Concurrent Infection of the Human Brain with Multiple Borrelia Species
Abstract
1. Introduction
2. Results
2.1. Patient History and Borrelia Serology
2.2. Post-Mortem Toxicology and Microbiology
2.3. PCR Detection of Borrelia DNA in Frozen Brain Tissue Samples
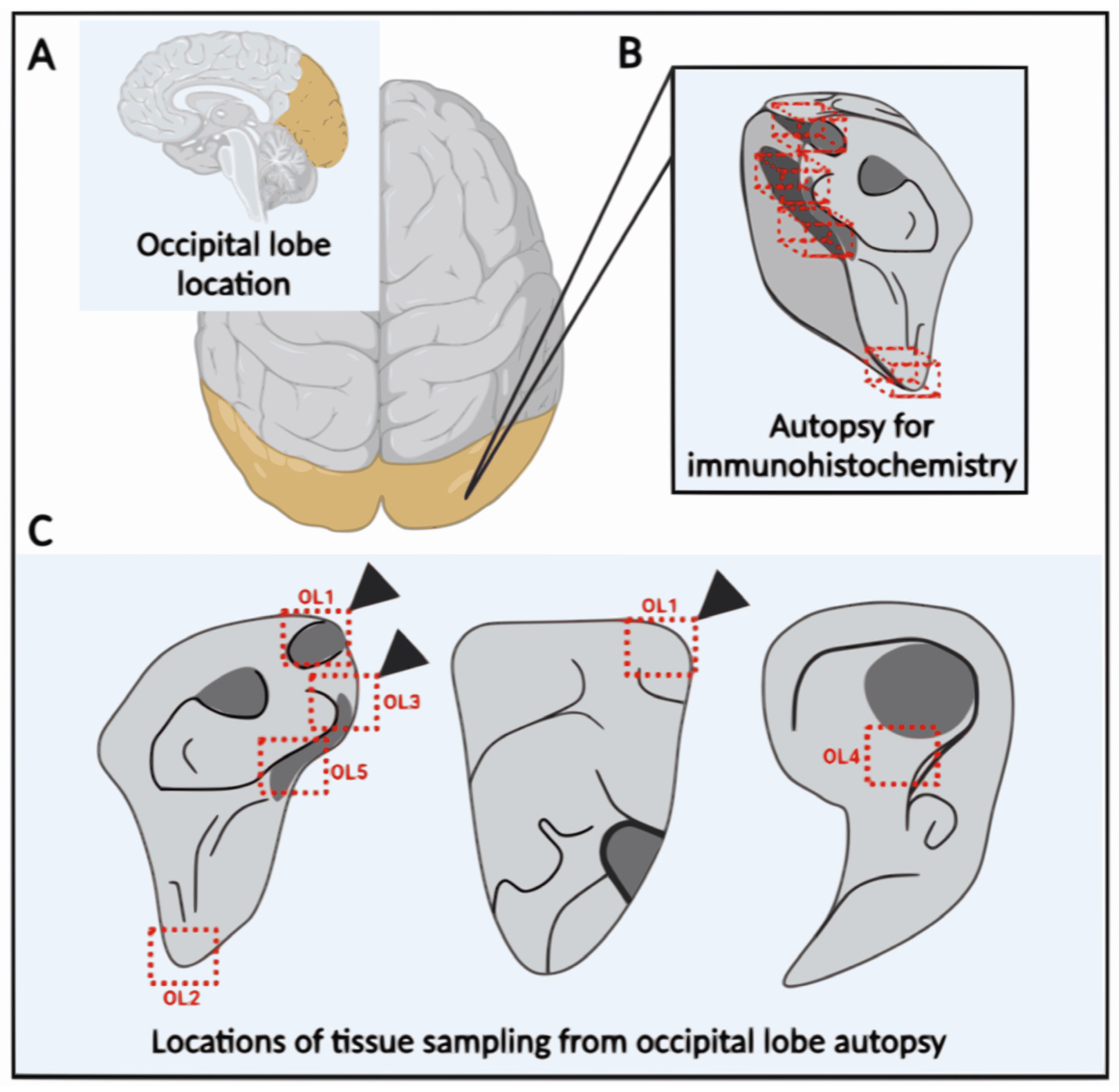
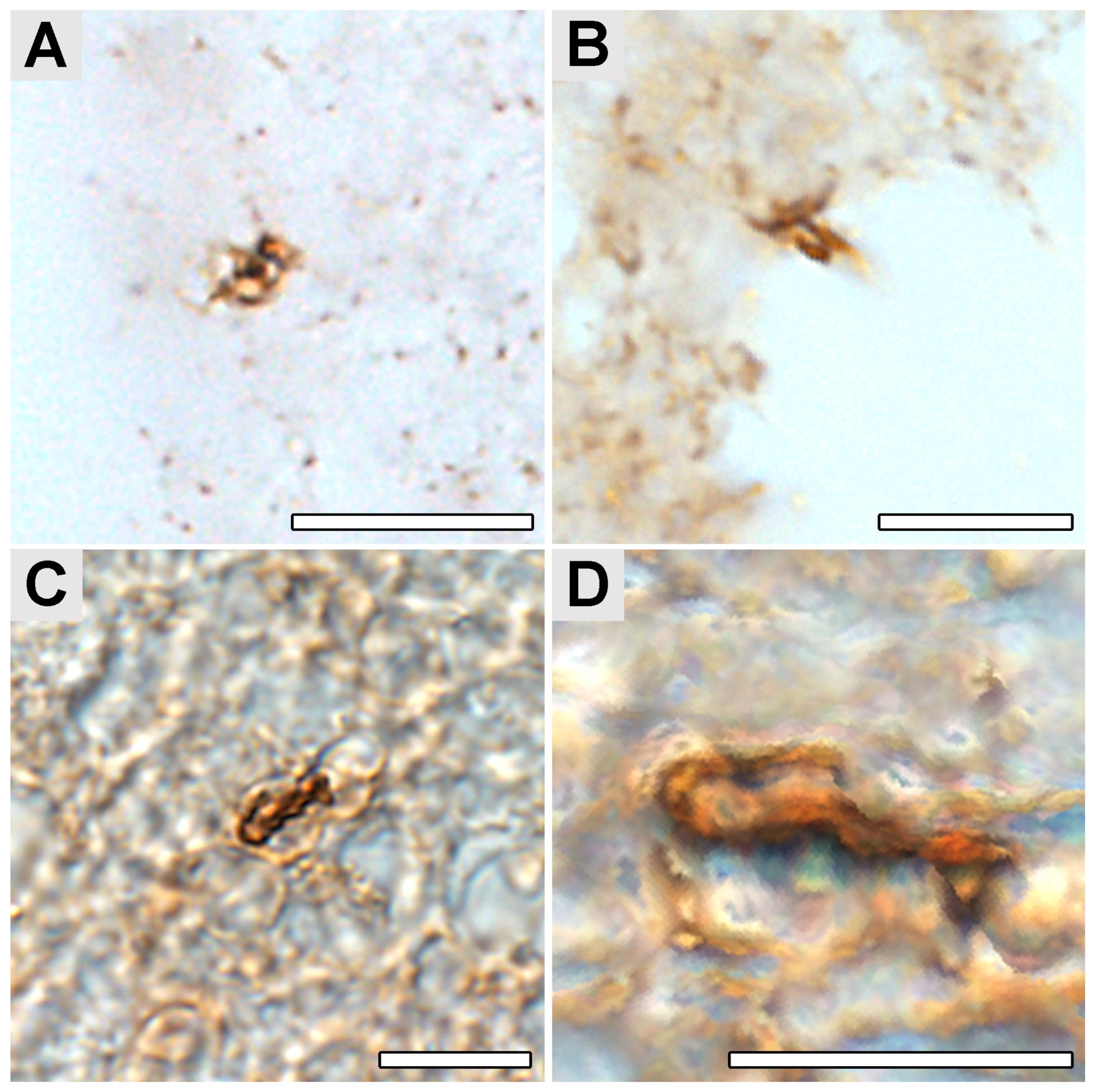
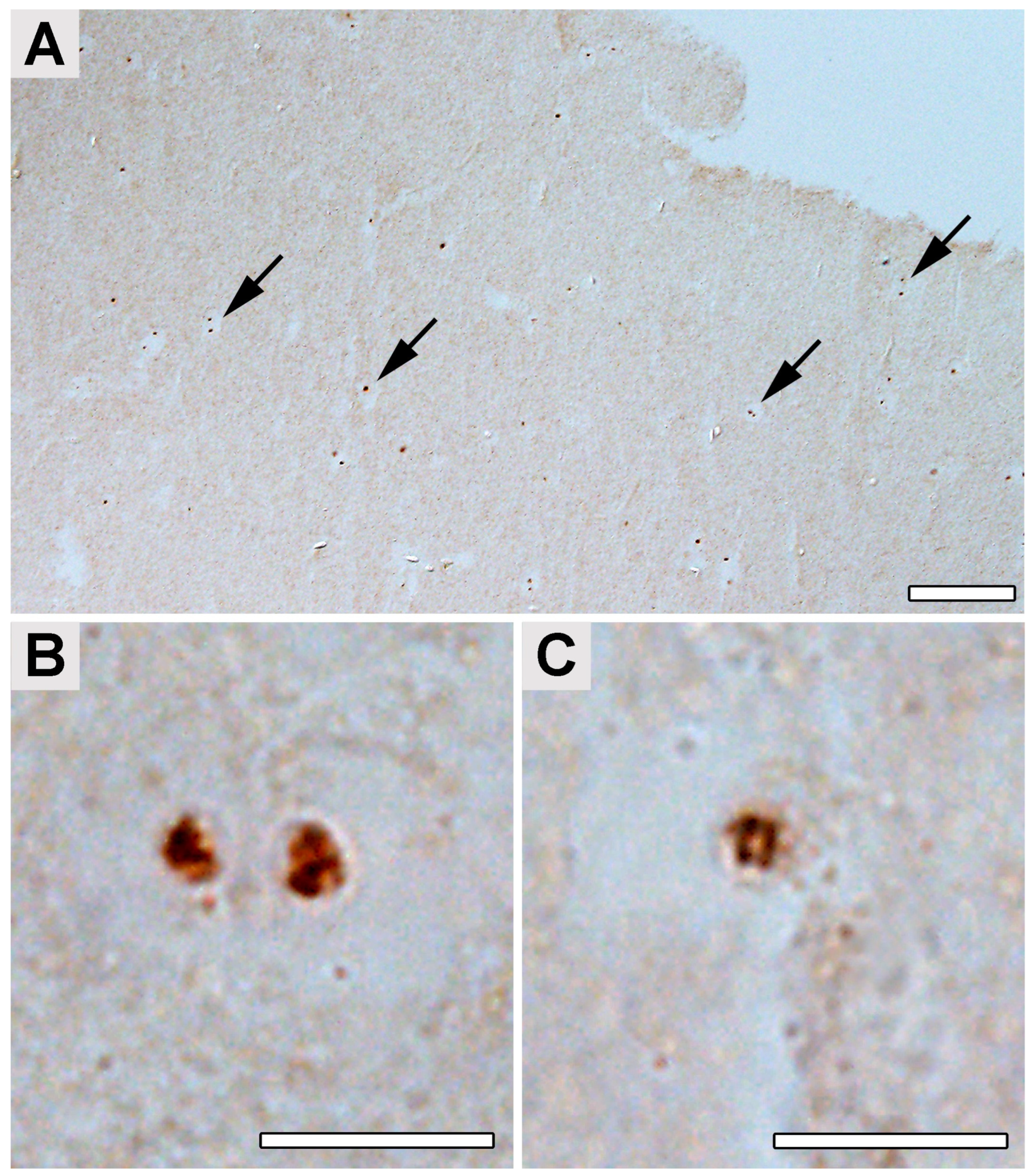
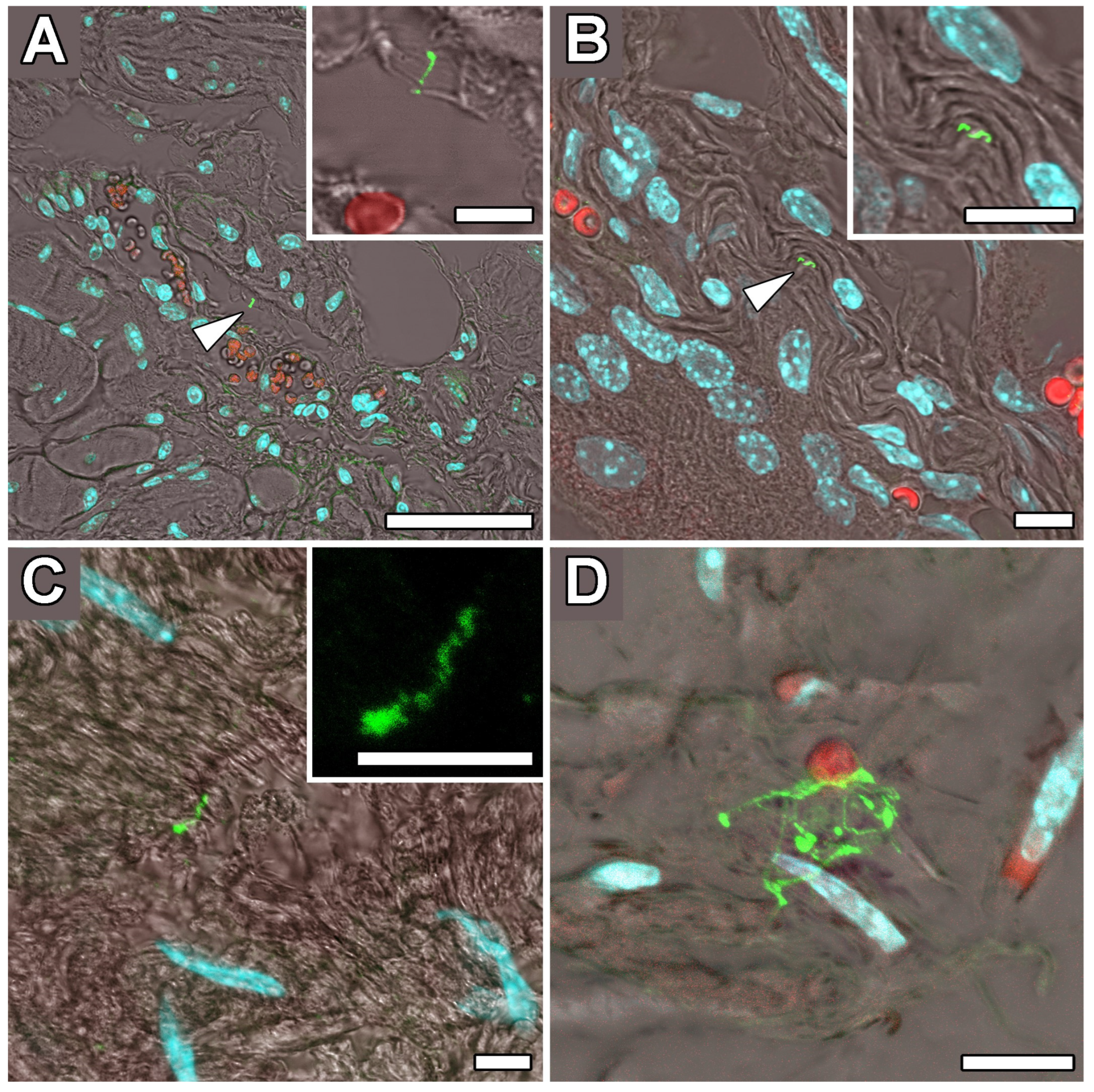
2.4. Occipital Lobe Tissue Exhibited Structures Consistent with Borrelia
3. Discussion
4. Materials and Methods
4.1. Ethical Statement
4.2. CSF and Blood Collection and Analysis
4.3. Brain Tissue Collection (Post-Mortem)
4.4. Analysis of Total DNA from Human Brain Tissues: Polymerase Chain Reactions (PCR)
4.5. Immunohistochemical Detection of Borrelia in the Paraplast Section of a Human Brain Autopsy and Infected Mouse Tissues
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Steere, A. Lyme disease. N. Engl. J. Med. 1989, 321, 586–596. [Google Scholar] [CrossRef]
- Bransfield, R.C. Neuropsychiatric Lyme borreliosis: An overview with a focus on a specialty psychiatrist’s clinical practice. Healthcare 2018, 6, 104. [Google Scholar] [CrossRef] [PubMed]
- Bransfield, R.C.; Aidlen, D.M.; Cook, M.J.; Javia, S. A clinical diagnostic system for late-stage neuropsychiatric Lyme borreliosis based upon an analysis of 100 patients. Healthcare 2020, 8, 13. [Google Scholar] [CrossRef]
- Fallon, B.A.; Madsen, T.; Erlangsen, A.; Benros, M.E. Lyme borreliosis and associations with mental disorders and suicidal behavior: A nationwide danish cohort study. Am. J. Psychiatry 2021, 178, 921–931. [Google Scholar] [CrossRef] [PubMed]
- Barthold, S.W.; Hodzic, E.; Imai, D.M.; Feng, S.; Yang, X.; Luft, B.J. Ineffectiveness of tigecycline against persistent Borrelia burgdorferi. Antimicrob. Agents Chemother. 2010, 54, 643–651. [Google Scholar] [CrossRef]
- Preac-Mursic, V.; Wilske, B.; Gross, B.; Weber, K.; Pfister, H.W.; Baumann, A.; Prokop, J. Survival of Borrelia burgdorferi in antibiotically treated patients with Lyme borreliosis. Infection 1989, 17, 355–359. [Google Scholar] [CrossRef] [PubMed]
- Shadick, N.A.; Phillips, C.B.; Logigian, E.L.; Steere, A.C.; Kaplan, R.F.; Berardi, V.P.; Duray, P.H.; Larson, M.G.; Wright, E.A.; Ginsburg, K.S.; et al. The long-term clinical outcomes of Lyme disease: A population-based retrospective cohort study. Ann. Intern. Med. 1994, 121, 560–567. [Google Scholar] [CrossRef] [PubMed]
- Fauvart, M.; De Groote, V.N.; Michiels, J. Role of persister cells in chronic infections: Clinical relevance and perspectives on anti-persister therapies. J. Med. Microbiol. 2011, 60 Pt 6, 699–709. [Google Scholar] [CrossRef]
- Nemeth, J.; Bernasconi, E.; Heininger, U.; Abbas, M.; Nadal, D.; Strahm, C.; Erb, S.; Zimmerli, S.; Furrer, H.; Delaloye, J.; et al. Update of the Swiss guidelines on post-treatment Lyme disease syndrome. Swiss Med. Wkly. 2016, 146, w14353. [Google Scholar] [CrossRef]
- Horowitz, R.I.; Freeman, P.R. Precision medicine: The role of the MSIDS model in defining, diagnosing, and treating chronic lyme disease/post treatment Lyme disease syndrome and other chronic illness: Part 2. Healthcare 2018, 6, 129. [Google Scholar] [CrossRef]
- Hastey, C.J.; Elsner, R.A.; Barthold, S.W.; Baumgarth, N. Delays and diversions mark the development of B cell responses to Borrelia burgdorferi Infection. J. Immunol. 2012, 188, 5612–5622. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Kybicova, K.; Vancova, M. Metamorphoses of Lyme disease spirochetes: Phenomenon of Borrelia persisters. Parasites Vectors 2019, 12, 237. [Google Scholar] [CrossRef]
- Sloupenska, K.; Koubkova, B.; Horak, P.; Hutyrova, B.; Racansky, M.; Mares, J.; Miklusova, M.; Schovanek, J.; Zapletalova, J.; Raska, M.; et al. Myositis autoantibodies in patients with suspected post-treatment Lyme disease syndrome. Life 2023, 13, 527. [Google Scholar] [CrossRef] [PubMed]
