Folate Receptor Targeted Photodynamic Therapy: A Novel Way to Stimulate Anti-Tumor Immune Response in Intraperitoneal Ovarian Cancer
Abstract
1. Introduction
2. Results
2.1. In Vitro Results
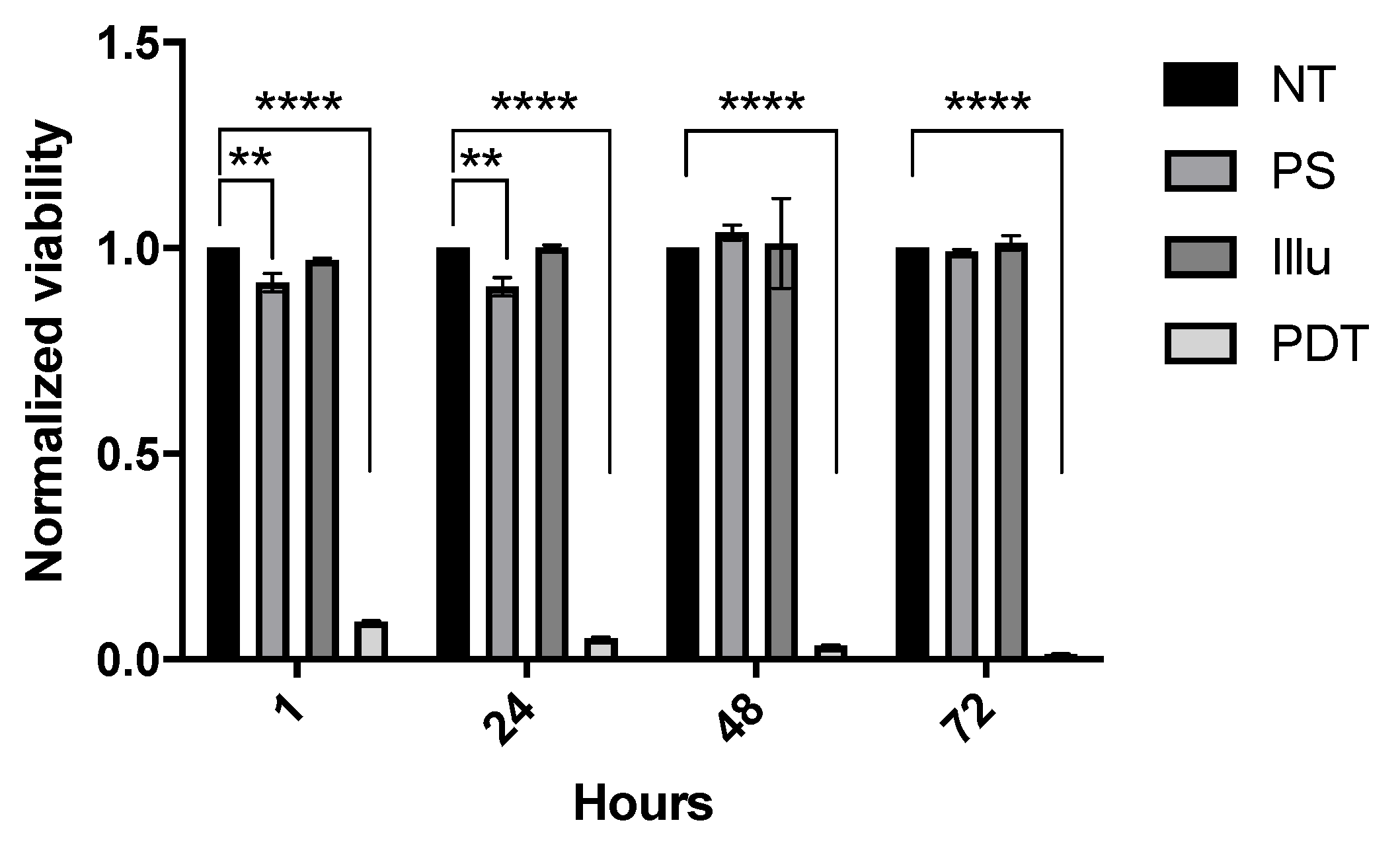
2.1.1. Evaluation of OVCAR3-Luc Viability upon PDT
2.2. In Vivo Results
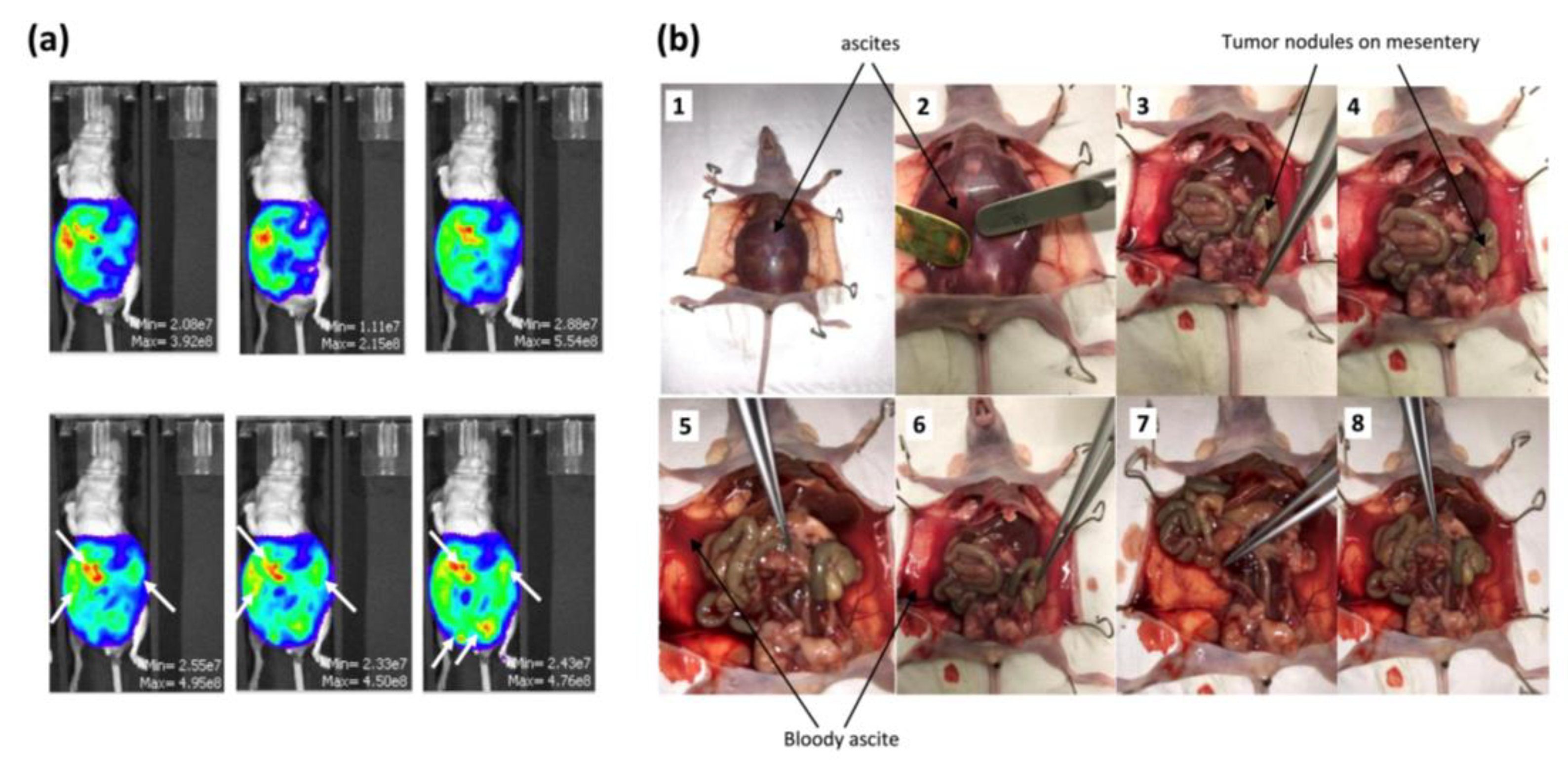
2.2.1. Development of a Humanized In Vivo Mouse Model of Intraperitoneal Ovarian Cancer
2.2.2. Before the Reconstitution of Immune System
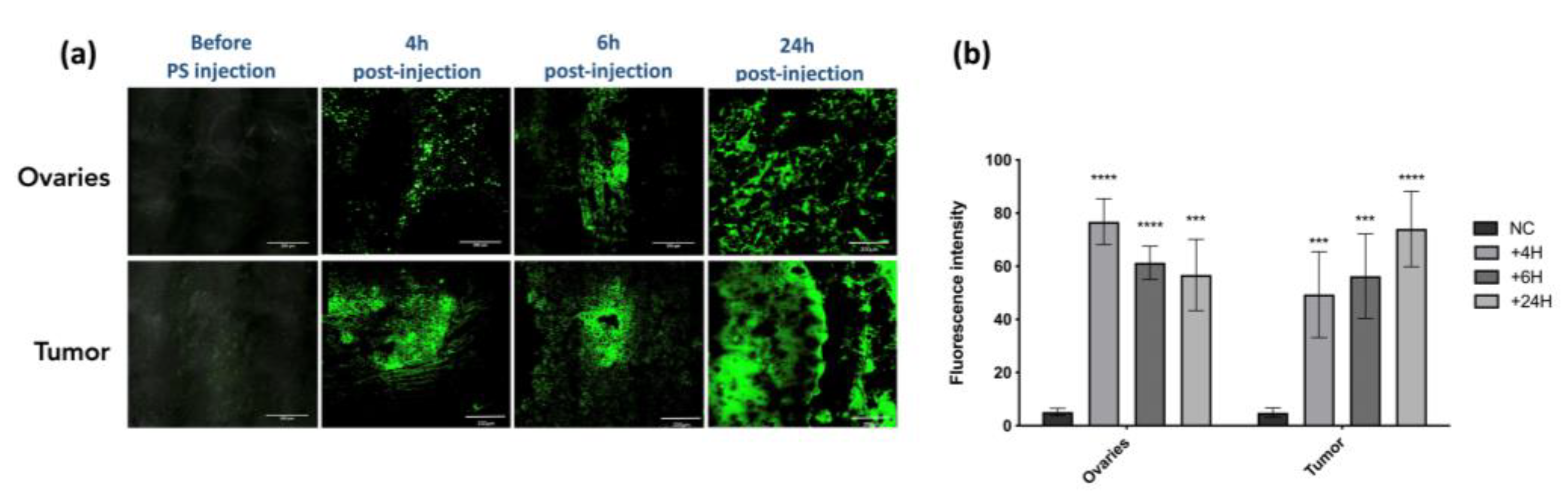
- Evaluation of the PS Uptake within Targeting Tissue
- Determination of the Fractionized Illumination Scheme
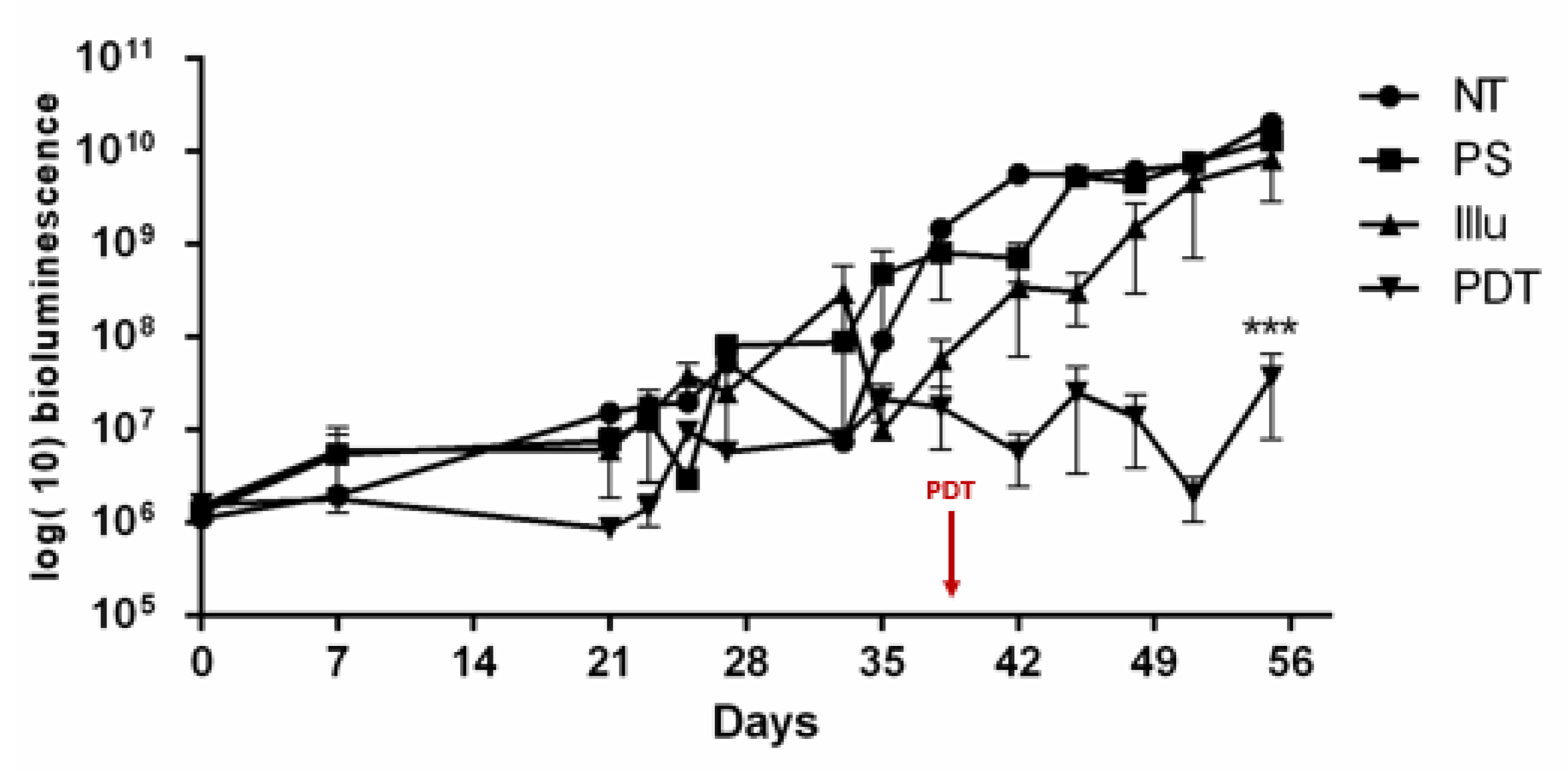
- Evaluation of PDT Efficacy
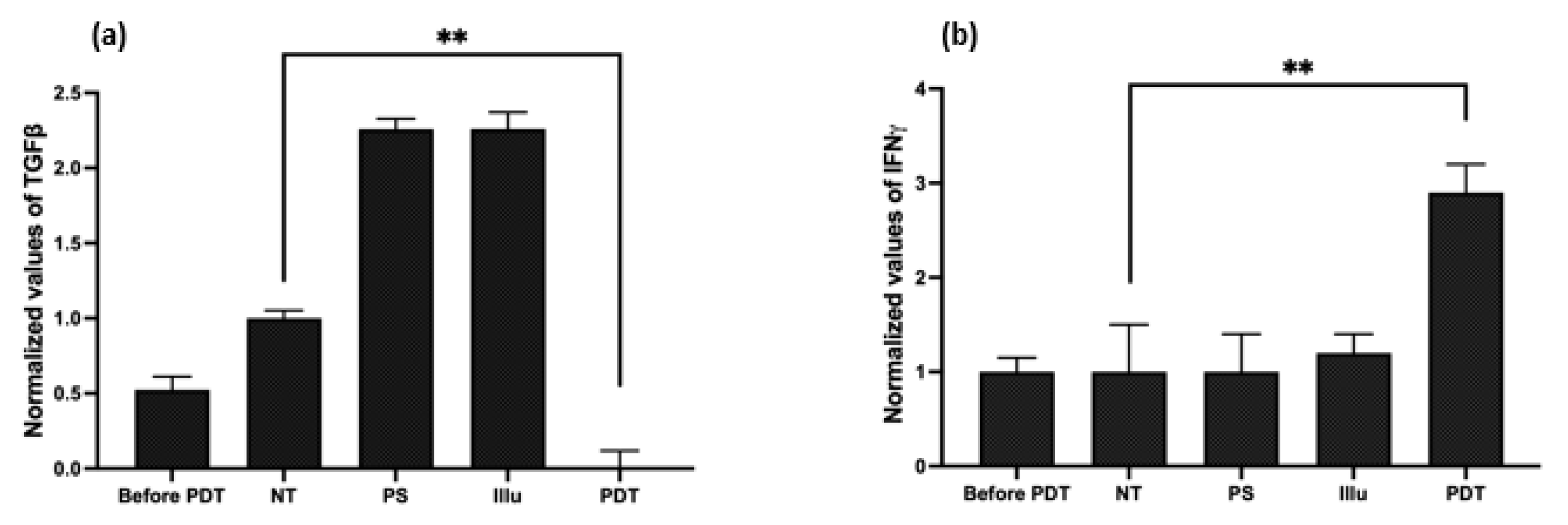
- Evaluation of the Cytokine Release upon PDT
2.2.3. After the Reconstitution of Immune System
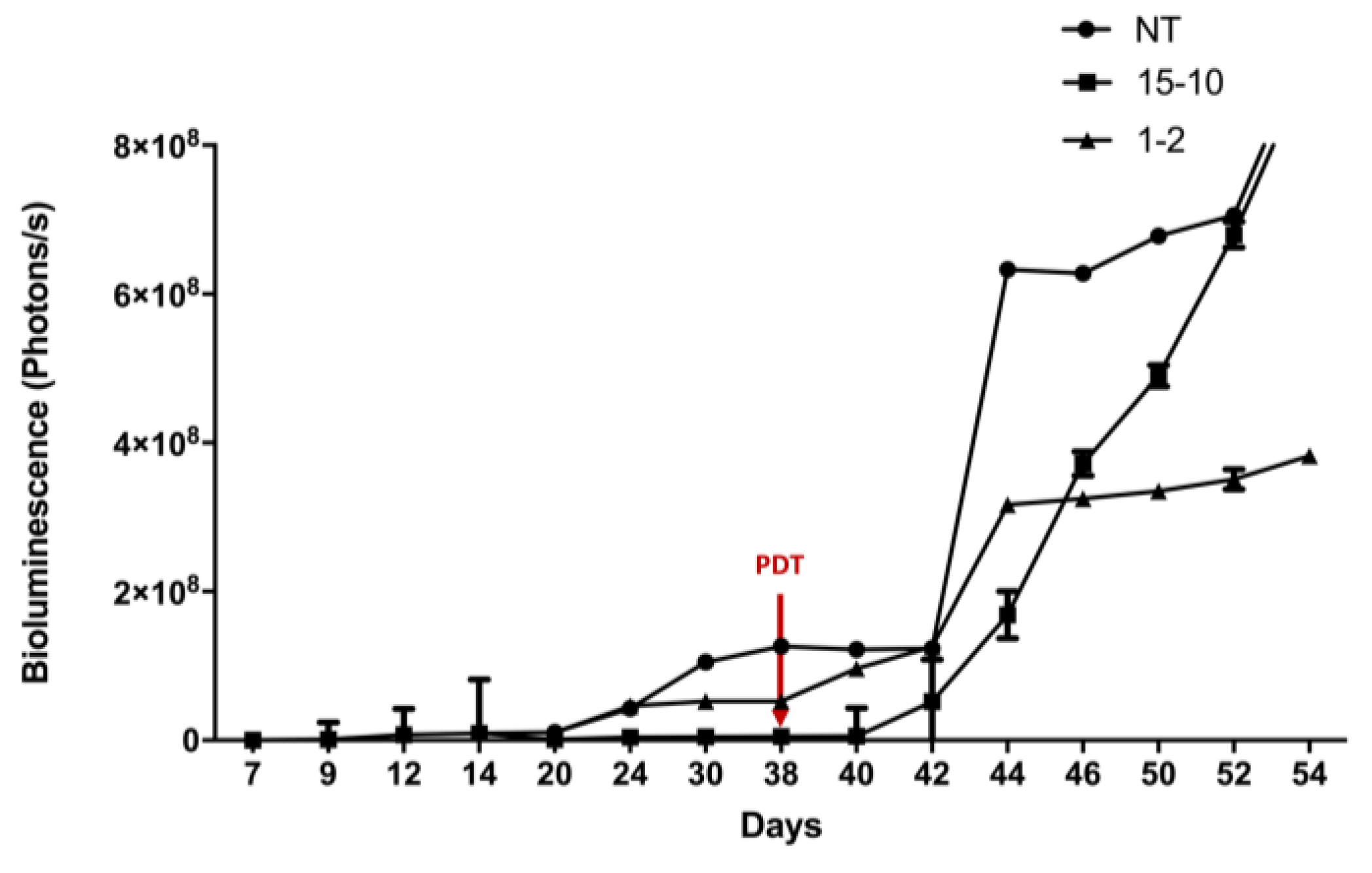
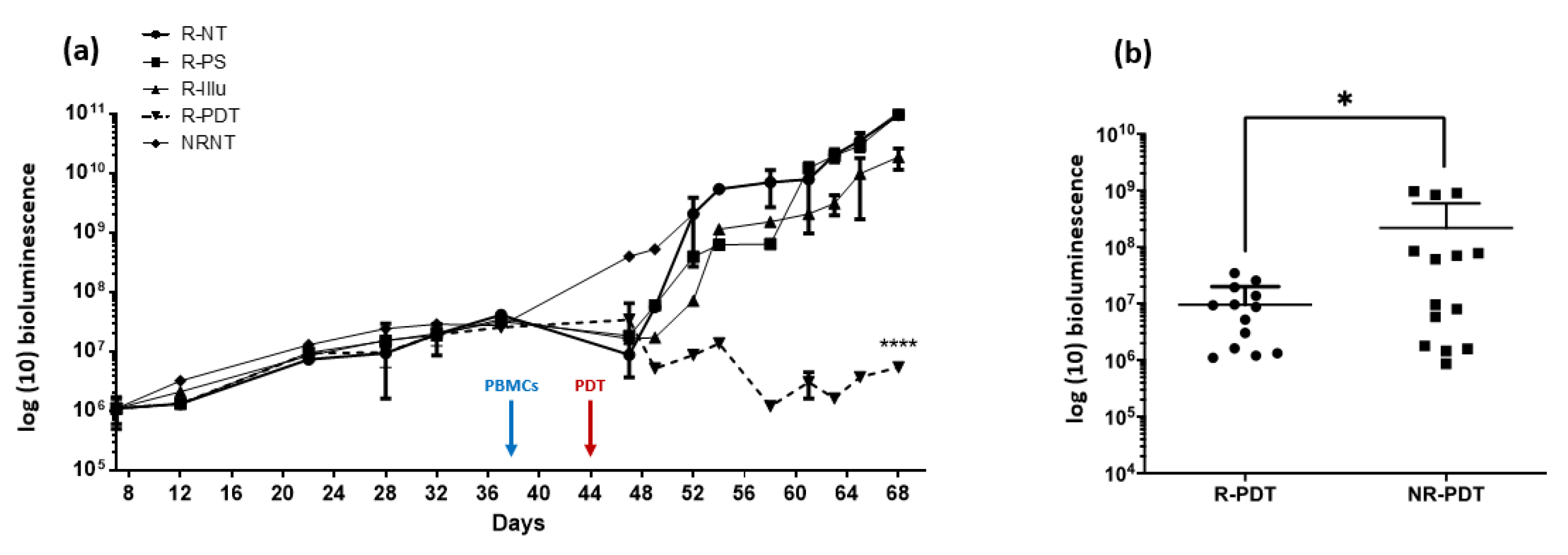
- Evaluation of PDT Efficacy
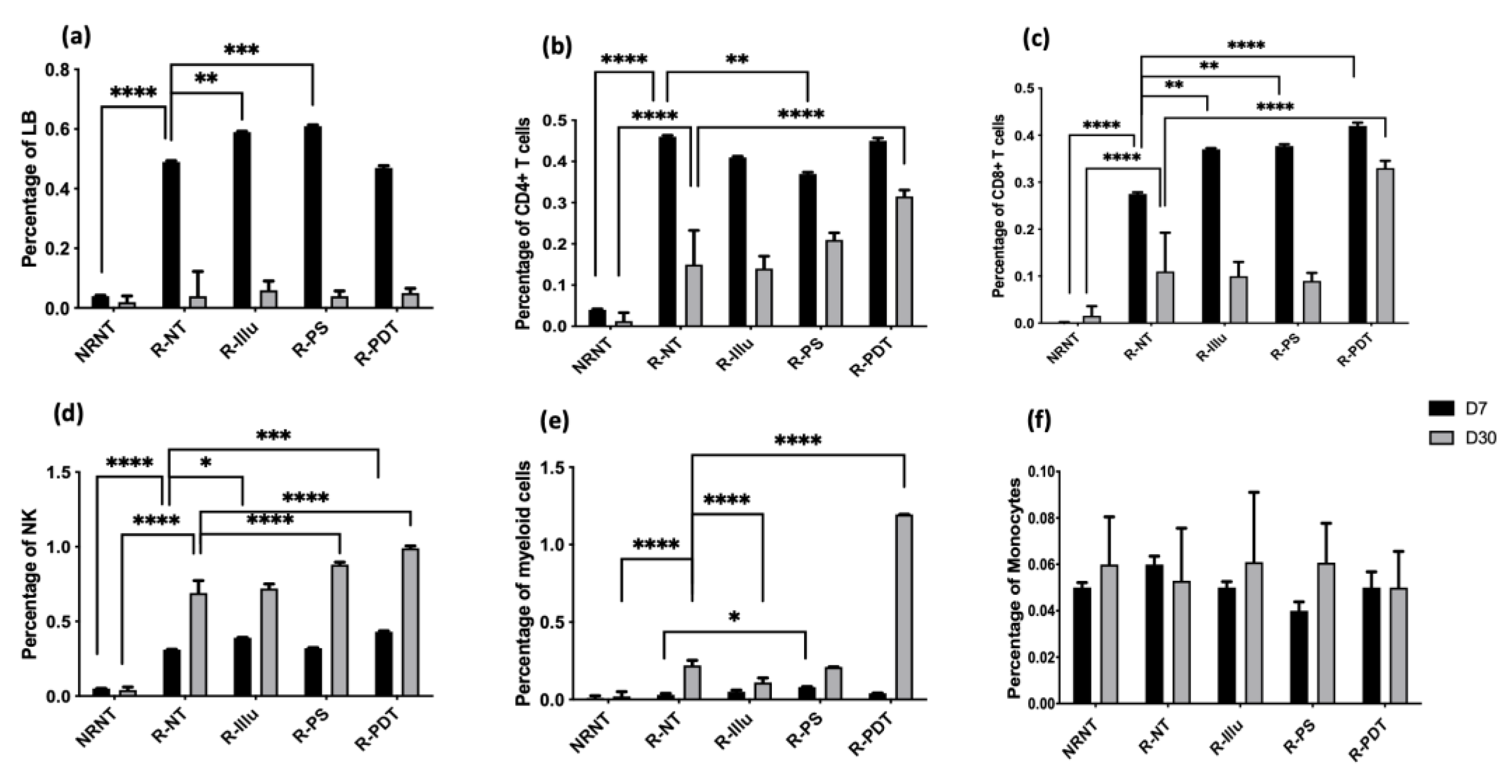
- Evaluation of the Immune Cell Response upon PDT
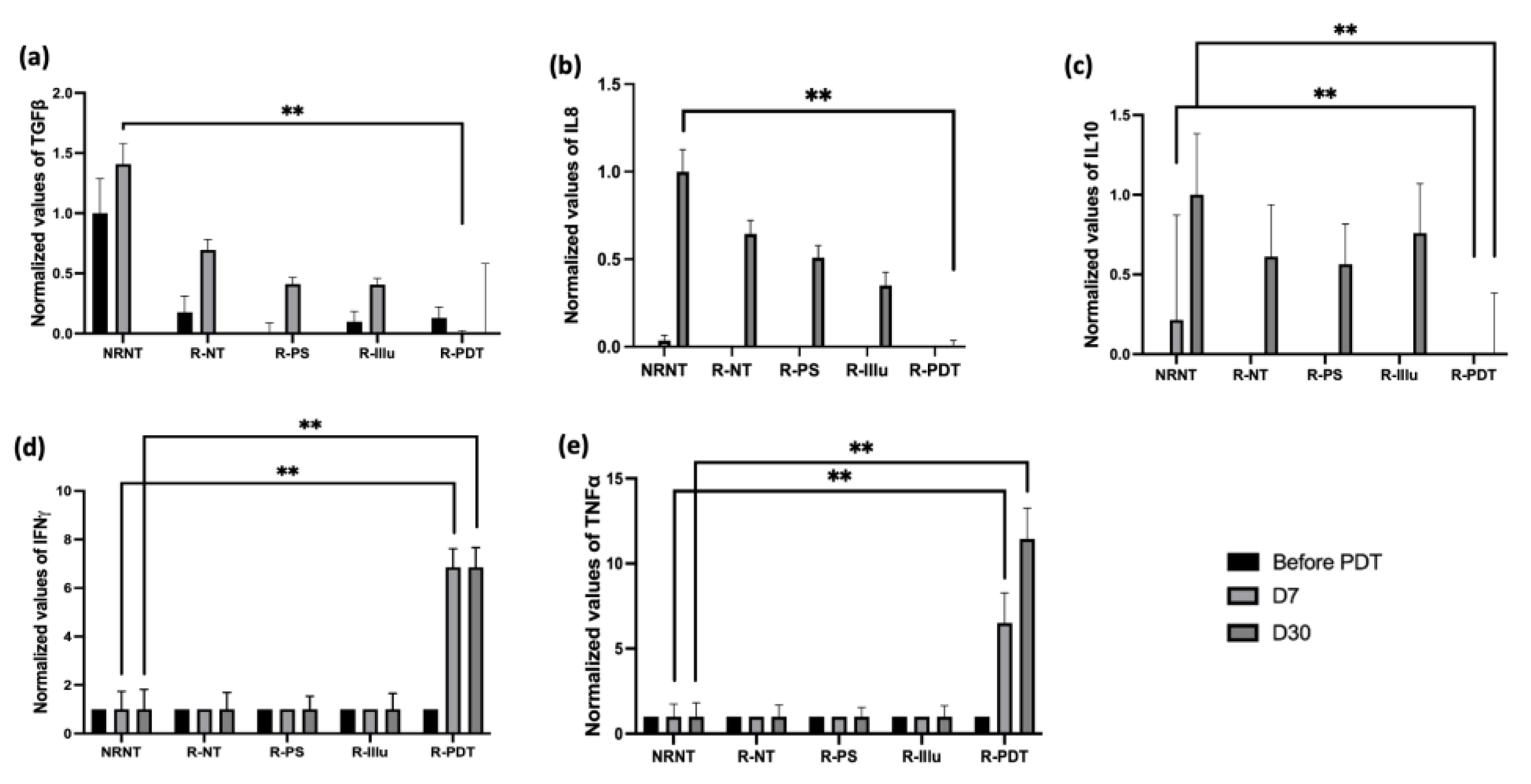
- Evaluation of the Cytokines’ Release upon PDT
3. Discussion
4. Materials and Methods
4.1. In Vitro
4.1.1. Cell Culture
4.1.2. Luciferase Transfection
4.1.3. Luciferase Assay
4.1.4. Photosensitizer
4.1.5. Photodynamic Therapy In Vitro
4.1.6. Viability Assays
4.2. In Vivo
4.2.1. Animals
4.2.2. Development of a Humanized Mouse Mode of Intraperitoneal Ovarian Cancer
4.2.3. Before the Reconstitution of Immune System (Non-Reconstituted (NR) Mice)
- PS Uptake
- Determination of the Fractionized Illumination Scheme
- Photodynamic Therapy Protocol
- ELISA
4.2.4. After the Reconstitution of Immune System (Reconstituted (R) Mice)
- Reconstitution of Immune System
- Photodynamic Therapy Protocol
- Flow Cytometric Analysis
- High Sensitivity Human Cytokine Magnetic Bead Kit
4.3. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Turubanova, V.D.; Balalaeva, I.V.; Mishchenko, T.A.; Catanzaro, E.; Alzeibak, R.; Peskova, N.N.; Efimova, I.; Bachert, C.; Mitroshina, E.V.; Krysko, O.; et al. Immunogenic Cell Death Induced by a New Photodynamic Therapy Based on Photosens and Photodithazine. J. Immunother. Cancer 2019, 7, 350. [Google Scholar] [CrossRef] [PubMed]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular Mechanisms of Cell Death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef] [PubMed]
