Simple Detection and Culture of Circulating Tumor Cells from Colorectal Cancer Patients Using Poly(2-Methoxyethyl Acrylate)-Coated Plates
Abstract
1. Introduction
2. Results
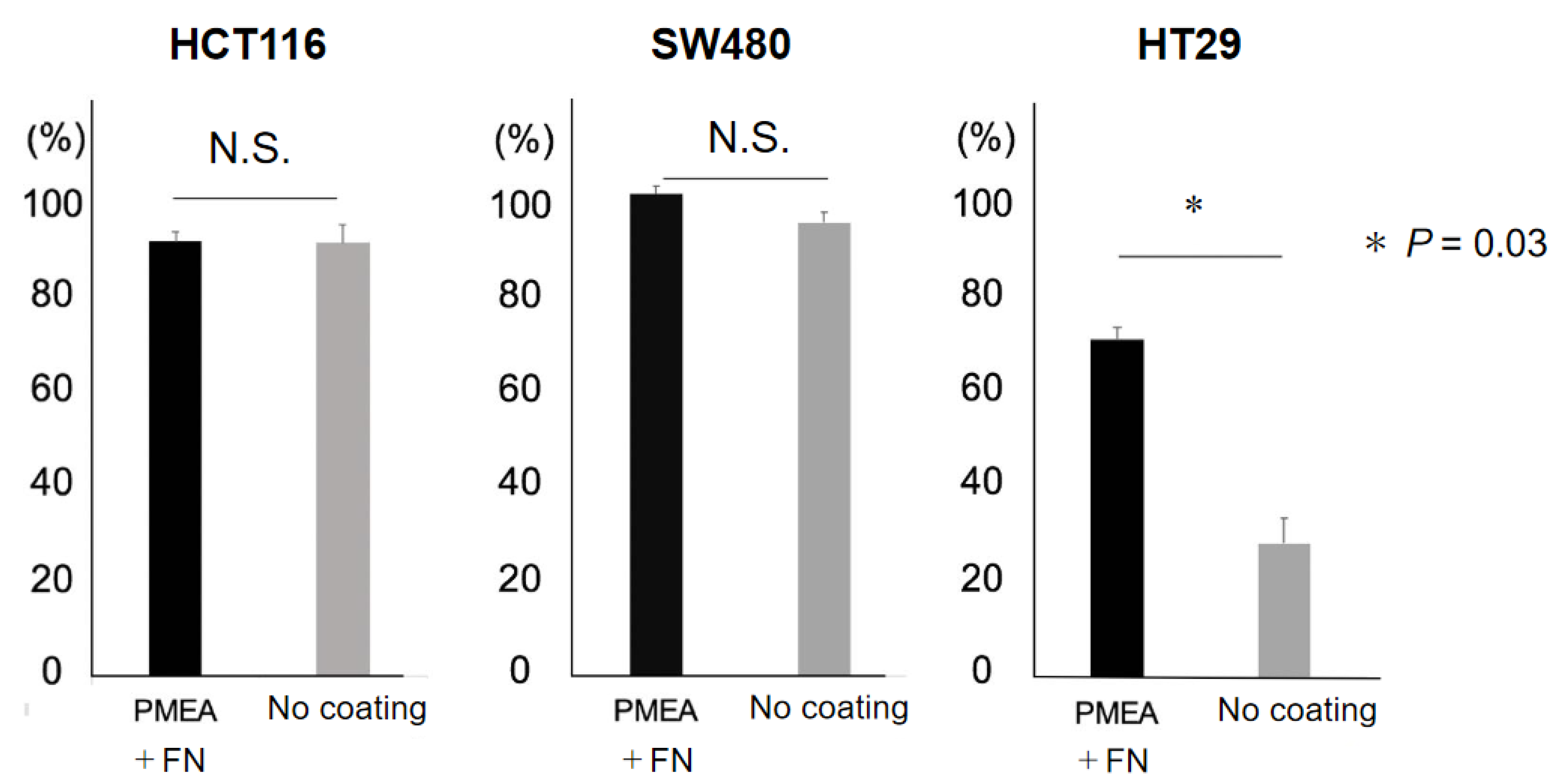
2.1. Adhesion of CRC Cell Lines to PMEA-Coated and Non-Coated Plates
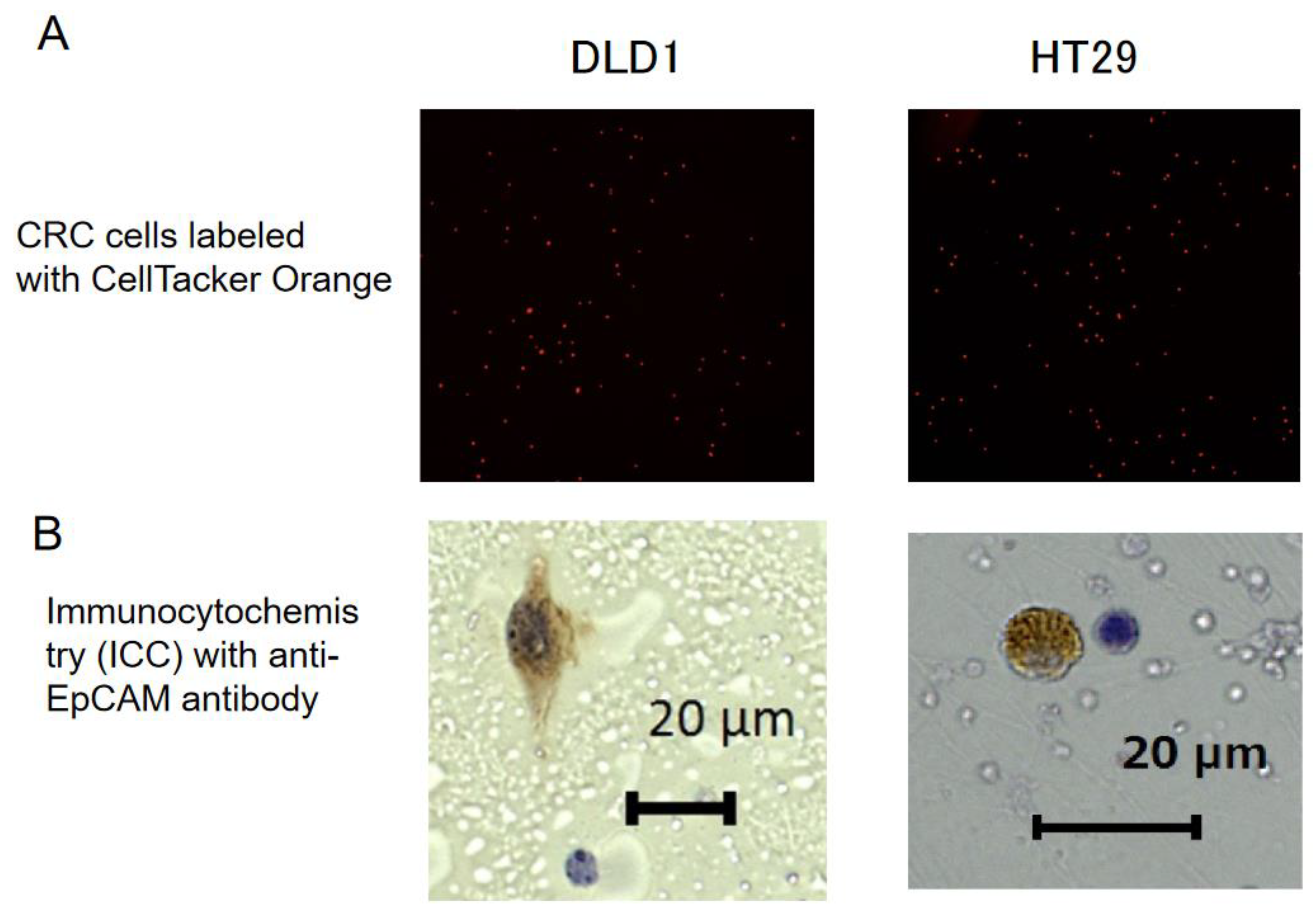
2.2. Spike Test
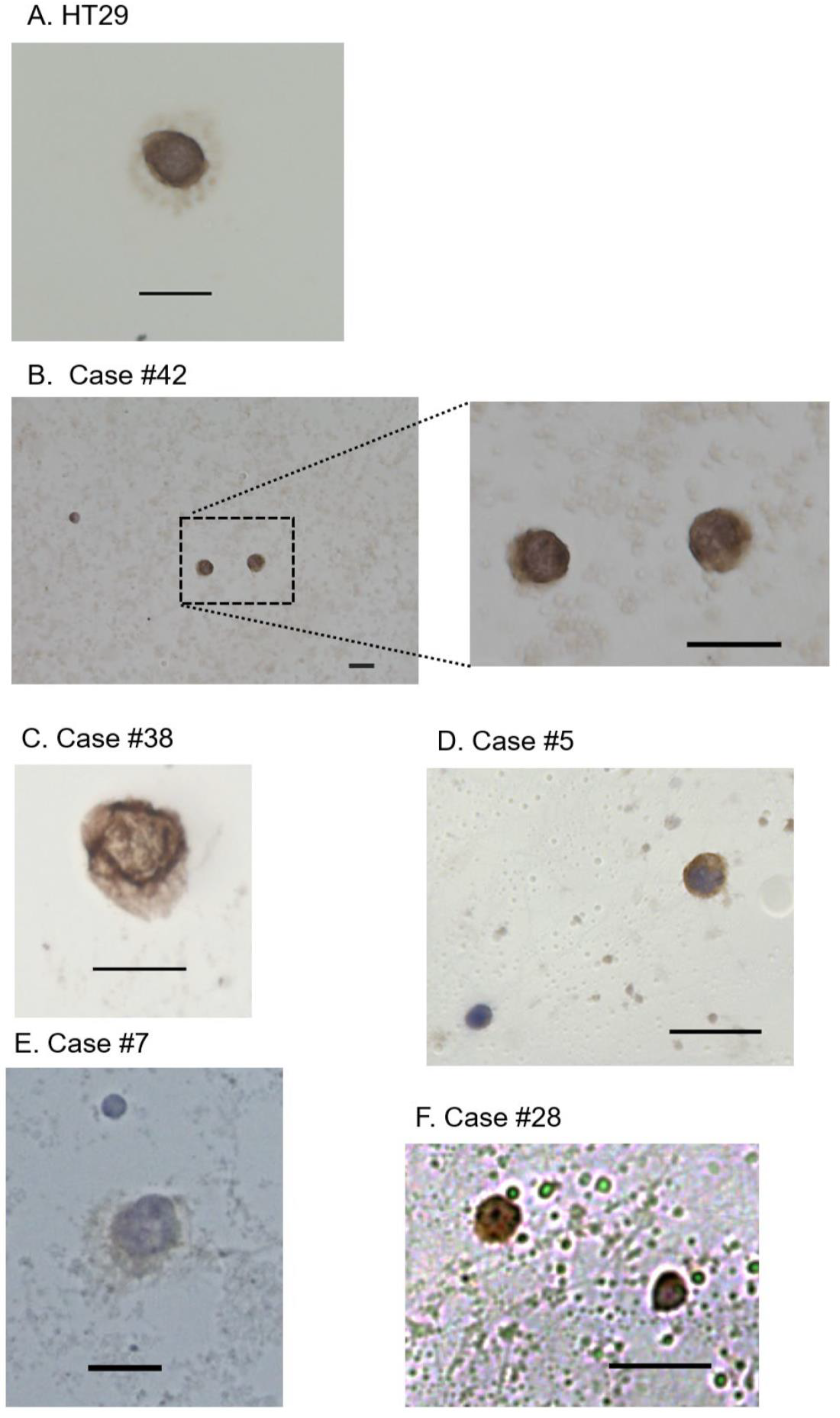
2.3. Immunocytochemistry for EpCAM and Cytological Examination
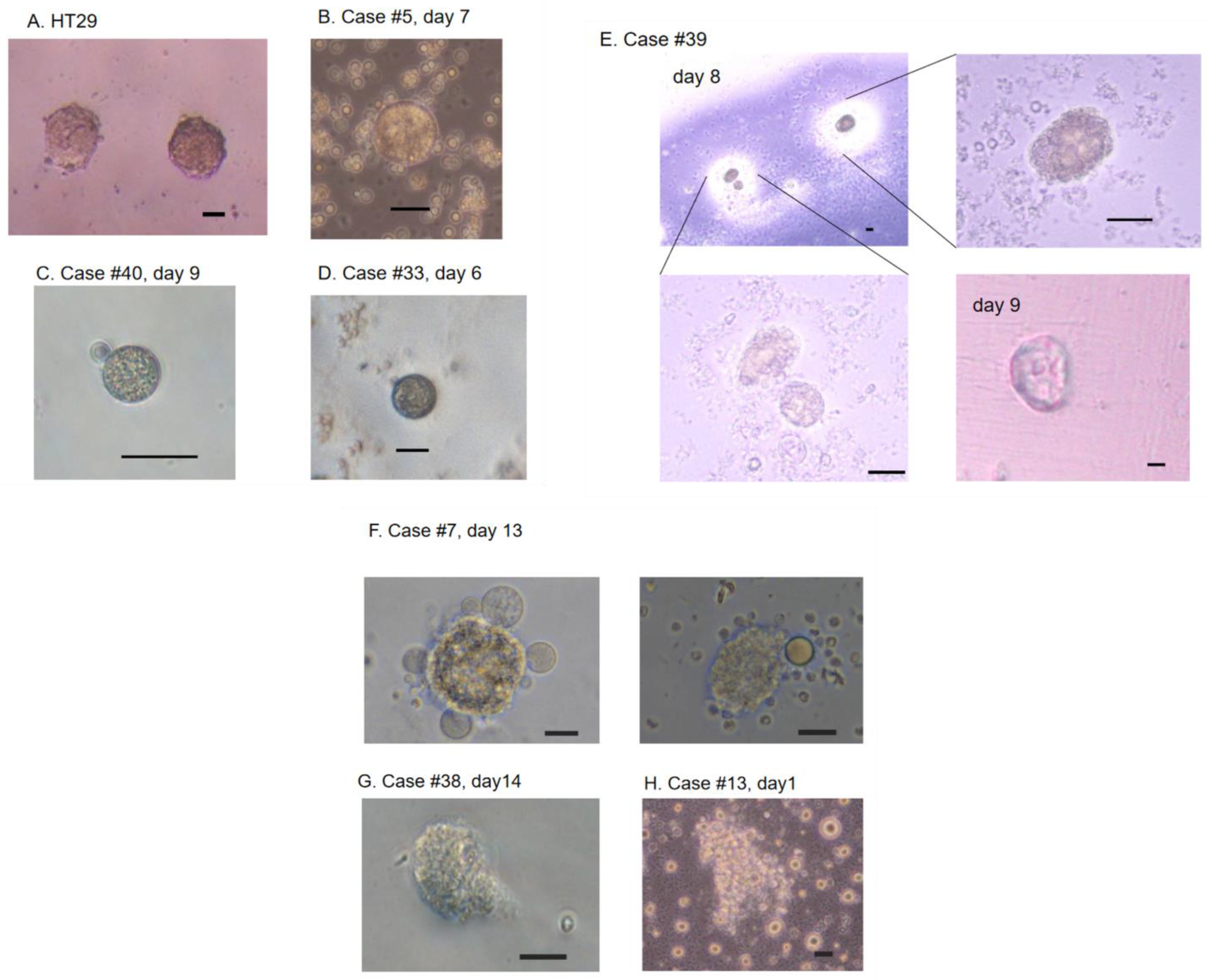
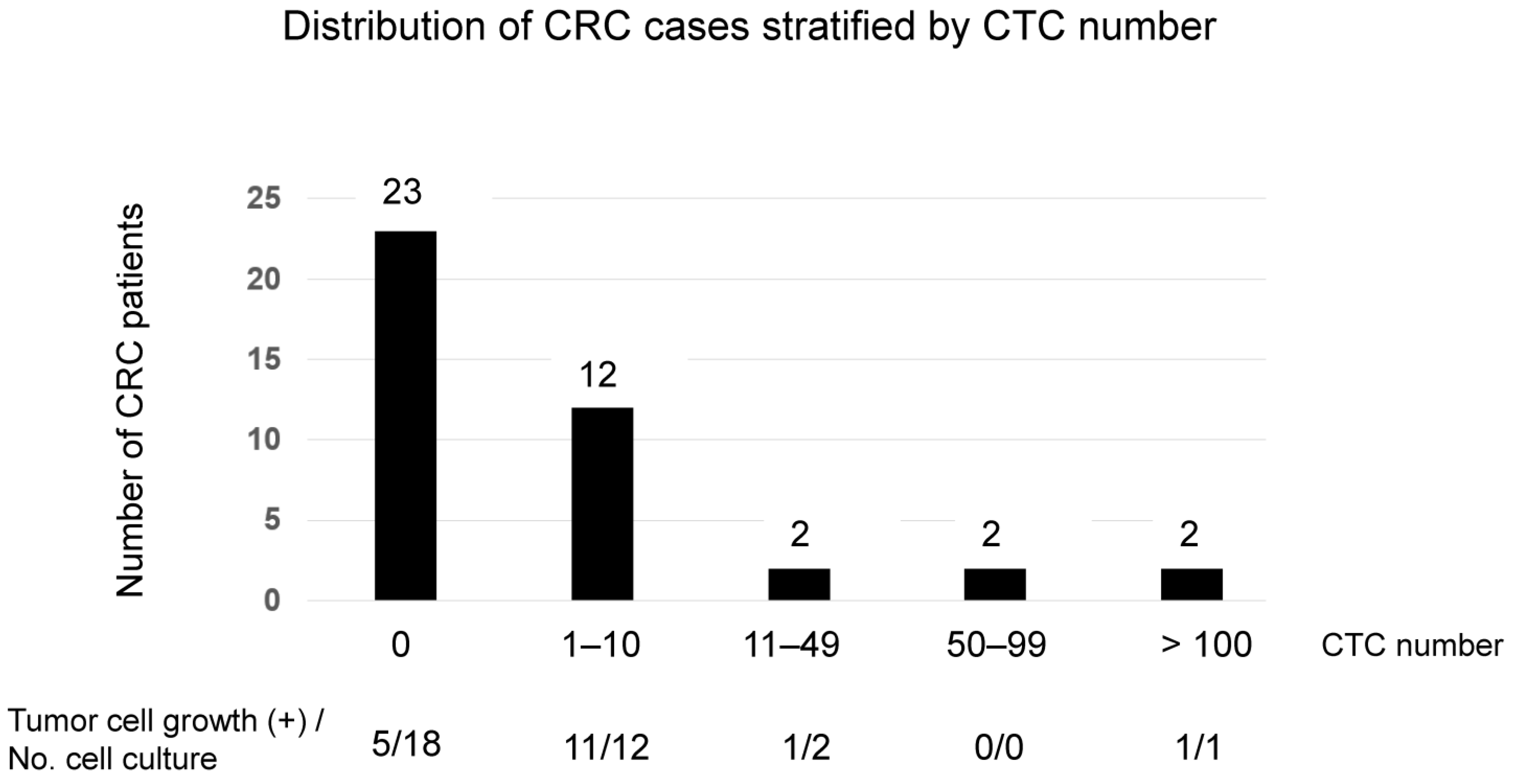
2.4. Cell Culture
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Preparation of PMEA-Coated Chamber Slides
4.3. Adhesion Test
4.4. Spike Test
4.5. Immunostaining
4.6. Patient Background Data
4.7. Enrichment of CTCs in Blood Samples
4.8. Culture of CTCs
4.9. Definition of CTC
4.10. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Wittekind, C.; Neid, M. Cancer invasion and metastasis. Oncology 2005, 69 (Suppl. 1), 14–16. [Google Scholar] [CrossRef]
- Colvin, H.; Mizushima, T.; Eguchi, H.; Takiguchi, S.; Doki, Y.; Mori, M. Gastroenterological surgery in Japan: The past, the present and the future. Ann. Gastroenterol. Surg. 2017, 1, 5–10. [Google Scholar] [CrossRef]
- Fabbri, F.; Carloni, S.; Zoli, W.; Ulivi, P.; Gallerani, G.; Fici, P.; Chiadini, E.; Passardi, A.; Frassineti, G.L.; Ragazzini, A.; et al. Detection and recovery of circulating colon cancer cells using a dielectrophoresis-based device: KRAS mutation status in pure CTCs. Cancer Lett. 2013, 335, 225–231. [Google Scholar] [CrossRef]
- Nakamura, Y.; Yoshino, T. Clinical Utility of Analyzing Circulating Tumor DNA in Patients with Metastatic Colorectal Cancer. Oncologist 2018, 23, 1310–1318. [Google Scholar] [CrossRef] [PubMed]
- Tie, J.; Cohen, J.D.; Wang, Y.; Christie, M.; Simons, K.; Lee, M.; Wong, R.; Kosmider, S.; Ananda, S.; McKendrick, J.; et al. Circulating Tumor DNA Analyses as Markers of Recurrence Risk and Benefit of Adjuvant Therapy for Stage III Colon Cancer. JAMA Oncol. 