Deficiency of ChPks and ChThr1 Inhibited DHN-Melanin Biosynthesis, Disrupted Cell Wall Integrity and Attenuated Pathogenicity in Colletotrichum higginsianum
Abstract
:1. Introduction
2. Results
2.1. Identification and Characterization of ChPks and ChThr1 in C. higginsianum
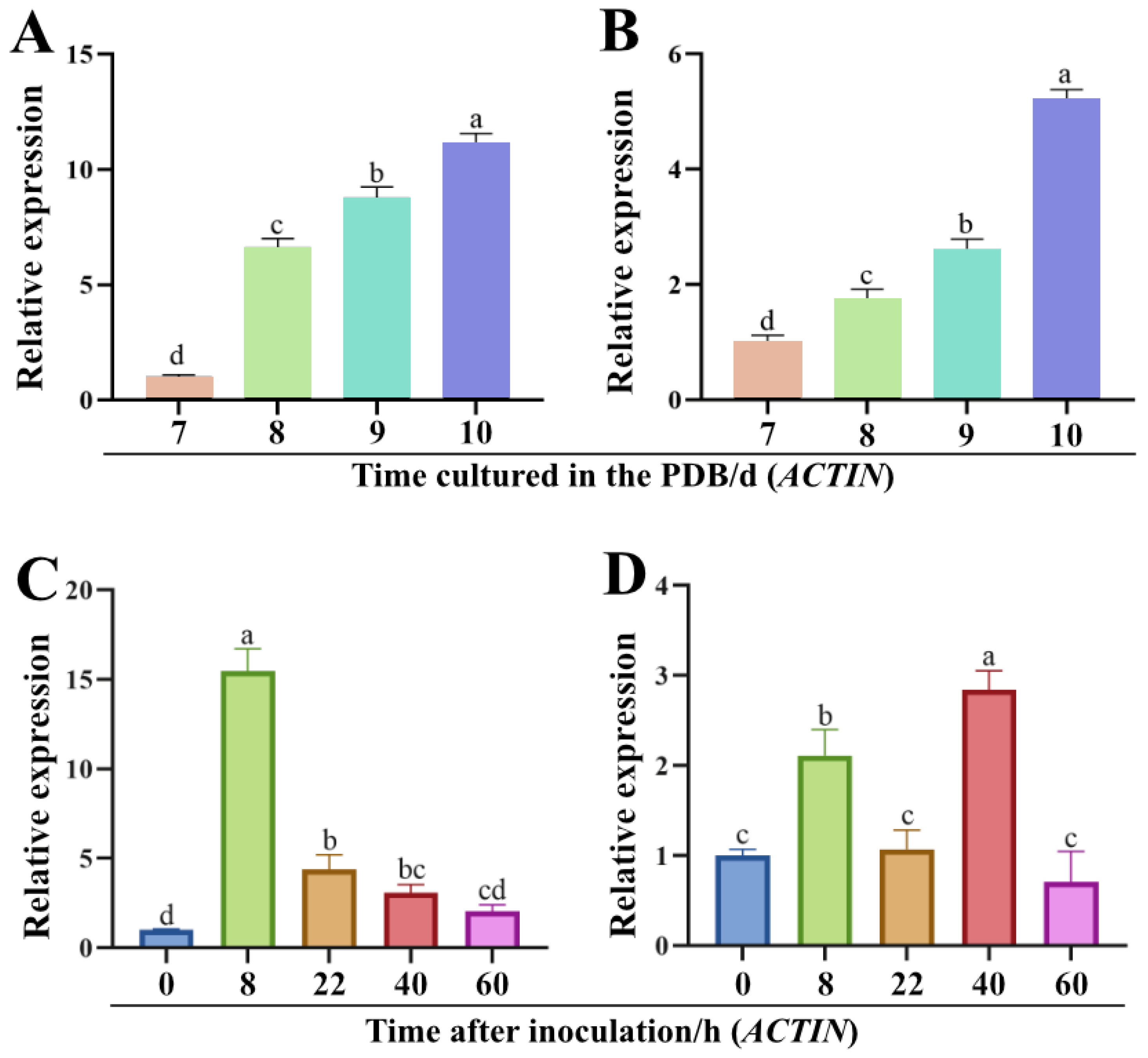
2.2. Expression of Genes ChPKS and ChTHR1 Is Up-Regulated during Melanization
2.3. ChPks and ChThr1 Are Involved in DHN-Melanin Biosynthesis
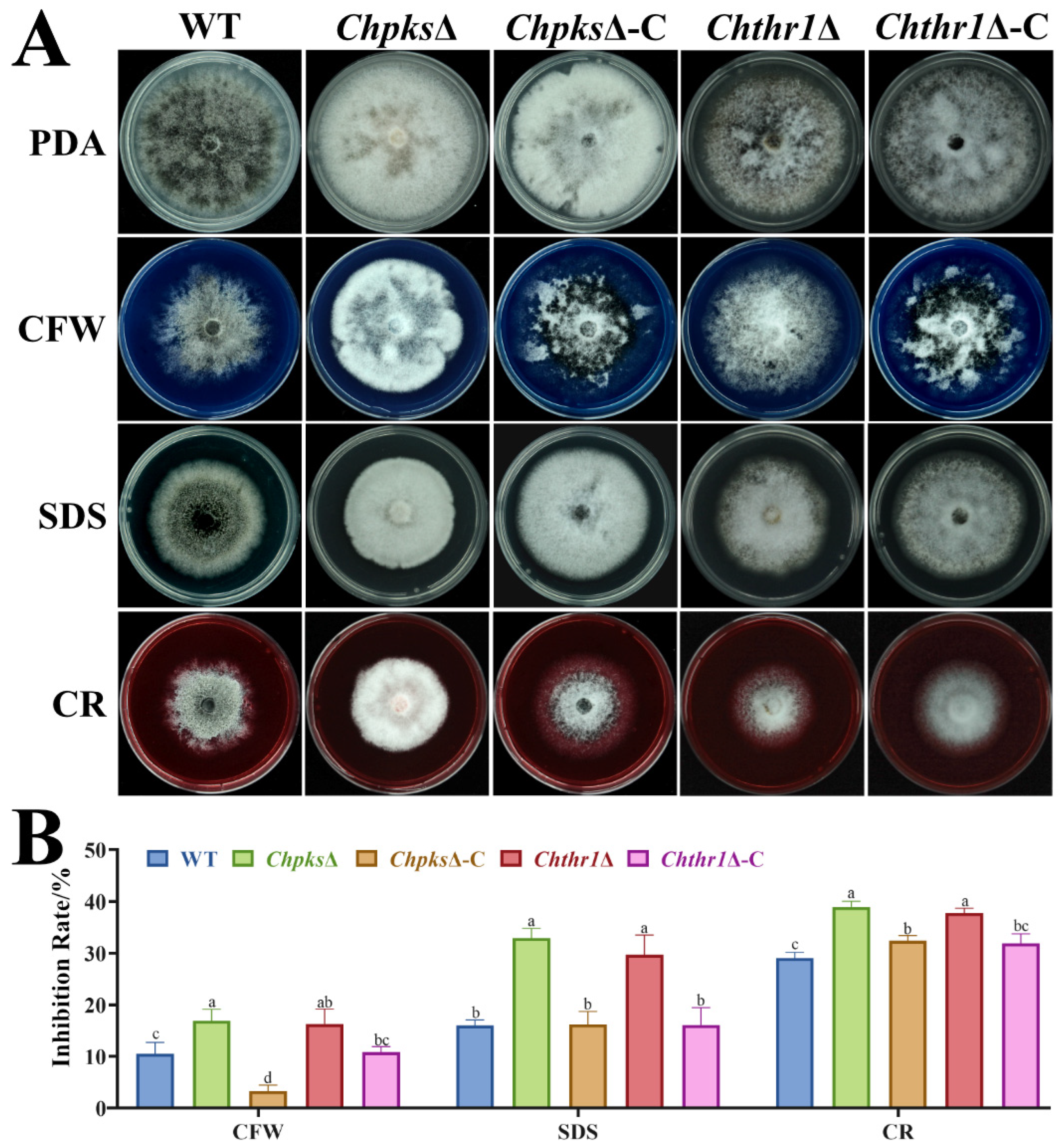
2.4. Melanin Is Essential for Cell Wall Integrity in C. higginsianum
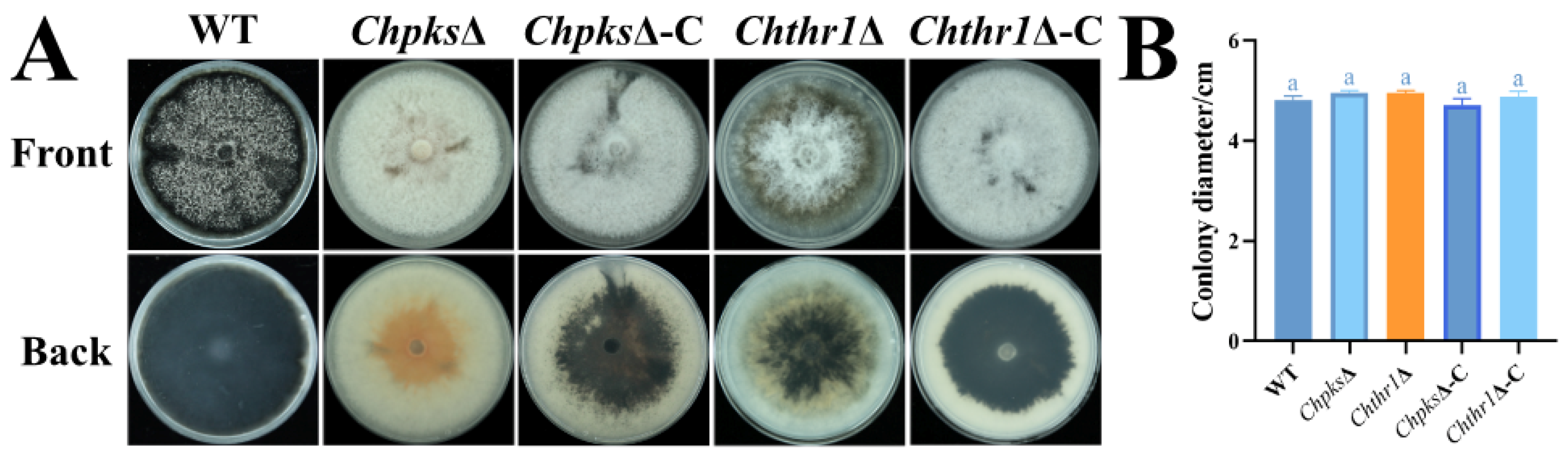
2.5. Analysis of Conidiation, Appressorium Formation and Morphology, Turgor Pressure in ChpksΔ and Chthr1Δ Mutants
2.6. ChPks and ChThr1 Are Enssential for Pathogenicity in C. higginsianum
3. Discussion
4. Materials and Methods
4.1. Strains, Plant and Culture Conditions
4.2. Bioinformatics Analysis of ChPks and ChThr1
4.3. Expression Pattern Analysis of Genes ChPKS and ChTHR1
4.4. Deletion and Complementation of ChPKS and ChTHR1
4.5. Phenotypic Analysis
4.6. Transmission Electron and Confocal Microscopy
4.7. Pathogenicity Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Liu, Z.; Zheng, Y.; Huang, H.; Zhou, E.; Yang, M. Identification of resistance to anthracnose in Chinese cabbage variety resources. Guangdong Agric. Sci. 2000, 48, 47–49. [Google Scholar] [CrossRef]
- Damm, U.; O’Connell, R.J.; Groenewald, J.Z.; Crous, P.W. The Colletotrichum destructivum species complex—Hemibiotrophic pathogens of forage and field crops. Stud. Mycol. 2014, 79, 49–84. [Google Scholar] [CrossRef]
- Yan, Y.; Yuan, Q.; Tang, J.; Huang, J.; Hsiang, T.; Wei, Y.; Zheng, L. Colletotrichum higginsianum as a Model for Understanding Host-Pathogen Interactions: A Review. Int. J. Mol. Sci. 2018, 19, 2142. [Google Scholar] [CrossRef]
