Impact of Physical Rehabilitation on Bone Biomarkers in Non-Metastatic Breast Cancer Women: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Registration
2.2. Search Strategy
2.3. Selection Criteria
- (P) Participants: adult women (18 years and older) with non-metastatic BC.
- Intervention: any rehabilitation treatment administered before, during, or after chemotherapy and/or radiotherapy treatments.
- (C) Comparator: any comparator including pharmacological, non-pharmacological, or no treatment.
- (O) Outcome: primary outcomes were bone metabolic biomarkers. Secondary outcomes were other bone health outcomes, including bone mineral density or trabecular bone score.
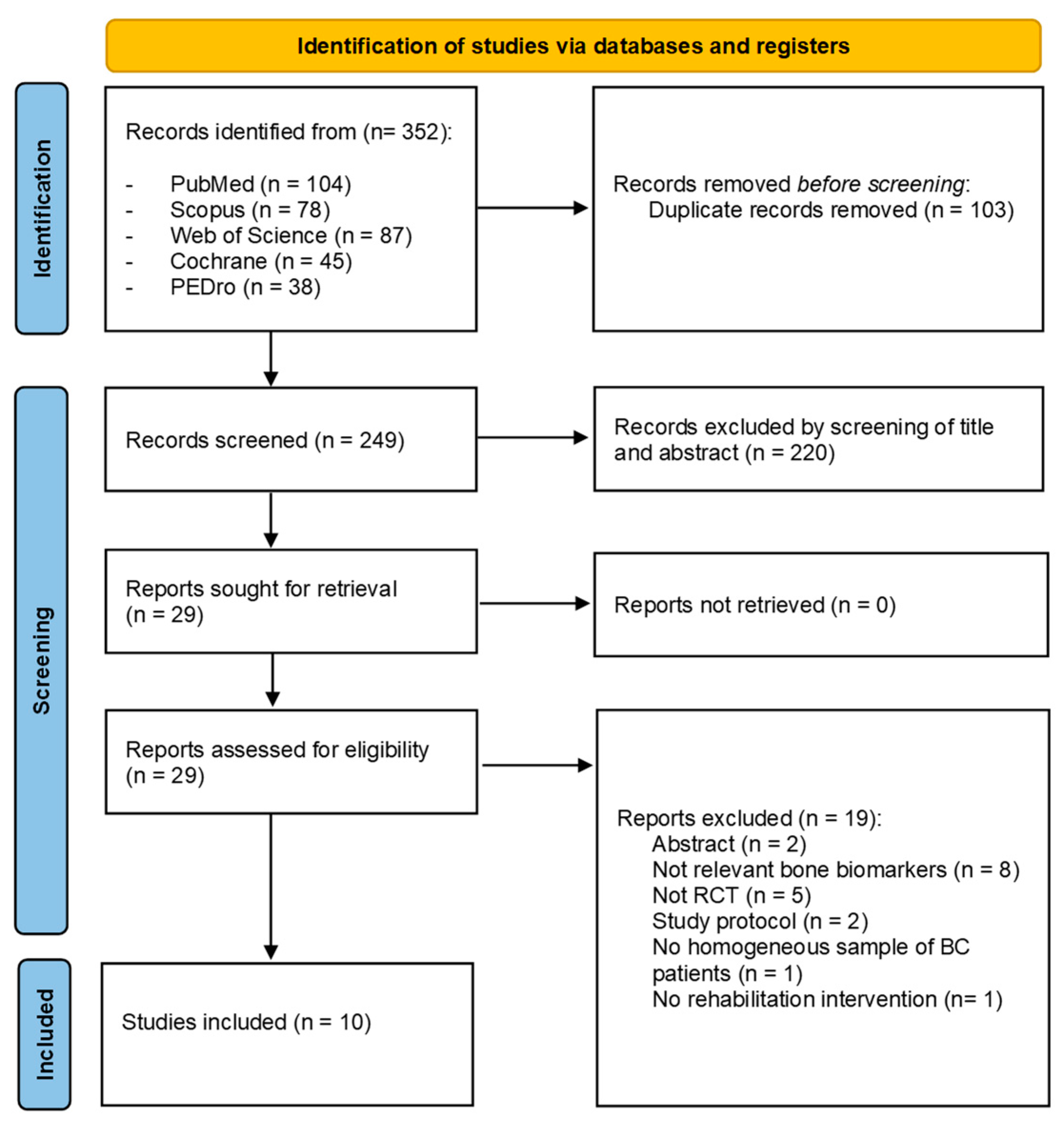
2.4. Study Screening and Eligibility Assessment
2.5. Data Extraction and Synthesis
2.6. Meta-Analysis
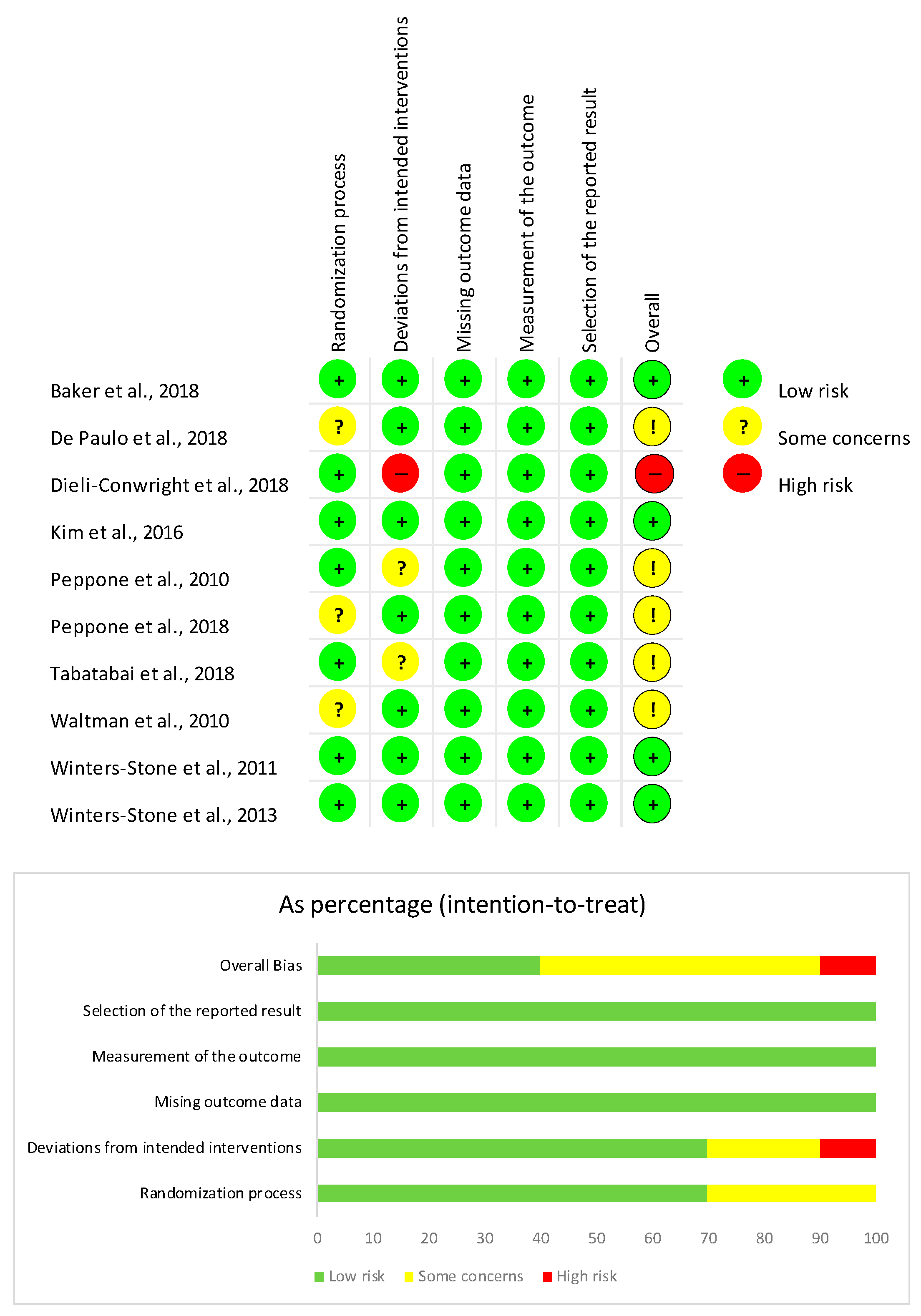
2.7. Quality Assessment and Risk of Bias
3. Results
3.1. Study Characteristics
3.2. Participants
3.3. Control Groups
3.4. Rehabilitation Therapy Interventions
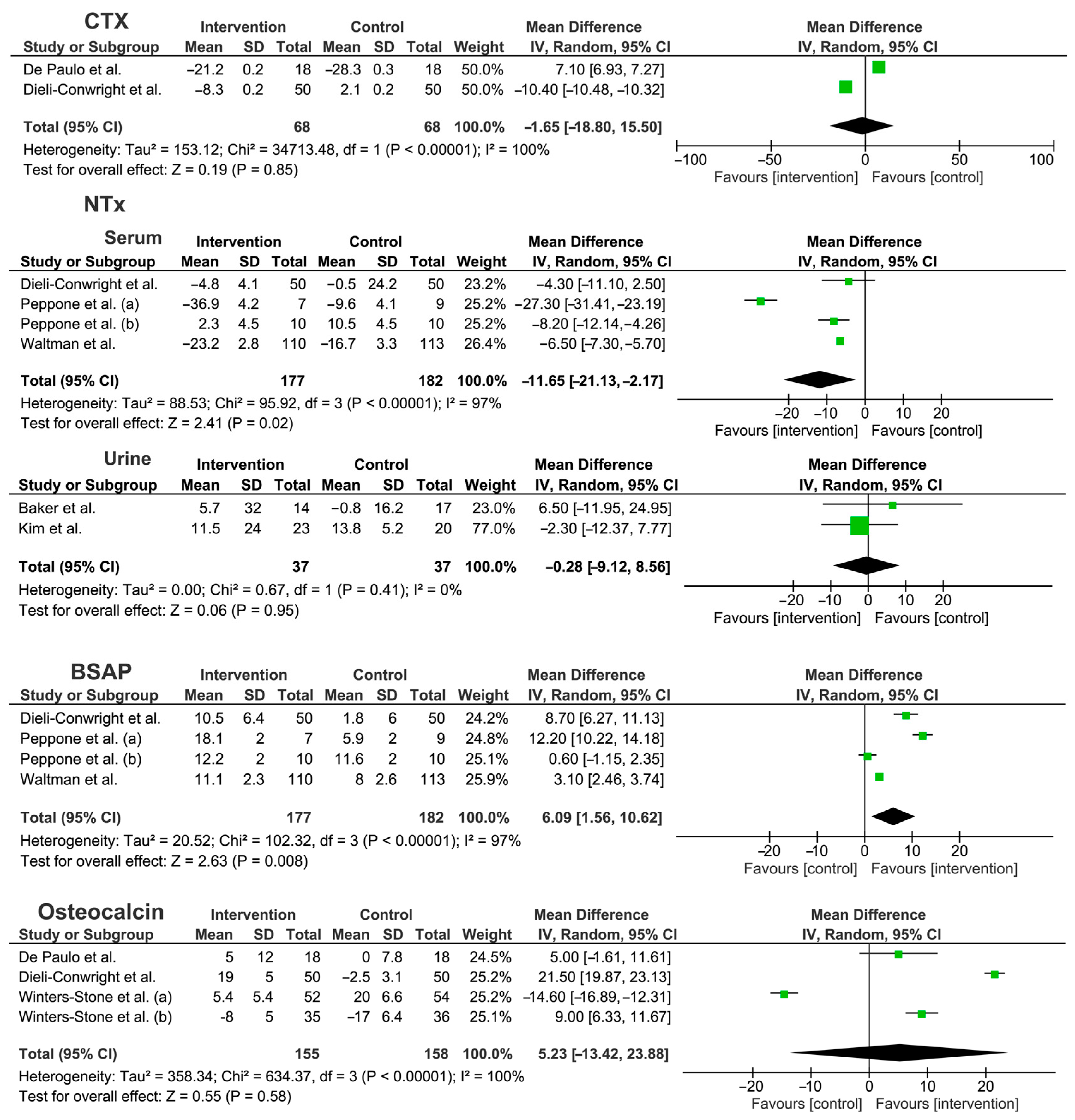
3.5. Primary Outcome—Bone Biomarkers Modifications
- Collagen type 1 cross-linked N-telopeptide (NTX) was assessed in four studies [33,35,36,38], but significant changes were reported only in one study [38]. In particular, Waltman et al. [38] reported significant changes (p < 0.05) in both the intervention group (RET + risedronate, calcium, and Vitamin D) and the control group (risedronate, calcium, and vitamin D).
- Osteocalcin was assessed in five papers [32,33,37,39,40]; out of these, two studies [32,33] reported a significant increase (both p < 0.05) in the intervention group after CET, while Winters-Stone et al., 2011, [39] reported a significative inter-group difference after RET combined with IET (p = 0.01). Lastly, Tabatabai et al. [37] reported a significant decrease in both intervention (CET) and control groups.
- Bone-specific alkaline phosphatase (BSAP) was assessed in four papers [33,35,36,38]; among these studies, Dieli-Conwright et al. [33] reported a significant increase in serum concentration in the CET intervention group compared to intervention; concurrently, Waltman et al. [38] reported a reduction in both the intervention group (RET + risedronate, calcium, and Vitamin D) and control group (risedronate, calcium, and Vitamin D). The remaining studies did not report a significant modification of BSAP values (p > 0.05).
- Receptor activator of nuclear factor (RANK) was assessed by Dieli-Conwright et al. [33], but the study did not report significant changes (p > 0.05).
- Receptor activator of nuclear factor ligand (RANKL) was assessed by Dieli-Conwright et al. [33], without reporting significant changes (p > 0.05).
3.6. Secondary Outcomes—Bone Mineral Density
- Lumbar spine BMD was assessed in seven studies [32,33,34,37,38,39,40]; more in-detail, Winters-Stone et al., 2011, [39] underlined significant changes (p < 0.01) in the intergroup analysis after RET combined with IET intervention after 12 months. Similarly, Waltman et al. [38] reported a percentage mean difference significant both in the intervention group (RET + risedronate, calcium, and vitamin D) and the control group (risedronate, calcium, and vitamin D) (both p < 0.05). Interestingly, Tabatabai et al. [37] reported a significant mean decrease in the control group, which received only a monthly health newsletter (p = 0.03).
- Radius (33% length) BMD was assessed by Waltman et al. [38], without reporting significant changes.
- Total radius BMD was assessed by Waltman et al. [38], with no significant changes after the intervention.
3.7. Meta-Analysis
3.8. Quality Assessment and Risk of Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Lima, S.M.; Kehm, R.D.; Terry, M.B. Global breast cancer incidence and mortality trends by region, age-groups, and fertility patterns. EClinicalMedicine 2021, 38, 100985. [Google Scholar] [CrossRef] [PubMed]
- Nardin, S.; Mora, E.; Varughese, F.M.; D’Avanzo, F.; Vachanaram, A.R.; Rossi, V.; Saggia, C.; Rubinelli, S.; Gennari, A. Breast Cancer Survivorship, Quality of Life, and Late Toxicities. Front. Oncol. 2020, 10, 864. [Google Scholar] [CrossRef] [PubMed]
- Paolucci, T.; Bernetti, A.; Bai, A.V.; Segatori, L.; Monti, M.; Maggi, G.; Ippolitoni, G.; Tinelli, L.; Santilli, V.; Paoloni, M. The sequelae of mastectomy and quadrantectomy with respect to the reaching movement in breast cancer survivors: Evidence for an integrated rehabilitation protocol during oncological care. Support Care Cancer. 2021, 29, 899–908. [Google Scholar] [CrossRef] [PubMed]
- de Sire, A.; Losco, L.; Cisari, C.; Gennari, A.; Boldorini, R.; Fusco, N.; Cigna, E.; Invernizzi, M. Axillary web syndrome in women after breast cancer surgery referred to an Oncological Rehabilitation Unit: Which are the main risk factors? A retrospective case-control study. Eur Rev Med Pharmacol Sci. 2020, 24, 8028–8035. [Google Scholar] [CrossRef]
- Senkus, E.; Kyriakides, S.; Ohno, S.; Penault-Llorca, F.; Poortmans, P.; Rutgers, E.; Zackrisson, S.; Cardoso, F.; Committee, E.G. Primary breast cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2015, 26 (Suppl. S5), v8–v30. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Aromatase inhibitors versus tamoxifen in early breast cancer: Patient-level meta-analysis of the randomised trials. Lancet 2015, 386, 1341–1352. [Google Scholar] [CrossRef]
- Burstein, H.J.; Temin, S.; Anderson, H.; Buchholz, T.A.; Davidson, N.E.; Gelmon, K.E.; Giordano, S.H.; Hudis, C.A.; Rowden, D.; Solky, A.J.; et al. Adjuvant endocrine therapy for women with hormone receptor-positive breast cancer: American society of clinical oncology clinical practice guideline focused update. J. Clin. Oncol. 2014, 32, 2255–2269. [Google Scholar] [CrossRef]
- Diana, A.; Carlino, F.; Giunta, E.F.; Franzese, E.; Guerrera, L.P.; Di Lauro, V.; Ciardiello, F.; Daniele, B.; Orditura, M. Cancer Treatment-Induced Bone Loss (CTIBL): State of the Art and Proper Management in Breast Cancer Patients on Endocrine Therapy. Curr. Treat. Options Oncol. 2021, 22, 45. [Google Scholar] [CrossRef]
