Physiological Analysis and Genetic Mapping of Short Hypocotyl Trait in Brassica napus L.
Abstract
1. Introduction
2. Results
2.1. Physiological Basis of Plant Responses to Low-Temperature Stress
2.2. Observation of Trypan Blue Staining and Counting of Dead Cell Area
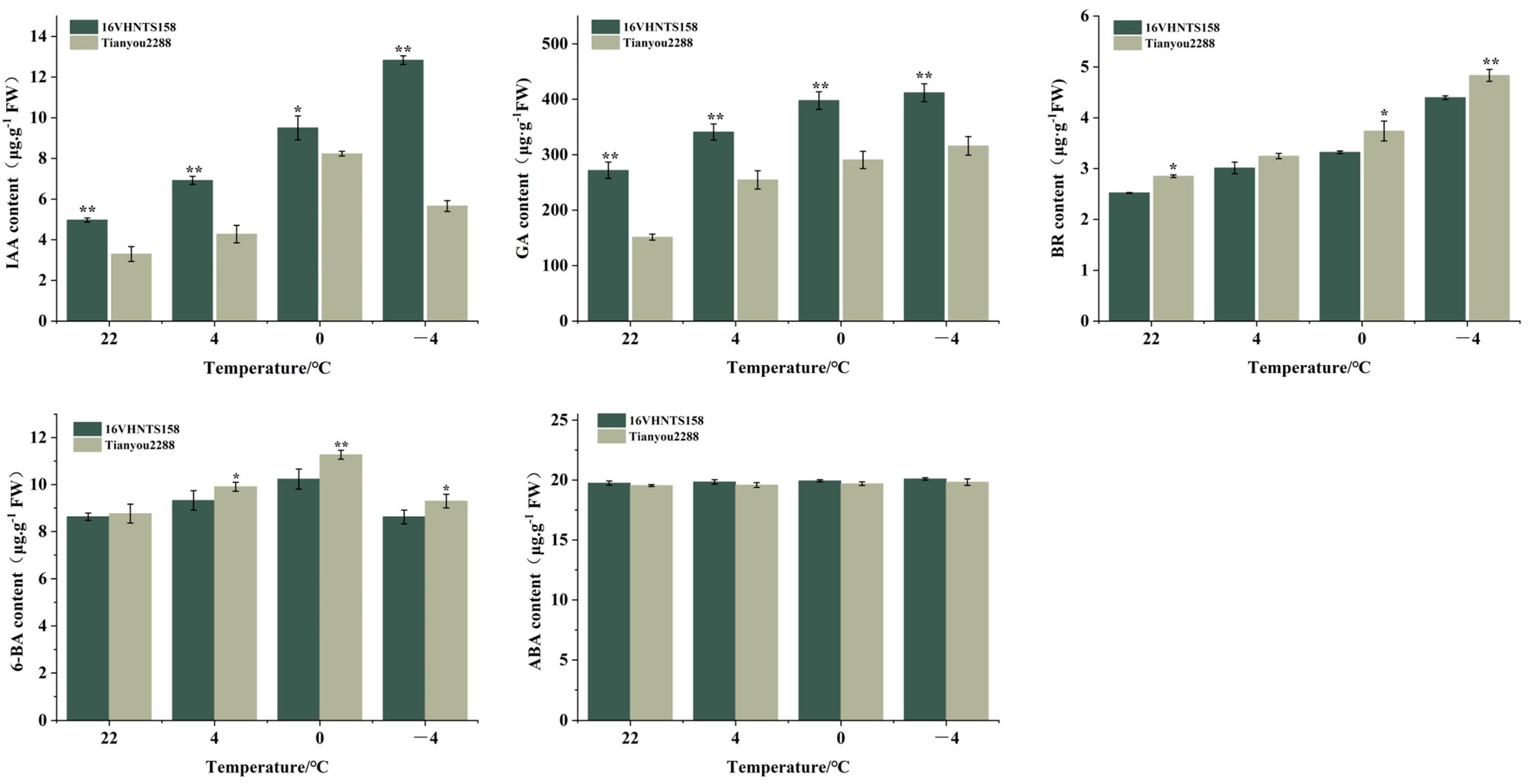
2.3. Differences in Endogenous Hormone Content in Embryonic Axes under Low-Temperature Stress
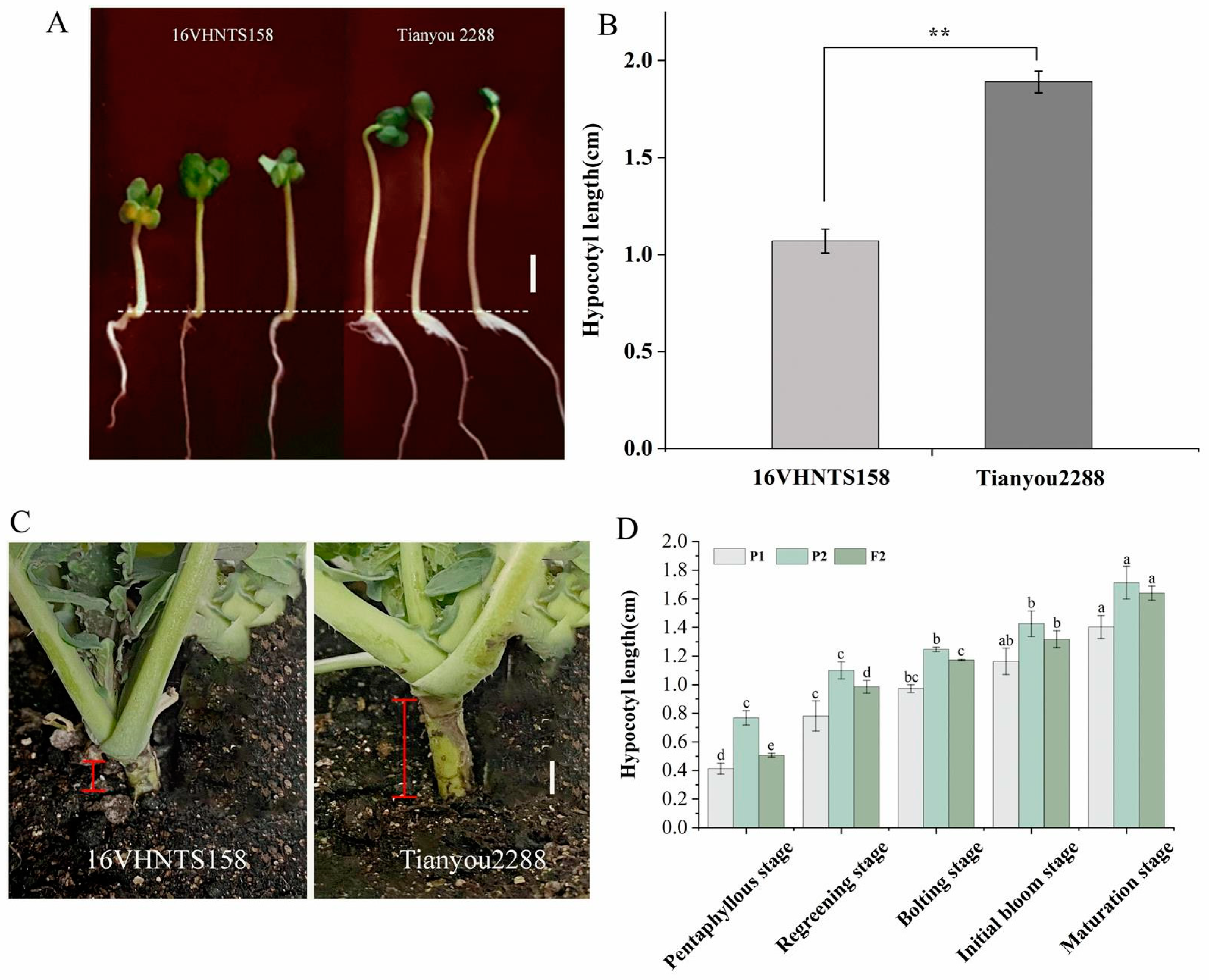
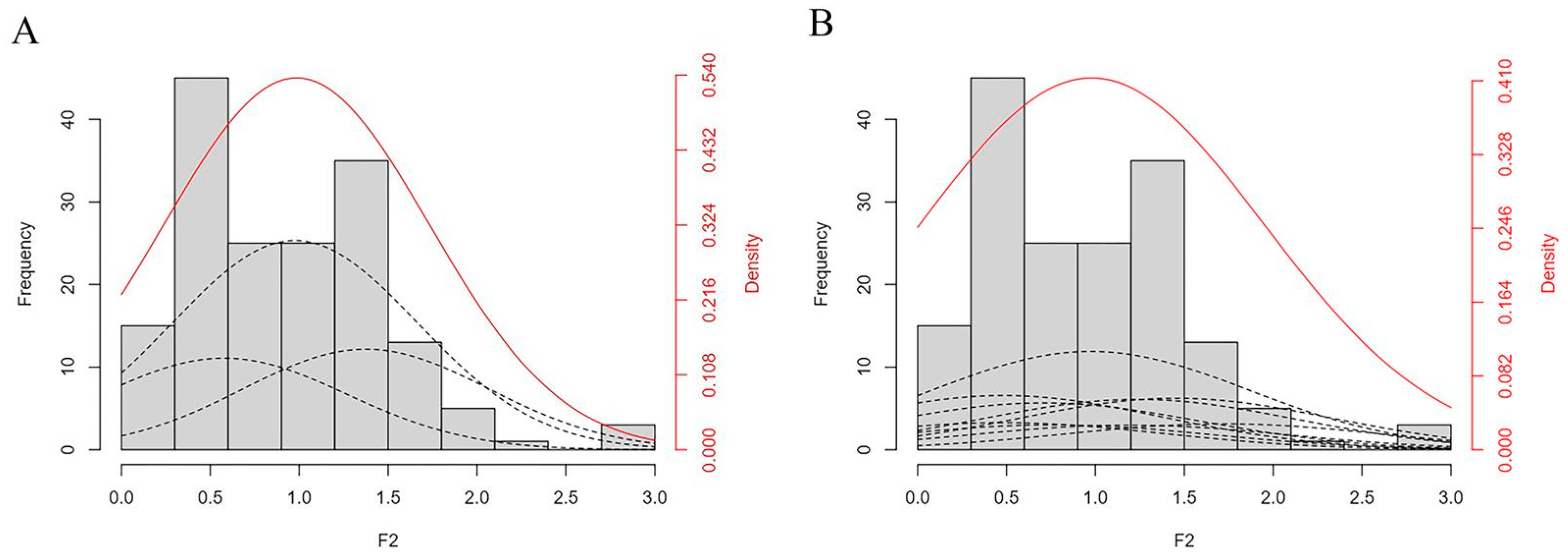
2.4. Measurement and Genetic Analysis of Hypocotyl Length in the Parents and F2 Populations
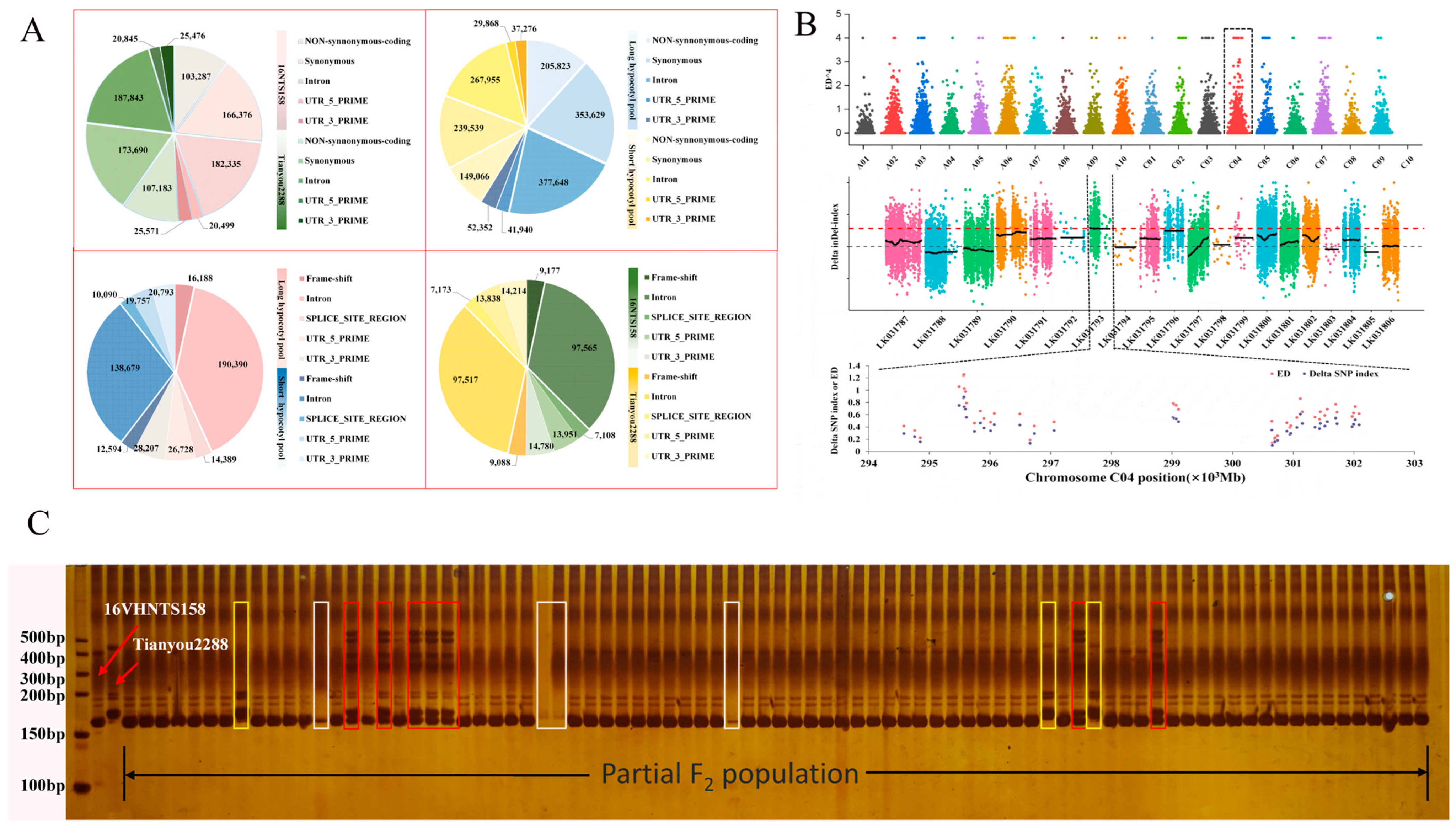
2.5. Mutant Locus Mapped on C04 Chromosome
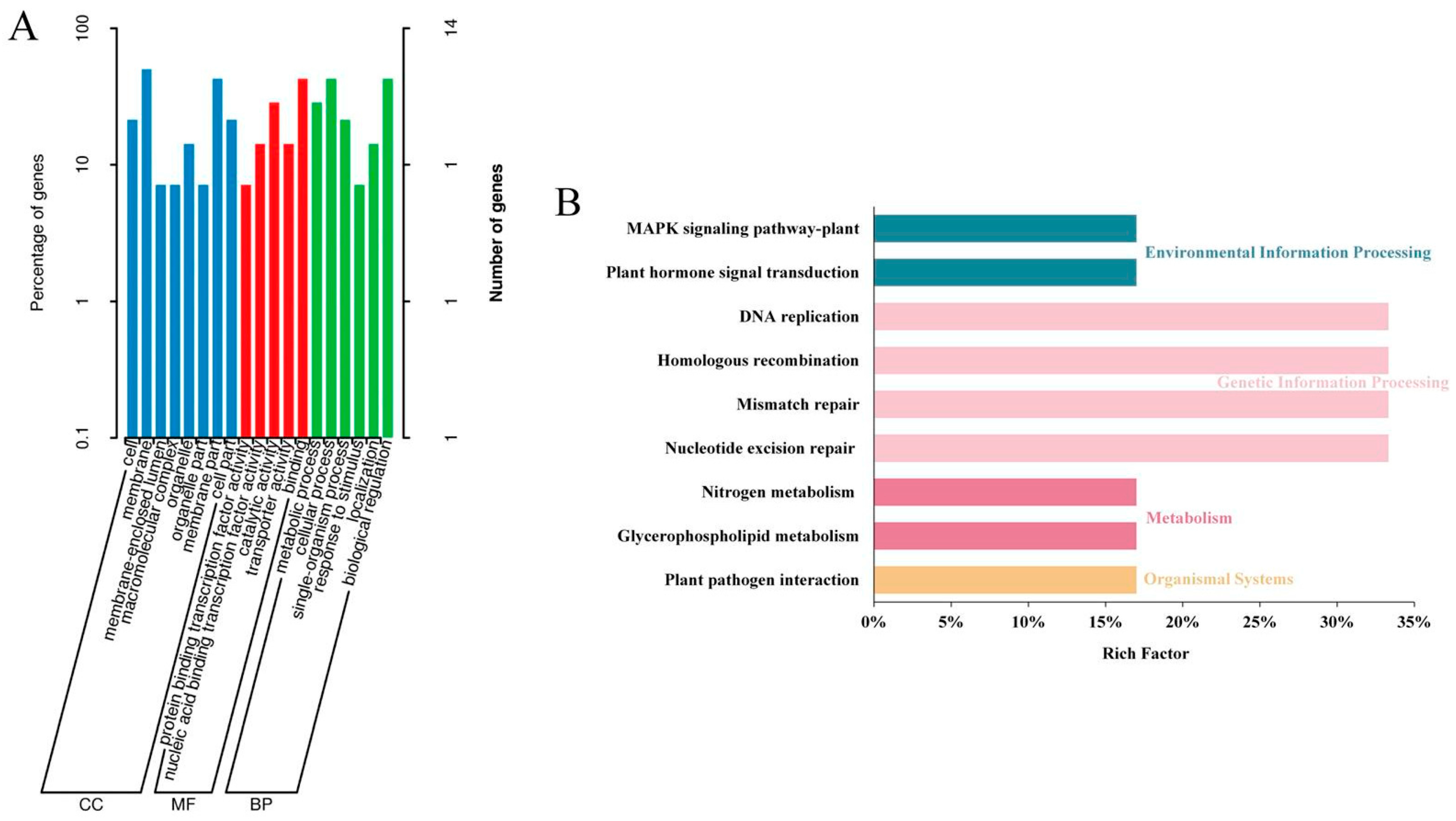
2.6. Selection of Candidate Regions and Functional Annotation
3. Discussion
4. Materials and Methods
4.1. Experiment Population
4.2. Determination of LT50 and Ion Leakage Rate
4.3. Observation of Trypan Blue Staining and Counting of Dead Cell Area
4.4. Measurement of Endogenous Hormone Content
4.5. Mixed Pool Construction and Sequencing Data Processing
4.6. DNA Extraction and PCR Amplication
4.7. Statistical and Functional Candidate Genes Analyses
4.7.1. Statistical Analyses
4.7.2. Functional Annotation of Candidate Genes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nakano, T. Hypocotyl Elongation: A Molecular Mechanism for the First Event in Plant Growth That Influences Its Physiology. Plant Cell Physiol. 2019, 60, 933–934. [Google Scholar] [CrossRef]
- Ragni, L.; Hardtke, C.S. Small but thick enough—The Arabidopsis hypocotyl as a model to study secondary growth. Physiol. Plant 2014, 151, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Zhang, Y.; Liu, R.; Hao, H.; Wang, Z.; Bi, Y. Phytochrome interacting factors (PIFs) are essential regulators for sucrose-induced hypocotyl elongation in Arabidopsis. J. Plant Physiol. 2011, 168, 1771–1779. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; He, J.; Yao, H.; Yu, Q.; Zhang, Q.; Li, K.; Huang, Y.; Chen, L.; Li, X.; Yang, Y.; et al. CARK3-mediated ADF4 regulates hypocotyl elongation and soil drought stress in Arabidopsis. Front. Plant Sci. 2022, 13, 1065677. [Google Scholar] [CrossRef] [PubMed]
- Chia, P.-L.; Kubota, C. End-of-day Far-red Light Quality and Dose Requirements for Tomato Rootstock Hypocotyl Elongation. HortScience 2010, 45, 1501–1506. [Google Scholar] [CrossRef]