- Zhu, Y.; Duan, L.; Zhu, C.; Wang, L.; He, Z.; Yang, M.; Zhou, E. Dual Transcriptome Analysis Reveals That ChATG8 Is Required for Fungal Development, Melanization and Pathogenicity during the Interaction between Colletotrichum higginsianum and Arabidopsis thaliana. Int. J. Mol. Sci. 2023, 24, 4376. [Google Scholar] [CrossRef]
- Zampounis, A.; Pigné, S.; Dallery, J.-F.; Wittenberg, A.H.J.; Zhou, S.; Schwartz, D.C.; Thon, M.R.; O’Connell, R.J. Genome Sequence and Annotation of Colletotrichum higginsianum, a Causal Agent of Crucifer Anthracnose Disease. Genome Announc. 2016, 4, e00821-16. [Google Scholar] [CrossRef]
- O’Connell, R.; Herbert, C.; Sreenivasaprasad, S.; Khatib, M.; Esquerre-Tugaye, M.T.; Dumas, B. A novel Arabidopsis-Colletotrichum pathosystem for the molecular dissection of plant-fungal interactions. Mol. Plant Microbe 2004, 17, 272–282. [Google Scholar] [CrossRef]
- Casadevall, A.; Cordero, R.J.B.; Bryan, R.; Nosanchuk, J.; Dadachova, E. Melanin, Radiation, and Energy Transduction in Fungi. Microbiol. Spectr. 2017, 5, 509–514. [Google Scholar] [CrossRef] [PubMed]
- Toledo, A.V.; Franco, M.E.E.; Lopez, S.M.Y.; Troncozo, M.I.; Saparrat, M.C.N.; Balatti, P.A. Melanins in fungi: Types, localization and putative biological roles. Physiol. Mol. Plant Pathol. 2017, 99, 2–6. [Google Scholar] [CrossRef]
- Lin, L.; Xu, J.P. Production of fungal pigments: Molecular processes and their applications. J. Fungi 2022, 9, 44. [Google Scholar] [CrossRef]
- Schumacher, J. DHN melanin biosynthesis in the plant pathogenic fungus Botrytis cinerea is based on two developmentally regulated key enzyme (PKS)-encoding genes. Mol. Microbiol. 2016, 99, 729–748. [Google Scholar] [CrossRef] [PubMed]
- Perez-Cuesta, U.; Aparicio-Fernandez, L.; Guruceaga, X.; Martin-Souto, L.; Abad-Diaz-de-Cerio, A.; Antoran, A.; Buldain, I.; Hernando, F.L.; Ramirez-Garcia, A.; Rementeria, A. Melanin and pyomelanin in Aspergillus fumigatus: From its genetics to host interaction. Int. Microbiol. 2020, 23, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Ren, D.D.; Guo, H.; Chen, X.; Zhu, P.K.; Nie, H.Z.; Xu, L. CgSCD1 Is Essential for Melanin Biosynthesis and Pathogenicity of Colletotrichum gloeosporioides. Pathogens 2020, 9, 141. [Google Scholar] [CrossRef]
