Osteoregenerative Potential of 3D-Printed Poly ε-Caprolactone Tissue Scaffolds In Vitro Using Minimally Manipulative Expansion of Primary Human Bone Marrow Stem Cells
Abstract
1. Introduction
1.1. Review of Literature
1.1.1. Human Bone Regeneration
1.1.2. 3D-Microfabrication
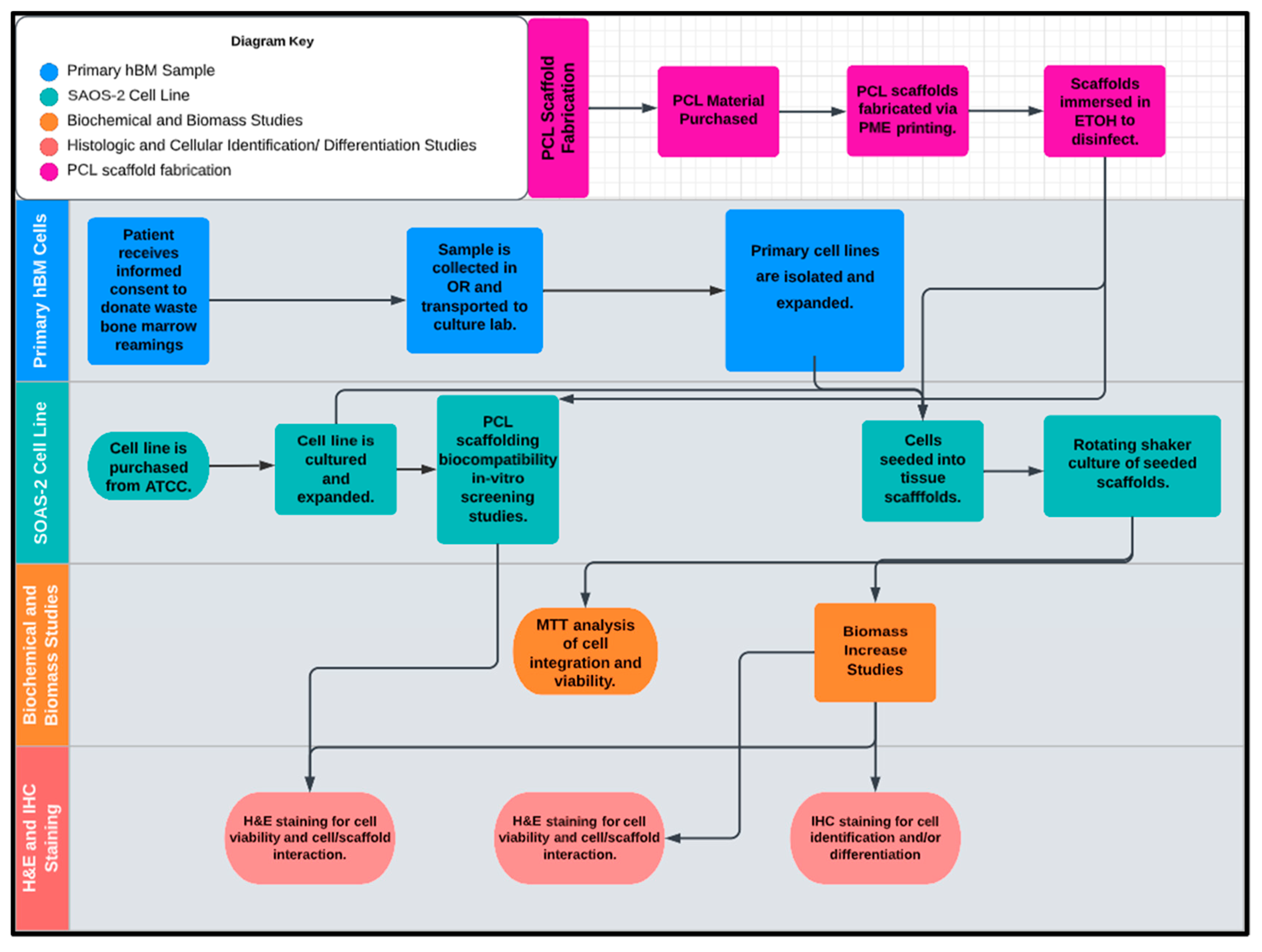
2. Results
2.1. Polycaprolactone (PCL) 3D-Printed Scaffold Fabrication
2.2. Human Bone Marrow (hBM) Sample Collection
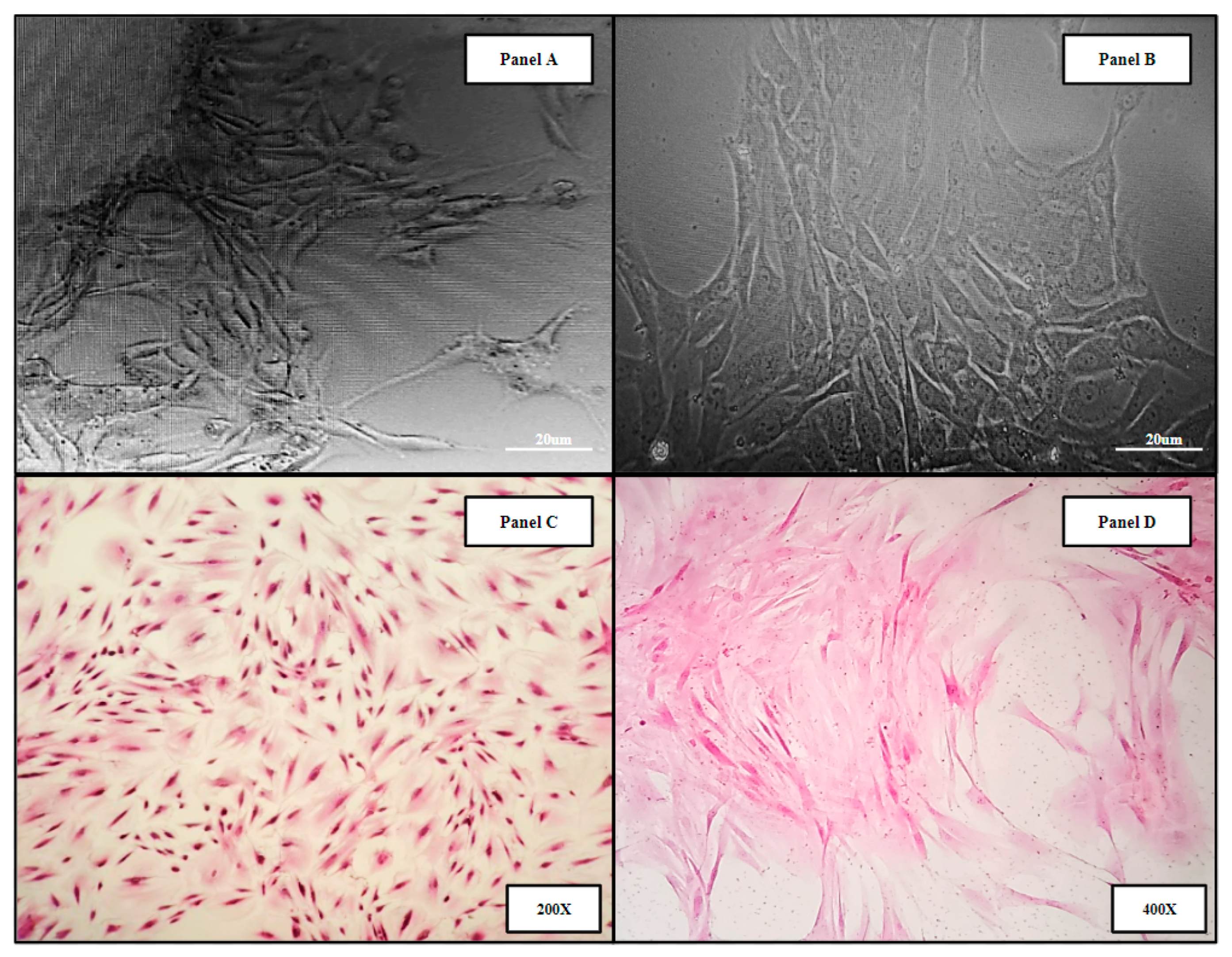
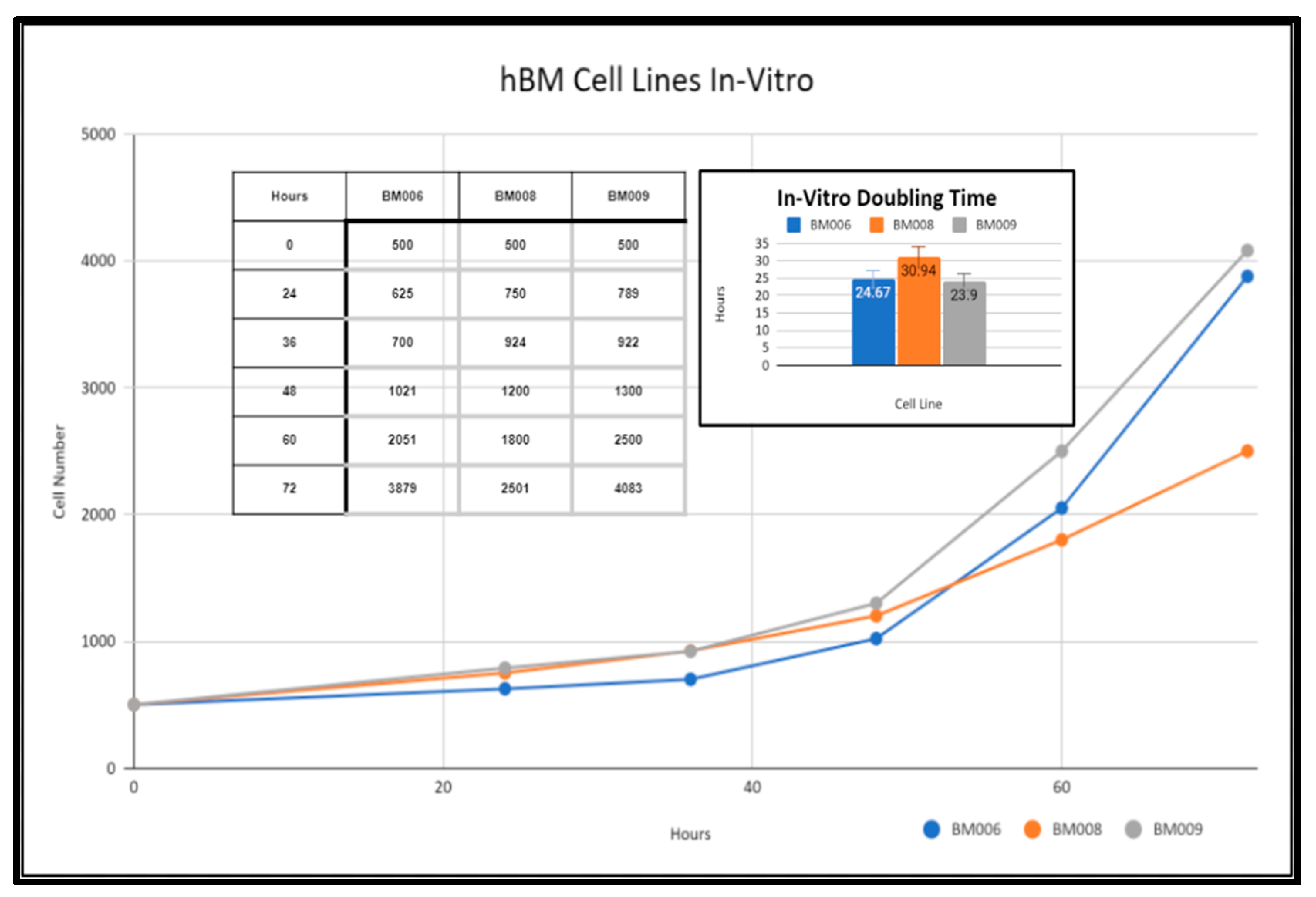
2.3. Primary Whole Bone Marrow Progenitor Cell Line Isolation and In Vitro Expansion
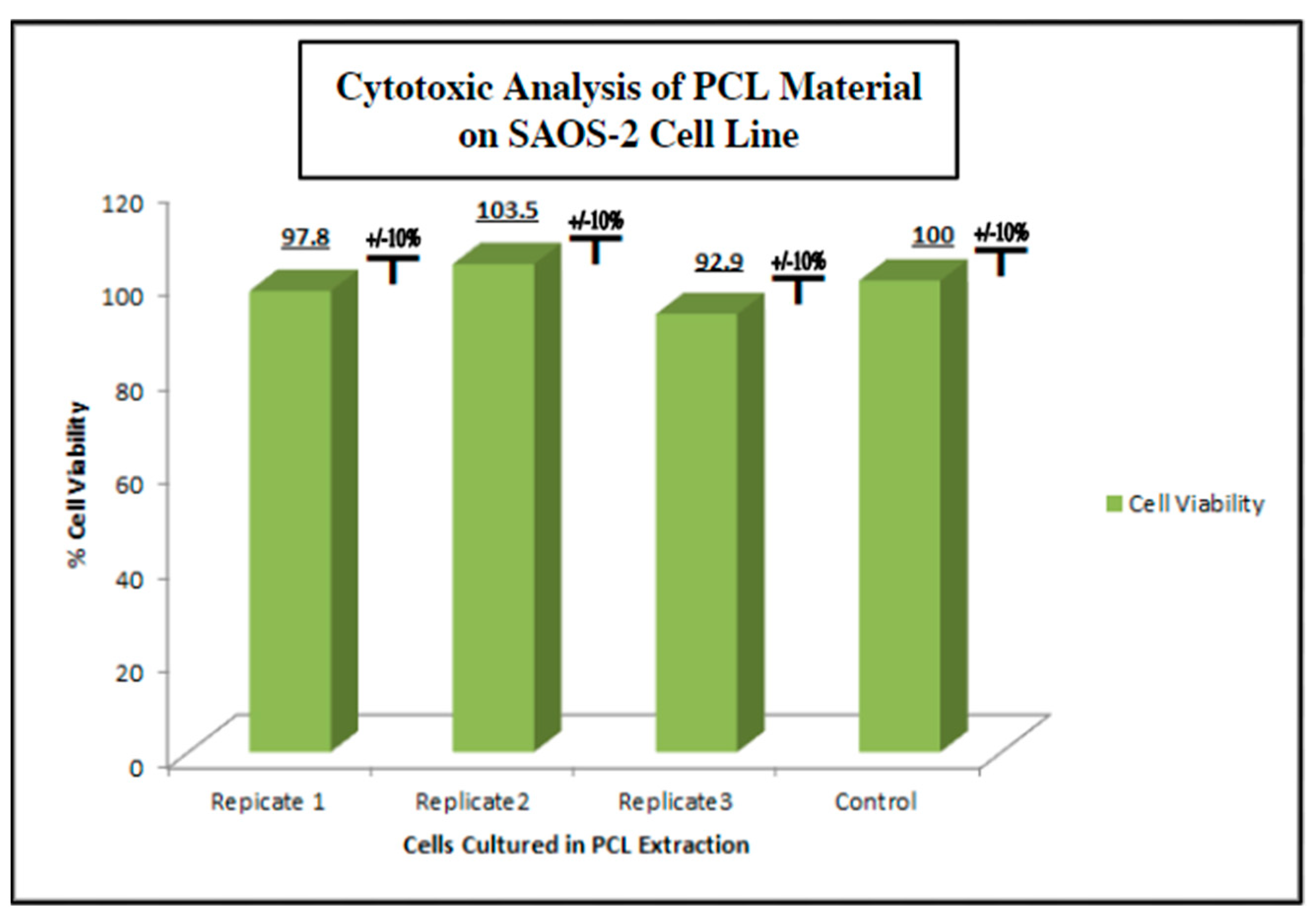
2.4. Screening of Biocompatibility of Fabricated Polycaprolactone (PCL) Scaffolds via Cytotoxic and Cytostatic Analysis of PCL Extract Exposure and Histological Analysis of Established Osteosarcoma Cell Line SAOS-2
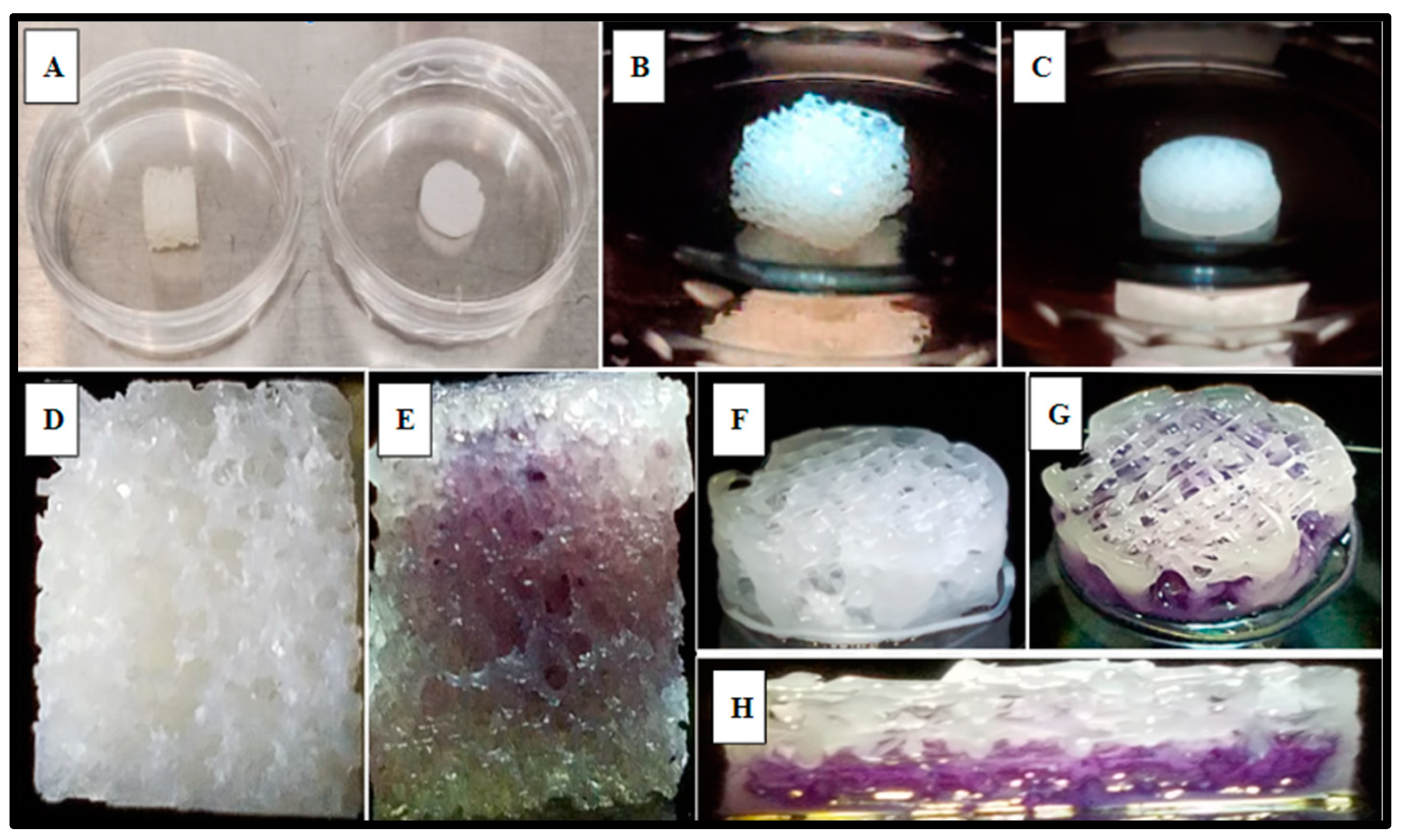
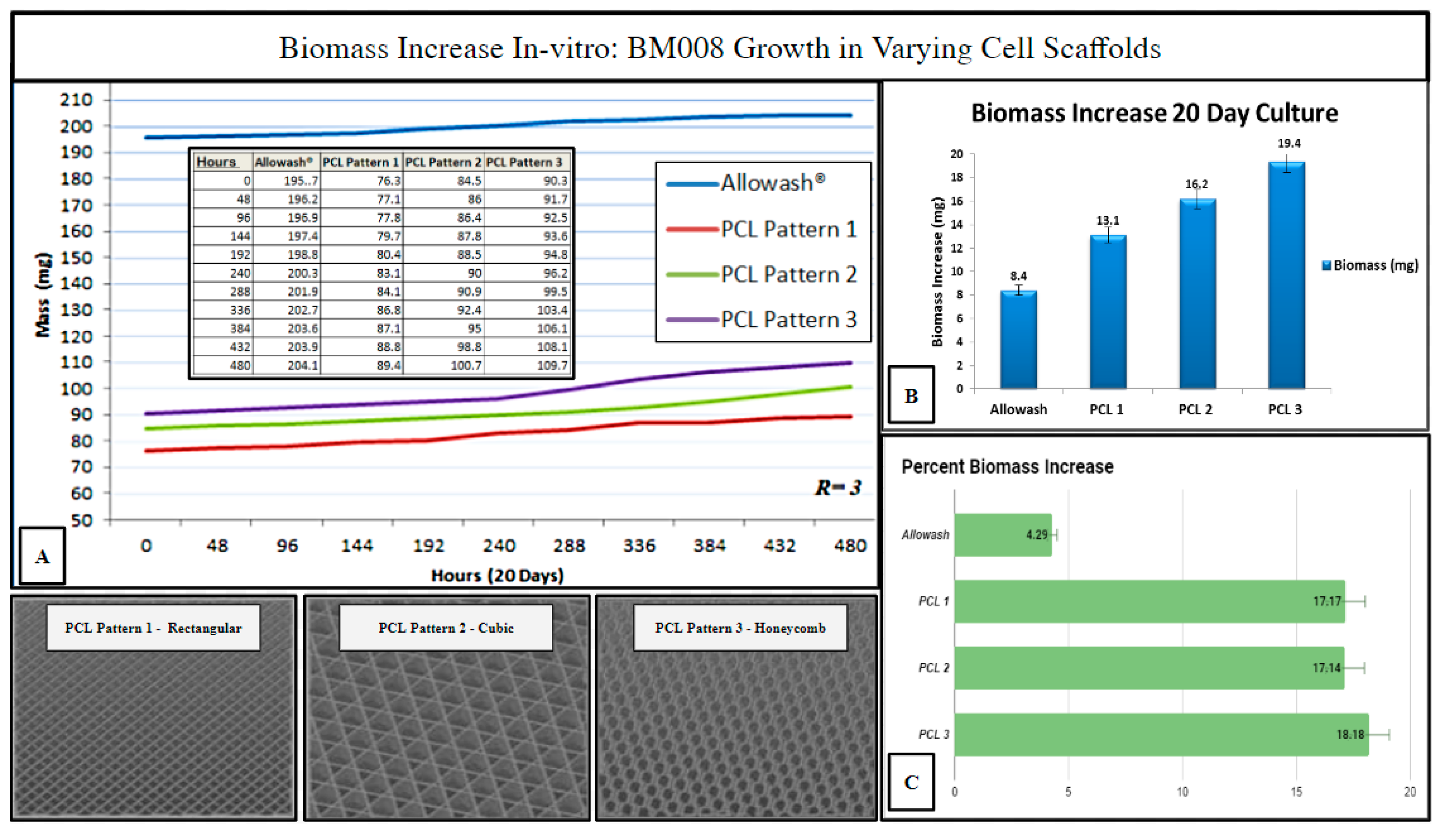
2.5. Investigation of Varying 3D-Printed PCL Scaffold Matrix Patterns on Cell Integration and Resulting Scaffold Biomass Increase In Vitro with Primary Human Bone Marrow Cell Culture as Compared to Standard-of-Care Allograft Cancellous Bone Cubes
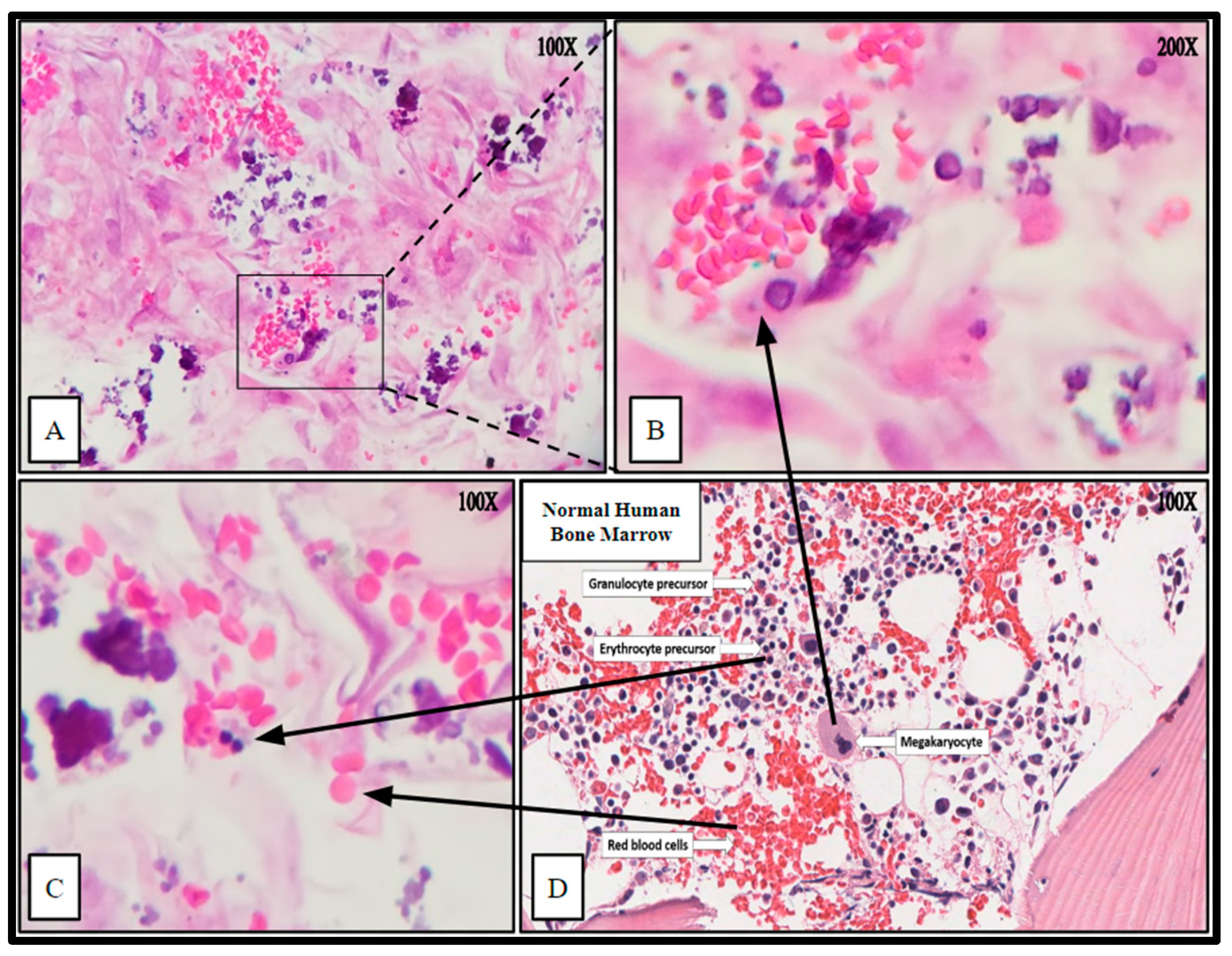
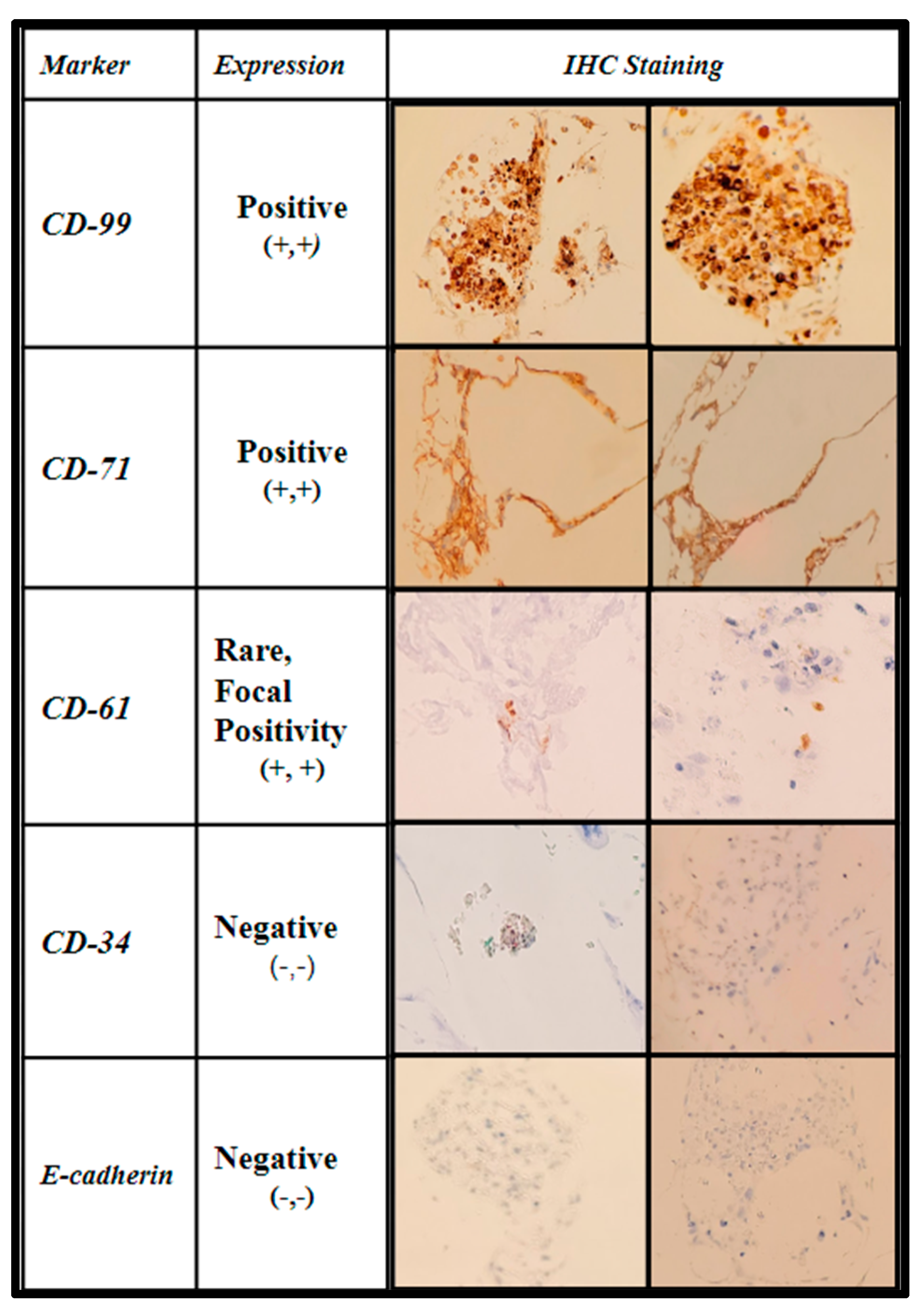
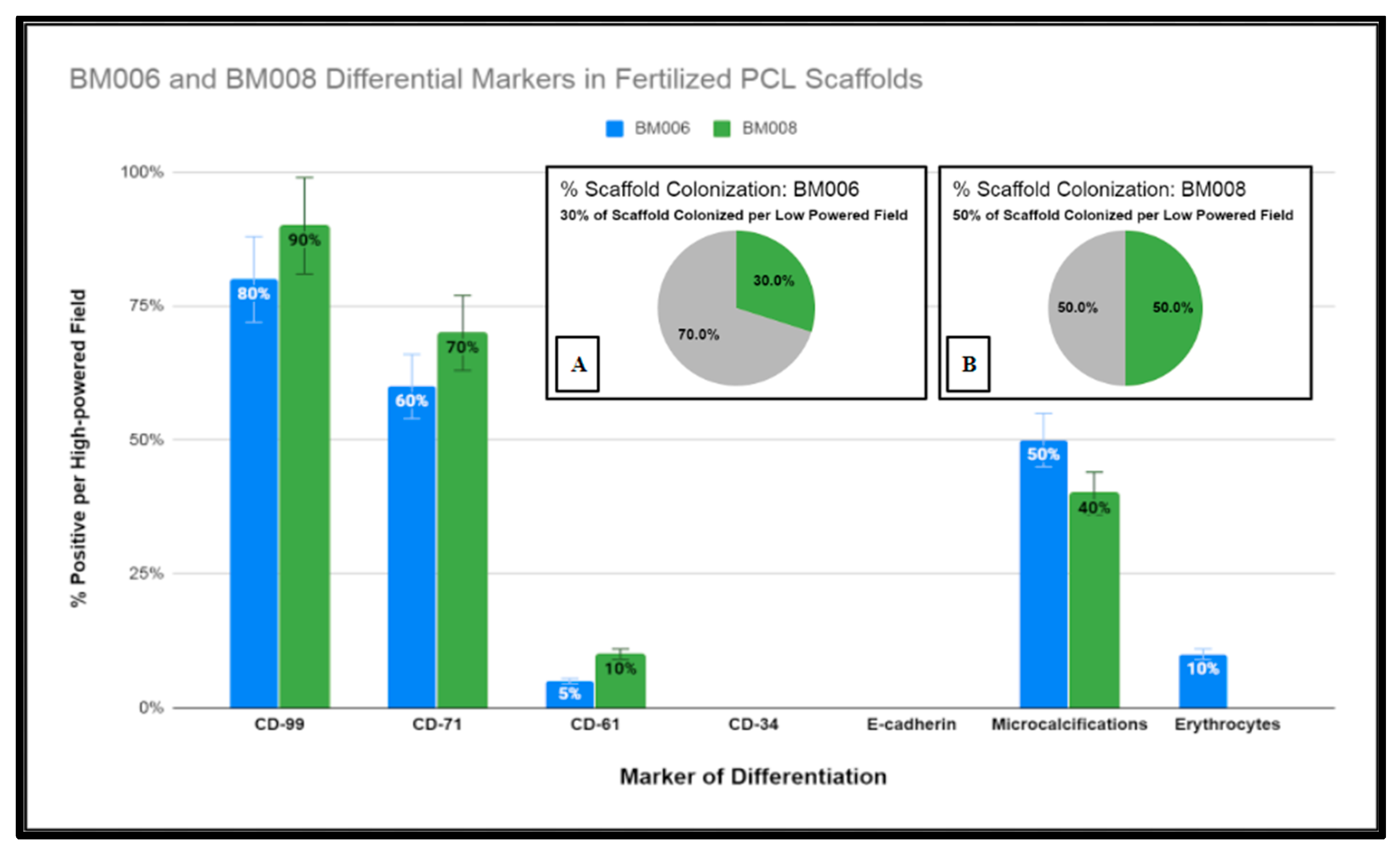
2.6. Histologic Analysis of Human Bone Marrow (hBM) Stem Cell Viability, Integration, and Differentiation in 3D-Printed PCL Honeycomb Scaffold
3. Discussion
4. Materials and Methods
4.1. Human Bone Marrow (hBM) Sample Collection
4.2. Patient Samples of Primary Bone Marrow (hBM) Stem Cell Colonies
4.3. Cell Lines and Growth Medium
4.4. Cytotoxic and Cytostatic Analysis of PCL Extract on Established Osteosarcoma Cell Line SAOS-2 via MTT Analysis
4.5. hBM Stem-Cell Culture and Colony Expansion
4.6. Cell Counting
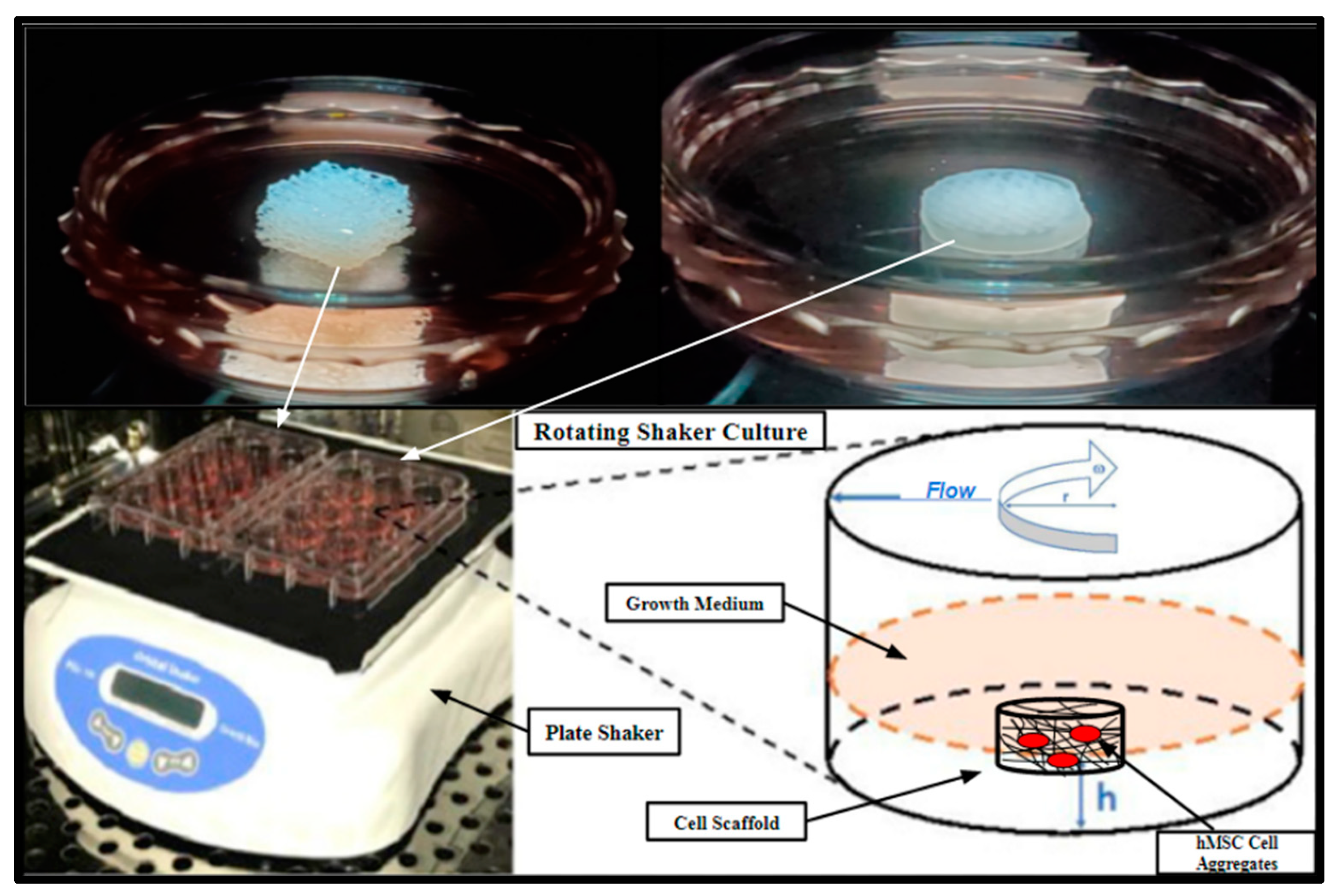
4.7. Scaffold Inoculation and Rotating Shaker Culture
4.8. PCL Scaffold Microfabrication and Specifications
4.9. In Vitro Biomass Increase
4.10. Histologic Processing: H&E and Immunohistochemical Evaluation of Cell Viability, Cell Integration, and Cell Differentiation of Primary (hBMs) in Fertilized Scaffolds
