The Rationale of Complement Blockade of the MCPggaac Haplotype following Atypical Hemolytic Uremic Syndrome of Three Southeastern European Countries with a Literature Review
Abstract
1. Introduction
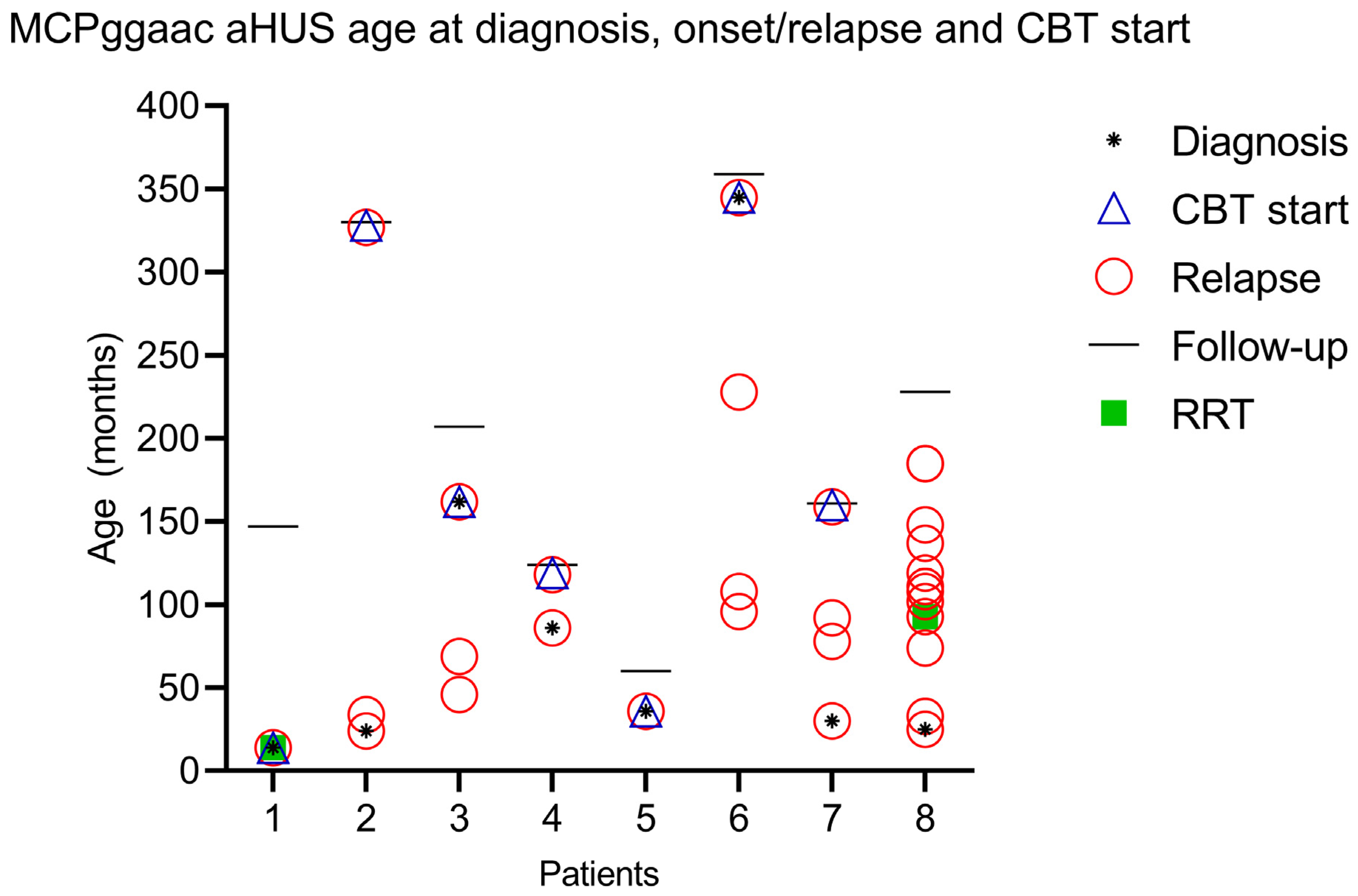
2. Case Series
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Patient | CD46, MCPggaac Risk Haplotype | Additional Variants |
|---|---|---|
| Patient 1 | homozygous MCPggaac | heterozygous C3 E1160K (LPV) heterozygous CFH H3 haplotype (risk haplotype *) |
| Patient 2 | homozygous MCPggaac | heterozygous CD46 S274I gene (VUS) heterozygous CFH H3 haplotype (risk haplotype *) |
| Patient 3 | homozygous MCPggaac | heterozygous CD46 S274I gene (VUS) |
| Patient 4 | homozygous MCPggaac | heterozygous CD46 c.286 + 2T > G (PV) heterozygous C3 gene N1229N (LBV) heterozygous CFH c.-331C > T (risk variant) |
| Patient 5 | homozygous MCPggaac | homozygous CFH H3 (risk haplotype *) |
| Patient 6 | homozygous MCPggaac | homozygous CD46 c.286 + 2T > G (PV) heterozygous CFB Y67H (VUS) |
| Patient 7 | heterozygous MCPggaac | heterozygous CFHR5 K144N (LBV) heterozygous CFH Q672Q and E936D (risk variants) heterozygous CFH V62I (protective variant) |
| Patient 8 | homozygous MCPggaac | homozygous CD46 c.286 + 2T > G (PV) heterozygous CFH c.-331C > T (risk variant) |
| PV | Pathogenic variant |
| LPV | Likely pathogenic variant |
| VUS | A variant of uncertain significance |
| LBV | Likely benign variant |
| BV | Benign variant |
References
- Esparza-Gordillo, J.; Jorge, E.; Garrido, C.A.; Carreras, L.; López-Trascasa, M.; Sánchez-Corral, P.; Decordoba, S. Insights into hemolytic uremic syndrome: Segregation of three independent predisposition factors in a large, multiple affected pedigree. Mol. Immunol. 2006, 43, 1769–1775. [Google Scholar] [CrossRef] [PubMed]