2019, 5, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.J.; Punt, C.J.A.; Iannotti, N.; Saidman, B.H.; Sabbath, K.D.; Gabrail, N.Y.; Picus, J.; Morse, M.; Mitchell, E.; Miller, M.C.; et al. Relationship of circulating tumor cells to tumor response, progression-free survival, and overall survival in patients with metastatic colorectal cancer. J. Clin. Oncol. 2008, 26, 3213–3221. [Google Scholar] [CrossRef]
- de Bono, J.S.; Scher, H.I.; Montgomery, R.B.; Parker, C.; Miller, M.C.; Tissing, H.; Doyle, G.V.; Terstappen, L.W.W.M.; Pienta, K.J.; Raghavan, D. Circulating tumor cells predict survival benefit from treatment in metastatic castration-resistant prostate cancer. Clin. Cancer Res. 2008, 14, 6302–6309. [Google Scholar] [CrossRef] [PubMed]
- Cristofanilli, M.; Budd, G.T.; Ellis, M.J.; Stopeck, A.; Matera, J.; Miller, M.C.; Reuben, J.M.; Doyle, G.V.; Allard, W.J.; Terstappen, L.W.M.M.; et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N. Engl. J. Med. 2004, 351, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Marcuello, M.; Vymetalkova, V.; Neves, R.P.L.; Duran-Sanchon, S.; Vedeld, H.M.; Tham, E.; Dalum, V.G.; Flugen, G.; Garcia-Barberan, V.; Fineman, R.J.; et al. Circulating biomarkers for early detection and clinical management of colorectal cancer. Mol. Aspects Med. 2019, 69, 107–122. [Google Scholar] [CrossRef]
- Vasseur, A.; Kiavue, N.; Bidard, F.C.; Pierga, J.Y.; Cabel, L. Clinical utility of circulating tumor cells: An update. Mol. Oncol. 2021, 15, 1647–1666. [Google Scholar] [CrossRef]
- Sun, Y.F.; Wu, L.; Liu, S.P.; Jiang, M.M.; Hu, B.; Zhou, K.Q.; Guo, W.; Xu, Y.; Zhong, Y.; Zhou, X.R.; et al. Dissecting spatial heterogeneity and the immune-evasion mechanism of CTCs by single-cell RNA-seq in hepatocellular carcinoma. Nat. Commun. 2021, 12, 4091. [Google Scholar] [CrossRef]
- Mout, L.; van Dessel, L.F.; Kraan, J.; de Jong, A.C.; Neves, R.P.L.; Erkens-Schulze, S.; Beaufort, C.M.; Sieuwerts, A.M.; van Riet, J.; Woo, T.L.C.; et al. Generating human prostate cancer organoids from leukapheresis enriched circulating tumour cells. Eur. J. Cancer 2021, 150, 179–189. [Google Scholar] [CrossRef]
