Dual Implications of Nanosilver-Induced Autophagy: Nanotoxicity and Anti-Cancer Effects
Abstract
:1. Introduction
2. The Autophagy Process and Its Importance for the Cell
The Role of Autophagy in Cancerous Process
3. Nanosilver-Induced Autophagy—Toxic or Protective Role?
3.1. Ultrastructural and Molecular Characteristics Indicative of Autophagy Induction by AgNPs In Vitro and In Vivo
3.2. Interference of AgNPs with Autophagic Flux in Different Models of Exposure
3.3. Evidence for Pro-Death and Pro-Survival Profiles of AgNP-Induced Autophagy in Normal Cells In Vitro and In Vivo
3.4. Evidence for Pro-Death and Pro-Survival Profiles of AgNP-Induced Autophagy in Cancer Cells In Vitro and In Vivo
3.5. AgNP-Related Factors of Potential Importance in Determining the Autophagy-Induced Cell Fate
3.6. Potential Medical Implementation of AgNP-Induced Autophagy in Cancer Therapy—Unanswered Questions
4. Concluding Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mao, B.-H.; Tsai, J.-C.; Chen, C.-W.; Yan, S.-J.; Wang, Y.-J. Mechanisms of silver nanoparticle-induced toxicity and important role of autophagy. Nanotoxicology 2016, 10, 1021–1040. [Google Scholar] [CrossRef] [PubMed]
- Strużyńska, L.; Skalska, J. Mechanisms underlying neurotoxicity of silver nanoparticles. In Cellular and Molecular Toxicology of Nanoparticles; Saquib, Q., Faisal, M., Al-Khedhairy, A.A., Alatar, A.A., Eds.; Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2018; Volume 1048, pp. 227–250. [Google Scholar] [CrossRef]
- Feng, X.; Zhang, Y.; Zhang, C.; Lai, X.; Zhang, Y.; Wu, J.; Hu, C.; Shao, L. Nanomaterial-mediated autophagy: Coexisting hazard and health benefits in biomedicine. Part. Fibre Toxicol. 2020, 17, 53. [Google Scholar] [CrossRef] [PubMed]
- Glick, D.; Barth, S.; Kay, F.M. Autophagy: Cellular and molecular mechanisms. J. Pathol. 2010, 221, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.-J.; Chen, Y.-Y.; Liao, M.-Y.; Lee, Y.-H.; Chen, Z.-Y.; Yan, S.-J.; Yeh, Y.-L.; Yang, L.-X.; Lee, Y.-L.; Wu, Y.-H.; et al. The current understanding of autophagy in nanomaterial toxicity and its implementation in safety assessment-related alternative testing strategies. Int. J. Mol. Sci. 2020, 21, 2387. [Google Scholar] [CrossRef]
- Mishra, A.R.; Zheng, J.; Tang, X.; Goering, P.L. Silver nanoparticle-induced autophagic-Lysosomal disruption and NLRP3-inflammasome activation in HepG2 cells is size-dependent. Toxicol. Sci. 2016, 150, 473–487. [Google Scholar] [CrossRef]
- Li, L.; Li, L.; Zhou, X.; Yu, Y.; Li, Z.; Zuo, D.; Wu, Y. Silver nanoparticles induce protective autophagy via Ca2+/CaMKKβ/AMPK/mTOR pathway in SH-SY5Y cells and rat brains. Nanotoxicology 2019, 13, 369–391. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, M.; Zhang, T.; Du, E.; Liu, Y.; Qi, S.; Xu, Y.; Zhang, Z. Autophagic effects and mechanisms of silver nanoparticles in renal cells under low dose exposure. Ecotoxicol. Environ. Saf. 2018, 166, 71–77. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, T.; Chen, S.; Qi, S.; Zhang, Z.; Xu, Y. Silver nanoparticles regulate autophagy through lysosome injury and cell hypoxia in prostate cancer cells. Biochem. Mol. Toxicol. 2020, 34, e22474. [Google Scholar] [CrossRef]