- Esparza-Gordillo, J.; Goicoechea de Jorge, E.; Buil, A.; Carreras Berges, L.; López-Trascasa, M.; Sánchez-Corral, P.; de Córdoba, S.R. Predisposition to atypical hemolytic uremic syndrome involves the concurrence of different susceptibility alleles in the regulators of complement activation gene cluster in 1q32. Hum. Mol. Genet. 2005, 14, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Le Clech, A.; Simon-Tillaux, N.; Provôt, F.; Delmas, Y.; Vieira-Martins, P.; Limou, S.; Halimi, J.-M.; Le Quintrec, M.; Lebourg, L.; Grangé, S.; et al. Atypical and secondary hemolytic uremic syndromes have a distinct presentation and no common genetic risk factors. Kidney Int. 2019, 95, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Fidalgo, T.; Martinho, P.; Pinto, C.S.; Oliveira, A.C.; Salvado, R.; Borràs, N.; Coucelo, M.; Manco, L.; Maia, T.; Mendes, M.J.; et al. Combined study of ADAMTS13 and complement genes in the diagnosis of thrombotic microangiopathies using next-generation sequencing. Res. Pract. Thromb. Haemost. 2017, 1, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Praga, M.; Rodríguez de Córdoba, S. Secondary atypical hemolytic uremic syndromes in the era of complement blockade. Kidney Int. 2019, 95, 1298–1300. [Google Scholar] [CrossRef]
- Bernabéu-Herrero, M.E.; Jiménez-Alcázar, M.; Anter, J.; Pinto, S.; Sánchez Chinchilla, D.; Garrido, S.; López-Trascasa, M.; de Córdoba, S.R.; Sánchez-Corral, P. Complement factor H, FHR-3 and FHR-1 variants associate in an extended haplotype conferring increased risk of atypical hemolytic uremic syndrome. Mol. Immunol. 2015, 67, 276–286. [Google Scholar] [CrossRef]
- Wu, X.; Liszewski, M.; Java, A.; Atkinson, J. Atypical Hemolytic Uremic Syndrome: Genetically-Based Insights into Pathogenesis through an Analysis of the Complement Regulator CD46. Annals of Blood. 2023. Available online: https://aob.amegroups.org/article/view/7737 (accessed on 24 July 2023).
