Dimethyl Fumarate Treatment Reduces the Amount but Not the Avidity of the Epstein–Barr Virus Capsid-Antigen-Specific Antibody Response in Multiple Sclerosis: A Pilot Study
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
2.2. Serum Levels of Anti-Epstein–Barr Virus Antibodies
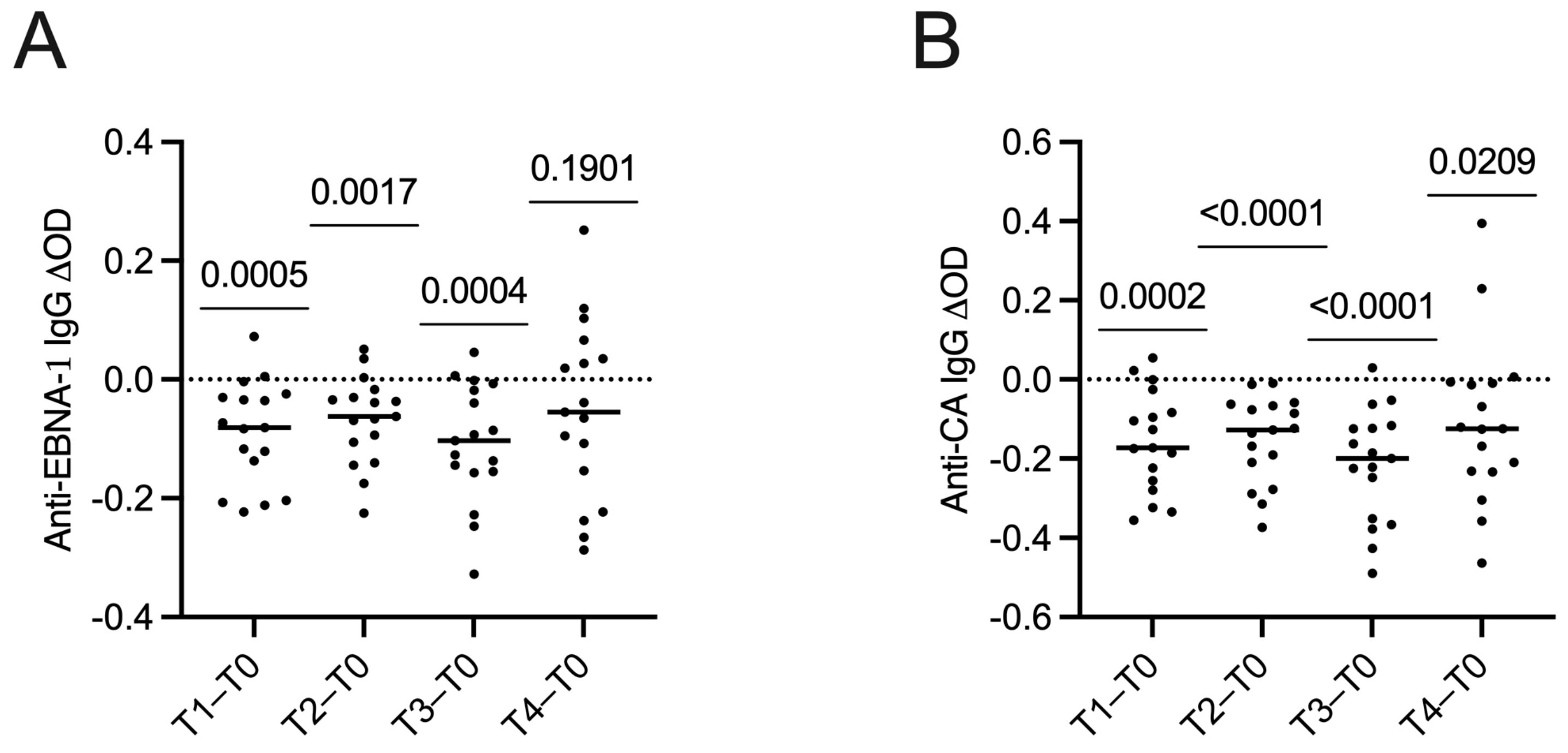
2.3. Changes in Serum Levels of Anti-Epstein–Barr Virus Antibodies
2.4. Anti-Epstein–Barr Virus Antibody Avidity
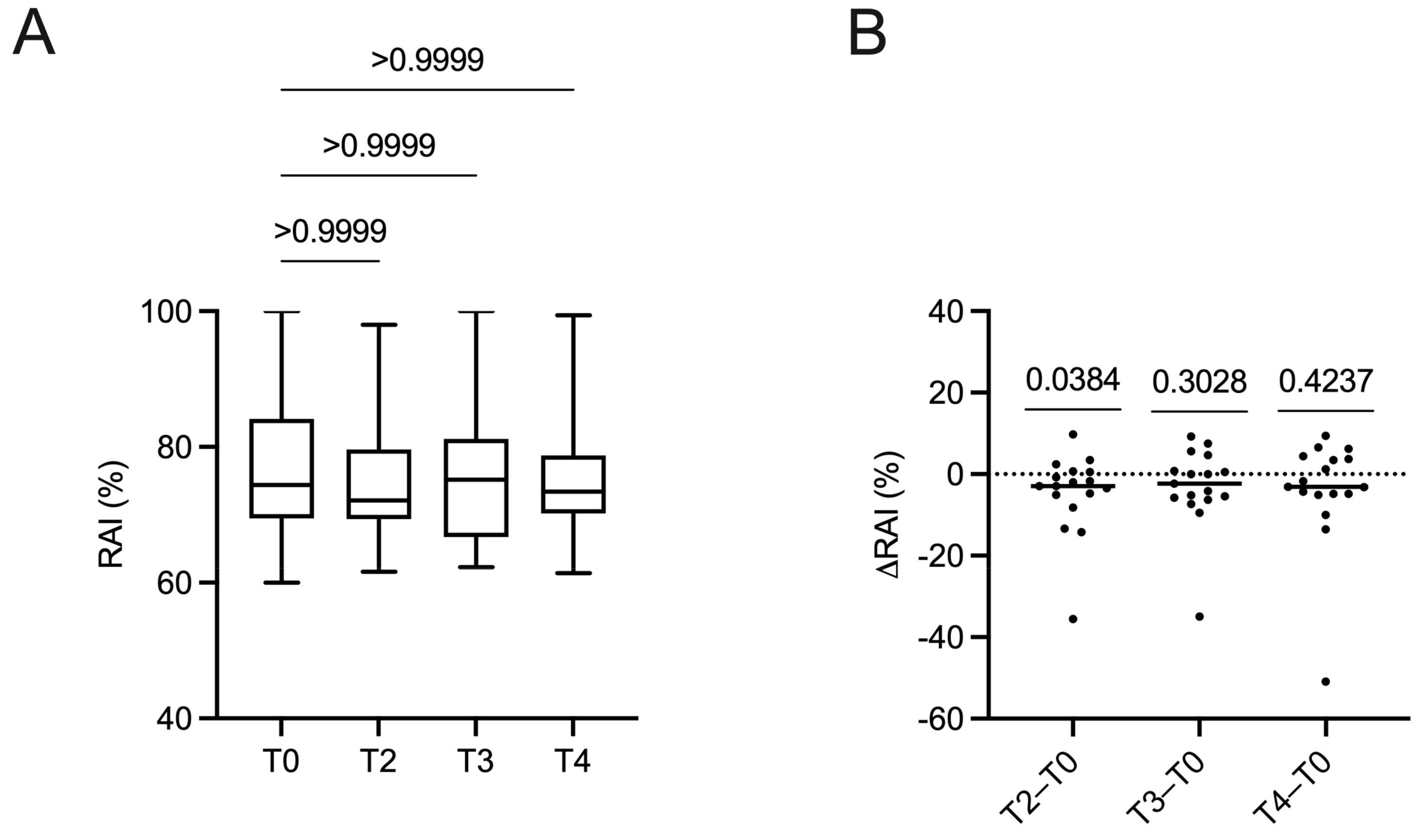
2.5. Changes in Anti-Epstein–Barr Virus Antibody Avidity
3. Discussion
4. Materials and Methods
4.1. Study Design, Population and Sampling
4.2. Serum Levels of Anti-Epstein–Barr virus Antibodies
4.3. Avidity Determination of Anti-Epstein–Barr Virus Antibodies
4.4. Delta Values
4.5. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thompson, A.J.; Baranzini, S.E.; Geurts, J.; Hemmer, B.; Ciccarelli, O. Multiple sclerosis. Lancet 2018, 391, 1622–1636. [Google Scholar] [CrossRef] [PubMed]
- McGinley, M.P.; Goldschmidt, C.H.; Rae-Grant, A.D. Diagnosis and Treatment of Multiple Sclerosis. JAMA 2021, 325, 765–779. [Google Scholar] [CrossRef] [PubMed]
- Voskuhl, R.R. The effect of sex on multiple sclerosis risk and disease progression. Mult. Scler. J. 2020, 26, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.J.; Banwell, B.L.; Barkhof, F.; Carroll, W.M.; Coetzee, T.; Comi, G.; Correale, J.; Fazekas, F.; Filippi, M.; Freedman, M.S.; et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018, 17, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Fainardi, E.; Castellazzi, M.; Seraceni, S.; Granieri, E.; Contini, C. Under the microscope: Focus on Chlamydia pneumoniae infection and multiple sclerosis. Curr. Neurovascular Res. 2008, 5, 60–70. [Google Scholar] [CrossRef]
- Lucas, R.M.; Hughes, A.M.; Lay, M.-L.J.; Ponsonby, A.-L.; Dwyer, D.E.; Taylor, B.V.; Pender, M. Epstein-Barr virus and multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2011, 82, 1142–1148. [Google Scholar] [CrossRef]