4.11. Sterilization of 3D-Printed PCL Scaffolds and Allowash® Cancellous Cubes
4.12. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MTT | 4, 5-dimethylthiazol-2-yl]-2, 5 diphenyl tetrazolium bromide |
| BVF | Bone Void Fillers |
| Cub | Cubic |
| DBM | Decellularized Bone Material |
| DIW | Direct Ink Writing |
| FFPE | Formalin Fixation and Paraffin Embedding |
| FDM | Fused Deposition Modeling |
| H&E | Hematoxylin & Eosin |
| HC | Honeycomb |
| hBM | Human Bone Marrow |
| IC | Immunohistochemistry |
| ICBG | Iliac Crest Bone Graft |
| LGDW | Laser-Guided Direct Writing |
| MSCs | Mesenchymal Stem Cells |
| PME | Pneumatic Micro-Extrusion |
| PCL | Polycaprolactone |
| RL | Rectilinear |
| 3D | Three-Dimensional |
| SLA | Stereolithography |
References
- Prevention, C. Transplant Safety—Overview—Key Facts. Available online: https://www.cdc.gov/transplantsafety/overview/key-facts.html (accessed on 25 January 2023).
- Albrektsson, T.; Johansson, C. Osteoinduction, osteoconduction and osseointegration. Eur. Spine J. 2001, 10, S96–S101. [Google Scholar] [CrossRef] [PubMed]
- Armiento, A.R.; Hatt, L.P.; Rosenberg, G.S.; Thompson, K.; Stoddart, M.J. Functional Biomaterials for Bone Regeneration: A Lesson in Complex Biology. Adv. Funct. Mater. 2020, 30, 1909874. [Google Scholar] [CrossRef]
- Baldwin, P.; Li, D.J.; Auston, D.A.; Mir, H.S.; Yoon, R.S.; Koval, K.J. Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery. J. Orthop. Trauma 2019, 33, 203–213. [Google Scholar] [CrossRef] [PubMed]
- Migliorini, F.; La Padula, G.; Torsiello, E.; Spiezia, F.; Oliva, F.; Maffulli, N. Strategies for large bone defect reconstruction after trauma, infections or tumour excision: A comprehensive review of the literature. Eur. J. Med. Res. 2021, 26, 118. [Google Scholar] [CrossRef]
- Muscolo, D.L.; Ayerza, M.A.; Aponte-Tinao, L.A. Massive Allograft Use in Orthopedic Oncology. Orthop. Clin. N. Am. 2006, 37, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Reznikov, N.; Bilton, M.; Lari, L.; Stevens, M.M.; Kröger, R. Fractal-like hierarchical organization of bone begins at the nanoscale. Science 2018, 360, 6388. [Google Scholar] [CrossRef]