- Gabriele, S.; Rizza, A.; Martone, J.; Circelli, P.; Costantino, P.; Vittorioso, P. The Dof protein DAG1 mediates PIL5 activity on seed germination by negatively regulating GA biosynthetic gene AtGA3ox1. Plant J. 2010, 61, 312–323. [Google Scholar] [CrossRef]
- Park, J.E.; Seo, P.J.; Lee, A.K.; Jung, J.H.; Kim, Y.S.; Park, C.M. An Arabidopsis GH3 gene, encoding an auxin-conjugating enzyme, mediates phytochrome B-regulated light signals in hypocotyl growth. Plant Cell Physiol. 2007, 48, 1236–1241. [Google Scholar] [CrossRef]
- Feng, S.; Martinez, C.; Gusmaroli, G.; Wang, Y.; Zhou, J.; Wang, F.; Chen, L.; Yu, L.; Iglesias-Pedraz, J.M.; Kircher, S.; et al. Coordinated regulation of Arabidopsis thaliana development by light and gibberellins. Nature 2008, 451, 475–479. [Google Scholar] [CrossRef]
- Schwechheimer, C. Understanding gibberellic acid signaling—Are we there yet? Curr. Opin. Plant Biol. 2008, 11, 9–15. [Google Scholar] [CrossRef]
- Sun, J.; Qi, L.; Li, Y.; Chu, J.; Li, C. PIF4-mediated activation of YUCCA8 expression integrates temperature into the auxin pathway in regulating arabidopsis hypocotyl growth. PLoS Genet. 2012, 8, e1002594. [Google Scholar] [CrossRef]
- Smets, R.; Le, J.; Prinsen, E.; Verbelen, J.P.; Van Onckelen, H.A. Cytokinin-induced hypocotyl elongation in light-grown Arabidopsis plants with inhibited ethylene action or indole-3-acetic acid transport. Planta 2005, 221, 39–47. [Google Scholar] [CrossRef]
- Luo, X.; Xue, Z.; Ma, C.; Hu, K.; Zeng, Z.; Dou, S.; Tu, J.; Shen, J.; Yi, B.; Fu, T. Joint genome-wide association and transcriptome sequencing reveals a complex polygenic network underlying hypocotyl elongation in rapeseed (Brassica napus L.). Sci. Rep. 2017, 7, 41561. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Panthee, D.R. PyBSASeq: A simple and effective algorithm for bulked segregant analysis with whole-genome sequencing data. BMC Bioinform. 2020, 21, 99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Yuan, M.; Xia, H.; He, L.; Ma, J.; Wang, M.; Zhao, H.; Hou, L.; Zhao, S.; Li, P.; et al. BSA-seq and genetic mapping reveals AhRt2 as a candidate gene responsible for red testa of peanut. Theor. Appl. Genet. 2022, 135, 1529–1540. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Tian, X.; Wang, Z.; Wei, X.; Zhao, Y.; Su, H.; Zhao, X.; Tian, B.; Yuan, Y.; Zhang, X.W. Fine Mapping and Candidate Gene Identification of a White Flower Gene BrWF3 in Chinese Cabbage (Brassica rapa L. ssp. pekinensis). Front. Plant Sci. 2021, 12, 646222. [Google Scholar] [CrossRef]
