Intensity Ratio of Kβ/Kα in Selected Elements from Mg to Cu, and the Chemical Effects of Cr Kα1,2 Diagram Lines and Cr Kβ/Kα Intensity Ratio in Cr Compounds
Abstract
1. Introduction
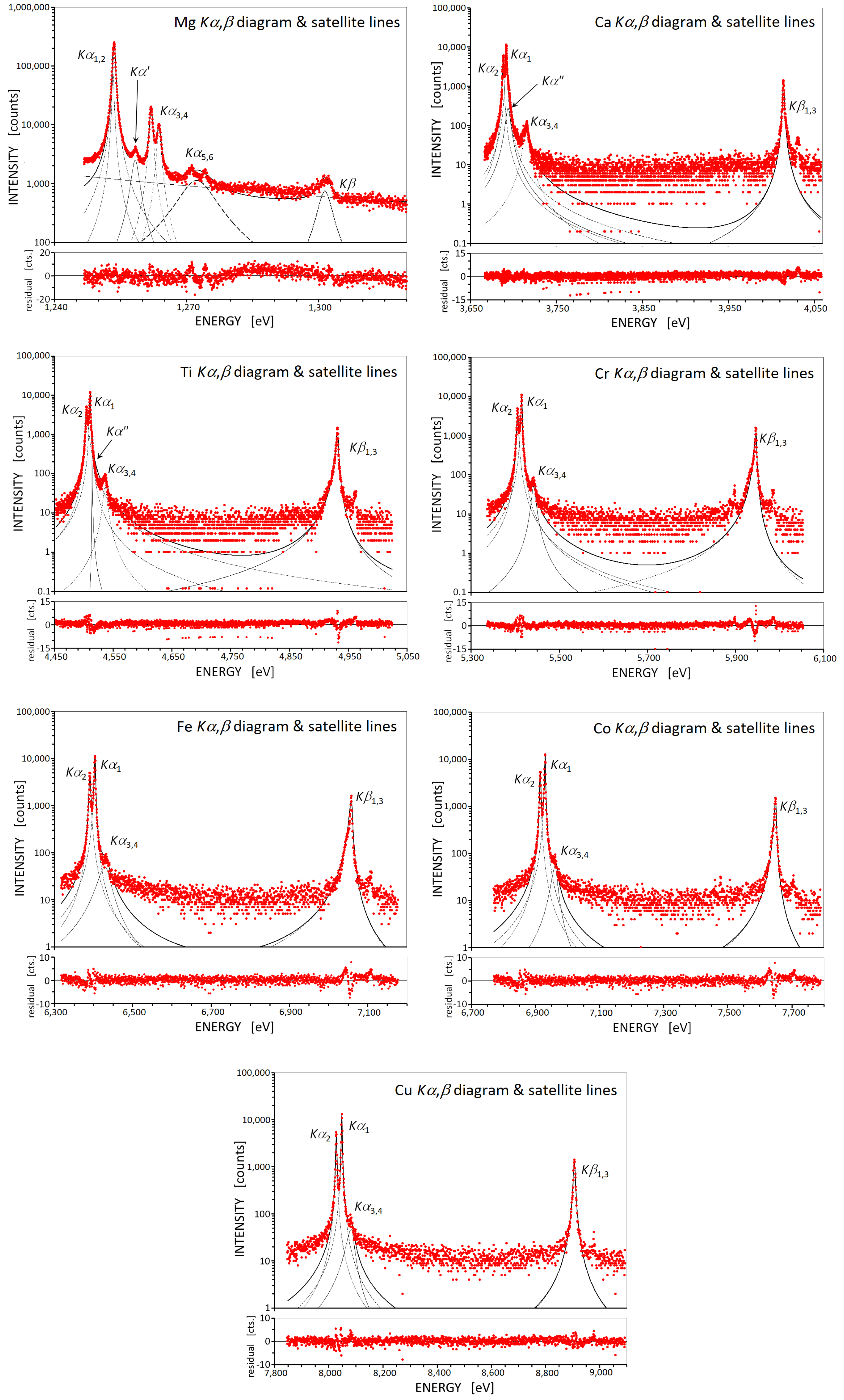
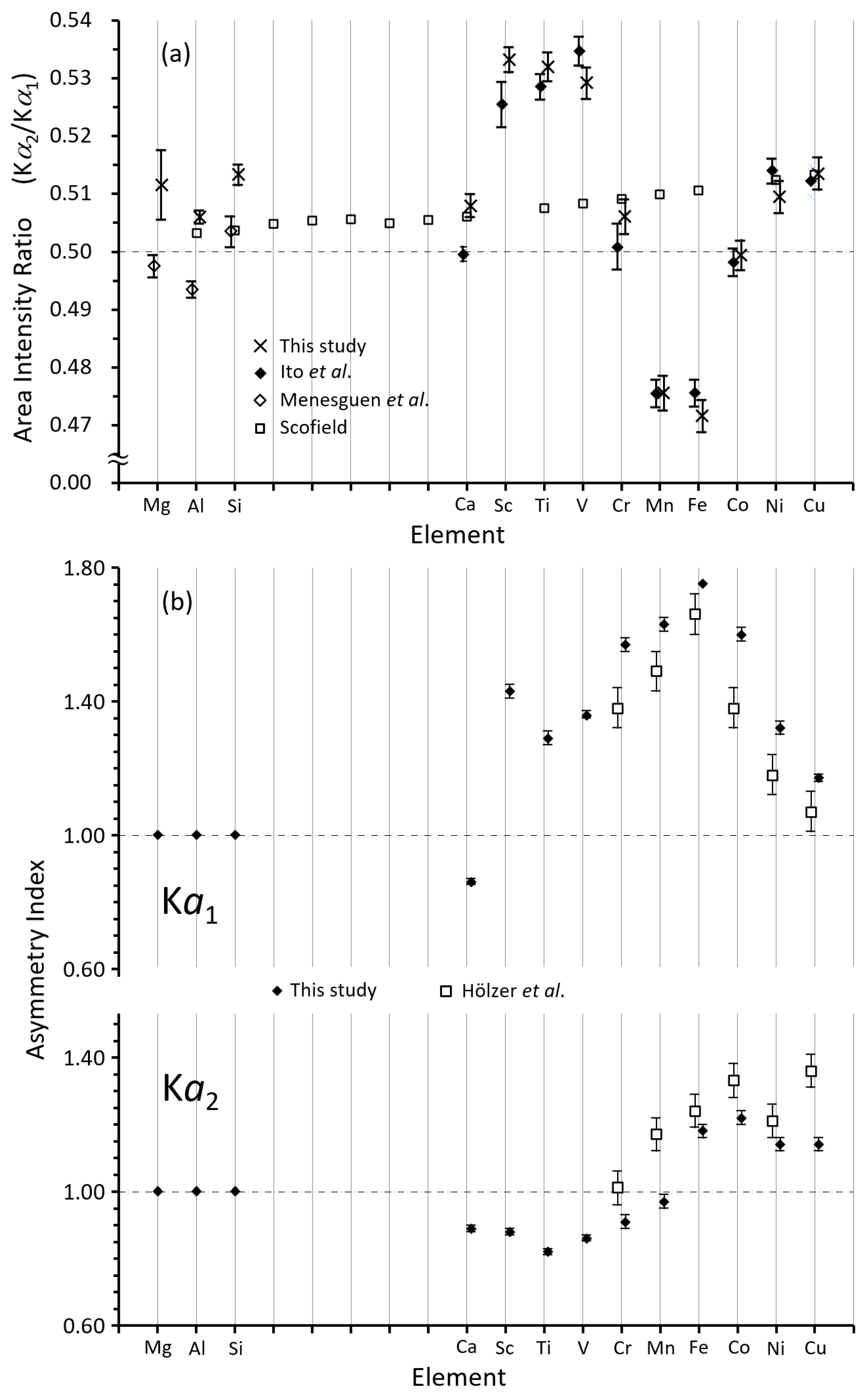
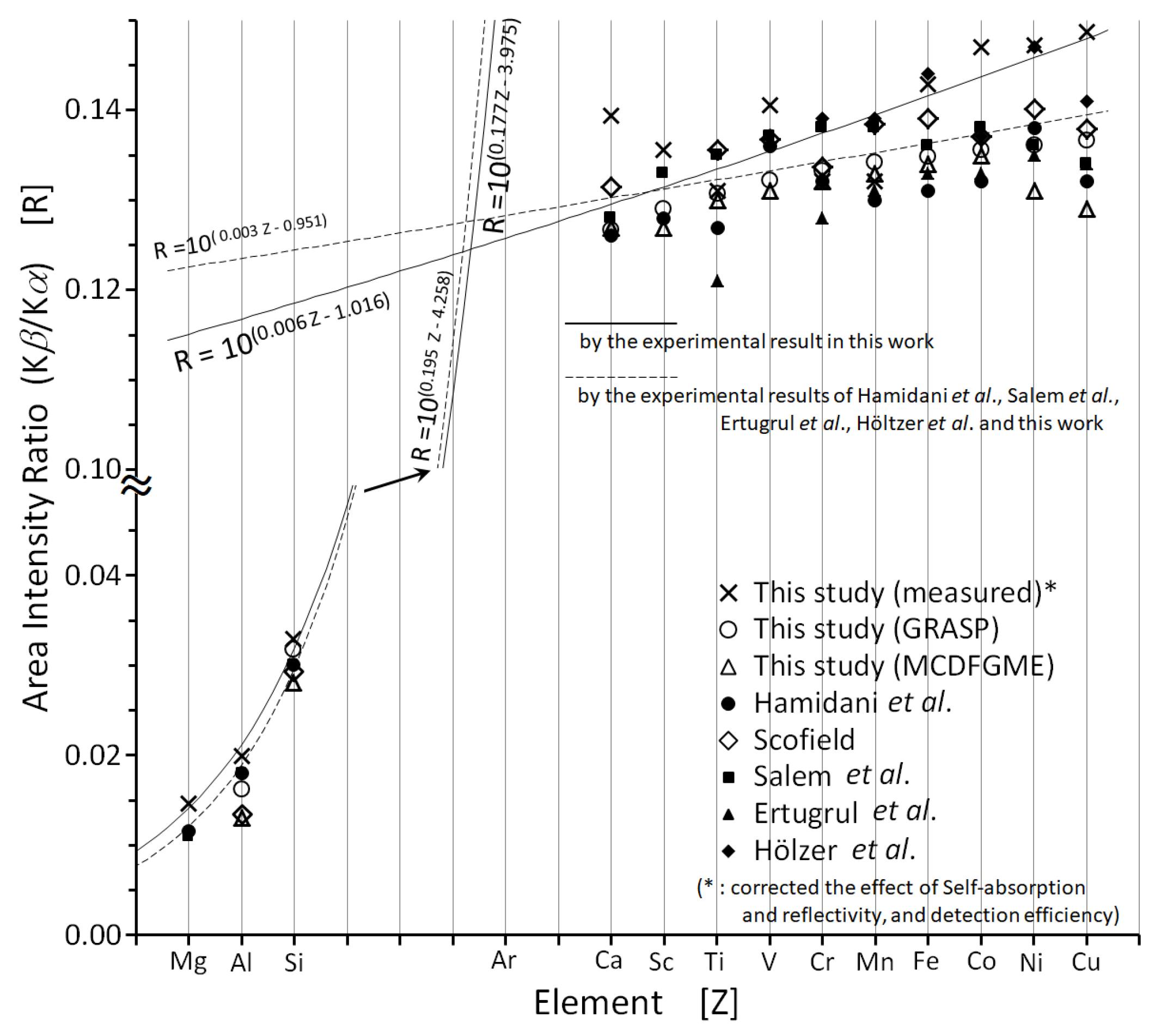
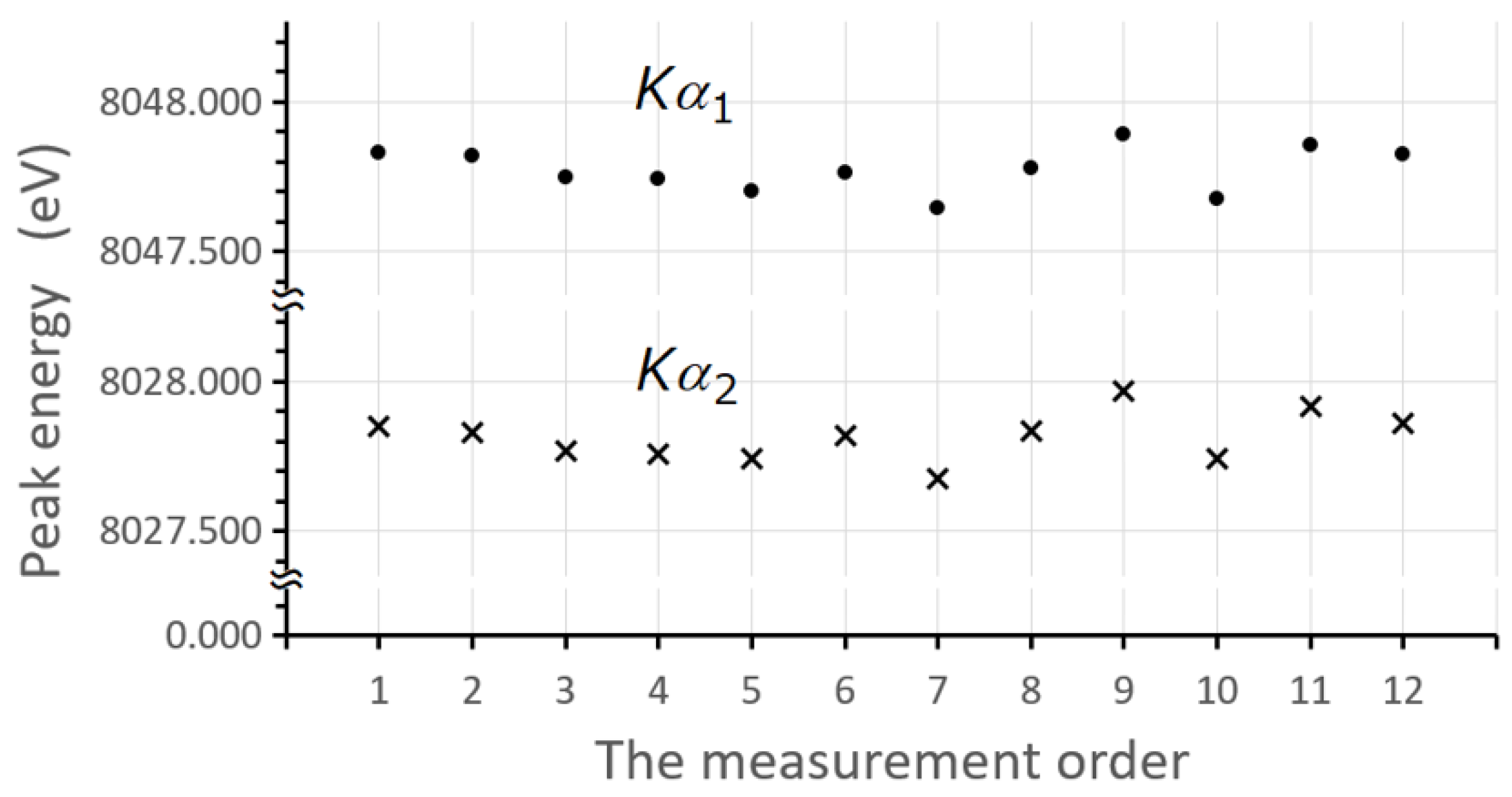
2. Results and Discussion
2.1. The Intensity Ratio in the Elements from Mg to Cu
2.2. Cr Compounds
3. Methods and Materials
3.1. Experimental Procedure
- CrO and FeO(CoO) are mixed in an agate mortar and ground well.
- Vacuum-seal the mixture in a glass tube and sinter it.
- Measure of the X-ray diffraction pattern of the resulting sample and repeat steps 1 and 2 until there are no extra peaks.
3.2. Theoretical Procedure
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hamidani, A.; Daoudi, S.; Kahoul, A.; Sampaio, J.M.; Marques, J.P.; Parente, F.; Croft, S.; Favalli, A.; Kup Aylikci, N.; Aylikci, V.; et al. Updated database, semi-empirical and theoretical calculation of Kβ/Kα intensity ratios for elements ranging from 11Na to 96Cm. At. Data Nucl. Data Tables 2023, 149, 101549. [Google Scholar] [CrossRef]
- Berenyi, D. Kα/Kβ ratio for X-ray transitions in higher energy collision processes. Bull. Inst. Chem. Res. Kyoto Univ. 1979, 57, 139. [Google Scholar]
- Scofield, J.H. Exchange corrections of K X-ray emission rates. Phys. Rev. A 1974, 9, 1041–1049. [Google Scholar] [CrossRef]
- Paić, G.; Pečar, V. Study of anomalies in Kβ/Kα ratios observed following K-electron capture. Phys. Rev. A 1976, 14, 2190–2192. [Google Scholar] [CrossRef]
- Arndt, E.; Brunner, G.; Hartmann, E. Intensity ratios for X-ray production in 3d elements by photoionisation and electron capture. J. Phys. B At. Mol. Phys. 1982, 15, L887. [Google Scholar] [CrossRef]
- Ito, Y.; Tochio, T.; Ohashi, H.; Yamashita, M.; Fukushima, S.; Polasik, M.; Słabkowska, K.; Syrocki, Ł.; Szymańska, E.; Rzadkiewicz, J.; et al. Kα1,2 X-ray linewidths, asymmetry indices, and [KM] shake probabilities in elements Ca to Ge and comparison with theory for Ca, Ti, and Ge. Phys. Rev. A 2016, 94, 042506. [Google Scholar] [CrossRef]