- Schmidt, A.H. Autologous bone graft: Is it still the gold standard? Injury 2021, 52, S18–S22. [Google Scholar] [CrossRef]
- Yamamoto, N.; Hayashi, K.; Tsuchiya, H. Progress in biological reconstruction and enhanced bone revitalization for bone defects. J. Orthop. Sci. 2019, 24, 387–392. [Google Scholar] [CrossRef]
- Bauer, T.W.; Muschler, G.F. Bone graft materials. An overview of the basic science. Clin. Orthop. Relat. Res. 2000, 371, 10–27. [Google Scholar] [CrossRef]
- Yu, M.; Yeow, Y.J.; Lawrence, L.; Claudio, P.P.; Day, J.B.; Salary, R. Investigation of the Effects of Design and Process Parameters on the Mechanical Properties of Biodegradable Bone Scaffolds, Fabricated Using Pneumatic Microextrusion Process. In Proceedings of the ASME International Mechanical Engineering Congress and Exposition, Virtual on Line, 16–19 November 2020; p. 7. [Google Scholar] [CrossRef]
- Chaffins, A.; Yu, M.; Claudio, P.P.; Day, J.B.; Salary, R. Investigation of the Functional Properties of Additively-Fabricated Triply Periodic Minimal Surface-Based Bone Scaffolds for the Treatment of Osseous Fractures. In Proceedings of the ASME 2021 International Manufacturing Science and Engineering Conference (MSEC 2021), Cincinnati, OH, USA, 21–25 June 2021. [Google Scholar] [CrossRef]
- Klemstine, C.; Abdelgaber, Y.; Lawrence, L.; Day, J.B.; Claudio, P.P.; Salary, R. Characterization of the Compressive Properties of Triply Periodic Minimal Surface PCL Scaffolds for Bone Tissue Engineering. In Proceedings of the ASME International Mechanical Engineering Congress & Exposition (IMECE 2021), Virtual, 20 February 2023. [Google Scholar] [CrossRef]
- Mandava, P.K.; Joyce, R.; Day, J.B.; Salary, R.R. Investigation of the Mechanical Properties of Additively Manufactured Bone Tissue Scaffolds, Composed of Polyamide, Polyolefin, and Cellulose Fibers. In Proceedings of the ASME 2022 International Manufacturing Science and Engineering Conference (MSEC 2022), West Lafayette, IN, USA, 27 June–1 July 2022. [Google Scholar]
