1. Nature 2019, 573, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Haselwandter, C.A.; Mackinnon, R. Piezo’s membrane footprint and its contribution to mechanosensitivity. eLife 2018, 7, e41968. [Google Scholar] [CrossRef]
- Gnanasambandam, R.; Gottlieb, P.A.; Sachs, F. The Kinetics and the Permeation Properties of Piezo Channels; Elsevier: Amsterdam, The Netherlands, 2017; Volume 79, ISBN 9780128093894. [Google Scholar]
- Coste, B.; Xiao, B.; Santos, J.S.; Syeda, R.; Grandl, J.; Spencer, K.S.; Kim, S.E.; Schmidt, M.; Mathur, J.; Dubin, A.E.; et al. Piezo proteins are pore-forming subunits of mechanically activated channels. Nature 2012, 483, 176–181. [Google Scholar] [CrossRef]
- Gottlieb, P.A.; Sachs, F. Piezo1. Channels 2012, 6, 214–219. [Google Scholar] [CrossRef]
- Advis, J.P.; White, S.S.; Ojeda, S.R. Delayed Puberty Induced by Chronic Suppression of Prolactin Release in the Female Rat*. Endocrinology 1981, 109, 1321–1330. [Google Scholar] [CrossRef]
- Buyan, A.; Cox, C.D.; Barnoud, J.; Li, J.; Chan, H.S.; Martinac, B.; Marrink, S.J.; Corry, B. Piezo1 Forms Specific, Functionally Important Interactions with Phosphoinositides and Cholesterol. Biophys. J. 2020, 119, 1683–1697. [Google Scholar] [CrossRef]
- Romero, L.O.; Massey, A.E.; Mata-Daboin, A.D.; Sierra-Valdez, F.J.; Chauhan, S.C.; Cordero-Morales, J.F.; Vásquez, V. Dietary fatty acids fine-tune Piezo1 mechanical response. Nat. Commun. 2019, 10, 1200. [Google Scholar] [CrossRef]
- Ridone, P.; Pandzic, E.; Vassalli, M.; Cox, C.D.; Macmillan, A.; Gottlieb, P.A.; Martinac, B. Disruption of membrane cholesterol organization impairs the activity of PIEZO1 channel clusters. J. Gen. Physiol. 2020, 152, e201912515. [Google Scholar] [CrossRef] [PubMed]
- Pathak, M.M.; Nourse, J.L.; Tran, T.; Hwe, J.; Arulmoli, J.; Dai Trang, T.L.; Bernardis, E.; Flanagan, L.A.; Tombola, F. Stretch-activated ion channel Piezo1 directs lineage choice in human neural stem cells. Proc. Natl. Acad. Sci. USA 2014, 111, 16148–16153. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, N.R.; Hermanson, O.; Heimrich, B.; Shastri, V.P. Stochastic nanoroughness modulates neuron–astrocyte interactions and function via mechanosensing cation channels. Proc. Natl. Acad. Sci. USA 2014, 111, 16124–16129. [Google Scholar] [CrossRef] [PubMed]
- Hung, W.-C.; Yang, J.R.; Yankaskas, C.L.; Wong, B.S.; Wu, P.-H.; Pardo-Pastor, C.; Serra, S.A.; Chiang, M.-J.; Gu, Z.; Wirtz, D.; et al. Confinement Sensing and Signal Optimization via Piezo1/PKA and Myosin II Pathways. Cell Rep. 2016, 15, 1430–1441. [Google Scholar] [CrossRef] [PubMed]
- Lai, A.; Thurgood, P.; Cox, C.D.; Chheang, C.; Peter, K.; Jaworowski, A.; Khoshmanesh, K.; Baratchi, S. Piezo1 Response to Shear Stress Is Controlled by the Components of the Extracellular Matrix. ACS Appl. Mater. Interfaces 2022, 14, 40559–40568. [Google Scholar] [CrossRef]
- Moroni, M.; Servin-Vences, M.R.; Fleischer, R.; Sánchez-Carranza, O.; Lewin, G.R. Voltage gating of mechanosensitive PIEZO channels. Nat. Commun. 2018, 9, 1096. [Google Scholar] [CrossRef]
- Poole, K.; Herget, R.; Lapatsina, L.; Ngo, H.-D.; Lewin, G.R. Tuning Piezo ion channels to detect molecular-scale movements relevant for fine touch. Nat. Commun. 2014, 5, 3520. [Google Scholar] [CrossRef]
- Wetzel, C.; Pifferi, S.; Picci, C.; Gök, C.; Hoffmann, D.; Bali, K.K.; Lampe, A.; Lapatsina, L.; Fleischer, R.; Smith, E.S.J.; et al. Small-molecule inhibition of STOML3 oligomerization reverses pathological mechanical hypersensitivity. Nat. Neurosci. 2016, 20, 209–218. [Google Scholar] [CrossRef]
- Lee, W.; Leddy, H.A.; Chen, Y.; Lee, S.H.; Zelenski, N.A.; McNulty, A.L.; Wu, J.; Beicker, K.N.; Coles, J.; Zauscher, S.; et al. Synergy between Piezo1 and Piezo2 channels confers high-strain mechanosensitivity to articular cartilage. Proc. Natl. Acad. Sci. USA 2014, 111, E5114–E5122. [Google Scholar] [CrossRef]
- Gottlieb, P.A.; Bae, C.; Sachs, F. Gating the mechanical channel Piezo1. Channels 2012, 6, 282–289. [Google Scholar] [CrossRef]