- Straubinger, R.K.; Straubinger, A.F.; Jacobson, R.H.; Chang, Y.; Summers, B.A.; Erb, H.N.; Appel, M.J.G. Two lessons from the canine model of Lyme disease: Migration of Borrelia burgdorferi in tissues and persistence after antibiotic treatment. J. Spirochetal Tick Borne Dis. 1997, 4, 24–31. [Google Scholar]
- Petzke, M.; Schwartz, I. Borrelia burgdorferi pathogenesis and the immune response. Clin. Lab. Med. 2015, 35, 745–764. [Google Scholar] [CrossRef]
- Tsao, J.I. Reviewing molecular adaptations of Lyme borreliosis spirochetes in the context of reproductive fitness in natural transmission cycles. Vet. Res. 2009, 40, 36. [Google Scholar] [CrossRef] [PubMed]
- Hyde, J.A. Borrelia burgdorferi keeps moving and carries on: A review of borrelial dissemination and invasion. Front. Immunol. 2017, 8, 114. [Google Scholar] [CrossRef]
- Livengood, J.A.; Gilmore, R.D. Invasion of human neuronal and glial cells by an infectious strain of Borrelia burgdorferi. Microbes Infect. 2006, 8, 2832–2840. [Google Scholar] [CrossRef]
- Ma, Y.; Sturrock, A.; Weis, J.J. Intracellular localization of Borrelia burgdorferi within human endothelial cells. Infect. Immun. 1991, 59, 671–678. [Google Scholar] [CrossRef]
- Montgomery, R.R.; Malawista, E.S. Entry of Borrelia burgdorferi into macrophages is end-on and leads to degradation in lysosomes. Infect. Immun. 1996, 64, 2867–2872. [Google Scholar] [CrossRef]
- Klempner, M.S.; Noring, R.; Rogers, R.A. Invasion of human skin fibroblasts by the Lyme disease spirochete, borrelia burgdorferi. J. Infect. Dis. 1993, 167, 1074–1081. [Google Scholar] [CrossRef]
- Gadila, S.K.G.; Rosoklija, G.; Dwork, A.J.; Fallon, B.A.; Embers, M.E. Detecting borrelia spirochetes: A case study with validation among autopsy specimens. Front. Neurol. 2021, 12, 707. [Google Scholar] [CrossRef]
- Hodzic, E.; Feng, S.; Holden, K.; Freet, K.J.; Barthold, S.W. Persistence of Borrelia burgdorferi following antibiotic treatment in mice. Antimicrob. Agents Chemother. 2008, 52, 1728–1736. [Google Scholar] [CrossRef] [PubMed]
- Hodzic, E.; Imai, D.; Feng, S.; Barthold, S.W. Resurgence of persisting non-cultivable Borrelia burgdorferi following antibiotic treatment in mice. PLoS ONE 2014, 9, e86907. [Google Scholar] [CrossRef]
- Straubinger, R.K.; Summers, A.B.; Chang, Y.F.; Appel, M.J. Persistence of Borrelia burgdorferi in experimentally infected dogs after antibiotic treatment. J. Clin. Microbiol. 1997, 35, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Embers, M.E.; Barthold, S.W.; Borda, J.T.; Bowers, L.; Doyle, L.; Hodzic, E.; Jacobs, M.B.; Hasenkampf, N.R.; Martin, D.S.; Narasimhan, S.; et al. Persistence of Borrelia burgdorferi in rhesus macaques following antibiotic treatment of disseminated infection. PLoS ONE 2012, 7, e29914. [Google Scholar] [CrossRef]
- Rizzoli, A.; Hauffe, H.C.; Carpi, G.; Vourc’h, I.G.; Neteler, M.; Rosà, R. Lyme borreliosis in europe. Eurosurveillance 2011, 16, 19906. [Google Scholar] [CrossRef] [PubMed]
- Hubálek, Z. Epidemiology of Lyme Borreliosis. In Lyme Borreliosis, Current Problems in Dermatology Volume 37; Lipsker, D., Jaulhac, B., Eds.; Karger: Basel, Switzerland, 2009; pp. 31–50. [Google Scholar]
- Gade, A.; Matin, T.; Rubenstein, R.; Robinson, C.A. Acrodermatitis Chronica Atrophicans. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. [Google Scholar]
- Janson, C.G. Blocking Borrelia in the brain. Sci. Transl. Med. 2016, 8, 324ec18. [Google Scholar] [CrossRef]
- Miklossy, J.; Khalili, K.; Gern, L.; Ericson, R.L.; Darekar, P.; Bolle, L.; Hurlimann, J.; Paster, B.J. Borrelia burgdorferi persists in the brain in chronic lyme neuroborreliosis and may be associated with Alzheimer disease. J. Alzheimers Dis. 2004, 6, 639–649; discussion 673–681. [Google Scholar] [CrossRef]