- Bar-Or, A.; Pender, M.P.; Khanna, R.; Steinman, L.; Hartung, H.-P.; Maniar, T.; Croze, E.; Aftab, B.T.; Giovannoni, G.; Joshi, M.A. Epstein–Barr Virus in Multiple Sclerosis: Theory and Emerging Immunotherapies. Trends Mol. Med. 2020, 26, 296–310. [Google Scholar] [CrossRef]
- Ascherio, A.; Munger, K.L. Environmental risk factors for multiple sclerosis. Part I: The role of infection. Ann. Neurol. 2007, 61, 288–299. [Google Scholar] [CrossRef]
- Santiago, O.; Gutiérrez-Fernández, J.; Sorlozano, A.; de Dios Luna, J.; Villegas, E.; Fernandez, O. Relation between Epstein-Barr virus and multiple sclerosis: Analytic study of scientific production. Eur. J. Clin. Microbiol. Infect. Dis. 2010, 29, 857–866. [Google Scholar] [CrossRef]
- Ascherio, A.; Munger, K.L. Epstein–Barr Virus Infection and Multiple Sclerosis: A Review. J. Neuroimmune Pharmacol. 2010, 5, 271–277. [Google Scholar] [CrossRef]
- Farrell, R.A.; Antony, D.; Wall, G.R.; Clark, D.A.; Fisniku, L.; Swanton, J.; Khaleeli, Z.; Schmierer, K.; Miller, D.H.; Giovannoni, G. Humoral immune response to EBV in multiple sclerosis is associated with disease activity on MRI. Neurology 2009, 73, 32–38. [Google Scholar] [CrossRef]
- Lunemann, J.; Tintore, M.; Messmer, B.; Strowig, T.; Rovira, A.; Perkal, H.; Caballero, E.; Münz, C.; Montalban, X.; Comabella, M. Elevated Epstein-Barr virus-encoded nuclear antigen-1 immune responses predict conversion to multiple sclerosis. Ann. Neurol. 2010, 67, 159–169. [Google Scholar] [CrossRef]
- Zivadinov, R.; Zorzon, M.; Weinstock-Guttman, B.; Serafin, M.; Bosco, A.; Bratina, A.; Maggiore, C.; Grop, A.; Tommasi, M.A.; Srinivasaraghavan, B.; et al. Epstein-Barr virus is associated with grey matter atrophy in multiple sclerosis. J. Neurol. Neurosurg. Psychiatry 2009, 80, 620–625. [Google Scholar] [CrossRef]
- Serafini, B.; Rosicarelli, B.; Franciotta, D.; Magliozzi, R.; Reynolds, R.; Cinque, P.; Andreoni, L.; Trivedi, P.; Salvetti, M.; Faggioni, A.; et al. Dysregulated Epstein-Barr virus infection in the multiple sclerosis brain. J. Exp. Med. 2007, 204, 2899–2912. [Google Scholar] [CrossRef]
