The Effects of Bisphenol A on Human Male Infertility: A Review of Current Epidemiological Studies
Abstract
1. Introduction
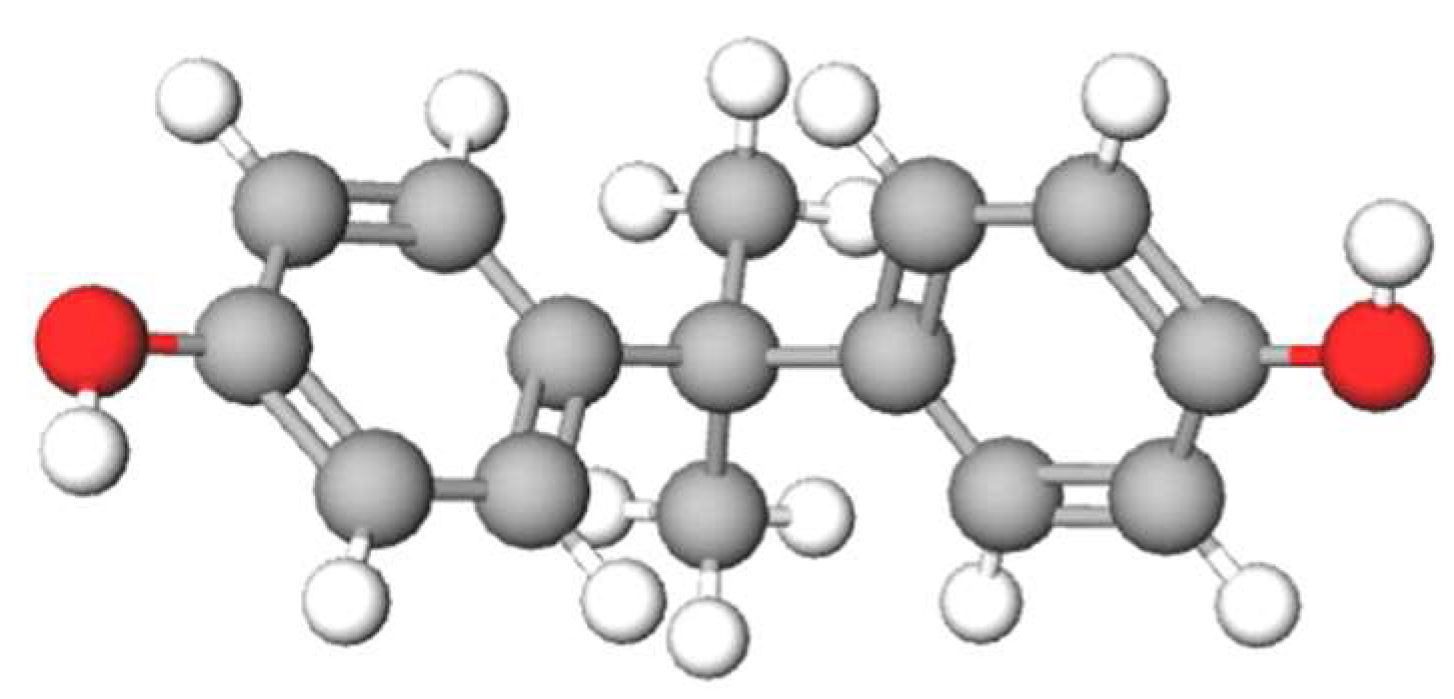
2. Physical and Chemical Properties of Bisphenol A
3. Production and Application
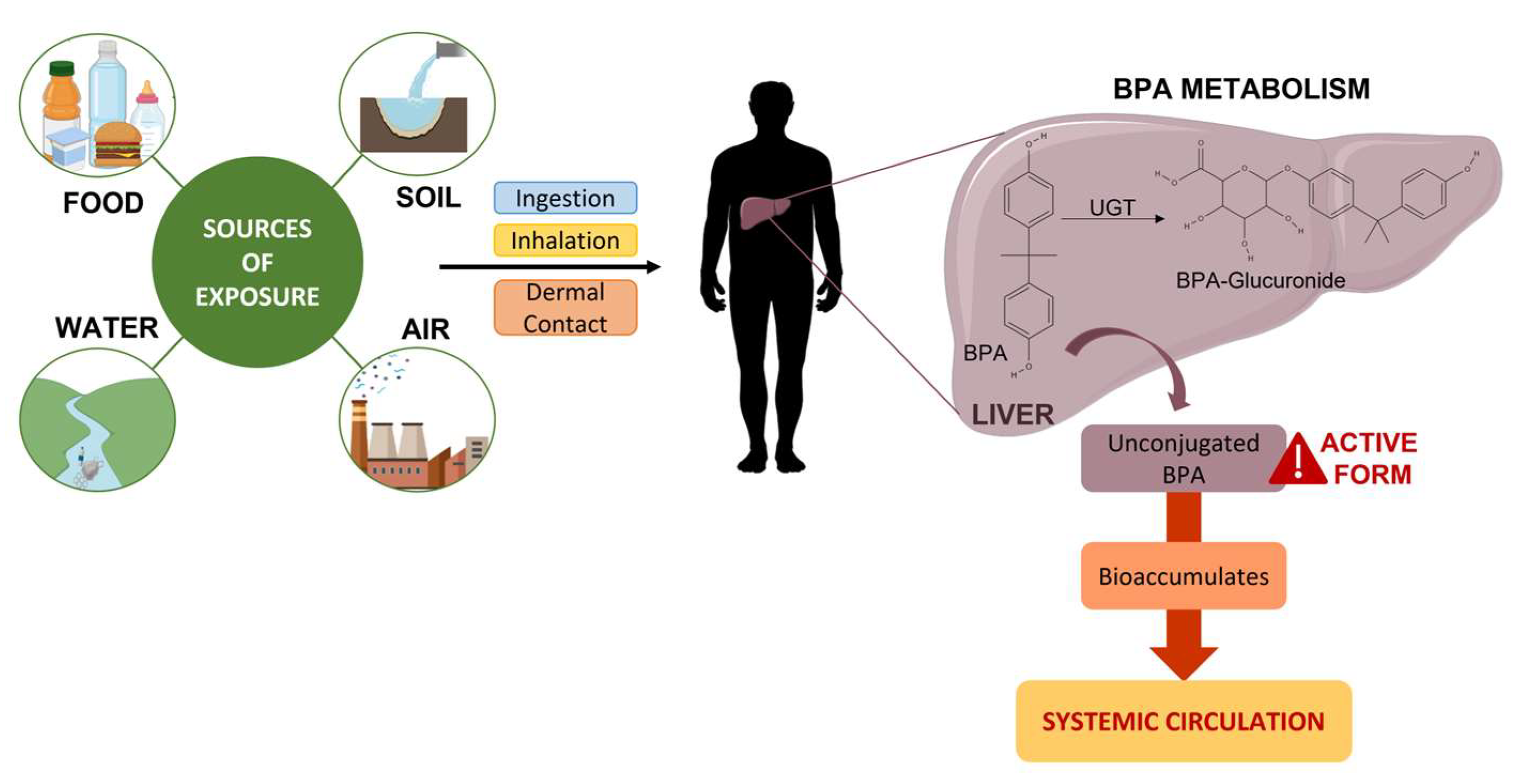
4. Sources of Exposure
4.1. Food
4.2. Water
4.3. Air
4.4. Soil
5. Conjugation, Metabolism, and Excretion
Detection in Biological Samples
6. Mechanisms of Action
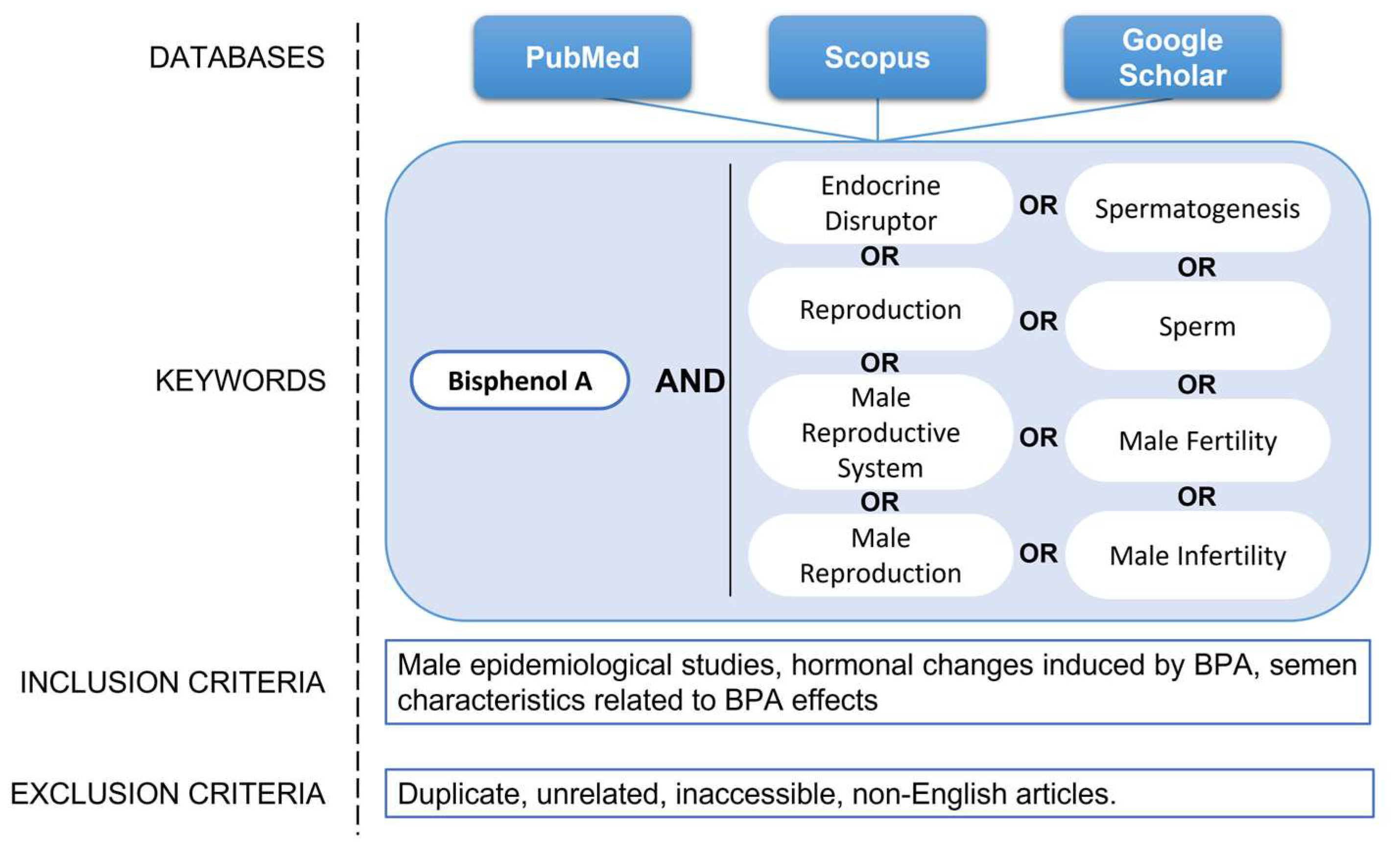
7. Methods
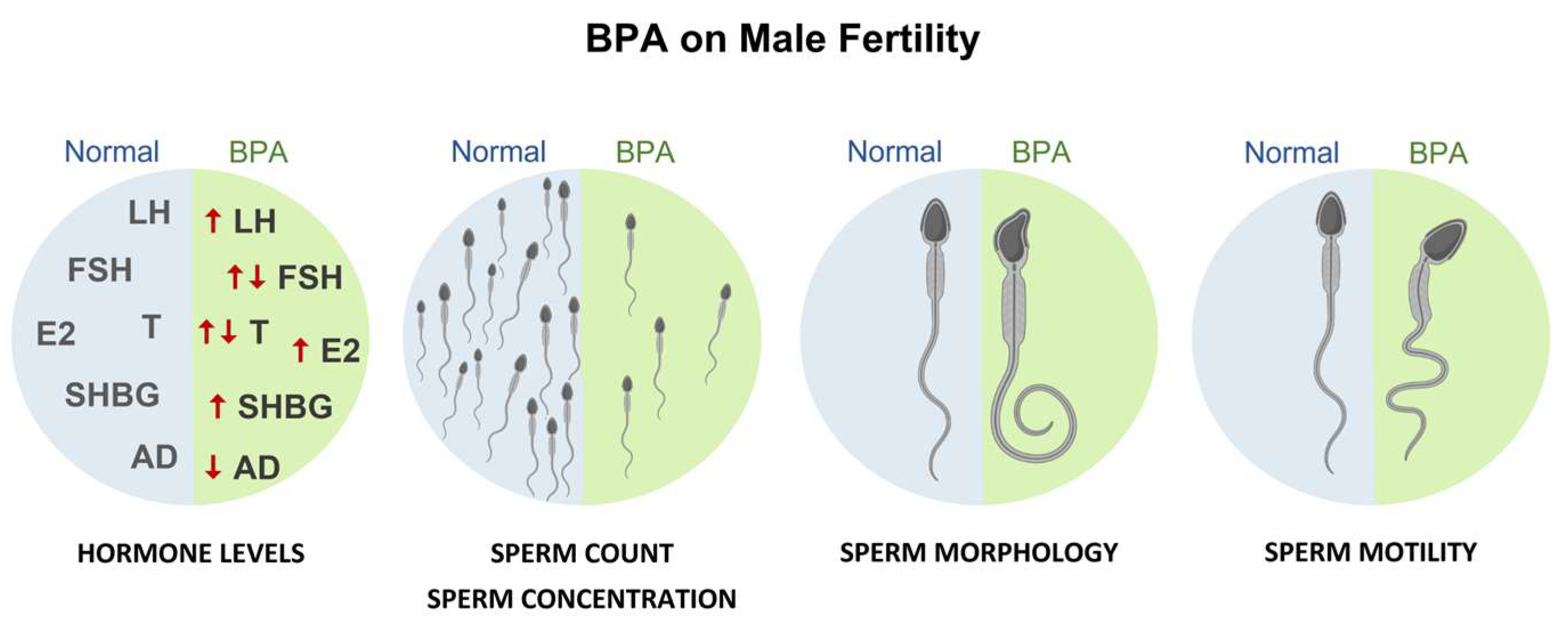
8. Effects on Human Male Fertility
Evidence from Epidemiological Studies
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Monneret, C. What is an endocrine disruptor? Comptes Rendus Biol. 2017, 340, 403–405. [Google Scholar] [CrossRef]
- Kabir, E.R.; Rahman, M.S.; Rahman, I. A review on endocrine disruptors and their possible impacts on human health. Environ. Toxicol. Pharmacol. 2015, 40, 241–258. [Google Scholar] [CrossRef]
- Kavlock, R.J.; Daston, G.P.; DeRosa, C.; Fenner-Crisp, P.; Gray, L.E.; Kaattari, S.; Lucier, G.; Luster, M.; Mac, M.J.; Maczka, C.; et al. Research needs for the risk assessment of health and environmental effects of endocrine disruptors: A report of the U.S. EPA-sponsored workshop. Environ. Health Perspect. 1996, 104 (Suppl. S4), 715–740. [Google Scholar] [CrossRef] [PubMed]
- Diamanti-Kandarakis, E.; Bourguignon, J.P.; Giudice, L.C.; Hauser, R.; Prins, G.S.; Soto, A.M.; Zoeller, R.T.; Gore, A.C. Endocrine-disrupting chemicals: An Endocrine Society scientific statement. Endocr. Rev. 2009, 30, 293–342. [Google Scholar] [CrossRef] [PubMed]
- Vandenberg, L.N.; Hunt, P.A.; Gore, A.C. Endocrine disruptors and the future of toxicology testing—Lessons from CLARITY-BPA. Nat. Rev. Endocrinol. 2019, 15, 366–374. [Google Scholar] [CrossRef]
- Abraham, A.; Chakraborty, P. A review on sources and health impacts of bisphenol A. Rev. Environ. Health 2020, 35, 201–210. [Google Scholar] [CrossRef]
- Rubin, B.S. Bisphenol A: An endocrine disruptor with widespread exposure and multiple effects. J. Steroid Biochem. Mol. Biol. 2011, 127, 27–34. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Maffini, M.V.; Sonnenschein, C.; Rubin, B.S.; Soto, A.M. Bisphenol-A and the great divide: A review of controversies in the field of endocrine disruption. Endocr. Rev. 2009, 30, 75–95. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, H.; Wu, J.; Yuan, L.; Wang, Y.; Du, X.; Wang, R.; Marwa, P.W.; Petlulu, P.; Chen, X.; et al. The adverse health effects of bisphenol A and related toxicity mechanisms. Environ. Res. 2019, 176, 108575. [Google Scholar] [CrossRef]
- Kang, J.H.; Kondo, F.; Katayama, Y. Human exposure to bisphenol A. Toxicology 2006, 226, 79–89. [Google Scholar] [CrossRef]
- Michalowicz, J. Bisphenol A—Sources, toxicity and biotransformation. Environ. Toxicol. Pharmacol. 2014, 37, 738–758. [Google Scholar] [CrossRef]
- Fonseca, M.I.; Lorigo, M.; Cairrao, E. Endocrine-Disrupting Effects of Bisphenol A on the Cardiovascular System: A Review. J. Xenobiot. 2022, 12, 181–213. [Google Scholar] [CrossRef]
- Konieczna, A.; Rutkowska, A.; Rachon, D. Health risk of exposure to Bisphenol A (BPA). Rocz. Panstw. Zakl. Hig. 2015, 66, 5–11. [Google Scholar] [PubMed]
- Eladak, S.; Grisin, T.; Moison, D.; Guerquin, M.J.; N’Tumba-Byn, T.; Pozzi-Gaudin, S.; Benachi, A.; Livera, G.; Rouiller-Fabre, V.; Habert, R. A new chapter in the bisphenol A story: Bisphenol S and bisphenol F are not safe alternatives to this compound. Fertil. Steril. 2015, 103, 11–21. [Google Scholar] [CrossRef]
- Altuwair, I. Production of Bisphenol A (BPA) By GreenTechnology. Eng. Technol. Open Access 2018, 1, 72–87. [Google Scholar] [CrossRef]
- Vandenberg, L.N.; Hauser, R.; Marcus, M.; Olea, N.; Welshons, W.V. Human exposure to bisphenol A (BPA). Reprod. Toxicol. 2007, 24, 139–177. [Google Scholar] [CrossRef]
- Manzoor, M.F.; Tariq, T.; Fatima, B.; Sahar, A.; Tariq, F.; Munir, S.; Khan, S.; Nawaz Ranjha, M.M.A.; Sameen, A.; Zeng, X.A.; et al. An insight into bisphenol A, food exposure and its adverse effects on health: A review. Front. Nutr. 2022, 9, 1047827. [Google Scholar] [CrossRef] [PubMed]
- Rochester, J.R.; Bolden, A.L. Bisphenol S and F: A Systematic Review and Comparison of the Hormonal Activity of Bisphenol A Substitutes. Environ. Health Perspect. 2015, 123, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, F.; Condorelli, R.A.; Mongioi, L.M.; Cannarella, R.; Aversa, A.; Calogero, A.E.; La Vignera, S. Effects of Bisphenols on Testicular Steroidogenesis. Front. Endocrinol. 2020, 11, 373. [Google Scholar] [CrossRef]