- Ito, Y.; Tochio, T.; Yamashita, M.; Fukushima, S.; Vlaicu, A.M.; Syrocki, Ł.; Słabkowska, K.; Weder, E.; Polasik, M.; Sawicka, K.; et al. Structure of high-resolution Kβ1,3 X-ray emission spectra for the elements from Ca to Ge. Phys. Rev. A 2018, 97, 052505. [Google Scholar] [CrossRef]
- Deutsch, M.; Hölzer, G.; Härtwig, J.; Wolf, J.; Fritsch, M.; Förster, E. Kα and Kβ X-ray emission spectra of copper. Phys. Rev. A 1995, 51, 283–296. [Google Scholar] [CrossRef]
- Hölzer, G.; Fritsch, M.; Deutsch, M.; Härtwig, J.; Förster, E. Kα1,2 and Kβ1,3 X-ray emission lines of the 3d transition metals. Phys. Rev. A 1997, 56, 4554–4568. [Google Scholar] [CrossRef]
- Tamaki, Y.; Omori, T.; Shiokawa, T. Chemical effect on the Kα/Kβ intensity ratios in the 51Cr- labelled chromium compounds. Radiochem. Radioanal. Lett. 1975, 20, 255–262. [Google Scholar]
- Lazzariimi, E.; Fantola, A.L.L.; Bettoni, M.M. On the Kα/Kβ Intensity Ratios for V and Mn Formed by EC Decay of 51Cr and 55Fe in Several Compounds and Doped Crystals. Radiochim. Acta 1978, 25, 81–84. [Google Scholar] [CrossRef]
- Collins, K.E.; Collins, C.H.; Heitz, C. Use of Kβ/Kα Intensity Ratios for the Study of Annealing Processes in 51Cr-doped Solids. Radiochim. Acta 1981, 28, 7–12. [Google Scholar] [CrossRef]
- Brunner, G.; Nagel, M.; Hartmann, E.; Arndt, E. Chemical sensitivity of the Kβ/Kα X-ray intensity ratio for 3d elements. J. Phys. B At. Mol. Phys. 1982, 15, 4517. [Google Scholar] [CrossRef]
- Urch, D.S. X-ray Emission Spectroscopy. In Electron Spectroscopy: Theory, Techniques and Applications; Brundle, C., Baker, A., Eds.; Academic Press: New York, NY, USA, 1979; Volume 3, p. 1. [Google Scholar]
- Meisel, A.; Leonhardt, G.; Szargan, R. Röntgenspektren und Chemische Bindung; Geest und Portig: Leipzig, Germany, 1977. [Google Scholar]
