Effect of Non-Modified as Well as Surface-Modified SiO2 Nanoparticles on Red Blood Cells, Biological and Model Membranes
Abstract
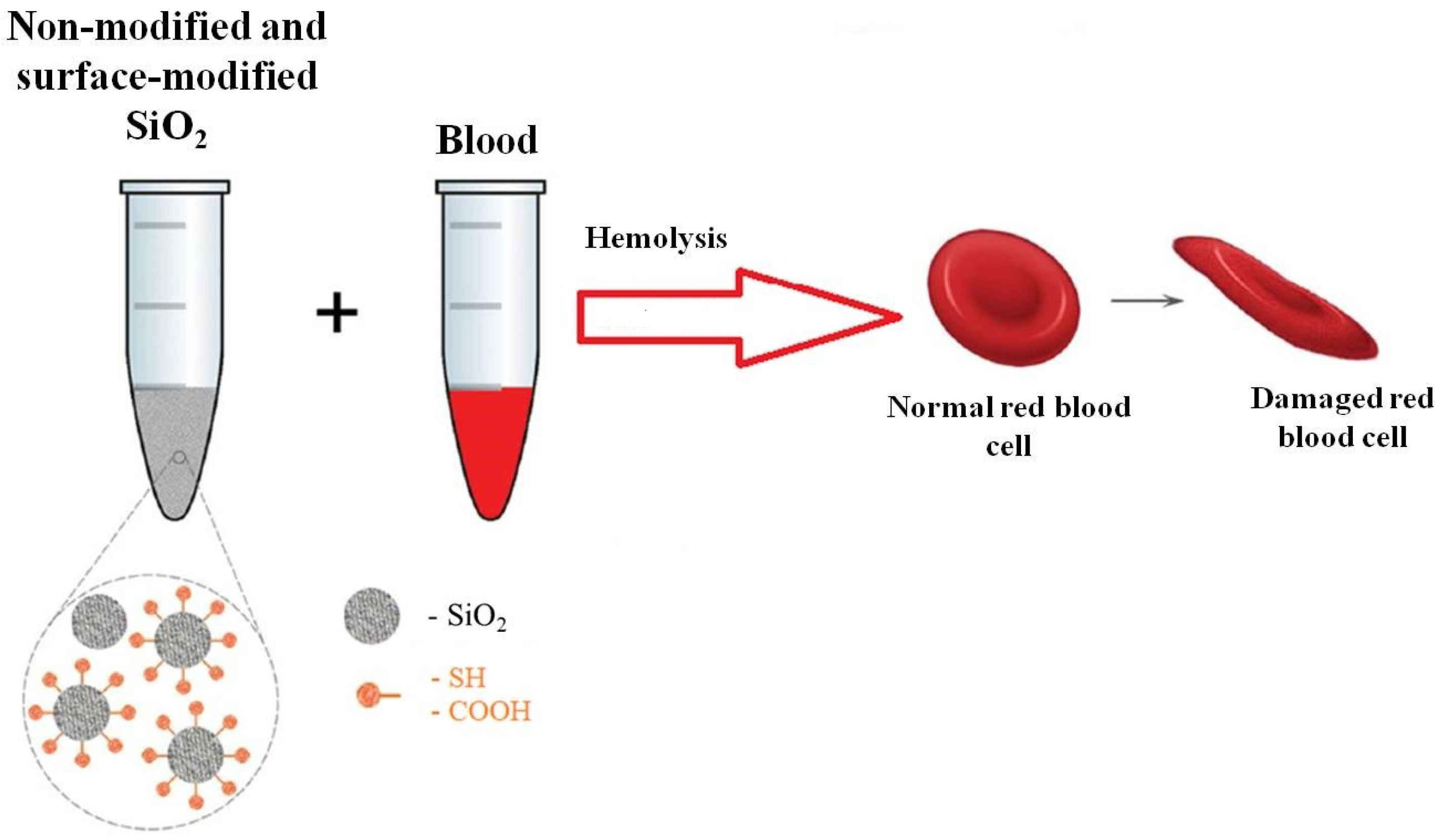
1. Introduction
2. Results
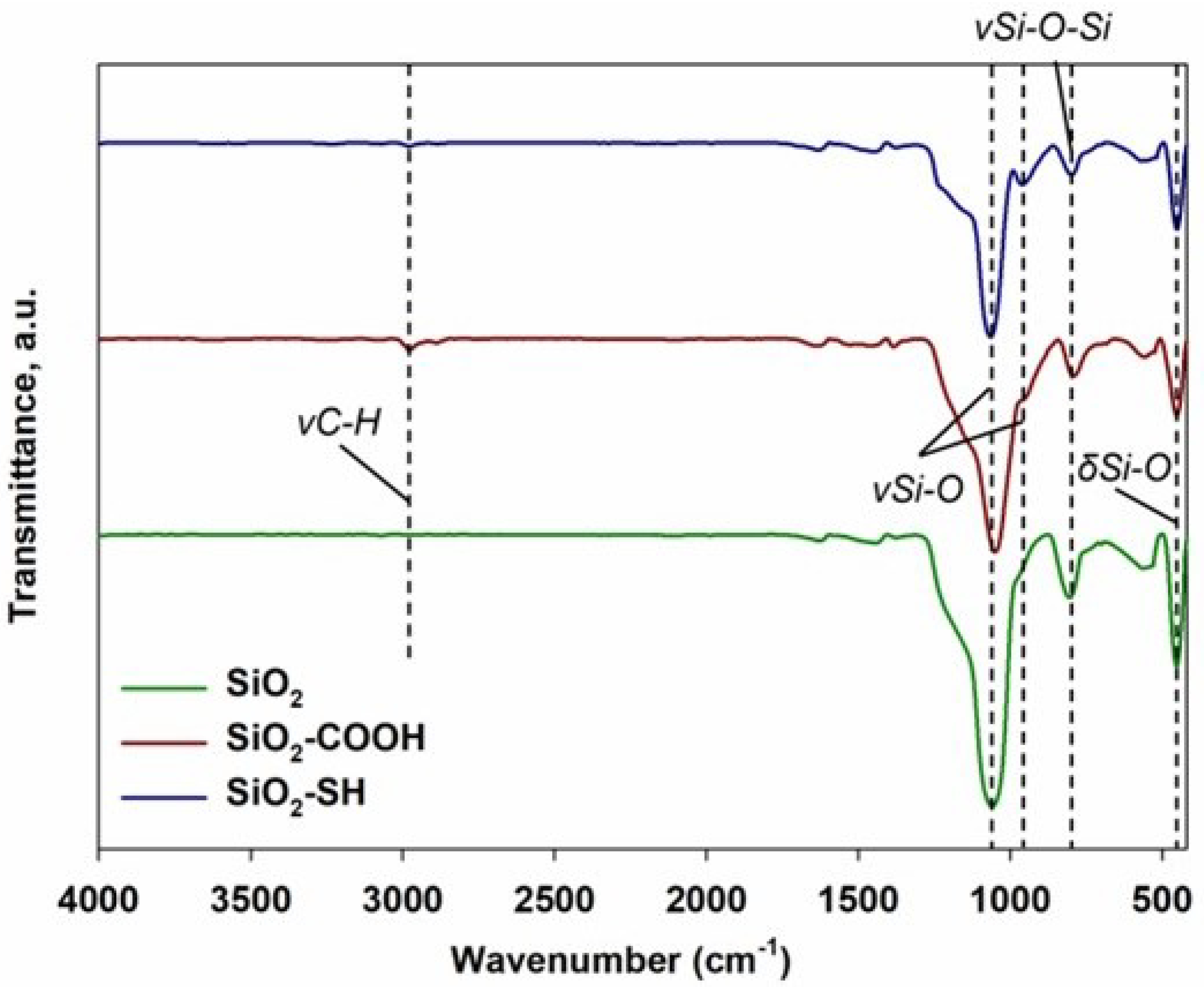
2.1. Attenuated Total Reflectance Infrared Spectroscopy Technique (ATR-FTIR)
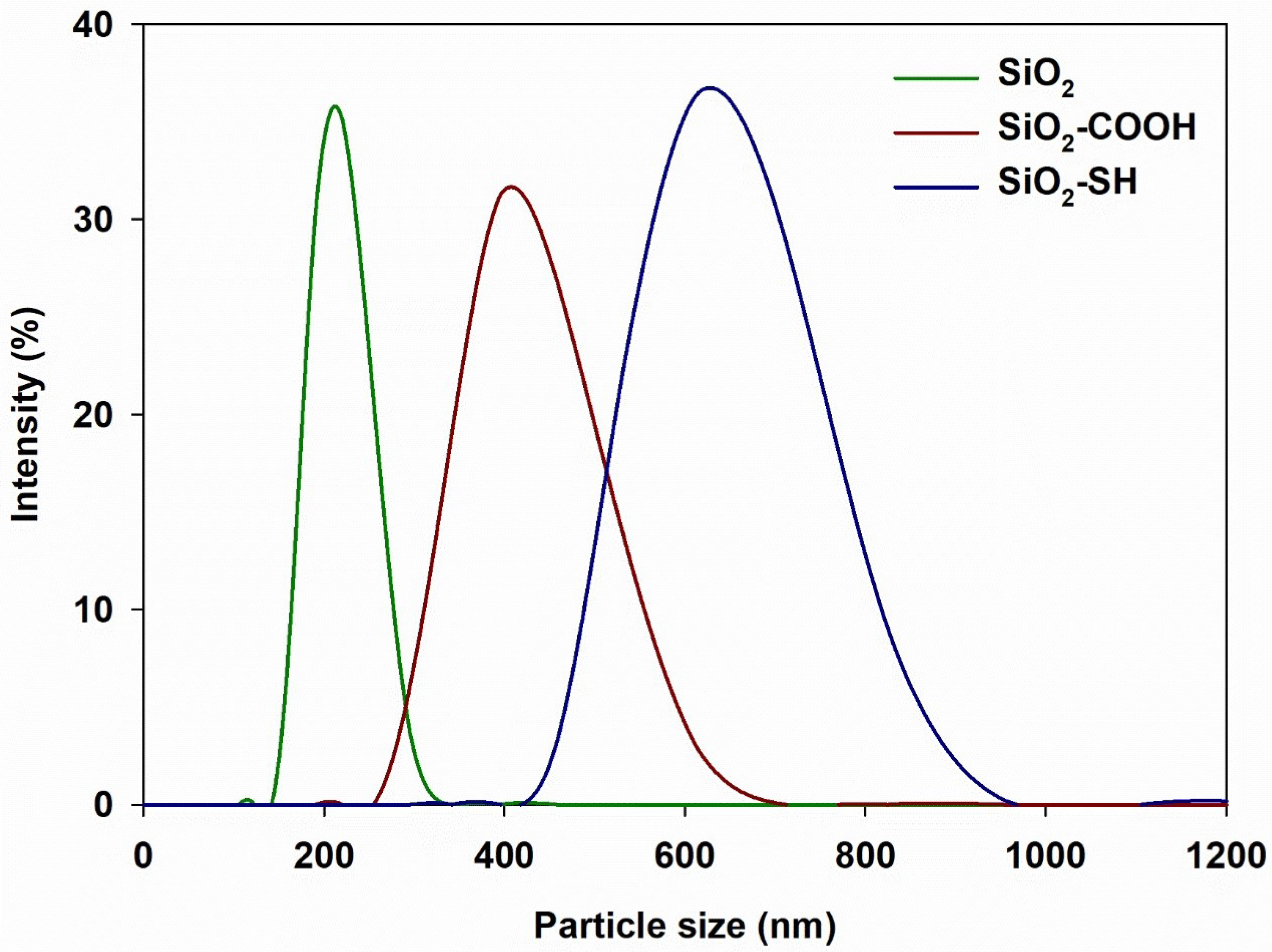
2.2. Particle Size Ranges Measurement
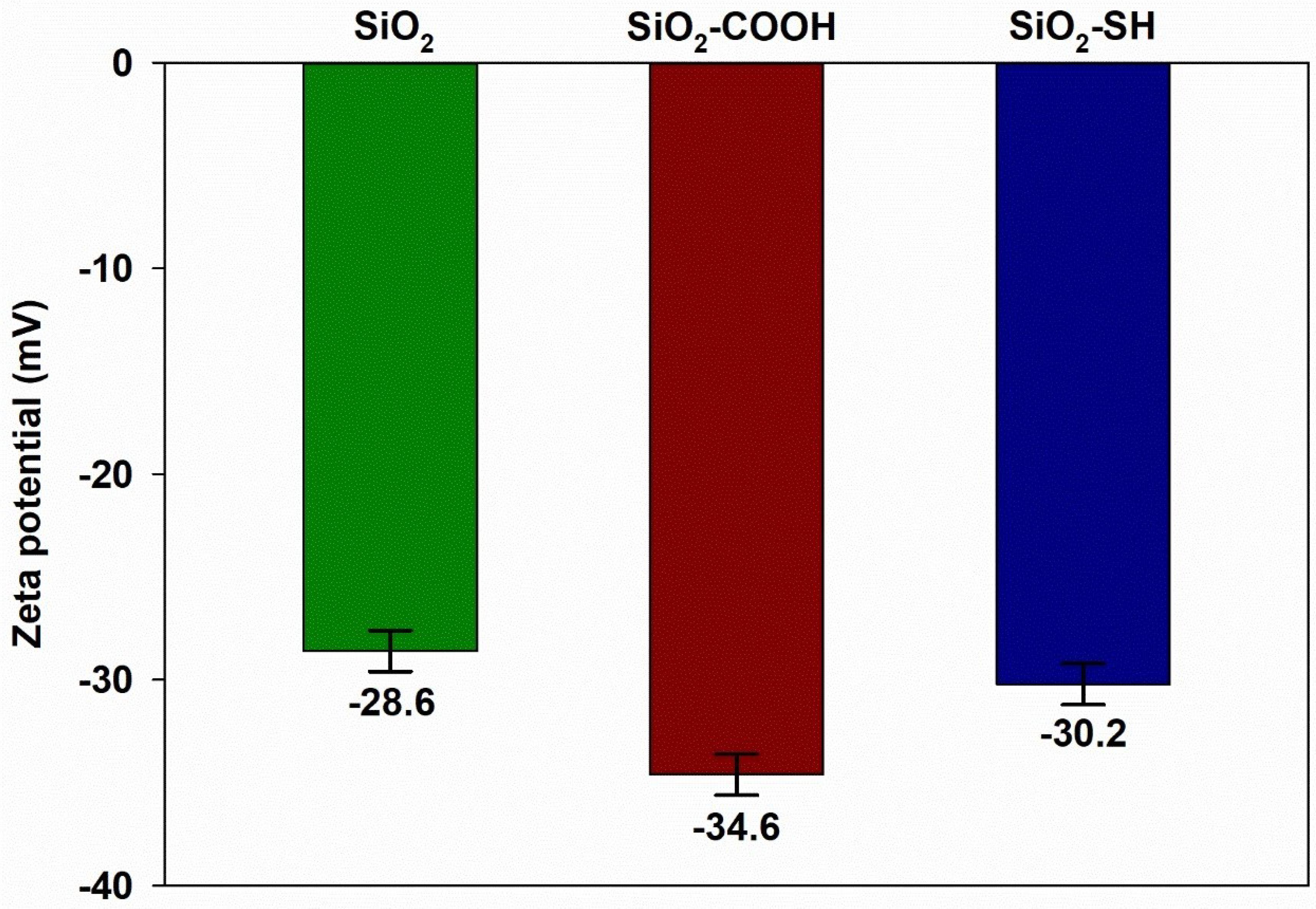
2.3. Electrokinetic Potential Measurement
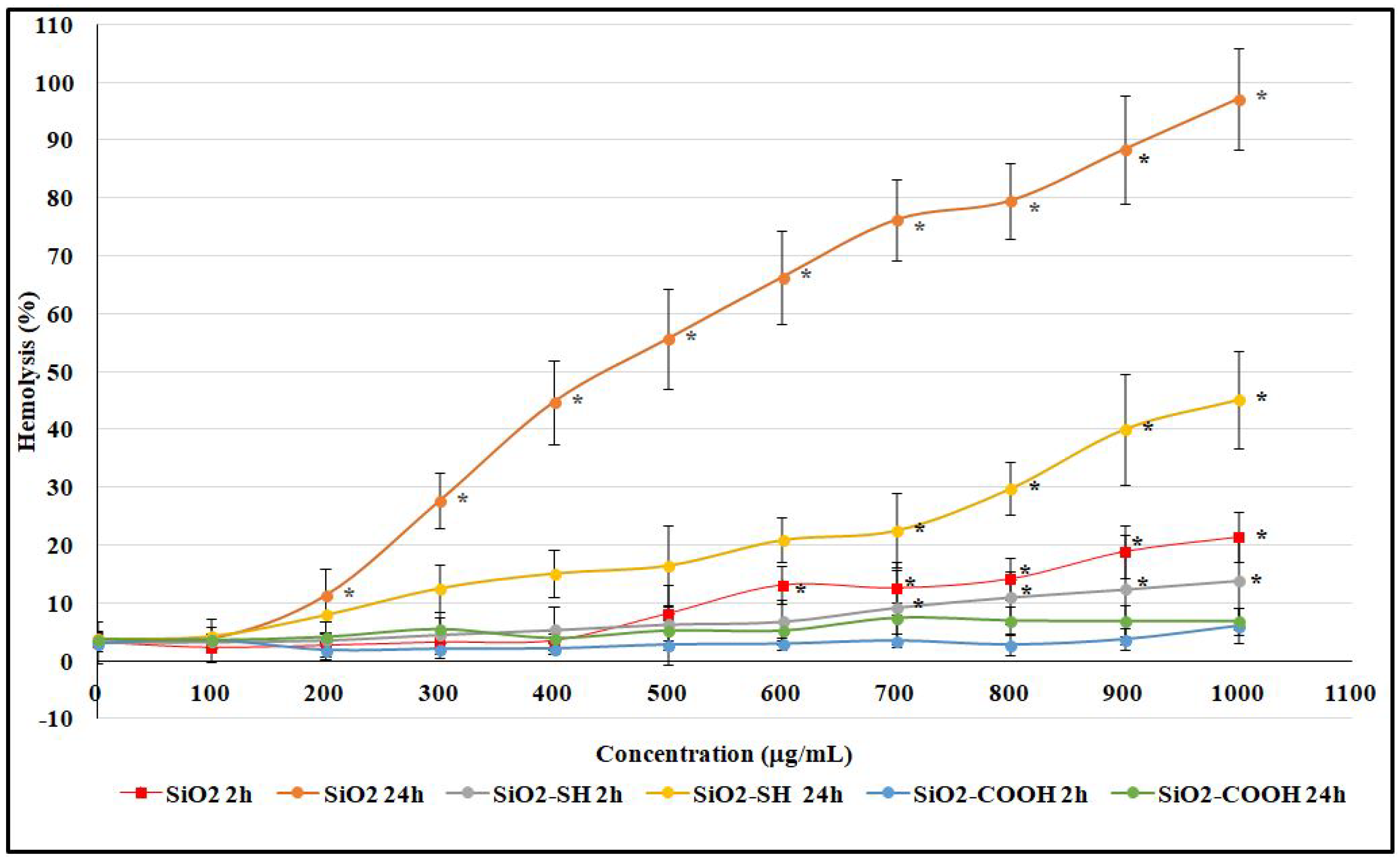
2.4. Hemolysis Assay of Erythrocytes
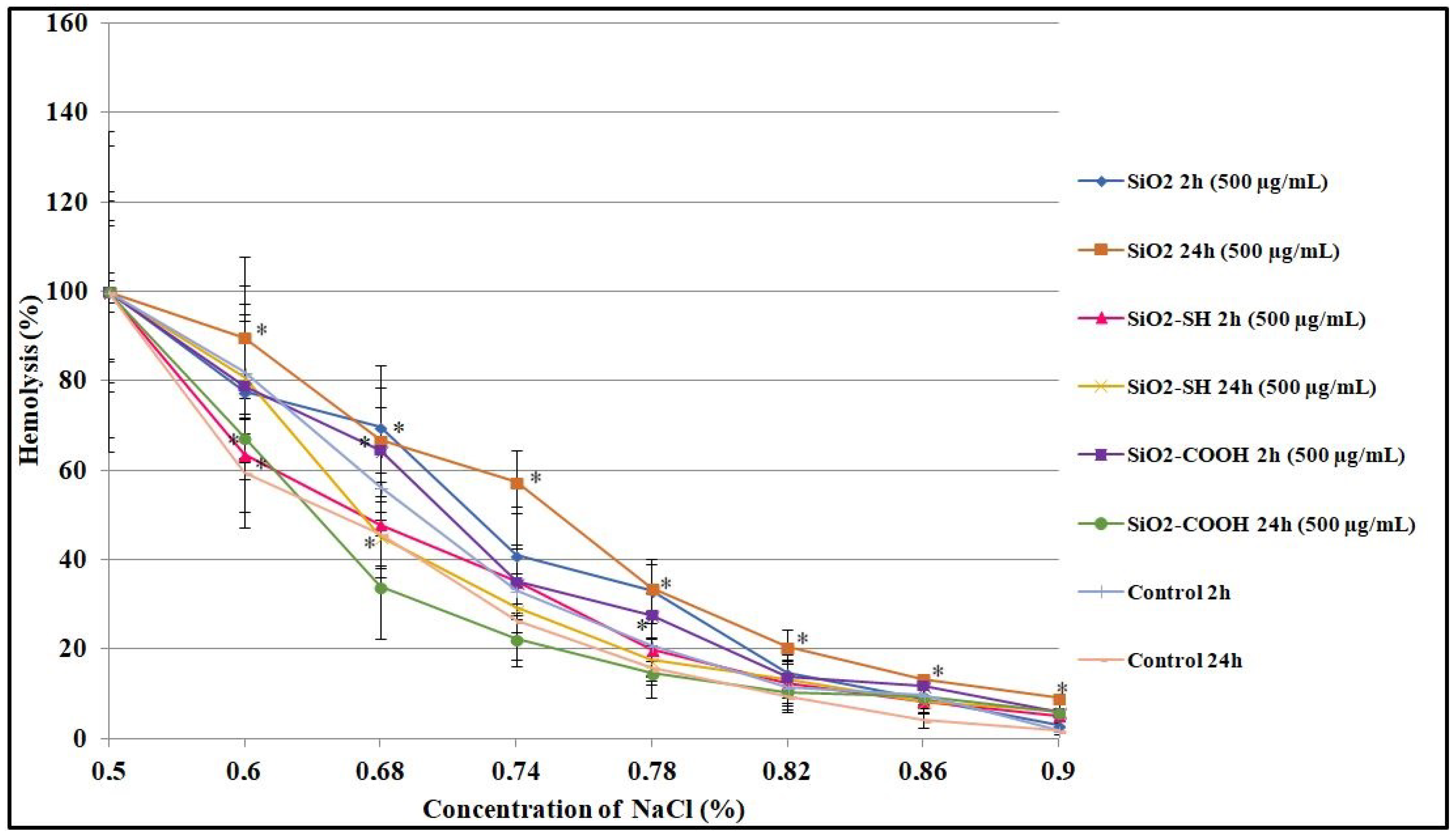
2.5. Osmotic Resistance of Erythrocytes
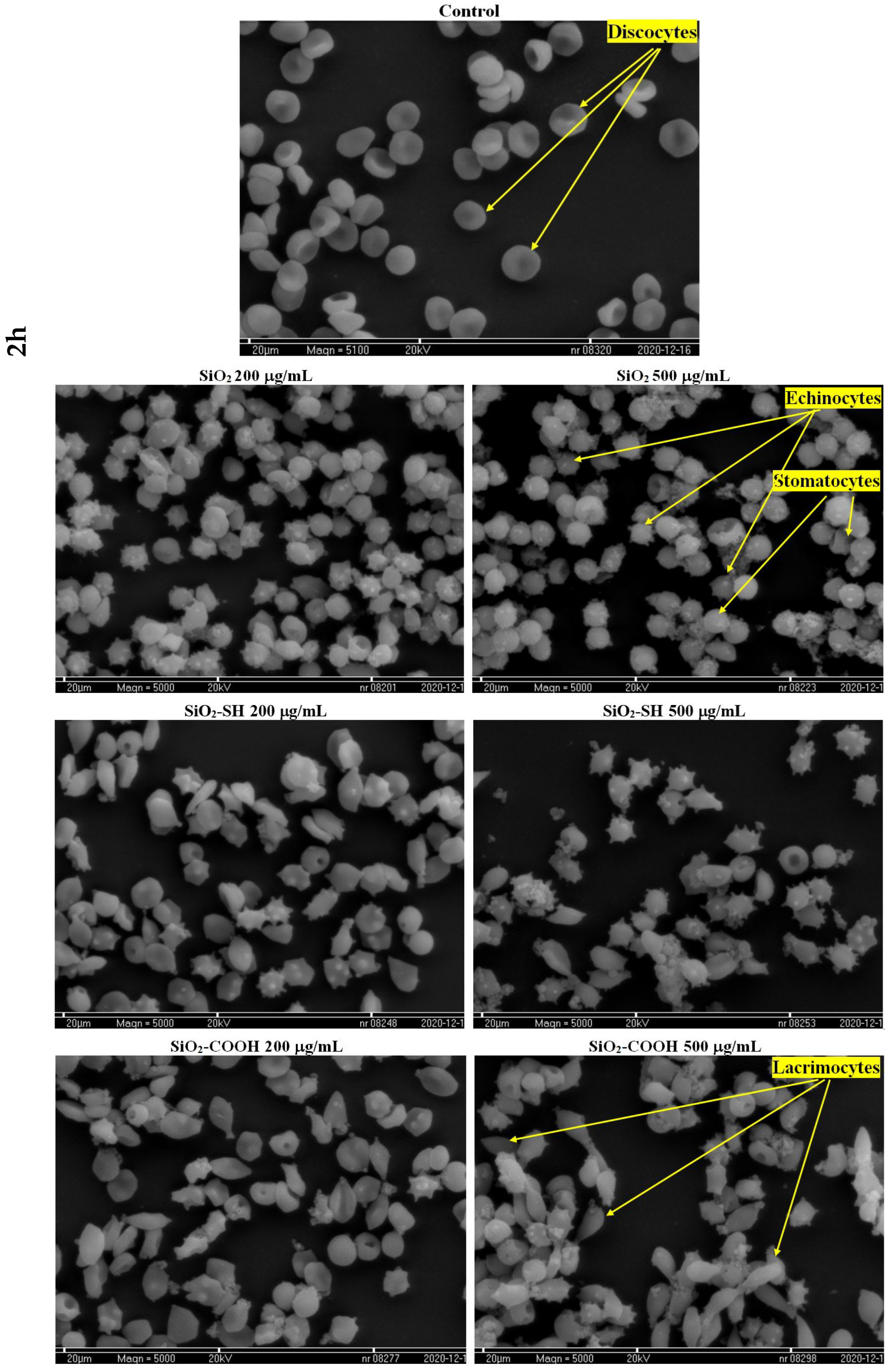
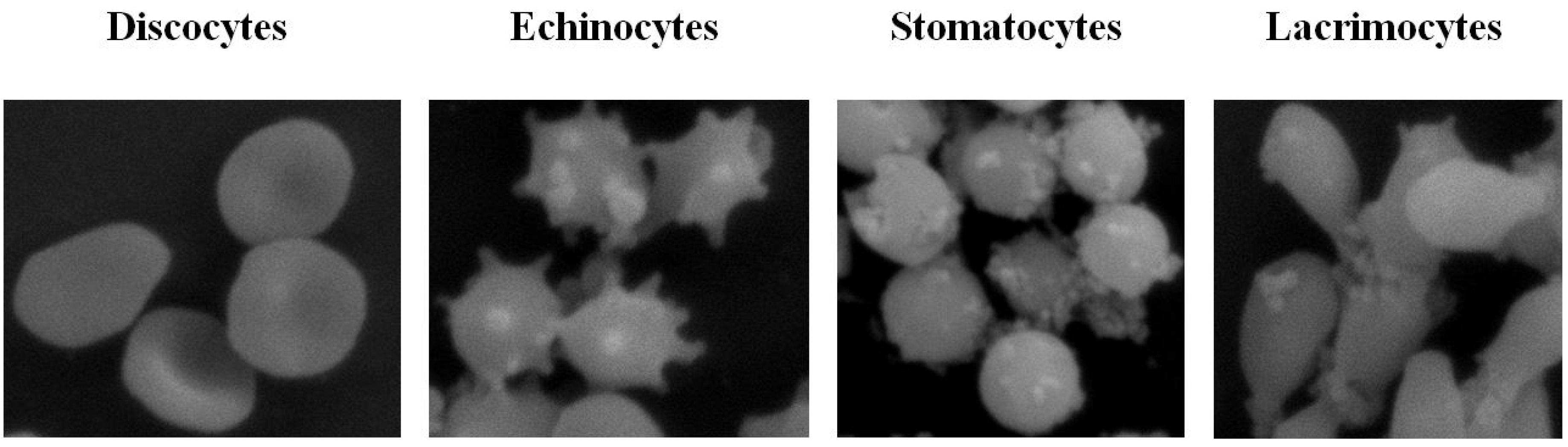
2.6. Microscopic Studies of Erythrocyte Shapes
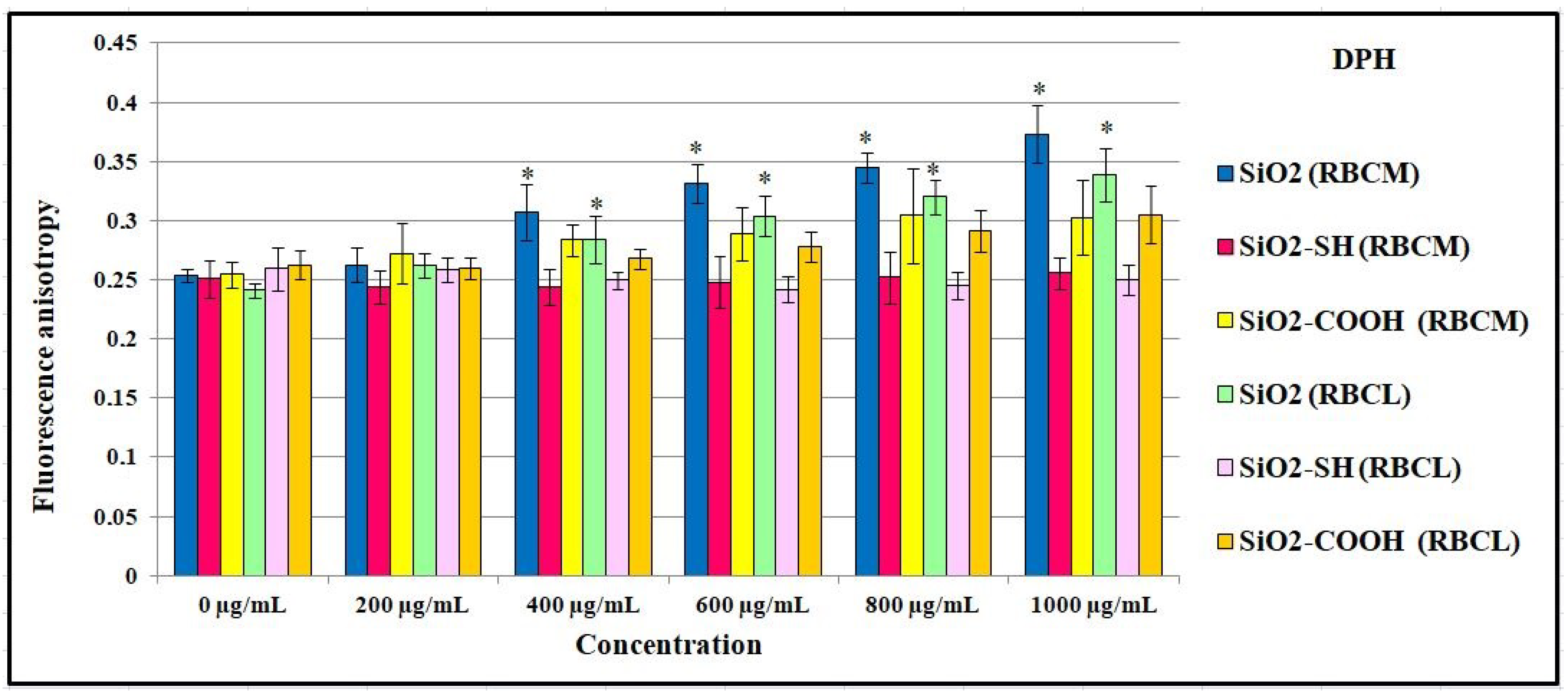
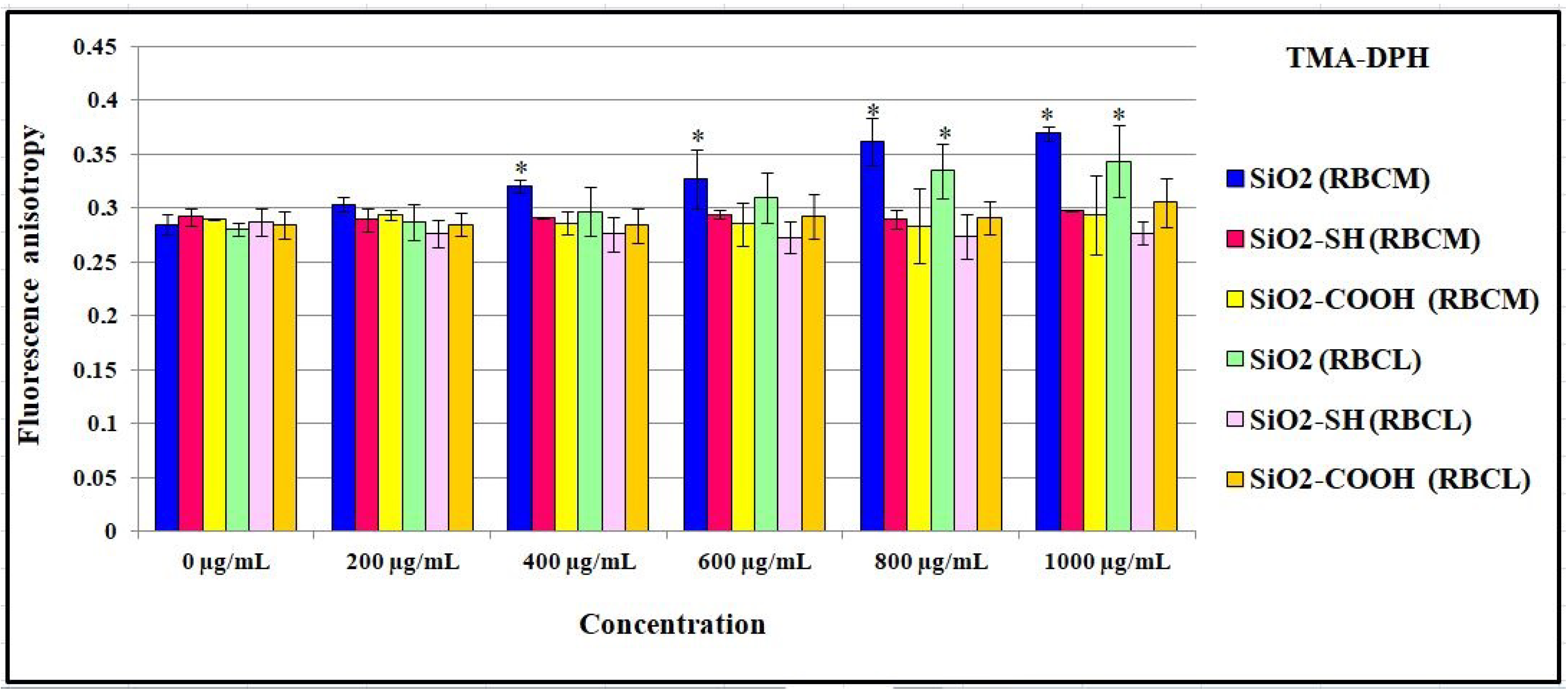
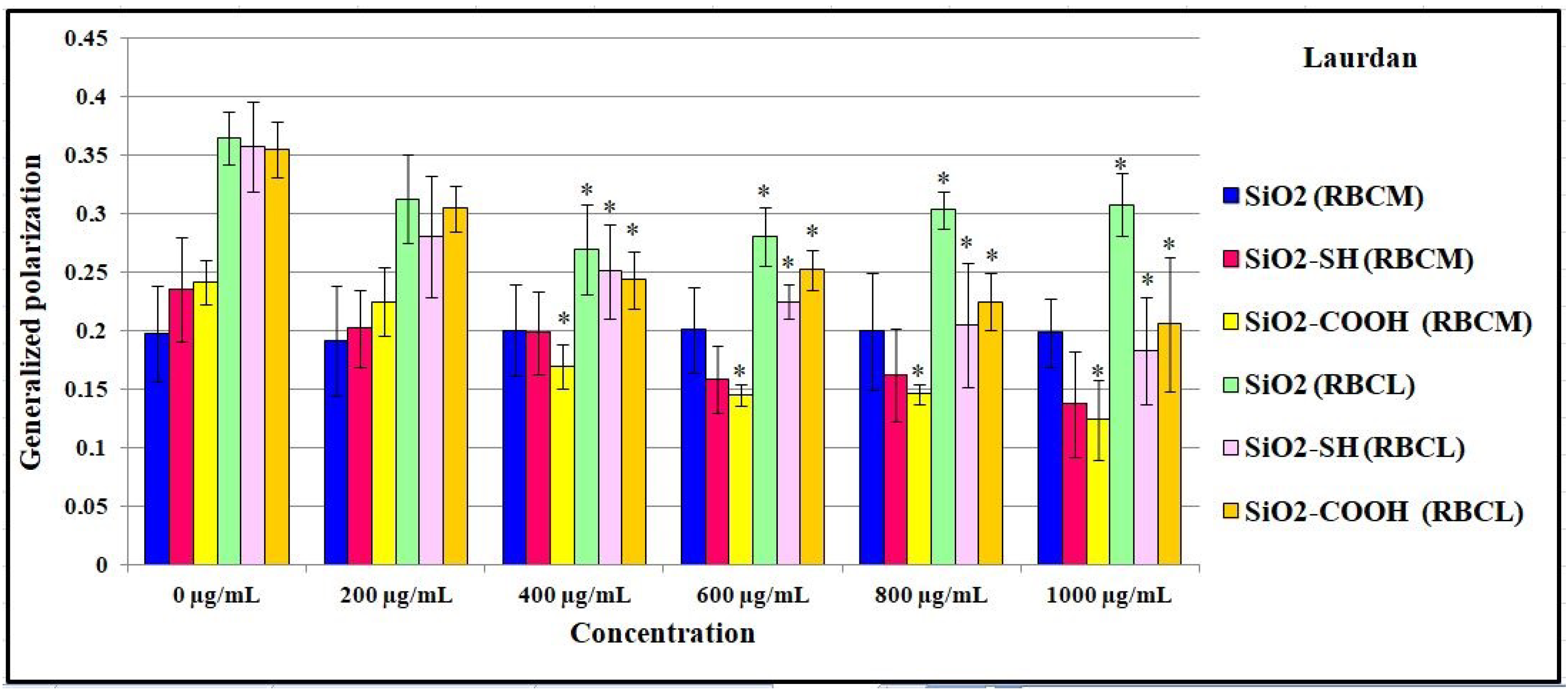
2.7. Influence of SiO2 Nanoparticles on the Physical Properties of the Membrane
3. Discussion
4. Materials and Methods
4.1. Silica Nanoparticles
4.2. Fourier Transform Infrared Spectroscopy
4.3. Particles Size Measurement
4.4. Electrokinetic Potential Measurement
4.5. Erythrocytes
4.5.1. Hemolysis of Erythrocytes
4.5.2. Osmotic Resistance Assay
4.5.3. Microscopic Study of Erythrocyte Shapes
4.5.4. Scanning Electron Microscopy (SEM)
4.5.5. Fluorimetric Studies of the Interaction of the Silica Nanoparticles with Biological and Model Membranes
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tan, Y.; Yu, D.; Feng, J.; You, H.; Bai, Y.; He, J.; Cao, H.; Che, Q.; Guo, J.; Su, Z. Toxicity evaluation of silica nanoparticles for delivery applications. Drug Deliv. Transl. Res. 2023, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Chauhan, S.; Manivasagam, G.; Kumar, P.; Ambasta, R.K. Cellular toxicity of mesoporous silica nanoparticle in SHSY5Y and BM-MNCs cell. Pharm. Nanotechnol. 2018, 6, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Napierska, D.; Thomassen, L.C.; Lison, D.; Martens, J.A.; Hoet, P.H. The nanosilica hazard: Another variable entity. Part. Fibre Toxicol. 