- Chang, X.; Wang, X.; Li, J.; Shang, M.; Niu, S.; Zhang, W.; Li, Y.; Sun, Z.; Gan, J.; Li, W.; et al. Silver nanoparticles induced cytotoxicity in HT22 cells through autophagy and apoptosis via PI3K/AKT/mTOR signaling pathway. Ecotoxicol. Environ. Saf. 2021, 208, 111696. [Google Scholar] [CrossRef]
- Zielinska, E.; Zauszkiewicz-Pawlak, A.; Wojcik, M.; Inkielewicz-Stepniak, I. Silver nanoparticles of different sizes induce a mixed type of programmed cell death in human pancreatic ductal adenocarcinoma. Oncotarget 2018, 9, 4675–4697. [Google Scholar] [CrossRef]
- Fageria, L.; Bambroo, V.; Mathew, A.; Mukherjee, S.; Chowdhury, R.; Pande, S. Functional autophagic flux regulates AgNP uptake and the internalized nanoparticles determine tumor cell fate by temporally regulating flux. Int. J. Nanomed. 2019, 14, 9063–9076. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.A.; Alanazi, A.; Alsaif, N.; Al-anazi, M.; Sayed, A.Y.A.; Bhat, M.A. Potential cytotoxicity of silver nanoparticles: Stimulation of autophagy and mitochondrial dysfunction in cardiac cells. Saudi J. Biol. Sci. 2021, 28, 2762–2771. [Google Scholar] [CrossRef] [PubMed]
- Buttacavoli, M.; Albanese, N.N.; Di Cara, G.; Alduina, R.; Faleri, C.; Gallo, M.; Pizzolanti, G.; Gallo, G.; Feo, S.; Baldi, F.; et al. Anticancer activity of biogenerated silver nanoparticles: An integrated proteomic investigation. Oncotarget 2018, 9, 9685–9705. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.K.; Gurunathan, S.; Kang, M.H.; Han, J.W.; Das, J.; Choi, Y.J.; Kwon, D.N.; Cho, S.G.; Park, C.; Seo, H.G.; et al. Hypoxia-mediated autophagic flux inhibits silver nanoparticle triggered apoptosis in human lung cancer cells. Sci. Rep. 2016, 6, 21688. [Google Scholar] [CrossRef]
- Lee, T.Y.; Liu, M.S.; Huang, L.J.; Lue, S.I.; Lin, L.C.; Kwan, A.L.; Yang, R.C. Bioenergetic failure correlates with autophagy and apoptosis in rat liver following silver nanoparticle intraperitoneal administration. Part. Fibre Toxicol. 2013, 10, 40. [Google Scholar] [CrossRef]
- Skalska, J.; Dabrowska-Bouta, B.; Frontczak-Baniewicz, M.; Sulkowski, G.; Struzynska, L. A low dose of nanoparticulate silver induces mitochondrial dysfunction and autophagy in adult rat brain. Neurotox. Res. 2020, 38, 650–664. [Google Scholar] [CrossRef]
- Dabrowska-Bouta, B.; Sulkowski, G.; Gewartowska, M.; Struzynska, L. Endoplasmic reticulum stress underlies nanosilver-induced neurotoxicity in immature rat brain. Int. J. Mol. Sci. 2022, 23, 13013. [Google Scholar] [CrossRef]
- Ravikumar, B.; Sarkar, S.; Davies, J.E.; Futter, M.; Garcia-Arencibia, M.; Green-Thompson, Z.W.; Jimenez-Sanchez, M.; Korolchuk, V.I.; Lichtenberg, M.; Luo, S.; et al. Regulation of mammalian autophagy in physiology and pathophysiology. Physiol. Rev. 2010, 90, 1383–1435. [Google Scholar] [CrossRef]
- Mulcahy Levy, J.M.; Thorburn, A. Autophagy in cancer: Moving from understanding mechanism to improving therapy responses in patients. Cell Death Differ. 2020, 27, 843–857. [Google Scholar] [CrossRef]
- Rosenfeldt, M.T.; Ryan, K.M. The multiple roles of autophagy in cancer. Carcinogenesis 2011, 32, 955–963. [Google Scholar] [CrossRef]
