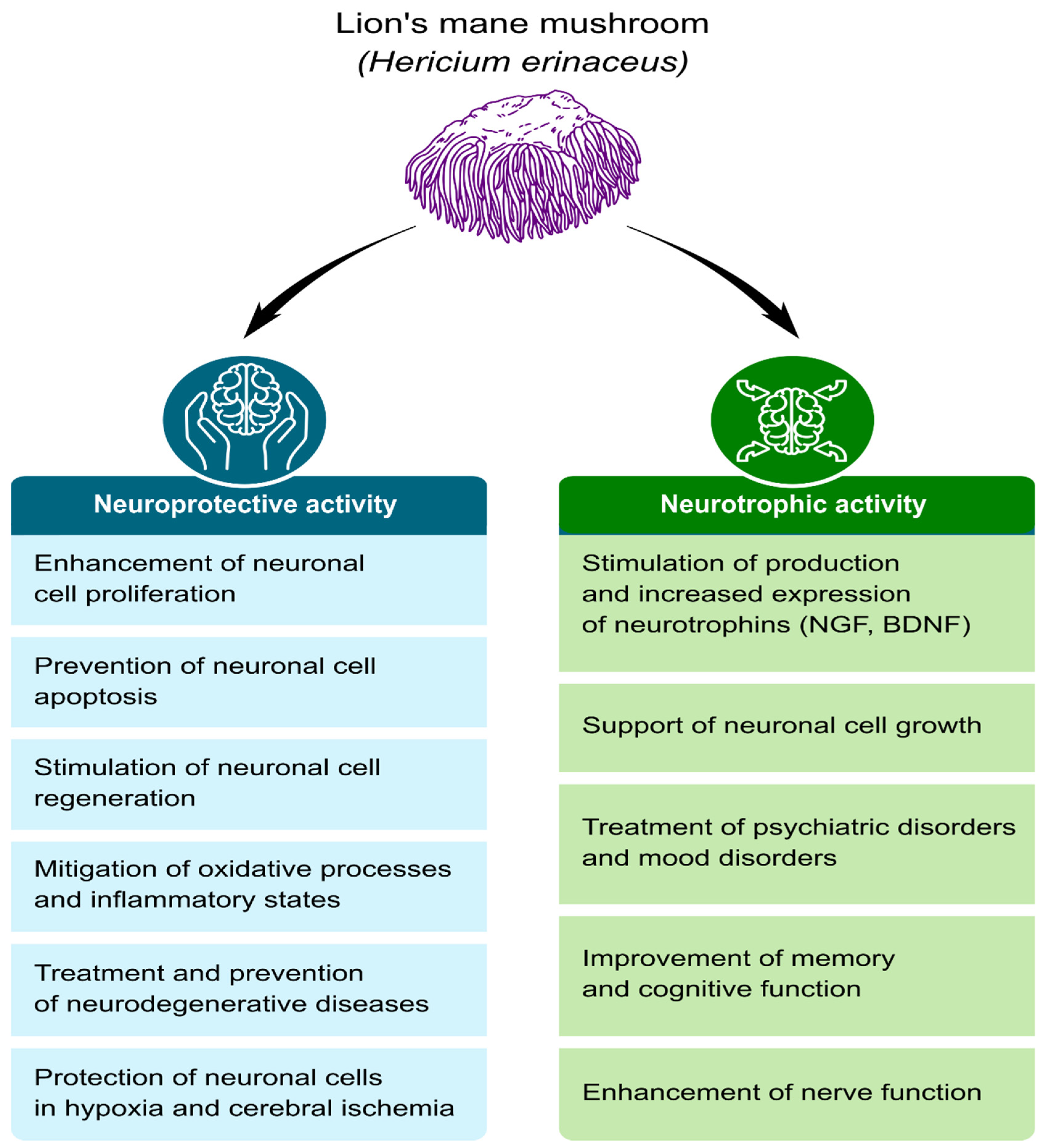
Neurotrophic and Neuroprotective Effects of Hericium erinaceus
Abstract
1. Introduction
2. Neurotrophins
2.1. NGF and BDNF
2.2. The Mechanisms of NGF and BDNF Activity
3. Activities of Components Isolated from Hericium erinaceus
3.1. Polysaccharides
3.2. Erinacines
3.3. Ergothioneine
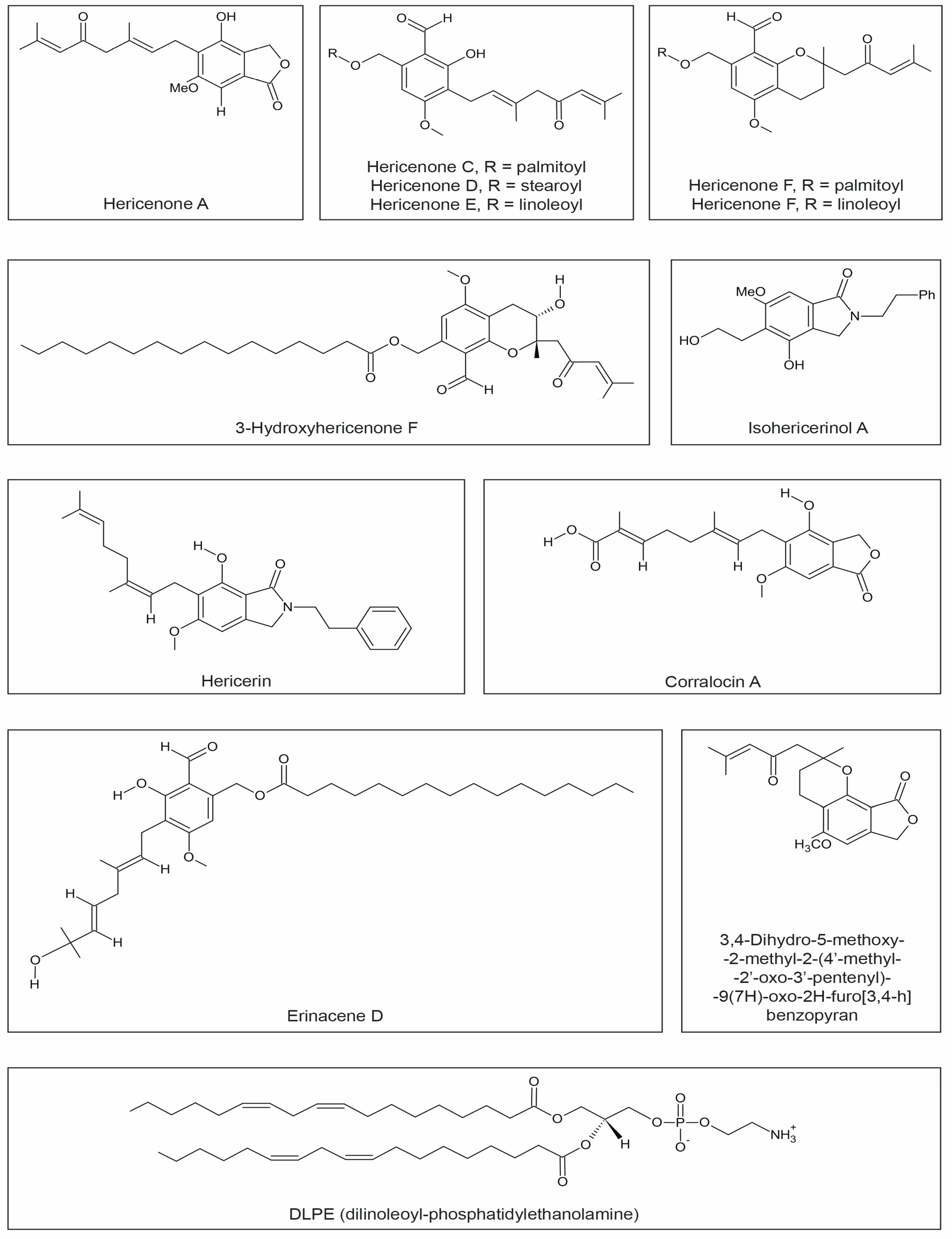
3.4. Hericenones
3.5. Dilinoleyl-Phosphatidylethanolamine (DLPE)
3.6. Other Compounds
4. The Neuroprotective and Neurotrophic Potential of H. erinaceus Components
4.1. Growth, Regeneration, and Protection of Nerve Cells
4.2. Cellular Aging Inhibition
4.3. Improvement of Cognitive Function
4.4. Anti-Neuroinflammatory and Antioxidant Effects
5. Toxicology Studies
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mattson, M.P. Neuroprotective signalling and the ageing brain: Take away my food and let me run. Brain Res. 2000, 886, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Lai, P.L.; Naidu, M.; Sabaratnam, V.; Wong, K.H.; David, R.P.; Kuppusamy, U.R.; Abdullah, N.; Malek, S.N. Neurotrophic properties of the Lion’s Mane medicinal mushroom, Hericium erinaceus (higher Basidiomycetes) from Malaysia. Int. J. Med. Mushrooms 2013, 15, 539–554. [Google Scholar] [CrossRef] [PubMed]
- Roda, E.; Priori, E.C.; Ratto, D.; De Luca, F.; Di Iorio, C.; Angelone, P.; Locatelli, C.A.; Desiderio, A.; Goppa, L.; Savino, E.; et al. Neuroprotective Metabolites of Hericium erinaceus Promote Neuro-Healthy Aging. Int. J. Mol. Sci. 2021, 22, 6379. [Google Scholar] [CrossRef]
- Brandalise, F.; Roda, E.; Ratto, D.; Goppa, L.; Gargano, M.L.; Cirlincione, F.; Priori, E.C.; Venuti, M.T.; Pastorelli, E.; Savino, E.; et al. Hericium erinaceus in Neurodegenerative Diseases: From Bench to Bedside and Beyond, How Far from the Shoreline? J. Fungi 2023, 9, 551. [Google Scholar] [CrossRef] [PubMed]
- Sokół, S.; Golak-Siwulska, I.; Sobieralski, K.; Siwulski, M.; Górka, K. Biology, cultivation, and medicinal functions of the mushroom Hericium erinaceum. Acta Mycol. 2016, 50, 1069. [Google Scholar] [CrossRef]
- Thongbai, B.; Rapior, S.; Hyde, K.D.; Wittstein, K.; Stadler, M. Hericium erinaceus, an amazing medicinal mushroom. Mycol. Prog. 2015, 14, 91. [Google Scholar] [CrossRef]
- Chatterjee, S.; Sarma, M.K.; Deb, U.; Steinhauser, G.; Walther, C.; Gupta, D.K. Mushrooms: From nutrition to mycoremediation. Environ. Sci. Pollut. Res. 2017, 24, 19480–19493. [Google Scholar] [CrossRef]
- Sabaratnam, V.; Phan, C.-W. Neuroactive Components of Culinary and Medicinal Mushrooms with Potential to Mitigate Age-Related Neurodegenerative Diseases. In Discovery and Development of Neuroprotective Agents from Natural Products; Nature Reviews Drug Discovery; Elsevier: Amsterdam, The Netherlands, 2018; pp. 401–413. [Google Scholar]
- Su, W.T.; Shih, Y.A. Nanofiber containing carbon nanotubes enhanced PC12 cell proliferation and neuritogenesis by electrical stimulation. Biomed. Mater. Eng. 2015, 26, 189–195. [Google Scholar] [CrossRef]
- Rahman, M.A.; Abdullah, N.; Aminudin, N. Inhibitory effect on in vitro LDL oxidation and HMG Co-a reductase activity of the liquid-liquid partitioned fractions of Hericium erinaceus (Bull.) persoon (Lion’s Mane Mushroom). BioMed Res. Int. 2014, 2014, 828149. [Google Scholar] [CrossRef]
- Mori, K.; Ouchi, K.; Hirasawa, N. The Anti-Inflammatory Effects of Lion’s Mane Culinary-Medicinal Mushroom, Hericium erinaceus (Higher Basidiomycetes) in a Coculture System of 3T3-L1 Adipocytes and RAW264 Macrophages. Int. J. Med. Mushrooms 2015, 17, 609–618. [Google Scholar] [CrossRef]
- Yang, B.K.; Park, J.B.; Song, C.H. Hypolipidemic effect of an exo-biopolymer produced from a submerged mycelial culture of Hericium erinaceus. Biosci. Biotechnol. Biochem. 2003, 67, 1292–1298. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Kikuchi, H.; Obara, Y.; Iwashita, M.; Azumi, Y.; Kinugasa, S.; Inatomi, S.; Oshima, Y.; Nakahata, N. Inhibitory effect of hericenone B from Hericium erinaceus on collagen-induced platelet aggregation. Phytomedicine 2010, 17, 1082–1085. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, R.N.; Tang, Q.J.; Zhang, J.S.; Yang, Y.; Shang, X.-D. A new diterpene from the fungal mycelia of Hericium erinaceus. Phytochem. Lett. 2015, 11, 151–156. [Google Scholar] [CrossRef]
- Kawagishi, H.; Zhuang, C. Compounds for dementia from Hericium erinaceum. Drugs Future 2008, 33, 149–155. [Google Scholar] [CrossRef]
- Liang, B.; Guo, Z.; Xie, F.; Zhao, A. Antihyperglycemic and antihyperlipidemic activities of aqueous extract of Hericium erinaceus in experimental diabetic rats. BMC Complement. Altern. Med. 2013, 13, 253. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, S.-l.; Wang, A.-h.; Sun, Z.-c.; Zhao, Y.-f.; Xu, Y.-t.; He, Y.-l. Protective effect of ethanol extracts of Hericium erinaceus on alloxan-induced diabetic neuropathic pain in rats. Evid.-Based Complement. Altern. Med. 2015, 2015, 595480. [Google Scholar]
- Wang, J.C.; Hu, S.H.; Su, C.H.; Lee, T.M. Antitumor and immunoenhancing activities of polysaccharide from culture broth of Hericium spp. Kaohsiung J. Med. Sci. 2001, 17, 461–467. [Google Scholar]
- Zhang, F.; Lu, H.; Zhang, X. Erinacerins, Novel Glioma Inhibitors from Hericium erinaceus, Induce Apoptosis of U87 Cells through Bax/Capase-2 Pathway. Anticancer Agents Med. Chem. 2020, 20, 2082–2088. [Google Scholar] [CrossRef]
- Lee, K.F.; Chen, J.H.; Teng, C.C.; Shen, C.H.; Hsieh, M.C.; Lu, C.C.; Lee, K.C.; Lee, L.Y.; Chen, W.P.; Chen, C.C.; et al. Protective Effects of Hericium erinaceus Mycelium and Its Isolated Erinacine A against Ischemia-Injury-Induced Neuronal Cell Death via the Inhibition of iNOS/p38 MAPK and Nitrotyrosine. Int. J. Mol. Sci. 2014, 15, 15073–15089. [Google Scholar] [CrossRef]
- Mori, K.; Inatomi, S.; Ouchi, K.; Azumi, Y.; Tuchida, T. Improving effects of the mushroom Yamabushitake (Hericium erinaceus) on mild cognitive impairment: A double-blind placebo-controlled clinical trial. Phytother. Res. 2009, 23, 367–372. [Google Scholar] [CrossRef]
- Tzeng, T.-T.; Chen, C.-C.; Lee, L.-Y.; Chen, W.-P.; Lu, C.-K.; Shen, C.-C.; Huang, F.C.-Y.; Chen, C.-C.; Young-Ji, S. Erinacine A-enriched Hericium erinaceus mycelium ameliorates Alzheimer’s disease-related pathologies in APPswe/PS1dE9 transgenic mice. J. Biomed. Sci. 2016, 23, 49. [Google Scholar]
- Lee, K.F.; Tung, S.Y.; Teng, C.C.; Shen, C.H.; Hsieh, M.C.; Huang, C.Y.; Lee, K.C.; Lee, L.Y.; Chen, W.P.; Chen, C.C.; et al. Post-Treatment with Erinacine A, a Derived Diterpenoid of H. erinaceus, Attenuates Neurotoxicity in MPTP Model of Parkinson’s Disease. Antioxidants 2020, 9, 137. [Google Scholar] [CrossRef] [PubMed]
- Chan, Y.C.; Chen, C.C.; Lee, L.Y.; Chen, W.P. Active Substances for Preventing Hearing Deterioration, the Composition Containing the Active Substances, and the Preparation Method Thereof. U.S. Patent 10,405,504, 10 September 2019. [Google Scholar]
