Advancements in Plasma Agriculture: A Review of Recent Studies
Abstract
:1. Introduction
2. Physical Plasma and Its Effect on Objects
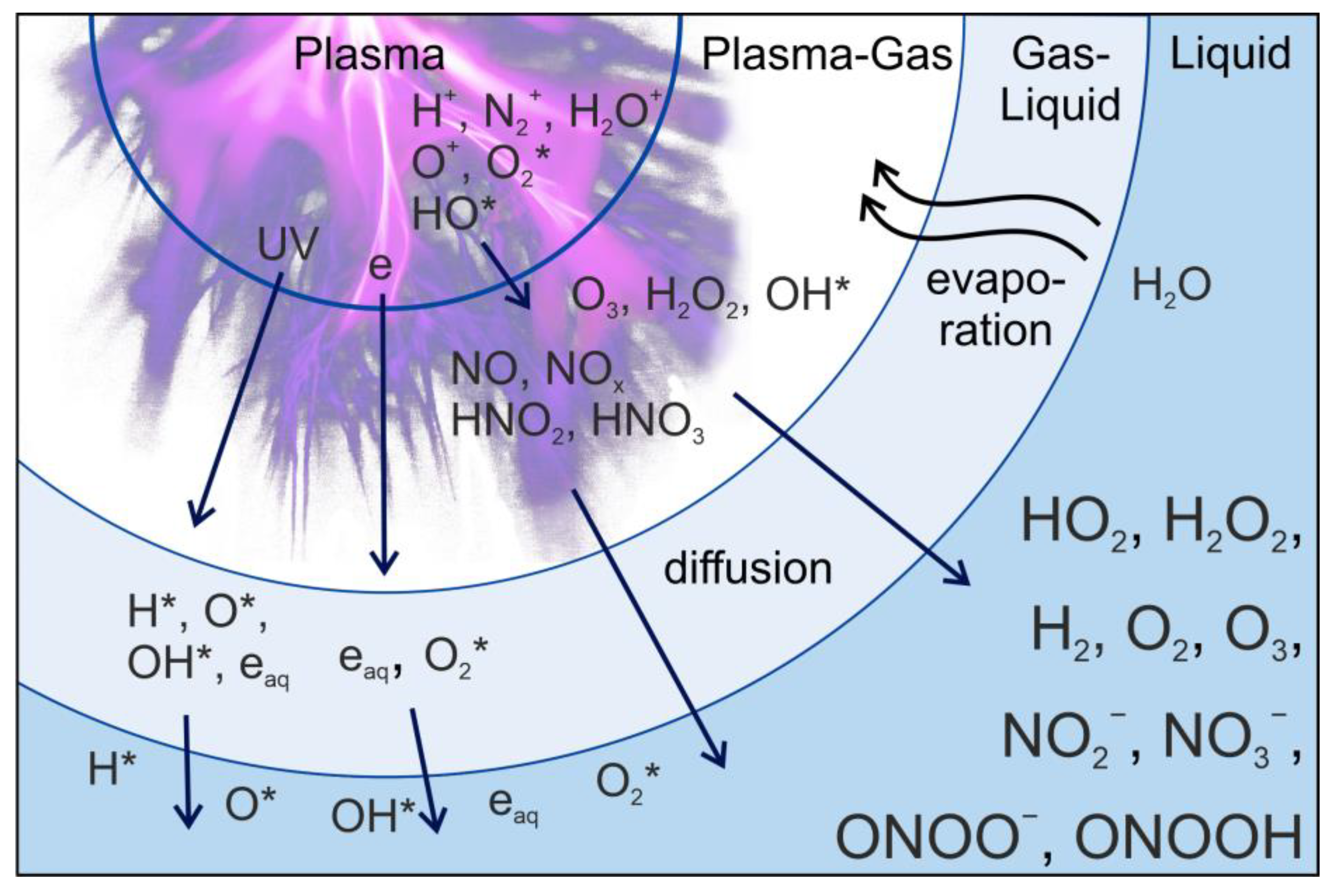
- Plasma contains high-energy electrons that can collide with biological objects, leading to energy transfer, excitation, and ionization of molecules. These processes can induce various chemical reactions within the biological material.
- The presence of charged particles in plasma generates electric fields that can influence the behavior of charged molecules and ions within the biological object.
- Plasma produces a multitude of reactive species, including free radicals (e.g., OH•, O•, N•), ions, and excited molecules. These species play a significant role in initiating chemical reactions within the biological material.
- The reactive oxygen and nitrogen species generated by plasma can lead to oxidative stress, affecting cellular components and signaling pathways.
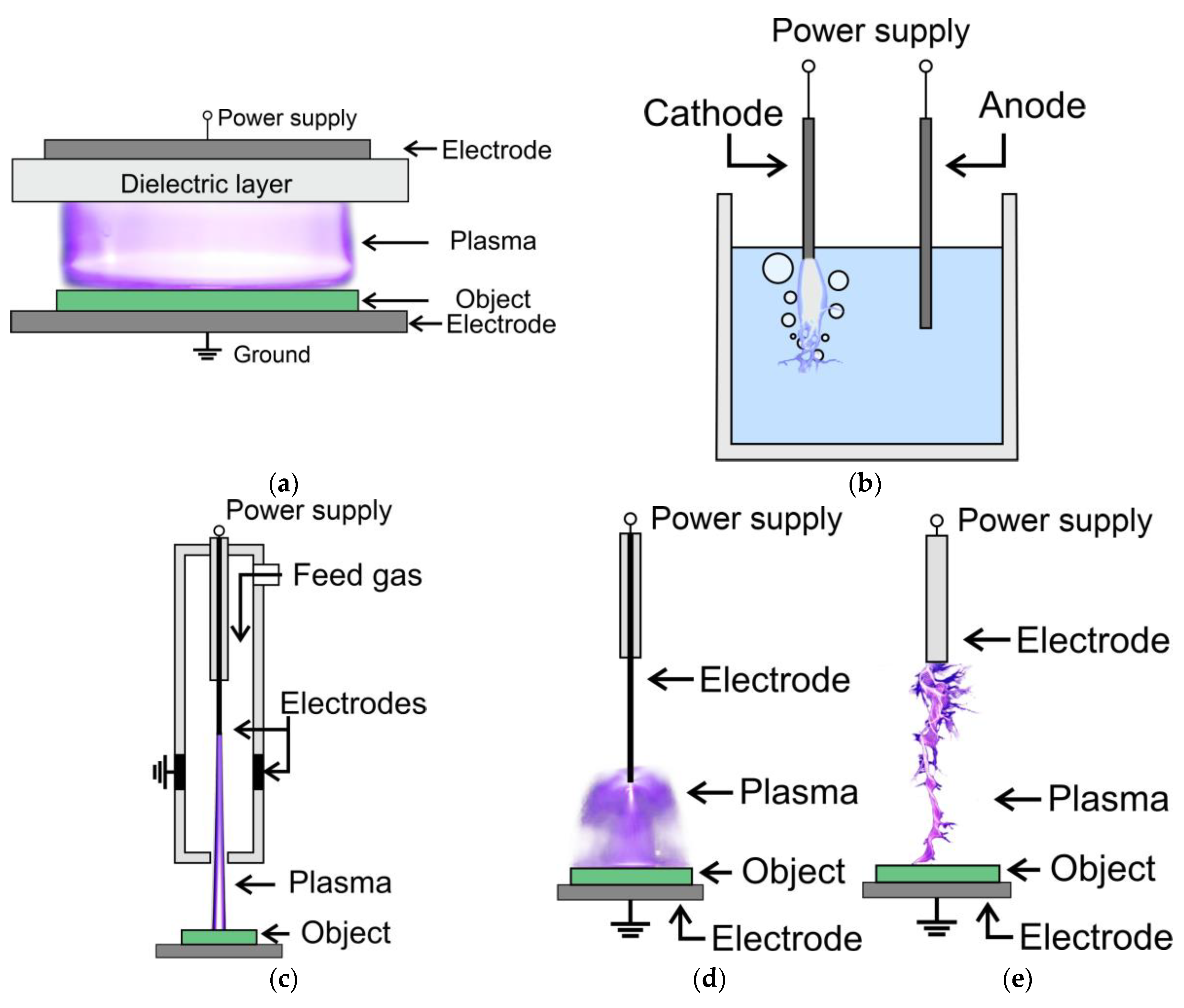
3. Plasma Sources in Biological Sciences and Agriculture
- dielectric barrier discharge (DBD),
- corona discharge,
- spark discharge,
- atmospheric pressure plasma jets (APPJ) and plasma torch,
- underwater discharge.
3.1. Dielectric Barrier Discharge
3.2. Corona Discharge
3.3. Spark Discharge
3.4. Atmospheric Pressure Plasma Jets and Plasma Torch
3.5. Underwater Discharge
4. Direct Plasma Treatment Results
4.1. Dielectric Barrier Discharge
4.2. Atmospheric Pressure Plasma Jets and Plasma Torch
4.3. Inductively Coupled Radio Frequency Plasma
4.4. Corona and Glow Discharges
5. Plasma-Activated Water Treatment Results
5.1. Dielectric Barrier Discharge
5.2. Atmospheric Pressure Plasma Jets and Plasma Torch
5.3. Corona Discharge
5.4. Gliding Arc Discharge
5.5. Spark and Glow Discharges
5.6. Underwater Discharge
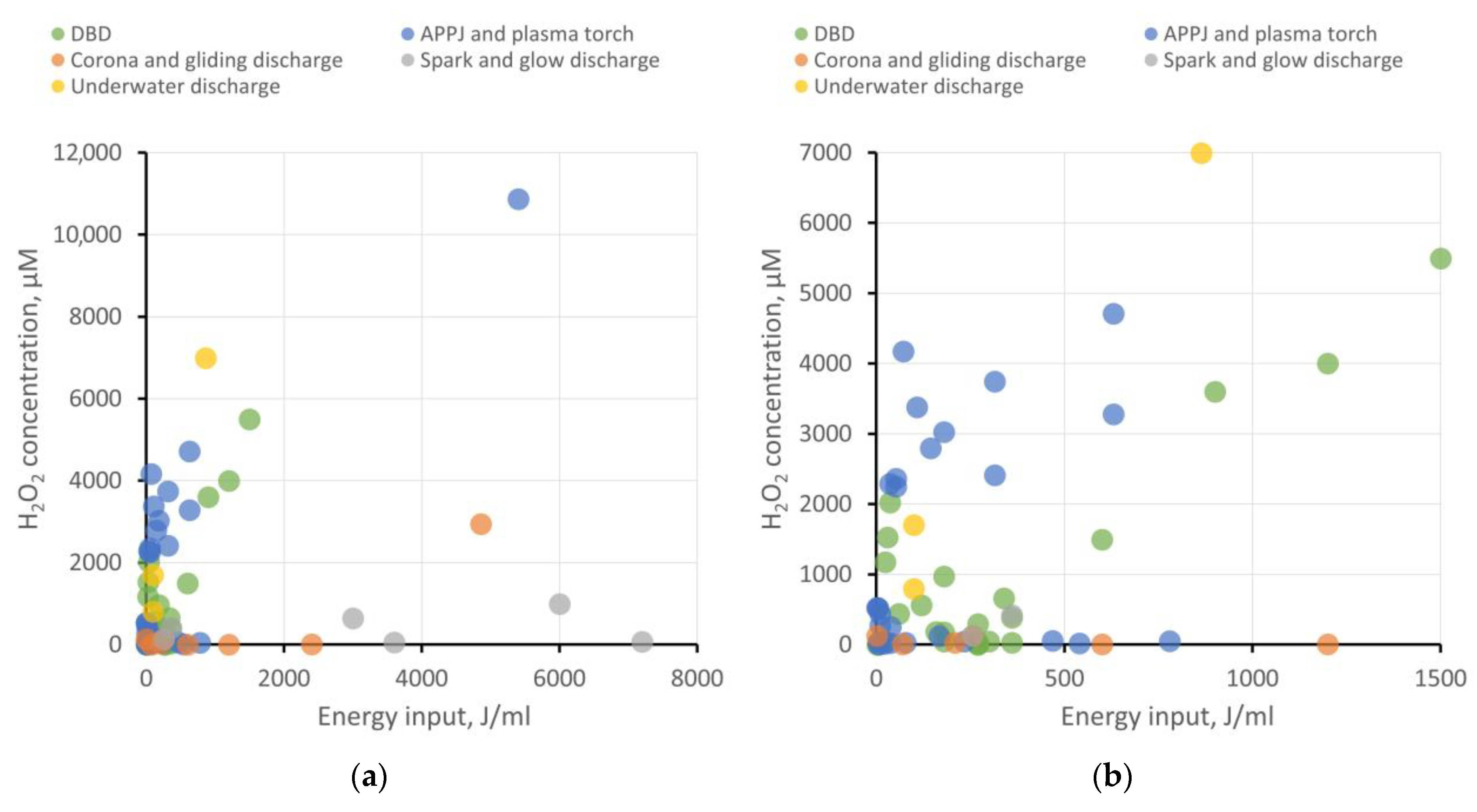
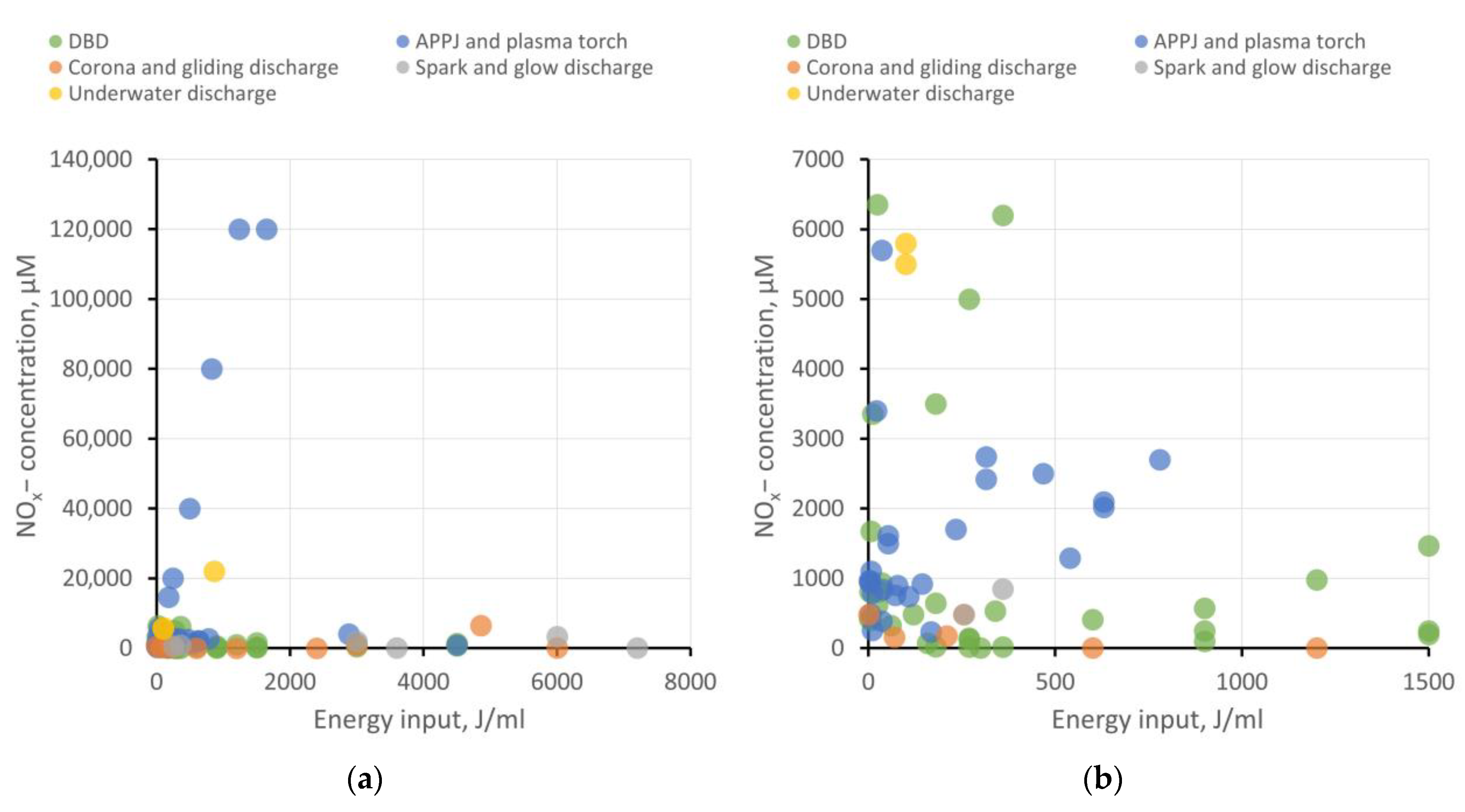
5.7. H2O2 and NOx− Generation Efficiency for Different Types of Plasma Sources
- DBD shows good energy efficiency when the goal is not to achieve ROS and RNS concentrations exceeding 5−6 mM and when large volumes of PAW are not required. To enhance productivity, increasing the plasma–liquid interaction surface area is necessary, transitioning from a planar electrode configuration to, for example, a coaxial one and implementing liquid flow.
