Effects of Exercise Training on Mitochondrial Fatty Acid β-Oxidation in the Kidneys of Dahl Salt-Sensitive Rats
Abstract
1. Introduction
2. Results
2.1. Effects of HS and Ex on Plasma Parameters and Body Weight
2.2. Effects of HS and Ex on SBP, Urinary Protein, and Creatinine Clearance
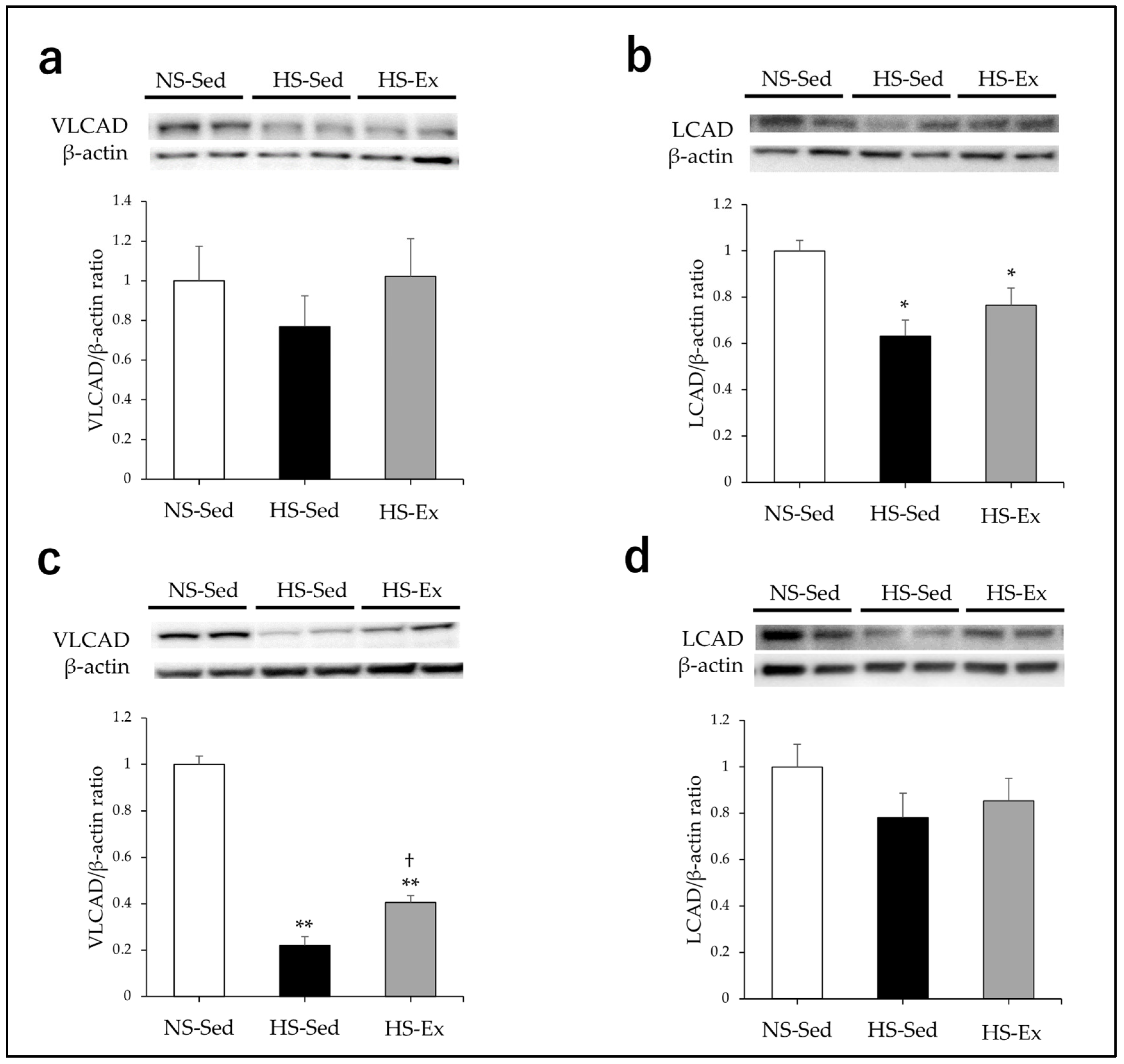
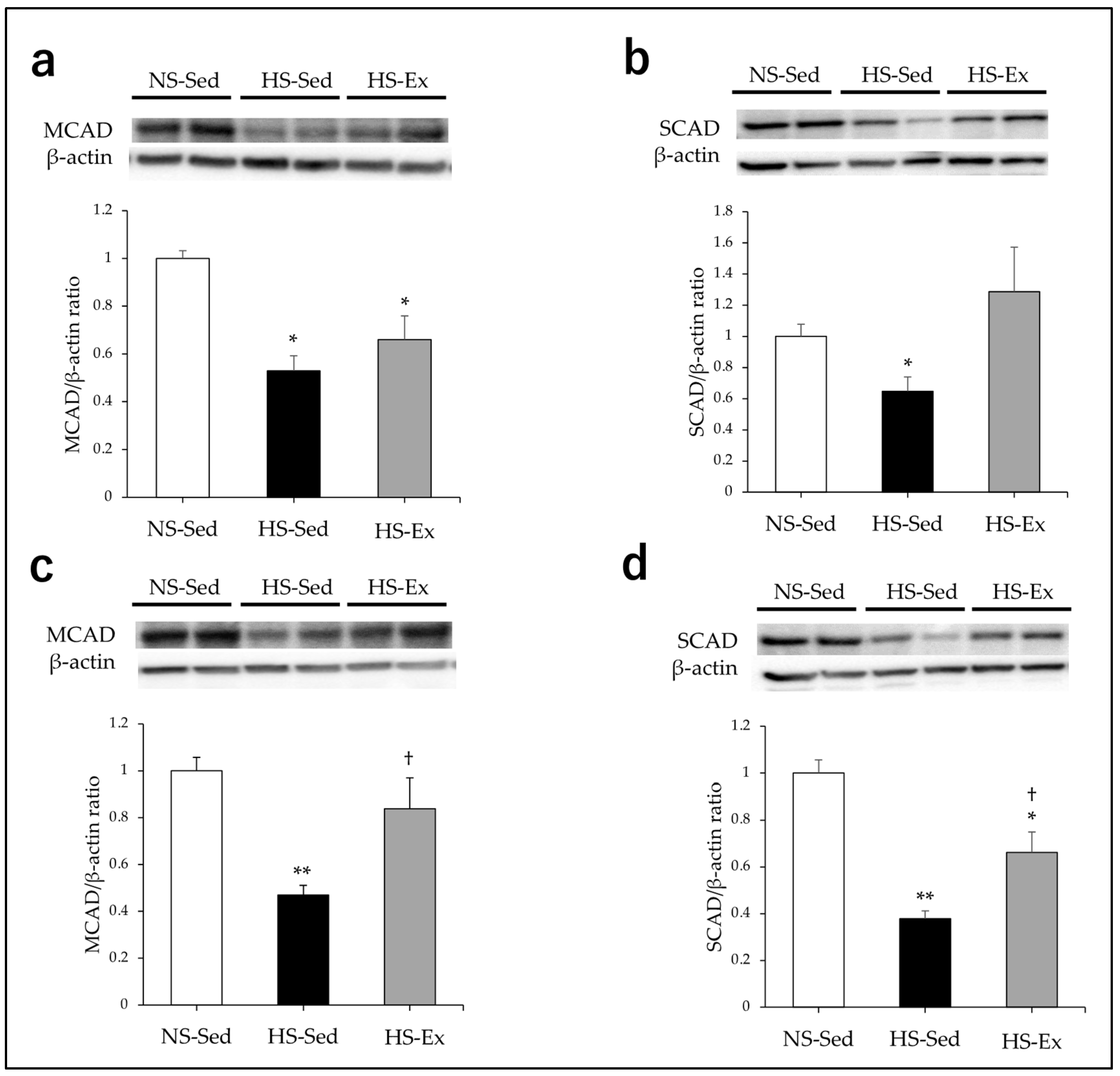
2.3. Effects of HS and Ex on the Expression of Acyl-CoA Dehydrogenase
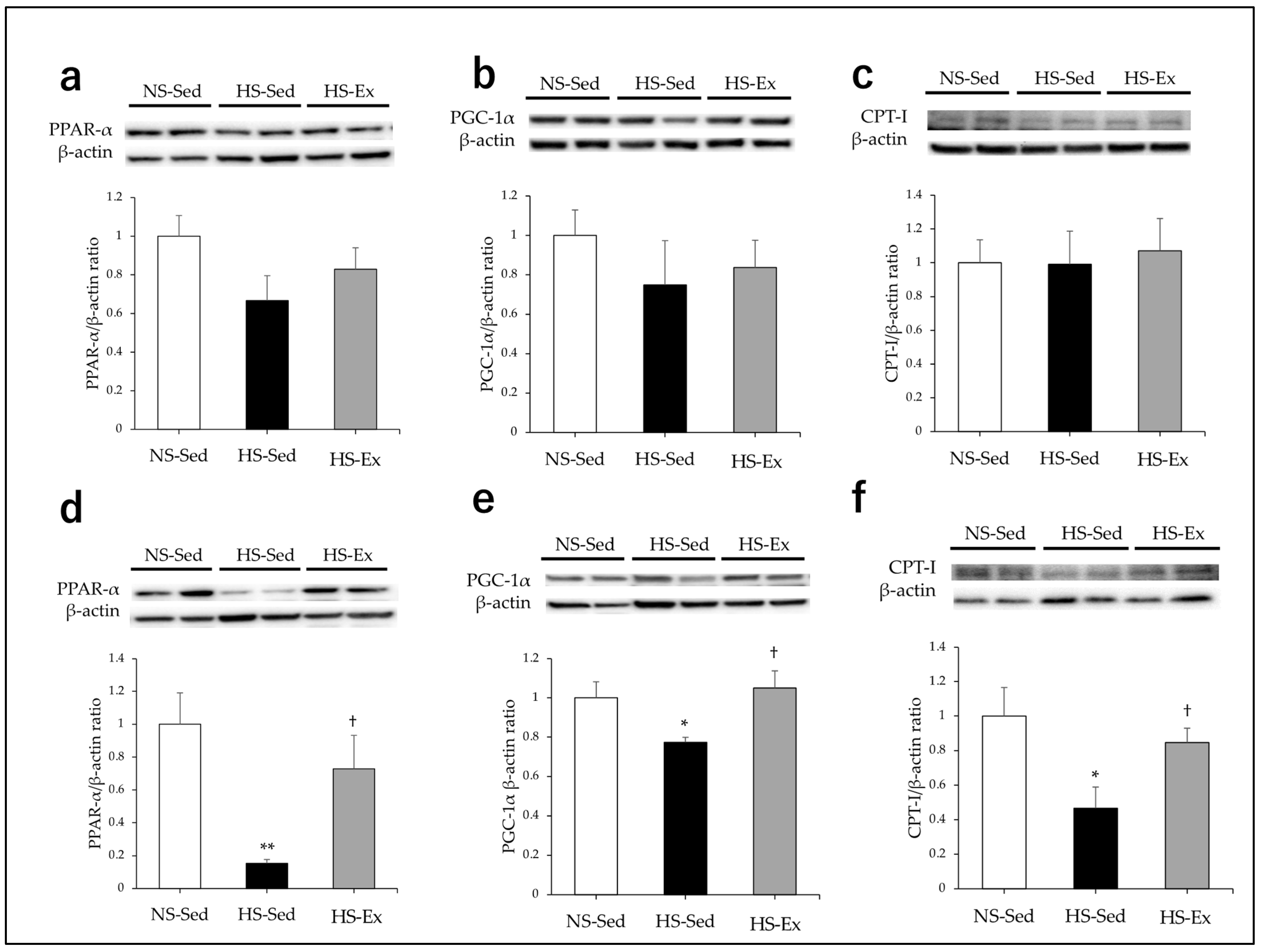
2.4. Effects of HS and Ex on the Expressions of CPT-I, PPAR-α, and PGC-1α
2.5. Effects of Ex on the Expression of ETC Enzymes
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Measurement of Blood Pressure and Plasma and Urine Parameters
4.3. Preparation of Kidney Samples
4.4. Western Blot Analysis
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pagliarini, D.; Calvo, S.E.; Chang, B.; Sheth, S.A.; Vafai, S.B.; Ong, S.-E.; Walford, G.A.; Sugiana, C.; Boneh, A.; Chen, W.K.; et al. A mitochondrial protein compendium elucidates complex I diseases biology. Cell 2008, 134, 112–123. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, P.M. Renal oxygen delivery: Matching delivery to metabolic demand. Clin. Exo Pharmacol. Physiol. 2006, 33, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Braga, P.C.; Alves, M.G.; Rodrigues, A.S.; Oliveria, P.F. Mitochondrial pathophysiology on chronic kidney disease. Int. J. Mol. Sci. 2002, 23, 1776. [Google Scholar] [CrossRef] [PubMed]
- Bhargava, P.; Schnellmann, R.G. Mitochondrial energetics in the kidney. Nat. Rev. Nephrol. 2017, 13, 629–646. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Kestenbaum, B. Proximal tubular secretory clearance: A neglected partner of kidney function. Clin. J. Am. Soc. Nephrol. 2018, 13, 1291–1296. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.R.; Tran, M.T.; Parikh, S.M. PGC1α in the kidney. Am. J. Physiol. Renal Physiol. 2018, 314, F1–F8. [Google Scholar] [CrossRef] [PubMed]