- Pihet, M.; Vandeputte, P.; Tronchin, G.; Renier, G.; Saulnier, P.; Georgeault, S.; Mallet, R.; Chabasse, D.; Symoens, F.; Bouchara, J.P. Melanin is an essential component for the integrity of the cell wall of Aspergillus fumigatus conidia. BMC Microbiol. 2009, 9, 177. [Google Scholar] [CrossRef]
- Eisenman, H.C.; Casadevall, A. Synthesis and assembly of fungal melanin. Appl. Microbiol. Biotechnol. 2012, 93, 931–940. [Google Scholar] [CrossRef] [PubMed]
- Nosanchuk, J.D.; Stark, R.E.; Casadevall, A. Fungal Melanin: What do We Know About Structure? Front. Microbiol. 2015, 6, 1463. [Google Scholar] [CrossRef]
- Bull, A.T. Inhibition of polysaccharases by melanin: Enzyme inhibition in relation to mycolysis. Arch. Biochem. Biophys. 1970, 137, 345–356. [Google Scholar] [CrossRef]
- Munro, C.A.; Gow, N.A.R. Chitin synthesis in human pathogenic fungi. Med. Mycol. 2001, 39, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Walker, C.A.; Gomez, B.L.; Mora-Montes, H.M.; Mackenzie, K.S.; Munro, C.A.; Brown, A.J.P.; Gow, N.A.R.; Kibbler, C.C.; Odds, F.C. Melanin Externalization in Candida albicans Depends on Cell Wall Chitin Structures. Eukaryot. Cell 2010, 9, 1329–1342. [Google Scholar] [CrossRef]
- Eisenman, H.C.; Greer, E.M.; McGrail, C.W. The role of melanins in melanotic fungi for pathogenesis and environmental survival. Appl. Microbiol. Biotechnol. 2020, 104, 4247–4257. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Song, J.J.; Wang, Y.C.; Yang, L.; Wu, M.D.; Li, G.Q.; Zhang, J. Biological characterization of the melanin biosynthesis gene Bcscd1 in the plant pathogenic fungus Botrytis cinerea. Fungal Genet. Biol. 2022, 160, 103693. [Google Scholar] [CrossRef] [PubMed]