- Lim, S.M.; Mohamad Hanif, E.A.; Chin, S.-F. Is targeting autophagy mechanism in cancer a good approach? The possible double-edge sword effect. Cell Biosci. 2021, 11, 56. [Google Scholar] [CrossRef] [PubMed]
- Cordani, M.; Somoza, Á. Targeting autophagy using metallic nanoparticles: A promising strategy for cancer treatment. Cell. Mol. Life Sci. 2019, 76, 1215–1242. [Google Scholar] [CrossRef] [PubMed]
- Kovács, D.; Igaz, N.; Gopisetty, M.K.; Kiricsi, M. Cancer therapy by silver nanoparticles: Fiction or reality? Int. J. Mol. Sci. 2022, 23, 839. [Google Scholar] [CrossRef]
- Kroemer, G.; Mariño, G.; Levine, B. Autophagy and the integrated stress response. Mol. Cell. 2010, 40, 280–293. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Klionsky, D.J. Autophagosome formation: Core machinery and adaptations. Nat. Cell Biol. 2007, 9, 1102–1109. [Google Scholar] [CrossRef] [PubMed]
- Menzies, F.M.; Fleming, A.; Caricasole, A.; Bento, C.F.; Andrews, S.P.; Ashkenazi, A.; Füllgrabe, J.; Jackson, A.; Jimenez Sanchez, M.; Karabiyik, C.; et al. Autophagy and neurodegeneration: Pathogenic mechanisms and therapeutic opportunities. Neuron 2017, 93, 1015–1034. [Google Scholar] [CrossRef]
- Lamark, T.; Johansen, T. Mechanisms of Selective Autophagy. Annu. Rev. Cell Dev. Biol. 2021, 37, 143–169. [Google Scholar] [CrossRef]
- Yorimitsu, T.; Nair, U.; Yang, Z.; Klionsky, D.J. Endoplasmic reticulum stress triggers autophagy. J. Biol. Chem. 2006, 281, 30299–30304. [Google Scholar] [CrossRef]
- Lebeau, J.; Saunders, J.M.; Moraes, V.W.R.; Madhavan, A.; Madrazo, N.; Anthony, M.C.; Wiseman, R.L. The PERK arm of the unfolded protein response regulates mitochondrial morphology during acute endoplasmic reticulum stress. Cell Rep. 2018, 22, 2827–2836. [Google Scholar] [CrossRef]
- Kabir, M.F.; Kim, H.-R.; Chae, H.-J. Endoplasmic reticulum stress and autophagy. In Endoplasmic Reticulum; Català, A., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Maiuri, M.C.; Zalckvar, E.; Kimchi, A.; Kroemer, G. Self-eating and self-killing: Crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell Biol. 2007, 8, 741–752. [Google Scholar] [CrossRef]
- Denton, D.; Kumar, S. Autophagy-dependent cell death. Cell Death Differ. 2019, 26, 605–616. [Google Scholar] [CrossRef]
- Rubinsztein, D.C.; Codogno, P.; Levine, B. Autophagy modulation as a potential therapeutic target for diverse diseases. Nat. Rev. Drug Discov. 2012, 11, 709–730. [Google Scholar] [CrossRef]
- Giordano, S.; Dodson, M.; Ravi, S.; Redmann, M.; Ouyang, X.; Darley Usmar, V.M.; Zhang, J. Bioenergetic adaptation in response to autophagy regulators during rotenone exposure. J. Neurochem. 2014, 131, 625–633. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; White, E. Role of autophagy in cancer prevention. Cancer Prev. Res. 2011, 4, 973–983. [Google Scholar] [CrossRef]
- Tan, Q.; Wang, M.; Yu, M.; Zhang, J.; Bristow, R.G.; Hill, R.P.; Tannock, J.F. Role of autophagy as a survival mechanism for hypoxic cells in tumors. Neoplasia 2016, 18, 347–355. [Google Scholar] [CrossRef]
- White, E. Deconvoluting the context-dependent role for autophagy in cancer. Nat. Rev. Cancer 2012, 12, 401–410. [Google Scholar] [CrossRef]