- Alzeibak, R.; Mishchenko, T.A.; Shilyagina, N.Y.; Balalaeva, I.V.; Vedunova, M.V.; Krysko, D.V. Targeting Immunogenic Cancer Cell Death by Photodynamic Therapy: Past, Present and Future. J. Immunother. Cancer 2021, 9, e001926. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Morse, M.A.; Gwin, W.R.; Mitchell, D.A. Vaccine Therapies for Cancer: Then and Now. Target. Oncol. 2021, 16, 121–152. [Google Scholar] [CrossRef]
- Ahmed, A.; Tait, S.W.G. Targeting Immunogenic Cell Death in Cancer. Mol. Oncol. 2020, 14, 2994–3006. [Google Scholar] [CrossRef]
- Castellani, A.; Pace, G.P.; Concioli, M. Photodynamic Effect of Haematoporphyrin on Blood Microcirculation. J. Pathol. Bacteriol. 1963, 86, 99–102. [Google Scholar] [CrossRef]
- Agarwal, M.L.; Clay, M.E.; Harvey, E.J.; Evans, H.H.; Antunez, A.R.; Oleinick, N.L. Photodynamic Therapy Induces Rapid Cell Death by Apoptosis in L5178Y Mouse Lymphoma Cells. Cancer Res. 1991, 51, 5993–5996. [Google Scholar]
- Luo, W.; Liu, R.-S.; Zhu, J.-G.; Li, Y.-C.; Liu, H.-C. Subcellular Location and Photodynamic Therapeutic Effect of Chlorin E6 in the Human Tongue Squamous Cell Cancer Tca8113 Cell Line. Oncol. Lett. 2015, 9, 551–556. [Google Scholar] [CrossRef]
- Hwang, H.S.; Shin, H.; Han, J.; Na, K. Combination of Photodynamic Therapy (PDT) and Anti-Tumor Immunity in Cancer Therapy. J. Pharm. Investig. 2018, 48, 143–151. [Google Scholar] [CrossRef]
- Michy, T.; Massias, T.; Bernard, C.; Vanwonterghem, L.; Henry, M.; Guidetti, M.; Royal, G.; Coll, J.-L.; Texier, I.; Josserand, V.; et al. Verteporfin-Loaded Lipid Nanoparticles Improve Ovarian Cancer Photodynamic Therapy In Vitro and In Vivo. Cancers 2019, 11, 1760. [Google Scholar] [CrossRef] [PubMed]
- Sitnik, T.M.; Hampton, J.A.; Henderson, B.W. Reduction of Tumour Oxygenation during and after Photodynamic Therapy in Vivo: Effects of Fluence Rate. Br. J. Cancer 1998, 77, 1386–1394. [Google Scholar] [CrossRef] [PubMed]
- Shams, M.; Owczarczak, B.; Manderscheid-Kern, P.; Bellnier, D.A.; Gollnick, S.O. Development of Photodynamic Therapy Regimens That Control Primary Tumor Growth and Inhibit Secondary Disease. Cancer Immunol. Immunother. 2015, 64, 287–297. [Google Scholar] [CrossRef]