2010, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Jafari, S.; Derakhshankhah, H.; Alaei, L.; Fattahi, A.; Varnamkhasti, B.S.; Saboury, A.A. Mesoporous silica nanoparticles for therapeutic/diagnostic applications. Biomed. Pharmacother. 2019, 109, 1100–1111. [Google Scholar] [CrossRef]
- Zhou, Y.; Quan, G.; Wu, Q.; Zhang, X.; Niu, B.; Wu, B.; Huang, Y.; Pan, X.; Wu, C. Mesoporous silica nanoparticles for drug and gene delivery. Acta Pharm. Sin. B 2018, 8, 165–177. [Google Scholar] [CrossRef]
- Ferenc, M.; Katir, N.; Milowska, K.; Bousmina, M.; Brahmi, Y.; Felczak, A.; Lisowska, K.; Bryszewska, M.; El Kadib, A. Impact of mesoporous silica surface functionalization on human serum albumin interaction, cytotoxicity and antibacterial activity. Microporous Mesoporous Mater. 2016, 231, 47–56. [Google Scholar] [CrossRef]
- Mukhopadhyay, S.; Veroniaina, H.; Chimombe, T.; Han, L.; Zhenghong, W.; Xiaole, Q. Synthesis and compatibility evaluation of versatile mesoporous silica nanoparticles with red blood cells: An overview. RSC Adv. 2019, 9, 35566–35578. [Google Scholar] [CrossRef]
- Repsold, L.; Joubert, A.M. Eryptosis: An erythrocyte’s suicidal type of cell death. BioMed Res. Int. 2018, 2018, 9405617. [Google Scholar] [CrossRef]
- Stasiuk, M.; Kijanka, G.; Kozubek, A. Zmiany kształtu erytrocytów i czynniki je wywołujące. Postępy Biochem. 2009, 55, 425–433. [Google Scholar]
- Spychalska, J. Membranopatie krwinek czerwonych—patogeneza, obraz kliniczny i diagnostyka. Hematologia 2012, 3, 81–119. [Google Scholar]
- Bruch, G.E.; Fernandes, L.F.; Bassi, B.L.; Alves, M.T.R.; Pereira, I.O.; Frézard, F.; Massensini, A.R. Liposomes for drug delivery in stroke. Brain Res. Bull. 2019, 152, 246–256. [Google Scholar] [CrossRef]
- Kozub, P. Otrzymywanie Oraz Charakterystyka Liposomów-Potencjalnych Nośników Leków w Celu Zwiększenia Skuteczności Terapii Fotodynamicznej. Doctoral Thesis, University of Silesia in Katowice, Katowice, Poland, 2015. [Google Scholar]
