Exploiting the Integrated Valorization of Eucalyptus globulus Leaves: Chemical Composition and Biological Potential of the Lipophilic Fraction before and after Hydrodistillation
Abstract
1. Introduction
2. Results and Discussion
2.1. Extraction Yields
2.2. Chemical Characterization of the Lipophilic Extracts
2.2.1. Triterpenic Compounds
2.2.2. Fatty Acids
2.2.3. Monoglycerides and Sterols
2.2.4. Long-Chain Aliphatic Alcohols
2.2.5. Other Compounds
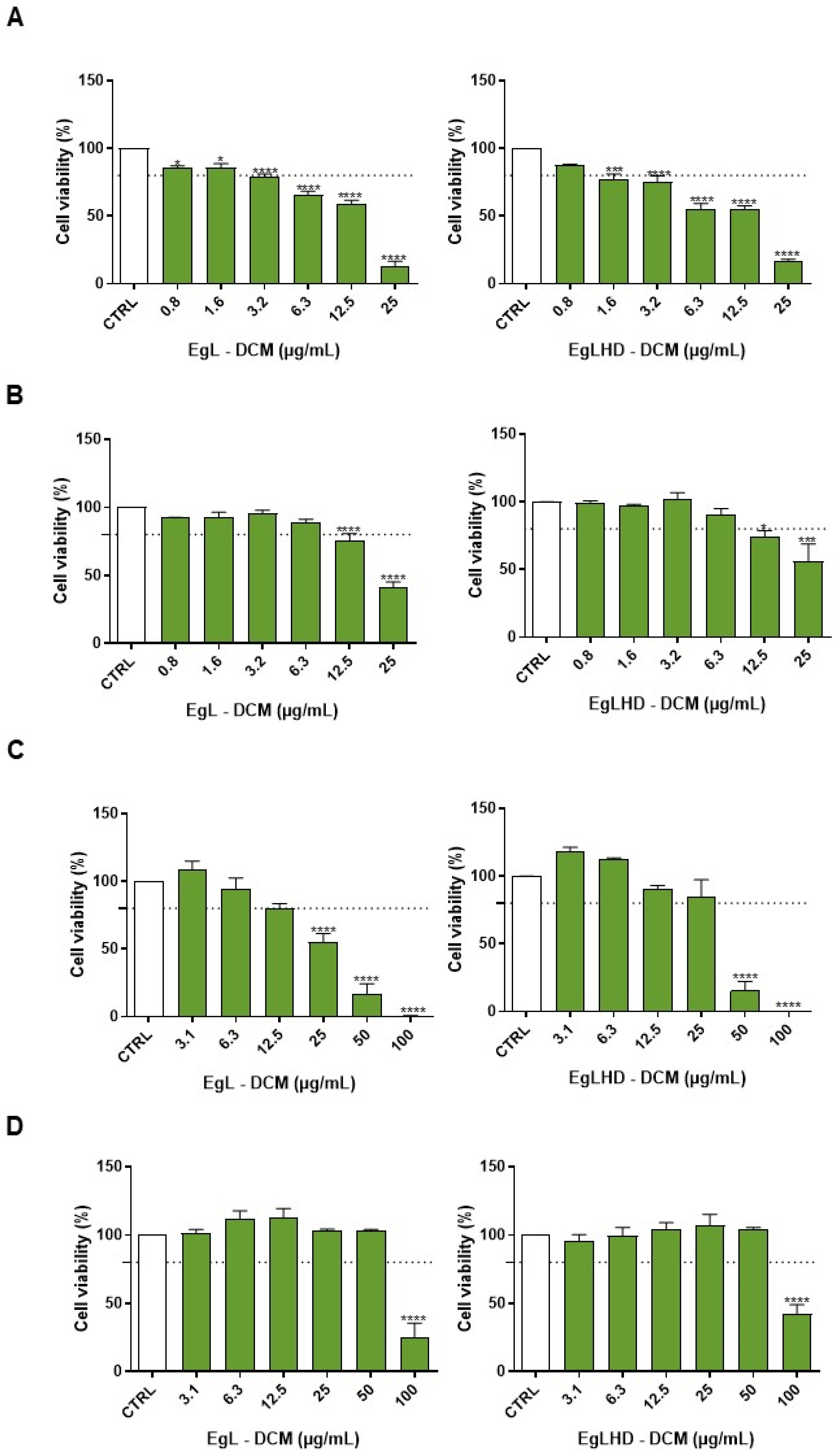
2.3. Cell Viability of Lipophilic Extracts
3. Materials and Methods
3.1. Reagents
3.2. Samples Collection
3.3. Hydrodistillation
3.4. Preparation of Lipophilic Extracts
3.5. GC–MS Analysis
3.6. Cell Culture
3.7. Cell Viability Evaluation
Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Dugmore, T.I.J.; Clark, J.H.; Bustamante, J.; Houghton, J.A.; Matharu, A.S. Valorisation of biowastes for the production of green materials using chemical methods. Top. Curr. Chem. 2017, 375, 46. [Google Scholar] [CrossRef] [PubMed]
- De Jong, E.; Higson, A.; Walsh, P.; Wellisch, M. Bio-based chemicals, value added products from biorefineries. In IEA Bioenergy Task 42 Report; IEA Bioenergy: Paris, France, 2012. [Google Scholar]
- Waldron, K.W. Advances in Biorefineries: Biomass and Waste Supply Chain Exploitation; Woodhead Publishing: Cambridge, UK, 2014; ISBN 9780857097385. [Google Scholar]
- Sadhukhan, J.; Ng, K.S.; Hernandez, E.M. Biorefineries and Chemical Processes: Design, Integration and Sustainability Analysis; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2014; ISBN 9781119990864. [Google Scholar]
- CELPA. Boletim Estatístico da CELPA—Indústria Papeleira Portuguesa; CELPA: Lisbon, Portugal, 2018. [Google Scholar]
- ICNF. IFN6—Principais resultados—relatório sumário. In Instituto da Conservação da Natureza e das Florestas; ICNF: Lisbon, Portugal, 2019; pp. 1–34. [Google Scholar]
- Neiva, D.M.; Araújo, S.; Gominho, J.; Carneiro, A.C.O.; Pereira, H. Potential of Eucalyptus globulus industrial bark as a biorefinery feedstock: Chemical and fuel characterization. Ind. Crops Prod. 2018, 123, 262–270. [Google Scholar] [CrossRef]
- Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P.; Cavaleiro, J.A.S. Lipophilic extractives of the inner and outer barks of Eucalyptus globulus. Holzforschung 2002, 56, 372–379. [Google Scholar] [CrossRef]
- Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P. Identification of new hydroxy fatty acids and ferulic acid esters in the wood of Eucalyptus globulus. Holzforschung 2002, 56, 143–149. [Google Scholar] [CrossRef]
- Domingues, R.M.A.; Patinha, D.S.J.; Sousa, G.D.A.; Villaverde, J.; Silva, C.M.; Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P. Eucalyptus biomass residues from agro-forest and pulping industries as sources of high-value triterpenic compounds. Cellul. Chem. Technol. 2011, 45, 475–481. [Google Scholar]
- Santos, S.A.O.; Freire, C.S.R.; Domingues, M.R.M.; Silvestre, A.J.D.; Pascoal Neto, C. Characterization of phenolic components in polar extracts of Eucalyptus globulus Labill. bark by high-performance liquid chromatography-mass spectrometry. J. Agric. Food Chem. 2011, 59, 9386–9393. [Google Scholar] [CrossRef]
