Therapeutic Efficiency of Proteins Secreted by Glial Progenitor Cells in a Rat Model of Traumatic Brain Injury
Abstract
1. Introduction
2. Results
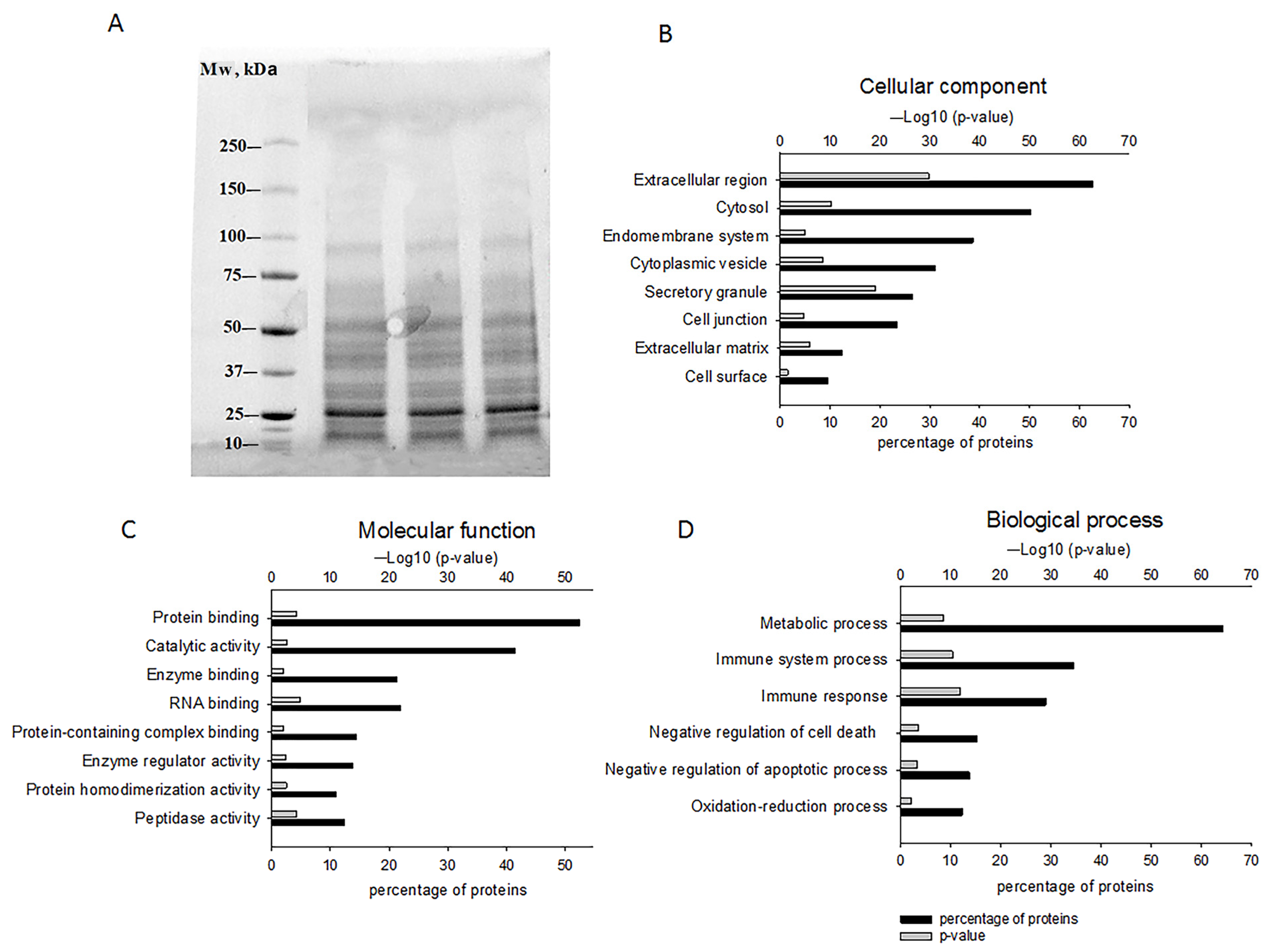
2.1. The Composition of GPC-Secreted Proteins
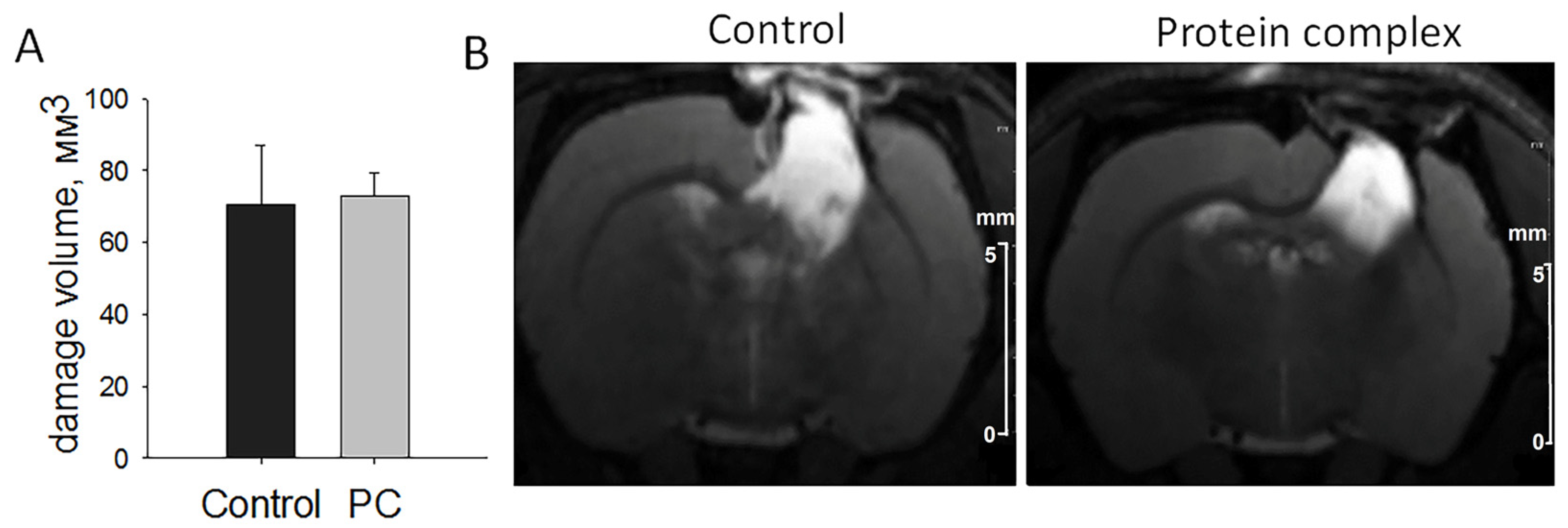
2.2. Assessment of Damage Volume
2.3. Neurological Outcomes
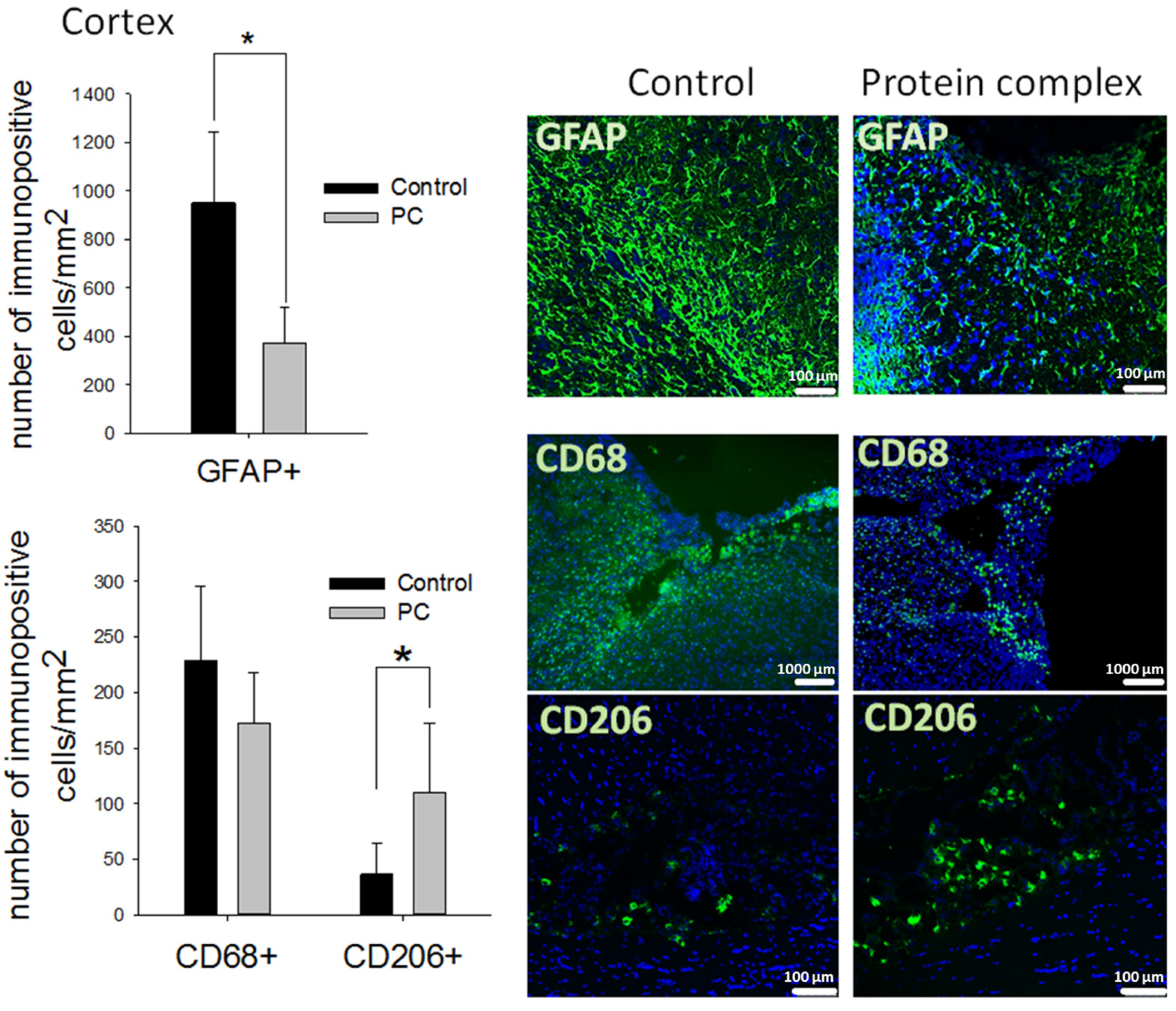
2.4. Histological Analysis of Brain Tissue
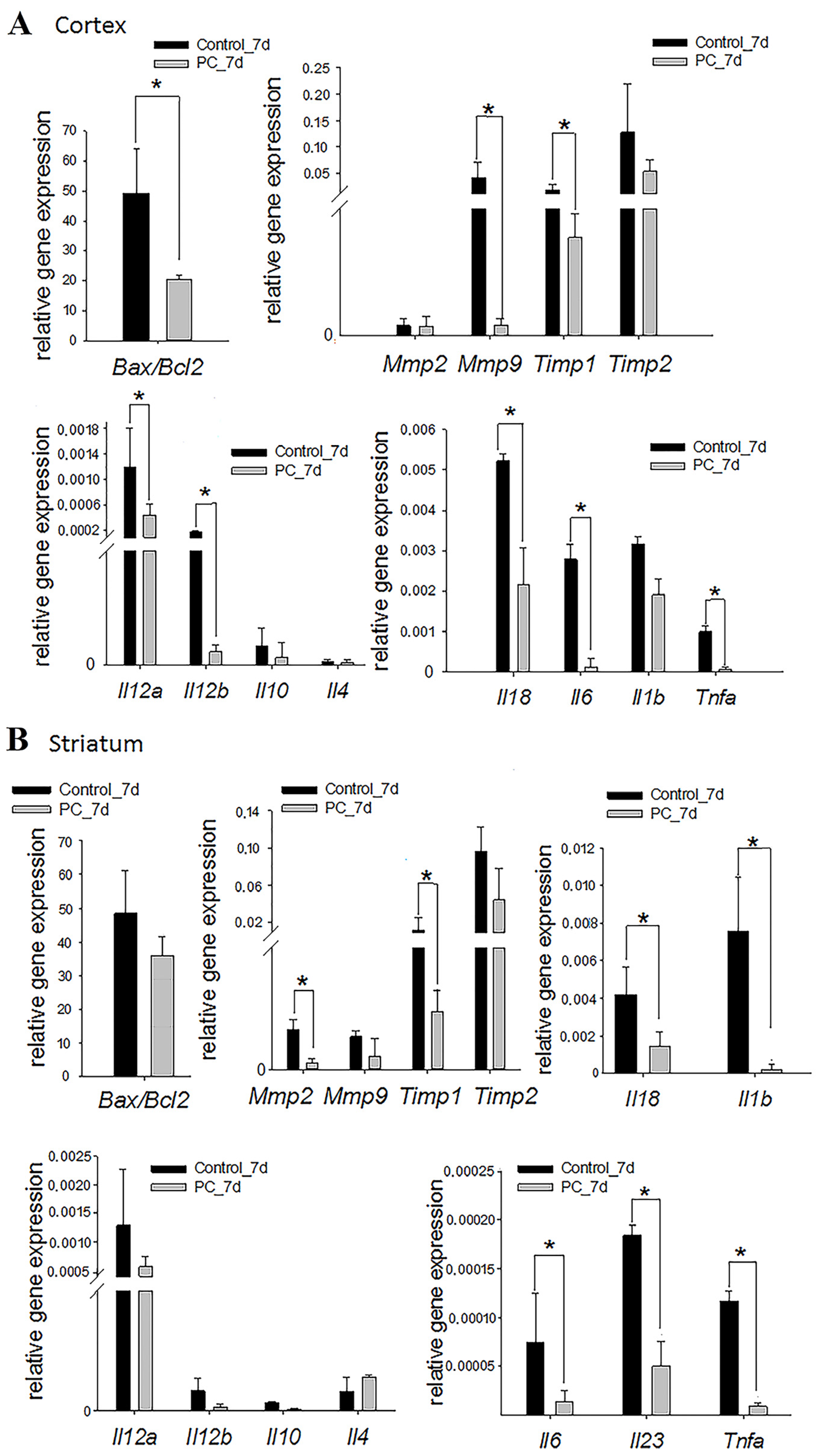
2.5. Gene Expression Assay
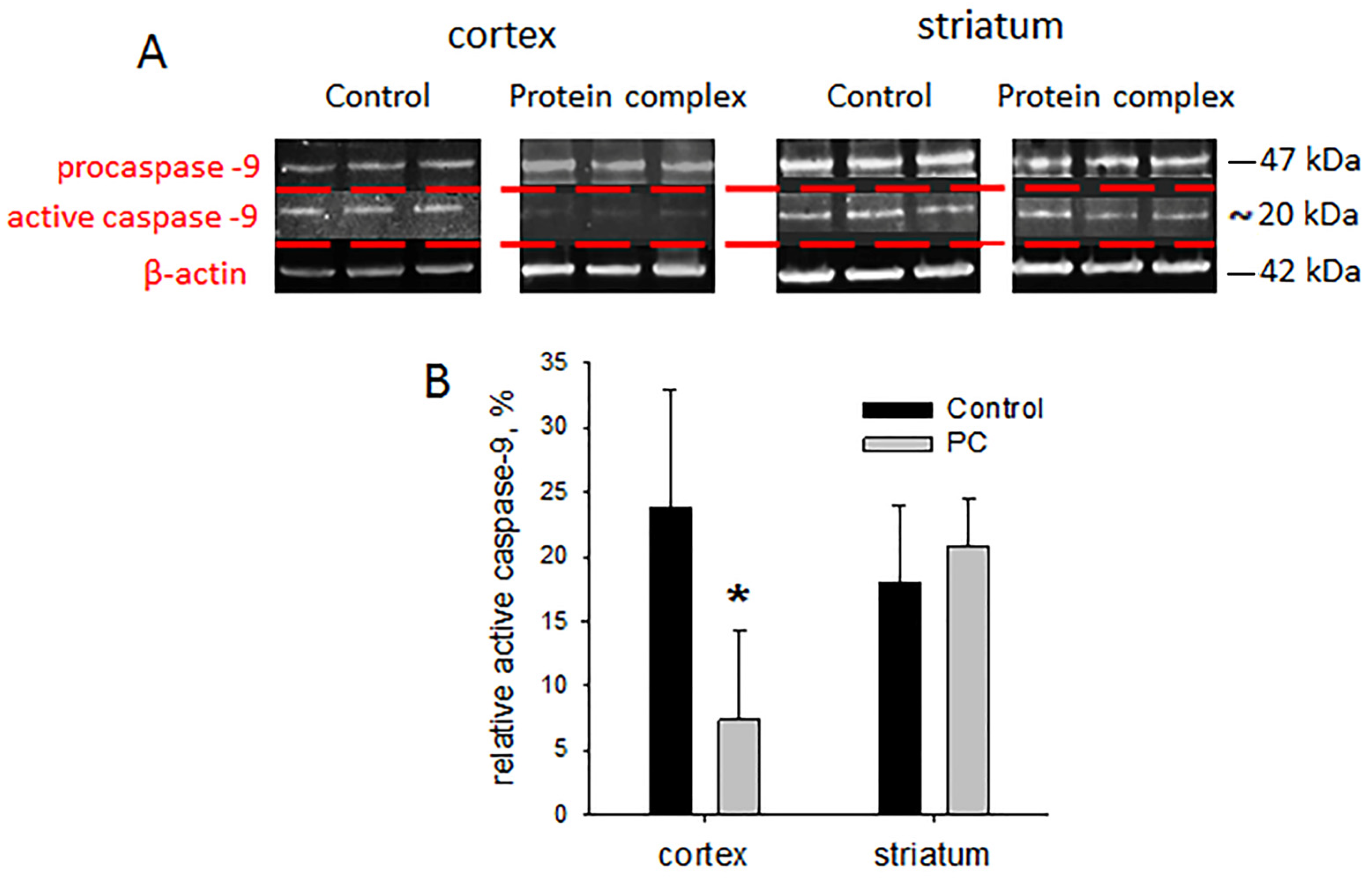
2.6. Tissue Levels of Active Caspase-9
2.7. Assessment of Neurodegeneration Markers
3. Discussion
4. Materials and Methods
4.1. PC Preparation
4.2. Proteomic Analysis
4.3. Animals
4.4. TBI Modeling
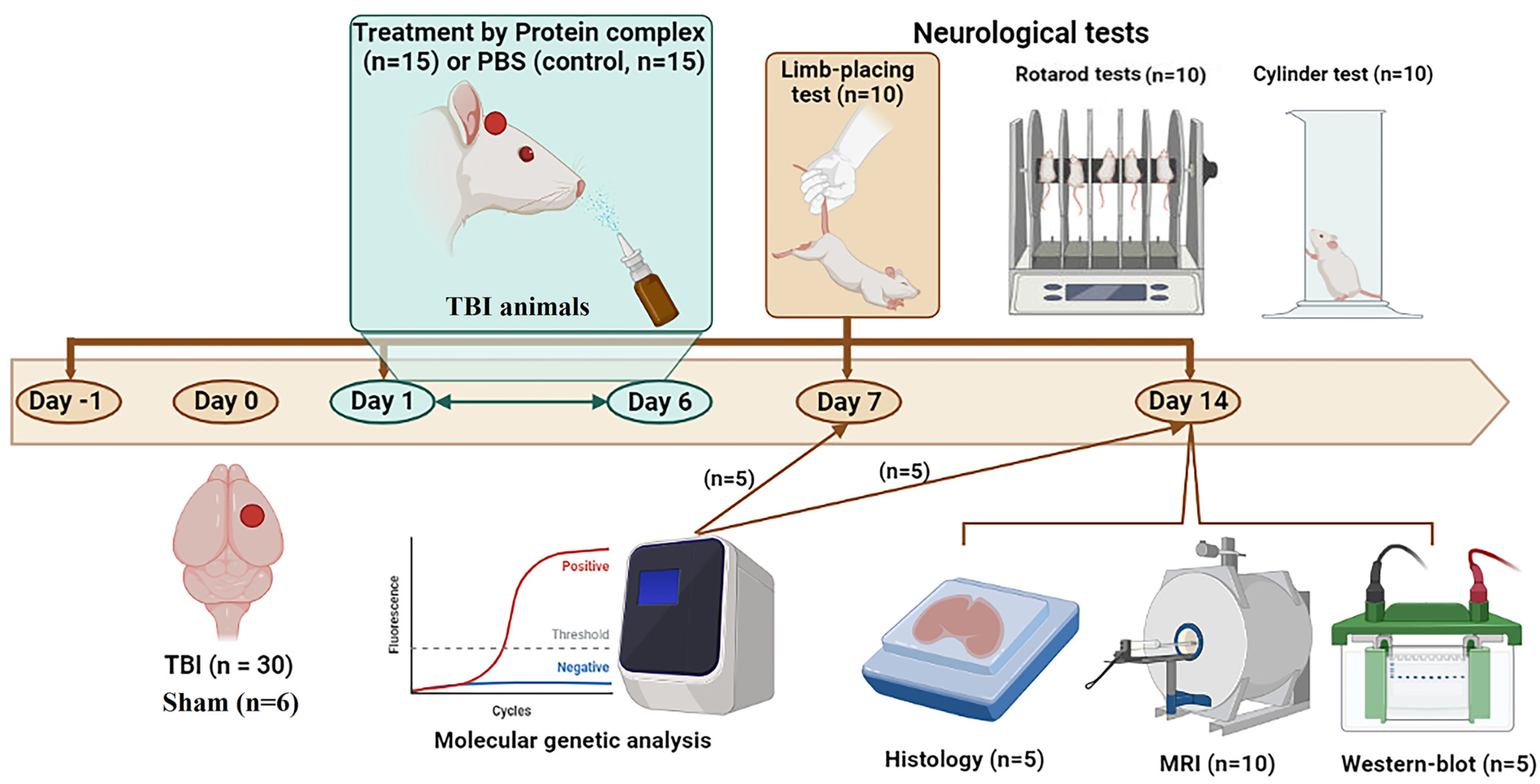
4.5. Animal Study Design
4.6. MRI Morphometry
4.7. Behavioral Tests
4.8. Immunohistochemistry
4.9. Immunoblotting
4.10. Reverse Transcription PCR Assay
4.11. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Burda, J.E.; Sofroniew, M.V. Reactive gliosis and the multicellular response to CNS damage and disease. Neuron 2014, 81, 229–248. [Google Scholar] [CrossRef] [PubMed]
- Humble, S.S.; Wilson, L.D.; Wang, L.; Long, D.A.; Smith, M.A.; Siktberg, J.C.; Patel, M.B. Prognosis of diffuse axonal injury with traumatic brain injury. J. Trauma Acute Care Surg. 2018, 85, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Sulhan, S.; Lyon, K.A.; Shapiro, L.A.; Huang, J.H. Neuroinflammation and blood–brain barrier disruption following traumatic brain injury: Pathophysiology and potential therapeutic targets. J. Neurosci. Res. 2020, 98, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Brett, B.L.; Gardner, R.C.; Godbout, J.; Dams-O’Connor, K.; Keene, C.D. Traumatic brain injury and risk of neurodegenerative disorder. Biol. Psychiatry 2022, 91, 498–507. [Google Scholar] [CrossRef]
