Biodegradable Water-Soluble Matrix for Immobilization of Biocidal 4-Hexylresorcinol
Abstract
:1. Introduction
2. Results and Discussion
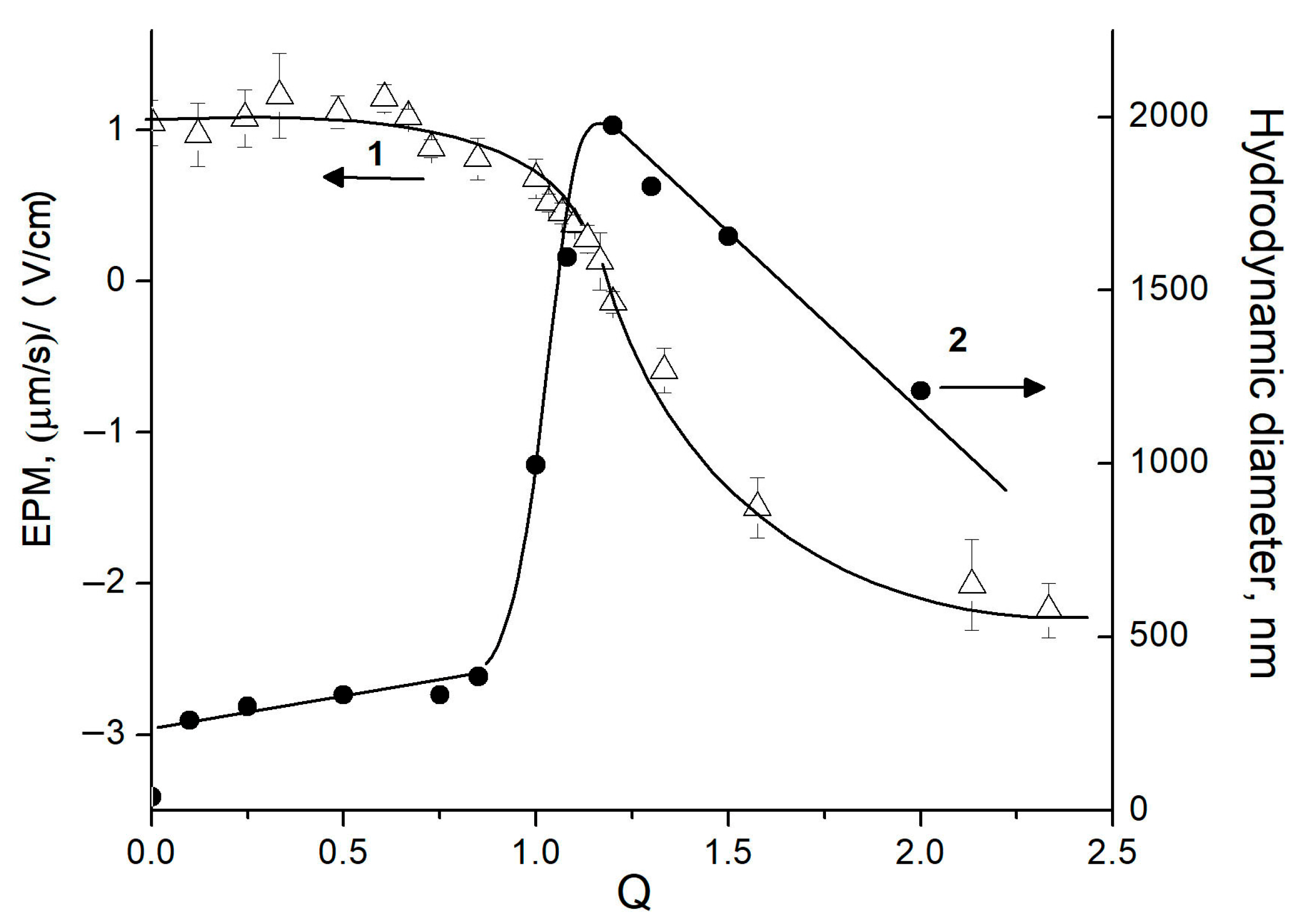
2.1. Electrostatic Interaction between Polysaccharides
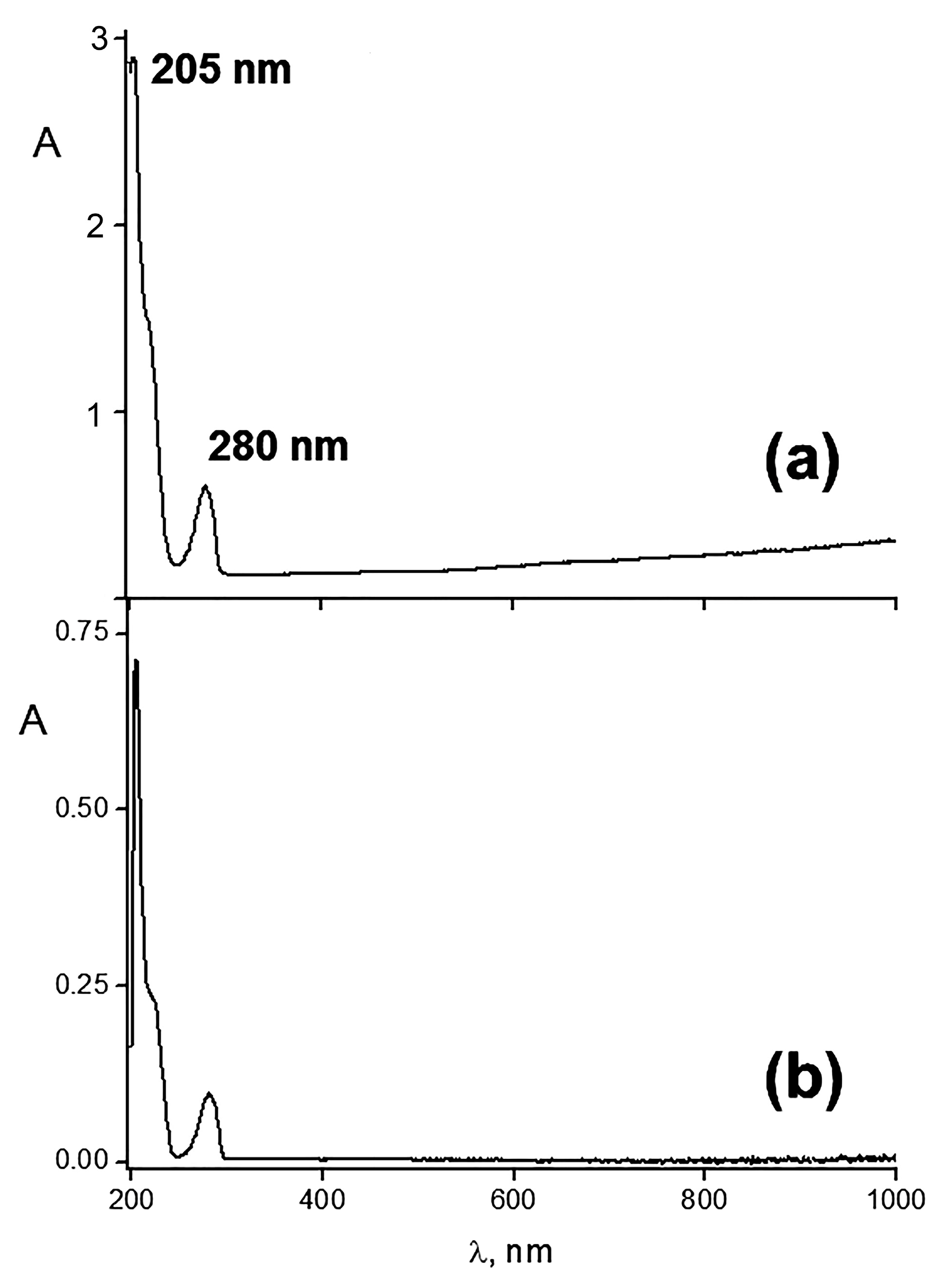
2.2. Incorporation of HR in Polycomplex Matrix
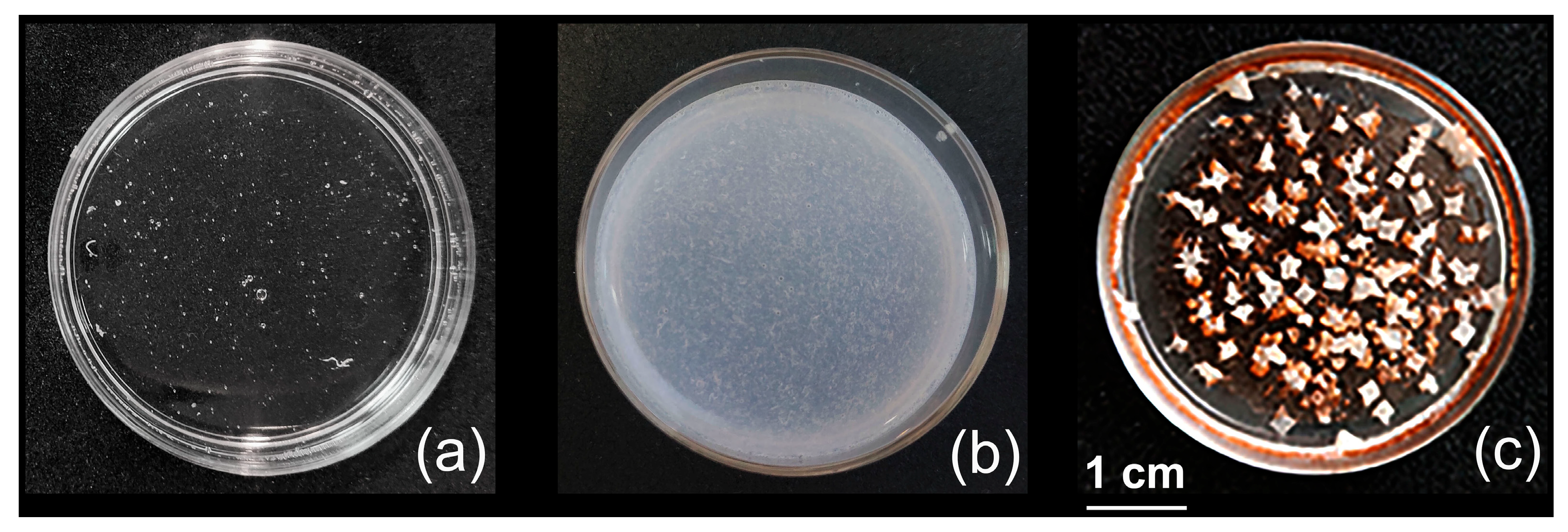
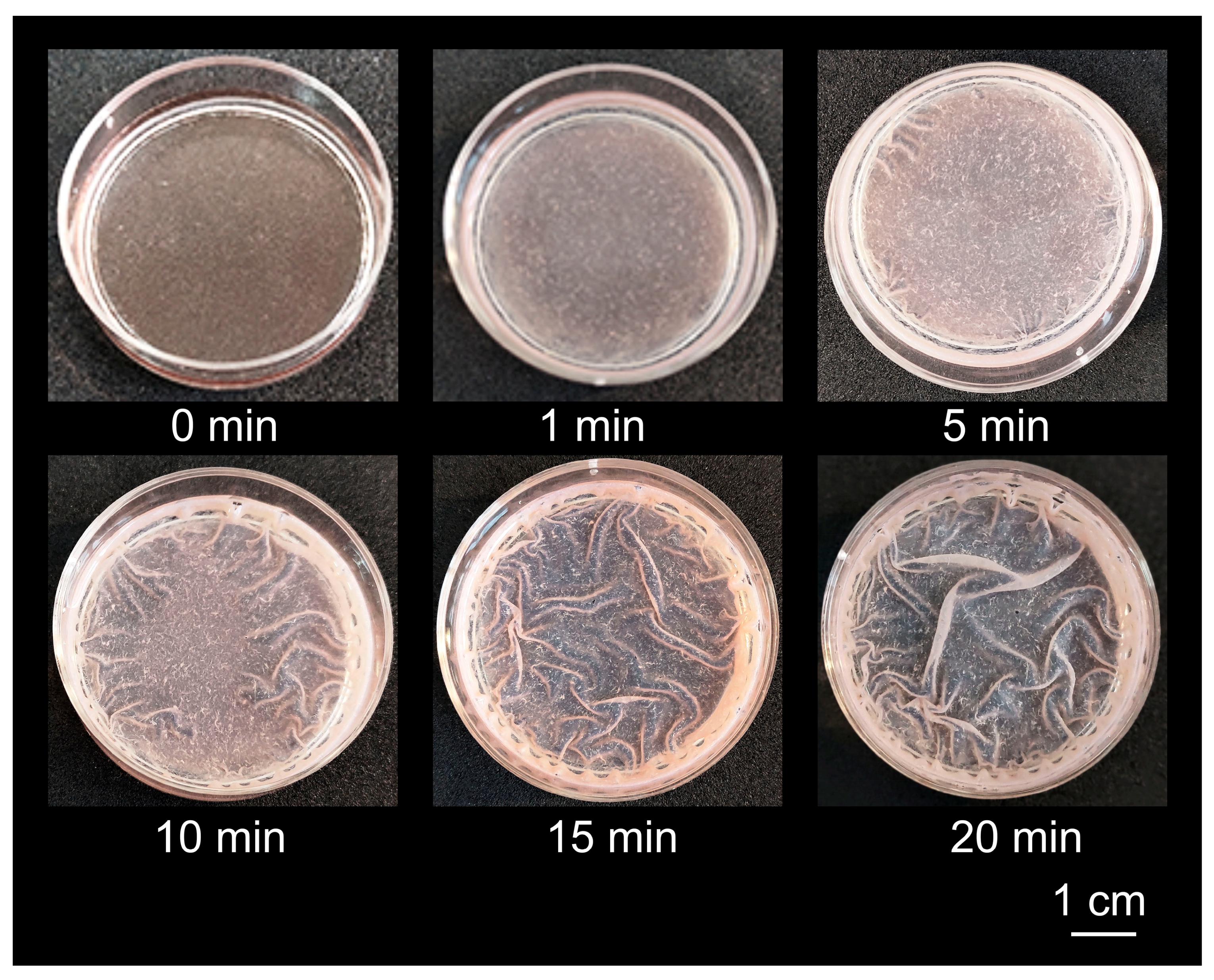
2.3. Formation of Composite Biocidal Coatings
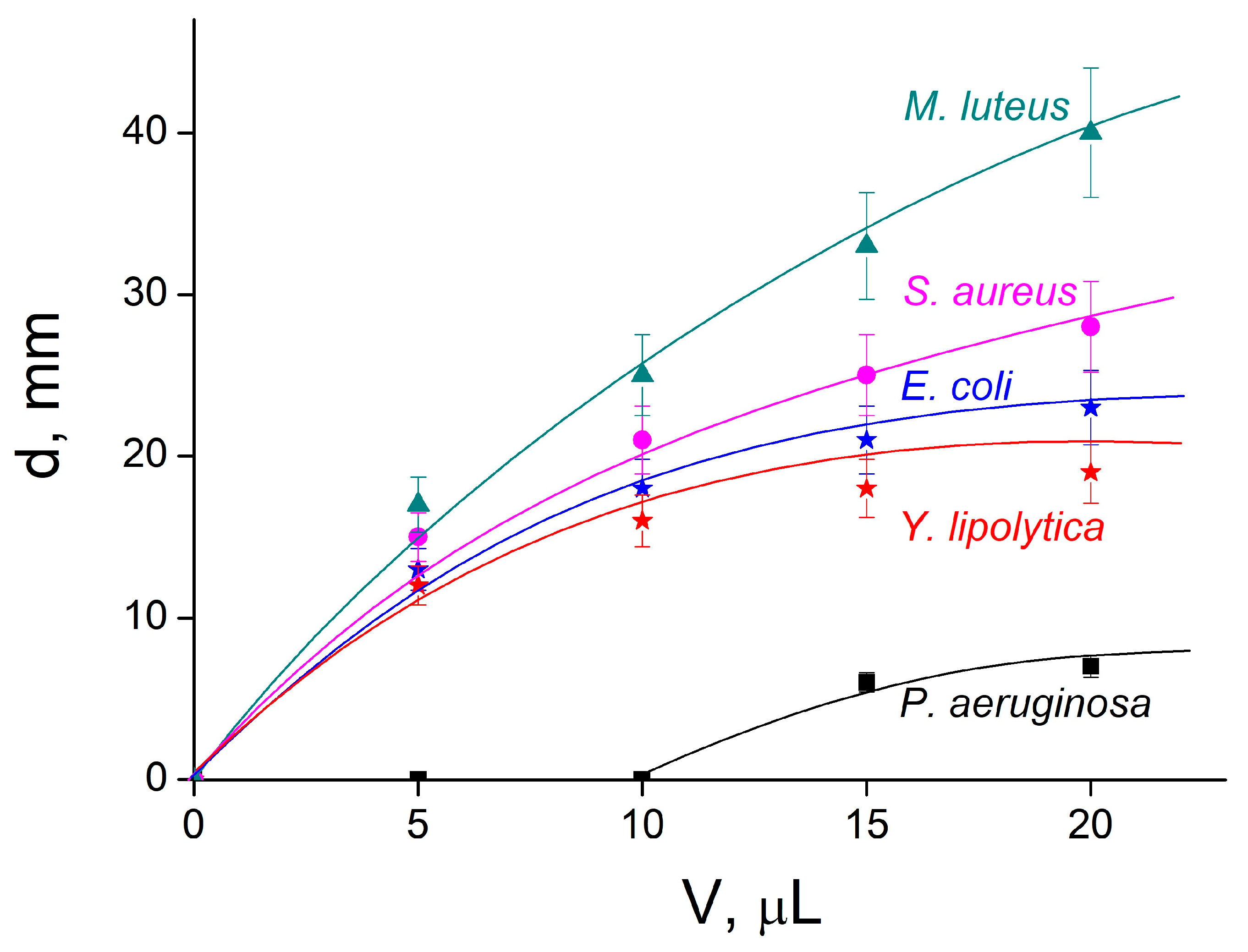
2.4. Antimicrobial Activity of IPEC–HR Composite
3. Materials and Methods
3.1. Materials
3.2. Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, L.; Xu, M.; Chen, H.; Li, Y.; Chen, S. Globalization, green economy and environmental challenges: State of the art review for practical implications. Front. Environ. Sci. 2022, 10, 870271. [Google Scholar] [CrossRef]
- Mutlu, H.; Barner, L. Getting the terms right: Green, sustainable, or circular chemistry? Macromol. Chem. Phys. 2022, 223, 2200111. [Google Scholar] [CrossRef]
- Groh, K.J.; Arp, H.P.H.; MacLeod, M.; Wang, Z. Assessing and managing environmental hazards of polymers: Historical development, science advances and policy options. Environ. Sci. Process. Impacts 2023, 25, 10–25. [Google Scholar] [CrossRef] [PubMed]
- Baranwal, J.; Barse, B.; Fais, A.; Delogu, G.L.; Kumar, A. Biopolymer: A sustainable material for food and medical applications. Polymers 2022, 14, 983. [Google Scholar] [CrossRef] [PubMed]
- Rinaudo, M. Main properties and current applications of some polysaccharides as biomaterials. Polym. Int. 2008, 57, 397–430. [Google Scholar] [CrossRef]
- Chen, T.; Sun, C.; Tian, X.; Jiang, X.; Zhang, M. Natural polysaccharide: Modification and application. Pap. Biomater. 2021, 6, 43–58. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Abasalizadeh, F.; Moghaddam, S.V.; Alizadeh, E.; Akbari, E.; Kashani, E.; Fazljou, S.M.B.; Torbati, M.; Akbarzadeh, A. Alginate-based hydrogels as drug delivery vehicles in cancer treatment and their applications in wound dressing and 3D bioprinting. J. Biol. Eng. 2020, 14, 8. [Google Scholar]
