The Regulatory Roles of Cerebellar Glycosphingolipid Microdomains/Lipid Rafts
Abstract
1. Introduction
2. Lipid Rafts
3. GPI-Anchored Proteins
4. Single-Molecule Imaging
5. Gangliosides
6. Ganglioside GD3-Binding Proteins
6.1. Heterotrimeric G Protein Goα
6.2. Src-Family Kinase Lyn
6.3. GPI-Anchored Neuronal Cell Adhesion Molecule TAG-1
6.4. Csk-Binding Protein Cbp
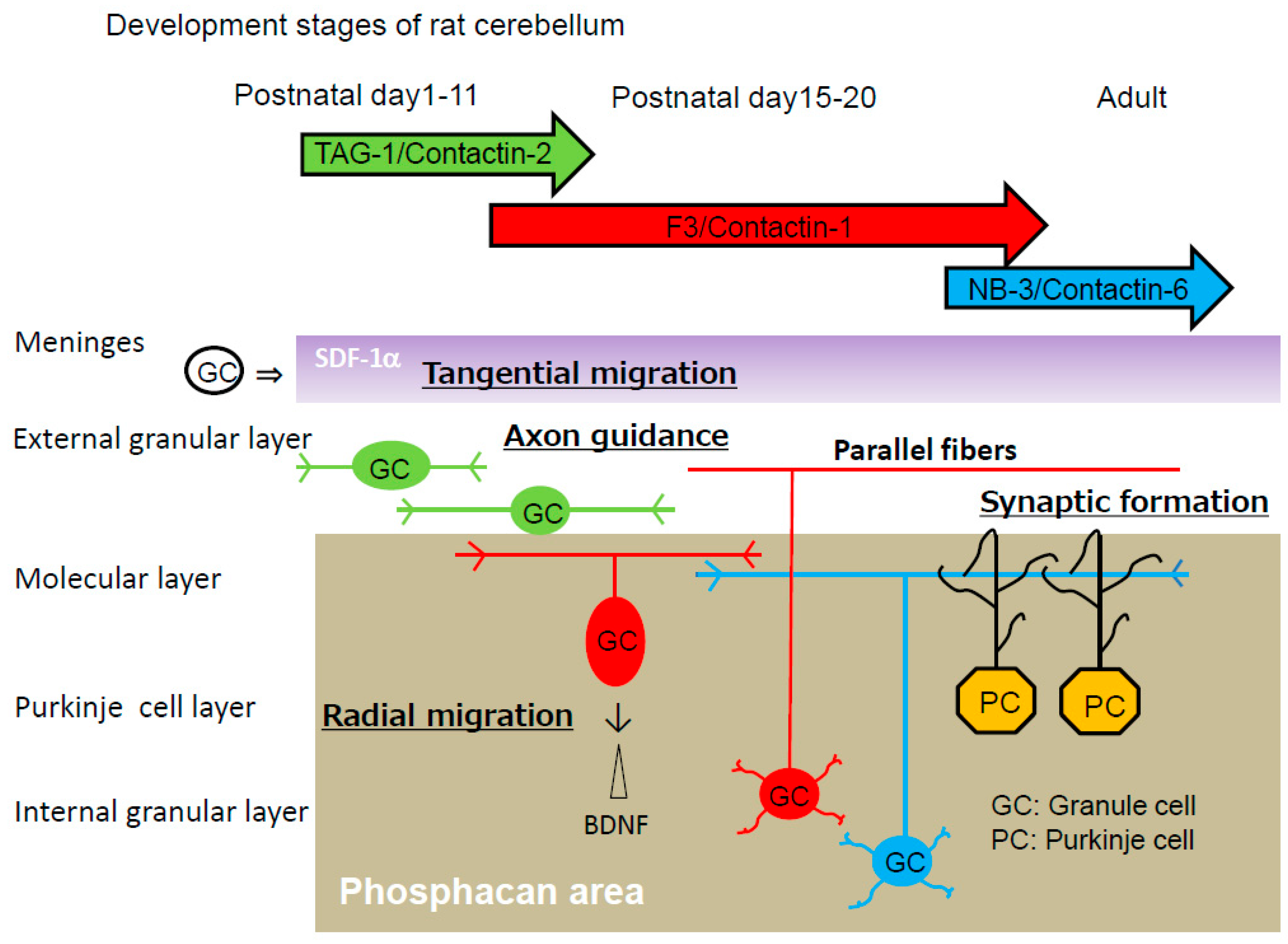
7. Cerebellar Development
8. Contactin Family Members in Developing Granule Cells
9. Cerebellar Raft-Binding Proteins
9.1. Cell Adhesion Molecule L1
9.2. Heterotrimeric G Protein Gsα
9.3. L-Type Voltage-Dependent Calcium Channels
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BDNF | brain-derived neurotrophic factor |
| CaMK-II | calmodulin-dependent protein kinase II |
| Cbp | Csk (C-terminal Src kinase)-binding protein |
| CGC | cerebellar granule cells |
| DKO | double knockout |
| DRM | detergent-resistant membrane |
| EGL | external granular layer |
| ERK | extracellular signal-regulated kinase |
| GC | granule cells |
| Goα | heterotrimeric G protein Go α-subunit |
| GPI | glycosylphosphatidylinositol |
| Gsα | heterotrimeric G protein Gs α-subunit |
| HIE | hypoxic-ischemic encephalopathy |
| IGL | internal granular layer |
| KO | knockout |
| L-VDCC | L-type voltage-dependent calcium channels |
| ML | molecular layer |
| PCL | Purkinje cell layer |
| PGAP | post GPI attachment to proteins |
| PIG | phosphatidyl inositol Glycan |
| PKA | protein kinase A |
| PKC | protein kinase C |
| SDF-1α | stromal-cell derived factor-1 |
| TAG-1 | transient axonal glycoprotein-1 |
References
- Yamakawa, T.; Nagai, Y. Glycolipids at the cell surface and their biological functions. Trends Biochem. Sci. 1978, 3, 128–131. [Google Scholar] [CrossRef]
- Hakomori, S. Glycosphingolipids in cellular interaction, differentiation, and oncogenesis. Annu. Rev. Biochem. 1981, 50, 733–764. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.A.; Rose, J.K. Sorting of GPI-anchored proteins to glycolipid-enriched membrane subdomains during transport to the apical cell surface. Cell 1992, 68, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Gerl, M.J. Revitalizing membrane rafts: New tools and insights. Nat. Rev. Mol. Cell Biol. 2010, 11, 688–699. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.G.N.; Kusumi, A. Refinement of Singer-Nicolson fluid-mosaic model by microscopy imaging: Lipid rafts and actin-induced membrane compartmentalization. Biochim. Biophys. Acta Biomembr. 2023, 1865, 184093. [Google Scholar] [CrossRef]
- Kasahara, K.; Sanai, Y. Functional roles of glycosphingolipids in signal transduction via lipid rafts. Glycoconj. J. 2000, 17, 153–162. [Google Scholar] [CrossRef]
- Kinoshita, T. Biosynthesis and biology of mammalian GPI-anchored proteins. Open Biol. 2020, 10, 190290. [Google Scholar] [CrossRef]
- Maeda, Y.; Tashima, Y.; Houjou, T.; Fujita, M.; Yoko-o, T.; Jigami, Y.; Taguchi, R.; Kinoshita, T. Fatty acid remodeling of GPI-anchored proteins is required for their raft association. Mol. Biol. Cell 2007, 18, 1497–1506. [Google Scholar] [CrossRef]