- Guo, J.Y.; Chen, H.-Y.; Mathew, R.; Fan, J.; Strohecker, A.M.; Karsli-Uzunbas, G.; Kamphorst, J.J.; Chen, G.; Lemons, J.M.S.; Karantza, V.; et al. Activated Ras requires autophagy to maintain oxidative metabolism and tumorigenesis. Genes Dev. 2011, 25, 460–470. [Google Scholar] [CrossRef]
- Marinković, M.; Šprung, M.; Buljubašić, M.; Novak, I. Autophagy modulation in cancer: Current knowledge on action and therapy. Oxidative Med. Cell. Longev. 2018, 2018, 8023821. [Google Scholar] [CrossRef]
- Bhutia, S.K.; Mukhopadhyay, S.; Sinha, N.; Nandini Das, D.; Panda, P.K.; Patra, S.K.; Maiti, T.K.; Mandal, M.; Dent, P.; Wang, X.-Y.; et al. Autophagy: Cancer’s friend or foe? Adv. Cancer Res. 2013, 118, 61–95. [Google Scholar] [CrossRef] [PubMed]
- Fung, C.; Lock, R.; Gao, S.; Salas, E.; Debnath, J. Induction of autophagy during extracellular matrix detachment promotes cell survival. Mol. Biol. Cell 2008, 19, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Debnath, J.; Gammoh, N.; Ryan, K.M. Autophagy and autophagy-related pathways in cancer. Nat. Rev. Mol. Cell Biol. 2023, 24, 560–575. [Google Scholar] [CrossRef]
- Pan, H.; Cai, N.; Li, M.; Liu, G.H.; Izpisua Belmonte, J.C. Autophagic control of cell ‘stemness’. EMBO Mol. Med. 2013, 5, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Ju, D. The role of autophagy in nanoparticles-induced toxicity and its related cellular and molecular mechanisms. Adv. Exp. Med. Biol. 2018, 1048, 71–84. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, L.; Bai, R.; Zhang, T.; Chen, C. Silver nanoparticles impede phorbol myristate acetate-induced monocyte-macrophage differentiation and autophagy. Nanoscale 2015, 7, 16100–16109. [Google Scholar] [CrossRef] [PubMed]
- Simard, J.-C.; Vallieres, F.; de Liz, R.; Lavastre, V.; Girard, D. Silver nanoparticles induce degradation of the endoplasmic reticulum stress sensor activating transcription factor-6 leading to activation of the NLRP-3 inflammasome. J. Biol. Chem. 2015, 290, 5926–5939. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-H.; Cheng, F.-Y.; Chiu, H.-W.; Tsai, J.-C.; Fang, C.-Y.; Chen, C.-W.; Wang, Y.-J. Cytotoxicity, oxidative stress, apoptosis and the autophagic effects of silver nanoparticles in mouse embryonic fibroblasts. Biomaterials 2014, 35, 4706–4715. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Zhao, L.; Tang, H.; He, X.; Ye, G.; Shi, F.; Kang, M.; Chen, H.; Li, Y. Silver Nanoparticles Induced Oxidative Stress and Mitochondrial Injuries Mediated Autophagy in HC11 Cells Through Akt/AMPK/mTOR Pathway. Biol. Trace Elem. Res. 2021, 199, 1062–1073. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Huang, Z.; Wu, H.; Zhou, W.; Jin, P.; Wei, P.; Zhang, Y.; Zheng, F.; Zhang, J.; Xu, J.; et al. Inhibition of autophagy enhances the anticancer activity of silver nanoparticles. Autophagy 2014, 10, 2006–2020. [Google Scholar] [CrossRef]
- Fageria, L.; Pareek, V.; Dilip, R.V.; Bhargava, A.; Pasha, S.S.; Laskar, I.R.; Saini, H.; Dash, S.; Chowdhury, R.; Panwar, J. Biosynthesized protein-capped silver nanoparticles induce ROS-dependent proapoptotic signals and prosurvival autophagy in cancer cells. ACS Omega 2017, 2, 1489–1504. [Google Scholar] [CrossRef]
- Miyayama, T.; Fujiki, K.; Matsuoka, M. Silver nanoparticles induce lysosomal-autophagic defects and decreased expression of transcription factor EB in A549 human lung adenocarcinoma cells. Toxicol. In Vitro 2018, 46, 148–154. [Google Scholar] [CrossRef]