- Abrantes-Soares, F.; Lorigo, M.; Cairrao, E. Effects of BPA substitutes on the prenatal and cardiovascular systems. Crit. Rev. Toxicol. 2022, 52, 469–498. [Google Scholar] [CrossRef]
- Sharma, B.M.; Bharat, G.K.; Chakraborty, P.; Martinik, J.; Audy, O.; Kukucka, P.; Pribylova, P.; Kukreti, P.K.; Sharma, A.; Kalina, J.; et al. A comprehensive assessment of endocrine-disrupting chemicals in an Indian food basket: Levels, dietary intakes, and comparison with European data. Environ. Pollut. 2021, 288, 117750. [Google Scholar] [CrossRef]
- Bolognesi, C.; Castle, L.; Cravedi, J.-P.; Engel, K.-H.; Fowler, P.A.F.; Franz, R.; Grob, K.; Gürtler, R.; Husøy, T.; Mennes, W.; et al. Scientific Opinion on the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs: Executive summary. EFSA J. 2015, 13, 3978. [Google Scholar]
- EFSA Panel on Food Contact Materials, Enzymes and Processing Aids (CEP); Lambre, C.; Barat Baviera, J.M.; Bolognesi, C.; Chesson, A.; Cocconcelli, P.S.; Crebelli, R.; Gott, D.M.; Grob, K.; Lampi, E.; et al. Re-evaluation of the risks to public health related to the presence of bisphenol A (BPA) in foodstuffs. EFSA J. 2023, 21, e06857. [Google Scholar] [CrossRef] [PubMed]
- Cannarella, R.; Gul, M.; Rambhatla, A.; Agarwal, A. Temporal decline of sperm concentration: Role of endocrine disruptors. Endocrine 2023, 79, 1–16. [Google Scholar] [CrossRef]
- Flint, S.; Markle, T.; Thompson, S.; Wallace, E. Bisphenol A exposure, effects, and policy: A wildlife perspective. J. Environ. Manag. 2012, 104, 19–34. [Google Scholar] [CrossRef]
- Dekant, W.; Volkel, W. Human exposure to bisphenol A by biomonitoring: Methods, results and assessment of environmental exposures. Toxicol. Appl. Pharmacol. 2008, 228, 114–134. [Google Scholar] [CrossRef]
- Santiago, J.; Silva, J.V.; Santos, M.A.S.; Fardilha, M. Fighting Bisphenol A-Induced Male Infertility: The Power of Antioxidants. Antioxidants 2021, 10, 289. [Google Scholar] [CrossRef] [PubMed]
- Lorigo, M.; Cairrao, E. Fetoplacental vasculature as a model to study human cardiovascular endocrine disruption. Mol. Asp. Med. 2022, 87, 101054. [Google Scholar] [CrossRef]
- Acconcia, F.; Pallottini, V.; Marino, M. Molecular Mechanisms of Action of BPA. Dose Response 2015, 13, 1559325815610582. [Google Scholar] [CrossRef]
- Rochester, J.R. Bisphenol A and human health: A review of the literature. Reprod. Toxicol. 2013, 42, 132–155. [Google Scholar] [CrossRef]
- Vitku, J.; Sosvorova, L.; Chlupacova, T.; Hampl, R.; Hill, M.; Sobotka, V.; Heracek, J.; Bicikova, M.; Starka, L. Differences in bisphenol A and estrogen levels in the plasma and seminal plasma of men with different degrees of infertility. Physiol. Res. 2015, 64, S303–S311. [Google Scholar] [CrossRef]
- Lassen, T.H.; Frederiksen, H.; Jensen, T.K.; Petersen, J.H.; Joensen, U.N.; Main, K.M.; Skakkebaek, N.E.; Juul, A.; Jorgensen, N.; Andersson, A.M. Urinary bisphenol A levels in young men: Association with reproductive hormones and semen quality. Environ. Health Perspect. 2014, 122, 478–484. [Google Scholar] [CrossRef] [PubMed]
- Manfo, F.P.; Jubendradass, R.; Nantia, E.A.; Moundipa, P.F.; Mathur, P.P. Adverse effects of bisphenol A on male reproductive function. Rev. Environ. Contam. Toxicol. 2014, 228, 57–82. [Google Scholar] [CrossRef] [PubMed]
- Peretz, J.; Vrooman, L.; Ricke, W.A.; Hunt, P.A.; Ehrlich, S.; Hauser, R.; Padmanabhan, V.; Taylor, H.S.; Swan, S.H.; VandeVoort, C.A.; et al. Bisphenol a and reproductive health: Update of experimental and human evidence, 2007–2013. Environ. Health Perspect. 2014, 122, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Yin, L.; Zhang, N.; Han, F.; Liu, W.B.; Zhang, X.; Chen, H.Q.; Cao, J.; Liu, J.Y. Bisphenol A induced male germ cell apoptosis via IFNbeta-XAF1-XIAP pathway in adult mice. Toxicol. Appl. Pharmacol. 2018, 355, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Wang, Z.; Liu, F. Chronic exposure of BPA impairs male germ cell proliferation and induces lower sperm quality in male mice. Chemosphere 2021, 262, 127880. [Google Scholar] [CrossRef]
- Mostari, M.H.; Rahaman, M.M.; Akhter, M.A.; Ali, M.H.; Sasanami, T.; Tokumoto, T. Transgenerational effects of bisphenol A on zebrafish reproductive tissues and sperm motility. Reprod. Toxicol. 2022, 109, 31–38. [Google Scholar] [CrossRef]