- Hadji, P.; Coleman, R.E.; Wilson, C.; Powles, T.J.; Clezardin, P.; Aapro, M.; Costa, L.; Body, J.J.; Markopoulos, C.; Santini, D.; et al. Adjuvant bisphosphonates in early breast cancer: Consensus guidance for clinical practice from a European Panel. Ann. Oncol. 2016, 27, 379–390. [Google Scholar] [CrossRef]
- Migliaccio, S.; Francomano, D.; Romagnoli, E.; Marocco, C.; Fornari, R.; Resmini, G.; Buffa, A.; Di Pietro, G.; Corvaglia, S.; Gimigliano, F.; et al. Persistence with denosumab therapy in women affected by osteoporosis with fragility fractures: A multicenter observational real practice study in Italy. J. Endocrinol. Invest. 2017, 40, 1321–1326. [Google Scholar] [CrossRef] [PubMed]
- Guise, T.A. Bone Loss and Fracture Risk Associated with Cancer Therapy. Oncologist 2006, 11, 1121–1131. [Google Scholar] [CrossRef] [PubMed]
- Kobza, A.O.; Herman, D.; Papaioannou, A.; Lau, A.N.; Adachi, J.D. Understanding and Managing Corticosteroid-Induced Osteoporosis. Open Access Rheumatol. Res. Rev. 2021, 13, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.M.; Heisey, R.; Srighanthan, J. Breast cancer and osteoporosis. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 532–538. [Google Scholar] [CrossRef]
- de Sire, A.; Ferrillo, M.; Gennari, A.; Cisari, C.; Pasqua, S.; Foglio Bonda, P.L.; Invernizzi, M.; Migliario, M. Bone health, vitamin D status and oral hygiene screening in breast cancer women before starting osteoporosis treatment: A cross-sectional study. J. Biol. Regul. Homeost. Agents 2021, 35, 397–402. [Google Scholar] [CrossRef]
- de Sire, A.; Lippi, L.; Venetis, K.; Morganti, S.; Sajjadi, E.; Curci, C.; Ammendolia, A.; Criscitiello, C.; Fusco, N.; Invernizzi, M. Efficacy of Antiresorptive Drugs on Bone Mineral Density in Post-Menopausal Women With Early Breast Cancer Receiving Adjuvant Aromatase Inhibitors: A Systematic Review of Randomized Controlled Trials. Front. Oncol. 2021, 11, 829875. [Google Scholar] [CrossRef] [PubMed]
- Coleman, R.; Hadji, P.; Body, J.J.; Santini, D.; Chow, E.; Terpos, E.; Oudard, S.; Bruland, O.; Flamen, P.; Kurth, A.; et al. Bone health in cancer: ESMO Clinical Practice Guidelines. Ann. Oncol. 2020, 31, 1650–1663. [Google Scholar] [CrossRef]
- Dhesy-Thind, S.; Fletcher, G.G.; Blanchette, P.S.; Clemons, M.J.; Dillmon, M.S.; Frank, E.S.; Gandhi, S.; Gupta, R.; Mates, M.; Moy, B.; et al. Use of Adjuvant Bisphosphonates and Other Bone-Modifying Agents in Breast Cancer: A Cancer Care Ontario and American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2017, 35, 2062–2081. [Google Scholar] [CrossRef] [PubMed]
- Duffy, M.J.; Mullooly, M.; Bennett, K.; Crown, J. Vitamin D Supplementation: Does It Have a Preventative or Therapeutic Role in Cancer? Nutr Cancer. 2022, 10, 1–11. [Google Scholar] [CrossRef]
- Benedetti, M.G.; Furlini, G.; Zati, A.; Letizia Mauro, G. The Effectiveness of Physical Exercise on Bone Density in Osteoporotic Patients. BioMed Res. Int. 2018, 2018, 4840531. [Google Scholar] [CrossRef]
- Hong, A.R.; Kim, S.W. Effects of Resistance Exercise on Bone Health. Endocrinol. Metab. 2018, 33, 435. [Google Scholar] [CrossRef]
- Kemmler, W.; Shojaa, M.; Kohl, M.; von Stengel, S. Effects of Different Types of Exercise on Bone Mineral Density in Postmenopausal Women: A Systematic Review and Meta-analysis. Calcif. Tissue Int. 2020, 107, 409–439. [Google Scholar] [CrossRef] [PubMed]
- Invernizzi, M.; Kim, J.; Fusco, N. Editorial: Quality of Life in Breast Cancer Patients and Survivors. Front. Oncol. 2020, 10, 620574. [Google Scholar] [CrossRef] [PubMed]
- Criscitiello, C.; Guerini-Rocco, E.; Viale, G.; Fumagalli, C.; Sajjadi, E.; Venetis, K.; Piciotti, R.; Invernizzi, M.; Malapelle, U.; Fusco, N. Immunotherapy in Breast Cancer Patients: A Focus on the Use of the Currently Available Biomarkers in Oncology. Anticancer Agents Med. Chem. 2022, 22, 787–800. [Google Scholar] [CrossRef] [PubMed]
- Ballinger, T.J.; Althouse, S.K.; Olsen, T.P.; Miller, K.D.; Sledge, J.S. A Personalized, Dynamic Physical Activity Intervention Is Feasible and Improves Energetic Capacity, Energy Expenditure, and Quality of Life in Breast Cancer Survivors. Front. Oncol. 2021, 11, 626180. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Huang, X.; Lin, J.; Demner-Fushman, D. Evaluation of PICO as a knowledge representation for clinical questions. AMIA Annu. Symp. Proc. 2006, 2006, 359–363. [Google Scholar]
- Hozo, S.P.; Djulbegovic, B.; Hozo, I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med. Res. Methodol. 2005, 5, 13. [Google Scholar] [CrossRef]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savovic, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Baker, M.K.; Peddle-McIntyre, C.J.; Galvão, D.A.; Hunt, C.; Spry, N.; Newton, R.U. Whole Body Vibration Exposure on Markers of Bone Turnover, Body Composition, and Physical Functioning in Breast Cancer Patients Receiving Aromatase Inhibitor Therapy: A Randomized Controlled Trial. Integr. Cancer Ther. 2018, 17, 968–978. [Google Scholar] [CrossRef] [PubMed]
- de Paulo, T.R.S.; Winters-Stone, K.M.; Viezel, J.; Rossi, F.E.; Simões, R.R.; Tosello, G.; Freitas, I.F.J. Effects of resistance plus aerobic training on body composition and metabolic markers in older breast cancer survivors undergoing aromatase inhibitor therapy. Exp. Gerontol. 2018, 111, 210–217. [Google Scholar] [CrossRef]
- Dieli-Conwright, C.M.; Courneya, K.S.; Demark-Wahnefried, W.; Sami, N.; Lee, K.; Sweeney, F.C.; Stewart, C.; Buchanan, T.A.; Spicer, D.; Tripathy, D.; et al. Aerobic and resistance exercise improves physical fitness, bone health, and quality of life in overweight and obese breast cancer survivors: A randomized controlled trial. Breast Cancer Res. 2018, 20, 124. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Cho, Y.U.; Kim, S.J.; Hong, S.; Han, M.S.; Choi, E. The Effect on Bone Outcomes of Adding Exercise to Supplements for Osteopenic Breast Cancer Survivors: A Pilot Randomized Controlled Trial. Cancer Nurs. 2016, 39, 144–152. [Google Scholar] [CrossRef] [PubMed]