- Mroz, P.; Hashmi, J.T.; Huang, Y.-Y.; Lange, N.; Hamblin, M.R. Stimulation of Anti-Tumor Immunity by Photodynamic Therapy. Expert. Rev. Clin. Immunol. 2011, 7, 75–91. [Google Scholar] [CrossRef]
- Henderson, B.W.; Busch, T.M.; Vaughan, L.A.; Frawley, N.P.; Babich, D.; Sosa, T.A.; Zollo, J.D.; Dee, A.S.; Cooper, M.T.; Bellnier, D.A.; et al. Photofrin Photodynamic Therapy Can Significantly Deplete or Preserve Oxygenation in Human Basal Cell Carcinomas during Treatment, Depending on Fluence Rate. Cancer Res. 2000, 60, 525–529. [Google Scholar] [PubMed]
- Kabingu, E.; Vaughan, L.; Owczarczak, B.; Ramsey, K.D.; Gollnick, S.O. CD8+ T Cell-Mediated Control of Distant Tumours Following Local Photodynamic Therapy Is Independent of CD4+ T Cells and Dependent on Natural Killer Cells. Br. J. Cancer 2007, 96, 1839–1848. [Google Scholar] [CrossRef] [PubMed]
- Henderson, B.W.; Gollnick, S.O.; Snyder, J.W.; Busch, T.M.; Kousis, P.C.; Cheney, R.T.; Morgan, J. Choice of Oxygen-Conserving Treatment Regimen Determines the Inflammatory Response and Outcome of Photodynamic Therapy of Tumors. Cancer Res. 2004, 64, 2120–2126. [Google Scholar] [CrossRef]
- Azaïs, H.; Vignion-Dewalle, A.-S.; Carrier, M.; Augustin, J.; Da Maïa, E.; Penel, A.; Belghiti, J.; Nikpayam, M.; Gonthier, C.; Ziane, L.; et al. Microscopic Peritoneal Residual Disease after Complete Macroscopic Cytoreductive Surgery for Advanced High Grade Serous Ovarian Cancer. J. Clin. Med. 2020, 10, 41. [Google Scholar] [CrossRef]
- Reid, B.M.; Permuth, J.B.; Sellers, T.A. Epidemiology of Ovarian Cancer: A Review. Cancer Biol. Med. 2017, 14, 9–32. [Google Scholar] [CrossRef]
- Gaona-Luviano, P.; Medina-Gaona, L.A.; Magaña-Pérez, K. Epidemiology of Ovarian Cancer. Chin. Clin. Oncol. 2020, 9, 47. [Google Scholar] [CrossRef]
- Kroeger, P.T.; Drapkin, R. Pathogenesis and Heterogeneity of Ovarian Cancer. Curr. Opin. Obstet. Gynecol. 2017, 29, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Della Pepa, C.; Tonini, G.; Pisano, C.; Di Napoli, M.; Cecere, S.C.; Tambaro, R.; Facchini, G.; Pignata, S. Ovarian Cancer Standard of Care: Are There Real Alternatives? Chin. J. Cancer 2015, 34, 17–27. [Google Scholar] [CrossRef] [PubMed]