- Marković, D.; Zarubica, A.; Stojković, N.; Vasić, M.; Cakić, M.; Nikolić, G. Alginates and similar exopolysaccharides in biomedical application and pharmacy: Controlled delivery of drugs. Adv. Technol. 2016, 5, 39–52. [Google Scholar] [CrossRef]
- Azucena Castro-Yobal, M.; Contreras-Oliva, A.; Saucedo-Rivalcoba, V.; Rivera-Armenta, J.L.; Hernández-Ramírez, G.; Salinas-Ruiz, J.; Herrera-Corredor, A. Evaluation of physicochemical properties of film-based alginate for food packing applications. e-Polymers 2021, 21, 82–95. [Google Scholar] [CrossRef]
- Abdullah, N.A.; Mohamad, Z.; Khan, Z.I.; Jusoh, M.; Zakaria, Z.Y.; Ngadi, N. Alginate based sustainable films and composites for packaging: A review. Chem. Eng. 2021, 83, 271–276. [Google Scholar]
- Barbut, S.; Harper, B.A. Dried Ca-alginate films: Effects of glycerol, relative humidity, soy fibers, and carrageenan. LWT 2019, 103, 260–265. [Google Scholar] [CrossRef]
- Senturk Parreidt, T.; Müller, K.; Schmid, M. Alginate-based edible films and coatings for food packaging applications. Foods 2018, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Koushki, M.R.; Azizi, M.H.; Azizkhani, M.; Koohy-Kamaly, P. Effect of different formulations on mechanical and physical properties of calcium alginate edible films. J. Food Qual. Hazards Control 2015, 2, 45–50. [Google Scholar]
- Trevisol, T.C.; Fritz, A.R.M.; de Souza, S.M.A.G.U.; Bierhalz, A.C.K.; Valle, J.A.B. Alginate and carboxymethyl cellulose in monolayer and bilayer films as wound dressings: Effect of the polymer ratio. J. Appl. Polym. Sci. 2019, 136, 46941. [Google Scholar] [CrossRef]
- Sun, J.; Tan, H. Alginate-based biomaterials for regenerative medicine applications. Materials 2013, 6, 1285–1309. [Google Scholar] [CrossRef]
- Ashimova, A.; Yegorov, S.; Negmetzhanov, B.; Hortelano, G. Cell encapsulation within alginate microcapsules: Immunological challenges and outlook. Front. Bioeng. Biotechnol. 2019, 7, 380. [Google Scholar] [CrossRef]