- Lukacs, M.; Blizzard, L.E.; Stottmann, R.W. CNS glycosylphosphatidylinositol deficiency results in delayed white matter development, ataxia and premature death in a novel mouse model. Hum. Mol. Genet. 2020, 29, 1205–1217. [Google Scholar] [CrossRef]
- Nguyen, T.T.M.; Murakami, Y.; Mobilio, S.; Niceta, M.; Zampino, G.; Philippe, C.; Moutton, S.; Zaki, M.S.; James, K.N.; Musaev, D.; et al. Bi-allelic Variants in the GPI Transamidase Subunit PIGK Cause a Neurodevelopmental Syndrome with Hypotonia, Cerebellar Atrophy, and Epilepsy. Am. J. Hum. Genet. 2020, 106, 484–495. [Google Scholar] [CrossRef]
- Zanni, G.; D’Abrusco, F.; Nicita, F.; Cascioli, S.; Tosi, M.; Corrente, F.; Serpieri, V.; Ciccone, R.; Motta, M.; Vasco, G.; et al. PIGQ-Related Glycophosphatidylinositol Deficiency Associated with Nonprogressive Congenital Ataxia. Cerebellum 2022, 21, 525–530. [Google Scholar] [CrossRef]
- Hirata, T.; Kobayashi, A.; Furuse, T.; Yamada, I.; Tamura, M.; Tomita, H.; Tokoro, Y.; Ninomiya, A.; Fujihara, Y.; Ikawa, M.; et al. Loss of the N-acetylgalactosamine side chain of the GPI-anchor impairs bone formation and brain functions and accelerates the prion disease pathology. J. Biol. Chem. 2022, 298, 101720. [Google Scholar] [CrossRef]
- Suzuki, K.G.; Fujiwara, T.K.; Sanematsu, F.; Iino, R.; Edidin, M.; Kusumi, A. GPI-anchored receptor clusters transiently recruit Lyn and G alpha for temporary cluster immobilization and Lyn activation: Single-molecule tracking study 1. J. Cell Biol. 2007, 177, 717–730. [Google Scholar] [CrossRef]
- Komura, N.; Suzuki, K.G.; Ando, H.; Konishi, M.; Koikeda, M.; Imamura, A.; Chadda, R.; Fujiwara, T.K.; Tsuboi, H.; Sheng, R.; et al. Raft-based interactions of gangliosides with a GPI-anchored receptor. Nat. Chem. Biol. 2016, 12, 402–410. [Google Scholar] [CrossRef]
- Ohmi, Y.; Tajima, O.; Ohkawa, Y.; Mori, A.; Sugiura, Y.; Furukawa, K. Gangliosides play pivotal roles in the regulation of complement systems and in the maintenance of integrity in nerve tissues. Proc. Natl. Acad. Sci. USA 2009, 106, 22405–22410. [Google Scholar] [CrossRef]
- Furukawa, K.; Ohmi, Y.; Yesmin, F.; Tajima, O.; Kondo, Y.; Zhang, P.; Hashimoto, N.; Ohkawa, Y.; Bhuiyan, R.H. Novel Molecular Mechanisms of Gangliosides in the Nervous System Elucidated by Genetic Engineering. Int. J. Mol. Sci. 2020, 21, 1906. [Google Scholar] [CrossRef]
- Kasahara, K.; Guo, L.; Nagai, Y.; Sanai, Y. Enzymatic assay of glycosphingolipid sialyltransferase using reverse-phase thin-layer chromatography. Anal. Biochem. 1994, 218, 224–226. [Google Scholar] [CrossRef]
- Nara, K.; Watanabe, Y.; Maruyama, K.; Kasahara, K.; Nagai, Y.; Sanai, Y. Expression cloning of a CMP-NeuAc:NeuAc alpha 2-3Gal beta 1-4Glc beta 1-1’Cer alpha 2,8-sialyltransferase (GD3 synthase) from human melanoma cells. Proc. Natl. Acad. Sci. USA 1994, 91, 7952–7956. [Google Scholar] [CrossRef]
