The Silkworm Carboxypeptidase Inhibitor Prevents Gastric Cancer Cells’ Proliferation through the EGF/EGFR Signaling Pathway
Abstract
1. Introduction
2. Results
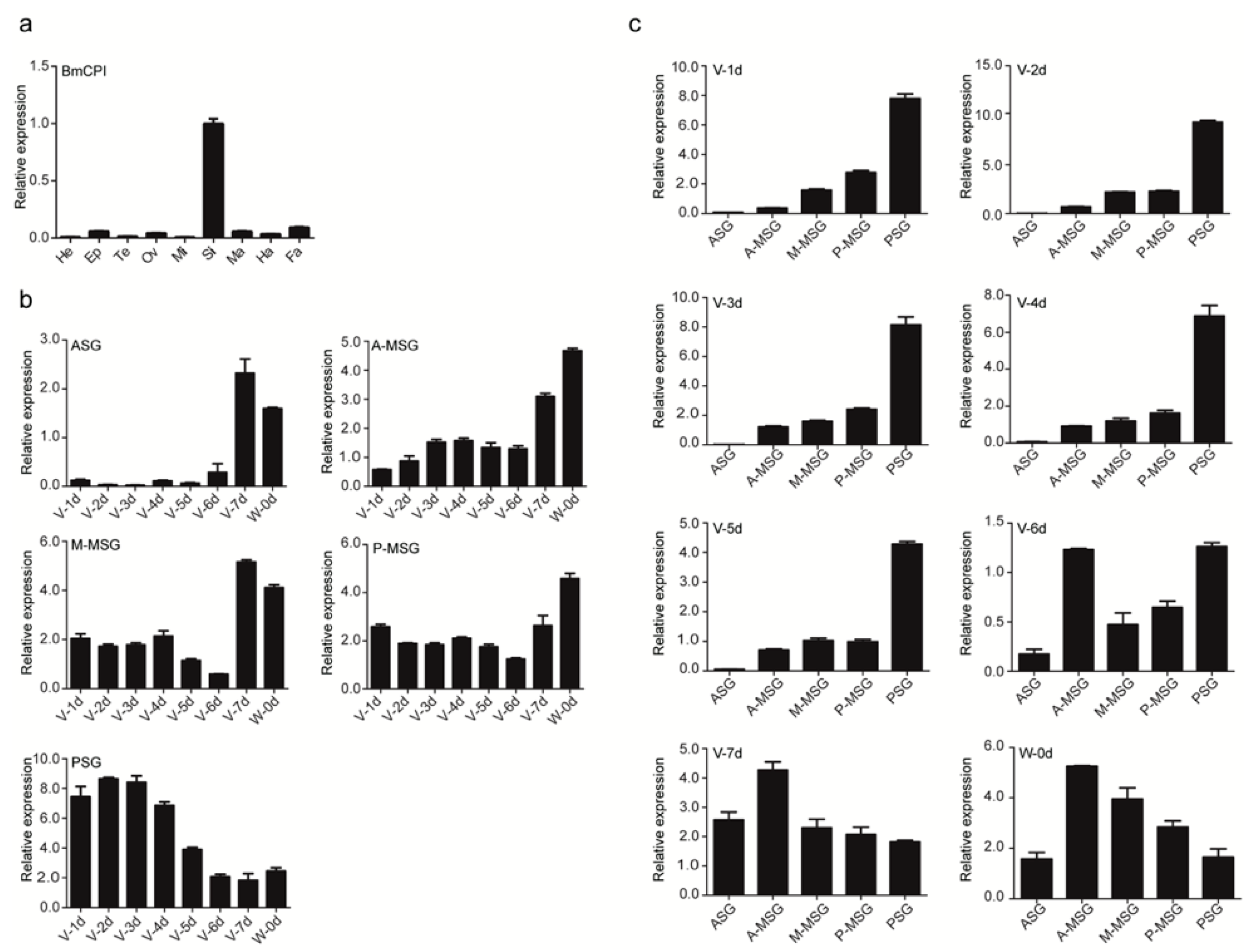
2.1. Subsection Isolation and Activity Evaluation of Bombyx mori Recombinant Carboxypeptidase Inhibitor
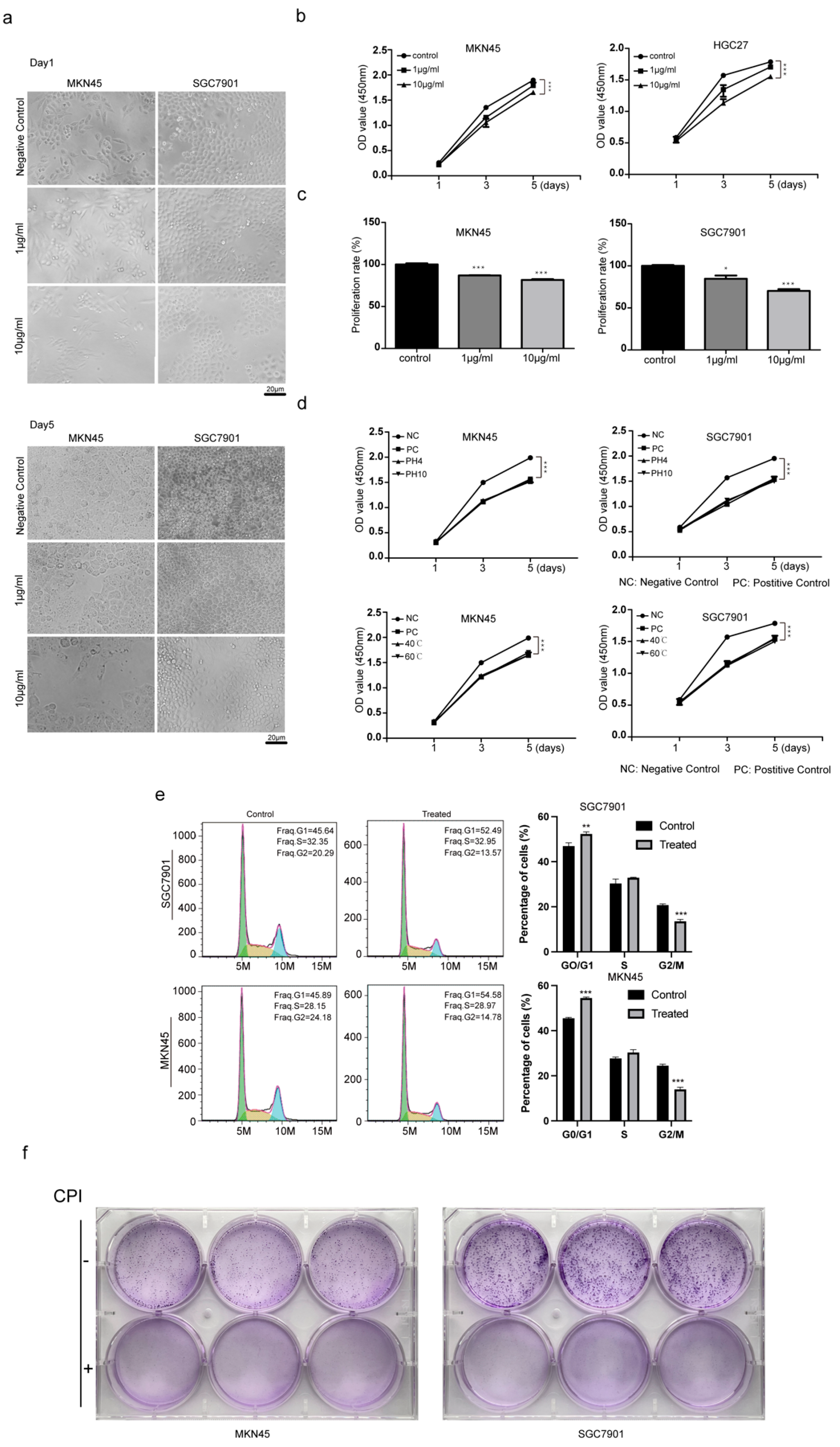
2.2. Evaluation of the Ability of Bombyx mori Carboxypeptidase Inhibitor Recombinant Protein to Inhibit the Proliferation of Gastric Cancer Tumors
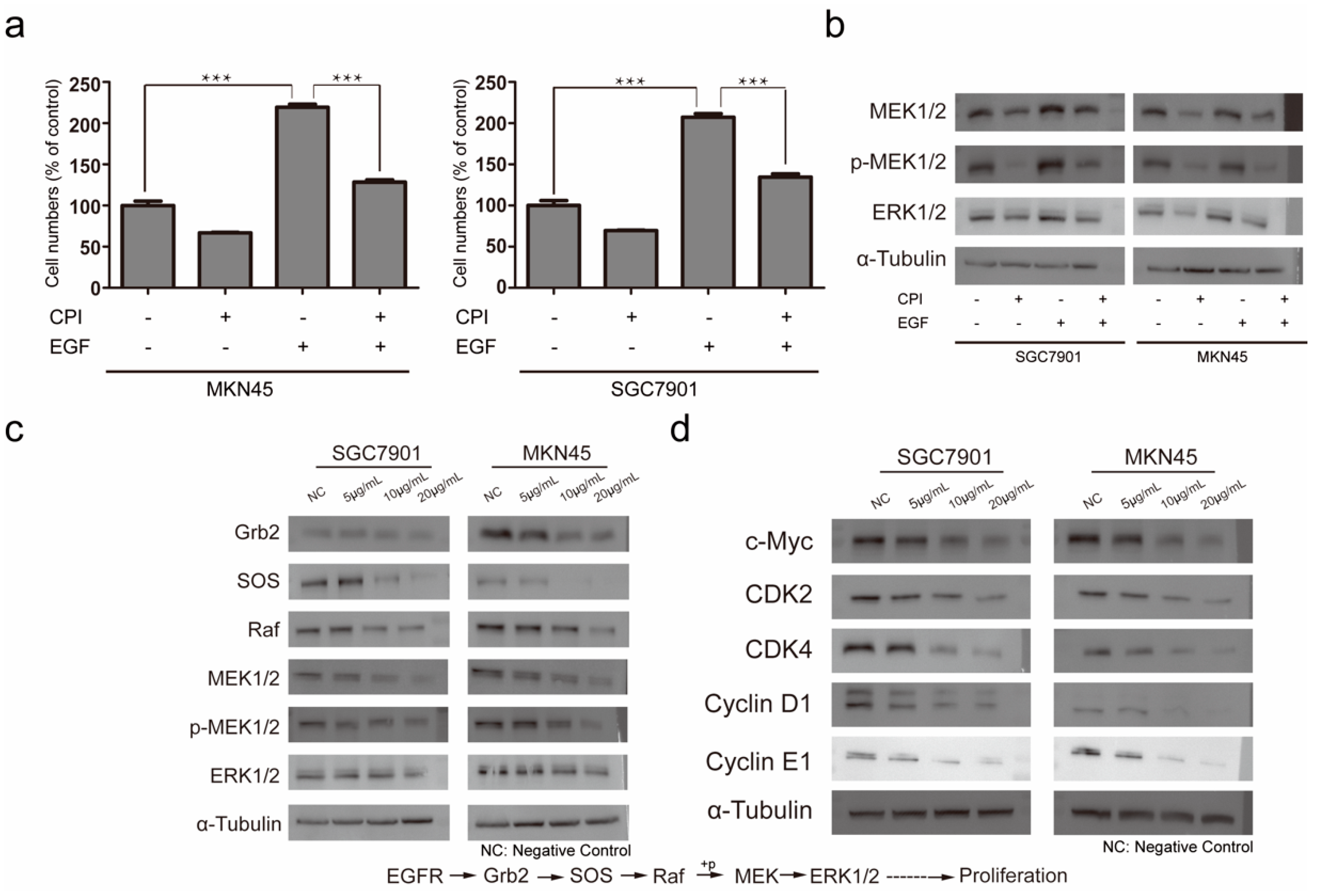
2.3. Molecular Mechanism of Silkworm Carboxypeptidase Inhibitor Recombinant Protein in Inhibiting Gastric Cancer Tumor Proliferation
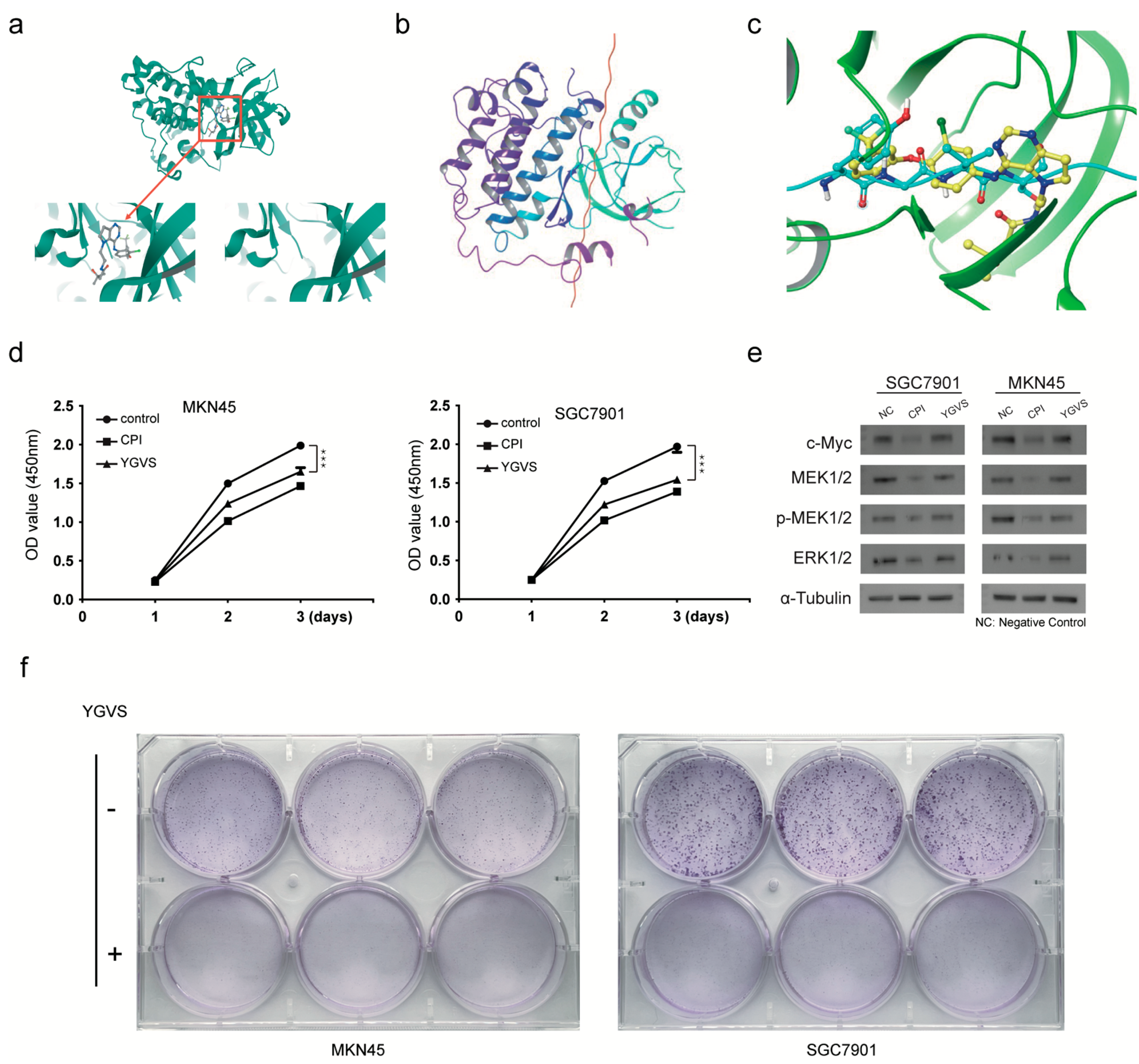
2.4. Screening of Lead Compounds for Gastric Cancer Based on Effective Peptides of Bombyx mori Carboxypeptidase Inhibitor Recombinant Protein
3. Discussion
4. Materials and Methods
4.1. Biological Materials
4.2. Western Blot
4.3. qRT-PCR
4.4. Acid–Base and Temperature Stability Analysis of Recombinant Silkworm Carboxypeptidase Inhibitor
4.5. Plate Cloning
4.6. CCK-8
4.7. Bioinformatics Analysis
4.8. Molecular Docking
- (1)
- Protein–protein docking
- (2)
- Small molecule–protein docking
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Machlowska, J.; Baj, J.; Sitarz, M.; Maciejewski, R.; Sitarz, R. Gastric Cancer: Epidemiology, Risk Factors, Classification, Genomic Characteristics and Treatment Strategies. Int. J. Mol. Sci. 2020, 21, 4012. [Google Scholar] [PubMed]
- Joshi, S.S.; Badgwell, B.D. Current treatment and recent progress in gastric cancer. CA Cancer J. Clin. 2021, 71, 264–279. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.; Torre, L.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries (vol 68, pg 394, 2018). CA-A Cancer J. Clin. 2020, 70, 313. [Google Scholar]