- Domingues, R.M.A.; Sousa, G.D.A.; Freire, C.S.R.; Silvestre, A.J.D.; Neto, C.P. Eucalyptus globulus biomass residues from pulping industry as a source of high value triterpenic compounds. Ind. Crops Prod. 2010, 31, 65–70. [Google Scholar] [CrossRef]
- Ko, C.-H.; Wang, Y.-N.; Chang, F.-C.; Chen, J.-J.; Chen, W.-H.; Hwang, W.-S. Potentials of lignocellulosic bioethanols produced from hardwood in Taiwan. Energy 2012, 44, 329–334. [Google Scholar] [CrossRef]
- Romaní, A.; Garrote, G.; Parajó, J.C. Bioethanol production from autohydrolyzed Eucalyptus globulus by simultaneous saccharification and fermentation operating at high solids loading. Fuel 2012, 94, 305–312. [Google Scholar] [CrossRef]
- Silvestre, A.J.D.; Cavaleiro, J.S.; Delmond, B.; Filliatre, C.; Bourgeois, G. Analysis of the variation of the essential oil composition of Eucalyptus globulus Labill. from Portugal using multivariate statistical analysis. Ind. Crops Prod. 1997, 6, 27–33. [Google Scholar] [CrossRef]
- Bachir, R.G.; Benali, M. Antibacterial activity of the essential oils from the leaves of Eucalyptus globulus against Escherichia coli and Staphylococcus aureus. Asian Pac. J. Trop. Biomed. 2012, 2, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Ghaffar, A.; Yameen, M.; Kiran, S.; Kamal, S.; Jalal, F.; Munir, B.; Saleem, S.; Rafiq, N.; Ahmad, A.; Saba, I.; et al. Chemical composition and in-vitro evaluation of the antimicrobial and antioxidant activities of essential oils extracted from seven Eucalyptus species. Molecules 2015, 20, 20487–20498. [Google Scholar] [CrossRef]
- González-Burgos, E.; Liaudanskas, M.; Viškelis, J.; Žvikas, V.; Janulis, V.; Gómez-Serranillos, M.P. Antioxidant activity, neuroprotective properties and bioactive constituents analysis of varying polarity extracts from Eucalyptus globulus leaves. J. Food Drug Anal. 2018, 26, 1293–1302. [Google Scholar] [CrossRef] [PubMed]
- Nile, S.H.; Keum, Y.S. Chemical composition, antioxidant, anti-inflammatory and antitumor activities of Eucalyptus globulus labill. Indian J. Exp. Biol. 2018, 56, 734–742. [Google Scholar]
- Boulekbache-Makhlouf, L.; Meudec, E.; Mazauric, J.P.; Madani, K.; Cheynier, V. Qualitative and semi-quantitative analysis of phenolics in Eucalyptus globulus leaves by high-performance liquid chromatography coupled with diode array detection and electrospray ionisation mass spectrometry. Phytochem. Anal. 2013, 24, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Dezsi, Ș.; Bădărău, A.; Bischin, C.; Vodnar, D.; Silaghi-Dumitrescu, R.; Gheldiu, A.-M.; Mocan, A.; Vlase, L. Antimicrobial and antioxidant activities and phenolic profile of Eucalyptus globulus Labill. and Corymbia ficifolia (F. Muell.) K.D. Hill & L.A.S. Johnson Leaves. Molecules 2015, 20, 4720–4734. [Google Scholar] [CrossRef]
- Rodrigues, V.H.; de Melo, M.M.R.; Portugal, I.; Silva, C.M. Extraction of Eucalyptus leaves using solvents of distinct polarity. Cluster analysis and extracts characterization. J. Supercrit. Fluids 2018, 135, 263–274. [Google Scholar] [CrossRef]