- Kossaï, M.; Leary, A.; Scoazec, J.-Y.; Genestie, C. Ovarian Cancer: A Heterogeneous Disease. Pathobiology 2018, 85, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Azaïs, H.; Estevez, J.P.; Foucher, P.; Kerbage, Y.; Mordon, S.; Collinet, P. Dealing with Microscopic Peritoneal Metastases of Epithelial Ovarian Cancer. A Surgical Challenge. Surg. Oncol. 2017, 26, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Sindelar, W.F.; DeLaney, T.F.; Tochner, Z.; Thomas, G.F.; Dachoswki, L.J.; Smith, P.D.; Friauf, W.S.; Cole, J.W.; Glatstein, E. Technique of Photodynamic Therapy for Disseminated Intraperitoneal Malignant Neoplasms. Phase I Study. Arch. Surg. 1991, 126, 318–324. [Google Scholar] [CrossRef]
- Delaney, T.F.; Sindelar, W.F.; Tochner, Z.; Smith, P.D.; Friauf, W.S.; Thomas, G.; Dachowski, L.; Cole, J.W.; Steinberg, S.M.; Glatstein, E. Phase I Study of Debulking Surgery and Photodynamic Therapy for Disseminated Intraperitoneal Tumors. Int. J. Radiat. Oncol. Biol. Phys. 1993, 25, 445–457. [Google Scholar] [CrossRef]
- Hahn, S.M.; Fraker, D.L.; Mick, R.; Metz, J.; Busch, T.M.; Smith, D.; Zhu, T.; Rodriguez, C.; Dimofte, A.; Spitz, F.; et al. A Phase II Trial of Intraperitoneal Photodynamic Therapy for Patients with Peritoneal Carcinomatosis and Sarcomatosis. Clin. Cancer Res. 2006, 12, 2517–2525. [Google Scholar] [CrossRef]
- Cheung, A.; Bax, H.J.; Josephs, D.H.; Ilieva, K.M.; Pellizzari, G.; Opzoomer, J.; Bloomfield, J.; Fittall, M.; Grigoriadis, A.; Figini, M.; et al. Targeting Folate Receptor Alpha for Cancer Treatment. Oncotarget 2016, 7, 52553–52574. [Google Scholar] [CrossRef]
- Moinard, M.; Augustin, J.; Carrier, M.; Da Maïa, E.; Penel, A.; Belghiti, J.; Nikpayam, M.; Gonthier, C.; Canlorbe, G.; Acherar, S.; et al. Residual Microscopic Peritoneal Metastases after Macroscopic Complete Cytoreductive Surgery for Advanced High-Grade Serous Ovarian Carcinoma: A Target for Folate Receptor Targeted Photodynamic Therapy? Pharmaceuticals 2022, 15, 1034. [Google Scholar] [CrossRef]
- Cortez, A.J.; Tudrej, P.; Kujawa, K.A.; Lisowska, K.M. Advances in Ovarian Cancer Therapy. Cancer Chemother. Pharmacol. 2018, 81, 17–38. [Google Scholar] [CrossRef]