- Yao, W.; Zhang, J.C.; Dong, C.; Zhuang, C.; Hirota, S.; Inanaga, K.; Hashimoto, K. Effects of amycenone on serum levels of tumor necrosis factor-α, interleukin-10, and depression-like behavior in mice after lipopolysaccharide administration. Pharmacol. Biochem. Behav. 2015, 136, 7–12. [Google Scholar] [CrossRef]
- Chiu, C.H.; Chyau, C.C.; Chen, C.C.; Lee, L.Y.; Chen, W.P.; Liu, J.L.; Lin, W.H.; Mong, M.C. Erinacine A-Enriched Hericium erinaceus Mycelium Produces Antidepressant-Like Effects through Modulating BDNF/PI3K/Akt/GSK-3β Signaling in Mice. Int. J. Mol. Sci. 2018, 19, 341. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.; Kim, H.G.; Kim, J.Y.; Kim, S.Y.; Cho, K.O. Hericium erinaceus Extract Reduces Anxiety and Depressive Behaviors by Promoting Hippocampal Neurogenesis in the Adult Mouse Brain. J. Med. Food 2018, 21, 174–180. [Google Scholar] [CrossRef] [PubMed]
- Chong, P.S.; Fung, M.L.; Wong, K.H.; Lim, L.W. Therapeutic Potential of Hericium erinaceus for Depressive Disorder. Int. J. Mol. Sci. 2019, 21, 163. [Google Scholar] [CrossRef]
- Martínez-Mármol, R.; Chai, Y.; Conroy, J.N.; Khan, Z.; Hong, S.M.; Kim, S.B.; Gormal, R.S.; Lee, D.H.; Lee, J.K.; Coulson, E.J.; et al. Hericerin derivatives activates a pan-neurotrophic pathway in central hippocampal neurons converging to ERK1/2 signalling enhancing spatial memory. J. Neurochem. 2023, 165, 791–808. [Google Scholar] [CrossRef]
- Friedman, M. Chemistry, Nutrition, and Health-Promoting Properties of Hericium erinaceus (Lion’s Mane) Mushroom Fruiting Bodies and Mycelia and Their Bioactive Compounds. J. Agric. Food Chem. 2015, 63, 7108–7123. [Google Scholar] [CrossRef]
- Ratto, D.; Corana, F.; Mannucci, B.; Priori, E.C.; Cobelli, F.; Roda, E.; Ferrari, B.; Occhinegro, A.; Di Iorio, C.; De Luca, F.; et al. Hericium erinaceus Improves Recognition Memory and Induces Hippocampal and Cerebellar Neurogenesis in Frail Mice during Aging. Nutrients 2019, 11, 715. [Google Scholar] [CrossRef]
- Wong, K.H.; Naidu, M.; David, R.P.; Bakar, R.; Sabaratnam, V. Neuroregenerative potential of lion’s mane mushroom, Hericium erinaceus (Bull.: Fr.) Pers. (higher Basidiomycetes), in the treatment of peripheral nerve injury (review). Int. J. Med. Mushrooms 2012, 14, 427–446. [Google Scholar] [CrossRef]
- Hazekawa, M.; Kataoka, A.; Hayakawa, K.; Uchimasu, T.; Furuta, R.; Irie, K.; Akitake, Y.; Yoshida, M.; Fujioka, T.; Egashira, N.; et al. Neuroprotective Effect of Repeated Treatment with Hericium erinaceum in Mice Subjected to Middle Cerebral Artery Occlusion. J. Health Sci. 2010, 56, 296–303. [Google Scholar] [CrossRef]
- Mori, K.; Obara, Y.; Moriya, T.; Inatomi, S.; Nakahata, N. Effects of Hericium erinaceus on amyloid β(25–35) peptideinduced learning and memory deficits in mice. Biomed. Res. 2011, 32, 67–72. [Google Scholar] [CrossRef]
- Phan, C.W.; Lee, G.S.; Hong, S.L.; Wong, Y.T.; Brkljača, R.; Urban, S.; Abd Malek, S.N.; Sabaratnam, V. Hericium erinaceus (Bull.: Fr) Pers. cultivated under tropical conditions: Isolation of hericenones and demonstration of NGF-mediated neurite outgrowth in PC12 cells via MEK/ERK and PI3K-Akt signalling pathways. Food Funct. 2014, 5, 3160–3169. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Obara, Y.; Hirota, M.; Azumi, Y.; Kinugasa, S.; Inatomi, S.; Nakahata, N. Nerve growth factor-inducing activity of Hericium erinaceus in 1321N1 human astrocytoma cells. Biol. Pharm. Bull. 2008, 31, 1727–1732. [Google Scholar] [CrossRef] [PubMed]
- Vigna, L.; Morelli, F.; Agnelli, G.M.; Napolitano, F.; Ratto, D.; Occhinegro, A.; Di Iorio, C.; Savino, E.; Girometta, C.; Brandalise, F.; et al. Hericium erinaceus Improves Mood and Sleep Disorders in Patients Affected by Overweight or Obesity: Could Circulating Pro-BDNF and BDNF Be Potential Biomarkers? Evid.-Based Complement. Altern. Med. 2019, 2019, 7861297. [Google Scholar] [CrossRef]
- Nagai, K.; Chiba, A.; Nishino, T.; Kubota, T.; Kawagishi, H. Dilinoleoyl-phosphatidylethanolamine from Hericium erinaceum protects against ER stress-dependent Neuro2a cell death via protein kinase C pathway. J. Nutr. Biochem. 2006, 17, 525–530. [Google Scholar] [CrossRef]
- Zhang, C.C.; Yin, X.; Cao, C.Y.; Wei, J.; Zhang, Q.; Gao, J.M. Chemical constituents from Hericium erinaceus and their ability to stimulate NGF-mediated neurite outgrowth on PC12 cells. Bioorg. Med. Chem. Lett. 2015, 25, 5078–5082. [Google Scholar] [CrossRef]
- Kawagishi, H.; Shimada, A.; Shirai, R.; Okamoto, K.; Ojima, F.; Sakamoto, H.; Ishiguro, Y.; Furukawa, S. Erinacines A, B, and C, strong stimulators of nerve growth factor (NGF)–synthesis, from the mycelia of Hericium erinaceum. Tetrahedron Lett. 1994, 35, 1569–1572. [Google Scholar] [CrossRef]
- Rupcic, Z.; Rascher, M.; Kanaki, S.; Köster, R.W.; Stadler, M.; Wittstein, K. Two New Cyathane Diterpenoids from Mycelial Cultures of the Medicinal Mushroom Hericium erinaceus and the Rare Species, Hericium flagellum. Int. J. Mol. Sci. 2018, 19, 740. [Google Scholar] [CrossRef]
- Wang, L.Y.; Huang, C.S.; Chen, Y.H.; Chen, C.C.; Chen, C.C.; Chuang, C.H. Anti-Inflammatory Effect of Erinacine C on NO Production through Down-Regulation of NF-kappaB and Activation of Nrf2-Mediated HO-1 in BV2 Microglial Cells Treated with LPS. Molecules 2019, 24, 3317. [Google Scholar] [CrossRef]
- Kawagishi, H.; Shimada, A.; Shizuki, K.; Ojima, F.; Mori, H.; Okamoto, K.; Sakamoto, H.; Furukawa, S. Erinacine D, a stimulator of NGF-synthesis, from the mycelia of Hericium erinaceum. Heterocycl. Commun. 1996, 2, 51–54. [Google Scholar] [CrossRef]
- Kawagishi, H.; Shimada, A.; Hosokawa, S.; Mori, H.; Sakamoto, H.; Ishiguro, Y.; Sakemi, S.; Bordner, J.; Kojima, N.; Furukawa, S. Erinacines E, F, and G, stimulators of nerve growth factor (NGF)-synthesis, from the mycelia of Hericium erinaceum. Tetrahedron Lett. 1996, 37, 7399–7402. [Google Scholar] [CrossRef]
- Lee, E.W.; Shizuki, K.; Hosokawa, S.; Suzuki, M.; Suganuma, H.; Inakuma, T.; Li, J.; Ohnishi-Kameyama, M.; Nagata, T.; Furukawa, S.; et al. Two novel diterpenoids, Erinacines H and I from the mycelia of Hericium erinaceum. Biosci. Biotechnol. Biochem. 2000, 64, 2402–2405. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, L.; Bao, L.; Yang, Y.; Ma, K.; Liu, H. Three new cyathane diterpenes with neurotrophic activity from the liquid cultures of Hericium erinaceus. J. Antibiot. 2018, 71, 818–821. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Zhang, Y.; Guo, C.; Yang, Y.; Han, J.; Yu, B.; Yin, W.; Liu, H. Reconstitution of biosynthetic pathway for mushroom-derived cyathane diterpenes in yeast and generation of new “non-natural” analogues. Acta Pharm. Sin. B 2021, 11, 2945–2956. [Google Scholar] [CrossRef] [PubMed]