- Guzmán, P.; Fernández, V.; Graça, J.; Cabral, V.; Kayali, N.; Khayet, M.; Gil, L. Chemical and structural analysis of Eucalyptus globulus and E. camaldulensis leaf cuticles: A lipidized cell wall region. Front. Plant Sci. 2014, 5, 481. [Google Scholar] [CrossRef]
- Singh, A.; Ahmad, A.; Bushra, R. Supercritical carbondioxide extraction of essential oils from leaves of Eucalyptus globulus L., their analysis and application. Anal. Methods 2016, 8, 1339–1350. [Google Scholar] [CrossRef]
- Gullón, B.; Muñiz-Mouro, A.; Lú-Chau, T.A.; Moreira, M.T.; Lema, J.M.; Eibes, G. Green approaches for the extraction of antioxidants from eucalyptus leaves. Ind. Crops Prod. 2019, 138, 111473. [Google Scholar] [CrossRef]
- Vecchio, M.G.; Loganes, C.; Minto, C. Beneficial and healthy properties of Eucalyptus plants: A great potential use. Open Agric. J. 2016, 10, 52–57. [Google Scholar] [CrossRef]
- Coppen, J.J.W. Eucalyptus: The Genus Eucalyptus; Taylor & Francis: London, UK, 2002; ISBN 9780415278799. [Google Scholar]
- Barbosa, L.C.A.; Filomeno, C.A.; Teixeira, R.R. Chemical variability and biological activities of Eucalyptus spp. essential oils. Molecules 2016, 21, 1671. [Google Scholar] [CrossRef]
- El-Ghorab, A.H.; El-Massry, K.F.; Marx, F.; Fadel, H.M. Antioxidant activity of Egyptian Eucalyptus camaldulensis var. brevirostris leaf extracts. Food/Nahrung 2003, 47, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Pereira, V.; Dias, C.; Vasconcelos, M.C.; Rosa, E.; Saavedra, M.J. Antibacterial activity and synergistic effects between Eucalyptus globulus leaf residues (essential oils and extracts) and antibiotics against several isolates of respiratory tract infections (Pseudomonas aeruginosa). Ind. Crops Prod. 2014, 52, 1–7. [Google Scholar] [CrossRef]
- Moreira, P.; Sousa, F.J.; Matos, P.; Brites, G.S.; Gonçalves, M.J.; Cavaleiro, C.; Figueirinha, A.; Salgueiro, L.; Batista, M.T.; Branco, P.C.; et al. Chemical composition and effect against skin alterations of bioactive extracts obtained by the hydrodistillation of Eucalyptus globulus leaves. Pharmaceutics 2022, 14, 561. [Google Scholar] [CrossRef] [PubMed]
- Domingues, R.; Guerra, A.; Duarte, M.; Freire, C.; Neto, C.; Silva, C.; Silvestre, A. Bioactive triterpenic acids: From agroforestry biomass residues to promising therapeutic tools. Mini Rev. Org. Chem. 2014, 11, 382–399. [Google Scholar] [CrossRef]
- Qian, W.; Wang, W.; Zhang, J.; Wang, T.; Liu, M.; Yang, M.; Sun, Z.; Li, X.; Li, Y. Antimicrobial and antibiofilm activities of ursolic acid against carbapenem-resistant Klebsiella pneumoniae. J. Antibiot. 2020, 73, 382–391. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Rebolledo, G.A.; Siordia-Reyes, A.G.; Meckes-Fischer, M.; Jiménez-Arellanes, A. Hepatoprotective properties of oleanolic and ursolic acids in antitubercular drug-induced liver damage. Asian Pac. J. Trop. Med. 2016, 9, 644–651. [Google Scholar] [CrossRef]