- Montanucci, P.; Terenzi, S.; Santi, C.; Pennoni, I.; Bini, V.; Pescara, T.; Basta, G.; Calafiore, R. Insights in behavior of variably formulated alginate-based microcapsules for cell transplantation. BioMed. Res. Int. 2015, 2015, 965804. [Google Scholar] [CrossRef]
- Adamiak, K.; Sionkowska, A. State of innovation in alginate-based materials. Mar. Drugs 2023, 21, 353. [Google Scholar] [CrossRef]
- Łętocha, A.; Miastkowska, M.; Sikora, E. Preparation and characteristics of alginate microparticles for food, pharmaceutical and cosmetic applications. Polymers 2022, 14, 3834. [Google Scholar] [CrossRef]
- Terescenco, D.; Hadj Benali, L.; Canivet, F.; Benoit le Gelebart, M.; Hucher, N.; Gore, E.; Picard, C. Bio-sourced polymers in cosmetic emulsions: A hidden potential of the alginates as thickeners and gelling agents. Int. J. Cosmet. Sci. 2021, 43, 573–587. [Google Scholar] [CrossRef] [PubMed]
- van der Merwe, R.D.T.; Goosen, N.J.; Pott, R.W.M. Macroalgal-derived alginate soil amendments for water retention, nutrient release rate reduction, and soil pH control. Gels 2022, 8, 548. [Google Scholar] [CrossRef] [PubMed]
- Borgogna, M.; Skjåk-Bræk, G.; Paoletti, S.; Donati, I. On the initial binding of alginate by calcium ions. The tilted egg-box hypothesis. J. Phys. Chem. B 2013, 117, 7277–7282. [Google Scholar] [CrossRef] [PubMed]
- Liling, G.; Di, Z.; Jiachao, X.; Xin, G.; Xiaoting, F.; Qing, Z. Effects of ionic crosslinking on physical and mechanical properties of alginate mulching films. Carbohydr. Polym. 2016, 136, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Deepa, B.; Abraham, E.; Pothan, L.A.; Cordeiro, N.; Faria, M.; Thomas, S. Biodegradable nanocomposite films based on sodium alginate and cellulose nanofibrils. Materials 2016, 9, 50. [Google Scholar] [CrossRef] [PubMed]
- Hurtado, A.; Aljabali, A.A.; Mishra, V.; Tambuwala, M.M.; Serrano-Aroca, Á. Alginate: Enhancement strategies for advanced applications. Int. J. Mol. Sci. 2022, 23, 4486. [Google Scholar] [CrossRef]
- Ye, J.; Ma, D.; Qin, W.; Liu, Y. Physical and antibacterial properties of sodium alginate–sodium carboxymethylcellulose films containing Lactococcus lactis. Molecules 2018, 23, 2645. [Google Scholar] [CrossRef]
