Expression of Selected miRNAs in Normal and Cancer-Associated Fibroblasts and in BxPc3 and MIA PaCa-2 Cell Lines of Pancreatic Ductal Adenocarcinoma
Abstract
1. Introduction
2. Results and Discussion
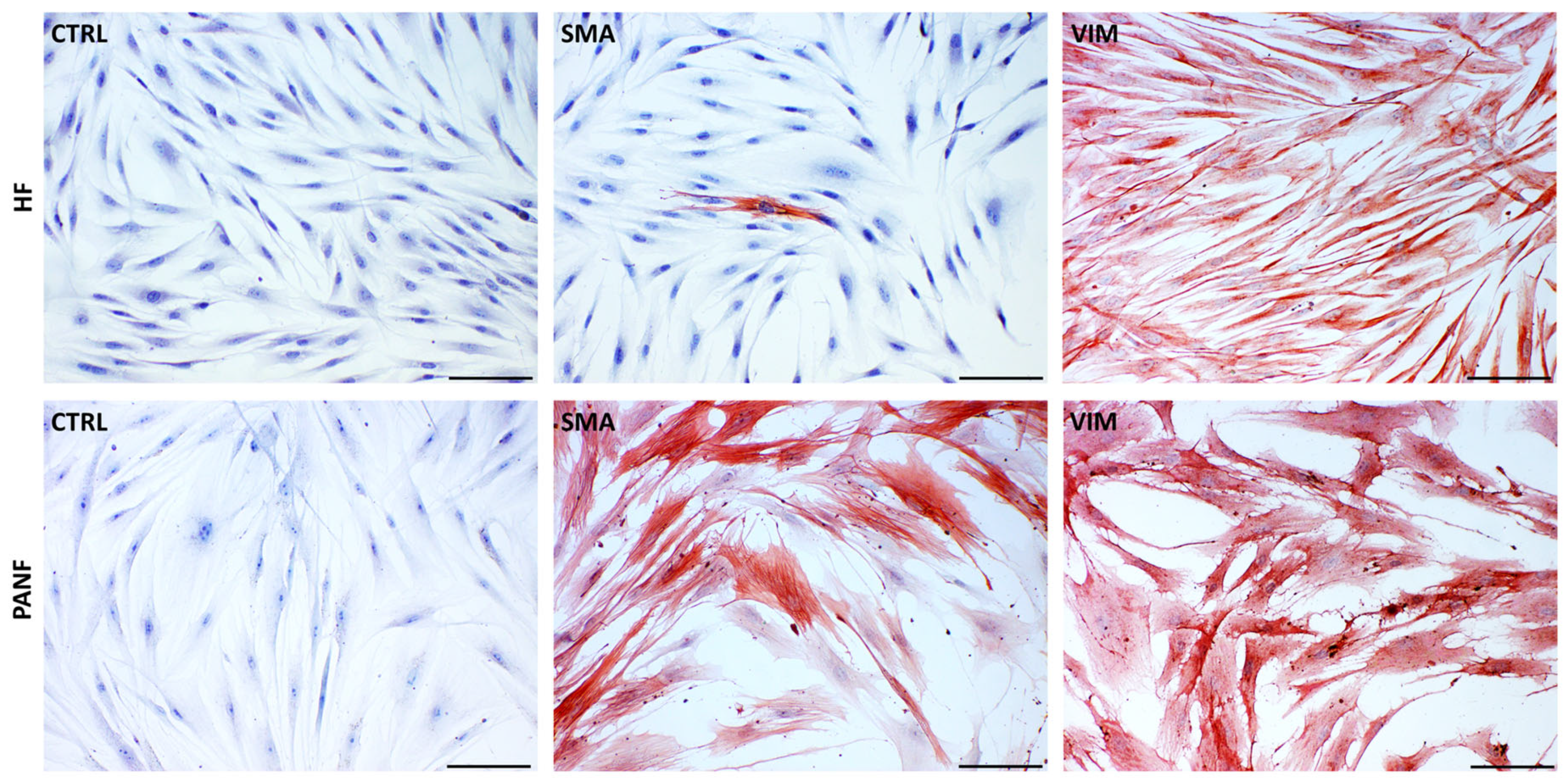
2.1. Immunocytochemical Characterisation of HFs and PANF
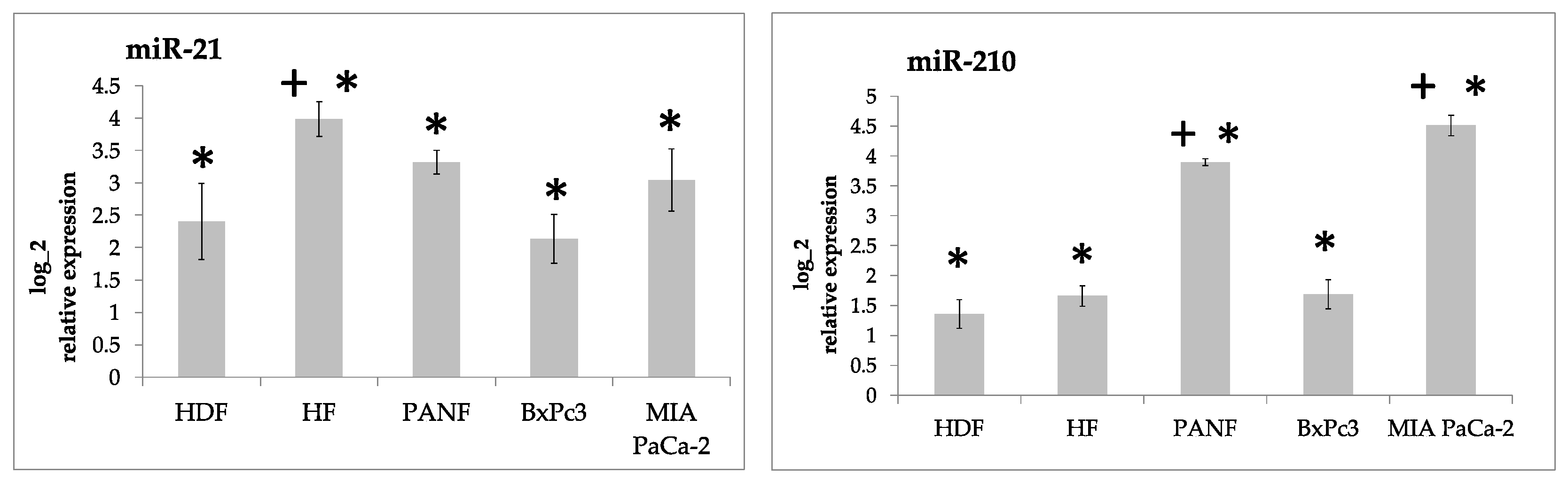
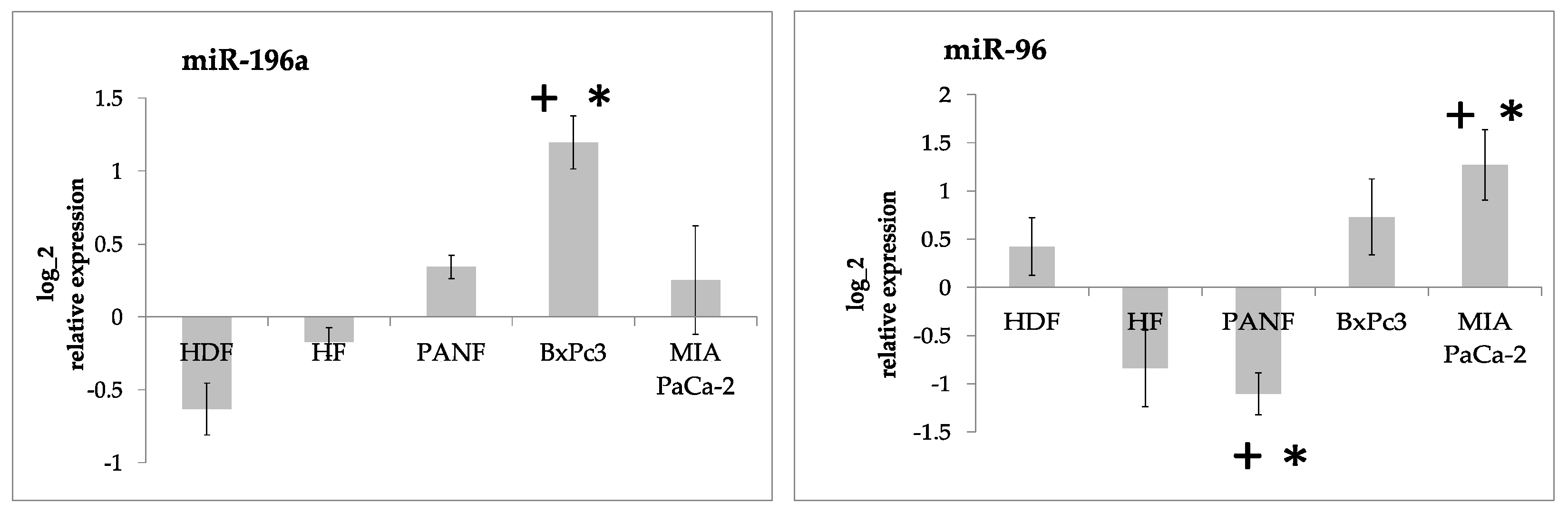
2.2. miRNA Expression
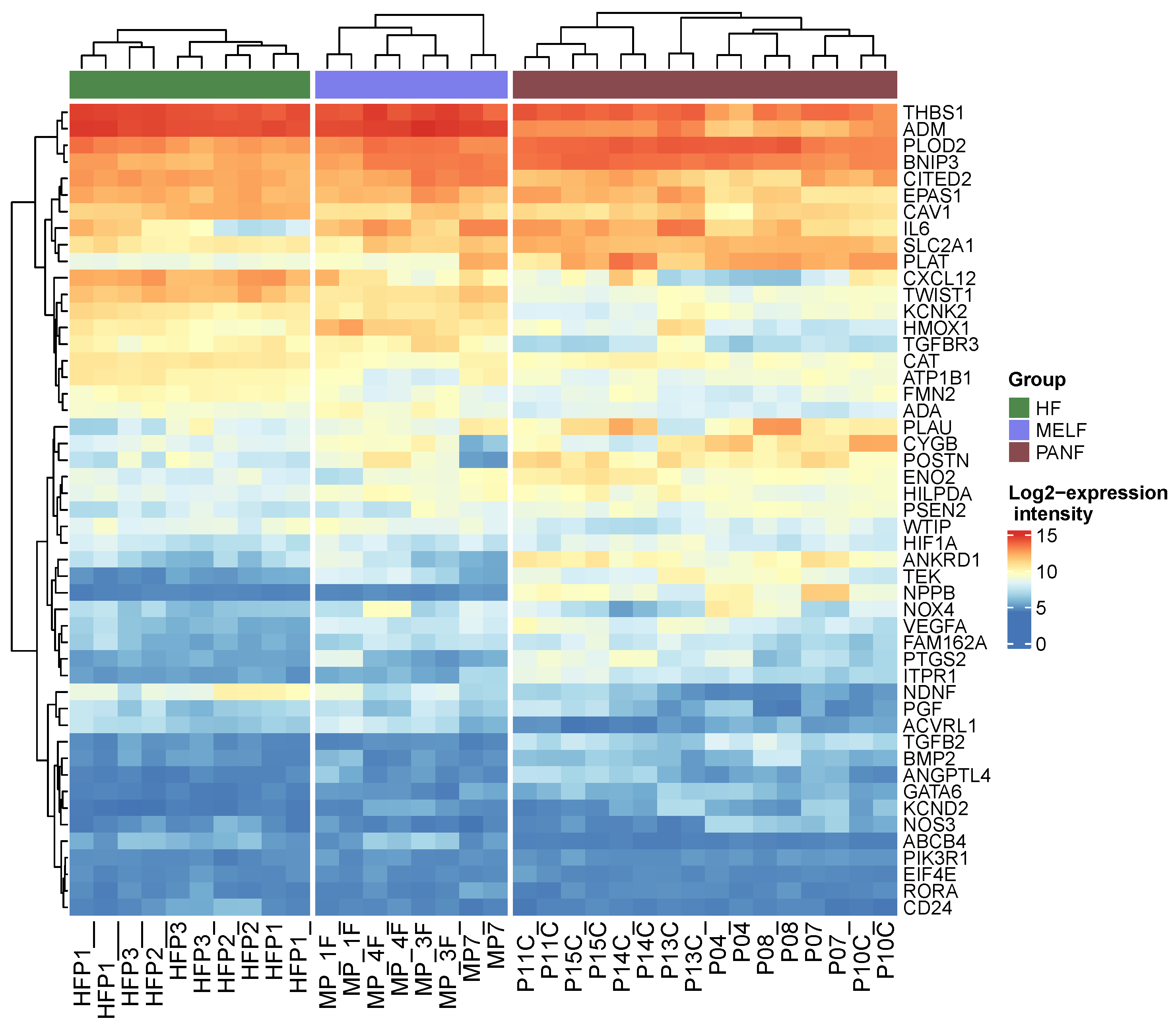
2.3. mRNA Expression in Normal Fibroblasts, CAFs, and PDAC Cell Lines
2.4. Cross-Talk of miR-21 and IL6 Signalling in Hypoxia
2.5. Consequences of Upregulation of miR-21 and miR-210 and Downregulation of miR-217 by Cancer Cells and CAFs Prepared from Pancreatic Cancer
3. Material and Methods
3.1. Cell Culture
3.2. Immunocytochemical Analysis
3.3. MicroRNA Isolation and Reverse Transcription
3.4. Real-Time qPCR
3.5. Comparison with the mRNA Expression Profile of PANF and CAFs from Melanoma (MELF) Based on [5]
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CAF | Cancer-associated fibroblast |
| CTRL | Negative controls |
| FDR | False discovery rate |
| GSEA | Gene set enrichment analysis |
| GO | The Gene Ontology |
| HDF | Human dermal fibroblast originating in ectomesenchyme derived from the neural crest |
| HF | Human dermal fibroblast originated from mesoderm |
| HIF-1α | Hypoxia-induced factor 1α |
| IL-6 | Interleukin 6 |
| LIF | Leukaemia inhibitory factor |
| MELF | CAF prepared from cutaneous malignant melanoma |
| miRNA | MicroRNA |
| miR-21 | MicroRNA 21 |
| mir-96 | MicroRNA 96 |
| mir-196a | MicroRNA 196a |
| mir-210 | MicroRNA 210 |
| mir-217 | MicroRNA 217 |
| MMP | Matrix metalloproteinase |
| PANF | CAF prepared from PDAC |
| PBS | Phosphate-buffered saline |
| PDAC | Pancreatic ductal adenocarcinoma |
| SMA | α-smooth muscle actin |
| VIM | Vimentin |
References
- Park, W.; Chawla, A.; O’Reilly, E.M. Pancreatic Cancer: A Review. JAMA 2021, 326, 851–862. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, A.; Popova, O.; Panchenkov, D.; Dyuzheva, T.; Ivanov, A. Pancreatic ductal adenocarcinoma: Tumor microenvironment and problems in the development of novel therapeutic strategies. Clin. Exp. Med. 2022. [Google Scholar] [CrossRef] [PubMed]
- Vokurka, M.; Lacina, L.; Brabek, J.; Kolar, M.; Ng, Y.Z.; Smetana, K., Jr. Cancer-Associated Fibroblasts Influence the Biological Properties of Malignant Tumours via Paracrine Secretion and Exosome Production. Int. J. Mol. Sci. 2022, 23, 964. [Google Scholar] [CrossRef] [PubMed]
- Podzolkov, V.I.; Pokrovskaya, A.E.; Vargina, T.S.; Ponomarev, A.B.; Kogan, Y.A.; Lerner, J.V. Anaplastic Carcinoma of the Pancreas: Clinical and Morphological Characteristics. Case Rep. Gastroenterol. 2020, 14, 624–631. [Google Scholar] [CrossRef]
- Novak, S.; Kolar, M.; Szabo, A.; Vernerova, Z.; Lacina, L.; Strnad, H.; Sachova, J.; Hradilova, M.; Havranek, J.; Spanko, M.; et al. Desmoplastic Crosstalk in Pancreatic Ductal Adenocarcinoma Is Reflected by Different Responses of Panc-1, MIAPaCa-2, PaTu-8902, and CAPAN-2 Cell Lines to Cancer-associated/Normal Fibroblasts. Cancer Genom. Proteom. 2021, 18, 221–243. [Google Scholar] [CrossRef]
- Chu, X.; Yang, Y.; Tian, X. Crosstalk between Pancreatic Cancer Cells and Cancer-Associated Fibroblasts in the Tumor Microenvironment Mediated by Exosomal MicroRNAs. Int. J. Mol. Sci. 2022, 23, 9512. [Google Scholar] [CrossRef]
- LaRue, M.M.; Parker, S.; Puccini, J.; Cammer, M.; Kimmelman, A.C.; Bar-Sagi, D. Metabolic reprogramming of tumor-associated macrophages by collagen turnover promotes fibrosis in pancreatic cancer. Proc. Natl. Acad. Sci. USA 2022, 119, e2119168119. [Google Scholar] [CrossRef]
- Dvorankova, B.; Szabo, P.; Lacina, L.; Kodet, O.; Matouskova, E.; Smetana, K., Jr. Fibroblasts prepared from different types of malignant tumors stimulate expression of luminal marker keratin 8 in the EM-G3 breast cancer cell line. Histochem. Cell Biol. 2012, 137, 679–685. [Google Scholar] [CrossRef]
- Kodet, O.; Kucera, J.; Strnadova, K.; Dvorankova, B.; Stork, J.; Lacina, L.; Smetana, K., Jr. Cutaneous melanoma dissemination is dependent on the malignant cell properties and factors of intercellular crosstalk in the cancer microenvironment (Review). Int. J. Oncol. 2020, 57, 619–630. [Google Scholar] [CrossRef]