- Miklossy, J.; Kis, A.; Radenovic, A.; Miller, L.; Forro, L.; Martins, R.; Reiss, K.; Darbinian, N.; Darekar, P.; Mihaly, L.; et al. Beta-amyloid deposition and Alzheimer’s type changes induced by Borrelia spirochetes. Neurobiol. Aging 2006, 27, 228–236. [Google Scholar] [CrossRef]
- Liegner, K.B.; Rosenkilde, C.; Campbell, G.; Quan, T.; Dennis, D. Culture confirmed treatment failure of cefotaxime and minocycline in a case of Lyme meningoencephalomyelitis in the United States. In Proceedings of the Program and Abstracts of the 5th International Conference on Lyme Borreliosis, Arlington, VA, USA, 30 May–2 June 1992. [Google Scholar]
- Sapi, E.; Kasliwala, R.S.; Ismail, H.; Torres, J.P.; Oldakowski, M.; Markland, S.; Gaur, G.; Melillo, A.; Eisendle, K.; Liegner, K.B.; et al. The long-term persistence of Borrelia burgdorferi antigens and DNA in the tissues of a patient with Lyme disease. Antibiotics 2019, 8, 183. [Google Scholar] [CrossRef] [PubMed]
- Miklossy, J.; Kasas, S.; Zurn, A.D.; McCall, S.; Yu, S.; McGeer, P.L. Persisting atypical and cystic forms of Borrelia burgdorferi and local inflammation in Lyme neuroborreliosis. J. Neuroinflamm. 2008, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Piesman, J.; Gern, L. Lyme borreliosis in Europe and North America. Parasitology 2004, 129, S191–S220. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Grubhoffer, L.; Oliver, J.H., Jr. Updates on Borrelia burgdorferi sensu lato complex with respect to public health. Ticks Tick-Borne Dis. 2011, 2, 123–128. [Google Scholar] [CrossRef]
- Omotosho, Y.B.; Sherchan, R.; Ying, G.W.; Shayuk, M. A unique case of bannwarth syndrome in early disseminated Lyme disease. Cureus 2021, 13, e14680. [Google Scholar] [CrossRef]
- Garcia-Monco, J.C.; Benach, J.L. Lyme neuroborreliosis: Clinical outcomes, controversy, pathogenesis, and polymicrobial infections. Ann. Neurol. 2018, 85, 21–31. [Google Scholar] [CrossRef]
- Adams, Y.; Clausen, A.S.; Jensen, P.; Lager, M.; Wilhelmsson, P.; Henningson, A.J.; Lindgren, P.-E.; Faurholt-Jepsen, D.; Mens, H.; Kraiczy, P.; et al. 3D blood-brain barrier-organoids as a model for Lyme neuroborreliosis highlighting genospecies dependent organotropism. iScience 2022, 26, 105838. [Google Scholar] [CrossRef] [PubMed]
- Ford, L.; Tufts, D.M. Lyme neuroborreliosis: Mechanisms of B. burgdorferi infection of the nervous system. Brain Sci. 2021, 11, 789. [Google Scholar] [CrossRef] [PubMed]
- Embers, M.E.; Hasenkampf, N.R.; Jacobs, M.B.; Tardo, A.C.; Doyle-Meyers, L.A.; Philipp, M.T.; Hodzic, E. Variable manifestations, diverse seroreactivity and post-treatment persistence in non-human primates exposed to Borrelia burgdorferi by tick feeding. PLoS ONE 2017, 12, e0189071. [Google Scholar] [CrossRef]
- Marques, A.; Telford, S.R., 3rd; Turk, S.-P.; Chung, E.; Williams, C.; Dardick, K.; Krause, P.J.; Brandeburg, C.; Crowder, C.D.; Carolan, H.E.; et al. Xenodiagnosis to detect Borrelia burgdorferi infection: A first-in-human study. Clin. Infect. Dis. 2014, 58, 937–945. [Google Scholar] [CrossRef]
- Middelveen, M.J.; Sapi, E.; Burke, J.; Filush, K.R.; Franco, A.; Fesler, M.C.; Stricker, R.B. Persistent borrelia infection in patients with ongoing symptoms of Lyme disease. Healthcare 2018, 6, 33. [Google Scholar] [CrossRef]
- CDC Post-Treatment Lyme Disease Syndrome. 2017. Available online: https://www.cdc.gov/lyme/postLDS/index.html (accessed on 7 March 2018).
- Adrion, E.R.; Aucott, J.; Lemke, K.W.; Weiner, J.P. Health care costs, utilization and patterns of care following Lyme disease. PLoS ONE 2015, 10, e0116767. [Google Scholar] [CrossRef]
- Zhang, Y. Persisters, persistent infections and the Yin–Yang model. Emerg. Microbes Infect. 2014, 3, e3. [Google Scholar] [CrossRef]