- Hefter, D.; Draguhn, A. APP as a protective factor in acute neuronal insults. Front. Mol. Neurosci. 2017, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Liang, P.; Liu, J.; Xiong, J.; Liu, Q.; Zhao, J.; Liang, H.; Zhao, L.; Tang, H. Neural Stem Cell-Conditioned Medium Protects Neurons and Promotes Propriospinal Neurons Relay Neural Circuit Reconnection after Spinal Cord Injury. Cell Transpl. 2014, 23, 45–56. [Google Scholar] [CrossRef]
- Yang, H.; Wang, C.; Chen, H.; Li, L.; Ma, S.; Wang, H.; Qu, T. Neural stem cell-conditioned medium ameliorated cerebral ischemia-reperfusion injury in rats. Stem Cells Int. 2018, 2018, 4659159. [Google Scholar] [CrossRef]
- Huang, Z.; Wong, L.W.; Su, Y.; Huang, X.; Wang, N.; Chen, H.; Yi, C. Blood-brain barrier integrity in the pathogenesis of Alzheimer’s disease. Front. Neuroendocrinol. 2020, 59, 100857. [Google Scholar] [CrossRef]
- Nhan, T.; Burgess, A.; Cho, E.E.; Stefanovic, B.; Lilge, L.; Hynynen, K. Drug delivery to the brain by focused ultrasound induced blood–brain barrier disruption: Quantitative evaluation of enhanced permeability of cerebral vasculature using two-photon microscopy. J. Control. Release 2013, 172, 274–280. [Google Scholar] [CrossRef]
- Pardridge, W.M. Treatment of Alzheimer’s disease and blood–brain barrier drug delivery. Pharmaceuticals 2020, 13, 394. [Google Scholar] [CrossRef]
- Weston, N.M.; Sun, D. The Potential of Stem Cells in Treatment of Traumatic Brain Injury. Curr. Neurol. Neurosci. Rep. 2018, 18, 1. [Google Scholar] [CrossRef] [PubMed]
- Hussain, A.A. Intranasal drug delivery. Advanced Drug Delivery Reviews 1998, 29, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Malerba, F.; Paoletti, F.; Capsoni, S.; Cattaneo, A. Intranasal delivery of therapeutic proteins for neurological diseases. Expert Opin. Drug Deliv. 2011, 8, 1277–1296. [Google Scholar] [CrossRef] [PubMed]
- Lawal, O.; Ulloa Severino, F.P.; Eroglu, C. The role of astrocyte structural plasticity in regulating neural circuit function and behavior. Glia 2022, 70, 1467–1483. [Google Scholar] [CrossRef] [PubMed]
- Ullian, E.M.; Christopherson, K.S.; Barres, B.A. Role for glia in synaptogenesis. Glia 2004, 47, 209–216. [Google Scholar] [CrossRef]
- Aitken, A. 14-3-3 proteins: A historic overview. Semin. Cancer Biol. 2006, 16, 162–172. [Google Scholar] [CrossRef]