- Smyth, E.C.; Nilsson, M.; Grabsch, H.I.; van Grieken, N.C.; Lordick, F. Gastric cancer. Lancet 2020, 396, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Sexton, R.E.; Al Hallak, M.N.; Diab, M.; Azmi, A.S. Gastric cancer: A comprehensive review of current and future treatment strategies. Cancer Metastasis Rev. 2020, 39, 1179–1203. [Google Scholar] [CrossRef]
- Ilson, D.H. Advances in the treatment of gastric cancer. Curr. Opin. Gastroenterol. 2018, 34, 465–468. [Google Scholar] [CrossRef]
- Kim, M.S.; Lim, J.S.; Hyung, W.J.; Lee, Y.C.; Rha, S.Y.; Keum, K.C.; Koom, W.S. Neoadjuvant chemoradiotherapy followed by D2 gastrectomy in locally advanced gastric cancer. World J. Gastroenterol. 2015, 21, 2711–2718. [Google Scholar] [CrossRef]
- Rabiee, N.; Akhavan, O.; Fatahi, Y.; Ghadiri, A.M.; Kiani, M.; Makvandi, P.; Rabiee, M.; Nicknam, M.H.; Saeb, M.R.; Varma, R.S.; et al. CaZnO-based nanoghosts for the detection of ssDNA, pCRISPR and recombinant SARS-CoV-2 spike antigen and targeted delivery of doxorubicin. Chemosphere 2022, 306, 135578. [Google Scholar] [CrossRef]
- Davies, L.C.; Friedlos, F.; Hedley, D.; Martin, J.; Ogilvie, L.M.; Scanlon, I.J.; Springer, C.J. Novel fluorinated prodrugs for activation by carboxypeptidase G2 showing good in vivo antitumor activity in gene-directed enzyme prodrug therapy. J. Med. Chem. 2005, 48, 5321–5328. [Google Scholar] [CrossRef]
- Ako, H.; Hass, G.M.; Grahn, D.T.; Neurath, H. Carboxypeptidase inhibitor from potatoes. Interaction with derivatives of carboxypeptidase A. Biochemistry 1976, 15, 2573–2578. [Google Scholar] [CrossRef]
- Arolas, J.L.; Lorenzo, J.; Rovira, A.; Castella, J.; Aviles, F.X.; Sommerhoff, C.P. A carboxypeptidase inhibitor from the tick Rhipicephalus bursa: Isolation, cDNA cloning, recombinant expression, and characterization. J. Biol. Chem. 2005, 280, 3441–3448. [Google Scholar] [CrossRef] [PubMed]