- Feng, J.; Li, T.; Yee, R.; Yuan, Y.; Bai, C.; Cai, M.; Shi, W.; Embers, M.; Brayton, C.; Saeki, H.; et al. Stationary Phase Persister/Biofilm Microcolony of Borrelia burgdorferi Causes More Severe Disease in a Mouse Model of Lyme Arthritis: Implications for understanding persistence, Post-Treatment Lyme Disease Syndrome (PTLDS), and Treatment Failure. Published in Discovery Medicine on 28 March 2019. Available online: http://www.discoverymedicine.com/Jie-Feng/2019/03/persister-biofilm-microcolony-borrelia-burgdorferi-causes-severe-lyme-arthritis-in-mouse-model/ (accessed on 9 October 2023).
- Feng, J.; Auwaerter, P.G.; Zhang, Y. Drug combinations against Borrelia burgdorferi Persisters in vitro: Eradication achieved by using daptomycin, cefoperazone and doxycycline. PLoS ONE 2015, 10, e0117207. [Google Scholar] [CrossRef]
- Feng, J.; Shi, W.; Zhang, S.; Zhang, Y. Identification of new compounds with high activity against stationary phase Borrelia burgdorferi from the NCI compound collection. Emerg. Microbes Infect. 2015, 4, 1–15. [Google Scholar] [CrossRef]
- Lewis, K. Persister cells, dormancy and infectious disease. Nat. Rev. Microbiol. 2006, 5, 48–56. [Google Scholar] [CrossRef]
- Horowitz, R.I.; Freeman, P.R. The use of dapsone as a novel “persister” drug in the treatment of chronic Lyme disease/post treatment Lyme disease syndrome. J. Clin. Exp. Dermatol. 2016, 7, 345. [Google Scholar] [CrossRef]
- Horowitz, R.I.; Murali, K.; Gaur, G.; Freeman, P.R.; Sapi, E. Effect of dapsone alone and in combination with intracellular antibiotics against the biofilm form of B. burgdorferi. BMC Res. Notes 2020, 13, 455. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.I.; Freeman, P.R. Efficacy of short-term high dose pulsed dapsone combination therapy in the treatment of chronic Lyme disease/post-treatment Lyme disease syndrome (PTLDS) and associated co-infections: A report of three cases and literature review. Antibiotics 2022, 11, 912. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Gong, Z.; Montesano, D.; Glazer, E.; Liegner, K. “Repurposing” disulfiram in the treatment of Lyme disease and babesiosis: Retrospective review of first 3 years’ experience in one medical practice. Antibiotics 2020, 9, 868. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, A.B. Borrelia in the brains of patients dying with dementia. JAMA 1986, 256, 2195–2196. [Google Scholar] [CrossRef]
- Fallon, A.B.; Nields, A.J. Lyme disease: A neuropsychiatric illness. Am. J. Psychiatry 1994, 151, 1571–1583. [Google Scholar] [CrossRef]
- Miklossy, J. Biology and neuropathology of dementia in syphilis and Lyme disease. Handb. Clin. Neurol. 2008, 89, 825–844. [Google Scholar]
- Blanc, F.; Philippi, N.; Cretin, B.; Kleitz, C.; Berly, L.; Jung, B.; Kremer, S.; Namer, I.J.; Sellal, F.; Jaulhac, B.; et al. Lyme neuroborreliosis and dementia. J. Alzheimers Dis. 2014, 41, 1087–1093. [Google Scholar] [CrossRef]
- Kristoferitsch, W.; Aboulenein-Djamshidian, F.; Jecel, J.; Rauschka, H.; Rainer, M.; Stanek, G.; Fischer, P. Secondary dementia due to Lyme neuroborreliosis. Wien. Klin. Wochenschr. 2018, 130, 468–478. [Google Scholar] [CrossRef]
- Garcia-Toro, M.; Aguirre, I. Biopsychosocial model in depression revisited. Med. Hypotheses 2007, 68, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Senejani, A.G.; Maghsoudlou, J.; El-Zohiry, D.; Gaur, G.; Wawrzeniak, K.; Caravaglia, C.; Khatri, V.A.; MacDonald, A.; Sapi, E. Borrelia burgdorferi co-localizing with amyloid markers in Alzheimer’s disease brain tissues. J. Alzheimers Dis. 2022, 85, 889–903. [Google Scholar] [CrossRef] [PubMed]
