Exploring Albumin Functionality Assays: A Pilot Study on Sepsis Evaluation in Intensive Care Medicine
Abstract
1. Introduction
2. Results
2.1. Study Population
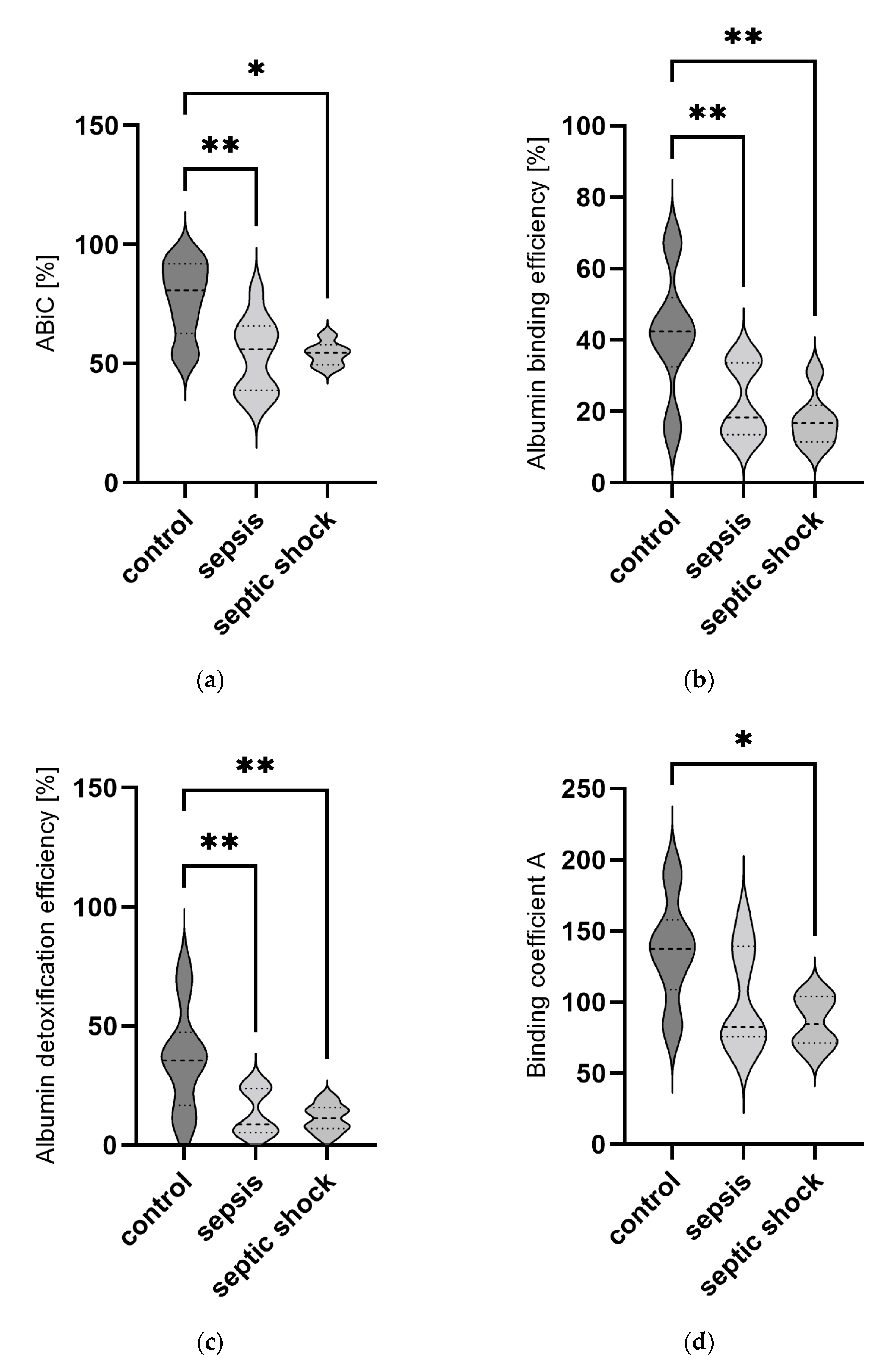
2.2. Albumin Binding in Patients on Intensive Care Unit (ICU) Admission
2.3. Comparison to Clinically Established Standard Parameters for the Assessment of the Patient’s Condition
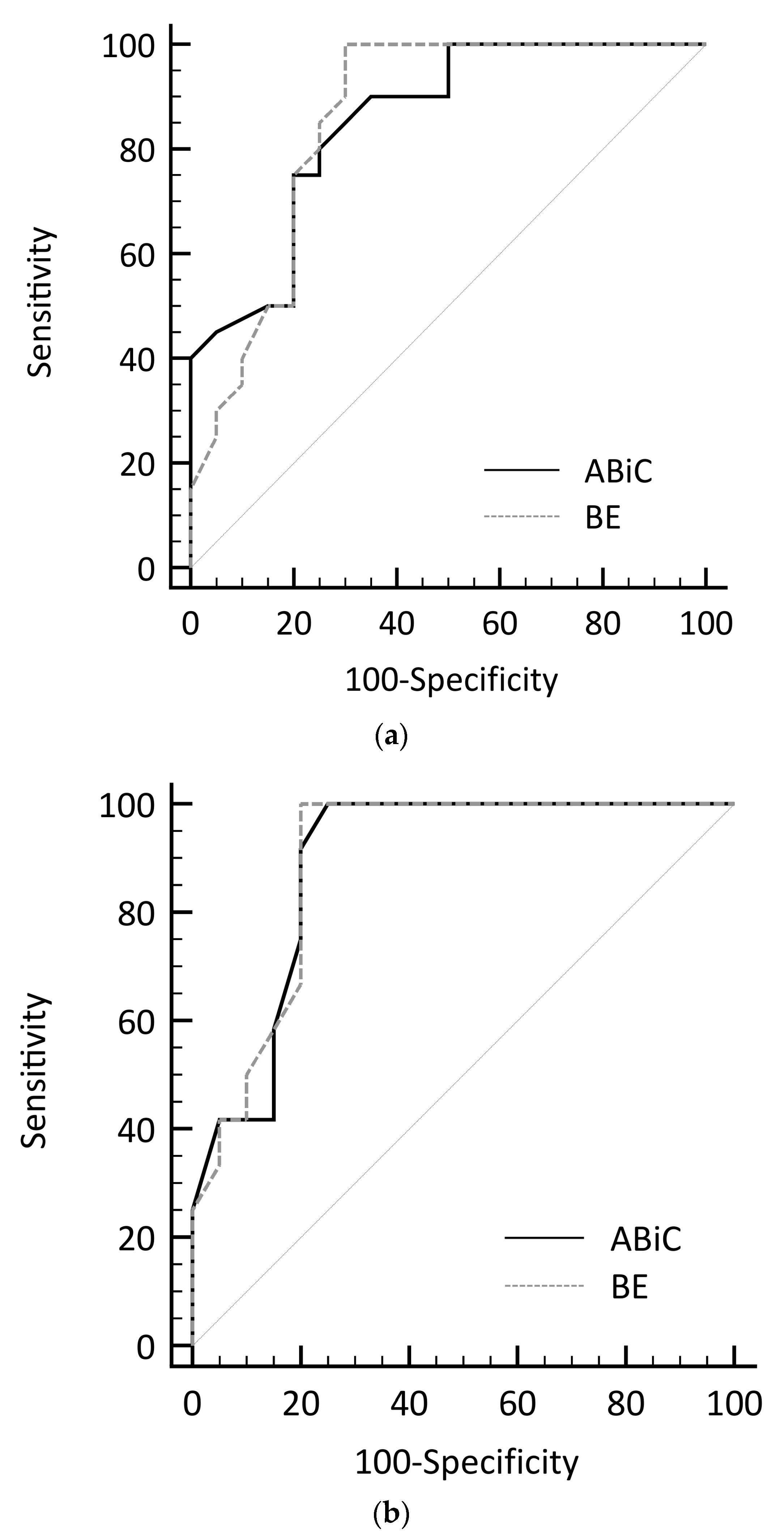
2.4. Patient Survival
2.5. Multiple Linear Regression