- Bao, J.; Jiang, Z.; Ding, W.; Cao, Y.; Yang, L.; Liu, J. Silver nanoparticles induce mitochondria-dependent apoptosis and late non-canonical autophagy in HT-29 colon cancer cells. Nanotechnol. Rev. 2022, 11, 1911–1926. [Google Scholar] [CrossRef]
- Gomes, L.C.; Di Benedetto, G.; Scorrano, L. During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat. Cell Biol. 2011, 13, 589–598. [Google Scholar] [CrossRef]
- Youle, R.J.; Narendra, D.P. Mechanisms of mitophagy. Nat. Rev. Mol. Cell Biol. 2011, 12, 9–14. [Google Scholar] [CrossRef]
- Kabeya, Y.; Mizushima, N.; Ueno, T.; Yamamoto, A.; Kirisako, T.; Noda, T.; Kominami, E.; Ohsumi, Y.; Yoshimori, T. LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J. 2000, 19, 5720–5728. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Xu, B.; Yao, M.; Mao, Z.; Zhang, Y.; Xu, G.; Tang, Q.; Wang, X.; Xia, Y. Graphene oxide quantum dots disrupt autophagic flux by inhibiting lysosome activity in GC-2 and TM4 cell lines. Toxicology 2016, 374, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yu, Y.; Lu, K.; Yang, M.; Li, Y.; Zhou, X.; Sun, Z. Silica nanoparticles induce autophagy dysfunction via lysosomal impairment and inhibition of autophagosome degradation in hepatocytes. Int. J. Nanomed. 2017, 12, 809–825. [Google Scholar] [CrossRef] [PubMed]
- Foghsgaard, L.; Wissing, D.; Mauch, D.; Lademann, U.; Bastholm, L.; Boes, M.; Elling, F.; Leist, M.; Jäättelä, M. Cathepsin B acts as a dominant execution protease in tumor cell apoptosis induced by tumor necrosis factor. J. Cell Biol. 2001, 153, 999–1010. [Google Scholar] [CrossRef]
- Uzhytchak, M.; Smolková, B.; Lunova, M.; Frtús, A.; Jirsa, M.; Dejneka, A.; Lunov, O. Lysosomal nanotoxicity: Impact of nanomedicines on lysosomal function. Adv. Drug Deliv. Rev. 2023, 197, 114828. [Google Scholar] [CrossRef]
- Yang, E.J.; Kim, S.; Kim, J.S.; Choi, I.H. Inflammasome formation and IL-1b release by human blood monocytes in response to silver nanoparticles. Biomaterials 2012, 33, 6858–6867. [Google Scholar] [CrossRef] [PubMed]
- Bjørkøy, G.; Lamark, T.; Pankiv, S.; Øvervatn, A.; Brech, A.; Johansen, T. Monitoring autophagic degradation of p62/SQSTM1. Methods Enzymol. 2009, 452, 181–197. [Google Scholar] [CrossRef]
- Gutierrez, M.G.; Munafó, D.B.; Berón, W.; Colombo, M.I. Rab7 is required for the normal progression of the autophagic pathway in mammalian cells. J. Cell Sci. 2004, 117, 2687–2697. [Google Scholar] [CrossRef] [PubMed]
- Shintani, T.; Klionsky, D.J. Autophagy in health and disease: A double-edged sword. Science 2004, 306, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Ziemińska, E.; Stafiej, A.; Strużyńska, L. The role of the glutamatergic NMDA receptor in nanosilver-evoked neurotoxicity in primary cultures of cerebellar granule cells. Toxicology 2014, 315, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Skalska, J.; Strużyńska, L. Toxic effects of silver nanoparticles in mammals—Does a risk of neurotoxicity exist? Folia Neuropathol. 2015, 53, 281–300. [Google Scholar] [CrossRef] [PubMed]