- van Herpt, T.T.W.; Timmermans, S.A.M.E.G.; van Mook, W.N.K.A.; van Bussel, B.C.T.; van der Horst, I.C.C.; Maessen, J.G.; Natour, E.; van Paassen, P.; Heuts, S. Postsurgical Thrombotic Microangiopathy and Deregulated Complement. J. Clin. Med. 2022, 11, 2501. [Google Scholar] [CrossRef]
- Arjona, E.; Huerta, A.; de Jorge, E.G.; de Córdoba, S.R. Familial risk of developing atypical hemolytic-uremic syndrome. Blood 2020, 136, 1558–1561. [Google Scholar] [CrossRef]
- Petr, V.; Csuka, D.; Hruba, P.; Szilágyi, Á.; Kollar, M.; Slavcev, A.; Prohászka, Z.; Viklicky, O. MCPggaac haplotype is associated with poor graft survival in kidney transplant recipients with de novo thrombotic microangiopathy. Front. Immunol. 2022, 13, 985766. [Google Scholar] [CrossRef]
- Park, M.; Kim, S.; Lee, T.; Lee, S.; Moon, J.; Ihm, C.; Kim, Y.; Kang, S.; Jeong, K.; Chung, J.-H. A Promoter Polymorphism in the CD46 Complement Regulatory Protein Gene Is Associated with Acute Renal Allograft Rejection. Transplant. Proc. 2016, 48, 809–812. [Google Scholar] [CrossRef]
- Verhave, J.C.; Westra, D.; van Hamersvelt, H.W.; van Helden, M.; Kar, N.C.; Wetzels, J.F. Living kidney transplantation in adult patients with atypical haemolytic uraemic syndrome. Neth. J. Med. 2013, 71, 342–347. [Google Scholar] [PubMed]
- Sánchez-Moreno, A.; Cerda, F.; Rodríguez-Barba, A.; Fijo, J.; Bedoya, R.; Arjona, E.; de Córdoba, S.R. Is the atypical hemolytic uremic syndrome risk polymorphism in Membrane Cofactor Protein MCPggaac relevant in kidney transplantation? A case report. Pediatr. Transplant. 2020, 25, e13903. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez de Córdoba, S. Genetic variability shapes the alternative pathway complement activity and predisposition to complement-related diseases. Immunol. Rev. 2022, 313, 71–90. [Google Scholar] [CrossRef] [PubMed]
- Timmermans, S.A.; Wérion, A.; Damoiseaux, J.G.; Morelle, J.; Reutelingsperger, C.P.; van Paassen, P. Diagnostic and Risk Factors for Complement Defects in Hypertensive Emergency and Thrombotic Microangiopathy. Hypertension 2020, 75, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.J.; Fremeaux-Bacchi, V.; Liszewski, M.K.; Pianetti, G.; Noris, M.; Goodship, T.H.J.; Atkinson, J.P. Membrane cofactor protein mutations in atypical hemolytic uremic syndrome (aHUS), fatal Stx-HUS, C3 glomerulonephritis, and the HELLP syndrome. Blood 2008, 111, 624–632. [Google Scholar] [CrossRef]
- Lhotta, K.; Janecke, A.R.; Scheiring, J.; Petzlberger, B.; Giner, T.; Fally, V.; Würzner, R.; Zimmerhackl, L.B.; Mayer, G.; Fremeaux-Bacchi, V. A Large Family with a Gain-of-Function Mutation of Complement C3 Predisposing to Atypical Hemolytic Uremic Syndrome, Microhematuria, Hypertension and Chronic Renal Failure. Clin. J. Am. Soc. Nephrol. 2009, 4, 1356–1362. [Google Scholar] [CrossRef][Green Version]
- Obando, I.; Camacho, M.S.; Falcon-Neyra, D.; Hurtado-Mingo, A.; Neth, O. Atypical Hemolytic Uremic Syndrome Associated with Bordetella pertussis Infection. Pediatr. Infect. Dis. J. 2012, 31, 1210. [Google Scholar] [CrossRef]