- Srivastava, S.; Gupta, P. Alteration in apoptotic rate of testicular cells and sperms following administration of Bisphenol A (BPA) in Wistar albino rats. Environ. Sci. Pollut. Res. Int. 2018, 25, 21635–21643. [Google Scholar] [CrossRef]
- Radwan, M.; Wielgomas, B.; Dziewirska, E.; Radwan, P.; Kaluzny, P.; Klimowska, A.; Hanke, W.; Jurewicz, J. Urinary Bisphenol A Levels and Male Fertility. Am. J. Mens. Health 2018, 12, 2144–2151. [Google Scholar] [CrossRef]
- Amir, S.; Shah, S.T.A.; Mamoulakis, C.; Docea, A.O.; Kalantzi, O.I.; Zachariou, A.; Calina, D.; Carvalho, F.; Sofikitis, N.; Makrigiannakis, A.; et al. Endocrine Disruptors Acting on Estrogen and Androgen Pathways Cause Reproductive Disorders through Multiple Mechanisms: A Review. Int. J. Environ. Res. Public Health 2021, 18, 1464. [Google Scholar] [CrossRef]
- Kotwicka, M.; Skibinska, I.; Piworun, N.; Jendraszak, M.; Chmielewska, M.; Jedrzejczak, P. Bisphenol A modifies human spermatozoa motility in vitro. J. Med. Sci. 2016, 85, 39–45. [Google Scholar] [CrossRef]
- Lagos-Cabre, R.; Moreno, R.D. Contribution of environmental pollutants to male infertily: A working model of germ cell apoptosis induced by plasticizers. Biol. Res. 2012, 45, 5–14. [Google Scholar] [CrossRef] [PubMed]
- Concepcion-Zavaleta, M.; Paz Ibarra, J.L.; Ramos-Yataco, A.; Coronado-Arroyo, J.; Concepcion-Urteaga, L.; Roseboom, P.J.; Williams, C.A. Assessment of hormonal status in male infertility. An update. Diabetes Metab. Syndr. 2022, 16, 102447. [Google Scholar] [CrossRef] [PubMed]
- Caporossi, L.; Vigano, P.; Paci, E.; Capanna, S.; Alteri, A.; Pigini, D.; Tranfo, G.; Papaleo, B. A Case-Control Study on the Effects of Plasticizers Exposure on Male Fertility. Int. J. Environ. Res. Public Health 2022, 20, 235. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Mruk, D.D.; Tang, E.I.; Wong, C.K.; Lee, W.M.; John, C.M.; Turek, P.J.; Silvestrini, B.; Cheng, C.Y. Environmental toxicants perturb human Sertoli cell adhesive function via changes in F-actin organization mediated by actin regulatory proteins. Hum. Reprod. 2014, 29, 1279–1291. [Google Scholar] [CrossRef]
- Vitku, J.; Heracek, J.; Sosvorova, L.; Hampl, R.; Chlupacova, T.; Hill, M.; Sobotka, V.; Bicikova, M.; Starka, L. Associations of bisphenol A and polychlorinated biphenyls with spermatogenesis and steroidogenesis in two biological fluids from men attending an infertility clinic. Environ. Int. 2016, 89–90, 166–173. [Google Scholar] [CrossRef]
- Zhuang, W.; Wu, K.; Wang, Y.; Zhu, H.; Deng, Z.; Peng, L.; Zhu, G. Association of serum bisphenol-A concentration and male reproductive function among exposed workers. Arch. Environ. Contam. Toxicol. 2015, 68, 38–45. [Google Scholar] [CrossRef]
- Shokry, D.A.; Mohamed, M.I.; Abdel-satar, M.F.; Selim, N.E.S.; El Salam, M.A.A. Correlation between Urinary Bisphenol A (BPA) Levels and Male Reproductive Functions among Sample of Egyptian Population. Indian J. Med. Forensic Med. Toxicol. 2020, 14, 1857–1862. [Google Scholar] [CrossRef]
- Adoamnei, E.; Mendiola, J.; Vela-Soria, F.; Fernandez, M.F.; Olea, N.; Jorgensen, N.; Swan, S.H.; Torres-Cantero, A.M. Urinary bisphenol A concentrations are associated with reproductive parameters in young men. Environ. Res. 2018, 161, 122–128. [Google Scholar] [CrossRef]
- Barbonetti, A.; Castellini, C.; Di Giammarco, N.; Santilli, G.; Francavilla, S.; Francavilla, F. In vitro exposure of human spermatozoa to bisphenol A induces pro-oxidative/apoptotic mitochondrial dysfunction. Reprod. Toxicol. 2016, 66, 61–67. [Google Scholar] [CrossRef]
- Zhou, Q.; Miao, M.; Ran, M.; Ding, L.; Bai, L.; Wu, T.; Yuan, W.; Gao, E.; Wang, J.; Li, G.; et al. Serum bisphenol-A concentration and sex hormone levels in men. Fertil. Steril. 2013, 100, 478–482. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Miao, M.; Zhou, Z.; Gao, E.; Chen, J.; Wang, J.; Sun, F.; Yuan, W.; Li, D.K. Exposure to bisphenol-A and reproductive hormones among male adults. Environ. Toxicol. Pharmacol. 2015, 39, 934–941. [Google Scholar] [CrossRef] [PubMed]
- Hanaoka, T.; Kawamura, N.; Hara, K.; Tsugane, S. Urinary bisphenol A and plasma hormone concentrations in male workers exposed to bisphenol A diglycidyl ether and mixed organic solvents. Occup. Environ. Med. 2002, 59, 625–628. [Google Scholar] [CrossRef]