- Allen, J.E.; EI-Deiry, W.S. Circulating tumor cells and colorectal cancer. Curr. Colorectal. Cancer Rep. 2010, 6, 212–220. [Google Scholar] [CrossRef]
- Huang, X.; Gao, P.; Song, Y.; Sun, J.; Chen, X.; Zhao, J.; Liu, J.; Xu, H.; Wang, Z. Relationship between circulating tumor cells and tumor response in colorectal cancer patients treated with chemotherapy: A meta-analysis. BMC Cancer 2014, 14, 976. [Google Scholar] [CrossRef] [PubMed]
- Peeters, D.J.E.; De Laere, B.; Van den Eynden, G.G.; Laere, S.J.V.; Rothe, F.; Ignatiadis, M.; Sieuwerts, A.M.; Lambrechts, D.; Rutten, A.; van Dam, P.A.; et al. Semiautomated isolation and molecular characterisation of single or highly purified tumour cells from CellSearch enriched blood samples using dielectrophoretic cell sorting. Br. J. Cancer 2013, 108, 1358–1367. [Google Scholar] [CrossRef] [PubMed]
- Pantel, K.; Brakenhoff, R.H.; Brandt, B. Detection, clinical relevance and specific biological properties of disseminating tumour cells. Nat. Rev. Cancer 2008, 8, 329–340. [Google Scholar] [CrossRef]
- Cayrefourcq, L.; Mazard, T.; Joosse, S.; Solassol, J.; Ramos, J.; Assenat, E.; Schumacher, U.; Costes, V.; Maudelonde, T.; Pantel, K.; et al. Establishment and characterization of a cell line from human circulating colon cancer cells. Cancer Res. 2015, 75, 892–901. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Gu, Y.; Yu, F.; Zhou, L.; Cheng, X.; Jiang, H.; Huang, Y.; Zhang, Y.; Xu, T.; Qian, W.; et al. The Number of Intraoperative Intestinal Venous Circulating Tumor Cells Is a Prognostic Factor for Colorectal Cancer Patients. Evid. Based Complement. Alternat. Med. 2022, 2022, 4162354. [Google Scholar] [CrossRef] [PubMed]
- Abdalla, T.S.A.; Meiners, J.; Riethdorf, S.; Konig, A.; Melling, N.; Gorges, T.; Karstens, K.F.; Izbicki, J.R.; Pantel, K.; Reeh, M. Prognostic value of preoperative circulating tumor cells counts in patients with UICC stage I-IV colorectal cancer. PLoS ONE 2021, 16, e0252897. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhou, S.; Zhang, W.; Wang, J.; Wang, M.; Hu, X.; Liu, F.; Zhang, Y.; Jiang, B.; Yuan, H. Circulating tumor cells as an independent prognostic factor in advanced colorectal cancer: A retrospective study in 121 patients. Int. J. Colorectal Dis. 2019, 34, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Hoshiba, T.; Nikaido, M.; Tanaka, M. Characterization of the attachment mechanisms of tissue-derived cell lines to blood-compatible polymers. Adv. Healthc. Mater. 2014, 3, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Hoshiba, T.; Nemoto, E.; Sato, K.; Orui, T.; Otaki, T.; Yoshihiro, A.; Tanaka, M. Regulation of the Contribution of Integrin to Cell Attachment on Poly(2-Methoxyethyl Acrylate) (PMEA) Analogous Polymers for Attachment-Based Cell Enrichment. PLoS ONE 2015, 10, e0136066. [Google Scholar] [CrossRef] [PubMed]
- Hoshiba, T.; Tanaka, M. Integrin-independent Cell Adhesion Substrates: Possibility of Applications for Mechanobiology Research. Anal. Sci. 2016, 32, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.-Y.; Aratsu, F.; Sekida, S.; Kobayashi, S.; Tanaka, M. Blood-Compatible Poly(2-methoxyethyl acrylate) Induces Blebbing-like Phenomenon and Promotes Viability of Tumor Cells in Serum-Free Medium. ACS Appl. Bio Mater. 2020, 3, 1858–1864. [Google Scholar] [CrossRef] [PubMed]
- Nishida, K.; Sekida, S.; Anada, T.; Tanaka, M. Modulation of Biological Responses of Tumor Cells Adhered to Poly(2-methoxyethyl acrylate) with Increasing Cell Viability under Serum-Free conditions. ACS Biomater. Sci. Eng. 2022, 8, 672–681. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Sugasaki, A.; Yamamoto, Y.; Shigenoi, Y.; Udaka, A.; Yamamoto, A.; Tanaka, M. Enrichment of Cancer Cells Based on Antibody-Free Selective Cell Adhesion. ACS Biomater. Sci. Eng. 2022, 8, 4547–4556. [Google Scholar] [CrossRef]
- Tanaka, M.; Motomura, T.; Kawada, M.; Anzai, T.; Kasori, Y.; Shiroya, T.; Shimura, K.; Onishi, M.; Mochizuki, A. Blood compatible aspects of Poly(2-methoxyethylacrylate) (PMEA) -Relationship between protein adsorption and platelet adhesion on PMEA surface. Biomaterials 2000, 21, 1471–1481. [Google Scholar] [CrossRef]