- Strnadova, K.; Pfeiferova, L.; Prikryl, P.; Dvorankova, B.; Vlcak, E.; Frydlova, J.; Vokurka, M.; Novotny, J.; Sachova, J.; Hradilova, M.; et al. Exosomes produced by melanoma cells significantly influence the biological properties of normal and cancer-associated fibroblasts. Histochem. Cell Biol. 2022, 157, 153–172. [Google Scholar] [CrossRef]
- Carthew, R.W.; Sontheimer, E.J. Origins and Mechanisms of miRNAs and siRNAs. Cell 2009, 136, 642–655. [Google Scholar] [CrossRef] [PubMed]
- Menon, A.; Abd-Aziz, N.; Khalid, K.; Poh, C.L.; Naidu, R. miRNA: A Promising Therapeutic Target in Cancer. Int. J. Mol. Sci. 2022, 23, 11502. [Google Scholar] [CrossRef] [PubMed]
- Cheung, P.Y.; Szafranska-Schwarzbach, A.E.; Schlageter, A.M.; Andruss, B.F.; Weiss, G.J. No miR quirk: Dysregulation of microRNAs in pancreatic ductal adenocarcinoma. Microrna 2012, 1, 49–58. [Google Scholar] [CrossRef]
- Szabo, A.; Gurlich, R.; Liberko, M.; Soumarova, R.; Vernerova, Z.; Mandys, V.; Popov, A. Expression of selected microRNAs in pancreatic ductal adenocarcinoma: Is there a relation to tumor morphology, progression and patient’s outcome? Neoplasma 2020, 67, 1170–1181. [Google Scholar] [CrossRef] [PubMed]
- Popov, A.; Mandys, V. Senescence-Associated miRNAs and Their Role in Pancreatic Cancer. Pathol. Oncol. Res. 2022, 28, 1610156. [Google Scholar] [CrossRef]
- Greither, T.; Grochola, L.F.; Udelnow, A.; Lautenschlager, C.; Wurl, P.; Taubert, H. Elevated expression of microRNAs 155, 203, 210 and 222 in pancreatic tumors is associated with poorer survival. Int. J. Cancer 2010, 126, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Negoi, I.; Hostiuc, S.; Sartelli, M.; Negoi, R.I.; Beuran, M. MicroRNA-21 as a prognostic biomarker in patients with pancreatic cancer—A systematic review and meta-analysis. Am. J. Surg. 2017, 214, 515–524. [Google Scholar] [CrossRef]
- Tomasek, J.J.; Gabbiani, G.; Hinz, B.; Chaponnier, C.; Brown, R.A. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat. Rev. Mol. Cell Biol. 2002, 3, 349–363. [Google Scholar] [CrossRef]
- Zhao, B.; Baloch, Z.; Ma, Y.; Wan, Z.; Huo, Y.; Li, F.; Zhao, Y. Identification of Potential Key Genes and Pathways in Early-Onset Colorectal Cancer Through Bioinformatics Analysis. Cancer Control 2019, 26, 1073274819831260. [Google Scholar] [CrossRef]
- Creuzet, S.; Couly, G.; Le Douarin, N.M. Patterning the neural crest derivatives during development of the vertebrate head: Insights from avian studies. J. Anat. 2005, 207, 447–459. [Google Scholar] [CrossRef]
- LeBleu, V.S.; Neilson, E.G. Origin and functional heterogeneity of fibroblasts. FASEB J. 2020, 34, 3519–3536. [Google Scholar] [CrossRef] [PubMed]
- Forte, E.; Ramialison, M.; Nim, H.T.; Mara, M.; Li, J.Y.; Cohn, R.; Daigle, S.L.; Boyd, S.; Stanley, E.G.; Elefanty, A.G.; et al. Adult mouse fibroblasts retain organ-specific transcriptomic identity. Elife 2022, 11, e71008. [Google Scholar] [CrossRef] [PubMed]
- Bera, A.; VenkataSubbaRao, K.; Manoharan, M.S.; Hill, P.; Freeman, J.W. A miRNA signature of chemoresistant mesenchymal phenotype identifies novel molecular targets associated with advanced pancreatic cancer. PLoS ONE 2014, 9, e106343. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Ali, S.; Ahmad, A.; Azmi, A.S.; Li, Y.; Banerjee, S.; Kong, D.; Sethi, S.; Aboukameel, A.; Padhye, S.B.; et al. Hypoxia-induced aggressiveness of pancreatic cancer cells is due to increased expression of VEGF, IL-6 and miR-21, which can be attenuated by CDF treatment. PLoS ONE 2012, 7, e50165. [Google Scholar] [CrossRef]
- Bavelloni, A.; Ramazzotti, G.; Poli, A.; Piazzi, M.; Focaccia, E.; Blalock, W.; Faenza, I. MiRNA-210: A Current Overview. Anticancer Res. 2017, 37, 6511–6521. [Google Scholar] [CrossRef]
- Geismann, C.; Arlt, A. Coming in the Air: Hypoxia Meets Epigenetics in Pancreatic Cancer. Cells 2020, 9, 2353. [Google Scholar] [CrossRef]
- Narayanan, S.; Eliasson Angelstig, S.; Xu, C.; Grunler, J.; Zhao, A.; Zhu, W.; Xu Landen, N.; Stahle, M.; Zhang, J.; Ivan, M.; et al. HypoxamiR-210 accelerates wound healing in diabetic mice by improving cellular metabolism. Commun. Biol. 2020, 3, 768. [Google Scholar] [CrossRef]
- Chen, X.; Peng, Y.; Xue, H.; Liu, G.; Wang, N.; Shao, Z. MiR-21 regulating PVT1/PTEN/IL-17 axis towards the treatment of infectious diabetic wound healing by modified GO-derived biomaterial in mouse models. J. Nanobiotechnol. 2022, 20, 309. [Google Scholar] [CrossRef]
