Sympathetic System in Wound Healing: Multistage Control in Normal and Diabetic Skin
Abstract
1. Introduction
2. Methods
- (1)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Autonomic Nervous System” [Mesh] OR autonom* nerv*)—458 results, 78 to analysis
- (2)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Receptors, Adrenergic” [Mesh])—59 results, 32 to analysis
- (3)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Epinephrine” [Mesh] OR “Norepinephrine” [Mesh] OR epinephrine OR norepinephrine)—365 results, 61 to analysis
- (4)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Receptors, Cholinergic” [Mesh])—41 results, 18 to analysis
- (5)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Acetylcholine” [Mesh])—50 results, 2 to analysis
- (6)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Neuropeptide Y” [Mesh] OR NPY)—33 results, 5 to analysis
- (7)
- (“wound healing” [Mesh] OR skin wound* OR diabet* wound* OR diabet* ulcer*) AND (“Vasoactive Intestinal Peptide” [Mesh] OR VIP)—257 results, 7 to analysis
3. Sympathetic Neurotransmitters: Neuronal and Paracrine Sources
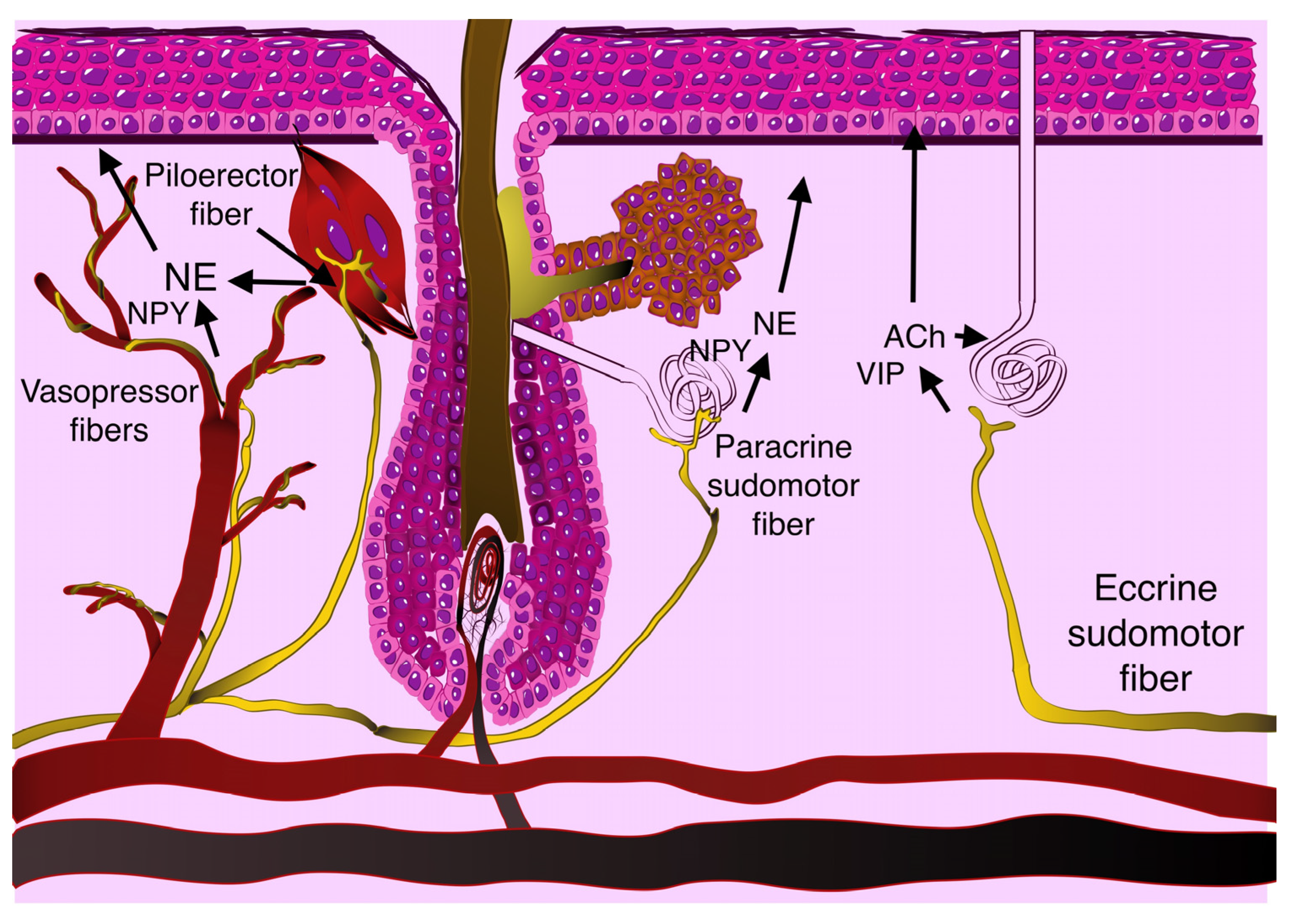
3.1. Sympathetic Regulation in Normal Skin
3.2. Neurotransmitter Synthesis in Skin Cells
4. Sympathetic Regulation in Normal Wound Healing
4.1. Inflammation and Immune Cells
4.2. Keratinocytes
4.3. Fibroblasts
4.4. Blood Vessel Cells
5. Wound Healing in Denervation Models
6. Sympathetic System in Diabetic Wounds Regeneration—Beneficial or Deleterious?
7. Targeting Sympathetic Nervous System in Diabetic Ulcers
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hicks, C.W.; Selvin, E. Epidemiology of Peripheral Neuropathy and Lower Extremity Disease in Diabetes. Curr. Diabetes Rep. 2019, 19, 86. [Google Scholar] [CrossRef] [PubMed]
- Rekha, P.-D.; Rao, S.S.; Sahana, T.G.; Prabhu, A. Diabetic wound management. Br. J. Community Nurs. 2018, 23, S16–S22. [Google Scholar] [CrossRef] [PubMed]
- Boulton, A.J.; Armstrong, D.G.; Kirsner, R.S.; Attinger, C.E.; Lavery, L.A.; Lipsky, B.A.; Mills, J.L., Sr.; Steinberg, J.S. Diagnosis and management of diabetic foot complications. Diabetes 2018, 2018, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Akkus, G.; Sert, M. Diabetic foot ulcers: A devastating complication of diabetes mellitus continues non-stop in spite of new medical treatment modalities. World J. Diabetes 2022, 13, 1106–1121. [Google Scholar] [CrossRef]
- Ghotaslou, R.; Memar, M.Y.; Alizadeh, N. Classification, microbiology and treatment of diabetic foot infections. J. Wound Care 2018, 27, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Matoori, S.; Veves, A.; Mooney, D.J. Advanced bandages for diabetic wound healing. Sci. Transl. Med. 2021, 13, eabe4839. [Google Scholar] [CrossRef]
- Jalilian, M.; Sarbarzeh, P.A.; Oubari, S. Factors Related to Severity of Diabetic Foot Ulcer: A Systematic Review. Diabetes Metab. Syndr. Obes. Targets Ther. 2020, 13, 1835–1842. [Google Scholar] [CrossRef]
- Wang, X.; Yuan, C.-X.; Xu, B.; Yu, Z. Diabetic foot ulcers: Classification, risk factors and management. World J. Diabetes 2022, 13, 1049–1065. [Google Scholar] [CrossRef]
- Okonkwo, U.A.; DiPietro, L.A. Diabetes and Wound Angiogenesis. Int. J. Mol. Sci. 2017, 18, 1419. [Google Scholar] [CrossRef] [PubMed]
- Mieczkowski, M.; Mrozikiewicz-Rakowska, B.; Kowara, M.; Kleibert, M.; Czupryniak, L. The Problem of Wound Healing in Diabetes—From Molecular Pathways to the Design of an Animal Model. Int. J. Mol. Sci. 2022, 23, 7930. [Google Scholar] [CrossRef]
- Urso, B.; Ghias, M.; John, A.; Khachemoune, A. Neuropathic ulcers: A focused review. Int. J. Dermatol. 2020, 60, e383–e389. [Google Scholar] [CrossRef]
- Rathur, H.M.; Boulton, A.J.M. The neuropathic diabetic foot. Nat. Clin. Pract. Endocrinol. Metab. 2007, 3, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Aldana, P.C.; Cartron, A.M.; Khachemoune, A. Reappraising Diabetic Foot Ulcers: A Focus on Mechanisms of Ulceration and Clinical Evaluation. Int. J. Low. Extremity Wounds 2020, 21, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Boulton, A.J.M. Diabetic Neuropathy and Foot Complications. In Handbook of Clinical Neurology; Elsevier B.V.: Amsterdam, The Netherlands, 2014; Volume 126, pp. 97–107. [Google Scholar]
- Guo, Q.; Ying, G.; Jing, O.; Zhang, Y.; Liu, Y.; Deng, M.; Long, S. Influencing factors for the recurrence of diabetic foot ulcers: A meta-analysis. Int. Wound J. 2022. [Google Scholar] [CrossRef]
- Stachura, A.; Khanna, I.; Krysiak, P.; Paskal, W.; Włodarski, P. Wound Healing Impairment in Type 2 Diabetes Model of Leptin-Deficient Mice—A Mechanistic Systematic Review. Int. J. Mol. Sci. 2022, 23, 8621. [Google Scholar] [CrossRef] [PubMed]
- Stino, A.M.; Smith, A.G. Peripheral neuropathy in prediabetes and the metabolic syndrome. J. Diabetes Investig. 2017, 8, 646–655. [Google Scholar] [CrossRef]
- Noor, S.; Zubair, M.; Ahmad, J. Diabetic foot ulcer—A review on pathophysiology, classification and microbial etiology. Diabetes Metab. Syndr. Clin. Res. Rev. 2015, 9, 192–199. [Google Scholar] [CrossRef]
- Deng, L.; Du, C.; Song, P.; Chen, T.; Rui, S.; Armstrong, D.G.; Deng, W. The Role of Oxidative Stress and Antioxidants in Diabetic Wound Healing. Oxidative Med. Cell. Longev. 2021, 2021, 8852759. [Google Scholar] [CrossRef]
- Steeper, R. A critical review of the aetiology of diabetic neuropathic ulcers. J. Wound Care 2005, 14, 101–103. [Google Scholar] [CrossRef]
- Schaper, N.C.; Huijberts, M.; Pickwell, K. Neurovascular control and neurogenic inflammation in diabetes. Diabetes/Metab. Res. Rev. 2008, 24, S40–S44. [Google Scholar] [CrossRef]
- Pradhan, L.; Nabzdyk, C.; Andersen, N.D.; LoGerfo, F.W.; Veves, A. Inflammation and neuropeptides: The connection in diabetic wound healing. Expert Rev. Mol. Med. 2009, 11, e2. [Google Scholar] [CrossRef] [PubMed]