- Sanglas, L.; Aviles, F.X.; Huber, R.; Gomis-Ruth, F.X.; Arolas, J.L. Mammalian metallopeptidase inhibition at the defense barrier of Ascaris parasite. Proc. Natl. Acad. Sci. USA 2009, 106, 1743–1747. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wong, K.H.; Tan, W.L.; Tay, S.V.; Wang, S.; Tam, J.P. Identification and characterization of a wolfberry carboxypeptidase inhibitor from Lycium barbarum. Food Chem. 2021, 351, 129338. [Google Scholar] [CrossRef] [PubMed]
- Quilis, J.; Meynard, D.; Vila, L.; Aviles, F.X.; Guiderdoni, E.; San Segundo, B. A potato carboxypeptidase inhibitor gene provides pathogen resistance in transgenic rice. Plant Biotechnol. J. 2007, 5, 537–553. [Google Scholar] [CrossRef]
- Muto, Y.; Suzuki, K.; Sato, E.; Ishii, H. Carboxypeptidase B inhibitors reduce tissue factor-induced renal microthrombi in rats. Eur. J. Pharmacol. 2003, 461, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Ote, M.; Mita, K.; Kawasaki, H.; Daimon, T.; Kobayashi, M.; Shimada, T. Identification of molting fluid carboxypeptidase A (MF-CPA) in Bombyx mori. Comp Biochem. Physiol. B Biochem. Mol. Biol. 2005, 141, 314–322. [Google Scholar] [CrossRef]
- Ye, J.; Li, Y.; Liu, H.W.; Li, J.; Dong, Z.; Xia, Q.; Zhao, P. Genome-Wide Identification and Characterization of Carboxypeptidase Genes in Silkworm (Bombyx mori). Int. J. Mol. Sci. 2016, 17, 1203. [Google Scholar] [CrossRef]
- Sabbah, D.A.; Hajjo, R.; Sweidan, K. Review on Epidermal Growth Factor Receptor (EGFR) Structure, Signaling Pathways, Interactions, and Recent Updates of EGFR Inhibitors. Curr. Top. Med. Chem. 2020, 20, 815–834. [Google Scholar] [CrossRef]
- Sigismund, S.; Avanzato, D.; Lanzetti, L. Emerging functions of the EGFR in cancer. Mol. Oncol. 2018, 12, 3–20. [Google Scholar] [CrossRef]
- Kothari, N.; Almhanna, K. Current status of novel agents in advanced gastroesophageal adenocarcinoma. J. Gastrointest. Oncol. 2015, 6, 60–74. [Google Scholar]
