Identification of Antibody-Mediated Hydrolysis Sites of Oligopeptides Corresponding to the SARS-CoV-2 S-Protein by MALDI-TOF Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Donor Groups and Antibody Preparations
2.2. MALDI Spectrometric Analysis of Oligopeptide Hydrolysis by Antibodies
2.3. Comparison of the Specificity of the Hydrolysis of Five Oligopeptides by Antibodies Formed upon Vaccination and COVID-19 Infection and of the One by Proteases
3. Materials and Methods
3.1. Donors and Patients
3.2. Characterization of the Oligopeptides
Visualization of the Oligopeptides
3.3. MALDI-TOF Analysis of Oligopeptide Hydrolysis Sites
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Yan, R.; Zhang, Y.; Li, Y.; Xia, L.; Guo, Y.; Zhou, Q. Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2. Science 2020, 367, 1444–1448. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Yu, X. An introduction to epitope prediction methods and software. Rev. Med. Virol. 2009, 19, 77–96. [Google Scholar] [CrossRef]
- Kozlova, E.E.G.; Viart, B.T.; de Avila, R.A.M.; Felicori, L.F.; Chavez-Olortegui, C. Classification epitopes in groups based on their protein family. BMC Bioinform. 2015, 16, S7. [Google Scholar] [CrossRef] [PubMed]
- Kringelum, J.V.; Nielsen, M.; Padkjær, S.B.; Lund, O. Structural analysis of B-cell epitopes in antibody:protein complexes. Mol. Immunol. 2013, 53, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Gershoni, J.M.; Roitburd-Berman, A.; Siman-Tov, D.D.; Tarnovitski Freund, N.; Weiss, Y. Epitope Mapping. BioDrugs 2007, 21, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Hamed, S.M.; Sakr, M.M.; El-Housseiny, G.S.; Wasfi, R.; Aboshanab, K.M. State of the art in epitope mapping and opportunities in COVID-19. Futur. Sci. OA 2023, 9, FSO832. [Google Scholar] [CrossRef]
- Irving, M.B.; Pan, O.; Scott, J.K. Random-peptide libraries and antigen-fragment libraries for epitope mapping and the development of vaccines and diagnostics. Curr. Opin. Chem. Biol. 2001, 5, 314–324. [Google Scholar] [CrossRef]
