Organ-Specificity of Breast Cancer Metastasis
Abstract
1. Introduction
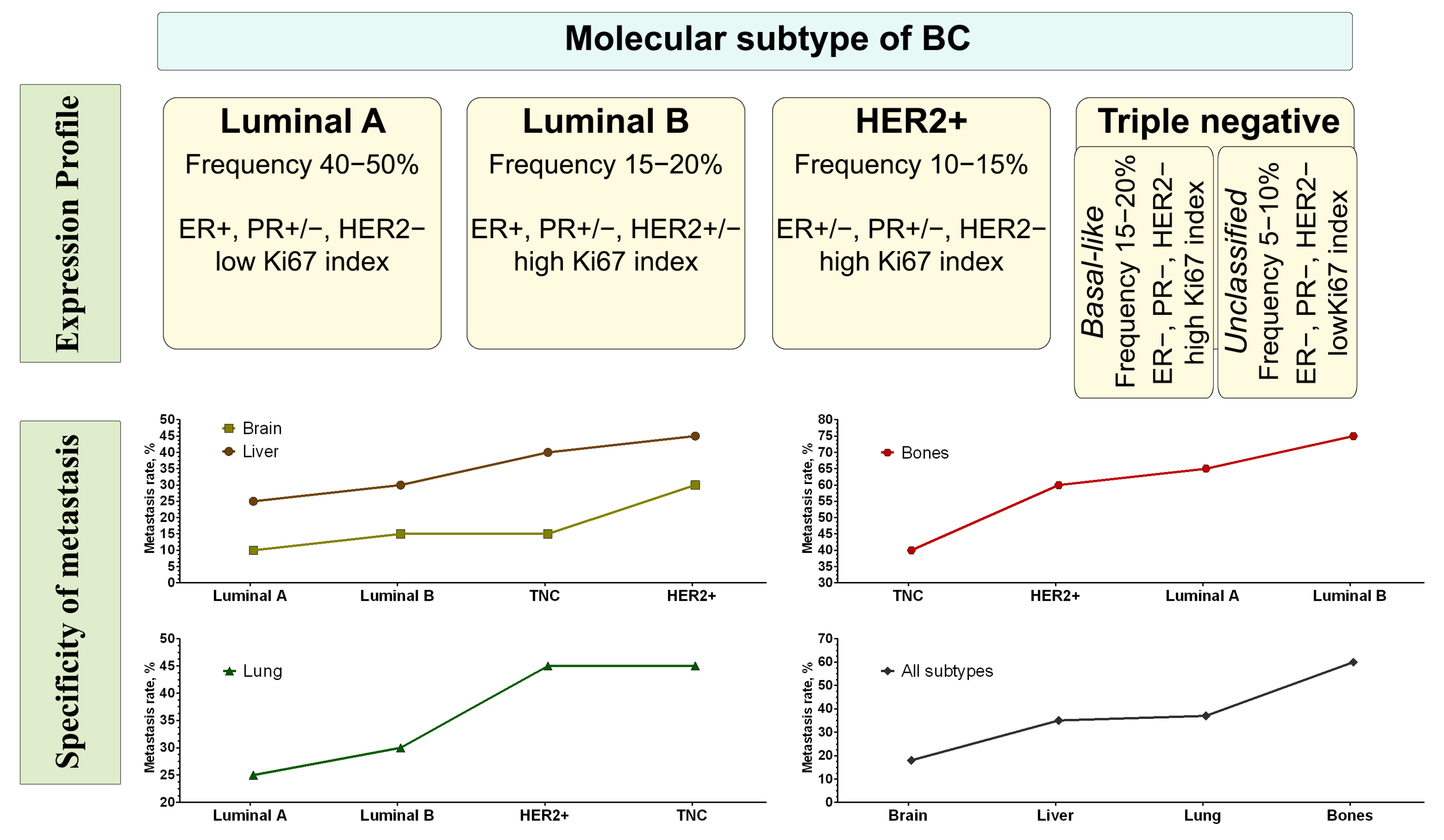
2. Metastatic Breast Cancer Signature
3. Organ-Specific Markers of Breast Cancer Metastasis to Distant Organs
4. Biomarker Profile of Rare Types of Breast Cancer Metastases to Distant Organs
4.1. Gynaecological Metastases
4.2. Metastases to the Pancreas
5. Genomic Profile of Breast Cancer Organ-Specific Metastasis
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Zielonke, N.; Gini, A.; Jansen, E.E.L.; Anttila, A.; Segnan, N.; Ponti, A.; Veerus, P.; de Koning, H.J.; van Ravesteyn, N.T.; Heijnsdijk, E.A.M. Evidence for reducing cancer-specific mortality due to screening for breast cancer in Europe: A systematic review. Eur. J. Cancer 2020, 127, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, C.; Conceição, F.; Jerónimo, C.; Lamghari, M.; Sousa, D.M. Stress in Metastatic Breast Cancer: To the Bone and Beyond. Cancers 2022, 14, 1881. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Li, J.; Zhu, S.; Wu, J.; Chen, C.; Liu, Q.; Wei, W.; Zhang, Y.; Sun, S. Breast cancer subtypes predict the preferential site of distant metastases: A SEER based study. Oncotarget 2017, 8, 27990–27996. [Google Scholar] [CrossRef] [PubMed]
- Buonomo, O.C.; Caredda, E.; Portarena, I.; Vanni, G.; Orlandi, A.; Bagni, C.; Petrella, G.; Palombi, L. New insights into the metastatic behavior after breast cancer surgery, according to well-established clinicopathological variables and molecular subtypes. PLoS ONE 2017, 12, e0184680. [Google Scholar] [CrossRef] [PubMed]
- Almubarak, M.M.; Laé, M.; Cacheux, W.; de Cremoux, P.; Pierga, J.-Y.; Reyal, F.; Bennett, S.P.; Falcou, M.-C.; Salmon, R.J.; Baranger, B.; et al. Gastric metastasis of breast cancer: A single centre retrospective study. Dig. Liver Dis. 2011, 43, 823–827. [Google Scholar] [CrossRef] [PubMed]
- Demirci, U.; Buyukberber, S.; Cakir, T.; Poyraz, A.; Baykara, M.; Karakus, E.; Tufan, G.; Benekli, M.; Coskun, U. Isolated mucinous adrenal metastasis in a breast cancer patient. J. Oncol. Pharm. Pract. 2011, 17, 444–447. [Google Scholar] [CrossRef]
- Bastings, L.; Beerendonk, C.C.; Westphal, J.R.; Massuger, L.F.; Kaal, S.E.; van Leeuwen, F.E.; Braat, D.D.M.; Peek, R. Autotransplantation of cryopreserved ovarian tissue in cancer survivors and the risk of reintroducing malignancy: A systematic review. Hum. Reprod. Update 2013, 19, 483–506. [Google Scholar] [CrossRef]
- Peters, I.T.; van Zwet, E.W.; Smit, V.T.; Liefers, G.J.; Kuppen, P.J.; Hilders, C.G.; Trimbos, J.B. Prevalence and risk factors of ovarian metastases in breast cancer patients < 41 years of age in the Netherlands: A nationwide retrospective cohort study. PLoS ONE 2017, 12, e0168277. [Google Scholar]
- Tian, W.; Zhou, Y.; Wu, M.; Yao, Y.; Deng, Y. Ovarian metastasis from breast cancer: A comprehensive review. Clin. Transl. Oncol. 2019, 21, 819–827. [Google Scholar] [CrossRef]
- Yadav, B.S.; Sharma, S.C.; Robin, T.P.; Sams, S.; Elias, A.D.; Kaklamani, V.; Marcom, P.K.; Schaefer, S.; Morris, G.J. Synchronous primary carcinoma of breast and ovary versus ovarian metastases. Semin. Oncol. 2015, 42, e13–e24. [Google Scholar] [CrossRef]
- Tamas, J.; Vereczkey, I.; Toth, E. Metastatic tumors in the ovary, difficulties of histologic diagnosis. Magyar Onkol. 2015, 59, 205–213. [Google Scholar]
- Xiong, Z.; Deng, G.; Huang, X.; Li, X.; Xie, X.; Wang, J.; Shuang, Z.; Wang, X. Bone metastasis pattern in initial metastatic breast cancer: A population-based study. Cancer Manag. Res. 2018, 10, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Caswell-Jin, J.L.; Plevritis, S.K.; Tian, L.; Cadham, C.J.; Xu, C.; Stout, N.K.; Sledge, G.W.; Mandelblatt, J.S.; Kurian, A.W. Change in Survival in Metastatic Breast Cancer with Treatment Advances: Meta-Analysis and Systematic Review. JNCI Cancer Spectr. 2018, 2, pky062. [Google Scholar] [CrossRef] [PubMed]
- Bale, R.; Putzer, D.; Schullian, P. Local Treatment of Breast Cancer Liver Metastasis. Cancers 2019, 11, 1341. [Google Scholar] [CrossRef] [PubMed]
- Sal, V.; Demirkiran, F.; Topuz, S.; Kahramanoglu, I.; Yalcin, I.; Bese, T.; Sozen, H.; Tokgozoglu, N.; Salihoglu, Y.; Turan, H.; et al. Surgical treatment of metastatic ovarian tumors from extragenital primary sites. Int. J. Gynecol. Cancer 2016, 26, 688–696. [Google Scholar] [CrossRef] [PubMed]
- Rabban, J.T.; Barnes, M.; Chen, L.-M.; Powell, C.B.; Crawford, B.; Zaloudek, C.J. Ovarian pathology in risk-reducing salpingo-oophorectomies from women with BRCA mutations, emphasizing the differential diagnosis of occult primary and metastatic carcinoma. Am. J. Surg. Pathol. 2009, 33, 1125–1136. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhu, Y.; Liu, X.; Liao, X.; He, J.; Niu, L. The Clinicopathological features and survival outcomes of patients with different metastatic sites in stage IV breast cancer. BMC Cancer 2019, 19, 1091. [Google Scholar] [CrossRef]
- Harbeck, N.; Penault-Llorca, F.; Cortes, J.; Gnant, M.; Houssami, N.; Poortmans, P.; Ruddy, K.; Tsang, J.; Cardoso, F. Breast cancer. Nat. Rev. Dis. Primers 2019, 5, 66. [Google Scholar] [CrossRef]
- Swain, S.M.; Miles, D.; Kim, S.-B.; Im, Y.-H.; Im, S.-A.; Semiglazov, V.; Ciruelos, E.; Schneeweiss, A.; Loi, S.; Monturus, E.; et al. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA): End-of-study results from a double-blind, randomised, placebo-controlled, phase 3 study. Lancet Oncol. 2020, 21, 519–530. [Google Scholar] [CrossRef]