- Ali, S.; Suresh, R.; Banerjee, S.; Bao, B.; Xu, Z.; Wilson, J.; Philip, P.A.; Apte, M.; Sarkar, F.H. Contribution of microRNAs in understanding the pancreatic tumor microenvironment involving cancer associated stellate and fibroblast cells. Am. J. Cancer Res. 2015, 5, 1251–1264. [Google Scholar]
- Xie, J.; Wu, W.; Zheng, L.; Lin, X.; Tai, Y.; Wang, Y.; Wang, L. Roles of MicroRNA-21 in Skin Wound Healing: A Comprehensive Review. Front. Pharmacol. 2022, 13, 828627. [Google Scholar] [CrossRef]
- Gal, P.; Varinska, L.; Faber, L.; Novak, S.; Szabo, P.; Mitrengova, P.; Mirossay, A.; Mucaji, P.; Smetana, K. How Signaling Molecules Regulate Tumor Microenvironment: Parallels to Wound Repair. Molecules 2017, 22, 1818. [Google Scholar] [CrossRef] [PubMed]
- Gal, P.; Brabek, J.; Holub, M.; Jakubek, M.; Sedo, A.; Lacina, L.; Strnadova, K.; Dubovy, P.; Hornychova, H.; Ryska, A.; et al. Autoimmunity, cancer and COVID-19 abnormally activate wound healing pathways: Critical role of inflammation. Histochem. Cell Biol. 2022, 158, 415–434. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Du, R.; Xiao, X.; Deng, Z.L.; Jian, D.; Xie, H.F.; Li, J. Microrna-217 modulates human skin fibroblast senescence by directly targeting DNA methyltransferase 1. Oncotarget 2017, 8, 33475–33486. [Google Scholar] [CrossRef] [PubMed]
- Chang, X.; Yu, C.; Li, J.; Yu, S.; Chen, J. hsa-miR-96 and hsa-miR-217 Expression Down-Regulates with Increasing Dysplasia in Pancreatic Intraepithelial Neoplasias and Intraductal Papillary Mucinous Neoplasms. Int. J. Med. Sci. 2017, 14, 412–418. [Google Scholar] [CrossRef][Green Version]
- Zhao, W.G.; Yu, S.N.; Lu, Z.H.; Ma, Y.H.; Gu, Y.M.; Chen, J. The miR-217 microRNA functions as a potential tumor suppressor in pancreatic ductal adenocarcinoma by targeting KRAS. Carcinogenesis 2010, 31, 1726–1733. [Google Scholar] [CrossRef]
- Prinz, C.; Fehring, L.; Frese, R. MicroRNAs as Indicators of Malignancy in Pancreatic Ductal Adenocarcinoma (PDAC) and Cystic Pancreatic Lesions. Cells 2022, 11, 2374. [Google Scholar] [CrossRef]
- Rahimi, H.R.; Mojarrad, M.; Moghbeli, M. MicroRNA-96: A therapeutic and diagnostic tumor marker. Iran J. Basic Med. Sci. 2022, 25, 3–13. [Google Scholar] [CrossRef]
- Cui, X.; Churchill, G.A. Statistical tests for differential expression in cDNA microarray experiments. Genome Biol. 2003, 4, 210. [Google Scholar] [CrossRef]
- Paczkowska, J.; Giefing, M. MicroRNA signature in classical Hodgkin lymphoma. J. Appl. Genet. 2021, 62, 281–288. [Google Scholar] [CrossRef]
- Chen, Z.Y.; Chen, X.; Wang, Z.X. The role of microRNA-196a in tumorigenesis, tumor progression, and prognosis. Tumour Biol. 2016. [Google Scholar] [CrossRef]
- Chen, W.Y.; Liu, W.J.; Zhao, Y.P.; Zhou, L.; Zhang, T.P.; Chen, G.; Shu, H. Induction, modulation and potential targets of miR-210 in pancreatic cancer cells. Hepatobiliary Pancreat. Dis. Int. 2012, 11, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Sabry, D.; El-Deek, S.E.M.; Maher, M.; El-Baz, M.A.H.; El-Bader, H.M.; Amer, E.; Hassan, E.A.; Fathy, W.; El-Deek, H.E.M. Role of miRNA-210, miRNA-21 and miRNA-126 as diagnostic biomarkers in colorectal carcinoma: Impact of HIF-1alpha-VEGF signaling pathway. Mol. Cell Biochem. 2019, 454, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Zhang, Y.; Li, B.; Yang, H. MTA1 promotes the invasion and migration of pancreatic cancer cells potentially through the HIF-alpha/VEGF pathway. J. Recept. Signal Transduct. Res. 2018, 38, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Juhasz, M.; Chen, J.; Lendeckel, U.; Kellner, U.; Kasper, H.U.; Tulassay, Z.; Pastorekova, S.; Malfertheiner, P.; Ebert, M.P. Expression of carbonic anhydrase IX in human pancreatic cancer. Aliment. Pharmacol. Ther. 2003, 18, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Neal, C.S.; Michael, M.Z.; Rawlings, L.H.; Van der Hoek, M.B.; Gleadle, J.M. The VHL-dependent regulation of microRNAs in renal cancer. BMC Med. 2010, 8, 64. [Google Scholar] [CrossRef]
- Nijsten, T.; Colpaert, C.G.; Vermeulen, P.B.; Harris, A.L.; Van Marck, E.; Lambert, J. Cyclooxygenase-2 expression and angiogenesis in squamous cell carcinoma of the skin and its precursors: A paired immunohistochemical study of 35 cases. Br. J. Dermatol. 2004, 151, 837–845. [Google Scholar] [CrossRef]