- Mello, T.; Materozzi, M.; Galli, A. PPARs and mitochondrial metabolism: From NAFLD to HCC. PPAR Res. 2016, 2016, 7403230. [Google Scholar] [CrossRef] [PubMed]
- Portilla, D.; Dai, G.; McClure, T.; Bates, L.; Kurten, R.; Megyesi, J.; Price, P.; Li, S. Alterations of PPARα and its coactivator PGC-1 in cisplatin-induced acute renal failure. Kidney Int. 2002, 62, 1208–1218. [Google Scholar] [CrossRef]
- Muroya, Y.; Ito, O.; Rong, R.; Takashima, K.; Ito, D.; Cao, P.; Nakamura, Y.; Joh, K.; Kohzuki, M. Disorder of fatty acid metabolism in the kidney of PAN-induced nephrotic rats. Am. J. Physiol. Renal Physiol. 2012, 303, F1070–F1079. [Google Scholar] [CrossRef]
- Kasiske, B.L.; O’Donnel, M.P.; Gravis, W.J.; Keane, W. Pharmacologic treatment of hyperlipidemia reduces glomerular injury in rat 5/6 nephrectomy model of chronic renal failure. Circ. Res. 1988, 62, 367–374. [Google Scholar] [CrossRef]
- Tanaka, Y.; Kume, S.; Araki, S.; Isshiki, K.; Chin-Kanasaki, M.; Sakaguchi, M.; Sugimoto, T.; Koya, D.; Haneda, M.; Kashiwagi, A.; et al. Fenofibrate, a PPARα agonist, has renoprotective effects in mice by enhancing renal lipolysis. Kidney Int. 2011, 79, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Tran, M.T.; Zsengeller, Z.K.; Berg, A.H.; Khankin, E.V.; Bhasin, M.K.; Kim, W.; Clish, C.B.; Stillman, I.E.; Karamanchi, S.A.; Rhee, E.P.; et al. PGC1α drives NAD biosynthesis linking oxidative metabolism to renal protection. Nature 2016, 531, 528–532. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, T.; Bennion, B.G.; Stenson, E.K.; Lynn, J.C.; Otinga, C.; Djukovic, D.; Raftery, D.; Fei, L.; Wong, H.R.; Liles, W.C.; et al. PPARα contributes to protection against metabolic and inflammatory derangements associated with acute kidney injury in experimental sepsis. Physiol. Rep. 2019, 7, e14078. [Google Scholar] [CrossRef] [PubMed]