- Raines, R.; Day, J.B.; Salary, R.R. Experimental Characterization of the Mechanical Properties of Medical-Grade Dental Implants, Fabricated Using Vat-Photopolymerization Additive Manufacturing Process. In Proceedings of the ASME 2022 International Manufacturing Science and Engineering Conference (MSEC 2022), West Lafayette, IN, USA, 27 June–1 July 2022. [Google Scholar]
- Karageorgiou, V.; Kaplan, D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials 2005, 26, 5474–5491. [Google Scholar] [CrossRef]
- Abdelgaber, Y.; Klemstine, C.; Salary, R. A Novel, Image-Based Method for Characterization of the Porosity of Additively Manufactured Bone Scaffolds with Complex Microstructures. J. Manuf. Sci. Eng. 2023, 145, 0411008. [Google Scholar] [CrossRef]
- Yamagiwa, H.; Endo, N. Bone fracture and the healing mechanisms. Histological aspect of fracture healing. Primary and secondary healing. Clin. Calcium 2009, 19, 627–633. [Google Scholar] [PubMed]
- ElHawary, H.; Baradaran, A.; Abi-Rafeh, J.; Vorstenbosch, J.; Xu, L.; Efanov, J.I. Bone Healing and Inflammation: Principles of Fracture and Repair. Semin. Plast. Surg. 2021, 35, 198–203. [Google Scholar] [CrossRef]
- Sathyendra, V.; Darowish, M. Basic Science of Bone Healing. Hand Clin. 2013, 29, 473–481. [Google Scholar] [CrossRef]
- Anderson, H.C. Matrix vesicles and calcification. Curr. Rheumatol. Rep. 2003, 5, 222–226. [Google Scholar] [CrossRef]
- Grover, V.; Malhotra, R.; Kapoor, A.; Sachdeva, S. Bone allografts: A review of safety and efficacy. Indian J. Dent. Res. 2011, 22, 496. [Google Scholar] [CrossRef] [PubMed]
- Oro, F.B.; Sikka, R.S.; Wolters, B.; Graver, R.; Boyd, J.L.; Nelson, B.J.; Swiontkowski, M. Autograft Versus Allograft: An Economic Cost Comparison of Anterior Cruciate Ligament Reconstruction. Arthrosc. J. Arthrosc. Relat. Surg. 2011, 27, 1219–1225. [Google Scholar] [CrossRef]
- Alford, A.I.; Nicolaou, D.; Hake, M.; McBride-Gagyi, S. Masquelet’s induced membrane technique: Review of current concepts and future directions. J. Orthop. Res. 2021, 39, 707–718. [Google Scholar] [CrossRef]
