Metabolic Oscillations and Glycolytic Phenotypes of Cancer Cells
Abstract
1. Introduction
2. Mathematical Model
3. Results
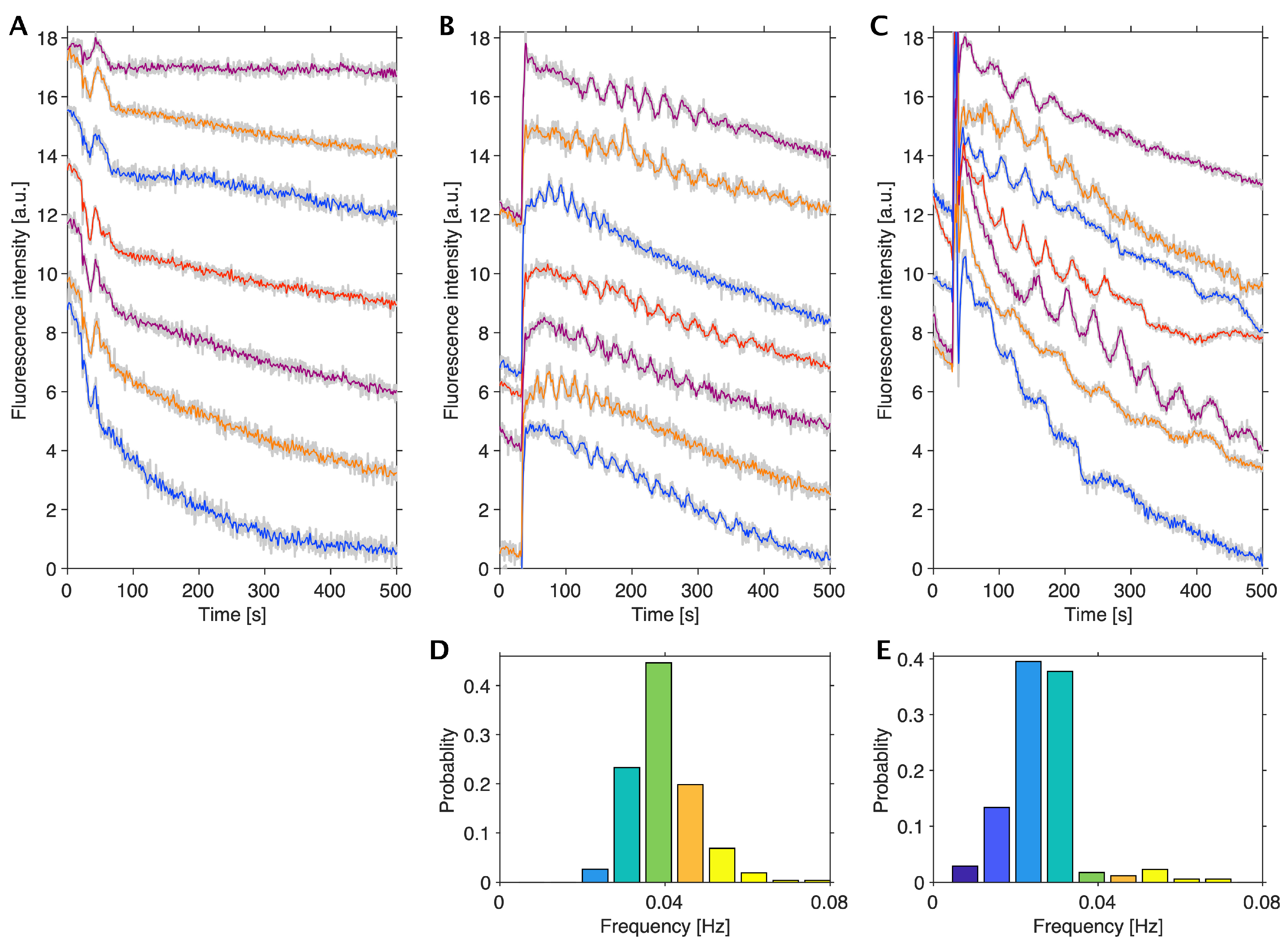
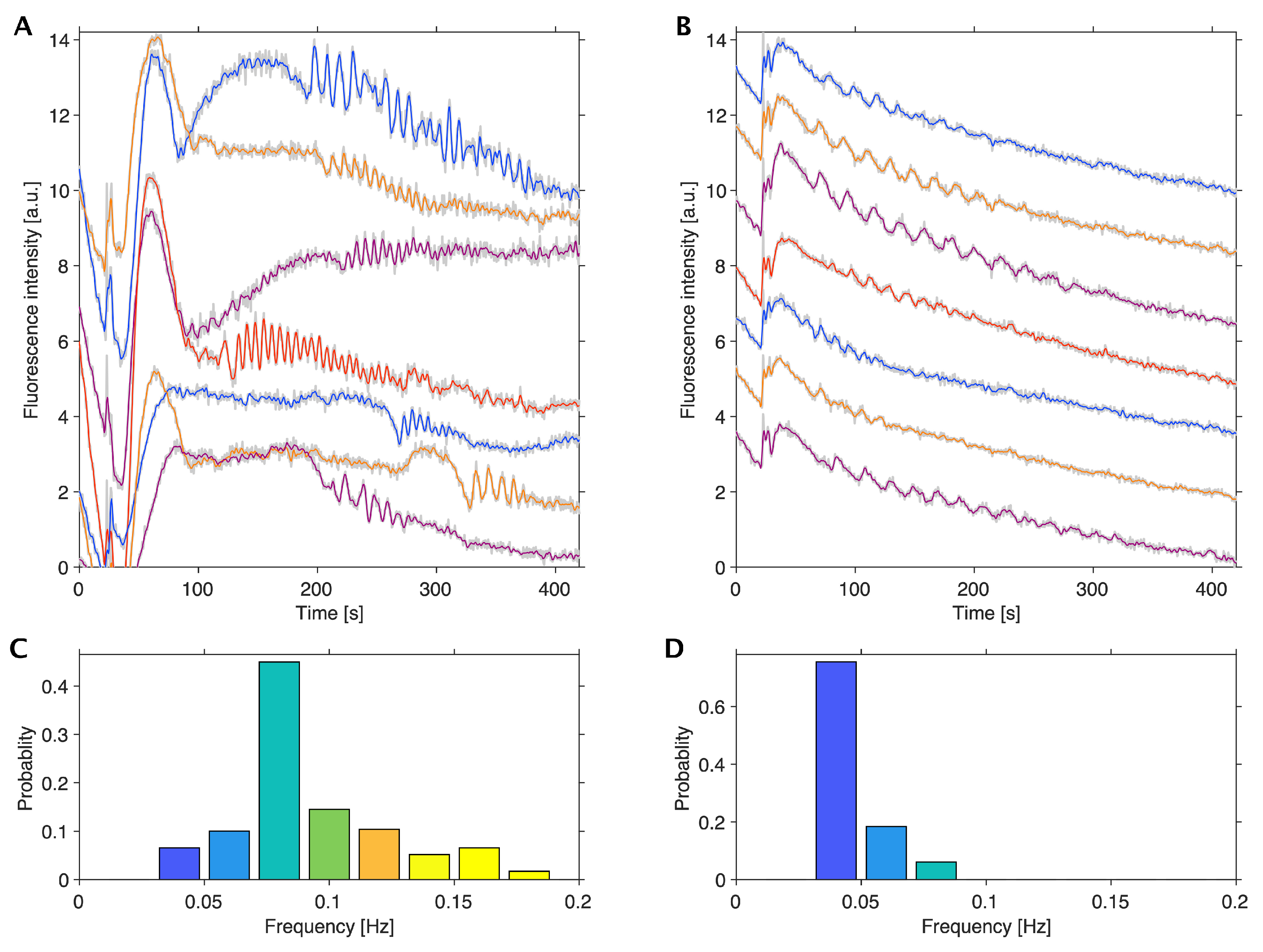
3.1. Glycolytic Oscillations in Monolayers of HeLa Cervical and DU145 Prostate Cancer Cells
3.2. Comparison of Spheroids and Monolayers of HeLa Cells
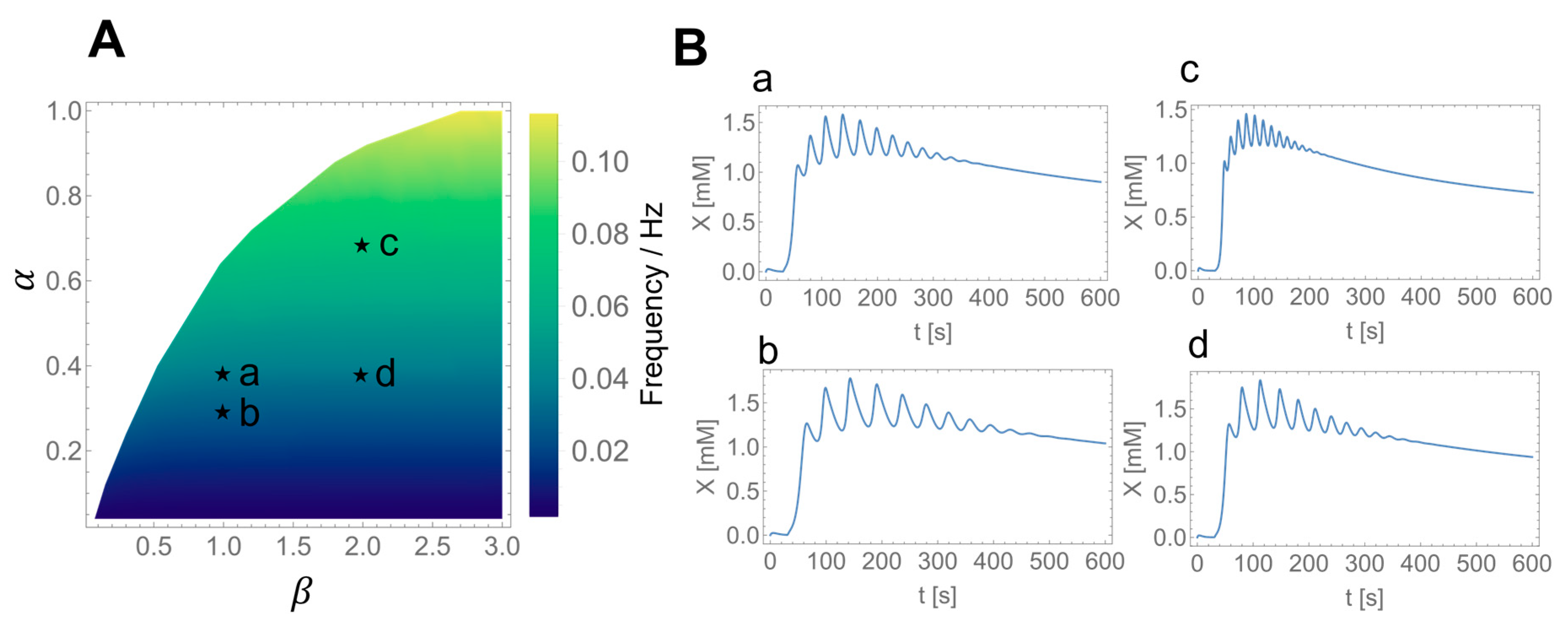
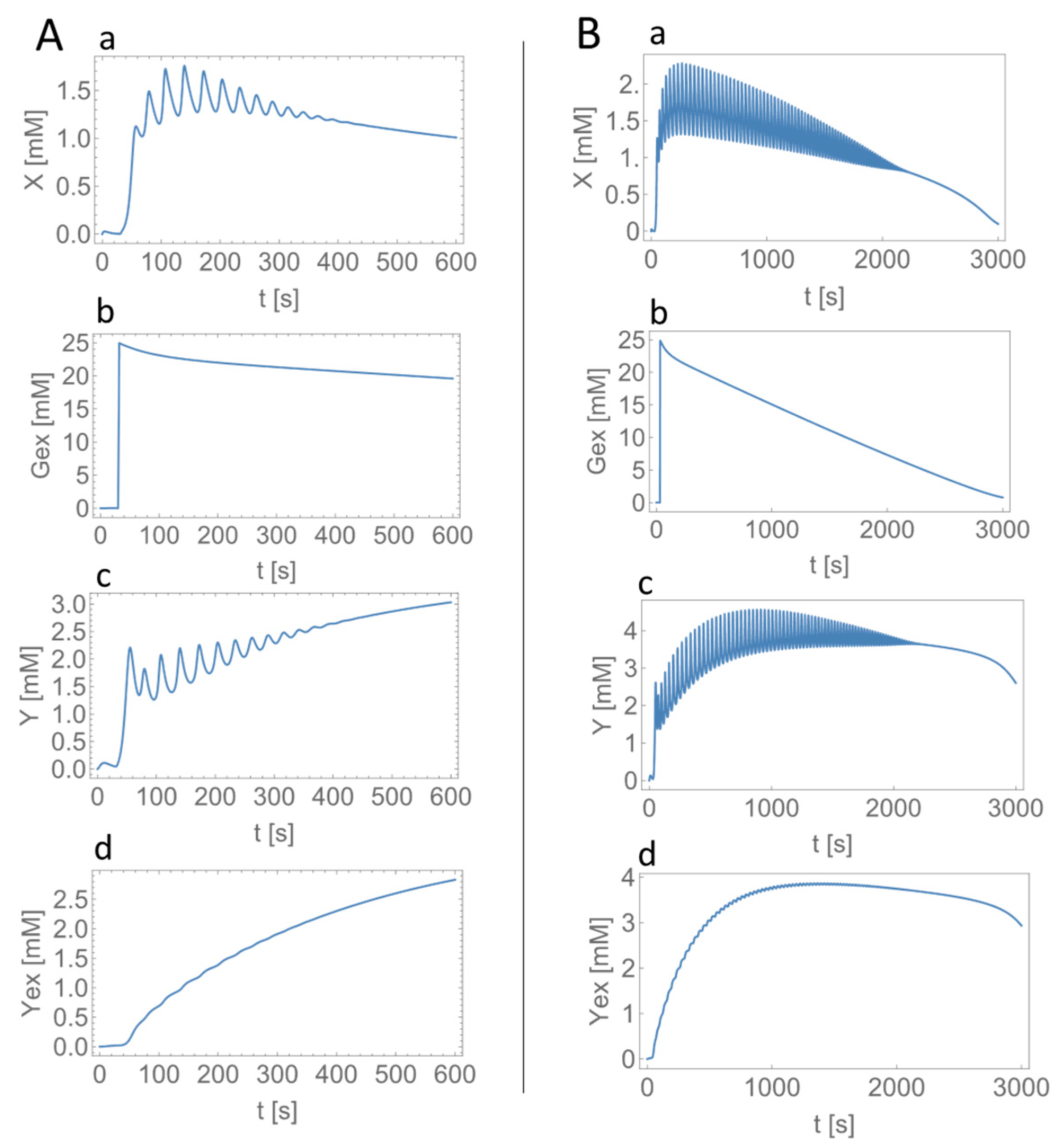
3.3. Numerical Simulations of Glycolytic Oscillations in HeLa and DU145 Cells
3.4. Effect of the Inhibitory Feedback Mechanism on the Glycolytic Oscillations
4. Discussion
5. Materials and Methods
5.1. Cultures and Starvation of Glucose for HeLa Cervical and DU145 Prostate Cancer Cells in Monolayers
5.2. Cultures and Starvation of Glucose for HeLa Cervical Cancer Cells in Spheroids and Monolayers
5.3. Fluorescence Microscopy
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- DeBerardinis, R.J.; Chandel, N.S. Fundamentals of cancer metabolism. Sci. Adv. 2016, 2, e1600200. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wang, J. Uncovering the underlying mechanisms of cancer metabolism through the landscapes and probability flux quantifications. iScience 2020, 23, 101002. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. Über den stoffwechsel der carcinomzelle. Klin. Wochenschr. 1925, 4, 534–536. [Google Scholar] [CrossRef]
- Warburg, O. On the origin of cancer cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Potter, M.; Newport, E.; Morten, K.J. The Warburg effect: 80 years on. Biochem. Soc. Trans. 2016, 44, 1499–1505. [Google Scholar]
- Feng, J.; Li, J.; Wu, L.; Yu, Q.; Ji, J.; Wu, J.; Dai, W.; Guo, C. Emerging roles and the regulation of aerobic glycolysis in hepatocellular carcinoma. J. Exp. Clin. Cancer Res. 2022, 39, 126. [Google Scholar] [CrossRef]
- Wu, M.; Neilson, A.; Swift, A.L.; Moran, R.; Tamagnine, J.; Parslow, D.; Armistead, S.; Lemire, K.; Orrell, J.; Teich, J.; et al. Multiparameter metabolic analysis reveals a close link between attenuated mitochondrial bioenergetic function and enhanced glycolysis dependency in human tumor cells. Am. J. Physiol. Cell Physiol. 2007, 292, C125–C136. [Google Scholar] [CrossRef]
- Grover-McKay, M.; Walsh, S.A.; Seftor, E.A.; Thomas, P.A.; Hendrix, M.J. Role for glucose transporter 1 protein in human breast cancer. Pathol. Oncol. Res. 1998, 4, 115–120. [Google Scholar] [CrossRef]