- Kunova, A.; Pizzatti, C.; Cortesi, P. Impact of tricyclazole and azoxystrobin on growth, sporulation and secondary infection of the rice blast fungus, Magnaporthe oryzae. Pest Manag. Sci. 2013, 69, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.J.; Xu, X.W.; Peng, F.; Yan, D.Z.; Zhang, S.P.; Xu, R.; Wu, J.; Li, X.; Wei, W.; Chen, W.D. The cyclase-associated protein ChCAP is important for regulation of hyphal growth, appressorial development, penetration, pathogenicity, conidiation, intracellular cAMP level, and stress tolerance in Colletotrichum higginsianum. Plant Sci. 2019, 283, 1–10. [Google Scholar] [CrossRef]
- Zhu, S.; Yan, Y.; Qu, Y.; Wang, J.; Feng, X.; Liu, X.; Lin, F.; Lu, J. Role refinement of melanin synthesis genes by gene knockout reveals their functional diversity in Pyricularia oryzae strains. Microbiol. Res. 2021, 242, 126620. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, N.; Löhrer, M.; Hempel, M.; Mathea, S.; Schliebner, I.; Menzel, M.; Kiesow, A.; Schaffrath, U.; Deising, H.B.; Horbach, R. Melanin Is Not Required for Turgor Generation but Enhances Cell-Wall Rigidity in Appressoria of the Corn Pathogen Colletotrichum graminicola. Mol. Plant Microbe 2014, 27, 315–327. [Google Scholar] [CrossRef]
- Perpetua, N.S.; Kubo, Y.; Yasuda, N.; Takano, Y.; Furusawa, I. Cloning and characterization of a melanin biosynthetic THR1 reductase gene essential for appressorial penetration of Colletotrichum lagenaium. Mol. Plant-Microbe Interact. J. 1996, 9, 323–329. [Google Scholar] [CrossRef]
- Wang, X.; Lu, D.; Tian, C. Analysis of melanin biosynthesis in the plant pathogenic fungus Colletotrichum gloeosporioides. Fungal Biol. 2021, 125, 679–692. [Google Scholar] [CrossRef]
- Qin, X.Y.; Tian, C.M.; Meng, F.L. Comparative Transcriptome Analysis Reveals the Effect of the DHN Melanin Biosynthesis Pathway on the Appressorium Turgor Pressure of the Poplar Anthracnose-Causing Fungus Colletotrichum gloeosporioides. Int. J. Mol. Sci. 2023, 24, 7411. [Google Scholar] [CrossRef] [PubMed]