- Kaur, G.; Yadav, I.S.; Bhatia, D.; Vikal, Y.; Neelam, K.; Dhillon, N.K.; Praba, U.P.; Mangat, G.S.; Singh, K. BSA-seq Identifies a Major Locus on Chromosome 6 for Root-Knot Nematode (Meloidogyne graminicola) Resistance from Oryza glaberrima. Front. Genet. 2022, 13, 871833. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Imai, R. Mechanisms of cold-induced immunity in plants. Physiol. Plant 2023, 175, e13846. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Tan, C.; Liu, Y.; Chen, X.; Li, X.; Jones, A.; Zhu, Y.; Song, Y. Physiology and Molecular Breeding in Sustaining Wheat Grain Setting and Quality under Spring Cold Stress. Int. J. Mol. Sci. 2022, 23, 14099. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, L.; Sun, Y.; Zheng, S.; Wang, J.; Zhang, T. Hydrogen peroxide is involved in strigolactone induced low temperature stress tolerance in rape seedlings (Brassica rapa L.). Plant Physiol. Biochem. 2020, 157, 402–415. [Google Scholar] [CrossRef]
- Armstrong, J.J.; Takebayashi, N.; Sformo, T.; Wolf, D.E. Cold tolerance in Arabidopsis kamchatica. Am. J. Bot. 2015, 102, 439–448. [Google Scholar] [CrossRef]
- Fang, J.C.; Tsai, Y.C.; Chou, W.L.; Liu, H.Y.; Chang, C.C.; Wu, S.J.; Lu, C.A. A CCR4-associated factor 1, OsCAF1B, confers tolerance of low-temperature stress to rice seedlings. Plant Mol. Biol. 2021, 105, 177–192. [Google Scholar] [CrossRef]
- Liu, Z.; Meng, J.; Sun, Z.; Su, J.; Luo, X.; Song, J.; Li, P.; Sun, Y.; Yu, C.; Peng, X. Zinc application after low temperature stress promoted rice tillers recovery: Aspects of nutrient absorption and plant hormone regulation. Plant Sci. 2022, 314, 111104. [Google Scholar] [CrossRef]
- Sadura, I.; Janeczko, A. Brassinosteroids and the Tolerance of Cereals to Low and High Temperature Stress: Photosynthesis and the Physicochemical Properties of Cell Membranes. Int. J. Mol. Sci. 2021, 23, 342. [Google Scholar] [CrossRef]
- Antonietta, M.; de Felipe, M.; Rothwell, S.A.; Williams, T.B.; Skilleter, P.; Albacete, A.; Borras, L.; Rufino, M.C.; Dodd, I.C. Prolonged low temperature exposure de-sensitises ABA-induced stomatal closure in soybean, involving an ethylene-dependent process. Plant Cell Environ. 2023, 46, 2128–2141. [Google Scholar] [CrossRef] [PubMed]