- Zou, S.; Tong, Q.; Liu, B.; Huang, W.; Tian, Y.; Fu, X. Targeting STAT3 in Cancer Immunotherapy. Mol. Cancer 2020, 19, 145. [Google Scholar] [CrossRef] [PubMed]
- Quilbe, A.; Moralès, O.; Baydoun, M.; Kumar, A.; Mustapha, R.; Murakami, T.; Leroux, B.; de Schutter, C.; Thecua, E.; Ziane, L.; et al. An Efficient Photodynamic Therapy Treatment for Human Pancreatic Adenocarcinoma. J. Clin. Med. 2020, 9, 192. [Google Scholar] [CrossRef] [PubMed]
- Azaïs, H.; Schmitt, C.; Tardivel, M.; Kerdraon, O.; Stallivieri, A.; Frochot, C.; Betrouni, N.; Collinet, P.; Mordon, S. Assessment of the Specificity of a New Folate-Targeted Photosensitizer for Peritoneal Metastasis of Epithelial Ovarian Cancer to Enable Intraperitoneal Photodynamic Therapy. A Preclinical Study. Photodiagnosis Photodyn. Ther. 2016, 13, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Baydoun, M.; Moralès, O.; Frochot, C.; Ludovic, C.; Leroux, B.; Thecua, E.; Ziane, L.; Grabarz, A.; Kumar, A.; de Schutter, C.; et al. Photodynamic Therapy Using a New Folate Receptor-Targeted Photosensitizer on Peritoneal Ovarian Cancer Cells Induces the Release of Extracellular Vesicles with Immunoactivating Properties. J. Clin. Med. 2020, 9, 1185. [Google Scholar] [CrossRef]
- Burger, R.A.; Fleming, G.F.; Mannel, R.S.; Greer, B.E.; Liang, S.X. Incorporation of Bevacizumab in the Primary Treatment of Ovarian Cancer. N. Engl. J. Med. 2011, 11, 2473–2483. [Google Scholar] [CrossRef] [PubMed]
- Perren, T.J.; Swart, A.M.; Pfisterer, J.; Ledermann, J.A.; Pujade-Lauraine, E.; Kristensen, G.; Carey, M.S.; Beale, P.; Cervantes, A.; Kurzeder, C.; et al. A Phase 3 Trial of Bevacizumab in Ovarian Cancer. N. Engl. J. Med. 2011, 365, 2484–2496. [Google Scholar] [CrossRef]
- Gulia, S.; Kannan, S.; Ghosh, J.; Rath, S.; Maheshwari, A.; Gupta, S. Maintenance Therapy with a Poly(ADP-Ribose) Polymerase Inhibitor in Patients with Newly Diagnosed Advanced Epithelial Ovarian Cancer: Individual Patient Data and Trial-Level Meta-Analysis. ESMO Open. 2022, 7, 100558. [Google Scholar] [CrossRef]
- Medina-Castro, J.M.; Ruiz-DeLeón, A. Role of Hyperthermic Intraperitoneal Chemotherapy in Ovarian Cancer. Chin. Clin. Oncol. 2020, 9, 44. [Google Scholar] [CrossRef]