- Vlassenko, A.G.; McConathy, J.; Couture, L.E.; Su, Y.; Massoumzadeh, P.; Leeds, H.S.; Chicoine, M.R.; Tran, D.D.; Huang, J.; Dahiya, S.; et al. Aerobic glycolysis as a marker of tumor aggressiveness: Preliminary data in high grade human brain tumors. Dis. Merkers 2015, 2015, 874904. [Google Scholar] [CrossRef]
- Chacon-Barahona, J.A.; MacKeigan, J.P.; Lanning, N.J. Unique metabolic contexts sensitize cancer cells and discriminate between glycolytic tumor types. Cancers 2023, 15, 1158. [Google Scholar] [CrossRef] [PubMed]
- Pavlova, N.N.; Thompson, C.B. The emerging hallmarks of cancer metabolism. Cell Metab. 2016, 23, 24–47. [Google Scholar] [CrossRef] [PubMed]
- Jia, D.; Park, J.H.; Jung, K.H.; Levine, H.; Kaipparettu, B.A. Elucidating the metabolic plasticity of cancer: Mitochondrial reprogramming and hybrid metabolic states. Cells 2018, 7, 21. [Google Scholar] [CrossRef]
- Jia, D.; Lu, M.; Jung, K.H.; Park, J.H.; Yu, L.; Onuchic, J.N.; Kaipparettu, B.A.; Levine, H. Elucidating cancer metabolic plasticity by coupling gene regulation with metabolic pathways. Proc. Natl. Acad. USA 2019, 116, 3909–3918. [Google Scholar] [CrossRef]
- Rossignol, R.; Gilkerson, R.; Aggeler, R.; Yamagata, K.; Remington, S.J.; Capaldi, R.A. Energy substrate modulates mitochondrial structure and oxidative capacity in cancer cells. Cancer Res. 2004, 64, 985–993. [Google Scholar] [CrossRef] [PubMed]
- Ibsen, K.H.; Schiller, K.W. Oscillations of nucleotides and glycolytic intermediates in aerobic suspensions of Ehrlich ascites tumor cells. Biochim. Biophys. Acta 1967, 131, 405–407. [Google Scholar] [CrossRef] [PubMed]
- Chance, B.B.; Estabrook, R.W.; Ghosh, A. Damped sinusoidal oscillations of cytoplasmic reduced pyridine nucleotide in yeast cell. Proc. Natl. Acad. Sci. USA 1964, 51, 1244–1251. [Google Scholar] [CrossRef]
- Frenkel, R. Control of reduced diphosphopyridine nucleotide oscillations in beef heart extracts. I. Effect of modifiers of phosphofructokinase activity. Arch. Biochem. Biophys. 1968, 125, 151–156. [Google Scholar] [CrossRef]