- Bjornevik, K.; Cortese, M.; Healy, B.C.; Kuhle, J.; Mina, M.J.; Leng, Y.; Elledge, S.J.; Niebuhr, D.W.; Scher, A.I.; Munger, K.L.; et al. Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science 2022, 375, 296–301. [Google Scholar] [CrossRef]
- Berg, L.P.; Eriksson, M.; Longhi, S.; Kockum, I.; Warnke, C.; Thomsson, E.; Bäckström, M.; Olsson, T.; Fogdell-Hahn, A.; Bergström, T. Serum IgG levels to Epstein-Barr and measles viruses in patients with multiple sclerosis during natalizumab and interferon beta treatment. BMJ Neurol. Open 2022, 4, e000271. [Google Scholar] [CrossRef]
- Andersson, A.; Vetter, V.; Kreutzer, L.; Bauer, G. Avidities of IgG directed against viral capsid antigen or early antigen: Useful markers for significant epstein-barr virus serology. J. Med. Virol. 1994, 43, 238–244. [Google Scholar] [CrossRef]
- Vetter, V.; Kreutzer, L.; Bauer, G. Differentiation of primary from secondary anti-EBNA-1-negative cases by determination of avidity of VCA-IgG. Clin. Diagn. Virol. 1994, 2, 29–39. [Google Scholar] [CrossRef]
- De Paschale, P.C.M. Serological diagnosis of Epstein-Barr virus infection: Problems and solutions. World J. Virol. 2012, 1, 31–43. [Google Scholar] [CrossRef]
- Kurtzke, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef]
- Gold, R.; Kappos, L.; Arnold, D.L.; Bar-Or, A.; Giovannoni, G.; Selmaj, K.; Tornatore, C.; Sweetser, M.T.; Yang, M.; Sheikh, S.I.; et al. Placebo-Controlled Phase 3 Study of Oral BG-12 for Relapsing Multiple Sclerosis. New Engl. J. Med. 2012, 367, 1098–1107. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Chuikov, S.; Taitano, S.; Wu, Q.; Rastogi, A.; Tuck, S.J.; Corey, J.M.; Lundy, S.K.; Mao-Draayer, Y. Dimethyl Fumarate Protects Neural Stem/Progenitor Cells and Neurons from Oxidative Damage through Nrf2-ERK1/2 MAPK Pathway. Int. J. Mol. Sci. 2015, 16, 13885–13907. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Buttari, B.; Panieri, E.; Profumo, E.; Saso, L. An Overview of Nrf2 Signaling Pathway and Its Role in Inflammation. Molecules 2020, 25, 5474. [Google Scholar] [CrossRef] [PubMed]
- Handel, A.E.; Williamson, A.J.; Disanto, G.; Handunnetthi, L.; Giovannoni, G.; Ramagopalan, S.V. An Updated Meta-Analysis of Risk of Multiple Sclerosis following Infectious Mononucleosis. PLoS ONE 2010, 5, e12496. [Google Scholar] [CrossRef] [PubMed]