- Di Domenico, E.G.; Cavallo, I.; Bordignon, V.; D’Agosto, G.; Pontone, M.; Trento, E.; Gallo, M.T.; Prignano, G.; Pimpinelli, F.; Toma, L.; et al. The emerging role of microbial biofilm in Lyme neuroborreliosis. Front. Neurol. 2018, 9, 1048. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.; Lebec, A.-M. The clinical and epidemiological profile of Lyme neuroborreliosis in Denmark 1985-1990: A prospective study of 187 patients with Borrelia burgdorferi specific intrathecal antibody production. Brain 1992, 115, 399–423. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, A.B. Spirochetal cyst forms in neurodegenerative disorders,…hiding in plain sight. Med. Hypotheses 2006, 67, 819–832. [Google Scholar] [CrossRef]
- Gruntar, I.; Malovrh, T.; Murgia, R.; Cinco, M. Conversion of Borrelia garinii cystic forms to motile spirochetes in vivo note. APMIS 2001, 109, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Murgia, R.; Cinco, M. Induction of cystic forms by different stress conditions in Borrelia burgdorferi. APMIS 2004, 112, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Cabello, F.C.; Godfrey, H.P.; Newman, S.A. Hidden in plain sight: Borrelia burgdorferi and the extracellular matrix. Trends Microbiol. 2007, 15, 350–354. [Google Scholar] [CrossRef] [PubMed]
- Tilly, K.; Rosa, P.A.; Stewart, P.E. Biology of Infection with Borrelia burgdorferi. Infect. Dis. Clin. N. Am. 2008, 22, 217–234. [Google Scholar] [CrossRef]
- Liang, F.T.; Brown, E.L.; Wang, T.; Iozzo, R.V.; Fikrig, E. Protective niche for Borrelia burgdorferi to evade humoral immunity. Am. J. Pathol. 2004, 165, 977–985. [Google Scholar] [CrossRef]
- Nordstrand, A.; Barbour, A.G.; Bergström, S. Borrelia pathogenesis research in the post-genomic and post-vaccine era. Curr. Opin. Microbiol. 2000, 3, 86–92. [Google Scholar] [CrossRef]
- Berndtson, K. Review of evidence for immune evasion and persistent infection in Lyme disease. Int. J. Gen. Med. 2013, 6, 291–306. [Google Scholar] [CrossRef] [PubMed]
- Liang, F.T.; Yan, J.; Mbow, M.L.; Sviat, S.L.; Gilmore, R.D.; Mamula, M.; Fikrig, E. Borrelia burgdorferi changes its surface antigenic expression in response to host immune responses. Infect. Immun. 2004, 72, 5759–5767. [Google Scholar] [CrossRef]
- Pachner, A.; Basta, J.; Delaney, E.; Hulinska, D. Localisation of Borrelia burgdorferi in murine Lyme borreliosis by electron microscopy. Am. J. Trop. Med. Hyg. 1995, 52, 128–133. [Google Scholar] [CrossRef]
- Girschick, H.J.; Huppertz, H.I.; Krenn, V.; Karch, H.; Rüssman, H. Intracellular persistence of Borrelia burgdorferi in human synovial cells. Rheumatol. Int. 1996, 16, 125–132. [Google Scholar] [CrossRef]
- Rupprecht, T.A.; Koedel, U.; Fingerle, V.; Pfister, H.-W. The pathogenesis of Lyme neuroborreliosis: From infection to inflammation. Mol. Med. 2008, 14, 205–212. [Google Scholar] [CrossRef]
- Rudenko, N.; Golovchenko, M.; Hönig, V.; Mallátová, N.; Krbková, L.; Mikulášek, P.; Fedorova, N.; Belfiore, N.M.; Grubhoffer, L.; Lane, R.S.; et al. Detection of Borrelia burgdorferi sensu stricto ospC alleles associated with human Lyme borreliosis worldwide in non-human-biting tick Ixodes affinis and rodent hosts in Southeastern United States. Appl. Environ. Microbiol. 2013, 79, 1444–1453. [Google Scholar] [CrossRef]
- Qiu, W.-G.; Bruno, J.F.; McCaig, W.D.; Xu, Y.; Livey, I.; Schriefer, M.E.; Luft, B.J. Wide Distribution of a High-Virulence Borrelia burgdorferi clone in Europe and North America. Emerg. Infect. Dis. 2008, 14, 1097–1104. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hanincovaá, K.; Liveris, D.; Sandigursky, S.; Wormser, G.P.; Schwartz, I. Borrelia burgdorferi sensu stricto is clonal in patients with early lyme borreliosis. Appl. Environ. Microbiol. 2008, 74, 5008–5014. [Google Scholar] [CrossRef] [PubMed]
- Strle, K.; Jones, K.L.; Drouin, E.E.; Li, X.; Steere, A.C. Borrelia burgdorferi RST1 (OspC type A) genotype is associated with greate rinflammation and more severe Lyme disease. Am. J. Pathol. 2011, 178, 2726–2739. [Google Scholar] [CrossRef] [PubMed]