- Amate, P.; Huchon, C.; Dessapt, A.L.; Bensaid, C.; Medioni, J.; Le Frère Belda, M.-A.; Bats, A.-S.; Lécuru, F.R. Ovarian Cancer: Sites of Recurrence. Int. J. Gynecol. Cancer 2013, 23, 1590–1596. [Google Scholar] [CrossRef]
- Thécua, E.; Ziane, L.; Grolez, G.P.; Fagart, A.; Kumar, A.; Leroux, B.; Baert, G.; Deleporte, P.; Vermandel, M.; Vignion-Dewalle, A.-S.; et al. A Warp-Knitted Light-Emitting Fabric-Based Device for In Vitro Photodynamic Therapy: Description, Characterization, and Application on Human Cancer Cell Lines. Cancers 2021, 13, 4109. [Google Scholar] [CrossRef]
- Rezniczek, G.A.; Buggisch, J.; Sobilo, J.; Launay, A.; Lerondel, S.; Le Pape, A.; Ouaissi, M.; Göhler, D.; Senkal, M.; Giger-Pabst, U.; et al. Establishment of a Mouse Ovarian Cancer and Peritoneal Metastasis Model to Study Intraperitoneal Chemotherapy. Cancers 2020, 12, 3818. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, E. Ovarian Cancer Development and Metastasis. Am. J. Pathol. 2010, 177, 1053–1064. [Google Scholar] [CrossRef] [PubMed]
- Ogawara, K.-I.; Higaki, K. Nanoparticle-Based Photodynamic Therapy: Current Status and Future Application to Improve Outcomes of Cancer Treatment. Chem. Pharm. Bull. 2017, 65, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Sai, D.L.; Lee, J.; Nguyen, D.L.; Kim, Y.-P. Tailoring Photosensitive ROS for Advanced Photodynamic Therapy. Exp. Mol. Med. 2021, 53, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Curnow, A.; Haller, J.C.; Bown, S.G. Oxygen Monitoring during 5-Aminolaevulinic Acid Induced Photodynamic Therapy in Normal Rat Colon. Comparison of Continuous and Fractionated Light Regimes. J. Photochem. Photobiol. B. 2000, 58, 149–155. [Google Scholar] [CrossRef]
- Klimenko, V.V.; Knyazev, N.A.; Moiseenko, F.V.; Rusanov, A.A.; Bogdanov, A.A.; Dubina, M.V. Pulse Mode of Laser Photodynamic Treatment Induced Cell Apoptosis. Photodiagnosis Photodyn. Ther. 2016, 13, 101–107. [Google Scholar] [CrossRef]
- Coutier, S.; Bezdetnaya, L.N.; Foster, T.H.; Parache, R.-M.; Guillemin, F. Effect of Irradiation Fluence Rate on the Efficacy of Photodynamic Therapy and Tumor Oxygenation in Meta-Tetra (Hydroxyphenyl) Chlorin (MTHPC)-Sensitized HT29 Xenografts in Nude Mice1. Radiat. Res. 2002, 158, 339–345. [Google Scholar] [CrossRef]