- Madzovska-Malagurski, I.; Vukasinovic-Sekulic, M.; Kostic, D.; Levic, S. Towards antimicrobial yet bioactive Cu-alginate hydrogels. Biomed. Mater. 2016, 11, 035015. [Google Scholar] [CrossRef]
- Chiaoprakobkij, N.; Suwanmajo, T.; Sanchavanakit, N.; Phisalaphong, M. Curcumin-loaded bacterial cellulose/alginate/gelatin as a multifunctional biopolymer composite film. Molecules 2020, 25, 3800. [Google Scholar] [CrossRef]
- Ye, Y.; Zhang, X.; Deng, X.; Hao, L.; Wang, W. Modification of alginate hydrogel films for delivering hydrophobic kaempferol. J. Nanomater. 2019, 2019, 9170732. [Google Scholar] [CrossRef]
- Mahcene, Z.; Khelil, A.; Hasni, S.; Akman, P.K.; Bozkurt, F.; Birech, K.; Goudjil, M.B.; Tornuk, F. Development and characterization of sodium alginate based active edible films incorporated with essential oils of some medicinal plants. Int. J. Biol. Macromol. 2020, 145, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.; Adegoke, O.; Shephard, A. Bactericidal activity of hexylresorcinol lozenges against oropharyngeal organisms associated with acute sore throat. BMC Res. Notes 2020, 13, 99. [Google Scholar] [CrossRef] [PubMed]
- Fidalgo, J.; Deglesne, P.A.; Arroya, R.; Ranneva, E.; Deprez, P. 4-Hexylresorcinol a new molecule for cosmetic application. J. Biomol. Res. Ther. 2018, 8, 170. [Google Scholar]
- Mosangi, D.; Pillai, S.K.; Moyo, L.; Ray, S.S. Inorganic layered double hydroxides as a 4-hexylresorcinol delivery system for topical applications. RSC Adv. 2016, 6, 77709–77716. [Google Scholar] [CrossRef]
- Shariff, R.; Du, Y.; Dutta, M.; Kumar, S.; Thimmaiah, S.; Doraiswamy, C.; Kumari, A.; Kale, V.; Nair, N.; Zhang, S.; et al. Superior even skin tone and anti-ageing benefit of a combination of 4-hexylresorcinol and niacinamide. Int. J. Cosmet. Sci. 2022, 44, 103–117. [Google Scholar] [CrossRef]
- Stasiuk, M.; Kozubek, A. Biological activity of phenolic lipids. Cell. Mol. Life Sci. 2010, 67, 841–860. [Google Scholar] [CrossRef]