- Zhang, T.; Chi, S.; Jiang, F.; Zhao, Q.; Xiao, B. A protein interaction mechanism for suppressing the mechanosensitive Piezo channels. Nat. Commun. 2017, 8, 1797. [Google Scholar] [CrossRef]
- Peyronnet, R.; Martins, J.R.; Duprat, F.; Demolombe, S.; Arhatte, M.; Jodar, M.; Tauc, M.; Duranton, C.; Paulais, M.; Teulon, J.; et al. Piezo1-dependent stretch-activated channels are inhibited by Polycystin-2 in renal tubular epithelial cells. EMBO Rep. 2013, 14, 1143–1148. [Google Scholar] [CrossRef] [PubMed]
- Nourse, J.L.; Pathak, M.M. How cells channel their stress: Interplay between Piezo1 and the cytoskeleton. Semin. Cell Dev. Biol. 2017, 71, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jiang, J.; Yang, X.; Zhou, G.; Wang, L.; Xiao, B. Tethering Piezo channels to the actin cytoskeleton for mechanogating via the cadherin-β-catenin mechanotransduction complex. Cell Rep. 2022, 38, 110342. [Google Scholar] [CrossRef]
- Syeda, R.; Xu, J.; Dubin, A.E.; Coste, B.; Mathur, J.; Huynh, T.; Matzen, J.; Lao, J.; Tully, D.C.; Engels, I.H.; et al. Chemical activation of the mechanotransduction channel Piezo1. eLife 2015, 4, e07369. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chi, S.; Guo, H.; Li, G.; Wang, L.; Zhao, Q.; Rao, Y.; Zu, L.; He, W.; Xiao, B. A lever-like transduction pathway for long-distance chemical- and mechano-gating of the mechanosensitive Piezo1 channel. Nat. Commun. 2018, 9, 1300. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Lu, W.; Chen, Y.; Duan, X.; Zhang, C.; Luo, X.; Lin, Z.; Chen, J.; Liu, S.; Yan, H.; et al. Upregulation of Piezo1 (Piezo Type Mechanosensitive Ion Channel Component 1) Enhances the Intracellular Free Calcium in Pulmonary Arterial Smooth Muscle Cells from Idiopathic Pulmonary Arterial Hypertension Patients. Hypertension 2021, 77, 1974–1989. [Google Scholar] [CrossRef] [PubMed]
- Suchyna, T.M. Piezo channels and GsMTx4: Two milestones in our understanding of excitatory mechanosensitive channels and their role in pathology. Prog. Biophys. Mol. Biol. 2017, 130, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, M.; Kimura, M.; Ouchi, T.; Nakamura, T.; Ohyama, S.; Ando, M.; Nomura, S.; Azuma, T.; Ichinohe, T.; Shibukawa, Y. Mechanical Stimulation-Induced Calcium Signaling by Piezo1 Channel Activation in Human Odontoblast Reduces Dentin Mineralization. Front. Physiol. 2021, 12, 704518. [Google Scholar] [CrossRef]
- Miron, T.R.; Flood, E.D.; Tykocki, N.R.; Thompson, J.M.; Watts, S.W. Identification of Piezo1 channels in perivascular adipose tissue (PVAT) and their potential role in vascular function. Pharmacol. Res. 2021, 175, 105995. [Google Scholar] [CrossRef]
- Relaix, F.; Zammit, P.S. Satellite cells are essential for skeletal muscle regeneration: The cell on the edge returns centre stage. Development 2012, 139, 2845–2856. [Google Scholar] [CrossRef]
- Chen, W.; Datzkiw, D.; Rudnicki, M.A. Satellite cells in ageing: Use it or lose it. Open Biol. 2020, 10, 200048. [Google Scholar] [CrossRef]
- Forcina, L.; Cosentino, M.; Musarò, A. Mechanisms Regulating Muscle Regeneration: Insights into the Interrelated and Time-Dependent Phases of Tissue Healing. Cells 2020, 9, 1297. [Google Scholar] [CrossRef]
- Baghdadi, M.B.; Tajbakhsh, S. Regulation and phylogeny of skeletal muscle regeneration. Dev. Biol. 2018, 433, 200–209. [Google Scholar] [CrossRef]
- Franco, A.; Lansman, J.B. Calcium entry through stretch-inactivated ion channels in mdx myotubes. Nature 1989, 13, 104–116. [Google Scholar] [CrossRef]
- Yeung, E.W.; Whitehead, N.P.; Suchyna, T.M.; Gottlieb, P.A.; Sachs, F.; Allen, D.G. Effects of stretch-activated channel blockers on [Ca2+]I and muscle damage in the mdx mouse. J. Physiol. 2005, 562, 367–380. [Google Scholar] [CrossRef]
- Tsuchiya, M.; Hara, Y.; Okuda, M.; Itoh, K.; Nishioka, R.; Shiomi, A.; Nagao, K.; Mori, M.; Mori, Y.; Ikenouchi, J.; et al. Cell surface flip-flop of phosphatidylserine is critical for PIEZO1-mediated myotube formation. Nat. Commun. 2018, 9, 2049. [Google Scholar] [CrossRef]