- Aydin, K.; Okutur, S.K.; Bozkurt, M.; Turkmen, I.; Namal, E.; Pilanci, K.; Ozturk, A.; Akcali, Z.; Dogusoy, G.; Demir, O.G. Effect of epidermal growth factor receptor status on the outcomes of patients with metastatic gastric cancer: A pilot study. Oncol. Lett. 2014, 7, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Ayati, A.; Moghimi, S.; Salarinejad, S.; Safavi, M.; Pouramiri, B.; Foroumadi, A. A review on progression of epidermal growth factor receptor (EGFR) inhibitors as an efficient approach in cancer targeted therapy. Bioorg. Chem. 2020, 99, 103811. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Wu, J.; Zhou, M.; Wu, J.; Wu, Z.; Lin, L.; Huang, N.; Liao, W.; Sun, L. Inhibition of Glutamine Uptake Improves the Efficacy of Cetuximab on Gastric Cancer. Integr. Cancer Ther. 2021, 20, 15347354211045349. [Google Scholar] [CrossRef] [PubMed]
- Janjigian, Y.Y. Lapatinib in Gastric Cancer: What Is the LOGiCal Next Step? J. Clin. Oncol. 2016, 34, 401–403. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Guo, D.; Jiang, Z.; Tong, R.; Jiang, P.; Bai, L.; Chen, L.; Zhu, Y.; Guo, C.; Shi, J.; et al. Novel HER2-Targeting Antibody-Drug Conjugates of Trastuzumab Beyond T-DM1 in Breast Cancer: Trastuzumab Deruxtecan(DS-8201a) and (Vic-)Trastuzumab Duocarmazine (SYD985). Eur. J. Med. Chem. 2019, 183, 111682. [Google Scholar] [CrossRef]
- Zhao, P.; Dong, Z.; Duan, J.; Wang, G.; Wang, L.; Li, Y.; Xiang, Z.; Xia, Q. Genome-wide identification and immune response analysis of serine protease inhibitor genes in the silkworm, Bombyx mori. PLoS ONE 2012, 7, e31168. [Google Scholar] [CrossRef]
- Dong, Z.; Zhao, P.; Zhang, Y.; Song, Q.; Zhang, X.; Guo, P.; Wang, D.; Xia, Q. Analysis of proteome dynamics inside the silk gland lumen of Bombyx mori. Sci. Rep. 2016, 6, 21158. [Google Scholar] [CrossRef]
- Kurioka, A.; Yamazaki, M.; Hirano, H. Primary structure and possible functions of a trypsin inhibitor of Bombyx mori. Eur. J. Biochem. 1999, 259, 120–126. [Google Scholar] [CrossRef]