- Barker, A.R.; Rosson, G.; Dellon, A.L. Wound Healing in Denervated Tissue. Ann. Plast. Surg. 2006, 57, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Theocharidis, G.; Veves, A. Autonomic Nerve Dysfunction and Impaired Diabetic Wound Healing: The Role of Neuropep-tides. Auton. Neurosci. 2020, 223, 102610. [Google Scholar] [CrossRef] [PubMed]
- Anand, P. Neurotrophic factors and their receptors in human sensory neuropathies. Prog. Brain Res. 2004, 146, 477–492. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, L.; Carvalho, E.; Cruz, M.T. Role of neuropeptides in skin inflammation and its involvement in diabetic wound healing. Expert Opin. Biol. Ther. 2010, 10, 1427–1439. [Google Scholar] [CrossRef]
- Sun, S.; Ma, J.; Ran, X. Mechanisms of Adrenergic β-Antagonist for Wounds and Its Application Prospect in Diabetic Foot Ulcers. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2020, 34, 1630–1634. [Google Scholar] [CrossRef]
- Nowak, N.C.; Menichella, D.M.; Miller, R.; Paller, A.S. Cutaneous innervation in impaired diabetic wound healing. Transl. Res. 2021, 236, 87–108. [Google Scholar] [CrossRef]
- Pan, L.; Tang, J.; Liu, H.; Cheng, B. Sympathetic nerves: How do they affect angiogenesis, particularly during wound healing of soft tissues? Clin. Hemorheol. Microcirc. 2016, 62, 181–191. [Google Scholar] [CrossRef]
- Ashrafi, M.; Baguneid, M.; Bayat, A. The Role of Neuromediators and Innervation in Cutaneous Wound Healing. Acta Dermato-Venereol. 2016, 96, 587–597. [Google Scholar] [CrossRef]
- Gupta, D.; Kaushik, D.; Mohan, V. Role of neurotransmitters in the regulation of cutaneous wound healing. Exp. Brain Res. 2022, 240, 1649–1659. [Google Scholar] [CrossRef]
- Slominski, A.T.; Slominski, R.M.; Raman, C.; Chen, J.Y.; Athar, M.; Elmets, C. Neuroendocrine signaling in the skin with a special focus on the epidermal neuropeptides. Am. J. Physiol. Physiol. 2022, 323, C1757–C1776. [Google Scholar] [CrossRef] [PubMed]
- Grando, S.A.; Pittelkow, M.R.; Schallreuter, K.U. Adrenergic and Cholinergic Control in the Biology of Epidermis: Physiological and Clinical Significance. J. Investig. Dermatol. 2006, 126, 1948–1965. [Google Scholar] [CrossRef] [PubMed]
- Kurzen, H.; Schallreuter, K.U. Novel aspects in cutaneous biology of acetylcholine synthesis and acetylcholine receptors. Exp. Dermatol. 2004, 13, 27–30. [Google Scholar] [CrossRef]
- Pongratz, G.; Straub, R.H. The sympathetic nervous response in inflammation. Arthritis Res. Ther. 2014, 16, 504. [Google Scholar] [CrossRef]
- Vinik, A.I.; Maser, R.E.; Mitchell, B.D.; Freeman, R. Diabetic Autonomic Neuropathy. Diabetes Care 2003, 26, 1553–1579. [Google Scholar] [CrossRef] [PubMed]
- Sharma, J.K.; Rohatgi, A.; Sharma, D. Diabetic Autonomic Neuropathy: A Clinical Update. J. R. Coll. Physicians Edinb. 2020, 50, 269–273. [Google Scholar] [CrossRef] [PubMed]
- Basra, R.; Papanas, N.; Farrow, F.; Karalliedde, J.; Vas, P. Diabetic Foot Ulcers and Cardiac Autonomic Neuropathy. Clin. Ther. 2022, 44, 323–330. [Google Scholar] [CrossRef]
- Anderson, C.; Bergner, A.; Murphy, S. How many types of cholinergic sympathetic neuron are there in the rat stellate ganglion? Neuroscience 2006, 140, 567–576. [Google Scholar] [CrossRef]
- Macefield, V.G. Sympathetic Microneurography. In Handbook of Clinical Neurology; Elsevier B.V.: Amsterdam, The Netherlands, 2013; Volume 117, pp. 353–364. [Google Scholar]
- Schmelz, M.; Schmidt, R.; Bickel, A.; Torebjörk, H.E.; Handwerker, H.O. Innervation territories of single sympathetic C fibers in human skin. J. Neurophysiol. 1998, 79, 1653–1660. [Google Scholar] [CrossRef]
- Donadio, V.; Incensi, A.; Vacchiano, V.; Infante, R.; Magnani, M.; Liguori, R. The autonomic innervation of hairy skin in humans: An in vivo confocal study. Sci. Rep. 2019, 9, 16982. [Google Scholar] [CrossRef]
- von Duuring, M.; Fricke, B. Organization of Peripheral Nerves in Skin, Musculoskeletal System and Viscera. In Neuronal Activity in Tumor Tissue; KARGER: Basel, Switzerland, 2007; Volume 39, pp. 30–44. [Google Scholar]
- Benarroch, E.E. Neuropeptides in the sympathetic system: Presence, plasticity, modulation, and implications. Ann. Neurol. 1994, 36, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Mao, Z.; Yang, L.; Sun, K. Co-staining Blood Vessels and Nerve Fibers in Adipose Tissue. J. Vis. Exp. 2019, 144, e59266. [Google Scholar] [CrossRef] [PubMed]
- Bartness, T.J.; Ryu, V. Neural control of white, beige and brown adipocytes. Int. J. Obes. Suppl. 2015, 5, S35–S39. [Google Scholar] [CrossRef] [PubMed]
- Katayama, K.; Saito, M. Muscle sympathetic nerve activity during exercise. J. Physiol. Sci. 2019, 69, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Di Bona, A.; Vita, V.; Costantini, I.; Zaglia, T. Towards a clearer view of sympathetic innervation of cardiac and skeletal muscles. Prog. Biophys. Mol. Biol. 2020, 154, 80–93. [Google Scholar] [CrossRef]
- Grando, S.A. Cholinergic control of epidermal cohesion. Exp. Dermatol. 2006, 15, 265–282. [Google Scholar] [CrossRef]
- Pullar, C.E.; Rizzo, A.; Isseroff, R.R. β-Adrenergic receptor antagonists accelerate skin wound healing: Evidence for a catecholamine synthesis network in the epidermis. J. Biol. Chem. 2006, 281, 21225–21235. [Google Scholar] [CrossRef]
- Steinkraus, V.; Mak, J.C.W.; Pichlmeier, U.; Mensing, H.; Ring, J.; Barnes, P.J. Autoradiographic mapping of beta-adrenoceptors in human skin. Arch. Dermatol. Res. 1996, 288, 549–553. [Google Scholar] [CrossRef]
- Sivamani, R.K.; Lam, S.T.; Isseroff, R.R. Beta Adrenergic Receptors in Keratinocytes. Dermatol. Clin. 2007, 25, 643–653. [Google Scholar] [CrossRef]
- Kurzen, H.; Wessler, I.; Kirkpatrick, C.J.; Kawashima, K.; Grando, S.A. The Non-neuronal Cholinergic System of Human Skin. Horm. Metab. Res. 2007, 39, 125–135. [Google Scholar] [CrossRef]