- Kaprelyants, A.S.; Suleimenov, M.K.; Sorokina, A.D.; Deborin, G.A.; El-Registan, G.I.; Stoyanovich, F.M.; Lille, Y.; Ostrovsky, D.N. Structural-functional changes in bacterial and model membranes induced by phenolic lipids. Biol. Membr. 1987, 4, 254–261. [Google Scholar]
- Tereshkin, E.V.; Tereshkina, K.B.; Loiko, N.G.; Generalova, A.A.; Kovalenko, V.V.; Krupyanskii, Y.F. Mechanisms of interaction of Escherichia coli biopolymers with 4-hexylresorcinol. Russ. J. Phys. Chem. B 2023, 17, 608–619. [Google Scholar] [CrossRef]
- Kim, S.G. 4-Hexylresorcinol: Pharmacologic chaperone and its application for wound healing. Maxillofac. Plast. Reconstr. Surg. 2022, 44, 5. [Google Scholar] [CrossRef]
- Kang, Y.J.; Jo, Y.Y.; Kweon, H.; Kim, S.G. Sericin and 4-hexylresorcinol combination ointment accelerates wound healing in the diabetic burn wound model. Int. J. Indust. Entomol. 2020, 40, 1–5. [Google Scholar]
- Kim, J.Y.; Seok, H. Role of 4-hexylresorcinol in the field of tissue engineering. Appl. Sci. 2020, 10, 3385. [Google Scholar] [CrossRef]
- Kim, S.G.; Hahn, B.D.; Park, D.S.; Lee, Y.C.; Choi, E.J.; Chae, W.S.; Baek, D.H.; Choi, J.Y. Aerosol deposition of hydroxyapatite and 4-hexylresorcinol coatings on titanium alloys for dental implants. J. Oral Maxillofac. Surg. 2011, 69, e354–e363. [Google Scholar] [CrossRef] [PubMed]
- Choo, K.W.; Dhital, R.; Mao, L.; Lin, M.; Mustapha, A. Development of polyvinyl alcohol/chitosan/modified bacterial nanocellulose films incorporated with 4-hexylresorcinol for food packaging applications. Food Packag. Shelf Life 2021, 30, 100769. [Google Scholar] [CrossRef]
- Macé, S.; Hansen, L.T.; Rupasinghe, H.V. Anti-bacterial activity of phenolic compounds against Streptococcus pyogenes. Medicines 2017, 4, 25. [Google Scholar] [CrossRef]
- Wang, K.; Ye, L. Structure and property of cationic hydroxyethyl cellulose. Polym. Plast. Technol. Eng. 2010, 49, 807–811. [Google Scholar] [CrossRef]
- Gao, W.; Liu, X.M.; Gross, R.A. Determination of molar mass and solution properties of cationic hydroxyethyl cellulose derivatives by multi-angle laser light scattering with simultaneous refractive index detection. Polym. Int. 2009, 58, 1115–1119. [Google Scholar] [CrossRef]