- Amemiya, T.; Shibata, K.; Itoh, Y.; Itoh, K.; Watanabe, M.; Yamaguchi, T. Primordial oscillations in life: Direct observation of glycolytic oscillations in individual HeLa cervical cancer cells. Chaos 2017, 27, 104602. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Racker, E. Regulatory mechanism in carbohydrate metabolism. IV. Pasteur effect and Crabtree effect in ascites tumor cells. J. Biol. Chem. 1959, 234, 1036–1041. [Google Scholar] [CrossRef] [PubMed]
- Weber, A.; Prokazov, Y.; Zuschratter, W.; Hauser, M.J.B. Desynchronisation of glycolytic oscillations in yeast cell populations. PLoS ONE 2012, 7, e43276. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, T.; Shibata, K.; Watanabe, M.; Nakata, S.; Nakamura, K.; Yamaguchi, T. Glycolytic oscillations in cancer cells. In Physics of Biological Oscillators; Stefanovska, A., McClintock, P.V.E., Eds.; Springer: Cham, Switzerland, 2021; pp. 245–259. [Google Scholar]
- Amemiya, T.; Shibata, K.; Takahashi, J.; Watanabe, M.; Nakata, S.; Nakamura, K.; Yamaguchi, T. Glycolytic oscillations in HeLa cervical cancer cell spheroids. FEBS J. 2022, 289, 5551–5570. [Google Scholar] [CrossRef]
- Janiszewska, M.; Primi, M.C.; Izard, T. Cell adhesion in cancer: Beyond the migration of single cells. J. Biol. Chem. 2020, 295, 2495–2505. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Enríquez, S.; Marín-Hernández, A.; Gallardo-Pérez, J.C.; Moreno-Sánchez, R. Kinetics of transport and phosphorylation of glucose in cancer cells. J. Cell Physiol. 2008, 216, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Pereira, P.M.R.; Berisha, N.; Bhupathiraju, N.V.S.D.K.; Fernandes, R.; Tomé, J.P.C.; Drain, C.M. Cancer cell spheroids are a better screen for the photodynamic efficiency of glycosylated photosensitizers. PLoS ONE 2017, 12, e0177737. [Google Scholar] [CrossRef] [PubMed]
- Stadler, M.; Scherzer, M.; Walter, S.; Holzner, S.; Pudelko, K.; Riedl, A.; Unger, C.; Kramer, N.; Weil, B.; Neesen, J.; et al. Exclusion from spheroid formation identifies loss of essential cell-cell adhesion molecules in colon cancer cells. Sci. Rep. 2018, 8, 1151. [Google Scholar] [CrossRef]
- Wolf, J.; Heinrich, R. Effect of cellular interaction on glycolytic oscillations in yeast: A theoretical investigation. Biochem. J. 2000, 345, 321–334. [Google Scholar] [CrossRef]
- Amemiya, T.; Shibata, K.; Du, Y.; Nakata, S.; Yamaguchi, T. Modeling studies of heterogeneities in glycolytic oscillations in HeLa cervical cancer cells. Chaos 2019, 29, 033132. [Google Scholar] [CrossRef]