- Sivamani, R.K.; Garcia, M.S.; Rivkah Isseroff, R. Wound Re-Epithelialization: Modulating Keratinocyte Migration in Wound Healing. Front. Biosci. 2007, 12, 2849–2868. [Google Scholar]
- Suh, D.Y.; Hunt, T.K. Time line of wound healing. Clin. Podiatr. Med. Surg. 1998, 15, 1–9. [Google Scholar] [PubMed]
- Engel, D. The influence of the sympathetic nervous system on capillary permeability. Res. Exp. Med. 1978, 173, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Coderre, T.J.; Basbaum, A.I.; Levine, J.D. Neural control of vascular permeability: Interactions between primary afferents, mast cells, and sympathetic efferents. J. Neurophysiol. 1989, 62, 48–58. [Google Scholar] [CrossRef]
- Lefrandt, J.D.; Bosma, E.; Oomen, P.H.N.; van der Hoeven, J.H.; van Roon, A.M.; Smit, A.J.; Hoogenberg, K. Sympathetic mediated vasomotion and skin capillary permeability in diabetic patients with peripheral neuropathy. Diabetologia 2003, 46, 40–47. [Google Scholar] [CrossRef]
- Sulakvelidze, I.; Baluk, P.; McDonald, D.M. Plasma extravasation induced in rat trachea by 6-OHDA is mediated by sensory nerves, not by sympathetic nerves. J. Appl. Physiol. 1994, 76, 701–707. [Google Scholar] [CrossRef]
- Rough, J.; Engdahl, R.; Opperman, K.; Yerrum, S.; Monroy, M.A.; Daly, J.M. β2 Adrenoreceptor blockade attenuates the hyperinflammatory response induced by traumatic injury. Surgery 2009, 145, 235–242. [Google Scholar] [CrossRef]
- Beta-Adrenergic Receptors in Human Leukocyte Subpopulations–PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/1333965/ (accessed on 12 June 2022).
- Scanzano, A.; Cosentino, M. Adrenergic regulation of innate immunity: A review. Front. Pharmacol. 2015, 6, 171. [Google Scholar] [CrossRef]
- Galvan, D.L.; Danesh, F.R. β2-adrenergic receptors in inflammation and vascular complications of diabetes. Kidney Int. 2017, 92, 14–16. [Google Scholar] [CrossRef]
- Ağaç, D.; Estrada, L.D.; Maples, R.; Hooper, L.V.; Farrar, J.D. The β2-adrenergic receptor controls inflammation by driving rapid IL-10 secretion. Brain Behav. Immun. 2018, 74, 176–185. [Google Scholar] [CrossRef]
- Kolmus, K.; Tavernier, J.; Gerlo, S. β2-Adrenergic receptors in immunity and inflammation: Stressing NF-κB. Brain Behav. Immun. 2015, 45, 297–310. [Google Scholar] [CrossRef]
- Gosain, A.; Gamelli, R.L.; DiPietro, L.A. Norepinephrine-Mediated Suppression of Phagocytosis by Wound Neutrophils. J. Surg. Res. 2009, 152, 311–318. [Google Scholar] [CrossRef] [PubMed]
- Gosain, A.; Muthu, K.; Gamelli, R.L.; DiPietro, L.A. Norepinephrine suppresses wound macrophage phagocytic efficiency through alpha- and beta-adrenoreceptor dependent pathways. Surgery 2007, 142, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Miksa, M.; Das, P.; Zhou, M.; Wu, R.; Dong, W.; Ji, Y.; Goyert, S.; Ravikumar, T.S.; Wang, P. Pivotal Role of the α2A-Adrenoceptor in Producing Inflammation and Organ Injury in a Rat Model of Sepsis. PLoS ONE 2009, 4, e5504. [Google Scholar] [CrossRef]
- Sharma, D.; Farrar, J.D. Adrenergic regulation of immune cell function and inflammation. Semin. Immunopathol. 2020, 42, 709–717. [Google Scholar] [CrossRef]
- Grisanti, L.A.; Woster, A.P.; Dahlman, J.; Sauter, E.R.; Combs, C.K.; Porter, J.E. α1-Adrenergic Receptors Positively Regulate Toll-Like Receptor Cytokine Production from Human Monocytes and Macrophages. J. Pharmacol. Exp. Ther. 2011, 338, 648–657. [Google Scholar] [CrossRef] [PubMed]
- Enten, G.A.; Gao, X.; Strzelinski, H.R.; Weche, M.; Liggett, S.B.; Majetschak, M. α 1B/D -adrenoceptors regulate chemokine receptor–mediated leukocyte migration via formation of heteromeric receptor complexes. Proc. Natl. Acad. Sci. USA 2022, 119, e2123511119. [Google Scholar] [CrossRef]
- Gao, X.; Albee, L.J.; Volkman, B.F.; Gaponenko, V.; Majetschak, M. Asymmetrical ligand-induced cross-regulation of chemokine (C-X-C motif) receptor 4 by α1-adrenergic receptors at the heteromeric receptor complex. Sci. Rep. 2018, 8, 2730. [Google Scholar] [CrossRef]
- Hübnera, G.; Brauchlea, M.; Smolab, H.; Madlenera, M.; Fässlerc, R.; Wernera, S. Differential regulation of pro-inflammatory cytokines during wound healing in normal and glucocorticoid-treated mice. Cytokine 1996, 8, 548–556. [Google Scholar] [CrossRef]
- Valls, M.D.; Cronstein, B.N.; Montesinos, M.C. Adenosine receptor agonists for promotion of dermal wound healing. Biochem. Pharmacol. 2009, 77, 1117–1124. [Google Scholar] [CrossRef]
- Almeida, T.; Pires, T.D.C.; Monte-Alto-Costa, A. Blockade of glucocorticoid receptors improves cutaneous wound healing in stressed mice. Exp. Biol. Med. 2015, 241, 353–358. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Ngo, Q.T.; Ramirez, R.P.; Nakamura, T.; Farman, N.; Aractingi, S.; Jaisser, F. The myeloid mineralocorticoid receptor regulates dermal angiogenesis and inflammation in glucocorticoid-induced impaired wound healing. Br. J. Pharmacol. 2022, 179, 5222–5232. [Google Scholar] [CrossRef]
- Tu, H.; Zhang, D.; Barksdale, A.N.; Wadman, M.C.; Muelleman, R.L.; Li, Y.-L. Dexamethasone Improves Wound Healing by Decreased Inflammation and Increased Vasculogenesis in Mouse Skin Frostbite Model. Wilderness Environ. Med. 2020, 31, 407–417. [Google Scholar] [CrossRef]
- Durand, M.; Hagimont, E.P.; Louis, H.; Asfar, P.M.; Frippiat, J.-P.; Singer, M.M.; Gauchotte, G.M.; Labat, C.B.; Lacolley, P.M.; Levy, B.M.; et al. The β1-Adrenergic Receptor Contributes to Sepsis-Induced Immunosuppression Through Modulation of Regulatory T-Cell Inhibitory Function. Crit. Care Med. 2022, 50, e707–e718. [Google Scholar] [CrossRef]
- Sitkauskiene, B. The Role of β2-Adrenergic Receptors in Inflammation and Allergy. Curr. Drug Targets Inflamm. Allergy 2005, 4, 157–162. [Google Scholar] [CrossRef]
- Van Der Jagt, M.; Miranda, D.R. Beta-blockers in Intensive Care Medicine: Potential Benefit in Acute Brain Injury and Acute Respiratory Distress Syndrome. Recent Patents Cardiovasc. Drug Discov. 2012, 7, 141–151. [Google Scholar] [CrossRef]