- Kabanov, V.A. Polyelectrolyte complexes in solution and in bulk. Russ. Chem. Rev. 2005, 74, 3–20. [Google Scholar] [CrossRef]
- Van der Gucht, J.; Spruijt, E.; Lemmers, M.; Stuart, M.A.C. Polyelectrolyte complexes: Bulk phases and colloidal systems. J. Colloid. Interface Sci. 2011, 361, 407–422. [Google Scholar] [CrossRef]
- Izumrudov, V.A. Self-assembly and molecular ‘recognition’ phenomena in solutions of (bio)polyelectrolyte complexes. Russ. Chem. Rev. 2008, 77, 381–393. [Google Scholar] [CrossRef]
- Fertah, M.; Belfkira, A.; Taourirte, M.; Brouillette, F. Extraction and characterization of sodium alginate from Moroccan Laminaria digitata brown seaweed. Arab. J. Chem. 2017, 10, S3707–S3714. [Google Scholar] [CrossRef]
- Trivedi, M.; Branton, A.; Trivedi, D.; Nayak, G.; Singh, R.; Jana, S. Characterisation of physical, spectral and thermal properties of biofield treated resorcinol. Org. Chem. Curr. Res. 2015, 4, 146. [Google Scholar]
- Izumrudov, V.A.; Mussabayeva, B.K.; Kassymova, Z.S.; Klivenko, A.N.; Orazzhanova, L.K. Interpolyelectrolyte complexes: Advances and prospects of application. Russ. Chem. Rev. 2019, 88, 1046. [Google Scholar] [CrossRef]
- Zezin, A.B.; Mikheikin, S.V.; Rogacheva, V.B.; Zansokhova, M.F.; Sybachin, A.V.; Yaroslavov, A.A. Polymeric stabilizers for protection of soil and ground against wind and water erosion. Adv. Colloid Interface Sci. 2015, 226, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Novoskoltseva, O.A.; Belov, A.A.; Loiko, N.G.; Nikolaev, Y.A.; Panova, I.G.; Yaroslavov, A.A. Biodegradable interpolycomplexes for anti-erosion stabilization of soil and sand. Polymers 2022, 14, 5383. [Google Scholar] [CrossRef] [PubMed]
- Şen, F.; Kahraman, M.V. Preparation and characterization of hybrid cationic hydroxyethyl cellulose/sodium alginate polyelectrolyte antimicrobial films. Polym. Adv. Technol. 2018, 29, 1895–1901. [Google Scholar] [CrossRef]
- Kemme, M.; Heinzel-Wieland, R. Quantitative assessment of antimicrobial activity of PLGA films loaded with 4-hexylresorcinol. J. Funct. Biomater. 2018, 9, 4. [Google Scholar] [CrossRef]