- Huo, L.; Chen, R.; Zhao, L.; Shi, X.; Bai, R.; Long, D.; Chen, F.; Zhao, Y.; Chang, Y.-Z.; Chen, C. Silver nanoparticles activate endoplasmic reticulum stress signaling pathway in cell and mouse models: The role in toxicity evaluation. Biomaterials 2015, 61, 307–315. [Google Scholar] [CrossRef]
- Zhang, R.; Piao, M.J.; Kim, K.C.; Kim, A.D.; Choi, J.Y.; Choi, J.; Hyun, J.W. Endoplasmic reticulum stress signaling is involved in silver nanoparticles-induced apoptosis. Int. J. Biochem. Cell Biol. 2012, 44, 224–232. [Google Scholar] [CrossRef]
- Skalska, J.; Frontczak-Baniewicz, M.; Struzynska, L. Synaptic degeneration in rat brain after prolonged oral exposure to silver nanoparticles. Neurotoxicology 2015, 46, 145–154. [Google Scholar] [CrossRef]
- Shutt, T.E.; McBride, H.M. Staying cool in difficult times: Mitochondrial dynamics, quality control and the stress response. Biochim. Biophys. Acta 2013, 1833, 417–424. [Google Scholar] [CrossRef]
- Yuan, Y.G.; Gurunathan, S. Combination of graphene oxide-silver nanoparticle nanocomposites and cisplatin enhances apoptosis and autophagy in human cervical cancer cells. Int. J. Nanomed. 2017, 12, 6537–6558. [Google Scholar] [CrossRef]
- Zhang, X.-F.; Gurunathan, S. Combination of salinomycin and silver nanoparticles enhances apoptosis and autophagy in human ovarian cancer cells: An effective anticancer therapy. Int. J. Nanomed. 2016, 11, 3655–3675. [Google Scholar] [CrossRef]
- Lin, J.; Liu, Y.; Wu, H.; Huang, Z.; Ma, J.; Guo, C.; Gao, F.; Jin, P.; Wei, P.; Zhang, Y.; et al. Key role of TFEB nucleus translocation for silver nanoparticle-induced cytoprotective autophagy. Small 2018, 14, e1703711. [Google Scholar] [CrossRef] [PubMed]
- Amaravadi, R.K.; Yu, D.; Lum, J.J.; Bui, T.; Christophorou, M.A.; Evan, G.I.; Thomas-Tikhonenko, A.; Thompson, C.B. Autophagy inhibition enhances therapy-induced apoptosis in a Myc-induced model of lymphoma. J. Clin. Investig. 2007, 117, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Kalyane, D.; Raval, N.; Maheshwari, R.; Tambe, V.; Kalia, K.; Tekade, R.K. Employment of enhanced permeability and retention effect (EPR): Nanoparticle-based precision tools for targeting of therapeutic and diagnostic agent in cancer. Mater. Sci. Eng. 2019, 98, 1252–1276. [Google Scholar] [CrossRef]
- Tran, S.; DeGiovanni, P.-J.; Piel, B.; Rai, P. Cancer nanomedicine: A review of recent success in drug delivery. Clin. Transl. Med. 2017, 6, e44. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Du, Z.; Ma, S.; Liu, Y.; Li, D.; Huang, H.; Jiang, S.; Cheng, S.; Wu, W.; Zhang, K.; et al. Effects of green-synthesized silver nanoparticles on lung cancer cells in vitro and grown as xenograft tumors in vivo. Int. J. Nanomed. 2016, 11, 1879–1887. [Google Scholar] [CrossRef]
- Dziedzic, A.; Kubina, R.; Bułdak, R.J.; Skonieczna, M.; Cholewa, K. Silver nanoparticles exhibit the dose-dependent anti-proliferative effect against human squamous carcinoma cells attenuated in the presence of berberine. Molecules 2016, 21, 365. [Google Scholar] [CrossRef]