- Sledge, G.W., Jr.; Toi, M.; Neven, P.; Sohn, J.; Inoue, K.; Pivot, X.; Burdaeva, O.; Okera, M.; Masuda, N.; Kaufman, P.A.; et al. The effect of abemaciclib plus fulvestrant on overall survival in hormone receptor-positive, ERBB2-negative breast cancer that progressed on endocrine therapy—MONARCH 2: A randomized clinical trial. JAMA Oncol. 2019, 6, 116–124. [Google Scholar] [CrossRef] [PubMed]
- O’Leary, B.; Cutts, R.J.; Liu, Y.; Hrebien, S.; Huang, X.; Fenwick, K.; André, F.; Loibl, S.; Loi, S.; Garcia-Murillas, I.; et al. The genetic landscape and clonal evolution of breast cancer resistance to palbociclib plus fulvestrant in the PALOMA-3 trial. Cancer Discov. 2018, 8, 1390–1403. [Google Scholar] [CrossRef] [PubMed]
- Lambert, A.W.; Pattabiraman, D.R.; Weinberg, R.A. Emerging biological principles of metastasis. Cell 2017, 168, 670–691. [Google Scholar] [CrossRef] [PubMed]
- Tabassum, D.P.; Polyak, K. Tumorigenesis: It takes a village. Nat. Rev. Cancer 2015, 15, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.G.; Vignjevic, D.M. Modes of cancer cell invasion and the role of the microenvironment. Curr. Opin. Cell Biol. 2015, 36, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Cheung, K.J.; Ewald, A.J. A collective route to metastasis: Seeding by tumor cell clusters. Science 2016, 352, 167–169. [Google Scholar] [CrossRef]
- Lin, D.; Shen, L.; Luo, M.; Zhang, K.; Li, J.; Yang, Q.; Zhu, F.; Zhou, D.; Zheng, S.; Chen, Y.; et al. Circulating tumor cells: Biology and clinical significance. Signal Transduct. Target. Ther. 2021, 6, 404. [Google Scholar] [CrossRef]
- Lawson, D.A.; Kessenbrock, K.; Davis, R.T.; Pervolarakis, N.; Werb, Z. Tumour heterogeneity and metastasis at single-cell resolution. Nat. Cell Biol. 2018, 20, 1349–1360. [Google Scholar] [CrossRef]
- Kimbung, S.; Loman, N.; Hedenfalk, I. Clinical and molecular complexity of breast cancer metastases. Semin. Cancer Biol. 2015, 35, 85–95. [Google Scholar] [CrossRef]
- Birkbak, N.J.; McGranahan, N. Cancer genome evolutionary trajectories in metastasis. Cancer Cell 2020, 37, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Denny, S.K.; Greenside, P.G.; Chaikovsky, A.C.; Brady, J.J.; Ouadah, Y.; Granja, J.M.; Jahchan, N.S.; Lim, J.S.; Kwok, S.; et al. Intertumoral heterogeneity in SCLC is influenced by the cell type of origin. Cancer Discov. 2018, 8, 1316–1331. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, K.; Basnet, H.; Kaygusuz, Y.; Laughney, A.M.; He, L.; Sharma, R.; O’Rourke, K.P.; Reuter, V.P.; Huang, Y.-H.; Turkekul, M.; et al. L1CAM defines the regenerative origin of metastasis-initiating cells in colorectal cancer. Nat. Cancer 2020, 1, 28–45. [Google Scholar] [CrossRef] [PubMed]
- The Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012, 490, 61–70. [Google Scholar] [CrossRef] [PubMed]
- International Cancer Genome Consortium. International network of cancer genome projects. Nature 2010, 464, 993–998. [Google Scholar] [CrossRef] [PubMed]
- Bertucci, F.; Ng, C.K.Y.; Patsouris, A.; Droin, N.; Piscuoglio, S.; Carbuccia, N.; Soria, J.C.; Dien, A.T.; Adnani, Y.; Kamal, M.; et al. Genomic characterization of metastatic breast cancers. Nature 2019, 569, 560–564. [Google Scholar] [CrossRef]
- Brown, D.; Smeets, D.; Székely, B.; Larsimont, D.; Szász, A.M.; Adnet, P.-Y.; Rothé, F.; Rouas, G.; Nagy, Z.I.; Faragó, Z.; et al. Phylogenetic analysis of metastatic progression in breast cancer using somatic mutations and copy number aberrations. Nat. Commun. 2017, 8, 14944. [Google Scholar] [CrossRef]
- Yates, L.R.; Knappskog, S.; Wedge, D.; Farmery, J.H.R.; Gonzalez, S.; Martincorena, I.; Alexandrov, L.B.; Van Loo, P.; Haugland, H.K.; Lilleng, P.K.; et al. Genomic evolution of breast cancer metastasis and relapse. Cancer Cell 2017, 32, 169–184. [Google Scholar] [CrossRef]
- De Mattos-Arruda, L.; Sammut, S.J.; Ross, E.M.; Bashford-Rogers, R.; Greenstein, E.; Markus, H.; Morganella, S.; Teng, Y.; Maruvka, Y.; Pereira, B.; et al. The genomic and immune landscapes of lethal metastatic breast cancer. Cell Rep. 2019, 27, 2690–2708. [Google Scholar] [CrossRef]
- Tahara, R.K.; Brewer, T.M.; Theriault, R.L.; Ueno, N.T. Bone Metastasis of Breast Cancer. In Breast Cancer Metastasis and Drug Resistance; Ahmad, A., Ed.; Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2019; Volume 1152. [Google Scholar]
- Leto, G. Current status and future directions in the treatment of bone metastases from breast cancer. Clin. Exp. Pharmacol. Physiol. 2019, 46, 968–971. [Google Scholar] [CrossRef]
- Medeiros, B.; Allan, A.L. Molecular Mechanisms of Breast Cancer Metastasis to the Lung: Clinical and Experimental Perspectives. Int. J. Mol. Sci. 2019, 20, 2272. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Ye, F.; Kong, Y.; Hu, X.; Deng, X.; Xie, J.; Song, C.; Ou, X.; Wu, S.; Wu, L.; et al. The Single-Cell Landscape of Intratumoral Heterogeneity and The Immunosuppressive Microenvironment in Liver and Brain Metastases of Breast Cancer. Adv. Sci. 2023, 10, 2203699. [Google Scholar] [CrossRef] [PubMed]
- Horn, S.R.; Stoltzfus, K.C.; Lehrer, E.J.; Dawson, L.A.; Tchelebi, L.; Gusani, N.J.; Sharma, N.K.; Chen, H.; Trifiletti, D.M.; Zaorsky, N.G.; et al. Epidemiology of liver metastases. Cancer Epidemiol. 2020, 67, 101760. [Google Scholar] [CrossRef] [PubMed]