- Bosutti, A.; Giniatullin, A.; Odnoshivkina, Y.; Giudice, L.; Malm, T.; Sciancalepore, M.; Giniatullin, R.; D’Andrea, P.; Lorenzon, P.; Bernareggi, A. “Time window” effect of Yoda1-evoked Piezo1 channel activity during mouse skeletal muscle differentiation. Acta Physiol. 2021, 233, e13702. [Google Scholar] [CrossRef]
- Ma, N.; Chen, D.; Lee, J.-H.; Kuri, P.; Hernandez, E.B.; Kocan, J.; Mahmood, H.; Tichy, E.D.; Rompolas, P.; Mourkioti, F. Piezo1 regulates the regenerative capacity of skeletal muscles via orchestration of stem cell morphological states. Sci. Adv. 2022, 8, eabn0485. [Google Scholar] [CrossRef]
- Peng, Y.; Du, J.; Günther, S.; Guo, X.; Wang, S.; Schneider, A.; Zhu, L.; Braun, T. Mechano-signaling via Piezo1 prevents activation and p53-mediated senescence of muscle stem cells. Redox Biol. 2022, 52, 102309. [Google Scholar] [CrossRef]
- Quiroga, H.P.O.; Ganassi, M.; Yokoyama, S.; Nakamura, K.; Yamashita, T.; Raimbach, D.; Hagiwara, A.; Harrington, O.; Breach-Teji, J.; Asakura, A.; et al. Fine-Tuning of Piezo1 Expression and Activity Ensures Efficient Myoblast Fusion during Skeletal Myogenesis. Cells 2022, 11, 393. [Google Scholar] [CrossRef]
- Wang, M.J.; Zhu, Y.C.; Shi, J. A crucial physiological role of Piezo1 channel in differentiation rather than proliferation during myogenesis. Acta Physiol. 2021, 233, e13728. [Google Scholar] [CrossRef]
- Jagasia, R.; Wagner, K.R. Piezo1: Opening the way to preventing muscle atrophy. J. Clin. Investig. 2022, 132, 15–17. [Google Scholar] [CrossRef]
- Hirano, K.; Tsuchiya, M.; Shiomi, A.; Takabayashi, S.; Suzuki, M.; Ishikawa, Y.; Kawano, Y.; Takabayashi, Y.; Nishikawa, K.; Nagao, K.; et al. The mechanosensitive ion channel PIEZO1 promotes satellite cell function in muscle regeneration. Life Sci. Alliance 2022, 6, e202201783. [Google Scholar] [CrossRef] [PubMed]
- Sciancalepore, M.; Luin, E.; Parato, G.; Ren, E.; Giniatullin, R.; Fabbretti, E.; Lorenzon, P. Reactive oxygen species contribute to the promotion of the ATP-mediated proliferation of mouse skeletal myoblasts. Free Radic. Biol. Med. 2012, 53, 1392–1398. [Google Scholar] [CrossRef]
- Espinosa, A.; Leiva, A.; Peña, M.; Müller, M.; Debandi, A.; Hidalgo, C.; Carrasco, M.A.; Jaimovich, E. Myotube depolarization generates reactive oxygen species through NAD(P)H oxidase; ROS-elicited Ca2+ stimulates ERK, CREB, early genes. J. Cell. Physiol. 2006, 209, 379–388. [Google Scholar] [CrossRef]
- Hirata, Y.; Nomura, K.; Kato, D.; Tachibana, Y.; Niikura, T.; Uchiyama, K.; Hosooka, T.; Fukui, T.; Oe, K.; Kuroda, R.; et al. A Piezo1/KLF15/IL-6 axis mediates immobilization-induced muscle atrophy. J. Clin. Investig. 2022, 132, 1–13. [Google Scholar] [CrossRef]
- Olsen, B.R.; Reginato, A.M.; Wang, W. Bone Development. Annu. Rev. Cell Dev. Biol. 2003, 16, 191–220. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, C.R.; Temiyasathit, S.; Castillo, A.B. Osteocyte Mechanobiology and Pericellular Mechanics. Annu. Rev. Biomed. Eng. 2010, 12, 369–400. [Google Scholar] [CrossRef]
- Bonewald, L.F. The amazing osteocyte. J. Bone Miner. Res. 2011, 26, 229–238. [Google Scholar] [CrossRef]
- Uda, Y.; Azab, E.; Sun, N.; Shi, C.; Pajevic, P.D. Osteocyte Mechanobiology. Curr. Osteoporos. Rep. 2017, 15, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Wolff, J. Gesetz der Transformation der Knochen; Verlag von August Hirschwald: Berlin, Germany, 1892. [Google Scholar]
- Shea, C.A.; Rolfe, R.A.; Murphy, P. The importance of foetal movement for co-ordinated cartilage and bone development in utero. Bone Jt. Res. 2015, 4, 105–116. [Google Scholar] [CrossRef]
- Rauch, F.; Schoenau, E. Changes in Bone Density During Childhood and Adolescence: An Approach Based on Bone’s Biological Organization. J. Bone Miner. Res. 2001, 16, 597–604. [Google Scholar] [CrossRef] [PubMed]