- Somova, L.O.; Nadar, A.; Rammanan, P.; Shode, F.O. Cardiovascular, antihyperlipidemic and antioxidant effects of oleanolic and ursolic acids in experimental hypertension. Phytomedicine 2003, 10, 115–121. [Google Scholar] [CrossRef]
- Pavlova, N.I.; Savinova, O.V.; Nikolaeva, S.N.; Boreko, E.I.; Flekhter, O.B. Antiviral activity of betulin, betulinic and betulonic acids against some enveloped and non-enveloped viruses. Fitoterapia 2003, 74, 489–492. [Google Scholar] [CrossRef]
- Alakurtti, S.; Bergström, P.; Sacerdoti-Sierra, N.; Jaffe, C.L.; Yli-Kauhaluoma, J. Anti-leishmanial activity of betulin derivatives. J. Antibiot. 2010, 63, 123–126. [Google Scholar] [CrossRef] [PubMed]
- De Sá, M.S.; Costa, J.F.O.; Krettli, A.U.; Zalis, M.G.; De Azevedo Maia, G.L.; Sette, I.M.F.; De Amorim Câmara, C.; Filho, J.M.B.; Giulietti-Harley, A.M.; Ribeiro Dos Santos, R.; et al. Antimalarial activity of betulinic acid and derivatives in vitro against Plasmodium falciparum and in vivo in P. berghei-infected mice. Parasitol. Res. 2009, 105, 275–279. [Google Scholar] [CrossRef]
- Chiang, Y.M.; Chang, J.Y.; Kuo, C.C.; Chang, C.Y.; Kuo, Y.H. Cytotoxic triterpenes from the aerial roots of Ficus microcarpa. Phytochemistry 2005, 66, 495–501. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, T.; Kashiwada, Y.; Kilkuskie, R.E.; Cosentino, L.M.; Bailas, L.M.; Jiang, J.B.; Janzen, W.P.; Chen, I.S.; Lee, K.H. Anti-AIDS agents, 11. Betulinic acid and platanic acid as anti-HIV principles from Syzigium claviflorum, and the anti-HIV activity of structurally related triterpenoids. J. Nat. Prod. 1994, 57, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen Recio, M.; Giner, R.M.; Manez, S.; Gueho, J.; Julien, H.R.; Hostettmann, K.; Rios, J.L. Investigations on the steroidal anti-inflammatory activity of triterpenoids from Diospyros leucomelas. Planta Med. 1995, 61, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Patinha, D.J.S.; Domingues, R.M.A.; Villaverde, J.J.; Silva, A.M.S.; Silva, C.M.; Freire, C.S.R.; Neto, C.P.; Silvestre, A.J.D. Lipophilic extractives from the bark of Eucalyptus grandis x globulus, a rich source of methyl morolate: Selective extraction with supercritical CO2. Ind. Crops Prod. 2013, 43, 340–348. [Google Scholar] [CrossRef]
- Cordeiro, N.; Freitas, N.; Faria, M.; Gouveia, M. Ipomoea batatas (L.) Lam.: A rich source of lipophilic phytochemicals. J. Agric. Food Chem. 2013, 61, 12380–12384. [Google Scholar] [CrossRef]
- Devappa, R.K.; Rakshit, S.K.; Dekker, R.F.H. Forest biorefinery: Potential of poplar phytochemicals as value-added co-products. Biotechnol. Adv. 2015, 33, 681–716. [Google Scholar] [CrossRef]
- Ramos, P.A.B.; Moreirinha, C.; Santos, S.A.O.; Almeida, A.; Freire, C.S.R.; Silva, A.M.S.; Silvestre, A.J.D. Valorisation of bark lipophilic fractions from three Portuguese Salix species: A systematic study of the chemical composition and inhibitory activity on Escherichia coli. Ind. Crop. Prod. 2019, 132, 245–252. [Google Scholar] [CrossRef]
- Chanioti, S.; Katsouli, M.; Tzia, C. β-Sitosterol as a functional bioactive. In A Centum of Valuable Plant Bioactives; Mushtaq, M., Anwar, F., Eds.; Academic Press: London, UK, 2021; pp. 193–212. ISBN 9780128229231. [Google Scholar]