- Galloway, T.; Cipelli, R.; Guralnik, J.; Ferrucci, L.; Bandinelli, S.; Corsi, A.M.; Money, C.; McCormack, P.; Melzer, D. Daily bisphenol A excretion and associations with sex hormone concentrations: Results from the InCHIANTI adult population study. Environ. Health Perspect. 2010, 118, 1603–1608. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Xu, W.; Chen, J.; Shi, H.; Zhu, J.; Liu, X.; Wang, J.; Miao, M.; Yuan, W. The Association between Exposure to Environmental Bisphenol A and Gonadotropic Hormone Levels among Men. PLoS ONE 2017, 12, e0169217. [Google Scholar] [CrossRef] [PubMed]
- Joensen, U.N.; Jorgensen, N.; Thyssen, J.P.; Szecsi, P.B.; Stender, S.; Petersen, J.H.; Andersson, A.M.; Frederiksen, H. Urinary excretion of phenols, parabens and benzophenones in young men: Associations to reproductive hormones and semen quality are modified by mutations in the Filaggrin gene. Environ. Int. 2018, 121, 365–374. [Google Scholar] [CrossRef]
- Den Hond, E.; Tournaye, H.; De Sutter, P.; Ombelet, W.; Baeyens, W.; Covaci, A.; Cox, B.; Nawrot, T.S.; Van Larebeke, N.; D’Hooghe, T. Human exposure to endocrine disrupting chemicals and fertility: A case-control study in male subfertility patients. Environ. Int. 2015, 84, 154–160. [Google Scholar] [CrossRef]
- Mendiola, J.; Jorgensen, N.; Andersson, A.M.; Calafat, A.M.; Ye, X.; Redmon, J.B.; Drobnis, E.Z.; Wang, C.; Sparks, A.; Thurston, S.W.; et al. Are environmental levels of bisphenol a associated with reproductive function in fertile men? Environ. Health Perspect. 2010, 118, 1286–1291. [Google Scholar] [CrossRef]
- Kiwitt-Cardenas, J.; Adoamnei, E.; Arense-Gonzalo, J.J.; Sarabia-Cos, L.; Vela-Soria, F.; Fernandez, M.F.; Gosalvez, J.; Mendiola, J.; Torres-Cantero, A.M. Associations between urinary concentrations of bisphenol A and sperm DNA fragmentation in young men. Environ. Res. 2021, 199, 111289. [Google Scholar] [CrossRef]
- Knez, J.; Kranvogl, R.; Breznik, B.P.; Voncina, E.; Vlaisavljevic, V. Are urinary bisphenol A levels in men related to semen quality and embryo development after medically assisted reproduction? Fertil. Steril. 2014, 101, 215–221.e215. [Google Scholar] [CrossRef]
- Meeker, J.D.; Ehrlich, S.; Toth, T.L.; Wright, D.L.; Calafat, A.M.; Trisini, A.T.; Ye, X.; Hauser, R. Semen quality and sperm DNA damage in relation to urinary bisphenol A among men from an infertility clinic. Reprod. Toxicol. 2010, 30, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Li, D.K.; Zhou, Z.; Miao, M.; He, Y.; Wang, J.; Ferber, J.; Herrinton, L.J.; Gao, E.; Yuan, W. Urine bisphenol-A (BPA) level in relation to semen quality. Fertil. Steril. 2011, 95, 625–630.e4. [Google Scholar] [CrossRef]
- La Rocca, C.; Tait, S.; Guerranti, C.; Busani, L.; Ciardo, F.; Bergamasco, B.; Perra, G.; Mancini, F.R.; Marci, R.; Bordi, G.; et al. Exposure to Endocrine Disruptors and Nuclear Receptors Gene Expression in Infertile and Fertile Men from Italian Areas with Different Environmental Features. Int. J. Environ. Res. Public Health 2015, 12, 12426–12445. [Google Scholar] [CrossRef]
- Ji, H.; Miao, M.; Liang, H.; Shi, H.; Ruan, D.; Li, Y.; Wang, J.; Yuan, W. Exposure of environmental Bisphenol A in relation to routine sperm parameters and sperm movement characteristics among fertile men. Sci. Rep. 2018, 8, 17548. [Google Scholar] [CrossRef]
- Freour, T.; Jean, M.; Mirallie, S.; Barriere, P. Computer-assisted sperm analysis parameters in young fertile sperm donors and relationship with age. Syst. Biol. Reprod. Med. 2012, 58, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Freour, T.; Jean, M.; Mirallie, S.; Dubourdieu, S.; Barriere, P. Computer-Assisted Sperm Analysis (CASA) parameters and their evolution during preparation as predictors of pregnancy in intrauterine insemination with frozen-thawed donor semen cycles. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 149, 186–189. [Google Scholar] [CrossRef]
- Wiser, A.; Sachar, S.; Ghetler, Y.; Shulman, A.; Breitbart, H. Assessment of sperm hyperactivated motility and acrosome reaction can discriminate the use of spermatozoa for conventional in vitro fertilisation or intracytoplasmic sperm injection: Preliminary results. Andrologia 2014, 46, 313–315. [Google Scholar] [CrossRef] [PubMed]
- Omran, G.A.; Gaber, H.D.; Mostafa, N.A.M.; Abdel-Gaber, R.M.; Salah, E.A. Potential hazards of bisphenol A exposure to semen quality and sperm DNA integrity among infertile men. Reprod. Toxicol. 2018, 81, 188–195. [Google Scholar] [CrossRef]