- Evans, P.R.; Farrants, G.W.; Hudson, P.J. Phosphofructokinase: Structure and control. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1981, 293, 53–62. [Google Scholar] [CrossRef]
- Ogawa, T.; Mori, H.; Tomita, M.; Yoshino, M. Inhibitory effect of phosphoenolpyruvate on glycolytic enzymes in Escherichia Coli. Res. Microbiol. 2007, 158, 159–163. [Google Scholar] [CrossRef]
- Leite, T.C.; Coelho, R.G.; Silva, D.D.; Coelho, W.S.; Marinho-Carvalho, M.M.; Sola-Penna, M. Lactate downregulates the glycolytic enzymes hexokinase and phosphofructokinase in diverse tissues from mice. FEBS Lett. 2011, 585, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, V.; Krishnamoorthy, G.; Govindarajulu, P. Enzymes of carbohydrate metabolism in human breast carcinoma: Relationship with serum hormones. Neoplasma 1994, 41, 49–54. [Google Scholar] [PubMed]
- Wang, L.; Schumann, U.; Liu, Y.; Prokopchuk, O.; Steinacker, J.M. Heat shock protein 70 (Hsp 70) inhibits oxidative phosphorylation and compensates ATP balance through enhanced glycolytic activity. J. Appl. Physiol. 2012, 113, 1669–1676. [Google Scholar] [CrossRef] [PubMed]
- Zheng, N.; Wang, K.; He, J.; Qiu, Y.; Xie, G.; Su, M.; Jia, W.; Li, H. Effects of ADMA on gene expression and metabolism in serum-starved LoVo cells. Sci. Rep. 2012, 6, 25892. [Google Scholar] [CrossRef] [PubMed]
- Hegarty, T.W. Temperature coefficient (q10), seed germination and germination and other biological processes. Nature 1973, 243, 305–306. [Google Scholar] [CrossRef]
- Elias, M.; Wieczorek, G.; Rosenne, S.; Tawfik, D.S. The universality of enzymatic rate-temperature dependency. Trends Biochem. Sci. 2014, 39, 1–7. [Google Scholar] [CrossRef]
- Yurkovich, J.T.; Zielinski, D.C.; Yang, L.; Paglia, G.; Rolfsson, O.; Sigurjónsson, Ó.E.; Broddrick, J.T.; Bordbar, A.; Wichuk, K.; Brynjólfsson, S.; et al. Quantitative time-course metabolomics in human red blood cells reveal the temperature dependence of human metabolic networks. J. Biol. Chem. 2017, 292, 19556–19564. [Google Scholar] [CrossRef]
- Deng, M.; Qiao, Y.; Liu, C.; Wang, Z.; Shi, J.; Pan, T.; Mao, Y.; Mei, Z.; Huang, F.; Tian, Y. Tricolor core/shell polymeric ratiometric nanosensors for intracellular glucose and oxygen dual sensing. Sens. Actuators B Chem. 2019, 286, 437–444. [Google Scholar] [CrossRef]