- Tanaka, M.; Motomura, T.; Kawada, M.; Anzai, T.; Kasori, Y.; Shiroya, T.; Shimura, K.; Onishi, M.; Mochizuki, A.; Okahata, Y. A new blood-compatible surface prepared by Poly(2-methoxyethyl acrylate) (PMEA) coating -Protein adsorption on PMEA surface. Jpn. J. Art. Org. 2000, 29, 209–216. [Google Scholar]
- Tanaka, M. Design of Novel BioInterfaces (I) -Blood compatibility of poly(2-methoxyethyl acrylate). Bio-Med. Mater. Eng. 2004, 14, 427–438. [Google Scholar]
- Tanaka, M.; Mochizuki, A. Clarification of the Blood Compatibility Mechanism by Controlling the Water Structure at the Blood–Poly(meth)acrylate Interface. J. Biomat. Sci. Polym. Ed. 2010, 21, 1849–1863. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Tanaka, M. Designing Smart Biomaterials for Tissue Engineering. Int. J. Mol. Sci. 2018, 19, 17. [Google Scholar] [CrossRef]
- Sato, K.; Kobayashi, S.; Kusakari, M.; Watahiki, S.; Oikawa, M.; Hoshiba, T.; Tanaka, M. The relationship between water structure and blood compatibility in poly(2-methoxyethyl acrylate) (PMEA) analogues. Macromol. Biosci. 2015, 15, 1296–1303. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Iglesias, L.; Barcia-Castro, L.; Rodríguez-Quiroga, M.; Páez de la Cadena, M.; Rodríguez-Berrocal, J.; Cordero, O.J. Surface expression marker profile in colon cancer cell lines and sphere-derived cells suggests complexity in CD26+ cancer stem cells subsets. Biol. Open 2019, 8, bio041673. [Google Scholar] [CrossRef]
- Sacchetti, A.; Teeuwssen, M.; Verhagen, M.; Joosten, R.; Xu, T.; Stabile, R.; van der Steen, B.; Watson, M.M.; Gusinac, A.; Kim, W.K.; et al. Phenotypic plasticity underlies local invasion and distant metastasis in colon cancer. Elife 2021, 10, e61461. [Google Scholar] [CrossRef]
- Ning, Y.; Zhang, W.; Hanna, D.L.; Yang, D.; Okazaki, S.; Berger, M.D.; Miyamoto, Y.; Suenaga, M.; Schirripa, M.; EI-Khoueiry, A.; et al. Clinical relevance of EMT and stem-like gene expression in circulating tumor cells of metastatic colorectal cancer patients. Pharm. J. 2018, 18, 29–34. [Google Scholar] [CrossRef]