- Tsilimigras, D.I.; Brodt, P.; Clavien, P.A.; Muschel, R.J.; D’Angelica, M.I.; Endo, I.; Parks, R.W.; Doyle, M.; de Santibañes, E.; Pawlik, T.M. Liver metastases. Nat. Rev. Dis. Primers 2021, 7, 27. [Google Scholar] [CrossRef] [PubMed]
- Hosonaga, M.; Saya, H.; Arima, Y. Molecular and cellular mechanisms underlying brain metastasis of breast cancer. Cancer Metastasis Rev. 2020, 39, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Corti, C.; Antonarelli, G.; Criscitiello, C.; Lin, N.U.; Carey, L.A.; Cortés, J.; Poortmans, P.; Curigliano, G. Targeting brain metastases in breast cancer. Cancer Treat. Rev. 2022, 103, 102324. [Google Scholar] [CrossRef] [PubMed]
- Lim, A.R.; Ghajar, C.M. Thorny ground, rocky soil: Tissue-specific mechanisms of tumor dormancy and relapse. Semin. Cancer Biol. 2022, 78, 104–123. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.; Rathbone, E.; Hinsley, S.; Gregory, W.; Gossiel, F.; Marshall, H.; Burkinshaw, R.; Shulver, H.; Thandar, H.; Bertelli, G.; et al. Associations between serum bone biomarkers in early breast cancer and development of bone metastasis: Results from the AZURE (BIG01/04) trial. J. Natl. Cancer Inst. 2018, 110, 871–879. [Google Scholar] [CrossRef]
- Holen, I.; Lefley, D.V.; Francis, S.E.; Rennicks, S.; Bradbury, S.; Coleman, R.E.; Ottewell, P. IL-1 drives breast cancer growth and bone metastasis in vivo. Oncotarget 2016, 7, 75571–75584. [Google Scholar] [CrossRef]
- Westbrook, J.A.; Cairns, D.A.; Peng, J.; Speirs, V.; Hanby, A.M.; Holen, I.; Wood, S.L.; Ottewell, P.D.; Marshall, H.; Banks, R.E.; et al. CAPG and GIPC1: Breast cancer biomarkers for bone metastasis development and treatment. J. Natl. Cancer Inst. 2016, 108, djv360. [Google Scholar] [CrossRef]
- Sutherland, A.; Forsyth, A.; Cong, Y.; Grant, L.; Juan, T.-H.; Lee, J.K.; Klimowicz, A.; Petrillo, S.K.; Hu, J.; Chan, A.; et al. The role of prolactin in bone metastasis and breast cancer cell-mediated osteoclast differentiation. J. Natl. Cancer Inst. 2016, 108, djv338. [Google Scholar] [CrossRef] [PubMed]
- Rafiei, S.; Tiedemann, K.; Tabaries, S.; Siegel, P.M.; Komarova, S.V. Peroxiredoxin 4: A novel secreted mediator of cancer induced osteoclastogenesis. Cancer Lett. 2015, 361, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, H.; Zhao, Y.; Wang, C.; Cheng, Z.; Tang, L.; Gao, Y.; Liu, F.; Li, J.; Li, Y.; et al. A mandatory role of nuclear PAK4–LIFR axis in breast-to-bone metastasis of ERα-positive breast cancer cells. Oncogene 2019, 38, 808–821. [Google Scholar] [CrossRef] [PubMed]
- Pavlovic, M.; Arnal-Estape, A.; Rojo, F.; Bellmunt, A.; Tarragona, M.; Guiu, M.; Planet, E.; Garcia-Albéniz, X.; Morales, M.; Urosevic, J.; et al. Enhanced MAF oncogene expression and breast cancer bone metastasis. J. Natl. Cancer Inst. 2015, 107, djv256. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.E.; Collinson, M.; Gregory, W.; Marshall, H.; Bell, R.; Dodwell, D.; Keane, M.; Gil, M.; Barrett-Lee, P.; Ritchie, D.; et al. Benefits and risks of adjuvant treatment with zoledronic acid in stage II/III breast cancer. 10 years follow-up of the AZURE randomized clinical trial (BIG 01/04). J. Bone Oncol. 2018, 13, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Westbrook, J.A.; Wood, S.L.; Cairns, D.A.; McMahon, K.; Gahlaut, R.; Thygesen, H.; Shires, M.; Roberts, S.; Marshall, H.; Oliva, M.R.; et al. Identification and validation of DOCK4 as a potential biomarker for risk of bone metastasis development in patients with early breast cancer. J. Pathol. 2019, 247, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Huang, J.; Lan, J.; Zhou, K.; Gao, Y.; Song, Z.; Deng, Y.; Liu, L.; Dong, Y.; Liu, X. Overexpression of CENPF correlates with poor prognosis and tumor bone metastasis in breast cancer. Cancer Cell Int. 2019, 19, 264. [Google Scholar] [CrossRef]
- Zhang, Y.; He, W.; Zhang, S. Seeking for Correlative Genes and Signaling Pathways with Bone Metastasis from Breast Cancer by Integrated Analysis. Front. Oncol. 2019, 9, 138. [Google Scholar] [CrossRef]
- Bahrami, A.; Aledavood, A.; Anvari, K.; Hassanian, S.M.; Maftouh, M.; Yaghobzade, A.; Salarzaee, O.; ShahidSales, S.; Avan, A. The prognostic and thera-peutic application of microRNAs in breast cancer: Tissue and circu-lating microRNAs. J. Cell Physiol. 2018, 233, 774–786. [Google Scholar] [CrossRef]
- Fan, X.; Chen, W.; Fu, Z.; Zeng, L.; Yin, Y.; Yuan, H. MicroRNAs, a sub-population of regulators, are involved in breast cancer progres-sion through regulating breast cancer stem cells. Oncol. Lett. 2017, 14, 5069–5076. [Google Scholar]
- Yuan, X.; Qian, N.; Ling, S.; Li, Y.; Sun, W.; Li, J.; Du, R.; Zhong, G.; Liu, C.; Yu, G.; et al. Breast cancer exosomes contribute to pre-metastatic niche formation and promote bone metastasis of tumor cells. Theranostics 2021, 11, 1429–1445. [Google Scholar] [CrossRef] [PubMed]
- Romero-Moreno, R.; Curtis, K.J.; Coughlin, T.R.; Miranda-Vergara, M.C.; Dutta, S.; Natarajan, A.; Facchine, B.A.; Jackson, K.M.; Nystrom, L.; Li, J.; et al. The CXCL5/CXCR2 axis is sufficient to promote breast cancer colonization during bone metastasis. Nat. Commun. 2019, 10, 4404. [Google Scholar] [CrossRef]
- Sousa, S.; Gineyts, E.; Geraci, S.; Croset, M.; Clézardin, P. RANK-RANKL signaling inhibition delays early breast cancer bone metastasis formation. AACR Cancer Res. 2018, 78, 29. [Google Scholar] [CrossRef]