- Middelburg, T.A.; Van Zaane, F.; De Bruijn, H.S.; Van Der Ploeg-van den Heuvel, A.; Sterenborg, H.J.C.M.; Neumann, H.A.M.; De Haas, E.R.M.; Robinson, D.J. Fractionated Illumination at Low Fluence Rate Photodynamic Therapy in Mice. Photochem. Photobiol. 2010, 86, 1140–1146. [Google Scholar] [CrossRef]
- Robertson, C.A.; Evans, D.H.; Abrahamse, H. Photodynamic Therapy (PDT): A Short Review on Cellular Mechanisms and Cancer Research Applications for PDT. J. Photochem. Photobiol. B. 2009, 96, 1–8. [Google Scholar] [CrossRef]
- Falk-Mahapatra, R.; Gollnick, S.O. Photodynamic Therapy and Immunity: An Update. Photochem. Photobiol. 2020, 96, 550–559. [Google Scholar] [CrossRef]
- Hendrzak-Henion, J.A.; Knisely, T.L.; Cincotta, L.; Cincotta, E.; Cincotta, A.H. Role of the Immune System in Mediating the Antitumor Effect of Benzophenothiazine Photodynamic Therapy. Photochem. Photobiol. 1999, 69, 575–581. [Google Scholar] [CrossRef]
- Li, W.; Yang, J.; Luo, L.; Jiang, M.; Qin, B.; Yin, H.; Zhu, C.; Yuan, X.; Zhang, J.; Luo, Z.; et al. Targeting Photodynamic and Photothermal Therapy to the Endoplasmic Reticulum Enhances Immunogenic Cancer Cell Death. Nat. Commun. 2019, 10, 3349. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, I.S.S.; Martins-Filho, A.; Micheli, D.C.; de Lima, C.A.; Tavares-Murta, B.M.; Murta, E.F.C.; Nomelini, R.S. IL-6 and IL-8 as Prognostic Factors in Peritoneal Fluid of Ovarian Cancer. Immunol. Investig. 2020, 49, 510–521. [Google Scholar] [CrossRef] [PubMed]
- Nowak, M.; Glowacka, E.; Szpakowski, M.; Szyllo, K.; Malinowski, A.; Kulig, A.; Tchorzewski, H.; Wilczynski, J. Proinflammatory and Immunosuppressive Serum, Ascites and Cyst Fluid Cytokines in Patients with Early and Advanced Ovarian Cancer and Benign Ovarian Tumors. Neuroendocrinol. Lett. 2010, 31, 375–383. [Google Scholar] [PubMed]
- Wen, J.; Zhao, Z.; Huang, L.; Wang, L.; Miao, Y.; Wu, J. IL-8 Promotes Cell Migration through Regulating EMT by Activating the Wnt/β-Catenin Pathway in Ovarian Cancer. J. Cell. Mol. Med. 2020, 24, 1588–1598. [Google Scholar] [CrossRef]
- Brencicova, E.; Jagger, A.L.; Evans, H.G.; Georgouli, M.; Laios, A.; Attard Montalto, S.; Mehra, G.; Spencer, J.; Ahmed, A.A.; Raju-Kankipati, S.; et al. Interleukin-10 and Prostaglandin E2 Have Complementary but Distinct Suppressive Effects on Toll-like Receptor-Mediated Dendritic Cell Activation in Ovarian Carcinoma. PLoS ONE 2017, 12, e0175712. [Google Scholar] [CrossRef]