- Kurakawa, T.; Ueda, N.; Maekawa, M.; Kobayashi, K.; Kojima, M.; Nagato, Y.; Sakakibara, H.; Kyozuka, J. Direct control of shoot meristem activity by a cytokinin-activating enzyme. Nature 2007, 445, 652–655. [Google Scholar] [CrossRef] [PubMed]
- Munnik, T.; Testerink, C. Plant phospholipid signaling: “in a nutshell”. J. Lipid Res. 2009, 50, S260–S265. [Google Scholar] [CrossRef]
- Saidi, Y.; Hearn, T.J.; Coates, J.C. Function and evolution of ‘green’ GSK3/Shaggy-like kinases. Trends Plant Sci. 2012, 17, 39–46. [Google Scholar] [CrossRef]
- Li, B.; Liu, X.; Guo, Y.; Deng, L.; Qu, L.; Yan, M.; Li, M.; Wang, T. BnaC01.BIN2, a GSK3-like kinase, modulates plant height and yield potential in Brassica napus. Theor. Appl. Genet. 2023, 136, 29. [Google Scholar] [CrossRef] [PubMed]
- Legris, M.; Klose, C.; Burgie, E.S.; Rojas, C.C.; Neme, M.; Hiltbrunner, A.; Wigge, P.A.; Schäfer, E.; Vierstra, R.D.; Casal, J.J. Phytochrome B integrates light and temperature signals in Arabidopsis. Science 2016, 354, 897–900. [Google Scholar] [CrossRef] [PubMed]
- Munnik, T. Phosphatidic acid: An emerging plant lipid second messenger. Trends Plant Sci. 2001, 6, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Nokhrina, K.; Ray, H.; Bock, C.; Georges, F. Metabolomic shifts in Brassica napus lines with enhanced BnPLC2 expression impact their response to low temperature stress and plant pathogens. GM Crops Food 2014, 5, 120–131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ma, L.; Xu, J.; Tao, X.; Wu, J.; Wang, W.; Pu, Y.; Yang, G.; Fang, Y.; Liu, L.; Li, X.; et al. Genome-Wide Identification of C2H2 ZFPs and Functional Analysis of BRZAT12 under Low-Temperature Stress in Winter Rapeseed (Brassica rapa). Int. J. Mol. Sci. 2022, 23, 12218. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Zhao, N.; Qin, M.; Xu, A. Mapping of quantitative trait loci related to cold resistance in Brassica napus L. J. Plant Physiol. 2018, 231, 147–154. [Google Scholar] [CrossRef]
- Yang, M.; He, J.; Wan, S.; Li, W.; Chen, W.; Wang, Y.; Jiang, X.; Cheng, P.; Chu, P.; Shen, W.; et al. Fine mapping of the BnaC04.BIL1 gene controlling plant height in Brassica napus L. BMC Plant Biol. 2021, 21, 359. [Google Scholar] [CrossRef]