- Miguel, V.; Tituaña, J.; Herrero, J.I.; Herrero, L.; Serre, D.; Cuevas, P.; Barbas, C.; Puyol, D.R.; Márquez-Expósito, L.; Ruiz-Ortega, M.; et al. Renal tubule Cpt1a overexpression protects from kidney fibrosis by restoring mitochondrial homeostasis. J. Clin. Investig. 2021, 131, e140695. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Kanazawa, M.; Ishida, A.; Tufescu, A.; Sasaki, Y.; Ito, O.; Kurosawa, H.; Sato, T.; Ootaka, T.; Kohzuki, M. Combination of chronic exercise and antihypertensive therapy enhances renoprotective effects in rats with renal ablation. Am. J. Hypertens. 2009, 22, 1101–1106. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kohzuki, M.; Kamimoto, M.; Wu, X.M.; Xu, H.L.; Kawamura, T.; Mori, Y.; Nagasaka, M.; Kurosawa, H.; Minamia, N.; Kanazawa, M.; et al. Renal protective effects of chronic exercise and antihypertensive therapy in hypertensive rats with chronic renal failure. J. Hyypertens 2001, 19, 1877–1882. [Google Scholar] [CrossRef] [PubMed]
- Ito, D.; Cao, P.; Kakihana, T.; Sato, E.; Suda, C.; Muroya, Y.; Ogawa, Y.; Hu, G.; Ishii, T.; Ito, O.; et al. Chronic running exercise alleviates early progression of nephropathy with upregulation of nitric oxide synthases and suppression of glycation in Zucker diabetic rats. PLoS ONE 2015, 10, e0138037. [Google Scholar] [CrossRef]
- Tufescu, A.; Kanazawa, M.; Ishida, A.; Lu, H.; Sasaki, Y.; Ootaka, T.; Sato, T.; Kohzuki, M. Combination of exercise and losartan enhances renoprotective and peripheral effects in spontaneously type 2 diabetes mellitus rats with nephropathy. J. Hypertens. 2008, 26, 312–321. [Google Scholar] [CrossRef]
- Hu, G.; Xu, L.; Ma, Y.; Kohzuki, M.; Ito, O. Chronic exercise provides renal-protective effects with upregulation of fatty acid oxidation in the kidney oh high fructose-fed rats. Am. J. Physiol. Renal Physiol. 2020, 318, F826–F834. [Google Scholar] [CrossRef]
- Rapp, J.P. Dahl salt-susceptible and salt-resistant rats. A review. Hypertension 1982, 4, 753–763. [Google Scholar] [CrossRef]
