Global Proteomics for Identifying the Alteration Pathway of Niemann–Pick Disease Type C Using Hepatic Cell Models
Abstract
1. Introduction
2. Results and Discussion
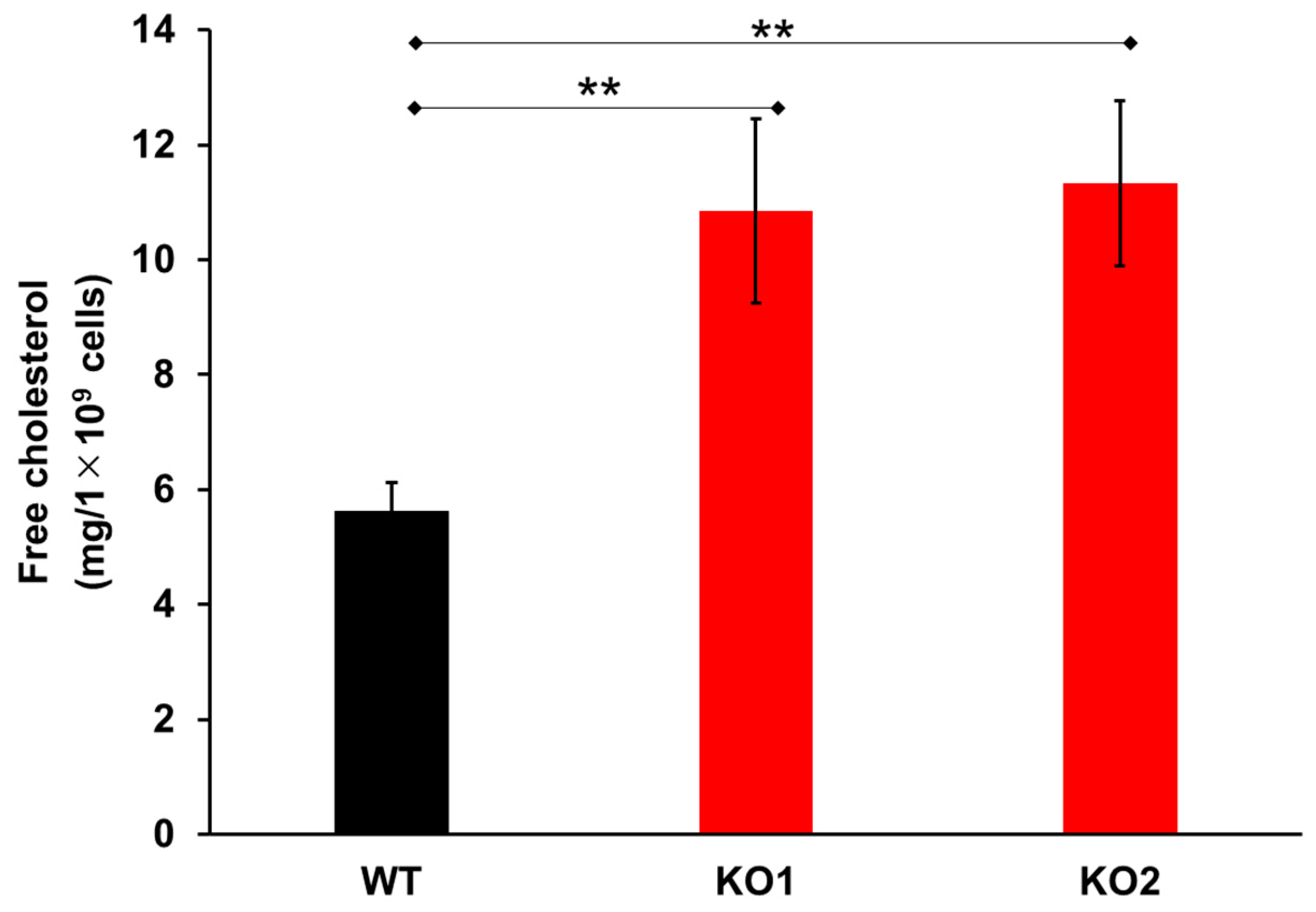
2.1. NPC Model Cell Development
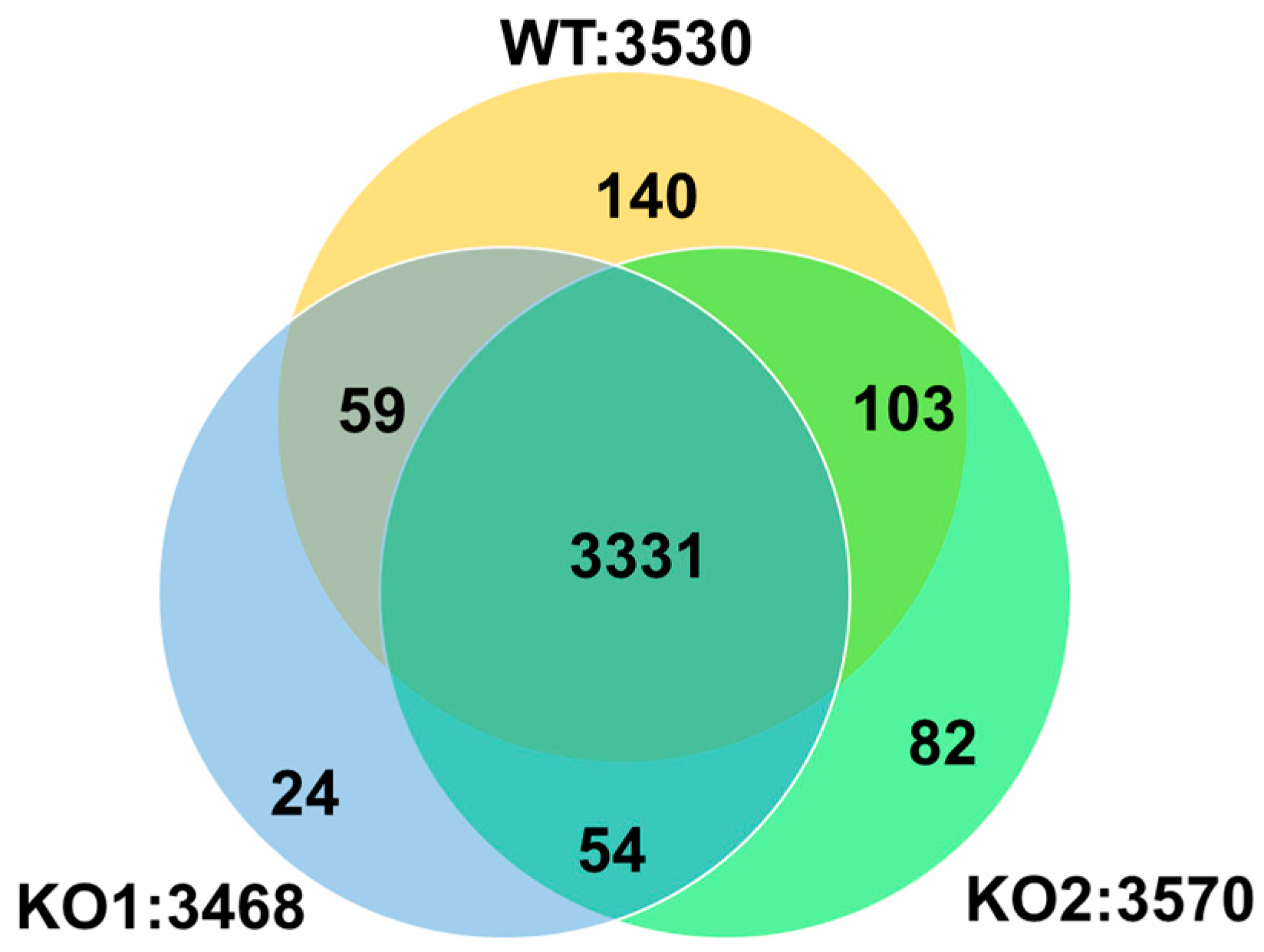
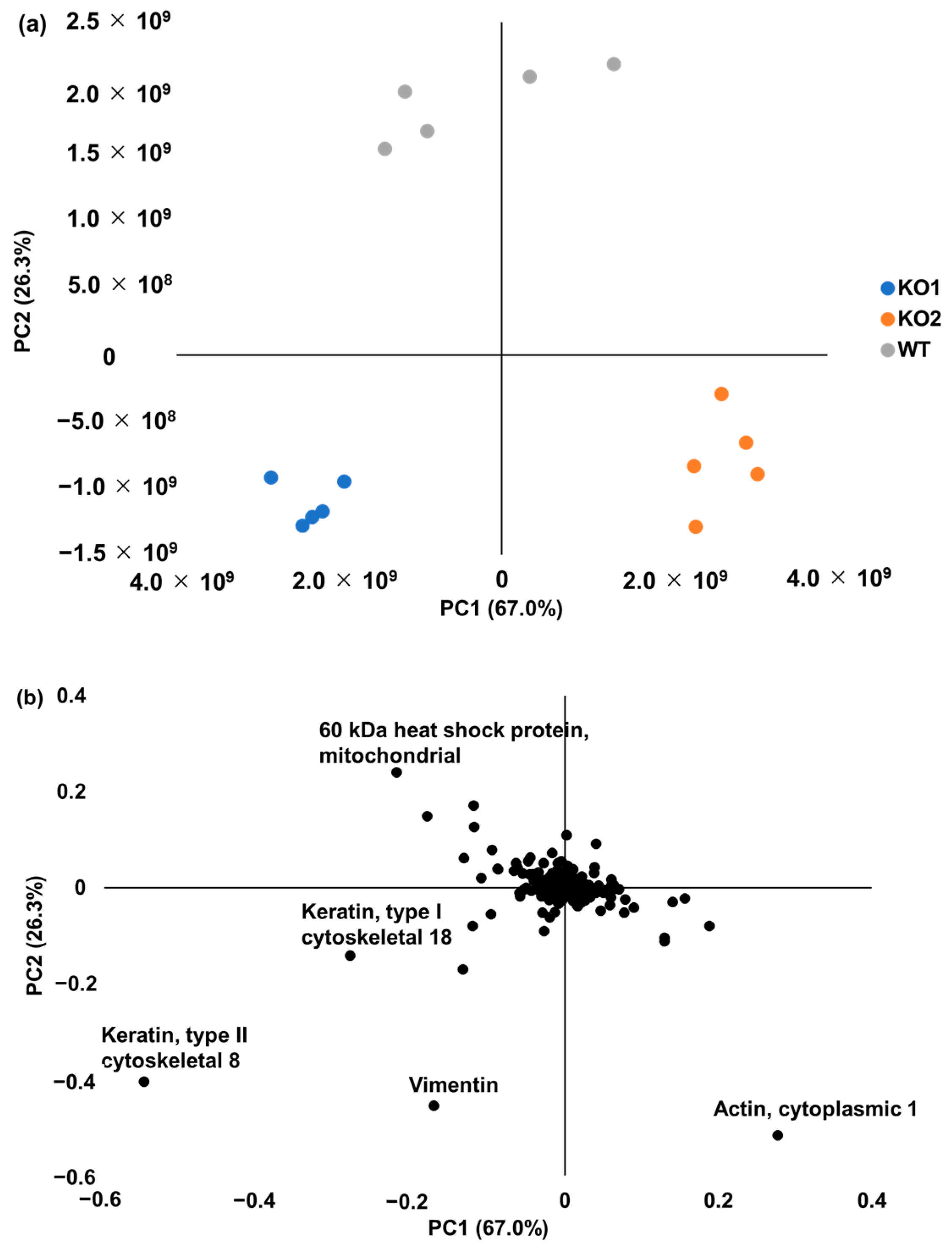
2.2. Label-Free Global Proteomics
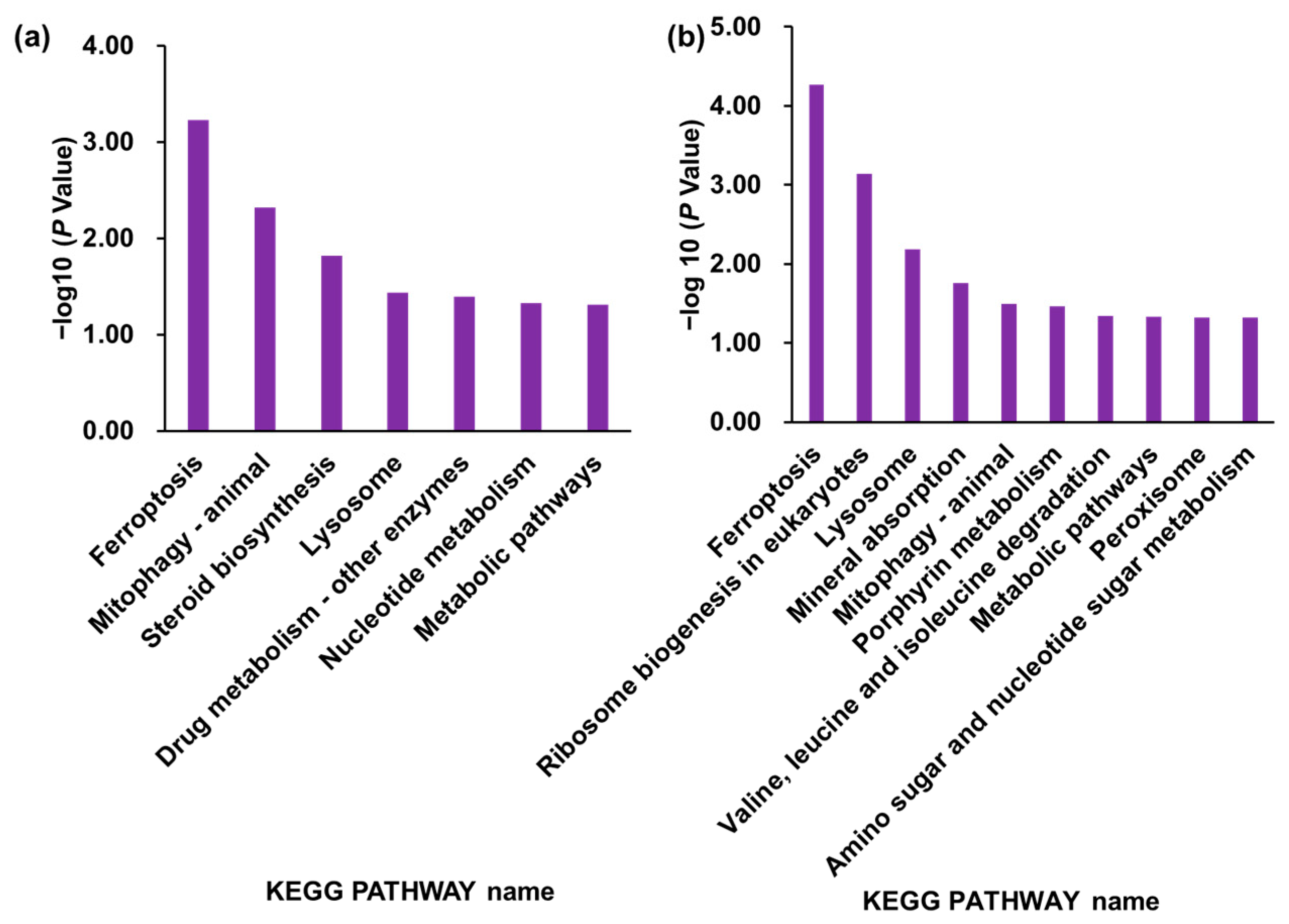
2.3. Bioinfomatics Analysis
2.4. The Accumulation of Lipid Peroxide Was Observed in Two Types of NPC Model Cells
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. LC/MS/MS Equipment, General Conditions, Data Acquisition, and Data Analysis
3.3. The Establishment of NPC Cell Models
3.4. Cell Culture and Cellular Protein Extraction
3.5. nLC Condition for Label-Free Global Proteomics
3.6. Cell Fluorescence Intensity Measurement
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vanier, M.T. Niemann-Pick Disease Type C. Orphanet J. Rare Dis. 2010, 5, 16. [Google Scholar] [CrossRef]
- Patterson, M.C.; Hendriksz, C.J.; Walterfang, M.; Sedel, F.; Vanier, M.T.; Wijburg, F. Recommendations for the Diagnosis and Management of Niemann-Pick Disease Type C: An Update. Mol. Genet. Metab. 2012, 106, 330–344. [Google Scholar] [CrossRef]
- Walkley, S.U.; Suzuki, K. Consequences of NPC1 and NPC2 Loss of Function in Mammalian Neurons. Biochim. Biophys. Acta 2004, 1685, 48–62. [Google Scholar] [CrossRef]
- Maekawa, M.; Mano, N. Identification and Evaluation of Biomarkers for Niemann-Pick Disease Type C Based on Chemical Analysis Techniques. Chromatography 2020, 41, 19–29. [Google Scholar] [CrossRef]
- Maekawa, M.; Iwahori, A.; Mano, N. Biomarker Analysis of Niemann-Pick Disease Type C Using Chromatography and Mass Spectrometry. J. Pharm. Biomed. Anal. 2020, 191, 113622. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, M.; Mano, N. Searching, Structural Determination, and Diagnostic Performance Evaluation of Biomarker Molecules for Niemann-Pick Disease Type C Using Liquid Chromatography/Tandem Mass Spectrometry. Mass Spectrom. 2022, 11, A0111. [Google Scholar] [CrossRef]
- Maekawa, M.; Mano, N. Cutting-Edge LC-MS/MS Applications in Clinical Mass Spectrometry: Focusing on Analysis of Drugs and Metabolites. Biomed. Chromatogr. 2022, 36, e5347. [Google Scholar] [CrossRef]
- Jiang, X.; Sidhu, R.; Orsini, J.J.; Farhat, N.Y.; Porter, F.D.; Berry-Kravis, E.; Schaffer, J.E.; Ory, D.S. Diagnosis of Niemann-Pick C1 by Measurement of Bile Acid Biomarkers in Archived Newborn Dried Blood Spots. Mol. Genet. Metab. 2019, 126, 183–187. [Google Scholar] [CrossRef]
- Sidhu, R.; Kell, P.; Dietzen, D.J.; Farhat, N.Y.; Do, A.N.D.; Porter, F.D.; Berry-Kravis, E.; Reunert, J.; Marquardt, T.; Giugliani, R.; et al. Application of a Glycinated Bile Acid Biomarker for Diagnosis and Assessment of Response to Treatment in Niemann-Pick Disease Type C1. Mol. Genet. Metab. 2020, 131, 405–417. [Google Scholar] [CrossRef]
- Geberhiwot, T.; Moro, A.; Dardis, A.; Ramaswami, U.; Sirrs, S.; Marfa, M.P.; Vanier, M.T.; Walterfang, M.; Bolton, S.; Dawson, C.; et al. Consensus Clinical Management Guidelines for Niemann-Pick Disease Type C. Orphanet J. Rare Dis. 2018, 13, 50. [Google Scholar] [CrossRef]
- Kwon, H.J.; Abi-Mosleh, L.; Wang, M.L.; Deisenhofer, J.; Goldstein, J.L.; Brown, M.S.; Infante, R.E. Structure of N-Terminal Domain of NPC1 Reveals Distinct Subdomains for Binding and Transfer of Cholesterol. Cell 2009, 137, 1213–1224. [Google Scholar] [CrossRef]
- Pentchev, P.G.; Comly, M.E.; Kruth, H.S.; Vanier, M.T.; Wenger, D.A.; Patel, S.; Brady, R.O. A Defect in Cholesterol Esterification in Niemann-Pick Disease (Type C) Patients. Proc. Natl. Acad. Sci. USA 1985, 82, 8247–8251. [Google Scholar] [CrossRef] [PubMed]