- Chen, P.P.; Liu, C.; Zhang, M.; Miao, Y.; Cui, F.P.; Deng, Y.L.; Luo, Q.; Zeng, J.Y.; Shi, T.; Lu, T.T.; et al. Associations between urinary bisphenol A and its analogues and semen quality: A cross-sectional study among Chinese men from an infertility clinic. Environ. Int. 2022, 161, 107132. [Google Scholar] [CrossRef]
- Mantzouki, C.; Bliatka, D.; Iliadou, P.K.; Margeli, A.; Papassotiriou, I.; Mastorakos, G.; Kousta, E.; Goulis, D.G. Serum Bisphenol A concentrations in men with idiopathic infertility. Food Chem. Toxicol. 2019, 125, 562–565. [Google Scholar] [CrossRef]
- Pollard, S.H.; Cox, K.J.; Blackburn, B.E.; Wilkins, D.G.; Carrell, D.T.; Stanford, J.B.; Porucznik, C.A. Male exposure to bisphenol A (BPA) and semen quality in the Home Observation of Periconceptional Exposures (HOPE) cohort. Reprod. Toxicol. 2019, 90, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Miao, M.; Zhou, X.; Li, Y.; Zhang, O.; Zhou, Z.; Li, T.; Yuan, W.; Li, R.; Li, D.K. LINE-1 hypomethylation in spermatozoa is associated with Bisphenol A exposure. Andrology 2014, 2, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Tian, Y.; Zhou, X.; Miao, M.; Li, D.K.; Wang, Z.; Li, R.; Liang, H.; Yuan, W. Association of Bisphenol A Exposure with LINE-1 Hydroxymethylation in Human Semen. Int. J. Environ. Res. Public Health 2018, 15, 1770. [Google Scholar] [CrossRef]
- Song, X.; Miao, M.; Zhou, X.; Li, D.; Tian, Y.; Liang, H.; Li, R.; Yuan, W. Bisphenol A Exposure and Sperm ACHE Hydroxymethylation in Men. Int. J. Environ. Res. Public Health 2019, 16, 152. [Google Scholar] [CrossRef]
- Palak, E.; Lebiedzinska, W.; Anisimowicz, S.; Sztachelska, M.; Pierzynski, P.; Wiczkowski, W.; Zelazowska-Rutkowska, B.; Niklinska, G.N.; Ponikwicka-Tyszko, D.; Wolczynski, S. The Association between Bisphenol A, Steroid Hormones, and Selected MicroRNAs Levels in Seminal Plasma of Men with Infertility. J. Clin. Med. 2021, 10, 5945. [Google Scholar] [CrossRef]
- Chen, M.; Tang, R.; Fu, G.; Xu, B.; Zhu, P.; Qiao, S.; Chen, X.; Xu, B.; Qin, Y.; Lu, C.; et al. Association of exposure to phenols and idiopathic male infertility. J. Hazard. Mater. 2013, 250–251, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, A.E.; Chen, Z.; Perry, M.J.; Kannan, K.; Louis, G.M. Urinary bisphenol A and semen quality, the LIFE Study. Reprod. Toxicol. 2015, 51, 7–13. [Google Scholar] [CrossRef]
- Hu, W.; Dong, T.; Wang, L.; Guan, Q.; Song, L.; Chen, D.; Zhou, Z.; Chen, M.; Xia, Y.; Wang, X. Obesity aggravates toxic effect of BPA on spermatogenesis. Environ. Int. 2017, 105, 56–65. [Google Scholar] [CrossRef]
- Kim, H.K.; Ko, D.H.; Lee, W.; Kim, K.R.; Chun, S.; Song, J.; Min, W.K. Body fluid concentrations of bisphenol A and their association with in vitro fertilization outcomes. Hum. Fertil. 2021, 24, 199–207. [Google Scholar] [CrossRef]
- Benson, T.E.; Gaml-Sorensen, A.; Ernst, A.; Brix, N.; Hougaard, K.S.; Haervig, K.K.; Ellekilde Bonde, J.P.; Tottenborg, S.S.; Lindh, C.H.; Ramlau-Hansen, C.H.; et al. Urinary Bisphenol A, F and S Levels and Semen Quality in Young Adult Danish Men. Int. J. Environ. Res. Public Health 2021, 18, 1742. [Google Scholar] [CrossRef]
- Caporossi, L.; Alteri, A.; Campo, G.; Paci, E.; Tranfo, G.; Capanna, S.; Papaleo, E.; Pigini, D.; Vigano, P.; Papaleo, B. Cross Sectional Study on Exposure to BPA and Phthalates and Semen Parameters in Men Attending a Fertility Center. Int. J. Environ. Res. Public Health 2020, 17, 489. [Google Scholar] [CrossRef] [PubMed]
- Hart, R.J.; Doherty, D.A.; Keelan, J.A.; Minaee, N.S.; Thorstensen, E.B.; Dickinson, J.E.; Pennell, C.E.; Newnham, J.P.; McLachlan, R.; Norman, R.J.; et al. The impact of antenatal Bisphenol A exposure on male reproductive function at 20–22 years of age. Reprod. BioMed. Online 2018, 36, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Holmboe, S.A.; Scheutz Henriksen, L.; Frederiksen, H.; Andersson, A.M.; Priskorn, L.; Jorgensen, N.; Juul, A.; Toppari, J.; Skakkebaek, N.E.; Main, K.M. Prenatal exposure to phenols and benzophenones in relation to markers of male reproductive function in adulthood. Front. Endocrinol. 2022, 13, 1071761. [Google Scholar] [CrossRef]
- Song, J.W.; Chung, K.C. Observational studies: Cohort and case-control studies. Plast. Reconstr. Surg. 2010, 126, 2234–2242. [Google Scholar] [CrossRef] [PubMed]

| Study | Country | Population Sample Characteristics (Number, Mean Age, Mean BMI, Progeny) | Biological Samples | Evaluated Parameters | General Conclusions | Author, Year Reference |