- Peppone, L.J.; Ling, M.; Huston, A.J.; Reid, M.E.; Janelsins, M.C.; Puzas, J.E.; Kamen, C.; Del Giglio, A.; Asare, M.; Peoples, A.R.; et al. The effects of high-dose calcitriol and individualized exercise on bone metabolism in breast cancer survivors on hormonal therapy: A phase II feasibility trial. Support. Care Cancer 2018, 26, 2675–2683. [Google Scholar] [CrossRef] [PubMed]
- Peppone, L.J.; Mustian, K.M.; Janelsins, M.C.; Palesh, O.G.; Rosier, R.N.; Piazza, K.M.; Purnell, J.Q.; Darling, T.V.; Morrow, G.R. Effects of a structured weight-bearing exercise program on bone metabolism among breast cancer survivors: A feasibility trial. Clin. Breast Cancer 2010, 10, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Tabatabai, L.S.; Bloom, J.; Stewart, S.; Sellmeyer, D.E. A Randomized Controlled Trial of Exercise to Prevent Bone Loss in Premenopausal Women with Breast Cancer. J. Women’s Health 2019, 28, 87–92. [Google Scholar] [CrossRef]
- Waltman, N.L.; Twiss, J.J.; Ott, C.D.; Gross, G.J.; Lindsey, A.M.; Moore, T.E.; Berg, K.; Kupzyk, K. The effect of weight training on bone mineral density and bone turnover in postmenopausal breast cancer survivors with bone loss: A 24-month randomized controlled trial. Osteoporos. Int. 2010, 21, 1361–1369. [Google Scholar] [CrossRef]
- Winters-Stone, K.M.; Dobek, J.; Nail, L.; Bennett, J.A.; Leo, M.C.; Naik, A.; Schwartz, A. Strength training stops bone loss and builds muscle in postmenopausal breast cancer survivors: A randomized, controlled trial. Breast Cancer Res. Treat. 2011, 127, 447–456. [Google Scholar] [CrossRef]
- Winters-Stone, K.M.; Dobek, J.; Nail, L.M.; Bennett, J.A.; Leo, M.C.; Torgrimson-Ojerio, B.; Luoh, S.W.; Schwartz, A. Impact + resistance training improves bone health and body composition in prematurely menopausal breast cancer survivors: A randomized controlled trial. Osteoporos. Int. 2013, 24, 1637–1646. [Google Scholar] [CrossRef]
- de Sire, A.; Lippi, L.; Ammendolia, A.; Cisari, C.; Venetis, K.; Sajjadi, E.; Fusco, N.; Invernizzi, M. Physical Exercise with or without Whole-Body Vibration in Breast Cancer Patients Suffering from Aromatase Inhibitor-Induced Musculoskeletal Symptoms: A Pilot Randomized Clinical Study. J. Pers. Med. 2021, 11, 1369. [Google Scholar] [CrossRef] [PubMed]
- Paolucci, T.; Bernetti, A.; Bai, A.V.; Capobianco, S.V.; Bonifacino, A.; Maggi, G.; Ippolitoni, G.; Tinelli, L.; Santilli, V.; Agostini, F. The recovery of reaching movement in breast cancer survivors: Two different rehabilitative protocols in comparison. Eur. J. Phys. Rehabil. Med. 2021, 57, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Moayyeri, A. The association between physical activity and osteoporotic fractures: A review of the evidence and implications for future research. Ann. Epidemiol. 2008, 18, 827–835. [Google Scholar] [CrossRef]
- de Kam, D.; Smulders, E.; Weerdesteyn, V.; Smits-Engelsman, B.C. Exercise interventions to reduce fall-related fractures and their risk factors in individuals with low bone density: A systematic review of randomized controlled trials. Osteoporos. Int. 2009, 20, 2111–2125. [Google Scholar] [CrossRef] [PubMed]
- Suominen, H. Muscle training for bone strength. Aging Clin. Exp. Res. 2006, 18, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Hlaing, T.T.; Compston, J.E. Biochemical markers of bone turnover–uses and limitations. Ann. Clin. Biochem. 2014, 51, 189–202. [Google Scholar] [CrossRef]
- Bauer, D.C.; Garnero, P.; Harrison, S.L.; Cauley, J.A.; Eastell, R.; Ensrud, K.E.; Orwoll, E. Biochemical Markers of Bone Turnover, Hip Bone Loss, and Fracture in Older Men: The MrOS Study. J. Bone Miner. Res. 2009, 24, 2032–2038. [Google Scholar] [CrossRef] [PubMed]
- Christenson, R.H. Biochemical markers of bone metabolism: An overview. Clin. Biochem. 1997, 30, 573–593. [Google Scholar] [CrossRef]
- Taylor, A.K.; Lueken, S.A.; Libanati, C.; Baylink, D.J. Biochemical markers of bone turnover for the clinical assessment of bone metabolism. Rheum. Dis. Clin. N. Am. 1994, 20, 589–607. [Google Scholar] [CrossRef]
- Nair, A.; Chuang, S.C.; Lin, Y.S.; Chen, C.H.; Fang, T.C.; Chiu, H.C.; Lien, C.H.; Chen, S.J. Characterization of collagen response to bone fracture healing using polarization-SHG. Sci. Rep. 2022, 12, 18453. [Google Scholar] [CrossRef]
- Roccuzzo, A.; Molinero-Mourelle, P.; Ferrillo, M.; Cobo-Vázquez, C.; Sanchez-Labrador, L.; Ammendolia, A.; Migliario, M.; de Sire, A. Type I Collagen-Based Devices to Treat Nerve Injuries after Oral Surgery Procedures. A Systematic Review. Appl.Sci. 2021, 11, 3927. [Google Scholar] [CrossRef]
- Migliorini, F.; Maffulli, N.; Spiezia, F.; Peretti, G.M.; Tingart, M.; Giorgino, R. Potential of biomarkers during pharmacological therapy setting for postmenopausal osteoporosis: A systematic review. J. Orthop. Surg. Res. 2021, 16, 351. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.; Krege, J.; Lane, N.; Leary, E.; Libanati, C.; Miller, P.; Myers, G.; Silverman, S.; Vesper, H.W.; Lee, D.; et al. National Bone Health Alliance Bone Turnover Marker Project: Current practices and the need for US harmonization, standardization, and common reference ranges. Osteoporos. Int. 2012, 23, 2425–2433. [Google Scholar] [CrossRef]
- Vasikaran, S.; Eastell, R.; Bruyère, O.; Foldes, A.J.; Garnero, P.; Griesmacher, A.; McClung, M.; Morris, H.A.; Silverman, S.; Trenti, T.; et al. Markers of bone turnover for the prediction of fracture risk and monitoring of osteoporosis treatment: A need for international reference standards. Osteoporos. Int. 2011, 22, 391–420. [Google Scholar] [CrossRef] [PubMed]
- Lee, N.K.; Sowa, H.; Hinoi, E.; Ferron, M.; Ahn, J.D.; Confavreux, C.; Dacquin, R.; Mee, P.J.; McKee, M.D.; Jung, D.Y.; et al. Endocrine regulation of energy metabolism by the skeleton. Cell 2007, 130, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Lacombe, J.; Ferron, M. Gamma-carboxylation regulates osteocalcin function. Oncotarget 2015, 6, 19924–19925. [Google Scholar] [CrossRef]
- Karsenty, G.; Ferron, M. The contribution of bone to whole-organism physiology. Nature 2012, 481, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Sattler, A.M.; Schoppet, M.; Schaefer, J.R.; Hofbauer, L.C. Novel aspects on RANK ligand and osteoprotegerin in osteoporosis and vascular disease. Calcif. Tissue Int. 2004, 74, 103–106. [Google Scholar] [CrossRef]