- Kasahara, K.; Watanabe, Y.; Yamamoto, T.; Sanai, Y. Association of Src family tyrosine kinase Lyn with ganglioside GD3 in rat brain. Possible regulation of Lyn by glycosphingolipid in caveolae-like domains. J. Biol. Chem. 1997, 272, 29947–29953. [Google Scholar] [CrossRef]
- Kasahara, K.; Sanai, Y. Possible roles of glycosphingolipids in lipid rafts. Biophys. Chem. 1999, 82, 121–127. [Google Scholar] [CrossRef]
- Kasahara, K.; Watanabe, K.; Takeuchi, K.; Kaneko, H.; Oohira, A.; Yamamoto, T.; Sanai, Y. Involvement of gangliosides in glycosylphosphatidylinositol-anchored neuronal cell adhesion molecule TAG-1 signaling in lipid rafts. J. Biol. Chem. 2000, 275, 34701–34709. [Google Scholar] [CrossRef] [PubMed]
- Kasahara, K.; Watanabe, K.; Kozutsumi, Y.; Oohira, A.; Yamamoto, T.; Sanai, Y. Association of GPI-anchored protein TAG-1 with src-family kinase Lyn in lipid rafts of cerebellar granule cells. Neurochem. Res. 2002, 27, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Yuyama, K.; Sekino-Suzuki, N.; Sanai, Y.; Kasahara, K. Translocation of activated heterotrimeric G protein Galpha(o) to ganglioside-enriched detergent-resistant membrane rafts in developing cerebellum. J. Biol. Chem. 2007, 282, 26392–26400. [Google Scholar] [CrossRef] [PubMed]
- Yuyama, K.; Sekino-Suzuki, N.; Yamamoto, N.; Kasahara, K. Ganglioside GD3 monoclonal antibody-induced paxillin tyrosine phosphorylation and filamentous actin assembly in cerebellar growth cones. J. Neurochem. 2011, 116, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Sekino-Suzuki, N.; Yuyama, K.; Miki, T.; Kaneda, M.; Suzuki, H.; Yamamoto, N.; Yamamoto, T.; Oneyama, C.; Okada, M.; Kasahara, K. Involvement of gangliosides in the process of Cbp/PAG phosphorylation by Lyn in developing cerebellar growth cones. J. Neurochem. 2013, 124, 514–522. [Google Scholar] [CrossRef]
- Miki, T.; Kaneda, M.; Iida, K.; Hasegawa, G.; Murakami, M.; Yamamoto, N.; Asou, H.; Kasahara, K. An anti-sulfatide antibody O4 immunoprecipitates sulfatide rafts including Fyn, Lyn and the G protein α subunit in rat primary immature oligodendrocytes. Glycoconj. J. 2013, 30, 819–823. [Google Scholar] [CrossRef]
- Kasahara, K.; Kaneda, M.; Miki, T.; Iida, K.; Sekino-Suzuki, N.; Kawashima, I.; Suzuki, H.; Shimonaka, M.; Arai, M.; Ohno-Iwashita, Y.; et al. Clot retraction is mediated by factor XIII-dependent fibrin-αIIbβ3-myosin axis in platelet sphingomyelin-rich membrane rafts. Blood 2013, 122, 3340–3348. [Google Scholar] [CrossRef]
- Ohtsuka, H.; Iguchi, T.; Hayashi, M.; Kaneda, M.; Iida, K.; Shimonaka, M.; Hara, T.; Arai, M.; Koike, Y.; Yamamoto, N.; et al. SDF-1α/CXCR4 Signaling in Lipid Rafts Induces Platelet Aggregation via PI3 Kinase-Dependent Akt Phosphorylation. PLoS ONE 2017, 12, e0169609. [Google Scholar] [CrossRef]
- Komatsuya, K.; Kaneko, K.; Kasahara, K. Function of Platelet Glycosphingolipid Microdomains/Lipid Rafts. Int. J. Mol. Sci. 2020, 21, 5539s. [Google Scholar] [CrossRef]