- Stricher, F.; Macri, C.; Ruff, M.; Muller, S. HSPA8/HSC70 chaperone protein: Structure, function, and chemical targeting. Autophagy 2013, 9, 1937–1954. [Google Scholar] [CrossRef]
- Robinson, M.B.; Taylor, A.R.; Gifondorwa, D.J.; Tytell, M.; Milligan, C.E. Exogenous Hsc70, but not thermal preconditioning, confers protection to motoneurons subjected to oxidative stress. Dev. Neurobiol. 2008, 68, 1–17. [Google Scholar] [CrossRef]
- Novoselova, T.V.; Margulis, B.A.; Novoselov, S.S.; Sapozhnikov, A.M.; Van Der Spuy, J.; Cheethamà, M.E.; Guzhova, I.V. Treatment with extracellular HSP70/HSC70 protein can reduce polyglutamine toxicity and aggregation. J. Neurochem. 2005, 94, 597–606. [Google Scholar] [CrossRef]
- Miroshnichenko, S.; Usynin, I.; Dudarev, A.; Nimaev, V.; Solovieva, A. Apolipoprotein ai supports mscs survival under stress conditions. Int. J. Mol. Sci. 2020, 21, 4062. [Google Scholar] [CrossRef]
- Ganfornina, M.D.; Do Carmo, S.; Lora, J.M.; Torres-Schumann, S.; Vogel, M.; Allhorn, M.; González, C.; Bastiani, M.J.; Rassart, E.; Sanchez, D. Apolipoprotein D is involved in the mechanisms regulating protection from oxidative stress. Aging Cell 2008, 7, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Boyles, J.K.; Notterpek, L.M.; Anderson, L.J. Accumulation of apolipoproteins in the regenerating and remyelinating mammalian peripheral nerve. Identification of apolipoprotein D, apolipoprotein A-IV, apolipoprotein E, and apolipoprotein AI. J. Biol. Chem. 1990, 265, 17805–17815. [Google Scholar] [CrossRef] [PubMed]
- Phani, S.; Jablonski, M.; Pelta-Heller, J.; Cai, J.; Iacovitti, L. Gremlin is a novel VTA derived neuroprotective factor for dopamine neurons. Brain Res. 2013, 1500, 88–98. [Google Scholar] [CrossRef]
- Pinto, V.I.; Senini, V.W.; Wang, Y.; Kazembe, M.P.; Mcculloch, C.A. Filamin A protects cells against force-induced apoptosis by stabilizing talin-and vinculin-containing cell adhesions. FASEB J. 2014, 28, 453–463. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Sengupta, A.; Glogauer, M.; McCulloch, C.A. Filamin A regulates cell spreading and survival via β1 integrins. Exp. Cell Res. 2008, 314, 834–846. [Google Scholar] [CrossRef] [PubMed]