- Barinski, R.; Nefedov, W. Röntgenspektroskopische Bestimmung der Atomladungen in Molekülen; Geest und Portig: Leipzig, Germany, 1969. [Google Scholar]
- Mukoyama, T.; Taniguchi, K.; Adachi, H. Chemical effect on Kβ:Kα X-ray intensity ratios. Phys. Rev. B 1986, 34, 3710–3716. [Google Scholar] [CrossRef]
- Mukoyama, T.; Taniguchi, K.; Adachi, H. Single-atom approximation for Kβ-to-Kα X-ray intensity ratios in chemical compounds of 3d elements. Phys. Rev. A 2001, 63, 042514. [Google Scholar] [CrossRef]
- Ishizuka, T.; Vlaicu, A.M.; Tochio, T.; Ito, Y.; Mukoyama, T.; Hayakawa, S.; Gohshi, Y.; Kawai, S.; Motoyama, M.; Shoji, T. X-ray emission spectra by a simple-quasi-two-crystal spectrometer. Adv.-Ray Chem. Anal. Jpn. 1998, 30, 21. [Google Scholar]
- Ito, Y.; Tochio, T.; Yamashita, M.; Fukushima, S.; Vlaicu, A.M.; Marques, J.P.; Sampaio, J.M.; Guerra, M.; Santos, J.P.; Syrocki, Ł.; et al. Structure of Kα1,2- and Kβ1,3-emission x-ray spectra for Se, Y, and Zr. Phys. Rev. A 2020, 102, 052820. [Google Scholar] [CrossRef]
- Shigemi, A.; Tochio, T.; Ishizuka, T.; Shigeoka, N.; Ito, K.; Vlaicu, A.M.; Ito, Y.; Mukoyama, T.; Gohshi, Y. K X-ray emission spectra of Ni in nickel(II) Schiff base complexes. X-ray Spectrom. 1999, 28, 478–483. [Google Scholar] [CrossRef]
- Chantler, C.T.; Kinnane, M.N.; Su, C.H.; Kimpton, J.A. Characterization of Kα spectral profiles for vanadium, component redetermination for scandium, titanium, chromium, and manganese, and development of satellite structure for Z=21 to Z=25. Phys. Rev. A 2006, 73, 012508. [Google Scholar] [CrossRef]
- Ménesguen, Y.; Lépy, M.C.; Ito, Y.; Yamashita, M.; Fukushima, S.; Tochio, T.; Polasik, M.; Słabkowska, K.; Syrocki, Ł.; Indelicato, P.; et al. Structure of single KL0–, double KL1–, and triple KL2 ionization in Mg, Al, and Si targets induced by photons, and their absorption spectra. Radiat. Phys. Chem. 2022, 194, 110048. [Google Scholar] [CrossRef]