- Komatsuya, K.; Iguchi, T.; Fukuyama, M.; Kawashima, I.; Ogura, K.; Kikuchi, N.; Shimoda, Y.; Takeda, Y.; Shimonaka, M.; Yamamoto, N.; et al. Phosphacan acts as a repulsive cue in murine and rat cerebellar granule cells in a TAG-1/GD3 rafts-dependent manner. J. Neurochem. 2022, 163, 375–390. [Google Scholar] [CrossRef]
- Chédotal, A. Should I stay or should I go? Becoming a granule cell. Trends Neurosci. 2010, 33, 163–172. [Google Scholar] [CrossRef]
- Ma, Q.; Jones, D.; Borghesani, P.R.; Segal, R.A.; Nagasawa, T.; Kishimoto, T.; Bronson, R.T.; Springer, T.A. Impaired B-lymphopoiesis, myelopoiesis, and derailed cerebellar neuron migration in CXCR4- and SDF-1-deficient mice. Proc. Natl. Acad. Sci. USA 1998, 95, 9448–9453. [Google Scholar] [CrossRef]
- Santiago, M.F.; Scemes, E. Neuroblast migration and P2Y(1) receptor mediated calcium signalling depend on 9-O-acetyl GD3 ganglioside. ASN Neuro 2012, 4, 357–369. [Google Scholar] [CrossRef]
- Galas, L.; Bénard, M.; Lebon, A.; Komuro, Y.; Schapman, D.; Vaudry, H.; Vaudry, D.; Komuro, H. Postnatal Migration of Cerebellar Interneurons. Brain Sci. 2017, 7, 62. [Google Scholar] [CrossRef]
- Zhu, Y.; Yu, T.; Zhang, X.C.; Nagasawa, T.; Wu, J.Y.; Rao, Y. Role of the chemokine SDF-1 as the meningeal attractant for embryonic cerebellar neurons. Nat. Neurosci. 2002, 5, 719–720. [Google Scholar] [CrossRef]
- Chen, A.I.; Zang, K.; Masliah, E.; Reichardt, L.F. Glutamatergic axon-derived BDNF controls GABAergic synaptic differentiation in the cerebellum. Sci. Rep. 2016, 6, 20201. [Google Scholar] [CrossRef]
- Borghesani, P.R.; Peyrin, J.M.; Klein, R.; Rubin, J.; Carter, A.R.; Schwartz, P.M.; Luster, A.; Corfas, G.; Segal, R.A. BDNF stimulates migration of cerebellar granule cells. Development 2002, 129, 1435–1442. [Google Scholar] [CrossRef]
- Consalez, G.G.; Goldowitz, D.; Casoni, F.; Hawkes, R. Origins, Development, and Compartmentation of the Granule Cells of the Cerebellum. Front. Neural Circuits 2020, 14, 611841. [Google Scholar] [CrossRef]
- Maeda, N.; Ishii, M.; Nishimura, K.; Kamimura, K. Functions of chondroitin sulfate and heparan sulfate in the developing brain. Neurochem. Res. 2011, 36, 1228–1240. [Google Scholar] [CrossRef]
- Maeda, N. Proteoglycans and neuronal migration in the cerebral cortex during development and disease. Front. Neurosci. 2015, 9, 98. [Google Scholar] [CrossRef]
- Milev, P.; Maurel, P.; Häring, M.; Margolis, R.K.; Margolis, R.U. TAG-1/axonin-1 is a high-affinity ligand of neurocan, phosphacan/protein-tyrosine phosphatase-zeta/beta, and N-CAM. J. Biol. Chem. 1996, 271, 15716–15723. [Google Scholar] [CrossRef] [PubMed]
- Stoeckli, E.T. Neural circuit formation in the cerebellum is controlled by cell adhesion molecules of the Contactin family. Cell Adhes. Migr. 2010, 4, 523–526. [Google Scholar] [CrossRef]