- Pelicano, M.B.; de Córdoba, S.R.; Diekmann, F.; Saiz, M.; Herrero, S.; Oppenheimer, F.; Campistol, J.M. Anti-C5 as Prophylactic Therapy in Atypical Hemolytic Uremic Syndrome in Living-Related Kidney Transplantation. Transplantation 2013, 96, e26–e29. [Google Scholar] [CrossRef]
- Szarvas, N.; Szilágyi, Á.; Tasic, V.; Nushi-Stavileci, V.; Sofijanova, A.; Gucev, Z.; Szabó, M.; Szabó, A.; Szeifert, L.; Reusz, G.; et al. First-line therapy in atypical hemolytic uremic syndrome: Consideration on infants with a poor prognosis. Ital. J. Pediatr. 2014, 40, 101. [Google Scholar] [CrossRef]
- Martínez-Barricarte, R.; Heurich, M.; López-Perrote, A.; Tortajada, A.; Pinto, S.; López-Trascasa, M.; Sánchez-Corral, P.; Morgan, B.P.; Llorca, O.; Harris, C.L.; et al. The molecular and structural bases for the association of complement C3 mutations with atypical hemolytic uremic syndrome. Mol. Immunol. 2015, 66, 263–273. [Google Scholar] [CrossRef]
- Valoti, E.; Alberti, M.; Tortajada, A.; Garcia-Fernandez, J.; Gastoldi, S.; Besso, L.; Bresin, E.; Remuzzi, G.; Rodríguez de Cordoba, S.; Noris, M. A Novel Atypical Hemolytic Uremic Syndrome–Associated Hybrid CFHR1/CFH Gene Encoding a Fusion Protein That Antagonizes Factor H–Dependent Complement Regulation. J. Am. Soc. Nephrol. 2015, 26, 209–219. [Google Scholar] [CrossRef]
- García Monteavaro, C.; Peralta Roselló, C.; Quiroga, B.; Baltar Martín, J.M.; Castillo Eraso Eraso, L.; de Álvaro Moreno, F.; Martínez Vea, A.; Visus-Fernández de Manzanos, M.T. Adjustment of Eculizumab Dosage Pattern in Patients with Atypical Hemolytic Uremic Syndrome with Suboptimal Response to Standard Treatment Pattern. Case Rep. Nephrol. 2016, 2016, 7471082. [Google Scholar] [CrossRef]
- Matošević, M.; Kos, I.; Davidović, M.; Ban, M.; Matković, H.; Jakopčić, I.; Brinar, I.V.; Szilágyi, Á.; Csuka, D.; Sinkovits, G.; et al. Hemolytic uremic syndrome in the setting of COVID-19 successfully treated with complement inhibition therapy: An instructive case report of a previously healthy toddler and review of literature. Front. Pediatr. 2023, 11, 1092860. [Google Scholar] [CrossRef] [PubMed]
- Flögelová, H.; Volejníková, J.; Hrachovinová, I.; Prohászka, Z.; Šeda, M.; Gumulec, J. Repeated spontaneous remission of atypical hemolytic-uremic syndrome caused by influenza—A case report. [Opakovaná spontánní remise atypického hemolyticko-uremického syndromu vyvolaného chřipkou—Kazuistika]. Czecho-Slovak Pediatr./Cesko-Slov. Pediatr. 2020, 75, 98–102. [Google Scholar]
- Fakhouri, F.; Fila, M.; Hummel, A.; Ribes, D.; Sellier-Leclerc, A.-L.; Ville, S.; Pouteil-Noble, C.; Coindre, J.-P.; Le Quintrec, M.; Rondeau, E.; et al. Eculizumab discontinuation in children and adults with atypical hemolytic-uremic syndrome: A prospective multicenter study. Blood 2021, 137, 2438–2449. [Google Scholar] [CrossRef]
- Lumbreras, J.; Subias, M.; Espinosa, N.; Ferrer, J.M.; Arjona, E.; Rodríguez de Córdoba, S. The Relevance of the MCP Risk Polymorphism to the Outcome of aHUS Associated with C3 Mutations. A Case Report. Front. Immunol. 2020, 11, 1348. [Google Scholar] [CrossRef] [PubMed]
- Galic, S.; Csuka, D.; Prohászka, Z.; Turudic, D.; Dzepina, P.; Milosevic, D. A case report of a child with sepsis induced multiorgan failure and massive complement consumption treated with a short course of Eculizumab: A case of crosstalk between coagulation and complement? Medicine 2019, 98, e14105. [Google Scholar] [CrossRef] [PubMed]