- Pentchev, P.G.; Boothe, A.D.; Kurth, H.S. A Genetic Storage Disorder in BALB/C Mice with a Metabolic Block in Esterification of Exogenous Cholesterol. J. Biol. Chem. 1984, 259, 5784–5791. [Google Scholar] [CrossRef] [PubMed]
- Kuchar, L.; Sikora, J.; Gulinello, M.E.; Poupetova, H.; Lugowska, A.; Malinova, V.; Jahnova, H.; Asfaw, B.; Ledvinova, J. Quantitation of Plasmatic Lysosphingomyelin and Lysosphingomyelin-509 for Differential Screening of Niemann-Pick A/B and C Diseases. Anal. Biochem. 2017, 525, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Iwamoto, T.; Hossain, M.A.; Akiyama, K.; Igarashi, J.; Miyajima, T.; Eto, Y. A Combination of 7-Ketocholesterol, Lysosphingomyelin and Bile Acid-408 to Diagnose Niemann-Pick Disease Type C Using LC-MS/MS. PLoS ONE 2020, 15, e0238624. [Google Scholar] [CrossRef] [PubMed]
- Vanier, M.T. Biochemical Studies in Niemann-Pick Disease. I. Major Sphingolipids of Liver and Spleen. Biochim. Biophys. Acta 1983, 750, 178–184. [Google Scholar] [CrossRef]
- Svennerholm, L.; Vanier, M.T. The Distribution of Lipids in the Human Nervous System. IV. Fatty Acid Composition of Major Sphingolipids of Human Infant Brain. Brain Res. 1973, 55, 413–423. [Google Scholar] [CrossRef]
- Patterson, M.C.; Vecchio, D.; Prady, H.; Abel, L.; Wraith, J.E. Miglustat for Treatment of Niemann-Pick C Disease: A Randomised Controlled Study. Lancet Neurol. 2007, 6, 765–772. [Google Scholar] [CrossRef]
- Jiang, X.; Sidhu, R.; Porter, F.D.; Yanjanin, N.M.; Speak, A.O.; Te Vruchte, D.T.; Platt, F.M.; Fujiwara, H.; Scherrer, D.E.; Zhang, J.; et al. A Sensitive and Specific LC-MS/MS Method for Rapid Diagnosis of Niemann-Pick C1 Disease from Human Plasma. J. Lipid Res. 2011, 52, 1435–1445. [Google Scholar] [CrossRef]
- Porter, F.D.; Scherrer, D.E.; Lanier, M.H.; Langmade, S.J.; Molugu, V.; Gale, S.E.; Olzeski, D.; Sidhu, R.; Dietzen, D.J.; Fu, R.; et al. Cholesterol Oxidation Products Are Sensitive and Specific Blood-Based Biomarkers for Niemann-Pick C1 Disease. Sci. Transl. Med. 2010, 2, 56ra81. [Google Scholar] [CrossRef]
- Welford, R.W.D.; Garzotti, M.; Lourenço, C.M.; Mengel, E.; Marquardt, T.; Reunert, J.; Amraoui, Y.; Kolb, S.A.; Morand, O.; Groenen, P. Plasma Lysosphingomyelin Demonstrates Great Potential as a Diagnostic Biomarker for Niemann-Pick Disease Type C in a Retrospective Study. PLoS ONE 2014, 9, e114669. [Google Scholar] [CrossRef]
- Sidhu, R.; Mondjinou, Y.; Qian, M.; Song, H.; Kumar, A.B.; Hong, X.; Hsu, F.F.; Dietzen, D.J.; Yanjanin, N.M.; Porter, F.D.; et al. N-Acyl-O-Phosphocholineserines: Structures of a Novel Class of Lipids That Are Biomarkers for Niemann-Pick C1 Disease. J. Lipid Res. 2019, 60, 1410–1424. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, M.; Jinnoh, I.; Matsumoto, Y.; Narita, A.; Mashima, R.; Takahashi, H.; Iwahori, A.; Saigusa, D.; Fujii, K.; Abe, A.; et al. Structural Determination of Lysosphingomyelin-509 and Discovery of Novel Class Lipids from Patients with Niemann-Pick Disease Type C. Int. J. Mol. Sci. 2019, 20, 5018. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, R.; Kell, P.; Dietzen, D.J.; Farhat, N.Y.; Do, A.N.D.; Porter, F.D.; Berry-Kravis, E.; Vite, C.H.; Reunert, J.; Marquardt, T.; et al. Application of N-Palmitoyl-O-Phosphocholineserine for Diagnosis and Assessment of Response to Treatment in Niemann-Pick Type C Disease. Mol. Genet. Metab. 2020, 129, 292. [Google Scholar] [CrossRef] [PubMed]