- Huang, Z.-R.; Lin, Y.-K.; Fang, J.-Y. Biological and pharmacological activities of squalene and related compounds: Potential uses in cosmetic dermatology. Molecules 2009, 14, 540–554. [Google Scholar] [CrossRef]
- Yang, X.; Li, Y.; Jiang, W.; Ou, M.; Chen, Y.; Xu, Y.; Wu, Q.; Zheng, Q.; Wu, F.; Wang, L.; et al. Synthesis and biological evaluation of novel ursolic acid derivatives as potential anticancer prodrugs. Chem. Biol. Drug Des. 2015, 86, 1397–1404. [Google Scholar] [CrossRef]
- Ramos, A.A.; Lima, C.F.; Pereira, M.L.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Antigenotoxic effects of quercetin, rutin and ursolic acid on HepG2 cells: Evaluation by the comet assay. Toxicol. Lett. 2008, 177, 66–73. [Google Scholar] [CrossRef]
- Li, T.; Chen, X.; Liu, Y.; Fan, L.; Lin, L.; Xu, Y.; Chen, S.; Shao, J. pH-Sensitive mesoporous silica nanoparticles anticancer prodrugs for sustained release of ursolic acid and the enhanced anti-cancer efficacy for hepatocellular carcinoma cancer. Eur. J. Pharm. Sci. 2017, 96, 456–463. [Google Scholar] [CrossRef]
- Dong, H.; Yang, X.; Xie, J.; Xiang, L.; Li, Y.; Ou, M.; Chi, T.; Liu, Z.; Yu, S.; Gao, Y.; et al. UP12, a novel ursolic acid derivative with potential for targeting multiple signaling pathways in hepatocellular carcinoma. Biochem. Pharmacol. 2015, 93, 151–162. [Google Scholar] [CrossRef]
- Zhang, T.-Y.; Li, C.-S.; Li, P.; Bai, X.-Q.; Guo, S.-Y.; Jin, Y.; Piao, S.-J. Synthesis and evaluation of ursolic acid-based 1,2,4-triazolo[1,5-a]pyrimidines derivatives as anti-inflammatory agents. Mol. Divers. 2022, 26, 27–38. [Google Scholar] [CrossRef]
- Jang, S.-E.; Jeong, J.-J.; Hyam, S.R.; Han, M.J.; Kim, D.-H. Ursolic acid isolated from the seed of Cornus officinalis ameliorates colitis in mice by inhibiting the binding of lipopolysaccharide to toll-like receptor 4 on macrophages. J. Agric. Food Chem. 2014, 62, 9711–9721. [Google Scholar] [CrossRef]
- Kim, M.-H.; Kim, J.N.; Han, S.N.; Kim, H.-K. Ursolic acid isolated from guava leaves inhibits inflammatory mediators and reactive oxygen species in LPS-stimulated macrophages. Immunopharmacol. Immunotoxicol. 2015, 37, 228–235. [Google Scholar] [CrossRef]
- Wójciak-Kosior, M.; Paduch, R.; Matysik-Woźniak, A.; Niedziela, P.; Donica, H. The effect of ursolic and oleanolic acids on human skin fibroblast cells. Folia Histochem. Cytobiol. 2011, 49, 664–669. [Google Scholar] [CrossRef]
- Qiang, Z.; Ye, Z.; Hauck, C.; Murphy, P.A.; McCoy, J.-A.; Widrlechner, M.P.; Reddy, M.B.; Hendrich, S. Permeability of rosmarinic acid in Prunella vulgaris and ursolic acid in Salvia officinalis extracts across Caco-2 cell monolayers. J. Ethnopharmacol. 2011, 137, 1107–1112. [Google Scholar] [CrossRef]
- Mlala, S.; Oyedeji, A.O.; Gondwe, M.; Oyedeji, O.O. Ursolic acid and its derivatives as bioactive agents. Molecules 2019, 24, 2751. [Google Scholar] [CrossRef]
- Kashyap, D.; Tuli, H.S.; Sharma, A.K. Ursolic acid (UA): A metabolite with promising therapeutic potential. Life Sci. 2016, 146, 201–213. [Google Scholar] [CrossRef]