- Jelicic, I.; Kovacic, V.; Luketin, M.; Mikacic, M.; Skaro, D.B. Atypical HUS with multiple complement system mutations triggered by synthetic psychoactive drug abuse: A case report. J Nephrol. 2023; 1–3, epub ahead of print. [Google Scholar] [CrossRef]
- Kavanagh, D.; Goodship, T.H.; Richards, A. Atypical Hemolytic Uremic Syndrome. Semin. Nephrol. 2013, 33, 508–530. [Google Scholar] [CrossRef]
- Bresin, E.; Rurali, E.; Caprioli, J.; Sanchez-Corral, P.; Fremeaux-Bacchi, V.; Rodríguez de Cordoba, S.; Pinto, S.; Goodship, T.H.; Alberti, M.; Ribes, D.; et al. Combined Complement Gene Mutations in Atypical Hemolytic Uremic Syndrome Influence Clinical Phenotype. J. Am. Soc. Nephrol. 2013, 24, 475–486. [Google Scholar] [CrossRef]
- Bu, F.; Borsa, N.; Gianluigi, A.; Smith, R.J.H. Familial Atypical Hemolytic Uremic Syndrome: A Review of Its Genetic and Clinical Aspects. Clin. Dev. Immunol. 2012, 2012, 370426. [Google Scholar] [CrossRef]
- Fremeaux-Bacchi, V.; Fakhouri, F.; Garnier, A.; Bienaimé, F.; Dragon-Durey, M.-A.; Ngo, S.; Moulin, B.; Servais, A.; Provot, F.; Rostaing, L.; et al. Genetics and Outcome of Atypical Hemolytic Uremic Syndrome: A nationwide French series comparing children and adults. Clin. J. Am. Soc. Nephrol. 2013, 8, 554–562. [Google Scholar] [CrossRef] [PubMed]
- Fremeaux-Bacchi, V.; Kemp, E.J.; Goodship, J.A.; Dragon-Durey, M.-A.; Strain, L.; Loirat, C.; Deng, H.-W.; Goodship, T.H.J. The development of atypical haemolytic-uraemic syndrome is influenced by susceptibility factors in factor H and membrane cofactor protein: Evidence from two independent cohorts. J. Med. Genet. 2005, 42, 852–856. [Google Scholar] [CrossRef] [PubMed]
- Manenti, L.; Gnappi, E.; Vaglio, A.; Allegri, L.; Noris, M.; Bresin, E.; Pilato, F.P.; Valoti, E.; Pasquali, S.; Buzio, C. Atypical haemolytic uraemic syndrome with underlying glomerulopathies. A case series and a review of the literature. Nephrol. Dial. Transplant. 2013, 28, 2246–2259. [Google Scholar] [CrossRef]
- Caprioli, J.; Castelletti, F.; Bucchioni, S.; Bettinaglio, P.; Bresin, E.; Pianetti, G.; Gamba, S.; Brioschi, S.; Daina, E.; Remuzzi, G.; et al. Complement factor H mutations and gene polymorphisms in haemolytic uraemic syndrome: The C-257T, the A2089G and the G2881T polymorphisms are strongly associated with the disease. Hum. Mol. Genet. 2003, 12, 3385–3395. [Google Scholar] [CrossRef] [PubMed]
- Goicoechea de Jorge, E.; Harris, C.L.; Esparza-Gordillo, J.; Carreras, L.; Arranz, E.A.; Garrido, C.A.; López-Trascasa, M.; Sánchez-Corral, P.; Morgan, B.P.; Rodríguez de Córdoba, S. Gain-of-function mutations in complement factor B are associated with atypical hemolytic uremic syndrome. Proc. Natl. Acad. Sci. USA 2007, 104, 240–245. [Google Scholar] [CrossRef]
- Frimat, M.; Roumenina, L.T.; Tabarin, F.; Halbwachs-Mecarelli, L.; Fremeaux-Bacchi, V. Membrane cofactor protein (MCP) haplotype, which predisposes to atypical hemolytic and uremic syndrome, has no consequence on neutrophils and endothelial cells MCP levels or on HUVECs ability to activate complement. Immunobiology 2012, 217, 1187–1188. [Google Scholar] [CrossRef]