- Iwahori, A.; Maekawa, M.; Narita, A.; Kato, A.; Sato, T.; Ogura, J.; Sato, Y.; Kikuchi, M.; Noguchi, A.; Higaki, K.; et al. Development of a Diagnostic Screening Strategy for Niemann–Pick Diseases Based on Simultaneous Liquid Chromatography-Tandem Mass Spectrometry Analyses of N-Palmitoyl-O-Phosphocholine-Serine and Sphingosylphosphorylcholine. Biol. Pharm. Bull. 2020, 43, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Sidhu, R.; Mydock-McGrane, L.; Hsu, F.F.; Covey, D.F.; Scherrer, D.E.; Earley, B.; Gale, S.E.; Farhat, N.Y.; Porter, F.D.; et al. Development of a Bile Acid-Based Newborn Screen for Niemann-Pick C Disease. Sci. Transl. Med. 2016, 8, 337ra63. [Google Scholar] [CrossRef]
- Mazzacuva, F.; Mills, P.; Mills, K.; Camuzeaux, S.; Gissen, P.; Nicoli, E.R.; Wassif, C.; te Vruchte, D.; Porter, F.D.; Maekawa, M.; et al. Identification of Novel Bile Acids as Biomarkers for the Early Diagnosis of Niemann-Pick C Disease. FEBS Lett. 2016, 590, 1651–1662. [Google Scholar] [CrossRef]
- Maekawa, M.; Misawa, Y.; Sotoura, A.; Yamaguchi, H.; Togawa, M.; Ohno, K.; Nittono, H.; Kakiyama, G.; Iida, T.; Hofmann, A.F.; et al. LC/ESI-MS/MS Analysis of Urinary 3β-Sulfooxy-7β-N-Acetylglucosaminyl-5-Cholen-24-Oic Acid and Its Amides: New Biomarkers for the Detection of Niemann-Pick Type C Disease. Steroids 2013, 78, 967–972. [Google Scholar] [CrossRef]
- Maekawa, M.; Shimada, M.; Ohno, K.; Togawa, M.; Nittono, H.; Iida, T.; Hofmann, A.F.; Goto, J.; Yamaguchi, H.; Mano, N. Focused Metabolomics Using Liquid Chromatography/Electrospray Ionization Tandem Mass Spectrometry for Analysis of Urinary Conjugated Cholesterol Metabolites from Patients with Niemann-Pick Disease Type C and 3β-Hydroxysteroid Dehydrogenase Deficiency. Ann. Clin. Biochem. 2015, 52, 576–587. [Google Scholar] [CrossRef]
- Maekawa, M.; Omura, K.; Sekiguchi, S.; Iida, T.; Saigusa, D.; Yamaguchi, H.; Mano, N. Identification of Two Sulfated Cholesterol Metabolites Found in the Urine of a Patient with Niemann-Pick Disease Type C as Novel Candidate Diagnostic Markers. Mass Spectrom. 2016, 5, S0053. [Google Scholar] [CrossRef]
- Maekawa, M.; Narita, A.; Jinnoh, I.; Iida, T.; Marquardt, T.; Mengel, E.; Eto, Y.; Clayton, P.T.; Yamaguchi, H.; Mano, N. Diagnostic Performance Evaluation of Sulfate-Conjugated Cholesterol Metabolites as Urinary Biomarkers of Niemann–Pick Disease Type C. Clin. Chim. Acta 2019, 494, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, M.; Jinnoh, I.; Narita, A.; Iida, T.; Saigusa, D.; Iwahori, A.; Nittono, H.; Okuyama, T.; Eto, Y.; Ohno, K.; et al. Investigation of Diagnostic Performance of Five Urinary Cholesterol Metabolites for Niemann-Pick Disease Type C. J. Lipid Res. 2019, 60, 2074–2081. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, M.; Miyoshi, K.; Narita, A.; Sato, T.; Sato, Y.; Kumondai, M.; Kikuchi, M.; Higaki, K.; Okuyama, T.; Eto, Y.; et al. Development of a Highly Sensitive and Rapid Liquid Chromatography-Tandem Mass Spectrometric Method Using a Basic Mobile Phase Additive to Determine the Characteristics of the Urinary Metabolites for Niemann-Pick Disease Type C. Biol. Pharm. Bull. 2022, 45, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhang, P.; Solari, F.A.; Sickmann, A.; Garcia, A.; Jurk, K.; Heemskerk, J.W.M. Molecular Proteomics and Signalling of Human Platelets in Health and Disease. Int. J. Mol. Sci. 2021, 22, 9860. [Google Scholar] [CrossRef]
- Pisitkun, T.; Shen, R.F.; Knepper, M.A. Identification and Proteomic Profiling of Exosomes in Human Urine. Proc. Natl. Acad. Sci. USA 2004, 101, 13368–13373. [Google Scholar] [CrossRef]
- Straussman, R.; Morikawa, T.; Shee, K.; Barzily-Rokni, M.; Qian, Z.R.; Du, J.; Davis, A.; Mongare, M.M.; Gould, J.; Frederick, D.T.; et al. Tumour Micro-Environment Elicits Innate Resistance to RAF Inhibitors through HGF Secretion. Nature 2012, 487, 500–504. [Google Scholar] [CrossRef]