- Baeriswyl, T.; Stoeckli, E.T. Axonin-1/TAG-1 is required for pathfinding of granule cell axons in the developing cerebellum. Neural Dev. 2008, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Xenaki, D.; Martin, I.B.; Yoshida, L.; Ohyama, K.; Gennarini, G.; Grumet, M.; Sakurai, T.; Furley, A.J. F3/contactin and TAG1 play antagonistic roles in the regulation of sonic hedgehog-induced cerebellar granule neuron progenitor proliferation. Development 2011, 138, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Faivre-Sarrailh, C.; Falk, J.; Pollerberg, E.; Schachner, M.; Rougon, G. NrCAM, cerebellar granule cell receptor for the neuronal adhesion molecule F3, displays an actin-dependent mobility in growth cones. J. Cell Sci. 1999, 112 Pt 18, 3015–3027. [Google Scholar] [CrossRef] [PubMed]
- Olive, S.; Dubois, C.; Schachner, M.; Rougon, G. The F3 neuronal glycosylphosphatidylinositol-linked molecule is localized to glycolipid-enriched membrane subdomains and interacts with L1 and fyn kinase in cerebellum. J. Neurochem. 1995, 65, 2307–2317. [Google Scholar] [CrossRef]
- Berglund, E.O.; Murai, K.K.; Fredette, B.; Sekerková, G.; Marturano, B.; Weber, L.; Mugnaini, E.; Ranscht, B. Ataxia and abnormal cerebellar microorganization in mice with ablated contactin gene expression. Neuron 1999, 24, 739–750. [Google Scholar] [CrossRef]
- Takeuchi, M.; Inoue, C.; Goshima, A.; Nagao, Y.; Shimizu, K.; Miyamoto, H.; Shimizu, T.; Hashimoto, H.; Yonemura, S.; Kawahara, A.; et al. Medaka and zebrafish contactin1 mutants as a model for understanding neural circuits for motor coordination. Genes Cells 2017, 22, 723–741. [Google Scholar] [CrossRef]
- Sakurai, K.; Toyoshima, M.; Ueda, H.; Matsubara, K.; Takeda, Y.; Karagogeos, D.; Shimoda, Y.; Watanabe, K. Contribution of the neural cell recognition molecule NB-3 to synapse formation between parallel fibers and Purkinje cells in mouse. Dev. Neurobiol. 2009, 69, 811–824. [Google Scholar] [CrossRef]
- Brügger, B.; Graham, C.; Leibrecht, I.; Mombelli, E.; Jen, A.; Wieland, F.; Morris, R. The membrane domains occupied by glycosylphosphatidylinositol-anchored prion protein and Thy-1 differ in lipid composition. J. Biol. Chem. 2004, 279, 7530–7536. [Google Scholar] [CrossRef]
- Poejo, J.; Salazar, J.; Mata, A.M.; Gutierrez-Merino, C. Binding of Amyloid β(1-42)-Calmodulin Complexes to Plasma Membrane Lipid Rafts in Cerebellar Granule Neurons Alters Resting Cytosolic Calcium Homeostasis. Int. J. Mol. Sci. 2021, 22, 1984. [Google Scholar] [CrossRef]
- Davies, A.; Douglas, L.; Hendrich, J.; Wratten, J.; Tran Van Minh, A.; Foucault, I.; Koch, D.; Pratt, W.S.; Saibil, H.R.; Dolphin, A.C. The calcium channel alpha2delta-2 subunit partitions with CaV2.1 into lipid rafts in cerebellum: Implications for localization and function. J. Neurosci. 2006, 26, 8748–8757. [Google Scholar] [CrossRef]
- Fortalezas, S.; Marques-da-Silva, D.; Gutierrez-Merino, C. Methyl-β-Cyclodextrin Impairs the Phosphorylation of the β₂ Subunit of L-Type Calcium Channels and Cytosolic Calcium Homeostasis in Mature Cerebellar Granule Neurons. Int. J. Mol. Sci. 2018, 19, 3667. [Google Scholar] [CrossRef]