- Alonso-Galicia, M.; Frohlich, B.; Roman, R.J. Induction of P4504A activity improves pressure-natriuresis in Dahl S rats. Hypertension 1998, 31, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Dahl, L.K.; Heine, M.; Tassinari, L. Effects of chronic salt ingestion. Evidence that genetic factors play an important role in susceptibility to experimental hypertension. J. Exp. Med. 1962, 115, 1173–1190. [Google Scholar] [CrossRef] [PubMed]
- Doi, R.; Masuyama, T.; Yamamoto, K.; Doi, Y.; Mano, T.; Sakata, Y.; Ono, K.; Kuzuya, T.; Hirota, S.; Koyama, T.; et al. Development of different phenotypes of hypertensive heart failure: Systolic versus diastolic failure in Dahl salt-sensitive rats. J. Hypertens. 2000, 18, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Manning, R.J., Jr.; Hu, L.; Tan, D.Y.; Meng, S. Role of abnormal nitric oxide system in salt-sensitive hypertension. Am. J. Hypertens. 2001, 14, 68S–73S. [Google Scholar] [CrossRef] [PubMed]
- Overton, J.M.; VanNess, J.; Takata, H.J. Effects of chronic exercise on blood pressure in Dahl salt-sensitive rats. Am. J. Hypertens. 1998, 11, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Elijovich, F.; Weinberger, M.H.; Anderson, C.A.M.; Appel, L.J.; Bursztyn, M.; Cook, N.R.; Dart, R.A.; Newton-Chen, C.H.; Sacks, F.M.; Laffer, C.L. Salt sensitivity of blood pressure: A scientific statement from the American Heart Association. Hypertension 2016, 68, e7–e46. [Google Scholar] [CrossRef] [PubMed]
- Roman, R.J. P-450 metabolites of arachidonic acid in the control of cardiovascular function. Physiol. Rev. 2002, 82, 131–185. [Google Scholar] [CrossRef] [PubMed]
- Tanada, Y.; Okuda, J.; Kato, T.; Minamino-Muta, E.; Murata, I.; Soga, T.; Shioi, T.; Kimura, T. The metabolic profile of a rat model of chronic kidney disease. PeerJ 2017, 5, e3352. [Google Scholar] [CrossRef]
- Roman, R.J.; Ma, Y.H.; Frohlich, B.; Markham, B. Clofibrate prevents the development of hypertension in Dahl salt-sensitive rats. Hypertension 1993, 21, 985–988. [Google Scholar] [CrossRef]
- Holloway, T.M.; Bloemberg, D.; Silva, M.L.; Quadrilatero, J.; Spriet, L.L. High-intensity interval and endurance training are associate with divergent skeletal muscle adaptations in a rodent model of hypertension. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R927–R934. [Google Scholar] [CrossRef]