- Seinost, G.; Dykhuizen, D.E.; Dattwyler, R.J.; Golde, W.T.; Dunn, J.J.; Wang, I.-N.; Wormser, G.P.; Schriefer, M.E.; Luft, B.J. Four clones of Borrelia burgdorferi sensu stricto cause invasive infection in humans. Infect. Immun. 1999, 67, 3518–3524. [Google Scholar] [CrossRef] [PubMed]
- Brisson, D.; Baxamusa, N.; Schwartz, I.; Wormser, G.P. Biodiversity of Borrelia burgdorferi strains in tissues of Lyme disease patients. PLoS ONE 2011, 6, e22926. [Google Scholar] [CrossRef]
- Dykhuizen, D.E.; Sandigursky, S.; Nadelman, R.B.; Nowakowski, J.; Brisson, D.; Wormser, G.P.; Schwartz, I. The propensity of different Borrelia burgdorferi sensu stricto genotypes to cause disseminated infections in humans. Am. J. Trop. Med. Hyg. 2008, 78, 806–810. [Google Scholar] [CrossRef]
- Ivanova, L.; Christova, I.; Neves, V.; Aroso, M.; Meirelles, L.; Brisson, D.; Gomes-Solecki, M. Comprehensive seroprofiling of sixteen B. burgdorferi OspC: Implications for Lyme disease diagnostics design. Clin. Immunol. 2009, 132, 393–400. [Google Scholar] [CrossRef]
- Casselli, T.; Divan, A.; Vomhof-DeKrey, E.E.; Tourand, Y.; Pecoraro, H.L.; Brissette, C.A. A murine model of Lyme disease demonstrates that Borrelia burgdorferi colonizes the dura mater and induces inflammation in the central nervous system. PLoS Pathog. 2021, 17, e1009256. [Google Scholar] [CrossRef]
- Divan, A.; Casselli, T.; Narayanan, S.A.; Mukherjee, S.; Zawieja, D.C.; Watt, J.A.; Brissette, C.A.; Newell-Rogers, M.K. Borrelia burgdorferi adhere to blood vessels in the dura mater and are associated with increased meningeal T cells during murine disseminated borreliosis. PLoS ONE 2018, 13, e0196893. [Google Scholar] [CrossRef] [PubMed]
- Persidsky, Y.; Ramirez, S.H.; Haorah, J.; Kanmogne, G.D. Blood–brain barrier: Structural components and function under physiologic and pathologic conditions. J. Neuroimmune Pharmacol. 2006, 1, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Dupin, N.; Lecuyer, H.; Carlotti, A.; Poyart, C.; Coureuil, M.; Chanal, J.; Schmitt, A.; Vacher-Lavenu, M.-C.; Taha, M.-K.; Nassif, X.; et al. Chronic meningococcemia cutaneous lesions involve meningococcal perivascular invasion through the remodeling of endothelial barriers. Clin. Infect. Dis. 2012, 54, 1162–1165. [Google Scholar] [CrossRef]
- Adams, Y.; Olsen, R.W.; Bengtsson, A.; Dalgaard, N.; Zdioruk, M.; Satpathi, S.; Behera, P.K.; Sahu, P.K.; Lawler, S.E.; Qvortrup, K.; et al. Plasmodium falciparum erythrocyte membrane protein 1 variants induce cell swelling and disrupt the blood–brain barrier in cerebral malaria. J. Exp. Med. 2021, 218, e20201266. [Google Scholar] [CrossRef]
- Disson, O.; Lecuit, M. Targeting of the central nervous system by Listeria monocytogenes. Virulence 2012, 3, 213–221. [Google Scholar] [CrossRef]
- Hussain, B.; Fang, C.; Chang, J. Blood–brain barrier breakdown: An emerging biomarker of cognitive impairment in normal aging and dementia. Front. Neurosci. 2021, 15, 688090. [Google Scholar] [CrossRef]
- Al-Bachari, S.; Naish, J.H.; Parker, G.J.M.; Emsley, H.C.A.; Parkes, L.M. Blood–brain barrier leakage is increased in parkinson’s disease. Front. Physiol. 2020, 11, 593026. [Google Scholar] [CrossRef]
- Horowitz, I.R.; Freeman, P.R. Precision medicine: Retrospective chart review and data analysis of 200 patients on dapsone combination therapy for chronic Lyme disease/post-treatment Lyme disease syndrome: Part 1. Int. J. Gen. Med. 2019, 12, 101–119. [Google Scholar] [CrossRef] [PubMed]
- Horowitz, R.I.; Freeman, P.R. Efficacy of double-dose dapsone combination therapy in the treatment of chronic Lyme disease/post-treatment Lyme disease syndrome (PTLDS) and associated co-infections: A report of three cases and retrospective chart review. Antibiotics 2020, 9, 725. [Google Scholar] [CrossRef] [PubMed]