- Lira, A.; Pinsky, M.R. Should β-blockers be used in septic shock? Crit. Care 2014, 18, 304. [Google Scholar] [CrossRef]
- Nguyen, L.P.; Omoluabi, O.; Parra, S.; Frieske, J.M.; Clement, C.; Ammar-Aouchiche, Z.; Ho, S.B.; Ehre, C.; Kesimer, M.; Knoll, B.J.; et al. Chronic Exposure to Beta-Blockers Attenuates Inflammation and Mucin Content in a Murine Asthma Model. Am. J. Respir. Cell Mol. Biol. 2008, 38, 256–262. [Google Scholar] [CrossRef]
- Novotny, N.M.; Lahm, T.; Markel, T.A.; Crisostomo, P.R.; Wang, M.; Wang, Y.; Ray, R.; Tan, J.; Al-Azzawi, D.; Meldrum, D.R. β-Blockers in Sepsis: Reexamining the evidence. Shock 2009, 31, 113–119. [Google Scholar] [CrossRef]
- Al-Kuraishy, H.M.; Al-Gareeb, A.I.; Mostafa-Hedeab, G.; Kasozi, K.I.; Zirintunda, G.; Aslam, A.; Allahyani, M.; Welburn, S.C.; Batiha, G.E.-S. Effects of β-Blockers on the Sympathetic and Cytokines Storms in Covid-19. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Hasegawa, D.; Sato, R.; Prasitlumkum, N.; Nishida, K.; Takahashi, K.; Yatabe, T.; Nishida, O. Effect of Ultrashort-Acting β-Blockers on Mortality in Patients With Sepsis With Persistent Tachycardia Despite Initial Resuscitation: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Chest 2021, 159, 2289–2300. [Google Scholar] [CrossRef]
- Henriquez, A.R.; Snow, S.J.; Schladweiler, M.C.; Miller, C.N.; Dye, J.A.; Ledbetter, A.D.; Richards, J.E.; Mauge-Lewis, K.; McGee, M.A.; Kodavanti, U.P. Adrenergic and glucocorticoid receptor antagonists reduce ozone-induced lung injury and inflammation. Toxicol. Appl. Pharmacol. 2017, 339, 161–171. [Google Scholar] [CrossRef]
- Souza, B.R.; Santos, J.S.; Costa, A.M. Blockade of beta1- and beta2-adrenoceptors delays wound contraction and re-epithelialization in rats. Clin. Exp. Pharmacol. Physiol. 2006, 33, 421–430. [Google Scholar] [CrossRef]
- Romana-Souza, B.; Nascimento, A.P.; Monte-Alto-Costa, A. Propranolol improves cutaneous wound healing in streptozotocin-induced diabetic rats. Eur. J. Pharmacol. 2009, 611, 77–84. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Wang, H.; Czura, C.J.; Friedman, S.G.; Tracey, K.J. The Cholinergic Anti-inflammatory Pathway: A Missing Link in Neuroimmunomodulation. Mol. Med. 2003, 9, 125–134. [Google Scholar] [CrossRef]
- Sato, E.; Koyama, S.; Okubo, Y.; Kubo, K.; Sekiguchi, M. Acetylcholine stimulates alveolar macrophages to release inflammatory cell chemotactic activity. Am. J. Physiol. Cell. Mol. Physiol. 1998, 274, L970–L979. [Google Scholar] [CrossRef]
- Fujii, T.; Mashimo, M.; Moriwaki, Y.; Misawa, H.; Ono, S.; Horiguchi, K.; Kawashima, K. Expression and Function of the Cholinergic System in Immune Cells. Front. Immunol. 2017, 8, 1085. [Google Scholar] [CrossRef]
- Koarai, A.; Traves, S.L.; Fenwick, P.S.; Brown, S.M.; Chana, K.K.; Russell, R.; Nicholson, A.G.; Barnes, P.J.; Donnelly, L.E. Expression of muscarinic receptors by human macrophages. Eur. Respir. J. 2011, 39, 698–704. [Google Scholar] [CrossRef]
- Kawashima, K.; Fujii, T.; Moriwaki, Y.; Misawa, H. Critical roles of acetylcholine and the muscarinic and nicotinic acetylcholine receptors in the regulation of immune function. Life Sci. 2012, 91, 1027–1032. [Google Scholar] [CrossRef]
- St-Pierre, S.; Jiang, W.; Roy, P.; Champigny, C.; Leblanc, E.; Morley, B.J.; Hao, J.; Simard, A.R. Nicotinic Acetylcholine Receptors Modulate Bone Marrow-Derived Pro-Inflammatory Monocyte Production and Survival. PLoS ONE 2016, 11, e0150230. [Google Scholar] [CrossRef]
- Lu, J.; Wu, W. Cholinergic modulation of the immune system–A novel therapeutic target for myocardial inflammation. Int. Immunopharmacol. 2021, 93, 107391. [Google Scholar] [CrossRef] [PubMed]
- Razani-Boroujerdi, S.; Singh, S.P.; Knall, C.; Hahn, F.F.; Peña-Philippides, J.C.; Kalra, R.; Langley, R.J.; Sopori, M.L. Chronic nicotine inhibits inflammation and promotes influenza infection. Cell. Immunol. 2004, 230, 1–9. [Google Scholar] [CrossRef]
- Delgado, M.; Pozo, D.; Ganea, D. The Significance of Vasoactive Intestinal Peptide in Immunomodulation. Pharmacol. Rev. 2004, 56, 249–290. [Google Scholar] [CrossRef]
- Gonzalez-Rey, E.; Chorny, A.; Fernandez-Martin, A.; Ganea, D.; Delgado, M. Vasoactive intestinal peptide generates human tolerogenic dendritic cells that induce CD4 and CD8 regulatory T cells. Blood 2006, 107, 3632–3638. [Google Scholar] [CrossRef]
- Delgado, M.; Chorny, A.; Gonzalez-Rey, E.; Ganea, D. Vasoactive intestinal peptide generates CD4+CD25+ regulatory T cells in vivo. J. Leukoc. Biol. 2005, 78, 1327–1338. [Google Scholar] [CrossRef] [PubMed]
- Dimitrijević, M.; Stanojević, S. The intriguing mission of neuropeptide Y in the immune system. Amino Acids 2011, 45, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Wheway, J.; Herzog, H.; Mackay, F. NPY and Receptors in Immune and Inflammatory Diseases. Curr. Top. Med. Chem. 2007, 7, 1743–1752. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekharan, B.; Nezami, B.G.; Srinivasan, S. Emerging neuropeptide targets in inflammation: NPY and VIP. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G949–G957. [Google Scholar] [CrossRef]
- El-Salhy, M.; Hausken, T. The role of the neuropeptide Y (NPY) family in the pathophysiology of inflammatory bowel disease (IBD). Neuropeptides 2016, 55, 137–144. [Google Scholar] [CrossRef]
- Makinde, T.O.; Steininger, R.; Agrawal, D.K. NPY and NPY receptors in airway structural and inflammatory cells in allergic asthma. Exp. Mol. Pathol. 2013, 94, 45–50. [Google Scholar] [CrossRef]
- Kunath, J.; Delaroque, N.; Szardenings, M.; Neundorf, I.; Straub, R.H. Sympathetic nerve repulsion inhibited by designer molecules in vitro and role in experimental arthritis. Life Sci. 2017, 168, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Koeck, F.-X.; Bobrik, V.; Fassold, A.; Grifka, J.; Kessler, S.; Straub, R.H. Marked loss of sympathetic nerve fibers in chronic Charcot foot of diabetic origin compared to ankle joint osteoarthritis. J. Orthop. Res. 2008, 27, 736–741. [Google Scholar] [CrossRef] [PubMed]