- Rolfe, R.; Roddy, K.; Murphy, P. Mechanical Regulation of Skeletal Development. Curr. Osteoporos. Rep. 2013, 11, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Responte, D.J.; Lee, J.K.; Hu, J.C.; Athanasiou, K.A. Biomechanics-driven chondrogenesis: From embryo to adult. FASEB J. 2012, 26, 3614–3624. [Google Scholar] [CrossRef]
- Qin, L.; Liu, W.; Cao, H.; Xiao, G. Molecular mechanosensors in osteocytes. Bone Res. 2020, 8, 23. [Google Scholar] [CrossRef]
- Guldberg, R.E.; Caldwell, N.J.; Guo, X.E.; Goulet, R.W.; Hollister, S.J.; Goldstein, S.A. Mechanical Stimulation of Tissue Repair in the Hydraulic Bone Chamber. J. Bone Miner. Res. 1997, 12, 1295–1302. [Google Scholar] [CrossRef] [PubMed]
- Sugisawa, E.; Takayama, Y.; Takemura, N.; Kondo, T.; Hatakeyama, S.; Kumagai, Y.; Sunagawa, M.; Tominaga, M.; Maruyama, K. RNA Sensing by Gut Piezo1 Is Essential for Systemic Serotonin Synthesis. Cell 2020, 182, 609–624.e21. [Google Scholar] [CrossRef]
- Hendrickx, G.; Fischer, V.; Liedert, A.; von Kroge, S.; Haffner-Luntzer, M.; Brylka, L.; Pawlus, E.; Schweizer, M.; Yorgan, T.; Baranowsky, A.; et al. Piezo 1 Inactivation in Chondrocytes Impairs Trabecular Bone Formation. J. Bone Miner. Res. 2020, 36, 369–384. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Han, L.; Nookaew, I.; Mannen, E.; Silva, M.J.; Almeida, M.; Xiong, J. Stimulation of Piezo1 by mechanical signals promotes bone anabolism. eLife 2019, 8, e49631. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Gao, B.; Fan, Y.; Liu, Y.; Feng, S.; Cong, Q.; Zhang, X.; Zhou, Y.; Yadav, P.S.; Lin, J.; et al. Piezo1/2 mediate mechanotransduction essential for bone formation through concerted activation of NFAT-YAP1-ß-catenin. eLife 2020, 9, e52779. [Google Scholar] [CrossRef] [PubMed]
- Ranade, S.S.; Woo, S.-H.; Dubin, A.E.; Moshourab, R.A.; Wetzel, C.; Petrus, M.; Mathur, J.; Bégay, V.; Coste, B.; Mainquist, J.; et al. Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature 2014, 516, 121–125. [Google Scholar] [CrossRef]
- Wang, L.; You, X.; Lotinun, S.; Zhang, L.; Wu, N.; Zou, W. Mechanical sensing protein PIEZO1 regulates bone homeostasis via osteoblast-osteoclast crosstalk. Nat. Commun. 2020, 11, 282. [Google Scholar] [CrossRef]
- Hillam, R.A.; Goodship, A.E.; Skerry, T.M. Peak strain magnitudes and rates in the tibia exceed greatly those in the skull: An in vivo study in a human subject. J. Biomech. 2015, 48, 3292–3298. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Tasdogan, A.; Ubellacker, J.M.; Zhang, J.; Nosyreva, E.D.; Du, L.; Murphy, M.M.; Hu, S.; Yi, Y.; Kara, N.; et al. A mechanosensitive peri-arteriolar niche for osteogenesis and lymphopoiesis. Nature 2021, 591, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Ziros, P.G.; Basdra, E.K.; Papavassiliou, A.G. Runx2: Of bone and stretch. Int. J. Biochem. Cell Biol. 2008, 40, 1659–1663. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Xiao, Z.; Luo, J.; He, N.; Mahlios, J.; Quarles, L.D. Dose-Dependent Effects of Runx2 on Bone Development. J. Bone Miner. Res. 2009, 24, 1889–1904. [Google Scholar] [CrossRef]
- Shu, B.; Zhao, Y.; Zhao, S.; Pan, H.; Xie, R.; Yi, D.; Lu, K.; Yang, J.; Xue, C.; Huang, J.; et al. Inhibition of Axin1 in osteoblast precursor cells leads to defects in postnatal bone growth through suppressing osteoclast formation. Bone Res. 2020, 8, 31. [Google Scholar] [CrossRef]
- Shekaran, A.; Shoemaker, J.T.; Kavanaugh, T.E.; Lin, A.S.; LaPlaca, M.C.; Fan, Y.; Guldberg, R.E.; García, A.J. The effect of conditional inactivation of beta 1 integrins using twist 2 Cre, Osterix Cre and osteocalcin Cre lines on skeletal phenotype. Bone 2014, 68, 131–141. [Google Scholar] [CrossRef]
- Cao, H.; Yan, Q.; Wang, D.; Lai, Y.; Zhou, B.; Zhang, Q.; Jin, W.; Lin, S.; Lei, Y.; Ma, L.; et al. Focal adhesion protein Kindlin-2 regulates bone homeostasis in mice. Bone Res. 2020, 8, 2. [Google Scholar] [CrossRef]