- Litwińczuk-Mammadova, A.; Cieślik-Boczula, K.; Rospenk, M. Budowa i funkcje układów białkowo-lipidowych. Wiadomości Chem. 2016, 70, 11–12. [Google Scholar]
- Bozzuto, G.; Molinari, A. Liposomes as nanomedical devices. Int. J. Nanomed. 2015, 10, 975. [Google Scholar] [CrossRef] [PubMed]
- Horakova, J.; Mikes, P.; Saman, A.; Svarcova, T.; Jencova, V.; Suchy, T.; Heczkova, B.; Jakubkova, S.; Jirousova, J.; Prochazkova, R. Comprehensive assessment of electrospun scaffolds hemocompatibility. Mater. Sci. Eng. C 2018, 82, 330–335. [Google Scholar] [CrossRef] [PubMed]
- Bhavsar, D.; Patel, V.; Sawant, K. Systematic investigation of in vitro and in vivo safety, toxicity and degradation of mesoporous silica nanoparticles synthesized using commercial sodium silicate. Microporous Mesoporous Mater. 2019, 284, 343–352. [Google Scholar] [CrossRef]
- Yildirim, A.; Ozgur, E.; Bayindir, M. Impact of mesoporous silica nanoparticle surface functionality on hemolytic activity, thrombogenicity and non-specific protein adsorption. J. Mater. Chem. B 2013, 1, 1909–1920. [Google Scholar] [CrossRef]
- Solarska-Ściuk, K.; Gajewska, A.; Glińska, S.; Studzian, M.; Michlewska, S.; Balcerzak, Ł.; Skolimowski, J.; Kolago, B.; Bartosz, G. Intracellular transport of nanodiamond particles in human endothelial and epithelial cells. Chem.-Biol. Interact. 2014, 219, 90–100. [Google Scholar] [CrossRef]
- Jędrzejczak, P.; Parus, A.; Balicki, S.; Kornaus, K.; Janczarek, M.; Wilk, K.A.; Jesionowski, T.; Ślosarczyk, A.; Klapiszewski, Ł. The influence of various forms of titanium dioxide on the performance of resultant cement composites with photocatalytic and antibacterial functions. Mater. Res. Bull. 2023, 160, 112139. [Google Scholar] [CrossRef]
- Feng, P.; Chang, H.; Liu, X.; Ye, S.; Shu, X.; Ran, Q. The significance of dispersion of nano-SiO2 on early age hydration of cement pastes. Mater. Des. 2020, 186, 108320. [Google Scholar] [CrossRef]