- Nomura, M.; Miyake, Y.; Inoue, A.; Yokoyama, Y.; Noda, N.; Kouda, S.; Hata, T.; Ogino, T.; Miyoshi, N.; Uemura, M.; et al. Single-Cell Analysis of Circulating Tumor Cells from Patients with Colorectal Cancer Captured with a Dielectrophoresis-Based Micropore System. Biomedicines 2023, 11, 203. [Google Scholar] [CrossRef] [PubMed]
- Yokobori, T.; Iinuma, H.; Shimamura, T.; Imoto, S.; Sugimachi, K.; Ishii, H.; Iwatsuki, M.; Ota, D.; Ohkuma, M.; Iwaya, T.; et al. Plastin3 is a novel marker for circulating tumor cells undergoing the epithelial-mesenchymal transition and is associated with colorectal cancer prognosis. Cancer Res. 2013, 73, 2059–2069. [Google Scholar] [CrossRef]
- Hoshiba, T.; Orui, T.; Endo, C.; Sato, K.; Yoshihiro, A.; Minagawa, Y.; Tanaka, M. Adhesion-Based Simple Capture and Recovery of Circulating Tumor Cells Using a Blood-Compatible and Thermo-Responsive Polymer-Coated Substrate. RSC Adv. 2016, 6, 89103–89112. [Google Scholar] [CrossRef]
- Nishida, K.; Nishimura, S.; Tanaka, M. Selective Accumulation to Tumor Cells with Coacervate Droplets Formed from a Water-Insoluble Acrylate Polymer. Biomacromolecules 2022, 23, 1569–1580. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Kobayashi, S.; Murakami, D.; Aratsu, F.; Kashiwazaki, A.; Hoshiba, T.; Fukushima, K. Design of Polymeric Biomaterials: The “Intermediate Water Concept”. Bull. Chem. Soc. Jpn. 2019, 92, 2043–2057. [Google Scholar] [CrossRef]
- Nishida, K.; Anada, T.; Tanaka, M. Roles of interfacial water states on advanced biomedical material design. Adv. Drug Deliv. Rev. 2022, 186, 114310. [Google Scholar] [PubMed]
- Kobayashi, S.; Wakui, M.; Iwata, Y.; Tanaka, M. Poly(ω-methoxyalkyl acrylate)s: A nonthrombogenic polymer family with tunable protein adsorption. Biomacromolecules 2017, 18, 4214–4223. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, A.; Nagano, H.; Sakon, M.; Fujiwara, Y.; Sugita, Y.; Eguchi, H.; Kondo, M.; Arai, I.; Morimoto, O.; Dono, K.; et al. Clinical application of quantitative analysis for detection of hematogenous spread of hepatocellular carcinoma by real-time PCR. Int. J. Oncol. 2001, 18, 527–532. [Google Scholar] [CrossRef]