- Wang, Y.; Yan, Q.; Zhao, Y.; Liu, X.; Lin, S.; Zhang, P.; Ma, L.; Lai, Y.; Bai, X.; Liu, C.; et al. Focal adhesion proteins Pinch1 and Pinch2 regulate bone homeostasis in mice. J. Clin. Investig. 2019, 4, e131692. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Zhou, B.; Yan, Q.; Tao, C.; Qin, L.; Wu, X.; Lin, S.; Chen, S.; Lai, Y.; Zou, X.; et al. Kindlin-2 regulates skeletal homeostasis by modulating PTH1R in mice. Signal Transduct. Target. Ther. 2020, 5, 297. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Cooper, P.R.; Walmsley, A.D.; Scheven, B.A. Role of Piezo Channels in Ultrasound-stimulated Dental Stem Cells. J. Endod. 2017, 43, 1130–1136. [Google Scholar] [CrossRef]
- Zhang, G.; Li, X.; Wu, L.; Qin, Y.-X. Piezo1 channel activation in response to mechanobiological acoustic radiation force in osteoblastic cells. Bone Res. 2021, 9, 16. [Google Scholar] [CrossRef] [PubMed]
- Nakamichi, R.; Ma, S.; Nonoyama, T.; Chiba, T.; Kurimoto, R.; Ohzono, H.; Olmer, M.; Shukunami, C.; Fuku, N.; Wang, G.; et al. The mechanosensitive ion channel PIEZO1 is expressed in tendons and regulates physical performance. Sci. Transl. Med. 2022, 14, eabj5557. [Google Scholar] [CrossRef]
- Passini, F.S.; Jaeger, P.K.; Saab, A.S.; Hanlon, S.; Chittim, N.A.; Arlt, M.J.; Ferrari, K.D.; Haenni, D.; Caprara, S.; Bollhalder, M.; et al. Shear-stress sensing by PIEZO1 regulates tendon stiffness in rodents and influences jumping performance in humans. Nat. Biomed. Eng. 2021, 5, 1457–1471. [Google Scholar] [CrossRef]
- Woo, S.-H.; Lukacs, V.; De Nooij, J.C.; Zaytseva, D.; Criddle, C.R.; Francisco, A.; Jessell, T.M.; Wilkinson, K.A.; Patapoutian, A. Piezo2 is the principal mechanotransduction channel for proprioception HHS Public Access Author manuscript. Nat. Neurosci. 2015, 18, 1756–1762. [Google Scholar] [CrossRef]
- Florez-Paz, D.; Bali, K.K.; Kuner, R.; Gomis, A. A critical role for Piezo2 channels in the mechanotransduction of mouse proprioceptive neurons. Sci. Rep. 2016, 6, 25923. [Google Scholar] [CrossRef]
- Jin, Y.; Li, J.; Wang, Y.; Ye, R.; Feng, X.; Jing, Z.; Zhao, Z. Functional role of mechanosensitive ion channel Piezo1 in human periodontal ligament cells. Angle Orthod. 2015, 85, 87–94. [Google Scholar] [CrossRef]
- Shen, Y.; Pan, Y.; Guo, S.; Sun, L.; Zhang, C.; Wang, L. The roles of mechanosensitive ion channels and associated downstream MAPK signaling pathways in PDLC mechanotransduction. Mol. Med. Rep. 2020, 21, 2113–2122. [Google Scholar] [CrossRef]
- Lin, W.; Xi, W.; Nan, J.; Haimei, L.; Shixin, C. Mechanisms of the mechanically activated ion channel Piezo1 protein in mediating osteogenic differentiation of perio? dontal ligament stem cells via the Notch signaling pathway Wang. West China J. Stomatol. 2020, 38, 628–636. [Google Scholar]
- Phan, M.N.; Leddy, H.A.; Votta, B.J.; Kumar, S.; Levy, D.S.; Lipshutz, D.B.; Lee, S.H.; Liedtke, W.; Guilak, F. Functional characterization of TRPV4 as an osmotically sensitive ion channel in porcine articular chondrocytes. Arthritis Rheum. 2009, 60, 3028–3037. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Nims, R.J.; Savadipour, A.; Zhang, Q.; Leddy, H.A.; Liu, F.; McNulty, A.L.; Chen, Y.; Guilak, F.; Liedtke, W.B. Inflammatory signaling sensitizes Piezo1 mechanotransduction in articular chondrocytes as a pathogenic feed-forward mechanism in osteoarthritis. Proc. Natl. Acad. Sci. USA 2021, 118, e2001611118. [Google Scholar] [CrossRef]
- Li, X.-F.; Zhang, Z.; Chen, Z.-K.; Cui, Z.-W.; Zhang, H.-N. Piezo1 protein induces the apoptosis of human osteoarthritis-derived chondrocytes by activating caspase-12, the signaling marker of ER stress. Int. J. Mol. Med. 2017, 40, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, K.M.; Jones, R.C.; Jackson, T.R.; Baylie, R.L.; Abbott, B.; Bruhn-Olszewska, B.; Board, T.N.; Locke, I.C.; Richardson, S.M.; Townsend, P.A. Chondroprotection by urocortin involves blockade of the mechanosensitive ion channel Piezo1. Sci. Rep. 2017, 7, 5147. [Google Scholar] [CrossRef] [PubMed]