- Klatt, S.; Fassold, A.; Straub, R.H. Sympathetic nerve fiber repulsion: Testing norepinephrine, dopamine, and 17β-estradiol in a primary murine sympathetic neurite outgrowth assay. Ann. N. Y. Acad. Sci. 2012, 1261, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Hoover, D.B.; Brown, T.C.; Miller, M.K.; Schweitzer, J.B.; Williams, D.L. Loss of Sympathetic Nerves in Spleens from Pa-tients with End Stage Sepsis. Front. Immunol. 2017, 8, 1712. [Google Scholar] [CrossRef]
- Usui, M.L.; Underwood, R.A.; Mansbridge, J.N.; Muffley, L.A.; Carter, W.G.; Olerud, J.E. Morphological evidence for the role of suprabasal keratinocytes in wound reepithelialization. Wound Repair Regen. 2005, 13, 468–479. [Google Scholar] [CrossRef]
- Galkowska, H.; Wojewodzka, U.; Olszewski, W.L. Chemokines, cytokines, and growth factors in keratinocytes and dermal endothelial cells in the margin of chronic diabetic foot ulcers. Wound Repair Regen. 2006, 14, 558–565. [Google Scholar] [CrossRef]
- Chen, J.; Hoffman, B.B.; Isseroff, R.R. β-Adrenergic Receptor Activation Inhibits Keratinocyte Migration via a Cyclic Adenosine Monophosphate-independent Mechanism. J. Investig. Dermatol. 2002, 119, 1261–1268. [Google Scholar] [CrossRef]
- Pullar, C.E.; Manabat-Hidalgo, C.G.; Bolaji, R.S.; Isseroff, R.R. β-Adrenergic receptor modulation of wound repair. Pharmacol. Res. 2008, 58, 158–164. [Google Scholar] [CrossRef]
- Donaldson, D.J.; Mahan, J.T. Influence of catecholamines on epidermal cell migration during wound closure in adult newts. Comp. Biochem. Physiol. Part C Comp. Pharmacol. 1984, 78, 267–270. [Google Scholar] [CrossRef]
- Pullar, C.E.; Chen, J.; Isseroff, R.R. PP2A Activation by β2-Adrenergic Receptor Agonists: Novel Regulatory Mecha-nism of Keratinocyte Migration. J. Biol. Chem. 2003, 278, 22555–22562. [Google Scholar] [CrossRef]
- Wu, C.-S.; Tsao, D.-A.; Chang, H.-R. Beta2-adrenergic receptor agonist inhibits keratinocyte proliferation by mechanisms involving nitric oxide. Adv. Dermatol. Allergol. 2021, 38, 396–403. [Google Scholar] [CrossRef] [PubMed]
- Dasu, M.R.; Ramirez, S.R.; La, T.D.; Gorouhi, F.; Nguyen, C.; Lin, B.R.; Mashburn, C.; Stewart, H.; Peavy, T.R.; Nolta, J.A.; et al. Crosstalk Between Adrenergic and Toll-Like Receptors in Human Mesenchymal Stem Cells and Keratinocytes: A Recipe for Impaired Wound Healing. STEM CELLS Transl. Med. 2014, 3, 745–759. [Google Scholar] [CrossRef] [PubMed]
- Parrado, A.C.; Canellada, A.; Gentile, T.; Rey-Roldán, E.B. Dopamine Agonists Upregulate IL-6 and IL-8 Production in Human Keratinocytes. Neuroimmunomodulation 2012, 19, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Sivamani, R.K.; Shi, B.; Griffiths, E.; Vu, S.M.; Lev-Tov, H.A.; Dahle, S.; Chigbrow, M.; La, T.D.; Mashburn, C.; Peavy, T.R.; et al. Acute Wounding Alters the Beta2-Adrenergic Signaling and Catecholamine Synthetic Pathways in Keratinocytes. J. Investig. Dermatol. 2014, 134, 2258–2266. [Google Scholar] [CrossRef]
- Sivamani, R.K.; Pullar, C.E.; Manabat-Hidalgo, C.G.; Rocke, D.M.; Carlsen, R.C.; Greenhalgh, D.G.; Isseroff, R.R. Stress-Mediated Increases in Systemic and Local Epinephrine Impair Skin Wound Healing: Potential New Indication for Beta Blockers. PLoS Med. 2009, 6, e1000012. [Google Scholar] [CrossRef]
- Romana-Souza, B.; Nascimento, A.P.; Brum, P.C.; Monte-Alto-Costa, A. Deletion of the α2A/α2C-adrenoceptors accelerates cutaneous wound healing in mice. Int. J. Exp. Pathol. 2014, 95, 330–341. [Google Scholar] [CrossRef]
- Yang, H.-Y.; Steenhuis, P.; Glucksman, A.M.; Gurenko, Z.; La, T.D.; Isseroff, R.R. Alpha and beta adrenergic receptors modulate keratinocyte migration. PLoS ONE 2021, 16, e0253139. [Google Scholar] [CrossRef]
- Kurzen, H.; Berger, H.; Jäger, C.; Hartschuh, W.; Maas-Szabowski, N. Alpha 9 acetylcholine receptors are essential for epidermal differentiation. Exp. Dermatol. 2005, 14, 155. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Chernyavsky, A.I.; Arredondo, J.; Bercovich, D.; Orr-Urtreger, A.; Vetter, D.; Wess, J.; Beaudet, A.L.; Kitajima, Y.; Grando, S.A. Synergistic control of keratinocyte adhesion through muscarinic and nicotinic acetylcholine receptor subtypes. Exp. Cell Res. 2004, 294, 534–549. [Google Scholar] [CrossRef]
- Buchli, R.; Ndoye, A.; Rodriguez, J.G.; Zia, S.; Webber, R.J.; Grando, S.A. Human Skin Fibroblasts Express M2, M4, and M5 Subtypes of Muscarinic Acetylcholine Receptors. J. Cell. Biochem. 1999, 74, 264–277. [Google Scholar] [CrossRef]
- Kishibe, M.; Griffin, T.M.; Radek, K.A. Keratinocyte nicotinic acetylcholine receptor activation modulates early TLR2-mediated wound healing responses. Int. Immunopharmacol. 2015, 29, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Ndoye, A.; Buchli, R.; Greenberg, B.; Nguyen, V.T.; Zia, S.; Rodriguez, J.G.; Webber, R.J.; Lawry, M.A.; Grando, S.A. Identification and Mapping of Keratinocyte Muscarinic Acetylcholine Receptor Subtypes in Human Epidermis. J. Investig. Dermatol. 1998, 111, 410–416. [Google Scholar] [CrossRef]
- Wollina, U.; Huschenbeck, J.; Knöll, B.; Sternberg, B.; Hipler, U.-C. Vasoactive intestinal peptide supports induced migration of human keratinocytes and their colonization of an artificial polyurethane matrix. Regul. Pept. 1997, 70, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Haegerstrand, A.; Jonzon, B.; Dalsgaard, C.J.; Nilsson, J. Vasoactive intestinal polypeptide stimulates cell proliferation and adenylate cyclase activity of cultured human keratinocytes. Proc. Natl. Acad. Sci. USA 1989, 86, 5993–5996. [Google Scholar] [CrossRef]