- Kawagishi, H.; Ando, M.; Sakamoto, H.; Yoshida, S.; Ojima, F.; Ishiguro, Y.; Ukai, N.; Furukawa, S. Hericenones C, D and E, stimulators of nerve growth factor (NGF)-synthesis, from the mushroom Hericium erinaceum. Tetrahedron Lett. 1991, 32, 4561–4564. [Google Scholar] [CrossRef]
- Lee, D.G.; Kang, H.-W.; Park, C.-G.; Ahn, Y.-S.; Shin, Y. Isolation and identification of phytochemicals and biological activities of Hericium ernaceus and their contents in Hericium strains using HPLC/UV analysis. J. Ethnopharmacol. 2016, 184, 219–225. [Google Scholar] [CrossRef]
- Wong, K.H.; Naidu, M.; David, P.; Abdulla, M.A.; Abdullah, N.; Kuppusamy, U.R.; Sabaratnam, V. Peripheral Nerve Regeneration Following Crush Injury to Rat Peroneal Nerve by Aqueous Extract of Medicinal Mushroom Hericium erinaceus (Bull.: Fr) Pers. (Aphyllophoromycetideae). Evid.-Based Complement. Altern. Med. 2011, 2011, 580752. [Google Scholar] [CrossRef]
- Li, I.C.; Chang, H.H.; Lin, C.H.; Chen, W.P.; Lu, T.H.; Lee, L.Y.; Chen, Y.W.; Chen, Y.P.; Chen, C.C.; Lin, D.P. Prevention of early Alzheimer’s disease by erinacine a-enriched Hericium erinaceus mycelia pilot double-blind placebo-controlled study. Front. Aging Neurosci. 2020, 12, 155. [Google Scholar] [CrossRef]
- Chen, C.C.; Tzeng, T.T.; Chen, C.C.; Ni, C.L.; Lee, L.Y.; Chen, W.P.; Shiao, Y.J.; Shen, C.C. Erinacine S, a Rare Sesterterpene from the Mycelia of Hericium erinaceus. J. Nat. Prod. 2016, 79, 438–441. [Google Scholar] [CrossRef]
- Lee, L.Y.; Chou, W.; Chen, W.P.; Wang, M.F.; Chen, Y.J.; Chen, C.C.; Tung, K.C. Erinacine A-Enriched Hericium erinaceus Mycelium Delays Progression of Age-Related Cognitive Decline in Senescence Accelerated Mouse Prone 8 (SAMP8) Mice. Nutrients 2021, 13, 3659. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.C.; Lin, Y.C.; Huang, C.C.; Villaflores, O.B.; Wu, T.Y.; Huang, S.M.; Chin, T.Y. Hericium erinaceus Mycelium and Its Isolated Compound, Erinacine A, Ameliorate High-Fat High-Sucrose Diet-Induced Metabolic Dysfunction and Spatial Learning Deficits in Aging Mice. J. Med. Food 2019, 22, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.H.; Sabaratnam, V.; Abdullah, N.; Naidu, M.; Keynes, R. Activity of aqueous extracts of lion’s mane mushroom Hericium erinaceus (Bull.: Fr.) Pers. (Aphyllophoromycetideae) on the neural cell line NG108-15. Int. J. Med. Mushrooms 2007, 9, 57–65. [Google Scholar] [CrossRef]
- Üstün, R.; Ayhan, P. Regenerative activity of Hericium erinaceus on axonal injury model using in vitro laser microdissection technique. Neurol. Res. 2019, 41, 265–274. [Google Scholar] [CrossRef]
- Rossi, P.; Cesaroni, V.; Brandalise, F.; Occhinegro, A.; Ratto, D.; Perrucci, F.; Lanaia, V.; Girometta, C.; Orrù, G.; Savino, E. Dietary Supplementation of Lion’s Mane Medicinal Mushroom, Hericium erinaceus (Agaricomycetes), and Spatial Memory in Wild-Type Mice. Int. J. Med. Mushrooms 2018, 20, 485–494. [Google Scholar] [CrossRef]
- Park, Y.S.; Lee, H.S.; Won, M.H.; Lee, J.H.; Lee, S.Y.; Lee, H.Y. Effect of an exo-polysaccharide from the culture broth of Hericium erinaceus on enhancement of growth and differentiation of rat adrenal nerve cells. Cytotechnology 2002, 39, 155–162. [Google Scholar] [CrossRef]
- Cheng, J.H.; Tsai, C.L.; Lien, Y.Y.; Lee, M.S.; Sheu, S.C. High molecular weight of polysaccharides from Hericium erinaceus against amyloid beta-induced neurotoxicity. BMC Complement. Altern. Med. 2016, 16, 170. [Google Scholar] [CrossRef]
- Zhang, J.; An, S.; Hu, W.; Teng, M.; Wang, X.; Qu, Y.; Liu, Y.; Yuan, Y.; Wang, D. The Neuroprotective Properties of Hericium erinaceus in Glutamate-Damaged Differentiated PC12 Cells and an Alzheimer’s Disease Mouse Model. Int. J. Mol. Sci. 2016, 17, 1810. [Google Scholar] [CrossRef]
- Trovato, A.; Siracusa, R.; Di Paola, R.; Scuto, M.; Ontario, M.L.; Bua, O.; Di Mauro, P.; Toscano, M.A.; Petralia, C.C.T.; Maiolino, L.; et al. Redox modulation of cellular stress response and lipoxin A4 expression by Hericium erinaceus in rat brain: Relevance to Alzheimer’s disease pathogenesis. Immun. Ageing 2016, 9, 13–23. [Google Scholar] [CrossRef]
- Howard, L.; Wyatt, S.; Nagappan, G.; Davies, A.M. ProNGF promotes neurite growth from a subset of NGF dependent neurons by a p75NTR-dependent mechanism. Development 2013, 140, 2108–2117. [Google Scholar] [CrossRef]
- Isaev, N.K.; Stelmashook, E.V.; Genrikhs, E.E. Role of Nerve Growth Factor in Plasticity of Forebrain Cholinergic Neurons. Biochemistry 2017, 82, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Chao, M.V. Neurotrophins and their receptors: A convergence point for many signalling pathways. Nat. Rev. Neurosci. 2003, 4, 299–309. [Google Scholar] [CrossRef]
- Mitra, S.; Behbahani, H.; Eriksdotter, M. Innovative Therapy for Alzheimer’s Disease-with Focus on Biodelivery of NGF. Front. Neurosci. 2019, 13, 38. [Google Scholar] [CrossRef]
- Crutcher, K.A.; Collins, F. In Vitro Evidence for Two Distinct Hippocampal Growth Factors: Basis of Neuronal Plasticity? Science 1982, 217, 67–68. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.J.; Shen, J.W.; Yu, H.Y.; Ruan, Y.; Wu, T.T.; Zhao, X. Hericenones and erinacines: Stimulators of nerve growth factor (NGF) biosynthesis in Hericium erinaceus. Mycology 2010, 1, 92–98. [Google Scholar] [CrossRef]
- Conner, J.M.; Franks, K.M.; Titterness, A.K.; Russell, K.; Merrill, D.A.; Christie, B.R.; Sejnowski, T.J.; Tuszynski, M.H. NGF Is Essential for Hippocampal Plasticity and Learning. J. Neurosci. 2009, 29, 10883–10889. [Google Scholar] [CrossRef]
- Birch, A.M.; Kelly, Á.M. Chronic intracerebroventricular infusion of nerve growth factor improves recognition memory in the rat. Neuropharmacology 2013, 75, 255–261. [Google Scholar] [CrossRef]
- Bathina, S.; Das, U.N. Brain-derived neurotrophic factor and its clinical implications. Arch. Med. Sci. 2015, 11, 1164–1178. [Google Scholar] [CrossRef]
- Chao, M.V.; Hempstead, B.L. p75 and Trk: A tworeceptor system. Trends Neurosci. 1995, 18, 321–326. [Google Scholar] [CrossRef]
- Esposito, D.; Patel, P.; Stephens, R.M.; Perez, P.; Chao, M.V.; Kaplan, D.R.; Hempstead, B.L. The Cytoplasmic and Transmembrane Domains of the p75 and Trk A Receptors Regulate High Affinity Binding to Nerve Growth Factor. J. Biol. Chem. 2001, 276, 32687–32695. [Google Scholar] [CrossRef]
- Roux, P.P.; Barker, P.A. Neurotrophin signaling through the p75 neurotrophin receptor. Prog. Neurobiol. 2002, 67, 203–233. [Google Scholar] [CrossRef] [PubMed]
- De la Cruz-Morcillo, M.A.; Berger, J.; Sánchez-Prieto, R.; Saada, S.; Naves, T.; Guillaudeau, A.; Perraud, A.; Sindou, P.; Lacroix, A.; Descazeaud, A.; et al. p75 neurotrophin receptor and pro-BDNF promote cell survival and migration in clear cell renal cell carcinoma. Oncotarget 2016, 7, 34480–34497. [Google Scholar] [CrossRef] [PubMed]
- Mau, J.L.; Lin, H.C.; Ma, J.T.; Song, S.F. Non-volatile taste components of several speciality mushrooms. Food Chem. 2001, 73, 461–466. [Google Scholar] [CrossRef]