- Nirmala, X.; Kodrik, D.; Zurovec, M.; Sehnal, F. Insect silk contains both a Kunitz-type and a unique Kazal-type proteinase inhibitor. Eur. J. Biochem. 2001, 268, 2064–2073. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, P.; Liu, S.; Dong, Z.; Chen, J.; Xiang, Z.; Xia, Q. A novel protease inhibitor in Bombyx mori is involved in defense against Beauveria bassiana. Insect Biochem. Mol. Biol. 2012, 42, 766–775. [Google Scholar] [CrossRef]
- Venerito, M.; Link, A.; Rokkas, T.; Malfertheiner, P. Gastric cancer—Clinical and epidemiological aspects. Helicobacter 2016, 21 (Suppl. S1), 39–44. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Martin, F.; Herrera-Leon, C.; D’Amelio, N. Molecular basis of the anticancer, apoptotic and antibacterial activities of Bombyx mori Cecropin A. Arch. Biochem. Biophys. 2022, 715, 109095. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Im, H.N.; An, D.R.; Yoon, J.Y.; Jang, J.Y.; Mobashery, S.; Hesek, D.; Lee, M.; Yoo, J.; Cui, M.; et al. The Cell Shape-determining Csd6 Protein from Helicobacter pylori Constitutes a New Family of L,D-Carboxypeptidase. J. Biol. Chem. 2015, 290, 25103–25117. [Google Scholar] [CrossRef]
- Srikumar, K.; Premalatha, R. Effect of gastrointestinal proteases on purified human intrinsic factor-vitamin B12 (IF-B12) complex. Indian J. Biochem. Biophys. 2003, 40, 139–142. [Google Scholar] [PubMed]
- Amato, A.; Porcelli, G.; Volpe, A.R.; Cotogni, P.; De Giovanni, L.; Civello, I.M.; Butti, A. Kininase I, kininase II and aminopeptidase levels in patients with gastrointestinal tumors. Ital. J. Surg. Sci. 1987, 17, 141–146. [Google Scholar] [PubMed]
- Li, Y.; Basang, Z.; Ding, H.; Lu, Z.; Ning, T.; Wei, H.; Cai, H.; Ke, Y. Latexin expression is downregulated in human gastric carcinomas and exhibits tumor suppressor potential. BMC Cancer 2011, 11, 121. [Google Scholar] [CrossRef]
- Tokunaga, A.; Onda, M.; Okuda, T.; Teramoto, T.; Fujita, I.; Mizutani, T.; Kiyama, T.; Yoshiyuki, T.; Nishi, K.; Matsukura, N. Clinical significance of epidermal growth factor (EGF), EGF receptor, and c-erbB-2 in human gastric cancer. Cancer 1995, 75 (Suppl. S6), 1418–1425. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; Peng, P.; Li, J.; Deng, H.; Zhan, N.; Zeng, Z.; Dong, W. SERPINH1 regulates EMT and gastric cancer metastasis via the Wnt/beta-catenin signaling pathway. Aging 2020, 12, 3574–3593. [Google Scholar] [CrossRef]