- APPJ also appears attractive in terms of energy efficiency. Using gases to create a plasma jet offers broad possibilities in enriching PAW with target active compounds. For instance, it is feasible to achieve high RNS production while keeping the liquid free from extraneous impurities. On the other hand, using a gas injection system places additional demands on workspace organization and slightly raises treatment costs. For creating larger volumes of PAW, using a microwave plasma torch as the plasma source seems more efficient.
- Corona, gliding, and spark discharges might not initially show high ROS and RNS production, but they provide the opportunity to create “pure” PAW without side impurities, which is in demand, for example, in medicine or food processing. To improve the energy efficiency of PAW generation, increasing the plasma–liquid interaction area can be achieved through multi-spark (multi-electrode) systems. Gas injection into the discharge region is also described to enhance the production of target bioactive compounds.
- Underwater discharges allow for the generation of large volumes of PAW with high concentrations of ROS and RNS. These discharges can be employed for creating concentrates to replace chemical fertilizers in agrobiological tasks or for disinfection purposes. However, close attention must be paid to studying electrode erosion and electrolysis processes, which significantly contribute to the chemical composition of PAW. The presence of metal nanoparticles and dissolved metal compounds can either help or hinder the benefits of PAW use.
6. Conclusions
- One of the primary challenges lies in the development and selection of plasma sources suitable for agricultural applications. These sources must generate stable and controllable plasma states while being energy-efficient, cost-effective, and scalable for deployment across various farming scales.
- Achieving consistent and reproducible results with plasma treatments across different settings remains a challenge. Standardizing parameters such as discharge power, treatment duration, and gas composition is essential to ensure the reliability and comparability of outcomes.
- Clarifying the underlying physiological, biochemical, and genetic mechanisms of plasma–plant interactions is critical for maximizing desired effects. The intricate interactions between plasma and plants are not yet fully understood. The variability of plant species, growth stages, and environmental conditions can influence the outcomes of plasma treatment.
- Determining the optimal treatment conditions for diverse agricultural contexts is complex. Tailoring plasma treatments to address specific crop types, growth stages, and environmental factors requires a nuanced understanding of how these variables influence plasma effects.
- In a broad range of tasks, direct plasma treatment can be substituted with the use of PAW treatment. However, it is crucial to identify and control the ROS and RNS and the dissolved ions of metals and nanoparticles of metals and other elements present initially in the electrodes and vessels used for generating PAW.
- Achieving precise and reproducible biological effects from the use of PAW requires studying the dynamics of changes in the concentration of ROS and RNS over the period necessary for delivering PAW to the consumer. Research demonstrates that the concentration of several important bioactive compounds can change by an order of magnitude (or more) within several tens of minutes after the completion of plasma treatment.
Author Contributions
Funding
Conflicts of Interest
References
- Adamovich, I.; Agarwal, S.; Ahedo, E.; Alves, L.L.; Baalrud, S.; Babaeva, N.; Bogaerts, A.; Bourdon, A.; Bruggeman, P.J.; Canal, C.; et al. The 2022 Plasma Roadmap: Low Temperature Plasma Science and Technology. J. Phys. D Appl. Phys. 2022, 55, 373001. [Google Scholar] [CrossRef]
- Hossain, M.M.; Robinson Junior, N.A.; Mok, Y.S.; Wu, S. Investigation of Silver Nanoparticle Synthesis with Various Nonthermal Plasma Reactor Configurations. Arab. J. Chem. 2023, 16, 105174. [Google Scholar] [CrossRef]
- Hossain, M.M.; Wu, S.; Nasir, A.; Mohotti, D.; Robinson, N.A.; Yuan, Y.; Agyekum-Oduro, E.; Akter, A.; Bhuiyan, K.A.; Ahmed, R.; et al. Superhydrophobic and Superoleophilic Surfaces Prepared by One-Step Plasma Polymerization for Oil-Water Separation and Self-Cleaning Function. Surf. Interfaces 2022, 35, 102462. [Google Scholar] [CrossRef]
- Nonnenmacher, L.; Fischer, M.; Haralambiev, L.; Bekeschus, S.; Schulze, F.; Wassilew, G.I.; Schoon, J.; Reichert, J.C. Orthopaedic Applications of Cold Physical Plasma. EFORT Open Rev. 2023, 8, 409–423. [Google Scholar] [CrossRef]
- Koga-Ito, C.Y.; Kostov, K.G.; Miranda, F.S.; Milhan, N.V.M.; Azevedo Neto, N.F.; Nascimento, F.; Pessoa, R.S. Cold Atmospheric Plasma as a Therapeutic Tool in Medicine and Dentistry. Plasma Chem. Plasma Process. 2023, 21, 2932. [Google Scholar] [CrossRef]
- Lotfi, M.; Khani, M.; Shokri, B. A Review of Cold Atmospheric Plasma Applications in Dermatology and Aesthetics. Plasma Med. 2023, 13, 39–63. [Google Scholar] [CrossRef]
- Ranieri, P.; Sponsel, N.; Kizer, J.; Rojas-Pierce, M.; Hernández, R.; Gatiboni, L.; Grunden, A.; Stapelmann, K. Plasma Agriculture: Review from the Perspective of the Plant and Its Ecosystem. Plasma Process. Polym. 2021, 18, 2000162. [Google Scholar] [CrossRef]
- Zhou, R.; Zhou, R.; Wang, P.; Xian, Y.; Mai-Prochnow, A.; Lu, X.; Cullen, P.J.; Ostrikov, K.; Bazaka, K. Plasma-Activated Water: Generation, Origin of Reactive Species and Biological Applications. J. Phys. D Appl. Phys. 2020, 53, 303001. [Google Scholar] [CrossRef]
- Shaji, M.; Rabinovich, A.; Surace, M.; Sales, C.; Fridman, A. Physical Properties of Plasma-Activated Water. Plasma 2023, 6, 45–57. [Google Scholar] [CrossRef]
- Lebedev, Y.A.; Shakhatov, V.A. Decomposition of Carbon Dioxide in Microwave Discharges (an Analytical Review). Russ. J. Appl. Chem. 2022, 95, 1–20. [Google Scholar] [CrossRef]
- Babaeva, N.Y.; Naidis, G.V.; Tereshonok, D.V.; Chernyshev, T.V.; Volkov, L.S.; Vasiliev, M.M.; Petrov, O.F. CO2 Conversion in a Microwave Plasma Torch: 2D vs 1D Approaches. Plasma Sources Sci. Technol. 2023, 32, 054001. [Google Scholar] [CrossRef]
- Artem’ev, K.V.; Batanov, G.M.; Berezhetskaya, N.K.; Borzosekov, V.D.; Gritsinin, S.I.; Davydov, A.M.; Kolik, L.V.; Konchekov, E.M.; Kossyi, I.A.; Lebedev, Y.A.; et al. Synthesis of Nitrogen Oxides in a Subthreshold Microwave Discharge in Air and in Air Mixtures with Methane. Plasma Phys. Rep. 2020, 46, 311–319. [Google Scholar] [CrossRef]
- Artem’ev, K.V.; Batanov, G.M.; Davydov, A.M.; Berezhetskaya, N.K.; Borzosekov, V.D.; Kolik, L.V.; Konchekov, E.M.; Kossyi, I.A.; Petrov, A.E.; Sarksyan, K.A.; et al. Characteristics of a Subthreshold Microwave Discharge in a Wave Beam in Air and the Efficiency of the Plasma-Chemical Reactor. Plasma Phys. Rep. 2021, 47, 498–502. [Google Scholar] [CrossRef]
- Dryakhlov, V.O.; Nikitina, M.Y.; Shaikhiev, I.G.; Galikhanov, M.F.; Shaikhiev, T.I.; Bonev, B.S. Effect of Parameters of the Corona Discharge Treatment of the Surface of Polyacrylonitrile Membranes on the Separation Efficiency of Oil-in-Water Emulsions. Surf. Eng. Appl. Electrochem. 2015, 51, 406–411. [Google Scholar] [CrossRef]
- Skvortsova, N.N.; Stepakhin, V.D.; Borzosekov, V.D.; Sorokin, A.A.; Malakhov, D.V.; Kachmar, V.V.; Kolik, L.V.; Konchekov, E.M.; Gusein-zade, N.G.; Akmadullina, N.S.; et al. Microwave Plasma Imitation Experiments on Deposition of Lunar Dust on Metal Plates. Plasma Phys. Rep. 2023, 49, 120–128. [Google Scholar] [CrossRef]
- Lebedev, Y.A. Microwave Discharges in Liquid Hydrocarbons: Physical and Chemical Characterization. Polymers 2021, 13, 1678. [Google Scholar] [CrossRef] [PubMed]
- Averin, K.A.; Borisov, R.S.; Lebedev, Y.A. Microwave Discharge in Liquid Hydrocarbons: Study of a Liquid Hydrocarbon after Its Discharge Treatment Including Air Bubbling. High Energy Chem. 2020, 54, 210–216. [Google Scholar] [CrossRef]
- Skvortsova, N.N.; Shishilov, O.N.; Akhmadullina, N.S.; Konchekov, E.M.; Letunov, A.A.; Malakhov, D.V.; Obraztsova, E.A.; Stepakhin, V.D. Synthesis of Micro- and Nanostructured Materials via Oscillating Reactions Initiated by High-Power Microwave Pulses. Ceram. Int. 2021, 47, 3978–3987. [Google Scholar] [CrossRef]
- Akhmadullina, N.S.; Skvortsova, N.N.; Obraztsova, E.A.; Stepakhin, V.D.; Konchekov, E.M.; Letunov, A.A.; Konovalov, A.A.; Kargin, Y.F.; Shishilov, O.N. Plasma-Chemical Processes under High-Power Gyrotron’s Discharge in the Mixtures of Metal and Dielectric Powders. Chem. Phys. 2019, 516, 63–70. [Google Scholar] [CrossRef]
- Sirotkin, N.; Khlyustova, A.; Costerin, D.; Naumova, I.; Titov, V.; Agafonov, A. Applications of Plasma Synthesized ZnO, TiO2, and Zn/TiOx Nanoparticles for Making Antimicrobial Wound-healing Viscose Patches. Plasma Process. Polym. 2022, 19, 2100093. [Google Scholar] [CrossRef]
- Sirotkin, N.A.; Khlyustova, A.V.; Titov, V.A.; Agafonov, A.V. The Use of a Novel Three-Electrode Impulse Underwater Discharge for the Synthesis of W-Mo Mixed Oxide Nanocomposites. Plasma Chem. Plasma Process. 2022, 42, 191–209. [Google Scholar] [CrossRef]
- Małajowicz, J.; Khachatryan, K.; Kozłowska, M. Properties of Water Activated with Low-Temperature Plasma in the Context of Microbial Activity. Beverages 2022, 8, 63. [Google Scholar] [CrossRef]
- Veerana, M.; Yu, N.; Ketya, W.; Park, G. Application of Non-Thermal Plasma to Fungal Resources. JoF 2022, 8, 102. [Google Scholar] [CrossRef]
- Soni, A.; Choi, J.; Brightwell, G. Plasma-Activated Water (PAW) as a Disinfection Technology for Bacterial Inactivation with a Focus on Fruit and Vegetables. Foods 2021, 10, 166. [Google Scholar] [CrossRef]
- Laroussi, M.; Bekeschus, S.; Keidar, M.; Bogaerts, A.; Fridman, A.; Lu, X.; Ostrikov, K.; Hori, M.; Stapelmann, K.; Miller, V.; et al. Low-Temperature Plasma for Biology, Hygiene, and Medicine: Perspective and Roadmap. IEEE Trans. Radiat. Plasma Med. Sci. 2022, 6, 127–157. [Google Scholar] [CrossRef]
- Fridman, G.; Friedman, G.; Gutsol, A.; Shekhter, A.B.; Vasilets, V.N.; Fridman, A. Applied Plasma Medicine. Plasma Process. Polym. 2008, 5, 503–533. [Google Scholar] [CrossRef]
- Vasilets, V.N.; Gutsol, A.; Shekhter, A.B.; Fridman, A. Plasma Medicine. High Energy Chem. 2009, 43, 229–233. [Google Scholar] [CrossRef]
- Pavlik, T.; Gudkova, V.; Razvolyaeva, D.; Pavlova, M.; Kostukova, N.; Miloykovich, L.; Kolik, L.; Konchekov, E.; Shimanovskii, N. The Role of Autophagy and Apoptosis in the Combined Action of Plasma-Treated Saline, Doxorubicin, and Medroxyprogesterone Acetate on K562 Myeloid Leukaemia Cells. Int. J. Mol. Sci. 2023, 24, 5100. [Google Scholar] [CrossRef] [PubMed]
- Akopdzhanov, A.G.; Sveshnikova, E.D.; Guseyn-zade, N.G.; Kolik, L.V.; Konchekov, E.M.; Shimanovskiy, N.L. Enhancement of the Cytotoxic Effect of Doxorubicin on Tumor Cells Upon Exposure to Atmospheric Cold Plasma. Pharm. Chem. J. 2021, 55, 11–13. [Google Scholar] [CrossRef]