- Valu, M.-V.; Soare, L.C.; Sutan, N.A.; Ducu, C.; Moga, S.; Hritcu, L.; Boiangiu, R.S.; Carradori, S. Optimization of Ultrasonic Extraction to Obtain Erinacine A and Polyphenols with Antioxidant Activity from the Fungal Biomass of Hericium erinaceus. Foods 2020, 9, 1889. [Google Scholar] [CrossRef]
- Eisenhut, R.; Fritz, D.; Tiefel, P. Investigations on nutritionally valuable constituents (mineral substances, amino acids, aromatic substances) of Hericium erinaceus (Bull.: Fr.) Pers. Eur. J. Hortic. Sci. 1995, 60, 212–218. [Google Scholar]
- Yang, Y.; Zhou, C.Y.; Zhang, J.S.; Tang, Q.J. Comparison of chemical component and biological activity of Hericium erinaceus fruit body and mycelial extracts. Junwu Yanjiu 2006, 4, 15–19. [Google Scholar]
- Yang, F.; Wang, H.; Feng, G.; Zhang, S.; Wang, J.; Cui, L. Rapid Identification of Chemical Constituents in Hericium erinaceus Based on LC-MS/MS Metabolomics. J. Food Qual. 2021, 2021, 5560626. [Google Scholar] [CrossRef]
- Hu, J.H.; Li, I.C.; Lin, T.W.; Chen, W.P.; Lee, L.Y.; Chen, C.C.; Kuo, C.F. Absolute Bioavailability, Tissue Distribution, and Excretion of Erinacine S in Hericium erinaceus Mycelia. Molecules 2019, 24, 1624. [Google Scholar] [CrossRef]
- Roda, E.; De Luca, F.; Ratto, D.; Priori, E.C.; Savino, E.; Bottone, M.G.; Rossi, P. Cognitive Healthy Aging in Mice: Boosting Memory by an Ergothioneine-Rich Hericium erinaceus Primordium Extract. Biology 2023, 12, 196. [Google Scholar] [CrossRef]
- Lin, C.Y.; Chen, Y.J.; Hsu, C.H.; Lin, Y.H.; Chen, P.T.; Kuo, T.H.; Ho, C.T.; Chen, H.H.; Huang, S.J.; Chiu, H.C.; et al. Erinacine S from Hericium erinaceus mycelium promotes neuronal regeneration by inducing neurosteroids accumulation. J. Food Drug Anal. 2023, 31, 32–54. [Google Scholar] [CrossRef]
- Kawagishi, H.; Ando, M.; Mizuno, T. Hericenone A and B as cytotoxic principles from the mushroom Hericium erinaceum. Tetrahedron Lett. 1990, 31, 373–376. [Google Scholar] [CrossRef]
- Kawagishi, H.; Ando, M.; Shinba, K.; Sakamoto, H.; Yoshida, S.; Ojima, F.; Ishiguro, Y.; Ukai, N.; Furukawa, S. Chromans, hericenones F, G and H from the mushroom Hericium erinaceum. Phytochemistry 1992, 32, 175–178. [Google Scholar] [CrossRef]
- Ueda, K.; Tsujimori, M.; Kodani, S.; Chiba, A.; Kubo, M.; Masuno, K.; Sekiya, A.; Nagai, K.; Kawagishi, H. An endoplasmic reticulum (ER) stress-suppressive compound and its analogues from the mushroom Hericium erinaceum. Bioorg. Med Chem. 2008, 16, 9467–9470. [Google Scholar] [CrossRef] [PubMed]
- Ryu, S.H.; Hong, S.M.; Khan, Z.; Lee, S.K.; Vishwanath, M.; Turk, A.; Yeon, S.W.; Jo, Y.H.; Lee, D.H.; Lee, J.K.; et al. Neurotrophic isoindolinones from the fruiting bodies of Hericium erinaceus. Bioorg. Med. Chem. Lett. 2021, 31, 127714. [Google Scholar] [CrossRef]
- Li, W.; Zhou, W.; Song, S.B.; Shim, S.H.; Kim, Y.H. Sterol fatty acid esters from the mushroom Hericium erinaceum and their PPAR transactivational effects. J. Nat. Prod. 2014, 77, 2611–2618. [Google Scholar] [CrossRef]
- Mizuno, T. Bioactive substances in Hericium erinaceus (Bull.: Fr.) Pers. and its medicinal utilization. Int. J. Med. Mushrooms 1999, 1, 105–119. [Google Scholar] [CrossRef]
- Jia, L.M.; Liu, L.; Dong, Q.; Fang, J.N. Structural investigation of a novel rhamnoglucogalactan isolated from the fruiting bodies of the fungus Hericium erinaceus. Carbohydr. Res. 2004, 339, 2667–2671. [Google Scholar] [CrossRef]
- Zhang, A.Q.; Zhang, J.S.; Tang, Q.J.; Jia, W.; Yang, Y.; Liu, Y.F.; Fan, J.M.; Pan, Y.J. Structural elucidation of a novel fucogalactan that contains 3-O-methyl rhamnose isolated from the fruiting bodies of the fungus, Hericium erinaceus. Carbohydr. Res. 2006, 341, 645–649. [Google Scholar] [CrossRef]
- Zhang, M.; Cui, S.W.; Cheung, P.C.K.; Wang, Q. Polysaccharides from mushrooms: A review on their isolation process, structural characteristics and antitumor activity. Trends Food Sci. Technol. 2007, 18, 4–19. [Google Scholar] [CrossRef]
- Keong, C.Y.; Amini Abdul Rashid, B.; Swee Ing, Y.; Ismail, Z. Quantification and identification of polysaccharide contents in Hericium erinaceus. Nutr. Food Sci. 2007, 37, 260–271. [Google Scholar] [CrossRef]
- Yang, Y.; Li, J.; Hong, Q.; Zhang, X.; Liu, Z.; Zhang, T. Polysaccharides from Hericium erinaceus Fruiting Bodies: Structural Characterization, Immunomodulatory Activity and Mechanism. Nutrients 2022, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Ding, X.; Hou, W. Composition and antioxidant activity of water-soluble oligosaccharides from Hericium erinaceus. Mol. Med. Rep. 2015, 11, 3794–3799. [Google Scholar] [CrossRef] [PubMed]
- Li, I.C.; Lee, L.Y.; Tzeng, T.T.; Chen, W.P.; Chen, Y.P.; Shiao, Y.J.; Chen, C.C. Neurohealth Properties of Hericium erinaceus Mycelia Enriched with Erinacines. Behav. Neurol. 2018, 2018, 5802634. [Google Scholar] [CrossRef] [PubMed]
- Li, I.C.; Chen, W.P.; Chen, Y.P.; Lee, L.Y.; Tsai, Y.T.; Chen, C.C. Acute and developmental toxicity assessment of erincine A-enriched Hericium erinaceus mycelia in Sprague-Dawley rats. Drug Chem. Toxicol. 2018, 41, 459–464. [Google Scholar] [CrossRef]
- Ma, B.J.; Zhou, Y.; Li, L.Z.; Li, H.M.; Gao, Z.M.; Ruan, Y. A new cyathanexyloside from the mycelia of Hericium erinaceum. Z. Naturforschung B 2008, 63, 1241–1242. [Google Scholar] [CrossRef]
- Corana, F.; Cesaroni, V.; Mannucci, B.; Baiguera, R.M.; Picco, A.M.; Savino, E.; Ratto, D.; Perini, C.; Kawagishi, H.; Girometta, C.E.; et al. Array of Metabolites in Italian Hericium erinaceus Mycelium, Primordium, and Sporophore. Molecules 2019, 24, 3511. [Google Scholar] [CrossRef]
- Kawagishi, H.; Masui, A.; Tokuyama, S.; Nakamura, T. Erinacines J and K from the mycelia of Hericium erinaceum. Tetrahedron 2006, 62, 8463–8466. [Google Scholar] [CrossRef]
- Kenmoku, H.; Sassa, T.; Kato, N. Isolation of erinacine P, a new parental metabolite of cyathane-xylosides, from Hericium erinaceum and its biomimetic conversion into erinacines A and B. Tetrahedron Lett. 2000, 41, 4389–4393. [Google Scholar] [CrossRef]
- Kenmoku, H.; Shimai, T.; Toyomasu, T.; Kato, N.; Sassa, T. Erinacine Q, a new erinacine from Hericium erinaceum and its biosynthetic route to erinacine C in the Basidiomycete. Biosci. Biotechnol. Biochem. 2002, 66, 571–575. [Google Scholar] [CrossRef]
- Smith, E.; Ottosson, F.; Hellstrand, S.; Ericson, U.; Orho-Melander, M.; Fernandez, C.; Melander, O. Ergothioneine is associated with reduced mortality and decreased risk of cardiovascular disease. Heart 2019, 106, 691–697. [Google Scholar] [CrossRef]
- Williamson, R.D.; McCarthy, F.P.; Manna, S.; Groarke, E.; Kell, D.B.; Kenny, L.C.; McCarthy, C.M. L-(+)-Ergothioneine Significantly Improves the Clinical Characteristics of Preeclampsia in the Reduced Uterine Perfusion Pressure Rat Model. Hypertension 2020, 75, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Song, T.-Y.; Chen, C.-L.; Liao, J.-W.; Ou, H.-C.; Tsai, M. Ergothioneine protects against neuronal injury induced by cisplatin both in vitro and in vivo. Food Chem. Toxicol. 2010, 48, 3492–3499. [Google Scholar] [CrossRef] [PubMed]