- Devignes, C.S.; Aslan, Y.; Brenot, A.; Devillers, A.; Schepers, K.; Fabre, S.; Chou, J.; Casbon, A.-J.; Werb, Z.; Provot, S. HIF signaling in osteoblast-lineage cells promotes systemic breast cancer growth and metastasis in mice. Proc. Natl. Acad. Sci. USA 2018, 115, E992–E1001. [Google Scholar] [CrossRef] [PubMed]
- Bartels, S.; Christgen, M.; Luft, A.; Persing, S.; Jödecke, K.; Lehmann, U.; Kreipe, H. Estrogen receptor (ESR1) mutation in bone metastases from breast cancer. Mod. Pathol. 2018, 31, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Endo, M.; Yamamoto, Y.; Odagiri, H.; Kadomatsu, T.; Nakamura, T.; Tanoue, H.; Ito, H.; Yugami, M.; Miyata, K.; et al. ANGPTL2 increases bone metastasis of breast cancer cells through enhancing CXCR4 signaling. Sci. Rep. 2015, 5, 9170. [Google Scholar] [CrossRef] [PubMed]
- Schrijver, W.A.M.E.; van Diest, P.J.; Dutch Distant Breast Cancer Metastases Consortium; Moelans, C.B. Unravelling site-specific breast cancer metastasis: A microRNA expression profiling study. Oncotarget 2017, 8, 3111–3123. [Google Scholar] [CrossRef]
- Wang, Z.; Li, T.-E.; Chen, M.; Pan, J.-J.; Shen, K.-W. MiR-106b-5p contributes to the lung metastasis of breast cancer via targeting CNN1 and regulating Rho/ROCK1 pathway. Aging 2020, 12, 1867–1887. [Google Scholar] [CrossRef]
- Tang, X.; Shi, L.; Xie, N.; Liu, Z.; Qian, M.; Meng, F.; Xu, Q.; Zhou, M.; Cao, X.; Zhu, W.-G.; et al. SIRT7 antagonizes TGF-beta signaling and inhibits breast cancer metastasis. Nat. Commun. 2017, 8, 318. [Google Scholar] [CrossRef]
- Pascual, G.; Avgustinova, A.; Mejetta, S.; Martín, M.; Castellanos, A.; Attolini, C.S.O.; Berenguer, A.; Prats, N.; Toll, A.; Hueto, J.A.; et al. Targeting metastasis-initiating cells through the fatty acid receptor CD36. Nature 2017, 541, 41–45. [Google Scholar] [CrossRef]
- Aleckovic, M.; Wei, Y.; LeRoy, G.; Sidoli, S.; Liu, D.D.; Garcia, B.A.; Kang, Y. Identification of Nidogen 1 as a lung metastasis protein through secretome analysis. Genes Dev. 2017, 31, 1439–1455. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Adorno-Cruz, V.; Chang, Y.-F.; Jia, Y.; Kawaguchi, M.; Dashzeveg, N.K.; Taftaf, R.; Ramos, E.K.; Schuster, E.J.; El-Shennawy, L.; et al. EGFR inhibition blocks cancer stem cell clustering and lung metastasis of triple negative breast cancer. Theranostics 2021, 11, 6632–6643. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Zhang, Z.; Zhao, S.; He, X.; Yu, H.; Yin, Q.; Zhang, Z.; Gu, W.; Chen, L.; Li, Y. Hydrophobic interaction mediating self-assembled nanoparticles of succinobucol suppress lung metastasis of breast cancer by inhibition of VCAM-1 expression. J. Control. Release 2015, 205, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, X.; Zhang, H.; Li, X.; Li, X.; Cong, M.; Peng, F.; Yu, J.; Zhang, X.; Yang, Q.; Hu, G. Differential effects on lung and bone metastasis of breast cancer by Wnt signalling inhibitor DKK1. Nat. Cell Biol. 2017, 19, 1274–1285. [Google Scholar] [CrossRef] [PubMed]
- Kazan, J.M.; El-Saghir, J.; Saliba, J.; Shaito, A.; Jalaleddine, N.; El-Hajjar, L.; Al-Ghadban, S.; Yehia, L.; Zibara, K.; El-Sabban, M. Cx43 Expression Correlates with Breast Cancer Metastasis in MDA-MB-231 Cells In Vitro, In a Mouse Xenograft Model and in Human Breast Cancer Tissues. Cancers 2019, 11, 460. [Google Scholar] [CrossRef]
- Chen, I.X.; Chauhan, V.P.; Posada, J.; Ng, M.R.; Wu, M.W.; Adstamongkonkul, P.; Huang, P.; Lindeman, N.; Langer, R.; Jain, R.K. Blocking CXCR4 alleviates desmoplasia, increases T-lymphocyte infiltration, and improves immunotherapy in metastatic breast cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 4558–4566. [Google Scholar] [CrossRef] [PubMed]
- Dupuy, F.; Tabariès, S.; Andrzejewski, S.; Dong, Z.; Blagih, J.; Annis, M.G.; Omeroglu, A.; Gao, D.; Leung, S.; Amir, E.; et al. PDK1-dependent metabolic reprogramming dictates metastatic potential in breast cancer. Cell Metab. 2015, 22, 577–589. [Google Scholar] [CrossRef]
- Liu, P.; Wang, Z.; Ou, X.; Wu, P.; Zhang, Y.; Wu, S.; Xiao, X.; Li, Y.; Ye, F.; Tang, H. The FUS/circEZH2/KLF5/feedback loop contributes to CXCR4-induced liver metastasis of breast cancer by enhancing epithelial-mesenchymal transition. Mol. Cancer 2022, 21, 198. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, L.; Wu, P.; Li, X.; Tang, Y.; Ou, X.; Zhang, Y.; Xiao, X.; Wang, J.; Tang, H. The circROBO1/KLF5/FUS feedback loop regulates the liver metastasis of breast cancer by inhibiting the selective autophagy of afadin. Mol. Cancer 2022, 21, 29. [Google Scholar] [CrossRef]
- Tabariès, S.; Annis, M.G.; Hsu, B.E.; Tam, C.E.; Savage, P.; Park, M.; Siegel, P.M. Lyn modulates Claudin-2 expression and is a therapeutic target for breast cancer liver metastasis. Oncotarget 2015, 6, 9476–9487. [Google Scholar] [CrossRef]
- Yang, J.; Wu, N.N.; Huang, D.J.; Luo, Y.C.; Huang, J.Z.; He, H.Y.; Lu, H.L.; Song, W.L. PPFIA1 is upregulated in liver metastasis of breast cancer and is a potential poor prognostic indicator of metastatic relapse. Tumour Biol. 2017, 39, 1010428317713492. [Google Scholar] [CrossRef] [PubMed]
- Tian, C.; Liu, S.; Wang, Y.; Song, X. Prognosis and Genomic Landscape of Liver Metastasis in Patients with Breast Cancer. Front. Oncol. 2021, 11, 588136. [Google Scholar] [CrossRef] [PubMed]
- Blazquez, R.; Wlochowitz, D.; Wolff, A.; Seitz, S.; Wachter, A.; Perera-Bel, J.; Bleckmann, A.; Beißbarth, T.; Salinas, G.; Riemenschneider, M.J.; et al. PI3K: A master regulator of brain metastasis-promoting macrophages/microglia. Glia 2018, 66, 2438–2455. [Google Scholar] [CrossRef]