- Samhan-Arias, A.K.; Marques-da-Silva, D.; Yanamala, N.; Gutierrez-Merino, C. Stimulation and clustering of cytochrome b5 reductase in caveolin-rich lipid microdomains is an early event in oxidative stress-mediated apoptosis of cerebellar granule neurons. J. Proteom. 2012, 75, 2934–2949. [Google Scholar] [CrossRef]
- Amadio, S.; Vacca, F.; Martorana, A.; Sancesario, G.; Volonté, C. P2Y1 receptor switches to neurons from glia in juvenile versus neonatal rat cerebellar cortex. BMC Dev. Biol. 2007, 7, 77. [Google Scholar] [CrossRef]
- Dalskov, S.M.; Immerdal, L.; Niels-Christiansen, L.L.; Hansen, G.H.; Schousboe, A.; Danielsen, E.M. Lipid raft localization of GABA A receptor and Na+, K+-ATPase in discrete microdomain clusters in rat cerebellar granule cells. Neurochem. Int. 2005, 46, 489–499. [Google Scholar] [CrossRef]
- Becher, A.; White, J.H.; McIlhinney, R.A. The gamma-aminobutyric acid receptor B, but not the metabotropic glutamate receptor type-1, associates with lipid rafts in the rat cerebellum. J. Neurochem. 2001, 79, 787–795. [Google Scholar] [CrossRef]
- Botto, L.; Masserini, M.; Palestini, P. Changes in the composition of detergent-resistant membrane domains of cultured neurons following protein kinase C activation. J. Neurosci. Res. 2007, 85, 443–450. [Google Scholar] [CrossRef]
- Donati, R.J.; Dwivedi, Y.; Roberts, R.C.; Conley, R.R.; Pandey, G.N.; Rasenick, M.M. Postmortem brain tissue of depressed suicides reveals increased Gs alpha localization in lipid raft domains where it is less likely to activate adenylyl cyclase. J. Neurosci. 2008, 28, 3042–3050. [Google Scholar] [CrossRef]
- Sui, Z.; Kovács, A.D.; Maggirwar, S.B. Recruitment of active glycogen synthase kinase-3 into neuronal lipid rafts. Biochem. Biophys. Res. Commun. 2006, 345, 1643–1648. [Google Scholar] [CrossRef]
- Chen, S.; Bawa, D.; Besshoh, S.; Gurd, J.W.; Brown, I.R. Association of heat shock proteins and neuronal membrane components with lipid rafts from the rat brain. J. Neurosci. Res. 2005, 81, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Laudati, E.; Gilder, A.S.; Lam, M.S.; Misasi, R.; Sorice, M.; Gonias, S.L.; Mantuano, E. The activities of LDL Receptor-related Protein-1 (LRP1) compartmentalize into distinct plasma membrane microdomains. Mol. Cell. Neurosci. 2016, 76, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Marques-da-Silva, D.; Gutierrez-Merino, C. Caveolin-rich lipid rafts of the plasma membrane of mature cerebellar granule neurons are microcompartments for calcium/reactive oxygen and nitrogen species cross-talk signaling. Cell Calcium. 2014, 56, 108–123. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Guo, W.; Feng, L. Segregation of Nogo66 receptors into lipid rafts in rat brain and inhibition of Nogo66 signaling by cholesterol depletion. FEBS Lett. 2004, 577, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Marques-da-Silva, D.; Gutierrez-Merino, C. L-type voltage-operated calcium channels, N-methyl-D-aspartate receptors and neuronal nitric-oxide synthase form a calcium/redox nano-transducer within lipid rafts. Biochem. Biophys. Res. Commun. 2012, 420, 257–262. [Google Scholar] [CrossRef]