|---|---|---|---|---|---|---|
| Cross-sectional | China | 137 men with and 153 men without occupational exposure (<30–>50 years old) | Blood | Reproductive hormones | Decrease AD, FT, and FAI Increase SHGB | Zhou et al.—2013 [51] |
| Case-control | China | 281 men with occupational exposure (34.12 years, 27.54 kg/m2) 278 men without occupational exposure (32.78 years, 26.41 kg/m2) | Blood: - Exposed workers—median [BPA] 18.75 ng/mL; - Unexposed workers—median [BPA] 3.37 ng/mL. | Reproductive hormones | Exposure >5 years—increased SHBG and decreased AD No changes between groups in SHBG, TT, or inhibin B or AD | Zhuang et al.—2015 [47] |
| Cross-sectional | China | 165 men with and 427 men without occupational exposure (31.7 years) | Blood Urine: - Exposed workers—median [BPA] 685.9 µg/g Cr; - Unexposed workers—median [BPA] 4.2 µg/g Cr. | Reproductive hormones | Increased prolactin, E2, and SHGB Decrease FSH, AD and FAI | Liu et al.—2015 [52] |
| Cross-sectional | Japan | 42 men (37 years) with and 42 men (38 years) without occupational exposure | Blood Urine: - Exposed workers—median [BPA] 1.06 mmol/mol Cr; - Unexposed workers—median [BPA] 0.52 mmol/mol Cr. | Reproductive hormones | Decreased FSH No association with LH and FT | Hanaoka et al.—2002 [53] |
| Cross-sectional | Denmark | 308 young men from the general population (≈ 18 years) | Semen Blood Urine (median [BPA] 3.25 ng/mL) | Characteristics of semen Reproductive hormones | Increase T, LH, E2, and FT Decreased sperm motility No evidence for other semen parameters | Lassen et al.—2014 [32] |
| Cross-sectional | Italy | 334 men from the general population (20-74 years old, BMI: < 18.5–> 35 kg/m2) | Blood Urine (geometric mean [BPA] 4.02 ng/mL) | Reproductive hormones | Increased TT No association with E2 | Galloway et al.—2010 [54] |
| Cross-sectional | China | 560 men (32.2 years, BMI: < 18.5–> 25 kg/m2) | Blood Urine (geometric mean [BPA] 0.44 µg/L) | Reproductive hormones | Increased LH and FSH in smokers Total T decreased in BMI >25 kg/m2 | Liang et al.—2017 [55] |
| Case-control | Denmark | 65 young men with FLG mutation 130 controls (19 years, 23 kg/m2) | Semen Blood Urine (median [BPA] 3.5 ng/mL) | Characteristics of semen Reproductive hormones | FLG mutation carriers—increased T and E2 and decreased FSH Decreased sperm motility | Joensen et al.—2018 [56] |
| Case-control | Belgium | 120 men from 4 fertility clinics 40 cases (31.6 years, 26.1 kg/m2) 80 controls (34.1 years, 24.6 kg/m2) | Semen Blood Urine: - Cases—geometric mean [BPA] 1.7 µg/L; - Controls—geometric mean [BPA] 1.5 µg/L, | Reproductive hormones | Decreased T and increased risk of subfertility | Den Hond et al.—2015 [57] |
| Cross-sectional | USA | 375 fertile men from a prenatal clinic (31.9 years, 28.3 kg/m2) | Semen Blood Urine (geometric mean [BPA] 1.5 µg/mL) | Characteristics of semen Reproductive hormones | Modest FT decrease No association with semen features or reproductive hormones | Mendiola et al.—2010 [58] |
| Cross-sectional | Spain | 215 healthy college students (median age of 20.4 years, median BMI of 23.7 kg/m2) | Semen Blood Urine (geometric mean [BPA] 2.3 ng/mL) | Characteristics of semen Reproductive hormones | Increased LH Decreased sperm concentration No evidence for other parameters of semen or other reproductive hormones | Adoamnei et al.—2018 [49] |
| Cross-sectional | Spain | 158 university students (median age of 20.4 years, median BMI of 23.6 kg/m2) | Semen Urine (mean [BPA] 2.05 μg/g) | Sperm DNA fragmentation | Increased SDF index in the SDF subgroup>30% | Kiwitt-Cardenas et al.—2021 [59] |
| Prospective Cohort | Slovenia | 149 men from couples undergoing IVF or ICSI treatment (34.05 years, 27.45 kg/m2) | Semen Urine (geometric mean [BPA] 1.55 ng/mL) | Characteristics of semen | Decreased sperm count, sperm concentration, and vitality | Knez et al.—2014 [60] |
| Cross-sectional | USA | 190 men from a fertility clinic (36.4 years, 27.3 kg/m2) | Semen Urine (geometric mean [BPA] 1.4 ng/mL) | Characteristics of semen | Decreased sperm concentration and motility Morphology changes Increased DNA sperm damage | Meeker et al.—2010 [61] |
| Cohort Case-control | China | 218 men with and without occupational exposure (<25–>45 years) | Semen Urine | Characteristics of semen | Decreased concentration, total count, vitality, and sperm motility No association with morphology changes | Li et al.—2011 [62] |