- Hohensee, I.; Chuang, H.N.; Grottke, A.; Werner, S.; Schulte, A.; Horn, S.; Lamszus, K.; Bartkowiak, K.; Witzel, I.; Westphal, M.; et al. PTEN mediates the cross talk between breast and glial cells in brain metastases leading to rapid disease progression. Oncotarget 2017, 8, 6155–6168. [Google Scholar] [CrossRef] [PubMed]
- Anders, C.K.; Rhun, E.L.; Bachelot, T.D.; Yardley, D.A.; Awada, A.; Conte, P.F.; Conte, P.F.; Kabos, P.; Bear, M.; Yang, Z.; et al. A phase II study of abemaciclib in patients (pts) with brain metastases (BM) secondary to HR+, HER2- metastatic breast cancer (MBC). J. Clin. Oncol. 2019, 37, 1017. [Google Scholar] [CrossRef]
- Priego, N.; Zhu, L.; Monteiro, C.; Mulders, M.; Wasilewski, D.; Bindeman, W.; Doglio, L.; Martínez, L.; Martínez-Saez, E.; Ramón y Cajal, S.; et al. STAT3 labels a subpopulation of reactive astrocytes required for brain metastasis. Nat. Med. 2018, 24, 1024–1035. [Google Scholar] [CrossRef] [PubMed]
- Ghoochani, A.; Schwarz, M.A.; Yakubov, E.; Engelhorn, T.; Doerfler, A.; Buchfelder, M.; Bucala, R.; Savaskan, N.E.; Eyüpoglu, I.Y. MIF-CD74 signaling impedes microglial M1 polarization and facilitates brain tumorigenesis. Oncogene 2016, 35, 6246–6261. [Google Scholar] [CrossRef]
- Wang, S.; Liang, K.; Hu, Q.; Li, P.; Song, J.; Yang, Y.; Yao, J.; Mangala, L.S.; Li, C.; Yang, W.; et al. JAK2-binding long noncoding RNA promotes breast cancer brain metastasis. J. Clin. Investig. 2017, 127, 4498–4515. [Google Scholar] [CrossRef]
- Wu, K.; Fukuda, K.; Xing, F.; Zhang, Y.; Sharma, S.; Liu, Y.; Chan, M.D.; Zhou, X.; Qasem, S.A.; Pochampally, R.; et al. Roles of the cyclooxygenase 2 matrix metalloproteinase 1 pathway in brain metastasis of breast cancer. J. Biol. Chem. 2015, 290, 9842–9854. [Google Scholar] [CrossRef]
- Cordero, A.; Kanojia, D.; Miska, J.; Panek, W.K.; Xiao, A.; Han, Y.; Bonamici, N.; Zhou, W.; Xiao, T.; Wu, M.; et al. FABP7 is a key metabolic regulator in HER2+ breast cancer brain metastasis. Oncogene 2019, 38, 6445–6460. [Google Scholar] [CrossRef]
- Sato, J.; Shimomura, A.; Kawauchi, J.; Matsuzaki, J.; Yamamoto, Y.; Takizawa, S.; Sakamoto, H.; Ohno, M.; Narita, Y.; Ochiya, T.; et al. Brain metastasis-related microRNAs in patients with advanced breast cancer. PLoS ONE 2019, 14, e0221538. [Google Scholar] [CrossRef]
- Figueira, I.; Galego, S.; Custódio-Santos, T.; Vicente, R.; Molnár, K.; Haskó, J.; Malhó, R.; Videira, M.; Wilhelm, I.; Krizbai, I.; et al. Picturing Breast Cancer Brain Metastasis Development to Unravel Molecular Players and Cellular Crosstalk. Cancers 2021, 13, 910. [Google Scholar] [CrossRef] [PubMed]
- Kutasovic, J.R.; McCart Reed, A.E.; Males, R.; Sim, S.; Saunus, J.M.; Dalley, A.; McEvoy, C.R.; Dedina, L.; Miller, G.; Peyton, S.; et al. Breast cancer metastasis to gynaecological organs: A clinico-pathological and molecular profiling study. J. Pathol. Clin. Res. 2019, 5, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Shee, K.; Strait, A.M.; Liu, X. Biomarkers to diagnose metastatic breast carcinoma to the pancreas: A case report and update. Diagn. Cytopathol. 2019, 47, 912–917. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, G.S.; Goswami, S.; Jones, J.G.; Oktay, M.H.; Condeelis, J.S. Signatures of breast cancer metastasis at a glance. J. Cell Sci. 2016, 129, 1751–1758. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.; Barraclough, R.; Vasieva, O. Gene Expression Meta-Analysis of Potential Metastatic Breast Cancer Markers. Curr. Mol. Med. 2017, 17, 200–210. [Google Scholar] [CrossRef][Green Version]
- Schulten, H.J.; Bangash, M.; Karim, S.; Dallol, A.; Hussein, D.; Merdad, A.; Al-Thoubaity, F.K.; Al-Maghrabi, J.; Jamal, A.; Al-Ghamdi, F.; et al. Comprehensive molecular biomarker identification in breast cancer brain metastases. J. Transl. Med. 2017, 15, 269. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, J.; Liu, E.; Kinnebrew, G.; Zhang, X.; Stover, D.; Huo, Y.; Zeng, Z.; Jiang, W.; Cheng, L.; et al. Identification of Alternatively-Activated Pathways between Primary Breast Cancer and Liver Metastatic Cancer Using Microarray Data. Genes 2019, 10, 753. [Google Scholar] [CrossRef] [PubMed]
- Paul, M.R.; Pan, T.-C.; Pant, D.K.; Shih, N.N.; Chen, Y.; Harvey, K.L.; Solomon, A.; Lieberman, D.; Morrissette, J.J.; Soucier-Ernst, D. Genomic landscape of metastatic breast cancer identifies preferentially dysregulated pathways and targets. J. Clin. Investig. 2020, 130, 4252–4265. [Google Scholar] [CrossRef]
- Aftimos, P.; Oliveira, M.; Irrthum, A.; Fumagalli, D.; Sotiriou, C.; Gal-Yam, E.N.; Robson, M.E.; Ndozeng, J.; Di Leo, A.; Ciruelos, E.M.; et al. Genomic and Transcriptomic Analyses of Breast Cancer Primaries and Matched Metastases in AURORA, the Breast International Group (BIG) Molecular Screening Initiative. Cancer Discov. 2021, 11, 2796–2811. [Google Scholar] [CrossRef]
- Spadazzi, C.; Mercatali, L.; Esposito, M.; Wei, Y.; Liverani, C.; De Vita, A.; Miserocchi, G.; Carretta, E.; Zanoni, M.; Cocchi, C.; et al. Trefoil factor-1 upregulation in estrogen-receptor positive breast cancer correlates with an increased risk of bone metastasis. Bone 2021, 144, 115775. [Google Scholar] [CrossRef]