- Man, A.; Gâz, A.S.; Mare, A.D.; Berţa, L. Effects of low-molecular weight alcohols on bacterial viability. Rev. Romana Med. Lab. 2017, 10, 335–343. [Google Scholar] [CrossRef]
- Mazzola, P.G.; Jozala, A.F.; Novaes, L.C.D.L.; Moriel, P.; Penna, T.C.V. Minimal inhibitory concentration (MIC) determination of disinfectant and/or sterilizing agents. Braz. J. Pharm. Sci. 2009, 45, 241–248. [Google Scholar] [CrossRef]
- Bonev, B.; Hooper, J.; Parisot, J. Principles of assessing bacterial susceptibility to antibiotics using the agar diffusion method. J. Antimicrob. Chemother. 2008, 61, 1295–1301. [Google Scholar] [CrossRef]
- Novoskoltseva, O.A.; Panova, I.G.; Loiko, N.G.; Nikolaev, Y.A.; Litmanovich, E.A.; Yaroslavov, A.A. Polyelectrolytes and polycomplexes for stabilizing sandy grounds. Polym. Sci. Ser. B 2021, 63, 488–495. [Google Scholar] [CrossRef]
- Kabanov, V.A.; Zezin, A.B.; Mustafaev, M.I.; Kasaikin, V.A. Soluble interpolymer complexes of polyamines and polyammonium salts. In Polymeric Amines and Ammonium Salts; Goethals, E.J., Ed.; Pergamon Press: Oxford, NY, USA, 1980; pp. 173–192. [Google Scholar]
- Litmanovich, E.A.; Kotova, E.V.; Efremov, V.V. Dilute-semidilute regime crossover in aqueous solutions of poly (acrylic acid)-sodium poly (styrene sulfonate) mixtures. Colloid. Polym. Sci. 2019, 297, 371–385. [Google Scholar] [CrossRef]
- Andrews, J.M. Determination of minimum inhibitory concentrations. J. Antimicrob. Chemother. 2002, 49, 1049. [Google Scholar] [CrossRef]
- Elbing, K.L.; Brent, R. Recipes and tools for culture of Escherichia coli. Curr. Protoc. Mol. Biol. 2019, 125, e83. [Google Scholar] [CrossRef] [PubMed]
- Loiko, N.; Danilova, Y.; Moiseenko, A.; Kovalenko, V.; Tereshkina, K.; Tutukina, M.; El-Registan, G.; Sokolova, O.; Krupyanskii, Y. Morphological peculiarities of the DNA-protein complexes in starved Escherichia coli cells. PLoS ONE 2020, 15, e0231562. [Google Scholar] [CrossRef]
- Panova, I.G.; Shevaleva, E.A.; Gritskova, I.A.; Loiko, N.G.; Nikolaev, Y.A.; Novoskoltseva, O.A.; Yaroslavov, A.A. Biocidal coatings from complexes of carboxylated latex particles and a linear cationic polymer. Polymers 2022, 14, 4598. [Google Scholar] [CrossRef]