- Schroder, K.; Hertzog, P.J.; Ravasi, T.; Hume, D.A. Interferon-Gamma: An Overview of Signals, Mechanisms and Functions. J. Leukoc. Biol. 2004, 75, 163–189. [Google Scholar] [CrossRef]
- Brackett, C.M.; Gollnick, S.O. Photodynamic Therapy Enhancement of Anti-Tumor Immunity. Photochem. Photobiol. Sci. 2011, 10, 649–652. [Google Scholar] [CrossRef]
- Garg, A.D.; Krysko, D.V.; Vandenabeele, P.; Agostinis, P. DAMPs and PDT-Mediated Photo-Oxidative Stress: Exploring the Unknown. Photochem. Photobiol. Sci. 2011, 10, 670–680. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baydoun, M.; Boidin, L.; Leroux, B.; Vignion-Dewalle, A.-S.; Quilbe, A.; Grolez, G.P.; Azaïs, H.; Frochot, C.; Moralès, O.; Delhem, N. Folate Receptor Targeted Photodynamic Therapy: A Novel Way to Stimulate Anti-Tumor Immune Response in Intraperitoneal Ovarian Cancer. Int. J. Mol. Sci. 2023, 24, 11288. https://doi.org/10.3390/ijms241411288
Baydoun M, Boidin L, Leroux B, Vignion-Dewalle A-S, Quilbe A, Grolez GP, Azaïs H, Frochot C, Moralès O, Delhem N. Folate Receptor Targeted Photodynamic Therapy: A Novel Way to Stimulate Anti-Tumor Immune Response in Intraperitoneal Ovarian Cancer. International Journal of Molecular Sciences. 2023; 24(14):11288. https://doi.org/10.3390/ijms241411288
Chicago/Turabian StyleBaydoun, Martha, Léa Boidin, Bertrand Leroux, Anne-Sophie Vignion-Dewalle, Alexandre Quilbe, Guillaume Paul Grolez, Henri Azaïs, Céline Frochot, Olivier Moralès, and Nadira Delhem. 2023. "Folate Receptor Targeted Photodynamic Therapy: A Novel Way to Stimulate Anti-Tumor Immune Response in Intraperitoneal Ovarian Cancer" International Journal of Molecular Sciences 24, no. 14: 11288. https://doi.org/10.3390/ijms241411288
APA StyleBaydoun, M., Boidin, L., Leroux, B., Vignion-Dewalle, A.-S., Quilbe, A., Grolez, G. P., Azaïs, H., Frochot, C., Moralès, O., & Delhem, N. (2023). Folate Receptor Targeted Photodynamic Therapy: A Novel Way to Stimulate Anti-Tumor Immune Response in Intraperitoneal Ovarian Cancer. International Journal of Molecular Sciences, 24(14), 11288. https://doi.org/10.3390/ijms241411288