- Masquelet, A.-C.; Fitoussi, F.; Begue, T.; Muller, G.P. Reconstruction of the long bones by the induced membrane and spongy autograft. Ann. Chir. Plast. Esthet. 2000, 45, 346–353. [Google Scholar]
- Verboket, R.D.; Leiblein, M.; Janko, M.; Schaible, A.; Brune, J.C.; Schröder, K.; Heilani, M.; Fremdling, C.; Busche, Y.; Irrle, T.; et al. From two stages to one: Acceleration of the induced membrane (Masquelet) technique using human acellular dermis for the treatment of non-infectious large bone defects. Eur. J. Trauma Emerg. Surg. 2020, 46, 317–327. [Google Scholar] [CrossRef]
- Carson, J.S.; Bostrom, M.P. Synthetic bone scaffolds and fracture repair. Injury 2007, 38, S33–S37. [Google Scholar] [CrossRef] [PubMed]
- Kretlow, J.D.; Mikos, A.G. Review: Mineralization of Synthetic Polymer Scaffolds for Bone Tissue Engineering. Tissue Eng. 2007, 13, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Ganguli, A.; Pagan-Diaz, G.J.; Grant, L.; Cvetkovic, C.; Bramlet, M.; Vozenilek, J.; Kesavadas, T.; Bashir, R. 3D printing for preoperative planning and surgical training: A review. Biomed. Microdevices 2018, 20, 65. [Google Scholar] [CrossRef] [PubMed]
- Kantaros, A. 3D Printing in Regenerative Medicine: Technologies and Resources Utilized. Int. J. Mol. Sci. 2022, 23, 14621. [Google Scholar] [CrossRef]
- Bishop, E.S.; Mostafa, S.; Pakvasa, M.; Luu, H.H.; Lee, M.J.; Wolf, J.M.; Ameer, G.A.; He, T.-C.; Reid, R.R. 3-D bioprinting technologies in tissue engineering and regenerative medicine: Current and future trends. Genes Dis. 2017, 4, 185–195. [Google Scholar] [CrossRef]
- Kantaros, A.; Piromalis, D. Fabricating Lattice Structures via 3D Printing: The Case of Porous Bio-Engineered Scaffolds. Appl. Mech. 2021, 2, 289–302. [Google Scholar] [CrossRef]
- Jammalamadaka, U.; Tappa, K. Recent Advances in Biomaterials for 3D Printing and Tissue Engineering. J. Funct. Biomater. 2018, 9, 22. [Google Scholar] [CrossRef]
- Salary, R. Advanced Manufacturing for Bone Tissue Engineering and Regenerative Medicine; IntechOpen: London, UK, 2022; p. 304. [Google Scholar] [CrossRef]