- Kihara, J.; Moriwaki, A.; Ito, M.; Arase, S.; Honda, Y. Expression of THR1, a 1,3,8-trihydroxynaphthalene reductase gene involved in melanin biosynthesis in the phytopathogenic fungus Bipolaris oryzae, is enhanced by near-ultraviolet radiation. Pigment. Cell Res. 2004, 17, 15–23. [Google Scholar] [CrossRef]
- Zhu, P.K.; LI, Q.; Zhang, C.H.; Na, Y.T.; Xu, L. Bcpks12 gene inactivation substantiates biological functions of sclerotium melanization in Botrytis cinerea. Physiol. Mol. Plant Pathol. 2017, 98, 80–84. [Google Scholar] [CrossRef]
- Verde-Yanez, L.; Vall-Llaura, N.; Usall, J.; Teixido, N.; Torreblanca-Bravo, E.; Torres, R. Identification and Biosynthesis of DHN-melanin Related Pigments in the Pathogenic Fungi Monilinia laxa, M. fructicola, and M. fructigena. J. Fungi 2023, 9, 138. [Google Scholar] [CrossRef]
- Langfelder, K.; Streibel, M.; Jahn, B.; Haase, G.; Brakhage, A.A. Biosynthesis of fungal melanins and their importance for human pathogenic fungi. Fungal Genet. Biol. 2003, 38, 143–158. [Google Scholar] [CrossRef]
- Li, H.; Wang, D.; Zhang, D.D.; Geng, Q.; Li, J.J.; Sheng, R.C.; Xue, H.S.; Zhu, H.; Kong, Z.Q.; Dai, X.F.; et al. A polyketide synthase from Verticillium dahliae modulates melanin biosynthesis and hyphal growth to promote virulence. BMC Biol. 2022, 20, 125. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; He, Y.; Zhu, P.; Chen, L.; Wang, Y.; Ni, B.; Xu, L. Loss of bcbrn1 and bcpks13 in Botrytis cinerea Not Only Blocks Melanization But Also Increases Vegetative Growth and Virulence. Mol. Plant Microbe Interact. 2015, 28, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Xu, J.P. Fungal Pigments and Their Roles Associated with Human Health. J. Fungi 2020, 6, 280. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.J.; Li, N.; Yang, J.Y.; Yang, L.; Wu, M.D.; Chen, W.D.; Li, G.Q.; Zhang, J. Contrast Between Orange- and Black-Colored Sclerotial Isolates of Botrytis cinerea: Melanogenesis and Ecological Fitness. Plant Dis. 2018, 102, 428–436. [Google Scholar] [CrossRef]