- Tochio, T.; Ito, Y.; Omote, K. Broadening of the X-ray emission line due to the instrumental function of the double-crystal spectrometer. Phys. Rev. A 2002, 65, 042502. [Google Scholar] [CrossRef]
- Salem, S.I.; Falconer, T.H.; Winchell, R.W. Kβ/Kα Radiative-Transition-Probability Ratios for Elements of Low Atomic Numbers in Amorphous and Crystal Forms. Phys. Rev. A 1972, 6, 2147–2150. [Google Scholar] [CrossRef]
- Ertugrul, M.; Sögüt, O.; Simsek, O.; Büyükkasap, E. Measurement of Kβ/Kα intensity ratios for elements in the range 22 ≤ Z ≤ 69 at 59.5 keV. J. Phys. B At. Mol. Opt. Phys. 2001, 34, 909. [Google Scholar] [CrossRef]
- Bearden, J.A. X-ray Wavelengths. Rev. Mod. Phys. 1967, 39, 78–124. [Google Scholar] [CrossRef]
- Deslattes, R.D.; Kessler, E.G.; Indelicato, P.; de Billy, L.; Lindroth, E.; Anton, J. X-ray transition energies: New approach to a comprehensive evaluation. Rev. Mod. Phys. 2003, 75, 35–99. [Google Scholar] [CrossRef]
- Shuvaev, A.T.; Kukulyabin, G.M. Effect of the change in valence on K emission spectra of chromium. Izv. Akad. Nauk SSSR Ser. Fiz. 1963, 27, 332. [Google Scholar]
- Oku, M.; Wagatsuma, K.; Konishi, T. Transition metal 2p X-ray photoelectron and high-resolution Kα X-ray emission spectra of K2CrO4 and KMnO4. X-ray Spectrom. 1999, 28, 464–469. [Google Scholar] [CrossRef]
- Leonhardt, G.; Meisel, A. Über den einflußder chemischen bindung auf die Kα1,2– und Kβ1,3–linien von eisen und chrom in ihren verbindungen–ein beitrag zur theoretischen interpretation röntgenspektroskopischer untersuchungen an verbindungen der 3d-übergangselemente. Spectrochim. Acta Part B At. Spectrosc. 1970, 25, 163–174. [Google Scholar] [CrossRef]
- Tsutsumi, K.; Nakamori, H. X-ray K Emission Spectra of Chromium in Various Chromium Compounds. J. Phys. Soc. Jpn. 1968, 25, 1418–1423. [Google Scholar] [CrossRef]
- Berger, M.J.; Hubbell, J.H.; Seltzer, S.M.; Chang, J.; Coursey, J.S.; Sukumar, R.; Zucker, D.S.; Olsen, K. XCOM: Photon Cross Sections Database, NIST Standard Reference Database 8 (XGAM); (Version 1.5); The U.S. Secretary of Commerce: Washington, DC, USA, 2010. [Google Scholar] [CrossRef]