- Bernhardt, I.; Ellory, J.C. Red Cell Membrane Transport in Health and Disease; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Jouyandeh, M.; Jazani, O.M.; Navarchian, A.H.; Shabanian, M.; Vahabi, H.; Saeb, M.R. Bushy-surface hybrid nanoparticles for developing epoxy superadhesives. Appl. Surf. Sci. 2019, 479, 1148–1160. [Google Scholar] [CrossRef]
- Solarska-Ściuk, K.; Adach, K.; Cyboran-Mikołajczyk, S.; Bonarska-Kujawa, D.; Rusak, A.; Cwynar-Zając, Ł.; Machałowski, T.; Jesionowski, T.; Grzywacz, K.; Fijałkowski, M. Are biogenic and pyrogenic mesoporous SiO2 nanoparticles safe for normal cells? Molecules 2021, 26, 1427. [Google Scholar] [CrossRef]
- Ranjbarzadeh, R.; Moradikazerouni, A.; Bakhtiari, R.; Asadi, A.; Afrand, M. An experimental study on stability and thermal conductivity of water/silica nanofluid: Eco-friendly production of nanoparticles. J. Clean. Prod. 2019, 206, 1089–1100. [Google Scholar] [CrossRef]
- Bois, L.; Bonhommé, A.; Ribes, A.; Pais, B.; Raffin, G.; Tessier, F. Functionalized silica for heavy metal ions adsorption. Colloids Surf. A Physicochem. Eng. Asp. 2003, 221, 221–230. [Google Scholar] [CrossRef]
- Mureseanu, M.; Reiss, A.; Stefanescu, I.; David, E.; Parvulescu, V.; Renard, G.; Hulea, V. Modified SBA-15 mesoporous silica for heavy metal ions remediation. Chemosphere 2008, 73, 1499–1504. [Google Scholar] [CrossRef] [PubMed]
- Jesionowski, T.; Ciesielczyk, F.; Krysztafkiewicz, A. Influence of selected alkoxysilanes on dispersive properties and surface chemistry of spherical silica precipitated in emulsion media. Mater. Chem. Phys. 2010, 119, 65–74. [Google Scholar] [CrossRef]
- Patwardhan, S.V.; Emami, F.S.; Berry, R.J.; Jones, S.E.; Naik, R.R.; Deschaume, O.; Heinz, H.; Perry, C.C. Chemistry of aqueous silica nanoparticle surfaces and the mechanism of selective peptide adsorption. J. Am. Chem. Soc. 2012, 134, 6244–6256. [Google Scholar] [CrossRef] [PubMed]
- Klapiszewski, Ł.; Nowacka, M.; Milczarek, G.; Jesionowski, T. Physicochemical and electrokinetic properties of silica/lignin biocomposites. Carbohydr. Polym. 2013, 94, 345–355. [Google Scholar] [CrossRef]