- Song, T.-Y.; Lin, H.-C.; Chen, C.-L.; Wu, J.-H.; Liao, J.-W.; Hu, M.-L. Ergothioneine and melatonin attenuate oxidative stress and protect against learning and memory deficits in C57BL/6J mice treated with D-galactose. Free Radic. Res. 2014, 48, 1049–1060. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Hamada, Y.; Yasumoto, T.; Hashino, Y.; Masuyama, A.; Nagai, K. Total syntheses and endoplasmic reticulum stress suppressive activities of hericenes A-C and their derivatives. Tetrahedron Lett. 2018, 59, 1733–1736. [Google Scholar] [CrossRef]
- Kobayashi, S.; Tamura, T.; Koshishiba, M.; Yasumoto, T.; Shimizu, S.; Kintaka, T.; Nagai, K. Total Synthesis, Structure Revision, and Neuroprotective Effect of Hericenones C-H and Their Derivatives. J. Org. Chem. 2021, 86, 2602–2620. [Google Scholar] [CrossRef]
- Olariu, A.; Yamada, K.; Nabeshima, T. Amyloid pathology and protein kinase C (PKC): Possible therapeutics effects of PKC activators. J. Pharmacol. Sci. 2005, 97, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.S.; Lee, B.K.; Park, H.S.; Shim, J.K.; Kim, S.U.; Lee, S.H.; Baik, E.J.; Moon, C.H. Activation of protein kinase C-y attenuates kinate-induced cell death of cortical neurons. Neuroreport 2005, 16, 741–744. [Google Scholar] [CrossRef]
- Martelli, A.M.; Mazzotti, G.; Capitani, S. Nuclear protein kinase C isoforms and apoptosis. Eur. J. Histochem. 2004, 48, 89–94. [Google Scholar] [CrossRef]
- Li, W.; Sun, Y.N.; Zhou, W.; Shim, S.H.; Kim, Y.H. Erinacene D, a new aromatic compound from Hericium erinaceum. J. Antibiot. 2014, 67, 727–729. [Google Scholar] [CrossRef]
- Zan, X.; Cui, F.; Li, Y.; Yang, Y.; Wu, D.; Sun, W.; Ping, L. Hericium erinaceus polysaccharide-protein HEG-5 inhibits SGC-7901 cell growth via cell cycle arrest and apoptosis. Int. J. Biol. Macromol. 2015, 76, 242–253. [Google Scholar] [CrossRef]
- Lu, C.C.; Huang, W.S.; Lee, K.F.; Lee, K.C.; Hsieh, M.C.; Huang, C.Y.; Lee, L.Y.; Lee, B.O.; Teng, C.C.; Shen, C.H.; et al. Inhibitory effect of Erinacines A on the growth of DLD-1 colorectal cancer cells is induced by generation of reactive oxygen species and activation of p70S6K and p21. J. Funct. Foods 2016, 21, 474–484. [Google Scholar] [CrossRef]
- Wu, Y.-L.; Chen, S.-C.; Chang, J.-C.; Lin, W.-Y.; Chen, C.-C.; Li, C.-C.; Hsieh, M.; Chen, H.-W.; Chang, T.-Y.; Liu, C.-S.; et al. The protective effect of erinacine A–enriched Hericium erinaceus mycelium ethanol extract on oxidative Stress–Induced neurotoxicity in cell and Drosophila models of spinocerebellar ataxia type 3. Free Radic. Biol. Med. 2023, 195, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kushairi, N.; Phan, C.W.; Sabaratnam, V.; David, P.; Naidu, M. Lion’s Mane Mushroom, Hericium erinaceus (Bull.: Fr.) Pers. Suppresses H2O2-Induced Oxidative Damage and LPS-Induced Inflammation in HT22 Hippocampal Neurons and BV2 Microglia. Antioxidants 2019, 8, 261. [Google Scholar] [CrossRef] [PubMed]
- Kolotushkina, E.V.; Moldavan, M.G.; Voronin, K.Y.; Skibo, G.G. The influence of Hericium erinaceus extract on myelination process in vitro. Fiziol. Zhurnal 2003, 49, 38–45. [Google Scholar]
- Noh, H.J.; Yang, H.H.; Kim, G.S.; Lee, S.E.; Lee, D.Y.; Choi, J.H.; Kim, S.Y.; Lee, E.S.; Ji, S.H.; Kang, K.S.; et al. Chemical constituents of Hericium erinaceum associated with the inhibitory activity against cellular senescence in human umbilical vascular endothelial cells. J. Enzym. Inhib. Med. Chem. 2015, 30, 934–940. [Google Scholar] [CrossRef]
- Lew, S.Y.; Lim, S.H.; Lim, L.W.; Wong, K.H. Neuroprotective effects of Hericium erinaceus (Bull.: Fr.) Pers. against high-dose corticosterone-induced oxidative stress in PC-12 cells. BMC Complement. Med. Ther. 2020, 20, 340. [Google Scholar] [CrossRef]
- Kim, M.K.; Choi, W.Y.; Lee, H.Y. Enhancement of the neuroprotective activity of Hericium erinaceus mycelium co-cultivated with Allium sativum extract. Arch. Physiol. Biochem. 2015, 121, 19–25. [Google Scholar] [CrossRef]
- Raman, J.; Lakshmanan, H.; John, P.A.; Zhijian, C.; Periasamy, V.; David, P.; Naidu, M.; Sabaratnam, V. Neurite outgrowth stimulatory effects of myco synthesized AuNPs from Hericium erinaceus (bull.: Fr.) Pers. on pheochromocytoma (PC-12) cells. Int. J. Nanomed. 2015, 10, 5853–5863. [Google Scholar]
- Amara, I.; Scuto, M.; Zappalà, A.; Ontario, M.L.; Petralia, A.; Abid-Essefi, S.; Maiolino, L.; Signorile, A.; Trovato Salinaro, A.; Calabrese, V. Hericium erinaceus Prevents DEHP-Induced Mitochondrial Dysfunction and Apoptosis in PC12 Cells. Int. J. Mol. Sci. 2020, 21, 2138. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, E.K.; Hwang, J.W.; Kim, C.G.; Choi, D.K.; Lim, B.O.; Moon, S.H.; Jeon, B.T.; Park, P.J. Neuroprotective effect of Hericium erinaceum against oxidative stress on PC12 cells. J. Korean Soc. Appl. Biol. Chem. 2010, 53, 283–289. [Google Scholar] [CrossRef]
- Shimbo, M.; Kawagishi, H.; Yokogoshi, H. Erinacine A increases catecholamine and nerve growth factor content in the central nervous system of rats. Nutr. Res. 2005, 25, 617–623. [Google Scholar] [CrossRef]
- Jang, H.J.; Kim, J.E.; Jeong, K.H.; Lim, S.C.; Kim, S.Y.; Cho, K.O. The Neuroprotective Effect of Hericium erinaceus Extracts in Mouse Hippocampus after Pilocarpine-Induced Status Epilepticus. Int. J. Mol. Sci. 2019, 20, 859. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.H.; Liao, E.C.; Chiang, W.C.; Wang, K.L. Antioxidative Activities of Micronized Solid-State Cultivated Hericium erinaceus Rich in Erinacine A against MPTP-Induced Damages. Molecules 2023, 28, 3386. [Google Scholar] [CrossRef] [PubMed]
- Brandalise, F.; Cesaroni, V.; Gregori, A.; Repetti, M.; Romano, C.; Orrù, G.; Botta, L.; Girometta, C.; Guglielminetti, M.L.; Savino, E.; et al. Dietary Supplementation of Hericium erinaceus Increases Mossy Fiber-CA3 Hippocampal Neurotransmission and Recognition Memory in Wild-Type Mice. Evid.-Based Complement. Altern. Med. 2017, 2017, 3864340. [Google Scholar] [CrossRef]
- Rodriguez, M.N.; Lippi, S.L.P. Lion’s Mane (Hericium erinaceus) Exerts Anxiolytic Effects in the rTg4510 Tau Mouse Model. Behav. Sci. 2022, 12, 235. [Google Scholar] [CrossRef]
- Saitsu, Y.; Nishide, A.; Kikushima, K.; Shimizu, K.; Ohnuki, K. Improvement of cognitive functions by oral intake of Hericium erinaceus. Biomed. Res. 2019, 40, 125–131. [Google Scholar] [CrossRef]
- Kasahara, K.; Kaneko, N.; Shimizu, K. Effects of Hericium erinaceum on aged patients with impairment. Gunma Med. Suppl. 2001, 76, 77–78. (In Japanese) [Google Scholar]
- Nagano, M.; Shimizu, K.; Kondo, R.; Hayashi, C.; Sato, D.; Kitagawa, K.; Ohnuki, K. Reduction of depression and anxiety by 4 weeks Hericium erinaceus intake. Biomed. Res. 2010, 31, 231–237. [Google Scholar] [CrossRef]