- Xiao, Z.; Hu, L.; Yang, L.; Wang, S.; Gao, Y.; Zhu, Q.; Yang, G.; Huang, D.; Xu, Q. TGFbeta2 is a prognostic-related biomarker and correlated with immune infiltrates in gastric cancer. J. Cell Mol. Med. 2020, 24, 7151–7162. [Google Scholar] [CrossRef]
- Blanco-Aparicio, C.; Molina, M.A.; Fernandez-Salas, E.; Frazier, M.L.; Mas, J.M.; Querol, E.; Aviles, F.X.; de Llorens, R. Potato carboxypeptidase inhibitor, a T-knot protein, is an epidermal growth factor antagonist that inhibits tumor cell growth. J. Biol. Chem. 1998, 273, 12370–12377. [Google Scholar] [CrossRef]
- da Cunha Santos, G.; Shepherd, F.A.; Tsao, M.S. EGFR mutations and lung cancer. Annu. Rev. Pathol. 2011, 6, 49–69. [Google Scholar] [CrossRef] [PubMed]
- de Castro-Carpeno, J.; Belda-Iniesta, C.; Casado Saenz, E.; Hernandez Agudo, E.; Feliu Batlle, J.; Gonzalez Baron, M. EGFR and colon cancer: A clinical view. Clin. Transl. Oncol. 2008, 10, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Harrison, P.T.; Vyse, S.; Huang, P.H. Rare epidermal growth factor receptor (EGFR) mutations in non-small cell lung cancer. Semin Cancer Biol. 2020, 61, 167–179. [Google Scholar] [CrossRef]
- Crombet Ramos, T.; Santos Morales, O.; Dy, G.K.; Leon Monzon, K.; Lage Davila, A. The Position of EGF Deprivation in the Management of Advanced Non-Small Cell Lung Cancer. Front. Oncol. 2021, 11, 639745. [Google Scholar] [CrossRef] [PubMed]
- McCubrey, J.A.; Steelman, L.S.; Chappell, W.H.; Abrams, S.L.; Wong, E.W.; Chang, F.; Lehmann, B.; Terrian, D.M.; Milella, M.; Tafuri, A.; et al. Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance. Biochim. Biophys. Acta 2007, 1773, 1263–1284. [Google Scholar] [CrossRef]