- Bowen, T.S.; Rolim, N.P.L.; Fischer, T.; Baekkerud, F.H.; Medeiros, A.; Werner, S.; Brφnstad, E.; Rognmo, O.; Mangner, N.; Linke, A.; et al. Heart failure with preserved ejection fraction induces molecular, mitochondrial, histological, and functional alterations in rat respiratory and limb skeletal muscle. Eur. J. Heart Fail. 2015, 17, 253–272. [Google Scholar] [CrossRef]
- Libonati, J.R.; Gaudhan, J.P. Low-intensity exercise training improves survival in Dahl salt hypertension. Med. Sci. Sports Exerc. 2006, 38, 856–858. [Google Scholar] [CrossRef] [PubMed]
- Miyachi, M.; Yazawa, H.; Furukawa, M.; Tshuboi, K.; Ohtake, M.; Nishizawa, T.; Hashimoto, K.; Yokoi, T.; Kojima, T.; Murate, T.; et al. Exercise training alters left ventricular geometry and attenuates heart failure in Dahl salt-sensitive hypertensive rats. Hypertension 2009, 53, 701–707. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ogawa, Y.; Takahashi, J.; Sakuyama, A.; Xu, L.; Miura, T.; Muroya, Y.; Ito, D.; Kohzuki, M.; Ito, O. Exercise training delays renal disorders with decreasing oxidative stress and increasing production of 20-hydroxyeicosatetraenoic acid in Dahl salt-sensitive rats. J. Hypertens. 2020, 38, 1136–1146. [Google Scholar] [CrossRef] [PubMed]
- Che, R.; Yuan, Y.; Huang, S.; Zhang, A. Mitochondrial dysfunction in the pathophysiology of renal diseases. Am. J. Physiol. Renal Physiol. 2013, 306, F367–F378. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Sun, Q.; Sun, N.; Liang, M.; Tian, Z. Mitochondrial dysfunction and altered renal metabolism in Dahl salt-sensitive rats. Kidney Blood Press. Res. 2017, 42, 587–597. [Google Scholar] [CrossRef]
- He, X.; Liu, Y.; Usa, K.; Tian, Z.; Cowlet, A.W., Jr.; Liang, M. Ultrastructure of mitochondria and the endoplasmic reticulum in renal tubules of Dahl-salt-sensitive rats. Am. J. Physiol. Renal Physiol. 2014, 306, F1190–F1197. [Google Scholar] [CrossRef]