- Dechaine, J.M.; Brock, M.T.; Weinig, C. QTL architecture of reproductive fitness characters in Brassica rapa. BMC Plant Biol. 2014, 14, 66. [Google Scholar] [CrossRef]
- Kim, J.A.; Moon, H.; Kim, H.S.; Choi, D.; Kim, N.S.; Jang, J.; Lee, S.W.; Baskoro Dwi Nugroho, A.; Kim, D.H. Transcriptome and QTL mapping analyses of major QTL genes controlling glucosinolate contents in vegetable- and oilseed-type Brassica rapa plants. Front. Plant Sci. 2022, 13, 1067508. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Su, T.; Zhang, B.; Li, P.; Xin, X.; Yue, X.; Cao, Y.; Wang, W.; Zhao, X.; Yu, Y.; et al. Identification and fine mapping of qSB.A09, a major QTL that controls shoot branching in Brassica rapa ssp. chinensis Makino. Theor. Appl. Genet. 2020, 133, 1055–1068. [Google Scholar] [CrossRef]
- Li, G.H.; Chen, H.C.; Liu, J.L.; Luo, W.L.; Xie, D.S.; Luo, S.B.; Wu, T.Q.; Akram, W.; Zhong, Y.J. A high-density genetic map developed by specific-locus amplified fragment (SLAF) sequencing and identification of a locus controlling anthocyanin pigmentation in stalk of Zicaitai (Brassica rapa L. ssp. chinensis var. purpurea). BMC Genom. 2019, 20, 343. [Google Scholar] [CrossRef]
- Liu, S.; Wang, R.; Zhang, Z.; Li, Q.; Wang, L.; Wang, Y.; Zhao, Z. High-resolution mapping of quantitative trait loci controlling main floral stalk length in Chinese cabbage (Brassica rapa L. ssp. pekinensis). BMC Genom. 2019, 20, 437. [Google Scholar] [CrossRef]
- Pu, Y.; Liu, L.; Wu, J.; Zhao, Y.; Bai, J.; Ma, L.; Yue, J.; Jin, J.; Niu, Z.; Fang, Y.; et al. Transcriptome Profile Analysis of Winter Rapeseed (Brassica napus L.) in Response to Freezing Stress, Reveal Potentially Connected Events to Freezing Stress. Int. J. Mol. Sci. 2019, 20, 2771. [Google Scholar] [CrossRef]
- Youssef, C.; Aubry, C.; Montrichard, F.; Beucher, D.; Juchaux, M.; Ben, C.; Prosperi, J.M.; Teulat, B. Cell length instead of cell number becomes the predominant factor contributing to hypocotyl length genotypic differences under abiotic stress in Medicago truncatula. Physiol. Plant 2016, 156, 108–124. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Lu, Z.; Wang, L.; Jin, B. Plant Responses to Heat Stress: Physiology, Transcription, Noncoding RNAs, and Epigenetics. Int. J. Mol. Sci. 2020, 22, 117. [Google Scholar] [CrossRef]