- Almohmeed, Y.H.; Avenell, A.; Aucott, L.; Vickers, M.A. Systematic Review and Meta-Analysis of the Sero-Epidemiological Association between Epstein Barr Virus and Multiple Sclerosis. PLoS ONE 2013, 8, e61110. [Google Scholar] [CrossRef]
- Bielekova, B.; Martin, R. Development of biomarkers in multiple sclerosis. Brain 2004, 127, 1463–1478. [Google Scholar] [CrossRef]
- Amanna, I.J.; Carlson, N.E.; Slifka, M.K. Duration of Humoral Immunity to Common Viral and Vaccine Antigens. New Engl. J. Med. 2007, 357, 1903–1915. [Google Scholar] [CrossRef]
- Raffel, J.; Dobson, R.; Gafson, A.; Mattoscio, M.; Muraro, P.; Giovannoni, G. Multiple sclerosis therapy and Epstein–Barr virus antibody titres. Mult. Scler. Relat. Disord. 2014, 3, 372–374. [Google Scholar] [CrossRef]
- Dominguez-Mozo, M.I.; Perez-Perez, S.; Villar, L.M.; Oliver-Martos, B.; Villarrubia, N.; Matesanz, F.; Costa-Frossard, L.; Pinto-Medel, M.J.; García-Sánchez, M.I.; Ortega-Madueño, I.; et al. Predictive factors and early biomarkers of response in multiple sclerosis patients treated with natalizumab. Sci. Rep. 2020, 10, 14244. [Google Scholar] [CrossRef]
- Castellazzi, M.; Delbue, S.; Elia, F.; Gastaldi, M.; Franciotta, D.; Rizzo, R.; Bellini, T.; Bergamaschi, R.; Granieri, E.; Fainardi, E. Epstein-Barr Virus Specific Antibody Response in Multiple Sclerosis Patients during 21 Months of Natalizumab Treatment. Dis. Markers 2015, 2015, 901312. [Google Scholar] [CrossRef]
- Hangartner, L.; Zinkernagel, R.M.; Hengartner, H. Antiviral antibody responses: The two extremes of a wide spectrum. Nat. Rev. Immunol. 2006, 6, 231–243. [Google Scholar] [CrossRef]
- Pender, M.P. The Essential Role of Epstein-Barr Virus in the Pathogenesis of Multiple Sclerosis. Neuroscientist 2011, 17, 351–367. [Google Scholar] [CrossRef]
- Horáková, D.; Zivadinov, R.; Weinstock-Guttman, B.; Havrdova, E.K.; Qu, J.; Tamaño-Blanco, M.; Badgett, D.; Tyblova, M.; Bergsland, N.; Hussein, S.; et al. Environmental Factors Associated with Disease Progression after the First Demyelinating Event: Results from the Multi-Center SET Study. PLoS ONE 2013, 8, e53996. [Google Scholar] [CrossRef]