- do Canto, A.M.; Robalo, J.R.; Santos, P.D.; Carvalho, A.J.P.; Ramalho, J.P.; Loura, L.M. Diphenylhexatriene membrane probes DPH and TMA-DPH: A comparative molecular dynamics simulation study. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2016, 1858, 2647–2661. [Google Scholar] [CrossRef]
- Wang, J.; Wang, W.; Kollman, P.A.; Case, D.A. Automatic atom type and bond type perception in molecular mechanical calculations. J. Mol. Graph. Model. 2006, 25, 247–260. [Google Scholar] [CrossRef]
- Li, Z.; Mu, Y.; Peng, C.; Lavin, M.F.; Shao, H.; Du, Z. Understanding the mechanisms of silica nanoparticles for nanomedicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2021, 13, e1658. [Google Scholar] [CrossRef]
- Chen, L.; Zhou, X.; He, C. Mesoporous silica nanoparticles for tissue-engineering applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2019, 11, e1573. [Google Scholar] [CrossRef] [PubMed]
- Ferenc, M.; Katir, N.; Miłowska, K.; Bousmina, M.; Majoral, J.P.; Bryszewska, M.; El Kadib, A. Haemolytic activity and cellular toxicity of SBA-15-type silicas: Elucidating the role of the mesostructure, surface functionality and linker length. J. Mater. Chem. B 2015, 3, 2714–2724. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Monaghan, S.A. Red blood cell/hemoglobin disorders. In Hematopathology; Elsevier: Amsterdam, The Netherlands, 2018; pp. 3–56. [Google Scholar]
- Elsabahy, M.; Wooley, K.L. Cytokines as biomarkers of nanoparticle immunotoxicity. Chem. Soc. Rev. 2013, 42, 5552–5576. [Google Scholar] [CrossRef] [PubMed]
- Labbaf, S.; Tsigkou, O.; Müller, K.H.; Stevens, M.M.; Porter, A.E.; Jones, J.R. Spherical bioactive glass particles and their interaction with human mesenchymal stem cells in vitro. Biomaterials 2011, 32, 1010–1018. [Google Scholar] [CrossRef]
- Zhai, W.; He, C.; Wu, L.; Zhou, Y.; Chen, H.; Chang, J.; Zhang, H. Degradation of hollow mesoporous silica nanoparticles in human umbilical vein endothelial cells. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 1397–1403. [Google Scholar] [CrossRef]