- Ciscato, F.; Sciacovelli, M.; Villano, G.; Turato, C.; Bernardi, P.; Rasola, A.; Pontisso, P. SERPINB3 protects from oxidative damage by chemotherapeutics through inhibition of mitochondrial respiratory complex I. Oncotarget 2014, 5, 2418–2427. [Google Scholar] [CrossRef] [PubMed]
- Levy, J.H.; Sniecinski, R.M.; Welsby, I.J.; Levi, M. Antithrombin: Anti-inflammatory properties and clinical applications. Thromb. Haemost. 2016, 115, 712–728. [Google Scholar] [CrossRef] [PubMed]
- Okajima, K.; Uchiba, M. The anti-inflammatory properties of antithrombin III: New therapeutic implications. Semin. Thromb. Hemost. 1998, 24, 27–32. [Google Scholar] [CrossRef]
- Mitchell, R.A.; Liao, H.; Chesney, J.; Fingerle-Rowsor, G.; Baugh, J.; David, J.; Bucala, R. Macrophage migration inhibitory factor (MIF) sustains macrophage proinflammatory function by inhibiting p53: Regulatory role in the innate immune response. Proc. Natl. Acad. Sci. USA 2002, 99, 345–350. [Google Scholar] [CrossRef]
- Camby, I.; Le Mercier, M.; Lefranc, F.; Kiss, R. Galectin-1: A small protein with major functions. Glycobiology 2006, 16, 137–157. [Google Scholar] [CrossRef]
- Dhirapong, A.; Lleo, A.; Leung, P.; Gershwin, M.E.; Liu, F.T. The immunological potential of galectin-1 and-3. Autoimmun. Rev. 2009, 8, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Vashishta, A.; Ohri, S.S.; Vetvicka, V. Pleiotropic effects of cathepsin D. Endocr. Metab. Immune Disord.-Drug Targets (Former. Curr. Drug Targets-Immune Endocr. Metab. Disord.) 2009, 9, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Conus, S. Cathepsins and their involvement in immune responses. Swiss Med. Wkly. 2010, 140, 13042. [Google Scholar] [CrossRef] [PubMed]
- Fesharaki-Zadeh, A. Oxidative Stress in Traumatic Brain Injury. Int. J. Mol. Sci. 2022, 23, 13000. [Google Scholar] [CrossRef] [PubMed]