- Brasó-Maristany, F.; Paré, L.; Chic, N.; Martínez-Sáez, O.; Pascual, T.; Mallafré-Larrosa, M.; Schettini, F.; González-Farré, B.; Sanfeliu, E.; Martínez, D.; et al. Gene expression profiles of breast cancer metastasis according to organ site. Mol. Oncol. 2021, 16, 69–87. [Google Scholar] [CrossRef]
- Lawler, K.; Papouli, E.; Naceur-Lombardelli, C.; Mera, A.; Ougham, K.; Tutt, A.; Kimbung, S.; Hedenfalk, I.; Zhan, J.; Zhang, H.; et al. Gene expression modules in primary breast cancers as risk factors for organotropic patterns of first metastatic spread: A case control study. Breast Cancer Res. 2017, 19, 113. [Google Scholar] [CrossRef]
- Cioce, M.; Sacconi, A.; Donzelli, S.; Bonomo, C.; Perracchio, L.; Carosi, M.; Telera, S.; Fazio, V.M.; Botti, C.; Strano, S.; et al. Breast cancer metastasis: Is it a matter of OMICS and proper ex-vivo models? Comput. Struct. Biotechnol. J. 2022, 20, 4003–4008. [Google Scholar] [CrossRef]
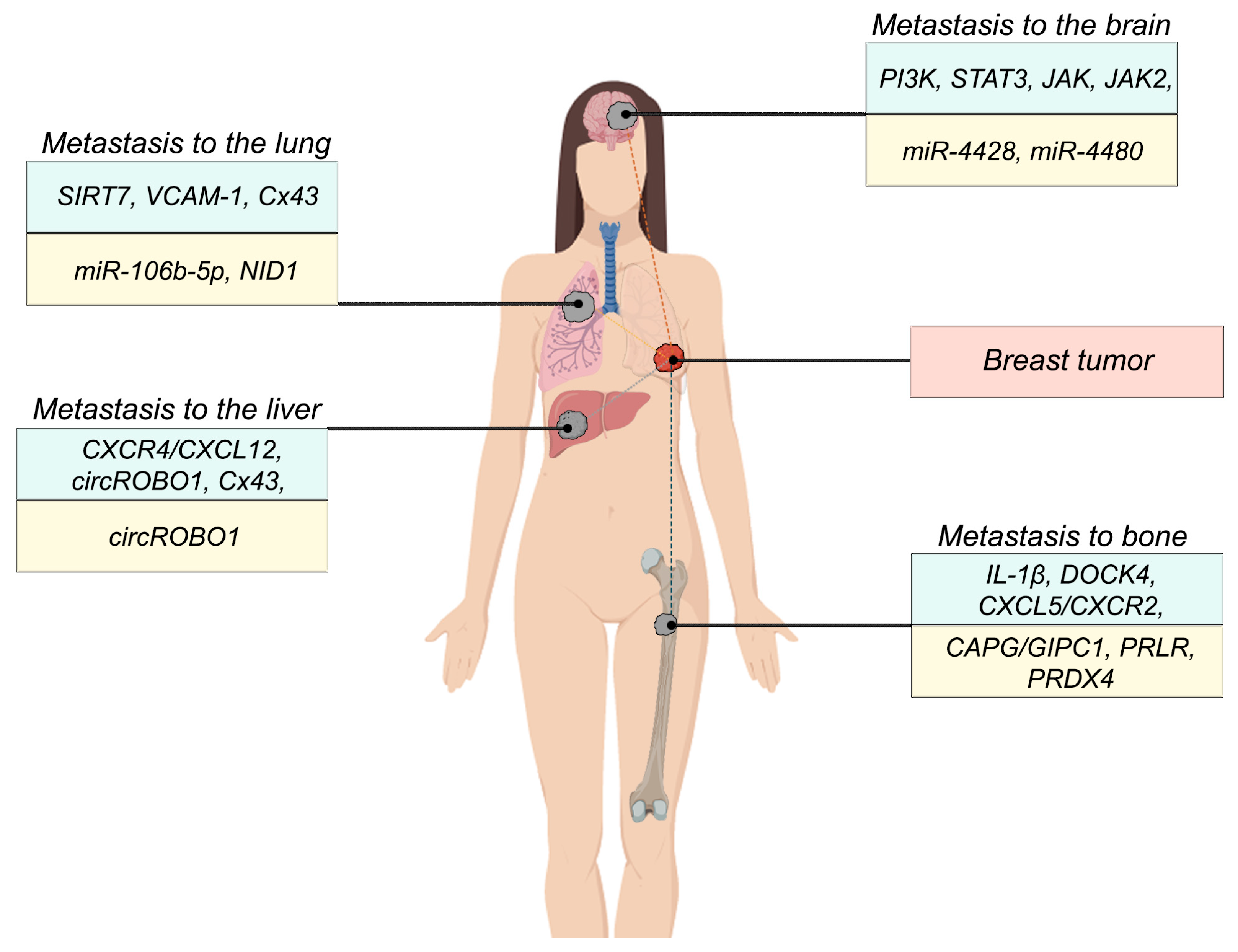
| Marker | Description | Source |
|---|---|---|
| BONES | ||
| P1NP, CTX, 1-CTP | Patients with high serum levels of P1NP, CTX and 1-CTP have been shown to have a high risk of metastasising to bone soon after diagnosis (p = 0.006, p = 0.009, p = 0.008, respectively). | [49] |
| IL-1β | In preclinical experimental mouse models, IL-1β inhibitors have been shown to prevent the development of bone metastases. | [50] |
| CAPG/GIPC1 | The identification of CAPG and GIPC1 in primary tumour samples (by IHC) was a strong prognostic indicator for the development of bone metastases of breast cancer. Cox regression analysis showed that control patients were more likely to develop first distant recurrence in bone (hazard ratio [HR] = 4.5, 95% confidence interval [CI] = 2.1 to 9.8, p < 0.001) and die (HR for overall survival = 1.8, 95% CI = 1.01 to 3.24, p = 0.045) if both proteins were highly expressed in the primary tumour. | [51] |
| PRLR | High PRLR expression in primary breast tumour is associated with shorter time to metastasis (p = 0.03). | [52] |
| PRDX4 | High expression of PRDX4 in primary breast tumour is associated with metastasis within 5 years. | [53] |
| PAK4 | PAK4 enhances the invasive potential of ERα-positive breast cancer cells in vitro and promotes metastasis in vivo. The status of the nuclear PAK4 (nPAK4) scores was significantly higher in the bone metastatic breast cancer group than in the non-bone metastatic breast cancer group (p = 2.22 × 10−9). | [54] |
| MAF | MAF is a molecular target for the prevention or treatment of bone metastases because MAF accumulation (16q23 amplification) plays a role in bone colonisation. 16q23 gain copy number alterations (CNA) encoding the transcription factor MAF mediate breast cancer bone metastasis through PTHrP control. 16q23 gain (hazard ratio (HR) for bone metastasis = 14.5, 95% confidence interval (CI) = 6.4 to 32.9, p < 0.001) as well as MAF overexpression (HR for bone metastasis = 2.5, 95% CI = 1.7 to 3.8, p < 0.001) in primary breast tumours were specifically associated with risk of metastasis to bone but not to other organs. | [55,56] |
| DOCK4 | In a triple-negative MDA-MB-231 cell line model, DOCK4 was identified as a biomarker of bone metastasis in early stages of breast cancer. Adjusted Cox regression analyses showed that high DOCK4 expression in the control arm was significantly prognostic for first recurrence in bone (HR 2.13, 95%CI 1.06–4.30, p = 0.034) (a clinical validation). High DOCK4 expression was not associated with metastasis to non-skeletal sites when these were assessed collectively. | [57] |
| CENPF | CENPF promotes breast cancer metastasis to bone by activating PI3K-AKT-mTORC1 signalling and represents a novel therapeutic target for breast cancer treatment. | [58] |
| MMP9, MMP13, TNFAIP6, CD200, DHRS3, ASS1, VIM | Together, they can be considered as specific prognostic markers of metastasis to bone in primary breast cancer. The relative expression of MMP9, MMP13, TNFAIP6 and CD200 were significantly up-regulated (p < 0.05), while DHRS3, ASS1 and VIM were significantly down-regulated in the bone metastasis compared with lung and liver metastasis (p < 0.05). | [59] |
| miR-200, -128, -99a, -29b, -600, -34, -30, let-7 miRNA | These miRs act as tumour suppressors and inhibit breast cancer metastasis to bone. | [60,61] |
| miR-21 | Exosomal miR-21 derived from SCP28 cells promotes osteoclastogenesis through regulation of PDCD4 protein levels. The level of miR-21 is significantly higher in serum exosomes of breast cancer patients with bone metastases than in other subpopulations. | [62] |
| CXCL5/CXCR2 | CXCL5 stimulates proliferation of breast cancer cells and their colonisation in bone. Inhibition of its CXCR2 receptor with an antagonist blocks the proliferation of metastatic cells. CXCL5 and CXCR2 inhibitors may be effective in the treatment of tumours with metastasis to bone. | [63] |
| RANKL/RANK | RANKL/RANK regulates breast cancer cell migration. RANKL acts as a chemoattractive agent on tumour cells which overexpress one of its receptors. Blocking signalling by AMG161 (IgG1) reduces micrometastasis formation in bone marrow in vivo. Daily subcutaneous injections of 1.5 mg/kg AMG161 antibody to MDA-MB231RANK tumour-bearing animals reduced bone micrometastases and early bone marrow colonization without affecting lung micrometastasis. | [64] |
| CXCL-12 | HIF signalling transduction in osteoporosis precursor cells increases blood levels of CXCL-12, promoting metastasis to bone. | [65] |