- Chandra, F.A.; Buzi, G.; Doyle, J.C. Glycolytic oscillations and limits on robust efficiency. Science 2011, 333, 187–192. [Google Scholar] [CrossRef]
- Dasgupta, B.; Chhipa, R.R. Evolving lessons on the complex role of AMPK in normal physiology and cancer. Trens Pharmacol. Sci. 2016, 37, 192–206. [Google Scholar] [CrossRef]
- Moldogazieva, N.T.; Mokhosoev, I.M.; Terentiev, A.A. Metabolic heterogeneity of cancer cells: An interplay between HIF-1, GLUTs, and AMPK. Cancers 2020, 12, 862. [Google Scholar] [CrossRef]
- Shaw, S.N.; Amos, H. Insulin stimulation of glucose entry in chick fibroblasts and Hela cells. Biochem. Biophys. Res. Commun. 1973, 53, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Roberts, D.J.; Miyamoto, S. Hexokinase II integrates energy metabolism and cellular protection: Akting on mitochondria and TORCing to autophagy. Cell Death Differ. 2015, 22, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Jurica, M.S.; Mesecar, A.; Heath, P.J.; Shi, W.; Nowak, T.; Stoddard, B.L. The allosteric regulation of pyruvate kinase by fructose-1,6-bisphosphate. Structure 1998, 6, 195–210. [Google Scholar] [CrossRef] [PubMed]
- Wood, T. The inhibition of pyruvate kinase by ATP. Biochem. Biophys. Res. Commun. 1968, 31, 779–785. [Google Scholar] [CrossRef]
- Aboagye, E.O.; Mori, N.; Bhujwalla, Z.M. Effect of malignant transformation on lactate levels of human mammary epithelial cells. Advan. Enzyme Regul. 2001, 41, 251–260. [Google Scholar] [CrossRef]
- Grashei, M.; Biechl, P.; Schilling, F.; Otto, A.M. Conversion of Hyperpolarized [1-13C] pyruvate in breast cancer cells depends on their malignancy, metabolic program and nutrient microenvironment. Cancers 2022, 14, 1845. [Google Scholar] [CrossRef]
- Liao, J.; Qian, F.; Tchabo, N.; Mhawech-Fauceglia, P.; Beck, A.; Qian, Z.; Wang, X.; Huss, W.J.; Lele, S.B.; Morrison, C.D.; et al. Ovarian cancer spheroid cells with stem cell-like properties contribute to tumor generation, metastasis and chemotherapy resistance through hypoxia-resistant metabolism. PLoS ONE 2014, 9, e84941. [Google Scholar]
- Gatenby, R.A.; Gillies, R.J. Why do caners have high aerobic glycolysis? Nat. Rev. Cancer 2004, 4, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Pelicano, H.; Zhang, W.; Liu, J.; Hammoudi, N.; Dai, J.; Xu, R.-H.; Pusztai, L.; Huang, P. Mitochondrial dysfunction in some triple-negative breast cancer cell lines: Role of mTOR pathway and therapeutic potential. Breast Cancer Res. 2014, 16, 434. [Google Scholar] [CrossRef]