- Caprioli, J.; Noris, M.; Brioschi, S.; Pianetti, G.; Castelletti, F.; Bettinaglio, P.; Mele, C.; Bresin, E.; Cassis, L.; Gamba, S.; et al. Genetics of HUS: The impact of MCP, CFH, and IF mutations on clinical presentation, response to treatment, and outcome. Blood 2006, 108, 1267–1279. [Google Scholar] [CrossRef]
- Provaznikova, D.; Rittich, S.; Malina, M.; Seeman, T.; Marinov, I.; Riedl, M.; Hrachovinova, I. Manifestation of atypical hemolytic uremic syndrome caused by novel mutations in MCP. Pediatr. Nephrol. 2011, 27, 73–81. [Google Scholar] [CrossRef]
- Rondeau, E.; Scully, M.; Ariceta, G.; Barbour, T.; Cataland, S.; Heyne, N.; Miyakawa, Y.; Ortiz, S.; Swenson, E.; Vallee, M.; et al. The long-acting C5 inhibitor, Ravulizumab, is effective and safe in adult patients with atypical hemolytic uremic syndrome naïve to complement inhibitor treatment. Kidney Int. 2020, 97, 1287–1296. [Google Scholar] [CrossRef]
- Ariceta, G.; Dixon, B.P.; Kim, S.H.; Kapur, G.; Mauch, T.; Ortiz, S.; Vallee, M.; Denker, A.E.; Kang, H.G.; Greenbaum, L.A.; et al. The long-acting C5 inhibitor, ravulizumab, is effective and safe in pediatric patients with atypical hemolytic uremic syndrome naïve to complement inhibitor treatment. Kidney Int. 2020, 100, 225–237. [Google Scholar] [CrossRef]
- Bouwmeester, R.N.; Duineveld, C.; Wijnsma, K.L.; Bemelman, F.J.; van der Heijden, J.W.; van Wijk, J.A.; Bouts, A.H.; van de Wetering, J.; Dorresteijn, E.; Berger, S.P.; et al. Early Eculizumab Withdrawal in Patients with Atypical Hemolytic Uremic Syndrome in Native Kidneys Is Safe and Cost-Effective: Results of the CUREiHUS Study. Kidney Int. Rep. 2022, 8, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Marini, S.C.; Gomes, M.; Guilherme, R.; Carda, J.P.; Pinto, C.S.; Fidalgo, T.; Ribeiro, M.L. Atypical hemolytic–uremic syndrome: Recurrent phenotypic expression of a patient with MCP gene mutation combined with risk haplotypes. Blood Coagul. Fibrinolysis 2019, 30, 68–70. [Google Scholar] [CrossRef] [PubMed]
- Turudic, D.; Milosevic, D.; Bilic, K.; Prohászka, Z.; Bilic, E. A Limited Course of Eculizumab in a Child with the Atypical He-molytic Uremic Syndrome and Pre-B Acute Lymphoblastic Leukemia on Maintenance Therapy Case Report and Literature Review. J. Clin. Med. 2022, 11, 2779. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, F.; O’halloran, A.; Alghamdi, A.; Chen, C.; Trissal, M.; Traum, A.; DeCourcey, D. Toddler with New Onset Diabetes and Atypical Hemolytic-Uremic Syndrome in the Setting of COVID-19. Pediatrics 2021, 147, e2020016774. [Google Scholar] [CrossRef]
- Mahajan, R.; Lipton, M.; Broglie, L.; Jain, N.G.; Uy, N.S. Eculizumab treatment for renal failure in a pediatric patient with COVID-19. J. Nephrol. 2020, 33, 1373–1376. [Google Scholar] [CrossRef]
- Aurora, T.; Joseph, N.; Bhoopalan, S.V.; Caniza, M.A.; Flerlage, T.; Ghafoor, S.; Hankins, J.; Hijano, D.R.; Jesudas, R.; Kirkham, J.; et al. The successful use of eculizumab for treatment of thrombotic microangiopathy in pediatric acute SARS-CoV-2 infection and multisystem inflammatory syndrome in children. Haematologica 2022, 107, 2517–2522. [Google Scholar] [CrossRef] [PubMed]