- Zhao, Y.; Vivero-Escoto, J.L.; Slowing, I.I.; Trewyn, B.G.; Lin, V.S. Capped mesoporous silica nanoparticles as stimuli-responsive controlled release systems for intracellular drug/gene delivery. Expert Opin. Drug Deliv. 2010, 7, 1013–1029. [Google Scholar] [CrossRef]
- Vivero-Escoto, J.L.; Slowing, I.I.; Trewyn, B.G.; Lin, V.S.Y. Mesoporous silica nanoparticles for intracellular controlled drug delivery. Small 2010, 6, 1952–1967. [Google Scholar] [CrossRef]
- Lin, Y.; Haynes, C. CH2-Hemolysis and size different MSN. J. Am. Chem. Soc. 2010, 132, 4834–4842. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.; Malugin, A.; Ghandehari, H. Impact of silica nanoparticle design on cellular toxicity and hemolytic activity. ACS Nano 2011, 5, 5717–5728. [Google Scholar] [CrossRef]
- Paula, A.J.; Martinez, D.S.T.; Araujo Júnior, R.T.; Souza Filho, A.G.; Alves, O.L. Suppression of the hemolytic effect of mesoporous silica nanoparticles after protein corona interaction: Independence of the surface microchemical environment. J. Braz. Chem. Soc. 2012, 23, 1807–1814. [Google Scholar] [CrossRef]
- Jiang, L.; Yu, Y.; Li, Y.; Yu, Y.; Duan, J.; Zou, Y.; Li, Q.; Sun, Z. Oxidative damage and energy metabolism disorder contribute to the hemolytic effect of amorphous silica nanoparticles. Nanoscale Res. Lett. 2016, 11, 57. [Google Scholar] [CrossRef]
- Shinto, H.; Fukasawa, T.; Yoshisue, K.; Tezuka, M.; Orita, M. Cell membrane disruption induced by amorphous silica nanoparticles in erythrocytes, lymphocytes, malignant melanocytes, and macrophages. Adv. Powder Technol. 2014, 25, 1872–1881. [Google Scholar] [CrossRef]
- Wei, X.; Jiang, W.; Yu, J.; Ding, L.; Hu, J.; Jiang, G. Effects of SiO2 nanoparticles on phospholipid membrane integrity and fluidity. J. Hazard. Mater. 2015, 287, 217–224. [Google Scholar] [CrossRef]