- Gray, J. Avidity of EBV VCA-specific IgG antibodies: Distinction between recent primary infection, past infection and reactivation. J. Virol. Methods 1995, 52, 95–104. [Google Scholar] [CrossRef]
- Longbrake, E.E.; Mao-Draayer, Y.; Cascione, M.; Zielinski, T.; Bame, E.; Brassat, D.; Chen, C.; Kapadia, S.; Mendoza, J.; Miller, C.; et al. Dimethyl fumarate treatment shifts the immune environment toward an anti-inflammatory cell profile while maintaining protective humoral immunity. Mult. Scler. J. 2021, 27, 883–894. [Google Scholar] [CrossRef]
- Sabatino, J.J.; Mittl, K.; Rowles, W.M.; McPolin, K.; Rajan, J.V.; Laurie, M.T.; Zamecnik, C.R.; Dandekar, R.; Alvarenga, B.D.; Loudermilk, R.P.; et al. Multiple sclerosis therapies differentially affect SARS-CoV-2 vaccine–induced antibody and T cell immunity and function. J. Clin. Investig. 2022, 7, e156978. [Google Scholar] [CrossRef]
- von Hehn, C.; Howard, J.; Liu, S.; Meka, V.; Pultz, J.; Mehta, D.; Prada, C.; Ray, S.; Edwards, M.R.; Sheikh, S.I. Immune response to vaccines is maintained in patients treated with dimethyl fumarate. Neurol. Neuroimmunol. Neuroinflamm. 2018, 5, e409. [Google Scholar] [CrossRef]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 Revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef]
| N = 17 | |
|---|---|
| Age at entry, years: median (IQR) | 40.0 (31.0–45.5) |
| Women: n (%) | 12 (70.6) |
| Disease duration, months: median (IQR) | 29.0 (14.0–103.5) |
| EDSS score: median (IQR) | 1.5 (1.0–1.5) |
| Time Points | |||||
|---|---|---|---|---|---|
| T0 (Pre-Therapy) | T1 (1 Week) | T2 (1 Month) | T3 (3 Months) | T4 (6 Months) | |
| Anti-EBNA-1 IgG levels, OD a: median (IQR) | 1.261 (1.061–1.477) | 1.231 (0.889–1.444) | 1.252 (0.966–1.423) | 1.238 b (0.857–1.425) | 1.221 (0.938–1.520) |
| Anti-CA IgG levels, OD c: median (IQR) | 1.308 (1.077–1.666) | 1.121 d 0.932–1554) | 1.186 e (0.942–1.514) | 1.067 f (0.911–1.319) | 1.182 g (0.954–1.601) |