- Chen, H.H.W.; Chiu, N.-T.; Su, W.-C.; Guo, H.-R.; Lee, B.-F. Prognostic value of whole-body total lesion glycolysis at pretreatment FDG PET/CT in non-small cell lung cancer. Radiology 2012, 264, 559–566. [Google Scholar] [CrossRef]
- Collisson, E.A.; Sadanandam, A.; Olson, P.; Gibb, W.J.; Truitt, M.; Gu, S.; Cooc, J.; Weinkle, J.; Kim, G.E.; Jakkula, L.; et al. Subtypes of pancreatic ductal adenocarcinoma and their differing responses to therapy. Nat. Med. 2011, 17, 500–504. [Google Scholar] [CrossRef]
- Daemen, A.; Peterson, D.; Sahu, N.; McCord, R.; Du, X.; Liu, B.; Kowanetz, K.; Hong, R.; Moffat, J.; Gao, M.; et al. Metabolic profiling stratifies pancreatic ductal adenocarcinomas into subtypes with distinct sensitivities to metabolic inhibitors. Proc. Natl. Acd. Sci. USA 2015, 112, E4410–E4417. [Google Scholar] [CrossRef] [PubMed]
- Aon, M.A.; Cortassa, S.; Marbán, E.; O’Rourke, B. Synchronized whole cell oscillations in mitochondrial metabolism triggered by a local release of reactive oxygen species in cardiac myocytes. J. Biol. Chem. 2003, 278, 44735–44744. [Google Scholar] [CrossRef] [PubMed]
- Lancaster, G.; Suprunenko, Y.F.; Jenkins, K.; Stefanovska, A. Modelling chronotaxicity of cellular energy metabolism to facilitate the identification of altered metabolic states. Sci. Rep. 2016, 6, 29584. [Google Scholar] [CrossRef] [PubMed]
- Compan, V.; Pierredon, S.; Vanderperre, B.; Krznar, P.; Marchiq, I.; Zamboni, N.; Pouyssegur, J.; Martinou, J.-C. Monitoring mitochondrial pyruvate carrier activity in real time using a BRET-based biosensor: Investigation of the Warburg effect. Mol. Cell 2015, 59, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Wu, D.; Walker, M.; Wang, P.; Tian, R.; Wang, W. Genetically encoded biosensors for evaluating NAD+/NADH ratio in cytosolic and mitochondrial compartments. Cell Rep. Methods 2021, 1, 100116. [Google Scholar] [CrossRef]
- Nakajima, E.C.; Van Houten, B. Metabolic symbiosis in cancer: Refocusing the Warburg lends. Mol. Carcinog. 2013, 52, 329–337. [Google Scholar] [CrossRef]