- Akopdzhanov, A.G.; Shimanovskii, N.L.; Stepanova, D.S.; Fedotcheva, T.A.; Pulish, A.V.; Gusein-zade, N.G.; Kolik, L.V.; Konchekov, E.M. The Cytotoxicity of Cold Atmospheric Plasma against HeLa Cancer Cells and Its Modification with Pharmaceutical Substances. Biophysics 2019, 64, 926–929. [Google Scholar] [CrossRef]
- Pavlik, T.I.; Gusein-zade, N.G.; Kolik, L.V.; Shimanovskii, N.L. Comparison of the Biological Properties of Plasma-Treated Solution and Solution of Chemical Reagents. Appl. Sci. 2022, 12, 3704. [Google Scholar] [CrossRef]
- Pavlik, T.; Gusein-Zade, N. Characterizing the Biological Effects of Plasma-Activated Physiological Saline. Plasma Med. 2022, 12, 1–11. [Google Scholar] [CrossRef]
- Adhikari, B.; Adhikari, M.; Park, G. The Effects of Plasma on Plant Growth, Development, and Sustainability. Appl. Sci. 2020, 10, 6045. [Google Scholar] [CrossRef]
- Leti, L.-I.; Gerber, I.C.; Mihaila, I.; Galan, P.-M.; Strajeru, S.; Petrescu, D.-E.; Cimpeanu, M.-M.; Topala, I.; Gorgan, D.-L. The Modulatory Effects of Non-Thermal Plasma on Seed’s Morphology, Germination and Genetics—A Review. Plants 2022, 11, 2181. [Google Scholar] [CrossRef] [PubMed]
- Mildaziene, V.; Ivankov, A.; Sera, B.; Baniulis, D. Biochemical and Physiological Plant Processes Affected by Seed Treatment with Non-Thermal Plasma. Plants 2022, 11, 856. [Google Scholar] [CrossRef]
- Pańka, D.; Jeske, M.; Łukanowski, A.; Baturo-Cieśniewska, A.; Prus, P.; Maitah, M.; Maitah, K.; Malec, K.; Rymarz, D.; Muhire, J.D.D.; et al. Can Cold Plasma Be Used for Boosting Plant Growth and Plant Protection in Sustainable Plant Production? Agronomy 2022, 12, 841. [Google Scholar] [CrossRef]
- Mumtaz, S.; Khan, R.; Rana, J.N.; Javed, R.; Iqbal, M.; Choi, E.H.; Han, I. Review on the Biomedical and Environmental Applications of Nonthermal Plasma. Catalysts 2023, 13, 685. [Google Scholar] [CrossRef]
- Rahman, M.; Hasan, M.S.; Islam, R.; Rana, R.; Sayem, A.; Sad, M.A.A.; Matin, A.; Raposo, A.; Zandonadi, R.P.; Han, H.; et al. Plasma-Activated Water for Food Safety and Quality: A Review of Recent Developments. Int. J. Environ. Res. Public Health 2022, 19, 6630. [Google Scholar] [CrossRef]
- Holc, M.; Mozetič, M.; Recek, N.; Primc, G.; Vesel, A.; Zaplotnik, R.; Gselman, P. Wettability Increase in Plasma-Treated Agricultural Seeds and Its Relation to Germination Improvement. Agronomy 2021, 11, 1467. [Google Scholar] [CrossRef]
- Starič, P.; Vogel-Mikuš, K.; Mozetič, M.; Junkar, I. Effects of Nonthermal Plasma on Morphology, Genetics and Physiology of Seeds: A Review. Plants 2020, 9, 1736. [Google Scholar] [CrossRef]
- Hossain, M.M.; Mok, Y.S.; Kim, S.-J.; Kim, Y.J.; Lee, J.H.; Kim, J.H.; Heo, I. Non-Thermal Plasma in Honeycomb Catalyst for the High-Throughput Removal of Dilute Styrene from Air. J. Environ. Chem. Eng. 2021, 9, 105780. [Google Scholar] [CrossRef]
- Aka, R.J.N.; Wu, S.; Mohotti, D.; Bashir, M.A.; Nasir, A. Evaluation of a Liquid-Phase Plasma Discharge Process for Ammonia Oxidation in Wastewater: Process Optimization and Kinetic Modeling. Water Res. 2022, 224, 119107. [Google Scholar] [CrossRef]
- Žigon, J.; Petrič, M.; Dahle, S. Dielectric Barrier Discharge (DBD) Plasma Pretreatment of Lignocellulosic Materials in Air at Atmospheric Pressure for Their Improved Wettability: A Literature Review. Holzforschung 2018, 72, 979–991. [Google Scholar] [CrossRef]
- Piskarev, I.M. Corona Electric Discharge as a Source of Chemically Active Species. Plasma Chem. Plasma Process. 2021, 41, 1415–1434. [Google Scholar] [CrossRef]
- Piskarev, I.M.; Ivanova, I.P. Comparison of Chemistry Induced by Direct and Indirect Plasma Treatment of Water to the Effect of UV Radiation. Plasma Chem. Plasma Process. 2021, 41, 447–475. [Google Scholar] [CrossRef]
- Bychkov, V.L.; Chernikov, V.A.; Deshko, K.I.; Zaitsev, F.S.; Esakov, I.I.; Vysikaylo, P.I. Corona Discharge Over Alcohol against Germs in Air. IEEE Trans. Plasma Sci. 2021, 49, 1028–1033. [Google Scholar] [CrossRef]
- Bychkov, V.L.; Chernikov, V.A.; Deshko, K.I.; Zaitsev, F.S.; Mikhailovskaya, T.O.; Shvarov, A.P.; Izotov, A.M.; Tarasenko, B.A.; Dudarev, D.P. Corona Discharge Influence on Winter Wheat and Winter Barley Seeds Infected by Fungal Disease. IEEE Trans. Plasma Sci. 2021, 49, 1034–1040. [Google Scholar] [CrossRef]
- Bychkov, V.L.; Bikmukhametova, A.R.; Chernikov, V.A.; Deshko, K.I.; Mikhailovskaya, T.O.; Shvarov, A.P. Corona Discharge Influence on Soil. IEEE Trans. Plasma Sci. 2020, 48, 350–354. [Google Scholar] [CrossRef]
- Piskarev, I.M. Chemical Transformations of Aqueous Solutions Activated by Remote Plasma Spark Discharge in Air, Nitrogen, or Oxygen. High Energy Chem. 2021, 55, 145–149. [Google Scholar] [CrossRef]
- Korzec, D.; Hoppenthaler, F.; Nettesheim, S. Piezoelectric Direct Discharge: Devices and Applications. Plasma 2020, 4, 1–41. [Google Scholar] [CrossRef]
- Konchekov, E.M.; Glinushkin, A.P.; Kalinitchenko, V.P.; Artem’ev, K.V.; Burmistrov, D.E.; Kozlov, V.A.; Kolik, L.V. Properties and Use of Water Activated by Plasma of Piezoelectric Direct Discharge. Front. Phys. 2021, 8, 616385. [Google Scholar] [CrossRef]
- Konchekov, E.M.; Gusein-Zade, N.G.; Kolik, L.V.; Artem’ev, K.V.; Pulish, A.V. Using of Direct Piezo-Discharge in Generation of Plasma-Activated Liquid Media. In IOP Conference Series: Materials Science and Engineering; Institute of Physics Publishing: Bristol, UK, 2020; Volume 848. [Google Scholar]
- Artem’ev, K.V.; Bogachev, N.N.; Gusein-zade, N.G.; Dolmatov, T.V.; Kolik, L.V.; Konchekov, E.M.; Andreev, S.E. Study of Characteristics of the Cold Atmospheric Plasma Source Based on a Piezo Transformer. Russ. Phys. J. 2020, 62, 2073–2080. [Google Scholar] [CrossRef]
- Korzec, D.; Hoppenthaler, F.; Shestakov, A.; Burger, D.; Shapiro, A.; Andres, T.; Lerach, S.; Nettesheim, S. Multi-Device Piezoelectric Direct Discharge for Large Area Plasma Treatment. Plasma 2021, 4, 281–293. [Google Scholar] [CrossRef]
- Barkhudarov, E.M.; Kossyi, I.A.; Kozlov, Y.N.; Temchin, S.M.; Taktakishvili, M.I.; Christofi, N. Multispark Discharge in Water as a Method of Environmental Sustainability Problems Solution. J. At. Mol. Phys. 2013, 2013, 429189. [Google Scholar] [CrossRef]
- Anpilov, A.M.; Barkhudarov, E.M.; Kossyi, I.A.; Misakyan, M.A.; Moryakov, I.V.; Smirnov, M.G.; Taktakishvili, I.M. High-Voltage Pulsed Discharge at the Gas–Liquid Interface in a Multiphase System. Tech. Phys. 2021, 66, 675–680. [Google Scholar] [CrossRef]
- Antipov, S.N.; Gadzhiev, M.K.; Sargsyan, M.A.; Tereshonok, D.V.; Tyuftyaev, A.S.; Yusupov, D.I.; Chistolinov, A.V.; Abramov, A.G.; Ugryumov, A.V. Interelectrode Microwave Glow Discharge in Atmospheric-Pressure Argon Flow. Phys. Scr. 2023, 98, 025604. [Google Scholar] [CrossRef]
- Antipov, S.N.; Chepelev, V.M.; Gadzhiev, M.K.; Abramov, A.G.; Ugryumov, A.V. Some Techniques for Diagnostics of the Cold Plasma Jet Generated on the Base of the Atmospheric-Pressure Microwave Discharge. Plasma Phys. Rep. 2023, 49, 559–562. [Google Scholar] [CrossRef]
- Tikhonov, V.N.; Gorbatov, S.A.; Ivanov, I.A.; Tikhonov, A.V. A New Type of Non-Thermal Atmospheric Pressure Plasma Source Based on a Waveguide Bridge. J. Phys. Conf. Ser. 2021, 2064, 012131. [Google Scholar] [CrossRef]
- Tikhonov, V.N.; Aleshin, S.N.; Ivanov, I.A.; Tikhonov, A.V. The Low-Cost Microwave Plasma Sources for Science and Industry Applications. J. Phys. Conf. Ser. 2017, 927, 012067. [Google Scholar] [CrossRef]
- Petrukhina, D.; Polyakova, I.; Gorbatov, S. Biocide Effect of Non-Thermal Atmospheric Pressure Plasma. Food Process. Tech. Technol. 2021, 51, 86–97. [Google Scholar] [CrossRef]
- Petrukhina, D.; Gorbatov, S.; Tkhorik, O.; Medzhidov, I.; Tikhonov, V.; Shishko, V.; Ivanov, I.; Tikhonov, A.; Gluchshenko, N.; Kharlamov, V. Non-Thermal Plasma Installation as a Pre-Treatment Method of Barley Seeds (Hordeum vulgare L.). J. Phys. Conf. Ser. 2022, 2270, 012012. [Google Scholar] [CrossRef]
- Svarnas, P.; Poupouzas, M.; Papalexopoulou, K.; Kalaitzopoulou, E.; Skipitari, M.; Papadea, P.; Varemmenou, A.; Giannakopoulos, E.; Georgiou, C.D.; Georga, S.; et al. Water Modification by Cold Plasma Jet with Respect to Physical and Chemical Properties. Appl. Sci. 2022, 12, 11950. [Google Scholar] [CrossRef]
- Reuter, S.; Von Woedtke, T.; Weltmann, K.-D. The kINPen—A Review on Physics and Chemistry of the Atmospheric Pressure Plasma Jet and Its Applications. J. Phys. D Appl. Phys. 2018, 51, 233001. [Google Scholar] [CrossRef]
- Babaeva, N.Y.; Naidis, G.V.; Tereshonok, D.V.; Zhang, C.; Huang, B.; Shao, T. Interaction of Helium Plasma Jet with Tilted Targets: Consequences of Target Permittivity, Conductivity and Incidence Angle. Plasma Sources Sci. Technol. 2021, 30, 115021. [Google Scholar] [CrossRef]
- Naumova, I.; Subbotkina, I.; Titov, V.; Sirotkin, N. Effect of Water Activated by Non-Equilibrium Gas-Discharge Plasma on the Germination and Early Growth of Cucumbers. Prikl. Fiz. 2021, 4, 40–46. [Google Scholar] [CrossRef]
- Kim, H.-J.; Bae, J.H.; Lee, S.; Kim, J.; Jung, S.; Jo, C.; Lee, J.Y.; Seo, J.H.; Park, S. Structural and Functional Changes in Soybean Protein via Remote Plasma Treatments. Molecules 2023, 28, 3882. [Google Scholar] [CrossRef]
- Lim, J.S.; Kim, D.; Ki, S.; Mumtaz, S.; Shaik, A.M.; Han, I.; Hong, Y.J.; Park, G.; Choi, E.H. Characteristics of a Rollable Dielectric Barrier Discharge Plasma and Its Effects on Spinach-Seed Germination. Int. J. Mol. Sci. 2023, 24, 4638. [Google Scholar] [CrossRef]
- Florescu, I.; Radu, I.; Teodoru, A.; Gurau, L.; Chireceanu, C.; Bilea, F.; Magureanu, M. Positive Effect Induced by Plasma Treatment of Seeds on the Agricultural Performance of Sunflower. Plants 2023, 12, 794. [Google Scholar] [CrossRef]
- Nicoletto, C.; Falcioni, V.; Locatelli, S.; Sambo, P. Non-Thermal Plasma and Soilless Nutrient Solution Application: Effects on Nutrient Film Technique Lettuce Cultivation. Horticulturae 2023, 9, 208. [Google Scholar] [CrossRef]
- Burducea, I.; Burducea, C.; Mereuta, P.-E.; Sirbu, S.-R.; Iancu, D.-A.; Istrati, M.-B.; Straticiuc, M.; Lungoci, C.; Stoleru, V.; Teliban, G.-C.; et al. Helium Atmospheric Pressure Plasma Jet Effects on Two Cultivars of Triticum aestivum L. Foods 2023, 12, 208. [Google Scholar] [CrossRef]
- Motrescu, I.; Ciolan, M.A.; Calistru, A.E.; Jitareanu, G. Germination and Growth Improvement of Some Micro-Greens under the Influence of Reactive Species Produced in a Non-Thermal Plasma (NTP). Agronomy 2023, 13, 150. [Google Scholar] [CrossRef]
- Rashid, M.; Rashid, M.M.; Alam, M.S.; Talukder, M.R. Enhancement of Growth, Enzymes, Nutrition and Yield of Eggplant: Combined Effects of Plasma Treatments. Plasma Chem. Plasma Process. 2023, 43, 163–181. [Google Scholar] [CrossRef]
- Guragain, R.P.; Baniya, H.B.; Pradhan, S.P.; Pandey, B.P.; Shrestha, B.; Fronczak, M.; Kierzkowska-Pawlak, H.; Subedi, D.P. Growth Enhancement of Radish Seed Induced by Low-Temperature Argon Plasma. Plasma Chem. Plasma Process. 2023, 43, 111–137. [Google Scholar] [CrossRef]
- Ding, Y.; Mo, W.; Deng, Z.; Kimatu, B.M.; Gao, J.; Fang, D. Storage Quality Variation of Mushrooms (Flammulina velutipes) after Cold Plasma Treatment. Life 2022, 13, 70. [Google Scholar] [CrossRef]