- Liang, Y.; Xiong, W.; Steinkellner, S.; Feng, J. Deficiency of the melanin biosynthesis genes SCD1 and THR1 affects sclerotial development and vegetative growth, but not pathogenicity, in Sclerotinia sclerotiorum. Mol. Plant Pathol. 2018, 19, 1444–1453. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, G.; Sugahara, T.; Fujii, I.; Mori, Y.; Ebizuka, Y.; Shiraishi, T.; Kubo, Y. Evidence for involvement of two naphthol reductases in the first reduction step of melanin biosynthesis pathway of Colletotrichum lagenarium. Mycol. Res. 2003, 107, 854–860. [Google Scholar] [CrossRef]




| Primer Name | Sequence (5′~3′) |
|---|---|
| ACTIN | CCCCAAGTCCAACAGAGAGA |
| CATCAGGTAGTCGGTCAAGTCA | |
| β-TUBULIN | GCCCTATTCTCGCTCGTCTTCC |
| GGGCTCCAAATCGCAGTAAATG | |
| PKS-qP | GTTCAAGTGGTTCTCATGGCTT |
| CTGGCGATGGGGATGTAATTAG | |
| THR1-qP | ACTTTGTCATCTCCAACTCGG |
| ATGGAGGAGGTGAGGAGGAT | |
| THNR-qP | GCACCATCGAGACATTTGT |
| GGTACATGTCGGTCTTGATG | |
| PKS-UP | ACGACGGCCAGTGCCAAGCTTCTGCTTTCAAACTCGTTTCACG |
| GACCTGCAGGCATGCAAGCTCAAGAACAAGATTTCTGGATGTCG | |
| PKS-DS | GACTCTAGAACTAGTGGATCCGGAACTTCATAGCGAAGCATATGA |
| AGCTCGGTACCCGGGGGATCCGGAGTATTCGGGGAAGGAGAAT | |
| THR1-UP | ACGACGGCCAGTGCCAAGCTTTTAAGCCAAAAAACGACTTGATAGA |
| GACCTGCAGGCATGCAAGCTTAGTTGCTATGAAAAGACTGTGGCG | |
| THR1-DS | CCGGGTACCGAGCTCGAATTCGTGCGATGATGCCACAAACTC |
| TATGGAGAAACTCGAGAATTCGACACCGGGAGAGGGGAGG | |
| PKSc-F1 | GAAACTCGAGCTCGAGAATTCATCGGTACCCTATCTGGCTGC |
| CCGGGTACCGAGCTCGAATTCGCTCGCTGGTTACCTCGTTCG | |
| PKSc-F2 | GACTCTAGATCTAGAGTCGACCTGCTTTCAAACTCGTTTCACG |
| CTTGCATGCCTGCAGGTCGACTTACTCGCAAACCGCTTCACG | |
| PKSc-F3 | GACCTGCAGGCATGCAAGCTTGGAACTTCATAGCGAAGCATATGA |
| ACGACGGCCAGTGCCAAGCTTGTAACGACAAAAAAAGCCACAGG | |
| THR1c-F1 | GAAACTCGAGCTCGAGAATTCCTCTAGCCGAAGGTATGAACCG |
| CCGGGTACCGAGCTCGAATTCCCGCCGCCACGGAGGGGC | |
| THR1c-F2 | ACTCTAGATCTAGAGTCGACCGGTGGCGGCGGCATGGTCGCG |