- Du, G.; Li, L.; Zhang, X.; Liu, J.; Hao, J.; Zhu, J.; Wu, H.; Chen, W.; Zhang, Q. Roles of TRPV4 and piezo channels in stretch-evoked Ca2+ response in chondrocytes. Exp. Biol. Med. 2019, 245, 180–189. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, W.; Chen, S.; Wang, B.; Wang, P.; Hu, B.; Lv, X.; Shao, Z. Extracellular matrix in intervertebral disc: Basic and translational implications. Cell Tissue Res. 2022, 390, 1–22. [Google Scholar] [CrossRef]
- Yang, Q.; Zhou, Y.; Wang, J.; Fu, W.; Li, X. Study on the mechanism of excessive apoptosis of nucleus pulposus cells induced by shRNA-Piezo1 under abnormal mechanical stretch stress. J. Cell. Biochem. 2018, 120, 3989–3997. [Google Scholar] [CrossRef]
- Sun, Y.; Leng, P.; Song, M.; Li, D.; Guo, P.; Xu, X.; Gao, H.; Li, Z.; Li, C.; Zhang, H. Piezo1 activates the NLRP3 inflammasome in nucleus pulposus cell-mediated by Ca2+/NF-κB pathway. Int. Immunopharmacol. 2020, 85, 106681. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Zheng, X.M.; Li, S.; Zeng, B.; Yang, J.; Ling, Z.M.; Liu, X.M.; Wei, F.M. Single Impact Injury of Vertebral Endplates Without Structural Disruption, Initiates Disc Degeneration Through Piezo1 Mediated Inflammation and Metabolism Dysfunction. Spine 2021, 47, E203–E213. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Ke, W.; Wang, K.; Li, G.; Ma, L.; Lu, S.; Xiang, Q.; Liao, Z.; Luo, R.; Song, Y.; et al. Mechanosensitive Ion Channel Piezo1 Activated by Matrix Stiffness Regulates Oxidative Stress-Induced Senescence and Apoptosis in Human Intervertebral Disc Degeneration. Oxidative Med. Cell. Longev. 2021, 2021, 8884922. [Google Scholar] [CrossRef]
- Wu, J.; Chen, Y.; Liao, Z.; Liu, H.; Zhang, S.; Zhong, D.; Qiu, X.; Chen, T.; Su, D.; Ke, X.; et al. Self-amplifying loop of NF-κB and periostin initiated by PIEZO1 accelerates mechano-induced senescence of nucleus pulposus cells and intervertebral disc degeneration. Mol. Ther. 2022, 30, 3241–3256. [Google Scholar] [CrossRef]
- Shi, S.; Kang, X.-J.; Zhou, Z.; He, Z.-M.; Zheng, S.; He, S.-S. Excessive mechanical stress-induced intervertebral disc degeneration is related to Piezo1 overexpression triggering the imbalance of autophagy/apoptosis in human nucleus pulpous. Thromb. Haemost. 2022, 24, 119. [Google Scholar] [CrossRef]
- Vergroesen, P.-P.; Kingma, I.; Emanuel, K.; Hoogendoorn, R.; Welting, T.; van Royen, B.; van Dieën, J.; Smit, T. Mechanics and biology in intervertebral disc degeneration: A vicious circle. Osteoarthr. Cartil. 2015, 23, 1057–1070. [Google Scholar] [CrossRef]
- Zhu, D.; Zhang, G.; Guo, X.; Wang, Y.; Liu, M.; Kang, X. A New Hope in Spinal Degenerative Diseases: Piezo1. BioMed Res. Int. 2021, 2021, 6645193. [Google Scholar] [CrossRef]
- Jiang, L.; Zhao, Y.-D.; Chen, W.-X. The Function of the Novel Mechanical Activated Ion Channel Piezo1 in the Human Osteosarcoma Cells. Experiment 2017, 23, 5070–5082. [Google Scholar] [CrossRef]
- Suzuki, T.; Muraki, Y.; Hatano, N.; Suzuki, H.; Muraki, K. PIEZO1 Channel Is a Potential Regulator of Synovial Sarcoma Cell-Viability. Int. J. Mol. Sci. 2018, 19, 1452. [Google Scholar] [CrossRef]
- Jiang, Y.; Guan, Y.; Lan, Y.; Chen, S.; Li, T.; Zou, S.; Hu, Z.; Ye, Q. Mechanosensitive Piezo1 in Periodontal Ligament Cells Promotes Alveolar Bone Remodeling During Orthodontic Tooth Movement. Front. Physiol. 2021, 12, 767136. [Google Scholar] [CrossRef]
- Beech, D.J.; Kalli, A.C. Force Sensing by Piezo Channels in Cardiovascular Health and Disease. Arter. Thromb. Vasc. Biol. 2019, 39, 2228–2239. [Google Scholar] [CrossRef]
- Yang, X.-N.; Lu, Y.-P.; Liu, J.-J.; Huang, J.-K.; Liu, Y.-P.; Xiao, C.-X.; Jazag, A.; Ren, J.-L.; Guleng, B. Piezo1 Is as a Novel Trefoil Factor Family 1 Binding Protein that Promotes Gastric Cancer Cell Mobility In Vitro. Dig. Dis. Sci. 2014, 59, 1428–1435. [Google Scholar] [CrossRef]
- Romac, J.M.-J.; Shahid, R.A.; Swain, S.M.; Vigna, S.R.; Liddle, R.A. Piezo1 is a mechanically activated ion channel and mediates pressure induced pancreatitis. Nat. Commun. 2018, 9, 1715. [Google Scholar] [CrossRef]