- Khan, M.J.; Jovicic, V.; Zbogar-Rasic, A.; Delgado, A. Enhancement of Wheat Flour and Dough Properties by Non-Thermal Plasma Treatment of Wheat Flour. Appl. Sci. 2022, 12, 7997. [Google Scholar] [CrossRef]
- Ambrico, P.F.; Zicca, S.; Ambrico, M.; Rotondo, P.R.; De Stradis, A.; Dilecce, G.; Saponari, M.; Boscia, D.; Saldarelli, P. Low Temperature Plasma Strategies for Xylella Fastidiosa Inactivation. Appl. Sci. 2022, 12, 4711. [Google Scholar] [CrossRef]
- Hasan, M.; Sohan, M.S.R.; Sajib, S.A.; Hossain, M.F.; Miah, M.; Maruf, M.M.H.; Khalid-Bin-Ferdaus, K.M.; Kabir, A.H.; Talukder, M.R.; Rashid, M.M.; et al. The Effect of Low-Pressure Dielectric Barrier Discharge (LPDBD) Plasma in Boosting Germination, Growth, and Nutritional Properties in Wheat. Plasma Chem. Plasma Process. 2022, 42, 339–362. [Google Scholar] [CrossRef]
- Molina-Hernandez, J.B.; Laika, J.; Peralta-Ruiz, Y.; Palivala, V.K.; Tappi, S.; Cappelli, F.; Ricci, A.; Neri, L.; Chaves-López, C. Influence of Atmospheric Cold Plasma Exposure on Naturally Present Fungal Spores and Physicochemical Characteristics of Sundried Tomatoes (Solanum lycopersicum L.). Foods 2022, 11, 210. [Google Scholar] [CrossRef]
- Waskow, A.; Ibba, L.; Leftley, M.; Howling, A.; Ambrico, P.F.; Furno, I. An In Situ FTIR Study of DBD Plasma Parameters for Accelerated Germination of Arabidopsis thaliana Seeds. Int. J. Mol. Sci. 2021, 22, 11540. [Google Scholar] [CrossRef]
- Abarghuei, F.M.; Etemadi, M.; Ramezanian, A.; Esehaghbeygi, A.; Alizargar, J. An Application of Cold Atmospheric Plasma to Enhance Physiological and Biochemical Traits of Basil. Plants 2021, 10, 2088. [Google Scholar] [CrossRef]
- Bafoil, M.; Yousfi, M.; Dunand, C.; Merbahi, N. Effects of Dielectric Barrier Ambient Air Plasma on Two Brassicaceae Seeds: Arabidopsis thaliana and Camelina sativa. Int. J. Mol. Sci. 2021, 22, 9923. [Google Scholar] [CrossRef]
- Motyka-Pomagruk, A.; Dzimitrowicz, A.; Orlowski, J.; Babinska, W.; Terefinko, D.; Rychlowski, M.; Prusinski, M.; Pohl, P.; Lojkowska, E.; Jamroz, P.; et al. Implementation of a Non-Thermal Atmospheric Pressure Plasma for Eradication of Plant Pathogens from a Surface of Economically Important Seeds. Int. J. Mol. Sci. 2021, 22, 9256. [Google Scholar] [CrossRef] [PubMed]
- Holubová, Ľ.; Švubová, R.; Slováková, Ľ.; Bokor, B.; Chobotová Kročková, V.; Renčko, J.; Uhrin, F.; Medvecká, V.; Zahoranová, A.; Gálová, E. Cold Atmospheric Pressure Plasma Treatment of Maize Grains—Induction of Growth, Enzyme Activities and Heat Shock Proteins. Int. J. Mol. Sci. 2021, 22, 8509. [Google Scholar] [CrossRef] [PubMed]
- Ivankov, A.; Naučienė, Z.; Degutytė-Fomins, L.; Žūkienė, R.; Januškaitienė, I.; Malakauskienė, A.; Jakštas, V.; Ivanauskas, L.; Romanovskaja, D.; Šlepetienė, A.; et al. Changes in Agricultural Performance of Common Buckwheat Induced by Seed Treatment with Cold Plasma and Electromagnetic Field. Appl. Sci. 2021, 11, 4391. [Google Scholar] [CrossRef]
- Renáta, Š.; Nicolette, V.; Monika, B.; Stanislav, K.; Eliška, G.; Veronika, M.; Ľudmila, S. Enhanced In Situ Activity of Peroxidases and Lignification of Root Tissues after Exposure to Non-Thermal Plasma Increases the Resistance of Pea Seedlings. Plasma Chem. Plasma Process. 2021, 41, 903–922. [Google Scholar] [CrossRef]
- Peťková, M.; Švubová, R.; Kyzek, S.; Medvecká, V.; Slováková, Ľ.; Ševčovičová, A.; Gálová, E. The Effects of Cold Atmospheric Pressure Plasma on Germination Parameters, Enzyme Activities and Induction of DNA Damage in Barley. Int. J. Mol. Sci. 2021, 22, 2833. [Google Scholar] [CrossRef] [PubMed]
- Šerá, B.; Šerý, M.; Zahoranová, A.; Tomeková, J. Germination Improvement of Three Pine Species (Pinus) After Diffuse Coplanar Surface Barrier Discharge Plasma Treatment. Plasma Chem. Plasma Process. 2021, 41, 211–226. [Google Scholar] [CrossRef]
- Tomeková, J.; Kyzek, S.; Medvecká, V.; Gálová, E.; Zahoranová, A. Influence of Cold Atmospheric Pressure Plasma on Pea Seeds: DNA Damage of Seedlings and Optical Diagnostics of Plasma. Plasma Chem. Plasma Process. 2020, 40, 1571–1584. [Google Scholar] [CrossRef]
- Nishime, T.M.C.; Wannicke, N.; Horn, S.; Weltmann, K.-D.; Brust, H. A Coaxial Dielectric Barrier Discharge Reactor for Treatment of Winter Wheat Seeds. Appl. Sci. 2020, 10, 7133. [Google Scholar] [CrossRef]
- Švubová, R.; Kyzek, S.; Medvecká, V.; Slováková, Ľ.; Gálová, E.; Zahoranová, A. Novel Insight at the Effect of Cold Atmospheric Pressure Plasma on the Activity of Enzymes Essential for the Germination of Pea (Pisum sativum L. Cv. Prophet) Seeds. Plasma Chem. Plasma Process. 2020, 40, 1221–1240. [Google Scholar] [CrossRef]
- Feizollahi, E.; Iqdiam, B.; Vasanthan, T.; Thilakarathna, M.S.; Roopesh, M.S. Effects of Atmospheric-Pressure Cold Plasma Treatment on Deoxynivalenol Degradation, Quality Parameters, and Germination of Barley Grains. Appl. Sci. 2020, 10, 3530. [Google Scholar] [CrossRef]
- Konchekov, E.M.; Kolik, L.V.; Danilejko, Y.K.; Belov, S.V.; Artem’ev, K.V.; Astashev, M.E.; Pavlik, T.I.; Lukanin, V.I.; Kutyrev, A.I.; Smirnov, I.G.; et al. Enhancement of the Plant Grafting Technique with Dielectric Barrier Discharge Cold Atmospheric Plasma and Plasma-Treated Solution. Plants 2022, 11, 1373. [Google Scholar] [CrossRef] [PubMed]
- Izmailov, A.; Khort, D.; Filippov, R.; Pishchalnikov, R.Y.; Simakin, A.V.; Shogenov, Y. Improvement of Winter Graft Techniques Using Cold Plasma and Plasma-Treated Solution on Cherry Cultures. Appl. Sci. 2022, 12, 4953. [Google Scholar] [CrossRef]
- Pogorzelska-Nowicka, E.; Hanula, M.M.; Brodowska-Trębacz, M.; Górska-Horczyczak, E.; Jankiewicz, U.; Mazur, T.; Marcinkowska-Lesiak, M.; Półtorak, A.; Wierzbicka, A. The Effect of Cold Plasma Pretreatment on Water-Suspended Herbs Measured in the Content of Bioactive Compounds, Antioxidant Activity, Volatile Compounds and Microbial Count of Final Extracts. Antioxidants 2021, 10, 1740. [Google Scholar] [CrossRef] [PubMed]
- Le, T.Q.X.; Nguyen, L.N.; Nguyen, T.T.; Choi, E.H.; Nguyen, Q.L.; Kaushik, N.K.; Dao, N.T. Effects of Cold Plasma Treatment on Physical Modification and Endogenous Hormone Regulation in Enhancing Seed Germination and Radicle Growth of Mung Bean. Appl. Sci. 2022, 12, 10308. [Google Scholar] [CrossRef]
- Terebun, P.; Kwiatkowski, M.; Starek, A.; Reuter, S.; Mok, Y.S.; Pawłat, J. Impact of Short Time Atmospheric Plasma Treatment on Onion Seeds. Plasma Chem. Plasma Process. 2021, 41, 559–571. [Google Scholar] [CrossRef]
- Pavasupree, S.; Chanchula, N.; Bootchanont, A.; Wattanawikkam, C.; Jitjing, P.; Boonyawan, D.; Porjai, P. Enhancement Propagation of Protocorms in Orchid (Cymbidium tracyanum L. Castle) by Cold Atmospheric Pressure Air Plasma Jet. Plasma Chem. Plasma Process. 2021, 41, 573–589. [Google Scholar] [CrossRef]
- Wiktor, A.; Hrycak, B.; Jasiński, M.; Rybak, K.; Kieliszek, M.; Kraśniewska, K.; Witrowa-Rajchert, D. Impact of Atmospheric Pressure Microwave Plasma Treatment on Quality of Selected Spices. Appl. Sci. 2020, 10, 6815. [Google Scholar] [CrossRef]
- Gutiérrez-León, D.G.; Serrano-Ramírez, T.; López-Callejas, R.; Rodríguez-Méndez, B.G. Non-Thermal Plasma as an Alternative to Enhance the Early Growth Structures in Lentil Plants. Agronomy 2022, 12, 2920. [Google Scholar] [CrossRef]
- Judickaitė, A.; Lyushkevich, V.; Filatova, I.; Mildažienė, V.; Žūkienė, R. The Potential of Cold Plasma and Electromagnetic Field as Stimulators of Natural Sweeteners Biosynthesis in Stevia Rebaudiana Bertoni. Plants 2022, 11, 611. [Google Scholar] [CrossRef]
- Mravlje, J.; Kobal, T.; Regvar, M.; Starič, P.; Zaplotnik, R.; Mozetič, M.; Vogel-Mikuš, K. The Sensitivity of Fungi Colonising Buckwheat Grains to Cold Plasma Is Species Specific. JoF 2023, 9, 609. [Google Scholar] [CrossRef]
- Starič, P.; Mravlje, J.; Mozetič, M.; Zaplotnik, R.; Šetina Batič, B.; Junkar, I.; Vogel Mikuš, K. The Influence of Glow and Afterglow Cold Plasma Treatment on Biochemistry, Morphology, and Physiology of Wheat Seeds. Int. J. Mol. Sci. 2022, 23, 7369. [Google Scholar] [CrossRef]
- Holc, M.; Gselman, P.; Primc, G.; Vesel, A.; Mozetič, M.; Recek, N. Wettability and Water Uptake Improvement in Plasma-Treated Alfalfa Seeds. Agriculture 2022, 12, 96. [Google Scholar] [CrossRef]
- Masiello, M.; Somma, S.; Lo Porto, C.; Palumbo, F.; Favia, P.; Fracassi, F.; Logrieco, A.F.; Moretti, A. Plasma Technology Increases the Efficacy of Prothioconazole against Fusarium Graminearum and Fusarium Proliferatum Contamination of Maize (Zea mays) Seedlings. Int. J. Mol. Sci. 2021, 22, 9301. [Google Scholar] [CrossRef]
- Recek, N.; Holc, M.; Vesel, A.; Zaplotnik, R.; Gselman, P.; Mozetič, M.; Primc, G. Germination of Phaseolus vulgaris L. Seeds after a Short Treatment with a Powerful RF Plasma. Int. J. Mol. Sci. 2021, 22, 6672. [Google Scholar] [CrossRef]
- Ivankov, A.; Zukiene, R.; Nauciene, Z.; Degutyte-Fomins, L.; Filatova, I.; Lyushkevich, V.; Mildaziene, V. The Effects of Red Clover Seed Treatment with Cold Plasma and Electromagnetic Field on Germination and Seedling Growth Are Dependent on Seed Color. Appl. Sci. 2021, 11, 4676. [Google Scholar] [CrossRef]
- Starič, P.; Grobelnik Mlakar, S.; Junkar, I. Response of Two Different Wheat Varieties to Glow and Afterglow Oxygen Plasma. Plants 2021, 10, 1728. [Google Scholar] [CrossRef] [PubMed]
- Marček, T.; Hamow, K.Á.; Janda, T.; Darko, E. Effects of High Voltage Electrical Discharge (HVED) on Endogenous Hormone and Polyphenol Profile in Wheat. Plants 2023, 12, 1235. [Google Scholar] [CrossRef]
- Sohan, M.S.R.; Hasan, M.; Hossain, M.F.; Sajib, S.A.; Miah, M.M.; Iqbal, M.A.; Karmakar, S.; Alam, M.J.; Khalid-Bin-Ferdaus, K.M.; Kabir, A.H.; et al. Improvement of Seed Germination Rate, Agronomic Traits, Enzymatic Activity and Nutritional Composition of Bread Wheat (Triticum aestivum) Using Low-Frequency Glow Discharge Plasma. Plasma Chem. Plasma Process. 2021, 41, 923–944. [Google Scholar] [CrossRef]
- Hui, Y.; Wang, D.; You, Y.; Shao, C.; Zhong, C.; Wang, H. Effect of Low Temperature Plasma Treatment on Biological Characteristics and Yield Components of Wheat Seeds (Triticum aestivum L.). Plasma Chem. Plasma Process. 2020, 40, 1555–1570. [Google Scholar] [CrossRef]
- Yemeli, G.B.N.; Janda, M.; Machala, Z. Non-Thermal Plasma as a Priming Tool to Improve the Yield of Pea in Outdoor Conditions. Plasma Chem. Plasma Process. 2022, 42, 1143–1168. [Google Scholar] [CrossRef]
- Astashev, M.E.; Konchekov, E.M.; Kolik, L.V.; Gudkov, S.V. Electric Impedance Spectroscopy in Trees Condition Analysis: Theory and Experiment. Sensors 2022, 22, 8310. [Google Scholar] [CrossRef]
- Filippov, R.A.; Solovchenko, A.; Khort, D.; Smirnov, I.G.; Kutyrev, A.I.; Pobedonostsev, R.V.; Yanykin, D.V. Application of the Impedance Measurement Method to Evaluate the Results of Winter Grafting of Pear Cuttings Using Cold Plasma. Appl. Sci. 2022, 13, 583. [Google Scholar] [CrossRef]
- Aihaiti, A.; Maimaitiyiming, R.; Wang, L.; Wang, J. Processing of Fresh-Cut Potato Using Plasma-Activated Water Prepared by Decreasing Discharge Frequency. Foods 2023, 12, 2285. [Google Scholar] [CrossRef] [PubMed]