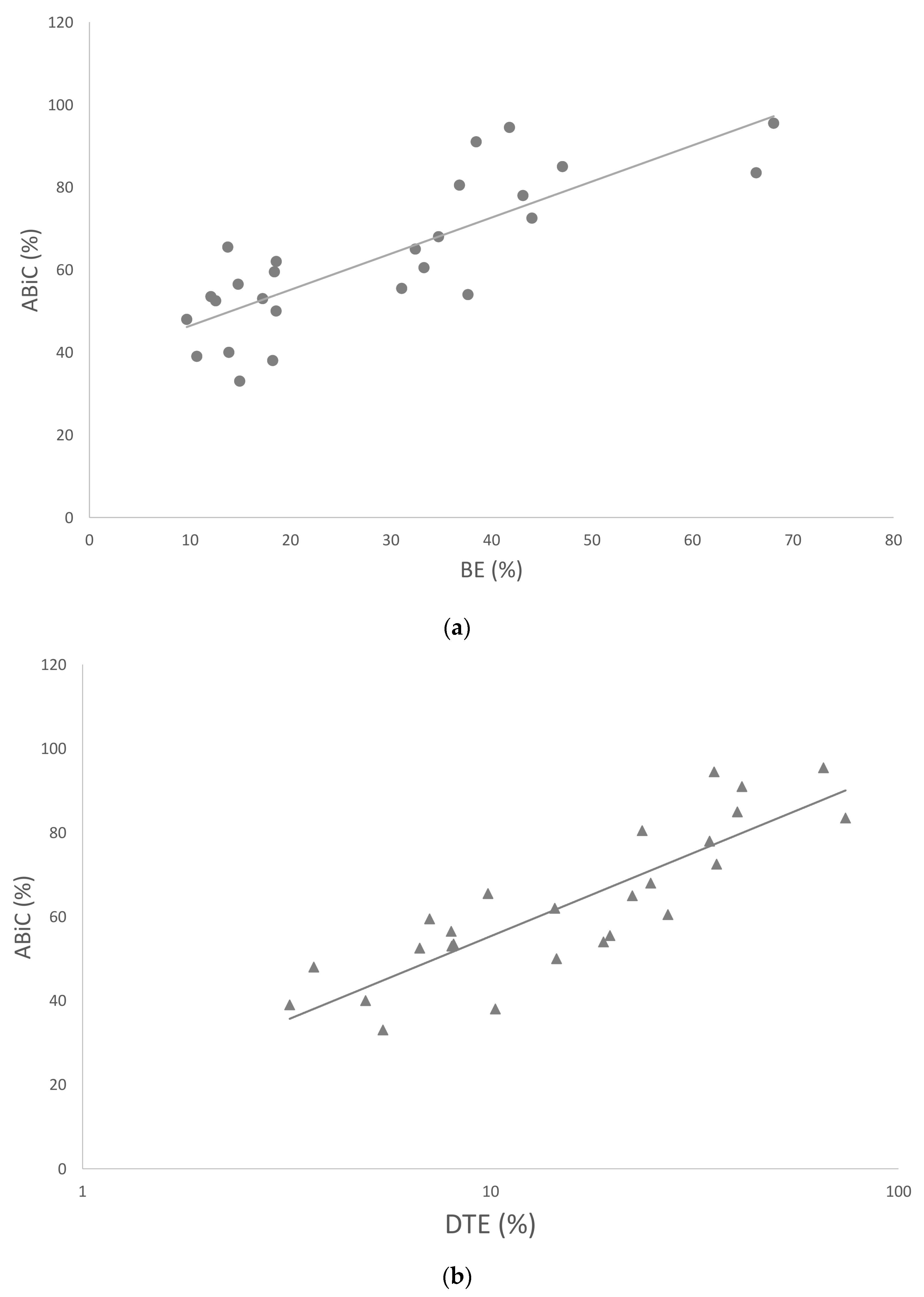
2.6. Comparison of the Applied Methods for Albumin Binding Efficiency
3. Discussion
4. Material and Methods
4.1. Design, Settings and Study Population
4.2. Measurement of Albumin Concentration
4.3. Estimation of ABiC
4.4. Determination of Binding and Detoxification Efficiency by Electron Paramagnetic Resonance (EPR)-Spectroscopy
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; Mcintyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Crit. Care Med. 2021, 49, e1063. [Google Scholar] [CrossRef] [PubMed]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Häufigste Vermeidbare Todesursache Im Land. Available online: https://www.aerztezeitung.de/Politik/Haeufigste-vermeidbare-Todesursache-im-Land-224930.html (accessed on 30 June 2023).
- Global Report on the Epidemiology and Burden of Sepsis. Available online: https://www.who.int/publications-detail-redirect/9789240010789 (accessed on 24 July 2023).