- Lee, S.; Jiang, X. Modeling miRNA-mRNA interactions that cause phenotypic abnormality in breast cancer patients. PLoS ONE 2017, 12, e0182666. [Google Scholar] [CrossRef]
- Noman, M.Z.; Buart, S.; Romero, P.; Ketari, S.; Janji, B.; Mari, B.; Mami-Chouaib, F.; Chouaib, S. Hypoxia-inducible miR-210 regulates the susceptibility of tumor cells to lysis by cytotoxic T cells. Cancer Res. 2012, 72, 4629–4641. [Google Scholar] [CrossRef]
- Luan, Y.; Zhang, X.; Zhang, Y.; Dong, Y. MicroRNA-210 Protects PC-12 Cells Against Hypoxia-Induced Injury by Targeting BNIP3. Front. Cell Neurosci. 2017, 11, 285. [Google Scholar] [CrossRef]
- Chen, Q.; Xie, X. Association of Exosomal miR-210 with Signaling Pathways Implicated in Lung Cancer. Genes 2021, 12, 1248. [Google Scholar] [CrossRef]
- Tarhini, A.A.; Lin, Y.; Yeku, O.; LaFramboise, W.A.; Ashraf, M.; Sander, C.; Lee, S.; Kirkwood, J.M. A four-marker signature of TNF-RII, TGF-alpha, TIMP-1 and CRP is prognostic of worse survival in high-risk surgically resected melanoma. J. Transl. Med. 2014, 12, 19. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.H.; Yue, J.; Pfeffer, S.R.; Fan, M.; Paulus, E.; Hosni-Ahmed, A.; Sims, M.; Qayyum, S.; Davidoff, A.M.; Handorf, C.R.; et al. MicroRNA-21 promotes glioblastoma tumorigenesis by down-regulating insulin-like growth factor-binding protein-3 (IGFBP3). J. Biol. Chem. 2014, 289, 25079–25087. [Google Scholar] [CrossRef] [PubMed]
- Lacina, L.; Kodet, O.; Dvorankova, B.; Szabo, P.; Smetana, K., Jr. Ecology of melanoma cell. Histol. Histopathol. 2018, 33, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Cancer Genome Atlas Research Network. Integrated Genomic Characterization of Pancreatic Ductal Adenocarcinoma. Cancer Cell 2017, 32, 185–203.e113. [Google Scholar] [CrossRef] [PubMed]
- Donahue, T.R.; Nguyen, A.H.; Moughan, J.; Li, L.; Tatishchev, S.; Toste, P.; Farrell, J.J. Stromal microRNA-21 levels predict response to 5-fluorouracil in patients with pancreatic cancer. J. Surg. Oncol. 2014, 110, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Chen, X.; Shan, T.; Ma, J.; Lin, W.; Li, W.; Kang, Y. MiR-21-mediated Metabolic Alteration of Cancer-associated Fibroblasts and Its Effect on Pancreatic Cancer Cell Behavior. Int. J. Biol. Sci. 2018, 14, 100–110. [Google Scholar] [CrossRef]
- Zhang, L.; Yao, J.; Li, W.; Zhang, C. Micro-RNA-21 Regulates Cancer-Associated Fibroblast-Mediated Drug Resistance in Pancreatic Cancer. Oncol. Res. 2018, 26, 827–835. [Google Scholar] [CrossRef]
- Savardashtaki, A.; Shabaninejad, Z.; Movahedpour, A.; Sahebnasagh, R.; Mirzaei, H.; Hamblin, M.R. miRNAs derived from cancer-associated fibroblasts in colorectal cancer. Epigenomics 2019, 11, 1627–1645. [Google Scholar] [CrossRef]
- Richards, K.E.; Xiao, W.; Hill, R.; On Behalf Of The Usc Pancreas Research, T. Cancer-Associated Fibroblasts Confer Gemcitabine Resistance to Pancreatic Cancer Cells through PTEN-Targeting miRNAs in Exosomes. Cancers 2022, 14, 2812. [Google Scholar] [CrossRef]
- Frampton, A.E.; Krell, J.; Jamieson, N.B.; Gall, T.M.; Giovannetti, E.; Funel, N.; Mato Prado, M.; Krell, D.; Habib, N.A.; Castellano, L.; et al. microRNAs with prognostic significance in pancreatic ductal adenocarcinoma: A meta-analysis. Eur. J. Cancer 2015, 51, 1389–1404. [Google Scholar] [CrossRef]
- Steele, C.W.; Oien, K.A.; McKay, C.J.; Jamieson, N.B. Clinical potential of microRNAs in pancreatic ductal adenocarcinoma. Pancreas 2011, 40, 1165–1171. [Google Scholar] [CrossRef]
- Hernandez, Y.G.; Lucas, A.L. MicroRNA in pancreatic ductal adenocarcinoma and its precursor lesions. World J. Gastrointest. Oncol. 2016, 8, 18–29. [Google Scholar] [CrossRef]
- Takikawa, T.; Masamune, A.; Hamada, S.; Nakano, E.; Yoshida, N.; Shimosegawa, T. miR-210 regulates the interaction between pancreatic cancer cells and stellate cells. Biochem. Biophys. Res. Commun. 2013, 437, 433–439. [Google Scholar] [CrossRef]
- Nielsen, B.S.; Jorgensen, S.; Fog, J.U.; Sokilde, R.; Christensen, I.J.; Hansen, U.; Brunner, N.; Baker, A.; Moller, S.; Nielsen, H.J. High levels of microRNA-21 in the stroma of colorectal cancers predict short disease-free survival in stage II colon cancer patients. Clin. Exp. Metastasis 2011, 28, 27–38. [Google Scholar] [CrossRef]