- Inanaga, K. Marked improvement of neurocognitive impairment after treatment with compounds from Hericium erinaceum: A case study of recurrent depressive disorder. Pers. Med. Universe 2014, 3, 46–48. [Google Scholar] [CrossRef]
- Okamura, H.; Anno, N.; Tsuda, A.; Inokuchi, T.; Uchimura, N.; Inanaga, K. The effects of Hericium erinaceus (Amyloban® 3399) on sleep quality and subjective well-being among female undergraduate students: A pilot study. Pers. Med. Universe 2015, 4, 76–78. [Google Scholar] [CrossRef]
- Tzeng, T.-T.; Chen, C.-C.; Chen, C.-C.; Tsay, H.-J.; Lee, L.-Y.; Chen, W.-P.; Shen, C.-C.; Shiao, Y.-J. The Cyanthin Diterpenoid and Sesterterpene Constituents of Hericium erinaceus Mycelium Ameliorate Alzheimer’s Disease-Related Pathologies in APP/PS1 Transgenic Mice. Int. J. Mol. Sci. 2018, 19, 598. [Google Scholar] [CrossRef] [PubMed]
- Grygansky, A.P.; Moldavan, M.G.; Kolotushkina, O.; Kirchhoff, B.; Skibo, G.G. Hericium erinaceus (Bull.: Fr.) Pers. extract effect on nerve cells. Int. J. Med. Mushrooms 2001, 3, 152. [Google Scholar] [CrossRef]
- Kempermann, G. Adult neurogenesis: An evolutionary perspective. Cold Spring Harb. Perspect. Biol. 2016, 8, a018986. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.P.; Nam, S.H.; Friedman, M. Correction to Hericium erinaceus (Lion’s Mane) Mushroom Extracts Inhibit Metastasis of Cancer Cells to the Lung in CT-26 Colon Cancer-Transplanted Mice. J. Agric. Food Chem. 2013, 61, 4898–4904. [Google Scholar] [CrossRef]
- Wong, K.H.; Naidu, M.; David, R.P.; Abdulla, M.A.; Kuppusamy, U.R. Functional recovery enhancement following Injury to rodent peroneal nerve by lion’s mane mushroom, Hericium erinaceus (Bull.: Fr.) Pers. (Aphyllophoromycetideae) . Int. J. Med. Mushrooms 2009, 11, 225–236. [Google Scholar] [CrossRef]
- Samuels, B.A.; Hen, R. Neurogenesis and affective disorders. Eur. J. Neurosci. 2011, 33, 1152–1159. [Google Scholar] [CrossRef]
- Malinski, T. Nitric oxide and nitroxidative stress in Alzheimer’s disease. J. Alzheimer’s Dis. 2007, 11, 207–218. [Google Scholar] [CrossRef]
- Gąsecka, M.; Mleczek, M.; Siwulski, M.; Niedzielski, P.; Kozak, L. Phenolic and flavonoid content in Hericium erinaceus, Ganoderma lucidum, and Agrocybe aegerita under selenium addition. Acta Aliment. 2016, 45, 300–308. [Google Scholar] [CrossRef]
- Lew, S.Y.; Yow, Y.Y.; Lim, L.W.; Wong, K.H. Antioxidant-mediated protective role of Hericium erinaceus (Bull.: Fr.) Pers. against oxidative damage in fibroblasts from Friedreich’s ataxia patient. Food Sci. Technol. 2020, 40, 264–272. [Google Scholar] [CrossRef]
- Ames, B.N.; Shigenaga, M.K.; Hagen, T.M. Oxidants, antioxidants, and the degenerative diseases of ageing. Proc. Natl. Acad. Sci. USA 1993, 90, 7915–7922. [Google Scholar] [CrossRef]
- Abdullah, N.; Ismail, S.M.; Aminudin, N.; Shuib, A.S.; Lau, B.F. Evaluation of Selected Culinary-Medicinal Mushrooms for Antioxidant and ACE Inhibitory Activities. Evid.-Based Complement. Altern. Med. 2012, 2012, 464238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lv, G.; Pan, H.; Pandey, A.; He, W.; Fan, L. Antioxidant and hepatoprotective potential of endo-polysaccharides from Hericium erinaceus grown on tofu whey. Int. J. Biol. Macromol. 2012, 51, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Li, I.-C.; Chen, Y.-L.; Lee, L.-Y.; Chen, W.-P.; Tsai, Y.-T.; Chen, C.-S. Evaluation of the toxicological safety of erinacine A-enriched Hericium erinaceus in a 28-day oral feeding study in Sprague–Dawley rats. Food Chem. Toxicol. 2014, 70, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, H.; Raman, J.; David, P.; Wong, K.-H.; Naidu, M.; Sabaratnam, V. Haematological, biochemical and histopatho-logical aspects of Hericium erinaceus ingestion in a rodent model: A sub-chronic toxi-cological assessment. J. Ethnopharmacol. 2016, 194, 1051–1059. [Google Scholar] [CrossRef]
- Chen, S.N.; Chang, C.S.; Yang, M.F.; Chen, S.; Soni, M.; Mahadevan, B. Subchronic toxicity and genotoxicity studies of Hericium erinaceus β-glucan extract preparation. Curr. Res. Toxicol. 2022, 3, 100068. [Google Scholar] [CrossRef]
- Li, I.C.; Chen, Y.L.; Chen, W.P.; Lee, L.-Y.; Tsai, Y.-T.; Chen, C.-C.; Chen, C.-S. Genotoxicity profile of erinacine A-enriched Hericium erinaceus mycelium. Toxicol. Rep. 2014, 1, 1195–1201. [Google Scholar] [CrossRef]

| Components | Biological Activities | Tests | Test Cell Lines/Organisms | Reference |
|---|---|---|---|---|
| Erinacine A | induced 250.1 ± 36.2 pg/mL NGF synthesis *; | In vitro | mouse astroglial cells | [40] |
| enhanced neurotrophin production (increased NGF mRNA) | 1321N1 human astrocytoma cells | [41] | ||
| Erinacine B | induced 129.7 ± 6.5 pg/mL NGF synthesis *; | In vitro | mouse astroglial cells | [40] |
| enhanced neurotrophin production (increased NGF mRNA) | 1321N1 human astrocytoma cells | [41] | ||
| Erinacine C | induced 299.1 ± 59.6 pg/mL NGF synthesis *; | In vitro | mouse astroglial cells | [40] |
| enhanced neurotrophin production (increased NGF mRNA, increased BDNF mRNA); | 1321N1 human astrocytoma cells | [41] | ||
| preventing neuroinflammation (reduced NO, IL-6, and TNF-α, inhibiting the expression of NF-κB and phosphorylation of IκBα) | BV2 microglial cells | [42] | ||
| Erinacine D | induced 141.5 ± 18.2 pg/mL NGF synthesis * | In vitro | rat astroglial cells | [43] |
| Erinacine E | induced 105 ± 5.2 pg/mL NGF synthesis *; | In vitro | rat astroglial cells | [44] |
| enhanced neurotrophin production | 1321N1 human astrocytoma cells | [41] | ||
| Erinacine F | induced 175 ± 52 pg/mL NGF synthesis * | In vitro | rat astroglial cells | [44] |
| Erinacine H | induced 31.5 ± 1.7 pg/mL NGF synthesis * | In vitro | rat astroglial cells | [45] |
| Erinacine P | significant neurite outgrowth-promoting effects | In vitro | PC12 cells | [46] |
| Erinacine S | neurite outgrowth of primary neurons from both the CNS and PNS are significantly enhanced | In vitro | Mouse cortical neuron cultures Rat dorsal root ganglion (DRG) neuron cultures | [82] |
| Erinacine T | significant neurite outgrowth-promoting effects | In vitro | PC12 cells | [46] |
| Erinacine U | ||||
| Erinacine V | ||||
| Erinacine W (non-natural analogue) | stimulated neurite outgrowth | In vitro | PC12 cells | [47] |
| Erinacine X (non-natural analogue) | ||||
| Erinacine Y (non-natural analogue) | ||||
| Erinacicne ZA (non-natural analogue) | ||||
| Erinacine Z1 | increased NGF mRNA | In vitro | 1321N1 human astrocytoma cells | [41] |
| Hericenone A | cytotoxicity | In vitro | HeLa cells | [83] |
| Hericenone C | induced 23.5 ± 1.0 pg/mL NGF synthesis * | In vitro | mouse astroglial cells | [48] |
| Hericenone D | induced 10.08 ± 0.8 pg/mL NGF synthesis * | |||
| Hericenone E | induced 13.9 ± 2.1 pg/mL NGF synthesis *; | mouse astroglial cells | [48] | |