- Schlapbach, L.J.; Kissoon, N.; Alhawsawi, A.; Aljuaid, M.H.; Daniels, R.; Gorordo-Delsol, L.A.; Machado, F.; Malik, I.; Nsutebu, E.F.; Finfer, S.; et al. World Sepsis Day: A Global Agenda to Target a Leading Cause of Morbidity and Mortality. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 319, L518–L522. [Google Scholar] [CrossRef]
- Yealy, D.M.; Mohr, N.M.; Shapiro, N.I.; Venkatesh, A.; Jones, A.E.; Self, W.H. Early Care of Adults With Suspected Sepsis in the Emergency Department and Out-of-Hospital Environment: A Consensus-Based Task Force Report. Ann. Emerg. Med. 2021, 78, 1–19. [Google Scholar] [CrossRef]
- Guarino, M.; Perna, B.; Cesaro, A.E.; Maritati, M.; Spampinato, M.D.; Contini, C.; De Giorgio, R. 2023 Update on Sepsis and Septic Shock in Adult Patients: Management in the Emergency Department. J. Clin. Med. 2023, 12, 3188. [Google Scholar] [CrossRef]
- Evans, T.W. Review Article: Albumin as a Drug--Biological Effects of Albumin Unrelated to Oncotic Pressure. Aliment. Pharmacol. Ther. 2002, 16 (Suppl. S5), 6–11. [Google Scholar] [CrossRef]
- Bihari, S.; Bannard-Smith, J.; Bellomo, R. Albumin as a Drug: Its Biological Effects beyond Volume Expansion. Crit. Care Resusc. 2020, 22, 257–265. [Google Scholar]
- Kragh-Hansen, U.; Chuang, V.T.G.; Otagiri, M. Practical Aspects of the Ligand-Binding and Enzymatic Properties of Human Serum Albumin. Biol. Pharm. Bull. 2002, 25, 695–704. [Google Scholar] [CrossRef]
- Paris, G.; Ramseyer, C.; Enescu, M. A Principal Component Analysis of the Dynamics of Subdomains and Binding Sites in Human Serum Albumin. Biopolymers 2014, 101, 561–572. [Google Scholar] [CrossRef]
- Fasano, M.; Curry, S.; Terreno, E.; Galliano, M.; Fanali, G.; Narciso, P.; Notari, S.; Ascenzi, P. The Extraordinary Ligand Binding Properties of Human Serum Albumin. IUBMB Life 2005, 57, 787–796. [Google Scholar] [CrossRef] [PubMed]
- Arnau-Barrés, I.; Güerri-Fernández, R.; Luque, S.; Sorli, L.; Vázquez, O.; Miralles, R. Serum Albumin Is a Strong Predictor of Sepsis Outcome in Elderly Patients. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 743–746. [Google Scholar] [CrossRef]
- Gurachevsky, A.; Kazmierczak, S.C.; Jörres, A.; Muravsky, V. Application of Spin Label Electron Paramagnetic Resonance in the Diagnosis and Prognosis of Cancer and Sepsis. Clin. Chem. Lab. Med. 2008, 46, 1203–1210. [Google Scholar] [CrossRef]
- Klammt, S.; Brinkmann, B.; Mitzner, S.; Munzert, E.; Loock, J.; Stange, J.; Emmrich, J.; Liebe, S. Albumin Binding Capacity (ABiC) Is Reduced in Commercially Available Human Serum Albumin Preparations with Stabilizers. Z. Gastroenterol. 2001, 39 (Suppl. S2), 24–27. [Google Scholar] [CrossRef] [PubMed]
- Klammt, S.; Mitzner, S.R.; Stange, J.; Loock, J.; Heemann, U.; Emmrich, J.; Reisinger, E.C.; Schmidt, R. Improvement of Impaired Albumin Binding Capacity in Acute-on-Chronic Liver Failure by Albumin Dialysis. Liver Transpl. 2008, 14, 1333–1339. [Google Scholar] [CrossRef] [PubMed]
- Klammt, S.; Wojak, H.-J.; Mitzner, A.; Koball, S.; Rychly, J.; Reisinger, E.C.; Mitzner, S. Albumin-Binding Capacity (ABiC) Is Reduced in Patients with Chronic Kidney Disease along with an Accumulation of Protein-Bound Uraemic Toxins. Nephrol. Dial. Transpl. 2012, 27, 2377–2383. [Google Scholar] [CrossRef] [PubMed]
- Seidel, P.; Gurachevsky, A.; Muravsky, V.; Schnurr, K.; Seibt, G.; Matthes, G. Recognition of Malignant Processes with Neural Nets from ESR Spectra of Serum Albumin. Z. Med. Phys. 2005, 15, 265–272. [Google Scholar] [CrossRef]