- Morimoto, O.; Nagano, H.; Miyamoto, A.; Fujiwara, Y.; Kondo, M.; Yamamoto, T.; Ota, H.; Nakamura, M.; Wada, H.; Damdinsuren, B.; et al. Association between recurrence of hepatocellular carcinoma and alpha-fetoprotein messenger RNA levels in peripheral blood. Surg. Today 2005, 35, 1033–1041. [Google Scholar] [CrossRef]
- Yamamoto, H.; Monden, T.; Ikeda, K.; Izawa, H.; Fukuda, K.; Fukunaga, M.; Tomita, N.; Shimano, T.; Shiozaki, H.; Monden, M. Coexpression of cdk2/cdc2 and retinoblastoma gene products in colorectal cancer. Br. J. Cancer 1995, 71, 1231–1236. [Google Scholar] [CrossRef] [PubMed]
- Brierley, J.D.; Gospodarowicz, M.K.; Wittekind, C. UICC TNM Classification of Malignant Tumors, 8th ed.; Wiley: New York, NY, USA, 2017. [Google Scholar]
- Sato, T.; Stange, D.E.; Ferrante, M.; Vries, R.G.J.; Es, J.H.V.; Brink, S.V.D.; Houdt, W.J.V.; Pronk, A.; Gorp, J.V.; Siersema, P.D.; et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett’s epithelium. Gastroenterology 2011, 141, 1762–1772. [Google Scholar] [CrossRef] [PubMed]
| N = 41 | |
|---|---|
| Age: years; median (range) | 67 (2–80) |
| Sex: Male/Female | 26/15 |
| CEA ng/mL: median (range) | 30 (2–2739) |
| CA19-9 U/mL: median (range) | 26.2 (0.4–4349.5) |
| Location: Right/Left | 8/33 |
| Stage II/III/IV | 1/2/38 |
| Primary tumor Max diameter mm; median (range) | 45 (20–140) |
| * Differentiation: tub/muc, por | 31/3 |
| Liver metastasis: yes/no | 31/10 |
| Lung metastasis: yes/no | 12/29 |
| Peritoneal dissemination: yes/no | 7/34 |
| Primary tumor resection: yes/no | 30/11 |
| Treatment experience of chemotherapy or radiation: yes/no | 11/30 |
| N = 41 | CTC (+) and/or Cell Growth (+), N = 23 | CTC (-) and Cell Growth (-) N = 18 | p Value | |
|---|---|---|---|---|
| Age: years; median (range) | 67 (25–80) | 67 (25–80) | 68 (43–80) | 0.64 |
| Sex: Male/Female | 26/15 | 15/8 | 11/7 | 0.79 |
| CEA ng/mL: median (range) | 30 (2–2739) | 21 (2–2739) | 33.5 (2–1956) | 0.89 |