- Guo, J.-Y.; Liu, I.-J.; Lin, H.-T.; Wang, M.-J.; Chang, Y.-L.; Lin, S.-C.; Liao, M.-Y.; Hsu, W.-C.; Lin, Y.-L.; Liao, J.C.; et al. Identification of COVID-19 B-cell epitopes with phage-displayed peptide library. J. Biomed. Sci. 2021, 28, 43. [Google Scholar] [CrossRef]
- Kar, T.; Narsaria, U.; Basak, S.; Deb, D.; Castiglione, F.; Mueller, D.M.; Srivastava, A.P. A candidate multi-epitope vaccine against SARS-CoV-2. Sci. Rep. 2020, 10, 10895. [Google Scholar] [CrossRef] [PubMed]
- Camerini, D.; Randall, A.Z.; Trappl-Kimmons, K.; Oberai, A.; Hung, C.; Edgar, J.; Shandling, A.; Huynh, V.; Teng, A.A.; Hermanson, G.; et al. Mapping SARS-CoV-2 Antibody Epitopes in COVID-19 Patients with a Multi-Coronavirus Protein Microarray. Microbiol. Spectr. 2021, 9, e0141621. [Google Scholar] [CrossRef]
- Farrera-Soler, L.; Daguer, J.-P.; Barluenga, S.; Vadas, O.; Cohen, P.; Pagano, S.; Yerly, S.; Kaiser, L.; Vuilleumier, N.; Winssinger, N. Identification of immunodominant linear epitopes from SARS-CoV-2 patient plasma. PLoS ONE 2020, 15, e0238089. [Google Scholar] [CrossRef] [PubMed]
- Timofeeva, A.M.; Sedykh, S.E.; Sedykh, T.A.; Nevinsky, G.A. Natural antibodies produced in vaccinated patients and COVID-19 convalescents recognize and hydrolyze oligopeptides corresponding to the S-protein of SARS-CoV-2. Vaccines. 2023, 11, 1494. [Google Scholar] [CrossRef]
- McConnell, S.A.; Sachithanandham, J.; Mudrak, N.J.; Zhu, X.; Farhang, P.A.; Cordero, R.J.B.; Wear, M.P.; Shapiro, J.R.; Park, H.-S.; Klein, S.L.; et al. Spike-protein proteolytic antibodies in COVID-19 convalescent plasma contribute to SARS-CoV-2 neutralization. Cell Chem. Biol. 2023, 30, 726–738.e4. [Google Scholar] [CrossRef] [PubMed]
- Paul, S. Natural catalytic antibodies. Mol. Biotechnol. 1996, 5, 197–207. [Google Scholar] [CrossRef]
- Ermakov, E.A.; Nevinsky, G.A.; Buneva, V.N. Immunoglobulins with Non-Canonical Functions in Inflammatory and Autoimmune Disease States. Int. J. Mol. Sci. 2020, 21, 5392. [Google Scholar] [CrossRef]
- Bezuglova, A.M.; Dmitrenok, P.S.; Konenkova, L.P.; Buneva, V.N.; Nevinsky, G.A. Multiple sites of the cleavage of 17- and 19-mer encephalytogenic oligopeptides corresponding to human myelin basic protein (MBP) by specific anti-MBP antibodies from patients with systemic lupus erythematosus. Peptides 2012, 37, 69–78. [Google Scholar] [CrossRef]