- Rysava, R.; Peiskerova, M.; Tesar, V.; Benes, J.; Kment, M.; Szilágyi, Á.; Csuka, D.; Prohászka, Z. Atypical hemolytic uremic syndrome triggered by mRNA vaccination against SARS-CoV-2: Case report. Front. Immunol. 2022, 13, 1001366. [Google Scholar] [CrossRef]
- Walle, J.V.; Delmas, Y.; Ardissino, G.; Wang, J.; Kincaid, J.F.; Haller, H. Improved renal recovery in patients with atypical hemolytic uremic syndrome following rapid initiation of eculizumab treatment. J. Nephrol. 2017, 30, 127–134. [Google Scholar] [CrossRef]
- Levart, T.K. A child with atypical hemolytic uremic syndrome: Is there a rationale to stop eculizumab? Clin. Nephrol. 2020, 93, 215–216. [Google Scholar] [CrossRef]
- Timmermans, S.A.; Damoiseaux, J.G.; Werion, A.; Reutelingsperger, C.P.; Morelle, J.; van Paassen, P. Functional and Genetic Landscape of Complement Dysregulation Along the Spectrum of Thrombotic Microangiopathy and its Potential Implications on Clinical Outcomes. Kidney Int. Rep. 2021, 6, 1099–1109. [Google Scholar] [CrossRef]
- Fakhouri, F.; Fila, M.; Provôt, F.; Delmas, Y.; Barbet, C.; Châtelet, V.; Rafat, C.; Cailliez, M.; Hogan, J.; Servais, A.; et al. Pathogenic Variants in Complement Genes and Risk of Atypical Hemolytic Uremic Syndrome Relapse after Eculizumab Discontinuation. Clin. J. Am. Soc. Nephrol. 2016, 12, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Cugno, M.; Gualtierotti, R.; Possenti, I.; Testa, S.; Tel, F.; Griffini, S.; Grovetti, E.; Tedeschi, S.; Salardi, S.; Cresseri, D.; et al. Complement functional tests for monitoring eculizumab treatment in patients with atypical hemolytic uremic syndrome. J. Thromb. Haemost. 2014, 12, 1440–1448. [Google Scholar] [CrossRef] [PubMed]
| Article | Population | Year | Gene: Variant or Haplotype | Risk Genotype | Additional Genotypes |
|---|---|---|---|---|---|
| Fang et al. [16] | 2008 | MCP: MCPggaac | homozygous | heterozygous R69W MCP and N151S CFI mutations, heterogzygous CFI and c. 905-925del21n mutations | |
| Lhotta [17] | 2009 | MCP: MCPggaac | homozygous | het C3 R570Q mutation, | |
| Obando et al. [18] | Spain | 2012 | MCP: MCPggaac | homozygous | |
| Pelicano et al. [19] | Spain | 2013 | MCP:MCPggaac | homozygous | het CFHcataag (protective haplotype) |
| Szarvas et al. [20] | 2014 | MCP: MCPggaac and CFH H3 | homozygous, heterozygous, compound heterozgyous | Het CFH Y402H; hom CFH Y402H; het CFH E936D; het C3 R102G; het C3 P314L; het CFB R32W; het CFB L9H; het CFH V62I; het CFB R32Q; | |
| Martínez-Barricarte et al. [21] | Spain | 2015 | MCP: MCPggaac | homozygous | |
| MCP: MCPggaac | heterozygous | CFHGATAAG | |||
| MCP: MCPggaac | heterozygous | CFHGATAAG | |||
| MCP: MCPggaac | heterozygous | CFHTGTGGT (homozygous) | |||
| MCP: MCPggaac | heterozygous | CFHTGTGGT (homozygous) | |||
| MCP: MCPggaac | heterozygous | CFHCATAAG (heterozygous) | |||
| Valoti et al. [22] | Italy | 2015 | MCPallele c.*897 T.C (rs7144) | homozygous | het CFH-H3 (TGTGT) |
| Monteavaro et al. [23] | Spain | 2016 | MCP: MCPggaac | homozygous | CFH/CFHR1 hybrid gene, heterozygous CFH (H3) |