- Dyall, K.; Grant, I.; Johnson, C.; Parpia, F.; Plummer, E. GRASP: A general-purpose relativistic atomic structure program. Comput. Phys. Commun. 1989, 55, 425–456. [Google Scholar] [CrossRef]
- Parpia, F.; Fischer, C.; Grant, I. GRASP92: A package for large-scale relativistic atomic structure calculations. Comput. Phys. Commun. 1996, 94, 249–271. [Google Scholar] [CrossRef]
- Jönsson, P.; He, X.; Froese Fischer, C.; Grant, I. The grasp2K relativistic atomic structure package. Comput. Phys. Commun. 2007, 177, 597–622. [Google Scholar] [CrossRef]
- Grant, I. Gauge invariance and relativistic radiative transitions. J. Phys. B At. Mol. Phys. 1974, 7, 1458. [Google Scholar] [CrossRef]
- Grant, I.; McKenzie, B.; Norrington, P.; Mayers, D.; Pyper, N. An atomic multiconfigurational Dirac-Fock package. Comput. Phys. Commun. 1980, 21, 207–231. [Google Scholar] [CrossRef]
- Grant, I.P.; McKenzie, B.J. The transverse electron-electron interaction in atomic structure calculations. J. Phys. B At. Mol. Phys. 1980, 13, 2671. [Google Scholar] [CrossRef]
- McKenzie, B.; Grant, I.; Norrington, P. A program to calculate transverse Breit and QED corrections to energy levels in a multiconfiguration Dirac-Fock environment. Comput. Phys. Commun. 1980, 21, 233–246. [Google Scholar] [CrossRef]
- Hata, J.; Grant, I.P. The representation of higher-order relativistic corrections in the MCDF-EAL method. J. Phys. B At. Mol. Phys. 1983, 16, 3713. [Google Scholar] [CrossRef]
- Grant, I.P. Relativistic atomic structure theory: Some recent work. Int. J. Quantum Chem. 1984, 25, 23–46. [Google Scholar] [CrossRef]
- Polasik, M. Theoretical multiconfiguration Dirac-Fock method study on the X-ray spectra of multiply ionized heavy atoms: The structure of the KαLn lines. Phys. Rev. A 1989, 39, 616–627. [Google Scholar] [CrossRef]
- Polasik, M. Theoretical simulation of the X-ray spectra of multiply ionized heavy atoms: The KαLn spectra of molybdenum. Phys. Rev. A 1989, 39, 5092–5097. [Google Scholar] [CrossRef]