- Gnant, M.; Pfeiler, G.; Steger, G.G.; Egle, D.; Greil, R.; Fitzal, F.; Wette, V.; Balic, M.; Haslbauer, F.; Melbinger-Zeinitzer, E.; et al. Adjuvant denosumab in postmenopausal patients with hormone receptor-positive breast cancer (ABCSG-18): Disease-free survival results from a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. Oncol. 2019, 20, 339–351. [Google Scholar] [CrossRef]
- Gnant, M.; Pfeiler, G.; Dubsky, P.C.; Hubalek, M.; Greil, R.; Jakesz, R.; Wette, V.; Balic, M.; Haslbauer, F.; Melbinger, E.; et al. Adjuvant denosumab in breast cancer (ABCSG-18): A multicentre, randomised, double-blind, placebo-controlled trial. Lancet 2015, 386, 433–443. [Google Scholar] [CrossRef]
- Ellis, G.K.; Bone, H.G.; Chlebowski, R.; Paul, D.; Spadafora, S.; Smith, J.; Fan, M.; Jun, S. Randomized trial of denosumab in patients receiving adjuvant aromatase inhibitors for nonmetastatic breast cancer. J. Clin. Oncol. 2008, 26, 4875–4882. [Google Scholar] [CrossRef]
- Sandino, C.; McErlain, D.D.; Schipilow, J.; Boyd, S.K. Mechanical stimuli of trabecular bone in osteoporosis: A numerical simulation by finite element analysis of microarchitecture. J. Mech. Behav. Biomed. Mater. 2017, 66, 19–27. [Google Scholar] [CrossRef]
- Ott, S.M. Cortical or Trabecular Bone: What’s the Difference? Am. J. Nephrol. 2018, 47, 373–375. [Google Scholar] [CrossRef]
- Howe, T.E.; Shea, B.; Dawson, L.J.; Downie, F.; Murray, A.; Ross, C.; Harbour, R.T.; Caldwell, L.M.; Creed, G. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst. Rev. 2011, 7, Cd000333. [Google Scholar] [CrossRef] [PubMed]
- Marini, S.; Barone, G.; Masini, A.; Dallolio, L.; Bragonzoni, L.; Longobucco, Y.; Maffei, F. Current Lack of Evidence for an Effect of Physical Activity Intervention Combined with Pharmacological Treatment on Bone Turnover Biomarkers in People with Osteopenia and Osteoporosis: A Systematic Review. J. Clin. Med. 2021, 10, 3442. [Google Scholar] [CrossRef] [PubMed]
- Daly, R.M.; Dalla Via, J.; Duckham, R.L.; Fraser, S.F.; Helge, E.W. Exercise for the prevention of osteoporosis in postmenopausal women: An evidence-based guide to the optimal prescription. Braz. J. Phys. 2019, 23, 170–180. [Google Scholar] [CrossRef] [PubMed]
- Kast, S.; Shojaa, M.; Kohl, M.; Von Stengel, S.; Gosch, M.; Jakob, F.; Kerschan-Schindl, K.; Kladny, B.; Klöckner, N.; Lange, U.; et al. Effects of different exercise intensity on bone mineral density in adults: A comparative systematic review and meta-analysis. Osteoporos. Int. 2022, 33, 1643–1657. [Google Scholar] [CrossRef] [PubMed]
- Massini, D.A.; Nedog, F.H.; De Oliveira, T.P.; Almeida, T.A.F.; Santana, C.A.A.; Neiva, C.M.; Macedo, A.G.; Castro, E.A.; Espada, M.C.; Santos, F.J.; et al. The Effect of Resistance Training on Bone Mineral Density in Older Adults: A Systematic Review and Meta-Analysis. Healthcare 2022, 10, 1129. [Google Scholar] [CrossRef]
- Williamson, S.; Landeiro, F.; McConnell, T.; Fulford-Smith, L.; Javaid, M.K.; Judge, A.; Leal, J. Costs of fragility hip fractures globally: A systematic review and meta-regression analysis. Osteoporos. Int. 2017, 28, 2791–2800. [Google Scholar] [CrossRef] [PubMed]
- de Sire, A.; Invernizzi, M.; Baricich, A.; Lippi, L.; Ammendolia, A.; Grassi, F.A.; Leigheb, M. Optimization of transdisciplinary management of elderly with femur proximal extremity fracture: A patient-tailored plan from orthopaedics to rehabilitation. World J. Orthop. 2021, 12, 456–466. [Google Scholar] [CrossRef]
- Muschitz, C.; Hummer, M.; Grillari, J.; Hlava, A.; Birner, A.H.; Hemetsberger, M.; Dimai, H.P. Epidemiology and economic burden of fragility fractures in Austria. Osteoporos. Int. 2022, 33, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Saarto, T.; Sievänen, H.; Kellokumpu-Lehtinen, P.; Nikander, R.; Vehmanen, L.; Huovinen, R.; Kautiainen, H.; Järvenpää, S.; Penttinen, H.M.; Utriainen, M.; et al. Effect of supervised and home exercise training on bone mineral density among breast cancer patients. A 12-month randomised controlled trial. Osteoporos. Int. 2012, 23, 1601–1612. [Google Scholar] [CrossRef] [PubMed]
- Vehmanen, L.; Sievänen, H.; Kellokumpu-Lehtinen, P.; Nikander, R.; Huovinen, R.; Ruohola, J.; Penttinen, H.M.; Utriainen, M.; Tokola, K.; Blomqvist, C.; et al. Five-year follow-up results of aerobic and impact training on bone mineral density in early breast cancer patients. Osteoporos. Int. 2021, 32, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Longobucco, Y.; Masini, A.; Marini, S.; Barone, G.; Fimognari, C.; Bragonzoni, L.; Dallolio, L.; Maffei, F. Exercise and Oxidative Stress Biomarkers among Adult with Cancer: A Systematic Review. Oxidative Med. Cell. Longev. 2022, 2022, 2097318. [Google Scholar] [CrossRef]
- Delrieu, L.; Touillaud, M.; Pérol, O.; Morelle, M.; Martin, A.; Friedenreich, C.M.; Mury, P.; Dufresne, A.; Bachelot, T.; Heudel, P.-E.; et al. Impact of Physical Activity on Oxidative Stress Markers in Patients with Metastatic Breast Cancer. Oxidative Med. Cell. Longev. 2021, 2021, 6694594. [Google Scholar] [CrossRef]
- Wennerberg, E.; Lhuillier, C.; Rybstein, M.D.; Dannenberg, K.; Rudqvist, N.-P.; Koelwyn, G.J.; Jones, L.W.; Demaria, S. Exercise reduces immune suppression and breast cancer progression in a preclinical model. Oncotarget 2020, 11, 452–461. [Google Scholar] [CrossRef]
- Eisen, A.; Somerfield, M.R.; Accordino, M.K.; Blanchette, P.S.; Clemons, M.J.; Dhesy-Thind, S.; Dillmon, M.S.; D’Oronzo, S.; Fletcher, G.G.; Frank, E.S.; et al. Use of Adjuvant Bisphosphonates and Other Bone-Modifying Agents in Breast Cancer: ASCO-OH (CCO) Guideline Update. J. Clin. Oncol. 2022, 40, 787–800. [Google Scholar] [CrossRef]

| PubMed: | |
|---|---|