- Zheleznova, N.N.; Yang, C.; Ryan, R.P.; Halligan, B.D.; Liang, M.; Greene, A.S.; Cowley, A.W., Jr. Mitochondrial proteomic analysis reveals deficiencies in oxygen utilization in medullary thick ascending limb of Henle in the Dahl salt-sensitive rat. Physiol. Genom. 2012, 44, 829–842. [Google Scholar] [CrossRef] [PubMed]
- Gu, Q.; Zhao, L.; Ma, Y.P.; Liu, J.D. Contribution of mitochondrial function to exercise-induced attenuation of renal dysfunction in spontaneously hypertensive rats. Mol. Cell Biochem. 2015, 406, 217–225. [Google Scholar] [CrossRef]
- Console, L.; Scalise, M.; Giangregorio, N.; Tonazzi, A.; Barlie, M.; Indiveri, C. The link between the mitochondrial fatty acid oxidation derangement and kidney injury. Front. Physiol. 2020, 11, 794. [Google Scholar] [CrossRef]
- Kang, H.M.; Ahn, S.H.; Choi, P.; Ko, Y.A.; Han, S.H.; Chinga, F.; Park, A.S.D.; Tao, J.; Sharma, K.; Pullman, J.; et al. Defective fatty acid oxidation in renal tubular epithelial cells has a key role in kidney fibrosis development. Nat. Med. 2015, 21, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Shimada, S.; Hoffmann, B.R.; Yang, C.; Kurth, T.; Greene, A.S.; Liang, M.; Dash, R.K.; Cowley, A.W., Jr. Metabolic responses of normal rat kidney to a high salt intake. Function 2023, 4, zqad031. [Google Scholar] [CrossRef] [PubMed]
- Choksi, K.B.; Nuss, J.E.; DeFord, J.H.; Papaconstantinou, J. Mitochondrial electron transport chain functions in long-lived Ames dwarf mice. Aging 2011, 3, 754–767. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ling, X.C.; Kuo, K.-L. Oxidative stress in chronic kidney disease. Ren. Replace. Ther. 2018, 4, 53. [Google Scholar] [CrossRef]
- Zhang, L.; Liu, J.; Zhou, F.; Wang, W.; Chen, N. PGC-1α ameliorates kidney fibrosis in mice with diabetic kidney disease through an antioxidative mechanism. Mol. Med. Rep. 2018, 17, 4490–4498. [Google Scholar] [CrossRef]