- Shin, T.H.; Ketebo, A.A.; Lee, D.Y.; Lee, S.; Kang, S.H.; Basith, S.; Manavalan, B.; Kwon, D.H.; Park, S.; Lee, G. Decrease in membrane fluidity and traction force induced by silica-coated magnetic nanoparticles. J. Nanobiotechnol. 2021, 19, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Pruchnik, H.; Włoch, A.; Bonarska-Kujawa, D.; Kleszczyńska, H. An In Vitro study of the effect of cytotoxic triorganotin dimethylaminophenylazobenzoate complexes on red blood cells. J. Membr. Biol. 2018, 251, 735–745. [Google Scholar] [CrossRef]
- Cyboran, S.; Oszmiański, J.; Kleszczyńska, H. Interaction between plant polyphenols and the erythrocyte membrane. Cell. Mol. Biol. Lett. 2012, 17, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Dodge, J.T.; Mitchell, C.; Hanahan, D.J. The preparation and chemical characteristics of hemoglobin-free ghosts of human erythrocytes. Arch. Biochem. Biophys. 1963, 100, 119–130. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Maddy, A.; Dunn, M.; Kelly, P. The characterisation of membrane proteins by centrifugation and gel electrophoresis a comparison of proteins prepared by different methods. Biochim. Et Biophys. Acta (BBA)-Biomembr. 1972, 288, 263–276. [Google Scholar] [CrossRef]
- Bonarska-Kujawa, D.; Pruchnik, H.; Oszmiański, J.; Sarapuk, J.; Kleszczyńska, H. Changes caused by fruit extracts in the lipid phase of biological and model membranes. Food Biophys. 2011, 6, 58–67. [Google Scholar] [CrossRef]
- Cyboran-Mikołajczyk, S.; Bonarska-Kujawa, D.; Męczarska, K.; Krawczyk-ebek, A.; Kostrzewa-Susłow, E. Novel O-Methylglucoside Derivatives of Flavanone in Interaction with Model Membrane and Transferrin. Membranes 2022, 12, 978. [Google Scholar] [CrossRef] [PubMed]
| (g/mL) | ||
|---|---|---|
| Time incubation | 2 h | 24 h |
| Control | 0.69% NaCl | 0.66% NaCl |
| 200 g/mL | 0.69% NaCl | 0.68% NaCl |
| 500 g/mL | 0.71% NaCl | 0.73% NaCl |
| 200 g/mL | 0.66% NaCl | 0.66% NaCl |
| 500 g/mL | 0.67% NaCl | 0.68% NaCl |
| 200 g/mL | 0.69% NaCl | 0.64% NaCl |
| 500 g/mL | 0.70% NaCl | 0.66% NaCl |
| Change of Erythrocyte’s Shape | ||||
|---|---|---|---|---|
| Time Incubation (2 h) | Discocytes | Echinocytes | Stomatocytes | Lacrimocytes |