- Vacca, F.; Amadio, S.; Sancesario, G.; Bernardi, G.; Volonté, C. P2X3 receptor localizes into lipid rafts in neuronal cells. J. Neurosci. Res. 2004, 76, 653–661. [Google Scholar] [CrossRef]
- Sepúlveda, M.R.; Berrocal-Carrillo, M.; Gasset, M.; Mata, A.M. The plasma membrane Ca2+-ATPase isoform 4 is localized in lipid rafts of cerebellum synaptic plasma membranes. J. Biol. Chem. 2006, 281, 447–453. [Google Scholar] [CrossRef]
- Buratta, S.; Felicetti, M.; Mozzi, R. Synthesis of phosphatidylserine by base exchange in Triton-insoluble floating fractions from rat cerebellum. J. Neurochem. 2007, 103, 942–951. [Google Scholar] [CrossRef]
- Palestini, P.; Pitto, M.; Tedeschi, G.; Ferraretto, A.; Parenti, M.; Brunner, J.; Masserini, M. Tubulin anchoring to glycolipid-enriched, detergent-resistant domains of the neuronal plasma membrane. J. Biol. Chem. 2000, 275, 9978–9985. [Google Scholar] [CrossRef]
- Nakai, Y.; Kamiguchi, H. Migration of nerve growth cones requires detergent-resistant membranes in a spatially defined and substrate-dependent manner. J. Cell Biol. 2002, 159, 1097–1108. [Google Scholar] [CrossRef]
- Ignelzi, M.A.; Miller, D.R.; Soriano, P.; Maness, P.F. Impaired neurite outgrowth of src-minus cerebellar neurons on the cell adhesion molecule L1. Neuron 1994, 12, 873–884. [Google Scholar] [CrossRef]
- Waddell, J.; Rickman, N.C.; He, M.; Tang, N.; Bearer, C.F. Neonatal hypoxia ischemia redistributes L1 cell adhesion molecule into rat cerebellar lipid rafts. Pediatr. Res. 2022, 92, 1325–1331. [Google Scholar] [CrossRef]
- Tang, N.; Farah, B.; He, M.; Fox, S.; Malouf, A.; Littner, Y.; Bearer, C.F. Ethanol causes the redistribution of L1 cell adhesion molecule in lipid rafts. J. Neurochem. 2011, 119, 859–867. [Google Scholar] [CrossRef]
- Schappi, J.M.; Rasenick, M.M. Gαs, adenylyl cyclase, and their relationship to the diagnosis and treatment of depression. Front. Pharmacol. 2022, 13, 1012778. [Google Scholar] [CrossRef]
- Dunham, J.S.; Deakin, J.F.; Miyajima, F.; Payton, A.; Toro, C.T. Expression of hippocampal brain-derived neurotrophic factor and its receptors in Stanley consortium brains. J. Psychiatr. Res. 2009, 43, 1175–1184. [Google Scholar] [CrossRef]
- Castrén, E.; Monteggia, L.M. Brain-Derived Neurotrophic Factor Signaling in Depression and Antidepressant Action. Biol. Psychiatry 2021, 90, 128–136. [Google Scholar] [CrossRef]
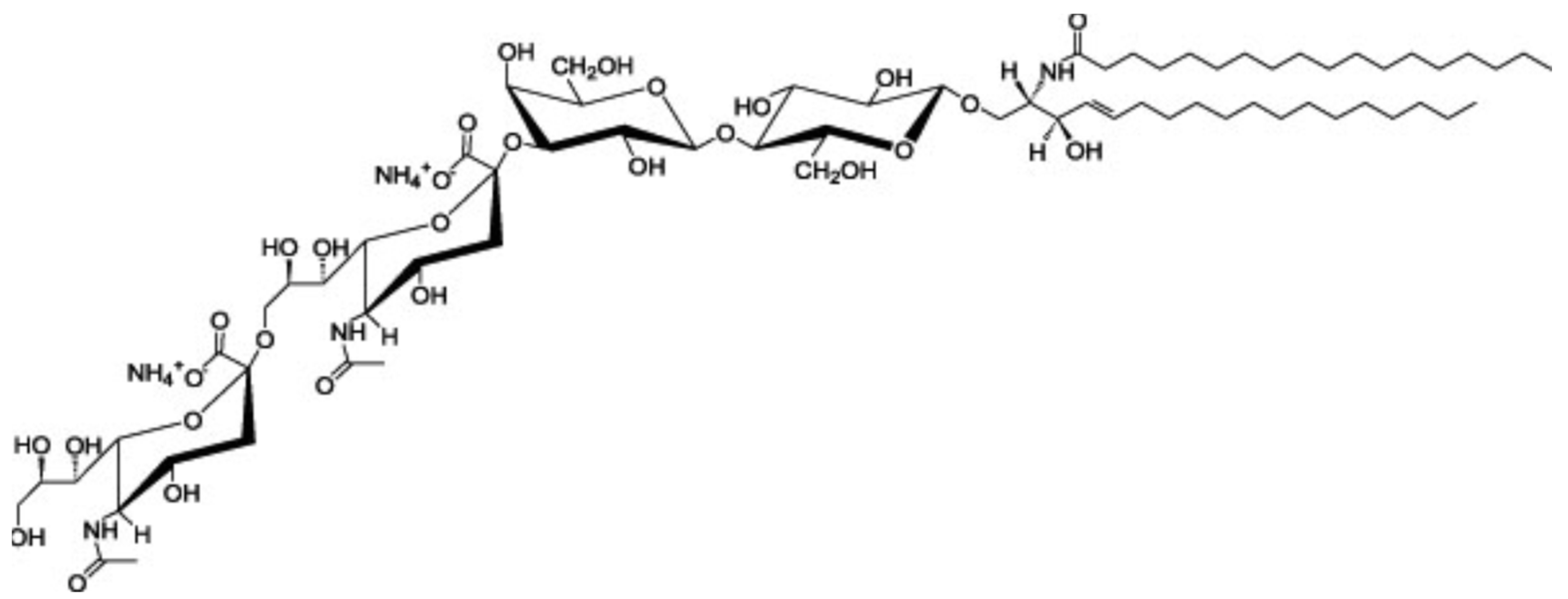


| Molecules | Function | Tissue/Cell | Ref. |
|---|---|---|---|
| Amyloid β(1–42) | Major plaque component in Alzheimer’s disease | CGC | [51] |
| α2δ-2 | Calcium channel | cerebellum | [52] |
| CaV2.1 | Calcium channel alpha1 subunit | cerebellum | [52] |
| Calmodulin | Multifunctional calcium-binding messenger protein | CGC | [51] |
| CaMK-II | Calmodulin-dependent protein kinase II | CGC | [53] |
| Cbp/PAG | Csk-binding protein | CGC | [25] |
| Cyb5R | Cytochrome b5 reductase | CGC | [54] |