| ((“breast neoplasms” [MeSH Terms] OR (“breast” [All Fields] AND “neoplasms” [All Fields]) OR “breast neoplasms” [All Fields] OR (“breast” [All Fields] AND “cancer” [All Fields]) OR “breast cancer” [All Fields] OR “breast tumor” [MeSH Terms] OR (“breast” [All Fields] AND “tumor” [All Fields]) OR “breast tumor” [All Fields])) AND ((“rehabilitant” [All Fields] OR “rehabilitants” [All Fields] OR “rehabilitate” [All Fields] OR “rehabilitated” [All Fields] OR “rehabilitates” [All Fields] OR “rehabilitating” [All Fields] OR “rehabilitation” [MeSH Terms] OR “rehabilitation” [All Fields] OR “rehabilitations” [All Fields] OR “rehabilitative” [All Fields] OR “rehabilitation” [MeSH Subheading] OR “rehabilitation s” [All Fields] OR “rehabilitational” [All Fields] OR “rehabilitator” [All Fields] OR “rehabilitators” [All Fields] OR “exercise” [MeSH Terms] OR “exercise” [All Fields] OR (“physical” [All Fields] AND “exercise” [All Fields]) OR “physical exercise” [All Fields] OR “training” [All Fields] OR “train” [All Fields] OR “train s” [All Fields] OR “trained” [All Fields] OR “training s” [All Fields] OR “trainings” [All Fields] OR “trains” [All Fields])) AND ((“bone and bones” [MeSH Terms] OR (“bone” [All Fields] AND “bones” [All Fields]) OR “bone and bones” [All Fields] OR “bone” [All Fields]) AND (“biomarker s” [All Fields] OR “biomarkers” [MeSH Terms] OR “biomarkers” [All Fields] OR “biomarker” [All Fields]) OR (“bone remodeling” [MeSH Terms] OR (“bone” [All Fields] AND “remodeling” [All Fields]) OR “bone remodeling” [All Fields] OR (“bone” [All Fields] AND “turnover” [All Fields]) OR “bone turnover” [All Fields] OR “osteogenesis” [MeSH Terms] OR “osteogenesis” [All Fields] OR (“bone” [All Fields] AND “formation” [All Fields]) OR “bone formation” [All Fields] OR “bone resorption” [MeSH Terms] OR (“bone” [All Fields] AND “resorption” [All Fields]) OR “bone resorption” [All Fields])) | |
| Scopus: | |
| TITLE-ABS-KEY (((((((breast AND neoplasms) OR (breast AND cancer) OR (breast AND tumor)) AND ((rehabilitation) OR (rehabilitation AND therapy) OR (exercise) OR (physical AND exercise)) AND ((bone AND (biomarker OR biomarkers)) OR (bone AND remodelling) OR (bone AND turnover))))))) | |
| Web of Science: | |
| (((((breast neoplasms) OR (breast cancer) OR (breast tumor)) AND ((rehabilitation) OR (rehabilitation therapy) OR (exercise) OR (physical exercise)) AND ((bone biomarker) OR (bone biomarkers) OR (bone remodelling) OR (bone turnover))))) | |
| Cochrane: | |
| ID | Search |
| #1 | MeSH descriptor: [breast neoplasms] explode all trees |
| #2 | MeSH descriptor: [breast cancer] explode all trees |
| #3 | MeSH descriptor: [breast tumor] explode all trees |
| #4 | MeSH descriptor: [rehabilitation] explode all trees |
| #5 | MeSH descriptor: [rehabilitation therapy] explode all trees |
| #6 | MeSH descriptor: [exercise] explode all trees |
| #7 | MeSH descriptor: [physical exercise] explode all trees |
| #8 | MeSH descriptor: [bone biomarker] explode all trees |
| #9 | MeSH descriptor: [bone biomarkers] explode all trees |
| #10 | MeSH descriptor: [bone remodelling] explode all trees |
| #11 | MeSH descriptor: [bone turnover] explode all trees |
| #12 | (#1 OR #2 OR #3) AND (#4 OR #5 OR #6 OR #7) AND (#8 OR #10 OR #11) |
| PEDro: | |
| breast cancer, exercise, bone * | |
| Authors Journal Year | Participants | Cancer Treatments | ||||||
|---|---|---|---|---|---|---|---|---|
| Sample Size | Age (Years) | BMI (kg/m2) | Cancer Characteristics | Breast Surgery | Chemotherapy | Radiation Therapy | Hormonal Therapy | |
| Baker et al. Integr. Cancer Ther. 2018 [31] | N = 31 IG: 14 CG: 17 | N = 61.6 ± 8.3 IG: 61.6 ± 9.2 CG: 61.6 ± 7.8 | N = 28.8 ± 4.7 IG: 28.4 ± 3.9 CG: 29.1 ± 5.3 | Non-metastatic breast cancer | NR | NR | NR | 100% |
| De Paulo et al. Exp. Gerontol. 2018 [32] | N = 36 IG: 18 CG: 18 | N = NR IG: 63.2 ± 7.1 CG: 66.6 ± 9.6 | N = NR IG: 28.9 ± 5.2 CG: 31.5 ± 6.3 | Stage I IG: 50% CG: 58.8% Stage II IG: 33.3% CG: 23.6% Stage IIIA IG: 17.6% CG: 17.6% | Partial Mastectomy IG: 47.4% CG: 58.8% Total Mastectomy IG: 52.6% CG: 41.2% | NR | NR | 100% |
| Dieli-Conwright et al. Breast Cancer Res. 2018 [33] | N = 100 IG: 50 CG: 50 | N = 53.5 ± 10.4 IG: NR CG: NR | N = 33.5 ± 5.5 IG: NR CG: NR | Stage I: 40% Stage II: 38% | NR | Chemotherapy and radiotherapy (76%) | Chemotherapy and radiotherapy (76%) | NR |
| Kim et al. Cancer Nurs. 2016 [34] | N = 43 IG: 23 CG: 20 | N = NR IG: 55.7 ± 5.3 CG: 56.3 ± 6.7 | N = NR IG: 23.3 ± 4.3 CG: 23.4 ± 2.5 | Stage 0-I: IG: 7 (31.8%) CG: 12 (60.0) Stage II-III: IG: 15 (68.2) CG: 8 (40.0) | Mastectomy: IG: 12 (52.2%) CG: 14 (20.0%) Breast-conserving surgery: IG: 11 (47.8%) CG: 16 (80.0%) | IG: 18 (78.3%) CG: 15 (75.0%) | IG: 14 (60.9%) CG: 16 (80.0%) | Selective estrogen receptor modulator IG: 13 (56.6%) CG: 9 (45%) Aromatase inhibitor IG: 5 (21.7%) CG: 8 (40.0%) |
| Peppone et al. Clin Breast Cancer 2010 [36] | N = 16 IG: 7 CG: 9 | N = NR IG: 53.8 CG: 52.6 | N = NR IG: 25.8 CG: 24.2 | Breast Cancer Stage 0-IIIB | Mastectomy IG: 57% CG: 33.3% Lumpectomy IG: 43% CG: 66.7% | NR | NR | IG: 42.9% CG: 66.7% |
| Peppone et al. Support. Care Cancer 2018 [35] | N = 41 IG1 (exercise + supplementation): 10 IG2 (exercise): 10 IG3 (supplementation): 10 CG: 11 | N = 53.5 ± 7.8 IG1: ≤48 = 18.2% 49–57 = 54.5% ≥58 = 27.3% IG2: ≤48 = 10.0% 49–57 = 40.0% ≥58 = 50.0% IG3: ≤48 = 50.0% 49–57 = 20.0% ≥58 = 30.0% CG: ≤48 = 60.0% 49–57 = 20.0% ≥58 = 20.0% | N = NR IG1: 31.3 IG2: 32.4 IG3: 28.1 CG: 28.4 p = 0.29 | Stage 0-I IG1: 63.6% IG2: 20% IG3: 50% CG: 30% Stage II IG1: 27.3% IG2: 60% IG3: 40% CG: 40% Stage III IG1: 9.1% IG2: 20% IG3: 10% CG: 30% | NR | IG1: 54.5% IG2: 70% IG3: 70% CG: 70% | IG1: 100% IG2: 100% IG3: 100% CG: 100% | Tamoxifen IG1: 36.4% IG2: 44.4% IG3: 60% CG: 70% Aromatase inhibitor IG1: 63.6% IG2: 55.6% IG3: 40% CG: 30% |
| Tabatabai et al. J. Clin. Endocrinol. Metab. 2016 [37] | N = 206 IG: 103 CG: 103 | N = NR IG: 46.0 ± 5.7 CG: 45.2 ± 5.9 | N = NR IG: 26.2 ± 6.0 CG: 25.7 ± 6.7 | Non-metastatic Breast Cancer | NR | 100% | NR | Tamoxifen IG: 49% CG: 72% |
| Waltman et al. Osteoporos Int. 2010 [38] | N = 223 IG: 110 CG: 113 | ≤60 years IG: 55 CG: 61 >60 years IG: 45 CG: 39 | ≤25 IG: 42 CG: 34 >25 IG: 58 CG: 66 | Stage I–II breast cancer | NR | NR | NR | NR |