- Lee, K.S.; Nam, S.K.; Koh, J.; Kim, D.W.; Kang, S.B.; Choe, G.; Kim, W.H.; Lee, H.S. Stromal Expression of MicroRNA-21 in Advanced Colorectal Cancer Patients with Distant Metastases. J. Pathol. Transl. Med. 2016, 50, 270–277. [Google Scholar] [CrossRef]
- Bhome, R.; Goh, R.W.; Bullock, M.D.; Pillar, N.; Thirdborough, S.M.; Mellone, M.; Mirnezami, R.; Galea, D.; Veselkov, K.; Gu, Q.; et al. Exosomal microRNAs derived from colorectal cancer-associated fibroblasts: Role in driving cancer progression. Aging 2017, 9, 2666–2694. [Google Scholar] [CrossRef]
- Yang, Y.; Gu, J.; Li, X.; Xue, C.; Ba, L.; Gao, Y.; Zhou, J.; Bai, C.; Sun, Z.; Zhao, R.C. HIF-1alpha promotes the migration and invasion of cancer-associated fibroblasts by miR-210. Aging Dis. 2021, 12, 1794–1807. [Google Scholar] [CrossRef] [PubMed]
- Kunita, A.; Morita, S.; Irisa, T.U.; Goto, A.; Niki, T.; Takai, D.; Nakajima, J.; Fukayama, M. MicroRNA-21 in cancer-associated fibroblasts supports lung adenocarcinoma progression. Sci. Rep. 2018, 8, 8838. [Google Scholar] [CrossRef] [PubMed]
- Marin, I.; Ofek, E.; Bar, J.; Prisant, N.; Perelman, M.; Avivi, C.; Lavy-Shahaf, G.; Onn, A.; Katz, R.; Barshack, I. MiR-21, EGFR and PTEN in non-small cell lung cancer: An in situ hybridisation and immunohistochemistry study. J. Clin. Pathol. 2020, 73, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Xu, G.; Chang, Z.; Zhu, L.; Yao, J. miR-210 transferred by lung cancer cell-derived exosomes may act as proangiogenic factor in cancer-associated fibroblasts by modulating JAK2/STAT3 pathway. Clin. Sci. 2020, 134, 807–825. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Yan, Y.; Yang, Y.; Hong, X.; Wang, M.; Yang, Z.; Liu, B.; Ye, L. MiR-210 in exosomes derived from CAFs promotes non-small cell lung cancer migration and invasion through PTEN/PI3K/AKT pathway. Cell Signal 2020, 73, 109675. [Google Scholar] [CrossRef] [PubMed]
- Rask, L.; Balslev, E.; Jorgensen, S.; Eriksen, J.; Flyger, H.; Moller, S.; Hogdall, E.; Litman, T.; Nielsen, B.S. High expression of miR-21 in tumor stroma correlates with increased cancer cell proliferation in human breast cancer. APMIS 2011, 119, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Tejchman, A.; Lamerant-Fayel, N.; Jacquinet, J.C.; Bielawska-Pohl, A.; Mleczko-Sanecka, K.; Grillon, C.; Chouaib, S.; Ugorski, M.; Kieda, C. Tumor hypoxia modulates podoplanin/CCL21 interactions in CCR7+ NK cell recruitment and CCR7+ tumor cell mobilization. Oncotarget 2017, 8, 31876–31887. [Google Scholar] [CrossRef] [PubMed]
- Uozaki, H.; Morita, S.; Kumagai, A.; Aso, T.; Soejima, Y.; Takahashi, Y.; Fukusato, T. Stromal miR-21 is more important than miR-21 of tumour cells for the progression of gastric cancer. Histopathology 2014, 65, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Rosenberg, A.Z.; Choi, S.M.; Fox-Talbot, K.; De Marzo, A.M.; Nonn, L.; Brennen, W.N.; Marchionni, L.; Halushka, M.K.; Lupold, S.E. Cell-type specific expression of oncogenic and tumor suppressive microRNAs in the human prostate and prostate cancer. Sci. Rep. 2018, 8, 7189. [Google Scholar] [CrossRef]
- Taddei, M.L.; Cavallini, L.; Comito, G.; Giannoni, E.; Folini, M.; Marini, A.; Gandellini, P.; Morandi, A.; Pintus, G.; Raspollini, M.R.; et al. Senescent stroma promotes prostate cancer progression: The role of miR-210. Mol. Oncol. 2014, 8, 1729–1746. [Google Scholar] [CrossRef]
- Andersen, S.; Richardsen, E.; Moi, L.; Donnem, T.; Nordby, Y.; Ness, N.; Holman, M.E.; Bremnes, R.M.; Busund, L.T. Fibroblast miR-210 overexpression is independently associated with clinical failure in Prostate Cancer—A multicenter (in situ hybridisation) study. Sci. Rep. 2016, 6, 36573. [Google Scholar] [CrossRef]
- Aubert, S.; Berdelou, A.; Gnemmi, V.; Behal, H.; Caiazzo, R.; D’Herbomez, M.; Pigny, P.; Wemeau, J.L.; Carnaille, B.; Renaud, F.; et al. Large sporadic thyroid medullary carcinomas: Predictive factors for lymph node involvement. Virchows Arch 2018, 472, 461–468. [Google Scholar] [CrossRef]
- Wang, C.; Wang, Y.; Chang, X.; Ba, X.; Hu, N.; Liu, Q.; Fang, L.; Wang, Z. Melanoma-Derived Exosomes Endow Fibroblasts with an Invasive Potential via miR-21 Target Signaling Pathway. Cancer Manag. Res. 2020, 12, 12965–12974. [Google Scholar] [CrossRef]