| TCACACCAGATCCGCCTGTGCCGCCACCGGTGACC | |
| THR1c-F3 | CACAGGCGGATCTGGTGTGAGCA |
| GGCATCATCGCACTTACTTGTACAGCTCGTCCATGCC | |
| THR1c-F4 | CAAGTAAGTGCGATGATGCCACAAACTC |
| CTTGCATGCCTGCAGGTCGACCCGACATCCGGGGAACAC | |
| PKS | GCACCGGTACCCAGG |
| GGTAGATGGTGTCGCTGG | |
| THR1 | ATGGCGCCCTCAGCGACTGAGAA |
| CCTAGATATAAGCTATGTTAGGTATCCC | |
| HPH1 | GGGCGTCGGTTTCCACTAT |
| GATATGTCCTGCGGGTAAATAGC | |
| NeoR | GATAGAAGGCGATGCGCT |
| ACCCGGTCATACCTTCTTAAG | |
| PKS-probe | CACAGGACATTACATGACGCA |
| CACCCTTGCTTATAACTTCGCA | |
| THR1-probe | ACATAGCTTATATCTAGGACAGGCG |
| TGTTACTAAACCTAGCTTGGGAAGG |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duan, L.; Wang, L.; Chen, W.; He, Z.; Zhou, E.; Zhu, Y. Deficiency of ChPks and ChThr1 Inhibited DHN-Melanin Biosynthesis, Disrupted Cell Wall Integrity and Attenuated Pathogenicity in Colletotrichum higginsianum. Int. J. Mol. Sci. 2023, 24, 15890. https://doi.org/10.3390/ijms242115890
Duan L, Wang L, Chen W, He Z, Zhou E, Zhu Y. Deficiency of ChPks and ChThr1 Inhibited DHN-Melanin Biosynthesis, Disrupted Cell Wall Integrity and Attenuated Pathogenicity in Colletotrichum higginsianum. International Journal of Molecular Sciences. 2023; 24(21):15890. https://doi.org/10.3390/ijms242115890
Chicago/Turabian StyleDuan, Lingtao, Li Wang, Weilun Chen, Zhenrui He, Erxun Zhou, and Yiming Zhu. 2023. "Deficiency of ChPks and ChThr1 Inhibited DHN-Melanin Biosynthesis, Disrupted Cell Wall Integrity and Attenuated Pathogenicity in Colletotrichum higginsianum" International Journal of Molecular Sciences 24, no. 21: 15890. https://doi.org/10.3390/ijms242115890
APA StyleDuan, L., Wang, L., Chen, W., He, Z., Zhou, E., & Zhu, Y. (2023). Deficiency of ChPks and ChThr1 Inhibited DHN-Melanin Biosynthesis, Disrupted Cell Wall Integrity and Attenuated Pathogenicity in Colletotrichum higginsianum. International Journal of Molecular Sciences, 24(21), 15890. https://doi.org/10.3390/ijms242115890