- Legostaeva, G.A.; Polosukhina, D.I.; Bezuglova, A.M.; Doronin, B.M.; Buneva, V.N.; Nevinsky, G.A. Affinity and catalytic heterogeneity of polyclonal myelin basic protein-hydrolyzing IgGs from sera of patients with multiple sclerosis. J. Cell. Mol. Med. 2009, 14, 699–709. [Google Scholar] [CrossRef]
- Odintsova, E.S.; Dmitrenok, P.S.; Baranova, S.V.; Timofeeva, A.M.; Buneva, V.N.; Nevinsky, G.A. Features of hydrolysis of specific and nonspecific globular proteins and oligopeptides by antibodies against viral integrase from blood of HIV-infected patients. Biochem. 2015, 80, 180–201. [Google Scholar] [CrossRef]
- Odintsova, E.S.; Dmitrenok, P.S.; Buneva, V.N.; Nevinsky, G.A. Specific anti-integrase abzymes from HIV-infected patients: A comparison of the cleavage sites of intact globular HIV integrase and two 20-mer oligopeptides corresponding to its antigenic determinants. J. Mol. Recognit. 2013, 26, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Timofeeva, A.; Sedykh, S.; Nevinsky, G. Post-Immune Antibodies in HIV-1 Infection in the Context of Vaccine Development: A Variety of Biological Functions and Catalytic Activities. Vaccines 2022, 10, 384. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.-J.; Lan, Y.-H.; Wu, P.-Y.; Wu, Y.-W.; Chen, Y.-H.; Tseng, S.-C.; Kuo, T.-J.; Sun, C.-P.; Jan, J.-T.; Ma, H.-H.; et al. A receptor-binding domain-based nanoparticle vaccine elicits durable neutralizing antibody responses against SARS-CoV-2 and variants of concern. Emerg. Microbes Infect. 2023, 12, 2149353. [Google Scholar] [CrossRef] [PubMed]
- Fox, T.; Geppert, J.; Dinnes, J.; Scandrett, K.; Bigio, J.; Sulis, G.; Hettiarachchi, D.; Mathangasinghe, Y.; Weeratunga, P.; Wickramasinghe, D.; et al. Antibody tests for identification of current and past infection with SARS-CoV-2. Cochrane Database Syst. Rev. 2022, 2022. [Google Scholar] [CrossRef]
- Shafie, M.H.; Antony Dass, M.; Ahmad Shaberi, H.S.; Zafarina, Z. Screening and confirmation tests for SARS-CoV-2: Benefits and drawbacks. Beni-Suef Univ. J. Basic Appl. Sci. 2023, 12, 6. [Google Scholar] [CrossRef]