| NGF-induced neurite outgrowth | PC12 cells | [35] | ||
| Hericenone F | reduced NO generation-anti-inflammatory effect | In vitro | RAW264.7 cells | [49] |
| Hericenone H | induced 45.1 ± 1.1 pg/mL NGF synthesis * | In vitro | mouse astroglial cells | [84] |
| 3-Hydroxyhericenone F | protective activity against endoplasmic reticulum (ER) stress | In vitro | culture medium of Neuro2a cells | [85] |
| Hericerin Isohericerinol A | increase of the NGF level in a dose-dependent manner; | In vitro | C6 glioma cells | [86] |
| Isohericerinol A Corallocin A | increased the expression of BDNF protein | |||
| Erinacene D | inhibited the induction of iNOS and ICAM-1 mRNA; suppress TNFα–induced NF-κB transcriptional activity | In vitro | HaCaT cells | [87] |
| 3,4-Dihydro-5-methoxy-2-methyl-2-(40-methyl-20-oxo-30-pentyl)-9(7H)-oxo-2h-furo[3,4-h]benzopyran | exhibited high neurite outgrowth-promoting activity | In vitro | PC12 cells | [39] |
| DLPE (dilinoleoyl-phosphatidylethanolamine) | protection against neuronal cell death caused by endoplasmic reticulum (ER) stress and oxidative stress | In vitro | Neuro2a cells | [38] |
| Tests | Type of Extract, Dose and Dosage | Biological Activities | Test Cell Lines/Organisms | References |
| In vitro | Ethanolic extract, H. erinaceus fruiting bodies | promotion of NGF mRNA expression in a concentration-dependent manner through activation of the JNK pathway | 1321N1 human astrocytoma cells | [36] |
| Ethanolic extract, H. erinaceus mycelia | inhibited the cell cycle G1 distribution as a result of the generation of the ROS and mTOR/p70S6K/NF-κB/p21 pathway | human colon cancer cell line DLD-1 (CCL-221) and human colorectal carcinoma cell line HCT-116 (CCL-247) | [113] | |
| Ethanolic extract, H. erinaceus mycelia, and a solution of erinacine A | modulate multiple signalling pathways involved in neuronal survival and cell death pathways | Neuro-2a cells | [23] | |
| Ethanolic extract, H. erinaceus mycelia enriched erinacine A | exerted an anti-apoptotic function by modulating the transcription factors p53 and NF-κB and their downstream events in cell lines, | neuroblastoma K-N-SHMJD78 cells | [114] | |
| enhancing neurite growth of primary cortical neurons in a dose-dependent manner | primary cultures of neonatal rat cortical neuronal cells | [96] | ||
| Ethanolic and hot water extract, H. erinaceus mycelia | exerted potent neuroprotection and NO-suppressing anti-inflammatory activity | HT22 cells | [115] | |
| Ethanolic extract from H. erinaceus | promoted the normal cultivation of the nerve and glial cells; influence on the process of myelination | cultured of WISTAR rat cerebellum cells | [116] | |
| Methanolic extract, H. erinaceus fruiting bodies | inhibitory effects against cellular senescence in human primary cells, | Human dermal fibroblasts (HDFs), human umbilical vein endothelial cells (HUVECs), endothelial cell growth medium-2(EGM-2) | [117] | |
| Aqueous extract, H. erinaceus fruiting bodies | induced 45.67 ± 0.79 pg/mL NGF synthesis * increased neurite extension | NG108-15 cells | [2,55] | |
| the protective abilities of H. erinaceus treatment and its combination with NGF were significantly higher than NGF treatment alone | mouse PNI model | [56] | ||
| Aqueous extract of Malaysian-grown H. erinaceus | increase neurite extension; protective effect against oxidative stress | NG108-15 cells | [2] | |
| neuroprotective effects against high-dose corticosterone-induced oxidative stress | PC12 cells | [118] | ||
| Aqueous extract, H. erinaceus mycelia and fruiting body, garlic extract | have the synergistic effect of the mycelium and garlic extracts on neuroprotective activity | PC12 cells | [119] | |
| Aqueous extract, H. erinaceus mycelium | neuroprotective effect | an L-Glu-induced DPC12 cellular apoptosis model | [60] | |
| AuNPs using the hot aqueous extract of H. erinaceus fruiting bodies | have potential neuronal differentiation and stimulated neurite outgrowth | PC12 cells | [120] | |
| H. erinaceus biomass, a powder containing mycelium and primordia | increased PC12 cell survival against DEHP insult; induces anti-apoptotic activity; reduces intracellular reactive oxygen species levels | PC12 cultured in DMEM | [121] | |
| Enzymatic hydrolysates from H. erinaceus | more effective antioxidative and superoxide radical scavenging activity (compared to water and organic solvent extracts); neuroprotective effects against H2O2 | PC12 cells | [122] | |
| Biopolymer from the liquid culture broth of H. erinaceus mycelium | enhanced the growth of rat adrenal nerve cells; both nerve growth factors also improved the growth of PC12 cells | PC12 cells | [58] | |
| Polysaccharide extracts from fruiting bodies H. erinaceus | antioxidant and neuroprotective effects on Aβ-induced neurotoxicity in neurons | PC12 cells | [59] | |
| In vivo | A solution of erinacine A (8 mg/kg body weight) dissolved in 5% ethanol and saline phosphate buffer, IGAS | Enhanced NGF and catecholamine secretion in the LC and hippocampus, and a decrease in the cerebral cortex | normal Wistar rats | [123] |
| Ethanolic extract, H. erinaceus mycelia (50, 300, and 1000 mg/kg body weight), PO, and a solution of erinacine A (1, 5, and 10 mg/kg), IP for 5 days | reduced infarcted volume in the cortex and subcortex; reduced levels of proinflammatory cytokines such as iNOS, IL-1β, IL-6, and TNF-α in the serum | ischemic stroke in Sprague-Dawley rat | [20] | |
| A solution of erinacine A (1, 2, and 5 mg/day) for 5 days, IP | decreased the growth of the xenografts of CRC cells in nude mice by inhibiting cell proliferation and promoting apoptosis | BALB/c-nu mice | [113] | |
| Ethanolic extract, H. erinaceus mycelia (10.76 mg and 21.52 mg), PO, and a solution of erinacine A (1 mg/kg body weight) IP for 5 days | the signalling molecules affected by erinacine A included the survival factors PAK1, cdc42, AKT, LIMK2, ERK, and Cofilin, IRE1á, TRAF2, ASK1, GADD45, and p21; a reduced number of apoptotic neurons | C57BL/6 mice | [23] | |
| Ethanolic extract, H. erinaceus mycelia, a solution of erinacine A and S (30 mg/kg/day) for 30 days, PO | reduced amyloid plaque burden in the cerebral cortex; increased the level of insulin-degrading enzyme (IDE) in the cerebral cortex | APPswe/PS1dE9 transgenic mice | [52] | |
| Ethanolic extract, H. erinaceus mycelia enriched erinacine A, PO | exerted an anti-apoptotic function by modulating the transcription factors p53 and NF-κB and their downstream events in Drosophila models of SCA3 disturbed by oxidative stress | Drosophila models of SCA3 (fly stocks, elav-Gal4, UAS-MJDtr-Q27, and UAS-MJDtr-Q78 flies) | [114] | |
| 300 mg/kg/day of mycelia powder and ethanolic extract for 30 days, PO | reduced amyloid plaque burden in the area, including the cerebral cortex and hippocampus; increased NGF/proNGF ratio; and promoted hippocampal neurogenesis | 5-month-old female APPswe/PS1dE9 transgenic mice | [22] | |