| Winters-Stone et al. Breast Cancer Res Treat. 2011 [39] | N = 106 IG: 52 CG: 54 | N = NR IG: 62.3 ± 6.7 CG: 62.2 ± 6.7 | N = NR IG: 29.5 ± 5.8 CG: 29.5 ± 5.6 | Stage 0–IIIA breast cancer Stage 0 IG: 7.7% CG: 3.7% Stage I IG: 38.5% CG: 40.7% Stage II IG: 48.1% CG: 35.2% Stage IIIa IG: 1.9% CG: 9.3% | NR | IG: 61.5% CG: 59.3% | IG: 92.3% CG: 83.3% | IG: 59.6% CG: 53.7% |
| Winters-Stone et al. Osteoporos Int. 2013 [40] | N = 71 IG: 35 CG: 36 | N = NR IG: 46.5 ± 5 CG: 46.4 ± 4.9 | N = NR IG: 27 ± 5.4 CG: 25.8 ± 4.6 | Stage I IG: 22.9% CG: 33.3% Stage II IG: 65.7% CG: 50% Stage III IG: 11.4% CG: 16.7% | NR | NR | IG: 62.9% CG: 61.1% | Aromatase inhibitors IG: 40% CG: 30.6% SERM IG: 37.1% CG: 44.4% |
| Authors Journal Year | Intervention | Comparator | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Type of Activity | Exercise Modality | Protocol Duration | Frequency | Volume (Session) | Intensity | Supervision or Home-Based | Timing | ||
| Baker et al. Integr. Cancer Ther. 2018 [31] | WBV | WBV | 12 weeks | Three sessions/wk | 20 min | Low-frequency, low-magnitude vibration (30 Hz, 0.1 mm, 0.3 g) | Supervised | After chemotherapy and/or radiotherapy | Usual care |
| De Paulo et al. Exp. Gerontol. 2018 [32] | CET |
| 36 weeks | Three sessions/wk | CET: 100 min (5 min warm-up, 55 min of RET, 30 min of AET, 10 min cooldown) | NR | Supervised | After chemotherapy and/or radiotherapy | Type of activity: low intensity stretching and relaxation Exercise modality: stretching and relaxation exercises Protocol duration: 36 weeks Frequency: Two sessions/wk Volume (session): 45 min per session Intensity: low intensity Supervised After chemotherapy and/or radiotherapy |
| Dieli-Conwright et al. Breast Cancer Res. 2018 [33] | CET |
| 16 weeks | Three sessions/wk | 5 min warm-up; Days one and three: approximately 80 min of CET; Day two: AET progressively increased from 30 min up to 50 min; 5-min cool down at 40–50% estimated VO2 max | 80% of estimated 1-RM for lower body exercises and 60% estimated 1-RM for upper body, keeping target HR in AET at 60–80% of maximum, increased every 4 weeks | Supervised | After chemotherapy and/or radiotherapy | Usual care |
| Kim et al. Cancer Nurs. 2016 [34] | CET |
| 6 months | AET: three nonconsecutive days; RET: two to three sessions/wk | AET: minimum 150 min/wk; RET: Two sets of 8–10 repetition | AET: 11 to 13 perceived exertions on a six to 20 point scale; RT: low to moderate intensity | Home-based + Telephone counseling | After chemotherapy and/or radiotherapy + Supplementation with 500 mg calcium and 1000 IU vitamin D | Supplementation with 500 mg calcium and 1000 IU vitamin D |
| Peppone et al. Clin Breast Cancer 2010 [36] | thai chi chuan (TCC) exercises | Warm up, stretches, Chi Kung; TCC sessions consisting of a 15-move short form sequence of Yang-style of TCC; cool down with regulatory breathing, imagery, and meditation | 12 weeks | Three sessions/wk | NR | 10 min warm up; 40 min TCC sessions; 10 min cool down | Supervised | After chemotherapy and/or radiotherapy | Usual care + Behavioral coping strategies, cohort support and group unity. Patients instructed not to change their pattern of physical activity in any manner for the duration of the intervention |
| Peppone et al. Support. Care Cancer 2018 [35] | CET |
| 12 weeks | RET: Three times per week with at least one rest day between the sessions; AET: Seven days/wk | RET: starting with an individually determined number of sets (7–10 repetitions) up to three sets for each exercise; AET: walking with a pedometer increasing daily the step count by 5–10%, up to 12,000 steps | Moderate intensity (60–70% HR) | Home-based | After chemotherapy and/or radiotherapy + Calcitriol supplementation | Usual care |
| Tabatabai et al. J. Clin. Endocrinol. Metab. 2016 [37] | CET |
| 12 months | Three times per week | AET: 20–30 min; RET: one set of 13 exercises, completing eight repetitions, up to two sets of exercises with 8–12 repetitions | NR | Supervised | After chemotherapy and/or radiotherapy | Monthly health newsletter |
| Waltman et al. Osteoporos Int. 2010 [38] | RET |
| 24 months |
| 30–45 min. two sets x 8–12 repetitions of exercises | increased weights based on individual response, adding weights after two consecutive training sessions at the maximum set and repetition |
| After chemotherapy and/or radiotherapy + Risedronate, calcium, and Vitamin D | Risedronate, calcium, and Vitamin D |
| Winters-Stone et al. Breast Cancer Res Treat. 2011 [39] | RET + IET |
| 12 months | Two supervised session/wk + 1 home-based session/wk | 45–60 min | RET: 60–70% of 1-RM for 1–3 sets of 8–12 rep; progressive increase by increasing band thickness, squat and lunge depth, and sets and repetitions | Supervised and home-based | After chemotherapy and/or radiotherapy | Type of activity: whole body stretching and relaxation exercises Exercise modality: in a seated or lying position to minimize weight-bearing forces Protocol duration: 12 months Frequency: 2 supervised session/wk + 1 home-based session/wk Volume (session): 45–60 min Intensity: NR Supervised and home-based After chemotherapy and/or radiotherapy |
| Winters-Stone et al. Osteoporos Int. 2013 [40] | RET + IET |
| 12 months | Two supervised session/wk + 1 home-based session/wk | 45–60 min | RET: 60–70% of 1-RM for 1–3 sets of 8–12 rep; progressive increase by increasing band thickness, squat and lunge depth, and sets and repetitions | Supervised and home-based | After chemotherapy and/or radiotherapy | Type of activity: whole body stretching and relaxation exercises Exercise modality: in a seated or lying position to minimize weight-bearing forces Protocol duration: 12 months Frequency: two supervised session/wk + one home-based session/wk Volume (session): 45–60 min Intensity: NR Supervised and home-based After chemotherapy and/or radiotherapy |
| Authors Journal Year | Results | ||
|---|---|---|---|
| Intragroup Analysis—IG | Intragroup Analysis—CG | Intergroups Analysis | |
| Baker et al. Integr. Cancer Ther. 2018 [31] | Molecular biomarkers NTX/Cr (BCE/mmol Cr): 43.3 ± 20.0 vs. 45.9 ± 25.6; p = NR P1NP (μg/L): 62.3 ± 27.0 vs. 64.0 ± 25.6; p = NR | Molecular biomarkers NTX/Cr (BCE/mmol Cr): 39.0 ± 16.4 vs. 38.7 ± 12.9; p = NR P1NP (μg/L): 62.2 ± 25.3 vs. 59.5 ± 26.2; p = NR | Molecular biomarkers NTX/Cr (BCE/mmol Cr): 45.9 ± 25.6 vs. 38.7 ± 12.9; p = 0.929 P1NP (μg/L): 64.0 ± 25.6 vs. 59.5 ± 26.2; p = 0.286 |