- Bennett, L.A.T.; Johnson, J.M.; Stephens, D.P.; Saad, A.R.; Kellogg, D.L. Evidence for a Role for Vasoactive Intestinal Peptide in Active Vasodilatation in the Cutaneous Vasculature of Humans. J. Physiol. 2003, 552, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Dumont, Y.; Bastianetto, S.; Duranton, A.; Breton, L.; Quirion, R. Immunohistochemical distribution of neuropeptide Y, peptide YY, pancreatic polypeptide-like immunoreactivity and their receptors in the epidermal skin of healthy women. Peptides 2015, 70, 7–16. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Nakanishi, S.; Imamura, S. Direct Effects of Cutaneous Neuropeptides on Adenylyl Cyclase Activity and Proliferation in a Keratinocyte Cell Line: Stimulation of Cyclic AMP Formation by CGRP and VIP/PHM, and Inhibition by NPY Through G Protein-Coupled Receptors. J. Investig. Dermatol. 1993, 101, 646–651. [Google Scholar] [CrossRef]
- Jiang, C.K.; Tomić-Canić, M.; Lucas, D.J.; Simon, M.; Blumenberg, M. TGF beta promotes the basal phenotype of epidermal keratinocytes: Transcriptional induction of K#5 and K#14 keratin genes. Growth Factors 1995, 12, 87–97. [Google Scholar]
- Hameedaldeen, A.; Liu, J.; Batres, A.; Graves, G.S.; Graves, D.T. FOXO1, TGF-β Regulation and Wound Healing. Int. J. Mol. Sci. 2014, 15, 16257–16269. [Google Scholar] [CrossRef]
- Le Provost, G.S.; Pullar, C.E. β2-Adrenoceptor Activation Modulates Skin Wound Healing Processes to Reduce Scarring. J. Investig. Dermatol. 2015, 135, 279–288. [Google Scholar] [CrossRef]
- Pullar, C.E.; Isseroff, R.R. ß2-adrenergic receptor activation delays dermal fibroblast-mediated contraction of collagen gels via a cAMP-dependent mechanism. Wound Repair Regen. 2005, 13, 405–411. [Google Scholar] [CrossRef] [PubMed]
- Pullar, C.E.; Isseroff, R.R. The β2-adrenergic receptor activates pro-migratory and pro-proliferative pathways in dermal fibroblasts via divergent mechanisms. J. Cell Sci. 2006, 119, 592–602. [Google Scholar] [CrossRef] [PubMed]
- Pullar, C.E.; Le Provost, G.S.; O’Leary, A.P.; Evans, S.E.; Baier, B.S.; Isseroff, R.R. β2AR Antagonists and β2AR Gene Deletion Both Promote Skin Wound Repair Processes. J. Investig. Dermatol. 2012, 132, 2076–2084. [Google Scholar] [CrossRef]
- Arredondo, J.; Nguyen, V.T.; Chernyavsky, A.I.; Bercovich, D.; Orr-Urtreger, A.; Vetter, D.E.; Grando, S.A. Functional role of α7 nicotinic receptor in physiological control of cutaneous homeostasis. Life Sci. 2003, 72, 2063–2067. [Google Scholar] [CrossRef] [PubMed]
- Shome, S.; Rana, T.; Ganguly, S.; Basu, B.; Choudhury, S.C.; Sarkar, C.; Chakroborty, D.; Dasgupta, P.S.; Basu, S. Dopamine Regulates Angiogenesis in Normal Dermal Wound Tissues. PLoS ONE 2011, 6, e25215. [Google Scholar] [CrossRef] [PubMed]
- Jacobi, J.; Jang, J.J.; Sundram, U.; Dayoub, H.; Fajardo, L.F.; Cooke, J.P. Nicotine Accelerates Angiogenesis and Wound Healing in Genetically Diabetic Mice. Am. J. Pathol. 2002, 161, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Chakroborty, D.; Goswami, S.; Basu, S.; Sarkar, C. Catecholamines in the regulation of angiogenesis in cutaneous wound healing. FASEB J. 2020, 34, 14093–14102. [Google Scholar] [CrossRef]
- Li, J.; Zhang, Y.-P.; Kirsner, R.S. Angiogenesis in wound repair: Angiogenic growth factors and the extracellular matrix. Microsc. Res. Tech. 2003, 60, 107–114. [Google Scholar] [CrossRef]
- Pullar, C.E.; Grahn, J.C.; Liu, W.; Isseroff, R.R. β2-Adrenergic receptor activation delays wound healing. FASEB J. 2006, 20, 76–86. [Google Scholar] [CrossRef]
- Johnson, R.; Webb, J.G.; Newman, W.H.; Wang, Z. Regulation of Human Vascular Smooth Muscle Cell Migration by Beta-Adrenergic Receptors. Am. Surg. 2006, 72, 51–54. [Google Scholar] [CrossRef]
- O’Leary, A.P.; Fox, J.M.; Pullar, C.E. Beta-Adrenoceptor Activation Reduces Both Dermal Microvascular Endothelial Cell Migration via a cAMP-Dependent Mechanism and Wound Angiogenesis. J. Cell. Physiol. 2014, 230, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Drummond, P.D.; Dawson, L.F.; Wood, F.M.; Fear, M.W. Up-regulation of α 1 -adrenoceptors in burn and keloid scars. Burns 2018, 44, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, L.; Cai, X.; Wu, S.; Andersen, N.D.; Martin, M.; Malek, J.; Guthrie, P.; Veves, A.; LoGerfo, F.W. Gene Expression of Pro-Inflammatory Cytokines and Neuropeptides in Diabetic Wound Healing. J. Surg. Res. 2009, 167, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, A.J.; Cao, R.; Björndahl, M.; Nyström, S.; Jönsson-Rylander, A.-C.; Hassani, H.; Hallberg, B.; Nordlander, M.; Cao, Y. Deletion of neuropeptide Y (NPY) 2 receptor in mice results in blockage of NPY-induced angiogenesis and delayed wound healing. Proc. Natl. Acad. Sci. USA 2003, 100, 6033–6038. [Google Scholar] [CrossRef]
- Polak, J.; Bloom, S. Regulatory peptides—The distribution of two newly discovered peptides: PHI and NPY. Peptides 1984, 5, 79–89. [Google Scholar] [CrossRef]
- Ranne, J.; Kalimo, H.; Pyykkö, K.; Scheinin, M.; Aaltonen, V.; Niinikoski, J.; Laato, M. Wound Healing in Denervated Rat Groin Skin Flap. Eur. Surg. Res. 2000, 32, 197–202. [Google Scholar] [CrossRef]
- Saburo, K.; Hiroshi, K.; Toshihiro, M. The role of sympathetic catecholaminergic nerves in wound healing. Burns 1982, 9, 135–141. [Google Scholar] [CrossRef]
- Romana-Souza, B.; Monte-Alto-Costa, A. Simultaneous blockade of alpha and beta adrenoceptors impairs cutaneous wound healing in rats. J. Eur. Acad. Dermatol. Venereol. 2010, 24, 349–352. [Google Scholar] [CrossRef]
- Souza, B.R.; Cardoso, J.F.; Amadeu, T.P.; Desmoulière, A.; Costa, A.M.A. Sympathetic denervation accelerates wound contraction but delays reepithelialization in rats. Wound Repair Regen. 2005, 13, 498–505. [Google Scholar] [CrossRef]
- Zheng, Z.; Wan, Y.; Liu, Y.; Yang, Y.; Tang, J.; Huang, W.; Cheng, B. Sympathetic Denervation Accelerates Wound Contraction but Inhibits Reepithelialization and Pericyte Proliferation in Diabetic Mice. J. Diabetes Res. 2017, 2017, 7614685. [Google Scholar] [CrossRef]