- Zheng, X.; Duan, R.H.; Gong, F.; Wei, X.; Dong, Y.; Chen, R.; Yue Liang, M.; Tang, C.; Lu, L. Accuracy of serological tests for COVID-19: A systematic review and meta-analysis. Front. Public Health 2022, 10, 923525. [Google Scholar] [CrossRef]
- Piccoli, L.; Park, Y.-J.; Tortorici, M.A.; Czudnochowski, N.; Walls, A.C.; Beltramello, M.; Silacci-Fregni, C.; Pinto, D.; Rosen, L.E.; Bowen, J.E.; et al. Mapping Neutralizing and Immunodominant Sites on the SARS-CoV-2 Spike Receptor-Binding Domain by Structure-Guided High-Resolution Serology. Cell 2020, 183, 1024–1042.e21. [Google Scholar] [CrossRef]
- Gushchin, V.A.; Dolzhikova, I.V.; Shchetinin, A.M.; Odintsova, A.S.; Siniavin, A.E.; Nikiforova, M.A.; Pochtovyi, A.A.; Shidlovskaya, E.V.; Kuznetsova, N.A.; Burgasova, O.A.; et al. Neutralizing Activity of Sera from Sputnik V-Vaccinated People against Variants of Concern (VOC: B.1.1.7, B.1.351, P.1, B.1.617.2, B.1.617.3) and Moscow Endemic SARS-CoV-2 Variants. Vaccines 2021, 9, 779. [Google Scholar] [CrossRef]
- Devi, M.J.; Gaffar, S.; Hartati, Y.W. A review post-vaccination SARS-CoV-2 serological test: Method and antibody titer response. Anal. Biochem. 2022, 658, 114902. [Google Scholar] [CrossRef]
- Mirdita, M.; Schütze, K.; Moriwaki, Y.; Heo, L.; Ovchinnikov, S.; Steinegger, M. ColabFold: Making protein folding accessible to all. Nat. Methods 2022, 19, 679–682. [Google Scholar] [CrossRef]
- Sehnal, D.; Bittrich, S.; Deshpande, M.; Svobodová, R.; Berka, K.; Bazgier, V.; Velankar, S.; Burley, S.K.; Koča, J.; Rose, A.S. Mol* Viewer: Modern web app for 3D visualization and analysis of large biomolecular structures. Nucleic Acids Res. 2021, 49, W431–W437. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, M.R.; Lindskog, I.; Gasteiger, E.; Bairoch, A.; Sanchez, J.-C.; Hochstrasser, D.F.; Appel, R.D. Detailed peptide characterization using—A World-Wide-Web-accessible tool. Electrophoresis 1997, 18, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Xiao, T.; Li, Z.; Xing, X.; He, F.; Huang, J.; Xue, D. Improving the activity and thermal stability of trypsin by the rational design. Process Biochem. 2023, 130, 227–235. [Google Scholar] [CrossRef]