- Oda, T.; Matsumoto, K. ichi Proteomic Analysis in Cardiovascular Research. Surg. Today 2016, 46, 285–296. [Google Scholar] [CrossRef]
- Yao, X.Q.; Liu, Z.Y.; Chen, J.Y.; Huang, Z.C.; Liu, J.H.; Sun, B.H.; Zhu, Q.A.; Ding, R.T.; Chen, J.T. Proteomics and Bioinformatics Reveal Insights into Neuroinflammation in the Acute to Subacute Phases in Rat Models of Spinal Cord Contusion Injury. FASEB J. 2021, 35, e21735. [Google Scholar] [CrossRef]
- Vardi, A.; Pri-Or, A.; Wigoda, N.; Grishchuk, Y.; Futerman, A.H. Proteomics Analysis of a Human Brain Sample from a Mucolipidosis Type IV Patient Reveals Pathophysiological Pathways. Orphanet J. Rare Dis. 2021, 16, 39. [Google Scholar] [CrossRef]
- Li, S.M.; Liu, W.T.; Yang, F.; Yi, Q.J.; Zhang, S.; Jia, H.L. Phosphorylated Proteomics Analysis of Human Coronary Artery Endothelial Cells Stimulated by Kawasaki Disease Patients Serum. BMC Cardiovasc. Disord. 2019, 19, 21. [Google Scholar] [CrossRef]
- Youssef, L.; Miranda, J.; Blasco, M.; Paules, C.; Crovetto, F.; Palomo, M.; Torramade-Moix, S.; García-Calderó, H.; Tura-Ceide, O.; Dantas, A.P.; et al. Complement and Coagulation Cascades Activation Is the Main Pathophysiological Pathway in Early-Onset Severe Preeclampsia Revealed by Maternal Proteomics. Sci. Rep. 2021, 11, 3048. [Google Scholar] [CrossRef] [PubMed]
- Langley, S.R.; Dwyer, J.; Drozdov, I.; Yin, X.; Mayr, M. Proteomics: From Single Molecules to Biological Pathways. Cardiovasc. Res. 2013, 97, 612–622. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Herrmann, J.M.; Becker, T. Quality Control of the Mitochondrial Proteome. Nat. Rev. Mol. Cell Biol. 2021, 22, 54–70. [Google Scholar] [CrossRef]
- Ross, A.B.; Langer, J.D.; Jovanovic, M. Proteome Turnover in the Spotlight: Approaches, Applications, and Perspectives. Mol. Cell. Proteom. 2021, 20, 100016. [Google Scholar] [CrossRef] [PubMed]
- Doherty, M.K.; Beynon, R.J. Protein Turnover on the Scale of the Proteome. Expert Rev. Proteom. 2006, 3, 97–110. [Google Scholar] [CrossRef]
- Nemutlu, E.; Zhang, S.; Juranic, N.O.; Terzic, A.; Macura, S.; Dzeja, P. 18O-Assisted Dynamic Metabolomics for Individualized Diagnostics and Treatment of Human Diseases. Croat. Med. J. 2012, 53, 529–534. [Google Scholar] [CrossRef]
- Dai, D.F.; Karunadharma, P.P.; Chiao, Y.A.; Basisty, N.; Crispin, D.; Hsieh, E.J.; Chen, T.; Gu, H.; Djukovic, D.; Raftery, D.; et al. Altered Proteome Turnover and Remodeling by Short-Term Caloric Restriction or Rapamycin Rejuvenate the Aging Heart. Aging Cell 2014, 13, 529–539. [Google Scholar] [CrossRef]
- Ivanov, M.V.; Bubis, J.A.; Gorshkov, V.; Tarasova, I.A.; Levitsky, L.I.; Solovyeva, E.M.; Lipatova, A.V.; Kjeldsen, F.; Gorshkov, M.V. DirectMS1Quant: Ultrafast Quantitative Proteomics with MS/MS-Free Mass Spectrometry. Anal. Chem. 2022, 94, 13068–13075. [Google Scholar] [CrossRef]
- White, N.M.A.; Masui, O.; DeSouza, L.V.; Krakovska, O.; Metias, S.; Romaschin, A.D.; John Honey, R.; Stewart, R.; Pace, K.; Lee, J.; et al. Quantitative Proteomic Analysis Reveals Potential Diagnostic Markers and Pathways Involved in Pathogenesis of Renal Cell Carcinoma. Oncotarget 2014, 5, 506–518. [Google Scholar] [CrossRef]
- Ishihama, Y. Proteomic LC-MS Systems Using Nanoscale Liquid Chromatography with Tandem Mass Spectrometry. J. Chromatogr. A 2005, 1067, 73–83. [Google Scholar] [CrossRef]
- Label-Free LC-MS/MS Quantitative Analysis of Aqueous Humor from Keratoconic and Normal Eyes-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/25999673/ (accessed on 26 September 2023).