- Javed, R.; Mumtaz, S.; Choi, E.H.; Han, I. Effect of Plasma-Treated Water with Magnesium and Zinc on Growth of Chinese Cabbage. Int. J. Mol. Sci. 2023, 24, 8426. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, C.; Li, Q.; Cheng, J.-H. Physicochemical Properties of Plasma-Activated Water and Its Control Effects on the Quality of Strawberries. Molecules 2023, 28, 2677. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Zhu, Y.; Zheng, Y.; Hu, J.; Chen, J.; Deng, S. The Effect of Dielectric Barrier Discharge Plasma Gas and Plasma-Activated Water on the Physicochemical Changes in Button Mushrooms (Agaricus bisporus). Foods 2022, 11, 3504. [Google Scholar] [CrossRef] [PubMed]
- Cortese, E.; Galenda, A.; Famengo, A.; Cappellin, L.; Roverso, M.; Settimi, A.G.; Dabalà, M.; De Stefani, D.; Fassina, A.; Serianni, G.; et al. Quantitative Analysis of Plant Cytosolic Calcium Signals in Response to Water Activated by Low-Power Non-Thermal Plasma. Int. J. Mol. Sci. 2022, 23, 10752. [Google Scholar] [CrossRef]
- Grainge, G.; Nakabayashi, K.; Iza, F.; Leubner-Metzger, G.; Steinbrecher, T. Gas-Plasma-Activated Water Impact on Photo-Dependent Dormancy Mechanisms in Nicotiana Tabacum Seeds. Int. J. Mol. Sci. 2022, 23, 6709. [Google Scholar] [CrossRef]
- Than, H.A.Q.; Pham, T.H.; Nguyen, D.K.V.; Pham, T.H.; Khacef, A. Non-Thermal Plasma Activated Water for Increasing Germination and Plant Growth of Lactuca sativa L. Plasma Chem. Plasma Process. 2022, 42, 73–89. [Google Scholar] [CrossRef]
- Ka, D.H.; Priatama, R.A.; Park, J.Y.; Park, S.J.; Kim, S.B.; Lee, I.A.; Lee, Y.K. Plasma-Activated Water Modulates Root Hair Cell Density via Root Developmental Genes in Arabidopsis thaliana L. Appl. Sci. 2021, 11, 2240. [Google Scholar] [CrossRef]
- Škarpa, P.; Klofáč, D.; Krčma, F.; Šimečková, J.; Kozáková, Z. Effect of Plasma Activated Water Foliar Application on Selected Growth Parameters of Maize (Zea mays L.). Water 2020, 12, 3545. [Google Scholar] [CrossRef]
- Šimečková, J.; Krčma, F.; Klofáč, D.; Dostál, L.; Kozáková, Z. Influence of Plasma-Activated Water on Physical and Physical–Chemical Soil Properties. Water 2020, 12, 2357. [Google Scholar] [CrossRef]
- Mahanta, S.; Habib, M.R.; Moore, J.M. Effect of High-Voltage Atmospheric Cold Plasma Treatment on Germination and Heavy Metal Uptake by Soybeans (Glycine max). Int. J. Mol. Sci. 2022, 23, 1611. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Feng, J.; Chen, S.; Qin, S.; Zang, Y.; Huang, H.; Wei, J. Effect of Plasma Activated Water on the Degradation of Bifenazate and Spirodiclofen Residues on Cuimi Kumquat and Impact on Its Quality. Agronomy 2023, 13, 1247. [Google Scholar] [CrossRef]
- Weihe, T.; Yao, Y.; Opitz, N.; Wagner, R.; Krall, J.; Schnabel, U.; Below, H.; Ehlbeck, J. Plasma-Treated Water: A Comparison with Analog Mixtures of Traceable Ingredients. Microorganisms 2023, 11, 932. [Google Scholar] [CrossRef] [PubMed]
- Hsu, S.-C.; Kong, T.-K.; Chen, C.-Y.; Chen, H.-L. Plasma-Activated Water Affects the Antioxidant Contents in Water Spinach. Appl. Sci. 2023, 13, 3341. [Google Scholar] [CrossRef]
- Ruamrungsri, S.; Sawangrat, C.; Panjama, K.; Sojithamporn, P.; Jaipinta, S.; Srisuwan, W.; Intanoo, M.; Inkham, C.; Thanapornpoonpong, S. Effects of Using Plasma-Activated Water as a Nitrate Source on the Growth and Nutritional Quality of Hydroponically Grown Green Oak Lettuces. Horticulturae 2023, 9, 248. [Google Scholar] [CrossRef]
- Borkar, S.B.; Negi, M.; Kaushik, N.; Abdul Munnaf, S.; Nguyen, L.N.; Choi, E.H.; Kaushik, N.K. Plasma-Generated Nitric Oxide Water Mediates Environmentally Transmitted Pathogenic Bacterial Inactivation via Intracellular Nitrosative Stress. Int. J. Mol. Sci. 2023, 24, 1901. [Google Scholar] [CrossRef]
- Romanjek Fajdetić, N.; Benković-Lačić, T.; Mirosavljević, K.; Antunović, S.; Benković, R.; Rakić, M.; Milošević, S.; Japundžić-Palenkić, B. Influence of Seed Treated by Plasma Activated Water on the Growth of Lactuca sativa L. Sustainability 2022, 14, 16237. [Google Scholar] [CrossRef]
- Japundžić-Palenkić, B.; Benković, R.; Benković-Lačić, T.; Antunović, S.; Japundžić, M.; Romanjek Fajdetić, N.; Mirosavljević, K. Pepper Growing Modified by Plasma Activated Water and Growth Conditions. Sustainability 2022, 14, 15967. [Google Scholar] [CrossRef]
- Sampaio, A.D.G.; Chiappim, W.; Milhan, N.V.M.; Botan Neto, B.; Pessoa, R.; Koga-Ito, C.Y. Effect of the pH on the Antibacterial Potential and Cytotoxicity of Different Plasma-Activated Liquids. Int. J. Mol. Sci. 2022, 23, 13893. [Google Scholar] [CrossRef]
- Heng, Y.; Wang, M.; Jiang, H.; Gao, S.; Zhang, J.; Wan, J.; Song, T.; Ren, Z.; Zhu, Y. Plasma-Activated Acidic Electrolyzed Water: A New Food Disinfectant for Bacterial Suspension and Biofilm. Foods 2022, 11, 3241. [Google Scholar] [CrossRef] [PubMed]
- Sawangrat, C.; Phimolsiripol, Y.; Leksakul, K.; Thanapornpoonpong, S.; Sojithamporn, P.; Lavilla, M.; Castagnini, J.M.; Barba, F.J.; Boonyawan, D. Application of Pinhole Plasma Jet Activated Water against Escherichia Coli, Colletotrichum Gloeosporioides, and Decontamination of Pesticide Residues on Chili (Capsicum annuum L.). Foods 2022, 11, 2859. [Google Scholar] [CrossRef] [PubMed]
- Rathore, V.; Tiwari, B.S.; Nema, S.K. Treatment of Pea Seeds with Plasma Activated Water to Enhance Germination, Plant Growth, and Plant Composition. Plasma Chem. Plasma Process. 2022, 42, 109–129. [Google Scholar] [CrossRef]
- Lee, G.J.; Lamichhane, P.; Ahn, S.J.; Kim, S.H.; Yewale, M.A.; Choong, C.E.; Jang, M.; Choi, E.H. Nitrate Capture Investigation in Plasma-Activated Water and Its Antifungal Effect on Cryptococcus Pseudolongus Cells. Int. J. Mol. Sci. 2021, 22, 12773. [Google Scholar] [CrossRef]
- Rathore, V.; Patel, D.; Shah, N.; Butani, S.; Pansuriya, H.; Nema, S.K. Inactivation of Candida Albicans and Lemon (Citrus limon) Spoilage Fungi Using Plasma Activated Water. Plasma Chem. Plasma Process. 2021, 41, 1397–1414. [Google Scholar] [CrossRef]
- Rashid, M.; Rashid, M.M.; Reza, M.A.; Talukder, M.R. Combined Effects of Air Plasma Seed Treatment and Foliar Application of Plasma Activated Water on Enhanced Paddy Plant Growth and Yield. Plasma Chem. Plasma Process. 2021, 41, 1081–1099. [Google Scholar] [CrossRef]
- Hou, C.-Y.; Kong, T.-K.; Lin, C.-M.; Chen, H.-L. The Effects of Plasma-Activated Water on Heavy Metals Accumulation in Water Spinach. Appl. Sci. 2021, 11, 5304. [Google Scholar] [CrossRef]
- Ki, S.H.; Noh, H.; Ahn, G.R.; Kim, S.H.; Kaushik, N.K.; Choi, E.H.; Lee, G.J. Influence of Nonthermal Atmospheric Plasma-Activated Water on the Structural, Optical, and Biological Properties of Aspergillus brasiliensis Spores. Appl. Sci. 2020, 10, 6378. [Google Scholar] [CrossRef]
- Lamichhane, P.; Veerana, M.; Lim, J.S.; Mumtaz, S.; Shrestha, B.; Kaushik, N.K.; Park, G.; Choi, E.H. Low-Temperature Plasma-Assisted Nitrogen Fixation for Corn Plant Growth and Development. Int. J. Mol. Sci. 2021, 22, 5360. [Google Scholar] [CrossRef]
- Cortese, E.; Settimi, A.G.; Pettenuzzo, S.; Cappellin, L.; Galenda, A.; Famengo, A.; Dabalà, M.; Antoni, V.; Navazio, L. Plasma-Activated Water Triggers Rapid and Sustained Cytosolic Ca2+ Elevations in Arabidopsis thaliana. Plants 2021, 10, 2516. [Google Scholar] [CrossRef] [PubMed]
- Sergeichev, K.F.; Lukina, N.A.; Sarimov, R.M.; Smirnov, I.G.; Simakin, A.V.; Dorokhov, A.S.; Gudkov, S.V. Physicochemical Properties of Pure Water Treated by Pure Argon Plasma Jet Generated by Microwave Discharge in Opened Atmosphere. Front. Phys. 2021, 8, 614684. [Google Scholar] [CrossRef]
- Ashurov, M.K.; Ashurov, E.M.; Astashev, M.E.; Baimler, I.V.; Gudkov, S.V.; Konchekov, E.M.; Lednev, V.N.; Lukina, N.A.; Matveeva, T.A.; Markendudis, A.G.; et al. Development of an Environmentally Friendly Technology for the Treatment of Aqueous Solutions with High-Purity Plasma for the Cultivation of Cotton, Wheat and Strawberries. ChemEngineering 2022, 6, 91. [Google Scholar] [CrossRef]
- Abouelenein, D.; Mustafa, A.M.; Nzekoue, F.K.; Caprioli, G.; Angeloni, S.; Tappi, S.; Castagnini, J.M.; Dalla Rosa, M.; Vittori, S. The Impact of Plasma Activated Water Treatment on the Phenolic Profile, Vitamins Content, Antioxidant and Enzymatic Activities of Rocket-Salad Leaves. Antioxidants 2022, 12, 28. [Google Scholar] [CrossRef] [PubMed]
- Jirešová, J.; Scholtz, V.; Julák, J.; Šerá, B. Comparison of the Effect of Plasma-Activated Water and Artificially Prepared Plasma-Activated Water on Wheat Grain Properties. Plants 2022, 11, 1471. [Google Scholar] [CrossRef]
- Ramazzina, I.; Lolli, V.; Lacey, K.; Tappi, S.; Rocculi, P.; Rinaldi, M. Fresh-Cut Eruca Sativa Treated with Plasma Activated Water (PAW): Evaluation of Antioxidant Capacity, Polyphenolic Profile and Redox Status in Caco2 Cells. Nutrients 2022, 14, 5337. [Google Scholar] [CrossRef] [PubMed]
- Mogo, J.P.K.; Fovo, J.D.; Sop-Tamo, B.; Mafouasson, H.N.A.; Ngwem, M.C.N.; Tebu, M.J.; Youbi, G.K.; Laminsi, S. Effect of Gliding Arc Plasma Activated Water (GAPAW) on Maize (Zea mays L.) Seed Germination and Growth. Seeds 2022, 1, 230–243. [Google Scholar] [CrossRef]
- Terebun, P.; Kwiatkowski, M.; Hensel, K.; Kopacki, M.; Pawłat, J. Influence of Plasma Activated Water Generated in a Gliding Arc Discharge Reactor on Germination of Beetroot and Carrot Seeds. Appl. Sci. 2021, 11, 6164. [Google Scholar] [CrossRef]
- Date, M.B.; Rivero, W.C.; Tan, J.; Specca, D.; Simon, J.E.; Salvi, D.A.; Karwe, M.V. Growth of Hydroponic Sweet Basil (O. basilicum L.) Using Plasma-Activated Nutrient Solution (PANS). Agriculture 2023, 13, 443. [Google Scholar] [CrossRef]
- Guragain, R.P.; Baniya, H.B.; Shrestha, B.; Guragain, D.P.; Subedi, D.P. Improvements in Germination and Growth of Sprouts Irrigated Using Plasma Activated Water (PAW). Water 2023, 15, 744. [Google Scholar] [CrossRef]
- Chumsri, P.; Panpipat, W.; Cheong, L.-Z.; Nisoa, M.; Chaijan, M. Comparative Evaluation of Hydrothermally Produced Rice Starch–Phenolic Complexes: Contributions of Phenolic Type, Plasma-Activated Water, and Ultrasonication. Foods 2022, 11, 3826. [Google Scholar] [CrossRef] [PubMed]
- Abouelenein, D.; Angeloni, S.; Caprioli, G.; Genovese, J.; Mustafa, A.M.; Nzekoue, F.K.; Petrelli, R.; Rocculi, P.; Sagratini, G.; Tappi, S.; et al. Effect of Plasma Activated Water on Selected Chemical Compounds of Rocket-Salad (Eruca sativa Mill.) Leaves. Molecules 2021, 26, 7691. [Google Scholar] [CrossRef] [PubMed]
- Laurita, R.; Gozzi, G.; Tappi, S.; Capelli, F.; Bisag, A.; Laghi, G.; Gherardi, M.; Cellini, B.; Abouelenein, D.; Vittori, S.; et al. Effect of Plasma Activated Water (PAW) on Rocket Leaves Decontamination and Nutritional Value. Innov. Food Sci. Emerg. Technol. 2021, 73, 102805. [Google Scholar] [CrossRef]
- Lukacova, Z.; Svubova, R.; Selvekova, P.; Hensel, K. The Effect of Plasma Activated Water on Maize (Zea mays L.) under Arsenic Stress. Plants 2021, 10, 1899. [Google Scholar] [CrossRef]
- Kostoláni, D.; Ndiffo Yemeli, G.B.; Švubová, R.; Kyzek, S.; Machala, Z. Physiological Responses of Young Pea and Barley Seedlings to Plasma-Activated Water. Plants 2021, 10, 1750. [Google Scholar] [CrossRef] [PubMed]