| De Paulo et al. Exp. Gerontol. 2018 [32] | Molecular Biomarkers CTX (ng/mL): 0.46 ± 0.2 vs. 0.46 ± 0.2; p = NS Osteocalcin (ng/mL): 19 ± 8 vs. 20 ± 9; p< 0.05 Bone Mineral Density Whole body BMD (g/cm2) 1.1 ± 0.1 vs. 1.1 ± 0.1; p = NS Lumbar spine BMD (g/cm2) 1.1 ± 0.1 vs. 1.0 ± 0.1; p = NS Total Hip BMD (g/cm2) 0.9 ± 0.1 vs. 0.9 ± 0.1; p = NS Trochanter BMD (g/cm2) 0.8 ± 0.1 vs. 0.9 ± 0.08; p = NS | Molecular Biomarkers CTX (ng/mL): 0.40 ± 0.1 vs. 0.33 ± 0.2; p = NS Osteocalcin (ng/mL): 17 ± 5 vs. 17 ± 6; p = NS Bone Mineral Density Whole body BMD (g/cm2) 1.1 ± 0.08 vs. 1.1 ± 0.1; p = NS Lumbar spine BMD (g/cm2) 1.1 ± 0.1 vs. 1.0 ± 0.1; p = NS Total Hip BMD (g/cm2) 0.9 ± 0.1 vs. 0.9 ± 0.1; p = NS Trochanter BMD (g/cm2) 0.9 ± 0.1 vs. 0.9 ± 0.1; p = NS | Molecular Biomarkers CTX (ng/mL): 0.46 ± 0.2 vs. 0.33 ± 0.2; p = 0.11 Osteocalcin (ng/mL): 20 ± 9 vs. 17 ± 6; p = 0.14 Bone Mineral Density Whole body BMD (g/cm2) 1.1 ± 0.1 vs. 1.1 ± 0.1; p = 0.5 Lumbar spine BMD (g/cm2) 1.0 ± 0.1 vs. 1.0 ± 0.1; p = 0.65 Total Hip BMD (g/cm2) 0.9 ± 0.1 vs. 0.9 ± 0.1; p = 0.54 Trochanter BMD (g/cm2) 0.9 ± 0.08 vs. 0.9 ± 0.1; p = 0.51 |
| Dieli-Conwright et al. Breast Cancer Res. 2018 [33] | Molecular Biomarkers Osteocalcin (ng/mL): 12.1 ± 3.1 vs. 15.0 ± 4.1; p = 0.01 BSAP (ng/mL): 16.1 ± 4 vs. 18.0 ± 5.0; p = 0.01 CTX (ng/mL): 0.48 ± 0.1 vs. 0.44 ± 0.2; p = 0.07 NTX (nM BCE/L): 18.6 ± 3.1 vs. 17.7 ± 2.8; p = 0.10 RANK (pg/mL): 27.4 ± 6.8 vs. 26.7 ± 6.4; p = 0.14 RANKL (pmol/L): 142.5 ± 18.9 vs. 146.1 ± 16.1; p = 0.09 Bone Mineral Density Whole body BMD (g/cm2): 1.22 ± 0.1 vs. 1.27 ± 0.1; p = 0.15 Lumbar spine BMD (g/cm2): 1.16 ± 0.09 vs. 1.20 ± 0.09; p = 0.09 Total Hip BMD (g/cm2): 0.91 ± 0.09 vs. 0.94 ± 0.09; p = 0.17 Trochanter BMD (g/cm2): 0.72 ± 0.07 vs. 0.74 ± 0.07; p = 0.18 Femoral neck BMD (g/cm2): 0.88 ± 0.1 vs. 0.90 ±0.1; p = 0.21 | Molecular Biomarkers Osteocalcin (ng/mL): 12.3 ± 3.4 vs. 12.0 ± 3.0; p = 0.61 BSAP (ng/mL): 16.2 ± 4.3 vs. 15.9 ± 4.2; p = 0.55 CTX (ng/mL): 0.47 ± 0.1 vs. 0.48 ± 0.2; p = 0.74 NTX (nM BCE/L): 18.4 ± 2.7 vs. 18.3 ± 2.5; p = 0.67 RANK (pg/mL): 26.9 ± 6.6 vs. 26.4 ± 6.5; p = 0.34 RANKL (pmol/L): 139.8 ± 18.1 vs. 148.8 ± 18.9; p = 0.47 Bone Mineral Density Whole body BMD (g/cm2): 1.20 ± 0.1 vs. 1.19 ± 0.1; p = 0.29 Lumbar spine BMD (g/cm2): 1.15 ± 0.09 vs. 1.14 ± 0.09; p = 0.57 Total Hip BMD (g/cm2): 0.90 ± 0.09 vs. 0.89 ± 0.08; p = 0.23 Trochanter BMD (g/cm2): 0.71 ± 0.06 vs. 0.70 ± 0.06; p = 0.43 Femoral neck BMD (g/cm2): 0.87 ± 0.1 vs. 0.86 ± 0.1; p = 0.23 | Molecular Biomarkers Osteocalcin (ng/mL): 15.0 ± 4.1 vs. 12.0 ± 3.0; MD: 3.1 (5.6 to 1.5); p = 0.01 BSAP (ng/mL): 18.0 ± 5.0 vs. 15.9 ± 4.2; MD: 1.9 (2.4 to 0.55); p = 0.001 CTX (ng/mL): 0.44 ± 0.2 vs. 0.48 ± 0.2; MD: −0.04 (−0.10 to −0.06); p = 0.10 NTX (nM BCE/L): 17.7 ± 2.8 vs. 18.3 ± 2.5; MD: −0.90 (−1.1 to −0.6); p = 0.12 RANK (pg/mL): 26.7 ± 6.4 vs. 26.4 ± 6.5; MD: −0.70 (−0.9 to −0.4); p = 0.20 RANKL (pmol/L): 146.1 ± 16.1 vs. 148.8 ± 18.9; MD: 3.6 (5.1 to 1.2); p = 0.14 Bone Mineral Density Total BMD (g/cm2): 1.27 ± 0.1 vs. 1.19 ± 0.1; MD: 0.05 (0.04 to 0.02); p = 0.15 Lumbar spine BMD (g/cm2): 1.20 ± 0.09 vs. 1.14 ± 0.09; MD: 0.04 (0.03 to 0.01); p = 0.10 Hip BMD (g/cm2): 0.94 ± 0.09 vs. 0.89 ± 0.08; MD: 0.03 (0.03 to 0.00); p = 0.18 Trochanter BMD (g/cm2): 0.74 ± 0.07 vs. 0.70 ± 0.06; MD: 0.02 (0.03 to 0.00); p = 0.22 Femoral neck BMD (g/cm2): 0.90 ± 0.1 vs. 0.86 ± 0.1; MD: 0.02 (0.03 to 0.00); p = 0.21 |
| Kim et al. Cancer Nurs. 2016 [34] | Molecular biomarkers NTX/Cr (nmol/mmol Cr): 45.98 ± 17.58 vs. 52.26 ± 17.78; p = NR Bone mineral density Lumbar spine BMD (g/cm2): 0.958 ± 0.080 vs. 0.966 ± 0.084; p = NR Total Hip BMD (g/cm2): 0.870 ± 0.089 vs. 0.876 ± 0.083; p = NR Femoral neck BMD (g/cm2): 0.797 ± 0.076 vs. 0.795 ± 0.075; p = NR | Molecular biomarkers NTX/Cr (nmol/mmol Cr): 47.85 ± 23.92 vs. 55.73 ± 26.86; p = NR Bone mineral density Lumbar spine BMD (g/cm2): 0.987 ± 0.064 vs. 0.985 ± 0.065; p = NR Total Hip BMD (g/cm2): 0.846 ± 0.075 vs. 0.845 ± 0.066; p = NR Femoral neck BMD (g/cm2): 0.798 ± 0.073 vs. 0.805 ± 0.080; p = NR | Molecular biomarkers NTX/Cr (nmol/mmol Cr): 52.26 ± 17.78 vs. 55.73 ± 26.86; p = 0.498 Bone mineral density Lumbar spine BMD (g/cm2): 0.966 ± 0.084 vs. 0.985 ± 0.065; p = 0.246 Total Hip BMD (g/cm2): 0.876 ± 0.083 vs. 0.845 ± 0.066; p = 0.506 Femoral neck BMD (g/cm2): 0.795 ± 0.075 vs. 0.805 ± 0.080; p = 0.352 |
| Peppone et al. Clin. Breast Cancer 2010 [36] | Molecular biomarkers BSAP (μg/L): 8.34 ± 0.8 vs. 10.21 ± 1.1; p = NR NTX (nmBCE): 17.6 ± 3.7 vs. 11.1 ± 2.9; p = NR | Molecular biomarkers BSAP (μg/L): 7.64 ± 0.7 vs. 8.12 ± 1.1; p = NR NTX (nmBCE): 20.8 ± 3.3 vs. 18.8 ± 2.5; p = NR | Molecular biomarkers BSAP (μg/L): 10.21 ± 1.1 vs. 8.12 ± 1.1; p = 0.17 NTX (nmBCE): 11.1 ± 2.9 vs. 18.8 ± 2.5; p = 0.14 |
| Peppone et al. Support. Care Cancer 2018 [35] | Molecular biomarkers NTX (nmBCE): 13 vs. 13.3; p = NR BSAP (ng/mL): 12.9 vs. 14.7; p = NR | Molecular biomarkers NTX (nmBCE): 12.8 vs. 14.3; p = NR BSAP (ng/mL): 12.2 vs. 13.8; p = NR | Molecular biomarkers NTX (nmBCE): 13.3 vs. 14.3; p = 0.86 BSAP (ng/mL): 14.7 vs. 13.8; p = 0.49 |
| Tabatabai et al. J. Clin. Endocrinol. Metab. 2016 [37] | Molecular biomarkers Osteocalcin: NR (decreased); p < 0.05 P1NP: NR (decreased); p < 0.05 NTX/Cr: NR (decreased); p < 0.05 CTX: NR (decreased); p < 0.05 Bone mineral density Lumbar spine BMD (g/cm2): MD: 0.001 ± 0.005; p = NS Total Hip BMD (g/cm2): NR; NS Femoral BMD (g/cm2): NR; NS | Molecular biomarkers Osteocalcin: NR (decreased); p < 0.05 P1NP (decreased): NR; p < 0.05 NTX/Cr: NR (decreased); p < 0.05 CTX: NR (decreased); p < 0.05 Bone mineral density Lumbar spine BMD (g/cm2): MD: −0.014 ± 0.005; p = 0.03 Total Hip BMD (g/cm2): NR; NS Femoral BMD (g/cm2): NR; NS | Molecular biomarkers Osteocalcin: NR; NS P1NP: NR; NS NTX/Cr: NR; NS CTX: NR; NS Bone mineral density Femoral BMD (g/cm2): NR; NS Lumbar spine BMD (g/cm2): NR; NS Hip BMD (g/cm2): NR; NS |