- Schirle, M.; Bantscheff, M.; Kuster, B. Mass Spectrometry-Based Proteomics in Preclinical Drug Discovery. Chem. Biol. 2012, 19, 72–84. [Google Scholar] [CrossRef] [PubMed]
- Ran, F.A.; Hsu, P.D.; Wright, J.; Agarwala, V.; Scott, D.A.; Zhang, F. Genome Engineering Using the CRISPR-Cas9 System. Nat. Protoc. 2013, 8, 2281–2308. [Google Scholar] [CrossRef] [PubMed]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A.; et al. Multiplex Genome Engineering Using CRISPR/Cas Systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef]
- Shirasago, Y.; Shimizu, Y.; Tanida, I.; Suzuki, T.; Suzuki, R.; Sugiyama, K.; Wakita, T.; Hanada, K.; Yagi, K.; Kondoh, M.; et al. Occludin-Knockout Human Hepatic Huh7.5.1-8-Derived Cells Are Completely Resistant to Hepatitis C Virus Infection. Biol. Pharm. Bull. 2016, 39, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Vanier, M.T.; Latour, P. Laboratory Diagnosis of Niemann-Pick Disease Type C: The Filipin Staining Test. Methods Cell Biol. 2015, 126, 357–375. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.A.; Zhang, D.; Coleman, K.G.; Nagle, J.; Pentchev, P.G.; Carstea, E.D. The Genomic Organization and Polymorphism Analysis of the Human Niemann-Pick C1 Gene. Biochem. Biophys. Res. Commun. 1999, 261, 493–498. [Google Scholar] [CrossRef]
- Yamamoto, T.; Nanba, E.; Ninomiya, H.; Higaki, K.; Taniguchi, M.; Zhang, H.; Akaboshi, S.; Watanabe, Y.; Takeshima, T.; Inui, K.; et al. NPC1 Gene Mutations in Japanese Patients with Niemann-Pick Disease Type C. Hum. Genet. 1999, 105, 10–16. [Google Scholar] [CrossRef]
- Kawazoe, T.; Yamamoto, T.; Narita, A.; Ohno, K.; Adachi, K.; Nanba, E.; Noguchi, A.; Takahashi, T.; Maekawa, M.; Eto, Y.; et al. Phenotypic Variability of Niemann-Pick Disease Type C Including a Case with Clinically Pure Schizophrenia: A Case Report. BMC Neurol. 2018, 18, 117. [Google Scholar] [CrossRef]
- Novel Compound Heterozygous Mutation in NPC1 Gene Cause Niemann-Pick Disease Type C with Juvenile Onset-PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/32482919/ (accessed on 26 September 2023).
- Polese-Bonatto, M.; Bock, H.; Farias, A.C.S.; Mergener, R.; Matte, M.C.; Gil, M.S.; Nepomuceno, F.; Souza, F.T.S.; Gus, R.; Giugliani, R.; et al. Niemann-Pick Disease Type C: Mutation Spectrum and Novel Sequence Variations in the Human NPC1 Gene. Mol. Neurobiol. 2019, 56, 6426–6435. [Google Scholar] [CrossRef]
- McKay Bounford, K.; Gissen, P. Genetic and Laboratory Diagnostic Approach in Niemann Pick Disease Type C. J. Neurol. 2014, 261 (Suppl. 2), 569–575. [Google Scholar] [CrossRef]
- Ntai, I.; Kim, K.; Fellers, R.T.; Skinner, O.S.; Smith, A.D.; Early, B.P.; Savaryn, J.P.; Leduc, R.D.; Thomas, P.M.; Kelleher, N.L. Applying Label-Free Quantitation to Top down Proteomics. Anal. Chem. 2014, 86, 4961–4968. [Google Scholar] [CrossRef] [PubMed]
- Schork, K.; Podwojski, K.; Turewicz, M.; Stephan, C.; Eisenacher, M. Important Issues in Planning a Proteomics Experiment: Statistical Considerations of Quantitative Proteomic Data. Methods Mol. Biol. 2021, 2228, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Liu, Y.; Huang, Q.; Sun, S.; Ji, Z.; Huang, L.; Li, Z.; Huang, X.; Deng, W.; Li, T. TMT-Based Quantitative Proteomics Analysis of Synovial Fluid-Derived Exosomes in Inflammatory Arthritis. Front. Immunol. 2022, 13, 800902. [Google Scholar] [CrossRef]
- Li, X.; Dong, Y.; Tu, K.; Wang, W. Proteomics Analysis Reveals the Interleukin-35-Dependent Regulatory Mechanisms Affecting CD8+ T-Cell Functions. Cell. Immunol. 2020, 348, 104022. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Kanehisa, M.; Furumichi, M.; Tanabe, M.; Sato, Y.; Morishima, K. KEGG: New Perspectives on Genomes, Pathways, Diseases and Drugs. Nucleic Acids Res. 2017, 45, D353–D361. [Google Scholar] [CrossRef]
- Mou, Y.; Wang, J.; Wu, J.; He, D.; Zhang, C.; Duan, C.; Li, B. Ferroptosis, a New Form of Cell Death: Opportunities and Challenges in Cancer. J. Hematol. Oncol. 2019, 12, 34. [Google Scholar] [CrossRef]
- Moujalled, D.; Strasser, A.; Liddell, J.R. Molecular Mechanisms of Cell Death in Neurological Diseases. Cell Death Differ. 2021, 28, 2029–2044. [Google Scholar] [CrossRef]
- Hung, Y.H.; Faux, N.G.; Killilea, D.W.; Yanjanin, N.; Firnkes, S.; Volitakis, I.; Ganio, G.; Walterfang, M.; Hastings, C.; Porter, F.D.; et al. Altered Transition Metal Homeostasis in Niemann-Pick Disease, Type C1. Metallomics 2014, 6, 542–553. [Google Scholar] [CrossRef]
- Liang, L.; Wang, H.; Yao, J.; Wei, Q.; Lu, Y.; Wang, T.; Cao, X. NPC1 Deficiency Contributes to Autophagy-Dependent Ferritinophagy in HEI-OC1 Auditory Cells. Front. Mol. Biosci. 2022, 9, 952608. [Google Scholar] [CrossRef]
- Mancias, J.D.; Wang, X.; Gygi, S.P.; Harper, J.W.; Kimmelman, A.C. Quantitative Proteomics Identifies NCOA4 as the Cargo Receptor Mediating Ferritinophagy. Nature 2014, 509, 105. [Google Scholar] [CrossRef]
- Hou, W.; Xie, Y.; Song, X.; Sun, X.; Lotze, M.T.; Iii, H.J.Z.; Kang, R.; Tang, D. Autophagy Promotes Ferroptosis by Degradation of Ferritin. Autophagy 2016, 12, 1425–1428. [Google Scholar] [CrossRef] [PubMed]
- Santana-Codina, N.; Mancias, J.D. The Role of NCOA4-Mediated Ferritinophagy in Health and Disease. Pharmaceuticals 2018, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Li, C.; Liao, S.; Yao, X.; Ouyang, Y.; Liu, Y.; Wang, Z.; Li, Z.; Yao, F. Ferritinophagy, a Form of Autophagic Ferroptosis: New Insights into Cancer Treatment. Front. Pharmacol. 2022, 13, 1043344. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Hou, W.; Song, X.; Yu, Y.; Huang, J.; Sun, X.; Kang, R.; Tang, D. Ferroptosis: Process and Function. Cell Death Differ. 2016, 23, 369–379. [Google Scholar] [CrossRef] [PubMed]
- Lamark, T.; Johansen, T. Mechanisms of Selective Autophagy. Annu. Rev. Cell Dev. Biol. 2021, 37, 143–169. [Google Scholar] [CrossRef]
- Yuan, X.; Fleming, M.D.; Hamza, I. Heme Transport and Erythropoiesis. Curr. Opin. Chem. Biol. 2013, 17, 204. [Google Scholar] [CrossRef]