- Dzimitrowicz, A.; Jamroz, P.; Pohl, P.; Babinska, W.; Terefinko, D.; Sledz, W.; Motyka-Pomagruk, A. Multivariate Optimization of the FLC-Dc-APGD-Based Reaction-Discharge System for Continuous Production of a Plasma-Activated Liquid of Defined Physicochemical and Anti-Phytopathogenic Properties. Int. J. Mol. Sci. 2021, 22, 4813. [Google Scholar] [CrossRef]
- Kučerová, K.; Henselová, M.; Slováková, Ľ.; Bačovčinová, M.; Hensel, K. Effect of Plasma Activated Water, Hydrogen Peroxide, and Nitrates on Lettuce Growth and Its Physiological Parameters. Appl. Sci. 2021, 11, 1985. [Google Scholar] [CrossRef]
- Talar-Krasa, M.; Wolski, K.; Radkowski, A.; Khachatryan, K.; Bujak, H.; Bocianowski, J. Effects of a Plasma Water and Biostimulant on Lawn Functional Value. Agronomy 2021, 11, 254. [Google Scholar] [CrossRef]
- Sajib, S.A.; Billah, M.; Mahmud, S.; Miah, M.; Hossain, F.; Omar, F.B.; Roy, N.C.; Hoque, K.M.F.; Talukder, M.R.; Kabir, A.H.; et al. Plasma Activated Water: The next Generation Eco-Friendly Stimulant for Enhancing Plant Seed Germination, Vigor and Increased Enzyme Activity, a Study on Black Gram (Vigna mungo L.). Plasma Chem. Plasma Process. 2020, 40, 119–143. [Google Scholar] [CrossRef]
- Gamaleev, V.; Iwata, N.; Ito, G.; Hori, M.; Hiramatsu, M.; Ito, M. Scalable Treatment of Flowing Organic Liquids Using Ambient-Air Glow Discharge for Agricultural Applications. Appl. Sci. 2020, 10, 801. [Google Scholar] [CrossRef]
- Padureanu, S.; Burlica, R.; Stoleru, V.; Beniuga, O.; Dirlau, D.; Cretu, D.E.; Astanei, D.; Patras, A. Non-Thermal Plasma-Activated Water: A Cytogenotoxic Potential on Triticum aestivum. Agronomy 2023, 13, 459. [Google Scholar] [CrossRef]
- Rashid, M.; Rashid, M.M.; Alam, M.S.; Talukder, M.R. Stimulating Effects of Plasma Activated Water on Growth, Biochemical Activity, Nutritional Composition and Yield of Potato (Solanum tuberosum L.). Plasma Chem. Plasma Process. 2022, 42, 131–145. [Google Scholar] [CrossRef]
- Belov, S.V.; Danyleiko, Y.K.; Glinushkin, A.P.; Kalinitchenko, V.P.; Egorov, A.V.; Sidorov, V.A.; Konchekov, E.M.; Gudkov, S.V.; Dorokhov, A.S.; Lobachevsky, Y.P.; et al. An Activated Potassium Phosphate Fertilizer Solution for Stimulating the Growth of Agricultural Plants. Front. Phys. 2021, 8, 616. [Google Scholar] [CrossRef]
- Kuzin, A.; Solovchenko, A.; Khort, D.; Filippov, R.; Lukanin, V.; Lukina, N.; Astashev, M.; Konchekov, E. Effects of Plasma-Activated Water on Leaf and Fruit Biochemical Composition and Scion Growth in Apple. Plants 2023, 12, 385. [Google Scholar] [CrossRef]
- Danilejko, Y.K.; Belov, S.V.; Egorov, A.B.; Lukanin, V.I.; Sidorov, V.A.; Apasheva, L.M.; Dushkov, V.Y.; Budnik, M.I.; Belyakov, A.M.; Kulik, K.N.; et al. Increase of Productivity and Neutralization of Pathological Processes in Plants of Grain and Fruit Crops with the Help of Aqueous Solutions Activated by Plasma of High-Frequency Glow Discharge. Plants 2021, 10, 2161. [Google Scholar] [CrossRef] [PubMed]
- Andreev, S.N.; Apasheva, L.M.; Ashurov, M.K.; Lukina, N.A.; Sapaev, B.; Sapaev, I.B.; Sergeichev, K.F.; Shcherbakov, I.A. Production of Pure Hydrogen Peroxide Solutions in Water Activated by the Plasma of Electrodeless Microwave Discharge and Their Application to Control Plant Growth. Phys. Wave Phenom. 2019, 27, 145–148. [Google Scholar] [CrossRef]
- Belov, S.V.; Danileiko, Y.K.; Egorov, A.B.; Lukanin, V.I.; Semenova, A.A.; Lisitsyn, A.B.; Revutskaya, N.M.; Nasonova, V.V.; Yushina, Y.K.; Tolordava, E.R.; et al. Sterilizer of Knives in the Meat Industry, Working by Activating Aqueous Solutions with Glow Discharge Plasma. Processes 2022, 10, 1536. [Google Scholar] [CrossRef]
| Plasma Source Parameters | Object | Key Results | Reference |
|---|---|---|---|
| DBD | Soybeans (Glycine max L.) | Increased protein extraction yield, water binding and oil absorption capacities, and improved emulsifying activity. | [67] |
| DBD | Spinach seeds | Increased water absorption and germination. | [68] |
| DBD | Sunflower (Helianthus annuus L.) seeds | Faster growth, taller seedlings, and increased yield in mature plants persisting even after storage for two weeks. | [69] |
| DBD | Lettuce (‘Kerlis’) | Increased yield, soluble solids, conductivity, and acidity. High ionization plasma raised nitrogen, phosphorus, and potassium content and increased leaf pigments. | [70] |
| DBD | Wheat (Triticum aestivum L. ‘Dacic’ and ‘Otilia’) seeds | Increased chlorophyll and flavonoid content | [71]. |
| DBD | Brassica oleracea and Lepidium sativum seeds | Enhancing hydrophilicity. Increased stem and root length in seedlings. | [72] |
| DBD | Eggplant (Solanum melongena L.) seeds | Increased seed germination plant growth, antioxidant activity, soluble sugars, proteins, total phenols, and certain mineral concentrations (Ca, Cu, Fe, Mn, K), while Zn content decreased. | [73] |
| DBD | White Radish (Raphanus sativus) | Changed seed coat morphology, enhancing water permeability. Improved germination, chlorophyll content, antioxidant activity, and seedling phenols. | [74] |
| DBD | Mushrooms (Flammulina velutipes) | Maintained weight parameters and superoxide anion formation rate. Improved antioxidant activity, enzyme activity, malondialdehyde levels, vitamin C retention, and extended shelf life. | [75] |
| DBD | Wheat (Triticum aestivum L.) flour | Improved wheat flour dough’s viscoelastic properties by strengthening gluten protein-starch networks. Increased flour hydration. | [76] |
| DBD | Xylella fastidiosa | Complete inhibition of bacterial growth. | [77] |
| DBD | Wheat (Triticum aestivum L.) seeds | [78] | |
| DBD | Sundried tomatoes (Solanum lycopersicum L.), Aspergillus rugulovalvus, Aspergillus niger | Reduced bacterial and fungal populations. Lycopene content increased. | [79] |
| DBD | Arabidopsis thaliana seeds | Improved seed germination. | [80] |
| DBD | Basil (Ocimum basilicum L. ‘Genovese Gigante’) | Increased leaf humidity, chlorophyll, carotenoids, antioxidant activity, flavonoids, and peroxidase activity. Reduced microbial load. | [81] |
| DBD | Arabidopsis thaliana, Camelina sativa seeds | A. thaliana benefits from longer exposure due to increased unsaturated fatty acids, while C. sativa’s optimal time is shorter due to reduced unsaturated fatty acids. | [82] |
| DBD | Pectobacterium carotovorum, Pectobacterium atrosepticum, Dickeya solani, inoculated either on mung bean seeds | Inactivated bacteria. A 2-min exposure stimulated seed germination and growth. A 4-min exposure hindered germination and growth. | [83] |
| DBD | Dried maize (Zea mays L.) ‘Ronaldinio’ grains | Enhanced seed viability and seedling growth through enzyme activity stimulation. Triggered heat shock proteins with minimal DNA damage. Surface hydrophilicity increased. | [84] |
| DBD | Buckwheat (Fagopyrum esculentum Moench) ’VB Vokiai’ and ’VB Nojai’ | Decreased emergence but increased growth, biomass, and yield. Enhanced seed weight per plant. | [85] |
| DBD | Dried pea (Pisum sativum L.) ’Prophet‘ seeds | Enhanced seed germination. Increased peroxidase activity in cell walls and mechanical strength. | [86] |
| DBD | Dried barley (Hordeum vulgare L.) ’Maltz‘ grains | Improved germination and enzyme activity. | [87] |
| DBD | Common pine (Pinus sylvestris L.), black pine (Pinus nigra Arnold), mountain pine (Pinus mugo Turra) seeds | Short treatment duration improved germinated and growing. Long treatment duration had a retarding effect. | [88] |
| DBD | Dried pea (Pisum sativum L.) seeds | Improved germination, minimized DNA damage | [89] |
| DBD | Winter wheat (Triticum aestivum L.) | Improved surface wettability and germination. | [90] |
| DBD | Pea (Pisum sativum L.) ’Prophet‘ seeds | Treatment for 60 s improved seed germination by enhancing surface wettability and activating enzymes. Shorter treatments stimulated germination without DNA damage. | [91] |
| DBD | Raw barley (Hordeum vulgare L.) grains | Reduced deoxynivalenol mycotoxin concentration. | [92] |
| DBD | Cuts of rootstock and scion of pear (Pyrus communis L.) | Enhanced scion growth. Improved vascular system differentiation. | [93] |
| DBD | Cuts of rootstock and scion of cherry | Enhanced scion growth. Improved vascular system differentiation | [94] |
| APPJ | Extracts from 12 herbs: Echinacea purpurea, Salvia officinalis, Urtica dioica, Polygonum aviculare, Vaccinium myrtillus, Taraxacum officinale, Hypericum perforatum, Achillea millefolium, Sanguisorba officinalis, Leonurus cardiaca, Ballota nigra, Andrographis paniculata | Enhanced antioxidant activity in extracts by promoting polyphenol extraction, increasing flavonoids and anthocyanins, while reducing volatile compounds and altering aroma. Lowered aerobic bacteria. Induced color and pH shifts. | [95] |
| APPJ | Mung bean (Vigna radiata) | Improved seed germination and stem length. The contact angle decreased, aiding water uptake. | [96] |
| APPJ | Bulb onions (Allium cepa L.) seeds | Improved germination and vigor. | [97] |
| APPJ | Orchid(Cymbidium tracyanum L. Castle) protocorms | Improved size, bud count, fresh and dry weights. Disrupted cell walls, aiding bud elongation and dormancy release. | [98] |
| Microwave-driven plasma jet | Whole black pepper seeds, whole allspice berries, and whole juniper berries | Inactivated fungi but did not achieve complete antibacterial effects. | [99] |
| Corona discharge | Lentil seeds | Improved germination and growth. Reduced bacteria. | [100] |
| DBD | Stevia rebaudiana | Enhanced seed water absorption, germination, and plant yield. Improved nutritional content and potential shelf life extension. | [101] |
| Inductively coupled RF discharge | Grains of common buckwheat (Fagopyrum esculentum Moench) infected with the following fungi: Alternaria alternata (GB002), Aspergillus flavus (GB005), Aspergillus niger (GB006), Cladosporium cladosporioides (GB007), Epicoccum nigrum (GB009), Fusarium fujikuroi (GB011), Fusarium graminearum (GB012), Fusarium oxysporum (GB013), Fusarium proliferatum (GB014), Fusarium sporotrichioides (GB015) | Reduced contamination for most fungal taxa, with Fusarium graminearum being the most sensitive and Fusarium fujikuroi the most resistant. | [102] |
| Inductively coupled RF discharge | Winter wheat (Triticum aestivum L.) ’Ingenio‘ seeds | Increased roughness and lowered contact angle. Enhanced water absorption. Hindered seedling germination, α-amylase activity. | [103] |
| Inductively coupled RF discharge | Alfalfa (Medicago sativa L.) seeds | Enhanced seed surface hydrophilicity. | [104] |
| Inductively coupled RF discharge | Fusarium graminearum and Fusarium proliferatum contaminated maize (Zea mays) seedlings | Fungicide (prothioconazole) combined with plasma effectively reduces fungi contamination. | [105] |
| RF discharge | Common bean (Phaseolus vulgaris L.), | Reduced fungal infection. Increased seed wettability. Decreased hydrophobicity. Increased root length. | [106] |
| Inductively coupled RF discharge | Red clover (Trifolium pratense L.) ’Arimaiciai‘ seeds | Improved germination. Phytohormone levels varied, not directly correlating with germination. Increased root nodule numbers. | [107] |
| RF discharge | Winter wheat (Triticum aestivum L.) ’Apache‘ and ’Bezostaya 1‘ seeds | Increased vigor index, root system, seedling wet weight, and germination rate. | [108] |