- Rinalducci, S.; Egidi, M.G.; Mahfoozi, S.; Godehkahriz, S.J.; Zolla, L. The influence of temperature on plant development in a vernalization-requiring winter wheat: A 2-DE based proteomic investigation. J. Proteom. 2011, 74, 643–659. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Liu, L.; Ma, X. CbADH1 improves plant cold tolerance. Plant Signal. Behav. 2019, 14, 1612680. [Google Scholar] [CrossRef]
- Wang, N.; Wang, K.L.; Liu, Q.H.; Liu, Q.C. Cold resistance of four evergreen broad-leaved tree species. Ying Yong Sheng Tai Xue Bao 2016, 27, 3114–3122. [Google Scholar]
- Li, H.; Ding, Y.; Shi, Y.; Zhang, X.; Zhang, S.; Gong, Z.; Yang, S. MPK3- and MPK6-Mediated ICE1 Phosphorylation Negatively Regulates ICE1 Stability and Freezing Tolerance in Arabidopsis. Dev. Cell 2017, 43, 630–642. [Google Scholar] [CrossRef]
- Müller, M.; Munné-Bosch, S. Rapid and sensitive hormonal profiling of complex plant samples by liquid chromatography coupled to electrospray ionization tandem mass spectrometry. Plant Methods 2011, 7, 37. [Google Scholar] [CrossRef]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, Y.; Shi, C.; Huang, Z.; Zhang, Y.; Li, S.; Li, Y.; Ye, J.; Yu, C.; Li, Z.; et al. SOAPnuke: A MapReduce acceleration-supported software for integrated quality control and preprocessing of high-throughput sequencing data. Gigascience 2018, 7, gix120. [Google Scholar] [CrossRef]
- Farooqi, M.Q.; Sa, K.J.; Hong, T.K.; Lee, J.K. Bulk segregant analysis (BSA) for improving cold stress resistance in maize using SSR markers. Genet. Mol. Res. 2016, 15, gmr15049326. [Google Scholar] [CrossRef] [PubMed]
- Wallqvist, A.; Fukunishi, Y.; Murphy, L.R.; Fadel, A.; Levy, R.M. Iterative sequence/secondary structure search for protein homologs: Comparison with amino acid sequence alignments and application to fold recognition in genome databases. Bioinformatics 2000, 16, 988–1002. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Zhang, T. Construction of customized sub-databases from NCBI-nr database for rapid annotation of huge metagenomic datasets using a combined BLAST and MEGAN approach. PLoS ONE 2013, 8, e59831. [Google Scholar] [CrossRef] [PubMed]