- Kim, J.; DeBerardinis, R.J. Mechanism and implications of metabolic heterogeneity in cancer. Cell Metab. 2019, 30, 434–446. [Google Scholar] [CrossRef] [PubMed]
- Pavlides, S.; Whitaker-Menezes, D.; Castello-Cros, R.; Flomenberg, N.; Witkiewicz, A.K.; Frank, P.G.; Casimiro, M.C.; Wang, C.; Fortina, P.; Addya, S. The reverse Warburg effect: Aerobic glycolysis in cancer associated fibroblasts and tumor stroma. Cell Cycle 2009, 8, 3984–4001. [Google Scholar] [CrossRef]
- Amemiya, T.; Yamaguchi, T. Oscillations and dynamic symbiosis in cellular metabolism in cancer. Front. Oncol. 2022, 12, 783908. [Google Scholar] [CrossRef] [PubMed]
- Relier, S.; Yazdani, L.; Ayad, O.; Choqet, A.; Bourgaux, J.-F.; Prudhomme, M.; Pannequin, J.; Macari, F.; David, A. Antibiotics inhibit sphere-forming ability in suspension culture. Cancer Cell Int. 2016, 16, 6. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to Image J: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Shibata, K.; Amemiya, T.; Kawakita, Y.; Obase, K.; Itoh, K.; Takinoue, M.; Nakata, S.; Yamaguchi, T. Promotion and inhibition of synchronous glycolytic oscillations in yeast by chitosan. FEBS J. 2018, 285, 2679–2690. [Google Scholar] [CrossRef]
- Shibata, K.; Amemiya, T. How to decide window-size of smoothing methods: A goodness of fit criterion for smoothing oscillation data. IEICE Trans. Electron. 2019, E102-C, 143–146. [Google Scholar]
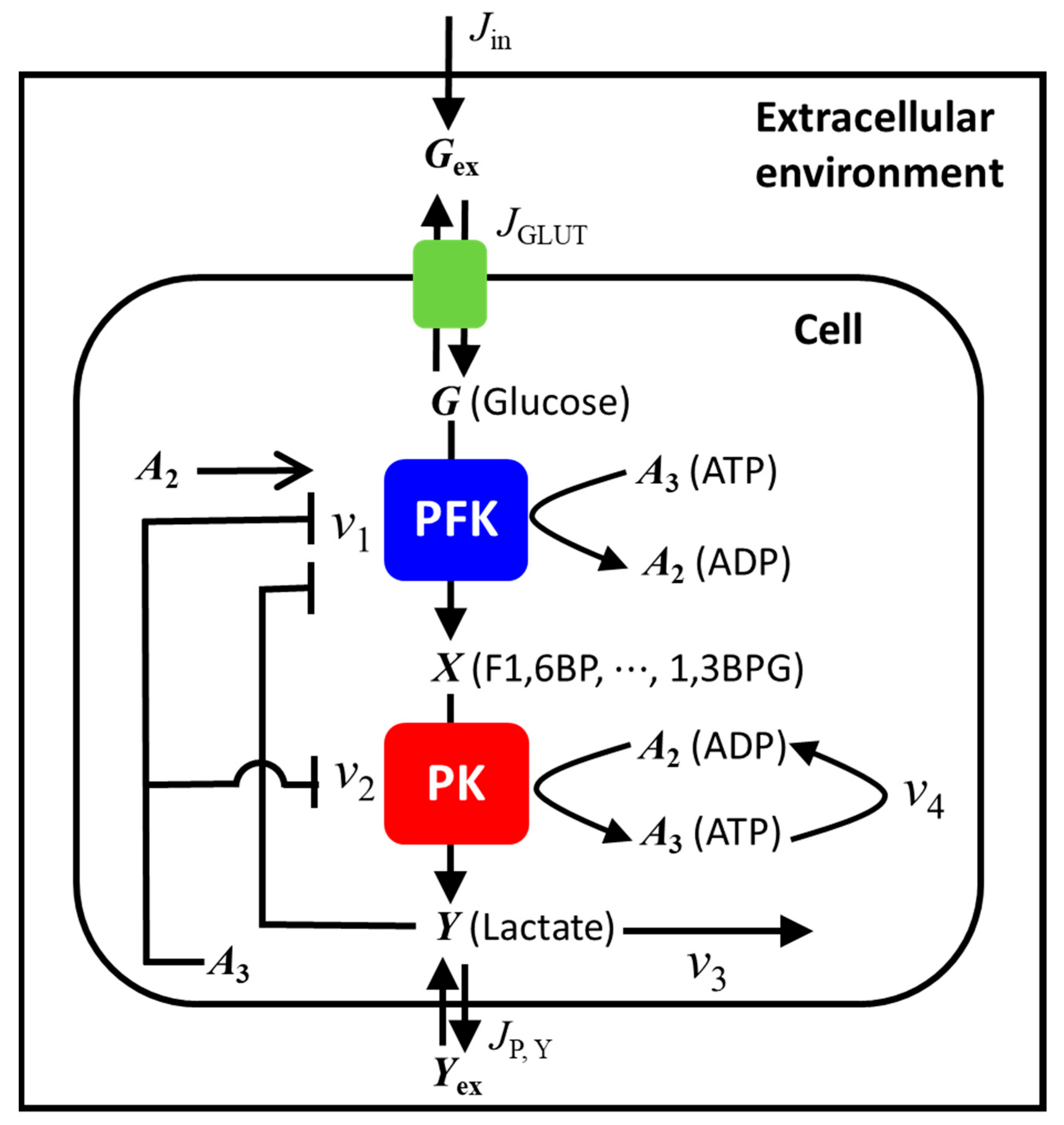
| Rate laws |
| Transport kinetics |
| Reaction rates |
, |
| Model-Step | Parameters | Meaning | Values | Sources |
|---|---|---|---|---|
| 20 or 25 mM | Experiments | |||
| Normalized maximum glucose uptake | 1.0 or 2.0 | Equation (2) | ||
| Ratio of cellular volume to extracellular volume | 0.1 | [23,29] | ||
| Michaelis constant of GLUT for intracellular glucose | 12 mM | [23,29] | ||
| Michaelis constant of GLUT for extracellular glucose | 10 mM | [23,29] | ||
| Equilibrium constant | 1.0 | [23,29] | ||
| Transport constant of MCT | [23,29] | |||
| Rate constant of PFK reaction | Equation (1) | |||
| A common parameter for the four rate constants | 0.27–0.68 | This work | ||
| [23,29] | ||||
| The number of substrate molecules bound to PFK | 4 | [23,29] | ||
| Dissociation constant for free PFK and m-molecules of ADP | [23,29] | |||
| Dissociation constant for free PFK and m-molecules of ATP | [23,29] | |||