- Brabek, J.; Jakubek, M.; Vellieux, F.; Novotny, J.; Kolar, M.; Lacina, L.; Szabo, P.; Strnadova, K.; Rosel, D.; Dvorankova, B.; et al. Interleukin-6: Molecule in the Intersection of Cancer, Ageing and COVID-19. Int. J. Mol. Sci. 2020, 21, 7937. [Google Scholar] [CrossRef]
- Spanko, M.; Strnadova, K.; Pavlicek, A.J.; Szabo, P.; Kodet, O.; Valach, J.; Dvorankova, B.; Smetana, K., Jr.; Lacina, L. IL-6 in the Ecosystem of Head and Neck Cancer: Possible Therapeutic Perspectives. Int. J. Mol. Sci. 2021, 22, 11027. [Google Scholar] [CrossRef] [PubMed]
- Bumrungthai, S.; Ekalaksananan, T.; Evans, M.F.; Chopjitt, P.; Tangsiriwatthana, T.; Patarapadungkit, N.; Kleebkaow, P.; Luanratanakorn, S.; Kongyingyoes, B.; Worawichawong, S.; et al. Up-Regulation of miR-21 Is Associated with Cervicitis and Human Papillomavirus Infection in Cervical Tissues. PLoS ONE 2015, 10, e0127109. [Google Scholar] [CrossRef]
- Waster, P.; Eriksson, I.; Vainikka, L.; Ollinger, K. Extracellular vesicles released by melanocytes after UVA irradiation promote intercellular signaling via miR21. Pigment. Cell Melanoma Res. 2020, 33, 542–555. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gutierrez, A.; Carbajal-Lopez, B.; Bui, T.M.; Mendoza-Rodriguez, M.; Campos-Parra, A.D.; Calderillo-Ruiz, G.; Cantu-De Leon, D.; Madrigal-Santillan, E.O.; Sumagin, R.; Perez-Plasencia, C.; et al. A microRNA panel that regulates proinflammatory cytokines as diagnostic and prognosis biomarkers in colon cancer. Biochem. Biophys. Rep. 2022, 30, 101252. [Google Scholar] [CrossRef] [PubMed]
- Lages, E.; Guttin, A.; El Atifi, M.; Ramus, C.; Ipas, H.; Dupre, I.; Rolland, D.; Salon, C.; Godfraind, C.; de Fraipont, F.; et al. MicroRNA and target protein patterns reveal physiopathological features of glioma subtypes. PLoS ONE 2011, 6, e20600. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Ren, Y.; Liu, A.; Han, L.; Zhang, K.; Li, S.; Li, P.; Li, P.; Kang, C.; Wang, X.; et al. STAT3 inhibitor WP1066 attenuates miRNA-21 to suppress human oral squamous cell carcinoma growth in vitro and in vivo. Oncol. Rep. 2014, 31, 2173–2180. [Google Scholar] [CrossRef]
- Ou, H.; Li, Y.; Kang, M. Activation of miR-21 by STAT3 induces proliferation and suppresses apoptosis in nasopharyngeal carcinoma by targeting PTEN gene. PLoS ONE 2014, 9, e109929. [Google Scholar] [CrossRef]
- Lu, X.; Luo, F.; Liu, Y.; Zhang, A.; Li, J.; Wang, B.; Xu, W.; Shi, L.; Liu, X.; Lu, L.; et al. The IL-6/STAT3 pathway via miR-21 is involved in the neoplastic and metastatic properties of arsenite-transformed human keratinocytes. Toxicol. Lett. 2015, 237, 191–199. [Google Scholar] [CrossRef]
- Lai, C.Y.; Yeh, K.Y.; Liu, B.F.; Chang, T.M.; Chang, C.H.; Liao, Y.F.; Liu, Y.W.; Her, G.M. MicroRNA-21 Plays Multiple Oncometabolic Roles in Colitis-Associated Carcinoma and Colorectal Cancer via the PI3K/AKT, STAT3, and PDCD4/TNF-alpha Signaling Pathways in Zebrafish. Cancers 2021, 13, 5565. [Google Scholar] [CrossRef]
- Zhao, Q.; Huang, L.; Qin, G.; Qiao, Y.; Ren, F.; Shen, C.; Wang, S.; Liu, S.; Lian, J.; Wang, D.; et al. Cancer-associated fibroblasts induce monocytic myeloid-derived suppressor cell generation via IL-6/exosomal miR-21-activated STAT3 signaling to promote cisplatin resistance in esophageal squamous cell carcinoma. Cancer Lett. 2021, 518, 35–48. [Google Scholar] [CrossRef]
- Rose-John, S. IL-6 trans-signaling via the soluble IL-6 receptor: Importance for the pro-inflammatory activities of IL-6. Int. J. Biol. Sci. 2012, 8, 1237–1247. [Google Scholar] [CrossRef] [PubMed]
- Kamohara, H.; Ogawa, M.; Ishiko, T.; Sakamoto, K.; Baba, H. Leukemia inhibitory factor functions as a growth factor in pancreas carcinoma cells: Involvement of regulation of LIF and its receptor expression. Int. J. Oncol. 2007, 30, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Di Giorgio, C.; Lupia, A.; Marchiano, S.; Bordoni, M.; Bellini, R.; Massa, C.; Urbani, G.; Roselli, R.; Moraca, F.; Sepe, V.; et al. Repositioning Mifepristone as a Leukaemia Inhibitory Factor Receptor Antagonist for the Treatment of Pancreatic Adenocarcinoma. Cells 2022, 11, 3482. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.; Zhao, Y.; Zhang, C.; Li, J.; Liu, Z.; Liu, J.; Hu, W. Leukemia inhibitory factor promotes EMT through STAT3-dependent miR-21 induction. Oncotarget 2016, 7, 3777–3790. [Google Scholar] [CrossRef]