- Jurjus, A.; Hourani, R.; Daouk, H.; Youssef, L.; Bou-Khalil, P.; Haidar, H.; Atiyeh, B.; Saade, N. Effect of denervation on burn wound healing. Ann. Burn. Fire Disasters 2018, 31, 278–291. [Google Scholar]
- Kim, L.R.; Pomeranz, B. The sympathomimetic agent, 6-hydroxydopamine, accelerates cutaneous wound healing. Eur. J. Pharmacol. 1999, 376, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.G.; Liu, M. Impaired cutaneous wound healing after sensory denervation in developing rats: Effects on cell proliferation and apoptosis. Cell Tissue Res. 2002, 307, 281–291. [Google Scholar] [CrossRef]
- Kim, L.R.; Whelpdale, K.; Zurowski, M.; Pomeranz, B. Sympathetic denervation impairs epidermal healing in cutaneous wounds. Wound Repair Regen. 1998, 6, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Wallengren, J.; Chen, D.; Sundler, F. Neuropeptide-containing C-fibres and wound healing in rat skin. Neither capsaicin nor peripheral neurotomy affect the rate of healing. Br. J. Dermatol. 1999, 140, 400–408. [Google Scholar] [CrossRef]
- Cheng, C.; Singh, V.; Krishnan, A.; Kan, M.; Martinez, J.A.; Zochodne, D.W. Loss of Innervation and Axon Plasticity Accompanies Impaired Diabetic Wound Healing. PLoS ONE 2013, 8, e75877. [Google Scholar] [CrossRef]
- Levy, D.M.; Karanth, S.S.; Springall, D.R.; Polak, J.M. Depletion of cutaneous nerves and neuropeptides in diabetes mellitus: An immunocytochemical study. Diabetologia 1989, 32, 427–433. [Google Scholar] [CrossRef]
- Reiber, G.E.; Vileikyte, L.; Boyko, E.J.; del Aguila, M.; Smith, D.G.; Lavery, L.A.; Boulton, A.J. Causal pathways for incident lower-extremity ulcers in patients with diabetes from two settings. Diabetes Care 1999, 22, 157–162. [Google Scholar] [CrossRef]
- Ahlborg, G.; Lundbergt, J.M. Exercise-induced changes in neuropeptide Y, noradrenaline and endothelin-1 levels in young people with type I diabetes. Clin. Physiol. Funct. Imaging 1996, 16, 645–655. [Google Scholar] [CrossRef]
- Burton, A.R.; Fazalbhoy, A.; Macefield, V.G. Sympathetic Responses to Noxious Stimulation of Muscle and Skin. Front. Neurol. 2016, 7, 109. [Google Scholar] [CrossRef]
- Oishi, M.; Mochizuki, Y.; Suzuki, Y.; Ogawa, K.; Naganuma, T.; Nishijo, Y.; Mizutani, T. Current Perception Threshold and Sympathetic Skin Response in Diabetic and Alcoholic Polyneuropathies. Intern. Med. 2002, 41, 819–822. [Google Scholar] [CrossRef] [PubMed]
- Ono, S.; Nishijo, Y.; Oishi, M.; Mizutani, T. Comparison of the Utility of Sympathetic Skin Response and Current Perception Threshold Examinations with Conventional Examinations for the Early Electrophysiological Diagnosis of Diabetic Poly-neuropathy. Electromyogr. Clin. Neurophysiol. 2006, 46, 401–407. [Google Scholar] [PubMed]
- Ziegler, D.; Papanas, N.; Roden, M.; GDC Study Group. Neuropad: Evaluation of three cut-off points of sudomotor dysfunction for early detection of polyneuropathy in recently diagnosed diabetes. Diabet. Med. 2011, 28, 1412–1415. [Google Scholar] [CrossRef] [PubMed]
- Schlereth, T.; Morellini, N.; Lismont, N.C.; Lemper, C.; Birklein, F.; Drummond, P.D. Alpha 1 adrenoceptor expression in skin, nerves and blood vessels of patients with painful diabetic neuropathy. Auton. Neurosci. 2021, 234, 102814. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, L.S.; Pabbidi, R.M. Diabetic Peripheral Neuropathy: Role of Reactive Oxygen and Nitrogen Species. Cell Biochem. Biophys. 2013, 67, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Di Marco, E.; Jha, J.C.; Sharma, A.; Wilkinson-Berka, J.L.; Jandeleit-Dahm, K.A.; de Haan, J.B. Are reactive oxygen species still the basis for diabetic complications? Clin. Sci. 2015, 129, 199–216. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Liang, H.; Clarke, E.; Jackson, C.; Xue, M. Inflammation in Chronic Wounds. Int. J. Mol. Sci. 2016, 17, 2085. [Google Scholar] [CrossRef]
- Zhao, G.; Usui, M.L.; Lippman, S.I.; James, G.A.; Stewart, P.S.; Fleckman, P.; Olerud, J.E. Biofilms and Inflammation in Chronic Wounds. Adv. Wound Care 2013, 2, 389–399. [Google Scholar] [CrossRef]
- Tellechea, A.; Kafanas, A.; Leal, E.; Tecilazich, F.; Kuchibhotla, S.; Auster, M.E.; Kontoes, I.; Paolino, J.; Carvalho, E.; Nabzdyk, L.P.; et al. Increased Skin Inflammation and Blood Vessel Density in Human and Experimental Diabetes. Int. J. Low. Extrem. Wounds 2013, 12, 4–11. [Google Scholar] [CrossRef]
- Wetzler, C.; Kämpfer, H.; Stallmeyer, B.; Pfeilschifter, J.; Frank, S. Large and Sustained Induction of Chemokines during Impaired Wound Healing in the Genetically Diabetic Mouse: Prolonged Persistence of Neutrophils and Macrophages during the Late Phase of Repair. J. Investig. Dermatol. 2000, 115, 245–253. [Google Scholar] [CrossRef]
- Albrecht, H.; Yang, H.-Y.; Kiuru, M.; Maksaereekul, S.; Durbin-Johnson, B.; Wong, M.S.; Stevenson, T.R.; Rocke, D.M.; Isseroff, R.R. The Beta 2 Adrenergic Receptor Antagonist Timolol Improves Healing of Combined Burn and Radiation Wounds. Radiat. Res. 2018, 189, 441–445. [Google Scholar] [CrossRef] [PubMed]
- Arbabi, S.; Ahrns, K.S.; Wahl, W.L.; Hemmila, M.R.; Wang, S.C.; Brandt, M.-M.; Taheri, P.A. Beta-Blocker Use Is Associated with Improved Outcomes in Adult Burn Patients. J. Trauma Inj. Infect. Crit. Care 2004, 56, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Beroukhim, K.; Rotunda, A.M. Topical 0.5% timolol heals a recalcitrant irradiated surgical scalp wound. Dermatol. Surg. 2014, 40, 924–926. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.C.; Dosal, J.; Kirsner, R.S. Topical Timolol for a Refractory Wound. Dermatol. Surg. 2012, 38, 135–138. [Google Scholar] [CrossRef]
- Braun, L.R.; Lamel, S.A.; Richmond, N.A.; Kirsner, R.S. Topical Timolol for Recalcitrant Wounds. JAMA Dermatol. 2013, 149, 1400–1402. [Google Scholar] [CrossRef] [PubMed]