| F3/contactin-1 | GPI-anchored adhesion molecule | cerebellum | [46] |
| Flotillin-1 | Scaffolding protein | cerebellum | [52] |
| Flotillin-2 | Scaffolding protein | cerebellum | [55] |
| Fyn | Src-family tyrosine kinase | cerebellum | [46] |
| GABA A receptor | Ligand-gated ion channel | CGC | [56] |
| GABA B receptor | G-protein coupled receptor | cerebellum | [57] |
| Goα | Heterotrimeric G protein | CGC | [23] |
| GAP-43 | Component of the axon and presynaptic terminal | CGC | [58] |
| Gsα | Heterotrimeric G protein | cerebellum | [59] |
| GSK-3 | Glycogen synthase kinase-3 | CGC | [60] |
| Hsc70 | Molecular chaperone | cerebellum | [61] |
| Hsp90 | Molecular chaperone | cerebellum | [61] |
| Hsp60 | Molecular chaperone | cerebellum | [61] |
| Hsp40 | Molecular chaperone | cerebellum | [61] |
| LRP1 | LDL receptor-related protein-1 | CGC | [62] |
| L-VDCC | L-type voltage-dependent calcium channels | CGC | [53] |
| Lyn | Src-family tyrosine kinase | CGC | [19] |
| NCAM 120 | GPI-anchored adhesion molecule | cerebellum | [46] |
| NCX | Sodium–calcium exchangers | CGC | [63] |
| NgR1 | GPI-anchored Nogo-66 receptor | CGC | [64] |
| NMDA receptor | Ionotropic glutamate receptor | CGC | [65] |
| nNOS | Neuronal nitric oxide synthase | CGC | [65] |
| P2X3 receptor | Ionotropic purinergic receptor | CGC | [66] |
| P2Y1 receptor | Metabotropic purinergic receptor | cerebellum | [55] |
| p75NTR | Low-affinity nerve growth factor receptor | CGC | [64] |
| PMCA4 | Ca2+-ATPase calcium pumps | cerebellum | [67] |
| Protein kinase A | cAMP-dependent protein kinase | CGC | [53] |
| Protein kinase C | Ca2+-activated phospholipid-dependent protein kinase | cerebellum | [68] |
| PrPC | Prion | CGC | [58] |
| SBEE | Serine base exchange enzyme | cerebellum | [68] |
| TAG-1/contactin-2 | GPI-anchored adhesion molecule | CGC | [21] |
| Thy-1 | GPI-anchored adhesion molecule | cerebellum | [46] |
| Tubulin | microtubule | CGC | [69] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komatsuya, K.; Kikuchi, N.; Hirabayashi, T.; Kasahara, K. The Regulatory Roles of Cerebellar Glycosphingolipid Microdomains/Lipid Rafts. Int. J. Mol. Sci. 2023, 24, 5566. https://doi.org/10.3390/ijms24065566
Komatsuya K, Kikuchi N, Hirabayashi T, Kasahara K. The Regulatory Roles of Cerebellar Glycosphingolipid Microdomains/Lipid Rafts. International Journal of Molecular Sciences. 2023; 24(6):5566. https://doi.org/10.3390/ijms24065566
Chicago/Turabian StyleKomatsuya, Keisuke, Norihito Kikuchi, Tetsuya Hirabayashi, and Kohji Kasahara. 2023. "The Regulatory Roles of Cerebellar Glycosphingolipid Microdomains/Lipid Rafts" International Journal of Molecular Sciences 24, no. 6: 5566. https://doi.org/10.3390/ijms24065566
APA StyleKomatsuya, K., Kikuchi, N., Hirabayashi, T., & Kasahara, K. (2023). The Regulatory Roles of Cerebellar Glycosphingolipid Microdomains/Lipid Rafts. International Journal of Molecular Sciences, 24(6), 5566. https://doi.org/10.3390/ijms24065566