| Fidalgo et al. [4] | Spain/Portugal | 2017 | CFH H3:MCPggaac | compound heterozygous | |
| Marini et al. [24] | Portugal | 2019 | MCP: MCPggaac | het (compound) | MCP c.287–2A > G (splice acceptor), MCPggaac and CFH-H3 (compound heterozygous) |
| Flögelová et al. [25] | Czechia | 2020 | MCPggaac haplotype of CD46 gene | heterogygous | MCP (CD46) p.C35Y (heterozygous) |
| Le Clech et al. [3] | 2020 | MCPggaac haplotype of CD46 gene | homozygous | ||
| Levart et al. [26] | Slovenia | 2020 | MCP: MCPggaac | homozygous | heterozygous variation (H508H), heterozygous CHF V621 missense variation |
| Lumbreras et al. [27] | Spain | 2020 | MCP: MCPggaac | homozygous, heterogyzous | MCP: Gly243Val, CFI: Gly162Asp, CFH:Arg885Serfs*13,THBD: (Ala43Thr) |
| Timmermans et al. [15] | 2020 | MCP: MCPggaac | homozygous, heterogyzous | C3 c.463A > C * | |
| Petr V et al. ** [10] | Czechia | 2022 | MCPggaac c.-652A/G, rs2796267 | heterogygous | CFH, CD46, C3, CFB |
| Czechia | 2022 | MCPggaac c.-366A/G, rs2796268 | heterogygous | CFH, CD46, C3, CFB | |
| Czechia | 2022 | MCPggaac IVS9-78G/A (c.989-78G > A), rs1962149 | heterogygous | CFH, CD46, C3, CFB | |
| Rysava et al. [28] | Czechia | 2022 | MCPggaac haplotype of CD46 gene | heterogygous | CFH (c.3096C > A, p.C1032X) (heterozygous) |
| Van. Herpt et al. [8] | Netherlands | 2022 | MCPggaac | heterogygous | C2 c.841_849+19del, deletion of CFHR1 and CFHR3 (all heterozygous) |
| Jelicic I. et al. [29] | Croatia | 2023 | MCPggaac | homozygous | Heterozygous CD46 gene (c.286+2 T > G) splice site mutation, rare heterozygous variant (c.463A > C), homozygous for the CFH H3 haplotype (with the rare alleles c.-331C > T, Q672Q and E936D polymorphisms) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Turudic, D.; Pokrajac, D.; Tasic, V.; Kasumovic, D.; Prohaszka, Z.; Milosevic, D. The Rationale of Complement Blockade of the MCPggaac Haplotype following Atypical Hemolytic Uremic Syndrome of Three Southeastern European Countries with a Literature Review. Int. J. Mol. Sci. 2023, 24, 13041. https://doi.org/10.3390/ijms241713041
Turudic D, Pokrajac D, Tasic V, Kasumovic D, Prohaszka Z, Milosevic D. The Rationale of Complement Blockade of the MCPggaac Haplotype following Atypical Hemolytic Uremic Syndrome of Three Southeastern European Countries with a Literature Review. International Journal of Molecular Sciences. 2023; 24(17):13041. https://doi.org/10.3390/ijms241713041
Chicago/Turabian StyleTurudic, Daniel, Danka Pokrajac, Velibor Tasic, Dino Kasumovic, Zoltan Prohaszka, and Danko Milosevic. 2023. "The Rationale of Complement Blockade of the MCPggaac Haplotype following Atypical Hemolytic Uremic Syndrome of Three Southeastern European Countries with a Literature Review" International Journal of Molecular Sciences 24, no. 17: 13041. https://doi.org/10.3390/ijms241713041
APA StyleTurudic, D., Pokrajac, D., Tasic, V., Kasumovic, D., Prohaszka, Z., & Milosevic, D. (2023). The Rationale of Complement Blockade of the MCPggaac Haplotype following Atypical Hemolytic Uremic Syndrome of Three Southeastern European Countries with a Literature Review. International Journal of Molecular Sciences, 24(17), 13041. https://doi.org/10.3390/ijms241713041