- Yu, M.; Yeow, Y.J.; Lawrence, L.; Claudio, P.P.; Day, J.B.; Salary, R. Characterization of the Functional Properties of Polycaprolactone Bone Scaffolds Fabricated Using Pneumatic Micro-Extrusion. J. Micro Nano Manuf. 2021, 9, 030905. [Google Scholar] [CrossRef]
- Kantaros, A. Bio-Inspired Materials: Exhibited Characteristics and Integration Degree in Bio-Printing Operations. Am. J. Eng. Appl. Sci. 2022, 15, 255–263. [Google Scholar] [CrossRef]
- Rodrίguez, F.J.; Gómez, N.; Perego, G.; Navarro, X. Highly permeable polylactide-caprolactone nerve guides enhance peripheral nerve regeneration through long gaps. Biomaterials 1999, 20, 1489–1500. [Google Scholar] [CrossRef]
- Putra, R.U.; Basri, H.; Prakoso, A.T.; Chandra, H.; Ammarullah, M.I.; Akbar, I.; Syahrom, A.; Kamarul, T. Level of Activity Changes Increases the Fatigue Life of the Porous Magnesium Scaffold, as Observed in Dynamic Immersion Tests, over Time. Sustainability 2023, 15, 823. [Google Scholar] [CrossRef]
- Salama, M.; Vaz, M.F.; Colaço, R.; Santos, C.; Carmezim, M. Biodegradable Iron and Porous Iron: Mechanical Properties, Degradation Behaviour, Manufacturing Routes and Biomedical Applications. J. Funct. Biomater. 2022, 13, 72. [Google Scholar] [CrossRef] [PubMed]
- Semenova, I.P.; Modina, Y.M.; Stotskiy, A.G.; Polyakov, A.V.; Pesin, M.V. Fatigue Properties of Ti Alloys with an Ultrafine Grained Structure: Challenges and Achievements. Metals 2022, 12, 312. [Google Scholar] [CrossRef]
- Sakthiabirami, K.; Soundharrajan, V.; Kang, J.-H.; Yang, Y.; Park, S.-W. Three-Dimensional Zirconia-Based Scaffolds for Load-Bearing Bone-Regeneration Applications: Prospects and Challenges. Materials 2021, 14, 3207. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Lai, J.; Li, K.; Zhu, S.; Lu, B.; Liu, J.; Tang, Y.; Wei, Y. Cryogenic 3D printing of dual-delivery scaffolds for improved bone regeneration with enhanced vascularization. Bioact. Mater. 2021, 6, 137–145. [Google Scholar] [CrossRef]
- Chen, V.J.; Smith, L.A.; Ma, P.X. Bone regeneration on computer-designed nano-fibrous scaffolds. Biomaterials 2006, 27, 3973–3979. [Google Scholar] [CrossRef]