- Khatri, N.; Thakur, M.; Pareek, V.; Kumar, S.; Sharma, S.; Datusalia, A.K. Oxidative stress: Major threat in traumatic brain injury. CNS Neurol. Disord.-Drug Targets (Former. Curr. Drug Targets-CNS Neurol. Disord.) 2018, 17, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Aeby, E.; Ahmed, W.; Redon, S.; Simanis, V.; Lingner, J. Peroxiredoxin 1 protects telomeres from oxidative damage and preserves telomeric DNA for extension by telomerase. Cell Rep. 2016, 17, 3107–3114. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, X.-S.; Zhou, X.-M.; Gao, Y.-Y.; Chen, C.-L.; Liu, J.-P.; Ye, Z.-N.; Zhang, Z.-H.; Wu, L.-Y.; Li, W.; et al. Peroxiredoxin 1/2 protects brain against H2O2-induced apoptosis after subarachnoid hemorrhage. FASEB J. 2019, 33, 3051–3062. [Google Scholar] [CrossRef]
- Tavender, T.J.; Bulleid, N.J. Peroxiredoxin IV protects cells from oxidative stress by removing H2O2 produced during disulphide formation. J. Cell Sci. 2010, 123, 2672–2679. [Google Scholar] [CrossRef]
- Immenschuh, S.; Baumgart-Vogt, E. Peroxiredoxins, oxidative stress, and cell proliferation. Antioxid. Redox Signal. 2005, 7, 768–777. [Google Scholar] [CrossRef]
- Schittek, B. The multiple facets of dermcidin in cell survival and host defense. J. Innate Immun. 2012, 4, 349–360. [Google Scholar] [CrossRef]
- Schallert, T. Behavioral Tests for Preclinical Intervention Assessment. NeuroRx 2006, 3, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Perlman, H.; Zhang, X.; Chen, M.W.; Walsh, K.; Buttyan, R. An elevated bax/bcl-2 ratio corresponds with the onset of prostate epithelial cell apoptosis. Cell Death Differ. 1999, 6, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, G.A. Matrix metalloproteinases in neuroinflammation. Glia 2002, 39, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Werner, C.; Engelhard, K. Pathophysiology of traumatic brain injury. BJA Br. J. Anaesth. 2007, 99, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Avila, J.; Lucas, J.J.; Pérez, M.; Hernández, F. Role of Tau Protein in Both Physiological and Pathological Conditions. Physiol. Rev. 2004, 84, 361–384. [Google Scholar] [CrossRef] [PubMed]