- Park, H.J.; Jo, D.S.; Choi, D.S.; Bae, J.-E.; Park, N.Y.; Kim, J.-B.; Chang, J.H.; Shin, J.J.; Cho, D.-H. Ursolic acid inhibits pigmentation by increasing melanosomal autophagy in B16F1 cells. Biochem. Biophys. Res. Commun. 2020, 531, 209–214. [Google Scholar] [CrossRef]
- Cho, J.; Rho, O.; Junco, J.; Carbajal, S.; Siegel, D.; Slaga, T.J.; DiGiovanni, J. Effect of combined treatment with ursolic acid and resveratrol on skin tumor promotion by 12-O-tetradecanoylphorbol-13-acetate. Cancer Prev. Res. 2015, 8, 817–825. [Google Scholar] [CrossRef]
- Samivel, R.; Nagarajan, R.P.; Subramanian, U.; Khan, A.A.; Masmali, A.; Almubrad, T.; Akhtar, S. Inhibitory effect of ursolic acid on ultraviolet B radiation-induced oxidative stress and proinflammatory response-mediated senescence in human skin dermal fibroblasts. Oxid. Med. Cell. Longev. 2020, 2020, 1246510. [Google Scholar] [CrossRef]
- Zheng, Y.; Huang, C.; Zhao, L.; Chen, Y.; Liu, F. Regulation of decorin by ursolic acid protects against non-alcoholic steatohepatitis. Biomed. Pharmacother. 2021, 143, 112166. [Google Scholar] [CrossRef]
- Wan, S.-Z.; Liu, C.; Huang, C.-K.; Luo, F.-Y.; Zhu, X. Ursolic acid improves intestinal damage and bacterial dysbiosis in liver fibrosis mice. Front. Pharmacol. 2019, 10, 1321. [Google Scholar] [CrossRef]
- Hao, W.; Kwek, E.; He, Z.; Zhu, H.; Liu, J.; Zhao, Y.; Ma, K.Y.; He, W.-S.; Chen, Z.-Y. Ursolic acid alleviates hypercholesterolemia and modulates the gut microbiota in hamsters. Food Funct. 2020, 11, 6091–6103. [Google Scholar] [CrossRef]
- Li, J.; Li, N.; Yan, S.; Liu, M.; Sun, B.; Lu, Y.; Shao, Y. Ursolic acid alleviates inflammation and against diabetes-induced nephropathy through TLR4-mediated inflammatory pathway. Mol. Med. Rep. 2018, 18, 4675–4681. [Google Scholar] [CrossRef]
- Wang, X.; Gong, Y.; Zhou, B.; Yang, J.; Cheng, Y.; Zhao, J.; Qi, M. Ursolic acid ameliorates oxidative stress, inflammation and fibrosis in diabetic cardiomyopathy rats. Biomed. Pharmacother. 2018, 97, 1461–1467. [Google Scholar] [CrossRef]
- Freire, C.S.R.; Coelho, D.S.C.; Santos, N.M.; Silvestre, A.J.D.; Pascoal Neto, C. Identification of Δ7 phytosterols and phytosteryl glucosides in the wood and bark of several Acacia species. Lipids 2005, 40, 317–322. [Google Scholar] [CrossRef]
- Santos, S.; Trindade, S.; Oliveira, C.; Parreira, P.; Rosa, D.; Duarte, M.; Ferreira, I.; Cruz, M.; Rego, A.; Abreu, M.; et al. Lipophilic fraction of cultivated Bifurcaria bifurcata R. Ross: Detailed composition and in vitro prospection of current challenging bioactive properties. Mar. Drugs 2017, 15, 340. [Google Scholar] [CrossRef] [PubMed]
- Sittampalam, G.S.; Coussens, N.B.P.; Brimacombe, K.; Grossman, A.B.; Arkin, M.B.; Auld, D.B.; Austin, C.; Baell, J.B.; Bejcek, B.B.; Chung, T.; et al. Assay Guidance Manual: Quantitative Biology and Pharmacology in Preclinical Drug Discovery; 2004; pp. 1–12. Available online: https://ascpt.onlinelibrary.wiley.com/doi/full/10.1111/cts.12570 (accessed on 11 January 2022).