| ESR1 | Mutations in the ESR1 gene have been observed in bone metastases, suggesting a potential causative role. In this study, bone metastases from breast cancer (n = 231) were analysed for ESR1 mutation. Activating ESR1 mutations were identified in 27 patients (12%). The most frequent mutation was p.D538G (53%), no mutations were found in exon 4 (K303) or 7 (S463). Metastatic breast cancer with activating mutations of ESR1 had a higher Ki67 labelling index than primary luminal cancers (median 30%, ranging from 5 to 60% with 85% of cases revealing ≥ 20% Ki67-positive cells). | [66] |
| ANGPTL2 | ANGPTL2 increases breast cancer cell metastasis to bone by enhancing CXCR4 signal transduction. | [67] |
| LUNGS | ||
| miR-106b-5p | It is an independent predictor of lung metastases (based on the expression level in the primary tumour). MiR-106b-5p promotes lung metastasis by suppressing CNN1 and activating the Rho/ROCK1 pathway. | [68,69] |
| SIRT7 | SIRT7 counteracts TGFβ signalling and inhibits breast cancer metastases to the lung. | [70] |
| Tumour stem cells (TSCs) (CD44hi CD36+) | The formation of lung metastases is associated with TSC function, metabolic changes and immune response. Lung metastasis can be mediated by TSCs with CD44hi CD36+ phenotype. | [71] |
| NID1 | Secretome analysis of lung metastases of breast cancer has shown that Nidogen 1 (NID1) is associated with poor treatment outcomes. NID1 promotes lung metastasis of breast cancer by increasing the motility of tumour cells and promoting their adhesion to the endothelium, thereby compromising its integrity and promoting angiogenesis. | [72] |
| EGFR | EGFR inhibition successfully blocks circulating tumour cells (by immunohistochemistry) clustering and triple-negative breast cancer metastasis to the lung. | [73] |
| VCAM-1 | VCAM-1 can be considered as a potential therapeutic target in lung metastasis of breast cancer. Selective inhibition of VCAM-1 has been successfully used to suppress the development of metastases. The experimental results showed that the SCB-loaded nanoparticles (SN) could greatly improve the oral delivery and suppress breast cancer metastasis to the lung. The cell migration and invasion abilities of metastatic 4T1 breast cancer cells were obviously inhibited by SN. Moreover, the VCAM-1 expression on 4T1 cells was significantly reduced by SN, and the binding ratio of RAW 264.7 cells to 4T1 cells was significantly decreased from 47.4% to 3.2%. Furthermore, the oral bioavailability of SCB was greatly increased 13-fold under the effect of SN, and the biodistribution in major organs was markedly improved. | [74] |
| DKK1 | In patients with breast cancer, low serological levels of DKK1 are associated with the risk of developing lung metastases. | [75] |
| Connexin43 (Cx43) | Mice injected with Cx43-shCx43-inhibited tumour cells exhibited more lung metastases compared to parental MDA-MB-231 cells. This observation was confirmed by qPCR analysis of human 18S RNA levels in secondary metastatic sites in the lungs. Higher levels of human 18S RNA were found in the lungs of mice injected with shCx43 cells compared to the lungs of mice injected with parental MDA-MB-231 cells. This observation indicates that suppression of Cx43 increases the metastatic potential of MDA-MB-231 cells. | [76] |
| LIVER | ||
| Connexin43 (Cx43) | Metastatic foci in the liver were almost absent in mice inoculated with parental MDA-MB-231 cells or Cx43D cells by week 9, compared to those clearly observed in mice inoculated with shCx43 cells. This result is consistent with the increased levels of human 18S RNA in the livers of mice inoculated with shCx43 cells. Inhibition of Cx43 induced metastasis of MDA-MB-231 cells to lung and liver at week 9, when the original MDA-MB-231 cells had not yet metastasised. These findings correlate with increased tumour volume and decreased survival of xenograft mice in vivo. | [76] |
| CXCR4/CXCL12 | CXCR4 inhibition doubles the response to immune checkpoint blockers in mice with metastatic triple-negative breast cancer (TNBC). CXCL12/CXCR4-mediated desmoplasia in metastatic breast cancer promotes immunosuppression and is a potential target to overcome therapeutic resistance to immune checkpoint blockade in MBC patients. | [77] |
| PDK1 | PDK1-dependent metabolic reprogramming is a key regulation of metabolism and metastasis to the liver in breast cancer. PDK1 is particularly required for metabolic adaptation to nutrient restriction and hypoxia as a HIF1α target of metastatic cells in the liver. | [78] |
| circRNA hsa_circ_0008324 (circEZH2) | CircEZH2 enhances oncogenesis and metastasis in vitro and in vivo by activating KLF5 protein expression, which in turn activates CXCR4 transcription, leading to the initiation of the EMT programme in breast cancer. | [79] |
| circRNA hsa_circ_0124696 (circROBO1) | Increased expression of circROBO1 was found in liver metastases in breast cancer and correlated with poor prognosis. Knockdown of circROBO1 strongly inhibited proliferation, migration and invasion of RRM cells, whereas circROBO1 overexpression showed opposite effects. circROBO1 overexpression promoted tumour growth and metastasis to the liver in vivo. | [80] |
| Lyn (Src-family kinase) | The Lyn-selective kinase inhibitor, bafetinib (INNO-406), reduces claudin-2 expression and suppresses breast cancer metastasis to the liver. | [81] |