- Song, G.; Zhang, S.; Tian, M.; Zhang, L.; Guo, R.; Zhuo, W.; Yang, M. Molecular Insights into the Human ABCB6 Transporter. Cell Discov. 2021, 7, 55. [Google Scholar] [CrossRef]
- Boswell-Casteel, R.C.; Fukuda, Y.; Schuetz, J.D. ABCB6, an ABC Transporter Impacting Drug Response and Disease. AAPS J. 2017, 20, 8. [Google Scholar] [CrossRef]
- Chen, K.Z.; Liu, S.X.; Li, Y.W.; He, T.; Zhao, J.; Wang, T.; Qiu, X.X.; Wu, H.F. Vimentin as a Potential Target for Diverse Nervous System Diseases. Neural Regen. Res. 2023, 18, 969. [Google Scholar] [CrossRef]
- Biskou, O.; Casanova, V.; Hooper, K.M.; Kemp, S.; Wright, G.P.; Satsangi, J.; Barlow, P.G.; Stevens, C. The Type III Intermediate Filament Vimentin Regulates Organelle Distribution and Modulates Autophagy. PLoS ONE 2019, 14, e0209665. [Google Scholar] [CrossRef]
- Mohanasundaram, P.; Coelho-Rato, L.S.; Modi, M.K.; Urbanska, M.; Lautenschläger, F.; Cheng, F.; Eriksson, J.E. Cytoskeletal Vimentin Regulates Cell Size and Autophagy through MTORC1 Signaling. PLoS Biol. 2022, 20, e3001737. [Google Scholar] [CrossRef]
- Walter, M.; Chen, F.W.; Tamari, F.; Wang, R.; Ioannou, Y.A. Endosomal Lipid Accumulation in NPC1 Leads to Inhibition of PKC, Hypophosphorylation of Vimentin and Rab9 Entrapment. Biol. Cell 2009, 101, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Sarria, A.J.; Panini, S.R.; Evans, R.M. A Functional Role for Vimentin Intermediate Filaments in the Metabolism of Lipoprotein-Derived Cholesterol in Human SW-13 Cells. J. Biol. Chem. 1992, 267, 19455–19463. [Google Scholar] [CrossRef] [PubMed]
- Tamari, F.; Chen, F.W.; Li, C.; Chaudhari, J.; Ioannou, Y.A. PKC Activation in Niemann Pick C1 Cells Restores Subcellular Cholesterol Transport. PLoS ONE 2013, 8, e74169. [Google Scholar] [CrossRef]
- Tharkeshwar, A.K.; Trekker, J.; Vermeire, W.; Pauwels, J.; Sannerud, R.; Priestman, D.A.; Te Vruchte, D.; Vints, K.; Baatsen, P.; Decuypere, J.P.; et al. A Novel Approach to Analyze Lysosomal Dysfunctions through Subcellular Proteomics and Lipidomics: The Case of NPC1 Deficiency. Sci. Rep. 2017, 7, 41408. [Google Scholar] [CrossRef]
- Wheeler, S.; Sillence, D.J. Niemann–Pick Type C Disease: Cellular Pathology and Pharmacotherapy. J. Neurochem. 2020, 153, 674–692. [Google Scholar] [CrossRef]
- Brady, R.O.; Kanfer, J.N.; Shapiro, D. Metabolism of Glucocerebrosides II. Evidence of an Enzymatic Deficiency in Gaucher’s Disease. Biochem. Biophys. Res. Commun. 1965, 18, 221–225. [Google Scholar] [CrossRef]
- Messner, M.C.; Cabot, M.C. Glucosylceramide in Humans. Adv. Exp. Med. Biol. 2010, 688, 156–164. [Google Scholar]
- Salvioli, R.; Scarpa, S.; Ciaffoni, F.; Tatti, M.; Ramoni, C.; Vanier, M.T.; Vaccaro, A.M. Glucosylceramidase Mass and Subcellular Localization Are Modulated by Cholesterol in Niemann-Pick Disease Type C. J. Biol. Chem. 2004, 279, 17674–17680. [Google Scholar] [CrossRef]
- Pentchev, P.G.; Gal, A.E.; Booth, A.D.; Omodeo-Sale, F.; Fours, J.; Neumeyer, B.A.; Quirk, J.M.; Dawson, G.; Brady, R.O. A Lysosomal Storage Disorder in Mice Characterized by a Dual Deficiency of Sphingomyelinase and Glucocerebrosidase. Biochim. Biophys. Acta (BBA)-Lipids Lipid Metab. 1980, 619, 669–679. [Google Scholar] [CrossRef]
- Marques, A.R.A.; Aten, J.; Ottenhoff, R.; Van Roomen, C.P.A.A.; Moro, D.H.; Claessen, N.; Veloz, M.F.V.; Zhou, K.; Lin, Z.; Mirzaian, M.; et al. Reducing GBA2 Activity Ameliorates Neuropathology in Niemann-Pick Type C Mice. PLoS ONE 2015, 10, e0135889. [Google Scholar] [CrossRef]
- Beck, M.; Olsen, K.J.; Wraith, J.E.; Zeman, J.; Michalski, J.C.; Saftig, P.; Fogh, J.; Malm, D. Natural History of Alpha Mannosidosis a Longitudinal Study. Orphanet J. Rare Dis. 2013, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hennermann, J.B.; Raebel, E.M.; Donà, F.; Jacquemont, M.L.; Cefalo, G.; Ballabeni, A.; Malm, D. Mortality in Patients with Alpha-Mannosidosis: A Review of Patients’ Data and the Literature. Orphanet J. Rare Dis. 2022, 17, 287. [Google Scholar] [CrossRef] [PubMed]
- Malm, D.; Riise Stensland, H.M.F.; Edvardsen, Ø.; Nilssen, Ø. The Natural Course and Complications of Alpha-Mannosidosis--a Retrospective and Descriptive Study. J. Inherit. Metab. Dis. 2014, 37, 79–82. [Google Scholar] [CrossRef]
- Karayel, O.; Virreira Winter, S.; Padmanabhan, S.; Kuras, Y.I.; Vu, D.T.; Tuncali, I.; Merchant, K.; Wills, A.M.; Scherzer, C.R.; Mann, M. Proteome Profiling of Cerebrospinal Fluid Reveals Biomarker Candidates for Parkinson’s Disease. Cell Rep. Med. 2022, 3, 100661. [Google Scholar] [CrossRef]
- Banno, Y.; Sasaki, N.; Miyawaki, S.; Kitagawa, T.; Nozawa, Y. Properties of Lysosomal Beta-Hexosaminidase Accumulated in Niemann-Pick Mouse Liver. Biochem. Med. Metab. Biol. 1986, 36, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Vischer, U.M.; Rosnoblet, C.; Lebrand, C.; Lindsay, M.; Parton, R.G.; Kruithof, E.K.O.; Gruenberg, J. The Tetraspanin CD63/Lamp3 Cycles between Endocytic and Secretory Compartments in Human Endothelial Cells. Mol. Biol. Cell 2000, 11, 1829–1843. [Google Scholar] [CrossRef]
- The Association of Ferritin and Its Cargo Receptor NCOA4 with CD63. CD63 orchestrates ferritin export. Blood 2021, 138, 1387–1389. [Google Scholar] [CrossRef]
- Yanatori, I.; Richardson, D.R.; Dhekne, H.S.; Toyokuni, S.; Kishi, F. CD63 Is Regulated by Iron via the IRE-IRP System and Is Important for Ferritin Secretion by Extracellular Vesicles. Blood 2021, 138, 1490–1503. [Google Scholar] [CrossRef]
- Leibold, E.A.; Laudano, A.; Yu, Y. Structural Requirements of Iron-Responsive Elements for Binding of the Protein Involved in Both Transferrin Receptor and Ferritin MRNA Post-Transcriptional Regulation. Nucleic Acids Res. 1990, 18, 1819. [Google Scholar] [CrossRef]
- Richardson, D.R.; Ponka, P. The Molecular Mechanisms of the Metabolism and Transport of Iron in Normal and Neoplastic Cells. Biochim. Biophys. Acta 1997, 1331, 1–40. [Google Scholar] [CrossRef] [PubMed]