- Inzana, J.A.; Olvera, D.; Fuller, S.M.; Kelly, J.P.; Graeve, O.A.; Schwarz, E.M.; Kates, S.L.; Awad, H.A. 3D printing of composite calcium phosphate and collagen scaffolds for bone regeneration. Biomaterials 2014, 35, 4026–4034. [Google Scholar] [CrossRef]
- Zhang, W.; Feng, C.; Yang, G.; Li, G.; Ding, X.; Wang, S.; Dou, Y.; Zhang, Z.; Chang, J.; Wu, C.; et al. 3D-printed scaffolds with synergistic effect of hollow-pipe structure and bioactive ions for vascularized bone regeneration. Biomaterials 2017, 135, 85–95. [Google Scholar] [CrossRef]
- Lawrence, L.M.; Cottrill, A.; Valluri, A.; Marenzi, G.; Denning, K.L.; Valluri, J.; Claudio, P.P.; Day, J.B. Minimally Manipulative Method for the Expansion of Human Bone Marrow Mesenchymal Stem Cells to Treat Osseous Defects. Int. J. Mol. Sci. 2019, 20, 612. [Google Scholar] [CrossRef]
- Lei, M.; Zhang, M.; Luo, N.; Ye, J.; Lin, F.; Chen, Y.; Chen, J.; Xiao, M. The clinical performance of ultra-low-dose shoulder CT scans: The assessment on image and physical 3D printing models. PLoS ONE 2022, 17, e0275297. [Google Scholar] [CrossRef]
- Lawrence, L.; Day, J.B.; Claudio, P.P.; Salary, R. Investigation of the Regenerative Potential of Human Bone Marrow Stem Cell-Seeded Polycaprolactone Bone Scaffolds, Fabricated Using Pneumatic Microextrusion Process. In Proceedings of the ASME 2021 International Manufacturing Science and Engineering Conference (MSEC 2021), Cincinnati, OH, USA, 21–25 June 2021. [Google Scholar] [CrossRef]
- Zhao, D.; Yu, M.; Lawrence, L.; Claudio, P.P.; Day, J.B.; Salary, R. Investigation of the Influence of Consequential Design Parameters on the Mechanical Performance of Biodegradable Bone Scaffolds, Fabricated Using Pneumatic Micro-Extrusion Additive Manufacturing Process. In Proceedings of the ASME 2020 International Manufacturing Science and Engineering Conference (MSEC 2020), Cincinnati, OH, USA, 22–26 June 2020. [Google Scholar] [CrossRef]