- McMillan, L.E.; Martin, A.C. Automatically extracting functionally equivalent proteins from SwissProt. BMC Bioinform. 2008, 9, 418. [Google Scholar] [CrossRef] [PubMed]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene ontology: Tool for the unification of biology. The Gene Ontology Consortium. Nat. Genet. 2000, 25, 25–29. [Google Scholar] [CrossRef]
- Tatusov, R.L.; Fedorova, N.D.; Jackson, J.D.; Jacobs, A.R.; Kiryutin, B.; Koonin, E.V.; Krylov, D.M.; Mazumder, R.; Mekhedov, S.L.; Nikolskaya, A.N.; et al. The COG database: An updated version includes eukaryotes. BMC Bioinform. 2003, 4, 41. [Google Scholar] [CrossRef] [PubMed]
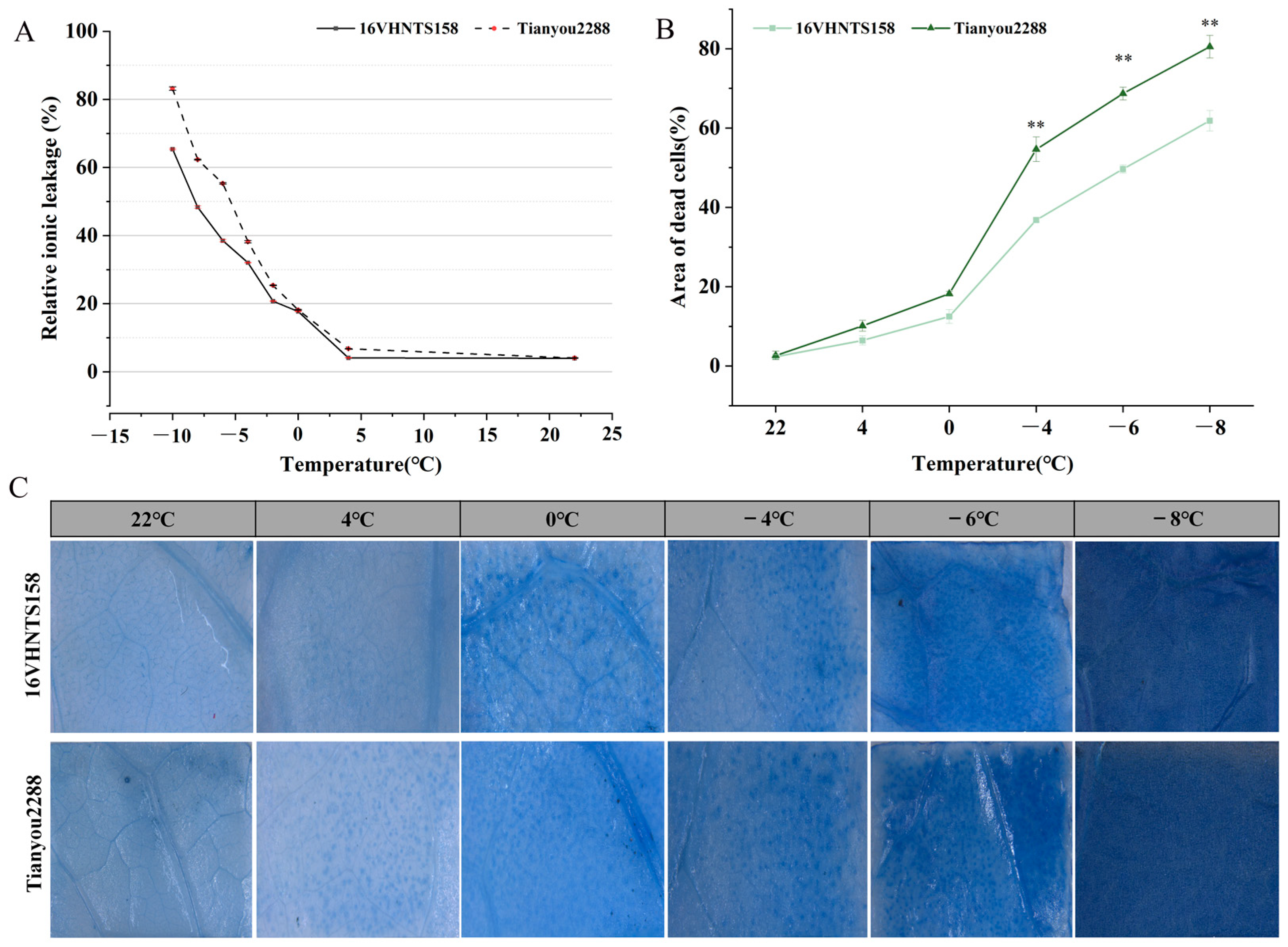
| Variety | Relative Electric Conductivity (%) | Regression Equation | R2 | LT50 (°C) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 4 °C | 0 °C | −2 °C | −4 °C | −6 °C | −8 °C | −10 °C | ||||
| 16VHNTS158 | 4.1 ± 0.1 | 17.7 ± 0.1 | 20.7 ± 0.1 | 32.0 ± 0.1 | 38.5 ± 0.4 | 48.3 ± 0.4 | 65.4 ± 0.2 | Y = 1.8786 − 0.250X | 0.97 | −7.5 |
| Tianyou 2288 | 6.8 ± 0.01 | 18.1 ± 0.1 | 25.8 ± 0.1 | 38.2 ± 0.3 | 55.3 ± 0.2 | 62.3 ± 0.1 | 83.2 ± 0.5 | Y = 1.5499 − 0.288X | 0.99 | −5.4 |
| Population | Average Length (cm) | Max | Min | Range | CV (%) | Kurtosis | Skewness |
|---|---|---|---|---|---|---|---|
| F2 | 0.51 | 1.5 | 0 | 1.5 | 46 | 2.42 | 0.64 |
| 16VHNTS158 | 0.41 | 0.7 | 0 | 0.7 | 37 | 3.02 | −1.1 |
| Tianyou 2288 | 0.77 | 1.2 | 0.4 | 1.2 | 30 | −0.87 | 0.3 |
| Model | AIC | nW2 | Dn | |||
|---|---|---|---|---|---|---|
| 1MG-AD | 263.2167 | 0.128 (0.96) | 0.0784 (0.85) | 0.0704 (0.35) | 0.1123 (0.16) | 0.0646 (0.03) |
| 1MG-A | 269.9081 | 0.0022 (0.93) | 0.0349 (0.86) | 0.8623 (0.69) | 0.2786 (0.32) | 0.1114 (0.15) |
| 1MG-EAD | 272.2328 | 0.0069 (0.70) | 0.0322 (0.72) | 0.1575 (0.97) | 0.1744 (0.14) | 0.0876 (0.03) |
| 1MG-NCD | 290.9928 | 0.1468 (0.87) | 0.1308 (0.85) | 0.0014 (0.16) | 0.302 (0.08) | 0.1105 (0.02) |
| 2MG-ADI | 294.5386 | 0.0281 (0.97) | 0.0347 (0.95) | 1.9439 (0.94) | 0.4003 (0.74) | 0.1198 (0.74) |
| 2MG-AD | 247.2805 | 0.0016 (0.87) | 0.0032 (0.97) | 0.0052 (0.42) | 0.0736 (0.60) | 0.0528 (0.47) |
| 2MG-A | 247.3956 | 0.0287 (0.37) | 0.0014 (0.55) | 0.6458 (0.26) | 0.0998 (0.18) | 0.0653 (0.03) |
| 2MG-EA | 259.385 | 0.808 (0.55) | 0.3494 (0.57) | 1.2482 (0.97) | 0.2654 (0.11) | 0.114 (0.01) |
| 2MG-CD | 294.5331 | 0.3587 (0.55) | 0.325 (0.57) | 0.0015 (0.97) | 0.3373 (0.11) | 0.1223 (0.01) |