- Kazmierczak, S.C.; Gurachevsky, A.; Matthes, G.; Muravsky, V. Electron Spin Resonance Spectroscopy of Serum Albumin: A Novel New Test for Cancer Diagnosis and Monitoring. Clin. Chem. 2006, 52, 2129–2134. [Google Scholar] [CrossRef]
- Muravsky, V.; Gurachevskaya, T.; Berezenko, S.; Schnurr, K.; Gurachevsky, A. Fatty Acid Binding Sites of Human and Bovine Albumins: Differences Observed by Spin Probe ESR. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 74, 42–47. [Google Scholar] [CrossRef]
- Jalan, R.; Schnurr, K.; Mookerjee, R.P.; Sen, S.; Cheshire, L.; Hodges, S.; Muravsky, V.; Williams, R.; Matthes, G.; Davies, N.A. Alterations in the Functional Capacity of Albumin in Patients with Decompensated Cirrhosis Is Associated with Increased Mortality. Hepatology 2009, 50, 555–564. [Google Scholar] [CrossRef]
- Baldassarre, M.; Naldi, M.; Zaccherini, G.; Bartoletti, M.; Antognoli, A.; Laggetta, M.; Gagliardi, M.; Tufoni, M.; Domenicali, M.; Waterstradt, K.; et al. Determination of Effective Albumin in Patients With Decompensated Cirrhosis: Clinical and Prognostic Implications. Hepatology 2021, 74, 2058–2073. [Google Scholar] [CrossRef]
- Boss, K.; Waterstradt, K.; Schnurr, K.; Paar, M.; Solpe, S.; Ickerott, P.; Wieneke, U.; Spitthöver, R.; Oettl, K.; Kribben, A. Binding and Detoxification Efficiency of Albumin Decline after Haemodialysis. Nephrol. Dial. Transplant. 2023. [Google Scholar]
- Wilkes, M.M.; Navickis, R.J. Patient Survival after Human Albumin Administration. A Meta-Analysis of Randomized, Controlled Trials. Ann. Intern. Med. 2001, 135, 149–164. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.-L.; Dubois, M.-J.; Navickis, R.J.; Wilkes, M.M. Hypoalbuminemia in Acute Illness: Is There a Rationale for Intervention? A Meta-Analysis of Cohort Studies and Controlled Trials. Ann. Surg. 2003, 237, 319–334. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.-L.; Wilkes, M.M.; Navickis, R.J. Safety of Human Albumin—Serious Adverse Events Reported Worldwide in 1998–2000. Br. J. Anaesth. 2003, 91, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Dubois, M.-J.; Orellana-Jimenez, C.; Melot, C.; De Backer, D.; Berre, J.; Leeman, M.; Brimioulle, S.; Appoloni, O.; Creteur, J.; Vincent, J.-L. Albumin Administration Improves Organ Function in Critically Ill Hypoalbuminemic Patients: A Prospective, Randomized, Controlled, Pilot Study. Crit. Care Med. 2006, 34, 2536–2540. [Google Scholar] [CrossRef]
- Wiedermann, C.J. Hypoalbuminemia as Surrogate and Culprit of Infections. Int. J. Mol. Sci. 2021, 22, 4496. [Google Scholar] [CrossRef]
- Sudlow, G.; Birkett, D.J.; Wade, D.N. The Characterization of Two Specific Drug Binding Sites on Human Serum Albumin. Mol. Pharmacol. 1975, 11, 824–832. [Google Scholar]
- Yamasaki, K.; Hyodo, S.; Taguchi, K.; Nishi, K.; Yamaotsu, N.; Hirono, S.; Chuang, V.T.G.; Seo, H.; Maruyama, T.; Otagiri, M. Long Chain Fatty Acids Alter the Interactive Binding of Ligands to the Two Principal Drug Binding Sites of Human Serum Albumin. PLoS ONE 2017, 12, e0180404. [Google Scholar] [CrossRef]
- Zampieri, F.G.; Kellum, J.A.; Park, M.; Ranzani, O.T.; Barbeiro, H.V.; de Souza, H.P.; da Cruz Neto, L.M.; da Silva, F.P. Relationship between Acid-Base Status and Inflammation in the Critically Ill. Crit. Care 2014, 18, R154. [Google Scholar] [CrossRef]
- Nasif, W.A.; Mukhtar, M.H.; El-Emshaty, H.M.; Alwazna, A.H. Redox State of Human Serum Albumin and Inflammatory Biomarkers in Hemodialysis Patients with Secondary Hyperparathyroidism During Oral Calcitriol Supplementation for Vitamin D. Open. Med. Chem. J. 2018, 12, 98–110. [Google Scholar] [CrossRef]