- Miyamoto, T.; Mochizuki, T.; Nakagomi, H.; Kira, S.; Watanabe, M.; Takayama, Y.; Suzuki, Y.; Koizumi, S.; Takeda, M.; Tominaga, M. Functional Role for Piezo1 in Stretch-evoked Ca2+ Influx and ATP Release in Urothelial Cell Cultures. J. Biol. Chem. 2014, 289, 16565–16575. [Google Scholar] [CrossRef]
- Wang, E.C.; Lee, J.-M.; Ruiz, W.G.; Balestreire, E.M.; von Bodungen, M.; Barrick, S.; Cockayne, D.A.; Birder, L.A.; Apodaca, G. ATP and purinergic receptor–dependent membrane traffic in bladder umbrella cells. J. Clin. Investig. 2005, 115, 2412–2422. [Google Scholar] [CrossRef]
- Zhao, X.; Li, C.; Wang, W.; Zhao, X.; Kong, Y.; Liang, B.; Xu, J.; Lin, Y.; Zhou, N.; Li, J.; et al. Mechanosensitive Piezo1 channels mediate renal fibrosis Find the latest version: Mechanosensitive Piezo1 channels mediate renal fibrosis. JCI Insight 2022, 2022, 7. [Google Scholar]
- Liang, G.-P.; Xu, J.; Cao, L.-L.; Zeng, Y.-H.; Chen, B.-X.; Yang, J.; Zhang, Z.-W.; Kang, Y. Piezo1 induced apoptosis of type II pneumocytes during ARDS. Respir. Res. 2019, 20, 118. [Google Scholar] [CrossRef]
- Friedrich, E.E.; Hong, Z.; Xiong, S.; Di, A.; Rehman, J.; Komarova, Y.A.; Malik, A.B. Endothelial cell Piezo1 mediates pressure-induced lung vascular hyperpermeability via disruption of adherens junctions. Proc. Natl. Acad. Sci. USA 2019, 116, 12980–12985. [Google Scholar] [CrossRef]
- McHugh, B.J.; Murdoch, A.; Haslett, C.; Sethi, T. Loss of the Integrin-Activating Transmembrane Protein Fam38A (Piezo1) Promotes a Switch to a Reduced Integrin-Dependent Mode of Cell Migration. PLoS ONE 2012, 7, e40346. [Google Scholar] [CrossRef]
- Chen, S.; Huang, P.; Qiu, Y.; Zhou, Q.; Li, X.; Zhu, M.; Hong, D. Phenotype variability and histopathological findings in patients with a novel DNM2 mutation. Neuropathology 2017, 38, 34–40. [Google Scholar] [CrossRef]
- Zhou, W.; Liu, X.; van Wijnbergen, J.W.M.; Yuan, L.; Liu, Y.; Zhang, C.; Jia, W. Identification of PIEZO1 as a potential prognostic marker in gliomas. Sci. Rep. 2020, 10, 16121. [Google Scholar] [CrossRef]
- Velasco, M.; Rolle, S.O.; Mampay, M.; Dev, K.K.; Sheridan, G.K. Piezo1 regulates calcium oscillations and cytokine release from astrocytes. Glia 2019, 68, 145–160. [Google Scholar] [CrossRef]
- Morozumi, W.; Inagaki, S.; Iwata, Y.; Nakamura, S.; Hara, H.; Shimazawa, M. Piezo channel plays a part in retinal ganglion cell damage. Exp. Eye Res. 2019, 191, 107900. [Google Scholar] [CrossRef] [PubMed]
- Dolgorukova, A.; Isaeva, J.E.; Verbitskaya, E.; Lyubashina, O.A.; Giniatullin, R.; Sokolov, A.Y. Differential effects of the Piezo1 agonist Yoda1 in the trigeminovascular system: An electrophysiological and intravital microscopy study in rats. Exp. Neurol. 2021, 339, 113634. [Google Scholar] [CrossRef]
- Mikhailov, N.; Plotnikova, L.; Singh, P.; Giniatullin, R.; Hämäläinen, R.H. Functional Characterization of Mechanosensitive Piezo1 Channels in Trigeminal and Somatic Nerves in a Neuron-on-Chip Model. Int. J. Mol. Sci. 2022, 23, 1370. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Fang, B.; Shan, S.; Xie, Y.; Wang, C.; Zhang, Y.; Zhang, X.; Li, Q. Mechanical stretch promotes hypertrophic scar formation through mechanically activated cation channel Piezo1. Cell Death Dis. 2021, 12, 226. [Google Scholar] [CrossRef]
- Holt, J.R.; Zeng, W.-Z.; Evans, E.L.; Woo, S.-H.; Ma, S.; Abuwarda, H.; Loud, M.; Patapoutian, A.; Pathak, M.M. Spatiotemporal dynamics of PIEZO1 localization controls keratinocyte migration during wound healing. eLife 2021, 10, e65415. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Sun, Q.; Tang, L.; Cao, Y.; Nourse, J.L.; Pathak, M.M.; Lu, X.; Yang, Q. Mechanosensitive Ion Channel Piezo1 Regulates Diet-Induced Adipose Inflammation and Systemic Insulin Resistance. Front. Endocrinol. 2019, 10, 373. [Google Scholar] [CrossRef]

| Agonists | Antagonists |
|---|---|
| Yoda1 | Gadolinium |
| Jedi1, Jedi2 | Ruthenium red |
| GsMTx4 | |
| Dooku1 | |
| Margaric acid | |
| Arachidonic acid | |
| Eicosapentaenoic acid |