| High voltage electrical discharge | Wheat (Triticum aestivum L.) ‘BC Opsesija’ | Enhanced germination and growth by altering hormone and metabolite levels. | [109] |
| Glow discharge | Wheat (Triticum aestivum L.) seeds | Increased seed germination. Superoxide dismutase, catalase, and ascorbate peroxidase activity increased in shoots. Increased content of soluble sugars, proteins, iron, manganese, fat, and ash. Moisture content decreased. | [110] |
| Glow discharge | Wheat (Triticum aestivum L.) ’Shannong 12’ | Improved seed germination index, plant height, bushiness, growth, and number of grains. | [111] |
| Transient spark discharge | Pea (Pisum sativum L.) ’Eso‘ seeds | Increased yields and sustained effect. | [112] |
| Plasma Source | Liquid | PTS Characteristics | Object | Key Results | References |
|---|---|---|---|---|---|
| DBD | Distilled water | pH: 3.42 ± 0.19 and 2.64 ± 0.05; ORV: 461.67 ± 18.18 and 547.33 ± 9.02 mV; Ec: 182.67 ± 16.8 and 883.33 ± 37.21 µs/cm; [O3]: 1.21 ± 0.24 and 6.05 ± 0.73 mg/L; [H2O2]: 181.67 ± 40.41 and 658.33 ± 28.87µM; [NO3−]: 4.03 ± 0.19 and 32.45 ± 5.43 mg/L; [NO2−]: 0.31 ± 0.04 and 0.46 ± 0.08 mg/L, When exposed to 10 kHz and 200 Hz, respectively | Fresh-cut potato | Disinfection. Antioxidant properties. Enzyme Inactivation. | [115] |
| DBD | Deionized water | pH: 7.17 ± 0.34, 8.04 ± 0.51, 3.97 ± 0.065, 6.807 ± 0.210; ORP: 176.5 ± 3.53, 194 ± 5.65, 301 ± 1.41, 241.5 ± 0.70 mV; TDS: 16.9 ± 1.27, 26.5 ± 1.31, 51.6 ± 1.44, 30.2 ± 1.74 ppm; Ec: 21 ± 2.83, 33.7 ± 1.62, 74 ± 2.12, 43.3 ± 2.99 µS/cm [NOx−]: 0.52 ± 0.04, 0.452 ± 0.2, 130.61 ± 1.39, 144.37 ± 1.48 µM; [H2O2]: 0.417 ± 0.01, 0.447 ± 0.03, 4.5 ± 0.26, 3.875 ± 0.441 µM for deionized water with Met+, deionized water with Met+, PAW without Met+, PAW with Met+, respectively | Pak Choi seeds (Brassica campestris L.) | Faster germination. Increased seedling length. Higher chlorophyll and protein content. Positive gene expression. | [116] |
| DBD | Deionized water | [NO3−]: ~25, 35, 60, 90, 110 mg/L [NO2−]: ~0.15, ~0.4, 0.5, 0.82, 0.9 mg/L [H2O2]: <1, ~1.5, 3.6, 4.0, 5.5 mM [O2−]: ~26, 34, 22, 7, 5 mM for 1, 2, 3, 4, 5 min. DBD treatment, respectively | Strawberry | Longer shelf life, reduced spoilage. No taste/texture change. Best quality after 4 days of PAW treatment. | [117] |
| DBD | Pure water | pH: 5.16 ± 0.03 | Button mushrooms (Agaricus bisporus) | Decreased champignon browning, inhibited enzymes, and maintained organoleptic quality for preservation. | [118] |
| DBD | Deionized water | [NO3−]: ~0.1, 0.2, 0.4, 0.7, 1.9 mM for 3, 5, 10, 15, 30 min. 12 kHz plasma treatment, respectively [NO3−]: ~0.25, 0.25, 1.2, 1.3, 3.3 mM For 3, 5, 10, 15, 30 min. 20 kHz plasma treatment, respectively | Arabidopsis thaliana seedlings | Low DBD-PAW doses influenced intracellular Ca2+ signals. | [119] |
| DBD | Distilled water | [H2O2]: 0, 0, 180.4 ± 7.2, 294.9 ± 18.4, 387.7 ± 24.5 μM [·OH]: 16.7 ± 3.3, 25.5 ± 2.9, 55 ± 4.0, 80.0 ± 2.9, 0 μM [NO2−]: 0, 0, 0, 0, 0 μM [NO3−]: 0, 0, 22.7 ± 3.6, 18.1 ± 3.8, 15.7 ± 1.9 μM With He/O2 plasma activation times of 10, 15, 30, 45, and 60 min, respectively [H2O2]: 0, 0, 47.8 ± 3.3, 33.3 ± 2.0, 30.2 ± 0.2 μM [·OH]: 14.5 ± 3.3, 23.1 ± 2.3, 42.7 ± 5.2, 54.3 ± 4.7, 0 μM [NO2−]: 0, 0, 56.5 ± 2.5, 47.3 ± 1.7, 35.4 ± 4.0 μM [NO3−]: 0, 0, 3420.7 ± 103.5, 4948.6 ± 74.5, 6191.1 ± 101.2 μM With air plasma activation times of 10, 15, 30, 45, and 60 min, respectively | Nicotiana tabacum ‘Havana 425’ seeds | Air and He/O2 PAW enhanced testa and endosperm rupture in low fluence conditions. Increased GA3ox2 and EXPA4 activity. | [120] |
| DBD | Distilled water | pH: ~5.8, 5.45, 4.8, 4.85, 4.8, 4.8 Ec: ~10, 13, 14, 15, 18, 17 μS/cm [H2O2]: ~15, 19, 33, 40, 52, 69 mg/L [NO2−]: <5 mg/L [NO3−]: ~20, 30, 40, 38, 50, 58 mg/L For 5, 10, 15, 20, 25, and 30 min plasma treatment, respectively | Lettuce (Lactuca sativa L.) seeds | Boosted lettuce seed germination with positive effects on seedling growth and chlorophyll content. | [121] |
| DBD | Deionized water | pH: 3.62 ± 0.02, 3.34 ± 0.03, 2.94 ± 0.08, 2.62 ± 0.07, 2.37 ± 0.04 Ec: 118.10 ± 2.26, 218.50 ± 9.64, 460.33 ± 15.25, 972.93 ± 32.41, 1847.00 ± 70.19 µS/cm [H2O2]: 0.09 ± 0.01, 0.14 ± 0.01, 0.27 ± 0.02, 0.88 ± 0.04, 1.31 ± 0.04 mg/L [NO2−]: 1.09 ± 0.11, 1.24 ± 0.12, 1.85 ± 0.07, 3.68 ± 0.12, 5.17 ± 0.16 mg/L [NO3−]: 25.29 ± 2.88, 49.05 ± 2.61, 102.67 ± 6.30, 204.87 ± 8.74, 389.08 ± 12.24 mg/L For 5, 7, 12, 19, and 40 min plasma treatment, respectively | Arabidopsis thaliana L. | Affected root hair density via gene regulation (COBL9, XTH9, XTH17). | [122] |
| DBD | Distilled water | [H2O2]: 0.7 ± 0.2 mg/L [NO2−]: 1.071 ± 0.005 mg/L [NO3−]: 24.7 ± 2.3 mg/L | Maize (Zea mays L. ‘SY ORPHEUS’) | Reduced leaf chlorophyll, changed fluorescence parameters, and increased nitrogen content. | [123] |
| DBD | Distilled water | pH: 6.7 Ec: 34 µS [H2O2]: 1.4 ± 0.4 mg/L [NO2−]: 0.753 ± 0.009 mg/L [NO3−]: 20.4 ± 1.8 mg/L | Soil | Minimal effects on soil, with slight changes in evaporation, pH, and water absorption. Higher PAW doses slowed tap water absorption but increased water retention. | [124] |
| DBD | Deionized water | pH: 4.3 [NO3−]: 25.7 mg/L [NO2−]: 16.4 mg/L [H2O2]: 2–5 mg/L | Xylella fastidiosa | Deactivation of Xylella fastidiosa cells. | [77] |
| DBD | Water | [H2O2]: 1,1,2,2,10,100, 100, >100, >100 ppm for 30 kV, 3 min., 30 kV, 5 min., 30 kV, 7 min., 50 kV, 3 min., 50 kV, 5 min., 50 kV, 7 min., 70 kV, 3 min., 70 kV, 5 min., 70 kV, 7 min. plasma treatment, respectively. | Soybeans (Glycine max) | Faster germination and growth. ZnO nanoparticles reduced heavy metal uptake in plants. | [125] |
| APPJ | Deionized water | pH: ~4.5, 3.5, 3 Ec: ~50, 270, 590 µS/cm For 10, 15, and 20 kV treatment, respectively | Cuimi kumquat | Reduced pesticide residues, preserved soluble solids, increased acidity, and maintained fruit color. | [126] |
| Microwave-driven plasma torch | Deionized water | [NO3−]: 72.3 mg/L [NO2−]: 1600.7 mg/L [H2O2]: 717.3 mg/L | Pseudomonas fluorescence suspended cells and P. fluorescence biofilms | Strong antibacterial effects. Effective against suspension cells and P. fluorescence biofilms. | [127] |
| APPJ | Reverse osmosis water | [NO3−]: 42.7 ± 0.70 mg/L [NO2−]: 14.7 ± 0.58 mg/L pH: 3.17 ± 0.06 Ec: 311.7 ± 12.01 µS/cm ORP: 554 ± 2.65 mV | Water spinach (Ipomoea aquatica) seeds | Phenols increase with PAW, more with Cd, and less with Pb. Flavonoids rise with PAW and Cd, with no change with Pb. Heavy metals impact PAW effects. | [128] |
| Pinhole plasma jet | Tap water | pH: 5.5–6 Ec: 1.5 µS/cm [NO3−]: 883.59 mg/L [NO2−]: 31.56 mg/L [H2O2]: 102.99 mg/L | Green oak lettuce (Lactuca sativa L.) | Growth parameters are mostly unaffected. Leaf area and greenness differed. Yields similar to commercial nitrate. Lower nitrate residues with PAW treatment. Plasma nitrate is converted to amino acids at higher concentrations than normal nitrate. | [129] |
| Microwave-driven plasma torch. | Deionized water | pH: ~4 ORP: ~502 mV Ec: ~1367 µS/cm [NOx−]: 4000 µM | Escherichia coli K-12 (KCTC 1116), Pseudomonas aeruginosa (KCTC 1636), Staphylococcus aureus (ATCC 12600) | Reduced viable cells, shifted gene expression (soxRS up, oxyR down), leads to nitric oxide accumulation, and alters bacterial cell morphology. | [130] |
| APPJ | Commercially purified water of pharmaceutical degree (Pharmacopoeia Europea, Ph. Eur. 9) | pH: 6.1, 6.1, 5.8 Ec: ~27 µS/cm [H2O2]: 4.1, 3.1, 0 mg/L [NO2−]: 3.3, 2.8, 1.4 mg/L [NO3−]: 11.2, 5.5, 5.4 mg/L For 10, 20, 30 min after plasma treatment, respectively | Lettuce (Lactuca sativa L.) | Better growth on day 7, but advantages faded by days 14, 21, and 28. No significant root system impact. Increased dry matter content. | [131] |
| APPJ | Commercially purified water of pharmaceutical degree (Pharmacopoeia Europea, Ph. Eur. 9) | pH: 6.1, 6.1, 5.8 [H2O2]: 4.1, 3.1, 0 mg/L [NO2−]: 3.3, 2.8, 1.4 mg/L [NO3−]: 11.2, 5.5, 5.4 mg/L For 10, 20, and 30 min after plasma treatment, respectively | Sweet pepper seeds (Capsicum annuum): ‘Bibic’ and ‘Bernita’ | Height, weight, leaf count, interleaf nodes, and buds increased. Dry matter content rose. Yield increased. | [132] |
| Gliding arc plasma jet | Tap water, deionized water, distilled water, filtered water, and 0.9% saline | Plasma-treated tap water: pH: 5.56, 3.55, 3.06, 2.57 ± 0.09 ORP: 83, 193, 221, 250 ± 5.0 mV TDS: 50, 160, 330, 720 ± 5.0 ppm Ec: 70, 220, 470, 720 ± 5 μS/cm [H2O2]: 15.0, 80.3, 127.3, 111.7 mg/L [NO2−]: 25.0, 47.6, 48.6, 55.6 mg/L [NO3−]: 24.0, 53.3, 120.4, 69.3 mg/L Plasma-treated deionized water: pH: 4.00, 3.30, 2.95, 2.47 ± 0.09 ORP: 169, 208, 228, 239 ± 5.0 mV TDS: 40, 170, 450, 560 ± 5.0 ppm Ec: 50, 240, 310, 800 ± 5 μS/cm [H2O2]: 9.3, 76.5, 82.0, 160.3 mg/L [NO2−]: 9.2, 46.2, 45.8, 62.0 mg/L [NO3−]: 7.3, 47.4, 106.3, 69.3 mg/L After 1.0, 5.0, 30.0, and 60.0 min., respectively | Escherichia coli Oral keratinocyte cell cultures | Strong antimicrobial efficacy. Low cytotoxicity on oral keratinocytes. | [133] |
| APPJ | Acidic electrolyzed water | pH: ~2.3, 2.5, 1.95, 1.91 ORP: ~605, 608, 610, 855 mV Ec: ~1600, 2470, 2750, 3200 μS/cm [H2O2]: 75, 82, 75, 72 mg/L [NO2−]: ~85, 90, 90, 90 mg/L [NO3−]: 2.02, 75.28, 138.7 and 219.6 mg/L With plasma activation times of 3, 6, 10, and 15 min, respectively | B. subtilis (ATCC6633) and E. coli (ATCC8739) | Plasma-activated acidic electrolyzed water (PA-AEW) effectively kills B. subtilis, surpassing PAW and AEW. | [134] |
| Pinhole plasma jet | Deionized water | [H2O2]: 369.12 mg/L | Escherichia coli and Colletotrichum gloeosporioides in chili (Capsicum annuum L.) | Carbendazim and chlorpyrifos reduction. 100% fungal spore germination inhibition. | [135] |
| APPJ | Ultrapure milli-Q | pH: 6.5, 6.0 Ec: 99.0 ± 13.5, 177.3 ± 11.0 µS/cm ORP: 423.3 ± 11.7, 483.3 ± 12.6 mV TDS: 36.3 ± 2.1, 68.3 ± 5.5 ppm [O3]: 2.8 ± 0.1, 2.1 ± 0.3 mg/L [H2O2]: 0.5 ± 0.1, 1.5 ± 0.2 mg/L [NO2−]: 5.1 ± 0.4, 10.5 ± 1.1 mg/L [NO3−]: 34 ± 2.6, 53.9 ± 3.6 mg/L For 5- and 10-min plasma treatment, respectively | Pea (Pisum sativum L.) seeds | PAW-treated pea seeds had improved germination, growth, and biochemical traits. Wax removal, increased hydrophilicity, and enhanced antioxidant enzyme activity. | [136] |
| APPJ | Deionized water and deionized water +Mg2+ | For PAW-Mg2+ pH: ~4.8, 5.2, 5.8, 6.0, 6.6 [NO3−]: ~25, 40, 40, 50, 60 mM For PAW pH: ~3.6, 3.2, 2.6, 2.2, 2.3 [NO3−]: ~20, 40, 80, 120, 120 mM For 3, 6, 10, 15, and 20 min plasma treatment, respectively | Cryptococcus pseudolongus | Magnesium-enriched PAW controls nitrate and exhibit antifungal activity against C. pseudolongus, with nitrate capture influencing its effectiveness. | [137] |
| APPJ | Ultrapure milli-Q | pH: ~2.5, 2.2, 2.0, 1.5, 1.5, 1.0 Ec: ~1, 3, 9, 22, 32, 39 mS/cm [NO3−]: ~200, 300, 500, 800, 900, 1200 mg/L [NO2−]: ~1500, 3000, 10,000, 14,000, 16,000, 16,000 µg/L [O3]: ~26, 24, 24, 17, 15, 14 mg/L [H2O2]: ~6, 4.5, 4.5, 3, 2.5, 2 mg/L For ORP 590, 630, 640, 700, 760, and 795 mV, respectively | C. albicans, Citrus limon | Reduced C. albicans by damaging cell membranes. Long-term fungicidal effects on C. albicans and Citrus limon spoilage fungi. | [138] |