- Sleat, D.E.; Wiseman, J.A.; Sohar, I.; El-Banna, M.; Zheng, H.; Moore, D.F.; Lobel, P. Proteomic Analysis of Mouse Models of Niemann-Pick C Disease Reveals Alterations in the Steady-State Levels of Lysosomal Proteins within the Brain. Proteomics 2012, 12, 3499–3509. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Pfeffer, S.R. Lysosomal Membrane Glycoproteins Bind Cholesterol and Contribute to Lysosomal Cholesterol Export. eLife 2016, 5, e21635. [Google Scholar] [CrossRef] [PubMed]
- Pereira, E.M.; do Monte, S.J.H.; do Nascimento, F.F.; de Castro, J.A.F.; Sousa, J.L.M.; Filho, H.C.S.A.L.C.; da Silva, R.N.; Labilloy, A.; Monte Neto, J.T.; da Silva, A.S. Lysosome-Associated Protein 1 (LAMP-1) and Lysosome-Associated Protein 2 (LAMP-2) in a Larger Family Carrier of Fabry Disease. Gene 2014, 536, 118–122. [Google Scholar] [CrossRef]
- Jehn, U.; Bayraktar, S.; Pollmann, S.; Van Marck, V.; Weide, T.; Pavenstädt, H.; Brand, E.; Lenders, M. α-Galactosidase a Deficiency in Fabry Disease Leads to Extensive Dysregulated Cellular Signaling Pathways in Human Podocytes. Int. J. Mol. Sci. 2021, 22, 11339. [Google Scholar] [CrossRef]
- Monticelli, M.; Hay Mele, B.; Allocca, M.; Liguori, L.; Lukas, J.; Monti, M.C.; Morretta, E.; Cubellis, M.V.; Andreotti, G. Curcumin Has Beneficial Effects on Lysosomal Alpha-Galactosidase: Potential Implications for the Cure of Fabry Disease. Int. J. Mol. Sci. 2023, 24, 1095. [Google Scholar] [CrossRef]
- Ashrafi, G.; Schwarz, T.L. The Pathways of Mitophagy for Quality Control and Clearance of Mitochondria. Cell Death Differ. 2013, 20, 31–42. [Google Scholar] [CrossRef]
- Düvel, K.; Yecies, J.L.; Menon, S.; Raman, P.; Lipovsky, A.I.; Souza, A.L.; Triantafellow, E.; Ma, Q.; Gorski, R.; Cleaver, S.; et al. Activation of a Metabolic Gene Regulatory Network Downstream of MTOR Complex 1. Mol. Cell 2010, 39, 171. [Google Scholar] [CrossRef]
- Settembre, C.; Zoncu, R.; Medina, D.L.; Vetrini, F.; Erdin, S.; Erdin, S.; Huynh, T.; Ferron, M.; Karsenty, G.; Vellard, M.C.; et al. A Lysosome-to-Nucleus Signalling Mechanism Senses and Regulates the Lysosome via MTOR and TFEB. EMBO J. 2012, 31, 1095. [Google Scholar] [CrossRef]
- Kim, J.; Kundu, M.; Viollet, B.; Guan, K.L. AMPK and MTOR Regulate Autophagy through Direct Phosphorylation of Ulk1. Nat. Cell Biol. 2011, 13, 132. [Google Scholar] [CrossRef] [PubMed]
- Elrick, M.J.; Yu, T.; Chung, C.; Lieberman, A.P. Impaired Proteolysis Underlies Autophagic Dysfunction in Niemann-Pick Type C Disease. Hum. Mol. Genet. 2012, 21, 4876–4887. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.E.; Madreiter, C.T.; Vishnu, N.; Malli, R.; Graier, W.F.; Karten, B. Adaptations of Energy Metabolism Associated with Increased Levels of Mitochondrial Cholesterol in Niemann-Pick Type C1-Deficient Cells. J. Biol. Chem. 2014, 289, 16278. [Google Scholar] [CrossRef]
- Yu, W.; Gong, J.S.; Ko, M.; Garver, W.S.; Yanagisawa, K.; Michikawa, M. Altered Cholesterol Metabolism in Niemann-Pick Type C1 Mouse Brains Affects Mitochondrial Function. J. Biol. Chem. 2005, 280, 11731–11739. [Google Scholar] [CrossRef]
- Ordonez, M.P. Defective Mitophagy in Human Niemann-Pick Type C1 Neurons Is Due to Abnormal Autophagy Activation. Autophagy 2012, 8, 1157–1158. [Google Scholar] [CrossRef] [PubMed]
- Davis, O.B.; Shin, H.R.; Lim, C.Y.; Wu, E.Y.; Kukurugya, M.; Maher, C.F.; Perera, R.M.; Ordonez, M.P.; Zoncu, R. NPC1-MTORC1 Signaling Couples Cholesterol Sensing to Organelle Homeostasis and Is a Targetable Pathway in Niemann-Pick Type C. Dev. Cell 2021, 56, 260–276.e7. [Google Scholar] [CrossRef] [PubMed]
- Thurston, T.L.M.; Wandel, M.P.; Von Muhlinen, N.; Foeglein, Á.; Randow, F. Galectin-8 Targets Damaged Vesicles for Autophagy to Defend Cells against Bacterial Invasion. Nature 2012, 482, 414. [Google Scholar] [CrossRef]
- Verlhac, P.; Viret, C.; Faure, M. Dual Function of CALCOCO2/NDP52 during Xenophagy. Autophagy 2015, 11, 965. [Google Scholar] [CrossRef]
- Towers, C.G.; Wodetzki, D.K.; Thorburn, J.; Smith, K.R.; Caino, M.C.; Thorburn, A. Mitochondrial Derived Vesicles Compensate for Loss of LC3-Mediated Mitophagy. Dev. Cell 2021, 56, 2029. [Google Scholar] [CrossRef]
- Weidberg, H.; Shvets, E.; Shpilka, T.; Shimron, F.; Shinder, V.; Elazar, Z. LC3 and GATE-16/GABARAP Subfamilies Are Both Essential yet Act Differently in Autophagosome Biogenesis. EMBO J. 2010, 29, 1792–1802. [Google Scholar] [CrossRef]
- Yamada, T.; Dawson, T.M.; Yanagawa, T.; Iijima, M.; Sesaki, H. SQSTM1/P62 Promotes Mitochondrial Ubiquitination Independently of PINK1 and PRKN/Parkin in Mitophagy. Autophagy 2019, 15, 2012–2018. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Long, H.; Hou, L.; Feng, B.; Ma, Z.; Wu, Y.; Zeng, Y.; Cai, J.; Zhang, D.; Zhao, G. The Mitophagy Pathway and Its Implications in Human Diseases. Signal Transduct. Target. Ther. 2023, 8, 304. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Ham, A.; Ma, T.C.; Kuo, S.H.; Kanter, E.; Kim, D.; Ko, H.S.; Quan, Y.; Sardi, S.P.; Li, A.; et al. Mitochondrial Dysfunction and Mitophagy Defect Triggered by Heterozygous GBA Mutations. Autophagy 2019, 15, 113. [Google Scholar] [CrossRef] [PubMed]
- Abe, A.; Maekawa, M.; Sato, T.; Sato, Y.; Kumondai, M.; Takahashi, H.; Kikuchi, M.; Higaki, K.; Ogura, J.; Mano, N. Metabolic Alteration Analysis of Steroid Hormones in Niemann-Pick Disease Type C Model Cell Using Liquid Chromatography/Tandem Mass Spectrometry. Int. J. Mol. Sci. 2022, 23, 4459. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, B.E.; Hundert, A.S.; Goguen, D.; Weaver, I.C.G.; Karten, B. Presymptomatic Alterations in Amino Acid Metabolism and DNA Methylation in the Cerebellum of a Murine Model of Niemann-Pick Type C Disease. Am. J. Pathol. 2016, 186, 1582–1597. [Google Scholar] [CrossRef] [PubMed]