- Loutfy, S.A.; Al-Ansary, N.A.; Abdel-Ghani, N.T.; Hamed, A.R.; Mohamed, M.B.; Craik, J.D.; Eldin, T.A.; Abdellah, A.M.; Hussein, Y.; Hasanin, M.T.; et al. Anti-proliferative activities of metallic nanoparticles in an in vitro breast cancer model. Asian Pac. J. Cancer Prev. 2015, 16, 6039–6046. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, J.-M. Altered energy metabolism in cancer. A unique opportunity for therapeutic intervention. Cancer Biol. Ther. 2013, 14, 81–89. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Fulda, S. Evasion of Apoptosis as a Cellular Stress Response in Cancer. Int. Cell Biol. 2010, 2010, 370835. [Google Scholar] [CrossRef]
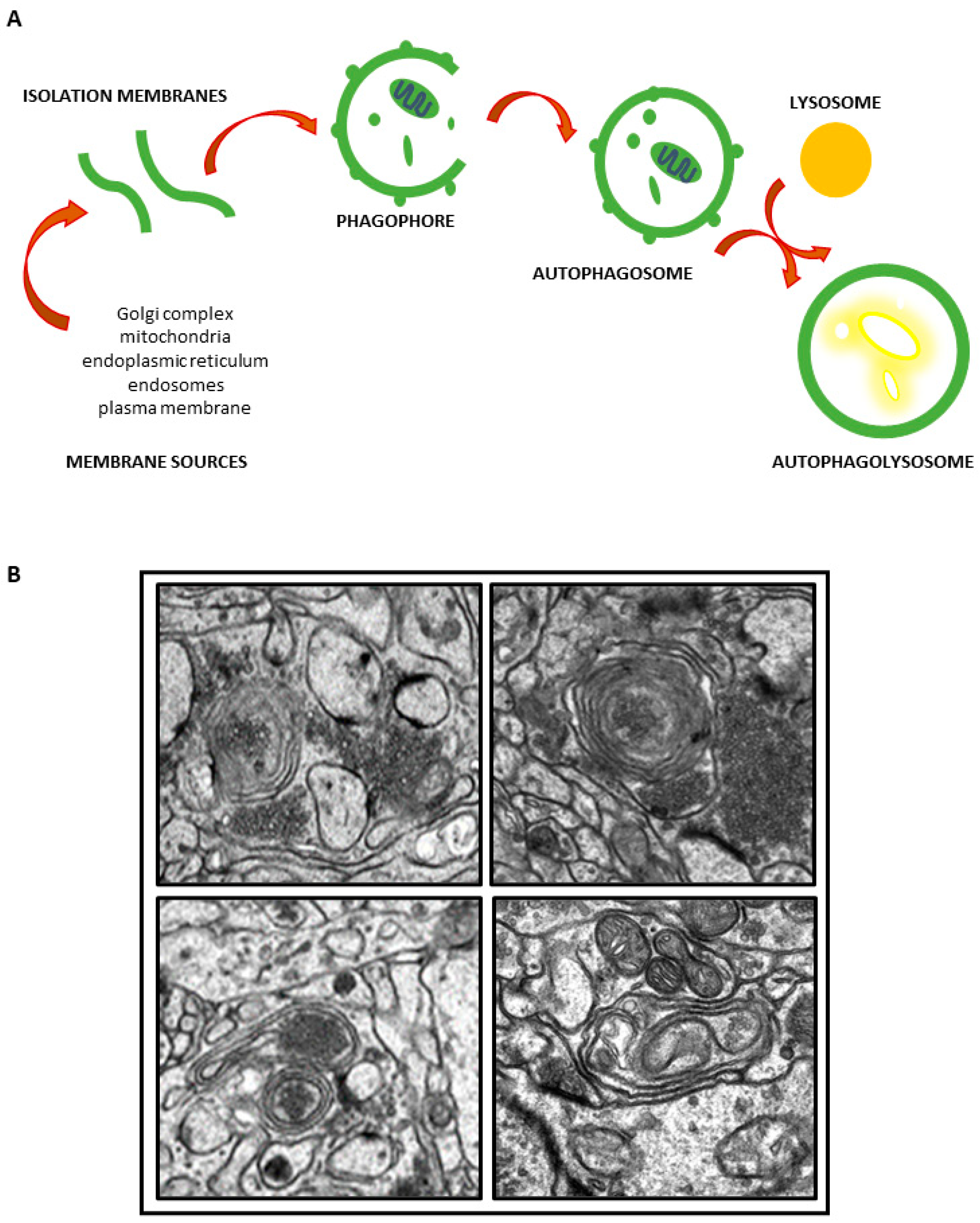
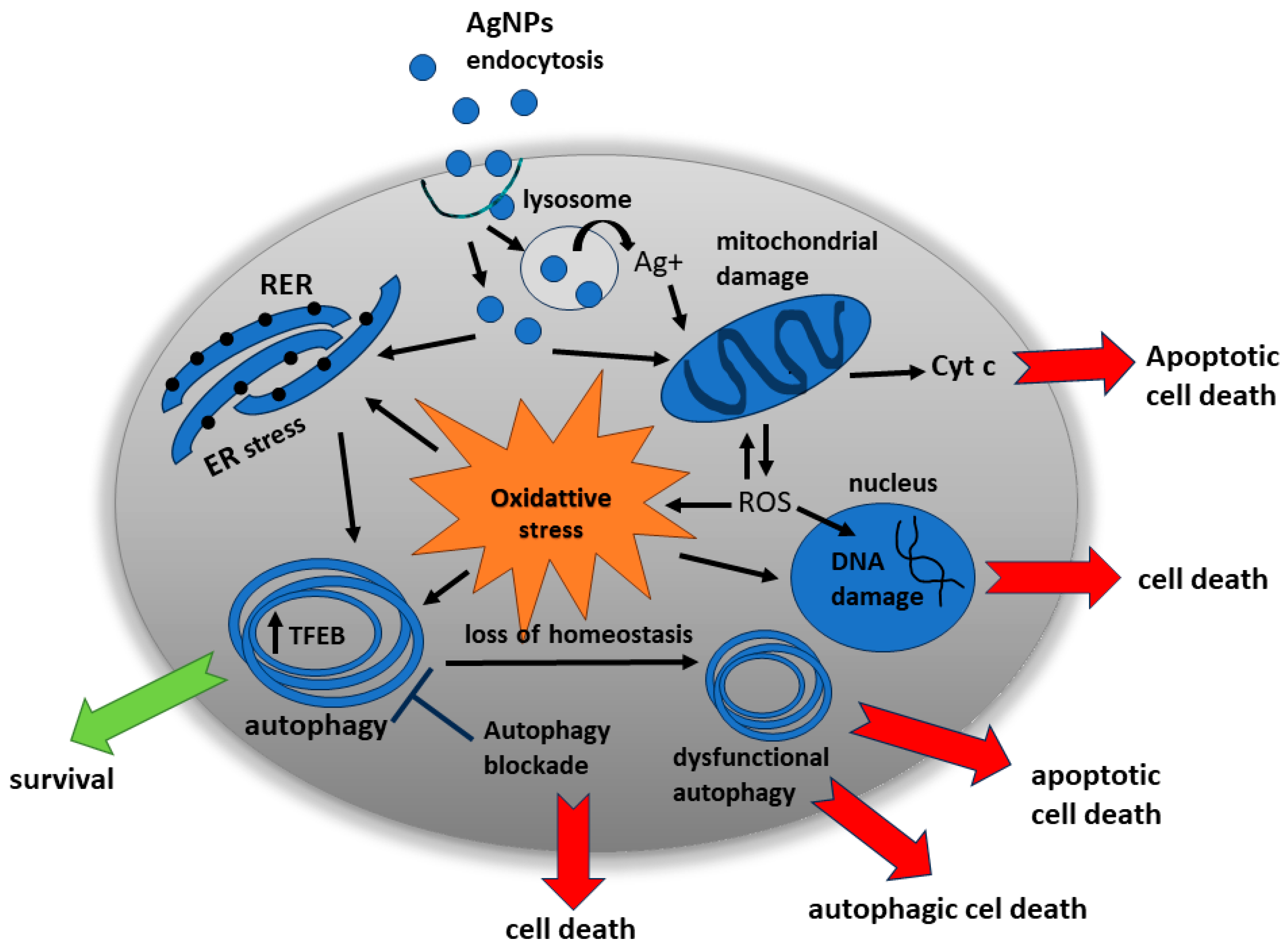
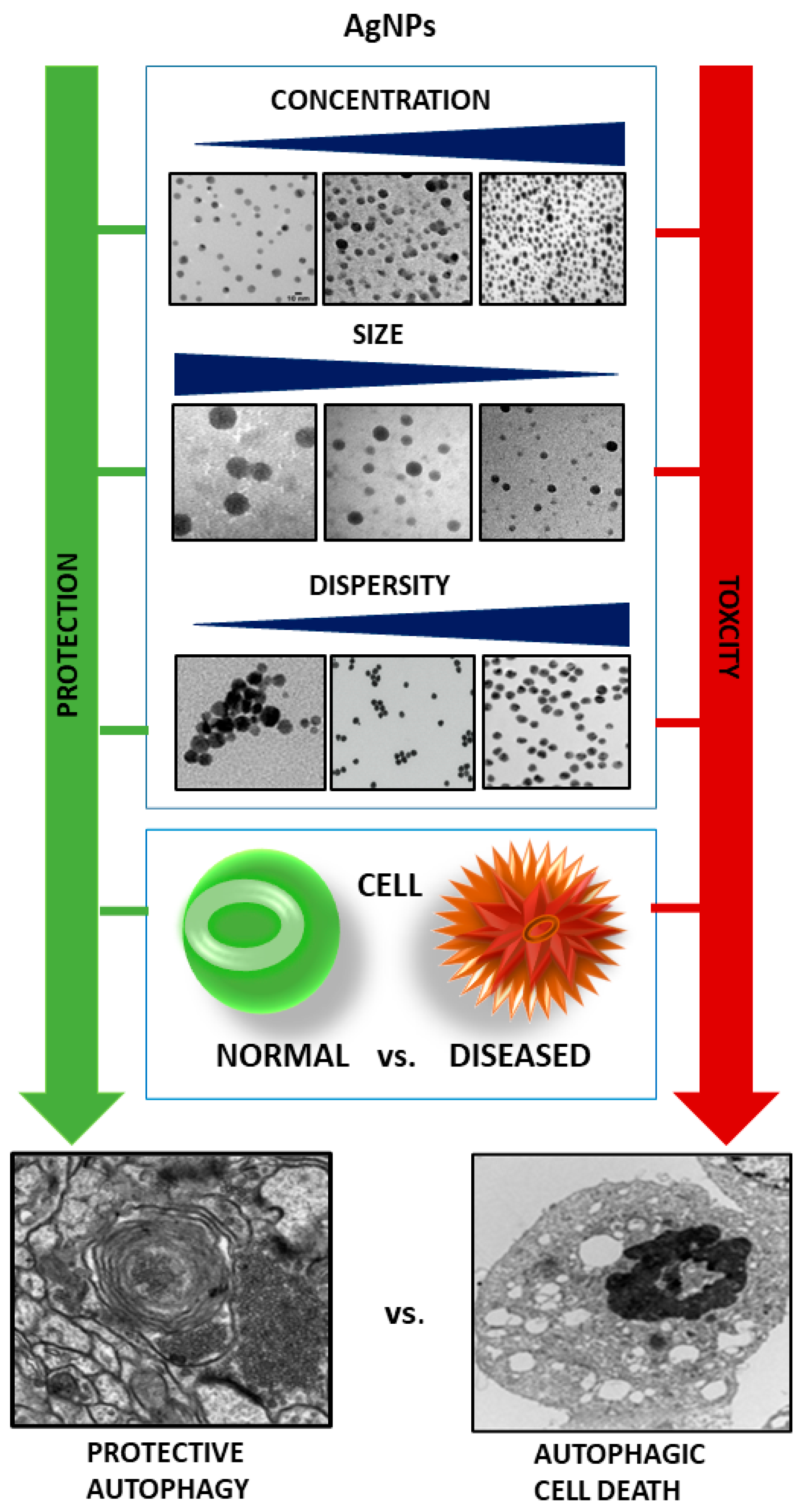
| Type of Cell/Tissue | AgNPs | Autophagy Markers | Autophagy Blockade | Cell Death/Apoptosis Markers | Ref. | |
|---|---|---|---|---|---|---|
| Diameter | Concentration/Dose | |||||
| IN VITRO | ||||||
| Human monocytes (THP-1) | 30 nm | 5, 10 µg/mL | ↑LC3-II Autophagosomes | Yes ↑p62 | N/A | [46] |
| Human monocytes (THP-1) | 15 nm | (a) 1–5 µg/mL (b) 25 µg/mL | ER stress (a) Atypical ER stress (b) | N/A | Absent (a) Pyroptosis caspase-1 and IL-1β activation (b) | [47] |
| Mouse hippocampal neuronal cell line (HT22) | 20 nm | 25, 50, 100 μg/mL | Autophagosomes ↑LC3 II/I | No ↓p62 | ↑Caspase-3; ↑Bax ↓Bcl-2 | [10] |
| Human embryonic kidney cell (HEK293T) | 25 nm (PVP-coated) | 2, 4, 6 μg/mL | ↑LC3II; impaired lysosome integrity and protease activity | Yes ↑p62 | N/A | [8] |
| Mouse embryonic fibroblasts (NIH 3T3) | 26 ± 7.6 nm | 2, 5, 10, 15 μg/mL | Autophagosomes, AVOs, ↑LC3-II (all conc. after 18 h) | Yes ↑p62 | Nuclear fragmentation; apoptotic cells ↑PARP and ↑caspase-3 (all conc. after 24 h) | [48] |
| Rat cardiomyoblasts (H9c2) | 4–12 nm | 3.5 μg/mL | ↑Atg5; ↑Beclin1; ↑LC3BII | N/A | Apoptosis (↑DNA fragmentation) | [13] |
| Mouse mammary epithelial cell line (HC11) | <100 nm | (a) 6.25, 12.5, (b) 25, 50 μg/mL | Autophagosomes ↑LC3-II/I and ↑Beclin-1 ↓MMP | No ↓p62 | Cell death (MTT) (b) | [49] |
| IN VIVO | ||||||
| Rat brain, neurons | 30 nm | 32, 80, or 200 mg/kg | ↑LC3-II/LC3-I; ↑pULK; ↑beclin-1 (all conc.) | No ↓p62 | Ultrastructural changes; ↑caspase-3; ↑caspase-9; ↑PARP (all conc.) | [7] |
| Rat liver | 10–30 nm | 500 mg/kg b.w. | Autophagic structures ↑LC3-II | N/A | Apoptosis ↑caspase-3; ↑TUNEL-positive cells | [16] |
| Neurons; adult rat brain | 10 ± 4 | 0.2 mg/kg b.w. | Mitochondrial elongation; autophagic structures; ↑LC3-II; ↑beclin-1 | Yes, lysosomal proteins cathepsin B and Rab7 do not increase | Morphologically not observed, ↓Bax/Bcl-2 | [17] |
| Neurons; immature rat brain | 10 ± 4 | 0.2 mg/kg b.w. | Mitochondrial elongation; autophagic structures | N/A | Morphologically not observed | [18] |
| Type of Cancer Cell Line | AgNPs | Autophagy Markers | Autophagy Blockade/Defect | Cell Death/Apoptosis Markers | Ref. | |
|---|---|---|---|---|---|---|
| Diameter (nm) | Concentration | |||||
| IN VITRO | ||||||
| Human liver cancer cells HepG2 | 10, 50, 100 nm | (a) 1 µg/mL (b) 10, 25, 50 µg/mL | ↑Lysosomal activity | N/A | (a) No (b) ↑Caspase-3 activity | [6] |
| Human pancreatic ductal adenocarcinoma (PANC-1) | 2.6 nm 18 nm | 0.5–3.5 μg/mL (a) 5 μg/mL (b) 10–50 μg/mL | ↑LC3-II Autophagosomes Autophagolysosomes ↑LC3-II | N/A | Morphologically—apoptosis, necrosis/necroptosis ↑Early and late apoptosis (Annexin V/PI) ↑Bax; ↓Bcl-2; ↑p53; ↑Necroptosis-related proteins: RIP-3, MLKL (a) Morphologically—apoptosis, necrosis and necroptosis; (b) ↑Early and late apoptosis (Annexin V/PI); ↑Bax, ↓Bcl-2; ↑p53; ↑necroptosis-related proteins: RIP-3, MLKL | [11] |