- Snider, W.D.; Johnson, E.M., Jr. Neurotrophic molecules. Ann. Neurol. Off. J. Am. Neurol. Assoc. Child Neurol. Soc. 1989, 26, 489–506. [Google Scholar] [CrossRef] [PubMed]
- Bendtzen, K. Cytokines and natural regulators of cytokines. Immunol. Lett. 1994, 43, 111–123. [Google Scholar] [CrossRef]
- Alcalá-Barraza, S.R.; Lee, M.S.; Hanson, L.R.; McDonald, A.A.; Frey, W.H.; McLoon, L.K. Intranasal delivery of neurotrophic factors BDNF, CNTF, EPO, and NT-4 to the CNS. J. Drug Target. 2010, 18, 179–190. [Google Scholar] [CrossRef]
- Chiu, C.C.; Liao, Y.E.; Yang, L.Y.; Wang, J.Y.; Tweedie, D.; Karnati, H.K.; Wang, J.Y. Neuroinflammation in animal models of traumatic brain injury. J. Neurosci. Methods 2016, 272, 38–49. [Google Scholar] [CrossRef]
- Liddelow, S.A.; Guttenplan, K.A.; Clarke, L.E.; Bennett, F.C.; Bohlen, C.J.; Schirmer, L.; Barres, B.A. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 2017, 541, 481–487. [Google Scholar] [CrossRef]
- Flevaris, P.; Vaughan, D. The Role of Plasminogen Activator Inhibitor Type-1 in Fibrosis. Semin. Thromb. Hemost. 2017, 43, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Yarza, R.; Vela, S.; Solas, M.; Ramirez, M.J. c-Jun N-terminal kinase (JNK) signaling as a therapeutic target for Alzheimer’s disease. Front. Pharmacol. 2016, 6, 321. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.P.; Chio, C.C.; Cheong, C.U.; Chao, C.M.; Cheng, B.C.; Lin, M.T. Hypoxic preconditioning enhances the therapeutic potential of the secretome from cultured human mesenchymal stem cells in experimental traumatic brain injury. Clin. Sci. 2013, 124, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Chuang, T.J.; Lin, K.C.; Chio, C.C.; Wang, C.C.; Chang, C.P.; Kuo, J.R. Effects of secretome obtained from normoxia-preconditioned human mesenchymal stem cells in traumatic brain injury rats. J. Trauma Acute Care Surg. 2012, 73, 1161–1167. [Google Scholar] [CrossRef] [PubMed]