| Erinacine A- enriched H. erinaceus mycelia (108, 215, and 431 mg/kg/day) for 13 weeks, PO | lower oxidative stress significantly improved learning and memory | 3-month-old male and female senescence-accelerated mice (SAMP8) | [53] | |
| Ethanolic extract, H. erinaceus (20 and 60 mg/kg) for 28 days, PO | increased hippocampal neurogenesis | male C57BL-6J mice | [27] | |
| Ethanolic extract of H. erinaceus (60, 120, and 300 mg/ kg body weight) for 21 days, PO | a reduction of COX2-expressing astrocytes; neuroprotective effect | male C57BL mice (a pilocarpine-induced SE model) | [124] | |
| An ethanolic mixture of lyophilized mycelium and sporophores of H. erinaceus (1 mg/supplement per day) for 2 months, PO | Increased recognition and memory performance in mice during aging; reduced cognitive decline | male C57BL-6J mice | [31] | |
| Methanolic extract, H. erinaceus mycelia (1 g/ kg), and a solution of erinacine A (43 mg/kg) for 18 weeks, PO | decreased neuronal loss; higher NGF biosynthesis; performed better in spatial learning; increased mRNA expression levels of TNFa and IL-1b in the hippocampus | C57BL/6 mice | [54] | |
| 5% freeze-dried powder of fruiting bodies of H. erinaceus, PO | increased NGF mRNA in the hippocampus | male ddY mice | [36] | |
| Powder of H. erinaceus mycelia (100, 200, and 400 mg/kg body weight) for 4 weeks, PO | antidepressant-like effect; increased BDNF, TrκB, and PI3K expressions in the hippocampus; reduced IL-6 and TNF-α levels | restraint stress-induced depression in ICR mice | [26] | |
| 400 mg mycelia and 100 mg dried fruiting body extract of H. erinaceus for 2 months, PO | increasing glutamatergic synaptic drive in the hippocampus; increased general locomotor activity but did not affect spatial memory | C57BL-6J mice | [57] | |
| The powdered fruiting bodies of H. erinaceus (5% w/w) for 23 days, PO | prevented the cognitive deficits induced by the administration of Aβ(25–35) | male 5-week-old ICR mice | [34] | |
| The powdered mycelia of H. erinaceus (0.1 g/kg, 0.3 g/kg, and 1 g/kg) for 30 days, PO | reduced oxidative stress; increase in dopamine levels | male C57BL/6Narl mice treated with 1-methyl-4-phenylpyridinium (MPTP) | [125] | |
| H. erinaceus biomass, a powder containing mycelium and primordia (200 mg/kg body weight) for 3 months, IGAS | neuroprotective effect; increased expression of genes, particularly HSP70, HO-1, and TRX), leading to an increase in LXA4 synthesis in various regions of the brain. | male Sprague-Dawley rats | [61] | |
| Aqueous extract, Malaysian-grown H. erinaceus fruiting bodies (10 or 20 mL kg−1 body weight per day) for 14 days, PO | promote the regeneration of injured rat peroneal nerves in the early stages of recovery | adult female Sprague-Dawley rats | [50] | |
| Aqueous extract, H. erinaceus fruiting bodies, for 14 days, PO | increased level of NGF in cortex, striatum and hippocampus | male ddY mice subjected to MCA Occlusion | [33] | |
| Aqueous extract, H. erinaceus mycelium (0.3, 1, and 3 g/kg body weight) for 4 weeks, PO | neuroprotective effect | AD mouse | [60] | |
| The supplement Micotherapy Hericium (Noceto, Parma, Italy) (contains mycelium and fruiting body extract of Hericium erinaceus in a ratio of 4/1), corresponding to 0.025 g/g body weight for 2 months, PO | increasing glutamatergic synaptic drive, novelty exploration behaviour, and recognition memory in the hippocampus | C57BL-6J mice | [126] | |
| Supplementation of H. erinaceus (Host Defense Mushrooms, Fungi Perfecti, LLC., Olympia, WA, USA) through wet food for 4 months, PO | anxiolytic effects; no improvements in spatial memory | rTg4510 tau mouse model | [127] | |
| In vivo/ Clinical trial | Aqueous and ethanolic extract, H. erinaceus supplementation (80% mycelia and 20% fruiting body), 1.2 g per capsule; 3 capsules/day for 8 weeks; PO | decreased depression, anxiety, and sleep disorders | seventy-seven volunteers (62 females and 15 males) with a body mass index (BMI) ≥ 25 kg/m2 | [37] |
| Aqueous extract, H. erinaceus mycelium, 350 mg/capsule containing 5 mg/g erinacine A (EAHE) for 49 weeks; PO | reduced cognitive decline | patients with age > 50 years and diagnosis of probable AD | [51] | |
| Dried fruiting bodies of H. erinaceus, 250 mg tablets containing 96% of H. erinaceus dry powder three times a day for 16 weeks, PO | improved cognitive function | a double-blind, parallel-group, placebo-controlled trial was performed on 50- to 80-year-old Japanese men and women diagnosed with mild cognitive impairment | [21] | |
| 0.8 g of the powdered fruiting body of H. erinaceus; 4 capsules/day for 12 weeks; PO | improved cognitive function | randomized, double-blind, placebo-controlled parallel-group | [128] | |
| 5 g/day of the lyophilized H. erinaceus for a 6-month period; PO | improved cognitive function | fifty elderly individuals with disabilities | [129] | |
| Aqueous extract, 0.5 g of the powdered fruiting bodies of H. erinaceus per cookie, 4 cookies daily for 4 weeks; PO | decreased depression, anxiety | a double-blind, parallel-group, placebo-controlled trial was performed on thirty middle-aged females in menopause | [130] | |
| Patented extraction, Amycenone®, H. erinaceus fruiting body extract (0.5% hericenones and 6% amyloban), 1950 mg/tablet (Amyloban® 3399) 6 tablets, divided into 2 or 3 doses /day for 6 months; PO | improved cognitive function | 86-year-old male patient | [131] | |
| Patented extraction, Amycenone®, H. erinaceus fruiting body extract (0.5% hericenones and 6% amyloban), 1950 mg/tablet (Amyloban® 3399) 6 tablets, divided into 2 or 3 doses /day for 4 weeks; PO | decreased depression, anxiety, and sleep disorders | 8 female healthy participants | [132] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szućko-Kociuba, I.; Trzeciak-Ryczek, A.; Kupnicka, P.; Chlubek, D. Neurotrophic and Neuroprotective Effects of Hericium erinaceus. Int. J. Mol. Sci. 2023, 24, 15960. https://doi.org/10.3390/ijms242115960
Szućko-Kociuba I, Trzeciak-Ryczek A, Kupnicka P, Chlubek D. Neurotrophic and Neuroprotective Effects of Hericium erinaceus. International Journal of Molecular Sciences. 2023; 24(21):15960. https://doi.org/10.3390/ijms242115960
Chicago/Turabian StyleSzućko-Kociuba, Izabela, Alicja Trzeciak-Ryczek, Patrycja Kupnicka, and Dariusz Chlubek. 2023. "Neurotrophic and Neuroprotective Effects of Hericium erinaceus" International Journal of Molecular Sciences 24, no. 21: 15960. https://doi.org/10.3390/ijms242115960
APA StyleSzućko-Kociuba, I., Trzeciak-Ryczek, A., Kupnicka, P., & Chlubek, D. (2023). Neurotrophic and Neuroprotective Effects of Hericium erinaceus. International Journal of Molecular Sciences, 24(21), 15960. https://doi.org/10.3390/ijms242115960