| Case-control | Italy | 70 infertile men and 83 fertile men from: Metropolitan (37.2 years, 25.0 kg/m2) Urban (34.02 years, 25.2 kg/m2) Rural (35.6 years, 26.3 kg/m2) | Semen Blood: - Infertile—mean [BPA] 9.3 ng/mL; - Fertile—mean [BPA] 5.7 ng/mL. | Gene expression nuclear receptors | Increased gene expression ERα, ERβ, AR, PXR, and AhR | La Rocca et al.—2015 [63] |
| Cross-sectional | China | 500 fertile men (18–55 years, BMI: < 18.5–> 25 kg/m2, ≥1 child) | Semen Urine (geometric mean [BPA] 0.38 µg/L) | Characteristics of semen | Decreased sperm concentration and motility Increased sperm velocity rates | Ji et al.—2018 [64] |
| Case-control | Egypt | 50 infertile men 50 fertile men (20–54 years old) | Semen Urine: - Infertile—median [BPA] 24.2 μg/L; - Fertile—median [BPA] 20.9 μg/L. | Characteristics of semen | Decreased semen quality | Omran et al.—2018 [68] |
| Case-control | Egypt | 100 infertile men (33.53 years, BMI < 30 kg/m2) 50 fertile men (35.20 years, BMI < 30 kg/m2) | Semen Urine: - Infertile—mean [BPA] 10.07 ng/mL; - Fertile—mean [BPA] 1.69 ng/mL. | Characteristics of semen Reproductive hormones | Increased BPA levels in infertility cases compared to controls Association between sperm motility and FSH, LH, TT, and E2 levels | Shokry et al.—2020 [48] |
| Cross-sectional | China | 984 men from a reproductive health clinic (32.0 years, 23.3 kg/m2) | Semen Urine (median [BPA] 2.24 µg/L) | Characteristics of semen | Decreased concentration and total sperm count and progressive and total motility | Chen et al.—2022 [69] |
| Cross-sectional Case-control | Greece | 55 infertile men: - 23 non-obstructive azoospermia (34.6 years); - 20 varicocele (37.4 years); - 12 cryptorchidism (33.7 years). 25 fertile men (30.7 years) | Semen Blood: - Infertile—median [BPA] 0.19 ng/mL; - Fertile—median [BPA] 0.18 ng/mL. | BPA concentration | Very high concentrations only in infertile men, but no differences in concentration between the two groups | Mantzouki et al.—2019 [70] |
| Cross-sectional | Czech Republic | 174 men with several degrees of infertility (35.97 years, 27.32 kg/m2) | Semen and blood [BPA]: - Fertile—66 and 47 pg/mL; - Slightly infertile—144 and 137 pg/mL; - Moderately infertile—132 and 114 pg/mL; - Severely infertile—179 and 33 pg/mL. | Characteristics of semen | Decreased sperm count and concentration | Vitku et al.—2015 [31] |
| Cross-sectional | Czech Republic | 191 men with various degrees of infertility: - Fertile (35.9 years, 27.7 kg/m2); - Slightly infertile (35.7 years, 26.9 kg/m2); - Moderately infertile (35.8 years, 26.1 kg/m2); - Severely infertile (35.2 years, 26.4 kg/m2). | Semen and blood [BPA]: - Fertile—0.075 and 0.029 ng/mL; - Slightly infertile—0.130 and 0.059 ng/mL; - Moderately infertile—0.153 and 0.072 ng/mL; - Severely infertile—0.158 and 0.019 ng/mL. | Characteristics of semen | Decreased concentration, total count, vitality, and sperm morphology Gonadal and adrenal steroidogenesis changes | Vitku et al.—2016 [46] |
| Cross-sectional | Poland | 315 men from a reproductive health clinic (32.14 years, 26.8 kg/m2) | Semen Urine (geometric mean [BPA] 1.84 µg/L) | Characteristics of semen | Decreased sperm motility Increased immature sperm Sperm chromosomal disomy | Radwan et al.—2018 [39] |
| Prospective Cohort | USA | 161 men (28.5 years, 27.1 kg/m2) | Semen Urine (geometric mean [BPA] 2.50 ng/mL) | Characteristics of semen | Changes in morphology (sperm tail) No evidence for other semen parameters | Pollard et al.—2019 [71] |
| Case-control | China | 77 men with and 72 men without occupational exposure (22–50 years old) | Semen Urine: - Exposed workers—mean [BPA] 36.23 µg/g Cr; - Unexposed workers—mean [BPA] 1.38 µg/g Cr. | Characteristics of semen Semen methylation | Decreased sperm concentration and methylation | Miao et al.—2014 [72] |
| Case-control | China | 72 men (34.1 years) with and 86 men (34.4 years) without occupational exposure | Semen Urine: - Exposed workers—geometric mean [BPA] 158.41 µg/g Cr; - Unexposed workers—geometric mean [BPA] 0.84 µg/g Cr. | Semen hydroxymethylation | Changes in semen hydroxymethylation | Tian et al.—2018 [73] |