- Himmelfarb, J.; McMonagle, E. Albumin Is the Major Plasma Protein Target of Oxidant Stress in Uremia. Kidney Int. 2001, 60, 358–363. [Google Scholar] [CrossRef] [PubMed]
- Colombo, G.; Clerici, M.; Giustarini, D.; Rossi, R.; Milzani, A.; Dalle-Donne, I. Redox Albuminomics: Oxidized Albumin in Human Diseases. Antioxid. Redox Signal. 2012, 17, 1515–1527. [Google Scholar] [CrossRef] [PubMed]
- Roohk, H.V.; Zaidi, A.R.; Patel, D. Glycated Albumin (GA) and Inflammation: Role of GA as a Potential Marker of Inflammation. Inflamm. Res. 2018, 67, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Klammt, S.; Mitzner, S.; Stange, J.; Brinkmann, B.; Drewelow, B.; Emmrich, J.; Liebe, S.; Schmidt, R. Albumin-Binding Function Is Reduced in Patients with Decompensated Cirrhosis and Correlates Inversely with Severity of Liver Disease Assessed by Model for End-Stage Liver Disease. Eur. J. Gastroenterol. Hepatol. 2007, 19, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Klinkmann, G.; Klammt, S.; Jäschke, M.; Henschel, J.; Gloger, M.; Reuter, D.A.; Mitzner, S. Impact of Albumin Binding Function on Pharmacokinetics and Pharmacodynamics of Furosemide. Medicina 2022, 58, 1780. [Google Scholar] [CrossRef] [PubMed]
- Woreta, T.A.; Alqahtani, S.A. Evaluation of Abnormal Liver Tests. Med. Clin. N. Am. 2014, 98, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, Z.; Yu, M.; Jiang, Y. The Predictive Ability of Liver Function Indexes on 18F-FDG Uptake in the Liver. Nuklearmedizin 2017, 56, 239–242. [Google Scholar] [CrossRef]
- Oettl, K.; Stadlbauer, V.; Petter, F.; Greilberger, J.; Putz-Bankuti, C.; Hallström, S.; Lackner, C.; Stauber, R.E. Oxidative Damage of Albumin in Advanced Liver Disease. Biochim. Biophys. Acta 2008, 1782, 469–473. [Google Scholar] [CrossRef]
- Paar, M.; Fengler, V.H.; Reibnegger, G.; Schnurr, K.; Waterstradt, K.; Schwaminger, S.P.; Stauber, R.E.; Oettl, K. Determination of Binding Characteristics as a Measure for Effective Albumin Using Different Methods. Biochim. Biophys. Acta Gen. Subj. 2023, 1867, 130427. [Google Scholar] [CrossRef]
- Barichello, T.; Generoso, J.S.; Singer, M.; Dal-Pizzol, F. Biomarkers for Sepsis: More than Just Fever and Leukocytosis—A Narrative Review. Crit. Care 2022, 26, 14. [Google Scholar] [CrossRef]
- Bhattacharya, A.A.; Grüne, T.; Curry, S. Crystallographic Analysis Reveals Common Modes of Binding of Medium and Long-Chain Fatty Acids to Human Serum Albumin. J. Mol. Biol. 2000, 303, 721–732. [Google Scholar] [CrossRef]
- Friedrichs, B. Th. Peters. Jr.: All about Albumin. Biochemistry, Genetics, and Medical Applications. XX and 432 Pages, Numerous Figures and Tables. Academic Press, Inc., San Diego, California, 1996. Price: 85.00 US $. Food Nahr. 1997, 41, 382. [Google Scholar] [CrossRef]
- Taboada, P.; Barbosa, S.; Castro, E.; Gutiérrez-Pichel, M.; Mosquera, V. Effect of Solvation on the Structure Conformation of Human Serum Albumin in Aqueous–Alcohol Mixed Solvents. Chem. Phys. 2007, 340, 59–68. [Google Scholar] [CrossRef]
- Reed, R.G.; Burrington, C.M. The Albumin Receptor Effect May Be Due to a Surface-Induced Conformational Change in Albumin. J. Biol. Chem. 1989, 264, 9867–9872. [Google Scholar] [CrossRef] [PubMed]
- Spin Labeling—1st Edition. Available online: https://shop.elsevier.com/books/spin-labeling/berliner/978-0-12-092350-2 (accessed on 30 June 2023).