- Polasik, M. Theoretical multiconfiguration Dirac-Fock method study on the X-ray spectra of multiply ionized heavy atoms: The structure of the KαL0Mr lines. Phys. Rev. A 1989, 40, 4361–4368. [Google Scholar] [CrossRef]
- Polasik, M. Theoretical multiconfiguration Dirac-Fock method study on the X-ray spectra of multiply ionized heavy atoms: The structure of the KαL1Mr satellite lines. Phys. Rev. A 1990, 41, 3689–3697. [Google Scholar] [CrossRef]
- Polasik, M. Systematic multiconfiguration-Dirac-Fock study of the X-ray spectra accompanying the ionization in collision processes: The structure of the Kβ1,3L0Mr lines. Phys. Rev. A 1995, 52, 227–235. [Google Scholar] [CrossRef]
- Polasik, M. Influence of changes in the valence electronic configuration on the Kβ-to-Kα X-ray intensity ratios of the 3d transition metals. Phys. Rev. A 1998, 58, 1840–1845. [Google Scholar] [CrossRef]
- Polasik, M.; Słabkowska, K.; Rzadkiewicz, J.; Kozioł, K.; Starosta, J.; Wiatrowska-Kozioł, E.; Dousse, J.C.; Hoszowska, J. Khα1,2 X-ray Hypersatellite Line Broadening as a Signature of K-Shell Double Photoionization Followed by Outer-Shell Ionization and Excitation. Phys. Rev. Lett. 2011, 107, 073001. [Google Scholar] [CrossRef]
- Desclaux, J. A multiconfiguration relativistic DIRAC-FOCK program. Comput. Phys. Commun. 1975, 9, 31–45. [Google Scholar] [CrossRef]
- Indelicato, P. Projection operators in multiconfiguration Dirac-Fock calculations: Application to the ground state of heliumlike ions. Phys. Rev. A 1995, 51, 1132–1145. [Google Scholar] [CrossRef]
- Löwdin, P.O. Quantum Theory of Many-Particle Systems. I. Physical Interpretations by Means of Density Matrices, Natural Spin-Orbitals, and Convergence Problems in the Method of Configurational Interaction. Phys. Rev. 1955, 97, 1474–1489. [Google Scholar] [CrossRef]
- Marques, J.P.; Parente, F.; Indelicato, P. Relativistic MCDF calculation of Kβ/Kα intensity ratios. J. Phys. B At. Mol. Opt. Phys. 2001, 34, 3487. [Google Scholar] [CrossRef]