| Organ System | Affected Organs | Diseases | Affected Cell Type | Experimental Setup | References |
|---|---|---|---|---|---|
| Skeletal system | Bone | Osteoporosis | Osteocytes, osteoblasts | In vitro Human and mouse MSCs | [147] |
| Bone | Osteosarcoma | Human osteosarcoma cells | In vitro Human osteosarcoma cell line MG63 and U2 cell line | [148] | |
| Joints | Synovial sarcoma | SW982 cells | In vitro Human synovial sarcoma SW982, human embryonic kidney 293 cell lines | [149] | |
| Joints | Joint disease | Chondrocytes | In vitro Primary chondrocytes from six-day-old mice | [138] | |
| Dental system | Orthodontic tooth movement | Periodontal ligament cells | Sprague-Dawley rats (8 week old) | [131,150] | |
| Heart | Cardiovascular disease | Cardiac fibroblasts, cardiomyocytes | In vitro Knockout Mice sm22Cre Piezo1−/− Mice | [151] | |
| Gastrointestinal system | Gastic region | Gastric cancer | Gastric cancer cell lines | In vitro Gastric cancer cell lines SGC-7901 and BGC-823 | [152] |
| Pancreas | Pancreatitis | Pancreatic acinar cells | In vitro C57BL/6J male mice 6–8 week old and Piezo1 knockout mice: Ptf1atm2(cre/ESR1)Cvw/J mice | [153] | |
| Pancreas | Pancreatic ductal adenocarcinoma | Pancreatic stellate cells | In vitro Human pancreatic cancer cell lines PCs, MiaPaCa-2 and Panc-1 | [154] | |
| Excretory system | Bladder | Bladder dysfunction | Bladder urothelial cells | In vitro C57BL/6Cr) mice and TRPV4-knockout mice In vitro Patients with prostate cancer or benign prostatic hyperplasia | [155] |
| Kidney | Renal fibrosis | HK2 cells | In vitro C57BL/6J mice In vitro Human kidney autopsy specimens Human proximal tubular cells (HK2 cells) | [156] | |
| Respiratory system | Lungs | Acute respiratory distress syndrome | Type II pneumocytes | In vitro Health adult Sprague-Dawley rats | [157] |
| Lungs | Lung capillary stress failure injury | Lung endothelial cells | In vitro Backcrossing Piezo1flox/flox mice with Endo-SCL-Cre mice | [158] | |
| Lungs | Lung cancer | Small cell lung cancer lines | In vitro 16HBE cells | [159] | |
| Nervous system | Central nervous system | Gliomas | Human gliomas cells | Human glioma tissues | [160,161] |
| Astrocytes | Neuroinflammation | Astrocytes | In vitro C57BL/6 mice; mixed glial cell cultures | [162] | |
| Eye | Glaucoma | Cornea, retinal ganglion cell layer, and lens epithelial cells | In vitro 7-week-old male albino ddY mice, 15-month-old C57BL/6 mice, 9- and 15-month-old DBA/2J mice, adult Sprague–Dawley rats | [163] | |
| Neurons | Migraine | Trigeminal ganglion | In vitro Wistar rats | [164,165] | |
| Connective tissue | Skin | Hypertrophic scar | Myofibroblasts | In vitro Human hypertrophic scar tissues and adjacent normal skin tissues from 9 people | [166] |
| Skin | Skin wound | Keratinocytes | In vitro Krt14Cre;Piezo1fl/fl (Piezo1-cKO) Krt14Cre;Piezo1cx/+ and Krt14Cre;Piezo1cx/cx (Piezo1-GoF) | [167] | |
| Adipose tissue | Inflammation | Adipocytes | In vitro Piezo1-flox/flox mice were crossed to adiponectin-Cre mice | [168] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dienes, B.; Bazsó, T.; Szabó, L.; Csernoch, L. The Role of the Piezo1 Mechanosensitive Channel in the Musculoskeletal System. Int. J. Mol. Sci. 2023, 24, 6513. https://doi.org/10.3390/ijms24076513
Dienes B, Bazsó T, Szabó L, Csernoch L. The Role of the Piezo1 Mechanosensitive Channel in the Musculoskeletal System. International Journal of Molecular Sciences. 2023; 24(7):6513. https://doi.org/10.3390/ijms24076513
Chicago/Turabian StyleDienes, Beatrix, Tamás Bazsó, László Szabó, and László Csernoch. 2023. "The Role of the Piezo1 Mechanosensitive Channel in the Musculoskeletal System" International Journal of Molecular Sciences 24, no. 7: 6513. https://doi.org/10.3390/ijms24076513
APA StyleDienes, B., Bazsó, T., Szabó, L., & Csernoch, L. (2023). The Role of the Piezo1 Mechanosensitive Channel in the Musculoskeletal System. International Journal of Molecular Sciences, 24(7), 6513. https://doi.org/10.3390/ijms24076513