- Yadid, G.; Sotnik-Barkai, I.; Tornatore, C.; Baker-Cairns, B.; Harvey-White, J.; Pentchev, P.G.; Goldin, E. Neurochemical Alterations in the Cerebellum of a Murine Model of Niemann-Pick Type C Disease. Brain Res. 1998, 799, 250–256. [Google Scholar] [CrossRef]
- Pergande, M.R.; Cougnoux, A.; Rathnayake, R.A.C.; Porter, F.D.; Cologna, S.M. Differential Proteomics Reveals MiR-155 as a Novel Indicator of Liver and Spleen Pathology in the Symptomatic Niemann-Pick Disease, Type C1 Mouse Model. Molecules 2019, 24, 994. [Google Scholar] [CrossRef]
- Balboa, E.; Marín, T.; Oyarzún, J.E.; Contreras, P.S.; Hardt, R.; van den Bosch, T.; Alvarez, A.R.; Rebolledo-Jaramillo, B.; Klein, A.D.; Winter, D.; et al. Proteomic Analysis of Niemann-Pick Type C Hepatocytes Reveals Potential Therapeutic Targets for Liver Damage. Cells 2021, 10, 2159. [Google Scholar] [CrossRef]
- Cougnoux, A.; Pergande, M.R.; Serna-Perez, F.; Cologna, S.M. Investigation of 2-Hydroxypropyl-β-Cyclodextrin Treatment in a Neuronal-Like Cell Model of Niemann-Pick Type C Using Quantitative Proteomics. J. Am. Soc. Mass Spectrom. 2023, 34, 668–675. [Google Scholar] [CrossRef]
- Tang, M.; Chen, Z.; Wu, D.; Chen, L. Ferritinophagy/Ferroptosis: Iron-Related Newcomers in Human Diseases. J. Cell. Physiol. 2018, 233, 9179–9190. [Google Scholar] [CrossRef]
- Wenz, C.; Faust, D.; Linz, B.; Turmann, C.; Nikolova, T.; Bertin, J.; Gough, P.; Wipf, P.; Schröder, A.S.; Krautwald, S.; et al. T-BuOOH Induces Ferroptosis in Human and Murine Cell Lines. Arch. Toxicol. 2017, 92, 759–775. [Google Scholar] [CrossRef] [PubMed]
- Stockwell, B.R.; Friedmann Angeli, J.P.; Bayir, H.; Bush, A.I.; Conrad, M.; Dixon, S.J.; Fulda, S.; Gascón, S.; Hatzios, S.K.; Kagan, V.E.; et al. Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease. Cell 2017, 171, 273–285. [Google Scholar] [CrossRef] [PubMed]
- Tsukui, T.; Chen, Z.; Fuda, H.; Furukawa, T.; Oura, K.; Sakurai, T.; Hui, S.P.; Chiba, H. Novel Fluorescence-Based Method to Characterize the Antioxidative Effects of Food Metabolites on Lipid Droplets in Cultured Hepatocytes. J. Agric. Food Chem. 2019, 67, 9934–9941. [Google Scholar] [CrossRef] [PubMed]
- Hung, Y.H.; Lotan, A.; Yeshurun, S.; Schroeder, A.; Bush, A.I. Iron Chelation by Deferiprone Does Not Rescue the Niemann-Pick Disease Type C1 Mouse Model. Biometals 2020, 33, 87–95. [Google Scholar] [CrossRef]
- Devos, D.; Moreau, C.; Devedjian, J.C.; Kluza, J.; Petrault, M.; Laloux, C.; Jonneaux, A.; Ryckewaert, G.; Garçon, G.; Rouaix, N.; et al. Targeting Chelatable Iron as a Therapeutic Modality in Parkinson’s Disease. Antioxid. Redox Signal. 2014, 21, 195–210. [Google Scholar] [CrossRef]
- Martin-Bastida, A.; Ward, R.J.; Newbould, R.; Piccini, P.; Sharp, D.; Kabba, C.; Patel, M.C.; Spino, M.; Connelly, J.; Tricta, F.; et al. Brain Iron Chelation by Deferiprone in a Phase 2 Randomised Double-Blinded Placebo Controlled Clinical Trial in Parkinson’s Disease. Sci. Rep. 2017, 7, 1398. [Google Scholar] [CrossRef]
- Wessling-Resnick, M. Excess Iron: Considerations Related to Development and Early Growth. Am. J. Clin. Nutr. 2017, 106, 1600S. [Google Scholar] [CrossRef]
- Dixon, S.J.; Lemberg, K.M.; Lamprecht, M.R.; Skouta, R.; Zaitsev, E.M.; Gleason, C.E.; Patel, D.N.; Bauer, A.J.; Cantley, A.M.; Yang, W.S.; et al. Ferroptosis: An Iron-Dependent Form of Nonapoptotic Cell Death. Cell 2012, 149, 1060–1072. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P.; et al. The STRING Database in 2021: Customizable Protein–Protein Networks, and Functional Characterization of User-Uploaded Gene/Measurement Sets. Nucleic Acids Res. 2021, 49, D605–D612. [Google Scholar] [CrossRef]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and Integrative Analysis of Large Gene Lists Using DAVID Bioinformatics Resources. Nat. Protoc. 2008, 4, 44–57. [Google Scholar] [CrossRef]
- Ong, Q.R.; Lim, M.L.; Chua, C.C.; Cheung, N.S.; Wong, B.S. Impaired Insulin Signaling in an Animal Model of Niemann-Pick Type C Disease. Biochem. Biophys. Res. Commun. 2012, 424, 482–487. [Google Scholar] [CrossRef] [PubMed]

| Upregulated DEPs (Numbers) | Downregulated DEPs (Numbers) | Total (Numbers) | |||
|---|---|---|---|---|---|
| Identified in KO1 (numbers) | 18 | 77 | 95 | ||
| 25 | (a) 2 | (b) 4 | 10 | 35 (41) | |
| Identified in KO2 (numbers) | (b) 4 | (a) 2 | |||
| 145 | 63 | 208 | |||
| Total | 188 | 6 | 150 | 338 (344) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyoshi, K.; Hishinuma, E.; Matsukawa, N.; Shirasago, Y.; Watanabe, M.; Sato, T.; Sato, Y.; Kumondai, M.; Kikuchi, M.; Koshiba, S.; et al. Global Proteomics for Identifying the Alteration Pathway of Niemann–Pick Disease Type C Using Hepatic Cell Models. Int. J. Mol. Sci. 2023, 24, 15642. https://doi.org/10.3390/ijms242115642
Miyoshi K, Hishinuma E, Matsukawa N, Shirasago Y, Watanabe M, Sato T, Sato Y, Kumondai M, Kikuchi M, Koshiba S, et al. Global Proteomics for Identifying the Alteration Pathway of Niemann–Pick Disease Type C Using Hepatic Cell Models. International Journal of Molecular Sciences. 2023; 24(21):15642. https://doi.org/10.3390/ijms242115642
Chicago/Turabian StyleMiyoshi, Keitaro, Eiji Hishinuma, Naomi Matsukawa, Yoshitaka Shirasago, Masahiro Watanabe, Toshihiro Sato, Yu Sato, Masaki Kumondai, Masafumi Kikuchi, Seizo Koshiba, and et al. 2023. "Global Proteomics for Identifying the Alteration Pathway of Niemann–Pick Disease Type C Using Hepatic Cell Models" International Journal of Molecular Sciences 24, no. 21: 15642. https://doi.org/10.3390/ijms242115642
APA StyleMiyoshi, K., Hishinuma, E., Matsukawa, N., Shirasago, Y., Watanabe, M., Sato, T., Sato, Y., Kumondai, M., Kikuchi, M., Koshiba, S., Fukasawa, M., Maekawa, M., & Mano, N. (2023). Global Proteomics for Identifying the Alteration Pathway of Niemann–Pick Disease Type C Using Hepatic Cell Models. International Journal of Molecular Sciences, 24(21), 15642. https://doi.org/10.3390/ijms242115642