| Case-control | China | 74 men with and 83 men without occupational exposure (<29–>36 years) | Semen Urine: - Exposed workers—geometric mean [BPA] 199.13 µg/g Cr; - Unexposed workers—geometric mean [BPA] 0.77 µg/g Cr. | Sperm hydroxymethylation | Increased BPA levels related to increased sperm acetylcholinesterase hydroxymethylation | Song et al.—2019 [74] |
| Case-control | Poland | 50 men, normospermic (31.30 years, 23.92 kg/m2) 46 men, oligoasthenoteratozoospermic (31.36 years, 23.69 kg/m2) 20 men, non-obstructive azoospermic (31.20 years, 23.3 kg/m2) | Semen | Characteristics of semen Reproductive hormones MiRNA levels | Decreased sperm concentration and morphology Decreased E2 and AD Increased miR-let-7a and miR-let-7c and decreased miR-518f | Palak et al.—2021 [75] |
| Case-control | China | 877 idiopathic infertile men (28.5 years, 23.47 kg/m2) 713 fertile men (29.83 years, 23.87 kg/m2) | Semen Urine: - Infertile—geometric mean [BPA] 0.612 ng/mL; - Fertile—geometric mean [BPA] 0.621 ng/mL. | Characteristics of semen | No evidence of an association between BPA exposure and male infertility | Chen et al.—2013 [76] |
| Prospective Cohort | USA | 418 men from couples trying to get pregnant (31.7 years, 29.5 kg/m2) | Semen Urine (geometric mean [BPA] 0.55—0.67 ng/mL) | Characteristics of semen | No evidence of an association between BPA exposure and semen quality | Goldstone et al.—2015 [77] |
| Cross-sectional | China | 357 subfertile men (28.7 years, 23.39 kg/m2) | Semen Urine (geometric mean [BPA] 0.52 ng/mL) | Characteristics of semen | No evidence of an association between BPA exposure and male infertility | Hu et al.—2017 [78] |
| Prospective Cohort | Republic of Korea | 146 men from couples undergoing IVF (36.3 years, 25.9 kg/m2) | Blood Urine (geometric mean [BPA] 1.1 ng/mL) | Characteristics of semen | No evidence of an association between BPA and sperm motility and concentration | Kim et al.—2021 [79] |
| Cross-sectional | Denmark | 556 young men (≈19 years old, BMI: <18.5–>25 kg/m2) | Semen Urine (median [BPA] 1.30 ng/mL) | Characteristics of semen | No evidence of an association between BPA exposure and semen quality | Benson et al.—2021 [80] |
| Cross-sectional | Italy | 105 men from a fertility clinic (40.5 years, BMI: 18.5–>25 kg/m2) | Semen Urine (mean [BPA] 0.24 µg/g Cr) | Characteristics of semen | No evidence of an association between BPA exposure and semen quality | Caporossi et al.—2020 [81] |
| Case-control | Italy | 155 infertile men (40.4 years) and 211 fertile men (36.1 years) (BMI: <18.5–>25 kg/m2) | Semen Urine: - Infertile—mean [BPA] 0.47 µg/g Cr; - Fertile—mean [BPA] 0.69 µg/g Cr. | Characteristics of semen | No evidence of an association between BPA exposure and semen quality | Caporossi et al.—2022 [44] |
| Prospective Cohort | Australia | 705 young men and available prenatal maternal serum samples (20–22 years old, ≈24 kg/m2) | Semen Blood | Characteristics of semen Reproductive hormones | Weak association between testicular function, sperm concentration, and motility No association between testicular volume, testicular and pituitary hormones, and total sperm output | Hart et al.—2018 [82] |
| Prospective Cohort | Denmark | 101 young men and prenatal maternal serum samples (median age of 19.3 years, median BMI of 21.2 kg/m2) | Semen Blood | Characteristics of semen Testicular function | Reduced Leydig cell function No association between anogenital distance and semen quality | Holmboe et al.—2022 [83] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Presunto, M.; Mariana, M.; Lorigo, M.; Cairrao, E. The Effects of Bisphenol A on Human Male Infertility: A Review of Current Epidemiological Studies. Int. J. Mol. Sci. 2023, 24, 12417. https://doi.org/10.3390/ijms241512417
Presunto M, Mariana M, Lorigo M, Cairrao E. The Effects of Bisphenol A on Human Male Infertility: A Review of Current Epidemiological Studies. International Journal of Molecular Sciences. 2023; 24(15):12417. https://doi.org/10.3390/ijms241512417
Chicago/Turabian StylePresunto, Mafalda, Melissa Mariana, Margarida Lorigo, and Elisa Cairrao. 2023. "The Effects of Bisphenol A on Human Male Infertility: A Review of Current Epidemiological Studies" International Journal of Molecular Sciences 24, no. 15: 12417. https://doi.org/10.3390/ijms241512417
APA StylePresunto, M., Mariana, M., Lorigo, M., & Cairrao, E. (2023). The Effects of Bisphenol A on Human Male Infertility: A Review of Current Epidemiological Studies. International Journal of Molecular Sciences, 24(15), 12417. https://doi.org/10.3390/ijms241512417