| Control | 95.18 ± 0.011% | 3.09 ± 0.013% | 1.23 ± 0.017% | - |
| 200 g/mL | 8.13 ± 0.015% | 74.21 ± 0.015% | 17.15 ± 0.014% | - |
| 500 g/mL | 5.11 ± 0.014% | 76.14 ± 0.011% | 19.13 ± 0.016% | - |
| 200 g/mL | 7.18 ± 0.020% | 65.23 ± 0.017% | 26.11 ± 0.011% | 2.24 ± 0.013% |
| 500 g/mL | 5.22 ± 0.011% | 60.33 ± 0.014% | 28.31 ± 0.013% | 5.14 ± 0.012% |
| 200 g/mL | 12.12 ± 0.011% | 52.14 ± 0.015% | 23.11 ± 0.013% | 13.32 ± 0.015% |
| 500 g/mL | 2.32 ± 0.011% | 55.13 ± 0.015% | 25.21 ± 0.015% | 18.21 ± 0.0143% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Solarska-Ściuk, K.; Męczarska, K.; Jencova, V.; Jędrzejczak, P.; Klapiszewski, Ł.; Jaworska, A.; Hryć, M.; Bonarska-Kujawa, D. Effect of Non-Modified as Well as Surface-Modified SiO2 Nanoparticles on Red Blood Cells, Biological and Model Membranes. Int. J. Mol. Sci. 2023, 24, 11760. https://doi.org/10.3390/ijms241411760
Solarska-Ściuk K, Męczarska K, Jencova V, Jędrzejczak P, Klapiszewski Ł, Jaworska A, Hryć M, Bonarska-Kujawa D. Effect of Non-Modified as Well as Surface-Modified SiO2 Nanoparticles on Red Blood Cells, Biological and Model Membranes. International Journal of Molecular Sciences. 2023; 24(14):11760. https://doi.org/10.3390/ijms241411760
Chicago/Turabian StyleSolarska-Ściuk, Katarzyna, Katarzyna Męczarska, Vera Jencova, Patryk Jędrzejczak, Łukasz Klapiszewski, Aleksandra Jaworska, Monika Hryć, and Dorota Bonarska-Kujawa. 2023. "Effect of Non-Modified as Well as Surface-Modified SiO2 Nanoparticles on Red Blood Cells, Biological and Model Membranes" International Journal of Molecular Sciences 24, no. 14: 11760. https://doi.org/10.3390/ijms241411760
APA StyleSolarska-Ściuk, K., Męczarska, K., Jencova, V., Jędrzejczak, P., Klapiszewski, Ł., Jaworska, A., Hryć, M., & Bonarska-Kujawa, D. (2023). Effect of Non-Modified as Well as Surface-Modified SiO2 Nanoparticles on Red Blood Cells, Biological and Model Membranes. International Journal of Molecular Sciences, 24(14), 11760. https://doi.org/10.3390/ijms241411760