- Hedstrom, L. Trypsin: A case study in the structural determinants of enzyme specificity. Biol. Chem. 1996, 377, 465–470. [Google Scholar] [PubMed]
- Vreeke, G.J.C.; Vincken, J.-P.; Wierenga, P.A. The path of proteolysis by bovine chymotrypsin. Food Res. Int. 2023, 165, 112485. [Google Scholar] [CrossRef]
- Bruice, T.C. The Mechanisms for Chymotrypsin. Proc. Natl. Acad. Sci. USA 1961, 47, 1924–1928. [Google Scholar] [CrossRef]
- Bezuglova, A.M.; Konenkova, L.P.; Doronin, B.M.; Buneva, V.N.; Nevinsky, G.A. Affinity and catalytic heterogeneity and metal-dependence of polyclonal myelin basic protein-hydrolyzing IgGs from sera of patients with systemic lupus erythematosus. J. Mol. Recognit. 2011, 24, 960–974. [Google Scholar] [CrossRef]
- Timofeeva, A.M.; Sedykh, S.E.; Ermakov, E.A.; Matveev, A.L.; Odegova, E.I.; Sedykh, T.A.; Shcherbakov, D.N.; Merkuleva, I.A.; Volosnikova, E.A.; Nesmeyanova, V.S.; et al. Natural IgG against S-Protein and RBD of SARS-CoV-2 Do Not Bind and Hydrolyze DNA and Are Not Autoimmune. Int. J. Mol. Sci. 2022, 23, 13681. [Google Scholar] [CrossRef]
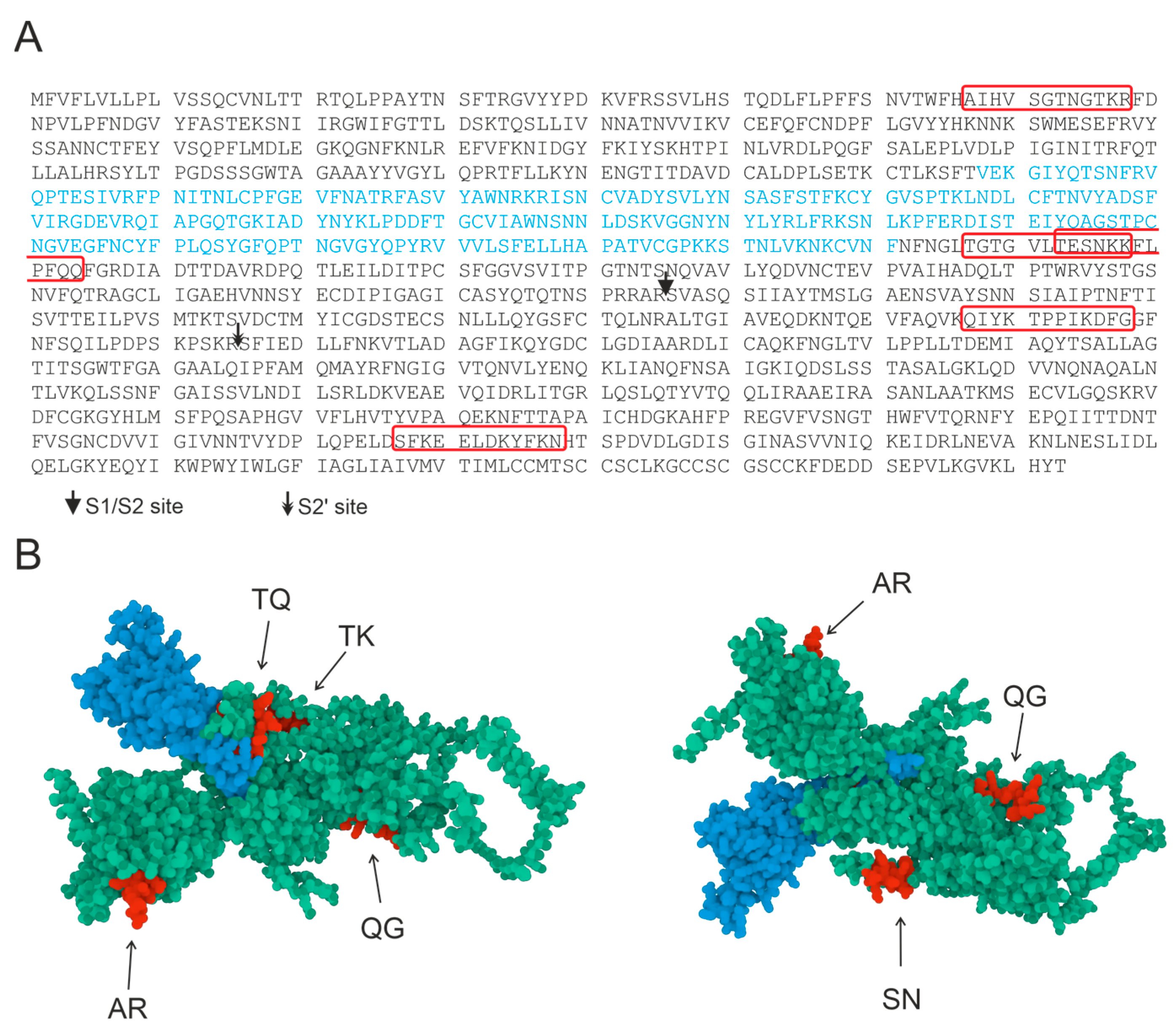

| Oligopeptide Fragment | Calculated Value kDa | Experimental Value m/z, kDa | Hydrolysis Site |
|---|---|---|---|
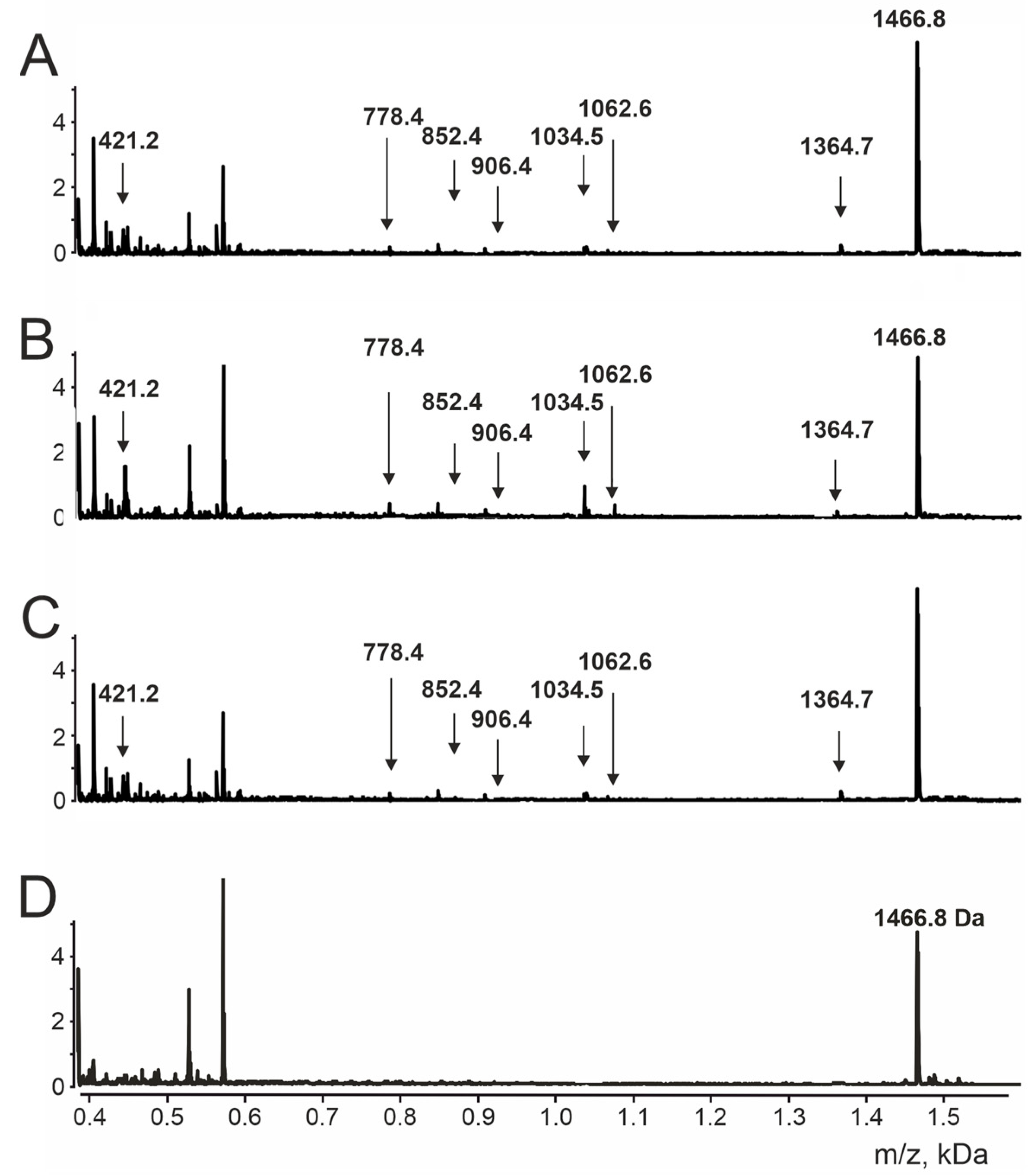
| FLPFQQ | 778.9 | 778.4 | K6↓F7 |
| TESNKKF | 852.9 | 852.4 | F7↓L8 |
| KKFLPFQ | 907.1 | 906.4 | N4↓K5, Q11↓Q12 |
| KKFLPFQQ | 1035.3 | 1034.5 | N4↓K5 |
| TESNKKFLP | 1063.2 | 1062.6 | P9↓F10 |
| ESNKKFLPFQQ | 1365.6 | 1364.7 | T1↓E2 |
| Oligopeptide | Con + Vac | Con | Vac |
|---|---|---|---|
| AR | A↓IHVS↓G↓TN↓G↓TK↓R | A↓IHVS↓G↓TN↓G↓TK↓R | A↓IHVS↓G↓TN↓G↓TK↓R |
| TK | TGTGVLT↓ES↓NKK | T↓GTGVLT↓ES↓NKK | T↓GTGVLT↓ES↓NKK |
| TQ | T↓ESN↓KK↓F↓LP↓FQ↓Q | T↓ESN↓KK↓F↓LP↓FQ↓Q | T↓ESN↓KK↓F↓LP↓FQ↓Q |
| QG | QIYKT↓P↓P↓IKDFG | QIYKT↓P↓P↓IKDFG | QIYKT↓P↓P↓IKDFG |
| SN | SFKEELDKYF↓K↓N | SFKEELDKYF↓K↓N | SFKEELDKYF↓K↓N |
| Oligopeptide | Amino Acids, after Which Hydrolysis by Antibodies Is Most Frequent (K, T, P) | Amino Acids, after Which Hydrolysis Is Rare (S, G, F) | Amino Acids, after Which Hydrolysis Is Extremely Rare (A, Q) |
|---|---|---|---|
| AR | A↓IHVS↓G↓TN↓G↓TK↓R | A↓IHVS↓G↓TN↓G↓TK↓R | A↓IHVS↓G↓TN↓G↓TK↓R |