- Kuphal, S.; Wallner, S.; Bosserhoff, A.K. Impact of LIF (leukemia inhibitory factor) expression in malignant melanoma. Exp. Mol. Pathol. 2013, 95, 156–165. [Google Scholar] [CrossRef]
- Vendrell-Flotats, M.; Garcia-Martinez, T.; Martinez-Rodero, I.; Lopez-Bejar, M.; LaMarre, J.; Yeste, M.; Mogas, T. In vitro maturation in the presence of Leukemia Inhibitory Factor modulates gene and miRNA expression in bovine oocytes and embryos. Sci. Rep. 2020, 10, 17777. [Google Scholar] [CrossRef]
- Tscherner, A.; Brown, A.C.; Stalker, L.; Kao, J.; Dufort, I.; Sirard, M.A.; LaMarre, J. STAT3 signaling stimulates miR-21 expression in bovine cumulus cells during in vitro oocyte maturation. Sci. Rep. 2018, 8, 11527. [Google Scholar] [CrossRef]
- Morales-Prieto, D.M.; Barth, E.; Murrieta-Coxca, J.M.; Favaro, R.R.; Gutierrez-Samudio, R.N.; Chaiwangyen, W.; Ospina-Prieto, S.; Gruhn, B.; Schleussner, E.; Marz, M.; et al. Identification of miRNAs and associated pathways regulated by Leukemia Inhibitory Factor in trophoblastic cell lines. Placenta 2019, 88, 20–27. [Google Scholar] [CrossRef]
- Hu, B.; Yang, X.B.; Sang, X.T. Development and Verification of the Hypoxia-Related and Immune-Associated Prognosis Signature for Hepatocellular Carcinoma. J. Hepatocell. Carcinoma 2020, 7, 315–330. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, S.; Zhang, X.; Wu, Z.; Li, Z.; Ding, Z.; Huang, X.; Chen, S.; Jing, Y.; Zhang, X.; et al. Identification and Validation of PLOD2 as an Adverse Prognostic Biomarker for Oral Squamous Cell Carcinoma. Biomolecules 2021, 11, 1842. [Google Scholar] [CrossRef]
- Sohrabi, E.; Rezaie, E.; Heiat, M.; Sefidi-Heris, Y. An Integrated Data Analysis of mRNA, miRNA and Signaling Pathways in Pancreatic Cancer. Biochem. Genet. 2021, 59, 1326–1358. [Google Scholar] [CrossRef] [PubMed]
- Bi, L.; Huang, Y.; Li, J.; Yang, X.; Hou, G.; Zhai, P.; Zhang, Q.; Alhaji, A.A.; Yang, Y.; Liu, B. Pirfenidone Attenuates Renal Tubulointerstitial Fibrosis through Inhibiting miR-21. Nephron 2022, 146, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhou, H.; Cheng, F.; Zhang, Z.; Long, S. MiR-21 regulates epithelial-mesenchymal transition in intestinal fibrosis of Crohn’s disease by targeting PTEN/mTOR. Dig. Liver Dis. 2022, 54, 1358–1366. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Tang, Z.; Ren, J.; Geng, J.; Guo, F.; Xu, Z.; Jia, J.; Chen, L.; Jia, Y. Downregulation of microRNA-21 contributes to decreased collagen expression in venous malformations via transforming growth factor-beta/Smad3/microRNA-21 signaling feedback loop. J. Vasc. Surg. Venous Lymphat. Disord. 2022, 10, 469–481.e462. [Google Scholar] [CrossRef]
- Li, Z.; Meng, D.; Li, G.; Xu, J.; Tian, K.; Li, Y. Overexpression of microRNA-210 promotes chondrocyte proliferation and extracellular matrix deposition by targeting HIF-3alpha in osteoarthritis. Mol. Med. Rep. 2016, 13, 2769–2776. [Google Scholar] [CrossRef]
- Morimoto, C.; Takedachi, M.; Kawasaki, K.; Shimomura, J.; Murata, M.; Hirai, A.; Kawakami, K.; Sawada, K.; Iwayama, T.; Murakami, S. Hypoxia stimulates collagen hydroxylation in gingival fibroblasts and periodontal ligament cells. J. Periodontol. 2021, 92, 1635–1645. [Google Scholar] [CrossRef]
- Yu, Q.; Xu, C.; Yuan, W.; Wang, C.; Zhao, P.; Chen, L.; Ma, J. Evaluation of Plasma MicroRNAs as Diagnostic and Prognostic Biomarkers in Pancreatic Adenocarcinoma: miR-196a and miR-210 Could Be Negative and Positive Prognostic Markers, Respectively. Biomed. Res. Int. 2017, 2017, 6495867. [Google Scholar] [CrossRef]
- Vychytilova-Faltejskova, P.; Kiss, I.; Klusova, S.; Hlavsa, J.; Prochazka, V.; Kala, Z.; Mazanec, J.; Hausnerova, J.; Kren, L.; Hermanova, M.; et al. MiR-21, miR-34a, miR-198 and miR-217 as diagnostic and prognostic biomarkers for chronic pancreatitis and pancreatic ductal adenocarcinoma. Diagn Pathol. 2015, 10, 38. [Google Scholar] [CrossRef]
- Guz, M.; Jeleniewicz, W.; Cybulski, M.; Kozicka, J.; Kurzepa, J.; Madro, A. Serum miR-210-3p can be used to differentiate between patients with pancreatic ductal adenocarcinoma and chronic pancreatitis. Biomed. Rep. 2021, 14, 10. [Google Scholar] [CrossRef]