| MG-EAD | 292.5332 | 0.3587 (0.55) | 0.325 (0.57) | 0.0015 (0.97) | 0.3372 (0.11) | 0.1223 (0.01) |
| Model | 1MG-AD | 1MG-A | 1MG-EAD | 2MG-AD | 2MG-A | 2MG-EA | |
|---|---|---|---|---|---|---|---|
| First-order genetic parameter | M | 0.78 | 0.98 | 0.83 | 1.31 | 1.00 | 1.04 |
| da | 0.26 | 0.40 | 0.30 | 0.40 | 0.22 | 0.29 | |
| db | - | - | - | 0.46 | 0.44 | - | |
| ha | 0.48 | - | - | −0.37 | - | - | |
| hb | - | - | −0.30 | - | - | ||
| I | - | - | - | - | - | - | |
| jab | - | - | - | - | - | - | |
| jba | - | - | - | - | - | - | |
| L | - | - | - | - | - | - | |
| Second-order genetic parameter | Major-Gene Var | 0.195 | |||||
| Heritability (%) | 59.54 | ||||||
| Name | Mapping Reads | Mapping Ratio (%) | Ave Depth | Properly Paired Ratio (%) | Q20% | Q30% | GC (%) |
|---|---|---|---|---|---|---|---|
| 16VHNTS158 | 141,541,608 | 99.15 | 23 | 88.85 | 97.65 | 93.42 | 36.51 |
| Tianyou 2288 | 146,156,508 | 98.97 | 23 | 88.39 | 97.17 | 92.38 | 36.69 |
| S-pool | 199,081,960 | 99.47 | 32 | 92 | 97.38 | 93.44 | 37.35 |
| L-pool | 203,088,138 | 99.22 | 33 | 90.76 | 97.99 | 92.21 | 37.34 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, M.; Hu, F.; Liu, L.; Lu, X.; Li, R.; Wang, J.; Wu, J.; Ma, L.; Pu, Y.; Fang, Y.; et al. Physiological Analysis and Genetic Mapping of Short Hypocotyl Trait in Brassica napus L. Int. J. Mol. Sci. 2023, 24, 15409. https://doi.org/10.3390/ijms242015409
Liu M, Hu F, Liu L, Lu X, Li R, Wang J, Wu J, Ma L, Pu Y, Fang Y, et al. Physiological Analysis and Genetic Mapping of Short Hypocotyl Trait in Brassica napus L. International Journal of Molecular Sciences. 2023; 24(20):15409. https://doi.org/10.3390/ijms242015409
Chicago/Turabian StyleLiu, Miaomiao, Fangdi Hu, Lijun Liu, Xiaoming Lu, Rong Li, Jingyu Wang, Junyan Wu, Li Ma, Yuanyuan Pu, Yan Fang, and et al. 2023. "Physiological Analysis and Genetic Mapping of Short Hypocotyl Trait in Brassica napus L." International Journal of Molecular Sciences 24, no. 20: 15409. https://doi.org/10.3390/ijms242015409
APA StyleLiu, M., Hu, F., Liu, L., Lu, X., Li, R., Wang, J., Wu, J., Ma, L., Pu, Y., Fang, Y., Yang, G., Wang, W., & Sun, W. (2023). Physiological Analysis and Genetic Mapping of Short Hypocotyl Trait in Brassica napus L. International Journal of Molecular Sciences, 24(20), 15409. https://doi.org/10.3390/ijms242015409