| TK | T↓GTGVLT↓ES↓NKK | T↓GTGVLT↓ES↓NKK | T↓GTGVLT↓ES↓NKK |
| TQ | T↓ESN↓KK↓F↓LP↓FQ↓Q | T↓ESN↓KK↓F↓LP↓FQ↓Q | T↓ESN↓KK↓F↓LP↓FQ↓Q |
| QG | QIYKT↓P↓P↓IKDFG | QIYKT↓P↓P↓IKDFG | QIYKT↓P↓P↓IKDFG |
| SN | SFKEELDKYF↓K↓N | SFKEELDKYF↓K↓N | SFKEELDKYF↓K↓N |
| AR | TK | TQ | QG | SN | |
|---|---|---|---|---|---|
| S-IgG | A↓IHVS↓G↓TN↓G↓TK↓R | T↓GTGVLT↓ES↓NKK | T↓ESN↓KK↓F↓LP↓FQ↓Q | QIYKT↓P↓P↓IKDFG | SFKEELDKYF↓K↓N |
| Trypsin | AIHVSGTNGTK↓R | TGTGVLTESNK↓K | TESNK↓K↓FLPFQQ | QIYK↓TPPIK↓DFG | SFK↓EELDK↓YFK↓N |
| Chymotrypsin | AIHVSGTNGTKR | TGTGVL↓TESNKK | TESNKKF↓LPF↓QQ | QIY↓KTPPIKDF↓G | SF↓KEEL↓DKY↓F↓KN |
| 12-mer OP | Position on S-Protein | Sequence |
|---|---|---|
| AR | 67–78 | AIHVSGTNGTKR |
| TK | 547–558 | TGTGVLTESNKK |
| TQ | 553–564 | TESNKKFLPFQQ |
| QG | 787–798 | QIYKTPPIKDFG |
| SN | 1147–1158 | SFKEELDKYFKN |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Timofeeva, A.M.; Sedykh, S.E.; Dmitrenok, P.S.; Nevinsky, G.A. Identification of Antibody-Mediated Hydrolysis Sites of Oligopeptides Corresponding to the SARS-CoV-2 S-Protein by MALDI-TOF Mass Spectrometry. Int. J. Mol. Sci. 2023, 24, 14342. https://doi.org/10.3390/ijms241814342
Timofeeva AM, Sedykh SE, Dmitrenok PS, Nevinsky GA. Identification of Antibody-Mediated Hydrolysis Sites of Oligopeptides Corresponding to the SARS-CoV-2 S-Protein by MALDI-TOF Mass Spectrometry. International Journal of Molecular Sciences. 2023; 24(18):14342. https://doi.org/10.3390/ijms241814342
Chicago/Turabian StyleTimofeeva, Anna M., Sergey E. Sedykh, Pavel S. Dmitrenok, and Georgy A. Nevinsky. 2023. "Identification of Antibody-Mediated Hydrolysis Sites of Oligopeptides Corresponding to the SARS-CoV-2 S-Protein by MALDI-TOF Mass Spectrometry" International Journal of Molecular Sciences 24, no. 18: 14342. https://doi.org/10.3390/ijms241814342
APA StyleTimofeeva, A. M., Sedykh, S. E., Dmitrenok, P. S., & Nevinsky, G. A. (2023). Identification of Antibody-Mediated Hydrolysis Sites of Oligopeptides Corresponding to the SARS-CoV-2 S-Protein by MALDI-TOF Mass Spectrometry. International Journal of Molecular Sciences, 24(18), 14342. https://doi.org/10.3390/ijms241814342