- Matthes, G.; Seibt, G.; Muravsky, V.; Hersmann, G.; Dornheim, G. Albumin Transport Analysis of Different Collected and Processed Plasma Products by Electron Spin Resonance Spectroscopy. Transfus. Apher. Sci. 2002, 27, 129–135. [Google Scholar] [CrossRef]

| Control (n = 10) | Sepsis (n = 10) | Septic Shock (n = 6) | p Value | |
|---|---|---|---|---|
| Anthropometric data | ||||
| Age (years) | 58.5 (49.3–62.0) | 62.5 (52.8–79.0) | 61 (54.8–68.0) | 0.551 |
| Male sex | 6 (60) | 7 (70) | 5 (83.3) | |
| Hematology, biochemistry, and prognostic scores | ||||
| Hemoglobin (mmol/L) | 6.9 (5.9–8.7) | 4.9 (4.5–5.5) | 5.9 (5.4–6.2) | 0.141 |
| Erythrocytes (1012/L) | 3.5 (2.8–3.9) | 2.7 (2.4–2.8) | 2.9 (2.8–3.2) | 0.026 |
| Hematocrit | 0.31 (0.26–0.33) | 0.26 (0.23–0.26) | 0.28 (0.26–0.30) | 0.005 |
| Platelets (109/L) | 227 (211–266) | 128 (86–272) | 114 (62–180) | 0.052 |
| Leukocytes (109/L) | 9.6 (7.1–11.6) | 10.2 (8.2–12.2) | 15.5 (13.0–17.0) | 0.382 |
| C-reactive protein (CRP) (mg/L) | 85 (56–120) | 151 (126–206) | 161 (122–242) | 0.037 |
| Procalcitonin (PCT) (ng/mL) | 0.1 (0.1–0.2) | 1.1 (0.6–3.0) | 11.5 (7.8–18.6) | <0.0001 |
| Lactate (mg/dL) | 9.0 (5.8–13.3) | 4.6 (0.7–10.6) | 21.6 (18.5–44.4) | 0.02 |
| Albumin (g/L) | 36 (28.7–38.1) | 22.5 (20.9–30.2) | 20.7 (19.7–22.1) | 0.005 |
| Bilirubin (µmol/L) | 8.5 (3.8–12.5) | 8.5 (6.0–11.8) | 30.0 (27.0–38.0) | <0.0001 |
| Creatinine (µmol/L) | 57.5 (54.3–75.5) | 79.0 (60.8–100.5) | 108.5 (83.3–149.5) | 0.107 |
| Urea (mmol/L) | 5.5 (4.6–6.8) | 8.6 (5.7–17.3) | 14.6 (9.5–21.9) | 0.029 |
| Sodium (mmol/L) | 139 (138–140) | 139 (136–140) | 134 (131–137) | 0.162 |
| Potassium (mmol/L) | 4.0 (3.8–4.2) | 3.9 (3.6–4.3) | 4.7 (4.4–4.9) | 0.002 |
| Calcium (mmol/L) | 1.2 (1.2–1.2) | 1.1 (1.1–1.2) | 1.2 (1.1–1.3) | 0.192 |
| Sequential organ failure assessment (SOFA) score | 1.0 (0.3–1.0) | 6.0 (4.3–7.5) | 10 (10.0–11.5) | <0.0001 |
| Reasons for ICU admission n (%) | ||||
| Hospital-aquired pneumonia | 0 (0) | 5 (50) | 2 (33.3) | |
| Soft tissue infection | 0 (0) | 2 (20) | 1 (16.6) | |
| Urosepsis | 0 (0) | 1 (10) | 0 (0) | |
| Abdominal infection | 0 (0) | 1 (10) | 0 (0) | |
| Spondylodiscitis | 0 (0) | 1 (10) | 0 (0) | |
| Mediastinitis | 0 (0) | 0 (0) | 2 (33.3) | |
| Endocarditis | 0 (0) | 0 (0) | 1 (16.6) | |
| ABiC (%) | BE (%) | DTE (%) | ||||
|---|---|---|---|---|---|---|
| Parameter | r | p-Value | r | p-Value | r | p-Value |
| PCT (ng/mL) | −0.55 | 0.0064 | −0.73 | 0.0001 | −0.62 | 0.0016 |
| CRP (mg/L) | −0.39 | 0.0656 | −0.48 | 0.0219 | −0.42 | 0.0455 |
| SOFA score | −0.53 | 0.0098 | −0.64 | 0.0010 | −0.56 | 0.0058 |
| Parameter | Survivors (n = 21) | Non-Survivors (n = 5) | p-Value |
|---|---|---|---|