| Z | Symb. | Experiment | Theory | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| This Work | Ref. [25] | Ref. [26] | Ref. [1] | Ref. [9] | This Work | Ref. [3] | ||||||
| GRASP | MCDFGME | |||||||||||
| 3pn | 3dn−24s2 | 3dn−14s1 | No Sat | With Sat | ||||||||
| 12 | Mg | |||||||||||
| 13 | Al | 2.2 | ||||||||||
| 14 | Si | 3 | ||||||||||
| 20 | Ca | |||||||||||
| 21 | Sc | |||||||||||
| 22 | Ti | |||||||||||
| 23 | V | |||||||||||
| 24 | Cr | |||||||||||
| 25 | Mn | |||||||||||
| 26 | Fe | |||||||||||
| 27 | Co | |||||||||||
| 28 | Ni | |||||||||||
| 29 | Cu | |||||||||||
| 2 Asymetric Lorentzian Fitting | |||||
| Energy | |||||
| FWHM | |||||
| AI | |||||
| 4 symetric Lorentzian fitting | |||||
| Energy | |||||
| FWHM | |||||
| CF | |||||
| Sample | Form | Time (s) | |
|---|---|---|---|
| Cr | plate | 5 | 90 |
| CrO | powder | 5 | 150 |
| FeCrO | powder | 5 | 150 |
| CoCrO | powder | 5 | 150 |
| KCrO | powder | 6 | 260 |
| KCrO | powder | 6 | 150 |
| Sample | Symmetry of Cr-O Part | Peak Pos. | Separation | FWHM | Asymm. Index | Int. Ratio | |||
|---|---|---|---|---|---|---|---|---|---|
| Cr metal | |||||||||
| KCrO | Td-like | ||||||||
| KCrO | Td-like | ||||||||
| CrO | Oh-like | ||||||||
| FeCrO | Oh-like | ||||||||
| CoCrO | Oh-like | ||||||||
| Sample | Peak Pos. | Separation | FWHM | Asymm. Index | ||||
|---|---|---|---|---|---|---|---|---|
| Cr metal | This work | |||||||
| Ref. [29] | ||||||||
| KCrO | This work | |||||||
| Ref. [29] | ||||||||
| CrO | This work | |||||||
| Ref. [29] | ||||||||
| Sample | Form | Crystal | Time (s) | Rh (kV, mA) |
|---|---|---|---|---|
| Mg | plate | ADP(101) | 30 | 40, 60 |
| Al | plate | ADP(101) | 30 | 40, 60 |
| Si | wafer | ADP(101) | 50 | 40, 60 |
| Ca | CaF powder | Si(220) | 100 | 40, 60 |
| Sc | foil | Si(220) | 6 | 40, 60 |
| Ti | plate | Si(220) | 6 | 40, 60 |
| V | foil | Si(220) | 8 | 40, 60 |
| Cr | plate | Si(220) | 5 | 40, 60 |