- Tajiri, N.; Acosta, S.A.; Shahaduzzaman, M.; Ishikawa, H.; Shinozuka, K.; Pabon, M.; Hernandez-Ontiveros, D.; Kim, D.W.; Metcalf, C.; Staples, M.; et al. Intravenous Transplants of Human Adipose-Derived Stem Cell Protect the Brain from Traumatic Brain Injury-Induced Neurodegeneration and Motor and Cognitive Impairments: Cell Graft Biodistribution and Soluble Factors in Young and Aged Rats. J. Neurosci. 2014, 34, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Diao, Y.F.; Wang, J.; Liang, J.; Xu, H.H.; Zhao, M.L.; Li, X.H. Intravenously infusing the secretome of adipose-derived mesenchymal stem cells ameliorates neuroinflammation and neurological functioning after traumatic brain injury. Stem Cells Dev. 2020, 29, 222–234. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Y.; Wei, M.G.; Liang, J.; Xu, H.H.; Wang, J.J.; Wang, J.; Yang, X.P.; Lv, F.F.; Wang, K.Q.; Duan, J.H.; et al. Injury-preconditioning secretome of umbilical cord mesenchymal stem cells amplified the neurogenesis and cognitive recovery after severe traumatic brain injury in rats. J. Neurochem. 2020, 153, 230–251. [Google Scholar] [CrossRef] [PubMed]
- Salikhova, D.; Bukharova, T.; Cherkashova, E.; Namestnikova, D.; Leonov, G.; Nikitina, M.; Gubskiy, I.; Akopyan, G.; Elchaninov, A.; Midiber, K.; et al. Therapeutic Effects of hiPSC-Derived Glial and Neuronal Progenitor Cells-Conditioned Medium in Experimental Ischemic Stroke in Rats. Int. J. Mol. Sci. 2021, 22, 4694. [Google Scholar] [CrossRef] [PubMed]
- Isaev, N.K.; Novikova, S.V.; Stelmashook, E.V.; Barskov, I.V.; Silachev, D.N.; Khaspekov, L.G.; Zorov, D.B. Mitochondria-targeted plastoquinone antioxidant SkQR1 decreases trauma-induced neurological deficit in rat. Biochemistry 2012, 77, 996–999. [Google Scholar] [CrossRef]
- Lee, D.H.; Hong, S.H.; Kim, S.K.; Lee, C.S.; Phi, J.H.; Cho, B.K.; Wang, K.C. Reproducible and persistent weakness in adult rats after surgical resection of motor cortex: Evaluation with limb placement test. Child’s Nerv. Syst. 2009, 25, 1547–1553. [Google Scholar] [CrossRef]
- Magno, L.A.V.; Collodetti, M.; Tenza-Ferrer, H.; Romano-Silva, M.A. Cylinder test to assess sensory-motor function in a mouse model of Parkinson’s disease. Bio-Protoc. 2019, 9, 3337. [Google Scholar] [CrossRef] [PubMed]