- Thomas, B.; Kurien, J.S.; Jose, T.; Ulahannan, S.E.; Varghese, S.A. Topical timolol promotes healing of chronic leg ulcer. J. Vasc. Surg. Venous Lymphat. Disord. 2017, 5, 844–850. [Google Scholar] [CrossRef]
- Ghanbarzamani, A.; Salehifar, E.; Jafarirad, A.; Hesamirostami, M.H.; Bagherzadehsaba, A.; Saeedi, M.; Ghazaeian, M.; Khorasani, G.; Moosazadeh, M. Efficacy and Safety of 0.25% Timolol Gel in HealinSplit-Thickness Skin Graft Site. Iran. J. Pharm. Res. 2021, 20, 178–186. [Google Scholar] [CrossRef]
- Baltazard, T.; Senet, P.; Momar, D.; Picard, C.; Joachim, C.; Adas, A.; Lok, C.; Chaby, G. Evaluation of timolol maleate gel for management of hard-to-heal chronic venous leg ulcers. Phase II randomised-controlled study. In Annales de Dermatologie et de Vénéréologie; Elsevier Masson: Paris, France, 2021; Volume 148, pp. 228–232. [Google Scholar] [CrossRef]
- Kaur, R.; Tchanque-Fossuo, C.; West, K.; Hadian, Y.; Gallegos, A.; Yoon, D.; Ismailyan, L.; Schaefer, S.; Dahle, S.E.; Isseroff, R.R. Beta-adrenergic antagonist for the healing of chronic diabetic foot ulcers: Study protocol for a prospective, randomized, double-blinded, controlled and parallel-group study. Trials 2020, 21, 496. [Google Scholar] [CrossRef]
- Jolivalt, C.G.; Frizzi, K.E.; Han, M.M.; Mota, A.J.; Guernsey, L.S.; Kotra, L.P.; Fernyhough, P.; Calcutt, N.A. Topical Delivery of Muscarinic Receptor Antagonists Prevents and Reverses Peripheral Neuropathy in Female Diabetic Mice. J. Pharmacol. Exp. Ther. 2020, 374, 44–51. [Google Scholar] [CrossRef]
- Dillon, R.S. Role of Cholinergic Nervous System in Healing Neuropathic Lesions: Preliminary Studies and Prospective, Double-Blinded, Placebo-Controlled Studies. Angiology 1991, 42, 767–778. [Google Scholar] [CrossRef]
- Kishibe, M.; Griffin, T.M.; Goslawski, M.; Sinacore, J.; Kristian, S.A.; Radek, K.A. Topical nicotinic receptor activation improves wound bacterial infection outcomes and TLR2-mediated inflammation in diabetic mouse wounds. Wound Repair Regen. 2018, 26, 403–412. [Google Scholar] [CrossRef] [PubMed]
- Dong, M.-W.; Li, M.; Chen, J.; Fu, T.-T.; Lin, K.-Z.; Ye, G.-H.; Han, J.-G.; Feng, X.-P.; Li, X.-B.; Yu, L.-S.; et al. Activation of α7nAChR Promotes Diabetic Wound Healing by Suppressing AGE-Induced TNF-α Production. Inflammation 2015, 39, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Li, J.-Y.; Jiang, S.-K.; Wang, L.-L.; Zhang, M.-Z.; Wang, S.; Jiang, Z.-F.; Liu, Y.-L.; Cheng, H.; Zhang, M.; Zhao, R.; et al. α7-nAChR Activation Has an Opposite Effect on Healing of Covered and Uncovered Wounds. Inflammation 2017, 41, 474–484. [Google Scholar] [CrossRef] [PubMed]
- Masuoka, H.; Morimoto, N.; Sakamoto, M.; Ogino, S.; Suzuki, S. Exploration of the wound healing effect of topical administration of nicotine in combination with collagen scaffold in a rabbit model. J. Artif. Organs 2015, 19, 167–174. [Google Scholar] [CrossRef]
- Morimoto, N.; Takemoto, S.; Kawazoe, T.; Suzuki, S. Nicotine at a Low Concentration Promotes Wound Healing. J. Surg. Res. 2008, 145, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Liem, P.H.; Morimoto, N.; Ito, R.; Kawai, K.; Suzuki, S. Treating a collagen scaffold with a low concentration of nicotine promoted angiogenesis and wound healing. J. Surg. Res. 2013, 182, 353–361. [Google Scholar] [CrossRef] [PubMed]
| Neurotransmitters | Norepinephrine/Epinephrine | Acetylcholine | VIP | NPY |
|---|---|---|---|---|
| Primary effect | Immunosuppressive | Immunosuppressive | Immunosuppressive | Pro-inflammatory |
| Source | Vasomotor fibers/keratinocytes | Sudomotor fibers/keratinocytes | Sudomotor fibers/keratinocytes | Vasomotor fibers/keratinocytes |
| Wound healing role—inflammation | Low concentration in wound area, high in healthy tissue around | Not clear | Not clear | Not clear |
| Primary receptor blockade | Switch to pro-inflammatory, better healing or hyperinflammation | Inflammation increase, better healing? | Inflammation increase, better healing? | Studied in some hyperinflammatory conditions only |
| Neurotransmitters | Norepinephrine/Epinephrine | Acetylcholine | VIP | NPY |
|---|---|---|---|---|
| Primary effect | Inhibits activation, stimulates cytokines | Mixed through different receptors, stimulates cytokines | Activates KC | Inhibits activation |
| Source | Vasomotor fibers/keratinocytes | Sudomotor fibers/keratinocytes | Sudomotor fibers/keratinocytes | Vasomotor fibers/keratinocytes |
| Wound healing role—re-epithelialization | Stimulates KC to produce cytokines, probably to stop migration | Stimulates KC to produce cytokines, to start migration | Stimulates KC | Inhibits KC |
| Primary receptor blockade | Ambiguous data | Ambiguous data | Worse healing? | Better healing? |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ivanov, E.; Akhmetshina, M.; Erdiakov, A.; Gavrilova, S. Sympathetic System in Wound Healing: Multistage Control in Normal and Diabetic Skin. Int. J. Mol. Sci. 2023, 24, 2045. https://doi.org/10.3390/ijms24032045
Ivanov E, Akhmetshina M, Erdiakov A, Gavrilova S. Sympathetic System in Wound Healing: Multistage Control in Normal and Diabetic Skin. International Journal of Molecular Sciences. 2023; 24(3):2045. https://doi.org/10.3390/ijms24032045
Chicago/Turabian StyleIvanov, Evgenii, Marina Akhmetshina, Aleksei Erdiakov, and Svetlana Gavrilova. 2023. "Sympathetic System in Wound Healing: Multistage Control in Normal and Diabetic Skin" International Journal of Molecular Sciences 24, no. 3: 2045. https://doi.org/10.3390/ijms24032045
APA StyleIvanov, E., Akhmetshina, M., Erdiakov, A., & Gavrilova, S. (2023). Sympathetic System in Wound Healing: Multistage Control in Normal and Diabetic Skin. International Journal of Molecular Sciences, 24(3), 2045. https://doi.org/10.3390/ijms24032045