| Human cell lines hepatocellular carcinoma (HCC) and human osteosarcoma (OS) | Biogenic bAgNPs 8.0 ± 2.7 nm | 15–40 µg/mL | ↑Autophagic vesicles ↑Autophagolysosomes ↑LC3B-II expression | N/A | Apoptosis Morphological markers -fragmented nuclei ↑Caspase-3 activity, the cleavage of PARP-1, ↑fragmented DNA | [51] |
| HeLa cells B16 melanoma cells | AgNPs-PVP 26.5 ± 8.4 | 10 µg/mL 50 μg/mL | ↑Autophagosomes ↑LC3-BII ↑Autophagosomes | No No | Apoptosis/necrosis ↑Caspase-3 enhanced by wortmannin Apoptosis/necrosis enhanced by wortmannin | [50] |
| A549 human lung adenocarcinoma | 60-nm AgNPs | Lysosomal pH alkalization and autophagosome formation. ↑LC3-BII | Yes ↑p62 | Cellular damage | [52] | |
| Human breast cancer cells MCF-7 and MDA-MB-468 | 9 ± 2.2 19 ± 2 | (a) 25 mM (b) 100 mM | ↑LC3B-II; ↑beclin1; ↑Atg3 (a, b) | No early (6 h) post-exposure Yes after prolonged exposure (24 h) ↑p62; ↑Rab7 ↓LAMP1; ↓AO fluorescence | ↓Mitochondrial potential, Apoptosis—AnnexinV/PI ↓PARP expression | [12] |
| SH-SY5Y | 30 nm | 12.5 μg/mL | Autophagosomes, acidic autophagic vacuoles, ↑beclin-1; ↑LC3-II | No ↓p62 | No | [7] |
| Human prostate cancer cell line (PC-3) | AgNPs-PVP 78.24 ± 0.58 | 2, 4, 6 µg/mL | ↑LC3-II/LC3-I | Yes ↑p62; Lysosomal degradation ↓cathepsin D ↓lysosome-related genes: CSTA, CSTD, CLCN7, MCOLN1 | No apoptotic cells (Annexin-V FITC/PI) | [9] |
| Breast cancer cells (SKBR3) | N/A | AgNPs-EPS 5 μg/mL | ↑ATG5; ↑ATG7; ↑LC3-II; ↑beclin-1 | No ↓AKT; ↓p-AKT; ↓p62; ↓HSP90 | No No DNA fragmentation | [14] |
| Lung epithelial cancer cells (A549) | 10 to 20 nm, | 32.33 μg/mL | Autophagosomes and autolysosomes ↑ATG5; ↑LC3-II | Yes ↑p62, | Activated caspase-3 | [15] |
| Colorectal adenocarcinoma cell line (HT-29) | <100 nm | 33.45 μg/mL | ↑Beclin-1; ↑XBP-1; ↑CHOP; ↑LC3-II | N/A | Apoptosis; ↑cyt-C; ↑p53; ↑ Bax; ↑ASP3, ↑CASP8; ↑CASP9; ↑CASP12 | [53] |
| IN VIVO | ||||||
| B16 cells injected to the C57BL/6 mice | AgNPs-PVP | 1.5 mg/kg Ag NPs, 1.5 mg/kg Ag NPs plus 25 nmol/kg wortmannin. | ↑LC3-II; Inhibited by wortmannin | No | ↓Tumor growth ↑Apoptosis (TUNEL) Enhanced by wortmannin | [50] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Strużyńska, L. Dual Implications of Nanosilver-Induced Autophagy: Nanotoxicity and Anti-Cancer Effects. Int. J. Mol. Sci. 2023, 24, 15386. https://doi.org/10.3390/ijms242015386
Strużyńska L. Dual Implications of Nanosilver-Induced Autophagy: Nanotoxicity and Anti-Cancer Effects. International Journal of Molecular Sciences. 2023; 24(20):15386. https://doi.org/10.3390/ijms242015386
Chicago/Turabian StyleStrużyńska, Lidia. 2023. "Dual Implications of Nanosilver-Induced Autophagy: Nanotoxicity and Anti-Cancer Effects" International Journal of Molecular Sciences 24, no. 20: 15386. https://doi.org/10.3390/ijms242015386
APA StyleStrużyńska, L. (2023). Dual Implications of Nanosilver-Induced Autophagy: Nanotoxicity and Anti-Cancer Effects. International Journal of Molecular Sciences, 24(20), 15386. https://doi.org/10.3390/ijms242015386