| ABiC (%) | 62 (52–81) | 54 (52–62) | 0.313 |
| BE (%) | 31 (15–42) | 19 (12–33) | 0.255 |
| DTE (%) | 19 (8–35) | 15 (8–24) | 0.720 |
| PCT (ng/mL) | 1.0 (0.1–3.5) | 4.4 (0.5–15) | 0.157 |
| CRP (mg/L) | 124 (61–189) | 148 (120–213) | 0.283 |
| SOFA score | 4 (1–8) | 8 (4–12) | 0.090 |
| Parameter | Description |
|---|---|
| BE (kbG × nG) | binding efficiency |
| DTE (LQ/UQ) | detoxification efficiency |
| KB A [(1 − C3)/(CAlb × C3)] | binding coefficient of albumin at highest ethanol concentration (17 Vol%) |
| KB B [(1 − C3)/(CAlb × C3)] | binding coefficient of albumin at highest ethanol concentration (19 Vol%) |
| KB C [(1 − C3)/(CAlb × C3)] | binding coefficient of albumin at highest ethanol concentration (22 Vol%) |
| kbG | global binding constant of whole albumin molecule, determined as regression of ratios between relative amounts of spin-probe molecules within both binding sides, strongly and weakly immobilized, to the concentration of ethanol |
| nG | amount of spin-probe molecules within both binding sides, strongly and weakly immobilized |
| LQ | loading quality—binding property under low ethanol (17 Vol%) and low spin probe (spin probe/albumin ratio 1) conditions |
| UQ | unloading quality—binding property under high ethanol (22 Vol%) and high spin probe (spin probe/albumin ratio 3) conditions |
| C3 | fraction of unbound fatty acids |
| CAlb | patient’s albumin concentration |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klinkmann, G.; Waterstradt, K.; Klammt, S.; Schnurr, K.; Schewe, J.-C.; Wasserkort, R.; Mitzner, S. Exploring Albumin Functionality Assays: A Pilot Study on Sepsis Evaluation in Intensive Care Medicine. Int. J. Mol. Sci. 2023, 24, 12551. https://doi.org/10.3390/ijms241612551
Klinkmann G, Waterstradt K, Klammt S, Schnurr K, Schewe J-C, Wasserkort R, Mitzner S. Exploring Albumin Functionality Assays: A Pilot Study on Sepsis Evaluation in Intensive Care Medicine. International Journal of Molecular Sciences. 2023; 24(16):12551. https://doi.org/10.3390/ijms241612551
Chicago/Turabian StyleKlinkmann, Gerd, Katja Waterstradt, Sebastian Klammt, Kerstin Schnurr, Jens-Christian Schewe, Reinhold Wasserkort, and Steffen Mitzner. 2023. "Exploring Albumin Functionality Assays: A Pilot Study on Sepsis Evaluation in Intensive Care Medicine" International Journal of Molecular Sciences 24, no. 16: 12551. https://doi.org/10.3390/ijms241612551
APA StyleKlinkmann, G., Waterstradt, K., Klammt, S., Schnurr, K., Schewe, J.-C., Wasserkort, R., & Mitzner, S. (2023). Exploring Albumin Functionality Assays: A Pilot Study on Sepsis Evaluation in Intensive Care Medicine. International Journal of Molecular Sciences, 24(16), 12551. https://doi.org/10.3390/ijms241612551