| Mn | plate | Si(220) | 5 | 40, 60 |
| Fe | plate | Si(220) | 8 | 40, 60 |
| Co | foil | Si(220) | 5 | 40, 60 |
| Ni | foil | Si(220) | 4 | 40, 60 |
| Cu | plate | Si(220) | 4 | 40, 60 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ito, Y.; Tochio, T.; Yamashita, M.; Fukushima, S.; Shoji, T.; Słabkowska, K.; Syrocki, Ł.; Polasik, M.; Gomilsek, J.P.; Marques, J.P.; et al. Intensity Ratio of Kβ/Kα in Selected Elements from Mg to Cu, and the Chemical Effects of Cr Kα1,2 Diagram Lines and Cr Kβ/Kα Intensity Ratio in Cr Compounds. Int. J. Mol. Sci. 2023, 24, 5570. https://doi.org/10.3390/ijms24065570
Ito Y, Tochio T, Yamashita M, Fukushima S, Shoji T, Słabkowska K, Syrocki Ł, Polasik M, Gomilsek JP, Marques JP, et al. Intensity Ratio of Kβ/Kα in Selected Elements from Mg to Cu, and the Chemical Effects of Cr Kα1,2 Diagram Lines and Cr Kβ/Kα Intensity Ratio in Cr Compounds. International Journal of Molecular Sciences. 2023; 24(6):5570. https://doi.org/10.3390/ijms24065570
Chicago/Turabian StyleIto, Yoshiaki, Tatsunori Tochio, Michiru Yamashita, Sei Fukushima, Takashi Shoji, Katarzyna Słabkowska, Łukasz Syrocki, Marek Polasik, Jana Padežnik Gomilsek, José Pires Marques, and et al. 2023. "Intensity Ratio of Kβ/Kα in Selected Elements from Mg to Cu, and the Chemical Effects of Cr Kα1,2 Diagram Lines and Cr Kβ/Kα Intensity Ratio in Cr Compounds" International Journal of Molecular Sciences 24, no. 6: 5570. https://doi.org/10.3390/ijms24065570
APA StyleIto, Y., Tochio, T., Yamashita, M., Fukushima, S., Shoji, T., Słabkowska, K., Syrocki, Ł., Polasik, M., Gomilsek, J. P., Marques, J. P., Sampaio, J. M., Guerra, M., Machado, J., Santos, J. P., Hamidani, A., Kahoul, A., Indelicato, P., & Parente, F. (2023). Intensity Ratio of Kβ/Kα in Selected Elements from Mg to Cu, and the Chemical Effects of Cr Kα1,2 Diagram Lines and Cr Kβ/Kα Intensity Ratio in Cr Compounds. International Journal of Molecular Sciences, 24(6), 5570. https://doi.org/10.3390/ijms24065570