- Dvorankova, B.; Lacina, L.; Smetana, K., Jr. Isolation of Normal Fibroblasts and Their Cancer-Associated Counterparts (CAFs) for Biomedical Research. Methods Mol. Biol. 2019, 1879, 393–406. [Google Scholar] [CrossRef]
- Shrestha, B.; Dunn, L. The Declaration of Helsinki on Medical Research involving Human Subjects: A Review of Seventh Revision. J. Nepal. Health Res. Counc. 2020, 17, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Mifkova, A.; Kodet, O.; Szabo, P.; Kucera, J.; Dvorankova, B.; Andre, S.; Koripelly, G.; Gabius, H.J.; Lehn, J.M.; Smetana, K., Jr. Synthetic polyamine BPA-C8 inhibits TGF-beta1-mediated conversion of human dermal fibroblast to myofibroblasts and establishment of galectin-1-rich extracellular matrix in vitro. Chembiochem 2014, 15, 1465–1470. [Google Scholar] [CrossRef] [PubMed]
- Szafranska, A.E.; Davison, T.S.; Shingara, J.; Doleshal, M.; Riggenbach, J.A.; Morrison, C.D.; Jewell, S.; Labourier, E. Accurate molecular characterisation of formalin-fixed, paraffin-embedded tissues by microRNA expression profiling. J. Mol. Diagn. 2008, 10, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Doleshal, M.; Magotra, A.A.; Choudhury, B.; Cannon, B.D.; Labourier, E.; Szafranska, A.E. Evaluation and validation of total RNA extraction methods for microRNA expression analyses in formalin-fixed, paraffin-embedded tissues. J. Mol. Diagn. 2008, 10, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Xi, Y.; Nakajima, G.; Gavin, E.; Morris, C.G.; Kudo, K.; Hayashi, K.; Ju, J. Systematic analysis of microRNA expression of RNA extracted from fresh frozen and formalin-fixed paraffin-embedded samples. RNA 2007, 13, 1668–1674. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Carvalho, B.S.; Irizarry, R.A. A framework for oligonucleotide microarray preprocessing. Bioinformatics 2010, 26, 2363–2367. [Google Scholar] [CrossRef]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Gentleman, R.C.; Carey, V.J.; Bates, D.M.; Bolstad, B.; Dettling, M.; Dudoit, S.; Ellis, B.; Gautier, L.; Ge, Y.; Gentry, J.; et al. Bioconductor: Open software development for computational biology and bioinformatics. Genome Biol. 2004, 5, R80. [Google Scholar] [CrossRef]

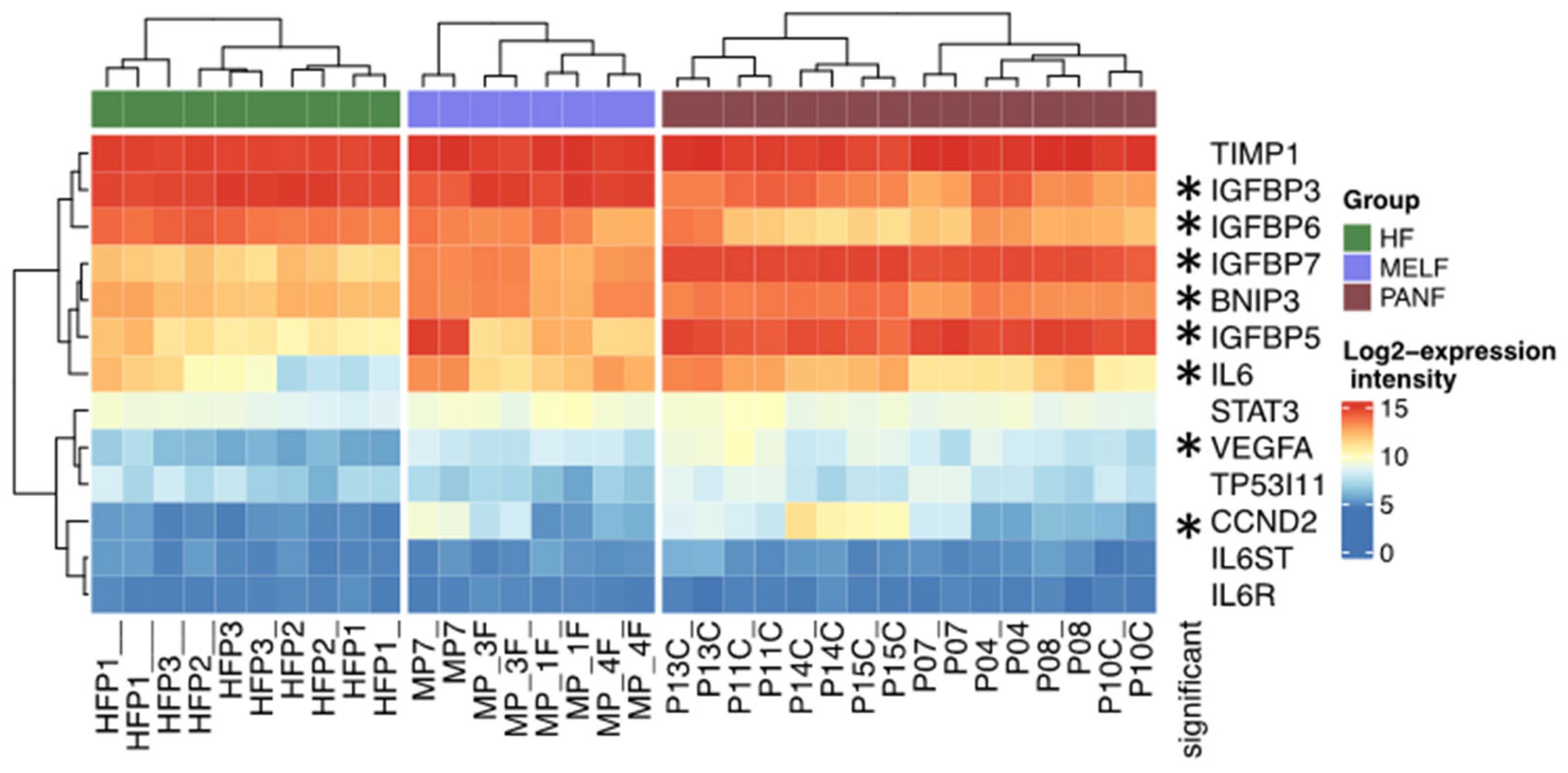
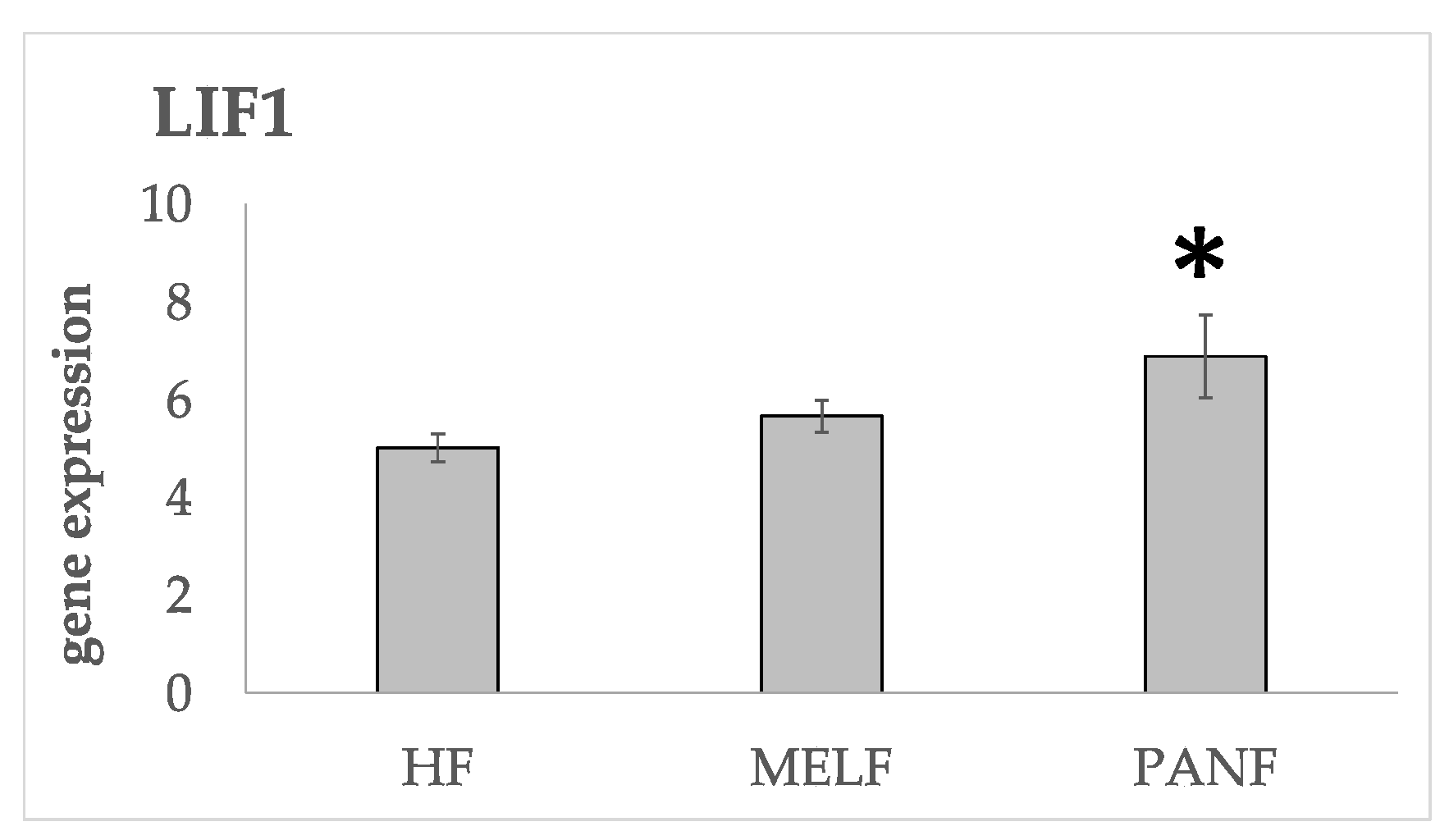
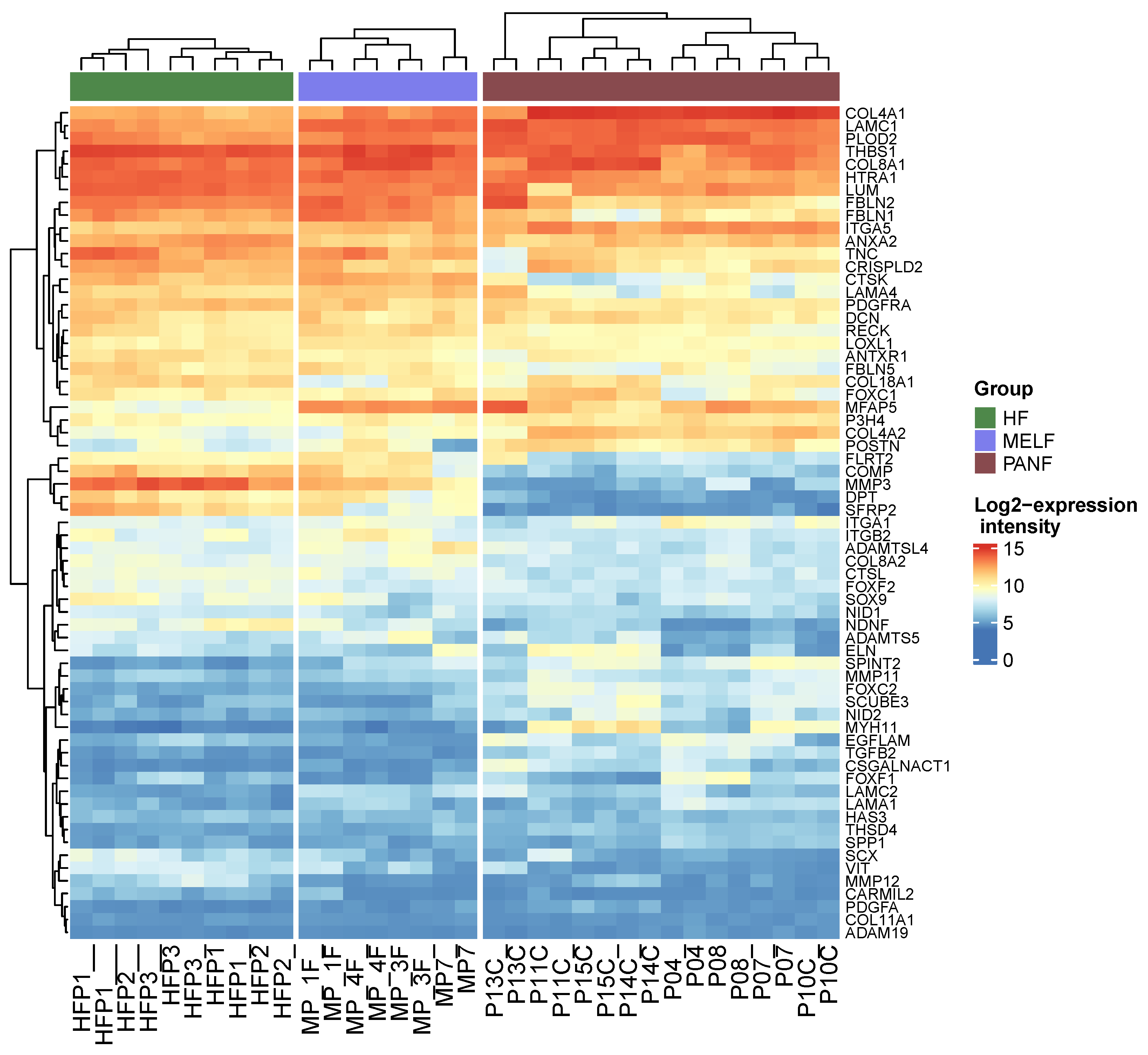
| Type of Cancer | Cancer-Supporting Effect of miR-21 | Cancer-Supporting Effect of miR-210 |
|---|---|---|
| Pancreatic ductal adenocarcinoma | [29,55,56,57,58,59,60,61,62] | [16,63] |
| Colorectal carcinoma | [58,64,65,66] | [67] |
| Lung adeno/non-small carcinoma | [68,69] | [70,71] |
| Breast | [72] | [73] |
| Gastric | [74] | Not available |
| Prostate | [75] | [76,77] |
| Thyroid medullary | [78] | Not available |
| Cutaneous malignant melanoma | Not reported | [79] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mandys, V.; Popov, A.; Gürlich, R.; Havránek, J.; Pfeiferová, L.; Kolář, M.; Vránová, J.; Smetana, K., Jr.; Lacina, L.; Szabo, P. Expression of Selected miRNAs in Normal and Cancer-Associated Fibroblasts and in BxPc3 and MIA PaCa-2 Cell Lines of Pancreatic Ductal Adenocarcinoma. Int. J. Mol. Sci. 2023, 24, 3617. https://doi.org/10.3390/ijms24043617
Mandys V, Popov A, Gürlich R, Havránek J, Pfeiferová L, Kolář M, Vránová J, Smetana K Jr., Lacina L, Szabo P. Expression of Selected miRNAs in Normal and Cancer-Associated Fibroblasts and in BxPc3 and MIA PaCa-2 Cell Lines of Pancreatic Ductal Adenocarcinoma. International Journal of Molecular Sciences. 2023; 24(4):3617. https://doi.org/10.3390/ijms24043617
Chicago/Turabian StyleMandys, Václav, Alexey Popov, Robert Gürlich, Jan Havránek, Lucie Pfeiferová, Michal Kolář, Jana Vránová, Karel Smetana, Jr., Lukáš Lacina, and Pavol Szabo. 2023. "Expression of Selected miRNAs in Normal and Cancer-Associated Fibroblasts and in BxPc3 and MIA PaCa-2 Cell Lines of Pancreatic Ductal Adenocarcinoma" International Journal of Molecular Sciences 24, no. 4: 3617. https://doi.org/10.3390/ijms24043617
APA StyleMandys, V., Popov, A., Gürlich, R., Havránek, J., Pfeiferová, L., Kolář, M., Vránová, J., Smetana, K., Jr., Lacina, L., & Szabo, P. (2023). Expression of Selected miRNAs in Normal and Cancer-Associated Fibroblasts and in BxPc3 and MIA PaCa-2 Cell Lines of Pancreatic Ductal Adenocarcinoma. International Journal of Molecular Sciences, 24(4), 3617. https://doi.org/10.3390/ijms24043617