- He, J.; Huang, Z.; Han, L.; Gong, Y.; Xie, C. Mechanisms and management of 3rdgeneration EGFRTKI resistance in advanced nonsmall cell lung cancer (Review). Int. J. Oncol. 2021, 59, 90. [Google Scholar] [CrossRef]
- Guan, Y.S.; He, Q.; Li, M. Icotinib: Activity and clinical application in Chinese patients with lung cancer. Expert Opin. Pharmacother. 2014, 15, 717–728. [Google Scholar] [CrossRef]
- Dang, C.V. MYC on the path to cancer. Cell 2012, 149, 22–35. [Google Scholar] [CrossRef]
- Anauate, A.C.; Leal, M.F.; Calcagno, D.Q.; Gigek, C.O.; Karia, B.T.R.; Wisnieski, F.; Dos Santos, L.C.; Chen, E.S.; Burbano, R.R.; Smith, M.A.C. The Complex Network between MYC Oncogene and microRNAs in Gastric Cancer: An Overview. Int. J. Mol. Sci. 2020, 21, 1782. [Google Scholar] [CrossRef]
- Wang, C.; Lisanti, M.P.; Liao, D.J. Reviewing once more the c-myc and Ras collaboration: Converging at the cyclin D1-CDK4 complex and challenging basic concepts of cancer biology. Cell Cycle 2011, 10, 57–67. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, D.; Mao, J.; Ke, X.X.; Zhang, R.; Yin, C.; Gao, N.; Cui, H. Morusin inhibits cell proliferation and tumor growth by down-regulating c-Myc in human gastric cancer. Oncotarget 2017, 8, 57187–57200. [Google Scholar] [CrossRef] [PubMed]
- Liao, D.J.; Thakur, A.; Wu, J.; Biliran, H.; Sarkar, F.H. Perspectives on c-Myc, Cyclin D1, and their interaction in cancer formation, progression, and response to chemotherapy. Crit. Rev. Oncog. 2007, 13, 93–158. [Google Scholar] [CrossRef] [PubMed]
- Butt, A.J.; McNeil, C.M.; Musgrove, E.A.; Sutherland, R.L. Downstream targets of growth factor and oestrogen signalling and endocrine resistance: The potential roles of c-Myc, cyclin D1 and cyclin E. Endocr. Relat. Cancer 2005, 12 (Suppl. S1), S47–S59. [Google Scholar] [CrossRef] [PubMed]
- Chilamakuri, R.; Agarwal, S. COVID-19: Characteristics and Therapeutics. Cells 2021, 10, 206. [Google Scholar] [CrossRef] [PubMed]