| CA19-9 U/mL: median (range) | 26.2 (0.4–4349.5) | 26.2 (0.4–2870.2) | 26.55 (0.4–4339.5) | 0.82 |
| Location: Right/Left | 8/33 | 5/18 | 3/15 | 0.68 |
| * Differentiation: tub/muc, por | 31/3 | 17/3 | 14/0 | 0.07 |
| Primary tumor resection: yes/no | 30/11 | 17/6 | 13/5 | 0.90 |
| Chemotherapy or radiation: yes/no | 11/30 | 3/20 | 8/10 | 0.02 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nomura, M.; Yokoyama, Y.; Yoshimura, D.; Minagawa, Y.; Yamamoto, A.; Tanaka, Y.; Sekiguchi, N.; Marukawa, D.; Ichihara, M.; Itakura, H.; et al. Simple Detection and Culture of Circulating Tumor Cells from Colorectal Cancer Patients Using Poly(2-Methoxyethyl Acrylate)-Coated Plates. Int. J. Mol. Sci. 2023, 24, 3949. https://doi.org/10.3390/ijms24043949
Nomura M, Yokoyama Y, Yoshimura D, Minagawa Y, Yamamoto A, Tanaka Y, Sekiguchi N, Marukawa D, Ichihara M, Itakura H, et al. Simple Detection and Culture of Circulating Tumor Cells from Colorectal Cancer Patients Using Poly(2-Methoxyethyl Acrylate)-Coated Plates. International Journal of Molecular Sciences. 2023; 24(4):3949. https://doi.org/10.3390/ijms24043949
Chicago/Turabian StyleNomura, Masatoshi, Yuhki Yokoyama, Daishi Yoshimura, Yasuhisa Minagawa, Aki Yamamoto, Yukiko Tanaka, Naoko Sekiguchi, Daiki Marukawa, Momoko Ichihara, Hiroaki Itakura, and et al. 2023. "Simple Detection and Culture of Circulating Tumor Cells from Colorectal Cancer Patients Using Poly(2-Methoxyethyl Acrylate)-Coated Plates" International Journal of Molecular Sciences 24, no. 4: 3949. https://doi.org/10.3390/ijms24043949
APA StyleNomura, M., Yokoyama, Y., Yoshimura, D., Minagawa, Y., Yamamoto, A., Tanaka, Y., Sekiguchi, N., Marukawa, D., Ichihara, M., Itakura, H., Matsumoto, K., Morimoto, Y., Tomihara, H., Inoue, A., Ogino, T., Miyoshi, N., Takahashi, H., Takahashi, H., Uemura, M., ... Yamamoto, H. (2023). Simple Detection and Culture of Circulating Tumor Cells from Colorectal Cancer Patients Using Poly(2-Methoxyethyl Acrylate)-Coated Plates. International Journal of Molecular Sciences, 24(4), 3949. https://doi.org/10.3390/ijms24043949