- Hamm, R.J.; Pike, B.R.; O’DELL, D.M.; Lyeth, B.G.; Jenkins, L.W. The rotarod test: An evaluation of its effectiveness in assessing motor deficits following traumatic brain injury. J. Neurotrauma 1994, 11, 187–196. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salikhova, D.I.; Golovicheva, V.V.; Fatkhudinov, T.K.; Shevtsova, Y.A.; Soboleva, A.G.; Goryunov, K.V.; Dyakonov, A.S.; Mokroysova, V.O.; Mingaleva, N.S.; Shedenkova, M.O.; et al. Therapeutic Efficiency of Proteins Secreted by Glial Progenitor Cells in a Rat Model of Traumatic Brain Injury. Int. J. Mol. Sci. 2023, 24, 12341. https://doi.org/10.3390/ijms241512341
Salikhova DI, Golovicheva VV, Fatkhudinov TK, Shevtsova YA, Soboleva AG, Goryunov KV, Dyakonov AS, Mokroysova VO, Mingaleva NS, Shedenkova MO, et al. Therapeutic Efficiency of Proteins Secreted by Glial Progenitor Cells in a Rat Model of Traumatic Brain Injury. International Journal of Molecular Sciences. 2023; 24(15):12341. https://doi.org/10.3390/ijms241512341
Chicago/Turabian StyleSalikhova, Diana I., Victoria V. Golovicheva, Timur Kh. Fatkhudinov, Yulia A. Shevtsova, Anna G. Soboleva, Kirill V. Goryunov, Alexander S. Dyakonov, Victoria O. Mokroysova, Natalia S. Mingaleva, Margarita O. Shedenkova, and et al. 2023. "Therapeutic Efficiency of Proteins Secreted by Glial Progenitor Cells in a Rat Model of Traumatic Brain Injury" International Journal of Molecular Sciences 24, no. 15: 12341. https://doi.org/10.3390/ijms241512341
APA StyleSalikhova, D. I., Golovicheva, V. V., Fatkhudinov, T. K., Shevtsova, Y. A., Soboleva, A. G., Goryunov, K. V., Dyakonov, A. S., Mokroysova, V. O., Mingaleva, N. S., Shedenkova, M. O., Makhnach, O. V., Kutsev, S. I., Chekhonin, V. P., Silachev, D. N., & Goldshtein, D. V. (2023). Therapeutic Efficiency of Proteins Secreted by Glial Progenitor Cells in a Rat Model of Traumatic Brain Injury. International Journal of Molecular Sciences, 24(15), 12341. https://doi.org/10.3390/ijms241512341