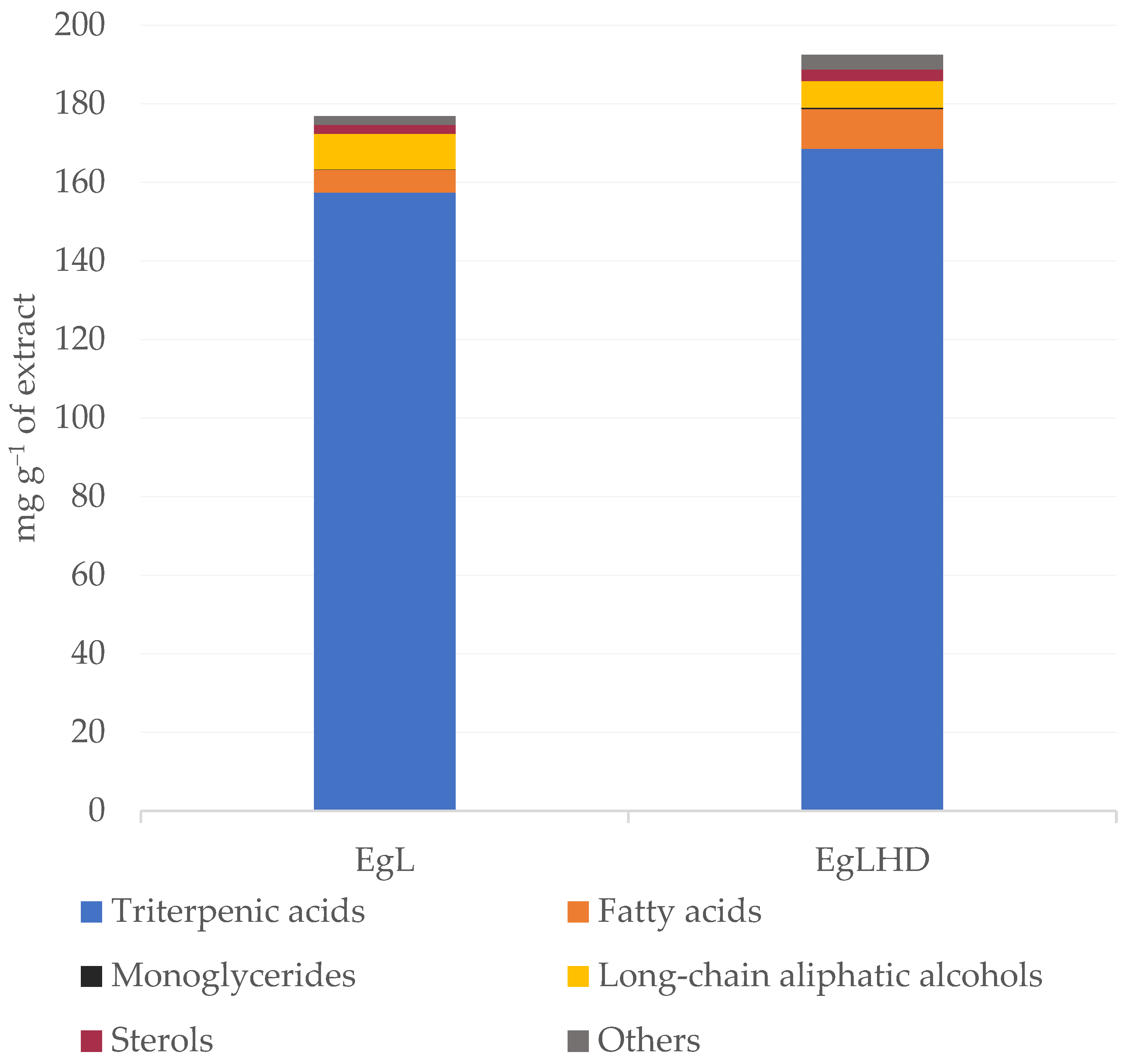
| Rt(min) | Compound | mg g−1 of Extract | g kg−1 of dw | ||
|---|---|---|---|---|---|
| EgL | EgLHD | EgL | EgLHD | ||
| Triterpenic acids | 157.42 | 168.53 | 30.63 | 37.14 | |
| 68.11 | Betulonic acid | 18.56 | 29.40 | 3.61 | 6.48 |
| 68.93 | Oleanolic acid | 28.02 | 28.70 | 5.45 | 6.32 |
| 69.52 | Betulinic acid | 7.05 | 6.49 | 1.37 | 1.43 |
| 70.46 | Ursolic acid | 89.73 | 89.93 | 17.46 | 19.82 |
| 73.09 | 3-Acetyloleanolic acid | 5.11 | 4.56 | 0.99 | 1.00 |
| 76.20 | 3-Acetylbetulinic acid | 6.85 | 7.14 | 1.33 | 1.57 |
| 77.25 | 3-Acetylursolic acid | 2.09 | 2.32 | 0.41 | 0.51 |
| Fatty acids | 5.79 | 10.14 | 1.13 | 2.23 | |
| Saturated fatty acids | 5.79 | 9.57 | 1.13 | 2.11 | |
| 30.83 | Tetradecanoic acid | 0.29 | 0.52 | 0.06 | 0.11 |
| 35.74 | Hexadecanoic acid | 0.59 | 2.61 | 0.11 | 0.58 |
| 38.03 | Heptadecanoic acid | tr | 0.12 | tr | 0.03 |
| 40.24 | Octadecanoic acid | 0.13 | 0.26 | 0.03 | 0.06 |
| 48.26 | Docosanoic acid | 0.21 | 0.29 | 0.04 | 0.06 |
| 51.94 | Tetracosanoic acid | 0.33 | 0.47 | 0.06 | 0.10 |
| 55.97 | Hexacosanoic acid | 1.68 | 1.61 | 0.33 | 0.35 |
| 60.37 | Octacosanoic acid | 0.54 | 1.29 | 0.11 | 0.28 |
| 65.28 | Triacontanoic acid | 2.02 | 2.41 | 0.39 | 0.53 |
| Unsaturated fatty acids | tr | 0.42 | tr | 0.09 | |
| 39.32 | Octadeca-9,12-dienoic acid | tr | tr | tr | tr |
| 39.51 | cis-Octadec-9-enoic acid | tr | 0.34 | tr | 0.07 |
| 39.65 | trans-Octadec-9-enoic acid | tr | 0.08 | tr | 0.02 |
| Diacids | tr | 0.15 | tr | 0.03 | |
| 29.37 | Nonanedioic acid | tr | 0.15 | tr | 0.03 |
| Long-chain aliphatic alcohols | 9.06 | 6.85 | 1.76 | 1.51 | |
| 50.41 | Tetracosan-1-ol | 0.32 | 0.19 | 0.06 | 0.04 |
| 54.24 | Hexacosan-1-ol | 1.23 | 0.96 | 0.24 | 0.21 |
| 58.49 | Octacosan-1-ol | 2.33 | 1.79 | 0.45 | 0.39 |
| 63.06 | Triacontan-1-ol | 5.18 | 3.91 | 1.01 | 0.86 |
| Monoglycerides | 0.07 | 0.29 | 0.01 | 0.03 | |
| 47.57 | 1-Monohexadecanoin | 0.07 | 0.29 | 0.01 | 0.03 |
| Sterols | 2.33 | 2.91 | 0.45 | 0.64 | |