- Novoskoltseva, O.A.; Loiko, N.G.; Nikolaev, Y.A.; Lisin, A.O.; Panova, I.G.; Yaroslavov, A.A. Interpolyelectrolyte complexes based on hydrolyzed polyacrylonitrile for anti-erosion stabilization of soils and ground. Polym. Int. 2022, 71, 697–705. [Google Scholar] [CrossRef]

| Tested Microorganism | MIC, wt.% | MBC, wt.% | ||
|---|---|---|---|---|
| IPEC–HR | HR | IPEC–HR | HR | |
| P. aeruginosa | 0.035 ± 0.003 | 0.035 ± 0.002 | 0.035 ± 0.002 | 0.035 ± 0.003 |
| E. coli | 0.0030 ± 0.0002 | 0.0030 ± 0.0001 | 0.0050 ± 0.0003 | 0.0030 ± 0.0002 |
| Y. lipolytica | 0.0020 ± 0.0001 | 0.0020 ± 0.0009 | ˃0.05 | ˃0.05 |
| S. aureus | 0.00100 ± 0.00006 | 0.0020 ± 0.0008 | 0.0040 ± 0.0003 | 0.0040 ± 0.0002 |
| M. luteus | 0.00070 ± 0.00004 | 0.0020 ± 0.0001 | 0.0030 ± 0.0001 | 0.0040 ± 0.0002 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Novoskoltseva, O.A.; Litmanovich, E.A.; Loiko, N.G.; Nikolaev, Y.A.; Yaroslavov, A.A. Biodegradable Water-Soluble Matrix for Immobilization of Biocidal 4-Hexylresorcinol. Int. J. Mol. Sci. 2023, 24, 14717. https://doi.org/10.3390/ijms241914717
Novoskoltseva OA, Litmanovich EA, Loiko NG, Nikolaev YA, Yaroslavov AA. Biodegradable Water-Soluble Matrix for Immobilization of Biocidal 4-Hexylresorcinol. International Journal of Molecular Sciences. 2023; 24(19):14717. https://doi.org/10.3390/ijms241914717
Chicago/Turabian StyleNovoskoltseva, Olga A., Ekaterina A. Litmanovich, Nataliya G. Loiko, Yury A. Nikolaev, and Alexander A. Yaroslavov. 2023. "Biodegradable Water-Soluble Matrix for Immobilization of Biocidal 4-Hexylresorcinol" International Journal of Molecular Sciences 24, no. 19: 14717. https://doi.org/10.3390/ijms241914717
APA StyleNovoskoltseva, O. A., Litmanovich, E. A., Loiko, N. G., Nikolaev, Y. A., & Yaroslavov, A. A. (2023). Biodegradable Water-Soluble Matrix for Immobilization of Biocidal 4-Hexylresorcinol. International Journal of Molecular Sciences, 24(19), 14717. https://doi.org/10.3390/ijms241914717