- Kirtipal, N.; Bharadwaj, S.; Kang, S.G. From SARS to SARS-CoV-2, insights on structure, pathogenicity and immunity aspects of pandemic human coronaviruses. Infect Genet Evol. 2020, 85, 104502. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, S.; Lipper, R.A. Role of the development scientist in compound lead selection and optimization. J. Pharm. Sci. 2000, 89, 145–154. [Google Scholar] [CrossRef]
- Kilpatrick, G.J. Remimazolam: Non-Clinical and Clinical Profile of a New Sedative/Anesthetic Agent. Front. Pharmacol. 2021, 12, 690875. [Google Scholar] [CrossRef]
- Xie, L.; Goto, M.; Chen, X.; Morris-Natschke, S.L.; Lee, K.H. Lead Optimization: Synthesis and Biological Evaluation of PBT-1 Derivatives as Novel Antitumor Agents. ACS Med. Chem. Lett. 2021, 12, 1948–1954. [Google Scholar] [CrossRef]
- Yonchev, D.; Bajorath, J. Integrating computational lead optimization diagnostics with analog design and candidate selection. Future Sci. OA 2020, 6, FSO451. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, J.; Li, J.; Zhao, P. The Silkworm Carboxypeptidase Inhibitor Prevents Gastric Cancer Cells’ Proliferation through the EGF/EGFR Signaling Pathway. Int. J. Mol. Sci. 2023, 24, 1078. https://doi.org/10.3390/ijms24021078
Ye J, Li J, Zhao P. The Silkworm Carboxypeptidase Inhibitor Prevents Gastric Cancer Cells’ Proliferation through the EGF/EGFR Signaling Pathway. International Journal of Molecular Sciences. 2023; 24(2):1078. https://doi.org/10.3390/ijms24021078
Chicago/Turabian StyleYe, Junhong, Jifu Li, and Ping Zhao. 2023. "The Silkworm Carboxypeptidase Inhibitor Prevents Gastric Cancer Cells’ Proliferation through the EGF/EGFR Signaling Pathway" International Journal of Molecular Sciences 24, no. 2: 1078. https://doi.org/10.3390/ijms24021078
APA StyleYe, J., Li, J., & Zhao, P. (2023). The Silkworm Carboxypeptidase Inhibitor Prevents Gastric Cancer Cells’ Proliferation through the EGF/EGFR Signaling Pathway. International Journal of Molecular Sciences, 24(2), 1078. https://doi.org/10.3390/ijms24021078