| APPJ | Distilled water | pH: 6.50 ± 0.07 [O3]: 0.45 ± 0.01 mg/L [H2O2]: 8.75 ± 0.09 mg/L [NO2−]: 6.00 ± 0.06 mg/L [NO3−]: 46.00 ± 0.47 mg/L | Paddy seeds (Oryza sativa L.‘BRRIdhan 28’) | Enhanced rice seed germination, improved plant growth, defense mechanisms, enzymatic activity, protein, sugar content, and yielding. | [139] |
| APPJ | Reverse osmotic water | pH: 6.79 ± 0.18, 4.22 ± 0.07, 3.37 ± 0.06, 3.17 ± 0.06 Ec: 104.9 ± 22.55, 117.9 ± 2.80, 208.3 ± 6.51, 311.7 ± 12.01 µS/cm ORP: 370 ± 7.51, 465 ± 9.29, 534 ± 13.0, 554 ± 2.65 mV [H2O2]: ~ 78, 142, 115, 95 mg/L [NO2−]: 10.3 ± 0.58, 17.3 ± 1.15, 13.0 ± 1.00, 14.7 ± 0.58 mg/L [NO3−]: 13.8 ± 0.15, 29.8 ± 1.67, 33.3 ± 0.87, 42.7 ± 0.70 mg/L For 5, 10, 15, and 20 min plasma treatment, respectively | Water spinach (Ipomoea aquatica) seeds | Reduced Cd uptake in spinach but not Pb. | [140] |
| APPJ | Sterile deionized water | pH: 3.53, 3.24, 3.10, 3.01 [H2O2]: ~25, 50, 60, 55 µM [NOx]: ~0.9, 1.7, 2.5, 2.7 mM For 1, 3, 6, and 10 min by the soft plasma jet treatment, respectively. | Aspergillus brasiliensis | Damaged cell walls and reduced spore viability | [141] |
| APPJ | Deionized water | pH: 4.3 ± 0.3, 4.7 ± 0.2, 5.1 ± 0.3, 6.2 ± 0.4 [NOx]: 490.0 ± 53.7, 520.7 ± 71.6, 450.1 ± 69.5, 597.5 ± 53.4 M [NH3]: 2.1 ± 0.1, 2.6 ± 0.3, 2.9 ± 0.2, 4.9 ± 0.3 mg/L [H2O2]: 38.2 ± 5.0, 35.3 ± 3.1, 32.6 ± 6.5, 28.7 ± 4.2 M For PAW, Zn- PAW, Al- PAW, and Mg- PAW, respectively. | Maize plants (Zea mays L.) | PAW with added Mg, Al, or Zn ions accelerated germination and stem growth. Metal ions enhanced nitrogen reduction, leading to increased chlorophyll and protein content in plants. | [142] |
| Plasma torch | Deionized water | pH: ~3 [H2O2]: ~0.5 mg/L [NO2−]: ~47 mg/L [NO3−]: ~33 mg/L | Arabidopsis thaliana | PAW exposure rapidly increased Ca2+ in cells. Nitrates, nitrites, and hydrogen peroxide at PAW concentrations did not affect Ca2+ dynamics. | [143] |
| Plasma torch | Distilled water | pH: ~3 [H2O2]: 70 µM [NOx−]: 15 mM | Strawberry seeds | Improved germination | [144] |
| Plasma torch | Distilled water | pH: ~3 [H2O2]: 22.8 µM [NOx−]: 5.7 mM | Cotton, wheat, and strawberry seeds | Improved germination. Protection against fusarium and hyperthermia. PAW is better than commercial seed germination stimulators. | [145] |
| Corona discharge | Distilled water | pH: 3.3. [H2O2]: 4.5 ± 0.1 [NO2−]: 30.4 ± 0.9 [O3]: 0.3 ± 0.1 mg/L | Fresh rocket (Eruca sativa) leaves | Preserved nutrients, increased some vitamins and polyphenols. Decreased antioxidant and catalase activity, aiding storage quality. | [146] |
| Corona discharge | Distilled water | [H2O2]: 97.4 ± 3.2 mg/L [NO3−]: 391.1 ± 9.3 mg/L [O3]: <3.8 mg/L, [NO2−]: <10−3 mg/L of NO2− mg/L | Wheat grains (Triticum aestivum L.), Escherichia coli, Saccharomyces cerevisiae | Improved germination, shoot growth, and decontamination of E. coli and S. cerevisiae. | [147] |
| Corona discharge | Distilled water | pH: 3.3 [H2O2]: 4.5 ± 0.1 mg/L [NO2−]: 30.4 ± 0.9 mg/L [O3]: 0.3 ± 0.1 mg/L | Fresh-cut Eruca sativa | Reduced radical scavenging activity over time but increased total phenols and glucosinolate percentage. PAW polyphenol extract showed no cytotoxicity and improved redox balance. | [148] |
| Gliding arc plasma | Tap water | pH: 3.4, 3.3 TDS: 90, 110 mg/L Ec: 190, 230 μS/cm [H2O2]: 0.01, 0.028 mM/L [NO3−]: 0.156, 0.176 mM/L For 5 and 15 min after plasma treatment, respectively | Maize (Zea mays L.) seeds | 15 min treatment led to 100% seed germination and improved stem length, leaf width, collar diameter, chlorophyll content, and water uptake. | [149] |
| Gliding arc plasma | Distilled water | pH: ~4.2 ± 0.2, 3.7 ± 0.1, 3.3 ± 0.3 [H2O2]: ~6 ± 1, 7 ± 3, 12 ± 5 µM [NO2−]: ~1.9 ± 0.4, 2.4 ± 0.3, 2.9 ± 0.6 mM For 5, 10, and 20 min plasma treatment, respectively | Beetroot (Beta vulgaris ‘CYLINDRA’ ), carrot (Daucus carota ‘AFALON F1’) seeds | Improved germination in Beta vulgaris and had mixed effects on Daucus carota seeds compared to NaOCl, also altering fungal species composition. | [150] |
| Gliding arc plasma | Nutrient solution | [NO3−]: 191.9 ± 3.1, 189.8 ± 1.7, 191.6 ± 1.6 [NO2−]: 18.8 ± 1.9, 18.6 ± 0.9, 18.7 ± 1.1, For one-time DBD processing and multiple processing, respectively | Ocimum basilicum L. | Boosted basil growth, enriched aroma compounds, and reduced algae. | [151] |
| Gliding arc plasma | Deionized water | pH: 4.69 ± 0.08, 4.48 ± 0.05, 4.37 ± 0.07, 4.29 ± 0.06 T: 16.5 ± 0.04, 16.59 ± 0.06, 16.62 ± 0.03, 16.64 ± 0.04 °C Ec: 50.2 ± 1.60; 60.2 ± 1.20, 70.6 ± 1.80, 76.8 ± 1.60 µS/cm [NO2−]: 0.49 ± 0.04, 0.60 ± 0.03, 0.80 ± 0.06, 0.86 ± 0.04 mg/L [NO3−]: 0.49 ± 0.14, 4.84 ± 0.12, 6.69 ± 0.16, 7.20 ± 0.12 mg/L [NH3]: 1.11 ± 0.07, 2.68 ± 0.09, 3.01 ± 0.07, 3.40 ± 0.08 mg/L for 5, 10, 15, 20. GAD treatment, respectively | Phapar (Fagopyrum esculentum), barley (Hordeum vulgare), mustard (Brassica nigra), and rayo (Brassica juncea) | Improved seed germination, uniformity, daily germination, increased water uptake, root/shoot length, and seed vigor. | [152] |
| RF discharge | Distilled water | pH: 3.0 [H2O2]: 100 ppm | Flour from Noui Khuea brown rice (Oryza sativa L.) conjugated with three different phenolic compounds—gallic acid, sinapic acid, and crude Mon-pu (Glochidion wallichianum Muell Arg) extract | Gallic acid boosts 1,1-diphenyl-2-picrylhydrazyl removal in starch, especially when PAW-synthesized, regardless of ultrasound. Complexation index, resistant starch lower than crude Mon-pu extract complex. | [153] |
| Direct discharge | Distilled water | pH: 3.3 [O3]: 0.3 ± 0.1 mg/L [H2O2]: 4.5 ± 0.1 mg/L [NO2−]: 30.4 ± 0.9 mg/L | Eruca sativa Mill | β-sitosterol and campesterol decreased, while β-carotene, luteolin, and chlorophyll b increased after 2-min PAW exposure. Chlorophyll content reduced at 20 min. | [154] |
| Direct discharge | Distilled water | pH: 3.3 [O3]: 0.3 ± 0.1 mg/L [H2O2]: 4.5 ± 0.1 mg/L [NO2−]: 30.4 ± 0.9 mg/L | Eruca sativa | Reduced bacteria by 1.7–3 Log CFU/g with minor quality changes, surpassing the antibacterial effect of hypochlorite. | [155] |
| Transient spark discharge | Tap water | pH: 7.5 [H2O2]: 0.5 ± 0.1 mM [NO2−]: 0.6 ± 0.1 mM [NO3−]: 1.7 ± 0.3 mM | Corns of maize (Zea mays L.) (hybrid Bielik) | Enhanced seedling growth. It affected chlorophyll, carotenoid levels, and leaf arsenic accumulation, not root accumulation. | [156] |
| Glow discharge | Tap water | [H2O2]: ~0.33, 0.64, 1.00 mM [NO2−]: ~0.93, 0.59, 0.95 mM [NO3−]: ~2.46, 1.40, 2.34 mM For transient spark, PAW of glow discharge at activation time 1 min and 2 min, respectively. | Dried barley (Hordeum vulgare L. ‘Kangoo’) grains and pea (Pisum sativum L. ‘Eso’) seeds | Enhanced pea growth and amylase activity without harm, while barley suffered DNA damage, growth reduction, and oxidative stress. | [157] |
| Glow discharge. | Re-distilled water and the inorganic salt concentration (ammonium nitrate, NH4NO3) in the solution turned out to be 0.50% (m/w) | − | Dickeya solani IFB0099, Pectobacterium atrosepticum IFB5103 strains | Bactericidal effects on Dickeya solani and Pectobacterium atrosepticum within 24 h. | [158] |
| Transient spark | Tap water | pH: ~ 7.5 [H2O2]: ~ 0.42 mM [NO3−]: ~ 0.85 mM | Lettuce (Lactuca sativa L. var. capitata ‘Král máje I’) | PAW-irrigated lettuce had similar dry weight but higher pigment content, photosynthetic rate, and lower antioxidant enzyme activity compared to H2O2 + NO3− irrigation. | [159] |
| Glow discharge | Tap water | − | Poa pratensis, Lolium perenne | Decreased fungal diseases, improved turf density and overwintering. Synergistic effect with bio-stimulant. | [160] |
| Spark discharge | Deionized water | pH: 6.7, 6.4, 6.0, 5.3, 3.7 [H2O2]: ~60, 75, 100, 150, 240 µM [NO2−]: 25, 30, 50, 75, 230 µM For 3, 6, 9,12, and 15 min plasma treatment, respectively. | Black gram (Vigna mungo L.) | Increased H2O2 and ROS levels in seeds, leaves, and roots. Elevated catalase levels linked to VmCAT gene activation. | [161] |
| Glow discharge | 80 mM L-phenylalanine | [H2O2]: 282 µM [NO2−]: 4.4 µM [NO3−]: 520 µM after 4 min plasma treatment | Raphanus sp., E. coli O1:K1:H7 | Seedlings stimulation and antibacterial effect. | [162] |
| T-shaped reactor | Distilled water | pH: 4.6; 4.4; 5.1; 4.1; 5.1; 3.9; 5.5; 3.8 [NO3−]: 30, 40, 14, 49, 15, 55, 8, 68 ± 10% mg/L [H2O2]: 7, 8, 3, 13, 5, 14, 1, 22 ± 5% mg/L During treatment in the modes: Frequency: 150, 250, 60, 250, 60, 250, 250, 250 Hz; Duration: 2, 2, 2, 2, 1.5, 1.5, 2, 2 ms; Airflow: 1, 1, 1, 1, 1, 1, 2.5, 2.5 L/min; Liquid flow: 10, 10, 20, 3.33, 25, 3.33; 85, 4 L/min, respectively | Wheat (Triticum aestivum) grains | Reduced mitotic activity in wheat sprouts’ cells. Genotoxicity tied to H2O2, NO3− levels. Positive effect on wheat germination | [163] |
| Underwater discharge | Distilled water | pH: ~3.91 ± 0.03, [O3]: ~0.25 ± 0.01 mg/L [H2O2]: ~7.50 ± 0.05 mg/L [NO2−]: ~4.59 ± 0.04 mg/L [NO3−]: ~26.58 ± 0.25 mg/L | Potato (Solanum tuberosum L.) | PAW spraying on potato foliage enhanced growth, enzyme activity, protein and sugar content, and tuber yield. | [164] |
| Underwater discharge | Distilled water with KH2PO4 | [H2O2]: 100 μM | Spring spelled seeds, spring rye seeds | Improved germination, improved root development. | [165] |
| (1) Underwater discharge. (2) Plasma torch | (1) Distilled water + KCl (2) Distilled water | (1) pH: 8.3 [H2O2]: 7.12 mM [NOx−]: 22.05 mM (2) pH: 4.5 [H2O2]: 0.11 mM [NOx−]: 87 mM | Apple tree | Increased primary nutrient content in fruits and leaves. Ca in the fruits increased significantly. | [166] |
| Underwater discharge | Distilled water + KCl | pH: 8.3 [H2O2]: 7.12 mM [NOx−]: 22.05 mM | Sorghum and barley seeds, strawberry crops | Improved germination and crop development. Increased drought resistance. | [167] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konchekov, E.M.; Gusein-zade, N.; Burmistrov, D.E.; Kolik, L.V.; Dorokhov, A.S.; Izmailov, A.Y.; Shokri, B.; Gudkov, S.V. Advancements in Plasma Agriculture: A Review of Recent Studies. Int. J. Mol. Sci. 2023, 24, 15093. https://doi.org/10.3390/ijms242015093
Konchekov EM, Gusein-zade N, Burmistrov DE, Kolik LV, Dorokhov AS, Izmailov AY, Shokri B, Gudkov SV. Advancements in Plasma Agriculture: A Review of Recent Studies. International Journal of Molecular Sciences. 2023; 24(20):15093. https://doi.org/10.3390/ijms242015093
Chicago/Turabian StyleKonchekov, Evgeny M., Namik Gusein-zade, Dmitriy E. Burmistrov, Leonid V. Kolik, Alexey S. Dorokhov, Andrey Yu. Izmailov, Babak Shokri, and Sergey V. Gudkov. 2023. "Advancements in Plasma Agriculture: A Review of Recent Studies" International Journal of Molecular Sciences 24, no. 20: 15093. https://doi.org/10.3390/ijms242015093
APA StyleKonchekov, E. M., Gusein-zade, N., Burmistrov, D. E., Kolik, L. V., Dorokhov, A. S., Izmailov, A. Y., Shokri, B., & Gudkov, S. V. (2023). Advancements in Plasma Agriculture: A Review of Recent Studies. International Journal of Molecular Sciences, 24(20), 15093. https://doi.org/10.3390/ijms242015093