| Waltman et al. Osteoporos Int. 2010 [38] | Molecular biomarkers BSAP (%MD): −11.10% ± 2.3; p < 0.001 NTX (%MD): −23.20% ± 2.8; p < 0.001 Bone mineral density Lumbar spine BMD (%MD): 3.08 ± 0.44; p < 0.0001 Total Hip BMD (%MD): 2.15 ± 0.28; p < 0.0001 Femoral neck BMD (%MD): 0.92 ± 0.50; p = 0.06 Radius (33%) BMD (%MD): −0.18 ± 0.41; p = 0.66 Total radius BMD (%MD): −0.27 ± 0.60; p = 0.66 | Molecular biomarkers BSAP (%MD): −08.70 ± 2.6; p < 0.001 NTX (%MD): −16.70 ± 3.3; p < 0.001 Bone mineral density Lumbar spine BMD (%MD): 2.85 ± 0.40; p < 0.0001 Total Hip BMD (%MD): 1.81 ± 0.36; p < 0.0001 Femoral neck BMD (%MD): 0.63 ± 0.42; p = 0.14 Radius (33%) BMD (%MD): −0.16 ± 0.56; p = 0.77 Total radius BMD (%MD): −0.57 ± 0.61; p = 0.35 | Molecular biomarkers BSAP (%MD): −11.10 ± 2.3 vs. −08.70 ± 2.6; p = NR NTX (%MD):−23.20 ± 2.8 vs. −16.70 ± 3.3; p = NR Bone mineral density Lumbar spine BMD (%MD): 3.08 ± 0.44 vs. 2.85 ± 0.40; p = NR Total Hip BMD (%MD): 2.15 ± 0.28 vs. 1.81 ± 0.36; p = NR Femoral neck BMD (%MD):0.92 ± 0.50 vs. 0.63 ± 0.42; p = NR Radius (33%) BMD (%MD): −0.18 ± 0.41 vs. −0.16 ± 0.56; p = NR Total radius BMD (%MD): −0.27 ± 0.60 vs. −0.57 ±0.61; p = NR |
| Winters-Stone et al. Breast Cancer Res Treat. 2011 [39] | Molecular biomarkers Osteocalcin (ng/mL): 12.6 ± 4.4 vs. 12.8 ± 3.8; p = NR Deoxypyridinoline (mMol/mMolCr): 21.4 ± 9.8 vs. 13.1 ± 4.2; p = NR Bone mineral density Lumbar spine BMD (g/cm2): 0.983 ± 0.146 vs. 0.987 ± 0.146; p = NR Total Hip BMD (g/cm2): 0.863 ± 0.101 vs. 0.860 ± 0.105; p = NR Trochanter BMD (g/cm2): 0.657 ± 0.088 vs. 0.654 ± 0.087; p = NR Femoral neck BMD (g/cm2): 0.731 ± 0.100 vs. 0.721 ± 0.101; p = NR | Molecular biomarkers Osteocalcin (ng/mL): 11.3 ± 4.1 vs. 14.3 ± 5.0; p = NR Deoxypyridinoline (mMol/mMolCr): 17.1 ± 5.6 vs. 12.2 ± 3.1; p = NR Bone mineral density Lumbar spine BMD (g/cm2): 0.971 ± 0.120 vs. 0.949 ± 0.108; p = NR Total Hip BMD (g/cm2): 0.848 ± 0.099 vs. 0.841 ± 0.096; p = NR Trochanter BMD (g/cm2): 0.642 ± 0.091 vs. 0.641 ± 0.089; p = NR Femoral neck BMD (g/cm2): 0.728 ± 0.091 vs. 0.713 ± 0.082; p = NR | Molecular biomarkers Osteocalcin (ng/mL): 12.8 ± 3.8 vs. 14.3 ± 5.0; p = 0.01 Deoxypyridinoline (mMol/mMolCr): 13.1 ± 4.2 vs. 12.2 ± 3.1; p = 0.22 Bone mineral density Lumbar spine BMD (g/cm2): 0.987 ± 0.146 vs. 0.949 ± 0.108; p < 0.01 Total Hip BMD (g/cm2): 0.860 ± 0.105 vs. 0.841 ± 0.096; p = 0.13 Trochanter BMD (g/cm2): 0.654 ± 0.087 vs. 0.641 ± 0.089; p = 0.15 Femoral neck BMD (g/cm2): 0.721 ± 0.101 vs. 0.713 ± 0.082; p = 0.27 |
| Winters-Stone et al. Osteoporos Int. 2013 [40] | Molecular biomarkers Osteocalcin (ng/mL): 10.6 ± 4.07 vs. 9.78 ± 4.5; p = NR Deoxypyrodinoline (nmol/mmol Cr): 13.0 ± 4.95 vs. 15.8 ± 12.1; p = NR Bone mineral density Lumbar spine BMD (g/cm2): 0.983 ± 0.113 vs. 0.972 ± 0.119; p = NR Total Hip BMD (g/cm2): 0.909 ± 0.095 vs. 0.899 ± 0.096; p = NR Trochanter BMD (g/cm2): 0.689 ± 0.065 vs. 0.683 ± 0.066; p = NR Femoral neck BMD (g/cm2): 0.809 ± 0.11 vs. 0.804 ± 0.108; p = NR | Molecular biomarkers Osteocalcin (ng/mL): 14.0 ± 3.78 vs. 11.9 ± 5.5; p = NR Deoxypyrodinoline (nmol/mmol Cr): 14.2 ± 4.57 vs. 15.0 ± 8.6; p = NR Bone mineral density Lumbar spine BMD (g/cm2): 0.988 ± 0.118 vs. 0.970 ± 0.126; p = NR Total Hip BMD (g/cm2): 0.892 ± 0.119 vs. 0.887 ± 0.119; p = NR Trochanter BMD (g/cm2): 0.666 ± 0.099 vs. 0.662 ± 0.101; p = NR Femoral neck BMD (g/cm2): 0.781 ± 0.093 vs. 0.773 ± 0.095; p = NR | Molecular biomarkers Osteocalcin (ng/mL): 9.78 ± 4.5 vs. 11.9 ± 5.5; p = 0.22 Deoxypyrodinoline (nmol/mmol Cr): 15.8 ± 12.1 vs. 15.0 ± 8.6; p = 0.39 Bone mineral density Lumbar spine BMD (g/cm2): 0.972 ± 0.119 vs. 0.970 ± 0.126 p = 0.18 Total Hip BMD (g/cm2): 0.899 ± 0.096 vs. 0.887 ± 0.887; p = 0.65 Trochanter BMD (g/cm2): 0.683 ± 0.066 vs. 0.662 ± 0.101; p = 0.90 Femoral neck BMD (g/cm2): 0.804 ± 0.108 vs. 0.773 ± 0.095; p = 0.68 |
| Articles | Domain | Score | ||||
|---|---|---|---|---|---|---|
| Random Sequence Generation | Appropriate Randomization | Blinding of Participants or Personnel | Blinding of Outcome Assessors | Withdrawals and Dropouts | ||
| Baker et al., 2018 [31] | 1 | 1 | 0 | 1 | 1 | 4 |
| De Paulo et al., 2018 [32] | 1 | 0 | 0 | 0 | 1 | 2 |
| Dieli-Conwright et al., 2018 [33] | 1 | 1 | 0 | 0 | 1 | 3 |
| Kim et al., 2016 [34] | 1 | 1 | 0 | 1 | 1 | 4 |
| Peppone et al., 2010 [35] | 1 | 1 | 0 | 0 | 1 | 3 |
| Peppone et al., 2018 [36] | 1 | 1 | 0 | 0 | 1 | 3 |
| Tabatabai et al., 2016 [37] | 1 | 1 | 0 | 1 | 1 | 4 |
| Waltman et al., 2010 [38] | 1 | 0 | 0 | 0 | 1 | 2 |
| Winters-Stone et al., 2011 [39] | 1 | 0 | 0 | 1 | 1 | 3 |
| Winters-Stone et al., 2013 [40] | 1 | 0 | 0 | 1 | 1 | 3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Sire, A.; Lippi, L.; Marotta, N.; Folli, A.; Calafiore, D.; Moalli, S.; Turco, A.; Ammendolia, A.; Fusco, N.; Invernizzi, M. Impact of Physical Rehabilitation on Bone Biomarkers in Non-Metastatic Breast Cancer Women: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2023, 24, 921. https://doi.org/10.3390/ijms24020921
de Sire A, Lippi L, Marotta N, Folli A, Calafiore D, Moalli S, Turco A, Ammendolia A, Fusco N, Invernizzi M. Impact of Physical Rehabilitation on Bone Biomarkers in Non-Metastatic Breast Cancer Women: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2023; 24(2):921. https://doi.org/10.3390/ijms24020921
Chicago/Turabian Stylede Sire, Alessandro, Lorenzo Lippi, Nicola Marotta, Arianna Folli, Dario Calafiore, Stefano Moalli, Alessio Turco, Antonio Ammendolia, Nicola Fusco, and Marco Invernizzi. 2023. "Impact of Physical Rehabilitation on Bone Biomarkers in Non-Metastatic Breast Cancer Women: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 24, no. 2: 921. https://doi.org/10.3390/ijms24020921
APA Stylede Sire, A., Lippi, L., Marotta, N., Folli, A., Calafiore, D., Moalli, S., Turco, A., Ammendolia, A., Fusco, N., & Invernizzi, M. (2023). Impact of Physical Rehabilitation on Bone Biomarkers in Non-Metastatic Breast Cancer Women: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 24(2), 921. https://doi.org/10.3390/ijms24020921