- Rasbach, K.A.; Schnellmann, R.G. Signaling of mitochondrial biogenesis following oxidant injury. J. Biol. Chem. 2007, 282, 2355–2362. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]


| NS-Sed | HS-Sed | HS-Ex | |
|---|---|---|---|
| Body Weight (g) | 384.8 ± 5.3 | 331.0 ± 9.0 ** | 323.9 ± 10.3 ** |
| Total protein (g/dL) | 5.8 ± 0.1 | 5.7 ± 0.1 | 5.7 ± 0.1 |
| Albumin (g/dL) | 3.4 ± 0.1 | 2.8 ± 0.1 ** | 3.1 ± 0.2 |
| Glucose (mg/dL) | 156.9 ± 7.0 | 135.5 ± 2.4 * | 155.4 ± 5.3 †† |
| Triglyceride (mg/dL) | 111.0 ± 11.8 | 144.8 ± 16.3 | 117 ± 11.3 |
| Free fatty acid (μEq/L) | 141.9 ± 9.1 | 196.5 ± 26.6 | 202.0 ± 16.6 ** |
| Total cholesterol (mg/dL) | 61.0 ± 3.3 | 154.8 ± 5.2 ** | 112.7 ± 14.4 ** †† |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Namai-Takahashi, A.; Takahashi, J.; Ogawa, Y.; Sakuyama, A.; Xu, L.; Miura, T.; Kohzuki, M.; Ito, O. Effects of Exercise Training on Mitochondrial Fatty Acid β-Oxidation in the Kidneys of Dahl Salt-Sensitive Rats. Int. J. Mol. Sci. 2023, 24, 15601. https://doi.org/10.3390/ijms242115601
Namai-Takahashi A, Takahashi J, Ogawa Y, Sakuyama A, Xu L, Miura T, Kohzuki M, Ito O. Effects of Exercise Training on Mitochondrial Fatty Acid β-Oxidation in the Kidneys of Dahl Salt-Sensitive Rats. International Journal of Molecular Sciences. 2023; 24(21):15601. https://doi.org/10.3390/ijms242115601
Chicago/Turabian StyleNamai-Takahashi, Asako, Junta Takahashi, Yoshiko Ogawa, Akihiro Sakuyama, Lusi Xu, Takahiro Miura, Masahiro Kohzuki, and Osamu Ito. 2023. "Effects of Exercise Training on Mitochondrial Fatty Acid β-Oxidation in the Kidneys of Dahl Salt-Sensitive Rats" International Journal of Molecular Sciences 24, no. 21: 15601. https://doi.org/10.3390/ijms242115601
APA StyleNamai-Takahashi, A., Takahashi, J., Ogawa, Y., Sakuyama, A., Xu, L., Miura, T., Kohzuki, M., & Ito, O. (2023). Effects of Exercise Training on Mitochondrial Fatty Acid β-Oxidation in the Kidneys of Dahl Salt-Sensitive Rats. International Journal of Molecular Sciences, 24(21), 15601. https://doi.org/10.3390/ijms242115601