| PPFIA1 | PPFIA1 is activated in breast cancer metastasis to the liver and is a potentially unfavourable prognostic sign of metastases development. Kaplan–Meier plotter results showed that although high PPFIA1 expression was generally associated with reduced distant metastasis-free survival in oestrogen receptor+ patients, subgroup analysis only confirmed significant association in an oestrogen receptor+/N− (node-negative) group (median survival, high PPFIA1 group vs. low PPFIA1 cohort: 191.21 vs. 236.22 months, hazard ratio: 2.23, 95% confidence interval: 1.42–3.5, p < 0.001), but not in an oestrogen receptor+/N+ (nodal positive) group (hazard ratio: 1.63, 95% confidence interval: 0.88–3.03, p = 0.12). In oestrogen receptor patients, there was no association between PPFIA1 expression and distant metastasis-free survival, regardless of Nm (mixed nodal status), N− or N+ subgroups. In bc-GenExMiner 4.0 programme using the Nottingham Prognostic Index and Adjuvant! Online-adjusted analysis validated the independent prognostic value of PPFIA1 in relation to the risk of metastasis in patients with oestrogen receptor+/N−. | [82] |
| ESR1, AKT1, ERBB2, FGFR4 | ESR1 (20%), AKT1 (8%), ERBB2 (7%) and FGFR4 (4%) were identified as driver genes for breast cancer metastasis. | [83] |
| BRAIN | ||
| PI3K | Activation of PI3K was found in a large proportion (77%) of brain metastases in patients with breast cancer, and activation of PI3K-Akt signalling in such metastases was associated with poor outcomes. Pharmacological inhibition of PI3K activity was found to attenuate the expression of PD-L1, CTLA4 and CSF1 genes, as well as the infiltration of metastatic breast cancer cells into the brains of mice. | [84,85] |
| CDK4 u CDK6 | Abemaciclib, an inhibitor of the cyclin-dependent kinases CDK4 and CDK6, has shown potential for the treatment of brain metastases in patients with breast cancer. The combination of abemaciclib with endocrine therapy was effective in patients with HER2-negative breast cancer and brain metastases, and 38% of patients had a reduction in metastatic tumour burden. | [86] |
| STAT3 | The STAT3 inhibitor silibinin, which penetrates the blood–brain barrier, impairs the viability of brain metastases in both mice and humans. This inhibitor is thought to block the growth of brain metastases by targeting STAT3 in tumour-associated astrocytes, thereby weakening their interaction with tumour cells and microglia. | [87] |
| JAK, JAK2 | The JAK inhibitor ruxolitinib limits the growth of primary brain tumours and also reduces the number of tumour-associated astrocytes in mice. JAK2/STAT3 signal transduction is hyperactivated when breast cancer metastasises to the brain. Inhibition of JAK2 results in reduced brain metastasis in vivo, suggesting that JAK2 may be a promising therapeutic target. | [88,89] |
| COX2 | COX2 can promote MMP1 expression, which is significantly correlated with brain metastasis. In addition, COX2 and prostaglandin activate astrocytes to release chemokine ligand, promoting self-renewal of tumour stem cells or tumour-initiating cells in the brain. | [90] |
| FABP7 | FABP7 is a key regulator of metabolism in HER2+ breast cancer metastasis to the brain. FABP7 has been shown to be required for the activation of key metastatic genes and pathways, such as integrins-Src and VEGFA, as well as for the growth of HER2+ breast cancer cells in the brain microenvironment in vivo. | [91] |
| miR-4428, miR-4480 | In a study of microRNAs in patients with advanced breast cancer with brain metastases, it was shown that the determination of miR-4428 and miR-4480 in serum may be useful as prognostic biomarkers. A total of 51 serum samples from patients with breast cancer and brain metastasis, and 28 serum samples from controls without brain metastasis were obtained. Two miRNAs, miR-4428 and miR-4480 could significantly distinguish patients with brain metastasis, with area under the receiver operating characteristic curve (AUC) values of 0.779 and 0.781, respectively, while a combination of miR-4428 and progesterone receptor had an AUC value of 0.884. | [92] |
| PLVAP | PLVAP staining was observed not only in isolated brain microvessels but also in brain metastases in breast cancer. Immune labelling for PLVAP was performed in 4T1 TNBC culture, where clear expression of this protein was observed. | [93] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibragimova, M.K.; Tsyganov, M.M.; Kravtsova, E.A.; Tsydenova, I.A.; Litviakov, N.V. Organ-Specificity of Breast Cancer Metastasis. Int. J. Mol. Sci. 2023, 24, 15625. https://doi.org/10.3390/ijms242115625
Ibragimova MK, Tsyganov MM, Kravtsova EA, Tsydenova IA, Litviakov NV. Organ-Specificity of Breast Cancer Metastasis. International Journal of Molecular Sciences. 2023; 24(21):15625. https://doi.org/10.3390/ijms242115625
Chicago/Turabian StyleIbragimova, Marina K., Matvey M. Tsyganov, Ekaterina A. Kravtsova, Irina A. Tsydenova, and Nikolai V. Litviakov. 2023. "Organ-Specificity of Breast Cancer Metastasis" International Journal of Molecular Sciences 24, no. 21: 15625. https://doi.org/10.3390/ijms242115625
APA StyleIbragimova, M. K., Tsyganov, M. M., Kravtsova, E. A., Tsydenova, I. A., & Litviakov, N. V. (2023). Organ-Specificity of Breast Cancer Metastasis. International Journal of Molecular Sciences, 24(21), 15625. https://doi.org/10.3390/ijms242115625