| 62.61 | β-Sitosterol | 2.33 | 2.91 | 0.45 | 0.64 |
| Others | 2.13 | 3.70 | 0.42 | 0.82 | |
| 14.14 | Glycerol | tr | 0.28 | tr | 0.06 |
| 22.95 | Tyrosol | n.d. | 0.05 | n.d. | 0.01 |
| 34.03 | Gallic acid | n.d. | 0.19 | n.d. | 0.04 |
| 57.23 | 1,6-Dihydroxy-2-methylanthraquinone | 1.16 | 0.99 | 0.23 | 0.22 |
| 58.03 | α-Tocopherol | 0.97 | 2.19 | 0.19 | 0.48 |
| TOTAL | 176.81 | 192.44 | 34.41 | 42.37 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, C.S.D.; Moreira, P.; Cruz, M.T.; Pereira, C.M.F.; Silva, A.M.S.; Santos, S.A.O.; Silvestre, A.J.D. Exploiting the Integrated Valorization of Eucalyptus globulus Leaves: Chemical Composition and Biological Potential of the Lipophilic Fraction before and after Hydrodistillation. Int. J. Mol. Sci. 2023, 24, 6226. https://doi.org/10.3390/ijms24076226
Oliveira CSD, Moreira P, Cruz MT, Pereira CMF, Silva AMS, Santos SAO, Silvestre AJD. Exploiting the Integrated Valorization of Eucalyptus globulus Leaves: Chemical Composition and Biological Potential of the Lipophilic Fraction before and after Hydrodistillation. International Journal of Molecular Sciences. 2023; 24(7):6226. https://doi.org/10.3390/ijms24076226
Chicago/Turabian StyleOliveira, Cátia. S. D., Patrícia Moreira, Maria T. Cruz, Cláudia M. F. Pereira, Artur M. S. Silva, Sónia A. O. Santos, and Armando J. D. Silvestre. 2023. "Exploiting the Integrated Valorization of Eucalyptus globulus Leaves: Chemical Composition and Biological Potential of the Lipophilic Fraction before and after Hydrodistillation" International Journal of Molecular Sciences 24, no. 7: 6226. https://doi.org/10.3390/ijms24076226
APA StyleOliveira, C. S. D., Moreira, P., Cruz, M. T., Pereira, C. M. F., Silva, A. M. S., Santos, S. A. O., & Silvestre, A. J. D. (2023). Exploiting the Integrated Valorization of Eucalyptus globulus Leaves: Chemical Composition and Biological Potential of the Lipophilic Fraction before and after Hydrodistillation. International Journal of Molecular Sciences, 24(7), 6226. https://doi.org/10.3390/ijms24076226