- Cadavid, D.; Barbour, A.G. Neuroborreliosis during relapsing fever: Review of the clinical manifestations, pathology, and treatment of infections in humans and experimental animals. Clin. Infect. Dis. 1998, 26, 151–164. [Google Scholar] [CrossRef]
- Sergent, A.; Richard, H. Spirochaeta hispanica peut persister plus de deux ans dans le cerveau d’un cobaye inocule experimentalement. Arch. Inst. Pasteur Alger. 1942, 20, 293–297. [Google Scholar]
- Sergent, E. Persistence de Spirochaete hispanica pendant trois ans dans le cerveau d’un cobaye. 3e note. Arch. Inst. Pasteur Alger. 1945, 23, 245–248. [Google Scholar] [PubMed]
- Schauder, H. Zur frage der spirochatenpersistenz in zentralnervensystem und ihrer chemotherapeutischen beeinflussbarkeit bei experimenteller rekurrens. Arch. Fuer Schiffs-Und Trop. Hyg. 1928, 32, 1–13. [Google Scholar]
- Adler, S.; Ashbel, R. Observations on Spirochaete sogdianum Nicolle and Anderson, 1928, in laboratory animals. Ann Trop Med Parasitol 1937, 31, 89–104. [Google Scholar] [CrossRef]
- Steiner, G.; Steinfeld, J.; Schauder, H. Zur frage der spirochatenpersistenzim zentralnervensystem bei experimenteller recurrens. Klin. Wochenschr. 1925, 4, 2288–2289. [Google Scholar] [CrossRef]
- Sergent, A. Fievre recurrente a Spirochaeta hispanicum en Algerie. Transmission par le rhipicephale du chien. Premunition. Serum de convalescents. Ann. Parasitol. Hum. Comparée 1938, 61, 217–254. [Google Scholar]
- Najera, L. Sobre la conservacion del Spirocheta hispanica. Boletın Real. Soc. Esp. Hist. Nat. Actas 1943, 41, 371–374. [Google Scholar]
- Steiner, G.; Steinfeld, J. Experimentelle untersuchungen zur pathologie und therapie der spirochatenkrankheiten. I. Die immunitatsverhaltnisse des gehirns und des serums in ihren beziehungen zueinander bei experimenteller recurrens. Klin. Wochenschr. 1925, 4, 1995–1997. [Google Scholar] [CrossRef]
- Cadavid, D.; Bundoc, V.; Barbour, A.G. Experimental infection of the mouse brain by a relapsing fever borrelia species: A molecular analysis. J. Infect. Dis. 1993, 168, 143–151. [Google Scholar] [CrossRef]
- Merritt, H.H.; Adams, R.D.; Solomon, H.C. Neurosyphilis; Oxford University Press: London, UK, 1946. [Google Scholar]
- Vinken, P.J.; Bruyn, G.W. Handbook of Neurology; Elsevier: Amsterdam, The Netherlands; New York, NY, USA, 1978. [Google Scholar]
- Miklossy, J. Alzheimer’s disease—A spirochetosis? Neuroreport 1993, 4, 841–848. [Google Scholar] [CrossRef]
- Loa, C.C.; Adelson, M.E.; Mordechai, E.; Lobrinus, J.A.; Fallon, B.A.; Tilton, R.C. Lyme disease associated with Alzheimer’s disease. Curr. Microbiol. 2006, 52, 330–332. [Google Scholar] [CrossRef]
- MacDonald, A.B.; Miranda, J.M. Concurrent neocortical borreliosis and Alzheimer’s disease. Hum. Pathol. 1987, 18, 759–761. [Google Scholar] [CrossRef] [PubMed]
- McDonald, A.B. Concurrent neocortical borreliosis and Alzheimer’s disease. Demonstration of a spirochetal cyst form. Ann. N. Y. Acad. Sci. 1988, 539, 468–470. [Google Scholar] [CrossRef]
- Stricker, R.B. Counterpoint: Long-term antibiotic therapy improves persistent symptoms associated with lyme disease. Clin. Infect. Dis. 2007, 45, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Margos, G.; Gatewood, A.G.; Aanensen, D.M.; Hanincová, K.; Terekhova, D.; Vollmer, S.A.; Cornet, M.; Piesman, J.; Donaghy, M.; Bormane, A.; et al. MLST of housekeeping genes captures geographic population structure and suggests a European origin of Borrelia burgdorferi. Proc. Natl. Acad. Sci. USA 2008, 105, 8730–8735. [Google Scholar] [CrossRef]
- Golovchenko, M.; Sima, R.; Hajdusek, O.; Grubhoffer, L.; Oliver, J.H.; Rudenko, N. Invasive potential of Borrelia burgdorferi sensu stricto ospC type L strains increases the possible disease risk to humans in the regions of their distribution. Parasites Vectors 2014, 7, 538. [Google Scholar] [CrossRef] [PubMed]