| Dissociation constant for ADP-activated PFK and glucose | 1.0 mM | [23,29] | ||
| Dissociation constant for ADP-activated PFK and ATP | 1.0 mM | [23,29] | ||
| (lactate) | This work | |||
| Rate constant of PK reaction | Equation (1) | |||
| A common parameter for the four rate constants | 0.27–0.68 | This work | ||
| [23,29] | ||||
| The number of substrate molecules Boud to PK | 4 | [23,29] | ||
| Dissociation constant for free PK and n-molecule of ATP | [23,29] | |||
| (pool of intermediates) | 20 mM | [23,29] | ||
| Dissociation constant for free PK and ADP | 20 mM | [23,29] | ||
| (lactate) | Equation (1) | |||
| A common parameter for the four rate constants | 0.27–0.68 | This work | ||
| [23,29] | ||||
| Rate constant of consumption of ATP | Equation (1) | |||
| A common parameter for the four rate constants | 0.27–0.68 | This work | ||
| [23,29] |
| Initial Concentrations | Sources | |
|---|---|---|
| Variables | Values | |
| 0.30 mM | [39] | |
| 0.30 mM | [23,29] | |
| 0.30 mM | [23,29] | |
| 0 mM | [23,29] | |
| 0 mM | [23,29] | |
| Total concentration of ATP and ADP | ||
| Constant | Value | |
| 3.0 mM | [28] | |
| (at 25 °C) | (at 37 °C) | ||||||
|---|---|---|---|---|---|---|---|
| HeLa Cells | DU145 Cells | Spheroids | Monolayers | ||||
| Exp. (Hz) | Sim. (Hz) | Exp. (Hz) | Sim. (Hz) | Exp. (Hz) | Sim. (Hz) | Exp. (Hz) | Sim. (Hz) |
| 0.034 | 0.035 | 0.023 | 0.024 | 0.070 | 0.072 | 0.031 | 0.034 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amemiya, T.; Shibata, K.; Yamaguchi, T. Metabolic Oscillations and Glycolytic Phenotypes of Cancer Cells. Int. J. Mol. Sci. 2023, 24, 11914. https://doi.org/10.3390/ijms241511914
Amemiya T, Shibata K, Yamaguchi T. Metabolic Oscillations and Glycolytic Phenotypes of Cancer Cells. International Journal of Molecular Sciences. 2023; 24(15):11914. https://doi.org/10.3390/ijms241511914
Chicago/Turabian StyleAmemiya, Takashi, Kenichi Shibata, and Tomohiko Yamaguchi. 2023. "Metabolic Oscillations and Glycolytic Phenotypes of Cancer Cells" International Journal of Molecular Sciences 24, no. 15: 11914. https://doi.org/10.3390/ijms241511914
APA StyleAmemiya, T., Shibata, K., & Yamaguchi, T. (2023). Metabolic Oscillations and Glycolytic Phenotypes of Cancer Cells. International Journal of Molecular Sciences, 24(15), 11914. https://doi.org/10.3390/ijms241511914