- Valtanen, R.S.; Yang, Y.P.; Gurtner, G.C.; Maloney, W.J.; Lowenberg, D.W. Synthetic and Bone tissue engineering graft substitutes: What is the future? Injury 2021, 52, S72–S77. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Cosme, J.G.; Xu, T.; Miszuk, J.M.; Picciani, P.H.; Fong, H.; Sun, H. Three dimensional electrospun PCL/PLA blend nanofibrous scaffolds with significantly improved stem cells osteogenic differentiation and cranial bone formation. Biomaterials 2017, 115, 115–127. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lawrence, L.M.; Salary, R.; Miller, V.; Valluri, A.; Denning, K.L.; Case-Perry, S.; Abdelgaber, K.; Smith, S.; Claudio, P.P.; Day, J.B. Osteoregenerative Potential of 3D-Printed Poly ε-Caprolactone Tissue Scaffolds In Vitro Using Minimally Manipulative Expansion of Primary Human Bone Marrow Stem Cells. Int. J. Mol. Sci. 2023, 24, 4940. https://doi.org/10.3390/ijms24054940
Lawrence LM, Salary R, Miller V, Valluri A, Denning KL, Case-Perry S, Abdelgaber K, Smith S, Claudio PP, Day JB. Osteoregenerative Potential of 3D-Printed Poly ε-Caprolactone Tissue Scaffolds In Vitro Using Minimally Manipulative Expansion of Primary Human Bone Marrow Stem Cells. International Journal of Molecular Sciences. 2023; 24(5):4940. https://doi.org/10.3390/ijms24054940
Chicago/Turabian StyleLawrence, Logan M., Roozbeh (Ross) Salary, Virginia Miller, Anisha Valluri, Krista L. Denning, Shannon Case-Perry, Karim Abdelgaber, Shannon Smith, Pier Paolo Claudio, and James B. Day. 2023. "Osteoregenerative Potential of 3D-Printed Poly ε-Caprolactone Tissue Scaffolds In Vitro Using Minimally Manipulative Expansion of Primary Human Bone Marrow Stem Cells" International Journal of Molecular Sciences 24, no. 5: 4940. https://doi.org/10.3390/ijms24054940
APA StyleLawrence, L. M., Salary, R., Miller, V., Valluri, A., Denning, K. L., Case-Perry, S., Abdelgaber, K., Smith, S., Claudio, P. P., & Day, J. B. (2023). Osteoregenerative Potential of 3D-Printed Poly ε-Caprolactone Tissue Scaffolds In Vitro Using Minimally Manipulative Expansion of Primary Human Bone Marrow Stem Cells. International Journal of Molecular Sciences, 24(5), 4940. https://doi.org/10.3390/ijms24054940