- Rudenko, N.; Golovchenko, M.; Vancova, M.; Clark, K.; Grubhoffer, L.; Oliver, J. Isolation of live Borrelia burgdorferi sensu lato spirochaetes from patients with undefined disorders and symptoms not typical for Lyme borreliosis. Clin. Microbiol. Infect. 2015, 22, 267.e9–267.e15. [Google Scholar] [CrossRef]
- Bunikis, J.; Garpmo, U.; Tsao, J.; Berglund, J.; Fish, D.; Barbour, A.G. Sequence typing reveals extensive strain diversity of the Lyme borreliosis agents Borrelia burgdorferi in north america and borrelia afzelii in europe. Microbiology 2004, 150 Pt 6, 1741–1755. [Google Scholar] [CrossRef]
- Clark, K.; Hendricks, A.; Burge, D. Molecular identification and analysis of Borrelia burgdorferi sensu lato in lizards in the southeastern united states. Appl. Environ. Microbiol. 2005, 71, 2616–2625. [Google Scholar] [CrossRef]
- Levine, J.D.; Sauman, I.; Imbalzano, M.; Reppert, S.M.; Jackson, F. Period protein from the giant silkmoth antheraea pernyi functions as a circadian clock element in drosophila melanogaster. Neuron 1995, 15, 147–157. [Google Scholar] [CrossRef][Green Version]
- Yuste, R.A.; Muenkel, M.; Axarlis, K.; Benito, M.J.G.; Reuss, A.; Blacker, G.; Tal, M.C.; Kraiczy, P.; Bastounis, E.E. Borrelia burgdorferi modulates the physical forces and immunity signaling in endothelial cells. iScience 2022, 25, 104793. [Google Scholar] [CrossRef]
- Sapiro, A.L.; Hayes, B.M.; Volk, R.F.; Zhang, J.Y.; Brooks, D.M.; Martyn, C.; Radkov, A.; Zhao, Z.; Kinnersley, M.; Secor, P.R.; et al. Longitudinal map of transcriptome changes in the Lyme pathogen Borrelia burgdorferi during tick-borne transmission. eLife 2023, 12, RP86636. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Preac-Mursic, V.; Wilske, B.; Schierz, G. European Borrelia burgdorferi isolated from humans and ticks. Culture conditions and antibiotic susceptibility. Zentralbl. Bakteriol. Mikrobiol. Hyg. 1986, 263, 112–118. [Google Scholar] [CrossRef] [PubMed]
| Gene Sample | fla B 388 bp | ospC 617 bp | clpA 706 bp | clpX 721 bp | nifS 629 bp | rplB 720 bp | pepX 666 bp | pyrG 687 bp | uvrA 677 bp |
|---|---|---|---|---|---|---|---|---|---|
| 1—temporal lobe | Bb s.s. | Bb s.s. | + | + | + | + | + | ||
| 2—choroid plexus | Bb s.s. | + | + | + | + | + | + | ||
| 3—occipital lobe | Bb s.s. | + | + | + | + | + | + | ||
| 4—frontal lobe | Bb s.s. | Bb s.s. | + | + | + | + | + | + | |
| 5—parietal lobe | Bb s.s. | Bb s.s. | + | + | + | + | + | + | + |
| 6—basal ganglia | B. gar | B. gar | + | + | + | + | + | ||
| 7—cerebellum | B. gar | + | + | + | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Golovchenko, M.; Opelka, J.; Vancova, M.; Sehadova, H.; Kralikova, V.; Dobias, M.; Raska, M.; Krupka, M.; Sloupenska, K.; Rudenko, N. Concurrent Infection of the Human Brain with Multiple Borrelia Species. Int. J. Mol. Sci. 2023, 24, 16906. https://doi.org/10.3390/ijms242316906
Golovchenko M, Opelka J, Vancova M, Sehadova H, Kralikova V, Dobias M, Raska M, Krupka M, Sloupenska K, Rudenko N. Concurrent Infection of the Human Brain with Multiple Borrelia Species. International Journal of Molecular Sciences. 2023; 24(23):16906. https://doi.org/10.3390/ijms242316906
Chicago/Turabian StyleGolovchenko, Maryna, Jakub Opelka, Marie Vancova, Hana Sehadova, Veronika Kralikova, Martin Dobias, Milan Raska, Michal Krupka, Kristyna Sloupenska, and Natalie Rudenko. 2023. "Concurrent Infection of the Human Brain with Multiple Borrelia Species" International Journal of Molecular Sciences 24, no. 23: 16906. https://doi.org/10.3390/ijms242316906
APA StyleGolovchenko, M., Opelka, J., Vancova, M., Sehadova, H., Kralikova, V., Dobias, M., Raska, M., Krupka, M., Sloupenska, K., & Rudenko, N. (2023). Concurrent Infection of the Human Brain with Multiple Borrelia Species. International Journal of Molecular Sciences, 24(23), 16906. https://doi.org/10.3390/ijms242316906