| Analysis Type | Data Analysed (Units) | Test |
|---|---|---|
| Comparison between groups | Anti-EBNA-1 IgG levels (OD) Anti-CA IgG levels (OD) RAI (%) | Friedman test |
| Multiple comparison (each time point vs. every other time point) | Anti-EBNA-1 IgG levels (OD) Anti-CA IgG levels (OD) RAI (%) | Dunn’s pots hoc test |
| Changes in repeated measures | ∆OD ∆RAI | Wilcoxon Signed Rank Test |
| Degree of association between variables | Anti-EBNA-1 and anti-CA IgG levels (OD) vs. EDSS score | Spearman’s rank |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castellazzi, M.; Ferri, C.; Piola, A.; Permunian, S.; Buscemi, G.; Laudisi, M.; Baldi, E.; Pugliatti, M. Dimethyl Fumarate Treatment Reduces the Amount but Not the Avidity of the Epstein–Barr Virus Capsid-Antigen-Specific Antibody Response in Multiple Sclerosis: A Pilot Study. Int. J. Mol. Sci. 2023, 24, 1500. https://doi.org/10.3390/ijms24021500
Castellazzi M, Ferri C, Piola A, Permunian S, Buscemi G, Laudisi M, Baldi E, Pugliatti M. Dimethyl Fumarate Treatment Reduces the Amount but Not the Avidity of the Epstein–Barr Virus Capsid-Antigen-Specific Antibody Response in Multiple Sclerosis: A Pilot Study. International Journal of Molecular Sciences. 2023; 24(2):1500. https://doi.org/10.3390/ijms24021500
Chicago/Turabian StyleCastellazzi, Massimiliano, Caterina Ferri, Alice Piola, Samantha Permunian, Gaia Buscemi, Michele Laudisi, Eleonora Baldi, and Maura Pugliatti. 2023. "Dimethyl Fumarate Treatment Reduces the Amount but Not the Avidity of the Epstein–Barr Virus Capsid-Antigen-Specific Antibody Response in Multiple Sclerosis: A Pilot Study" International Journal of Molecular Sciences 24, no. 2: 1500. https://doi.org/10.3390/ijms24021500
APA StyleCastellazzi, M., Ferri, C., Piola, A., Permunian, S., Buscemi, G., Laudisi, M., Baldi, E., & Pugliatti, M. (2023). Dimethyl Fumarate Treatment Reduces the Amount but Not the Avidity of the Epstein–Barr Virus Capsid-Antigen-Specific Antibody Response in Multiple Sclerosis: A Pilot Study. International Journal of Molecular Sciences, 24(2), 1500. https://doi.org/10.3390/ijms24021500







